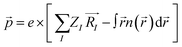Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Orientation control of Cu3(HHTP)2 MOF films using a dual working electrode electrochemical synthesis method
Yu Wang,
Abigail M. Lister ,
Christopher E. Patrick
,
Christopher E. Patrick ,
Sparsh Tyagi,
Shengming Zhang,
Axel Forssberg,
Neil Young,
Weishuo Li
,
Sparsh Tyagi,
Shengming Zhang,
Axel Forssberg,
Neil Young,
Weishuo Li and
Martin R. Castell
and
Martin R. Castell *
*
Department of Materials, University of Oxford, Parks Road, Oxford OX1 3PH, UK. E-mail: martin.castell@materials.ox.ac.uk
First published on 24th February 2026
Abstract
Materials that consist of stacked two-dimensional electronically conductive metal–organic frameworks (2D-cMOFs) have potential for integration into novel electronic devices. These layered materials have crystal structures and charge transport properties that are highly anisotropic. For effective use as functional device materials it is therefore important to achieve control over the crystallographic orientation of the films and their growth mechanisms. Here we report an electrochemical method to synthesise Cu3(HHTP)2 films in situ on ITO glass with controllable film orientation and thickness using a dual working electrode setup. By adjusting the ligand concentration in the electrolyte, different Cun(HHTP)m complexes form, which interact differently with the electrode upon which the MOF is grown. This leads to the consistent formation of edge-on or face-on oriented MOF films. For high ligand concentrations the 2D layers grow perpendicular to the electrode substrate, whereas for low ligand concentrations the layers are parallel to the substrate. Extensive characterisation and theoretical simulations of this material system lead to the conclusion that growth of the different MOF orientations is a result of the electric dipoles on the Cun(HHTP)m complexes. Cu(HHTP) and Cu2(HHTP) complexes are created under high ligand concentration conditions and have electric dipoles that result in edge-on nucleation due to electrostatic interactions with the potential on the electrode. Under low ligand concentrations Cu3(HHTP) complexes are formed that do not have a dipole and nucleate face-on via van der Waals interactions.
1. Introduction
Electrically conductive two-dimensional metal–organic frameworks (2D MOFs) are an evolving class of materials with potential for use in a variety of electronic devices such as chemiresistive sensors, field-effect transistors, and thermoelectric devices.1,2 Achieving precise control over the morphology, contact resistance, thickness, and orientation of 2D MOF films is important for the optimisation of the performance of these devices. The thin film form factor is required to maximize surface-interaction-driven functionality for sensors, and enable reliable device operation for transistors.3 Orientation control during materials integration into the device is especially relevant for stacked crystalline 2D MOF materials, where both structure and electrical conductivity are highly anisotropic.4–6Various synthesis methods, such as layer-by-layer methods, vapor-assisted synthesis, electrochemical growth, and chemical vapor deposition have been developed to fabricate large area 2D MOF thin films.7–13 Some of these methods have successfully achieved face-on oriented 2D MOF films where the 2D layers are parallel to the substrate surface. However, consistent fabrication of edge-on films, where the 2D layers are perpendicular to the substrate, remains challenging.5,14,15 One effective strategy for achieving edge-on orientated films is to use the hydrophilic functional group of the ligand to induce the ligand to stand upright on the surface of –OH functionalized substrates and use layer-by-layer deposition to grow the films.4,5,16 This method enables the fabrication of nanoscale-thickness films, however producing dense thicker films remain challenging due to the method's spraying process, which requires highly accurate repetitive operations. An alternative method for fabricating 2D MOF films is electrochemical synthesis.17 This method has the advantages of high efficiency, relatively simple operation, scalability, and for enabling the control of film morphology and thickness.10,14,18 Recently, a catalyst-assisted approach was introduced to controllably synthesize both face-on and edge-on oriented Cu3(HHTP)2 films by applying catalyst layers to modulate ligand oxidation kinetics.19 This method requires catalytically active substrates and additional fabrication steps, which limits its direct applicability in the context of device integration.
The MOF synthesis method described here, enables orientation control of Cu3(HHTP)2 films through a catalyst-free electrochemical route by modulating the ligand concentration in solution. We grow edge-on and face-on oriented Cu3(HHTP)2 films on indium-doped tin oxide (ITO) substrates with controllable film orientation and thickness. This is achieved using a dual working electrode setup that enables the MOF formation and anodic oxidation of Cu to occur on separate surfaces. We also elucidate the growth mechanism responsible for the orientation control.16,20 Our strategy provides flexibility in synthesis, broad compatibility with device substrates, and a mechanistic basis for tuning film orientation, offering a scalable route to high-quality, oriented 2D MOF films.
