 Open Access Article
Open Access ArticleAdvances in fluorescence-based point-of-care diagnostics: probes, nanostructures and integrated systems
Manuela
Cedrún-Morales
*ac,
Enrica
Soprano
ac,
Giulia
Vanoni
ac,
Anil
Chandra
 b,
Helena
Iuele
ac,
Francesco
Colella
cd,
Stefania
Forciniti
ac,
Valentina
Onesto
ac,
Giuliana
Grasso
ac,
Giuseppe
Gigli
acd and
Loretta L.
del Mercato
b,
Helena
Iuele
ac,
Francesco
Colella
cd,
Stefania
Forciniti
ac,
Valentina
Onesto
ac,
Giuliana
Grasso
ac,
Giuseppe
Gigli
acd and
Loretta L.
del Mercato
 *ac
*ac
aInstitute of Nanotechnology, National Research Council (CNR-NANOTEC), c/o Campus Ecotekne, via Monteroni, Lecce 73100, Italy. E-mail: manuelacedrunmorales@cnr.it; loretta.delmercato@cnr.it
bDepartment of Biotechnology, School of Life Sciences, Central University of Kashmir, Ganderbal 191201, Jammu and Kashmir, India
cTecnomed Puglia – Technopole for Precision Medicine (Biotech Lecce Hub), c/o Ecotekne Campus, via Monteroni, Lecce, 73100, Italy
dDepartment of Experimental Medicine, University of Salento, c/o Campus Ecotekne, via Monteroni, Lecce 73100, Italy
First published on 14th February 2026
Abstract
Fluorescence-based point-of-care (POC) technologies are rapidly evolving through advances in materials chemistry. This review provides a comprehensive overview of fluorescent molecular probes, proteins, hybrid nanostructures, metal–organic frameworks, silica platforms, carbon dots, and upconverting nanoparticles engineered for portable diagnostics. We highlight fundamental photophysical mechanisms, design strategies for improving brightness, stability, and multiplexing, and the integration of fluorescent materials with microfluidics, wearable interfaces, and smartphone readouts. Current limitations, translational challenges, and emerging trends, including AI-enabled data analysis and next-generation NIR and AIE fluorophores, are discussed. Together, these insights outline how material innovation is driving the next generation of high-performance POC sensing systems.
1. Introduction
The rapid advancement of medical technologies in recent decades has caused a significant change in the field of healthcare. Among these innovations, POC technologies have emerged as valuable tools for improving diagnostic efficiency, accessibility, and patient outcomes. Traditionally, medical testing required the collection of specimens and their transport to laboratories for processing, often resulting in significant delays between testing and diagnosis.1 POC technologies address this limitation by decentralising testing, allowing clinicians, and in many cases patients themselves, to obtain results rapidly in clinical settings, or even at home.2This shift has transformed medical diagnostics by delivering rapid results in a matter of minutes, which is critical for time-sensitive conditions and immediate clinical decision-making.3,4 Early POC devices included simple tools such as glucometers and urine pregnancy dipsticks. Over time, advances in microfluidics and Lab-on-Chip (LoC) technologies have expanded the capabilities of POC devices to detect a wide array of biomarkers, pathogens, and physiological parameters. Among available detection strategies, fluorescence-based methods offer distinct advantages that make them well-suited for a wide range of diagnostic applications.5 One of the key strengths of fluorescence-based detection lies in its high sensitivity and specificity, enabling accurate quantification of diverse analytes in complex biological samples. These methods are based on the exploitation of fluorescent molecules, or fluorophores, which are capable of emitting light upon excitation, allowing for the detection and quantification of target analytes with a high degree of precision. Additionally, fluorescence detection boasts a robust signal-to-noise ratio, rapid response time, and compatibility with miniaturized devices, further enhancing its appeal for POC applications.6 The improvements in fluorophore design and molecular biology techniques have facilitated the creation of an array of fluorescence-based POC assays, encompassing a diverse range of techniques such as fluorescence resonance energy transfer (FRET), time-resolved fluorescence (TRF), quantum dot-based detection, and single-molecule fluorescence.7–9 Each of these techniques offers unique advantages; for example, FRET-based assays excel in detecting protein-protein interactions and monitoring enzymatic activities,10 while quantum dot-based techniques provide exceptional sensitivity and multiplexing capabilities, allowing for the simultaneous detection of multiple targets.11
Integrating fluorescence detection with microfluidic and LoC systems has significantly expanded the potential of POC diagnostics.12 These miniaturized platforms facilitate the analysis of multiple samples while minimizing the volume of reagents and samples required for testing. Moreover, the development of novel fluorescent probes, such as upconverting nanoparticles (UCNPs) and aggregation-induced emission (AIE) fluorophores, has opened new avenues for enhancing the performance of fluorescence-based POC assays in terms of sensitivity, specificity, and stability.13,14 Combining fluorescence detection with other emerging technologies, such as smartphone-based imaging and artificial intelligence (AI), has resulted in the development of user-friendly, cost-effective, and highly accurate POC devices. Smartphone-based imaging systems can capture and analyse fluorescence signals, while artificial intelligence algorithms can interpret the data and provide diagnostic results, enabling healthcare professionals to make well-informed decisions regarding patient care.15,16 These developments also facilitate remote monitoring and telemedicine, broadening access to diagnostic services for underserved and rural populations.
The primary objective of this review is to provide a comprehensive and critical overview of fluorescence-based POC technologies. We discuss the fundamental detection principles, major technological advances, and broad landscape of current applications. Particular emphasis is placed on the integration of fluorescence readout with microfluidic and LoC architectures, which enables rapid, automated, and multiplexed analysis. We then examine the evolution of advanced fluorescent probes, including quantum dots (QDs), UCNPs, and AIE fluorophores, and their impact on assay sensitivity, specificity, and operational robustness. Current limitations and practical constraints, spanning probe stability, matrix interference, standardization, and device miniaturization, are critically assessed. Finally, we outline emerging opportunities and future directions that may drive the next generation of fluorescence-enabled POC platforms, ultimately aiming to enhance diagnostic performance, patient care, and accessibility across clinical, environmental, and resource-limited settings.
2. Fluorescence detection techniques
This section provides an overview of fluorescence-based detection techniques, focusing on the fundamental principles most relevant to sensing strategies, material innovation, and integration into point-of-care (POC) diagnostic platforms.2.1. Fluorescence spectroscopy
Fluorescence spectroscopy is a powerful analytical technique that exploits the photophysical properties of fluorescent molecules to detect and quantify target analytes in biological samples.17,18 Upon optical excitation, fluorophores emit light at longer wavelengths, enabling efficient separation of excitation and emission signals and highly sensitive optical readout (Fig. 1).19,20 The fluorescence response depends on several physicochemical parameters, including temperature, pH, fluorophore concentration, and interactions with the surrounding microenvironment.21 These dependencies make fluorescence spectroscopy particularly attractive for chemical and biological sensing. Measurements can be performed using steady-state or time-resolved approaches;20,22 however, steady-state fluorescence is most widely adopted due to its simpler implementation and suitability for miniaturized and portable POC devices.23–25 Beyond small-molecule fluorophores, fluorescence spectroscopy increasingly relies on nano- and microparticle-based systems, fluorescent proteins, and hybrid constructs. In nanoparticle-based sensors, fluorescent dyes can be incorporated or coupled to the nanostructure, enabling enhanced brightness, improved photostability, and resistance to photobleaching.26,27 Moreover, the intrinsic optical properties of certain nanomaterials allow spatially resolved detection using microscopy-based readouts, which is advantageous for device-integrated sensing applications. | ||
| Fig. 1 Jablonski diagram showing excitation, emission and other kinds of transitions of a fluorophore with their associated time scales. | ||
Quenching mechanisms are commonly classified as static or dynamic. Static quenching occurs when the fluorophore and quencher form a non-fluorescent complex before the excitation takes place. In this case, the fluorescence quenching is independent of the fluorophore concentration and is described by the Stern–Volmer equation:35
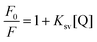 | (1) |
Dynamic quenching occurs when the fluorophore and quencher interact with each other in a collisional encounter. In this case, the fluorescence quenching is dependent on the fluorophore concentration and is described by the modified Stern–Volmer equation:
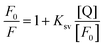 | (2) |
The distinction between static and dynamic quenching is therefore essential for sensor design, calibration, and interpretation of fluorescence responses in material-integrated sensing platforms.36
2.2. Fluorescence microscopy
Fluorescence microscopy is an imaging technique widely used to visualize the spatial distribution and dynamics of biomolecules in cells and tissues.39–42 Its application in diagnostics has expanded alongside the development of advanced probes and imaging strategies compatible with live and minimally invasive measurements.19,43–45 Despite its high sensitivity and specificity, fluorescence microscopy is limited by photobleaching, phototoxicity, and diffraction-limited spatial resolution.46 Considerable efforts have therefore been directed toward improving image resolution, leading to the development of super-resolution techniques capable of achieving lateral resolutions in the 20–50 nm range.2.3. Fluorescence lifetime imaging (FLIM)
Fluorescence lifetime imaging (FLIM) is an advanced optical technique that combines spatial resolution with time-resolved detection to probe molecular interactions and microenvironmental changes beyond intensity-based imaging.54,55 By measuring fluorescence decay times, FLIM provides concentration-independent contrast that is particularly valuable for sensing applications involving parameters such as pH, viscosity, ion concentration, and protein–protein interactions.56–58In its simplest formulation, the temporal decay of fluorescence intensity following pulsed excitation can be described by a mono-exponential function:
| I(t) = αe−t/τ |
The fluorescence lifetime reflects the average time a fluorophore remains in the excited state and is highly sensitive to its local molecular environment. As a result, lifetime-based measurements are less affected by variations in fluorophore concentration, excitation intensity, photobleaching, or uneven illumination compared to intensity-based fluorescence imaging.57
Fluorescence lifetime can be measured using time-domain or frequency-domain approaches. In time-domain FLIM, the samples are excited with short light pulses and the fluorescence decay is directly recorded, whereas in frequency-domain FLIM, the excitation light is intensity-modulated and lifetime information is extracted from phase shifts in the emitted signal.55
FLIM implementations range from widefield to scanning configurations and are compatible with both imaging and non-imaging modalities, including endoscopic and fiber-based systems.57,60–63 Owing to its robustness and sensitivity to biochemical changes, FLIM has found increasing application in tissue diagnostics and fluorescence-guided procedures, where lifetime contrast enables non-invasive or minimally invasive characterization of biological tissues.64
3. Fluorescent probes and labels
Fluorescent probes have been extensively employed not only for sensing, as optical imaging tools, but also for therapy as optical contrast agents or phototherapy agents. Fluorescent molecular probes and labels are made up of two components: a reporter and a recognition unit. The fluorophore, which works as a reporter, emits light upon excitation, while the recognition unit selectively recognizes the given molecular process or targets of interest, translating the recognition event in a well-defined manner into changes of the fluorescence properties of the reporter.65 Thus, fluorophore reporters are the centerpiece of each fluorescence molecular probe/label that in turn, represents the key components underlying the fluorescence-based POC technologies.A wide range of probes and labels are available, and the choice depends on the desired application, detection modality, and sample properties (Fig. 4). Fluorescent compounds can be generally classified into four categories considering their molecular complexity and synthetic methods: small fluorophores made up of molecules of 20–100 atoms,66 such as organic compounds,67 and larger fluorophores that include proteins and peptides,68–70 synthetic oligomers and polymers such as polyfluorenes,71 and polyphenylene vinylenes,72 and multi-component systems such as QDs73,74 and UCNPs.75,76 These fluorescence probes work by adopting different sensing mechanisms that include FRET, photo-induced electron transfer (PET), intramolecular charge transfer (ICT) and excited state intramolecular proton transfer (ESIPT).77
The fluorescence intensity change that occurs when the emission spectrum of a donor overlaps with the adsorption spectrum of an acceptor and the distance between the donor and the acceptor is suitable (<100 Å) is called fluorescence resonance energy transfer and it results in the fluorescence quenching of the donor and fluorescence enhancing of the acceptor.77,78 Bioluminescence resonance energy transfer (BRET) is a type of FRET where the donor luminophore is represented by a bioluminescent molecule and its electrons are excited by a bioluminescent chemical reaction.79 The introduction of an analyte can inhibit the energy transfer by changing the dipole, molecular orientation and so on, causing the fluorescence quenching of the acceptor and fluorescence enhancement of the donor.80,81
The electron transfer from a receptor (donor) to a fluorophore (acceptor) by intermolecular transfer, called the PET mechanism, leads to fluorescence quenching of the fluorophore. Like FRET, the PET process can be partially or completely inhibited when the receptor binds to the target, causing the restoration of the fluorophore fluorescence.82
The electron transfer from a receptor (donor) to a fluorophore (acceptor) by intramolecular transfer, called the ICT mechanism, represents a PET process where the donor and the acceptor are connected by a conjugate linker to form a conjugate system. The electronegativity of the receptor unit changes once the ICT probe interacts with the target and causes a change in the donor–acceptor system. As a consequence, the excitation of the target bound to the ICT probe produces a larger dipole and a change in the excited state due to the change in electron distribution.83 Hence, ICT probes possess changes in fluorescence intensity and shifts in emission wavelengths as well when they detect analytes.84
The four-level photocycle process of the photoinduced tautomerization of a specific enol–ketone is usually referred to as the ESIPT mechanism.85 The ground state of the ESIPT molecule usually exists in the form of an enol (E) and its photoexcitation leads to intramolecular charge rearrangement. In turn, the intramolecular charge rearrangement causes a simultaneous enhancement of the acidity of the proton donor and the alkalinity of the proton acceptor that rapidly converts the excited enol form (E*) into the excited ketol form (K*).86 Lastly, the excited ketol K* returns to the original E form after the radiative decay back to the electronic ground state, by reverse proton transfer (RPT).86
Recently, POC technologies based on fluorescence imaging have been developed employing the different classes of fluorophores and the different mechanisms of fluorescence. Fluorescence in fact appeared as one of the most promising approaches among the different bioimaging and biosensing techniques, such as magnetic resonance imaging (MRI), ultrasound (US), positron emission tomography/computed tomography (PET/CT), single-photon emission computed tomography (SPECT) and computed tomography (CT). Fluorescence allows extreme customization, and selective69,87 and sensitive88,89 target analyte detection in complex biological samples, it provides abundant information regarding biomolecules, and it results in a more holistic approach for the imaging of targets in living organisms.41,90
3.1. Organic fluorophores
Fluorescent organic fluorophores have been extensively used as raw materials in various biomedical, food industry91 and textile92 applications, thanks to their high versatility and their simple operation.93Organic fluorophores include a diverse class of synthetic organic compounds characterized by their ability to absorb light at specific wavelengths and emit fluorescence upon returning to their ground state.94 Their chemical structure is distinguished by the presence of several combined aromatic groups, or planar or cyclic molecules with different π bonds that generate rigid structures.94 According to the Woodward–Fieser rules, conjugated systems with fewer than eight double bonds absorb only in the ultraviolet region and appear colorless. Adding more double bonds causes a redshift in absorption, lowering the energy and shifting the color from yellow to red. Fluorogens are dyes that are colorless and non-fluorescent in solution but become fluorescent upon binding to a target, and can also serve as selective fluorescent probes.95 The Woodward–Fieser rules and other empirically derived guidelines help predict the absorption maximum wavelength in the UV–visible spectrum of a compound, allowing for flexible and customizable design of organic fluorophores.96 Hence, the photophysical properties of the organic fluorophores can be tuned to obtain specific wavelengths in their absorption and emission spectra,97 to increase their Stokes shift,98 to improve fluorophore photostability99 and to ensure high quantum yields.100 Additionally, organic fluorophores can be easily designed with functional groups that facilitate their conjugation to scaffolds or target biomolecules, allowing selective labelling, detection and their successful administration into cells.101–105 Moreover, organic fluorophores exhibit an inherent high sensitivity and strong specificity with low interference from sample matrices.69,106 Lastly, organic fluorophores are dyes that can be employed as inks.107 Thus, they can be printed on absorbent materials to assemble cheap POC devices.
Cai et al. developed a reliable lateral flow immunoassay (LFIA) by a facile one-step synthesis of red fluorescent microspheres containing Nile Red, enhancing stability and reducing background noise (Fig. 5A).129 By combining this with immunochromatography, they established a new POC testing platform for the rapid and early detection of cardiac troponin I (cTnI), a cardiac-specific biomarker for acute myocardial infarction (AMI).
 | ||
| Fig. 5 (A). PS microspheres functionalized with carboxyl groups via copolymerization of styrene and acrylic acid, labeled with Nile Red, and bioconjugated with anti-cTnI antibodies. (i) Schematic of the preparation. (ii) Structure of Nile Red. (iii) TEM images showing uniform morphology. (iv) Sandwich-type LFIA for cTnI detection: the sample migrates, binds fluorescent probe-McAb1, and is captured by immobilized McAb2 on the test line; the control line captures excess probes; fluorescence quantified under UV. Reproduced with permission129 copyright 2018 MDPI. (B) Skin-interfaced microfluidic device for sweat analysis of Cl−, Na+, and Zn2+. (i) Stable fluorescent reference dye in ionic liquid with excitation/emission spectra. (ii) Schematic of the multilayer microfluidic platform for sweat collection and in situ assays. (iii) Fluorescence images of the embedded chemical probes. (iv) Peeling the detachable black shield reveals sweat indicator channels. Reproduced with permission130 copyright 2018 Royal Society of Chemistry. C. Fluorescent compound array for portable chemical classification. (i) Kaleidolizine (KIz) structure and HOMO/LUMO orbital illustration. (ii) Array of 75 KIz derivatives on wax-printed cellulose under ambient and UV light; inset shows size comparison. (iii) 3D cradle device for smartphone-based detection. (iv) Photograph of the device in operation. (v) Automated fluorescence pattern analysis using machine learning for VOC classification. Reproduced with permission139 copyright 2021 Elsevier. | ||
Sekine et al. developed a wearable PDMS microfluidic device with fluorescent probes for real-time sweat analysis (Fig. 5B).130 Paired with a smartphone, it detects chloride, sodium, and zinc, demonstrating portable, continuous biomarker monitoring.
Ghassemi et al. explored contrast-enhanced near-infrared (NIR) fluorescence imaging using a mobile phone-based POC testing platform, employing indocyanine green (ICG) to visualize tissue perfusion and vasculature. Imaging on tissue phantoms and an ex vivo model confirmed the feasibility of mobile NIR imaging and highlighted performance variations based on wavelength, pixel color, and image processing.131
Lei He et al. developed a compact, integrated device for rapid and accurate screening of bovine mastitis via somatic cell counting (SCC). The system combines a custom cell-counting chamber pre-loaded with acridine orange dye and a miniature fluorescent microscope, providing a simple and practical POC diagnostic solution.132
Pharmacogenetic (PGx) testing and theranostics are essential components of personalized medicine, leveraging genetic variations such as single nucleotide polymorphisms (SNPs) to explain individual differences in drug response.133 For instance, the Spartan RX CYP2C19 system enables POC genotyping to guide antiplatelet therapy. Buccal swabs are collected in individual kits containing swabs and reagent tubes specific for the CYP2C19 *2, *3, and *17 alleles. The platform automates DNA extraction, PCR amplification, allele detection, and genotyping, using fluorescent-labeled probes and optical detection channels to identify each variant, with a sensitivity of 0.1 ng µL−1.134
Continuous monitoring of body fluids plays a vital role in modern healthcare by enabling early detection of health changes, improving treatment outcomes, and reducing the risk of complications. Tears, for example, known to closely reflect blood biomarker levels, are a valuable, non-invasive source for assessing eye health.135
Yetisen et al. developed a fluorescent wearable scleral lens sensor for POC diagnosis of ocular diseases through quantitative tear analysis. The device measures pH and five key ions (Na+, K+, Ca2+, Mg2+, and Zn2+) at physiological levels using specialized fluorescent probes embedded in an engraved scleral lens. The system includes a portable readout device with LEDs and optical filters, coupled with smartphone-based data processing and user interface. This integrated approach enables assessment of dry eye severity stages and differentiation of disease subtypes.136,137
Finally, the AFIAS IFX is an automated fluorescence lateral flow immunoassay for rapid POC monitoring of infliximab levels in patients with intestinal bowel disease (IBD). Unlike traditional ELISA testing that takes days, this system delivers quantitative results in 20 minutes using an all-in-one cartridge (comprising sample dilution buffer, antibody conjugators, and a strip). This enables clinicians to check infliximab levels immediately before infusion and make real-time dosage decisions, improving therapeutic drug monitoring and patient outcomes.137
Beyond clinical diagnostics, fluorescence-based POCT devices have recently expanded into the environmental and food safety sectors. In the field of water quality monitoring, Leonard et al. introduced a smartphone-enabled POC platform for the rapid quantification of microplastics in water using Nile Red fluorescence staining. A 3D-printed attachment and custom app enable semi-automated analysis of particles as small as 20 µm within 10 minutes. The portable, cost-effective system showed strong agreement with lab microscopy and works on synthetic and environmental samples, making it suitable for field monitoring and community-led research.138
Fluorescence-based sensing platforms are increasingly being adapted for POC chemical detection in air, proving their high sensitivity and rapid response. Kim et al. developed a fluorescent sensor array combined with machine learning to enable portable, low-cost, and multiplex detection of VOCs (Fig. 5C).139 Environment-sensitive dyes printed on a substrate generate unique fluorescence “fingerprints” upon analyte exposure, captured by a smartphone or portable imager. Convolutional neural networks (CNNs) analyze the patterns, accurately identifying VOCs such as toluene, acetone, and ethanol within ≤5 minutes, demonstrating a smart, field-ready platform for air quality monitoring.
Zhang et al. developed a wearable fluorescent POC sensor based on a newly synthesized hydroxynaphthalene benzophenone Schiff base (HNPBS).140 The fluorophore exhibits strong and selective emission responses to airborne VOCs such as acetone, acetonitrile, ethanol, and formaldehyde. Integrated into a flexible polymeric film, it functions as a low-cost, portable platform for real-time on-body detection. The sensor operates via fluorescence shifts from blue to green upon VOC exposure, arising from specific interactions between analytes and amine groups in the matrix. It provides distinct optical signatures for various VOCs, fast response (<30 s), good reversibility, and high selectivity under ambient conditions, making it highly suitable for personal exposure and indoor air quality monitoring within decentralized POC diagnostics.
Fluorescence based POC technologies have shown exceptional effectiveness for food safety monitoring, enabling the sensitive detection of pathogens, toxins, heavy metals, and antibiotic residues even at trace levels.15,141,142 Cheng et al. developed a dual-target lateral flow immunoassay (LFIA) enhanced with peroxidase-mimicking Pd@Pt core–shell nanozymes for the simultaneous detection of Salmonella Enteritidis and Escherichia coli O157:H7 in food samples.143 Acting as catalytic antibody labels, the nanozymes replaced conventional enzymes such as horseradish peroxidase and generated a strong colorimetric signal upon reaction with TMB/H2O2. This amplification strategy enabled ultrasensitive detection limits of 20 CFU mL−1 for Salmonella and 34 CFU mL−1 for E. coli. The portable assay was fully compatible with a 3D-printed smartphone-based reader, demonstrating high recovery and accuracy in real food matrices.
Further expanding the application scope to toxic metal detection, Shan et al. developed a portable fluorescence biosensor for selective and sensitive detection of Hg2+ ions in food, based on thymine–Hg2+–thymine (T–Hg2+–T) coordination chemistry.144 The sensor uses a fluorescent DNA probe that changes intensity upon binding mercury, ensuring high selectivity over other metals. Integrated with a miniaturized smartphone-based fluorescence microscope, the system excites samples with LED light and captures emission via the phone camera for real-time quantification. With a low detection limit of 0.33 nM, the compact and user-friendly platform enables on-site mercury monitoring in food without lab equipment, ideal for resource-limited settings.
3.2. Fluorescent proteins
Fluorescent proteins (FPs) have revolutionized the field of molecular and cellular biology by providing researchers with a unique and powerful colour palette to visualize and monitor a wide range of biological processes with unprecedented precision. The first FP, green fluorescent protein or GFP, was discovered in 1962 by the Japanese biochemist Osamu Shimomura, from the bioluminescent jellyfish Aequorea Victoria.145 Thanks to the discovery of such a powerful tool, he was awarded with the Nobel prize in chemistry in 2008, along with Roger Y. Tsien and Martin Chalfie. The wild type GFP is a 238 amino acid long protein, with a molecular weight of 27 kDa. It displays a β-barrel tertiary structure composed of 11 β-strands and 2 small α-alpha helices, one on the top of the barrel and one on the bottom. The chromophore portion of the protein is located between these α-helix segments. The chromophore of the GFPs derives from the subsequent cyclization and oxidation of a tripeptide (Ser65, Tyr66 and Gly67) that leads to the formation of an imidazoline moiety with conjugated double bonds. A similar structure is also present in the red fluorescent protein or DsRed, first isolated from the red coral of the Discosoma genus.145 | ||
| Fig. 6 (A). YC-Nano Ca2+ indicators. Top-left: peptide linker design connecting CaM and M13; YC3.60 and Nano140 contain CaM(E104Q), M13 binds Ca2+-loaded CaM, ECFPΔC11 is truncated CFP, cp173Venus is circularly permuted Venus. Top-right: Ca2+ titration curves (ΔR/R0vs. [Ca2+]; n = 3). Bottom: Confocal images of living zebrafish embryos injected with YC-Nano50 (left) or YC3.60 (right) showing spontaneous motor activity (yellow fluorescence), with corresponding Y/C ratio images; segmented musculature (SM, FM) lies between the spinal cord and epidermis. Scale bars, 50 µm. Reproduced with permission151 copyright 2010 Nature Publishing Group. (B) Nanoparticle (NP) sensor mechanisms. Schematic of protein interactions with quenched GFP–NP complexes versus protein aggregation, yielding fluorescence enhancement or additional quenching. NP1: cationic; NP2/NP4: hydrophobic; NP3: hydrogen bonding; NP5: aromatic π–π recognition. Reproduced with permission153 copyright 2009 Nature Publishing Group. (C) Engineering small GFPs. Left: Directed evolution strategy of flavin-binding miniGFPs in E. coli using FACS. Center: absorbance, excitation/emission spectra, and biochemical profiling relative to phiLOV3, CreiLOV, and BR1. Right: Representative fluorescence images of CHO cells expressing miniGFPs in the green/red channels. Scale bar, 20 µm. Reproduced with permission154 copyright 2022 Frontiers Media S.A. | ||
3.3. Fluorescent nanomaterials
Besides QDs or UCNPs, fluorescence POC technologies can be based on other nanomaterials, such as metal nanoparticles (MNPs),155 metal–organic frameworks (MOFs),156 silica nanoparticles (SiNPs), or carbon-based nanomaterials (Fig. 7).157 These systems combine structural tunability, high surface-to-volume ratios, and unique optical behaviours, properties that make them adaptable for sensing and diagnostic applications. Unlike molecular dyes or proteins, particle-based probes offer enhanced photostability, opportunities for surface functionalization, and strong signal amplification mechanisms that improve sensitivity in POC biosensors.158 Their fluorescence properties can originate from intrinsic photoluminescence, dye encapsulation, plasmonic effects, or hybrid organic–inorganic interactions, providing a wide range of detection strategies. In the next section, we will show an overview regarding these nanomaterials.3.3.1.1. Metal nanoparticles (MNPs). MNPs, particularly gold (AuNPs) and silver nanoparticles (AgNPs), are among the most widely explored fluorescent platforms for POC applications due to their unique plasmonic properties.159 These particles exhibit strong localized surface plasmon resonance (LSPR), which generates intense electromagnetic fields in their immediate vicinity. When placed near fluorescent molecules, these fields can significantly enhance excitation and emission rates, a phenomenon known as metal-enhanced fluorescence (MEF). The enhancement depends critically on particle parameters such as size, shape, composition, and architecture, including core–shell structures, alloys, or surface-decorated designs, allowing precise tuning of optical responses.
The interaction between metallic NPs and fluorophores creates hybrid systems, where the emission of photons from the fluorophore can excite plasmonic oscillations in the metal, producing new radiative decay pathways.160 In this configuration, both excitation and radiative decay rates increase, leading to stronger emission signals. As a result, fluorescence detection achieves higher sensitivity, allowing the observation of weakly emitting species that would remain undetectable in other cases. Moreover, photostability is improved because the reduced fluorescence lifetime decreases the time of probes in the excited state. In addition, MNPs can also act as scattering centers or energy donors, enabling sensitive detection even at the single-molecule level.161
In POC diagnostics, MNPs have been widely incorporated into lateral flow assays, microfluidic devices, and chip-based sensors.162 Their strong plasmonic properties not only provide rapid and ultrasensitive detection of biomolecules such as proteins, nucleic acids, and pathogens, but also enable multiplexed readouts, improved signal-to-noise ratios, and lower detection limits compared to conventional fluorescence-based assays. Furthermore, their tunable optical responses, ease of surface functionalization, and compatibility with portable detection systems make them highly attractive for the development of cost-effective, miniaturized, and field-deployable diagnostic platforms.163
Beyond biosensing, MNPs are also applied in forensics, food safety, drug testing, and environmental monitoring, highlighting their versatility as fluorescent nanoprobes. The combination of tunable optical properties, biocompatibility, and facile surface functionalization makes MNPs indispensable tools in the development of next-generation POC technologies.164
3.3.1.2. Metal–organic frameworks (MOFs). MOFs are a distinctive class of crystalline hybrid nanomaterials formed by the coordination of metal ions or clusters with multidentate organic ligands, giving rise to highly ordered, porous architectures.165,166 Their modularity allows enormous structural and chemical diversity, enabling precise control over pore size, surface area, and chemical functionality. Importantly, many MOFs exhibit intrinsic photoluminescence due to their organic linkers or metal centers, while others can be engineered as hybrid fluorescent platforms by incorporating dyes, QDs or rare-earth elements into their frameworks.156 This tunability provides MOFs with a high degree of flexibility for optical sensing and POC applications.
The luminescence mechanisms in MOFs may arise from several processes, including ligand-centered emission, metal–ligand charge transfer, or guest–host interactions within their porous cavities.167 These properties can be further modulated by tailoring the organic linker chemistry or by selecting specific metals (e.g., Zn, Zr, and lanthanides) to introduce new electronic transitions. Moreover, the porous structure of MOFs not only improves their fluorescence stability by shielding encapsulated fluorophores from quenching, but also provides selective recognition sites for analytes, creating turn-on, turn-off, or ratiometric fluorescent responses.165
In the POC landscape, fluorescent MOFs are gaining attention for biomarker detection, environmental monitoring, and multiplexed sensing, as they can combine signal generation with selective analyte capture.168 Thanks to their porous structure and tunable optical properties, MOFs can combine signal generation with selective analyte capture. Their large surface area allows efficient loading of biomolecules or fluorescent reporters, while their modular composition makes it possible to adjust emission characteristics and recognition sites. These features contribute to improving sensitivity and specificity in complex samples. In addition, the ability to design MOFs with tailored luminescence responses supports their integration into portable and multiplexed POC devices.169
3.3.1.3. Silica nanoparticles (SiNPs). SiNPs represent one of the most established classes of nanomaterials in fluorescence-based sensing due to their particular features, such as their tunable structures, optical transparency, high biocompatibility and ease of surface functionalization.170,171 Their popularity arises from their role as carriers that can encapsulate fluorescent dyes or QDs within a protective silica matrix, shielding them from photobleaching, solvent effects, and environmental degradation. This strategy not only enhances the photostability and brightness of the fluorophores but also ensures reproducible emission over long-term measurements, which is crucial for POC applications.172
What makes SiNPs fascinating from a nanotechnology point of view is their well-defined and tunable structures (i.e. size, morphology, and porosity) and surface chemistry. By introducing new functional groups via well-established siloxane chemistry, the silica surface can be modified to impart new properties to the particles, such as diagnostic and therapeutic capabilities. SiNPs are effectively ‘‘transparent’’ in the sense that they do not absorb light in the NIR, visible, and ultraviolet regions or interfere with magnetic fields. In addition, SiNPs are economic, easy to prepare, relatively chemically inert, biocompatible, and water dispersible.
Fluorescent SiNPs are generally prepared either by dye-doping during synthesis (e.g., via sol–gel or Stöber methods) or by surface conjugation with organic fluorophores or biomolecules. Both approaches allow fine control over size, porosity, and surface charge, making SiNPs adaptable to a wide variety of detection strategies.173–175 In addition, their surfaces can be easily modified with silane chemistry, polymers, or biomolecular ligands, enabling specific recognition of nucleic acids, proteins, or pathogens. The use of mesoporous SiNPs (MSNs) further expands their potential, as the porous architecture offers high loading capacity for dyes, drugs, or enzymes, while also facilitating efficient diffusion of analytes.
In the POC setting, dye-doped SiNPs have been successfully integrated into lateral flow assays,176 microfluidic devices,177 and multiplex immunoassays,178 where they serve as bright, stable, and cost-effective probes.179 Their strong fluorescence signals improve sensitivity compared to conventional organic dyes, while their robustness ensures compatibility with portable devices and long-term storage. Moreover, SiNP-based probes are being explored for ratiometric sensing,180–182 real-time monitoring, and combined therapeutic–diagnostic (theranostic) applications, underscoring their versatility.183
3.3.1.4. Quantum dots (QDs). Quantum dots are nanosized semiconductor particles with exceptional optical and electronic properties that arise from quantum confinement effects, which occur when charge carriers are confined within dimensions smaller than the exciton Bohr radius.184,185 This confinement produces discrete energy levels, allowing precise control of the absorption and emission spectra based on particle size and composition.
One of the most distinctive features of QDs is their size-tunable fluorescence: smaller QDs emit light at shorter wavelengths (blue region), while larger ones emit at longer wavelengths (red region).185,186 This tunability, combined with their broad absorption bands and narrow, symmetric emission peaks, enables simultaneous excitation of multiple QDs and highly multiplexed detection in fluorescence-based assays. Additionally, QDs exhibit exceptional photostability, high quantum yield, and resistance to photobleaching, outperforming conventional organic dyes and many other fluorophores.
From a materials standpoint, QDs are generally composed of group II–VI semiconductors (e.g., ZnS, CdS, CdSe, CdTe, and ZnO), group III–V compounds (e.g., InP, GaN, and GaAs), or group IV–VI materials (e.g., Si, Ge, and carbon-based QDs).186,187 The selection of the semiconductor core determines the emission range, while core–shell architectures are often employed to improve quantum yield, chemical stability, and biocompatibility. In these systems, a semiconductor core is encapsulated by another material with a wider band gap (e.g., CdSe/ZnS or InP/ZnS), which passivates surface defects and minimizes non-radiative recombination.
QDs can be synthesized using top-down methods (such as lithography or etching) or bottom-up techniques, including wet-chemical and vapor-phase routes.188 Surface modification plays a crucial role in determining colloidal stability, solubility, and biological compatibility. Functionalization with polymers, ligands, peptides, or antibodies allows target-specific recognition and facilitates integration into biosensors or microfluidic systems.
In the context of POC diagnostics, QDs serve as robust and bright fluorescent probes,189 enabling sensitive detection of nucleic acids, proteins, and pathogens.184 Their narrow emission bandwidths make them ideal for multiplexed assays, while their photostability ensures reliable signal retention over prolonged analyses. Despite these advantages, challenges such as potential cytotoxicity (particularly for cadmium-based QDs) and fluorescence intermittence (blinking) remain areas of ongoing research.
3.3.1.5. Carbon-based particles. Carbon-based NPs, including CDs, graphene QDs (GQDs), and nanodiamonds, have emerged as a versatile class of fluorescent nanomaterials, attracting attention due to their low toxicity, biocompatibility, aqueous solubility, and sustainable synthesis.190 Unlike traditional semiconductor QDs, which often contain toxic heavy metals, carbon-based nanostructures offer an environmentally friendly alternative without compromising photoluminescent performance.191 Their fluorescence typically arises from a combination of quantum confinement effects, surface defect states, and functional groups, which can be modulated by heteroatom doping (e.g., N, S, and P) or by chemical surface modifications.192
Among them, CDs are the most studied, typically synthesized through simple and scalable methods such as hydrothermal carbonization, microwave-assisted reactions, or pyrolysis of organic precursors, including biomass.193 CDs exhibit excitation-dependent emission, high photostability, and tunable fluorescence, making them promising candidates for multiplexed detection strategies.194 Similarly, GQDs combine the optical properties of QDs with the electronic and mechanical advantages of graphene, while nanodiamonds offer stable fluorescence from nitrogen-vacancy centers, suitable for bioimaging and sensing under demanding conditions.195
In the POC diagnostic landscape, carbon-based NPs have been integrated into fluorescence biosensors, lateral flow assays, and microfluidic devices for the detection of nucleic acids, proteins, and microbial pathogens.196,197 Their strong quenching ability has also been exploited in FRET–based assays, where target binding induces measurable changes in emission. Furthermore, their intrinsic biocompatibility allows safe use in intracellular imaging, drug delivery tracking, and metabolite monitoring, expanding their role beyond conventional biosensing. Taken together, carbon-based nanomaterials combine eco-friendliness, low cost, and tunable photophysics, positioning them as sustainable alternatives for next-generation POC fluorescence technologies.
3.3.1.6. Upconverting nanoparticles (UCNPs). UCNPs or upconverting nanoparticles have emerged as a promising class of nanomaterials, gaining significant interest in various scientific disciplines due to their unique ability to convert low-energy excitation into high-energy emission.198,199 UCNPs exhibit anti-Stokes luminescence, wherein two or more low-energy photons are sequentially absorbed, leading to the emission of a single higher-energy photon. This property makes them particularly attractive for fluorescence-based applications, as they exhibit minimal background autofluorescence, high photostability and deeper penetration in living tissues, which could make them ideal for real-time in vivo imaging.
UCNPs are typically composed of inorganic NPs based on NaYF4, doped with lanthanide cations in a core–shell manner or in a host–matrix system.198 Lanthanide cations (Ln3+) are essential for the upconversion phenomenon and the most widely used are Ytterbium (Yb3+), Erbium (Er3+) and Thulium (Tm3+).200 The photophysical properties of UCNPs are strictly connected to their size, shape and surface functionalization; these properties are related to the synthetic methods.
The synthesis of lanthanide-doped UCNPs can be performed with different strategies; usually, thermal decomposition and hydro(solvo)thermal synthesis allow monodisperse and regular nanoparticles to be obtained.201 However, they require high temperature and pressure, and the use of highly hydrophobic solvents (e.g., oleate), producing UCNPs with hydrophobic surfaces that can be dispersed only in nonpolar solvents. Microwave-assisted synthesis offers milder reaction conditions and can be performed in the liquid or solid phase. The easiest way to synthesize UCNPs is coprecipitation, using EDTA as a capping agent, although this method typically yields particles with a broad size distribution, irregular morphology, and low quantum yield.
Despite being considered highly promising for bioimaging, with several advantages compared to QDs, UCNP technology still requires substantial improvement. Their quantum yield remains significantly lower than that of organic fluorophores and QDs, and they typically require excitation with a 980 nm laser and specialized, non-commercial instrumentation. Furthermore, their composition, often involving lanthanides, rare-earth elements, and transition metals, raises concerns about potential toxicity, limiting their suitability for diagnostic applications.
In viral diagnostics, Xu et al. introduced fluorescent carbon dot–based silica (FCS) lateral flow strips for detecting the dengue virus NS1 antigen (Fig. 8A).202 These test strips, read under UV light, demonstrated a detection limit 100 times lower than conventional gold nanoparticle (AuNP)-based strips, with excellent reproducibility and stability. Similarly, Guo et al. developed a fluorescent lateral flow immunoassay LFIA platform using UCNPs encapsulated in mesoporous silica (UCNPs@mSiO2) to simultaneously detect the spike (S) and nucleocapsid (N) proteins of SARS-CoV-2.203 Coupled with a 5G-enabled fluorescent reader, the system achieved detection limits of 1.6 ng mL−1 for the S protein and 2.2 ng mL−1 for the N protein.
 | ||
| Fig. 8 (A). Schematic illustration of the synthetic procedures of CD embedded SiO2 spheres and the TEM image of dendritic SiO2 and HRTEM images of FCS spheres. Schematic representation of Zika NS1 protein detection using the iFCS-based lateral flow immunoassay. Reproduced with permission202 copyright 2021 Royal Society of Chemistry. (B) Schematic illustration of the preparation of RNs@Au and corresponding characterization: TEM image of RNs@Au (blue and red arrows indicate QD and Au, respectively) and UV-vis absorption spectra of Au, RNs, and RNs@Au. Below: schematic illustration of EBOV detection using an enhanced RNs@Au-based test strip: the target was labeled with Ab–RNs@Au–SA and captured by the test line of antibody to glycoprotein via antigen–antibody reaction. In the second step, biotin–RNs@ Au solution was applied to the sample pad. Biotin–RNs@Au were adhered to the test line by Ab–RNs@Au–SA via biotin–SA affinity, and then a network of RNs@Au can form on the test line through multivalent interactions of biotin–SA, in which a darker and brighter signal was obtained. Reproduced with permission204 copyright 2017 American Chemical Society. (C) Experimental process for the preparation of ZrMOF@CdTe–Ab conjugates and HTEM-mapping image. Corresponding schematic representation of the LFA for cTnT and h-FABP detection. Reproduced with permission208 copyright 2021 Royal Society of Chemistry. | ||
Beyond single-mode sensing, multimodal POC strategies have demonstrated superior performance by combining complementary signal outputs (colorimetric, fluorescent, and magnetic) to improve sensitivity, robustness, and resistance to interference. For example, Hu et al. reported a multimodal LFIA integrating colorimetric, fluorescent, and magnetic NPs, resulting in improved assay performance (Fig. 8B).204 They demonstrated a dual-signal LFIA for Ebola virus detection using RNs@Au (QDs + gold) nanospheres, achieving rapid and sensitive analysis of spiked samples within 20 minutes, detectable both visually and via fluorescence.
Fluorescent nanoparticle-based POC systems have also shown promise for cancer diagnostics. Liang et al. used aptamer-conjugated SiNPs coated with QDs on graphene oxide surfaces.205 Fluorescence is quenched by FRET but restored in the presence of target cancer cells. By changing aptamers and quantum dot colours, different cancer cells can be detected simultaneously with one light source. Likewise, Jimenez et al. developed a biosensor using gold-coated superparamagnetic nanomaghemite (AuMNPs) functionalized with DNA probes targeting the HPV-16 E6 oncogene.206 Detection relies on binding viral DNA and signalling via peptide-modified QDs. Tested for specificity against other viruses, it was successfully applied to patient samples and confirmed by PCR. This system offers a low-cost, sensitive alternative to traditional PCR for HPV detection, offering a low-cost alternative for HPV-related cancer diagnostics.
In the field of cardiovascular diseases (CVDs), Y. Fan Shi et al. developed a magnetic multilayer lanthanide MOF-based sensor (MagMOF) designed for the simultaneous detection of acute myocardial infarction (AMI) biomarkers such as CK-MB, troponin I (cTnI), and myoglobin (Mb), along with aspirin, a common treatment.207 Using a layer-by-layer assembly strategy, this hybrid nanosensor integrates antibodies and mRNA probes with MagMOF nanoballs, enabling highly sensitive detection within clinically relevant ranges. The platform offers rapid, stable, and multiplexed detection, showing strong potential for POC AMI diagnosis and therapeutic monitoring. Similarly, Jian Zou et al. studied a ZrMOF-based lateral flow assay using fluorescent ZrMOF@CdTe nanoparticles that enables rapid (8 min) and multiplexed detection of AMI biomarkers like h-FABP and cTnT (Fig. 8C).208 With a visual detection limit of 1 µg L−1 for h-FABP, it is simple, low-cost, and effective for POC use in serum samples.
Fluorescent POC sensing has also advanced in continuous glucose monitoring (CGM). To address limitations in traditional electrochemical CGMs, including instability, interference, and electrode degradation, a nanodiamond-boronic hydrogel system has been developed for integration into porous microneedles.209 These highly photostable and biocompatible nanodiamonds emit fluorescence in response to glucose levels. The hydrogel-based sensor offers long-term signal stability in vivo, as shown in animal models, and avoids common issues of electrochemical sensors. This proof-of-concept marks a promising step toward reliable, minimally invasive CGM using fluorescence technology.
Magnetic and fluorescent nanocomposites have further expanded the capabilities of optical POC systems. Tombelli et al. reported a compact fluorescence-based immunoassay device using disposable plastic chips for multi-analyte detection of immunosuppressants.210 The chip integrates ten parallel microchannels, on-chip microfluidics, and photodiodes for direct fluorescence readout. Submicron fluorescent magnetic particles accelerate the reaction kinetics through magnetic concentration while enhancing sensitivity through optical labelling. Reusability and integration with microdialysis sampling allowed continuous therapeutic monitoring for up to 48 hours, providing valuable pharmacokinetic insights.
Combined with MoS2 and Eu3+, QDs were employed to develop a ratiometric fluorescent probe for sensitive tetracycline (TC) detection in biological samples. The method relies on energy transfer between MoS2 QDs and Eu–TC complexes, enhancing luminescence at 620 nm while quenching at 470 nm, enabling ratiometric detection (F620/F470). It offers a 10 nM–60 µM linear range, 2 nM detection limit, high recovery (94.4–108.4%), and precision (<5.36% RSD). A smartphone-based platform enables visual, portable quantification with a 0.05 µM detection limit, providing a fast, low-cost solution for antibiotic monitoring.211
Beyond biomedical applications, nanoparticle-based POC platforms are also showing remarkable potential in environmental monitoring. Liang et al. developed a multicolor fluorescent probe for visual and smartphone-assisted detection of common water additives.212 The ratiometric system combines blue-emitting CDs and red-emitting gold NCs, producing visible color changes under UV light upon exposure to analytes such as ascorbic acid, sodium sulfite, and hydrogen peroxide. The probe exhibited high selectivity, rapid response (<5 min), and excellent performance in real water samples, demonstrating the versatility of fluorescent POC technologies for on-site environmental assessment.
Jin et al. developed a multiplex paper-based aptasensor for POC water quality monitoring, integrating UCNPs and specific aptamers for the simultaneous detection of multiple contaminants, including pathogens (Salmonella), toxins (ochratoxin A, microcystin-LR), and heavy metals (Hg2+, Pb2+).213 The system achieves high sensitivity, with detection limits as low as 115 CFU mL−1 for Salmonella, and 4 nM are read using a compact, smartphone-based device, enabling portable, rapid (≤30 min), and instrument-free analysis. The platform showed high specificity and minimal cross-reactivity, and has been successfully tested in real water samples (tap and pond water), making it a promising tool for decentralized and field-based water quality assessment.
3.4. Comparative analysis
Table 1 summarizes the principal characteristics of the fluorescent probe classes discussed in Sections 3.1–3.3, providing a comparative overview of performance-related parameters relevant to fluorescence-based POC applications, including sensitivity, photostability, biocompatibility, and practical advantages and limitations. Across the different probe categories, engineered nanomaterials generally exhibit higher signal robustness and photostability than organic fluorophores and fluorescent proteins, which can be advantageous for applications requiring high sensitivity or prolonged signal acquisition. However, superior optical performance does not necessarily correlate with biocompatibility, regulatory acceptance, or translational suitability. For example, QDs represent the highest-performing class in terms of optical properties, achieving detection limits down to the aM range while maintaining exceptional photostability and narrow, size-tunable emission spectra. These features enable highly multiplexed detection and prolonged signal acquisition, making QDs attractive for ultra-trace and multi-analyte analysis. Nevertheless, their heavy-metal composition raises cytotoxicity and regulatory concerns that currently limit widespread clinical translation, highlighting the gap that can exist between analytical performance and practical implementability.222| Probe type | Sensitivity | Photostability | Biocompatibility | Key advantages | Limitations | Ref. |
|---|---|---|---|---|---|---|
| Organic fluorophores | Moderate (nM) | Moderate to low | Good | Small size; well-known photophysics | Photobleaching; narrow Stokes shift | 81 and 214–216 |
| Fluorescent proteins | Moderate (nM) | Limited | Excellent | Genetic encoding; intracellular targeting | Photobleaching; narrow spectral range | 48, 217 and 218 |
| MNPs | High (fM) | Excellent | Variable | Plasmonic fluorescence enhancement | Potential toxicity; less explored | 162, 164 and 219 |
| MOFs | Moderate–high (pM) | Good | Generally good | High loading capacity; multifunctionality | Stability issues in biological media | 156 and 220 |
| SiNPs | Moderate (nM) | Good | Good | Biocompatible; easy surface modification | Potential aggregation | 120, 170, 173 and 183 |
| QDs | Very high (aM) | Excellent | Moderate (toxicity concerns) | Bright, narrow tunable emission spectra | Heavy metal toxicity; regulatory issues | 191 and 192 |
| Carbon-based NPs | Moderate–high (pM) | Very good | Excellent | Low toxicity; versatile excitation/emission | Lower brightness than QDs | 157, 190 and 191 |
| UCNPs | Moderate–high (pM) | Excellent | Good | NIR excitation, low autofluorescence | Lower quantum yield; complex synthesis | 198, 199 and 221 |
MNPs offer an alternative route to high analytical sensitivity, frequently reaching fM detection levels through plasmon-enhanced fluorescence mechanisms. Their ability to amplify emission signals and improve signal-to-noise ratios supports their use in rapid immunoassays and lateral-flow-based POC platforms. At the same time, performance strongly depends on surface chemistry, particle architecture, and probe–particle spacing, which can introduce variability and reproducibility challenges when transitioning from laboratory prototypes to standardized diagnostic formats.162,164
MOFs, carbon-based NPs, and UCNPs represent platforms that seek to balance optical performance with biocompatibility and functional versatility. MOFs offer high loading capacity and multifunctionality, enabling integrated analyte recognition and signal transduction, although their stability in biological fluids remains a critical limitation for long-term operation in complex matrices.156,220 Carbon-based NPs, including carbon dots and graphene derivatives, address several limitations associated with metal-containing probes by providing enhanced biocompatibility and chemical stability while maintaining sufficient sensitivity for most clinically relevant POC assays, despite lower brightness than QDs.157,190 UCNPs introduce a distinct optical advantage by enabling near-infrared excitation, which reduces background autofluorescence and improves tissue penetration. However, their relatively low quantum yield and the requirement for specialized excitation sources complicate integration into low-cost, miniaturized diagnostic platforms.199
Organic fluorophores and fluorescent proteins remain highly relevant for applications prioritizing simplicity, genetic encoding capability, or intracellular monitoring.223 Organic fluorophores benefit from well-characterized photophysics, straightforward functionalization, and compatibility with disposable, low-cost devices,81,214–216 but their susceptibility to photobleaching and limited sensitivity constrain their use in long-term or ultra-low-concentration detection. Fluorescent proteins provide unmatched specificity for intracellular sensing and genetically encoded biosensing strategies, yet their limited photostability and dependence on biological expression systems restrict their applicability in cell-free or disposable POC devices.48,217,218
Beyond analytical metrics, practical considerations play a decisive role in probe selection. Highly sensitive nanoparticle-based POC systems often require complex synthesis protocols, surface functionalization, and advanced instrumentation, whereas molecular probes are generally easier to integrate into portable and cost-effective devices. Overall, the discussion in Sections 3.1–3.3 and the comparison in Table 1 underscore that the selection of fluorescent probes for POC technologies is inherently application-dependent and requires a careful balance between optical performance, biocompatibility, stability, and implementation constraints.
4. Challenges and future perspectives
Fluorescence-based POC devices enable rapid, sensitive, and portable analyses across diverse application domains, including diagnostics, therapeutic monitoring, environmental analysis, and wearable systems (Fig. 9). However, despite these advantages, several challenges must still be addressed to unlock their full translational potential. Critical issues include probe and signal stability, reduced sensitivity in complex matrices, limited standardization, and the need for more robust, user-friendly readout systems. These aspects are discussed in detail below.4.1. Limitations and challenges of fluorescence-based POC technologies
Despite recent progress, several limitations hinder the widespread adoption and long-term performance of fluorescence-based POC systems.A key limitation of fluorescent probes is their susceptibility to environmental conditions, as factors such as temperature, pH, ionic strength, and solvent polarity can significantly affect both quantum yield and emission spectra. These factors cause instability and impair measurement reproducibility, especially in uncontrolled field circumstances, such as complex media (human serum, whole blood, urine, saliva, etc.).224 Thus, while fluorescence-enhancing systems have been widely explored to improve biosensor sensitivity, their translation to clinical practice remains limited, with few systems tested in biologically complex fluids or validated in real clinical conditions.225,226 Moreover, the same labelling processes frequently require laborious procedures for attaching fluorescent tags to biomolecules like proteins. This step is mandatory for the detection of analytes lacking intrinsic fluorescence; however, its performance may be adversely affected by interfering substances within the sample matrix, potentially compromising signal reliability and analytical accuracy. To minimize procedural steps and their associated challenges, several studies have focused on integrating microfluidic systems capable of performing sequential operations, including sample separation or filtration, reagent mixing, target labelling, and washing, to streamline sample preparation and improve assay efficiency.227–229
Photobleaching remains another major limitation, resulting in progressive signal decay during repeated or prolonged excitation. Reactive oxygen species or radical intermediates commonly contribute to photodegradation, limiting time-lapse detection and reducing suitability for long-term monitoring. Furthermore, matrix-related effects such as quenching by biomolecules (e.g., proteins, lipids, and nucleic acids) and high background autofluorescence further complicate measurements. These interferences lower signal-to-noise ratios and frequently demand complex sample pretreatment or filtration processes.
Beyond technical constraints, standardization of measurement protocols, validation procedures, and quality assurance measures remains critical for widespread adoption of these technologies. The development of reference standards and inter-laboratory comparison programs will support regulatory acceptance and commercial deployment. The integration of fluorescence-based sensors with emerging technologies, including artificial intelligence, blockchain for data integrity, and Internet of Things (IoT) platforms, promises to enhance the capabilities and applications of these monitoring systems.230,231 Advanced data analytics and machine learning algorithms will improve the interpretation of complex fluorescence signatures and enable predictive monitoring capabilities.
Finally, regulatory validation and clinical translation of fluorescence-based POC devices require rigorous performance benchmarking, repeatability investigations, and safety assessments. These steps are extremely time-consuming and expensive, especially when applied to multiplexed or nanomaterial-based platforms, impeding their road to market and limiting acceptance in resource-constrained environments.
Overcoming these constraints will necessitate collaborative work in materials science, optical engineering, microfluidics, and artificial intelligence to create next-generation POC systems that are robust, scalable, and comply with international diagnostic standards.
The development and validation of emerging microfluidic and POC fluorescence platforms critically depend on established fluorescence-based reference techniques. Flow cytometry, plate-reader fluorescence assays, and conventional fluorescence microscopy serve as gold standards to benchmark sensitivity, specificity, dynamic range, and reproducibility.232
Flow cytometry offers high-throughput multiparametric analysis using well-established fluorescence signals, making it a reliable reference technique for validating novel microfluidic devices. Relying on such established methodologies helps ensure that emerging platforms meet rigorous analytical standards and supports their translational development. Recent studies have shown that high-throughput fluorescence lifetime imaging flow cytometry can acquire detailed lifetime and intensity images at rates that overcome conventional speed limitations, improving the discrimination of cellular subpopulations in dynamic biological settings.233 In parallel, work on the immunomagnetic separation of B-type acute lymphoblastic leukemia blast cells from bone marrow has used flow cytometry to validate capture efficiency and benchmark microfluidic chip performance, highlighting how combining targeted separation strategies with established cytometric readouts enables quantitative evaluation of device performance.234
From this perspective, flow cytometry serves as an effective bridge between traditional fluorescence-based methods and emerging point-of-care technologies.
4.2. Emerging trends and novel POC technologies
Fluorescence-based POC technologies are rapidly evolving through the development of novel materials, advanced detection strategies, and improved system integration.235,236One of the main drivers of innovation is the emergence of next-generation fluorescent probes with improved brightness, photostability, and selectivity compared to traditional organic dyes. QDs with their size-tunable emission and exceptional photostability, allow highly sensitive and multiplexed biomarker detection with negligible spectral overlap. Their broad absorption and narrow emission profiles make them ideal for high-content, multi-analyte assays.
In parallel, AIE fluorophores have emerged as a complementary class of probes. Unlike conventional dyes that suffer from fluorescence quenching upon aggregation, AIE molecules become highly emissive upon aggregation, which significantly reduces background noise and enhances the signal-to-noise ratio. This property is particularly beneficial for detecting low-abundance targets in complex biological samples.237
Another key technological advancement is the incorporation of smartphone-based fluorescence readers, leveraging the widespread availability of mobile devices to create portable, cost-effective, and user-friendly diagnostic platforms.16,237 These systems often combine compact optical components with sophisticated image processing algorithms, facilitating real-time quantification and data sharing, which is critical for remote healthcare and telemedicine applications.
Moreover, microfluidics and LoC technologies are increasingly integrated with fluorescent detection to automate sample handling and reduce assay times, enhancing the practicality of POC testing in diverse settings.238
The use of AI and machine learning algorithms to analyse fluorescence signals is also emerging as a powerful tool to improve diagnostic accuracy and interpret complex data patterns.239 AI algorithms process diverse fluorescence data types including intensity-based signals, spectral profiles, fluorescence lifetime measurements, and imaging datasets.240
Key AI roles encompass feature extraction, noise reduction, classification, regression, and multiplexed target discrimination.241 For example, convolutional neural networks (CNNs) excel in analyzing fluorescence microscopy images for cell identification and sorting, while machine learning classifiers enhance spectral unmixing for complex multiplex assays.242
These AI-driven approaches yield measurable gains such as improved sensitivity, increased robustness against background interference, reduced analysis time, and enhanced multiplexing capacity. As AI methods evolve, they are expected to further expand the capabilities and clinical utility of fluorescence-based POC diagnostics.
On the other hand, the transition from proof-of-concept prototypes to clinically deployable devices remains a considerable challenge.243 For example, lateral flow assays incorporating fluorescent NPs have reached commercial availability, offering improved sensitivity over traditional colorimetric tests.244 Conversely, novel microfluidic platforms integrating fluorescence lifetime measurements often remain at early developmental stages, requiring further validation in clinical matrices to confirm robustness.245
Technologies at more advanced stages often feature durable materials, optimized probe chemistries, and fully integrated microfluidic systems that have been validated under realistic sample conditions, demonstrating scalability and adherence to regulatory standards. In contrast, earlier-stage platforms mainly focus on proof-of-principle demonstrations or novel sensing modalities, lacking extensive validation in clinically relevant matrices.246 Recognizing these developmental distinctions is essential to prioritize research directions, inform investment strategies, and set realistic timelines for clinical adoption. Additionally, recent advances in AI-assisted fluorescence analysis have shown considerable promise for improving diagnostic accuracy, though they are still under evaluation for robustness and feasibility in real-world deployment.247 Clearly distinguishing between these stages is critical to accelerating translation and ensuring that fluorescence-based innovations fulfil the clinical and operational requirements of decentralized healthcare.
5. Conclusions
This review provides a comprehensive overview of fluorescence-based technologies and their evolution into advanced applications in POC diagnostic technologies. It analyses the fundamental principles and instrumentation of key fluorescence techniques, including spectroscopy, microscopy, time-resolved fluorescence, and FLIM, highlighting their increasing sensitivity, spatial and temporal resolution, and multiplexing capabilities. Moreover, this review presents a detailed analysis of the main classes of fluorescent probes, such as organic fluorophores, fluorescent proteins, QDs, and UCNPs, discussing their unique photophysical features and suitability for portable diagnostic formats. This review also explores the broad range of POC applications enabled by these systems, spanning clinical diagnostics, therapeutic drug monitoring, environmental analysis and wearable biosensors. Collectively, fluorescence-based POC devices stand out for their portability, rapid detection capability, analytical sensitivity and suitability for use in resource-limited environments.Looking ahead, the future of fluorescence-based technologies in POC diagnostics and monitoring appears highly promising. The integration of smart biosensors, artificial intelligence, and microfluidic architectures is transforming how biomarkers are detected and quantified, in real time and with high specificity. There is a clear trend toward more portable, customizable, and user-friendly devices that enable continuous, minimally invasive monitoring and seamless connectivity to digital healthcare infrastructures. Advances in miniaturization, alongside the development of more stable, biocompatible, and near-infrared-emitting probes, are expected to enhance their translational potential across clinical and environmental settings.
However, key challenges remain. Regulatory approval, large-scale clinical validation, and robust standardization are essential to ensure reliability and reproducibility. Overcoming these barriers will require sustained collaboration between materials scientists, optical engineers, clinicians, and policymakers. With continued interdisciplinary progress, fluorescence-based POC systems have the potential to redefine personalized and preventive healthcare, expanding diagnostic access and improving outcomes on a global scale.
Author contributions
Conceptualization and original outline: A. C.; conceptualization and manuscript restructuring: L. L. d. M., M. C.-M., E. S., and G. V.; data curation: H. I., F. C., S. F., V. O., G. G., and A. C.; funding acquisition: Gi. Gi., A. C., and L. L. d. M.; project administration: L. L. d. M.; resources: Gi. Gi., A. C., and L. L. d. M.; supervision: L. L. d. M.; visualization: M. C.-M. and L. L. d. M.; writing – original draft: M. C.-M., E. S., G. V., A. C., H. I., F. C., S. F., V. O., and G. G.; writing – review and editing: M. C.-M., E. S., G. V., A. C., F. C., and L. L. d. M.Conflicts of interest
There are no conflicts to declare.Data availability
No new data were generated or analyzed in this study. Data sharing is not applicable.Acknowledgements
This work was supported by the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program ERC Proof of Concept “HySENSE” (no. 101212914), the Associazione Italiana per la Ricerca contro il Cancro (AIRC) (MFAG-2019, no. 22902), by the Italian Ministry of University and Research, under the complementary actions to the NRRP ‘Fit4MedRob - Fit for Medical Robotics’ Grant (#PNC0000007), the PNRR-MCNT2-2023-12377885 (CUP MASTER: F53C23001360008), the PNRR-MR1-2022-12376725 (CUP B53C22009590001), and the PRIN 2022 (2022CRFNCP_PE11_PRIN2022). Support was also provided by the “Tecnopolo per la medicina di precision” (TecnoMed Puglia) - Regione Puglia: DGR n.2117 of 21/11/2018, (CUP: B84I18000540002) and the Italian Ministry of Health FSC 2014-2020, Code T4-AN-01 (CUP: F83C22001560003). Dr. Anil Chandra would like to acknowledge the support received from the Anusandhan National Research Foundation (ANRF), Government of India, under the Startup Research Grant (SRG) scheme, File No: SRG/2023/002081.References
- S.-H. Huang, Y.-H. Huang and L.-C. Chiu, Int. J. Syst. Sci.-Oper. Logist., 2024, 2374968 Search PubMed.
- B. Heidt, W. F. Siqueira, K. Eersels, H. Diliën, B. Van Grinsven, R. T. Fujiwara and T. J. Cleij, Biosensors, 2020 DOI:10.3390/BIOS10100133.
- K. Lakshmanan and B. M. Liu, Diagnostics, 2025, 123 CrossRef CAS PubMed.
- T. S. Pillay, A. I. Khan and S. Yenice, Clin. Chim. Acta, 2025, 120341 CrossRef CAS PubMed.
- Y. Liu, X. Zhao, M. Liao, G. Ke and X.-B. Zhang, Sens. Diagn., 2024, 3, 1789–1806 RSC.
- A. Teymur, I. Hussain, C. Tang, R. Saxena, D. Erickson and T. Wu, Micromachines, 2025, 16, 156 CrossRef PubMed.
- D. Geißler, L. J. Charbonnière, R. F. Ziessel, N. G. Butlin, H. Löhmannsröben and N. Hildebrandt, Angew. Chem., Int. Ed., 2010, 49, 1396–1401 CrossRef PubMed.
- R. B. Sekar and A. Periasamy, J. Cell Biol., 2003, 629–633 CrossRef CAS PubMed.
- J. Chu, A. Ejaz, K. M. Lin, M. R. Joseph, A. E. Coraor, D. A. Drummond and A. H. Squires, Nat. Nanotechnol., 2024, 19, 1150–1157 CrossRef CAS PubMed.
- J. Aich, S. Singh, S. Chandrashekhar, V. Srivastav, S. Devarajan and S. Basu, in Smart Diagnostics for Neurodegenerative Disorders, Elsevier, 2024, pp. 179–196 Search PubMed.
- F. Shalileh, N. Shamani, M. Golbashy, M. Dadmehr and M. Hosseini, Nanotechnology, 2025, 36, 052002 CrossRef CAS PubMed.
- Y. Xie, L. Dai and Y. Yang, Biosens. Bioelectron. X, 2022, 100109 CAS.
- H. Wang, Q. Li, P. Alam, H. Bai, V. Bhalla, M. R. Bryce, M. Cao, C. Chen, S. Chen, X. Chen, Y. Chen, Z. Chen, D. Dang, D. Ding, S. Ding, Y. Duo, M. Gao, W. He, X. He, X. Hong, Y. Hong, J. J. Hu, R. Hu, X. Huang, T. D. James, X. Jiang, G. I. Konishi, R. T. K. Kwok, J. W. Y. Lam, C. Li, H. Li, K. Li, N. Li, W. J. Li, Y. Li, X. J. Liang, Y. Liang, B. Liu, G. Liu, X. Liu, X. Lou, X. Y. Lou, L. Luo, P. R. McGonigal, Z. W. Mao, G. Niu, T. C. Owyong, A. Pucci, J. Qian, A. Qin, Z. Qiu, A. L. Rogach, B. Situ, K. Tanaka, Y. Tang, B. Wang, D. Wang, J. Wang, W. Wang, W. X. Wang, W. J. Wang, X. Wang, Y. F. Wang, S. Wu, Y. Wu, Y. Xiong, R. Xu, C. Yan, S. Yan, H. B. Yang, L. L. Yang, M. Yang, Y. W. Yang, J. Yoon, S. Q. Zang, J. Zhang, P. Zhang, T. Zhang, X. Zhang, X. Zhang, N. Zhao, Z. Zhao, J. Zheng, L. Zheng, Z. Zheng, M. Q. Zhu, W. H. Zhu, H. Zou and B. Z. Tang, Aggregation-Induced Emission (AIE), Life and Health, American Chemical Society, 2023 DOI:10.1021/acsnano.3c03925.
- W. Chen, H. Guan, Y. Lu, G. Zeng, D. Gu, K. Guo, C. Jiang and H. Liu, Aggregate, 2025, e70008 CrossRef CAS.
- D. Zhang and Q. Liu, Biosens. Bioelectron., 2016, 75, 273–284 CrossRef CAS PubMed.
- J. F. Bergua, R. Álvarez-Diduk, A. Idili, C. Parolo, M. Maymó, L. Hu and A. Merkoçi, Anal. Chem., 2022, 94, 1271–1285 CrossRef CAS PubMed.
- K. E. Sapsford, L. Berti and I. L. Medintz, in Wiley Encyclopedia of Chemical Biology, Wiley, 2008, pp. 1–23 Search PubMed.
- S. Deshayes and G. Divita, Prog. Mol. Biol. Transl. Sci, 2013, 113, 109–143 CAS.
- M. J. Sanderson, I. Smith, I. Parker and M. D. Bootman, Cold Spring Harb. Protoc., 2014, 2014, pdb.top071795 CrossRef PubMed.
- J. R. Lakowicz, Principles of fluorescence spectroscopy, Springer, 2006 Search PubMed.
- D.-E. Zacharioudaki, I. Fitilis and M. Kotti, Molecules, 2022, 27, 4801 CrossRef CAS PubMed.
- H. H. Telle and Á. G. Ureña, Laser Spectroscopy and Laser Imaging, CRC Press, 2018 Search PubMed.
- M. Y. Berezin and S. Achilefu, Chem. Rev., 2010, 110, 2641–2684 CrossRef CAS PubMed.
- D. J. S. Birch, Y. Chen and O. J. Rolinski, in Photonics, Wiley, 2015, pp. 1–58 Search PubMed.
- S. Nagl, M. Schaeferling and O. S. Wolfbeis, Microchim. Acta, 2005, 151, 1–21 CrossRef CAS.
- Y. Fu, T. Liu, H. Wang, Z. Wang, L. Hou, J. Jiang and T. Xu, J. Sci.: Adv. Mater. Devices, 2024, 9, 100694 CAS.
- S. Dolai, S. K. Bhunia, S. Rajendran, V. UshaVipinachandran, S. C. Ray and P. Kluson, Crit. Rev. Solid State Mater. Sci., 2021, 46, 349–370 CrossRef CAS.
- G. Feng, G.-Q. Zhang and D. Ding, Chem. Soc. Rev., 2020, 49, 8179–8234 RSC.
- H. Wang, J. Tang, X. Wan, X. Wang, Y. Zeng, X. Liu and D. Tang, Anal. Chem., 2024, 15503 CAS.
- D. P. O’Neal, M. A. Meledeo, J. R. Davis, B. L. Ibey, V. A. Gant, M. V. Pishko and G. L. Cote, IEEE Sens. J., 2004, 4, 728–734 CrossRef.
- A. Kumar, V. S. Goudar, B. K. Nahak, P. Tsai, H. Lin and F. Tseng, Small, 2024, 2307955 CrossRef CAS PubMed.
- H. Iuele, S. Forciniti, V. Onesto, F. Colella, A. C. Siciliano, A. Chandra, C. Nobile, G. Gigli and L. L. del Mercato, ACS Appl. Mater. Interfaces, 2024, 55071 Search PubMed.
- G. Grasso, S. Forciniti, V. Onesto, L. Pierantoni, D. Caballero, E. D’Amone, G. Gigli, R. L. Reis, J. M. Oliveira and L. L. del Mercato, Biosens. Bioelectron., 2025, 283, 117481 CrossRef CAS PubMed.
- G. Grasso, V. Onesto, S. Forciniti, E. D’Amone, F. Colella, L. Pierantoni, V. Famà, G. Gigli, R. L. Reis, J. M. Oliveira and L. L. del Mercato, Biodes. Manuf, 2024, 7, 292–306 CrossRef CAS.
- M. H. Gehlen, J. Photochem. Photobiol., C, 2020, 42, 100338 CrossRef CAS.
- S. E. Webber, Photochem. Photobiol., 1997, 65, 33–38 CrossRef CAS.
- B. I. Ipe, K. Yoosaf and K. G. Thomas, J. Am. Chem. Soc., 2006, 128, 1907–1913 CrossRef CAS PubMed.
- Q. Tan, R. Zhang, G. Zhang, X. Liu, F. Qu and L. Lu, Anal. Bioanal. Chem., 2020, 412, 1317–1324 CrossRef CAS PubMed.
- E. A. Specht, E. Braselmann and A. E. Palmer, Annu. Rev. Physiol., 2017, 79, 93–117 CrossRef CAS PubMed.
- M. H. Jennison and A. F. Morgan, Tubercle, 1950, 31, 84–87 CrossRef.
- S. Prasad, A. Chandra, M. Cavo, E. Parasido, S. Fricke, Y. Lee, E. D’Amone, G. Gigli, C. Albanese, O. Rodriguez and L. L. del Mercato, Nanotechnology, 2021, 32, 062001 CrossRef CAS PubMed.
- F. Alemanno, M. Cavo, D. Delle Cave, A. Fachechi, R. Rizzo, E. D’Amone, G. Gigli, E. Lonardo, A. Barra and L. L. del Mercato, Proc. Natl. Acad. Sci. U. S. A., 2023, e2122352120 CrossRef CAS.
- F. Carnevali, S. Forciniti, V. Onesto, A. C. Siciliano, H. Iuele, G. Grasso, A. F. da Cruz, I. Serra, N. Depalma, S. D’Ugo, P. Piscitelli, M. G. Spampinato, G. Gigli, C. Camargo de Oliveira and L. L. del Mercato, Adv. Ther., 2024, 2400351 CrossRef CAS.
- R. Kasprowicz, R. Suman and P. O’Toole, Int. J. Biochem. Cell Biol., 2017, 84, 89–95 CrossRef CAS PubMed.
- H. Yokota, Biochim. Biophys. Acta Gen. Subj., 1864, 2020, 129362 Search PubMed.
- A. L. Robson, P. C. Dastoor, J. Flynn, W. Palmer, A. Martin, D. W. Smith, A. Woldu and S. Hua, Front. Pharmacol., 2018, 9, 1–8 Search PubMed.
- S. J. Sahl, S. W. Hell and S. Jakobs, Nat. Rev. Mol. Cell Biol., 2017, 18, 685–701 CrossRef CAS PubMed.
- B. C. Campbell, M. G. Paez-Segala, L. L. Looger, G. A. Petsko and C. F. Liu, Nat. Methods, 2022, 19, 1612–1621 CrossRef CAS PubMed.
- S. Y. Kim, Y. Arai, T. Tani, H. Takatsuka, Y. Saito, T. Kawashima, S. Kawakami, A. Miyawaki and T. Nagai, Microscopy, 2017, 66, 110–119 CAS.
- N. V. dos Santos, C. F. Saponi, T. M. Ryan, F. L. Primo, T. L. Greaves and J. F. B. Pereira, Int. J. Biol. Macromol., 2020, 164, 3474–3484 CrossRef CAS PubMed.
- J. W. Lichtman and J. A. Conchello, Nat. Methods, 2005, 2, 910–919 CrossRef CAS PubMed.
- W. Justyna, Curr Protoc Cytom, 2017, 176, 139–148 Search PubMed.
- S. P. Poland, N. Krstajić, S. Coelho, D. Tyndall, R. J. Walker, V. Devauges, P. E. Morton, N. S. Nicholas, J. Richardson, D. D.-U. Li, K. Suhling, C. M. Wells, M. Parsons, R. K. Henderson and S. M. Ameer-Beg, Opt. Lett., 2014, 39, 6013 CrossRef CAS PubMed.
- V. I. Shcheslavskiy, M. V. Shirmanova, K. S. Yashin, A. C. Rück, M. C. Skala and W. Beker, J. Biophotonics, 2025, 18(12), e202400450 CrossRef CAS PubMed.
- R. Datta, T. M. Heaster, J. T. Sharick, A. A. Gillette and M. C. Skala, J. Biomed. Opt., 2020, 25, 1 Search PubMed.
- J. Bleeker, A. P. Kahn, L. M. Baumgartner, F. C. Grozema, D. A. Vermaas and W. F. Jager, ACS Sens., 2023, 8, 2050–2059 CrossRef CAS PubMed.
- K. Suhling, L. M. Hirvonen, W. Becker, S. Smietana, H. Netz, J. Milnes, T. Conneely, A. Le Marois, O. Jagutzki, F. Festy, Z. Petrášek and A. Beeby, Nucl. Instrum. Methods Phys. Res. A, 2019, 942, 162365 CrossRef CAS.
- W. R. Algar, N. Hildebrandt, S. S. Vogel and I. L. Medintz, Nat. Methods, 2019, 16, 815–829 CrossRef CAS PubMed.
- R. Datta, T. M. Heaster, J. T. Sharick, A. A. Gillette and M. C. Skala, J. Biomed. Opt., 2020, 25, 1 Search PubMed.
- T. Hinsdale, C. Olsovsky, J. J. Rico-Jimenez, K. C. Maitland, J. A. Jo and B. H. Malik, Biomed. Opt. Express, 2017, 8, 1455 CrossRef CAS PubMed.
- N. Oleksiievets, C. Mathew, J. C. Thiele, J. I. Gallea, O. Nevskyi, I. Gregor, A. Weber, R. Tsukanov and J. Enderlein, Nano Lett., 2022, 22, 6454–6461 CrossRef CAS PubMed.
- J. E. Phipps, J. Bec and L. Marcu, Multimodality Imaging, Springer Singapore, Singapore, 2020, pp. 153–171 Search PubMed.
- A. Alfonso-Garcia, J. Bec, S. Sridharan Weaver, B. Hartl, J. Unger, M. Bobinski, M. Lechpammer, F. Girgis, J. Boggan and L. Marcu, J. Biophotonics, 2020, 13, e201900108 CrossRef CAS PubMed.
- A. Alfonso-Garcia, J. Bec, B. Weyers, M. Marsden, X. Zhou, C. Li and L. Marcu, J. Biophotonics, 2021, 14, e202000472 CrossRef PubMed.
- A. P. Demchenko, Introduction to fluorescence sensing, 2009, 65–118 Search PubMed.
- R. Kang, L. Talamini, E. D’Este, B. M. Estevao, L. De Cola, W. Klopper and F. Biedermann, Chem. Sci., 2021, 12, 1392–1397 RSC.
- S. Yang, Y. Zhan, W. Shou, L. Chen, Z. Lin and L. Guo, ACS Appl. Bio Mater., 2021, 4, 6065–6072 CrossRef CAS PubMed.
- J. Zhang, S. Shikha, Q. Mei, J. Liu and Y. Zhang, Microchim. Acta, 2019, 186, 1–21 Search PubMed.
- X. Gao, H. Xu, M. Baloda, A. S. Gurung, L.-P. Xu, T. Wang, X. Zhang and G. Liu, Biosens. Bioelectron., 2014, 54, 578–584 CrossRef CAS PubMed.
- F. G. Prendergast and K. G. Mann, Biochemistry, 1978, 17, 3448–3453 CrossRef CAS.
- H. Chen, J. Yu, X. Men, J. Zhang, Z. Ding, Y. Jiang, C. Wu and D. T. Chiu, Angewandte Chemie, 2021, 133, 12114–12119 CrossRef.
- N. T. Harrison, D. R. Baigent, I. D. W. Samuel, R. H. Friend, A. C. Grimsdale, S. C. Moratti and A. B. Holmes, Phys. Rev. B, 1996, 53, 15815 CrossRef CAS PubMed.
- Z. Li, X. Zhao, C. Huang and X. Gong, J. Mater. Chem. C Mater., 2019, 7, 12373–12387 RSC.
- S. Singh, A. Dhawan, S. Karhana, M. Bhat and A. K. Dinda, Micromachines, 2020, 11, 1058 CrossRef PubMed.
- M. Cordeiro, F. Ferreira Carlos, P. Pedrosa, A. Lopez and P. V. Baptista, Diagnostics, 2016, 6, 43 CrossRef PubMed.
- B. Della Ventura, M. Gelzo, E. Battista, A. Alabastri, A. Schirato, G. Castaldo, G. Corso, F. Gentile and R. Velotta, ACS Appl. Mater. Interfaces, 2019, 11, 3753–3762 CrossRef CAS PubMed.
- Q. Liu, Q. Sun, J. Shen, H. Li, Y. Zhang, W. Chen, S. Yu, X. Li and Y. Chen, Coord. Chem. Rev., 2023, 482, 215078 CrossRef CAS.
- P. P. Pompa, R. Chiuri, L. Manna, T. Pellegrino, L. L. del Mercato, W. J. Parak, F. Calabi, R. Cingolani and R. Rinaldi, Chem. Phys. Lett., 2006, 417, 351–357 CrossRef CAS.
- K. Tomimuro, K. Tenda, Y. Ni, Y. Hiruta, M. Merkx and D. Citterio, ACS Sens., 2020, 5, 1786–1794 CrossRef CAS PubMed.
- L. Wu, C. Huang, B. P. Emery, A. C. Sedgwick, S. D. Bull, X.-P. He, H. Tian, J. Yoon, J. L. Sessler and T. D. James, Chem. Soc. Rev., 2020, 49, 5110–5139 RSC.
- X. Tian, L. C. Murfin, L. Wu, S. E. Lewis and T. D. James, Chem. Sci., 2021, 12, 3406–3426 RSC.
- W. Zhang, Z. Ma, L. Du and M. Li, Analyst, 2014, 139, 2641–2649 RSC.
- J. F. Callan, A. P. De Silva and D. C. Magri, Tetrahedron, 2005, 61, 8551–8588 CrossRef CAS.
- H. S. Jung, P. Verwilst, W. Y. Kim and J. S. Kim, Chem. Soc. Rev., 2016, 45, 1242–1256 RSC.
- P.-Y. Fu, B.-N. Li, Q.-S. Zhang, J.-T. Mo, S.-C. Wang, M. Pan and C.-Y. Su, J. Am. Chem. Soc., 2022, 144, 2726–2734 CrossRef CAS PubMed.
- A. C. Sedgwick, L. Wu, H.-H. Han, S. D. Bull, X.-P. He, T. D. James, J. L. Sessler, B. Z. Tang, H. Tian and J. Yoon, Chem. Soc. Rev., 2018, 47, 8842–8880 RSC.
- A. W. Martinez, S. T. Phillips, G. M. Whitesides and E. Carrilho, Diagnostics for the Developing World: Microfluidic Paper-Based Analytical Devices, ACS Publications, 2010 Search PubMed.
- J. P. Devadhasan and J. Kim, Sens. Actuators, B, 2018, 273, 18–24 CrossRef CAS.
- Y. Hang, J. Boryczka and N. Wu, Chem. Soc. Rev., 2022, 51, 329–375 RSC.
- G. Hong, A. L. Antaris and H. Dai, Nat. Biomed. Eng., 2017, 1, 0010 CrossRef CAS.
- J. R. Choi, K. W. Yong, J. Y. Choi and A. C. Cowie, Sensors, 2019, 19, 817 CrossRef PubMed.
- A. Patti and D. Acierno, Encyclopedia, 2023, 3, 665–676 CrossRef CAS.
- T. Yan, G. Zhang, H. Chai, L. Qu and X. Zhang, Front. Bioeng. Biotechnol., 2021, 9, 753692 CrossRef PubMed.
- J. C. Stockert and A. Blázquez-Castro, Fluorescence microscopy in life sciences, Bentham Science Publishers, 2017 Search PubMed.
- M. S. Baranov, K. A. Lukyanov, A. O. Borissova, J. Shamir, D. Kosenkov, L. V. Slipchenko, L. M. Tolbert, I. V. Yampolsky and K. M. Solntsev, J. Am. Chem. Soc., 2012, 134, 6025–6032 CrossRef CAS PubMed.
- M. Ptaszek, Prog. Mol. Biol. Transl. Sci, 2013, 113, 59–108 CAS.
- E. Horak, M. Robić, A. Šimanović, V. Mandić, R. Vianello, M. Hranjec and I. M. Steinberg, Dyes Pigm., 2019, 162, 688–696 CrossRef CAS.
- X. Liu, B. Cho, L.-Y. Chan, W. L. Kwan and C.-L. K. Lee, RSC Adv., 2015, 5, 106868–106876 RSC.
- Q. Zheng, M. F. Juette, S. Jockusch, M. R. Wasserman, Z. Zhou, R. B. Altman and S. C. Blanchard, Chem. Soc. Rev., 2014, 43, 1044–1056 RSC.
- H. Lu, F. Su, Q. Mei, Y. Tian, W. Tian, R. H. Johnson and D. R. Meldrum, J. Mater. Chem., 2012, 22, 9890–9900 RSC.
- H.-B. Cheng, Y. Li, B. Z. Tang and J. Yoon, Chem. Soc. Rev., 2020, 49, 21–31 RSC.
- S. Xu, H.-W. Liu, S.-Y. Huan, L. Yuan and X.-B. Zhang, Mater. Chem. Front., 2021, 5, 1076–1089 RSC.
- L. L. del Mercato, M. Moffa, R. Rinaldi and D. Pisignano, Small, 2015, 11, 6417–6424 CrossRef CAS PubMed.
- I. L. Moldero, A. Chandra, M. Cavo, C. Mota, D. Kapsokalyvas, G. Gigli, L. Moroni and L. L. del Mercato, Small, 2020, 2002258 CrossRef CAS PubMed.
- R. Rizzo, V. Onesto, G. Morello, H. Iuele, F. Scalera, S. Forciniti, G. Gigli, A. Polini, F. Gervaso and L. L. del Mercato, Mater. Today Bio, 2023, 20, 100655 CrossRef CAS PubMed.
- L. Hou, Y. Qin, J. Li, S. Qin, Y. Huang, T. Lin, L. Guo, F. Ye and S. Zhao, Biosens. Bioelectron., 2019, 143, 111605 CrossRef CAS PubMed.
- A. W. Martinez, S. T. Phillips, G. M. Whitesides and E. Carrilho, Diagnostics for the Developing World: Microfluidic Paper-Based Analytical Devices, ACS Publications, 2010 Search PubMed.
- J. Kaur and P. Singh, Expert Opin. Ther. Pat., 2011, 21, 437–454 CrossRef CAS PubMed.
- Z. Ma, G. Saluta, G. L. Kucera and U. Bierbach, Bioorg. Med. Chem. Lett., 2008, 18, 3799–3801 CrossRef CAS PubMed.
- J. Xu, G. Niu, X. Wei, M. Lan, L. Zeng, J. M. Kinsella and R. Sheng, Dyes Pigm., 2017, 139, 166–173 CrossRef CAS.
- A. Santoro, F. Tuyèras, G. Dupeyre, P. P. Laine, I. Ciofini, F. Nastasi, F. Puntoriero and S. Campagna, Dyes Pigm., 2018, 159, 619–636 CrossRef CAS.
- Y.-L. Tsai, C.-C. Chang, C.-C. Kang and T.-C. Chang, J. Lumin., 2007, 127, 41–47 CrossRef CAS.
- L. Xie, Y. Chen, W. Wu, H. Guo, J. Zhao and X. Yu, Dyes Pigm., 2012, 92, 1361–1369 CrossRef CAS.
- E. V. Antina, N. A. Bumagina, A. I. V’yugin and A. V. Solomonov, Dyes Pigm., 2017, 136, 368–381 CrossRef CAS.
- M. Mojzych and M. Henary, Heterocyclic polymethine dyes: Synthesis, properties and applications, 2008, pp. 1–9 Search PubMed.
- R. Irshad, S. Asim, A. Mansha and Y. Arooj, J. Fluoresc., 2023, 1–31 Search PubMed.
- M. Homocianu and A. Airinei, J. Fluoresc., 2016, 26, 1617–1635 CrossRef CAS PubMed.
- V. Martinez and M. Henary, Chem. – Eur. J., 2016, 22, 13764–13782 CrossRef CAS PubMed.
- K. Ayyavoo and P. Velusamy, New J. Chem., 2021, 45, 10997–11017 RSC.
- A. Chandra, S. Prasad, H. Iuele, F. Colella, R. Rizzo, E. D’Amone, G. Gigli and L. L. Del Mercato, Chem. – Eur. J., 2021, 27, 13318–13324 CrossRef PubMed.
- W. Qiao and Z. Li, Symmetry, 2022, 14, 966 CrossRef CAS.
- O. Karaman, G. A. Alkan, C. Kizilenis, C. C. Akgul and G. Gunbas, Coord. Chem. Rev., 2023, 475, 214841 CrossRef CAS.
- A. Chandra, S. Prasad, F. Alemanno, M. De Luca, R. Rizzo, R. Romano, G. Gigli, C. Bucci, A. Barra and L. L. Del Mercato, ACS Appl. Mater. Interfaces, 2022, 14, 18133–18149 CrossRef CAS PubMed.
- R. Rizzo, V. Onesto, S. Forciniti, A. Chandra, S. Prasad, H. Iuele, F. Colella, G. Gigli and L. L. Del Mercato, Biosens. Bioelectron., 2022, 212, 114401 CrossRef CAS PubMed.
- D. Liu, Z. He, Y. Zhao, Y. Yang, W. Shi, X. Li and H. Ma, J. Am. Chem. Soc., 2021, 143, 17136–17143 CrossRef CAS PubMed.
- Q. Liu, Q. Sun, J. Shen, H. Li, Y. Zhang, W. Chen, S. Yu, X. Li and Y. Chen, Coord. Chem. Rev., 2023, 482, 215078 CrossRef CAS.
- W. Huang, L. U. O. Shenglin, Y. Dong and S. Zhang, J. Zhejiang Univ. Sci. B, 2021, 22, 171 CrossRef PubMed.
- A. Goncharov, H. Joung, R. Ghosh, G. Han, Z. S. Ballard, Q. Maloney, A. Bell, C. T. Z. Aung, O. B. Garner and D. Di Carlo, Small, 2023, 2300617 CrossRef CAS PubMed.
- Y. Cai, K. Kang, Q. Li, Y. Wang and X. He, Molecules, 2018, 23, 1102 CrossRef PubMed.
- Y. Sekine, S. B. Kim, Y. Zhang, A. J. Bandodkar, S. Xu, J. Choi, M. Irie, T. R. Ray, P. Kohli and N. Kozai, Lab Chip, 2018, 18, 2178–2186 RSC.
- P. Ghassemi, B. Wang, J. Wang, Q. Wang, Y. Chen and T. J. Pfefer, IEEE Trans. Biomed. Eng., 2017, 64, 1650–1653 Search PubMed.
- L. He, B. Chen, Y. Hu, B. Hu, Y. Li and X. Yang, Anal. Bioanal. Chem., 2023, 415, 5499–5509 CrossRef CAS PubMed.
- A. A. Alzu’bi, L. Zhou and V. J. M. Watzlaf, Perspect. Health Inf. Manag., 2019, 16, 1a Search PubMed.
- B. H. Davis, G. DeFrank, N. A. Limdi and S. Harada, Clin. Transl. Sci., 2020, 13, 260–264 CrossRef CAS PubMed.
- R. C. Tseng, C.-C. Chen, S.-M. Hsu and H.-S. Chuang, Sensors, 2018, 18, 2651 CrossRef PubMed.
- A. K. Yetisen, N. Jiang, C. M. Castaneda Gonzalez, Z. I. Erenoglu, J. Dong, X. Dong, S. Stößer, M. Brischwein, H. Butt, M. F. Cordeiro, M. Jakobi, O. Hayden and A. W. Koch, Adv. Mater., 2020, 1906762 CrossRef CAS PubMed.
- E. S. Kim, H. Chon, Y. Kwon, M. Lee, M. J. Kim and Y. H. Choe, Ther. Drug Monit., 2024, 46, 460–467 CrossRef CAS PubMed.
- J. Leonard, H. C. Koydemir, V. S. Koutnik, D. Tseng, A. Ozcan and S. K. Mohanty, J. Hazard. Mater. Lett., 2022, 3, 100052 CrossRef CAS.
- H. Kim, S.-K. Choi, J. Ahn, H. Yu, K. Min, C. Hong, I.-S. Shin, S. Lee, H. Lee, H. Im, J. Ko and E. Kim, Sens. Actuators, B, 2021, 329, 129248 CrossRef CAS PubMed.
- M. Zhang, L. Gao, X. Zhao, Y. Duan, Y. Liao and T. Han, Dyes Pigm., 2022, 205, 110561 CrossRef CAS.
- Y. Lu, Z. Shi and Q. Liu, Curr. Opin. Food Sci., 2019, 28, 74–81 CrossRef.
- G. Rateni, P. Dario and F. Cavallo, Sensors, 2017, 17, 1453 CrossRef PubMed.
- N. Cheng, Y. Song, M. M. A. Zeinhom, Y.-C. Chang, L. Sheng, H. Li, D. Du, L. Li, M.-J. Zhu, Y. Luo, W. Xu and Y. Lin, ACS Appl. Mater. Interfaces, 2017, 9, 40671–40680 CrossRef CAS PubMed.
- Y. Shan, B. Wang, H. Huang, D. Jian, X. Wu, L. Xue, S. Wang and F. Liu, Biosens. Bioelectron., 2019, 132, 238–247 CrossRef CAS PubMed.
- N. C. Shaner, P. A. Steinbach and R. Y. Tsien, Nat. Methods, 2005, 2, 905–909 CrossRef CAS PubMed.
- D. M. Chudakov, M. V. Matz, S. Lukyanov and K. A. Lukyanov, Physiol. Rev., 2010, 1103 CrossRef CAS PubMed.
- S. J. Remington, Protein Sci., 2011, 1509 CrossRef CAS PubMed.
- E. Balleza, J. M. Kim and P. Cluzel, Nat. Methods, 2018, 15, 47–51 CrossRef CAS PubMed.
- A. Ibraheem and R. E. Campbell, Curr. Opin. Chem. Biol., 2010, 30 CrossRef CAS PubMed.
- Z. Chen, T. M. Truong and H. W. Ai, Chemosensors, 2017, 32 CrossRef PubMed.
- K. Horikawa, Y. Yamada, T. Matsuda, K. Kobayashi, M. Hashimoto, T. Matsu-ura, A. Miyawaki, T. Michikawa, K. Mikoshiba and T. Nagai, Nat. Methods, 2010, 7, 729–732 CrossRef CAS PubMed.
- M. Deng, J. Yuan, H. Yang, X. Wu, X. Wei, Y. Du, G. Wong, Y. Tao, G. Liu, Z. Jin and J. Chu, ACS Nano, 2021, 15, 17602–17612 CrossRef CAS PubMed.
- M. De, S. Rana, H. Akpinar, O. R. Miranda, R. R. Arvizo, U. H. F. Bunz and V. M. Rotello, Nat. Chem., 2009, 1, 461–465 CrossRef CAS PubMed.
- G.-T. Liang, C. Lai, Z. Yue, H. Zhang, D. Li, Z. Chen, X. Lu, L. Tao, F. V. Subach and K. D. Piatkevich, Front. Bioeng. Biotechnol., 2022, 1039317 CrossRef PubMed.
- M. Wang, L. Jin, P. Hang-Mei Leung, F. Wang-Ngai Chow, X. Zhao, H. Chen, W. Pan, H. Liu and S. Li, Front. Bioeng. Biotechnol., 2024, 1393789 CrossRef PubMed.
- F. R. Mansour, S. F. Hammad, I. A. Abdallah, A. Bedair, R. M. Abdelhameed and M. Locatelli, TrAC, Trends Anal. Chem., 2024, 172, 117596 CrossRef CAS.
- A. T. Krasley, E. Li, J. M. Galeana, C. Bulumulla, A. G. Beyene and G. S. Demirer, Chem. Rev., 2024, 124, 3085–3185 CrossRef CAS PubMed.
- L. N. Thwala, S. C. Ndlovu, K. T. Mpofu, M. Y. Lugongolo and P. Mthunzi-Kufa, Nanomaterials, 2023, 13, 1247 CrossRef CAS PubMed.
- Y. Hang, A. Wang and N. Wu, Chem. Soc. Rev., 2024, 53, 2932–2971 RSC.
- Q. Yang, Y. Wu, J. Chen, M. Lu, X. Wang, Z. Zhang, H. Xiong, J. Choo and L. Chen, Coord. Chem. Rev., 2024, 507, 215768 CrossRef CAS.
- M. Bartolomé, M. J. Villaseñor and Á. Ríos, Anal. Chim. Acta, 2025, 344693 Search PubMed.
- M. Jouyandeh, S. M. Sajadi, F. Seidi, S. Habibzadeh, M. T. Munir, O. Abida, S. Ahmadi, D. Kowalkowska-Zedler, N. Rabiee, M. Rabiee, G. Heidari, M. Hassanpour, E. Nazarzadeh Zare and M. R. Saeb, OpenNano, 2022, 8, 100104 CrossRef CAS.
- C. Jin, Z. Wu, J. H. Molinski, J. Zhou, Y. Ren and J. X. J. Zhang, Mater. Today Bio, 2022, 14, 100263 CrossRef CAS PubMed.
- K. M. Ganesh, S. Bhaskar, V. S. K. Cheerala, P. Battampara, R. Reddy, S. C. Neelakantan, N. Reddy and S. S. Ramamurthy, Nanomaterials, 2024, 14, 111 CrossRef CAS PubMed.
- M. Cedrún-Morales, M. Ceballos, E. Polo, P. del Pino and B. Pelaz, Chem. Commun., 2023, 59, 2869–2887 RSC.
- M. Ceballos, S. Funes-Hernando, G. Zampini, M. Cedrún-Morales, J. M. Vila-Fungueiriño, B. Pelaz and P. del Pino, Small Struct., 2024, 2300464 CrossRef CAS.
- M. Pamei and A. Puzari, Nano-Struct. Nano-Objects, 2019, 19, 100364 CrossRef CAS.
- A. Kidanemariam and S. Cho, Sensors, 2025, 25, 5081 CrossRef CAS PubMed.
- J. Zhang, Y. Li, F. Chai, Q. Li, D. Wang, L. Liu, B. Z. Tang and X. Jiang, Sci. Adv., 2022, abo1874 CrossRef PubMed.
- J. L. Vivero-Escoto, R. C. Huxford-Phillips and W. Lin, Chem. Soc. Rev., 2012, 41, 2673 RSC.
- X. Ji, H. Wang, B. Song, B. Chu and Y. He, Front. Chem., 2018, 38 CrossRef PubMed.
- A. C. Siciliano, S. Forciniti, V. Onesto, H. Iuele, D. D. Cave, F. Carnevali, G. Gigli, E. Lonardo and L. L. del Mercato, Adv. Healthcare Mater., 2024, 2401138 CrossRef CAS PubMed.
- R. Prieto-Montero, A. Katsumiti, M. P. Cajaraville, I. López-Arbeloa and V. Martínez-Martínez, Sensors, 2020, 20, 5590 CrossRef CAS PubMed.
- H. Iuele, V. Onesto, A. C. Siciliano, S. Forciniti, F. Colella, G. Grasso, G. Gigli and L. L. del Mercato, Chem. Eng. J., 2025, 525, 170576 CrossRef CAS.
- F. Colella, S. Forciniti, V. Onesto, G. Grasso, H. Iuele, G. Gigli and L. L. del Mercato, J. Mater. Chem. B, 2024, 12, 10573–10583 RSC.
- S. Kakkar, P. Gupta, S. P. Singh Yadav, D. Raj, G. Singh, S. Chauhan, M. K. Mishra, E. Martín-Ortega, S. Chiussi and K. Kant, Mater. Today Bio, 2024, 28, 101188 CrossRef CAS PubMed.
- S. Siavashy, M. Soltani, S. Rahimi, M. Hosseinali, Z. Guilandokht and K. Raahemifar, Biosens. Bioelectron. X, 2024, 19, 100489 CAS.
- V. Gubala, G. Giovannini, F. Kunc, M. P. Monopoli and C. J. Moore, Cancer Nanotechnol., 2020, 11, 1 CrossRef CAS.
- V. Secchi, A. Armanni, L. Barbieri, A. Bruno, A. Colombo, S. Fumagalli, E. A. Kukushkina, R. Lorenzi, L. Marchesi, H. Moukham, A. Paleari, A. Ronchi, G. Tomaino, F. Tripodi, M. Colombo, L. Sironi and A. Monguzzi, Front. Nanotechnol., 2025, 1465429 CrossRef.
- M. De Luca, M. M. Ferraro, R. Hartmann, P. Rivera-Gil, A. Klingl, M. Nazarenus, A. Ramirez, W. J. Parak, C. Bucci, R. Rinaldi and L. L. del Mercato, ACS Appl. Mater. Interfaces, 2015, 7, 15052–15060 CrossRef CAS PubMed.
- L. Ouologuem, A. Kübler, S. Ouologuem, A. Hadzic, J. B. Stöckl, A. C. Siciliano, S. Forciniti, S. Nigro, H. Iuele, V. Onesto, A. Nguyen, D. Matzek, C. Abrahamian, C. Grimm, B. Popper, G. Gigli, L. L. del Mercato, O. M. Merkel, T. Fröhlich, S. Theurich and K. Bartel, Front. Immunol., 2025, 1668066 CrossRef CAS PubMed.
- V. Onesto, S. Forciniti, F. Alemanno, K. Narayanankutty, A. Chandra, S. Prasad, A. Azzariti, G. Gigli, A. Barra, A. De Martino, D. De Martino and L. L. del Mercato, ACS Nano, 2023, 17, 3313–3323 CrossRef CAS PubMed.
- F. Chen, G. Hableel, E. R. Zhao and J. V. Jokerst, J. Colloid Interface Sci., 2018, 521, 261–279 CrossRef CAS PubMed.
- S. Singh, A. Dhawan, S. Karhana, M. Bhat and A. K. Dinda, Micromachines, 2020, 11, 1058 CrossRef PubMed.
- V. G. Reshma and P. V. Mohanan, J. Lumin., 2019, 205, 287–298 CrossRef CAS.
- Z. Ramezani, F. K. Ghalehsardi and S. Noorizadeh, in Quantum Dots in Bioanalytical Chemistry and Medicine, Royal Society of Chemistry, 2023, pp. 1–36 Search PubMed.
- M. Nabil and F. Megahed, J. Fluoresc., 2024, 34, 2467–2484 CrossRef PubMed.
- M. Ussia, V. Privitera and S. Scalese, Adv. Mater. Interfaces, 2024, 2300970 CrossRef.
- G. Grasso, R. Scarfiello, F. Colella, V. Onesto, S. Forciniti, G. Gigli, L. Carbone and L. L. del Mercato, Sens Actuators Rep., 2025, 9, 100303 CrossRef.
- Z. A. Qureshi, H. Dabash, D. Ponnamma and M. K. G. Abbas, Heliyon, 2024, 10, e31634 CrossRef CAS PubMed.
- N. A. Pechnikova, K. Domvri, K. Porpodis, M. S. Istomina, A. V. Iaremenko and A. V. Yaremenko, Aggregate, 2025, e707 CrossRef CAS.
- M. Tavan, Z. Yousefian, Z. Bakhtiar, M. Rahmandoust and M. H. Mirjalili, Ind. Crops Prod., 2025, 231, 121207 CrossRef CAS.
- H. Tang, C. Lu, O. Kodra and J. Zhang, Carbon Trends, 2025, 100579 CrossRef CAS.
- T.-Q. Nguyen and A. Trentin, Mater. Today Commun., 2025, 47, 113204 CrossRef CAS.
- N. Sohal, S. Singla, S. J. Malode, S. Basu, B. Maity and N. P. Shetti, ACS Appl. Nano Mater., 2023, 6, 10925–10943 CrossRef CAS.
- H. Tavakoli, S. Mohammadi, X. Li, G. Fu and X. Li, TrAC, Trends Anal. Chem., 2022, 157, 116806 CrossRef CAS PubMed.
- J. Jin, J. Guo, J. Guo and D. Li, Adv. Sens. Res., 2024, 2400037 CrossRef CAS.
- S. Wilhelm, ACS Nano, 2017, 11, 10644–10653 CrossRef CAS PubMed.
- M. S. Arai and A. S. S. de Camargo, Nanoscale Adv., 2021, 3, 5135–5165 RSC.
- J. Jin, Y.-J. Gu, C. W.-Y. Man, J. Cheng, Z. Xu, Y. Zhang, H. Wang, V. H.-Y. Lee, S. H. Cheng and W.-T. Wong, ACS Nano, 2011, 5, 7838–7847 CrossRef CAS PubMed.
- K. S. Janbandhu, V. B. Pawade, A. M. Pethe and S. J. Dhoble, Sci. Rep., 2025, 15, 34517 CrossRef CAS PubMed.
- L.-D. Xu, F.-L. Du, J. Zhu and S.-N. Ding, Analyst, 2021, 146, 706–713 RSC.
- J. Guo, S. Chen, S. Tian, K. Liu, J. Ni, M. Zhao, Y. Kang, X. Ma and J. Guo, Biosens. Bioelectron., 2021, 181, 113160 CrossRef CAS PubMed.
- J. Hu, Y.-Z. Jiang, L.-L. Wu, Z. Wu, Y. Bi, G. Wong, X. Qiu, J. Chen, D.-W. Pang and Z.-L. Zhang, Anal. Chem., 2017, 89, 13105–13111 CrossRef CAS PubMed.
- L. Liang, M. Su, L. Li, F. Lan, G. Yang, S. Ge, J. Yu and X. Song, Sens. Actuators, B, 2016, 229, 347–354 CrossRef CAS.
- A. M. Jimenez Jimenez, M. A. M. Rodrigo, V. Milosavljevic, S. Krizkova, P. Kopel, Z. Heger and V. Adam, Sens. Actuators, B, 2017, 240, 503–510 CrossRef CAS.
- Y. F. Shi, Y. P. Jiang, X. Z. Wang, Z. Q. Zhang, J. Z. Huo, Y. Y. Liu, X. R. Wang and B. Ding, ACS Appl. Nano Mater., 2022, 5, 15629–15641 CrossRef CAS.
- J. Zou, X. Liu, X. Ren, L. Tan, C. Fu, Q. Wu, Z. Huang and X. Meng, Nanoscale, 2021, 13, 7844–7850 RSC.
- J. Zhang, Y. Zheng, J. Lee, A. Hoover, S. A. King, L. Chen, J. Zhao, Q. Lin, C. Yu, L. Zhu and X. Wu, Adv. Sci., 2023, 2203943 CrossRef CAS PubMed.
- S. Tombelli, C. Trono, S. Berneschi, C. Berrettoni, A. Giannetti, R. Bernini, G. Persichetti, G. Testa, G. Orellana, F. Salis, S. Weber, P. B. Luppa, G. Porro, G. Quarto, M. Schubert, M. Berner, P. P. Freitas, S. Cardoso, F. Franco, V. Silverio, M. Lopez-Martinez, U. Hilbig, K. Freudenberger, G. Gauglitz, H. Becker, C. Gärtner, M. T. O’Connell and F. Baldini, Anal. Bioanal. Chem., 2022, 414, 3243–3255 CrossRef CAS PubMed.
- J. Zhang and G. Shi, Anal. Chim. Acta, 2022, 339572 CrossRef CAS PubMed.
- H. Liang, Y. Li, B. Lin, Y. Yu, Y. Wang, L. Zhang, Y. Cao and M. Guo, Microchem. J., 2022, 175, 107221 CrossRef CAS.
- B. Jin, Z. Li, G. Zhao, J. Ji, J. Chen, Y. Yang and R. Xu, Anal. Chim. Acta, 2022, 1192, 339388 CrossRef CAS PubMed.
- Q. Zheng, M. F. Juette, S. Jockusch, M. R. Wasserman, Z. Zhou, R. B. Altman and S. C. Blanchard, Chem. Soc. Rev., 2014, 43, 1044–1056 RSC.
- J. Liu, C. Liu and W. He, Curr. Org. Chem., 2013, 17, 564–579 CrossRef CAS.
- H.-B. Cheng, Y. Li, B. Z. Tang and J. Yoon, Chem. Soc. Rev., 2020, 49, 21–31 RSC.
- B. C. Campbell, M. G. Paez-Segala, L. L. Looger, G. A. Petsko and C. F. Liu, Nat. Methods, 2022, 19, 1612–1621 CrossRef CAS.
- J. K. Heppert, D. J. Dickinson, A. M. Pani, C. D. Higgins, A. Steward, J. Ahringer, J. R. Kuhn and B. Goldstein, Mol. Biol. Cell, 2016, 27, 3385–3394 CrossRef CAS PubMed.
- D. Semeniak, D. F. Cruz, A. Chilkoti and M. H. Mikkelsen, Adv. Mater., 2023, 35, e2107986 CrossRef PubMed.
- M. A. Farzin, S. M. Naghib and N. Rabiee, TrAC, Trends Anal. Chem., 2025, 191, 118345 CrossRef CAS.
- V. D. Punetha, P. Sanghani, M. Punetha and V. Pathak, Upconversion Nanoparticles for Biomedical Applications, Elsevier, 2026, pp. 1–43 Search PubMed.
- P. Navid, H. R. Akbari-Hasanjani and R. Akbari-Hasanjani, Nano Select, 2025, e70046 CrossRef CAS.
- J. K. Heppert, D. J. Dickinson, A. M. Pani, C. D. Higgins, A. Steward, J. Ahringer, J. R. Kuhn and B. Goldstein, Mol. Biol. Cell, 2016, 27, 3385–3394 CrossRef CAS PubMed.
- D. Semeniak, D. F. Cruz, A. Chilkoti and M. H. Mikkelsen, Adv. Mater., 2025, 2107986 Search PubMed.
- B. Liu, Y. Li, H. Wan, L. Wang, W. Xu, S. Zhu, Y. Liang, B. Zhang, J. Lou, H. Dai and K. Qian, Adv. Funct. Mater., 2016, 26, 7994–8002 CrossRef CAS.
- F. Zang, Z. Su, L. Zhou, K. Konduru, G. Kaplan and S. Y. Chou, Adv. Mater., 2019, 1902331 CrossRef.
- W. Hu, Z. Lu, Y. Liu, T. Chen, X. Zhou and C. M. Li, Lab Chip, 2013, 13, 1797 RSC.
- N. Yamaguchi, Y. Tokunaga, S. Goto, Y. Fujii, F. Banno and A. Edagawa, Sci. Rep., 2017, 7, 3092 CrossRef PubMed.
- S. Shin, B. Kim, Y.-J. Kim and S. Choi, Biosens. Bioelectron., 2019, 133, 169–176 CrossRef CAS.
- Z. Göröcs, M. Tamamitsu, V. Bianco, P. Wolf, S. Roy, K. Shindo, K. Yanny, Y. Wu, H. C. Koydemir, Y. Rivenson and A. Ozcan, Light Sci. Appl., 2018, 7, 66 CrossRef PubMed.
- H. Wang, Y. Rivenson, Y. Jin, Z. Wei, R. Gao, H. Günaydın, L. A. Bentolila, C. Kural and A. Ozcan, Nat. Methods, 2019, 16, 103–110 CrossRef CAS.
- M. Wang, H. Liang, X. Chen, D. Chen, J. Wang, Y. Zhang and J. Chen, Biosensors, 2022 DOI:10.3390/bios12070443.
- H. Kanno, K. Hiramatsu, H. Mikami, A. Nakayashiki, S. Yamashita, A. Nagai, K. Okabe, F. Li, F. Yin, K. Tominaga, O. F. Bicer, R. Noma, B. Kiani, O. Efa, M. Büscher, T. Wazawa, M. Sonoshita, H. Shintaku, T. Nagai, S. Braun, J. P. Houston, S. Rashad, K. Niizuma and K. Goda, Nat. Commun., 2024, 15, 7376 CrossRef CAS PubMed.
- K. İçöz, A. Eken, S. Çınar, A. Murat, S. Özcan, E. Ünal and G. Deniz, Sep. Sci. Technol., 2021, 56, 2659–2666 CrossRef.
- C. P. Y. Chan, W. C. Mak, K. Y. Cheung, K. K. Sin, C. M. Yu, T. H. Rainer and R. Renneberg, Annu. Rev. Anal. Chem., 2013, 6, 191–211 CrossRef CAS.
- M. Liu and Y. Wen, Heliyon, 2024, 10, e38444 CrossRef CAS PubMed.
- S. Rasheed, T. Kanwal, N. Ahmad, B. Fatima, M. Najam-ul-Haq and D. Hussain, TrAC, Trends Anal. Chem., 2024, 173, 117640 CrossRef CAS.
- D. Strohmaier-Nguyen, C. Horn and A. J. Baeumner, Biosens. Bioelectron., 2025, 267, 116795 CrossRef CAS PubMed.
- G.-R. Han, A. Goncharov, M. Eryilmaz, S. Ye, B. Palanisamy, R. Ghosh, F. Lisi, E. Rogers, D. Guzman, D. Yigci, S. Tasoglu, D. Di Carlo, K. Goda, R. A. McKendry and A. Ozcan, Nat. Commun., 2025, 16, 3165 CrossRef CAS PubMed.
- T. Akkaş, M. Reshadsedghi, M. Şen, V. Kılıç and N. Horzum, Adv. Mater., 2025, 2504796 CrossRef PubMed.
- Y. Zhao, T. Sun, H. Zhang, W. Li, C. Lian, Y. Jiang, M. Qu, Z. Zhao, Y. Wang, Y. Sun, H. Duan, Y. Ren, P. Liu, X. Lang and S. Chen, Biosensors, 2025, 15, 565 CrossRef CAS.
- R. W. Oei, G. Hou, F. Liu, J. Zhong, J. Zhang, Z. An, L. Xu and Y. Yang, PLoS One, 2019, 14, e0213626 CrossRef CAS PubMed.
- A. Ait Lahcen, J. Rajendran and G. Slaughter, Biosens. Bioelectron. X, 2026, 28, 100728 CAS.
- H. Liu, E. Dai, R. Xiao, Z. Zhou, M. Zhang, Z. Bai, Y. Shao, K. Qi, J. Tu, C. Wang and S. Wang, Sens. Actuators, B, 2021, 329, 129196 CrossRef CAS PubMed.
- F. Lin, C. Zhang, Z. Huang, Y. Wang, M. Yi, J. Li, X. Weng, Y. Chen, P. Lai and J. Qu, Appl. Phys. Rev., 2026, 011303 Search PubMed.
- Y. Zu, H. Chang and Z. Cui, Nexus, 2025, 2, 100059 CrossRef.
- R. El-Khoury and G. Zaatari, Diagnostics, 2025, 15, 2308 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





