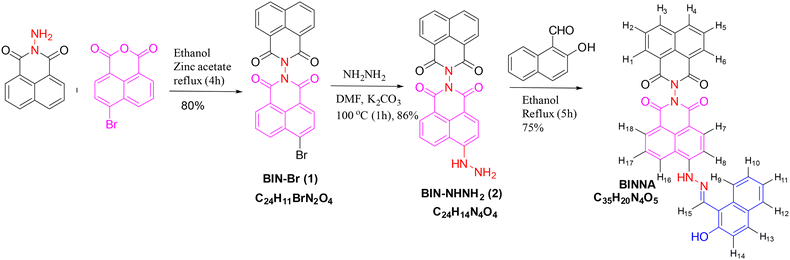
Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A next-generation fluorescent chemosensor for dual-mode detection of aluminium ions and DCP: a bridge between sensing and bio-imaging
Saurabh
Gupta†
a,
Abhishek
Kaundal†
b,
Gulshan
Kumar
 c,
Vijay
Luxami
c,
Vijay
Luxami
 *b and
Kamaldeep
Paul
*b and
Kamaldeep
Paul
 *ab
*ab
aTIET-VT, Centre of Excellence in Emerging Materials, Thapar Institute of Engineering and Technology, Patiala, Punjab 147004, India
bDepartment of Chemistry and Biochemistry, Thapar Institute of Engineering and Technology, Patiala, 147001, India. E-mail: vluxami@thapar.edu; kpaul@thapar.edu
cDepartment of Chemistry, Banasthali University, Banasthali, Newai, 304022, Rajasthan, India
First published on 19th January 2026
Abstract
In the present work, we have rationally designed and synthesized a bis-naphthalimide-based “turn-on” fluorescent chemosensor, BINNA, capable of dual-analyte recognition with high sensitivity and selectivity for aluminium ions (Al3+) and the nerve agent mimic diethyl chlorophosphate (DCP) in methanol medium. Distinct photophysical responses are observed: the addition of DCP induces a strong emission band at 370 nm, whereas the addition of Al3+ ions displays an intense emission band at 450 nm, allowing for clear spectral discrimination. The probe demonstrates outstanding detection capabilities, with detection limits reaching as low as 8.5 nM for DCP and 18.2 nM for Al3+. High-resolution mass spectrometry (HRMS) analyses provide mechanistic insights into the interaction pathways of BINNA with each target analyte. Complementary theoretical investigations using computational modelling further corroborate the experimental findings. Beyond its role in solution-phase sensing, BINNA's photonic properties are employed to construct a molecular logic gate system based on fluorescence signal outputs. The practical applicability of the sensor is validated through the successful detection of DCP and Al3+ in soil samples. Moreover, BINNA's ability to visualize cellular uptake and fluorescence in live bacterial cells underscores its potential as a multifunctional probe for environmental analysis and advanced bio-imaging applications.
1. Introduction
Chemosensors have emerged as pivotal tools in modern science, offering innovative solutions for environmental sustainability, health monitoring, and food quality control.1 Their ability to provide rapid, cost-effective, and selective detection of various analytes has revolutionized traditional sensing methodologies.2 Traditional detection methods like atomic absorption spectroscopy (AAS),3 voltammetry,4 and inductively coupled plasma mass spectrometry (ICP-MS)5 have been instrumental in analytical chemistry. However, these techniques often require sophisticated instrumentation, are time-consuming, and lack the desired sensitivity and selectivity.6,7 In contrast, chemosensors, especially those based on fluorophores, offer simplicity in synthesis, cost-effectiveness, and rapid detection capabilities.8–10 Their detection limits, selectivity, and recoverability make them ideal for detecting various metal and biological ions.11–13 Among the fluorophores, naphthalimide has garnered significant attention due to its unique properties: a flat aromatic ring system, π-electron deficiency, strong absorption in the visible region, high fluorescence quantum yield, significant Stokes shift, outstanding photostability, rapid response, high sensitivity and selectivity, and ease of structural modification. These characteristics make naphthalimide an excellent candidate for developing chemosensors.14–18Aluminium ions (Al3+) are prevalent in daily life, found in cooking vessels, the paper industry, textiles, agriculture, and food additives.19 Although Al3+ is non-essential for humans, elevated concentrations can lead to severe health issues, including Parkinson's disease, Alzheimer's disease, osteomalacia, and central nervous system malfunctions.20,21 The World Health Organization (WHO) recommends a permissible limit of 7.14 µM in water and 3–10 mg per day for human intake.22 Currently, most of the Al3+ detecting sensors face challenges due to interference from other metal ions like Hg2+, Cu2+, Zn2+, and Fe3+. Therefore, there is an urgent need to develop novel chemosensors that can selectively detect Al3+ ions without interference.23,24 Recent studies have shown that naphthalimide-based chemosensors exhibit high selectivity and sensitivity for Al3+ detection, making them promising candidates for practical applications.25,26
Moreover, organophosphorus compounds (OPs), including diethyl chlorophosphate (DCP), are highly toxic substances used as pesticides, herbicides, and chemical warfare agents.27 These compounds inhibit acetylcholine esterase (AchE), leading to the accumulation of acetylcholine, which disrupts nerve function and causes respiratory failure and death.28 DCP is often used as a simulant for G-series nerve agents like soman (GD), sarin (GB), and tabun (GA) due to its similar structure and reactivity.29,30 Given the high toxicity of OPs, researchers have focused on developing chemosensors for their detection.31 Recent advancements have led to the development of chemosensors capable of detecting DCP with high sensitivity and selectivity, offering potential for real-time monitoring and rapid response in case of exposure.32 Therefore, the development of chemosensors, particularly those based on fluorophores like naphthalimide, offers a promising avenue for the selective and sensitive detection of harmful ions and nerve agents. These advancements not only enhance the ability to monitor environmental and biological systems but also contribute significantly to public health and safety.
1.1. Design of BINNA and novelty of the work
In recent years, a variety of chemosensors have been designed and synthesized for the detection of Al3+ ions and DCP.33–35 These chemosensors can detect either Al3+ or DCP, with detection limits ranging from 23 µM to 98 nM (Table S1). These chemosensors are generally Schiff bases incorporated into some fluorophores such as naphthalimide, rhodamine, phenanthroimidazole, etc., but none of the developed chemosensors has the ability to detect Al3+ and DCP simultaneously, thereby increasing the synthesis cost of developing different chemosensors for both analytes and decreasing their multifunctional applications in biological detection and environmental monitoring. Thus, there is a need for the development of new chemosensors that can simultaneously detect both analytes and have better detection limits (Fig. 1). Recently, Zhang et al.25 reported a naphthalimide-based chemosensor containing a Schiff base unit formed from (N,N-diethylamino)benzaldehyde, while an alkyl group (ethyl morpholine) was introduced at the anhydride position of naphthalimide. The reported sensor detected the Al3+ ions in MeOH with a limit of detection in the micromolar range. Similarly, in 2023, Jain et al.26 developed a naphthalimide-based chemosensor appended with a Schiff base unit derived from (N,N-diethylamino)salicylaldehyde. They have used butylamine as an alkyl group at the anhydride position of naphthalimide; their developed sensor detects DCP in CH3CN with a detection limit in the micromolar range. Over the past few years, naphthalimide with a Schiff base unit has been largely explored for its sensing ability, but up to now, no report on a bis-naphthalimide moiety bearing Schiff base units has been published. In this context, we have developed a novel chemosensor (BINNA) by modifying a previously reported naphthalimide-based probe, specifically replacing the alkyl chain with an additional naphthalimide unit and substituting (N,N-diethylamino)salicylaldehyde with naphthaldehyde to create a bis-naphthalimide architecture. Our developed chemosensor sequentially detects both the metal ion, i.e. Al3+, and the nerve agent DCP in methanol, which could effectively reduce the synthesis cost of different chemosensors. The probe BINNA exhibits better selectivity and sensitivity than the reported chemosensors with a limit of detection in the nanomolar range for both analytes. Moreover, BINNA also exhibited aggregation effects, which were supported by DLS and SEM studies. Initially, BINNA is non-fluorescent, but upon interacting with Al3+ or DCP, an enhancement in the fluorescence intensity of the probe is observed, thus building a “turn-on” fluorescent probe for detecting metal ions and nerve agents. This capability not only improves sensitivity but also offers a dependable fluorescence-based approach for the selective detection of Al3+ ions and DCP. The probe also demonstrates strong performance in identifying both analytes in live bacterial cells, highlighting its versatility and effectiveness for practical and real-world applications.2. Experimental section
2.1. Materials and characterisation
The chemicals used for the synthesis of BINNA were purchased from various suppliers, including Sigma-Aldrich Ltd, Loba Chemie, and Spectrochem, depending on their availability. All solvents utilized in the synthesis were of spectroscopic grade and procured from Spectrochem. Reaction progress was monitored using thin-layer chromatography (TLC) on silica plates coated with silica gel GF-254, obtained from Spectrochem Pvt Ltd, India. UV light at wavelengths of 254 or 365 nm was employed to visualize changes on the TLC plates. Compound purification was carried out using column chromatography with silica gel (60–120 mesh). 1H and 13C NMR spectra were recorded at room temperature using a BRUKER ECS-400 MHz spectrometer (400 MHz for 1H and 100 MHz for 13C). NMR samples were prepared in deuterated DMSO (DMSO-d6) using tetramethylsilane (TMS) as the internal standard, with chemical shifts reported in parts per million (ppm). High-resolution mass spectrometry (HRMS) data were obtained using a XEVO G2-XS QTOF spectrometer.2.2. Computational methods
Geometry optimization of the probe BINNA in its ground state (S0) was performed using the B3LYP functional with the 6-31G(d) basis set as implemented in Gaussian 16. To account for experimental conditions, the solvation effect of methanol was incorporated using the integral equation formalism polarizable continuum model (IEFPCM). The absence of imaginary vibrational frequencies confirmed the stability of the optimized molecular structures. Excitation energies were calculated employing time-dependent density functional theory (TD-DFT). Given the molecular flexibility of BINNA at –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and –N–N– linkages, multiple conformers were explored at the S0 state. Two stable conformers, designated BINNA-C1 and BINNA-C2, were identified with relative energies of 0.00 and 2.57 kcal mol−1, respectively. Boltzmann population analysis at 298 K indicated distributions of 98% for BINNA-C1 and 2% for BINNA-C2, leading to the selection of BINNA-C1 for subsequent analysis (Fig. S1). Further geometry optimizations and TD-DFT calculations were carried out using various exchange–correlation functionals (CAM-B3LYP, B3PW91, PBE0, and M06-2X) and basis sets (6-31G(d), 6-311G(d) and 6-311G(d,p)). The CAM-B3LYP/6-31G(d) level of theory yielded results in the closest agreement with experimental data and was therefore employed for a detailed discussion of the electronic and optical properties. Molecular electrostatic potential (MEP) mapping was also conducted to assess charge distribution across the molecule.
N– and –N–N– linkages, multiple conformers were explored at the S0 state. Two stable conformers, designated BINNA-C1 and BINNA-C2, were identified with relative energies of 0.00 and 2.57 kcal mol−1, respectively. Boltzmann population analysis at 298 K indicated distributions of 98% for BINNA-C1 and 2% for BINNA-C2, leading to the selection of BINNA-C1 for subsequent analysis (Fig. S1). Further geometry optimizations and TD-DFT calculations were carried out using various exchange–correlation functionals (CAM-B3LYP, B3PW91, PBE0, and M06-2X) and basis sets (6-31G(d), 6-311G(d) and 6-311G(d,p)). The CAM-B3LYP/6-31G(d) level of theory yielded results in the closest agreement with experimental data and was therefore employed for a detailed discussion of the electronic and optical properties. Molecular electrostatic potential (MEP) mapping was also conducted to assess charge distribution across the molecule.
2.3 Synthesis of BINNA
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) mixture as the eluent. This process yielded a white solid of compound 1 with 80% yield, an Rf value of 0.6 (chloroform), and a melting point of 289–292 °C.36
2) mixture as the eluent. This process yielded a white solid of compound 1 with 80% yield, an Rf value of 0.6 (chloroform), and a melting point of 289–292 °C.36
2.4. Preparation of stock solutions
Stock solutions of individual anions and metal ions were prepared in acetonitrile (CH3CN) and distilled water at a concentration of 1 × 10−1 M. The ligand stock solution was prepared separately in a DMSO/CH3CN mixture at a concentration of 1 × 10−3 M. These stock solutions were then further diluted to the required concentrations using the appropriate solvents.2.5. Absorption and emission studies
The absorption and emission spectra of BINNA were analysed at a concentration of 20 µM. Absorption measurements were performed using a UV-2600 spectrophotometer and quartz cuvettes of 1 cm path length. Emission spectra were recorded with a Varian Cary Eclipse fluorescence spectrophotometer (G9800A) using slit widths of 10 nm for excitation and 20 nm for emission at the stated excitation wavelength. Time-resolved fluorescence measurements were carried out using a DeltaFlex Modular Fluorescence Lifetime Spectrofluorimeter (HORIBA Scientific – PPD-850). The stoichiometric ratios between the ligand and metal ions/nerve agent were determined using Job's plot analysis.3. Results and discussion
3.1. Photophysical behavior of BINNA
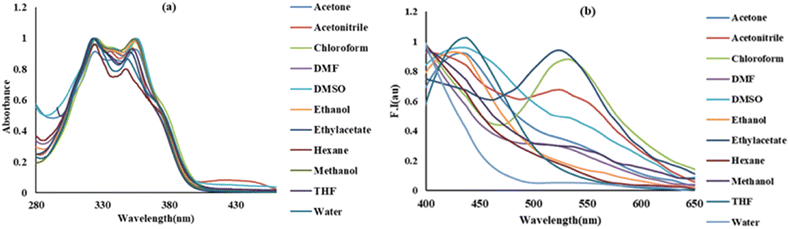
The photophysical studies of BINNA were conducted using spectroscopic techniques of absorption and emission. In CH3OH solvent, two absorption bands were displayed by BINNA (20 µM) at 325 and 355 nm, while weak fluorescence was observed, as shown in Fig. 2a and b. Different solvents were used to explore the spectral behaviour of BINNA with different polarities, such as MeOH, THF, toluene, DMF, CH3CN, CHCl3, cyclohexane, IPA, ethyl acetate, ethanol, DMSO, and hexane. No significant change in the absorption and emission spectra of BINNA on moving from low to high polarity was observed.3.2. Aggregation-induced emission enhancement (AIEE) behaviour of BINNA
To investigate the aggregation behaviour of the synthesized BINNA, absorption and emission spectra were recorded in CH3CN with increasing amounts of water. Initially, BINNA solution (20 µM in CH3CN) displayed two absorption bands at 325 and 355 nm. As the water content was increased from 0 to 60%, a significant reduction in absorption intensity was observed. However, with further addition of water (70 to 100%), a new absorption band at 375 nm was observed along with a tail-off absorption between 500 nm and 700 nm, suggesting aggregate formation in the mixed solvent system (Fig. 3a). Fluorescence measurements further confirmed the aggregation behaviour. BINNA exhibited very low fluorescence intensity in pure CH3CN, and no significant change in the fluorescence intensity was observed upon gradual addition of water up to 50% (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; H2O
1; H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN). Upon further incremental addition of water from 60 to 90%, a significant increase in fluorescence intensity at 380 nm was observed. In pure water, the fluorescence intensity of BINNA was quenched significantly as compared to 90% H2O
CH3CN). Upon further incremental addition of water from 60 to 90%, a significant increase in fluorescence intensity at 380 nm was observed. In pure water, the fluorescence intensity of BINNA was quenched significantly as compared to 90% H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN (v/v), indicating significant aggregation-induced emission enhancement effects (Fig. 3b). These results confirmed the formation of aggregates upon increasing water concentration.
CH3CN (v/v), indicating significant aggregation-induced emission enhancement effects (Fig. 3b). These results confirmed the formation of aggregates upon increasing water concentration.
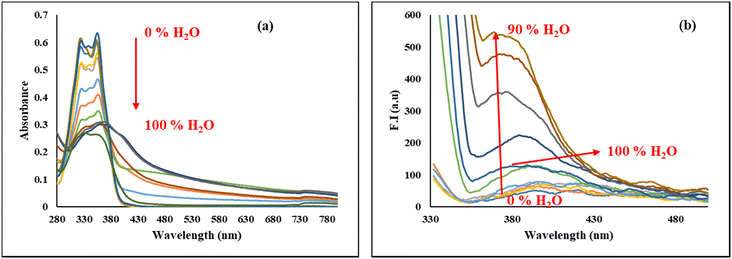 | ||
| Fig. 3 (a) Absorption spectra of BINNA (20 µM) and (b) emission spectra of BINNA (20 µM) with incremental addition of H2O in CH3CN. | ||
A dynamic light scattering (DLS) study was conducted to explore how varying water content influences the particle size and aggregation behaviour of BINNA. In pure acetonitrile (CH3CN), BINNA displayed a relatively uniform particle size, averaging 145 ± 3 nm, with a polydispersity index (PDI) of 0.372 (Fig. 4a). However, as water was incrementally introduced into the system, a clear trend of particle growth emerged, signifying enhanced aggregation. At 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 H2O
50 H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN ratio, the average hydrodynamic diameter expanded to 844 ± 10 nm, accompanied by a PDI of 0.479 (Fig. 4b). Further increasing the water content to 70% resulted in an increase of particle size to 1185 ± 16 nm, with a PDI of 0.587 (Fig. 4c). Moreover, at a water content of 90%, the particle size of BINNA increased to 2608 ± 8 nm (Fig. 4d), and in pure water, BINNA exhibited an average particle size of 3092 ± 10 nm with a PDI value of 0.717 (Fig. 4e). This progressive increase in both particle size and PDI underscores the aggregation of BINNA in aqueous environments, suggesting a growing heterogeneity in particle size distribution as the water content increases.
CH3CN ratio, the average hydrodynamic diameter expanded to 844 ± 10 nm, accompanied by a PDI of 0.479 (Fig. 4b). Further increasing the water content to 70% resulted in an increase of particle size to 1185 ± 16 nm, with a PDI of 0.587 (Fig. 4c). Moreover, at a water content of 90%, the particle size of BINNA increased to 2608 ± 8 nm (Fig. 4d), and in pure water, BINNA exhibited an average particle size of 3092 ± 10 nm with a PDI value of 0.717 (Fig. 4e). This progressive increase in both particle size and PDI underscores the aggregation of BINNA in aqueous environments, suggesting a growing heterogeneity in particle size distribution as the water content increases.
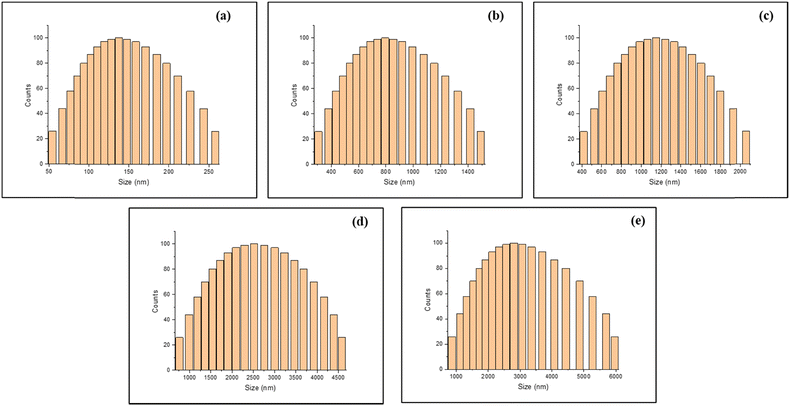 | ||
| Fig. 4 Hydrodynamic size of BINNA in (a) CH3CN, (b) 50% H2O, (c) 70% H2O, (d) 90% H2O and (e) H2O only. | ||
Furthermore, to explore how water content influences the structure of BINNA, FE-SEM images were recorded with increasing concentration of water in CH3CN. FE-SEM clearly demonstrated that the particle size increased with a higher H2O ratio. In a pure CH3CN environment, BINNA displayed small rod-like mixed morphology, i.e., small needle-like as well as rod-like shapes with particle size between 130 and 140 nm (Fig. 5a). However, upon increasing the water content to 50%, these small rod-like structures start accumulating together to form aggregates with a particle size between 800 and 870 nm; hence, an increase in size can be observed (Fig. 5b). Furthermore, upon increasing the water concentration to 90%, BINNA exhibited an elongated rod-like structure with a particle size between 2400 and 2800 nm and a diameter between 180 and 220 nm, clearly indicating an increase in the particle size of BINNA (Fig. 5c), whereas in pure water, these rod structures started accumulating, leading to the formation of aggregates with a particle size between 3000 and 3300 nm (Fig. 5d). The particle size determined from FE-SEM and DLS were consistent. These evolving structures indicate the progressive self-assembly and aggregation of the molecules.
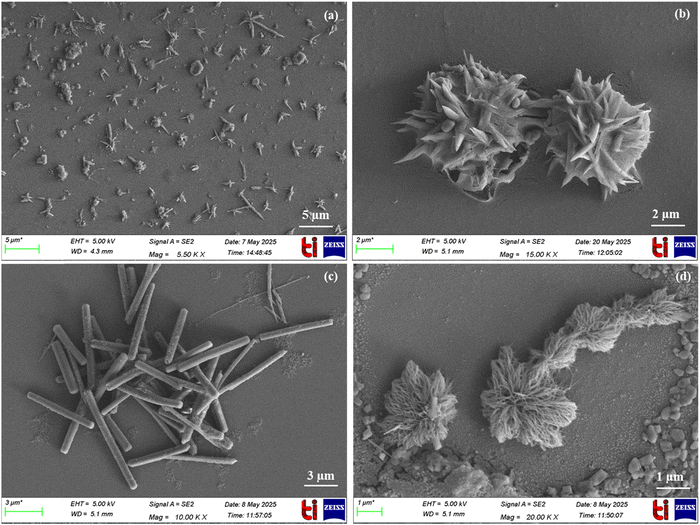 | ||
Fig. 5 FE-SEM images of BINNA in (a) CH3CN; (b) 50% (H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN); (c) 90% (H2O CH3CN); (c) 90% (H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN); and (d) H2O only. CH3CN); and (d) H2O only. | ||
To further elucidate the aggregation-induced emission enhancement (AIEE) behavior of BINNA, time-correlated single photon counting (TCSPC) measurements were conducted by varying the water content in CH3CN. Initially, in pure CH3CN, BINNA displayed an average fluorescence lifetime of 0.42 ns, having three decay components with lifetimes and populations of 0.81 ns and 3%, 4.15 ns and 2%, and 0.24 ns and 95% (Table 1). Upon increasing the water content to 50%, i.e., CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), a noticeable increase in the average lifetime of BINNA was observed with a lifetime value of 0.57 ns, corresponding to three decay components with lifetimes and populations of 0.57 ns and 50%, 1.18 ns and 0%, and 0.57 ns and 50%. Furthermore, a significant enhancement in the average fluorescence lifetime of BINNA was observed upon increasing the water content to 90% (CH3CN
1), a noticeable increase in the average lifetime of BINNA was observed with a lifetime value of 0.57 ns, corresponding to three decay components with lifetimes and populations of 0.57 ns and 50%, 1.18 ns and 0%, and 0.57 ns and 50%. Furthermore, a significant enhancement in the average fluorescence lifetime of BINNA was observed upon increasing the water content to 90% (CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O = 1
H2O = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), with lifetime reaching 1.06 ns, having three decay components with lifetimes and populations of 0.46 ns and 0%, 1.06 ns and 50%, and 1.06 ns and 50%. In pure water, the lifetime of BINNA was decreased and the average lifetime was found to be 0.51 ns with three decay components having lifetime and population values of 0.56 ns and 63%, 1.44 ns and 6%, and 0.29 ns and 31%. The progressive enhancement in fluorescence lifetime with increasing water content suggests the formation of molecular aggregates, leading to an enhancement of the excited state. The enhancement of fluorescence lifetime (from 0.42 ns to 1.06 ns) with increasing water content is a direct consequence of the molecular packing mode. The formation of intermolecular π–π stacked aggregates in the poor solvent (high water content) restricts the intramolecular rotation, which, in turn, reduces the non-radiative transitions and enhances the probability of radiative decay, thereby increasing the fluorescence lifetime. These findings are consistent with the results obtained from steady-state fluorescence, dynamic light scattering (DLS), and scanning electron microscopy (SEM), collectively reinforcing the hypothesis of aggregation-induced emission enhancement in BINNA.
9), with lifetime reaching 1.06 ns, having three decay components with lifetimes and populations of 0.46 ns and 0%, 1.06 ns and 50%, and 1.06 ns and 50%. In pure water, the lifetime of BINNA was decreased and the average lifetime was found to be 0.51 ns with three decay components having lifetime and population values of 0.56 ns and 63%, 1.44 ns and 6%, and 0.29 ns and 31%. The progressive enhancement in fluorescence lifetime with increasing water content suggests the formation of molecular aggregates, leading to an enhancement of the excited state. The enhancement of fluorescence lifetime (from 0.42 ns to 1.06 ns) with increasing water content is a direct consequence of the molecular packing mode. The formation of intermolecular π–π stacked aggregates in the poor solvent (high water content) restricts the intramolecular rotation, which, in turn, reduces the non-radiative transitions and enhances the probability of radiative decay, thereby increasing the fluorescence lifetime. These findings are consistent with the results obtained from steady-state fluorescence, dynamic light scattering (DLS), and scanning electron microscopy (SEM), collectively reinforcing the hypothesis of aggregation-induced emission enhancement in BINNA.
| τ 1 (ns) | τ 2 (ns) | τ 3 (ns) | α 1 | α 2 | α 3 | τ av (ns) | χ 2 | |
|---|---|---|---|---|---|---|---|---|
| CH3CN | 0.81 | 4.15 | 0.24 | 0.03 | 0.02 | 0.95 | 0.42 | 1.10 |
CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
0.57 | 1.88 | 0.57 | 0.50 | 0.00 | 0.50 | 0.57 | 1.05 |
CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ) ) |
0.46 | 1.06 | 1.06 | 0.00 | 0.50 | 0.50 | 1.06 | 0.99 |
| H2O | 0.56 | 1.44 | 0.29 | 0.63 | 0.06 | 0.31 | 0.51 | 1.11 |
3.3. Sensing properties of BINNA towards metal ions
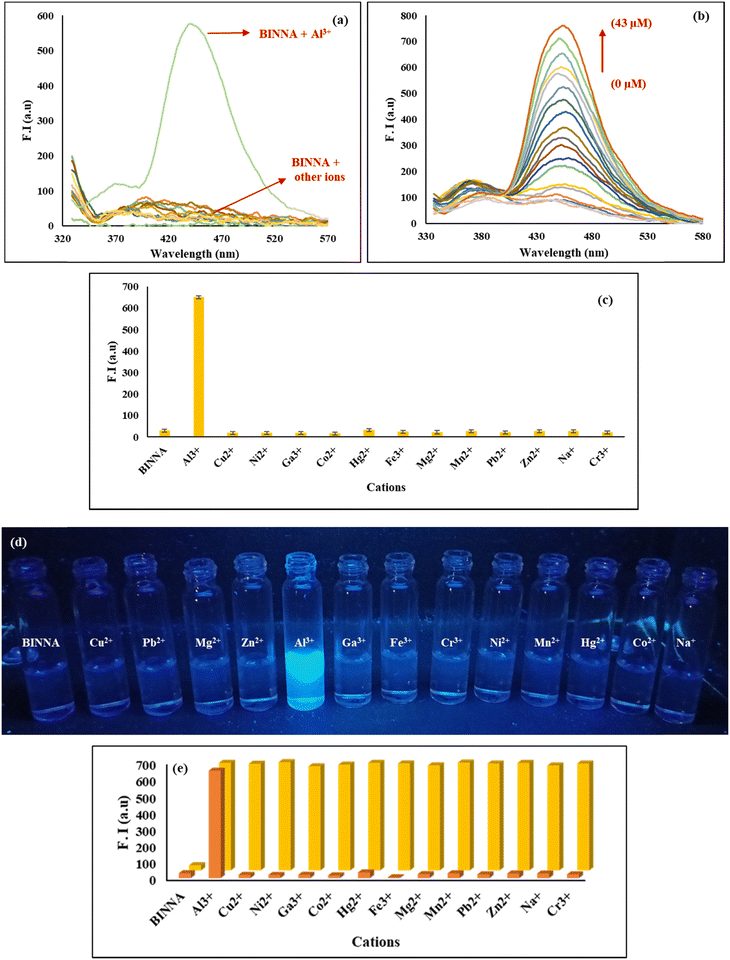
To assess metal ion detection, UV-vis absorption spectra of BINNA (20 µM, MeOH) were recorded in the absence and presence of various metal ions, including Cu2+, Ni2+, Ga3+, Al3+, Pb2+, Mg2+, Cr3+, Mn2+, Fe3+, Co2+, Zn2+, Na+, and Hg2+ (Fig. 6a and c). In its native state, BINNA exhibited two absorption bands at 325 and 355 nm. Upon the addition of aluminium ions (Al3+), a new band at 340 nm along with a red shift was observed, with the emergence of a new absorption band at 408 nm, accompanied by a noticeable light-yellow colour visible to the naked eye (Fig. 6d), whereas no significant spectral change was observed with other metal ions, suggesting that BINNA exhibits selectivity towards Al3+ ions. Further titrations with increasing concentrations of Al3+ ions (0–43 µM) revealed a gradual increase in the absorption band at 338 nm and the concurrent appearance of a 408 nm band, along with a clear isosbestic point at 388 nm, indicating the presence of two or more species in dynamic equilibrium at that wavelength (Fig. 6b).
3.4. Sensing properties of BINNA towards anions and nerve agents
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding mode between BINNA and DCP (Fig. S7c).
1 binding mode between BINNA and DCP (Fig. S7c).
To validate BINNA's selectivity, interference studies were performed by co-incubating BINNA with DCP (10 µM) in methanol, followed by the addition of various competing nerve agents and anions at equivalent concentrations (10 µM). We further checked the selectivity of BINNA for DCP ions in the presence of a higher concentration of interfering ions (50 µM). The fluorescence signal at 371 nm remained virtually unaffected (Fig. 8f and Fig. S8), clearly demonstrating that BINNA maintains high reliability in DCP detection even in the presence of potentially disruptive nerve agents and anions. This robust selectivity, combined with its low detection limit, reinforces BINNA's utility as a reliable and high-performance sensor for DCP.
3.5. Time-dependent study of BINNA
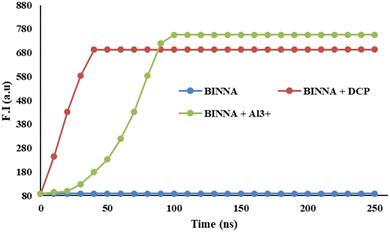
The response time of a fluorescence probe is crucial for the real-time detection of cations and nerve agents. Accordingly, time-dependent emission studies were performed to evaluate the response of BINNA toward Al3+ ions and DCP. As shown in Fig. 9, BINNA displayed weak fluorescence at 405 nm in the absence of Al3+ ions and DCP, with no significant change over time. However, upon the introduction of DCP, a pronounced increase in fluorescence intensity at 370 nm was observed, reaching a steady plateau within 40 s, whereas, upon the introduction of Al3+ ions, a marked increase in emission intensity at 450 nm was observed, reaching a steady plateau within 100 s. This rapid enhancement clearly demonstrates the fast response of the BINNA probe toward DCP and Al3+ ions, confirming its suitability for real-time detection applications.3.6. Plausible mechanism
Furthermore, to gain insight into the mode of interaction of BINNA with Al3+ ions and DCP, we recorded the HRMS spectra of BINNA in the absence and presence of Al3+ ions and DCP. The mass spectrum of free BINNA displayed an m/z peak of 577.3213 (M + H)+, whereas upon the addition of Al3+ ions to the solution of BINNA, the peak corresponding to the mass of BINNA at m/z 577.3213 diminished with the appearance of new peaks at m/z 194.9906 corresponding to the mass of compound 3 + Na+, whereas the peak at m/z 273.0049 corresponds to the mass of compound 3 + Al3+ + CH3OH + CH3CN and the peak at m/z 459.9230 is the peak corresponding to the mass of compound 2 + 2H2O. These results inferred that BINNA is hydrolyzed in the presence of Al3+ ions (Fig. S9). We also recorded the absorption and emission spectra of BINNA, BINNA + Al3+ and compound 3 to support the hydrolysis of BINNA in the presence of Al3+ ions (Fig. S10). The results of absorption and emission spectroscopy are in accordance with the HRMS results of BINNA in the presence of Al3+ ions.Furthermore, in the presence of DCP, new peaks at m/z 733.1271 corresponding to the mass of 4 + H2O and one peak at m/z 753.0900 corresponding to the mass of 4 + CH3CN were observed. These results indicated that DCP binds with BINNA in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry (Fig. S11). The plausible mechanisms for the detection of Al3+ ions and DCP by BINNA are shown in Fig. 10.
1 stoichiometry (Fig. S11). The plausible mechanisms for the detection of Al3+ ions and DCP by BINNA are shown in Fig. 10.
3.7. Time-correlated single photon counting (TCSPC) study
To gain further understanding of BINNA's interaction with Al3+ ions and DCP at 325 nm, a time-correlated single photon counting (TCSPC) experiment was conducted. Both BINNA and its complexes with Al3+ ions and DCP exhibited tri-exponential decay profiles, suggesting the presence of three distinct decay pathways (Fig. S12). For BINNA alone, three decay components with lifetime values of 1.38 ns, 6.20 ns, and 0.25 ns and populations of 18%, 7%, and 75% were observed, resulting in an average lifetime of 0.85 ns (Table 2). Upon interaction with DCP, the same three decay components were obtained with lifetimes of 0.81 ns, 5.10 ns, and 0.13 ns and populations of 3%, 4%, and 94%, with an average lifetime of 0.32 ns. Similarly, upon the addition of Al3+ ions, the complex also exhibited three decay components with lifetime values of 1.27 ns, 0.32 ns, and 5.90 ns and populations of 29%, 68%, and 3%, respectively, having an average lifetime value of 0.79 ns. The average lifetime of BINNA changed largely upon interaction with Al3+ and DCP, suggesting that its binding with metal ions and nerve agents occurred through a purely dynamic mechanism.| τ1 (ns) | τ2 (ns) | τ3 (ns) | α1 | α2 | α3 | χ 2 | τav (ns) | |
|---|---|---|---|---|---|---|---|---|
| BINNA | 1.38 | 6.20 | 0.25 | 0.18 | 0.07 | 0.75 | 1.09 | 0.85 |
| BINNA + DCP | 0.81 | 5.10 | 0.13 | 0.03 | 0.04 | 0.94 | 1.03 | 0.32 |
| BINNA + Al3+ | 1.27 | 0.32 | 5.90 | 0.29 | 0.68 | 0.03 | 1.12 | 0.79 |
3.8. Theoretical studies
The ground-state optimized geometries of BINNA and its derivative BINNA·DCP are shown in Fig. 11. Additionally, three low-lying Franck–Condon excitations (FCEs) were computed at S0 geometry, as summarized in Table 3. The time-dependent density functional theory (TDDFT) calculations revealed S0 → S1 excitation at 397 nm (experimental λmax ≈ 355 nm), primarily comprising HOMO → LUMO+1 (∼60%) and HOMO → LUMO (∼24%) transitions, with an oscillator strength of 1.1718. Molecular orbital analysis indicates electron density transfer from the naphthaldehyde moiety (HOMO) to the naphthalimide moiety (LUMO+1), suggesting an intramolecular charge transfer (ICT) process. This is supported by hole–electron analysis showing a modest spatial overlap (Sr = 0.69183) and a centroid separation of 1.676 Å. Additional transitions, S0 → S2 and S0 → S3, were calculated at 316 nm and 303 nm, with oscillator strengths of 0.0967 and 0.3561, respectively. Solvent effects (chloroform, acetone, DMSO, and methanol) produced only minor spectral shifts (392–399 nm) and the corresponding dipole moment variations (4.52–5.38 D), though these modest shifts appreciably influence emission behaviour (Table S2).| Probe | Transition | Absorption peak (λ; nm) | Oscillation strength (f) | Contributing molecular orbitals (MOs, %) |
|---|---|---|---|---|
| BINNA-C1 | S0 → S1 | 397.56 | 1.1718 | H → L + 1 (59.7%), H → L (24.3%) |
| S0 → S2 | 316.64 | 0.0967 | H → L + 2 (63.6%), H-1 → L + 1 (17.5%), H-1 → L (7.6%) | |
| S0 → S3 | 303.65 | 0.3561 | H-3 → L (72.9%), H-3 → L + 1 (23.4%) | |
| BINNA-C1-DCP | S0 → S1 | 390.07 | 1.12560 | H → L + 1 (73.5%), H → L (16.6%) |
| S0 → S2 | 305.59 | 0.15290 | H → L + 2 (67.2%), H-1 → L + 1 (13.9%) | |
| S0 → S3 | 303.38 | 0.37480 | H-2 → L (81.7%), H-2 → L + 1 (13.7%) | |
| S0 → S1 | 397.21 | 1.1688 | H → L + 1 59.6%, H → L 24.5% |
The molecular electrostatic potential (MEP) analysis identifies significant negative regions at the oxygen atoms of the naphthalimide and naphtholic units, indicating sites of electrophilic attack, while positive potential regions are localized around hydrogen atoms attached to the naphthalene, azine, and terminal naphthalimide rings, possible sites for nucleophilic interactions (Fig. 11). The analysis further suggests that the DCP analyte can engage in electrophilic interaction with the hydroxyl group on the naphthol ring.
Following geometry optimization of BINNA·DCP, TDDFT computations of electronic spectra show S0 → S1 excitation at 390 nm, with dominant contributions from HOMO → LUMO+1 (∼73%) and HOMO → LUMO (∼17%), and an oscillator strength of 1.1294. Similar to BINNA, this excitation reflects electron density migration from the naphthaldehyde to the naphthalimide moiety. However, hole–electron analysis reveals slightly reduced orbital overlap (Sr = 0.67792) and increased centroid separation (D = 1.903 Å), suggesting mildly hindered ICT character upon DCP binding. S0 → S2 and S0 → S3 transitions in BINNA·DCP were computed at 305 nm and 303 nm, with oscillator strengths of 0.1507 and 0.3795, respectively. Collectively, these results elucidate the electronic structures and solvent-responsive photophysical properties of BINNA and BINNA·DCP, underscoring their ICT potential and the modulation of their excited-state characteristics by solvent effects and analyte binding.
3.9. Practical applications for the detection of Al3+ ions and the nerve agent
 | ||
| Fig. 13 (a) Emission profile at 370 and 450 nm in the presence of different inputs. (b) Truth table and (c) fabrication of a logic gate integrating YES and INHIBIT gates. | ||
We constructed a logic circuit with two inputs (In DCP and In Al) and two outputs, measured as λem (output-1) and λem (output-2). As shown in the truth table in Fig. 13b, monitoring the emission band at 450 nm (output-1) results in an INHIBIT logic gate, where a fluorescence signal is observed only in the presence of Al3+ and is suppressed when DCP is concurrently present. Mechanistically, DCP possesses a highly reactive phosphonyl chloride group that can competitively react with the hydroxyl site of BINNA with a response time of only 40 s, forming a stable phosphorylated BINNA–DCP product, preventing the hydrolysis of BINNA in the presence of Al3+ as the response time for Al3+ to react with BINNA is 100 s, which is higher than that of DCP. Consequently, when both Al3+ and DCP are present, the fluorescence is strongly quenched (“OFF” state) at 450 nm, matching the behaviour of an INHIBIT logic gate, where DCP acts as the inhibitory input. This INHIBIT behaviour arises from a logical integration of AND and NOT operations, where the negation is applied to the input rather than the output. However, the analysis of output-2 indicates a YES logic gate response, where the presence of DCP and Al3+ or their combination leads to an emission band above the threshold. The integration of YES and INHIBIT gates within this molecular system enables the formation of a combinational logic circuit (Fig. 13c), demonstrating the potential of BINNA as a functional molecular logic element.
3.9.3.1. Detection of DCP in soil samples. We evaluated BINNA's ability to detect DCP in actual soil samples. Upon UV irradiation at 325 nm, all soil samples exhibited a marked fluorescence enhancement at 370 nm (Fig. 14a and Fig. S14a–c). Notably, sand samples exhibited a rapid and pronounced increase in fluorescence, while field and clay soils demonstrated progressive responses, reaching peak intensity within 10 and 20 minutes, respectively. These variations in response kinetics are attributed to intrinsic differences in soil characteristics, such as particle size, surface area, dispersibility, and light scattering behaviour in MeOH medium. Following the above procedure, the soil samples were spiked with DCP at various known concentrations, and the recovery rate was examined (Fig. S15a and Table 4). These results highlight BINNA's strong potential for sensitive and selective detection of DCP in real-world environmental samples, particularly in soil contexts with recovery rates >95%.
 | ||
| Fig. 14 The bar plots of the emission intensity unspiked and spiked (a) at 370 nm for DCP and (b) at 450 nm for Al3+ ions in different soil samples such as sand, field, and clay. | ||
| Al3+/DCP (spiked, µM) | Al3+ (found, µM) | Recovery (%) | DCP (found, µM) | Recovery (%) | |
|---|---|---|---|---|---|
| Sand soil | 10 | 9.04 | 90.4 | 9.87 | 98.7 |
| 20 | 18.24 | 91.2 | 19.54 | 97.7 | |
| 30 | 27.5 | 91.6 | 29.47 | 98.2 | |
| Field soil | 10 | 9.21 | 92.1 | 9.65 | 96.5 |
| 20 | 18.20 | 91.0 | 19.56 | 97.8 | |
| 30 | 28.55 | 95.1 | 28.98 | 96.6 | |
| Clay soil | 10 | 9.43 | 94.3 | 9.75 | 97.5 |
| 20 | 18.65 | 93.2 | 19.23 | 96.1 | |
| 30 | 28.85 | 96.1 | 29.12 | 97.0 |
3.9.3.2. Detection of Al3+ in soil samples. Furthermore, we investigated the capacity of the receptor BINNA to detect Al3+ ions in actual soil samples. Upon exposure to UV light at 325 nm, all tested soil samples displayed a significant increase in fluorescence at 450 nm (Fig. 14b and Fig. S16a–c). Among them, clay soil samples responded the fastest and most intensely, whereas field and sandy soils showed a slower, gradual rise in fluorescence. Following the above procedure, the soil samples were spiked with various known concentrations of Al3+ ions and the recovery rate was examined (Fig. S15b and Table 4). Overall, these findings underscore the strong capability of BINNA for the sensitive and selective detection of Al3+ in real environmental soil samples with a recovery rate > 90%.
4. Conclusion
In this study, we have successfully developed a bis-naphthalimide-based “turn-on” fluorescent probe, BINNA, for highly sensitive and selective detection of Al3+ ions and DCP in an MeOH solvent system, representing excellent aggregation-induced emission enhancement. BINNA responds distinguishably towards both analytes; upon the addition of DCP, a strong emission band at 370 nm was observed, while an intense emission band at 450 nm was observed upon the addition of Al3+. BINNA exhibits exceptional sensing performance towards aluminium ions (Al3+) and DCP with detection limits as low as 18.2 and 8.5 nm, respectively. HRMS spectra of BINNA were recorded in the absence and presence of the analytes to gain insight into the sensing mechanism. In addition, theoretical studies were conducted to support the experimental results. In addition to solution-based sensing, the optical properties of BINNA were harnessed to fabricate a logic gate by integrating “Yes” and “INHIBIT” gates using fluorescence inputs. The probe's applicability extends to real-world scenarios, successfully detecting DCP and Al3+ in environmental soil matrices. Moreover, its effectiveness in live-cell imaging of bacterial systems highlights its potential utility for environmental monitoring and bioimaging applications.Conflicts of interest
The authors declare no competing financial interests.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Material and Methods, Efficiency of BINNA, NMR and HRMS data. See DOI: https://doi.org/10.1039/d5tc04533a.Acknowledgements
KP thanks the SERB, New Delhi (CRG/2023/004080), and CEEMS (Project No. TIET/CEEMS/Regular/2021/018) for providing funds. SAI Labs, Acal Lab, and TIET for NMR and DST-FIST (SR/FST/CS-II/2018/69) for HRMS analysis are also acknowledged. Authors are also thankful to Prof. Rajesh Kumar, Central University of Punjab, Bathinda, for providing confocal laser scanning microscope facility. SG thanks CEEMS/TIET for providing the fellowship.References
- F. Zhang, N. W. Chan, C. Liu, X. Wang, J. Shi, H. T. Kung, X. Li, T. Guo, W. Wang and N. Cao, Water Quality Index (Wqi) as a Potential Proxy for Remote Sensing Evaluation of Water Quality in Arid Areas, Water, 2021, 13(22), 3250 CrossRef CAS.
- C. Immanuel David, N. Bhuvanesh, H. Jayaraj, A. Thamilselvan, D. Parimala Devi, A. Abiram, J. Prabhu and R. Nandhakumar, Experimental and Theoretical Studies on a Simple S-S-Bridged Dimeric Schiff Base: Selective Chromo-Fluorogenic Chemosensor for Nanomolar Detection of Fe2+ & Al3+ Ions and Its Varied Applications, ACS Omega, 2020, 5(6), 3055–3072 Search PubMed.
- B. T. Ji, L. P. Liu, J. H. Chen, L. L. Gao, Y. Sun, J. J. Wang, Z. P. Deng and Y. X. Sun, Ratiometric Fluorescence Sensor Based on Dye-Encapsulated Zn-MOF for Highly Sensitive Detection of Diquat in Tap Water and Apple Samples, Microchem. J., 2024, 207, 111663 Search PubMed.
- T. Zangmo and A. Siripinyanond, Exploring the Applicability of Nano-Selenium for Capture of Mercury Vapor: Paper Based Sorbent and a Chemical Modifier in Graphite Furnace Atomic Absorption Spectrometry, Anal. Chim. Acta, 2019, 1085, 29–38 CrossRef CAS PubMed.
- Z. P. Deng, W. Q. Hu, J. L. Yuan, Y. Sun, Q. Wang, Y. X. Sun, J. J. Wang, S. Z. Zhang and L. Xu, Dual-Ligand Zn-Based MOF as a Fluorescent Probe for the Detection of HSO4−, J. Mol. Struct., 2025, 1319, 139607 CrossRef CAS.
- Z. Qin, J. A. Caruso, B. Lai, A. Matusch and J. S. Becker, Trace Metal Imaging with High Spatial Resolution: Applications in Biomedicine, Metallomics, 2011, 3(1), 28–37 CrossRef CAS PubMed.
- B. Shen, xing; Qian, Y. Click Synthesis, Hg2+ Sensor and Intramolecular Fluorescence Resonance Energy Transfer in Novel BODIPY Dendrons, Sens. Actuators, B, 2017, 239, 226–234 Search PubMed.
- B. Li, H. Mei, Y. Chang, K. Xu and L. Yang, A Novel Near-Infrared “Turn-On” Fluorescent Probe for the Detection of Fe3+ and Al3+ and Its Applications in Living Cells Imaging, Spectrochim. Acta, Part A, 2020, 239, 118552 CrossRef CAS PubMed.
- A. Roy, U. Shee, A. Mukherjee, S. K. Mandal and P. Roy, Rhodamine-Based Dual Chemosensor for Al3+ and Zn2+ Ions with Distinctly Separated Excitation and Emission Wavelengths, ACS Omega, 2019, 4, 6864–6875 Search PubMed.
- P. Srisuwan, A. Sappasombut, W. Thongyod, T. Jantarat, V. Tipmanee, N. Leesakul and D. Sooksawat, Highly Sensitive and Selective Coumarin-Based Fluorescent Chemosensor for Cu2+ Detection, J. Photochem. Photobiol., A, 2022, 427, 113841 CrossRef CAS.
- Y. X. Sun, B. T. Ji, J. H. Chen, L. L. Gao, Y. Sun, Z. P. Deng, B. Zhao and J. G. Li, Ratiometric Emission of Tb(III)-Functionalized Cd-Based Layered MOFs for Portable Visual Detection of Trace Amounts of Diquat in Apples, Potatoes and Corn, Food Chem., 2024, 449, 139259 Search PubMed.
- Y. X. Sun, X. X. Li, L. L. Gan, M. X. Du, B. Zhao, W. D. Li, J. G. Li and W. K. Dong, A Novel Oligo(N,O-Donor) Fluorescent Probe Possessed the CHEF Effect for Identification of Aluminum Ions in the Environment, Chem. Pap., 2024, 78, 7001–7013 Search PubMed.
- X. Nan, Y. Huyan, H. Li, S. Sun and Y. Xu, Reaction-Based Fluorescent Probes for Hg2+, Cu2+ and Fe3+/Fe2+, Coord. Chem. Rev., 2021, 426, 213580 CrossRef CAS.
- H. Q. Dong, T. B. Wei, X. Q. Ma, Q. Y. Yang, Y. F. Zhang, Y. J. Sun, B. B. Shi, H. Yao, Y. M. Zhang and Q. Lin, 1,8-Naphthalimide-Based Fluorescent Chemosensors: Recent Advances and Perspectives, J. Mater. Chem. C, 2020, 8, 13501–13529 Search PubMed.
- S. Gupta, G. Kumar, V. Luxami and K. Paul, A Naphthalimide-Derived Chemosensor for Ratiometric Detection of Sulphide Ions: Insights into the S2−-Driven Reduction Cascade, Real-Time Applications and Live Cell Imaging of Bacterial Cells, Mater. Adv., 2025, 6, 4389–4401 RSC.
- S. Rani, S. Gupta, V. Luxami and K. Paul, A Novel Target and Biomarker Benzothiazolyl-Naphthalimide Probes for Precise and Selective Detection of Serum Albumin and Anticancer Activity, New J. Chem., 2022, 46, 12082–12092 RSC.
- S. Gupta and K. Paul, DNA Damage and Intercalation by Elinafide Modified Bis-Naphthalimides for Their Anticancer Activity, J. Mol. Liq., 2023, 382, 121980 CrossRef CAS.
- S. Gupta, V. Luxami and K. Paul, Deciphering Binding Potential of Naphthalimide-Coumarin Conjugate with c-MYC G-Quadruplex for Developing Anticancer Agents: A Spectroscopic and Molecular Modeling Approach, ACS Appl. Bio Mater., 2025, 8, 1077–1096 CrossRef CAS PubMed.
- B. Zhao, A. P. Luo, L. P. Liu, X. Bai, Y. Sun, Z. P. Deng and Y. X. Sun, A Non-Fluorescent Group-Based “off-on-off” Chemosensor for Detection of Al3+ and Fe3+ and Zebrafish Bioimaging, J. Mol. Struct., 2025, 1327, 141237 CrossRef CAS.
- B. Yao, J. Zhang, M. Han, L. Liang, X. Li, X. Cai and Y. Leng, Two Novel “Turn On” Fluorescent Probes for the Determination of Al3+ and Its Applications, Inorg. Chem. Commun., 2023, 153, 110798 Search PubMed.
- G. Kumar, I. Singh, R. Goel, K. Paul and V. Luxami, Dual-Channel Ratiometric Recognition of Al3+ and F− Ions through an ESIPT-ESICT Signalling Mechanism, Spectrochim. Acta, Part A, 2021, 247, 119112 CrossRef CAS PubMed.
- Y. Pan, P. Zhao, C. Zhou, L. Yan and X. Wu, A Dual-Functional Fluorescent Probe Based on Kaolin Nanosheets for the Detection and Separation of Aluminum Ions, Spectrochim. Acta, Part A, 2023, 295, 122636 CrossRef CAS PubMed.
- J. Song, X. Yang, S. Xie, G. Zhu, X. Rao, P. Zhao and Q. Jiang, A Novel “Turn On” Fluorescence Probe Based on a Caffeic Acid Skeleton for Detecting Al3+ and Bioimaging Application, Anal. Methods, 2023, 15, 3233–3239 RSC.
- Z. Ou, M. Wang, Z. Zhang, J. Liang, J. Tang, X. Lei, L. Yang, H. Liu and L. J. Ma, A Fluorescence and Colorimetric Bifunctional Probe for Recognition of Al3+ and Cu2+ and Its Applications in Cells and Environmental Water Systems, Microchem. J., 2023, 195, 109386 CrossRef CAS.
- Q. Zhang, D. Zhang, Z. Shi, C. Jin and Y. Jiang, Construction of Naphthalimide-Based Fluorescent Probe for Sequential Detection of Al3+ and NOR and Its Application in Tea and Honey, Spectrochim. Acta, Part A, 2025, 330, 125685 Search PubMed.
- N. Jain and N. Kaur, Smartphone Coupled, Solution, Solid and Vapour Phase Sensing of Nerve Agent Mimic by 1,8-Naphthalimide Derived Schiff Base, Results Opt., 2023, 12, 100466 CrossRef.
- Y. He, H. Wang, C. He, R. Zeng, F. Cheng, Y. Hao, S. Chen and P. Zhang, A Porphyrin-Based Colorimetric and near Infrared Fluorescent Probe for Reversible and Rapid Detection of Diethyl Chlorophosphate, Sens. Actuators, B, 2025, 425, 136983 Search PubMed.
- S. Rai, S. Lama, S. Pradhan, U. Darnal, J. Chourasia, S. Ahamed, N. Tohora and S. K. Das, A Donor-π-Donor Type Ratiometric Chromogenic Molecular Optode for Efficient Recognition of Sarin Analog, Diethylchlorophosphate, J. Fluoresc., 2025, 35, 11209–11219 CrossRef CAS PubMed.
- L. Zhang, K. Zhang, M. Wu, J. Ding, M. Feng, X. Li, S. Yang and H. Ma, Ultrasensitive Recognition of Trace Nerve Agents Enabled via a Thermally Activated Delayed Fluorescence-Based Fluorescent Probe, Anal. Chem., 2025, 97, 3344–3351 CrossRef CAS PubMed.
- M. Kumar, N. Kaur and N. Singh, NiCr2O4 Nanozyme-Based Portable Sensor Kit for On-Site Quantification of Nerve Agent Mimic for Environment Monitoring, Sens. Actuators, B, 2023, 392, 134080 CrossRef CAS.
- F. Xiao, D. Lei, C. Liu, Y. Li, W. Ren, J. Li, D. Li, B. Zu and X. Dou, Coherent Modulation of the Aggregation Behavior and Intramolecular Charge Transfer in Small Molecule Probes for Sensitive and Long-Term Nerve Agent Monitoring, Angew. Chem., Int. Ed., 2024, 63, 00453 Search PubMed.
- Q. Chen, J. Liu, S. Liu, J. Zhang, L. He, R. Liu, H. Jiang, X. Han and K. Zhang, Visual and Rapid Detection of Nerve Agent Mimics in Gas and Solution Phase by a Simple Fluorescent Probe, Anal. Chem., 2023, 95, 4390–4394 Search PubMed.
- A. Palta, P. Thakur, G. Kumar, P. Vashisht and V. Luxami, 1,10-Phenanthroline Appended Novel Schiff Base as a Selective Fluorescent Chemosensor for Nerve Agent Stimulants, Spectrochim. Acta, Part A, 2025, 329, 125601 Search PubMed.
- A. Palta, G. Kumar and V. Luxami, Excited State Double Proton Transfer Efficient Probe: Theoretical Investigation and Sensing Ability towards Pb2+ and Al3+ Ions, J. Photochem. Photobiol., A, 2022, 433, 114198 CrossRef CAS.
- A. Palta, G. Kumar and V. Luxami, Intramolecular Dual Hydrogen Bonded Fluorescent “Turn-On” Probe for Al3+ and HSO4− Ions: Applications in Real Water Samples and Molecular Keypad Lock, Spectrochim. Acta, Part A, 2023, 300, 122873 CrossRef CAS PubMed.
- S. Gupta, V. Luxami and K. Paul, Bacterial Cell Death to Overcome Drug Resistance with Multitargeting Bis-Naphthalimides as Potent Antibacterial Agents against Enterococcus Faecalis, J. Mater. Chem. B, 2024, 12(23), 5645–5660 Search PubMed.
Footnote |
| † Contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |