 Open Access Article
Open Access ArticleHealth risk assessment of polycyclic aromatic hydrocarbons in indoor dust from Okerenkoko community, Warri, Nigeria
Sisanmi Samuel
Aghomi
 *a,
Okpoebi Kenneth
Berezi
b and
Chilaka
Diepreye
c
*a,
Okpoebi Kenneth
Berezi
b and
Chilaka
Diepreye
c
aDepartment of Environmental Management and Pollution, Nigeria Maritime University, Okerenkoko, Delta State, Nigeria. E-mail: samaghomi@yahoo.com; Tel: +234(0)74848825
bDepartment of Environmental Management and Pollution, Nigeria Maritime University, Okerenkoko, Delta State, Nigeria
cDepartment of Environmental Management and Pollution, Nigeria Maritime University, Okerenkoko, Delta State, Nigeria
First published on 11th March 2026
Abstract
This study investigated the occurrence, sources, and health risks of polycyclic aromatic hydrocarbons (PAHs) in indoor dust from the Okerenkoko Community, Warri, Nigeria. Dust samples were collected from three locations: NMU Campus/Lodge (NMU CL), Zion Residential Area (ZA), and Okerenkoko II Area (Okk). Total PAH concentrations (∑PAHs) ranged from 0.0546 to 0.1005 mg kg−1, with higher levels at ZA and Okk, reflecting spatial variability influenced by local anthropogenic activities, including artisanal crude oil refining, biomass burning, combustion-related emissions from marine transport, and domestic fuel use. Compositional analysis revealed a predominance of 3- to 5-ring PAHs, with low- and high-molecular-weight (LMW and HMW) species contributing almost equally (50.8% and 49.2%, respectively), suggesting mixed pyrogenic–petrogenic sources. Diagnostic ratios (e.g., Ant/(Ant + Phe), Flt/(Flt + Pyr), BaA/(BaA + Chr), IP/(IP + BghiP)) indicated that NMU CL and ZA were mainly influenced by pyrogenic sources, whereas Okk reflected both pyrogenic and petroleum-related inputs. Principal component analysis (PCA) confirmed distinct contributions, with HMW PAHs associated with combustion processes and LMW PAHs linked to volatilization or indoor emissions. Health risk assessment showed that adults exhibited the highest incremental lifetime cancer risks (ILCRs), followed by children and adolescents, with total ILCRs ranging from 1.05 × 10−7 to 1.90 × 10−7 across all sites. Dermal contact and ingestion were identified as the dominant pathways, while inhalation contributed negligibly (≤10−12). For example, adults at Okk recorded the highest ILCR (1.90 × 10−7), driven largely by dermal exposure, whereas children's ILCRs ranged between 1.26 × 10−7 and 1.75 × 10−7. Non-cancer risk evaluation indicated hazard indices (HI) for all age groups remained far below 1 (children: 3.61 × 10−5 to 4.88 × 10−5), suggesting negligible non-carcinogenic risk. These findings reveal the complex nature of PAH sources in indoor dust and highlight the importance of dermal exposure alongside ingestion, particularly for adults. The results underscore the heightened exposure risks for vulnerable groups and the urgency of targeted environmental controls and community health interventions in the Niger Delta.
Environmental significanceIndoor environments represent critical but often overlooked reservoirs of polycyclic aromatic hydrocarbons (PAHs), especially in vulnerable communities within the Niger Delta. This study demonstrates that PAH concentrations in household dust of Okerenkoko, Nigeria, are influenced by both pyrogenic and petrogenic sources, including biomass burning, vehicular emissions, and petroleum-related activities. The findings reveal elevated risks for children, with hazard indices exceeding safe limits and incremental lifetime cancer risks approaching U.S. EPA thresholds. These results underscore the urgent need for improved environmental management, community health protection, and policy interventions to mitigate exposure. By linking local anthropogenic activities to measurable indoor contamination and health risks, this work advances understanding of environmental exposure pathways in resource-rich but pollution-burdened regions. |
1 Introduction
Human well-being is significantly affected by indoor environmental conditions. From infancy to old age, most individuals spend up to 90% of their lives indoors, primarily at home or in the workplace.1 Furthermore, indoor environments have been scientifically shown to be two to five times more polluted than outdoor environments.2 Dust particles are ubiquitous in indoor settings,3 where they act as a major sink for various semi-volatile organic compounds (SVOCs).4 Dust consists of solid, fine particles of matter ranging in size from less than 1 µm to at least 100 µm, which may become airborne depending on their origin, physical characteristics, and ambient conditions.5 It is a heterogeneous mixture of inorganic, organic, and biological materials.6 Dust can be suspended in the air or settle on surfaces and originates from multiple sources, including soil, material abrasion, pesticides, asbestos, pollen, bacteria, shed skin, cigarette smoke, and dust mites.7 It plays a crucial role in the deterioration of indoor air quality, as it carries allergens and airborne contaminants that adversely affect human health.8 Indoor dust refers to a complex mixture of materials and particulate matter deposited from indoor aerosols and soil particles transported indoors via foot traffic or air movement.9 It represents a heterogeneous matrix of debris and particles of both anthropogenic and natural origin.Despite its importance, indoor dust remains an underexplored vector of human exposure in the indoor environment.10 It is relatively persistent and can absorb contaminants over extended periods, ranging from weeks to years.11 Additionally, the resuspension of settled particles can mobilize potentially toxic compounds.12 Indoor dust particles can act as carriers for a wide array of pollutants, including organic, inorganic, and biological materials.13 Organic pollutants such as polycyclic aromatic hydrocarbons (PAHs)—by-products of incomplete combustion—adhere to dust particles because of their hydrophobic properties.14 Inorganic pollutants, including heavy metals such as lead, cadmium, and mercury, may originate from building materials, paints, electronics, and outdoor sources that infiltrate indoor spaces.15 Biological contaminants such as bacteria, fungi, pollen, and allergens also accumulate in indoor dust.16 These pollutants attach through physical adsorption or chemical interactions, posing health risks when inhaled, ingested, or absorbed dermally. Their composition and concentration are influenced by factors such as human activity, indoor materials, ventilation systems, and proximity to pollution sources.17 Once absorbed, pollutants may persist or degrade more slowly in indoor settings compared to outdoor environments.18 Consequently, indoor dust serves as a long-term repository of contaminants, increasing human exposure risks through air resuspension, ingestion, or dermal contact.19
Among indoor pollutants, PAHs are particularly notable due to their ubiquity across environmental media—including air, water, soil, sediment, vegetation, and dust—where they persist as trace organic contaminants.20 Because of their persistence, bioaccumulation potential, and carcinogenic and mutagenic properties, PAHs are among the most extensively studied environmental pollutants.4 Structurally, PAHs are hydrophobic organic compounds composed of two or more fused aromatic rings arranged linearly, angularly, or in clusters.21 They are typically formed through incomplete combustion or high-temperature pyrolysis of organic materials.22 PAH sources include anthropogenic activities such as fossil fuel and wood combustion, cooking, industrial operations, and engine exhausts, as well as natural events like volcanic eruptions and wildfires.23 Their mobilization and environmental transport through human activities play key roles in their geochemical cycling.4
In Nigeria's Niger Delta region, PAH contamination is further exacerbated by oil and gas-related activities, particularly illegal oil bunkering and non-standardized refining.24 These activities release soot, which readily binds with indoor dust particles. During combustion, PAHs preferentially adsorb onto soot due to their hydrophobicity and strong affinity for carbonaceous materials.25 These PAH-soot complexes are transported atmospherically and eventually deposited on indoor surfaces, contributing to their persistence and potential for human exposure. This interaction heightens health risks associated with airborne pollutants.26 Although the general health effects of PAHs are well documented—including endocrine disruption, reproductive toxicity, developmental and neurological impairments, asthma, skin allergies, and premature birth—there is limited understanding of the specific risks posed by PAHs bound to indoor dust particles, particularly under long-term exposure or in combination with other indoor pollutants.27 Nonetheless, PAHs remain a significant carcinogenic threat in indoor settings, particularly for children.28 Given this, timely and systematic evaluation of soot–dust interactions is critical. Indoor dust serves as a key reservoir of chemical pollutants due to its large surface area and ability to retain contaminants for extended periods. This capacity is especially pronounced in indoor environments, where dilution, leaching, and degradation processes are minimal.29 Thus, indoor dust acts as a vital indicator of environmental contamination, offering valuable insights into exposure levels, particularly in industrial or high-risk communities.
Investigating PAHs in indoor dust and understanding both their short- and long-term health implications has therefore become imperative. The primary objectives of this study are: (i) to determine, for the first time, the levels of the 16 USEPA priority PAHs in indoor dust from the Okerenkoko community; (ii) to identify possible sources of these contaminants; and (iii) to assess the associated health risks, including cancer and non-cancer risks. This study hypothesizes that PAHs in the Okerenkoko community primarily originate from outdoor sources and are introduced into indoor spaces through air exchange processes.
2 Materials and methods
2.1 Study area and sampling
Okerenkoko lies along the Escravos Estuary in Warri South-West Local Government Area of Delta State, Nigeria. It is located at coordinates 5°37′0.06″N, 5°23′17.412″E and 5°38′6.132″N, 5°23′45.732″E (Fig. 1). The area is a tidal flat and a transitional environment between the continent and the deep marine zone. The main occupations of the indigenes include fishing, hunting, and lumbering. The Escravos Estuary is a mangrove ecosystem that serves as a habitat for many aquatic organisms, a breeding ground for seasonal migratory birds, and a source of livelihood for community residents, particularly fishermen.30 The vegetation cover is mainly mangrove trees present in and around the creeks. The mangroves function as offshore long-drift and wave-energy breakers, thereby protecting the coastal environment and the communities within it from flooding.The combined extracts were concentrated, solvent-exchanged into hexane, and further reduced to a volume of 1–3 mL. Fractionation into the aromatic fraction was carried out using silica gel cartridges or columns packed with 10 g of 100–200 mesh silica gel, which had been pre-baked at 105 °C overnight and conditioned with 10–20 mL of dichloromethane immediately before use. The aromatic fractions were collected, concentrated to a final volume of 2 mL, and transferred into clean vials. At this stage, the samples were ready for GC-MS analysis of PAHs.
2.2 Instrumentation
The GC-MS method used for determining the concentrations of 16 priority polycyclic aromatic hydrocarbons (PAHs) in indoor dust was validated in accordance with international guidelines. The validation addressed calibration linearity, recovery, precision, and detection limits, with all results expressed in milligrams per kilogram (mg kg−1).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (LOD) and 10
1 (LOD) and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (LOQ).32,33
1 (LOQ).32,33
2.3 Gas chromatography analysis
After extraction, separation, identification, and quantification of PAHs were performed using an Agilent 7890A gas chromatograph coupled with a 5975C mass selective detector (MSD) (Agilent Technologies, CT, USA). The column employed was a DB-17 capillary column (30 m length × 0.25 mm internal diameter × 0.25 µm film thickness) (J&W Scientific, USA). Helium was used as the carrier gas at a constant flow rate of 1.0 mL min−1.The oven temperature was programmed as follows: initial temperature of 50 °C held for 2 min, ramped to 150 °C at 15 °C min−1, increased to 240 °C at 7 °C min−1, and finally raised to 280 °C at 4 °C min−1 and held. The MS detector was operated in selected ion monitoring (SIM) mode. The ion source temperature was set at 230 °C, while the quadrupole and transfer line temperatures were maintained at 150 °C and 280 °C, respectively.
An aliquot of 1.0 µL of extract was injected into the GC in splitless mode. Target PAHs were identified by comparing retention times and characteristic ions with those of authentic PAH standards, while quantification was performed using calibration curves prepared from the standards.
• Internal standards: deuterated PAHs (naphthalene-d8, phenanthrene-d10, chrysene-d12, and perylene-d12) were added to each sample prior to extraction to correct for extraction efficiency and instrumental variability.
• Calibration and secondary standards: multi-point calibration curves (five levels) were prepared for all target PAHs, with R2 consistently >0.995. Independent secondary check standards, prepared from separate stock solutions, were analyzed daily to verify calibration accuracy. Instrument response deviations remained within ±10%, consistent with USEPA criteria.
• Duplicate samples: at least 10% of the indoor dust samples were analyzed in duplicate. The relative percent difference (RPD) between duplicates was consistently <15%, indicating good reproducibility.
• Spike recovery: matrix spikes and spiked blanks were analyzed to evaluate recovery efficiency. Recoveries for target PAHs ranged from 72% to 108%, within the acceptable QA/QC limits (70–120%).
• Method detection limits (MDLs) and LOQs: MDLs were determined as three times the standard deviation of seven replicate low-level spiked samples, while LOQs were defined as ten times the standard deviation. Values for each PAH are reported in the results.
• Blanks and contamination control: laboratory reagent blanks were processed with each sample batch to assess contamination. No target PAHs were detected above MDL levels.
