Open Access Article
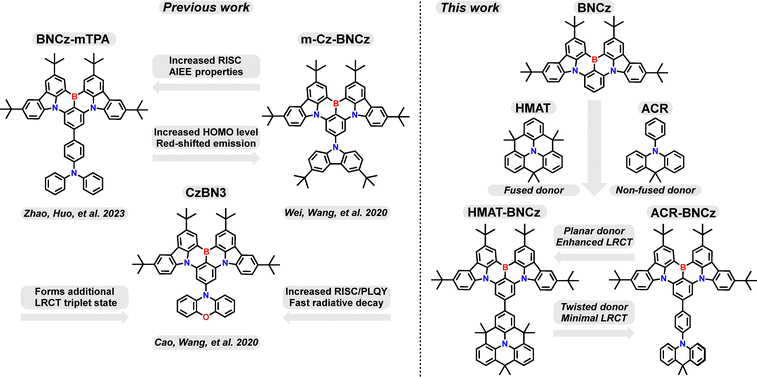
Open Access ArticleInvestigating the effects of planarized versus twisted donors on a multiple resonance thermally activated delayed fluorescence core
Mitchell J.
Demchuk
 ,
Ryoga
Hojo
,
Changfeng
Si
and
Zachary M.
Hudson
,
Ryoga
Hojo
,
Changfeng
Si
and
Zachary M.
Hudson
 *
*
Department of Chemistry, The University of British Columbia, 2036 Main Mall, Vancouver, British Columbia V6T 1Z1, Canada. E-mail: zhudson@chem.ubc.ca; Fax: +1-604-822-2847; Tel: +1-604-822-3266
First published on 19th February 2026
Abstract
Multiple resonance thermally activated delayed fluorescence (MR-TADF) emitters are known for their short-range charge transfer excited states and rigid, planar π-frameworks. In contrast, donor–acceptor TADF materials are typically dominated by long-range charge transfer excited states and twisted π-frameworks. An emerging design strategy that seeks to combine these two approaches can be achieved by appending a donor moiety to an MR-TADF core. This strategy has been shown to enhance desirable photophysical properties, such as increasing the rate of reverse intersystem crossing, due to spin-vibronic coupling between excited states of differing orbital symmetry. Here, two donor-appended materials based on the BNCz MR-TADF core are investigated using the donors HMAT (hexamethylazatriangulene) and ACR (9,9-dimethyl-9,10-dihydroacridine). Though structurally similar, the HMAT donor is a fused π-skeleton that provides a higher degree of planarity. In contrast, the ACR donor remains unfused and adopts a twisted structure. This difference leads to altered solution-state photophysical properties which are further investigated using a Lippert–Mataga analysis and theoretical calculations. Solid-state measurements of both emitters, HMAT-BNCz and ACR-BNCz, are then performed to elucidate the impact of donor rigidity on their TADF properties.
Introduction
Thermally activated delayed fluorescence (TADF) materials have undergone substantial development over the past decade, in particular for application in organic light emitting diodes (OLEDs).1–7 OLEDs are leading the field of display technology owing to their fast response time, high contrast ratio, and thin form factors.6,8 TADF materials provide several advantages over other classes of materials that can be used as emitters in OLEDs, namely that they can utilize both singlet and triplet excitons for radiative processes through reverse intersystem crossing (rISC), without the need for rare metal elements.2,9,10 Since singlet and triplet excitons are formed in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 in electroluminescent devices, organic TADF materials can theoretically achieve 100% internal quantum efficiencies in fully organic systems.1,10–12 There is however still potential to improve these materials in terms of their external quantum efficiencies, and the availability of stable, pure blue TADF emitters at display-relevant chromaticities. Therefore, the development and testing of various design strategies for TADF emitters is critical to full scale industrial implementation.13
3 in electroluminescent devices, organic TADF materials can theoretically achieve 100% internal quantum efficiencies in fully organic systems.1,10–12 There is however still potential to improve these materials in terms of their external quantum efficiencies, and the availability of stable, pure blue TADF emitters at display-relevant chromaticities. Therefore, the development and testing of various design strategies for TADF emitters is critical to full scale industrial implementation.13
In terms of molecular design, TADF is achieved by minimizing the energy gap between the lowest excited singlet (S1) and triplet (T1) excited states, termed ΔEST. This is achieved through spatial separation of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO), correlating to a decrease in the electron exchange energy that separates the S1 and T1 energy levels.1,10 A ΔEST < 0.3 eV generally allows for appreciable rISC rates, resulting in delayed fluorescence. To achieve this, donor–acceptor frameworks can be utilized, where the HOMO and LUMO are localized on the donor and acceptor moieties, respectively.1,11,14,15 Imposing a large torsion angle between the donor and acceptor moieties ensures sufficient separation of the HOMO and LUMO, inducing TADF character.15,16 However, their usefulness in OLEDs is challenged by the inherently broad emission spectra produced by compounds with long-range charge transfer (LRCT), leading to low colour purity.
To address this problem, Hatakeyama and coworkers developed multiple-resonance TADF (MR-TADF) which incorporates a donor atom (e.g. nitrogen) and acceptor atom (e.g. boron) in a rigid, polycyclic aromatic framework.17 This design allows the HOMO and LUMO to become atomically localized in an alternating fashion, which minimizes molecular vibrations to provide narrowband emission.2,3,17–19 When incorporated into OLEDs, these features provide high colour purity, high device efficiencies, and improved stability, enhancing overall device performance. Recent work has largely focused on extending the π-conjugation of these materials or increasing spin–orbit coupling (SOC) by introducing heavy atoms such as sulfur, selenium and bromine.14,20,21
Another promising strategy to enhance photophysical properties and tune emission colour is through appending donor moieties to MR-TADF cores.22–24 One of the most important advantages of this strategy is that appended donors can introduce LRCT character to these systems – which can in turn increase the rate constant for rISC (krISC) by altering the excited state character of S1 and T1.20,25–28 In the case of BNCz, this can be achieved through fusing an electron-donating unit para- to the boron atom, introducing a LRCT triplet excited state to the material.25,26,29 This then facilitates the forbidden transition between triplet and singlet SRCT states in the MR-TADF core through a higher-order spin-vibronic mechanism, enhancing the rate of rISC.3,25,30 The drawback of this approach is that if too much LRCT character is introduced, it can lead to spectral broadening and lower photoluminescent quantum yields (PLQYs).
