 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Spin-polarized carriers and ferromagnetism induced by Pt incorporation in ZnO: an experimental and DFT assessment
Subhash
Sharma
 *ab,
Gabriel
Rojas-George
c,
C. A.
Corona-García
*ab,
Gabriel
Rojas-George
c,
C. A.
Corona-García
 b,
Sunil
Chauhan
b,
Sunil
Chauhan
 d,
Santosh
Kumar
d,
Santosh
Kumar
 e,
Rajesh
Kumar
e,
Rajesh
Kumar
 e,
Javier Alonso Lopez
Medina
e,
Javier Alonso Lopez
Medina
 a,
Rodrigo Ponce
Perez
a,
Rodrigo Ponce
Perez
 b,
O. Raymond
Herrera
b,
O. Raymond
Herrera
 b and
J. Guerrero
Sanchez
b and
J. Guerrero
Sanchez
 *b
*b
aSECIHTI – IxM – Centro de Nanociencias y Nanotecnología, Universidad Nacional Autónoma de México, Km 107 Carretera Tijuana-Ensenada, AP 14, Ensenada 22800, B.C., México. E-mail: subhash@ens.cnyn.unam.mx
bCentro de Nanociencias y Nanotecnología, Universidad Nacional Autónoma de, México, Km 107 Carretera Tijuana-Ensenada, AP 14, Ensenada 22800, B.C., México. E-mail: guerrero@ens.cnyn.unam.mx
cSECIHTI – IxM, Centro de Investigación en Materiales Avanzados, S.C., Miguel de Cervantes 120, Chihuahua, Chih 31136, México
dDepartment of Physics & Environmental Sciences, Sharda School of Engineering & Science, Sharda University, Uttar Pradesh 201310, India
eDepartment of Chemistry, Himachal Pradesh University, Summer Hill, Shimla-5, Himachal Pradesh 171005, India
First published on 27th January 2026
Abstract
In the present work, we report the synthesis and detailed investigation on the structural, microstructural, and magnetic properties of ZnO–xPt (x = 0.0, 0.02, 0.06, 0.10 wt%) nanocomposites, using experimental and theoretical approaches. The structural analysis, using Rietveld-refined X-ray diffraction patterns, shows that all samples exhibit a wurtzite phase with a hexagonal structure and a P63mc symmetry. Transmission electron microscopy analysis revealed a consistent morphology for different Pt percentages, with nanoparticle diameters consistently less than 100 nm. Room-temperature magnetic measurements revealed superparamagnetic behavior, with the saturation magnetization increasing with the Pt percentage. However, the loops exhibited open regions in the low magnetic field range, where the remanent magnetization and coercivity also increased with the Pt weight percentage. The magnetic behavior is partly attributed to the bound magnetic polaron mechanism, and Thamm–Hesse analysis ruled out significant interparticle interactions, suggesting that clustering is not the dominant contribution. Density functional theory calculations indicate that Pt incorporation at Zn sites is energetically stable, regardless of the presence of oxygen vacancies. Spin-density isosurfaces reveal localized magnetic moments around Pt and adjacent oxygen atoms. The projected density of states exhibits a transition from semiconducting to half-metal behavior, producing spin-polarized carriers that can mediate long-range magnetic interactions. The close agreement between experimental observations and theoretical predictions highlights low-concentration Pt-doped ZnO as a promising nanoscale material for spintronic applications.
Introduction
Zinc oxide (ZnO) is well-known as a wide-bandgap semiconductor with a bandgap of 3.27–3.37 eV, characterized by a large exciton binding energy of 60 meV, which makes it a suitable material for application in optoelectronics, sensors, and ultraviolet light emitters.1–8 The progress in diluted magnetic semiconductors (DMSs), which combine magnetic ordering with semiconducting behavior, is the center of attraction for spintronic devices. DMSs are a class of materials in which transition metals are introduced into the cationic sites to tailor their physical properties, and they are receiving significant attention due to their potential for application in spintronic devices.9–11 Spintronics devices offer various benefits over traditional electronic devices, utilizing spin carriers to transport, store, and process information, instead of electrons as the primary charge carriers. However, in DMS-based devices, achieving long spin lifetimes and efficient spin injection and detection at room temperature is very challenging. It is a well-established fact that doping strategies are an effective way to alter physical properties, and in the case of ZnO, transition metal substitution has been explored to induce ferromagnetism in this material. Among these dopants, a noble metal (Pt) has emerged as a promising candidate due to its unique electronic configuration and potential to alter the carrier concentration and defects within the ZnO matrix.12–15 Therefore, Pt addition on the ZnO surfaces is expected to induce weak ferromagnetic ordering, which is crucial for the development of spin-based devices. The weak ferromagnetic ordering in ZnO-based materials is attributed to the interaction of localized magnetic moments with conduction electrons, facilitated by lattice distortions and defect states induced by dopants.13 Pt doping has also a center of attraction in recent times due to its sensing and photocatalytic properties; however, there are fewer reports in this direction, specifically regarding its magnetic properties.15–17 The promising magnetic ordering at room temperature observed in Pt–ZnO nanomaterials opens new horizons for next-generation spintronic devices based on ZnO. Therefore, in the present work, we investigate these properties comprehensively, using the sol–gel route to prepare ZnO–xPt nanocomposites for different values of x. The detailed structural analysis using X-ray diffraction (XRD), including Rietveld refinements, was performed to determine the correct lattice parameters. Magnetic properties are also discussed in detail for all studied samples. Furthermore, density functional theory (DFT) calculations were employed to understand the observed magnetic properties in the studied samples, revealing how the dopant-induced structural distortion influences the electronic band and, consequently, the magnetic behavior.18–20Experimental and characterization section
The nanocomposites in the ZnO–xPt system with x = 0.0, 0.02, 0.06, and 0.10 wt% (samples labeled as ZnO, Pt2, Pt6, and Pt10, respectively) were prepared by a simple, low-cost sol–gel route. The precursors, Zn(NO3)2·6H2O (Merck, >99%), a 3 nm Pt nanoparticle dispersion in water at 1000 ppm (Sigma-Aldrich, 99.99%), and citric acid as a complexing agent (C6H8O7·H2O, Merck, >99%), were used. An appropriate amount of Zn nitrate was dissolved in deionized water and stirred for 20–25 minutes on a magnetic stirrer, followed by the addition of an appropriate amount of Pt-sol. The citric acid was added in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of metal ion to citrate and kept at 70 °C on a hot plate with magnetic stirring for 2 hours. Then, a gel was obtained after 2 hours, followed by heating at ∼110 °C in an oven for 12 hours to form a dry gel. Finally, the dry gel was ground to make a fine powder and subjected to calcination at 525 °C to promote the crystalline phase. An XRD diffractometer (Cu Kα radiation, λ = 1.5406 Å) was used to determine the crystal phase of the samples under study. Rietveld analysis was performed using FullProf software to determine the lattice parameters. The morphology was studied using scanning transmission electron microscopy (STEM), employing a Hitachi HT7700 microscope (EXALENS System) with operating voltages ranging from 40 to 120 kV and a maximum spatial resolution of 0.14 nm. A physical property measurement system (PPMS) with a VMS probe of Quantum Design was used to study the room-temperature (300 K) magnetic properties.