2.4 Source identification
Diagnostic ratios are among the most widely used approaches for identifying emission sources of PAHs due to their simplicity.34 Studies show that the relative concentrations of PAHs with similar molecular weights and structures can effectively indicate pollutant sources.35 Based on isomeric ratios, ANT/(ANT + PHE) < 0.1 indicates petroleum sources, whereas a ratio > 0.1 suggests combustion.36 Similarly, FLA/(FLA + PYR) < 0.4 points to petroleum, values between 0.4 and 0.5 suggest liquid fossil-fuel combustion (e.g., engine exhausts and crude oil), and ratios > 0.5 indicate coal, grass, or wood combustion.2.5 Health risk assessment
The potential health impacts of exposure to PAHs in indoor dust from the Okerenkoko community were systematically evaluated using both carcinogenic and non-carcinogenic metrics. For carcinogenic risk, three indices were applied: (1) the benzo(a)pyrene equivalent concentration (BaPE), which quantifies relative carcinogenic potency; (2) the total toxicity equivalence (TEQ) derived from BaPE; and (3) the incremental lifetime cancer risk (ILCR), which estimates the probability of developing cancer over a lifetime. Non-carcinogenic risks were evaluated by estimating the average daily dose (ADD) of exposure, calculating the hazard quotient (HQ) for individual PAHs, and deriving the hazard index (HI) to represent cumulative effects from aggregated exposures.| BaPE = 0.06 × BaA + 0.07 × BbF + BkF + BaP + 0.6 × DahA + 0.08 × IcdP | (1) |
| BaPeq(TEQ) = Cn × TEF | (2) |
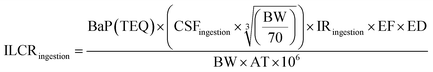 | (3) |
 | (4) |
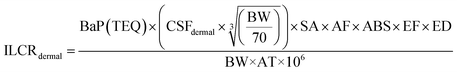 | (5) |
| ILCRtotal = ILCRingestion + ILCRinhalation + ILCRdermal | (6) |
| S/N | PAH | TEF | NMU A | ZA A | Okk A | |||
|---|---|---|---|---|---|---|---|---|
| Conc. (mg kg−1) | BaPeq (mg kg−1) | Conc. (mg kg−1) | BaPeq (mg kg−1) | Conc. (mg kg−1) | BaPeq (mg kg−1) | |||
| 1 | Nap | 0.001 | 0.004 | 0.000004 | 0.005 | 0.000005 | 0.003 | 0.000003 |
| 2 | Acy | 0.001 | 0.004 | 0.000004 | 0.006 | 0.000006 | 0.003 | 0.000003 |
| 3 | Ace | 0.001 | 0.003 | 0.000003 | 0.003 | 0.000003 | 0.003 | 0.000003 |
| 4 | Fluo | 0.001 | 0.003 | 0.000003 | 0.003 | 0.000003 | 0.002 | 0.000002 |
| 5 | Ant | 0.01 | 0.002 | 0.000020 | 0.003 | 0.000030 | 0.002 | 0.000020 |
| 6 | Phen | 0.001 | 0.004 | 0.000004 | 0.006 | 0.000006 | 0.005 | 0.000005 |
| 7 | Flt | 0.001 | 0.004 | 0.000004 | 0.005 | 0.000005 | 0.005 | 0.000005 |
| 8 | Pyr | 0.001 | 0.004 | 0.000004 | 0.005 | 0.000005 | 0.004 | 0.000004 |
| 9 | BaA | 0.1 | 0.006 | 0.000600 | 0.008 | 0.000800 | 0.009 | 0.000900 |
| 10 | Chrys | 0.01 | 0.005 | 0.000050 | 0.007 | 0.000070 | 0.007 | 0.000070 |
| 11 | BbF | 0.1 | 0.004 | 0.000400 | 0.007 | 0.000700 | 0.007 | 0.000700 |
| 12 | BkF | 0.1 | 0.004 | 0.000400 | 0.008 | 0.000800 | 0.006 | 0.000600 |
| 13 | BaP | 1.0 | 0.005 | 0.005000 | 0.006 | 0.006000 | 0.008 | 0.008000 |
| 14 | IndP | 1.0 | 0.005 | 0.005000 | 0.007 | 0.007000 | 0.006 | 0.006000 |
| 15 | DahA | 0.1 | 0.005 | 0.000500 | 0.007 | 0.000700 | 0.007 | 0.000700 |
| 16 | BghiP | 0.01 | 0.004 | 0.000040 | 0.006 | 0.000060 | 0.008 | 0.000080 |
| ∑BaPeq | 0.012036 | 0.016193 | 0.017095 | |||||
(a) Average daily dose (ADD) is a central parameter in non-cancer risk assessment, representing the estimated amount of a contaminant an individual is exposed to per unit body weight per day. It is calculated separately for different exposure routes, ingestion, dermal contact, and inhalation, since each pathway contributes uniquely to the total body burden of contaminants.44,45
Ingestion:
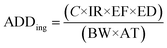 | (7) |
Dermal contact:
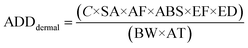 | (8) |
Inhalation:
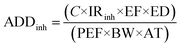 | (9) |
Non-cancer risk assessment of PAHs in indoor dust relies on established exposure parameters that quantify human contact through ingestion, dermal contact, and inhalation (Table 2). Risk characterization compares ADD values to the reference dose (RfD, mg per kg per day) (Table 3). Hazard quotients (HQ) are calculated as ADD/RfD, while the hazard index (HI) represents the sum of HQs across all pathways.46,47
| Exposure variable | Definitions | Unit | Children | Adolescents | Adults | Reference |
|---|---|---|---|---|---|---|
| Dust ingestion rate (IR) | Average amount of soil/dust ingested per day by an individual | mg per day | 200 | 100 | 100 | 52 and 53 |
| Exposure frequency (EF) | Number of exposure events per year | day per year | 350 | 350 | 350 | 54 and 55 |
| Exposure duration (ED) | Total period over which exposure occurs | year | 6 | 14 | 30 | 55 and 56 |
| Cancer slope factor (CSFingestion) | Upper-bound estimate of increased cancer risk from lifetime oral exposure | (per mg per kg per day) | 7.3 | 7.3 | 7.3 | 57 and 58 |
| Cancer slope factor (CSFinhalation) | Upper-bound estimate of increased cancer risk from lifetime inhalation exposure | (per mg per kg per day) | 3.85 | 3.85 | 3.85 | 57 and 58 |
| Cancer slope factor (CSFdermal) | Upper-bound estimate of increased cancer risk from lifetime dermal exposure | (per mg per kg per day) | 25 | 25 | 25 | 57 |
| Body weight (BW) | Average body mass of an exposed individual | kg | 15 | 45 | 62 | 57 and 58 |
| Average lifespan (AT) | Averaging time used in risk calculations (70 years × 365 days for carcinogenic risk) | day | 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 280 280 |
26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 280 280 |
26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 280 280 |
54 and 55 |
| Particle emission factor (PEF) | Factor estimating contaminant concentrations in air from soil/dust sources | m3 kg−1 | 1.36 × 109 | 1.36 × 109 | 1.36 × 109 | 57 and 58 |
| Dermal exposure area (SA) | Skin surface area in contact with soil/dust | cm per day | 2800 | 2800 | 5700 | 57 and 58 |
| Skin adherence factor (AF) | Mass of soil/dust adhering to skin per unit area | mg cm−2 | 0.2 | 0.2 | 0.07 | 57 and 58 |
| Dermal absorption fraction (ABS) | Fraction of contaminant absorbed into body after dermal contact | Unitless | 0.13 | 0.13 | 0.13 | 57 and 58 |
| Inhalation rate (IRinhalation) | Average volume of air breathed per day by an individual | m per day | 10.9 | 17.7 | 17.5 | 57 and 58 |
| PAH | Ingestion RfD (mg per kg per day) | Dermal RfD (mg per kg per day) | Inhalation RfD (mg m−3) |
|---|---|---|---|
| a ** Dermal and inhalation RfDs were not directly available from toxicological studies; instead, they were derived by route-to-route extrapolation from oral RfD values. NA (not available): no RfD value has been established for that exposure route.59,60 | |||
| Naphthalene (Nap) | 0.04 | 0.02** | 0.003 |
| Acenaphthene (Ace) | 0.006 | 0.02** | 0.88** |
| Acenaphthylene (Acy) | 0.06 | 0.06** | 0.88** |
| Fluorene (Flu) | 0.04 | 0.04** | NA |
| Phenanthrene (Phen) | 0.04 | NA | NA |
| Anthracene (Ant) | 0.3 | 0.3** | NA |
| Fluoranthene (Flt) | 0.04 | 0.04** | 0.88** |
| Pyrene (Pyr) | 0.03 | 0.03** | 0.88** |
| Benzo[a]anthracene (BaA) | 0.03 | 0.03** | 0.88** |
| Chrysene (Cry) | 0.03 | 0.03** | 0.88** |
| Benzo[b]fluoranthene (BbF) | 0.03 | 0.03** | 0.88** |
| Benzo[k]fluoranthene (BkF) | 0.03 | 0.03** | 0.88** |
| Benzo[a]pyrene (BaP) | 0.03 | 0.03** | 0.88** |
| Dibenzo[a,h]anthracene (DBA) | 0.03 | 0.03** | 0.88** |
| Indeno[1,2,3-cd]pyrene (IND) | 0.03 | 0.03** | 0.88** |
| Benzo[ghi]perylene (BghiP) | 0.03 | 0.03** | 0.88** |
(b) Hazard quotient (HQ): the hazard quotient (HQ) is a risk assessment metric used to evaluate the potential non-carcinogenic effects of a chemical substance. It is defined as the ratio of the average daily dose (ADD) of a contaminant to its reference dose (RfD). An HQ value less than 1 indicates an insignificant risk, whereas values greater than 1 suggest a likelihood of adverse health effects due to excessive exposure.48 The non-carcinogenic risk for each exposure pathway was calculated using the following equation:
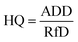 | (10) |
(c) Hazard index (HI): the hazard index (HI) extends the HQ approach by summing the HQ values of multiple chemicals or multiple exposure pathways to assess cumulative non-carcinogenic risk. An HI less than 1 implies that combined exposures are unlikely to cause significant health effects, whereas an HI above 1 suggests possible health concerns, especially for sensitive populations.49 The HI represents the combined effect of all PAHs and exposure pathways:
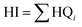 | (11) |
2.6 Threshold values for PAHs in indoor dust
Polycyclic aromatic hydrocarbons (PAHs) in indoor dust represent a significant exposure pathway, especially for children. Although specific indoor dust threshold values are limited, WHO and USEPA soil- and air-based standards are commonly referenced. Benzo[a]pyrene (BaP), a highly carcinogenic PAH, has a WHO unit cancer risk of 8.7 × 10−5 per ng m−3 (≈0.025 mg kg−1) and a USEPA residential soil screening level (SSL) of 0.06 mg kg−1. Other carcinogenic PAHs are assessed using Relative Potency Factors (RPFs) to calculate BaP equivalents, with SSLs of 0.01–0.2 mg kg−1. Non-carcinogenic PAHs have SSLs of ∼0.2 mg kg−1 and low RPFs (0.001–0.01), but still contribute to total exposure.50,512.7 Method of statistical analysis
Descriptive statistics, including mean, standard deviation, median, minimum, maximum, upper confidence limit (UCL), and coefficient of variation (CV%), were used to characterize the distribution of the data. All statistical analyses were performed using Microsoft Excel and IBM SPSS Statistics 30. Data visualization were performed using Python 3.11 with custom scripts executed in Google Colab.3 Results and discussions
The concentrations of the 16 US EPA priority PAHs were determined in indoor dust samples collected from three designated areas: NMU Campus/Lodge (NMU CL), Zion Residential Area (ZA), and Okerenkoko II Area (OKK) within the Okerenkoko Community. The results of the analysis are presented in Tables 3–8, Fig. 2 and 3. The minimum detection limit of the GC/MS used in the analysis was 0.001 mg kg−1. Mean concentrations, standard deviations, maximum and minimum values, upper confidence limits (UCL), and coefficients of variation (CV%) were calculated for both individual PAHs and total PAH levels at each location. In addition, composition profiles, source apportionment using diagnostic ratios, principal component analysis (PCA), incremental lifetime cancer risk (ILCR), and non-cancer risk (NCR) were evaluated to provide a comprehensive understanding of the sources, health risks, and potential impacts of PAH contamination in these areas.| Locations | NMU campus/lodge (NMU CL) | Zion residential area (ZA) | Okerenkoko II area (Okk) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Component | L1 | L2 | L3 | L4 | L5 | L6 | L7 | L8 | L9 | L10 | L11 | L12 | L13 | L14 | L15 |