The MR-TADF emitter BNCz (alternatively known as DtBuCzN) has received much attention since its discovery in 2020 by Wang, Zhang and coworkers.31 The polycyclic aromatic core is based on the fusion of two di-tert-butyl carbazole moieties with a central boron atom (Scheme 1). The BNCz motif has many attractive properties, including a narrow emission spectrum (λmax = 481 nm, FWHM = 22 nm), low ΔEST (0.13 eV), and high PLQY (91% in toluene).31 The ease of synthesizing and modifying this molecule has also proven advantageous, allowing many groups to tune its structure to achieve desired properties. For example, Zhao, Huo and coworkers recently reported BNCz modified with triphenylamine donors appended to the central benzene ring para to boron, which resulted in accelerated rISC and aggregation-induced emission enhancement (Scheme 1).26 This can be attributed to an increase of the charge transfer character of high-lying triplet excited states in BNCz, facilitated by the donor moieties. In 2020, Wang et al. synthesized m-Cz-BNCz, which has a carbazole donor attached to the meta position of the benzene rings with respect to boron (Scheme 1).27m-Cz-BNCz was significantly red-shifted compared to BNCz and showed narrowband green emission. A large torsion angle between the donor and BNCz core promoted combined LRCT and MR-TADF properties. The authors also found that the HOMO energy of m-Cz-BNCz was significantly increased compared to BNCz alone.27 This can be attributed to the fact that the central benzene ring in BNCz has the LUMO localized at the para position, while the HOMO is localized at the meta positions. Thus, di-tert-butylcarbazole appended to the meta position can influence the frontier molecular orbitals of the BNCz core.
Another prominent example of donor-functionalized MR-TADF cores comes from Yang, Cao and coworkers in 2023, where they attached a phenoxazine donor on the central benzene ring of BNCz, para to the boron atom (Scheme 1).25 The authors demonstrated that appending a phenoxazine donor to BNCz can boost rISC rates, while alteration of the electronic/steric properties in this molecule leads to emission from excited states of varying character (SRCT, LRCT or a hybrid SRCT/LRCT state).25 Many more strategies for tuning the properties of BNCz have been exploited, such as appending acceptor moieties, incorporating donors positioned for through-space charge transfer, and improving π-conjugation and molecular orbital delocalization.18,26,32–35
Herein, we describe two donor-appended MR-TADF molecules using BNCz as the core and either HMAT (hexamethylazatriangulene) or ACR (9,9-dimethyl-9,10-dihydroacridine) as donors on the central benzene ring para to boron. (Scheme 1). Our group has previously reported the use of HMAT and ACR donors in donor–acceptor TADF emitters, which have applications in OLED design, biological imaging, and energy transfer photocatalysis.36–41 We were interested to explore how the photophysical properties of the ACR donor linked to BNCz through a phenyl spacer would differ from that of the planar HMAT donor, which is fused to the phenyl spacer with dimethylmethylene groups. This fused structure encourages increased conjugation between HMAT and the MR-TADF core, while the unfused ACR donor remains twisted with respect to the BNCz unit (Scheme 1). We find that HMAT-BNCz and ACR-BNCz both have slightly red-shifted emission spectra with similar FWHM values when compared to BNCz. Additionally, while ACR-BNCz retains a PLQY of 91% in degassed toluene (identical to BNCz), the PLQY of HMAT-BNCz is somewhat lower at 80%. Notably, the solution photophysics of HMAT-BNCz indicates significant LRCT character, whereas ACR-BNCz retains MR-type SRCT character. A Lippert–Mataga analysis in concert with theoretical calculations point to emission from an alternative, LRCT excited state when stabilized in polar solvents for HMAT-BNCz, but not in ACR-BNCz. Finally, solid state measurements of both emitters doped into a 1 wt% PMMA film confirm their TADF nature, with delayed lifetimes of 96.8 µs and 101.9 µs for HMAT-BNCz and ACR-BNCz, respectively. Overall, this work showcases how structurally similar donors can influence the photophysical properties of the MR-TADF emitter BNCz, by adopting either a planar or twisted orientation, facilitated by ring fusion.
Results and discussion
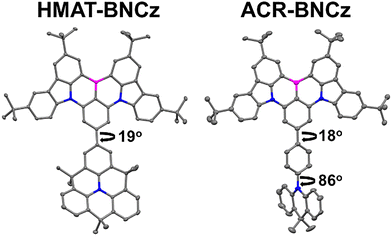
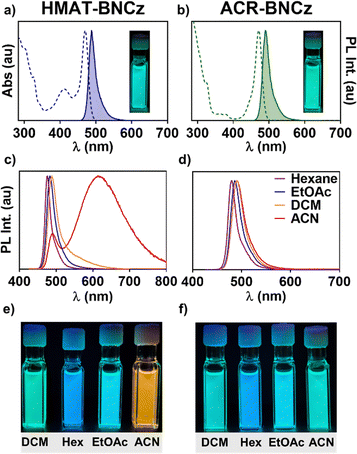
HMAT-BNCz and ACR-BNCz were synthesized through a Suzuki–Miyaura cross coupling reaction between BNCz-bromide and pinacolboranes of HMAT or ACR, respectively. BNCz-Br, as well as HMAT-BPin and ACR-BPin were prepared as per literature reports.35,42,43 Both reactions occur in good yield, 79% for HMAT-BNCz and 88% for ACR-BNCz (Fig. S1–S8). Single crystals of both emitters were obtained through slow evaporation from hexanes at room temperature and single crystal X-ray diffraction was used to obtain their solid-state structures. The main structural difference between in these two emitters is the torsion angle between the MR-TADF core and the donor group. In ACR-BNCz, the benzene ring linking ACR to the core is approximately planar, suggesting it is in conjugation with the BNCz moiety (torsion angle ∼18°). The torsion angle between the BNCz core and the acridine donor is significantly more twisted at ∼86° (Fig. 1). In contrast, for HMAT-BNCz the benzene ring linked to the MR-TADF core is fused by two dimethylmethylene groups, which locks it in a more planar configuration with a torsion angle of ∼19° between the HMAT donor and the BNCz core (Fig. 1).In the ultraviolet-visible (UV-Vis) absorption spectrum, HMAT-BNCz has a λmax = 471 nm (ε = 8.03 × 104 cm−1 M−1) and ACR-BNCz has a similar λmax = 470 nm (ε = 6.22 × 104 cm−1 M−1) (Fig. 2a and b). Though the λmax values are nearly identical, HMAT-BNCz has an additional large charge-transfer band from 375–430 nm, whereas ACR-BNCz has none. The PL spectra in a 10 µM toluene solution showed a λmax = 489 nm for HMAT-BNCz and λmax = 491 nm for ACR-BNCz, with narrow full width at half maximum (FWHM) of 22 nm and 24 nm, and small Stokes shifts of 18 nm and 21 nm, respectively. Both emitters showed evidence of positive solvatochromism, with hexanes showing the greatest blue shift and acetonitrile the greatest red shift (Fig. 2c and d). Supporting the hypothesis that HMAT induces more CT character than ACR, when HMAT-BNCz is placed in a 10 µM acetonitrile solution, its colour shifts to bright orange (Fig. 2e) and exhibits dual emission, with a new broad charge transfer (FWHM = 140 nm) at λmax = 615 nm, in addition to a narrow SRCT band at 494 nm. This is significantly different from the nature of ACR-BNCz in a 10 µM acetonitrile solution, which stays a blue/green in colour and has a λmax = 490 nm, indicative of SRCT type emission (Fig. 2f).
We then conducted a Lippert–Mataga analysis to further investigate the photophysical response of each emitter to solvent polarity, by examining the relationship between Stokes shift (νa − νf) and solvent orientational polarizability (Δf). The Lippert–Mataga equation (eqn (S1)) can then be used to determine the molecule's excited-state dipole moment (µe).44 The excited-state dipole moment value is known to be larger for LRCT-type emitters, when compared to emitters whose luminescence originates from locally excited or SRCT states.25,44,45HMAT-BNCz exhibits dual emission upon dissolution in the four most polar solvents tested, one with narrowband SRCT character and one with a broad,-redshifted emission peak showing LRCT character (Fig. 3). We predict this is most likely caused by stabilization of a low-lying S2 state which exhibits LRCT, due to the fused, planar HMAT donor. This is in contrast to ACR-BNCz, an unfused, twisted donor exhibiting only SRCT emission character even in the most polar solvents. The HMAT-BNCz emitter was found to have two excited-state dipole moments (µe) of 7.4 D and 39.5 D (D = Debye), while ACR-BNCz shows only one µe of 6.9 D (Fig. 3) (µe values calculated using eqn (S1)–(S4) and Table S1). The first µe of HMAT-BNCz and only µe of ACR-BNCz are smaller values, as expected of the narrowband SRCT emission originating from the MR-TADF core. The larger µe of 39.5 D for HMAT-BNCz corresponds to a new red-shifted broad emission band in more-polar solvents and can most likely be attributed to additional LRCT character induced by the HMAT donor on the BNCz core (see DFT analysis below).
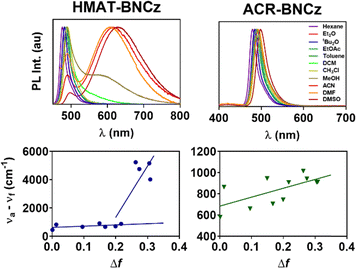 | ||
| Fig. 3 Normalized photoluminescent spectra of HMAT-BNCz and ACR-BNCz taken in each solvent as a 10 µM solution (top). Lippert–Mataga plots of HMAT-BNCz and ACR-BNCz (bottom). | ||
Subsequently, phosphorimetry studies were performed in a 10 µM 2-MeTHF glass at 77 K. The experimentally determined S1, T1 and ΔEST values were calculated by the onset of fluorescent and phosphorescent emission through obtaining steady-state and time-gated spectra (Fig. S11). HMAT-BNCz shows S1 and T1 energies of 2.65 eV and 2.48 eV respectively, giving a ΔEST = 0.17 eV. For ACR-BNCz, the S1 and T1 energies lie at 2.64 eV and 2.48 eV, giving a ΔEST = 0.16 eV. These ΔEST values are slightly larger compared to that of the parent BNCz compound, which has a ΔEST of 0.13 eV, yet are still well within range to showcase TADF behavior. In a 10 µM toluene solution, the PLQY of HMAT-BNCz was 80% (under N2) and 59% (in air), while the values for ACR-BNCz were slightly higher at 91% (under N2) and 66% (in air). PL decays in a toluene solution were measured using time correlated single-photon counting (TCSPC), while the delayed fluorescence component was monitored using multi-channel scaling (MCS). We found that HMAT-BNCz had a prompt lifetime of 5.14 ns (inert) and 4.48 ns (air), while ACR-BNCz had a prompt lifetime of 5.38 ns (inert) and 4.68 ns (air) (Fig. S13). The delayed fluorescence components were found to be 4.7 µs for HMAT-BNCz and 3.1 µs for ACR-BNCz under inert conditions (Fig. S14). Table 1 summarizes the solution state photophysics for each compound. Intrigued by the fact that HMAT-BNCz shows dual emission with a broad CT band while the more twisted and flexible ACR-BNCz does not, fluorescence lifetime measurements for each emitter were obtained in 10 µM acetonitrile solutions (Fig. S15 and S16). Under inert conditions the prompt fluorescence lifetimes for HMAT-BNCz were found to be 11.4 ns (at λmax = 615 nm) and 10.9 ns (at λmax = 494 nm), while ACR-BNCz has a prompt lifetime of 9.4 ns in acetonitrile. Interestingly, HMAT-BNCz was found to have a completely quenched delayed lifetime in acetonitrile (identical PL decay curves under nitrogen vs air) for both λmax = 615 nm and λmax = 494 nm emission bands, while ACR-BNCz retained a delayed fluorescence lifetime of 7.8 µs under nitrogen, while quenched in air.
| Entry | λ abs (nm) ε (104 cm−1 M−1) | λ max,em (nm) | FWHM (nm) | Φ air/ΦN2 | CIE (x,y) | τ airp (ns) | τ N2p (ns) | τ N2d (us) |
|---|---|---|---|---|---|---|---|---|
| All measurements were performed in 10 µM toluene solutions (λex = 375 nm). Absolute photoluminescence quantum yields were determined using an integrating sphere. | ||||||||
| HMAT-BNCz | 470 (6.2) | 489 | 22 | 0.59/0.80 | (0.085, 0.384) | 4.5 | 5.1 | 4.7 |
| ACR-BNCz | 471 (8.0) | 491 | 24 | 0.66/0.91 | (0.083, 0.423) | 4.7 | 5.4 | 3.1 |
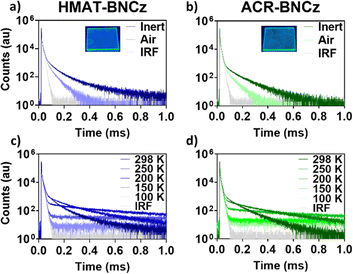
The solid state photophysical properties of these emitters were then investigated by doping them into a poly(methyl methacrylate) (PMMA) film using 1 wt% emitter. Both emitters showcased a blue-shifted emission in the solid state, with λmax at 486 nm for HMAT-BNCz and 485 nm for ACR-BNCz (Fig. S17). The PLQY of both compounds in the PMMA film were measured to be lower than in solution, with HMAT-BNCzΦPL = 21.6% and ACR-BNCzΦPL = 20.2%. The prompt fluorescence lifetime for HMAT-BNCz remained unchanged from the solution state under inert conditions at 5.1 ns, while ACR-BNCz showed a slight increase to τArp = 6.1 ns (Fig. S20). At 298 K, both emitters showed a delayed component, with HMAT-BNCz having a shorter delayed lifetime of 96.8 µs, and ACR-BNCz with a slightly longer delayed lifetime of 101.9 µs. Upon cooling from 298 K to 100 K, suppression of the delayed lifetime is observed confirming TADF character (Fig. 4). Summarized solid-state emission and lifetime data for 1 wt% doped PMMA films can be found in Table S2. Emission spectra and lifetime measurements for HMAT-BNCz and ACR-BNCz were also collected for 1 wt% doped Zeonex films to assess if there would be any change in photophysical properties within a less polar thin film. The emission spectra slightly blueshifts for HMAT-BNCz with λmax = 481 nm, while ACR-BNCz emission remains nearly unchanged at λmax = 486 nm. Under inert conditions, HMAT-BNCz shows a τArd = 111.1 µs and a τArp = 5.0 ns, while ACR-BNCz shows a τArd = 91.9 µs and a τArp = 7.3 ns. Overall, doping these emitters into PMMA versus Zeonex films was found to have little effect on their photophysical properties in the solid state (Fig. S21–S23 and Table S3). We then tested the emitters doped into a 1 wt% film of the OLED host material 1,3-bis(N-carbazolyl)benzene (mCP). Both HMAT-BNCz and ACR-BNCz exhibit a redshift compared to their PMMA and Zeonex film counterparts, with an λmax = 496 nm and 497 nm, respectively. Under inert conditions, HMAT-BNCz shows a τArd = 71.4 µs and a τArp = 5.5 ns, while ACR-BNCz shows a τArd = 80.9 µs and a τArp = 5.4 ns. Overall, the mCP host induces a significant redshift in both emitters and a shortened delayed lifetime in the solid state (Fig. S24–S26 and Table S4).
Photostability studies were then conducted on toluene solutions of both compounds to gauge their relative stabilities, using both 365 nm and 450 nm LED irradiation (ensuring both compounds had the same initial absorbance values at each excitation wavelength). UV-Vis absorption and PL spectra were taken at varying time intervals until decomposition was observed. Under 365 nm irradiation, HMAT-BNCz decomposed faster compared to ACR-BNCz over the course of 360 minutes (Fig. S27 and S28). Under these conditions it was determined that ACR-BNCz is approximately 35% more stable than HMAT-BNCz when comparing the decomposition slopes of UV-Vis spectra. Under 450 nm irradiation both compounds degraded significantly faster over 150 minutes (Fig. S29 and S30).
HOMO and LUMO energies were determined experimentally using cyclic voltammetry in a degassed DCM solution. HMAT-BNCz shows two reversible one-electron oxidations and one irreversible reduction, while ACR-BNCz shows two quasi-reversible one electron oxidations and one irreversible reduction (Fig. S31). From the oxidation and reduction potentials (referenced to ferrocene/ferrocenium, Fc0/+), HOMO/LUMO values of −5.27/−2.63 eV for HMAT-BNCz (Egap = 2.57 eV) and −5.41/−2.57 eV for ACR-BNCz (Egap = 2.56 eV) were obtained (Table S5). When compared to the parent MR-TADF material BNCz (HOMO = −5.40 eV, LUMO = 3.35 eV and Egap = 3.35 eV) both emitters have a lower LUMO level. However, the planar HMAT donor induces a significantly higher HOMO level, while the twisted ACR donor HOMO level is nearly identical.
Theoretical calculations
Density functional theory (DFT) calculations were performed to further investigate the electronic and photophysical properties of both emitters. Geometry optimizations were performed at the TPSSh/def2SVP level, followed by use of the Tamm–Dancoff approximation (TDA) for time-dependent DFT calculations at the RI-B2PLYP/def2-TZVP/CPCM(toluene) level of theory. TDA-DFT was used for excited state theoretical calculations, as it has proved suitable for large molecules which may suffer from triplet instability problems.46 In general, the results yielded by DFT match well with trends seen in experimental data. For HMAT-BNCz, the calculated S1/T1 values were 2.75 eV and 2.65 eV, respectively, with a ΔEST value of 0.10 eV. For ACR-BNCz, the theoretically calculated S1/T1 values were 2.74 eV and 2.61 eV, respectively, with a ΔEST value of 0.13 eV. Both emitters predicted ΔEST values are slightly lower than their experimentally determined values (less than 0.07 eV for each). To support the experimentally observed trends in fluorescent lifetime data and Lippert–Mataga analysis, excited state singlet energies were calculated at the RI-B2PLYP/def2-TZVP/CPCM(toluene) level of theory. HMAT-BNCz has a calculated S1 = 2.75 eV and S2 = 2.96 eV (ΔS1/S2 = 0.21 eV), while ACR-BNCz has a calculated S1 = 2.74 eV and S2 = 3.11 eV (ΔS1/S2 = 0.37 eV). Additionally, electron–hole diagrams were generated at the RI-B2PLYP/def2-TZVP/CPCM(toluene) level of theory.As seen in Fig. 5, the S1 state for both HMAT-BNCz and ACR-BNCz is dominated by MR-type SRCT character, with the electron and hole distributions localized on alternating atoms of the BNCz-core. Considering the electron–hole distribution for the S2 state, both molecules show more prominent LRCT character, like that of D/A TADF compounds. Using these theoretical predictions, we can determine that in polar solvents, the S2 excited state of HMAT-BNCz is likely stabilized to an energy lower than or close to the original S1 state, resulting in the observed LRCT emission. To further validate this theory, the oscillator strength (f) for HMAT-BNCz was calculated to be 0.69 for the S0 → S1 transition and 0.67 for the S0 → S2 transition. The oscillator strength of ACR-BNCz for the S0 → S1 transition was calculated to be 0.71, and zero for the S0 → S2 transition (Table S6). This information allows us to deduce that emission from the S2 state is much more likely in HMAT-BNCz than ACR-BNCz due to its significantly greater f (S0 → S2) and smaller S1/S2 energy gap, supporting experimental observations in the Lippert–Mataga analysis. The SOC values between S0/S1/S2 and T1/T2 were calculated at the TPSSH def2-TZVP level of theory in toluene using a conductor-like polarizable continuum (CPCM). The SOC between S1/T1 were found to be moderately larger for HMAT-BNCz at 0.17 cm−1, when compared to ACR-BNCz, with a very small value of 0.03 cm−1 (Table S7). This can likely be attributed to the greater difference in nature of orbital types introduced by the planar HMAT donor, when compared to the twisted ACR donor. Finally, to confirm the S1 and S2 excited state character, a percent charge transfer analysis was performed for both emitters at the RI-B2PLYP/def2-TZVP/CPCM(toluene) level of theory, using the interfragment charge transfer method (Table S8). For HMAT-BNCz, the S1 state has 95.2% SRCT and 4.8% LRCT character, while the S2 state has 38.1% SRCT and 61.9% LRCT character. For ACR-BNCz the S1 state was found to have 94.7% SRCT and 5.3% LRCT character, while the S2 state shows 46.9% SRCT and 53.1% LRCT character.
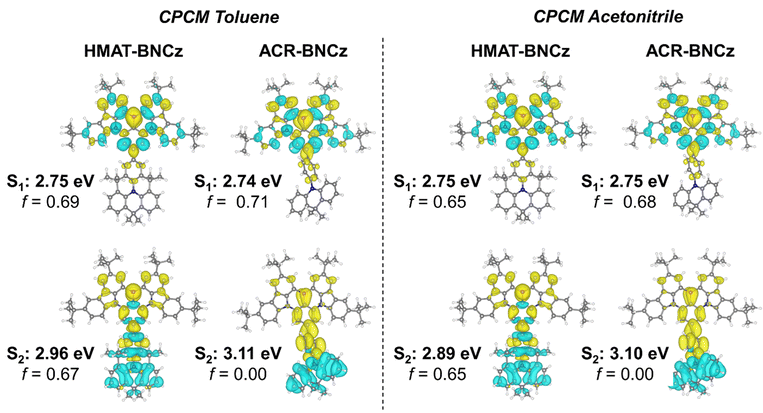 | ||
| Fig. 5 Hole–electron (blue-yellow) diagrams and oscillator strengths were calculated using TDA-TDDFT at the RI-B2PLYP/def2-TZVP level of theory using CPCM(toluene) and CPCM(acetonitrile). | ||
To further lend support to these findings, excited state calculations were repeated using the solvent acetonitrile within the CPCM model instead of toluene, at the RI-B2PLYP/def2-TZVP/CPCM(acetonitrile) level of theory. Calculations revealed HMAT-BNCz and ACR-BNCz ΔEST values of 0.12 eV and 0.14 eV, respectively, both less than 0.02 eV greater than those calculated in toluene. Importantly, calculations using acetonitrile show that HMAT-BNCz has an S1 = 2.75 eV and S2 = 2.89 eV (ΔS1/S2 = 0.14 eV), where the S1 energy does not change versus the theoretical calculations in toluene, but the S2 energy is stabilized by 0.07 eV. For ACR-BNCz, the S1/S2 values were changed by only 0.01 eV each with no change in ΔS1/S2. Calculated oscillator strengths in acetonitrile were found to be 0.65 for both the S0 → S1 and S0 → S2 transition in HMAT-BNCz, while values of 0.68 for the S0 → S1 transition and zero for the S0 → S2 transition were calculated for ACR-BNCz, consistent with experimental observations (see Tables S9–S11 for summarized theoretical data using CPCM(acetonitrile) and percent charge transfer analysis). Finally, electron–hole diagrams were generated at the RI-B2PLYP/def2-TZVP/CPCM(acetonitrile) level of theory (Fig. 5). Similar to the toluene CPCM model, the electron–hole distribution map using acetonitrile depicts an MR-type SRCT character dominated S1 state and a D/A-type LRCT character dominated S2 state.
Conclusions
In conclusion, TADF emitters HMAT-BNCz and ACR-BNCz have been synthesized utilizing the donor appended MR-TADF core design strategy. The fused and planarized nature of the HMAT donor leads to altered photophysical properties in solution when compared to the non-fused and twisted ACR donor. Solid-state measurements confirm the TADF activity of both compounds, with HMAT-BNCz having a τArd = 96.8 µs (ΔEST = 0.17 eV) and ACR-BNCz having a τArd = 101.9 µs (ΔEST = 0.16 eV) in 1 wt% PMMA films. Notably, HMAT-BNCz shows a large solvatochromic response in high-polarity solvents, leading to dual emission peaks with a significantly red-shifted and broadened emission spectra. This was investigated using a Lippert–Mataga analysis, showing HMAT-BNCz has two excited state dipole moments of µe = 7.4 D (SRCT character) and µe = 39.5 D (LRCT character), while ACR-BNCz has only one µe = 6.9 D (SRCT character). Theoretical calculations reveal that this difference in emission character is likely due to the S2 state of HMAT-BNCz being stabilized in more polar solvents, leading to dual emission. Although a TICT state cannot be fully ruled out, we believe there is strong evidence for dual emission originating from S1 and a stabilized S2 state in polar solvents such as acetonitrile for HMAT-BNCz – owing to its fused and planarized structure when compared to ACR-BNCz. This is supported by the fact that HMAT-BNCz has identical calculated oscillator strengths for both the S0–S1 and S0–S2 transitions of 0.65, with a small ΔS1/S2 in acetonitrile. This study enhances our understanding of how the donor-appended MR-TADF design strategy can be used to alter emission character between MR dominated SRCT and D/A dominated LRCT excited states, by exploring the difference between two structurally similar donors in a planar versus twisted geometry.Experimental details
General considerations
All reactions and material manipulation were carried out under a nitrogen atmosphere using either standard Schlenk line or glovebox techniques unless stated otherwise. Dry and degassed toluene was obtained using an Innovative Technologies Inc. solvent purification system. Ethanol (99%) and deionized water were degassed using nitrogen for 30 minutes prior to use. All reagents were obtained from Sigma-Aldrich, TCI America and Oakwood Chemical, and used as received unless otherwise stated. HMAT-BPin, ACR-BPin and BNCz-Br were prepared according to literature procedures.35,42,43 High resolution mass spectrometry (HRMS) data were obtained with field desorption (FD+) using a Jeol JMS-T100GCV AccuTOF GCv 4G spectrometer. The 1H and 13C{1H} nuclear magnetic resonance (NMR) spectra were measured on a Bruker AV III HD 400 MHz spectrometer with benzene-d6 (C6D6) or dichloromethane-d2 as the solvent.General photophysical characterization
Absorbance measurements were taken on a Cary 60 spectrometer and fluorescence measurements were performed using an Edinburgh Instruments FS5 spectrofluorometer or Edinburgh Instruments FLS1000 spectrofluorometer. Absolute photoluminescence quantum yields were determined using an Edinburgh Instruments SC–30 Integrating Sphere Module, with optical densities less than 0.1. Lifetimes were obtained using the Edinburgh Instruments FLS1000 spectrofluorometer and an EPLED (λexc = 375 nm or 300 nm) coupled with time-correlated single-photon counting (TCSPC), or a Xe µF lamp coupled with multichannel scaling (MCS). Concentrations of 10 µM were used unless otherwise stated. All solution state UV-Visible and fluorescence data were obtained using standard quartz cuvettes. Phosphorimetry was performed using a quartz tube and films were prepared on quartz or sapphire slides.Electrochemical methods
Cyclic voltammograms were measured using BASi Epsilon Eclipse potentiostat at room temperature with a standard cell three-electrode configuration. The working electrode was a 3 mm diameter glassy carbon electrode. The reference electrode was a Ag wire in a 0.2 M tetrabutylammonium hexafluorophosphate ([nBu4][PF6]) DCM solution, referenced externally to ferrocene/ferrocenium. The counter electrode was a Pt wire. Measurements were performed in a 0.2 M ([nBu4][PF6]) DCM solution with 1 mM analyte at scan rates of 100 and 200 mV s−1.Density functional theory
Quantum chemical calculations were conducted using Gaussian 16 with default settings unless otherwise stated. Ground state energies and corresponding geometries were calculated at the TPSSh/def2SVP level of theory. Tamm–Dancoff approximation (TDA) DFT was performed from the optimized frequencies at the B2PLYP/def2-TZVP level of theory using CPCM(toluene) and CPCM(acetonitrile) with ORCA 6.1.0. Electron–hole analysis and was performed using Multiwfn version 3.8 (dev) and visualized in VESTA (Visualization for Electronic Structural Analysis) version 3.5.8. Interfragment charge transfer analysis performed using Multiwfn version 3.8 (dev).Synthesis
HMAT-BNCz (C73H74BN3, Mw = 1004.2 g mol−1): a dry 50 mL Schlenk flask was charged with BNCz-Br (66 mg, 0.092 mmol, 1 equiv.), HMAT-BPin (45 mg, 0.092 mmol, 1 equiv.), Pd(PPh3)4 (11 mg, 0.0095 mmol, 0.1 equiv.), and K2CO3 (139 mg, 1.01 mmol, 11 equiv.), under a positive N2 atmosphere. In a separate 50 mL round bottom flask, a combination of 6 mL toluene, 2 mL EtOH and 2 mL deionized water were sparged with N2 for 30 minutes before being transferred via cannula to the reaction vessel. The reaction was then heated to 96 °C in an oil bath for 6 hours and completion was assessed by TLC. The reaction was then diluted with 20 mL of deionized water and extracted with toluene (1 × 30 mL) and DCM (2 × 30 mL). The combined organic phase was then evaporated under reduced pressure to give a dark yellow solid. The solid was redissolved in 30 mL DCM and then washed with deionized water (1 × 30 mL) and a brine solution (2 × 30 mL). The organic phase was then dried over anhydrous MgSO4 and evaporated under reduced pressure to give a yellow solid. The crude product was purified by silica column chromatography (gradient column starting at 100% hexanes to 90% hexanes: 10% DCM) and then precipitated out of hexanes to give a bright yellow solid (yield: 73 mg, 79%). 1H NMR (400 MHz, C6D6, 298 K): δH = 9.53 (d, 2H; JH,H = 1.70 Hz), 8.80 (s, 2H), 8.64 (d, 2H; JH,H = 1.66 Hz), 8.57 (d, 2H; JH,H = 8.87 Hz), 8.44 (d, 2H; JH,H = 1.93 Hz), 8.09 (s, 2H), 7.66 (2 H, dd; JH,H = 8.76, 1.96 Hz), 7.33 (m, 4H), 7.12 (t, 2H; JH,H = 7.63 Hz), 1.74 (s, 12H; iPr CH3), 1.68 (s, 18H; tBu CH3), 1.59 (s, 6H; iPr CH3), 1.46 (s, 18H; tBu CH3). 13C{1H} NMR (101 MHz, C6D6, 298 K): δC = 147.2, 145.5, 145.4, 144.9, 142.6, 139.2, 137.2, 132.9, 132.2, 131.1, 130.6, 130.5, 130.2, 129.0, 124.8, 124.4, 124.3, 124.1, 123.8, 123.5, 122.8, 120.9, 117.8, 114.8, 107.7, 36.1, 35.8, 35.3, 35.0, 34.8, 33.5, 33.4, 32.4, 31.9. HRMS (FD): m/z calc. for [C73H73BN3]+ 1002.6012; found 1002.6005; difference 0.7 ppm.ACR-BNCz (C67H66BN3, Mw = 924.1 g mol−1): a dry 50 mL Schlenk flask was charged with BNCz-Br (67 mg, 0.093 mmol, 1 equiv.), ACR-BPin (38 mg, 0.093 mmol, 1 equiv.), Pd(PPh3)4 (11 mg, 0.0093 mmol, 0.1 equiv.), and K2CO3 (141 mg, 1.02 mmol, 11 equiv.), under a positive N2 atmosphere. In a separate 50 mL round bottom flask, a combination of 6 mL toluene, 2 mL EtOH and 2 mL deionized water were sparged with N2 for 30 minutes before being transferred via cannula to the reaction vessel. The reaction was then heated to 96 °C in an oil bath for 6 hours and completion was assessed by TLC. The reaction was then diluted with 20 mL of deionized water and extracted with toluene (1 × 30 mL) and DCM (2 × 30 mL). The combined organic phase was then evaporated under reduced pressure to give a dark yellow solid. The solid was redissolved in 30 mL DCM and then washed with deionized water (1 × 30 mL) and a brine solution (2 × 30 mL). The organic phase was then dried over anhydrous MgSO4 and evaporated under reduced pressure to give a yellow solid. The crude product was purified by silica column chromatography (gradient column starting at 96% hexanes: 4% DCM to 70% hexanes: 30% DCM) and then washed with cold hexanes to give a bright yellow solid (yield: 76 mg, 88%). 1H NMR (400 MHz, CD2Cl2, 298 K): δH = 9.05 (d, 2H; JH,H = 1.60 Hz), 8.52 (s, 2H), 8.47 (d, 2H; JH,H = 1.67 Hz), 8.45 (d, 2H; JH,H = 8.96 Hz), 8.30 (d, 2H; JH,H = 1.88 Hz), 8.18 (s, 2H; JH,H = 8.26 Hz), 7.71 (2 H, dd; JH,H = 8.78, 1.99 Hz), 7.58 (d, 2H; JH,H = 8.22 Hz), 7.53 (dd, 2H; JH,H = 9.05, 1.31 Hz), 7.07 (dd, 2H; JH,H = 6.99, 1.35 Hz), 6.98 (dd, 2H; JH,H = 6.99, 0.95 Hz), 6.52 (dd, 2H; JH,H = 8.18, 0.71 Hz), 1.75 (s, 6H; iPr CH3), 1.66 (s, 18H; tBu CH3), 1.54 (s, 18H; tBu CH3). 13C{1H} NMR (101 MHz, CD2Cl2, 298 K): δC = 145.9, 145.4, 145.2, 145.1, 142.0, 141.9, 141.8, 141.4, 138.6, 132.4, 130.7, 130.6, 130.1, 127.5, 126.8, 125.7, 125, 124.0, 122.5, 121.9, 121.3, 121.1, 117.9, 114.7, 114.6, 107.3, 36.4, 35.5, 35.1, 32.3, 31.9, 31.5. HRMS (FD): m/z calc. for [C67H65BN3]+ 922.5386; found 922.5411; difference 2.5 ppm.
Conflicts of interest
The authors declare no conflict of interest.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information: Fig. S1–S26, Tables S1–S9 and further experimental details. See DOI: https://doi.org/10.1039/d5tc04366b.CCDC 2505730 and 2505731 contain the supplementary crystallographic data for this paper.47
Acknowledgements
The authors thank the Natural Sciences and Engineering Research Council of Canada (NSERC) for financial support of this work. MJD and RH are grateful for NSERC Graduate Scholarships, and ZMH is grateful for a Killam Accelerator Research Fellowship from the University of British Columbia. The authors also thank Dr Brian Patrick for his assistance with X-ray diffraction analysis, Sydney Mikulin for her assistance with density functional theory calculations, and Elvie Beaumont for her assistance with designing the TOC graphic.References
- H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Nature, 2012, 492, 234–238 CrossRef CAS PubMed.
- X. Wu, B.-K. Su, D.-G. Chen, D. Liu, C.-C. Wu, Z.-X. Huang, T.-C. Lin, C.-H. Wu, M. Zhu, E. Y. Li, W.-Y. Hung, W. Zhu and P.-T. Chou, Nat. Photonics, 2021, 15, 780–786 CrossRef CAS.
- H.-J. Cheon, S.-J. Woo, S.-H. Baek, J.-H. Lee and Y.-H. Kim, Adv. Mater., 2022, 34, 2207416 Search PubMed.
- S. Madayanad Suresh, L. Zhang, D. Hall, C. Si, G. Ricci, T. Matulaitis, A. M. Z. Slawin, S. Warriner, Y. Olivier, I. D. W. Samuel and E. Zysman-Colman, Angew. Chem., Int. Ed., 2023, 62, e202215522 CrossRef CAS PubMed.
- Y. Tang, Y. Liu, W. Ning, L. Zhan, J. Ding, M. Yu, H. Liu, Y. Gao, G. Xie and C. Yang, J. Mater. Chem. C, 2022, 10, 4637–4645 RSC.
- X. Yang, X. Xu and G. Zhou, J. Mater. Chem. C, 2015, 3, 913–944 RSC.
- Z. Xie, C. Cao, Y. Zou, X. Cao, C. Zhou, J. He, C.-S. Lee and C. Yang, Adv. Funct. Mater., 2022, 32, 2112881 CrossRef CAS.
- P. E. Burrows, G. Gu, V. Bulovic, Z. Shen, S. R. Forrest and M. E. Thompson, IEEE Trans. Electron Devices, 1997, 44, 1188–1203 CrossRef CAS.
- N. Aizawa, Y. Harabuchi, S. Maeda and Y.-J. Pu, Nat. Commun., 2020, 11, 3909 CrossRef CAS PubMed.
- C. M. Marian, Annu. Rev. Phys. Chem., 2021, 72, 617–640 CrossRef CAS PubMed.
- X. Cai and S.-J. Su, Adv. Funct. Mater., 2018, 28, 1802558 CrossRef.
- D. Zhang and L. Duan, Nat. Photonics, 2021, 15, 173–174 CrossRef CAS.
- Y. Im, M. Kim, Y. J. Cho, J.-A. Seo, K. S. Yook and J. Y. Lee, Chem. Mater., 2017, 29, 1946–1963 CrossRef CAS.
- Z. Yang, Z. Mao, Z. Xie, Y. Zhang, S. Liu, J. Zhao, J. Xu, Z. Chi and M. P. Aldred, Chem. Soc. Rev., 2017, 46, 915–1016 RSC.
- X.-K. Chen, Y. Tsuchiya, Y. Ishikawa, C. Zhong, C. Adachi and J.-L. Brédas, Adv. Mater., 2017, 29, 1702767 CrossRef PubMed.
- S. Weissenseel, N. A. Drigo, L. G. Kudriashova, M. Schmid, T. Morgenstern, K.-H. Lin, A. Prlj, C. Corminboeuf, A. Sperlich, W. Brütting, M. K. Nazeeruddin and V. Dyakonov, J. Phys. Chem. C, 2019, 123, 27778–27784 CrossRef CAS.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Adv. Mater., 2016, 28, 2777–2781 CrossRef CAS PubMed.
- J.-M. Jin, C. Shi, W.-C. Chen and Y. Huo, Chem. Commun., 2025, 61, 10731–10746 RSC.
- J. Han, Z. Huang, X. Lv, J. Miao, Y. Qiu, X. Cao and C. Yang, Adv. Opt. Mater., 2022, 10, 2102092 CrossRef CAS.
- X. Lv, J. Miao, M. Liu, Q. Peng, C. Zhong, Y. Hu, X. Cao, H. Wu, Y. Yang, C. Zhou, J. Ma, Y. Zou and C. Yang, Angew. Chem., Int. Ed., 2022, 61, e202201588 Search PubMed.
- S. A. Elgadi, S. Mikulin and Z. M. Hudson, Adv. Opt. Mater., 2025, 13, 2500683 CrossRef CAS.
- H. J. Kim and T. Yasuda, Adv. Opt. Mater., 2022, 10, 2201714 Search PubMed.
- S. A. Elgadi, D. M. Mayder, R. Hojo and Z. M. Hudson, Adv. Opt. Mater., 2023, 11, 2202754 Search PubMed.
- S. Wu, W. Li, K. Yoshida, D. Hall, S. Madayanad Suresh, T. Sayner, J. Gong, D. Beljonne, Y. Olivier, I. D. W. Samuel and E. Zysman-Colman, ACS Appl. Mater. Interfaces, 2022, 14, 22341–22352 CrossRef CAS PubMed.
- Z. Huang, H. Xie, J. Miao, Y. Wei, Y. Zou, T. Hua, X. Cao and C. Yang, J. Am. Chem. Soc., 2023, 145, 12550–12560 CrossRef CAS PubMed.
- G. Chen, J. Wang, W.-C. Chen, Y. Gong, N. Zhuang, H. Liang, L. Xing, Y. Liu, S. Ji, H.-L. Zhang, Z. Zhao, Y. Huo and B. Z. Tang, Adv. Funct. Mater., 2023, 33, 2211893 CrossRef CAS.
- Y. Xu, C. Li, Z. Li, Q. Wang, X. Cai, J. Wei and Y. Wang, Angew. Chem., Int. Ed., 2020, 59, 17442–17446 CrossRef CAS PubMed.
- Q. Wu, J. Li, D. Liu, Y. Mei, B. Liu, J. Wang, M. Xu and Y. Li, Dyes Pigm., 2023, 217, 111421 Search PubMed.
- Y. Liu, X. Xiao, Z. Huang, D. Yang, D. Ma, J. Liu, B. Lei, Z. Bin and J. You, Angew. Chem., Int. Ed., 2022, 61, e202210210 CrossRef CAS PubMed.
- T. J. Penfold, E. Gindensperger, C. Daniel and C. M. Marian, Chem. Rev., 2018, 118, 6975–7025 Search PubMed.
- Y. Xu, Z. Cheng, Z. Li, B. Liang, J. Wang, J. Wei, Z. Zhang and Y. Wang, Adv. Opt. Mater., 2020, 8, 1902142 Search PubMed.
- Y. Qu, Y. Xu, T. Huang, X. Song, K. Ye and Y. Wang, Angew. Chem., Int. Ed., 2025, 64, e202506201 Search PubMed.
- S. Luo, J. Wang, N. Li, X.-F. Song, X. Wan, K. Li and C. Yang, Angew. Chem., 2023, 135, e202310943 Search PubMed.
- T. Huang, Y. Xu, Y. Qu, X. Lu, K. Ye, X. Zhuang and Y. Wang, Adv. Mater., 2025, 37, 2503383 CrossRef CAS PubMed.
- S. Li, Z. Yang, Y. Xie, L. Hua, S. Ying, Y. Liu, Z. Ren and S. Yan, Chem. Sci., 2024, 15, 18335–18346 RSC.
- R. Hojo, K. Bergmann, S. A. Elgadi, D. M. Mayder, M. A. Emmanuel, M. S. Oderinde and Z. M. Hudson, J. Am. Chem. Soc., 2023, 145, 18366–18381 CrossRef CAS PubMed.
- E. R. Sauvé, D. M. Mayder, S. Kamal, M. S. Oderinde and Z. M. Hudson, Chem. Sci., 2022, 13, 2296–2302 RSC.
- R. Hojo, D. M. Mayder and Z. M. Hudson, J. Mater. Chem. C, 2022, 10, 13871–13877 RSC.
- A. M. Polgar, S. H. Huang and Z. M. Hudson, Polym. Chem., 2022, 13, 3892–3903 RSC.
- D. M. Mayder, C. M. Tonge, G. D. Nguyen, M. V. Tran, G. Tom, G. H. Darwish, R. Gupta, K. Lix, S. Kamal, W. R. Algar, S. A. Burke and Z. M. Hudson, J. Am. Chem. Soc., 2021, 143, 16976–16992 CrossRef CAS PubMed.
- N. R. Paisley, S. V. Halldorson, M. V. Tran, R. Gupta, S. Kamal, W. R. Algar and Z. M. Hudson, Angew. Chem., Int. Ed., 2021, 60, 18630–18638 CrossRef CAS PubMed.
- R. Hojo, D. M. Mayder and Z. M. Hudson, J. Mater. Chem. C, 2021, 9, 14342–14350 RSC.
- F.-Y. Hao, Y.-Z. Shi, K. Wang, X.-C. Fan, L. Wu, J. Ye, C.-J. Zheng, Y.-Q. Li, X.-M. Ou and X.-H. Zhang, J. Mater. Chem. C, 2020, 8, 10416–10421 RSC.
- N. Mataga, H. Chosrowjan and S. Taniguchi, J. Photochem. Photobiol. C Photochem. Rev., 2005, 6, 37–79 Search PubMed.
- J. Wiefermann, J. M. Kaminski, E. Pankert, D. Hertel, K. Meerholz, C. M. Marian and T. J. J. Müller, ChemPhotoChem, 2023, 7, e202200265 CrossRef CAS.
- F. J. Avila Ferrer, J. Cerezo, E. Stendardo, R. Improta and F. Santoro, J. Chem. Theory Comput., 2013, 9, 2072–2082 CrossRef CAS PubMed.
- (a) CCDC 2505730: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q3f09; (b) CCDC 2505731: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q3f1b.
| This journal is © The Royal Society of Chemistry 2026 |