2. Experimental and theoretical techniques
2.1. Materials
2,3,6,7,10,11-Hexahydroxytriphenylene (HHTP) was purchased from Fluorochem Ltd, UK. Before use, the HHTP powder was dissolved in methanol, filtered through a sterile syringe filter with a 450 nm pore size, and subsequently dried under argon flow. Tributyl methyl ammonium methyl sulfate (TBMAMS) was purchased from Santa Cruz Biotechnology, USA. 3M copper tape (1181 series) with an acrylic adhesive was purchased from RS Components, UK. The copper tape was ground with an abrasive disk, and rinsed with water and ethanol before use. ITO-coated glass slides with a surface resistivity of 8–12 Ω sq−1 were purchased from Sigma Aldrich, UK. Before use, the ITO glass substrates were sonicated with DI water, acetone, and ethanol for 10 min each and then dried in air. No additional surface treatment was applied.2.2. Synthesis methods
Solvothermal synthesis of Cu3(HHTP)2 powder was carried out using the following method. A solid mixture of HHTP (7.5 mg) and copper(II) acetate (10.5 mg) was suspended in 2 mL of deionized water in a glass vial. The vial was then sonicated for 10 min. The mixture was heated at 85 °C for 72 h resulting in dark blue crystals. The resulting powder was washed with deionized water, ethanol, and acetone three times and dried in air overnight.Cyclic voltammetry (CV) was performed using a three-electrode set-up, with a Pt counter electrode and an Ag/AgCl reference electrode. For the HHTP CV measurement, the electrolyte solution contained 2.6 mM HHTP and 0.02 M TBMAMS in a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water and ethanol mixture, with ITO glass serving as the working electrode. In the case of the Cu tape measurement, the electrolyte solution contained 0.02 M TBMAMS in the same 4
1 water and ethanol mixture, with ITO glass serving as the working electrode. In the case of the Cu tape measurement, the electrolyte solution contained 0.02 M TBMAMS in the same 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water and ethanol mixture, with Cu tape used as the working electrode.
1 water and ethanol mixture, with Cu tape used as the working electrode.
The same CV set-up was used for electrochemical synthesis of the Cu3(HHTP)2 oriented films on ITO as shown schematically in Fig. 1. Cu tape was adhered on the opposite side of the ITO glass, with the same size as the ITO substrate. We refer to this setup as a dual working electrode because the electrode is formed from two separate active surfaces. Different concentrations of ligand were employed in the synthesis of the oriented films. For the edge-on films, 2.6 mM HHTP powder and 0.02 M TBMAMS were dispersed in a mixed solution of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water and ethanol. For the face-on films, 1.3 mM HHTP powder and 0.02 M TBMAMS were dispersed in mixed solution of 4
1 water and ethanol. For the face-on films, 1.3 mM HHTP powder and 0.02 M TBMAMS were dispersed in mixed solution of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water and ethanol. The solution was sonicated for 5 min before synthesis. The deposition was conducted for various times from 2 h to 10 h. The final film on ITO glass was washed using acetone.
1 water and ethanol. The solution was sonicated for 5 min before synthesis. The deposition was conducted for various times from 2 h to 10 h. The final film on ITO glass was washed using acetone.
2.3. Characterization
Scanning electron microscope (SEM) images were obtained using a Zeiss Merlin SEM. Samples for transmission electron microscopy (TEM) were dispersed in ethanol, and drop-cast onto a carbon coated copper grid. TEM images were obtained using a JEOL-2100 microscope at 200 kV. Powder X-ray diffraction (XRD) measurements were performed on a Rigaku Miniflex XRD with a 1.54 Å Cu Kα X-ray source. Film XRD measurements were performed on an Empyrean XRD. UV-vis absorption spectra were measured by using a Varian Cary 5000 UV-visible-NIR spectrometer. A Bruker Dimension Icon atomic force microscope (AFM) was used to characterise all samples in a glovebox filled with argon (<0.1 ppm H2O, <0.1 ppm O2) at room temperature. All AFM probes (SCM-PIT-V2) were calibrated according to standard samples (sapphire and Ti roughness sample) interpolating the actual spring constant and tip radius (∼5 nm). The PeakForce Tunneling AFM (TUNA) method was conducted to capture both the topography of the samples and the corresponding TUNA current data (average current during a full tapping cycle). 3D topography images were reconstructed using NanoScope Analysis 2.0 software. The film thickness data were obtained from the height difference between the plated samples and the substrates. The current value used to calculate the resistance was the average TUNA current response in a field of view of 5 µm × 5 µm under different applied voltages between the AFM tip and the sample. The gradient of I vs. V was extracted by linear fitting to calculate the resistance, R = (I/V)−1. With additional information of tip radius and thickness of the plated sample, the conductivity σ (S cm−1) was calculated with the following formula,16 where d (cm) is the film thickness, A (cm2) is the tip area, R(Ω) is the resistance: σ = d/(R × A). X-ray photoelectron spectroscopy (XPS) was performed on a PHI VersaProbe III with a monochromatic Al Kα X-ray source (1486.6 eV).2.4. Theoretical modelling
The electric dipole moments of the model complexes, discussed in Section 3.3, were calculated using density-functional theory (DFT), using plane wave basis sets and ultrasoft pseudopotentials as implemented in the Quantum ESPRESSO software package.21–23 The complexes were placed in a cubic simulation cell of side length 20 Å and allowed to relax until the force on each atom was smaller than 26 meV Å−1. The relaxed positions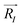 and the electronic charge density n(
and the electronic charge density n(![[r with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0072_20d1.gif) ) were then used to calculate the electric dipole moment
) were then used to calculate the electric dipole moment ![[p with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0070_20d1.gif) , as
, aswhere e is the electronic charge, and ZI is the valence charge of the atom located at position
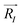 . In the structural relaxations, exchange and correlation were treated within the generalized-gradient approximation using the PBE functional.24 In order to evaluate the sensitivity of the electric dipole moments to the treatment of exchange and correlation, calculations were carried out both with the standard PBE functional and also with the hybrid PBE0 functional.25 The PBE0 functional is more computationally expensive, but expected to be more accurate due to its inclusion of exact Hartree–Fock exchange. The same qualitative results were found with both functionals, with the dipoles of the complexes found to be 1.25 eÅ and 0.26 eÅ within PBE, and 1.46 eÅ and 0.10 eÅ within PBE0.
. In the structural relaxations, exchange and correlation were treated within the generalized-gradient approximation using the PBE functional.24 In order to evaluate the sensitivity of the electric dipole moments to the treatment of exchange and correlation, calculations were carried out both with the standard PBE functional and also with the hybrid PBE0 functional.25 The PBE0 functional is more computationally expensive, but expected to be more accurate due to its inclusion of exact Hartree–Fock exchange. The same qualitative results were found with both functionals, with the dipoles of the complexes found to be 1.25 eÅ and 0.26 eÅ within PBE, and 1.46 eÅ and 0.10 eÅ within PBE0.
3. Results and discussion
3.1. Electrochemical MOF synthesis using a dual-working-electrode setup
As illustrated in Fig. 1a, we used a dual-working-electrode design where the working electrode is composed of copper tape adhered to the opposite side of an ITO substrate. The copper side of the working electrode acts as the Cu ion source, and the ITO side is where the oriented MOF grows. The electrolyte solution contains HHTP and TBMAMS in a mixed solvent of water/ethanol (v/v, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
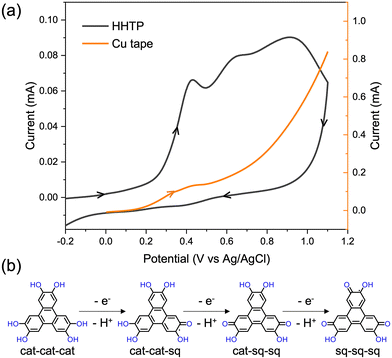
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). MOF formation requires an appropriate voltage to ionize the Cu and oxidize the ligand on the ITO electrode. The electrochemical behaviour of Cu and HHTP was investigated using cyclic voltammetry (CV) to determine the appropriate potential for electrochemical deposition. Previous studies have reported six one-electron-oxidation processes for HHTP after deprotonation, from fully reduced HHTP6− to fully oxidized HHTP0.26,27 As the CV scans in Fig. 2a show, the HHTP solution shows three anodic peaks at 0.43 V, 0.65 V, and 0.9 V, which are attributed to three one-electron-oxidation processes of HHTP from CatCatCat sequentially to CatCatSq, CatSqSq, and SqSqSq (Fig. 2b).14,28,29 The ionization of the Cu tape starts at around 0.2 V and shows a peak around 0.43 V, indicating the oxidation of Cu in this potential window.18 Following the CV scans we selected a potential of 0.43 V for our subsequent electrochemical MOF growth, which is the minimum potential that enables oxidation of both the Cu and the ligand. We ran the electrochemical cell for 1 hour at 0.43 V, following which a dark film was observed to have grown on both the Cu side and the ITO side of the working electrode.
1). MOF formation requires an appropriate voltage to ionize the Cu and oxidize the ligand on the ITO electrode. The electrochemical behaviour of Cu and HHTP was investigated using cyclic voltammetry (CV) to determine the appropriate potential for electrochemical deposition. Previous studies have reported six one-electron-oxidation processes for HHTP after deprotonation, from fully reduced HHTP6− to fully oxidized HHTP0.26,27 As the CV scans in Fig. 2a show, the HHTP solution shows three anodic peaks at 0.43 V, 0.65 V, and 0.9 V, which are attributed to three one-electron-oxidation processes of HHTP from CatCatCat sequentially to CatCatSq, CatSqSq, and SqSqSq (Fig. 2b).14,28,29 The ionization of the Cu tape starts at around 0.2 V and shows a peak around 0.43 V, indicating the oxidation of Cu in this potential window.18 Following the CV scans we selected a potential of 0.43 V for our subsequent electrochemical MOF growth, which is the minimum potential that enables oxidation of both the Cu and the ligand. We ran the electrochemical cell for 1 hour at 0.43 V, following which a dark film was observed to have grown on both the Cu side and the ITO side of the working electrode.
We then investigated the black film that had grown on the ITO side of the working electrode to confirm that it is the expected Cu3(HHTP)2 MOF. The powder was scraped off for PXRD characterization. As shown in Fig. 3a, peaks at 4.9°, 9.6°, 12.6°, 16.6° and a double peak at 28.2°/28.55° correspond to the (100), (200), (210), (220) and (001) planes of Cu3(HHTP)2, respectively.9 The XRD scan of the electrochemically synthesized Cu3(HHTP)2 powder (Fig. 3a, blue) is consistent with both the solvent synthesized Cu3(HHTP)2 powder (Fig. 3a, grey) and the XRD data reported in the literature.9,10 Detailed insight into the electrochemically synthesized Cu3(HHTP)2 was also obtained using high resolution TEM analysis. The TEM images show two distinct types of ordered image. The image showing layers (Fig. 3b) is of the MOF in the orientation when viewed perpendicular to the c-axis i.e. along the MOF planes. The layer periodicity is measured to be 0.33 nm which corresponds to the (001) reflection in the XRD pattern (0.32 nm periodicity). The hexagonal structure shown in Fig. 3c is of the MOF viewed parallel to the c-axis i.e. perpendicular to the direction of the image in Fig. 3b showing the lattice structure of Cu3(HHTP)2.30 The FFT pattern in the inset of Fig. 3c shows bright spots indicating periodicities ranging between 1.75 nm and 2.03 nm, corresponding to the 4.9° (100) reflection in the XRD pattern that has a 1.80 nm periodicity. The XRD and TEM results confirm the successful synthesis of Cu3(HHTP)2 on the ITO surface using our dual-working-electrode strategy.
3.2. Orientation control of the MOF
Having confirmed the successful formation of Cu3(HHTP)2 using the dual-working-electrode electrochemical synthesis method described above, we varied the ligand concentration in the solution and found that this gives rise to distinct differences in the orientation and morphology of the MOF film on the ITO substrate. We applied the same 0.43 V potential to the working electrode throughout our experiments to ensure consistency. The SEM images of Cu3(HHTP)2 synthesized from a relatively high ligand concentration (2.6 mM HHTP powder in solution) show vertically oriented edge-on crystals (Fig. 4a) with their c-axis parallel to the ITO substrate surface. The Cu3(HHTP)2 film synthesized from a relatively low ligand concentration (1.3 mM HHTP powder in solution) forms face-on crystals with their c-axis perpendicular to the electrode surface (Fig. 4b).PXRD measurements were performed to probe the stacking orientation of both the orientations of the Cu3(HHTP)2 MOF films. As shown in Fig. 5 (blue), the MOF film synthesized with a high ligand concentration exhibits strong diffraction peaks at 4.9°, 9.6°, 12.6°, and 16.6°, corresponding to the (100), (200), (210), and (220) planes, respectively. The presence of these peaks and the absence of a peak around 28° indicate edge-on orientation of the 2D layers i.e. the c-axes of the MOF crystals are parallel to the substrate surface.4 In contrast, the film synthesized with a low ligand concentration has a strong double diffraction peak at 27.28° and 27.86° (Fig. 5 red), corresponding to interlayer stacking periodicities of 0.328 nm and 0.321 nm, repectively.6 This double peak can be indexed to the (001) reflections of Cu3(HHTP)2, confirming the face-on orientation of the 2D layers. The two distinct c-axis periodicities correspond to coexisting slightly different planar stacking arrangements, previously reported as AA (or eclipsed) and AB (or near-eclipsed) stacking.30,31 Our results confirm the formation of highly oriented and crystalline Cu3(HHTP)2 films using our electrochemical synthesis method, with a high ligand concentration promoting an edge-on orientation, and a low ligand concentration resulting in face-on stacking of Cu3(HHTP)2 layers.
XPS measurements were conducted to determine the oxidation state of the elements in the oriented Cu3(HHTP)2 films. As shown in Fig. S1a, the Cu spectrum shows a strong binding energy peak at 934.8 eV, corresponding to Cu2+, along with a weak peak at 932.5 eV, suggesting the presence of Cu+.32 Both edge-on and face-on films show only low amounts of Cu+, likely originating from structural defects, which indicate a well-coordinated crystalline structure.33,34 The C 1s and O 1s spectra indicate the coexistence of C–O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, showing that catechol is partially oxidized to semiquinone in the HHTP ligand during coordination with Cu (Fig. S1b and c).14,35 Our XPS results corroborate the XRD data to further demonstrate that we have grown highly crystalline films of Cu3(HHTP)2.
O bonds, showing that catechol is partially oxidized to semiquinone in the HHTP ligand during coordination with Cu (Fig. S1b and c).14,35 Our XPS results corroborate the XRD data to further demonstrate that we have grown highly crystalline films of Cu3(HHTP)2.
The growth of the oriented Cu3(HHTP)2 films was further investigated by varying the reaction times. In a solution with high ligand concentration, edge-on films with 2 h and 10 h reaction times are shown to have thicknesses of 0.56 µm and 2.5 µm, respectively (Fig. S2a and b). The MOF growth rate for the low ligand concentration solution was significantly faster, achieving 1.5 µm and 9.6 µm thicknesses over the same durations (Fig. S2c and d). As the reaction time is increased the roughness of the film also increases as shown in the AFM images in Fig. S2. It is interesting that the film synthesized from the low ligand concentration, which utilizes fewer reactants, exhibits more rapid growth compared with the film grown from a high ligand concentration. Presumably this is because the face-on orientation is smoother, enabling a Cun(HHTP)m molecule to land on the MOF surface and diffuse laterally to find an attachment site. This type of diffusion is inhibited for the edge-on orientation and this results in slower growth.
The local electrical conductivity of the films was determined through conductive AFM mapping (Fig. S3). This technique measures the local conductivity of the films with high spatial resolution.16,36,37 The local average vertical conductivity through the edge-on film is (6.78 ± 0.45) × 10−2 S cm−1 (Fig. S3e), while for the face-on film it is (2.56 ± 0.97) × 10−5 S cm−1 (Fig. S3f). The in-plane charge transport is more than 103 times higher than the π–π stacking charge transport. These results are consistent with published transport properties in other MOFs where there is also a 2–3 orders of magnitude difference in the conductivity between the in-layer and across-layer orientations.16 It should be noted that our absolute conductivity results are not as reliable as the relative conductivity ratio comparing face-on and edge-on films.
3.3. Growth mechanism of the oriented MOFs
The growth mechanism of the oriented films can be understood by examining the reactions and interactions occurring in solution and on the two sides (Cu tape and ITO slide) of the working electrode. The MOF growth mechanism on the copper side differs from that occurring on the ITO side. On the copper side, at the applied potential of +0.43 V, the Cu is ionised to Cu2+ and the HHTP is ionised from its catechol to semiquinone form. This enables Cu3(HHTP)2 MOF formation on the Cu tape through the coordination between the (HHTP)3− and Cu2+ ions. Face-on oriented films are grown on the Cu tape side of the working electrode no matter the ligand concentration, as shown in the SEM images in Fig. S4.On the ITO side of the working electrode, no direct Cu source is available for MOF formation, and since the same voltage is applied to both the Cu tape and the ITO, there is no potential gradient between these two surfaces. For MOF growth to occur on the ITO electrode concentration-mediated Cu diffusion must occur to this electrode via either solvated Cu ions or Cun(HHTP)m complexes. Two experiments show that it is Cun(HHTP)m complexes that diffuse. The first experiment involves the preparation of separate solutions of Cu(NO3)2 and HHTP in a mixture of water and ethanol. When a drop of Cu(NO3)2 water:ethanol solution is added to an HHTP water:ethanol solution, an immediate blue colour change is observed. Since extended MOF formation is unlikely to occur this rapidly at room temperature without a base to facilitate deprotonation or higher temperatures to promote the reaction, the colour change suggests that intermediate Cun(HHTP)m complexes are formed. These complexes would also form in our solution for oriented growth following ionisation of the Cu electrode. This result further suggests that HHTP coordinates with a larger number of Cu ions in complexes formed under the low ligand concentration condition compared with the high ligand concentration.
The second experiment showing the existence of Cun(HHTP)m complexes involves UV-vis measurements. As shown in Fig. 6, the Cu(NO3)2 solution exhibits a light blue colour, with a broad peak at 810 nm. The ligand itself does not show any peaks in the metal–ligand charge transfer region. The MOF spectrum displays a broad absorption peak at about 664 nm, extending to the near-infrared (NIR) region, which can be attributed to the ligand-to-metal charge transfer transition (LMCT).38,39 After 30 minutes of electrochemically-oriented MOF growth, a solution near the ITO surface was collected, both under high and low ligand concentration conditions. As shown in Fig. 6, the UV-vis spectra of the electrolyte solutions differ from those of HHTP, Cu(NO3)2, and solvothermally synthesized Cu3(HHTP)2, confirming the presence of intermediate Cun(HHTP)m complexes. Compared to Cu3(HHTP)2 MOF, the complex formed in the high ligand concentration solution shows a blue shift in the LMCT band from 664 nm to 642 nm and a reduced NIR intensity. In contrast, the complexes formed in the low ligand concentration solution exhibit a red shift of the LMCT band shifting to 686 nm and an adsorption tail extending into the NIR region. The red shift of the LMCT band observed for complexes formed at low ligand concentration, relative to those formed at high ligand concentration, indicates an increased coordination number for the Cu ions.40 The higher NIR to LMCT peak intensity ratio observed at low ligand concentration suggests more extensive metal–ligand charge transfer and greater conjugation within the resulting complexes.41 Together, these results indicate that under low ligand concentration solution, HHTP coordinates with more Cu ions, resulting in larger complexes with higher Cu coordination number and increased electronic delocalization. The UV-vis results are compelling, but not definitive, because they do not provide direct structural information or coordination number. In addition, the low ligand concentration solution is a darker shade of blue than the high ligand concentration solution (Fig. S5). These results combined suggest that HHTP coordinates with a larger number of Cu ions in complexes formed under the low ligand concentration condition. Overall, the evidence indicates that Cu ions and HHTP molecules form different intermediate complexes under different ligand concentrations.
To gain further insight into the reaction occurring at the ITO side, we examined the importance of the applied voltage. A control experiment was performed where we applied the electrochemical potential only to the Cu surface, and left the ITO surface at ground potential. The formation of Cu3(HHTP)2 was observed on the Cu side, but not on the ITO side, indicating that a charged electrode surface is necessary for MOF growth on the ITO. The formation of MOF on the ITO electrode is therefore an electrochemically driven process between the Cun(HHTP)m complexes, solvated Cu ions, and HHTP ligands.
We now turn to the origin of the mechanism that results in orientated MOF growth. From the experiments reported above we know that Cun(HHTP)m complexes exist in the solution, and that these complexes have different colours and UV-vis signatures depending on the amount of ligand in the solution. The smallest complexes involve only one HHTP molecule with Cu ions bound to the dehydrogenated sites as shown in Fig. 7: Cu(HHTP), Cu2(HHTP), and Cu3(HHTP). We did not model higher Cun(HHTP)m complexes involving more than one HHTP molecule, although these are also likely to be present in the solution. As can be seen in the annotations in Fig. 7, there are in-plane electric dipole moments associated with the Cu(HHTP) and Cu2(HHTP) molecules. The Cu(HHTP) dipole is particularly strong at 1.25 eÅ, and the Cu2(HHTP) dipole is weaker at 0.26 eÅ. These dipole strengths can be compared with that of H2O molecules, which in their liquid state is around 0.6 eÅ, and in the gas phase 0.38 eÅ.42 The DFT calculations were performed using both the PBE and PBE0 functionals, and the same qualitative results were found. For HHTP coordinated with a single Cu ion, the electric dipole moment was 1.25 eÅ within PBE and 1.46 eÅ within PBE0. Coordination to two Cu atoms reduces the electric dipole moment to 0.26 eÅ within PBE and 0.1 eÅ within PBE0, while a fully coordinated HHTP shows no electric dipole moment. The out-of-plane electric dipole moment perpendicular to the CunHHTPm complex plane remains zero in all cases.
On diffusing to the ITO electrode, the Cu(HHTP) and Cu2(HHTP) complex molecules will align their planar dipoles with the electric field that is created by the applied electrochemical potential. This will result in an edge-on orientation for the nucleating MOF film. Fully coordinated Cu3(HHTP) molecules do not have a dipole associated with them and will therefore maximise their van der Waals interaction by attaching face-on to the ITO electrode resulting in a face-on nucleated MOF film. We reason that low ligand concentrations result in mainly fully-coordinated Cu3(HHTP) molecules because all the solvated Cu ions can find an HHTP binding site. Conversely, for high ligand concentrations there are not sufficient numbers of Cu ions to fully coordinate all the ligand binding sites, and this results in undercoordinated Cu(HHTP) and Cu2(HHTP) molecules with strong planar dipoles. The UV-vis spectra in Fig. 6 support this interpretation.
To investigate whether the formation of the oriented films is dominated by crystal nucleation or growth, we performed a control experiment by switching the synthesis conditions for edge-on and face-on films after 3 minutes of synthesis. ITO glass with Cu tape on the opposite side was submerged in an electrolyte solution with a high ligand concentration, and a voltage of 0.43 V was applied for 3 minutes. The combined working electrode was then transferred to a low ligand concentration solution. After 60 further minutes of synthesis, an edge-on film was observed. Conversely, when the electrode was initially placed in a low ligand concentration solution for 3 minutes under 0.43 V, and then transferred to the high ligand concentration condition, after 60 further minutes of synthesis, a face on film was observed. These results show that the film orientation is determined during the initial nucleation stage, and that once a film is oriented in a particular growth direction, then this orientation is maintained even if the growth conditions are subsequently changed to favour an alternative nucleation condition.
4. Conclusions
We have developed a dual-working electrode electrochemical synthesis method to fabricate Cu3(HHTP)2 films, enabling oriented MOF growth. This setup allows the oxidation of the metal and the MOF formation to occur at separate electrode surfaces, enabling highly crystalline and pure MOF films of varying thicknesses to be grown. By tuning the ligand concentration, well-oriented edge-on and face-on Cu3(HHTP)2 films were grown on an ITO electrode surface. Our investigation into the growth mechanism of oriented Cu3(HHTP)2 under different ligand concentration conditions emphasizes the importance of the intermediate Cun(HHTP)m complexes formed before MOF formation. The oriented edge-on film and face-on films exhibit anisotropic conductivity. The local conductivity along the in-plane direction was found ∼103 times higher than across the planes.This study presents a new strategy for electrochemical growth of oriented 2D MOF films on electrodes, providing a deeper understanding of their growth mechanisms. The results provide further opportunities for the design and synthesis of 2D MOF films with highly anisotropic charge transport properties. For device integration it may be necessary to tune the surface roughness of the films, which could be achieved through post-growth processing such as solvent vapour annealing43 or plasma etching.44
Author contributions
M. R. C and Y. W. designed the research. Y. W. performed the synthesis and characterization. A. M. L assisted with MOF powder synthesis and data interpretation. C. E. P conducted the DFT simulations. S. T. and W. L. assisted with data interpretation and manuscript preparation. S. Z. performed the AFM experiment. A. F. conducted the XPS analysis. N. Y. performed TEM image capture and analysis.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the supplementary information (SI) file and on request from the corresponding author. Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc04056f.Acknowledgements
We are grateful for computational support from the UK national high performance computing service, ARCHER2, for which access was obtained via the UKCP consortium and funded by EPSRC Grant ref EP/P022561/1.References
- L. S. Xie, G. Skorupskii and M. Dincă, Chem. Rev., 2020, 120, 8536–8580 CrossRef CAS PubMed.
- J. Liu, X. Song, T. Zhang, S. Liu, H. Wen and L. Chen, Angew. Chem., Int. Ed., 2021, 60, 5612–5624 CrossRef CAS PubMed.
- H. Jeong, G. Park, J. Jeon and S. S. Park, Acc. Chem. Res., 2024, 57, 2336–2346 CrossRef CAS PubMed.
- Z.-Z. Ma, Q.-H. Li, Z. Wang, Z.-G. Gu and J. Zhang, Nat. Commun., 2022, 13, 6347 CrossRef CAS PubMed.
- M.-S. Yao, K.-i Otake, T. Koganezawa, M. Ogasawara, H. Asakawa, M. Tsujimoto, Z.-Q. Xue, Y.-H. Li, N. C. Flanders and P. Wang, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2305125120 Search PubMed.
- T. Hong, C. Lee, Y. Bak, G. Park, H. Lee, S. Kang, T. H. Bae, D. K. Yoon and J. G. Park, Small, 2024, 20, 2309469 CrossRef CAS PubMed.
- J. Liu, Y. Chen, X. Huang, Y. Ren, M. Hambsch, D. Bodesheim, D. Pohl, X. Li, M. Deconinck and B. Zhang, Nat. Synth., 2024, 3, 715–726 CrossRef CAS.
- M. Choe, J. Y. Koo, I. Park, H. Ohtsu, J. H. Shim, H. C. Choi and S. S. Park, J. Am. Chem. Soc., 2022, 144, 16726–16731 CrossRef CAS PubMed.
- M. S. Yao, X. J. Lv, Z. H. Fu, W. H. Li, W. H. Deng, G. D. Wu and G. Xu, Angew. Chem., Int. Ed., 2017, 56, 16510–16514 CrossRef CAS PubMed.
- Y. Liu, Y. Wei, M. Liu, Y. Bai, X. Wang, S. Shang, J. Chen and Y. Liu, Angew. Chem., Int. Ed., 2021, 60, 2887–2891 Search PubMed.
- S. H. R. Shin, J. Tao, N. L. Canfield, M. E. Bowden, J. Heo, D. Li, J. Liu, J. J. De Yoreo, P. K. Thallapally and M. L. Sushko, Adv. Mater. Interfaces, 2023, 10, 2202031 CrossRef CAS.
- R. M. Stolz, A. F. Kolln, B. C. Rocha, A. Brinks, A. M. Eagleton, L. Mendecki, H. Vashisth and K. A. Mirica, ACS Nano, 2022, 16, 13869–13883 CrossRef CAS PubMed.
- A. M. Lister, Y. Wang, B. I. Armitage, W. Li and M. R. Castell, J. Phys. Mater., 2025, 8, 025004 CrossRef CAS.
- M. Song, J. Jia, P. Li, J. Peng, X. Pang, M. Qi, Y. Xu, L. Chen, L. Chi and G. Lu, J. Am. Chem. Soc., 2023, 145, 25570–25578 Search PubMed.
- A. Mähringer, A. C. Jakowetz, J. M. Rotter, B. J. Bohn, J. K. Stolarczyk, J. Feldmann, T. Bein and D. D. Medina, ACS Nano, 2019, 13, 6711–6719 Search PubMed.
- Z. Wang, L. S. Walter, M. Wang, P. S. Petkov, B. Liang, H. Qi, N. N. Nguyen, M. Hambsch, H. Zhong and M. Wang, J. Am. Chem. Soc., 2021, 143, 13624–13632 CrossRef CAS PubMed.
- A. M. Lister, B. I. Armitage, Y. Wang, R. Chen, W. Li and M. R. Castell, ACS Appl. Nano Mater., 2025, 8, 15114–15121 Search PubMed.
- M. de Lourdes Gonzalez-Juarez, E. Flores, M. Martin-Gonzalez, I. Nandhakumar and D. Bradshaw, J. Mater. Chem. A, 2020, 8, 13197–13206 RSC.
- M. Song, Y. Wu, J. Jia, J. Peng, Y. Ren, J. Cheng, Y. Xu, W. Liu, S. Kang and Y. Fang, J. Am. Chem. Soc., 2025, 147, 17058–17067 CrossRef CAS PubMed.
- R. Zheng, Z.-H. Fu, W.-H. Deng, Y. Wen, A.-Q. Wu, X.-L. Ye and G. Xu, Angew. Chem., Int. Ed., 2022, 61, e202212797 CrossRef CAS PubMed.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni and I. Dabo, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- D. Vanderbilt, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 41, 7892–7895 CrossRef PubMed.
- A. Dal Corso, Comput. Mater. Sci., 2014, 95, 337–350 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 Search PubMed.
- C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158–6170 CrossRef CAS.
- C. S. Grange, A. J. Meijer and M. D. Ward, Dalton Trans., 2010, 39, 200–211 RSC.
- L. Yang and M. Dincă, Angew. Chem., Int. Ed., 2021, 60, 23784–23789 CrossRef CAS PubMed.
- X. Du, J. Zhang, H. Wang, Z. Huang, A. Guo, L. Zhao, Y. Niu, X. Li, B. Wu and Y. Liu, Mater. Chem. Front., 2020, 4, 243–251 RSC.
- K. P. Naidek, C. R. Zuconelli, O. M. Cruz, R. Ribeiro, S. M. Winnischofer and H. Winnischofer, Biochem. Cell Biol., 2016, 94, 205–211 CrossRef CAS PubMed.
- R. W. Day, D. K. Bediako, M. Rezaee, L. R. Parent, G. Skorupskii, M. Q. Arguilla, C. H. Hendon, I. Stassen, N. C. Gianneschi and P. Kim, ACS Cent. Sci., 2019, 5, 1959–1964 Search PubMed.
- J. W. Gittins, C. J. Balhatchet, S. M. Fairclough and A. C. Forse, Chem. Sci., 2022, 13, 9210–9219 RSC.
- A. C. Gómez-Herrero, C. Sánchez-Sánchez, F. Chérioux, J. I. Martínez, J. Abad, L. Floreano, A. Verdini, A. Cossaro, E. Mazaleyrat and V. Guisset, Chem. Sci., 2021, 12, 2257–2267 RSC.
- Y. Misumi, A. Yamaguchi, Z. Zhang, T. Matsushita, N. Wada, M. Tsuchiizu and K. Awaga, J. Am. Chem. Soc., 2020, 142, 16513–16517 CrossRef CAS PubMed.
- T. Chen, J.-H. Dou, L. Yang, C. Sun, N. J. Libretto, G. Skorupskii, J. T. Miller and M. Dincă, J. Am. Chem. Soc., 2020, 142, 12367–12373 Search PubMed.
- Y. Chen, Q. Zhu, K. Fan, Y. Gu, M. Sun, Z. Li, C. Zhang, Y. Wu, Q. Wang and S. Xu, Angew. Chem., Int. Ed., 2021, 60, 18769–18776 Search PubMed.
- F. Giannazzo, G. Greco, F. Roccaforte, C. Mahata and M. Lanza, Electrical Atomic Force Microscopy for Nanoelectronics, 2019 Search PubMed.
- E. Osuna, P. Ares, J. Gómez-Herrero, G. Llauradó-Capdevila, D. Rodríguez-San-Miguel, S. Pané, J. Puigmartí-Luis and C. Gómez-Navarro, Small, 2025, 21, 2407945 CrossRef CAS PubMed.
- Q. Zuo, T. Liu, C. Chen, Y. Ji, X. Gong, Y. Mai and Y. Zhou, Angew. Chem., Int. Ed., 2019, 58, 10198–10203 CrossRef CAS PubMed.
- Y.-M. Jo, K. Lim, J. W. Yoon, Y. K. Jo, Y. K. Moon, H. W. Jang and J.-H. Lee, ACS Cent. Sci., 2021, 7, 1176–1182 Search PubMed.
- M. J. Sever and J. J. Wilker, Dalton Trans., 2004, 1061–1072 RSC.
- J. Wang, T. Chen, M. Jeon, J. J. Oppenheim, B. Tan, J. Kim and M. Dincă, J. Am. Chem. Soc., 2024, 146, 20500–20507 Search PubMed.
- C. Adamo, M. Cossi, G. Scalmani and V. Barone, Chem. Phys. Lett., 1999, 307, 265–271 CrossRef CAS.
- S. Iwamoto, R. Nakayama, S. Chon, R. Shimizu and T. Hitosugi, J. Mater. Chem. A, 2024, 12, 17492–17500 RSC.
- P. V. Alekseevskiy, M. Timofeeva, S. Bachinin, R. Peignier, C. Noel, P. Boulet, T. Belmonte and V. A. Milichko, Opt. Mater., 2024, 154, 115666 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |