 Open Access Article
Open Access ArticleRed to blue OLEDs based on highly luminescent 1,3-bis(4-phenylpyridin-2-yl)-4,6-difluorobenzene platinum(II) complexes: the key role of substituents on the 4-phenylpyridine unit†
Francesco
Fagnani
 a,
Alessia
Colombo
a,
Alessia
Colombo
 *a,
Giulia
De Soricellis
ab,
Claudia
Dragonetti
*a,
Giulia
De Soricellis
ab,
Claudia
Dragonetti
 a,
Dominique
Roberto
a,
Dominique
Roberto
 a,
Daniele
Marinotto
a,
Daniele
Marinotto
 c,
Véronique
Guerchais
d,
Francesca
Tinti
e and
Massimo
Cocchi
e
c,
Véronique
Guerchais
d,
Francesca
Tinti
e and
Massimo
Cocchi
e
aDipartimento di Chimica dell’Università degli Studi di Milano and UdR-INSTM di Milano, Via C. Golgi 19, I-20133 Milan, Italy. E-mail: alessia.colombo@unimi.it
bDipartimento di Chimica, Università di Pavia, Via Taramelli 12, 27100 Pavia, Italy
cIstituto di Scienze e Tecnologie Chimiche (SCITEC) “Giulio Natta”, Consiglio Nazionale delle Ricerche (CNR), via C. Golgi 19, I-20133 Milan, Italy
dUniversité de Rennes 1, CNRS, ISCR (Institut des Sciences Chimiques de Rennes) - UMR 6226, F-35000 Rennes, France
eIstituto per la Sintesi Organica e la Fotoreattività (ISOF), Consiglio Nazionale delle Ricerche (CNR), via P. Gobetti 101, 40129 Bologna, Italy
First published on 30th January 2026
Abstract
Four 1,3-bis(4-phenylpyridin-2-yl)-4,6-difluorobenzene platinum(II) complexes, bearing various substituents on the phenyl group, were employed for the preparation of novel organic light-emitting diodes (OLEDs): the complex with a simple phenyl substituent, Pt(1,3-bis(4-phenylpyridin-2-yl)-4,6-difluoro-benzene) (PtL1Cl), Pt(1,3-bis(4-(4-tert-butylphenyl)pyridin-2-yl)-4,6-difluoro-benzene)Cl (PtL3Cl), Pt(1,3-bis(4-(3,5-di-tert-butylphenyl)-pyridin-2-yl)-4,6-difluoro-benzene)Cl (PtL4Cl), and Pt(1,3-bis(4-(4-methoxy-2,6-dimethylphenyl)pyridin-2-yl)-4,6-difluoro-benzene)Cl (PtL6Cl). OLEDs fabricated with PtL4Cl as the emitter (6% wt in the emitting layer) produce the highest performing devices, putting in evidence the remarkable effect caused by the steric hindrance of the two tert-butyl groups on the 4-phenylpyridine moieties; it is characterized by an “emerald green” colour emission (CIE coordinates = 0.19, 0.44), a maximun external efficiency (EQEmax) of 20%, and a luminance of ca 23.000 cd m−2 under a 12 V driving voltage. Besides, OLEDs prepared with PtL6Cl (6% wt) exhibit a “sky-blue” colour emission (CIE coordinates = 0.13, 0.26), an EQEmax of 8%, and a luminance of ca 27.000 cd m−2 under a 10 V driving voltage. The design strategy of adding steric bulk to the platinum complexes is a useful route for producing highly efficient OLEDs whereas the introduction of methyls ortho to the interannular C–C bond is a way to go towards blue OLEDs. Besides, red OLEDs are fabricated by using a film of the pure complexes as emitting layer.
1. Introduction
In the last thirty years, coordination compounds have attracted much attention for various fields of photonics such as dye-sensitized solar cells,1–4 photocatalysis,5–8 nonlinear optics,9–12 sensing,13 electroluminescent devices,14–16 bio-imaging,17,18 and photodynamic therapy.19,20 In particular, phosphorescent iridium(III) and platinum(II) complexes are often used as red and green emitters for the fabrication of organic light-emitting diodes (OLEDs), which are renovating the lighting and display industries and find an important application in full-colour displays, whereas fluorescent organic molecules are usually chosen as blue emitters.21–23 Despite their large commercial success, the most important issue for OLEDs devices is their lifetime, which is restricted by the stability of the blue pixel.24,25 The most recent significant advance in the search for novel efficient blue organic emitters is the use of TADF (thermally activated delayed fluorescence) organic compounds26,27 and MRTADF (multiresonant TADF) compounds, typically polycyclic aromatic hydrocarbon molecules containing boron and nitrogen or oxygen.28,29In parallel, although there is a rapid growth of green and red phosphorescent OLEDs, the development of efficient blue ones is still a challenge because the prepared emissive metal complexes either do not possess sufficiently high photoluminescence quantum yields or are characterized by unsatisfactory Commission Internationale de L’Éclairage (CIE) coordinates 1931.24 Recently, promising “deep-blue” emitting OLEDs have been fabricated with iridium(III) carbene complexes (CIE = 0.16, 0.08) with a maximum external quantum efficiency (EQEmax) of 14.9%, which decreases to 11.7% at 100 cd m−2.30
The search for blue-emitting OLEDs based on platinum complexes is of growing interest but remains in its infancy.31,32 Tetradentate Pt(II) complexes show great potential as OLED emitters;33,34 however, their synthesis is often demanding, and precise molecular design to achieve tailored optoelectronic properties presents significant challenges. In contrast, N^C^N tridentate cyclometalated Pt(II) complexes offer relatively simple synthetic accessibility, high photoluminescence quantum yields, and easily tunable optoelectronic properties, making them attractive candidates for further investigation as OLED emitters.
Yet, a lot of work has been devoted to platinum(II) complexes in the field of OLEDs because (i) like for iridium, the important spin–orbit coupling (SOC) associated with the platinum center favors intersystem crossing and therefore radiative decay from triplet excited states and (ii) their square planar geometry promotes the formation of bimolecular species such as dimers (in the ground state) or excimers (in the excited state), by means of Pt⋯Pt or ligand⋯ligand intermolecular interactions, characterized by an emission at lower energies with respect to monomolecular species. These characteristics play a central role not only for the fabrication of deep-red/near infrared or white light emitting materials but also for the preparation of variously colored OLEDs, the emission colour being controlled by the amount of the platinum(II) compound in the emissive layer, due to the parallel emissions coming from the bimolecular and monomolecular excited states.35 In particular, brightly luminescent chlorido platinum(II) compounds bearing a rigid tridentate N^C^N cyclometalated 1,3-bis(pyridin-2-yl)-benzene (bpyb) ligand turned out to be a precious tool for tailoring the emission colour of efficient OLEDs.36,37 They are characterized by two emissive bands, usually located in the bluish-green (phosphorescence from the monomeric species) and red (phosphorescence from excimer and/or aggregate species) regions of the visible spectrum. The contribution of these phosphorescence bands to the total emission can be tuned by the quantity of the platinum(II) complex in the matrix employed for the fabrication of the emissive layer of the OLEDs.38
The appeal of these Pt(bpyb)Cl complexes is enhanced by the ease of synthesis and by the facile control of the emission colour of the monomeric species upon introduction of suitable substituents on the aryl or pyridine rings. It has been reported that introducing electron-acceptor fluorine atoms at the 4 and 6 positions of the central phenyl ring of the cyclometalated 1,3-bis(pyridin-2-yl)-benzene ligand causes a blue shift of the emission,39 a comportment improved by the presence of electron-donor groups like dimethylamino40 or methoxy36 on the pyridine rings. Remarkably, in these platinum(II) complexes bearing a tridentate ligand, the blue-shift of the emission occurs without the drop-off in quantum yield detected for related bidentate ligands. The origin of this blue-shift is an augmentation of the highest occupied molecular orbital (HOMO) – lowest unoccupied molecular orbital (LUMO) gap, as shown by means of density functional theory (DFT) calculations which put in evidence that the LUMO is largely located on the pyridine rings whereas the HOMO is mostly localized on the platinum center and the cyclometalated phenyl ring. Thus, electron-donor substituents on the pyridine rings raise the LUMO energy whereas electron-acceptor groups in position meta to the Pt–C bond are able to decrease the HOMO energy, both effects contributing to an increase of the HOMO–LUMO gap.41
Besides, it is known that the presence of a bulky substituent on the position 5 of the phenyl ring of the bpyb ligand causes steric hindrance which can prevent the face-to-face approach of the monomeric species, reducing self-quenching at elevated concentrations; such a steric effect is useful for the fabrication of OLEDs, in which local concentrations can be elevated, because self-quenching decreases device efficiencies.42 The fact that a suitable spatial arrangement of bulky groups could tune the excimer or aggregate formation, and consequently the phosphorescence characteristics, appeared as a springboard to decrease Pt⋯Pt interactions and/or π⋯π stacking and the concomitant red emission, and therefore to obtain efficient OLEDs with emission in the blue region of the visible spectrum. These observations, and the fact that the effect of the introduction of a π−delocalized polarizable substituent on position 4 of the pyridine rings was unknown, prompted us to synthesize and characterize complexes with more or less bulky substituents on phenyl groups linked to position 4 of the pyridine rings of Pt(F2dpyb)Cl39 (F2dpybH = 1,3-bis(pyridin-2-yl)-4,6-difluorobenzene; complexes PtL1–6Cl; Fig. 1).43–46
Preliminary results showed that complex PtL5Cl allows the fabrication of blue OLEDs46 whereas PtL2Cl can lead to yellow-green and deep red OLEDs.44 These preliminary investigations encouraged us to fabricate OLEDs based on the other complexes with different steric hindrance on the 4-phenylpyridine unit, namely, PtL1Cl, PtL3Cl, PtL4Cl, and PtL6Cl, in order to get a better understanding of the effect of steric hindrance on the OLED colour and performance. The results achieved in the fabrication of these OLEDs are reported in the present paper. Remarkably, an efficient (EQEmax= 8%) “sky-blue” OLED was realized with PtL6Cl whereas an “emerald green” OLED with an excellent performance (EQEmax= 20%.) was fabricated with PtL4Cl.
2. Results and discussion
Fig. S19–S24 in the SI report the UV-vis absorption spectra for the investigated Pt(II) complexes at different concentrations in dichloromethane.The influence on the phosphorescence properties of a more or less bulky π-delocalized polarizable substituent on position 4 of the pyridine rings of Pt(1,3-bis(pyridin-2-yl)-4,6-difluoro-benzene)Cl (PtL1–6Cl, Fig. 1) is summarized in Table 1.
| Ref. | Complex | λ max,abs/nm [ε/103 M−1 cm−1] | λ max,em/nmb monomer [bimolecular excited state]c | τ/µs | Φ lum | k r /s−1 [knre/s−1] |
|---|---|---|---|---|---|---|
| a At 298 K in degassed CH2Cl2, at 5 × 10−6 M. b Excitation at 374 nm for PtL1Cl and PtL3–6Cl, excitation at 422 nm for PtL2Cl. c Excimer/aggregate at 2 × 10−4 M. d Absolute Φlum measured with an integrating sphere. e Radiative and non-radiative rate constants obtained from the emission decay lifetimes and quantum yields, according to kr = Φlum/τ and knr = (1 − Φlum)/τ. | ||||||
| 43 | PtL1Cl | 277 [5.7] | 480 [∼704] | 3.52 | 0.89 | 2.5 × 105 [3.1 × 104] |
| 308 [3.4] | ||||||
| 340 [1.4] | ||||||
| 382 [2.1] | ||||||
| 471 [0.28] | ||||||
| 44 | PtL2Cl | 293 [45] | 562 [∼696] | 79.3 | 0.85 | 1.1 × 104 [1.9 × 103] |
| 423 [56] | ||||||
| 45 | PtL3Cl | 280 [50] | 480 [∼697] | 3.91 | 0.89 | 2.3 × 105 [2.8 × 104] |
| 312 [38] | ||||||
| 341 [17] | ||||||
| 383 [23] | ||||||
| 470 [0.31] | ||||||
| 45 | PtL4Cl | 278 [35] | 478 [∼690] | 3.96 | 0.95 | 2.4 × 105 [1.3 × 104] |
| 311 [24] | ||||||
| 340 [11] | ||||||
| 382 [16] | ||||||
| 470 [0.22] | ||||||
| 46 | PtL5Cl | 263 [23] | 471 [∼680] | 4.77 | 0.97 | 2.0 × 105 [6.3 × 103] |
| 287 [15] | ||||||
| 376 [7.8] | ||||||
| 438 [0.18] | ||||||
| 467 [0.12] | ||||||
| 43 | PtL6Cl | 238 [39] | 471 [∼680] | 4.48 | 0.98 | 2.2 × 105 [4.4 × 103] |
| 264 [38] | ||||||
| 335 [16] | ||||||
| 377 [14] | ||||||
| 467 [0.21] | ||||||
In dilute dichloromethane solution, all these complexes are characterized by luminescent quantum yields close to unity and luminescence lifetimes in the range 3–5 µs (Fig. S26 and S28-S31 in the SI) with the exception of complex PtL2Cl which has a very long luminescence lifetime (79.3 µs, Fig. S27), reasonably due to the presence of the electron-rich anilino pendants that render the lowest-energy excited states of intraligand character (πAr → πpy*, where Ar = Ph2N–C6H4–), suppressing the degree of metal character and leading to an abnormally long observed lifetime τ and low kr (Table 1); thus, it has been reported that aniline-based electron-rich pendants on the central aryl ring of related tridentate platinum(II) complexes lead to enhanced intraligand charge-transfer character, at the expense of the metal, and thus to longer lifetimes.42,47 In dilute dichloromethane solution (5 × 10−6 M), PtL1Cl and PtL3–6Cl show a vibrationally structured band (Fig. 2) with a high-energy emission maximum (471–480 nm) similar to that observed for Pt(1,3-bis(pyridin-2-yl)-4,6-difluoro-benzene)Cl (472 nm).39 In contrast, PtL2Cl is characterized by a broad emission at much lower-energy (562 nm).
 | ||
| Fig. 2 Normalized emission spectra in degassed dilute (top) and concentrated (bottom) dichloromethane solution, at 298 K. | ||
For complexes PtL1–3Cl, an increase of the concentration (2 × 10−4 M) leads to the formation of a structureless broad band at lower energy (λmax = ca 700 nm, Table 1), due to emissive bimolecular excited states. At the same concentration, the corresponding band is much less intense for PtL4Cl, as shown in Fig. 2, a behaviour that can be explained by the presence of two bulky tert-butyl groups, in positions 3 and 5 of the phenyl linked to the pyridine rings, which steric hindrance inhibits the association between two molecules in solution. It is worth pointing out that the introduction of a tert-butyl or diphenylamino group in position 4 of the phenylpyridine unit does not inhibit this association, as evidenced by the similar intensity of the low energy band of complexes PtL1–3Cl. In Fig. S25 the normalized emission spectra of dilute and concentrated solutions are reported for the single complexes. In order to get a better understanding of the steric effect in position 3 and 5 of the 4-phenylpyridine unit, it would be worthwhile to investigate the effect of other more or less bulky groups (for example adamantyl, isopropyl but even methyl). Besides, it turned out that the presence of a mesityl group in para of the pyridine rings is even more efficient to hamper the association of the monomeric species, as evidenced by a much less intense low energy band for PtL5Cl than for PtL4Cl. A similar low energy band intensity is observed with PtL6Cl. Clearly, the methyls ortho to the interannular C–C bond hamper efficiently the planar conformation that favors face-to-face intermolecular interactions. In agreement with this observation, in the complex PtL5Cl, there is a torsion angle of ca 62° between the mesityl groups and the pyridines, as shown from the X-ray structure, which hinders the neighbouring of the metal atoms and therefore the Pt⋯Pt interactions; the shortest Pt⋯Pt distance is 8.59 Å much longer than that reported for PtL1Cl (4.9 Å) or for the related platinum(II) chlorido complex with a cyclometalated 5-mesityl-1,3-bis(pyridin-2-yl)-benzene ligand (4.4 Å).43,46 Unfortunately, various attempts to obtain crystals of PtL4Cl suitable for X-ray determination failed.
Clearly, the nature of the substituents on the 4-phenylpyridine unit can tune the facility of association between two platinum complexes, Pt⋯Pt interactions and/or π⋯π stacking and thus the formation of excimer/aggregate species and concomitant red emission. Such an observation could be of particular interest for the development of OLED technologies, characterized by exceptional characteristics such as high colour purity, high contrast, wide view-angle, fast response, and energy-saving, required for emerging solid-state lighting and high-quality flat panel displays. Materials emitting red, green, and blue primary colours are typically used because a flat panel display has to produce all the different colours surrounding us even though other emitting colours may improve the displays quality. Regrettably, emitters in the blue region often show a much lower electroluminescence performance with respect to charge-carrier injection/transport, colour quality, efficiencies, and life expectancy.48 In this regard, phosphorescent metal complexes are particularly promising due to their long-lived excited state lifetimes which can harvest both triplet and singlet excitons, a behaviour that allows, from a theoretical point of view, to obtain in such devices a 100% internal quantum efficiency.
It turned out that Pt(II) complexes bearing a 1,3-bis(pyridin-2-yl) 4,6-difluoro-benzene ligand have a good potential for the fabrication of OLEDs. Thus, bluish-green OLEDs (CIE coordinates = 0.19, 0.42) have been prepared with Pt(1,3-bis(pyridin-2-yl)-4,6-difluoro-benzene)Cl working with a 5 wt% of platinum(II) compound in the matrix.38 A colour (CIE coordinates = 0.18, 0.35)38 close to that of “sky blue” CIE coordinates (0.17, 0.32)49 was obtained upon introduction of methyl substituents in position 4 of the pyridine rings whereas the presence of electron-donating methoxy or dimethylamino substituents allows the preparation of “sky-blue” OLEDs, with CIE coordinates (0.18,0.27)36 and (0.20,0.30),40 respectively (see later Table 2). The use of a pure film of these complexes as emitter led to the preparation of red OLEDs.36,38,40 Besides, recently, we reported that whereas the introduction of a triphenylamino substituent on the position 4 of the pyridine rings (complex PtL2Cl) allows the fabrication of yellow-green OLEDs,44 that of a mesityl group (complex PtL5Cl) leads to “sky-blue” OLEDs (CIE = 0.13, 0.29, see Table 2)46 with an admirable performance, putting in evidence that the introduction of bulky substituents in N^C^N Pt(II) complexes is a springboard for the fabrication of efficient blue OLEDs.
| Ref. | Complex | R | % w/w | CIE (x,y) | EQE (%) | LE (cd A−1) | PE (lm W−1) | J (mA cm−2) |
|---|---|---|---|---|---|---|---|---|
| 38 | — | H | 5% | 0.19, 0.42 | 7.8 | 15.9 | 6.9 | 3.1 |
| 100% | 0.63, 0.35 | 5.3 | 1.6 | 0.47 | 31.3 | |||
| 38 | — | Me | 5% | 0.18, 0.35 | 13.0 | 26.0 | 9.3 | 1.5 |
| 100% | 0.61, 0.38 | 14.7 | 13.9 | 5.0 | 3.7 | |||
| 36 | — | OMe | 5% | 0.18, 0.27 | 6.1 | 10.7 | 2.8 | 4.7 |
| 100% | 0.58, 0.41 | 16 | 27.7 | 8.1 | 1.8 | |||
| 40 | — | NMe2 | 5% | 0.20, 0.30 | 3.9 | 6.9 | 2.4 | 6.0 |
| 100% | 0.52, 0.47 | 6.4 | 14 | 5.7 | 3.9 | |||
| This work | PtL1Cl |
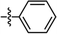
|
6% | 0.15, 0.40 | 5.8 | 12.3 | 7.7 | 3.2 |
| 100% | 0.64, 0.34 | 1.4 | 0.4 | 0.3 | 10.0 | |||
| 44 | PtL2Cl |
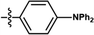
|
8% | 0.38, 0.56 | 5.8 | 18.9 | 11.4 | 2.3 |
| 100% | 0.65, 0.34 | 3.2 | 1.0 | 0.6 | 50.1 | |||
| This work | PtL3Cl |

|
6% | 0.20, 0.46 | 7.5 | 17.5 | 9.2 | 3.5 |
| 100% | 0.63, 0.35 | 1.1 | 0.4 | 0.2 | 127.9 | |||
| This work | PtL4Cl |
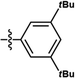
|
6% | 0.19, 0.44 | 16.0 | 34.0 | 19.5 | 1.2 |
| 100% | 0.60, 0.37 | 2.1 | 1.0 | 0.3 | 48.2 | |||
| 46 | PtL5Cl |
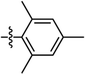
|
8% | 0.13, 0.29 | 10.2 | 18.3 | 12.0 | 2.4 |
| 100% | 0.56, 0.36 | 1.1 | 0.8 | 0.3 | 67.2 | |||
| This work | PtL6Cl |
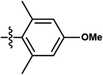
|
6% | 0.13, 0.26 | 7.0 | 12.0 | 7.5 | 3.5 |
| 100% | 0.52, 0.36 | 1.6 | 1.3 | 0.9 | 32.0 |
Therefore, the will to get a better understanding of the effect on the OLED colour and performance of the 4-phenylpyridine unit steric hindrance in 1,3-bis(4-phenylpyridin-2-yl)-4,6-difluorobenzene platinum(II) complexes and the desire to develop new efficient phosphorescent OLEDs, prompted us to study the behaviour of complexes PtL1Cl, PtL3Cl, PtL4Cl, and PtL6Cl for the preparation of the emissive layer.
OLEDs were prepared (see SI for details) with an emitting layer containing either a bis-4-((N-carbazolyl)phenyl)phenylphosphine oxide (BCPO) matrix hosting the platinum(II) complex (6% wt) or a film of the pure complex. Holes were injected from the indium tin oxide/MoOx anode (2 nm) and passed through a 50 nm thick 4,4′,4″-tris(N-carbazolyl)triphenylamine (TCTA) transporting layer. Electrons were injected from a cathode Al(100 nm)/LiF (0.5 nm) and transported to the emitting layer (EML) through a layer of 2,2′,2″-(1,3,5-benzinetriyl)-tris(1-phenyl-1-H-benzimidazole) (TPBi, 30 nm thick). Charges recombined in the EML (30 nm thick) made of the pure platinum complex or of a BCPO matrix hosting the platinum complex (6% wt) (see Fig. S32 for the OLED structure and the energy level diagram). Electroluminance spectra and images of the related OLEDs are shown in Fig. 3 whereas their luminance and external efficiency, as function of applied voltage and current density, are presented in Fig. 4.
 | ||
| Fig. 3 EL spectra, images and CIE coordinates of the OLED devices having 6% (black plot) and 100% (red plot) Pt complex. | ||
 | ||
| Fig. 4 Luminance vs. applied voltage and external EL efficiency vs. current density for the OLED devices having 6% (black plot) and 100% (red plot) Pt complex. | ||
In all the fabricated OLEDs, there is a good charge carrier confinement within the emitting layer and excellent energy transfer from the excited states of BCPO (generated by recombination of charge carriers) to the platinum compounds, with negligeable contribution from the electron-transporting (hole-blocking) or TCTA binder layers to the EML emission bands. Besides, as shown in Fig. 4, there is the characteristic roll-off of the EL efficiency at high applied voltage, caused by high field induced exciton dissociation and to exciton/charge and/or exciton–exciton interactions.50 The unexpected different trend EQE vs. current density observed for the OLED based on PtL3Cl as emitter (Fig. 4) can be attributed to lower recombination probability and/or higher dissociation probability of charge pairs at low current density.51
In Table 2, the performance of emerald green/blue and red OLEDs fabricated with complexes PtL1Cl, PtL3Cl, PtL4Cl, and PtL6Cl are compared with that of previously reported OLEDs based on N^C^N Pt complexes. Our investigation put in evidence that, during the fabrication of OLEDs with a low percentage of 1,3-bis(substituted 4-phenylpyridin-2-yl)-4,6-difluorobenzene platinum(II) complexes (6 wt%), the nature of the substituents on the 4-phenylpyridine plays a key role in the achievement of the desired OLED colour, brightness, and performance. Thus, an increase of the steric hindrance on the 4-phenylpyridine allows to reach a better performance of the devices, as shown by comparing the novel OLEDs fabricated with PtL1Cl (with a non-substituted 4-phenylpyridine moiety), PtL3Cl (with one tert-butyl group on the phenyl linked to the pyridine rings), and PtL4Cl (with two tert-butyl groups). The complex with the largest steric hindrance, PtL4Cl, allows the preparation of the most efficient OLEDs, in terms of EQE, PE, and LE with an “emerald green” emission (CIE coordinates = 0.19, 0.44), providing high color purity and contributing to improved display performance.52 Its performance is excellent, reaching an EQEmax of 20% and a luminance of ca 23.000 cd m−2 at a driving voltage of 12 V. This represents the highest EQEmax of N^C^N Pt(II) complexes, putting in evidence the remarkable effect caused by the steric hindrance of the two tert-butyl groups on the 4-phenylpyridine moieties.
Besides, the new OLED fabricated with PtL6Cl (6% wt) exhibits a “sky-blue” emission with CIE coordinates (0.13, 0.26) closer to those of “true blue” (CIE coordinates = 0.16, 0.13)53 with respect to the previously reported OLED based on PtL5Cl,46 in agreement with a more difficult formation of excimer/aggregate species with red emission due to a more difficult association between two platinum complexes, as confirmed by DFT calculations performed on the dimers of these complexes,43 with a Pt–Pt distance of 6.31 Å and 6.54 Å, for PtL5Cl and PtL6Cl, respectively. The performance reached with PtL6Cl (in terms of EQE, PE, and LE) is very good when compared to OLEDs with N^C^N platinum complexes bearing a methoxy or dimethylamino group on the pyridine rings (Table 2). It reaches an EQEmax of 8% and shows a luminance of ca 27.000 cd m−2 at a driving voltage of 10 V. This performance, although lower than that reported for a tetradentate cyclometalated N-heterocyclic carbene Pt(II) complex bearing tert-butyl groups (called PtON-tb-DTB, EQEmax of 20.9% with CIE coordinates = 0.14, 0.22),32 is very good when compared to that recently reached with PtL5Cl (EQEmax of 12.5% and luminance of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 cd m−2 at a driving voltage of 9 V)46 or blue OLEDs fabricated with other efficient Pt complexes bearing cyclometalated N-pyridyl carbazole ligands with monocarborane clusters (at 1010 cd m−2, EQEmax = 6.2%)54 or with a N-heterocyclic carbene Pt(II) complex bearing a phenyl acetylene ligand (at low luminance, EQEmax = 8% whereas at higher luminance there is a large efficiency roll off, reaching ca 2% at 500 cd m−2).55 The “sky-blue” colour observed in the case of OLEDs based on PtL5Cl and PtL6Cl put in evidence that the introduction of methyls ortho to the interannular C–C bond, which hamper the planar conformation that favours face-to-face intermolecular interactions, is a way to go towards blue OLEDs.
500 cd m−2 at a driving voltage of 9 V)46 or blue OLEDs fabricated with other efficient Pt complexes bearing cyclometalated N-pyridyl carbazole ligands with monocarborane clusters (at 1010 cd m−2, EQEmax = 6.2%)54 or with a N-heterocyclic carbene Pt(II) complex bearing a phenyl acetylene ligand (at low luminance, EQEmax = 8% whereas at higher luminance there is a large efficiency roll off, reaching ca 2% at 500 cd m−2).55 The “sky-blue” colour observed in the case of OLEDs based on PtL5Cl and PtL6Cl put in evidence that the introduction of methyls ortho to the interannular C–C bond, which hamper the planar conformation that favours face-to-face intermolecular interactions, is a way to go towards blue OLEDs.
Regarding the red OLEDs fabricated with a film of the pure complex as emitting layer, the best performance (in terms of EQE, LE, PE) is reached with the complex bearing a simple methyl group in position 4 of the pyridines (Table 2), as expected due to the lack of steric hindrance for the association of two platinum(II) molecules.
3. Conclusion
In conclusion, this work put in evidence the good potential of 1,3-bis(4-phenylpyridin-2-yl)-4,6-difluorobenzene platinum(II) complexes for the fabrication of both “emerald green” and “sky-blue” OLEDs. It shows the key role of substituents on the 4-phenylpyridine unit in tuning the colour of the emission and in achieving a high OLED's performance. As a matter of fact, the presence of two tert-butyl groups in position 3 and 5 of the phenylpyridine moiety affords a suitable steric hindrance to boost the external quantum efficiency whereas the introduction of methyls in ortho position to the interannular C–C bond is a way to switch the colour from “emerald green” to “sky-blue”. As a step forward, it would be important to verify the effect of a combination of these two factors on the colour and performance of the OLEDs since it could lead to highly efficient blue OLEDs. It would also be of interest to investigate the effect of the introduction of strongly electron-donor bulky groups on the pyridines because such a combination of bulkiness and electron-donor behaviour could allow the preparation not only of efficient “sky-blue” and “true-blue” OLEDs but also “deep-blue” ones. Thus, the present work gives important guidelines for the design of new members of this complexes family which represents a precious tool for the development of novel efficient phosphorescent OLEDs. In the near future, we plan to produce OLEDs with a larger surface area in collaboration with a company that will perform lifetime tests.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the Manuscript and in the supplementary information (SI). Supplementary information: Synthesis and characterization of platinum complexes; fabrication of OLEDs. See DOI: https://doi.org/10.1039/d6tc00125d.Acknowledgements
The work was supported by the National Interuniversity Consortium of Materials Science and Technology (Project TRI_25_073 Dragonetti and TRI_25_173 Colombo), the Università degli Studi di Milano (Project PSR2023_DIP_005_PI_FTESS), and CNR (PNRR MUR project ECS_00000033_ECOSISTER). We acknowledge the Ministero dell'Istruzione, dell'Università e della Ricerca—Fondo per Progetti di Ricerca di Rilevante Interesse Nazionale 2022 (PRIN 2022NMSFHN).Notes and references
- N. Tomar, A. Agrawal, V. S. Dhaka and P. K. Surolia, Sol. Energy, 2020, 207, 59–76 CrossRef CAS.
- C. Dragonetti, M. Magni, A. Colombo, F. Fagnani, D. Roberto, F. Melchiorre, P. Biagini and S. Fantacci, Dalton Trans., 2019, 48, 9703–9711 RSC.
- A. B. Muñoz-García, I. Benesperi, G. Boschloo, J. J. Concepcion, J. H. Delcamp, E. A. Gibson, G. J. Meyer, M. Pavone, H. Pettersson and A. Hagfeldt, Chem. Soc. Rev., 2021, 50, 12450–12550 RSC.
- C. E. Housecroft and E. C. Constable, Chem. Sci., 2022, 13, 1225–1262 RSC.
- C. Minozzi, A. Caron, J.-C. Grenier-Petel, J. Santandrea and S. K. Collins, Angew. Chem., Int. Ed., 2018, 57, 5477–5481 CrossRef CAS PubMed.
- X. Huang and E. Meggers, Acc. Chem. Res., 2019, 52, 833–847 CrossRef CAS PubMed.
- D. N. Tritton, F.-K. Tang, G. B. Bodedla, F.-W. Lee, C.-S. Kwan, K. C.-F. Leung, X. Zhu and W.-Y. Wong, Coord. Chem. Rev., 2022, 459, 214390 CrossRef CAS.
- D. C. Cabanero and T. Rovis, Nat. Rev. Chem., 2025, 9, 28–45 CrossRef CAS PubMed.
- S. Kaur, M. Kaur, P. Kaur, K. Clays and K. Singh, Coord. Chem. Rev., 2017, 343, 185–219 CrossRef CAS.
- S. Di Bella, A. Colombo, C. Dragonetti, S. Righetto and D. Roberto, Inorganics, 2018, 6, 133 Search PubMed.
- A. Colombo, C. Dragonetti, V. Guerchais and D. Roberto, Coord. Chem. Rev., 2021, 446, 214113 CrossRef CAS.
- X. Xue, H. Wang, Y. Han and H. Hou, Dalton Trans., 2018, 47, 13–22 RSC.
- Y. Y. Ning, G. Q. Jin, M. X. Wang, S. Gao and J. L. Zhang, Curr. Opin. Chem. Biol., 2022, 66, 102097–102107 CrossRef CAS PubMed.
- C. Cebrian and M. Mauro, Beilstein J. Org. Chem., 2018, 14, 1459–1481 CrossRef CAS PubMed.
- C. Bizzarri, E. Spuling, D. M. Knoll, D. Volz and S. Bräse, Coord. Chem. Rev., 2018, 373, 49–82 CrossRef CAS.
- X. Li, Y. Xie and Z. Li, Chem. – Asian J., 2021, 16, 2817–2829 CrossRef CAS PubMed.
- L. C.-C. Lee and K. K.-W. Lo, Chem. Rev., 2024, 124, 8825–9014 CrossRef CAS PubMed.
- V. W.-W. Yam and A. S.-Y. Law, Coord. Chem. Rev., 2020, 414, 213298 CrossRef CAS.
- L. K. McKenzie, H. E. Bryant and J. A. Weinstein, Coord. Chem. Rev., 2019, 379, 2–29 CrossRef CAS.
- G. De Soricellis, F. Fagnani, A. Colombo, C. Dragonetti and D. Roberto, Inorg. Chim. Acta, 2022, 541, 121082 CrossRef CAS.
- L. F. Gildea and J. A. G. Williams, in Iridium and platinum complexes for OLEDs, ed A. Buckley, Organic Light-Emitting Diodes: Materials, Devices and Applications, Woodhead, Cambridge, 2013 Search PubMed.
- A. K. Pal, S. Krotkus, M. Fontani, C. F. R. Mackenzie, D. B. Cordes, A. M. Z. Slawin, I. D. W. Samuel and E. Zysman-Colman, Adv. Mater., 2018, 30, 1804231 CrossRef PubMed.
- A. Jouaiti, D.-C. Huang, V. Giuso, C. Cebrián, P. Mercandelli, K.-H. Wang, C.-H. Chang and M. Mauro, ACS Appl. Electron. Mater., 2023, 5, 2781–2792 CrossRef CAS.
- E. Tankelevičiūtė, I. D. W. Samuel and E. Zysman-Colman, J. Phys. Chem. Lett., 2024, 15, 1034–1047 CrossRef PubMed.
- X. Yang, G. Mu, K. Weng and X. Tang, Photonics, 2024, 11, 864 CrossRef CAS.
- F. Hundemer, E. Crovini, Y. Wada, H. Kaji, S. Bräse and E. Zysman-Colman, Mater. Adv., 2020, 1, 2862 Search PubMed.
- E. Duda, S. M. Suresh, D. Hall, S. Bagnich, R. Saxena, D. B. Cordes, A. M. Z. Slawin, D. Beljonne, Y. Olivier, A. Köhler and E. Zysman-Colman, Chem. Mater., 2023, 35, 2027–2037 CrossRef CAS PubMed.
- S. Wu, L. Zhang, J. Wang, A. K. Gupta, I. D. W. Samuel and E. Zysman-Colman, Angew. Chem., Int. Ed., 2023, 62, e202305182 CrossRef CAS PubMed.
- R. W. Weerasinghe, J. Marques dos Santos, Y. Chitose, T. Matulaitis, S. L. Warriner, D. Barman, C.-Y. Chan, Y. Tsuchiya, E. Zysman-Colman and C. Adachi, Phys. Chem. Chem. Phys., 2024, 26, 21337 Search PubMed.
- C. F. R. Mackenzie, L. Zhang, D. B. Cordes, A. M. Z. Slawin, I. D. W. Samuel and E. Zysman-Colman, Adv. Opt. Mater., 2023, 11, 2201495 CrossRef CAS.
- T. Maganti and K. Venkatesan, ChemPlusChem, 2022, 87, e202200014 CrossRef CAS PubMed.
- Y. H. Jung, G. S. Lee, S. Muruganantham, H. R. Kim, J. H. Oh, J. H. Ham, S. B. Yadav, J. H. Lee, M. Y. Chae and Y.-H. Kim, Nat. Commun., 2024, 15, 2977 CrossRef CAS PubMed.
- F. Zhan, K. Xu, J. Zheng, Q. Chen, G. Shen, J. Dai, Q. Feng, H. Guo, S. Liu, J. Ying, J. Tong, W. Lou, Y.-F. Yang, S. Chen, Y. She and G. Li, Chem. Eng. J., 2025, 524, 169759 Search PubMed.
- Q. Chu, H. Yao, J. Tong, K. Xu, Y. She and G. Li, Chem. Mater., 2025, 37, 6404–6413 CrossRef CAS.
- V. Adamovich, J. Brooks, A. Tamayo, A. M. Alexander, P. I. Djurovich, B. W. D’Andrade, C. Adachi, S. R. Forrest and M. E. Thompson, New J. Chem., 2002, 26, 1171–1178 Search PubMed.
- M. Cocchi, J. Kalinowski, L. Murphy, J. A. G. Williams and V. Fattori, Org. Electron., 2010, 11, 388–396 Search PubMed.
- J. Kalinowski, V. Fattori, M. Cocchi and J. A. G. Williams, Coord. Chem. Rev., 2011, 255, 2401–2425 CrossRef CAS.
- M. Cocchi, J. Kalinowski, V. Fattori, J. A. G. Williams and L. Murphy, Appl. Phys. Lett., 2009, 94, 073309 CrossRef.
- A. F. Rausch, L. Murphy, J. A. G. Williams and H. Yersin, Inorg. Chem., 2012, 51, 312–319 CrossRef CAS PubMed.
- L. Murphy, P. Brulatti, V. Fattori, M. Cocchi and J. A. G. Williams, Chem. Commun., 2012, 48, 5817–5819 RSC.
- W. Sotoyama, T. Satoh, H. Sato, A. Matsuura and N. Sawatari, J. Phys. Chem. A, 2005, 109, 9760–9766 CrossRef CAS PubMed.
- S. J. Farley, D. L. Rochester, A. L. Thompson, J. A. K. Howard and J. A. G. Williams, Inorg. Chem., 2005, 44, 9690–9703 CrossRef CAS PubMed.
- G. De Soricellis, B. Carboni, V. Guerchais, J. A. G. Williams, D. Marinotto, A. Colombo, C. Dragonetti, F. Fagnani, S. Fantacci and D. Roberto, Dalton Trans., 2025, 54, 10566–10573 RSC.
- A. Colombo, G. De Soricellis, F. Fagnani, C. Dragonetti, M. Cocchi, B. Carboni, V. Guerchais and D. Marinotto, Dalton Trans., 2022, 51, 12161–12169 Search PubMed.
- G. De Soricellis, V. Guerchais, A. Colombo, C. Dragonetti, F. Fagnani, D. Roberto and D. Marinotto, Molecules, 2025, 30, 1498 CrossRef CAS PubMed.
- A. Colombo, G. De Soricellis, C. Dragonetti, F. Fagnani, D. Roberto, B. Carboni, V. Guerchais, T. Roisnel, M. Cocchi, S. Fantacci, E. Radicchi and D. Marinotto, J. Mater. Chem. C, 2024, 12, 9702–9715 Search PubMed.
- D. L. Rochester, S. Develay, S. Zalis and J. A. G. Williams, Dalton Trans., 2009, 1728–1741 Search PubMed.
- X. Yang, X. Xu and G. Zhou, J. Mater. Chem. C, 2015, 3, 913–944 RSC.
- Z. Zhang, E. Crovini, P. L. dos Santos, B. A. Naqvi, D. B. Cordes, A. M. Z. Slawin, P. Sahay, W. Brütting, I. D. W. Samuel, S. Bräse and E. Zysman-Colman, Adv. Opt. Mater., 2020, 8, 2001354 CrossRef CAS.
- J. Kalinowski, W. Stampor, J. Szmytkowski, D. Virgili, M. Cocchi, V. Fattori and C. Sabatini, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 74, 85316 CrossRef.
- J. Kalinowski, M. Cocchi, V. Fattori, L. Murphy and J. A. G. Williams, Org. Electron., 2010, 11, 724–730 CrossRef CAS.
- https://dot-color.com/tag/cie-1931/ .
- R. He, Z. Xu, S. Valandro, H. D. Arman, J. Xue and K. S. Schanze, ACS Appl. Mater. Interfaces, 2021, 13, 5327–5337 CrossRef CAS.
- Y. Shen, X. Kong, F. Yang, H.-D. Bian, G. Cheng, T. R. Cook and Y. Zhang, Inorg. Chem., 2022, 61, 16707–16717 CrossRef CAS PubMed.
- J. D. Bullock, A. Salehi, C. J. Zeman IV, K. A. Abboud, F. So and K. S. Schanze, ACS Appl. Mater. Interfaces, 2017, 9, 41111–41114 Search PubMed.
Footnote |
| † In memoriam of Bertrand Carboni. |
| This journal is © The Royal Society of Chemistry 2026 |