| Nap | 0.0034 | 0.0042 | 0.0031 | 0.0043 | 0.0032 | 0.0028 | 0.0075 | 0.0046 | 0.006 | 0.0057 | 0.0037 | 0.0036 | 0.0031 | 0.0024 | 0.0027 |
| Acy | 0.0061 | 0.0026 | 0.002 | 0.002 | 0.0065 | 0.006 | 0.0054 | 0.0074 | 0.0049 | 0.0052 | 0.0027 | 0.0036 | 0.0027 | 0.0038 | 0.0016 |
| Ace | 0.0024 | 0.0021 | 0.0023 | 0.0033 | 0.0026 | 0.0029 | 0.0024 | 0.0029 | 0.0027 | 0.002 | 0.0057 | 0.003 | 0.0025 | 0.0033 | 0.0018 |
| Fluo | 0.003 | 0.0032 | 0.0016 | 0.0034 | 0.0023 | 0.003 | 0.0025 | 0.0035 | 0.0028 | 0.0028 | 0.0018 | 0.0019 | 0.0015 | 0.0016 | 0.0012 |
| Ant | 0.003 | 0.0026 | 0.0016 | 0.0029 | 0.0023 | 0.0053 | 0.0014 | 0.0026 | 0.0035 | 0.0027 | 0.0026 | 0.0023 | 0.0025 | 0.0022 | 0.0018 |
| Phen | 0.0028 | 0.0029 | 0.0055 | 0.0042 | 0.0048 | 0.0071 | 0.0018 | 0.0072 | 0.0068 | 0.0088 | 0.0023 | 0.0043 | 0.0051 | 0.0069 | 0.0047 |
| Flt | 0.0013 | 0.004 | 0.0067 | 0.0041 | 0.0033 | 0.0075 | 0.0041 | 0.0067 | 0.0035 | 0.005 | 0.0063 | 0.0042 | 0.0022 | 0.0065 | 0.0038 |
| Pyr | 0.0023 | 0.004 | 0.0041 | 0.0031 | 0.0042 | 0.0055 | 0.0062 | 0.0036 | 0.004 | 0.0032 | 0.0028 | 0.0052 | 0.0032 | 0.0059 | 0.0018 |
| BaA | 0.0081 | 0.0038 | 0.0056 | 0.0052 | 0.0059 | 0.0072 | 0.0084 | 0.0092 | 0.007 | 0.0083 | 0.0098 | 0.0085 | 0.0095 | 0.0092 | 0.0082 |
| Chrys | 0.0031 | 0.0042 | 0.0043 | 0.0062 | 0.0052 | 0.0066 | 0.0085 | 0.0071 | 0.0096 | 0.0027 | 0.0028 | 0.0072 | 0.009 | 0.0087 | 0.007 |
| BbF | 0.0021 | 0.0076 | 0.0066 | 0.0038 | 0.0021 | 0.0075 | 0.0074 | 0.0081 | 0.0048 | 0.0084 | 0.0043 | 0.0087 | 0.0067 | 0.0087 | 0.0072 |
| BkF | 0.0029 | 0.0034 | 0.0036 | 0.0031 | 0.0049 | 0.0068 | 0.0088 | 0.0097 | 0.0096 | 0.0074 | 0.0065 | 0.006 | 0.0099 | 0.0043 | 0.0047 |
| BaP | 0.0034 | 0.0043 | 0.0068 | 0.006 | 0.0026 | 0.0071 | 0.0054 | 0.0071 | 0.0051 | 0.0042 | 0.0091 | 0.0078 | 0.0086 | 0.0098 | 0.0068 |
| IndP | 0.0032 | 0.0036 | 0.0099 | 0.0032 | 0.0062 | 0.0088 | 0.0084 | 0.0069 | 0.0038 | 0.0094 | 0.0037 | 0.0087 | 0.0064 | 0.0013 | 0.0083 |
| DahA | 0.0035 | 0.0052 | 0.0057 | 0.0067 | 0.0046 | 0.0059 | 0.0061 | 0.0061 | 0.0054 | 0.0094 | 0.0077 | 0.0083 | 0.0079 | 0.0065 | 0.0045 |
| BghiP | 0.004 | 0.0037 | 0.0036 | 0.005 | 0.006 | 0.0078 | 0.0054 | 0.0078 | 0.0054 | 0.0044 | 0.0054 | 0.0094 | 0.0079 | 0.0087 | 0.0071 |
| ∑PAHs | 0.0546 | 0.0614 | 0.073 | 0.0665 | 0.0667 | 0.0978 | 0.0897 | 0.1005 | 0.0849 | 0.0896 | 0.0772 | 0.0927 | 0.0887 | 0.0898 | 0.0732 |
| Component | Mean | Standard deviation | Median | Minimum (min) | Maximum (Max) | (Upper confidence limit) (UCL) | CV% (coefficient of variation) |
|---|---|---|---|---|---|---|---|
| Nap | 0.0040 | 0.0014 | 0.0036 | 0.0024 | 0.0075 | 0.0064 | 35.29% |
| Acy | 0.0042 | 0.0019 | 0.0038 | 0.0016 | 0.0074 | 0.0073 | 45.04% |
| Ace | 0.0028 | 0.0009 | 0.0026 | 0.0018 | 0.0057 | 0.0043 | 32.88% |
| Fluo | 0.0024 | 0.0008 | 0.0025 | 0.0012 | 0.0035 | 0.0037 | 31.51% |
| Ant | 0.0026 | 0.0009 | 0.0026 | 0.0014 | 0.0053 | 0.0041 | 35.02% |
| Phen | 0.0050 | 0.0021 | 0.0048 | 0.0018 | 0.0088 | 0.0084 | 40.89% |
| Flt | 0.0046 | 0.0018 | 0.0041 | 0.0013 | 0.0075 | 0.0076 | 38.90% |
| Pyr | 0.0039 | 0.0013 | 0.0040 | 0.0018 | 0.0062 | 0.0061 | 32.92% |
| BaA | 0.0076 | 0.0018 | 0.0082 | 0.0038 | 0.0098 | 0.0105 | 23.28% |
| Chrys | 0.0061 | 0.0023 | 0.0066 | 0.0027 | 0.0096 | 0.0100 | 37.79% |
| BbF | 0.0063 | 0.0023 | 0.0072 | 0.0021 | 0.0087 | 0.0100 | 36.27% |
| BkF | 0.0061 | 0.0025 | 0.0060 | 0.0029 | 0.0099 | 0.0102 | 41.22% |
| BaP | 0.0063 | 0.0021 | 0.0068 | 0.0026 | 0.0098 | 0.0097 | 33.61% |
| IndP | 0.0061 | 0.0028 | 0.0064 | 0.0013 | 0.0099 | 0.0107 | 45.26% |
| DahA | 0.0062 | 0.0016 | 0.0061 | 0.0035 | 0.0094 | 0.0088 | 25.31% |
| BghiP | 0.0061 | 0.0019 | 0.0054 | 0.0036 | 0.0094 | 0.0092 | 30.84% |
| ∑PAHs | 0.0804 | 0.0140 | 0.0849 | 0.0546 | 0.1005 | 0.1034 | 17.37 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Ring type | |||||||
| 2-Ring | 0.0040 | 0.0014 | 0.0036 | 0.0024 | 0.0075 | 0.0064 | 35.29% |
| 3-Ring | 0.0170 | 0.0040 | 0.0158 | 0.0111 | 0.0243 | 0.0236 | 23.62 |
| 4-Ring | 0.0223 | 0.0045 | 0.0217 | 0.0148 | 0.0303 | 0.0296 | 20.01 |
| 5-Ring | 0.0249 | 0.0062 | 0.0273 | 0.0119 | 0.0331 | 0.0351 | 24.93 |
| 6-Ring | 0.0122 | 0.0035 | 0.0135 | 0.0072 | 0.0181 | 0.0180 | 28.53 |
| ∑PAHs | 0.0804 | 0.0140 | 0.0849 | 0.0546 | 0.1005 | 0.1034 | 17.37 |
| S/N | Location | Concentration range (mg kg−1) | Mean/median (mg kg−1) | Reference |
|---|---|---|---|---|
| 1 | Okerenkoko community | 0.0546–1.005 | 0.0804 | Present study |
| 2 | Residential dust, Osogbo metropolis, Nigeria | 0.000023–0.02603 | — | 69 |
| 3 | Port Harcourt city, Nigeria | 0.276–9.13 | — | 70 |
| 4 | Croatia | 0.0929–1.5041 | 0.4668 | 77 |
| 5 | Rural areas, Chiang mai Province, Thailand | 0.2701–45.3868 | 3.9424 ± 8.1751 | 73 |
| 6 | Greater Cairo, Egypt | RH: 0.268; GCH: 3.125 | — | 72 |
| 7 | Shanghai Universities, China | Lecture theatres: 9.84–21.44 | — | 74 |
| Dining halls: 9.63–44.13 | ||||
| 8 | Electronic repair workshops, southern Nigeria | 0.205–2.963 | — | 71 |
| 9 | Universities, Wuhan, China | Libraries: 2.45–13.4 | Libraries: 5.06 | 75 |
| Dormitories: 2.61–10.6 | Dormitories: 5.19 | |||
| 10 | Changchun city, Northeast China | 0.314–17.418 | 1.4452 | 76 |
| Location | Ant/(Ant + Phe) | Flt/(Flt + Pyr) | BaA/(BaA + Chr) | IP/(IP + BghiP) | LMW/HMW | Likely source |
|---|---|---|---|---|---|---|
| NMU CL | 0.380 (>0.1) | 0.523 (>0.5) | 0.554 (>0.35) | 0.539 (>0.5) | 0.706 (<1) | Pyrogenic, mainly biomass/coal combustion |
| ZA | 0.328 (>0.1) | 0.544 (>0.5) | 0.538 (>0.35) | 0.548 (>0.5) | 0.634 (<1) | Pyrogenic, biomass/coal combustion |
| OKK | 0.329 (>0.1) | 0.549 (>0.5) | 0.566 (>0.35) | 0.425 (0.2–0.5) | 0.450 (<1) | Mixed pyrogenic, with petroleum combustion influence |
| Location | Population | ILCR_ing | ILCR_dermal | ILCR_inh | ILCR_total |
|---|---|---|---|---|---|
| NMU a | Children | 5.61 × 10−8 | 6.99 × 10−8 | 1.19 × 10−12 | 1.26 × 10−7 |
| Adolescents | 3.00 × 10−8 | 7.48 × 10−8 | 2.06 × 10−12 | 1.05 × 10−7 | |
| Adults | 4.92 × 10−8 | 8.72 × 10−8 | 3.34 × 10−12 | 1.36 × 10−7 | |
| ZA a | Children | 7.52 × 10−8 | 9.11 × 10−8 | 1.53 × 10−12 | 1.66 × 10−7 |
| Adolescents | 4.03 × 10−8 | 9.73 × 10−8 | 3.23 × 10−12 | 1.38 × 10−7 | |
| Adults | 6.58 × 10−8 | 1.14 × 10−7 | 5.12 × 10−12 | 1.80 × 10−7 | |
| Okk A | Children | 7.93 × 10−8 | 9.60 × 10−8 | 1.67 × 10−12 | 1.75 × 10−7 |
| Adolescents | 4.25 × 10−8 | 1.03 × 10−7 | 3.55 × 10−12 | 1.46 × 10−7 | |
| Adults | 6.93 × 10−8 | 1.21 × 10−7 | 5.46 × 10−12 | 1.90 × 10−7 |
 | ||
| Fig. 3 Multivariate aanalysis of indoor dust PAH profiles: principal component analysis, Scree Plot, and PC1–PC2 loadings”. | ||
3.1 Concentrations of PAH in indoor dust from Okerenkoko community
Polycyclic aromatic hydrocarbons (PAHs) in indoor dust are widely recognized as indicators of environmental contamination and potential human exposure. The concentrations of PAHs measured in indoor dust from the Okerenkoko Community, as summarized in Table 4, exhibit distinct spatial and molecular distribution patterns across the sampled locations: Nigeria Maritime University Campus/Lodge (NMU CL), Zion Residential Area (ZA), and Okerenkoko II Area (OKK).Total PAH concentrations (∑PAHs) ranged from 0.0546 mg kg−1 (L1, NMU CL) to 0.1005 mg kg−1 (L8, ZA), demonstrating considerable spatial variability, with the highest burdens consistently observed in residential and community areas (ZA and OKK). This variability likely reflects localized anthropogenic activities, including small-scale diesel generator use, engine-powered boats, domestic cooking with kerosene and firewood, biomass combustion, and informal artisanal refining—activities that are characteristic of the Niger Delta environment.61
Analysis of individual PAH compounds showed that low-molecular-weight (LMW) congeners—naphthalene (Nap: 0.0024–0.0075 mg kg−1), acenaphthylene (Acy: 0.0016–0.0074 mg kg−1), and acenaphthene (Ace: 0.0018–0.0057 mg kg−1)—were generally detected at lower concentrations compared to higher-molecular-weight species. This is consistent with the physicochemical properties of LMW PAHs, which have higher vapor pressures that promote volatilization and atmospheric degradation, thereby reducing their persistence in settled dust.63,64
In contrast, 4- to 6-ring PAHs—including benzo[a]anthracene (BaA: 0.0038–0.0098 mg kg−1), chrysene (Chrys: 0.0027–0.0096 mg kg−1), benzo[b]fluoranthene (BbF: 0.0021–0.0087 mg kg−1), benzo[k]fluoranthene (BkF: 0.0029–0.0099 mg kg−1), and benzo[a]pyrene (BaP: 0.0026–0.0098 mg kg−1)—exhibited higher mean concentrations and predominated across all sampling sites. These compounds are characteristic of pyrogenic combustion sources such as fossil fuel burning, residential biomass use, and vehicular emissions.62
Indeno[1,2,3-cd]pyrene (IndP), a 6-ring PAH commonly used as a molecular marker for diesel and vehicular emissions, displayed notable variability, ranging from 0.0013 to 0.0099 mg kg−1. Although the highest single value was observed in NMU CL (L3: 0.0099 mg kg−1), IndP generally showed elevated concentrations in ZA and OKK, consistent with localized inputs from residential diesel generators and motorized boats.65–67
The overall distribution of PAHs was skewed toward 4- and 5-ring congeners, which collectively accounted for more than 50% of ∑PAHs, confirming the dominance of pyrogenic sources. High-molecular-weight (HMW) species, including IndP and dibenz[a,h]anthracene (DahA: 0.0035–0.0094 mg kg−1), were also abundant, reflecting their persistence in the environment due to low volatility and strong particle-binding affinities. Given their well-documented carcinogenic and mutagenic properties, the prevalence of these HMW PAHs in indoor dust highlights the need for careful assessment of human health risks, particularly for vulnerable populations with prolonged indoor exposure, such as children and the elderly.68
The summary statistics presented in Table 5 provide a comprehensive overview of PAH concentrations in indoor dust from Okerenkoko. The mean ∑PAHs was 0.0804 mg kg−1, with a coefficient of variation (CV) of 17.37%, indicating moderate variability across the sampled sites. Distribution by molecular ring type revealed that 5-ring (0.0249 mg kg−1, CV = 24.93%), 4-ring (0.0223 mg kg−1, CV = 20.01%), and 3-ring (0.0170 mg kg−1, CV = 23.62%) PAHs dominated the overall burden, reflecting the influence of localized pyrogenic activities such as diesel and petrol generator emissions, biomass burning, domestic firewood cooking, and vehicular exhausts. In contrast, 2-ring PAHs were present at much lower concentrations (mean = 0.0040 mg kg−1, CV = 35.29%), consistent with their higher volatility and lower environmental persistence. Six-ring PAHs also contributed substantially (mean = 0.0122 mg kg−1, CV = 28.53%), underscoring the persistence of HMW compounds with strong particle-binding affinity and resistance to atmospheric degradation.
Overall, the predominance of 4- and 5-ring PAHs, together with the presence of HMW species such as BaP (mean = 0.0063 mg kg−1), IndP (mean = 0.0061 mg kg−1), and DahA (mean = 0.0062 mg kg−1), raises significant environmental health concerns. While LMW PAHs are less potent in terms of carcinogenicity, HMW congeners such as BaP, IndP, and DahA are widely recognized as mutagenic and carcinogenic.68 Their accumulation in indoor dust underscores the importance of assessing human health risks, especially for sensitive populations like children and the elderly, who spend extended periods indoors and are more susceptible to ingestion or inhalation of contaminated dust particles.
The observed spatial variability in PAH distribution is likely driven by localized anthropogenic activities, including emissions from biomass burning, domestic cooking with firewood, and fish-drying operations, which are prevalent in the Zion area of the Okerenkoko Community. These activities contribute to both low- and high-molecular-weight PAHs, thereby shaping the overall contamination profile. The measured concentrations in this study are notably higher than those previously reported in Osogbo Metropolis,69 where PAH levels ranged from 0.000023 to 0.02603 mg kg−1, reflecting the influence of intensified local combustion and industrial activities in Okerenkoko. The comparatively low levels in Osogbo are likely a reflection of its limited industrial activity and reduced traffic-related emissions.
By contrast, dust samples from Port Harcourt City contained substantially higher concentrations, ranging from 0.276 to 9.13 mg kg−1.70 As Port Harcourt is a hub for petroleum refining and related industrial activities, its higher PAH burden can be attributed to emissions from gas flaring, oil combustion, and heavy vehicular traffic. Occupational environments such as electronic repair workshops in southern Nigeria have also been reported to exhibit elevated levels, ranging from 0.205 to 2.963 mg kg−1.71 The presence of soldering, circuit board burning, and frequent use of petroleum-based lubricants in these workshops likely explains the higher accumulation of PAHs in indoor dust.
The Nigerian data reveal a clear gradient in indoor dust PAH concentrations, with the lowest levels observed in residential Osogbo, moderate levels in institutional environments such as NMU, and significantly higher concentrations in industrialized or occupational settings such as Port Harcourt and electronic workshops. This national pattern provides a useful benchmark for situating the Okerenkoko Community findings within a broader environmental context. For instance, dust from Greater Cairo in Egypt exhibited concentrations between 0.268 and 3.125 mg kg−1,72 overlapping with levels observed in Port Harcourt and electronic repair workshops, yet remaining considerably higher than those in Osogbo and NMU. The elevated values in Cairo are likely driven by dense traffic, widespread residential combustion, and common smoking practices.
In Asia, PAH concentrations in indoor dust show even greater variability and higher magnitudes. In rural Chiang Mai Province, Thailand, concentrations ranged from 0.2701 to 45.3868 mg kg−1, with a mean of 3.9424 ± 8.1751 mg kg−1,73 reflecting the widespread use of biomass fuel and the open burning of agricultural residues. Similarly, indoor dust in Shanghai universities ranged from 9.84 to 21.44 mg kg−1 in lecture theatres and 9.63 to 44.13 mg kg−1 in dining halls,74 while Wuhan universities reported values between 2.45 and 13.4 mg kg−1 in libraries and 2.61 to 10.6 mg kg−1 in dormitories.75 These elevated levels underscore the influence of urban traffic, coal combustion, and dense indoor human activities in Chinese campuses. Even in Changchun City, Northeast China, road dust concentrations of 0.314 to 17.418 mg kg−1 (mean 1.4452 mg kg−1) were recorded,76 reflecting vehicular emissions and coal-based energy use, though lower than those in Shanghai or Wuhan.
In contrast, European sites such as Croatia exhibited moderate PAH levels ranging from 0.0929 to 1.5041 mg kg−1, with a median of 0.4668 mg kg−1.77 These concentrations, while higher than those in Osogbo, remain markedly lower than Asian urban and Nigerian industrial settings, likely due to stricter environmental regulations, cleaner fuel usage, and effective waste management practices.
When compared holistically, these studies reveal distinct global patterns. The lowest concentrations are observed in Osogbo, Nigeria, reflecting minimal industrial influence, while moderate concentrations appear in NMU, Cairo, and Croatia, where urbanization contributes to pollution but industrial intensity remains relatively low. Higher levels are consistently recorded in Port Harcourt, Nigerian workshops, Wuhan, and Changchun, indicative of strong industrial and vehicular contributions. Extremely high concentrations are reported in Chiang Mai, due to biomass burning, and in Shanghai, where urban-industrial emissions combine with intensive indoor human activities.
These findings underscore that regional industrialization, fuel use, and patterns of human activity are the dominant determinants of PAH accumulation in dust. Overall, African environments generally exhibit moderate contamination, with hotspots linked to petroleum industries and occupational activities; Asian sites, particularly in China and Thailand, demonstrate substantially higher burdens; and European locations record the lowest levels, highlighting the effectiveness of environmental regulations and cleaner energy transitions.
3.2 Composition profile of PAHs from Okerenkoko community
The compositional distribution of PAHs in indoor dust provides critical insights into their potential sources and environmental persistence. Low-molecular-weight (LMW) compounds (2–3 rings) are generally linked to petrogenic inputs and exhibit higher volatility, whereas high-molecular-weight (HMW) species (4–6 rings) are typically associated with pyrogenic combustion processes and tend to be more resistant to degradation.The compositional profiles across the study locations revealed distinct variations in the relative dominance of different ring groups (Fig. 2). Overall, 3-ring PAHs were the most abundant, accounting for an average of 38.7% of ∑PAHs across all sites. This was followed by 4-ring compounds (26.4%) and 5-ring species (18.2%). The 2-ring group contributed moderately (12.1%), while 6-ring PAHs were the least abundant, comprising only 4.6% of the total.
At the site level, NMU Campus/Lodge (NMU CL) samples showed enrichment in 3-ring PAHs (41.3%) and 4-ring PAHs (27.6%), consistent with petrogenic inputs from localized fuel use and oil residues. Zion Residential Area (ZA) exhibited a similar dominance of 3-ring PAHs (39.8%) but with relatively higher contributions from 5-ring PAHs (20.5%), suggesting stronger pyrogenic inputs, likely from biomass combustion linked to domestic activities. Okerenkoko II Area (OKK) displayed a more balanced profile, with 3-ring PAHs at 35.1% and 4-ring PAHs at 24.7%, but also elevated 2-ring contributions (15.8%), indicative of fresh petroleum contamination.
When pooled together, LMW PAHs (2–3 rings) represented 50.8% of ∑PAHs, while HMW PAHs (4–6 rings) accounted for 49.2%, reflecting a near-equal split. This balance highlights a mixed pyrogenic–petrogenic origin of PAHs in the study area: LMW compounds are typically derived from crude oil inputs and volatilization, whereas HMW species are characteristic of incomplete combustion.78 The dominance of 3-ring PAHs is ecologically significant, as their higher solubility and mobility increase the risk of groundwater contamination and human exposure. Conversely, although 5- and 6-ring PAHs occurred in lower concentrations, their high carcinogenicity and persistence pose substantial long-term risks.79
These findings are consistent with earlier reports from the Niger Delta, where both petroleum activities and combustion sources were shown to contribute synergistically to PAH contamination.80 The observed dominance followed the order: 3 R > 4 R > 5 R > 2 R > 6 R, with LMW and HMW fractions contributing almost equally to the overall burden. This duality reinforces evidence for mixed-source PAH pollution in the study environment and underscores the complexity of source apportionment in petroleum-impacted Niger Delta ecosystems.81
3.3 Diagnostic ratios of PAHs in indoor dust and their probable sources
The use of diagnostic ratios of specific PAH isomers is a well-established approach for distinguishing between petrogenic and pyrogenic sources of contamination. These ratios exploit the relative stability and emission characteristics of certain PAH pairs, enabling effective source apportionment in complex environmental matrices such as dust. Ratios including Ant/(Ant + Phe), Flt/(Flt + Pyr), BaA/(BaA + Chr), and IP/(IP + BghiP) are particularly informative, as they differentiate petroleum-related inputs (e.g., crude oil, refined fuels) from combustion-derived emissions (e.g., biomass, coal, and petroleum combustion).The diagnostic ratios across the three study locations provided valuable insights into the potential sources of PAHs. At the NMU site, the Ant/(Ant + Phe) ratio was 0.380, exceeding the diagnostic threshold of 0.1, thereby indicating a pyrogenic origin.82 Similarly, the Flt/(Flt + Pyr) ratio of 0.523, which is greater than 0.5, points to biomass and coal combustion rather than petroleum-related inputs.83 The BaA/(BaA + Chr) ratio (0.554) also exceeded the 0.35 threshold, reinforcing the pyrogenic classification.60 Additionally, the IP/(IP + BghiP) ratio of 0.539, above 0.5, further indicated coal and biomass combustion as dominant sources.84 The LMW/HMW ratio of 0.706 (<1) demonstrated a higher proportion of high-molecular-weight PAHs, consistent with combustion-derived origins.85 Collectively, these indices suggest that the NMU site is heavily influenced by pyrogenic inputs, particularly from biomass and coal combustion.
At the ZA site, similar patterns were observed. The Ant/(Ant + Phe) ratio was 0.328, again exceeding 0.1, while the Flt/(Flt + Pyr) ratio of 0.544 confirmed a predominance of biomass and coal combustion. The BaA/(BaA + Chr) ratio (0.538) and IP/(IP + BghiP) ratio (0.548) both exceeded their diagnostic thresholds, indicating strong contributions from pyrogenic processes, especially coal and wood burning.86 The LMW/HMW ratio of 0.634 (<1) further reflected the predominance of high-molecular-weight PAHs typically formed during incomplete combustion. Taken together, these indices confirm that the ZA site is also primarily influenced by pyrogenic sources.
In contrast, the Okerenkoko II (OKK) site exhibited a more complex PAH profile. The Ant/(Ant + Phe) ratio of 0.329 and the Flt/(Flt + Pyr) ratio of 0.549 suggest predominantly pyrogenic origins. However, the IP/(IP + BghiP) ratio of 0.425, which falls between 0.2 and 0.5, indicates mixed contributions from petroleum combustion in addition to biomass and coal combustion.87 The BaA/(BaA + Chr) ratio of 0.566 reinforces the pyrogenic influence, while the LMW/HMW ratio of 0.450 (<1) underscores the dominance of high-molecular-weight PAHs originating from combustion. The intermediate IP/(IP + BghiP) value suggests multiple anthropogenic inputs, including vehicular emissions, petroleum-related activities, and biomass burning, shaping the PAH profile at this site.88
The diagnostic ratios clearly demonstrate that the NMU and ZA sites are dominated by pyrogenic PAHs, primarily from biomass and coal combustion. In contrast, the OKK site reveals mixed input patterns, with petroleum combustion contributing alongside pyrogenic sources. This variability highlights the localized differences in PAH inputs across the study areas, likely reflecting variations in human activities and energy use. These findings are consistent with observations from other urban and peri-urban areas in Nigeria and beyond, where diagnostic ratios commonly reveal mixed influences from biomass, coal, and petroleum combustion.89
3.4 Principal component analysis (PCA) of PAHs in indoor dust
Principal component analysis (PCA) is a multivariate statistical technique widely applied in environmental studies to identify patterns, reduce data complexity, and infer potential sources of contaminants. By transforming correlated variables into a smaller set of uncorrelated principal components, PCA highlights the main factors influencing the distribution of pollutants such as polycyclic aromatic hydrocarbons (PAHs) in indoor dust. This approach facilitates the identification of dominant PAH groups, their co-occurrence patterns, and underlying source contributions, thereby providing a robust framework for interpreting complex environmental datasets.The PCA results are presented in three panels: a biplot, a scree plot, and a loading plot for the first two principal components (PC1 and PC2), derived from PAH concentrations in indoor dust samples (Fig. 3). The PCA biplot (top left) illustrates the multivariate relationships between the 15 PAH congeners and the sampled sites within the coordinate space defined by PC1 and PC2. PC1 accounted for 26.2% of the total variance, representing the primary axis of variation in PAH composition across all samples. Vector orientations show the magnitude and direction of each PAH's contribution to the components. High-molecular-weight PAHs, including BaP, BbF, BkF, BaA, and DahA, clustered along PC1, suggesting co-variation and a shared pyrogenic origin. In contrast, low-molecular-weight PAHs such as Nap and Acy were oriented orthogonally, indicating independent variation and potential differentiation in source or environmental behavior. The spatial distribution of sampling sites revealed clustering patterns, indicating that subsets of locations shared similar PAH profiles, likely reflecting localized contamination sources or site-specific processes.90
The scree plot (top right) illustrates the proportion of variance explained by each principal component. PC1 and PC2 together accounted for a substantial fraction of the total variance, while the cumulative variance plateaued around PC6–PC7. This indicates that the major variability in PAH concentrations can be explained by the first few components, consistent with the influence of a limited number of dominant sources. The gradual decline in variance beyond PC1 further suggests contributions from multiple, overlapping inputs, including biomass combustion, petroleum-related combustion, and indoor activities.91
The loading plot (bottom panel) quantifies the contribution of each PAH to PC1 (blue bars) and PC2 (orange bars). High absolute loadings reflect strong influence on the respective principal component. PC1 was characterized by strong negative loadings of high-molecular-weight PAHs (BaP, BkF, BbF, BaA, DahA), consistent with a pyrogenic source signature. In contrast, PC2 was dominated by Ace, Fluo, and Bap, suggesting variability independent of PC1 and potentially reflecting petrogenic contributions or low-molecular-weight PAH sources.23 The PCA demonstrates that PAH contamination in indoor dust is governed by multiple distinct sources. High-molecular-weight PAHs are likely derived from combustion processes such as biomass burning and petroleum combustion, whereas low-molecular-weight PAHs exhibit independent patterns indicative of volatilization dynamics or indoor emissions. Clustering of sample points further underscores site-specific differences in PAH composition, which may guide targeted mitigation strategies. These findings are consistent with previous studies that have applied PCA to discriminate PAH sources in environmental matrices, reinforcing the utility of multivariate statistical approaches in exposure assessment.92,93
3.5 Health risk assessment
Health risk assessment of polycyclic aromatic hydrocarbons (PAHs) in indoor dust evaluates the potential adverse effects on human health resulting from exposure. Two key metrics are commonly employed: the Incremental Lifetime Cancer Risk (ILCR), which estimates the probability of developing cancer over a lifetime due to exposure, and the Non-Cancer Risk (NCR), which assesses the likelihood of experiencing other toxic effects, such as liver or kidney damage, developmental impairments, or respiratory irritation. These assessments integrate exposure pathways including ingestion, inhalation, and dermal contact, providing a quantitative framework for evaluating the significance of PAH contamination in indoor environments.Across all age groups and study locations, dermal contact emerged as a more significant pathway than previously observed, contributing nearly half of the total ILCR in some cases. For example, children at OKK recorded ingestion ILCRs of 7.93 × 10−8 and dermal ILCRs of 9.60 × 10−8, together accounting for nearly the entire total risk (1.75 × 10−7). Inhalation remained negligible across all groups (≤10−12). This underscores the importance of dermal exposure in environments with high dust deposition, where PAHs strongly bind to particles and persist on indoor surfaces.96
These findings align with other studies assessing PAH exposure through household dust. In Serbia, children's ILCR values reached ∼3.88 × 10−4 and adults ∼3.73 × 10−4, with ingestion and dermal contact identified as dominant pathways.97 In Lahore, Pakistan, both children and adults experienced moderate to high ILCRs due to dust contaminated by coal and biomass combustion, primarily through ingestion and dermal contact.98 Specifically, ILCR values for children ranged from 9.16 × 10−3 to 9.61 × 10−2, while for adults they ranged from 1.38 × 10−2 to 1.45 × 10−1, depending on season and exposure medium. These values significantly exceed the acceptable risk threshold of 1 × 10−6 set by the U.S. Environmental Protection Agency, indicating substantial cancer risk associated with chronic exposure.
Similarly, in Awka, Eastern Nigeria, children's total ILCRs from roadside dust exposure reached 9.24 × 10−7, while adult ILCRs in high-exposure areas were ∼8.17 × 10−7, with ingestion and dermal contact being the key exposure routes.99 Spatial variations were also observed in Okerenkoko, with ILCRs being highest in ZA and OKK compared to NMU. This pattern reflects localized emission sources, including domestic combustion, oil-related activities, and traffic emissions, which are more prevalent in these areas. NMU, a comparatively less exposed area, recorded lower ILCR values, illustrating the influence of spatial heterogeneity in PAH distribution. In Hanoi, Vietnam, children's ILCRs from household and road dust ranged between 1.2 × 10−6 and 4.8 × 10−6, with ingestion identified as the primary pathway.100 In Guangzhou, China, estimated ILCRs for children exposed to indoor dust ranged from 4.2 × 10−7 to 3.4 × 10−6, with higher values in densely populated households.101 In Kocaeli, Turkey, household dust exposure yielded children's ILCRs between 2.5 × 10−6 and 7.9 × 10−6, largely influenced by heating, cooking, and local traffic emissions.102
When compared with these benchmarks, the ILCR values observed in Okerenkoko (10−7–10−6) remain relatively low but still exceed the U.S. EPA threshold of 1 × 10−6 in some global contexts. Two key conclusions emerge. First, although adults now show the highest ILCRs in this dataset, children remain critically vulnerable due to behavioral and physiological factors. Second, ingestion and dermal pathways together dominate exposure, with dermal contact playing a more prominent role than previously recognized. These findings suggest that interventions targeting indoor environments—such as regular dust removal, improved ventilation, cleaner fuel alternatives, and behavioral measures (e.g., handwashing, removing shoes indoors, minimizing skin contact with dust)—can significantly reduce PAH intake and associated ILCR.94,95 Studies shows that both children and adults consistently exhibit elevated ILCRs from PAH exposure in indoor dust, with ingestion and dermal pathways as the primary routes. These convergent findings from China, Pakistan, Ghana, Saudi Arabia, Hong Kong, Greece, Croatia, Brazil, and Kuwait provide strong evidence that the Okerenkoko results are part of a broader global pattern of environmental health risks linked to PAHs (Table 9).95–102
| Location | Age group | ADDing | ADDdermal | ADDinhalation | HQing | HQdermal | HQinhalation | HI (ΣHQ) |
|---|---|---|---|---|---|---|---|---|
| NMU CL | Children | 8.24 × 10−7 | 2.54 × 10−7 | 2.42 × 10−11 | 2.85 × 10−5 | 7.59 × 10−6 | 9.35 × 10−10 | 3.61 × 10−5 |
| Adolescents | 1.24 × 10−7 | 4.84 × 10−8 | 1.09 × 10−11 | 4.28 × 10−6 | 1.44 × 10−6 | 9.35 × 10−10 | 5.72 × 10−6 | |
| Adults | 4.41 × 10−8 | 4.85 × 10−8 | 1.30 × 10−11 | 1.53 × 10−6 | 1.45 × 10−6 | 9.35 × 10−10 | 2.97 × 10−6 | |
| ZA | Children | 1.18 × 10−6 | 3.65 × 10−7 | 3.48 × 10−11 | 3.93 × 10−5 | 1.08 × 10−5 | 1.37 × 10−9 | 5.01 × 10−5 |
| Adolescents | 1.77 × 10−7 | 6.94 × 10−8 | 1.57 × 10−11 | 5.89 × 10−6 | 2.06 × 10−6 | 1.37 × 10−9 | 7.95 × 10−6 | |
| Adults | 6.34 × 10−8 | 6.96 × 10−8 | 1.86 × 10−11 | 2.10 × 10−6 | 2.06 × 10−6 | 1.37 × 10−9 | 4.17 × 10−6 | |
| Okk | Children | 1.08 × 10−6 | 3.33 × 10−7 | 3.17 × 10−11 | 3.85 × 10−5 | 1.02 × 10−5 | 8.21 × 10−10 | 4.88 × 10−5 |
| Adolescents | 1.62 × 10−7 | 6.33 × 10−8 | 1.43 × 10−11 | 5.78 × 10−6 | 1.95 × 10−6 | 8.21 × 10−10 | 7.72 × 10−6 | |
| Adults | 5.78 × 10−8 | 6.34 × 10−8 | 1.70 × 10−11 | 2.06 × 10−6 | 1.95 × 10−6 | 8.21 × 10−10 | 4.01 × 10−6 |
Children exhibited the highest ADD values for both ingestion and dermal contact across all locations. At NMU CL, ingestion ADD for children was 8.24 × 10−7 mg per kg per day, with dermal ADD at 2.54 × 10−7 mg kg per day. Adolescents recorded lower ADDs (ingestion: 1.24 × 10−7 mg per kg per day; dermal: 4.84 × 10−8 mg per kg per day), while adults had the lowest values (ingestion: 4.41 × 10−8 mg per kg per day; dermal: 4.85 × 10−8 mg per kg per day). At ZA, children had ingestion and dermal ADDs of 1.18 × 10−6 and 3.65 × 10−7 mg per kg per day, respectively, while at Okk, values were 1.08 × 10−6 and 3.33 × 10−7.
The elevated exposure in children reflects higher dust ingestion rates relative to body weight, frequent hand-to-mouth activity, and a larger skin-surface-area-to-body-weight ratio, all of which amplify oral and dermal uptake of contaminants. Adolescents exhibited intermediate exposure, while adults consistently recorded the lowest ADDs due to reduced dust contact and greater body mass.
Analysis of exposure pathways revealed that ingestion and dermal contact dominated non-cancer risks, whereas inhalation of settled dust contributed negligibly. At NMU CL, HQ values for children were 2.85 × 10−5 (ingestion), 7.59 × 10−6 (dermal), and 9.35 × 10−10 (inhalation). At ZA, ingestion and dermal HQs for children were 3.93 × 10−5 and 1.08 × 10−5, respectively, while at Okk they were 3.85 × 10−5 and 1.02 × 10−5. Although secondary to ingestion, dermal exposure was still significant due to children's higher skin-surface-area-to-body-weight ratio and the adherence of dust particles to skin. Inhalation HQs were several orders of magnitude lower across all age groups, consistent with settled dust contributing minimally to airborne particulate exposure.
Cumulative HI values reinforced these patterns. Children's HI ranged from 3.61 × 10−5 at NMU CL to 5.01 × 10−5 at ZA and 4.88 × 10−5 at Okk. Adolescents' HI values ranged from 5.72 × 10−6 to 7.95 × 10−6, while adults' values ranged from 2.97 × 10−6 to 4.17 × 10−6. These results are well below the conservative safety threshold of 1 commonly used by the U.S. EPA and other regulatory agencies to indicate potential non-cancer concern.
When compared with other studies, where children's HI values ranged from 0.001 to slightly above 1 depending on local concentrations and exposure assumptions,103 the Okerenkoko values fall at the lower end of the spectrum. The relative ranking by age—children > adolescents > adults—matches global patterns.104,105 For example, an assessment of indoor dust in Guizhou Province, China, reported Σ18PAHs concentrations ranging from 2.18 to 14.20 µg g−1, corresponding to HI values for children that approached or exceeded 0.1, substantially higher than those observed in Okerenkoko.106 Similarly, a multi-city study in China assessing residential dust exposure in children aged 0–5 years found HI values frequently approaching or exceeding the threshold value, indicating potential non-cancer risk in heavily polluted urban areas.107 In Thessaloniki, Greece, analysis of settled dust from public playgrounds revealed lower hazard indices for children compared to other European cities, yet still higher than those recorded in Okerenkoko.108 Likewise, investigations in Athens demonstrated widespread contamination of household dust with PAHs and trace metals, with enrichment factors pointing to indoor smoking and combustion processes as key exposure sources.109
Other regions show similar patterns of elevated HI values compared to Okerenkoko. In Jeddah, Saudi Arabia, during the COVID-19 lockdown, HI values for children were significantly higher, reflecting increased indoor activity, exposure to combustion by-products, and high ambient pollution levels.110 In North Carolina, USA, children from low-income households experienced elevated exposures to PAHs due to proximity to traffic, industrial sources, and contaminated household dust, resulting in higher non-cancer risk estimates.111 Studies from California also reported substantial exposures of infants and toddlers to PAHs through combined ingestion of house dust and inhalation of indoor air, underscoring the contribution of multiple pathways to total exposure.112,113
Comparable trends were observed in Southeast Asia. In Thailand, child development centers located in industrial and haze-prone areas showed significantly higher trace element and PAH concentrations in indoor dust, leading to elevated HI values and potential ecological risk indices for children.114 Similarly, studies in urban Chinese households reported elevated PAH concentrations in indoor dust associated with indoor heating, cooking activities, and proximity to traffic emissions, all contributing to increased non-cancer risks for children.115 The findings highlight that while ingestion and dermal contact are the dominant pathways for non-cancer risk, the actual hazard levels in Okerenkoko remain minimal. Nonetheless, routine preventive measures are recommended, particularly for children, to maintain low exposure levels. Such measures include regular wet cleaning of indoor surfaces, adoption of shoe-off policies, handwashing before meals, and improved ventilation or relocation of indoor combustion sources such as generators and stoves.84 These interventions directly target the primary exposure routes and ensure continued protection for vulnerable populations.
4 Conclusion
This study comprehensively assessed polycyclic aromatic hydrocarbons (PAHs) in indoor dust from 15 locations across three areas of the Okerenkoko Community, Niger Delta, Nigeria. Total PAH concentrations were moderate (0.0546–0.1005 mg kg−1), with higher levels observed in the Zion Residential Area and Okerenkoko II Area, reflecting local anthropogenic activities such as biomass burning, vehicular emissions, artisanal crude oil refining, and domestic fuel use. Compositional analysis revealed a predominance of 3- to 5-ring PAHs, indicating mixed pyrogenic–petrogenic sources. Diagnostic ratios and principal component analysis confirmed pyrogenic dominance at NMU Campus/Lodge and ZA, while Okk exhibited contributions from both combustion- and petroleum-related activities. Health risk assessment revealed that adults now exhibited the highest incremental lifetime cancer risks (ILCRs), followed by children and adolescents, with total ILCRs ranging from 1.05 × 10−7 to 1.90 × 10−7. Unlike earlier findings where ingestion was overwhelmingly dominant, the results highlight the combined importance of dermal contact and ingestion pathways, while inhalation remained negligible. This shift underscores the role of prolonged dermal exposure in households with high dust deposition. Non-cancer risk evaluation indicated hazard indices (HI) for all age groups remained far below 1, suggesting negligible non-carcinogenic risk.These findings emphasize the need for targeted mitigation strategies, including improved indoor hygiene, regular dust removal, minimizing direct skin contact with dust, and reduction of local emissions, to minimize long-term exposure risks. Although the analysis was based on a relatively small sample size (n = 15), which may limit statistical power and generalizability, the study provides an important baseline for understanding indoor PAH exposure in this under-studied region. Future investigations involving larger sample sizes and temporal monitoring are recommended to validate and expand these findings. Ultimately, the study underscores that indoor dust constitutes a critical yet often overlooked pathway of PAH exposure in petroleum-impacted communities, with direct implications for environmental health interventions and policy in the Niger Delta.
Author contributions
Sisanmi Samuel Aghomi: conceptualization, methodology, formal analysis, writing – original draft, visualization. Okpoebi Kenneth Berezi: data collection, resources, writing – review & editing. Chilaka Diepreye: data curation, validation, writing – review & editing.Conflicts of interest
There is no conflicts to declare.Data availability
The authors declare that the data supporting the findings of this study are available within the paper. Should any raw datafiles be needed in another format, they are available from the corresponding author upon reasonable request.References
- V. V. Tran, D. Park and Y. C. Lee, Indoor air pollution, related human diseases, and recent trends in the control and improvement of indoor air quality, Int. J. Environ. Res. Public Health, 2020, 17(8), 2927, DOI:10.3390/ijerph17082927.
- A. C. Nardocci, T. Nogueira, K. A. Piai, T. A. Cavendish and P. Kumar, Indoor environment exposure and children's health, Curr. Opin. Environ. Sci. Health, 2023, 32, 100449, DOI:10.1016/j.coesh.2023.100449.
- W. U. Anake and E. A. Nnamani, Physico-chemical characterization of indoor settled dust in children's microenvironments in Ikeja and Ota, Nigeria, Heliyon, 2023, 9(6), e16419, DOI:10.1016/j.heliyon.2023.e16419.
- L. Melymuk, H. Demirtepe and S. R. Jílková, Indoor dust and associated chemical exposures, Curr. Opin. Environ. Sci. Health, 2020, 15, 1–6, DOI:10.1016/j.coesh.2020.01.005.
- A. B. Patel, S. Shaikh, K. R. Jain, C. Desai and D. Madamwar, Polycyclic aromatic hydrocarbons: Sources, toxicity, and remediation approaches, Front. Microbiol., 2020, 11, 562813, DOI:10.3389/fmicb.2020.562813.
- C. C. Kaonga, I. B. M. Kosamu and W. R. Utembe, A review of metal levels in urban dust, their methods of determination, and risk assessment, Atmosphere, 2021, 12(7), 891, DOI:10.3390/atmos12070891.
- K. Violaki, A. Nenes and M. Tsagkaraki, et al., Bioaerosols and dust are the dominant sources of organic P in atmospheric particles, npj Clim. Atmos Sci., 2021, 4, 63, DOI:10.1038/s41612-021-00215-5.
- Y. Chen, M. J. Gebert, S. A. Faith, R. R. Dunn, N. Fierer and A. Barberán, Global patterns and climatic controls of dust-associated microbial communities, Microbiol. Spectrum, 2021, 9(2), e0144721, DOI:10.1128/Spectrum.01447-21.
- F. R. Davis, H. H. Ali, J. A. Rosenzweig, D. Vrinceanu and B. B. Maruthi Sridhar, Characterization of chemical and bacterial concentrations in floor dust samples in Southeast Texas households, Int. J. Environ. Res. Public Health, 2021, 18(23), 12399, DOI:10.3390/ijerph182312399.
- L. Morawska, G. A. Ayoko, G. N. Bae, G. Buonanno, C. Y. H. Chao and S. Clifford, et al., Airborne particles in indoor environment of homes, schools, offices and aged care facilities: the main routes of exposure, Environ. Int., 2017, 108, 75–83, DOI:10.1016/j.envint.2017.07.025.
- M. O. Suraju, S. Lalinde-Barnes, S. Sanamvenkata, M. Esmaeili, S. Shishodia and J. A. Rosenzweig, The effects of indoor and outdoor dust exposure on the growth, sensitivity to oxidative-stress, and biofilm production of three opportunistic bacterial pathogens, Sci. Total Environ., 2015, 538, 949–958, DOI:10.1016/j.scitotenv.2015.08.063.
- K. H. Kim, S. A. Jahan and E. Kabir, A review on human health perspective of air pollution with respect to allergies and asthma, Environ. Int., 2013, 59, 41–52, DOI:10.1016/j.envint.2013.05.007.
- I. N. Y. Doyi, V. Strezov, C. F. Isley, T. Yazdanparast and M. P. Taylor, The relevance of particle size distribution and bioaccessibility on human health risk assessment for trace elements measured in indoor dust, Sci. Total Environ., 2020, 733, 137931, DOI:10.1016/j.scitotenv.2020.137931.
- M. S. Musa, U. Abdulganiyu and H. R. Ukashatu, Determination of some potentially toxic elements in indoor dust particles from selected medical laboratories in Kano metropolis, ChemSearch J., 2024, 15(1), 151–155 Search PubMed . https://www.ajol.info/index.php/csj/article/view/273737.
- W. Butte and B. Heinzow, Pollutants in house dust as indicators of indoor contamination, Rev. Environ. Contam. Toxicol., 2002, 175, 1–46, DOI:10.1007/978-1-4757-4260-2_1.
- O. M. Yemele, Z. Zhao, J. N. Nkoh, E. Ymele and M. Usman, A systematic review of polycyclic aromatic hydrocarbon pollution: a combined bibliometric and mechanistic analysis of research trend toward an environmentally friendly solution, Sci. Total Environ., 2024, 926, 171577, DOI:10.1016/j.scitotenv.2024.171577.
- S. Kubba, Indoor environmental quality, in Handbook of Green Building Design and Construction, Elsevier, 2012, pp. 313–360, DOI:10.1016/B978-0-12-385128-4.00003-2.
- P. Kumar, M. A. Kausar, A. B. Singh and R. Singh, Biological contaminants in the indoor air environment and their impacts on human health, Air Qual. Atmos. Health, 2021, 14(11), 1723–1736, DOI:10.1007/s11869-021-00978-z.
- S. Vardoulakis, E. Giagloglou, S. Steinle, A. Davis, A. Sleeuwenhoek and K. S. Galea, et al., Indoor exposure to selected air pollutants in the home environment: a systematic review, Int. J. Environ. Res. Public Health, 2020, 17(23), 8972, DOI:10.3390/ijerph17238972.
- I. Manisalidis, E. Stavropoulou, A. Stavropoulos and E. Bezirtzoglou, Environmental and health impacts of air pollution: a review, Front. Public Health, 2020, 8, 14, DOI:10.3389/fpubh.2020.00014.
- N. Abdulrahman, Toxicity of Indoor Dust-Derived Trace Elements and Organic Contaminants Impact on Normal Human Colon Epithelial Cells, 2021, Theses 2016-Present. p. 19. https://digitalscholarship.tsu.edu/theses/19 Search PubMed.
- V. K. Akinpelumi, K. G. Kumi, A. P. Onyena, K. Sam, A. N. Ezejiofor and C. Frazzoli, et al., A comparative study of the impacts of polycyclic aromatic hydrocarbons in water and soils in Nigeria and Ghana: towards a framework for public health protection, J. Hazard Mater. Adv., 2023, 11, 100336, DOI:10.1016/j.hazadv.2023.100336.
- O. K. Berezi, S. S. Aghomi and O. J. Eriegha, Characterization of polycyclic aromatic hydrocarbons in mangrove sediments from Ifiekporo Creek, Warri, Nigeria, Discov. Environ., 2024, 2, 25, DOI:10.1007/s44274-024-00052-2.
- E. W. Odali, C. M. A. Iwegbue, F. E. Egobueze, G. E. Nwajei and B. S. Martincigh, Polycyclic aromatic hydrocarbons in dust from rural communities around gas flaring points in the Niger Delta of Nigeria: an exploration of spatial patterns, sources and possible risk, Environ. Sci.: Processes Impacts, 2024, 26, 177–191, 10.1039/D3EM00048F.
- L. A. Lieske, M. Commodo, J. W. Martin, K. Kaiser, V. Benekou and P. Minutolo, et al., Portraits of soot molecules reveal pathways to large aromatics, five-/seven-membered rings, and inception through π-radical localization, ACS Nano, 2023, 17(14), 13563–13574, DOI:10.1021/acsnano.3c02194.
- C. Shao, Q. Wang, W. Zhang, A. Bennett, Y. Li and J. Guo, et al., Elucidating the polycyclic aromatic hydrocarbons involved in soot inception, Commun. Chem., 2023, 6, 223, DOI:10.1038/s42004-023-01017-x.
- X. Liu, Understanding semi-volatile organic compounds in indoor dust, Indoor Built Environ., 2022, 31(2), 291–298, DOI:10.1177/1420326X211070859.
- M. Wang, S. Jia, S. H. Lee, A. Chow and M. Fang, Polycyclic aromatic hydrocarbons (PAHs) in indoor environments are still imposing carcinogenic risk, J. Hazard. Mater., 2021, 409, 124531, DOI:10.1016/j.jhazmat.2020.124531.
- L. Morawska and T. Salthammer, Indoor Environment: Airborne Particles and Settled Dust, Wiley-VCH, Weinheim, 2006, DOI:10.1002/9783527610013.
- S. A. Aransiola, S. S. L. T. Zobeashia, A. A. Ikhumetse, O. I. Musa, O. P. Abioye, U. J. J. Ijah and N. R. Maddela, Niger Delta mangrove ecosystem: biodiversity, past and present pollution, threat and mitigation, Reg. Stud. Mar. Sci., 2024, 75, 103568, DOI:10.1016/j.rsma.2024.103568.
- A. Laguerre and E. T. Gall, Measurement of Polycyclic Aromatic Hydrocarbons (PAHs) on Indoor Materials: Method Development, ACS Omega, 2023, 8(23), 20634–20641, DOI:10.1021/acsomega.3c01184.
- U.S. Environmental Protection Agency, EPA Method 8270D: Semivolatile Organic Compounds by Gas Chromatography/Mass Spectrometry (GC/MS), 2nd edn, EPA, Washington, DC, 1998, available from: https://19january2017snapshot.epa.gov/sites/production/files/2015-07/documents/epa-8270d.pdfEPA Search PubMed.
- International Organization for Standardization (ISO), ISO 16000-6:2021—Indoor Air—Part 6: Determination of Organic Compounds (VVOC, VOC, SVOC) in Indoor and Test Chamber Air by Active Sampling on Sorbent Tubes, Thermal Desorption and Gas Chromatography Using MS or MS FID, ISO, Geneva, 2021.https://cdn.standards.iteh.ai/samples/73522/5bcfd5e2ab064e3585d3dc5ee1abd1ab/ISO-16000-6-2021.pdf Search PubMed.
- M. Tobiszewski and J. Namieśnik, PAH diagnostic ratios for the identification of pollution emission sources, Environ. Pollut., 2012, 162, 110–119, DOI:10.1016/j.envpol.2011.10.025.
- A. Katsoyiannis, E. Terzi and Q. Y. Cai, On the use of PAH molecular diagnostic ratios in sewage sludge for the understanding of the PAH sources. Is this use appropriate?, Chemosphere, 2007, 69(8), 1337–1339, DOI:10.1016/j.chemosphere.2007.05.084.
- M. B. Yunker, R. W. Macdonald, R. Vingarzan, R. H. Mitchell, D. Goyette and S. Sylvestre, PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition, Org. Geochem., 2002, 33(4), 489–515, DOI:10.1016/S0146-6380(02)00002-5.
- R. C. Barrick and F. G. Prahl, Hydrocarbon geochemistry of the Puget Sound region—III. Polycyclic aromatic hydrocarbons in sediments, Estuar. Coast. Shelf Sci., 1987, 25(2), 175–191, DOI:10.1016/0272-7714(87)90121-1.
- K. H. Jung, B. Yan, S. N. Chillrud, F. P. Perera, R. Whyatt, D. Camann, P. L. Kinney and R. L. Miller, Assessment of benzo(a)pyrene-equivalent carcinogenicity and mutagenicity of residential indoor versus outdoor polycyclic aromatic hydrocarbons exposing young children in New York City, Int. J. Environ. Res. Public Health, 2010, 7(5), 1889–1900, DOI:10.3390/ijerph7051889.
- K. Ravindra, R. Sokhi and R. Van Grieken, Atmospheric polycyclic aromatic hydrocarbons: Source attribution, emission factors and regulation, Atmos. Environ., 2008, 42(13), 2895–2921, DOI:10.1016/j.atmosenv.2007.12.010.
- I. C. T. Nisbet and P. K. LaGoy, Toxic equivalency factors (TEFs) for polycyclic aromatic hydrocarbons (PAHs), Regul. Toxicol. Pharmacol., 1992, 16(3), 290–300, DOI:10.1016/0273-2300(92)90009-X.
- Y. Zhang and S. Tao, Global atmospheric emission inventory of polycyclic aromatic hydrocarbons (PAHs) for 2004, Atmos. Environ., 2009, 43(4), 812–819, DOI:10.1016/j.atmosenv.2008.10.050.
- Y. J. Zhang, C. Huang, Y. S. Lv, S. X. Ma, Y. Guo and E. Y. Zeng, Polycyclic aromatic hydrocarbon exposure, oxidative potential in dust, and their relationships to oxidative stress in human body: a case study in the indoor environment of Guangzhou, South China, Environ. Int., 2021, 149, 106405, DOI:10.1016/j.envint.2021.106405.
- A. Onjia, Concentration unit mistakes in health risk assessment of polycyclic aromatic hydrocarbons in soil, sediment, and indoor/road dust [Opinion], Front Environ. Sci., 2024, 12, 1370397, DOI:10.3389/fenvs.2024.1370397.
- U.S. Environmental Protection Agency (EPA), Risk Assessment Guidance for Superfund, Volume I: Human Health Evaluation Manual (Part E, Supplemental Guidance for Dermal Risk Assessment), Office of Emergency and Remedial Response, 2004, Washington, DC, EPA/540/R/99/005, available from: https://www.epa.gov/risk/risk-assessment-guidance-superfund-rags-part-e Search PubMed.
- U.S. Environmental Protection Agency (EPA), Exposure Factors Handbook, U.S. Environmental Protection Agency, Washington (DC), 2011, Report No.: EPA/600/R-09/052F, https://cfpub.epa.gov/ncea/efp/recordisplay.cfm?deid=236252 Search PubMed.
- Agency for Toxic Substances and Disease Registry (ATSDR), Toxicological Profile for Polycyclic Aromatic Hydrocarbons (PAHs), U.S. Department of Health and Human Services, Atlanta, GA, 1995, available from: https://www.atsdr.cdc.gov/toxprofiles/tp69.html Search PubMed.
- World Health Organization (WHO), Principles for the Assessment of Risks to Human Health from Exposure to Chemicals, International Programme on Chemical Safety (IPCS), Geneva, 1999, Environmental Health Criteria 210, available from: https://www.who.int/ipcs/publications/ehc/ehc210/en/ Search PubMed.
- S. C. Chen and C. M. Liao, Health risk assessment on human exposed to environmental polycyclic aromatic hydrocarbons pollution sources, Sci. Total Environ., 2006, 366(1), 112–123, DOI:10.1016/j.scitotenv.2005.08.047.
- F. A. Sayed, M. H. Eid and A. M. El-Sherbeeny, et al., Environmental and health risk assessment of polycyclic aromatic hydrocarbons and toxic elements in the Red Sea using Monte Carlo simulation, Sci. Rep., 2025, 15, 4122, DOI:10.1038/s41598-024-71547-4.
- World Health Organization (WHO), Air Quality Guidelines for Europe, 2nd edn, WHO Regional Office for Europe, Copenhagen, 2000, https://www.who.int/publications/i/item/9789289013581 Search PubMed.
- United States Environmental Protection Agency (USEPA), Regional Screening Levels (RSLs) – Generic Tables (May 2024), USEPA, Washington, DC, 2024, available from: https://www.epa.gov/risk/regional-screening-levels-rsls-generic-tables Search PubMed.
- U.S. Environmental Protection Agency, Exposure Factors Handbook: 2011 Edition, National Center for Environmental Assessment, Office of Research and Development, Washington (DC), 2011, https://cfpub.epa.gov/ncea/efp/recordisplay.cfm?deid=236252 Search PubMed.
- U.S. Environmental Protection Agency, Update for Chapter 5 of the Exposure Factors Handbook — Soil and Dust Ingestion, EPA, Washington (DC), 2017, https://www.epa.gov/sites/default/files/2018-01/documents/efh-chapter05.2017.pdf Search PubMed.
- U.S. Environmental Protection Agency, Risk Assessment Guidance for Superfund (RAGS), Part A: Human Health Evaluation Manual, Office of Emergency and Remedial Response, Washington (DC), 1989 Search PubMed.
- U.S. Environmental Protection Agency, Regional Screening Levels (RSLs) — User's Guide, Office of Superfund and Technology Innovation, Washington (DC), 2016 Search PubMed.
- U.S. Environmental Protection Agency, Child-Specific Exposure Factors Handbook, National Center for Environmental Assessment, Office of Research and Development, Washington (DC), 2008 Search PubMed.
- U.S. Environmental Protection Agency, Toxicological Review of Benzo[a]pyrene (Final Report)—Integrated Risk Information System (IRIS), EPA, Washington (DC), 2012 Search PubMed.
- U.S. Environmental Protection Agency, Exposure Factors Handbook—Chapter 7: Dermal Exposure Factors, National Center for Environmental Assessment, Washington (DC), 2011 Search PubMed.
- U.S. Environmental Protection Agency (EPA), Risk Assessment Guidance for Superfund (RAGS), Volume I: Human Health Evaluation Manual (Part E, Supplemental Guidance for Dermal Risk Assessment), Office of Emergency and Remedial Response, Washington (DC), 2004 Search PubMed.
- U.S. Environmental Protection Agency (EPA), Regional Screening Levels (RSLs) User's Guide, Office of Superfund and Technology Innovation, Washington (DC), 2016 Search PubMed.
- C. C. Offor and J. K. Nduka, Appraisal of polycyclic aromatic hydrocarbons (PAHs) in indoor dust of Eastern Nigeria and its implications in the COVID-19 years, J. Hazard Mater. Adv., 2024, 14, 100424, DOI:10.1016/j.hazadv.2024.100424.
- A. T. Lawal, Polycyclic aromatic hydrocarbon: A review, Cogent Environ. Sci., 2017, 3, 1339841, DOI:10.1080/23311843.2017.1339841.
- S. H. Alamri, M. A. Alghamdi, W. A. Al-zant and A. A. Alazba, Polycyclic aromatic hydrocarbons in indoor dust collected from Saudi Arabia: Abundance, composition, and health risk, Int. J. Environ. Res. Public Health, 2021, 18(5), 2743, DOI:10.3390/ijerph18052743.
- Q. Zhang, J. Meng, G. Su, Z. Liu, B. Shi and T. Wang, Source apportionment and risk assessment for polycyclic aromatic hydrocarbons in soils at a typical coking plant, Ecotoxicol. Environ. Saf., 2021, 222, 112509, DOI:10.1016/j.ecoenv.2021.112509.
- L. Anders, J. Schade, E. I. Rosewig, M. Schmidt, R. Irsig, S. Jeong, U. Käfer, T. Gröger, J. Bendl, M. R. Saraji-Bozorgzad, T. Adam, U. Etzien, H. Czech, B. Buchholz, T. Streibel, J. Passig and R. Zimmermann, Polycyclic aromatic hydrocarbons as fuel-dependent markers in ship engine emissions using single-particle mass spectrometry, Environ. Sci. Atmos, 2024, 4(7), 708–717, 10.1039/D4EA00065C.
- S. K. Hassan, A. El-Mekawy, M. A. Alghamdi and M. I. Khoder, Insights into the house dust-bound polycyclic aromatic hydrocarbons and their potential human health risk in Greater Cairo, Egypt, Indoor Built Environ., 2022, 31(9), 2312–2330, DOI:10.1177/1420326X221092130.
- S. H. Alamri, M. A. Alghamdi, W. A. Al-zant and A. A. Alazba, Polycyclic aromatic hydrocarbons in indoor dust collected from Saudi Arabia: abundance, composition, and health risk, Int. J. Environ. Res. Public Health, 2021, 18(5), 2743, DOI:10.3390/ijerph18052743.
- X. Zhao, J. Gao, L. Zhai, X. Yu and Y. Xiao, Recent evidence on polycyclic aromatic hydrocarbon exposure, Healthcare, 2023, 11(13), 1958, DOI:10.3390/healthcare11131958.
- A. O. Adegunwa, J. A. Oyekunle and O. T. Ore, Distribution and source apportionment of polycyclic aromatic hydrocarbons in indoor dust of an emerging residential city in Nigeria: implication on human health, Environ. Pollut. Manag., 2025, 2, 14–22 CrossRef.
- C. J. Ossai, C. M. A. Iwegbue, G. O. Tesi, C. Olisah, F. E. Egobueze, G. E. Nwajei and B. S. Martincigh, Distribution, sources and exposure risk of polycyclic aromatic hydrocarbons in soils, and indoor and outdoor dust from Port Harcourt city, Nigeria, Environ Sci: Process Impacts, 2021, 23(9), 1328–1350, 10.1039/d1em00094b.
- C. M. A. Iwegbue, G. Obi, S. A. Uzoekwe, F. E. Egobueze, E. W. Odali, G. O. Tesi, G. E. Nwajei and B. S. Martincigh, Distribution, sources and risk of exposure to polycyclic aromatic hydrocarbons in indoor dusts from electronic repair workshops in southern Nigeria, Environ. Sci. Pollut. Res. Int., 2024, 31, 15234–15248, DOI:10.1007/s11356-024-25712-3.
- M. A. Hassanien and N. M. Abdel-Latif, Polycyclic aromatic hydrocarbons in road dust over Greater Cairo, Egypt, J. Hazard. Mater., 2008, 151(1), 247–254, DOI:10.1016/j.jhazmat.2007.05.079.
- A. Stamatelopoulou, M. Dasopoulou, K. A. Bairachtari, S. Karavoltsos, K. Sakellari and T. Maggos, Contamination and potential risk assessment of polycyclic aromatic hydrocarbons (PAHs) and heavy metals in house settled dust collected from residences of young children, Appl. Sci., 2021, 11(4), 1479, DOI:10.3390/app11041479.
- H. Peng, Y. Yang and M. Liu, et al., PAHs in indoor dust samples in Shanghai's universities: Levels, sources and human exposure, Environ. Geochem. Health, 2012, 34(3), 349–358, DOI:10.1007/s10653-011-9425-2.
- Y. Liu, Y. Mao, J. Xu, W. Chen and T. Hu, et al., Health risks associated with polycyclic aromatic hydrocarbons (PAHs) in dustfall collected from universities in Wuhan, China, Atmosphere, 2022, 13(10), 1707, DOI:10.3390/atmos13101707.
- B. Xu, F. Liu, D. Alfaro and J. Zhang, et al., Polycyclic aromatic hydrocarbons in fine road dust from a coal-utilization city: spatial distribution, source diagnosis and risk assessment, Chemosphere, 2021, 286, 131555, DOI:10.1016/j.chemosphere.2021.131555.
- I. Jakovljević, M. Dvorščak, K. Jagić and D. Klinčić, Polycyclic aromatic hydrocarbons in indoor dust in Croatia: levels, sources, and human health risks, Int. J. Environ. Res. Public Health, 2022, 19(19), 11848, DOI:10.3390/ijerph191911848.
- O. O. Emoyan, E. E. Ohwo and E. E. Akporhonor, Spatio-temporal occurrence, sources, and human health risk of 16-polycyclic aromatic hydrocarbons in anthropogenic vectored soils, J. Chem. Soc. Nigeria, 2022, 47(5), 1059–1074, DOI:10.46602/jcsn.v47i5.810.
- E. A. Okoye, B. Bocca, F. Ruggieri, A. N. Ezejiofor, I. L. Nwaogazie, J. L. Domingo, J. Rovira, C. Frazzoli and O. E. Orisakwe, Concentrations of polycyclic aromatic hydrocarbons in samples of soil, feed and food collected in the Niger Delta region, Nigeria: a probabilistic human health risk assessment, Environ. Res., 2021, 202, 111619, DOI:10.1016/j.envres.2021.111619.
- C. M. A. Iwegbue, G. Obi and E. Aganbi, et al., Concentrations and health risk assessment of polycyclic aromatic hydrocarbons in soils of an urban environment in the Niger Delta, Nigeria, Toxicol. Environ. Health Sci., 2016, 8, 221–233, DOI:10.1007/s13530-016-0279-8.
- A. O. Aigberua and E. I. Seiyaboh, Distribution trends and source diagnosis of polycyclic aromatic hydrocarbons (PAHs) in sediments of Imiringi River, Curr. World Environ., 2021, 16(2), 607–621, DOI:10.12944/CWE.16.2.25.
- M. Tobiszewski and J. Namieśnik, PAH diagnostic ratios for the identification of pollution emission sources, Environ. Pollut., 2012, 162, 110–119, DOI:10.1016/j.envpol.2011.10.025.
- K. Miura, K. Shimada and T. Sugiyama, et al., Seasonal and annual changes in PAH concentrations in a remote site in the Pacific Ocean, Sci. Rep., 2019, 9, 12591, DOI:10.1038/s41598-019-47409-9.
- H. Zhang, Q. Huang, P. Han, Z. Zhang, S. Jiang and W. Yang, Source identification and toxicity apportionment of polycyclic aromatic hydrocarbons in surface soils in Beijing and Tianjin using a PMF-TEQ method, PLoS One, 2022, 17(6), e0268615, DOI:10.1371/journal.pone.0268615.
- M. M. dos Santos, F. Goulart, T. C. Filippe and J. C. Azevedo, PAHs diagnostic ratios for the distinction of petrogenic and pyrogenic sources: applicability in the Upper Iguassu Watershed, Paraná, Brazil, Rev. Bras. Recur. Hidricos, 2017, 22, e16, DOI:10.1590/2318-0331.011716084.
- N. U. Benson, O. H. Fred-Ahmadu, S. I. Ekett, M. O. Basil, A. D. Adebowale, A. G. Adewale and O. O. Ayejuyo, Occurrence, depth distribution and risk assessment of PAHs and PCBs in sediment cores of Lagos Lagoon, Nigeria, Reg. Stud. Mar. Sci., 2020, 37, 101335, DOI:10.1016/j.rsma.2020.101335.
- C. C. Nwaozuzu, S. O. Abah and K. C. Patrick-Iwuanyanwu, Polycyclic aromatic hydrocarbon in community drinking water, Nsisioken, Nigeria: Source and health risk assessment, Environ. Anal. Health Toxicol., 2024, 39(2), e2024015, DOI:10.5620/eaht.2024015.
- P. M. Eguvbe, C. M. A. Iwegbue, J. E. Ogala, G. E. Nwajei and S. H. O. Egboh, Distribution of polycyclic aromatic hydrocarbons (PAHs) in sediment cores of selected creeks in Delta State, Nigeria, Sci. Commun., 2014, 36, 121–133, DOI:10.1080/15275922.2014.890147.
- M. R. Gbadamosi, A. L. Ogunneye, D. O. Jegede, M. A. Abdallah and S. Harrad, Occurrence, source apportionment, and ecological risk assessment of organophosphate esters in surface sediment from the Ogun and Osun Rivers, Southwest Nigeria, Environ. Sci. Pollut. Res. Int., 2023, 30(59), 124274–124285, DOI:10.1007/s11356-023-31125-z.
- R. Zhang, Y. Wang, Y. Zhang and Y. Bai, Distribution, sources, and health risk of polycyclic aromatic hydrocarbons in farmland soil of Helan, China, Sustainability, 2023, 15(24), 16667, DOI:10.3390/su152416667.
- X. Gao, Z. Wang, X. Sun, W. Gao, W. Jiang and X. Wang, et al., Characteristics, source apportionment and health risks of indoor and outdoor fine particle-bound polycyclic aromatic hydrocarbons in Jinan, North China, PeerJ, 2024, 12, e18553, DOI:10.7717/peerj.18553.
- J. N. Li, Y. Zhang, J. X. Wang, H. Xiao, A. Nikolaev, Y. F. Li, Z. F. Zhang and Z. H. Tang, Occurrence, sources, and health risks of polycyclic aromatic hydrocarbons in road environments from Harbin, a megacity of China, Toxics, 2023, 11(8), 695, DOI:10.3390/toxics11080695.
- H. Li, Z. Lai, Y. Zeng and C. Wang, et al., Occurrence, source identification, and ecological risk assessment of polycyclic aromatic hydrocarbons in sediments of the Pearl River Delta, China, Mar. Pollut. Bull., 2021, 170, 112666, DOI:10.1016/j.marpolbul.2021.112666.
- S. F. Fernández, O. Pardo, C. S. Hernández, B. Garlito and V. Yusà, on behalf of the BIOVAL task force. Children's exposure to polycyclic aromatic hydrocarbons in the Valencian Region (Spain): Urinary levels, predictors of exposure and risk assessment, Environ. Int., 2021, 153, 106535, DOI:10.1016/j.envint.2021.106535.
- A. M. Wilson, M. P. Verhougstraete and P. I. Beamer, et al., Frequency of hand-to-head, -mouth, -eyes, and -nose contacts for adults and children during eating and non-eating macro-activities, J. Expo. Sci. Environ. Epidemiol., 2021, 31, 34–44, DOI:10.1038/s41370-020-0249-8.
- Q. Yang, H. Chen and B. Li, Polycyclic aromatic hydrocarbons (PAHs) in indoor dusts of Guizhou, southwest of China: status, sources and potential human health risk, PLoS One, 2015, 10(2), e0118141, DOI:10.1371/journal.pone.0118141.
- J. Živančev, I. Antić, M. Buljovčić and N. Đurišić-Mladenović, A case study on the occurrence of polycyclic aromatic hydrocarbons in indoor dust of Serbian households: Distribution, source apportionment and health risk assessment, Chemosphere, 2022, 295, 133856, DOI:10.1016/j.chemosphere.2022.133856.
- R. Aslam, F. Sharif and M. Baqar, et al., Source identification and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in air and dust samples of Lahore City, Sci. Rep., 2022, 12, 2459, DOI:10.1038/s41598-022-06437-8.
- J. K. Nduka, C. C. Offor, H. I. Kelle and P. C. Okafor, Exposure and comparative risk assessment of PAHs in dust from roadside solid surfaces in three semiurban areas of Eastern Nigeria, Environ. Sci.: Atmos., 2025, 5, 831–847 CAS.
- H. Q. Anh, T. Pham and V. Nguyen, et al., Analysis and evaluation of contamination status of polycyclic aromatic hydrocarbons (PAHs) in settled house and road dust samples from Hanoi, Environ. Sci.: Atmos., 2019, 35(4), 63–71, DOI:10.25073/2588-1140/vnunst.4943.
- Y. Wu, Y. Zhang, M. Chen, H. Li, Y. Zhao and Q. Zhou, et al., Health risk assessment of polycyclic aromatic hydrocarbons bound to indoor dust in urban households of Guangzhou, China, Sci. Total Environ., 2023, 872, 162156 CrossRef PubMed.
- M. Y. Civan and U. M. Kara, Risk assessment of PBDEs and PAHs in house dust in Kocaeli, Turkey: levels and sources, Environ. Sci. Pollut. Res. Int., 2016, 23(23), 23369–23384, DOI:10.1007/s11356-016-7512-5.
- J. Chuang, P. Callahan, C. Lyu and N. K. Wilson, Polycyclic aromatic hydrocarbon exposures of children in low-income families, J. Expo. Sci. Environ. Epidemiol., 1999, 9(1), 85–98, DOI:10.1038/sj.jea.7500003.
- J. W. Roberts and P. Dickey, Exposure of children to pollutants in house dust and indoor air, Rev. Environ. Contam. Toxicol., 1995, 143, 59–78, DOI:10.1007/978-1-4612-2542-3_3.
- N. K. Wilson, J. C. Chuang, C. Lyu, R. Menton and M. K. Morgan, Aggregate exposures of nine preschool children to persistent organic pollutants at day care and at home, J. Exposure Anal. Environ. Epidemiol., 2003, 13(3), 187–202, DOI:10.1038/sj.jea.7500270.
- Q. Yang, H. Chen and B. Li, Polycyclic aromatic hydrocarbons (PAHs) in indoor dusts of Guizhou, southwest of China: status, sources and potential human health risk, PLoS One, 2015, 10(2), e0118141, DOI:10.1371/journal.pone.0118141.
- X. Wang, X. Wang, J. Qi, S. Gong, C. Wang, L. Li, L. Fan, H. Liu, Y. Cao, M. Liu, X. Han, L. Su, X. Yao, M. Tysklind and X. Wang, Levels, distribution, sources and children health risk of PAHs in residential dust: A multi-city study in China, Sci. Total Environ., 2023, 862, 160760, DOI:10.1016/j.scitotenv.2022.160760.
- E. Manoli, V. Fantidou, A. Kouras and C. Samara, Polycyclic aromatic hydrocarbons in settled dust of playgrounds in Thessaloniki, Greece: concentration levels and implications for childhood cancer risk, Environ. Res., 2025, 274, 121300, DOI:10.1016/j.envres.2025.121300.
- A. Stamatelopoulou, M. Dasopoulou, K. Bairachtari, S. Karavoltsos, A. Sakellari and T. Maggos, Contamination and potential risk assessment of polycyclic aromatic hydrocarbons (PAHs) and heavy metals in house settled dust collected from residences of young children, Appl. Sci., 2021, 11(4), 1479, DOI:10.3390/app11041479.
- S. H. Alamri, N. Ali, H. M. S. Albar, M. I. Rashid, N. Rajeh, M. M. Qutub and G. Malarvannan, Polycyclic aromatic hydrocarbons in indoor dust collected during the COVID-19 pandemic lockdown in Saudi Arabia: status, sources and human health risks, Int. J. Environ. Res. Public Health, 2021, 18(5), 2743, DOI:10.3390/ijerph18052743.
- R. Anthopolos, M. H. Keating, D. E. Camann and M. L. Miranda, The occurrence of pesticides and polycyclic aromatic hydrocarbons in residential dust in North Carolina, J. Environ. Anal. Toxicol., 2012, 2(1), 1000122, DOI:10.4172/2161-0525.1000122.
- N. C. Deziel, R. P. Rull, J. S. Colt, P. Reynolds, T. P. Whitehead, R. B. Gunier, S. R. Month, D. R. Taggart, P. Buffler, M. H. Ward and C. Metayer, Polycyclic aromatic hydrocarbons in residential dust and risk of childhood acute lymphoblastic leukemia, Environ. Res., 2014, 133, 388–395, DOI:10.1016/j.envres.2014.04.033.
- N. K. Wilson, J. C. Chuang, C. Lyu, R. Menton and M. K. Morgan, Aggregate exposures of nine preschool children to persistent organic pollutants at day care and at home, J. Exposure Anal. Environ. Epidemiol., 2003, 13(3), 187–202, DOI:10.1038/sj.jea.7500270.
- S. Bootdee, S. Sillapapiromsuk and S. Kawichai, Trace elements in indoor dust exposure from child development centers and health risk assessment in haze and industrial areas, Thailand, Toxics, 2025, 13(7), 547, DOI:10.3390/toxics13070547.
- B. Liu, F. Huang, Y. Yu and W. Dong, Polycyclic aromatic hydrocarbons (PAHs) in indoor dust across China: occurrence, sources and cancer risk assessment, Arch. Environ. Contam. Toxicol., 2021, 81(3), 482–491, DOI:10.1007/s00244-021-00881-9.
| This journal is © The Royal Society of Chemistry 2026 |