1 molar ratio of metal ion to citrate and kept at 70 °C on a hot plate with magnetic stirring for 2 hours. Then, a gel was obtained after 2 hours, followed by heating at ∼110 °C in an oven for 12 hours to form a dry gel. Finally, the dry gel was ground to make a fine powder and subjected to calcination at 525 °C to promote the crystalline phase. An XRD diffractometer (Cu Kα radiation, λ = 1.5406 Å) was used to determine the crystal phase of the samples under study. Rietveld analysis was performed using FullProf software to determine the lattice parameters. The morphology was studied using scanning transmission electron microscopy (STEM), employing a Hitachi HT7700 microscope (EXALENS System) with operating voltages ranging from 40 to 120 kV and a maximum spatial resolution of 0.14 nm. A physical property measurement system (PPMS) with a VMS probe of Quantum Design was used to study the room-temperature (300 K) magnetic properties.
Computational methods
Spin-polarized total energy calculations within the framework of density functional theory were carried out using the Vienna ab initio simulation package (VASP).21–24 Exchange and correlation effects were treated according to the generalized gradient approximation (GGA) with the Perdew–Burke–Ernzerhof (PBE) parametrization.25 A plane-wave basis set with a cut-off energy of 400 eV was adopted to represent the electronic wavefunctions. The projector augmented wave (PAW) method was employed to account for the frozen-core approximation.26,27 It is well-known that standard GGA and LDA approaches significantly underestimate the ZnO bandgap and its magnetic properties when doped with magnetic atoms.26–30 Due to the highly localized nature of Zn 3d electrons, various functionals and correction methods have been proposed in recent studies to improve the accuracy of ZnO property descriptions. These include GGA+U,31–33 LDA+U,34 and hybrid functionals such as PBE0,35 HSE,36 and HSE06.30 Among these approximations, the Hubbard correction is the most widely used due to its precision in describing magnetic properties and its lower computational cost compared to hybrid functionals or HSE approximations.37–39 In this work, we adopted the DFT+U approach, applying the Hubbard correction as formulated by Dudarev et al.,40 with a U parameter of 6 eV for Zn atoms, consistent with previous reports.41–44 For Pt atoms, although they also present localized 5d electrons, previous DFT reports have shown that standard DFT calculations give excellent results.43,44 The inclusion of the Hubbard correction does not significantly affect the magnetic properties of the Pt atoms. Additionally, our calculations confirm that the magnetic moment in Pt-doped ZnO arises primarily from the Pt atoms themselves, which induce a small magnetization in the neighboring O atoms even without applying the Hubbard correction on Pt. Thus, introducing a Hubbard correction on Pt atoms would not qualitatively change the magnetic behavior or the conclusions of this study. Structural optimization was achieved when the residual force on each atom was less than 0.01 eV Å−1, and the total energy difference converged below 10−4 eV. Brillouin zone sampling was performed using a 3 × 3 × 2 Monkhorst–Pack k-point mesh.45 To enhance the reliability of the simulations, the surface was modeled using the supercell method. A 3 × 3 × 3 periodic ideal hexagonal wurtzite supercell was employed to explore the effects on the magnetic properties of pristine ZnO, Zn and O vacancies, and Pt doping at Zn and O substitutional sites, as well as at interstitial positions. In addition to single Pt substitutions, a configuration with two Pt atoms substituting two Zn atoms was also considered. This configuration enables us to investigate the impact of Pt concentration on the magnetic moment. Increasing the number of Pt atoms beyond one would lead to a large number of possible configurations, making such an analysis beyond the scope of this work. Therefore, only one representative configuration with two Pt atoms was selected to illustrate the experimentally observed trend in magnetization.Defect formation energy formalism
To investigate the thermodynamic stability of Pt incorporation into the ZnO matrix, the defect formation energy (DFE) formalism was employed. This approach, which is independent of the total number of atoms in the system, depends only on the chemical potentials (µi) of the constituent species. The formalism used here is adapted from previous studies42,46,47 to suit the characteristics of our system. It is essential to assume thermodynamic equilibrium between the ZnO bulk and its constituents to apply the DFE formalism. This implies:| μbulkZn + μmoleculeO − ΔHZnOf = μbulkZnO = μZn + μO | (1) |
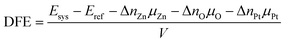 | (2) |
Results and discussion
Structural properties
X-ray diffraction measurements were performed to determine the crystal structure and calculate the lattice parameters. All studied samples, after being calcined at 550 °C, were subjected to XRD analysis. Fig. 1a shows the XRD patterns, which clearly indicate the hexagonal wurtzite structure belonging to space group P63mc as demonstrated by the observation of all standard planes [i.e., (100), (002), (101), (102), (110), (103), (200), (112), and (201)] as per the JCPDS 079-2205 card.4,9 It is worth noting that all samples exhibit a pure phase, with no other phases present. Furthermore, to calculate the lattice parameters, the Rietveld method was employed, as shown in Fig. 1b. The fitting parameters (i.e., χ2, Rp, and Rwp) are in an acceptable range, as shown in Table 1. | ||
| Fig. 1 (a) X-ray diffraction patterns and (b) Rietveld refined patterns for all ZnO–xPt samples with x = 0.00 (ZnO sample), 0.02 (Pt2), 0.06 (Pt6), and 0.10 (Pt10) wt%. | ||
| wt% of Pt | a (Å) | c (Å) | V C (Å3) | Rietveld-factors | D (nm) | R | η × 10−3 |
|---|---|---|---|---|---|---|---|
| 0.00 | 3.2511 | 5.2099 | 47.6882 | R p = 6.96, | 35 | 1.0190 | 1.52 |
| R wp = 9.12, | |||||||
| R exp = 5.33, | |||||||
| χ 2 = 2.93 | |||||||
| 0.02 | 3.2509 | 5.2094 | 47.6800 | R p = 6.70, | 33 | 1.0190 | 1.30 |
| R wp = 8.55, | |||||||
| R exp = 5.37, | |||||||
| χ 2 = 2.53 | |||||||
| 0.06 | 3.2512 | 5.2105 | 47.6987 | R p = 6.92, | 34 | 1.0189 | 1.10 |
| R wp = 8.86, | |||||||
| R exp = 5.39, | |||||||
| χ 2 = 2.70 | |||||||
| 0.10 | 3.2516 | 5.2108 | 47.7112 | R p = 7.25, | 25 | 1.0190 | 1.10 |
| R wp = 8.93, | |||||||
| R exp = 5.41, | |||||||
| χ 2 = 2.72 | |||||||
The lattice parameters and fitting parameters are listed in Table 1. It is found that the lattice parameters have no significant changes, as expected, since the Pt concentration is very low and the radius of the Pt2+ ions (0.60Å) is nearly the same as that of the Zn2+ ions (0.60Å) in four-coordination.50 The average crystallite size was calculated for all samples from the XRD peak broadening using the Scherrer formula:42,46
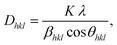 | (3) |
To determine the strain in the samples, the Williams–Hall (W–H) approach was used as per the following relation:51
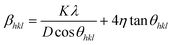 | (4) |
 | (5) |
In this approach, the W–H method defines the two contributions to the broadening of the diffraction peak βhkl: the first one (βsize) comes from crystallite size (i.e., Scherrer formula), and the other one (βstrain = 4η![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tan
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θhkl) corresponds to microstrains due to crystal imperfections and distortions. Eqn (4) represents the uniform deformation model (UDM), assuming that the strain is uniform in all crystallographic directions.49 Therefore, the plots of βhkl
θhkl) corresponds to microstrains due to crystal imperfections and distortions. Eqn (4) represents the uniform deformation model (UDM), assuming that the strain is uniform in all crystallographic directions.49 Therefore, the plots of βhkl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θhklvs. 4
θhklvs. 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θhkl have been graphed for each sample and are shown in Fig. 2. Using the linear fitting in the plots, the η and D values have been determined from the slope and the y-intercepts, respectively. The corresponding values are tabulated in Table 1. It is found that there is a decrease in strain, which may be attributed to the role of Pt incorporation in ZnO. The strain detected in the W–H analysis supports the presence of local lattice distortions, which, according to our DFT calculations (see the theoretical results section), play a central role in stabilizing the magnetic states upon Pt incorporation near oxygen vacancies. The crystal distortion induced by doping can be calculated as per the relation:
θhkl have been graphed for each sample and are shown in Fig. 2. Using the linear fitting in the plots, the η and D values have been determined from the slope and the y-intercepts, respectively. The corresponding values are tabulated in Table 1. It is found that there is a decrease in strain, which may be attributed to the role of Pt incorporation in ZnO. The strain detected in the W–H analysis supports the presence of local lattice distortions, which, according to our DFT calculations (see the theoretical results section), play a central role in stabilizing the magnetic states upon Pt incorporation near oxygen vacancies. The crystal distortion induced by doping can be calculated as per the relation: 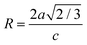 , where a and c are the lattice constants. Thus, R describes the nature of the crystal structure, i.e., indicates if the crystal structure is ideal or distorted, depending on the values of R (R = 1 for an ideal wurtzite structure). In the present case, the value of R is very close to 1, indicating a nearly undistorted structure expected when the Pt content is very low.
, where a and c are the lattice constants. Thus, R describes the nature of the crystal structure, i.e., indicates if the crystal structure is ideal or distorted, depending on the values of R (R = 1 for an ideal wurtzite structure). In the present case, the value of R is very close to 1, indicating a nearly undistorted structure expected when the Pt content is very low.
 | ||
| Fig. 2 The Williamson–Hall (W–H) plots for all ZnO–xPt samples with x = 0.00 (ZnO sample), 0.02 (Pt2), 0.06 (Pt6), and 0.10 (Pt10) wt%. | ||
The nanostructures exhibit a high degree of morphological uniformity, characterized by spherical and monodispersed particles across all Pt concentrations (Fig. 3(a–d)). This consistent morphology, irrespective of the Pt content, suggests that the synthesis process was highly controlled and reproducible. In support of this, the results of particle size distribution analysis are shown in Fig. 3(a–d), corresponding to ZnO–xPt particles with 0.00, 0.02, 0.06, and 0.10 wt% of Pt; the measured average particle sizes were 35.60 nm, 35.03 nm, 44.58 nm, and 44.00 nm, respectively. These values indicate relatively minor variation in particle size among samples with a low to moderate Pt content, highlighting the robustness of the synthesis procedure. The notable size reduction observed at 10 wt% of Pt may suggest a threshold concentration beyond which Pt influences nucleation dynamics. Particle sizes were statistically evaluated using ImageJ software, analyzing over 30 particles per sample. The size distributions were fitted to a Gaussian function, confirming a narrow dispersion and average diameters of less than 90 nm in most cases. This level of morphological precision is critical for applications such as photocatalysis and environmental remediation, where particle size and uniformity directly influence functional performance. These results demonstrate that the adopted synthetic strategy enables fine control over nanoparticle morphology and size, ensuring reproducibility and scalability for practical nanocomposite-based applications.
 | ||
| Fig. 3 Transmission electron microscopy micrographs and size distribution of ZnO–xPt nanocomposites synthesized with varying Pt wt% of (a) 0, (b) 0.02, (c) 0.06, and (d) 0.10. | ||
Magnetic properties
To investigate the magnetic properties, VSM measurements were performed at RT with applied magnetic fields in the range of +20 kOe to −20 kOe. Fig. 4a shows the RT magnetization curves as a function of the applied magnetic field (M–H curve) for all studied samples. As expected for ZnO (curve in the upper inset of Fig. 4a), a diamagnetic behavior is observed as reported by other researchers10; nevertheless, it exhibits a paramagnetic-like behavior close to zero magnetic field. Surprisingly, the ZnO–xPt samples show RT superparamagnetic behavior ascribed to single-domain ferromagnetic nanoparticles with a critical size.52,53 Moreover, the magnetic saturation enhances with increasing concentration of Pt in the ZnO matrix. The bottom inset in Fig. 4a illustrates an opening of the M–H loop at the low magnetic field, suggesting a nonzero coercivity. The samples for x = 0.06 and 0.10 wt% show a saturated M–H curve, indicating that all magnetic moments are aligned at higher values of the magnetic field. The remanent magnetization is found to increase with increasing Pt content. In diluted magnetic semiconductors (DMS materials), the RT ferromagnetism remains poorly understood due to an incomplete understanding of whether it is an extrinsic effect resulting from direct interaction between the local moments in the magnetic impurity or an intrinsic property caused by exchange coupling between the spin of carriers and local moments.42,46 The enhancement of ferromagnetism with Pt incorporation, together with the opening of the M–H loops, suggests that spin-polarized carriers mediate long-range interactions between localized Pt moments, as observed in N-doped ZnO.54 Thamm–Hesse analysis was conducted to determine the type of such magnetic interactions between nanoparticles.The Thamm–Hesse method (THM) is based on analyzing the difference ΔM between the initial magnetization curve and the average of the upper and lower branches of the hysteresis loop for systems composed of single-domain particles.44–46 In this approach, the M–H curve is segmented into five regions of the applied magnetic field: (I) from 0 → Hmax (initial magnetization curve); (II) from Hmax → 0 (positive descending branch); (III) from 0 → −Hmax (excluded); (IV) from −Hmax → 0 (excluded); and (V) from 0 → Hmax (ascending branch). Only segments I, II, and V are considered for the analysis. Thus, the deviation ΔM from ideal behavior is calculated using the following expression:
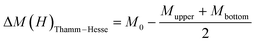 | (6) |
Fig. 4b presents the ΔMThamm–Hesse plots for ZnO–xPt nanocomposites synthesized with x = 0.02 and 0.10 wt%, as a function of the applied magnetic field. The nearly zero ΔM values observed across the applied magnetic field range for both Pt concentrations indicate minimal interaction between magnetic domains, consistent with isolated single-domain behavior. Thus, the values of ΔM ≈ 0 confirm the superparamagnetic behavior, observed in the M–H curves, characteristic of the single-domain non-interacting particles. Additionally, a slight negative deviation is observed at low magnetic fields, more pronounced for 10 wt% Pt, indicating the presence of dipolar interactions that promote magnetization reversal, possibly due to the closer proximity of magnetic Pt domains or a higher density of magnetic centers.55–57 All this suggests that ZnO–xPt nanoparticles are magnetically isolated, and the magnetic interactions between them are negligible.
On the other hand, several theories have been proposed over time by various researchers, especially to explain the origin of the observed magnetic behavior of DMS materials, including carrier-mediated exchange interactions (RKKY), bound magnetic polarons (BMP) linked with oxygen-vacancy trapped carriers, double-exchange or super exchange between transition-metal ions, and the defect-induced magnetism (DIM) arising from intrinsic vacancies or non-stoichiometry.1–8 Collectively, these mechanisms account for the diverse magnetic responses, ranging from weak ferromagnetism to superparamagnetic, commonly reported in doped and defect-engineered ZnO.1–8 Notably, it is well-known that ZnO-based materials are characterized by the presence of oxygen vacancies, which lead to the formation of free localized electrons. Thus, a possible mechanism to explain the observed superparamagnetic behavior coming from ferromagnetic nanoparticles with a critical size is to consider the BMP model. The BMP model suggests that magnetic moments from dopant ions become aligned when they are trapped around localized charge carriers (usually electrons bound to oxygen vacancies). When several such defect–carrier complexes overlap, they form percolating magnetic clusters, giving rise to long-range ferromagnetism in dilute magnetic oxides.11
Therefore, we carried out the theoretical fit for the experimental M–H data to the following equation:9,10
| M = MoL(x) + χmH | (7) |
In eqn (7), the first term denotes the BMP contribution, and the second term is due to the observed paramagnetic part at a higher field; Mo = N × ms, where N is the number of BMP involved, and ms denotes the effective spontaneous moment per BMP. L(x) = coth(x) − 1/x denotes the Langevin function with x = meffH/(kBT), where meff is the true magnetic spontaneous moment per BMP; at higher temperatures, the interactions between BMP can be avoided, and in that situation, ms = meff. χm is the susceptibility of the BMP matrix. In the fitting process, Mo, meff, and χm are variables. As shown in Fig. 5(a–c), the initial M–H data are in good agreement with the BMP model. The extracted parameters are tabulated in Table 2. It is found that the number of BMPs is 5.25 × 1014, 1.58 × 1015, and 1.55 × 1016 per cm3 for the Pt2, Pt6, and Pt10 samples, respectively. It is well-known that these ranges (i.e., 1014–1016 per cm3) are not good enough for making a contribution to magnetism. The required concentration for significant percolation of BMPs is in the range of 1020 per cm3.58 Therefore, it is not fully manifested by BMP-mediated magnetism.
 | ||
| Fig. 5 BMP model fit of magnetization for values of Pt wt% of (a) x = 0.02 (Pt2), (b) x = 0.06 (Pt6), and (c) x = 0.10 (Pt10). (d) Fit of the M–H data using eqn (8) for x = 0.06 (Pt6), with the deconvoluted contributions of the paramagnetic (upper inset) and ferromagnetic (bottom inset) behaviors. | ||
| Pt wt% | BMP model | Experimental parameters | ||||
|---|---|---|---|---|---|---|
| M o (10−3) (emu g−1) | m eff (10−18) (emu) | N (cm−3) | χ (10−7) (cgs) | M r (10−4) (emu g−1) | H c (Oe) | |
| x = 0.02 | 5.39 | 1.83 | 5.25 × 1014 | 7.20 | 3.74 | 137 |
| x = 0.06 | 23.63 | 2.66 | 1.58 × 1015 | 3.97 | 16.4 | 113 |
| x = 0.10 | 33.4 | 0.383 | 1.55 × 1016 | 3.00 | 26.4 | 123 |
To explain the possible magnetic behavior in more depth, we carried out DFT calculations and have discussed the results in the following section. Furthermore, we extracted the paramagnetic (PM) and ferromagnetic (FM) contributions from the M–H curve using a fitting function by the following equation59:
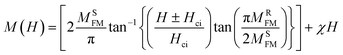 | (8) |
Here, χ is the magnetic susceptibility of the PM contribution. In eqn (8), the first term on the right-hand side, MSFM, denotes the ferromagnetic saturation magnetization, MRFM is the remnant magnetization, and Hc is the intrinsic coercivity. Fig. 5d shows the fitted M–H data to eqn (8) with PM and FM contributions for the Pt6 sample. It is observed from the fitting and the difference between experimental and theoretical parameters that the observed magnetic properties are purely FM in nature due to very little contribution from PM.
Theoretical results
The calculated lattice parameters of pristine ZnO are a = 3.20 Å and c = 5.16 Å, with a Zn–O bond length of 1.95 Å. These values are consistent with previous reports and our experimental results.25,38,49,60 For reference, the ZnO-(3 × 3 × 3) supercell and its projected density of states (PDOS) are shown in Fig. 6. According to the PDOS, ZnO does not exhibit magnetic properties since the spin-up and spin-down states are symmetric, indicating a magnetic null moment, and a bandgap of approximately 1.58 eV, in good agreement with previous reports.38 The valence band is mainly composed of O-2p orbitals, while the Zn-3d orbitals also contribute. In the conduction band, both elements contribute equally. For Pt atoms, although they also present localized 5d electrons, previous DFT reports have shown that standard DFT calculations give excellent results.43,44We employed the DFE formalism to analyze the thermodynamic stability of ZnO doped with Pt atoms under different chemical potential conditions. Four doping configurations were considered, as shown in Fig. 7: (a) Pt substituting a Zn atom, (b) Pt substituting an O atom, (c) Pt replacing a Zn atom with an adjacent O vacancy, and (d) Pt occupying an interstitial site. Fig. 7(f) displays the 2D DFE plot under O-poor to O-rich conditions. The energy reference corresponds to the pristine ZnO-(3 × 3 × 3) system.
According to the DFE formalism, the most thermodynamically favorable (i.e., experimentally probable) configurations are those with the lowest DFE values. Negative DFE values indicate spontaneous reactions, while positive values denote non-spontaneous processes. Our results show that the most stable structure for all chemical potential ranges is the two Zn substitution with an O vacancy (purple line), followed by the O substitution (red line) under O-poor to O-intermediate conditions, whereas the one Zn substitution with an O vacancy becomes stable under O-intermediate to O-rich conditions (blue line), demonstrating that the doping substitution with and without O vacancies is the most probable experimentally. Our results show that the most stable structure under O-poor to O-intermediate conditions is the O substitution (red line), whereas the Zn substitution becomes stable under O-intermediate to O-rich conditions (blue line). Interestingly, under intermediate conditions, a triple point involving O substitution, Zn substitution, and Zn substitution with an O vacancy is observed (green line), as shown in Fig. 7e. Additionally, the interstitial Pt configuration is more stable than the pristine system. These findings are consistent with previous theoretical studies suggesting that ZnO structures with vacancies are more stable than the pristine material, making it feasible to introduce doping atoms into the ZnO matrix.43,61 In all cases, the energy differences are less than 0.004 eV Å−3, suggesting that all these configurations are experimentally feasible.
Furthermore, we calculated the Bader charge transfer to evaluate the interactions among the atomic species. In bare ZnO, Zn atoms donate 1.18e, while O atoms gain −1.18e, confirming that Zn acts as a donor and O as an acceptor. In the O substitution system, Pt has a charge transfer of −0.74e, acting as an acceptor or anion. In the Zn substitution system, Pt donates 0.70e, acting as a donor or cation. In the interstitial system, Pt shows a negligible charge transfer of −0.06e; in the Zn substitution with an O vacancy with one and two Pt atoms, Pt shows a transfer of 0.07e. In the last two cases, the minimal charge transfer suggests that Pt remains in its metallic state.
According to our experimental results, Pt atoms induce magnetism in the ZnO matrix. To investigate the origin of the magnetic properties in Pt-doped systems, we calculated the spin-density isosurfaces (Fig. 8) and the PDOS (Fig. 9) exclusively for the Zn substitution and for the O vacancy configuration with one and two Pt atoms, as these are the only systems that exhibit a significant and localized magnetic moment. The negligible interparticle interaction observed in the THM analysis is consistent with our localized magnetization at the Pt atom and nearby O, rather than delocalized on the ZnO lattice.
 | ||
| Fig. 8 Spin-density isosurfaces for the Zn substitution configurations: (a) a Pt atom without an O vacancy, (b) a Pt atom with an O vacancy, and (c) two Pt atoms. | ||
 | ||
| Fig. 9 Orbital-resolved densities of states for the Zn substitution configurations: (a) without an O vacancy and (b) with an O vacancy. | ||
The spin-density isosurfaces reveal that the Pt atom exhibits local magnetization in all configurations, with nearby oxygen atoms showing weakly induced magnetization. The total magnetic moments are as follows: 1.87µB for one Zn substitution, 3.75µB for two Zn substitutions, 0.12µB for O substitution, 0.23µB for interstitial Pt, and 1.84µB for Zn substitution with an O vacancy. This trend in total magnetization aligns well with the observed increase in magnetic saturation as the Pt content increases in the ZnO matrix. The orbital-resolved PDOS for all Zn substitutional systems confirms the emergence of ferromagnetism and a transition from semiconducting to half-metallic behavior. The spin-up states near the Fermi level are mainly associated with Pt atoms, while impurity states at the Fermi level for the spin-down channel originate primarily from the Pt-d and -p orbitals, which are responsible for the half-metallic character of Pt-doped ZnO (see Fig. 9). Additionally, the O-p orbitals of neighboring O atoms contribute to the magnetic behavior through induced spin polarization caused by their proximity to Pt, representing the third most relevant contribution to the magnetic transition. In contrast, Zn atoms have a negligible effect on magnetization, with their electronic states lying predominantly at negative energy levels. The same tendency is found for the substitution of two Zn atoms with one O vacancy.
Combining all the experimental and theoretical evidence, we see that magnetism in ZnO cannot be explained by BMP contributions or clustering effects. While BMP fitting accounts only for part of the magnetic response, the estimated polaron density is insufficient to explain the observed magnetism. Additionally, THM analysis confirms small or negligible particle interactions, eliminating the possibility of clustering formation, which is also supported by the XRD measurements showing no extra phases. The enhancement of ferromagnetism with Pt doping points to other mechanisms. Our theoretical calculations helped us understand this behavior, revealing that Pt substitution at Zn sites drives a semiconductor to a half-metal transition, with spin asymmetry at the Fermi level. This electronic character partially explains the magnetism observed in the experimental loops, as spin-polarized carriers mediate the long-range interactions between the localized Pt atom moments, possibly giving rise to the enhanced magnetism observed at higher Pt concentrations.
Conclusions
Here, we successfully synthesized Pt-doped ZnO nanomaterials using the sol–gel method and systematically studied their structural, microstructural, and magnetic properties, supported by density functional theory calculations. XRD with Rietveld analyses confirmed that all samples have a wurtzite structure with uniform particle morphology. Additionally, Williamson–Hall analysis indicated local lattice distortions caused by Pt incorporation. Magnetic measurements revealed superparamagnetic behavior at room temperature, characterized by a consistent increase in remnant magnetization and loop saturation as the Pt concentration increased. The Bound Magnetic Polaron model explains part of the magnetic response, while the Thamm–Hesse method excluded significant interparticle interactions, suggesting that clustering is not a dominant mechanism. Our theoretical results offer a detailed atomic-scale explanation of these findings. The stability analysis revealed that Pt incorporation remains stable in Zn sites, regardless of the presence of O vacancies. Spin density isosurfaces displayed localized magnetic moments around the Pt and neighboring O atoms, and the projected density of states confirmed the transition from semiconductor to half-metal. These electronic properties show clear spin-polarized carriers, which could mediate long-range interactions between localized Pt moments. Finally, the experimental results align with the theoretical predictions, showing an increase in magnetization as Pt concentration increases, making low-concentration Pt-doped ZnO a promising candidate for spintronics devices with tailored properties at the nanoscale.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All the data in this study are within the paper.Acknowledgements
This work was partially supported by DGAPA-UNAM, through research Grants DGAPA-PAPIIT IT100525, IA100226, IN110424, and IG101124. Calculations were performed in the DGCTIC-UNAM Supercomputing Center via projects LANCAD-UNAM-DGTIC-452, 150, 286, 368, 422, and 457. The authors would like to thank Pedro Casillas, Luis Alejandro Arce Saldaña, Aldo Rodriguez, Luis Angel De Leon Alanis, and Samuel Flores Garcia, for their valuable technical support. Subhash Sharma and Javier Alonso Lxopez Medina acknowledge support from the SECIHTI Program via the Researchers for Mexico initiative through Projects 352-2018 and 146–2016, respectively. C. A. Corona-García thanks DGAPA-UNAM for the postdoctoral grant.References
- Ü. Özgür, et al., A comprehensive review of ZnO materials and devices, J. Appl. Phys., 2005, 98, 041301 CrossRef.
- D. C. Look, Recent advances in ZnO materials and devices, Mater. Sci. Eng. B, 2001, 80, 383–387 Search PubMed.
- I. Jabbar, et al., Diluted magnetic semiconductor properties in TM-doped ZnO nanoparticles, RSC Adv., 2022, 12, 13456–13463 RSC.
- A. Sukumaran and N. Gopalakrishnan, Donor-defect-induced ferromagnetism in Nb-doped ZnO thin films grown by RF magnetron sputtering, Phys. Status Solidi A, 2024, 221, 2400219 CrossRef CAS.
- M. Mohamed, A. Sedky and M. S. Alshammari, An investigation on structural, optical, and magnetic properties of Zn1−xCoxO nanorods fabricated by electrochemical deposition, Sci. Rep., 2025, 15, 37947 CrossRef CAS PubMed.
- N. Ali, B. Singh, A. R. Vijaya, S. Lal, C. S. Yadav, K. Tarafder and S. Ghosh, Ferromagnetism in Mn-doped ZnO: A joint theoretical and experimental investigation, J. Phys. Chem. C, 2021, 125, 7734–7745 Search PubMed.
- R. Khan, et al., Room-temperature dilute magnetic semiconductor behavior in co-doped (Gd/Co etc.) ZnO nanotubes and related systems, RSC Adv., 2022, 12, 36126–36137 Search PubMed.
- M. A. Wahb, Robust ferromagnetic V0.05-XCoxZn0.95O (x = 0.01, 0.02, 0.03, 0.04, 0.05) nano-compounds: New dilute magnetic-semiconductors with tailored optical activity, J. Magn. Magn. Mater., 2024, 25, 175503 Search PubMed.
- T. Dietl, et al., Zener model description of ferromagnetism in zinc-blende magnetic semiconductors, Science, 2000, 287, 1019–1022 CrossRef CAS PubMed.
- G. Srinet, R. Kumar and V. Sajal, Structural, optical, vibrational, and magnetic properties of sol-gel derived Ni doped ZnO nanoparticles, J. Appl. Phys., 2013, 114, 033912 Search PubMed.
- B. Pal and P. K. Giri, High temperature ferromagnetism and optical properties of Co doped ZnO nanoparticles, J. Appl. Phys., 2010, 108, 084322 CrossRef.
- S. J. Pearton, et al., Recent progress in processing and properties of ZnO, Superlattices Microstruct., 2003, 34, 3–32 Search PubMed.
- M. Venkatesan, et al., Unexpected magnetism in a dielectric oxide, Nature, 2004, 430, 630 CrossRef CAS PubMed.
- S. B. Patil, G. E. Patil and P. V. Dalal, Enhanced gas sensing performance of ZnO and Pt-doped ZnO nanostructured films by chemical spray pyrolysis, Surf. Interfaces, 2024, 54, 105173 CrossRef CAS.
- A. Mezn, et al., Pt–ZnO/M (M = Fe, Co, Ni or Cu): A New Promising Hybrid-Doped Noble Metal/Semiconductor Photocatalysts, J. Inorg. Organomet. Polym. Mater., 2020, 30, 4627–4636 CrossRef.
- J. Rodríguez-Carvajal, Recent advances in magnetic structure determination by neutron powder diffraction, Physica B, 1993, 192, 55–69 Search PubMed.
- J. A. L. Medina, et al., ZnO@Pt nanocomposites for enhanced amaranth dye degradation under UV–vis irradiation, Appl. Surf. Sci., 2026, 716, 164715 Search PubMed.
- P. Hohenberg and W. Kohn, Inhomogeneous electron gas, Phys. Rev., 1964, 136, B864 Search PubMed.
- Q. L. Lin, et al., First-principles study of defect-induced ferromagnetism in ZnO, J. Chem. Phys., 2019, 150, 094704 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Ab initio molecular dynamics for liquid metals, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 47, 558 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Ab initio molecular-dynamics simulation of the liquid-metal-amorphous-semiconductor transition in germanium, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 9, 14251 CrossRef PubMed.
- G. Kresse and J. Furthmüller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set, Comput. Mater. Sci., 1996, 6(1), 15–50 CrossRef CAS.
- G. Kresse and J. Furthmüller, Efficient iterative schemes for ab initiototal-energy calculations using a plane-wave basis set, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953 CrossRef PubMed.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758 CrossRef CAS.
- S. Lardjane, G. Merad, N. Fenineche, A. Billard and H. I. Faraoun, Ab initio study of ZnCoO diluted magnetic semiconductor and its magnetic properties, J. Alloys Compd., 2013, 551, 306–311 Search PubMed.
- P. Sikam, P. Moontragoon, J. Jumpatam, S. Pinitsoontorn, P. Thongbai and T. Kamwanna, Structural, Optical, Electronic and Magnetic Properties of Fe-Doped ZnO Nanoparticles Synthesized by Combustion Method and First-Principle Calculation, J. Supercond. Novel Magn., 2016, 29, 3155–3166 Search PubMed.
- P. Moontragoon, S. Pinitsoontorn and P. Thongbai, Mn-doped ZnO nanoparticles: Preparation, characterization, and calculation of electronic and magnetic properties, Microelectron. Eng., 2013, 108, 158–162 CrossRef CAS.
- M. V. Gallegos, C. R. Luna, M. A. Peluso, L. C. Damonte, J. E. Sambeth and P. V. Jasen, Effect of Mn in ZnO using DFT calculations: Magnetic and electronic changes, J. Alloys Compd., 2019, 795, 254–260 CrossRef CAS.
- E. Ozugurlu, Cd-doped ZnO nanoparticles: An experimental and first-principles DFT studies, J. Alloys Compd., 2021, 861, 158620 CrossRef CAS.
- F. Maldonado, L. Villamagua and R. Rivera, DFT Analysis of the Adsorption of Phenol on the Nonpolar (1-010) ZnO Surface, J. Phys. Chem. C, 2019, 123, 12296–12304 CrossRef CAS.
- A. Janotti and C. G. Van de Walle, Oxygen vacancies in ZnO, Appl. Phys. Lett., 2005, 87, 122102 CrossRef.
- S. Masoumi, E. Nadimi and F. Hossein-Babaei, Electronic properties of Ag-doped ZnO: DFT hybrid functional study, Phys. Chem. Chem. Phys., 2018, 20, 14688 RSC.
- K. Qi, X. Xing, A. Zada, M. Li, Q. Wang, S. Liu, H. Lin and G. Wang, Transition metal doped ZnO nanoparticles with enhanced photocatalytic and antibacterial performances: Experimental and DFT studies, Ceram. Int., 2020, 46, 1494–1502 CrossRef CAS.
- E. Mamani-Flores, M. L. Moreira and M. Jeomar-Piotrowski, Structural and Electronic Properties of Bulk ZnX (X = O, S, Se, Te), ZnF2 and ZnO/ZnF2: A DFT Investigation within PBE, PBE + U, and Hybrid HSE Functionals, J. Phys. Chem. A, 2020, 124(19), 3778–3785 CrossRef PubMed.
- D. S. Lambert and D. D. O'Regan, Use of DFT+U + J with linear response parameters to predict non-magnetic oxide band gaps with hybrid-functional accuracy, Phys. Rev. Res., 2023, 5, 013160 CrossRef CAS.
- W. Li, X. Xin, H. Wang, C. Guo, H. Jiang and Y. Zhao, Description of light-element magnetic systems via density functional theory plus U with an example system of fluorinated boron nitride: An efficient alternative to hybrid functional approach, Comput. Mater. Sci., 2018, 146, 84–89 CrossRef CAS.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA + U study, Phys. Rev. B:Condens. Matter Mater. Phys., 1998, 57, 1505 CrossRef CAS.
- M. V. Gallegos, C. R. Luna, M. A. Peluso, L. C. Damonte, J. E. Sambeth and P. V. Jasen, Effect of Mn in ZnO using DFT calculations: Magnetic and electronic changes, J. Alloys Compd., 2019, 795, 254–260 CrossRef CAS.
- A. Walsh, J. L. F. Da Silva and S.-H. Wei, Theoretical Description of Carrier Mediated Magnetism in Cobalt Doped ZnO, Phys. Rev. Lett., 2008, 100, 256401 CrossRef PubMed.
- S. Sharma, R. Ponce-Pérez, M. G. Moreno-Armenta, J. Guerrero-Sánchez, R. Kumar, S. Kumar, J. M. Siqueiros and O. Raymond-Herrera, An Atomic-Scale Justification for the Weak Ferromagnetism Observed in Nanostructured Zn0.96-xCoxMn0.04 Powders, J. Phys. Chem. C, 2024, 128, 11835–11844 CrossRef CAS.
- N. Seriani, Z. Jin, W. Pompe and L. Colombi Ciacchi, Density functional theory study of platinum oxides: From infinite crystals to nanoscopic particles, Phys. Rev. B:Condens. Matter Mater. Phys., 2007, 76, 155421 CrossRef.
- W. E. I. Han, et al., DFT Study on Electronic Interactions of Pt, Pd and Au Atoms with γ-Al2O3, Mater. Sci., 2018, 24(3), 239–242 Search PubMed.
- H. J. Monkhorst and J. D. Pack, Special points for Brillouin-zone integrations, Phys. Rev. B, 1976, 13, 5188 CrossRef.
- G. Srinet, S. Sharma, J. Guerrero-Sanchez, R. Garcia-Diaz, R. Ponce-Perez, J. M. Siqueiros and O. Raymond Herrera, Room-temperature ferromagnetism on ZnO nanoparticles doped with Cr: An experimental and theoretical analysis, J. Alloys Compd., 2020, 849, 156587 CrossRef CAS.
- C. Belman-Rodriguez, R. Ponce-Perez, A. M. Reyes, C. A. Galindez-Jamioy, G. Soto, M. H. Farías, J. Guerrero-Sánchez, M. G. Moreno-Armenta, A. Reyes-Serrato and S. A. Aguila, Experimental and theoretical assessment of the Eu3+ doped Bi4Ge3O12, J. Alloys Compd., 2023, 966, 171567 CrossRef CAS.
- O. Warschkow, K. Chuasiripattana, M. J. Lyle, B. Delley and C. Stampfl, Cu/ZnO(0001) under oxidating and reducing conditions: A first-principles survey of surface structures, Phys. Rev. B:Condens. Matter Mater. Phys., 2011, 84, 125311 CrossRef.
- J. I. Paez-Ornelas, R. Ponce-Perez, H. N. Fernández-Escamilla, E. Murillo Bracamontes, H. A. Borbón-Nuñez, J. P. Corbett, M. G. Moreno Armenta and J. Guerrero-Sánchez, Understanding the Role of Oxygen Vacancies in the Stability of ZnO(0001)-(1 × 3) Surface Reconstructions, J. Phys. Chem. C, 2021, 125, 7980–7989 CrossRef CAS.
- R. D. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., 1976, A32, 751–767 CrossRef CAS.
- S. Brandstetter, P. M. Derlet, S. Van Petegem and H. Van Swygenhoven, Williamson–Hall anisotropy in nanocrystalline metals: X-ray diffraction experiments and atomistic simulations, Acta Mater., 2008, 56, 165–176 CrossRef CAS.
- C. Reyes-Damián, R. Álvarez-Chimal, F. Ascencio, J. León-Flores and J. Arenas-Alatorre, Magnetic and optical properties of ZnO nanoparticles and nanorods synthesized by green chemistry, Nano Exp., 2024, 5, 025006 CrossRef.
- S. Sharma, C. F. Sánchez Valdés, J. L. Sánchez Llamazares, J. M. Siqueiros and O. Raymond Herrera, Unveiling quantum superparamagnetism by interacting monodomains in multiferroic Er-doped bismuth ferrate nanostructured particles, J. Phys. Chem. C, 2021, 125, 6449–6460 CrossRef CAS.
- L. Shen, et al., Mechanism of ferromagnetism in nitrogen-doped ZnO: First-principles calculations, Phys. Rev. B:Condens. Matter Mater. Phys., 2008, 78, 073306 CrossRef.
- J. Geshev, Interaction plots obtained from in-field magnetization instead of remanence measurements, J. Magn. Magn. Mater., 2018, 467, 135–138 CrossRef CAS.
- S. Thamm and J. Hesse, A simple plot indicating interactions, J. Magn. Magn. Mater., 1996, 154, 254–262 CrossRef CAS.
- J. Alonso Lopez Medina, et al., Magnetic, structural, and morphological properties behavior of Ni1–xCoxFe2O4 magnetic nanoparticles: Theoretical and experimental study, Mater. Charact., 2024, 216, 114296 CrossRef.
- G. Srinet, et al., Structural, optical and magnetic properties of Zn1-xCoxO prepared by the sol–gel route, Ceram. Int., 2013, 39(6), 6077–6085 CrossRef CAS.
- M. B. Stearns and Y. Cheng, J. Appl. Phys., 1994, 75, 6894 CrossRef CAS.
- R. Wahl, J. V. Lauritsen, F. Besenbacher and G. Kresse, Stabilization mechanism for the polar ZnO(000−1)-O Surface, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 87, 085313 CrossRef.
- J. M. Jacobo-Fernández, C. A. Corona-García, R. Ponce-Pérez, H. A. Borbón-Nuñez, D. M. Hoat, A. Reyes-Serrato and J. Guerrero-Sánchez, Spin-Polarized Total-Energy Calculations on Designing Magnetic Single-Atom Catalysts on the ZnO(000-1) Surface with Pt and Pd, ACS Appl. Nano Mater., 2023, 6, 16740–16748 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |



