Monolayer and bilayer tin monoxide in edge contact with common metals: a first-principles investigation
Binhao
Wang†
 ab,
Yujia
Tian†
ab,
Yujia
Tian†
 a,
Devesh R.
Kripalani
a,
Swee Lee
Gan
b,
Ming
Xue
b and
Kun
Zhou
a,
Devesh R.
Kripalani
a,
Swee Lee
Gan
b,
Ming
Xue
b and
Kun
Zhou
 *ac
*ac
aSchool of Mechanical and Aerospace Engineering, Nanyang Technological University, 50 Nanyang Avenue, Singapore 639798, Singapore. E-mail: kzhou@ntu.edu.sg
bInfineon Technologies Asia Pacific Pte. Ltd., 168 Kallang Way, Singapore 349282, Singapore
cCSIJRI-Qingyang Innovation Joint Lab, China-Singapore International Joint Research Institute, Guangzhou 510700, China
First published on 24th November 2025
Abstract
The high hole mobility and layer-dependent properties of two-dimensional (2D) tin monoxide (SnO) make it a promising candidate for use as a channel material in field effect transistors. However, the widely used top contact (TC) configuration in such transistors often faces high contact resistance due to weak van der Waals interaction at the interface. In contrast, the edge contact (EC) configuration offers improved charge injection efficiency through chemical bonding at the interface. This study provides a comprehensive investigation of the electronic properties of monolayer (ML) and bilayer (BL) SnO ECs with different metal electrodes (silver, aluminium, gold, copper, and nickel) via first-principles calculations. Our results show that SnO undergoes clear metallisation at the edge. Tunnelling barriers (TBs) are found within ML SnO instead of at the metal–semiconductor interface, whereas they are eliminated in BL SnO. Schottky barriers (SBs) are also observed near the TB locations. Metallisation is confined to Sn and O atoms near the interface, while distant regions remain semiconducting. The calculated Fermi level pinning factor for ML SnO ECs is 0.48, which is higher than the mean (0.31) and median (0.28) values reported in theoretical studies of ECs and TCs of 2D semiconductors. The carrier mobilities of BL SnO under ECs appear to be higher than those of its ML counterpart, as indicated by the more dispersive band structures of the former. This behaviour is likely attributed to the intrinsic layer-dependent properties of SnO. These findings offer robust guidance for the design of SnO-based EC transistors.
1. Introduction
The further downscaling of silicon-based transistors to keep up with Moore's law has become challenging because of the unfavourable quantum effects that may arise, such as the short-channel effect,1 which commands the exploration of alternative materials. Since the realisation of graphene in 2004 via mechanical exfoliation,2 a wide variety of two-dimensional (2D) semiconductors have been discovered with potential roles in next-generation nanoelectronics.3,4 Among these semiconductors, 2D metal oxides stand out for their stability in air and tunable physical properties.5 In particular, tin monoxide (SnO) has recently garnered significant interest due to its remarkable properties, including high hole mobility and layer-dependent characteristics.6,7 These attributes make SnO a promising candidate for a channel material in transistors. A thin-film transistor utilising SnO as the channel material has previously been fabricated, demonstrating a relatively high field effect mobility of 7.6 cm2 V−1 s−1.8 An earlier study by Saji et al. has also demonstrated multilayer SnO as a suitable channel material for field effect transistors (FETs).9It is well established that when a metal electrode comes into direct contact with a semiconductor, it alters the interfacial properties, including the Schottky barrier height (SBH) and tunnelling barrier (TB). These modifications are strongly influenced by the type of contact made. Currently, there are two common types of contacts in transistors based on 2D semiconductors: the top contact (TC) and edge contact (EC). In TC configurations, the planar surface of the 2D semiconductor comes into contact with the surface of the bulk metal electrode, whereas in EC configurations, the atomically thin edge of the 2D semiconductor comes into contact with the surface of the bulk metal electrode.
TC configurations often suffer from high contact resistance, primarily due to the van der Waals (vdW) interaction between the dangling-bond-free surface of 2D materials and the bulk metal electrode.10 For example, a relatively low tunnelling probability was observed in SnS/TaS211 and PdPSe/graphene12 TC configurations because of the weak orbital overlap or vdW interaction at the interface. This configuration effectively reduces the charge injection efficiency into the semiconductor. In comparison, EC configurations offer better charge injection through strong orbital overlap at the interface and generally lower contact resistance than TC configurations.13,14 This strong orbital overlap arises from the presence of dangling bonds at the semiconductor edge, which result from its abrupt edge termination, and leads to strong bonds with the interfacial metal atoms. Generally, stronger bonds translate into better charge injection efficiency.
Given the benefits of 2D SnO and EC configurations, we provide a comprehensive theoretical investigation into the electronic performance of monolayer (ML) and bilayer (BL) SnO EC transistors using various metal electrodes (silver, aluminium, gold, copper, and nickel). First-principles calculations were performed to systematically investigate the geometry, binding strength, metallisation, charge transfer, and energy barriers of the SnO EC configuration.
2. Computational details
This section presents the detailed methodology used to model the EC heterostructures and perform density functional theory (DFT) calculations. It covers the lattice matching between the bulk metal and SnO layers, determination of the optimal supercell configuration, structural relaxation of the supercell, and essential parameters used in the DFT calculations.2.1. Lattice matching
Five different metal electrodes (Ag, Al, Au, Cu, and Ni) were chosen to cover a wide range of work functions, ranging from 4.01 to 5.18 eV, as shown in Table 1. The calculated lattice constants for pristine ML SnO are a = b = 3.83 Å, which agree well with the previous theoretical studies.15,16| ML SnO | BL SnO | ||||||
|---|---|---|---|---|---|---|---|
| Ag | Al | Au | Cu | Ni | Al | Cu | |
| ε (%) | −0.93 | 0.68 | −2.52 | 1.03 | 4.01 | 0.68 | 1.03 |
| E b (eV per atom) | −6.15 | −8.43 | −8.41 | −8.62 | −12.78 | −5.34 | −5.47 |
| d min,m–Sn (Å) | 2.92 | 3.12 | 2.73 | 2.71 | 2.50 | 3.02 | 2.69 |
| d min,m–O (Å) | 2.60 | 2.39 | 2.54 | 2.13 | 2.47 | 2.37 | 2.19 |
| s m–Sn | 0.97 | 1.04 | 0.90 | 0.95 | 0.89 | 1.00 | 0.94 |
| s m–O | 1.06 | 0.95 | 1.05 | 1.05 | 1.24 | 1.09 | 1.08 |
| W m (eV) | 4.41 | 4.01 | 5.18 | 4.77 | 5.11 | 4.01 | 4.77 |
| Φ e (eV) | 1.93 | 1.64 | 1.98 | 1.96 | 2.12 | 0.49 | 0.92 |
| Φ h (eV) | 1.09 | 1.37 | 0.77 | 1.06 | 0.84 | 0.66 | 0.41 |
| E g (eV) | 3.02 | 3.01 | 2.75 | 3.02 | 2.96 | 1.15 | 1.33 |
| Penetration depth (nm) | 0.57 | 0.61 | 0.63 | 0.58 | 0.59 | 0.61 | 0.59 |
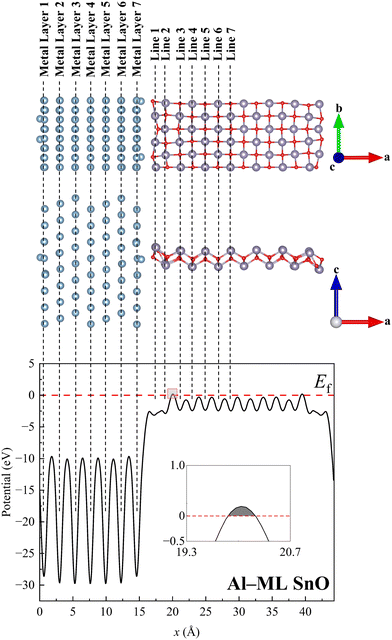
The (111) crystal plane of the metals with face-centred cubic structures, which is the most stable surface plane,17 was chosen to make a direct EC with SnO. Fig. 1 shows the top and side views of the heterostructure formed by Al and ML SnO as an example. Strain has been shown to significantly influence the electronic properties of 2D semiconductors, even when it is minimal.15,18–20 To preserve the intrinsic properties of SnO, strain was applied to the metals along the b direction rather than to SnO, enabling lattice matching between SnO and the metals. The applied strains ranging from −2.52% to 4.01%, as listed in Table 1, remain within the acceptable limit, causing negligible disruption to the electronic properties of the metals.21
2.2. Construction of the edge contact supercell
The construction of the EC supercell is rather tedious in view of the high degree of freedom in the EC configuration. To achieve the most stable configuration for the ML SnO EC, the edge of the SnO layer was initially positioned randomly on the interfacial layer of the metal electrode, and the position of the SnO layer along the a, b, and c directions was then optimised for each metal. In this subsection, only one ionic iteration was performed instead of the full relaxation, unless otherwise stated, to reduce the computational cost, and the structure with the lowest energy is taken as the most stable configuration.The distance along the a direction was first optimised, specifically the distance between the interfacial metal layer and the contacting edge of SnO, defined as dm–s. This optimisation was achieved by gradually decreasing dm–s at an interval of 0.1 Å, starting from an initial value of 3 Å. The optimum dm–s value was chosen at which the minimum energy is obtained. Next, to optimise the b and c positions, the Sn and O atoms at the edge were aligned over various high-symmetry sites of the metal layer using the previously optimised dm–s values; the high-symmetry sites are labelled in Fig. S1. The energy of these configurations was calculated, and the configuration with the lowest energy was selected to be the ideal configuration in terms of b and c positions. Last, dm–s was validated with the optimised b and c positions. There is no change to its optimised value.
For the construction of the BL SnO EC configuration, the interlayer distance between the two pristine layers of SnO must first be optimised before they are brought into contact with the metal electrode. The interlayer distance of the fully relaxed pristine BL SnO is 2.52 Å, which is consistent with the reported value in the literature.22 Subsequently, the same optimisation procedures used for the ML SnO EC are applied along the a, b, and c directions. During this process, the interlayer distance between the SnO layers is tuned both before and after each step of optimisation along the three directions.
2.3. Structural relaxation of the supercell
A total of seven layers of metal were used to model a bulk metal electrode. SnO mainly interacts with the metal layers near it. Thus, Metal Layers 1–3 and 5–7, as labelled in the top panel of Fig. 1, were allowed to relax while the middle layer (Metal Layer 4) was kept fixed to model the bulk metal. Similar approaches have been adopted in previous studies of TCs.15,23,24 Metal Layers 1–3 were allowed to move as these layers interact with the right-hand edge of SnO because of the periodic boundary condition applied in DFT calculations. Full relaxation of this heterostructure is computationally expensive because of the large number of atoms involved, with more than 200 atoms for every heterostructure, as listed in Table S1. Therefore, this study employed a layer-by-layer approach for structural relaxation to reduce the computational cost. With the optimised dm–s value in the a direction and locations of SnO in the b and c directions obtained in Section 2.2, relaxation was first performed with Metal Layers 3–5 kept fixed while the rest of the metal layers were allowed to relax. A final round of optimisation was then performed by relaxing the obtained structure once again with only Metal Layer 4 kept fixed.2.4. Density functional theory calculation settings
The optimised geometry and electronic properties in this work were obtained based on DFT calculations using the Vienna ab initio simulation package.25 The generalised gradient approximation (GGA) method was employed with the Perdew–Burke–Ernzerhof formulation26 for both geometry optimisation and electronic property calculation. The vdW interaction between atoms has been accounted for using the DFT-D2 method.27 The energy cutoff for the plane wave basis set was set to be 500 eV for all the models. The full relaxation of structures was performed until the energy difference converged to 10−4 eV and the maximum residual force per atom was less than 0.025 eV Å−1. Different gamma-centred k point grids were used for different heterostructures owing to the different sizes of the supercells, as listed in Table S1. To mitigate the spurious interaction between periodic images, a vacuum thickness of not less than 14 Å was used in the z direction (normal to the plane of SnO) for all the heterostructures. The Visualization for Electronic and STructural Analysis software28 was used for visualisation of the structures.3. Results and discussion
This section presents a systematic analysis of the DFT calculation results and comparison of SnO ECs across various metal electrodes, focusing on the structural distortions, bonding behaviours, and electronic properties.3.1. Structural distortion and binding energy
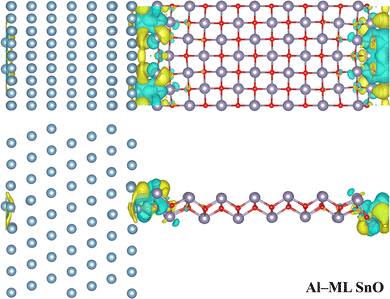
Significant structural distortion is observed near the interface between SnO and metal, as shown in Fig. 1 and Fig. S2. This severe distortion likely arises from the strong bonds formed between the SnO edge and the metal surface, causing the metal, Sn, and O atoms to be drawn towards one another to achieve the most stable configuration.Considering the highly distorted interface, the minimum distance is defined as the average of the shortest distances between interfacial metal atoms and the nearest interfacial Sn or O atoms, termed as dmin,m–Sn or dmin,m–O, respectively, as listed in Table 1. However, to examine the binding at the interface, using dmin,m–Sn and dmin,m–O alone for comparison across different metal electrodes is insufficient because of variations in the atomic radius among different metal elements. To address this, the dmin,m–Sn and dmin,m–O values were further normalised by the sum of the radii of the respective metal atom and Sn or O atoms. It is now expressed as sm–Sn and sm–O for Sn and O atoms, respectively, which indicate the degree of overlap between the interfacial atoms. When the degree of overlap is less than 1, strong covalent bonds should be formed between the interfacial metal atoms and Sn or O atoms. A smaller value indicates a higher degree of overlap.
The calculated results show that the degree of overlap, either sm–Sn or sm–O, is smaller than or equal to 1 for all the heterostructures as listed in Table 1, suggesting that covalent bonds are likely to form between the interfacial metal atoms and the edge of SnO. Interfacial Sn atoms tend to be the main contributor to the strong bonds formed in Ag–ML SnO, Au–ML SnO, Cu–ML SnO, Ni–ML SnO, Al–BL SnO, and Cu–BL SnO, as suggested by the higher degree of overlap with Sn atoms than with O atoms (sm–Sn < sm–O). In contrast, interfacial O atoms are the main contributor to the strong bonds in Al–ML SnO, as shown by the higher degree of overlap between Al and O atoms (sm–O = 0.95) than between Al and Sn atoms (sm–Sn = 1.04).
We have also calculated the binding energy Eb in eV atom−1 at the interface based on the formula
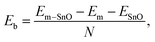 | (1) |
It was observed that the Ni–ML SnO system exhibits stronger binding than other ML heterostructures. This higher magnitude is attributed to the greater number of unpaired valence electrons in isolated Ni (2 electrons) than in other isolated metals in this study (1 electron), which are available to form bonds with SnO. Notably, among the rest of the metals, Al, Au, and Cu form stronger bonds with ML SnO than Ag despite having the same number of unpaired electrons in their isolated states. This higher strength is probably a result of charge redistribution caused by orbital hybridisation within the Al, Au, and Cu atoms themselves, leading to an increase in the number of unpaired electrons when they are in contact with the edge of SnO. This phenomenon has been observed in other theoretical studies based on Mulliken charge analysis.30–32
BL heterostructures seem to have weaker binding than their ML counterparts, as observed from the calculated binding energy of Al–ML SnO (−8.43 eV atom−1), Al–BL SnO (−5.34 eV atom−1), Cu–ML SnO (−8.62 eV atom−1), and Cu–BL SnO (−5.47 eV atom−1). This trend is in contrast to the theoretically calculated values for TCs in the literature, where BL heterostructures generally have stronger binding than their ML counterparts.29,33,34 The reason lies in the definition of binding energy. As stated in eqn (1), its absolute value is inversely proportional to the number of contacting atoms. For EC configurations, as the number of channel layers increases, the number of contacting atoms increases as the edges of all the layers are in direct contact with the metal electrode. Thus, a smaller binding energy magnitude per contacting atom is obtained than those of the ML counterparts. In contrast, for TC configurations, the number of contacting atoms remains the same when the ML is changed to a multilayer structure, as the layers below the interfacial layer do not make direct contact with the metal electrode. Therefore, the magnitude of the binding energy per atom for BL TCs is higher than that of the ML counterpart. Since the binding energy of the BL EC heterostructures, without being normalised by the number of contacting atoms, still has greater magnitudes than those of their ML counterparts (101.15, 128.23, 68.98, and 87.54 eV for Al–ML SnO, Al–BL SnO, Cu–ML SnO, and Cu–BL SnO), a stronger bond is actually expected within the BL heterostructures than in their ML counterparts.
3.2. Electronic properties
This section focuses on the electronic properties of the SnO EC, including the tunnelling behaviour, charge transfer performance, contributions of different orbital states in the band gap region, metallisation, SBHs, the FLP effect, and charge carrier mobility. These results offer a better understanding of the electronic performance of SnO-based EC devices.| Δρ = ρm−SnO − ρm − ρSnO, | (2) |
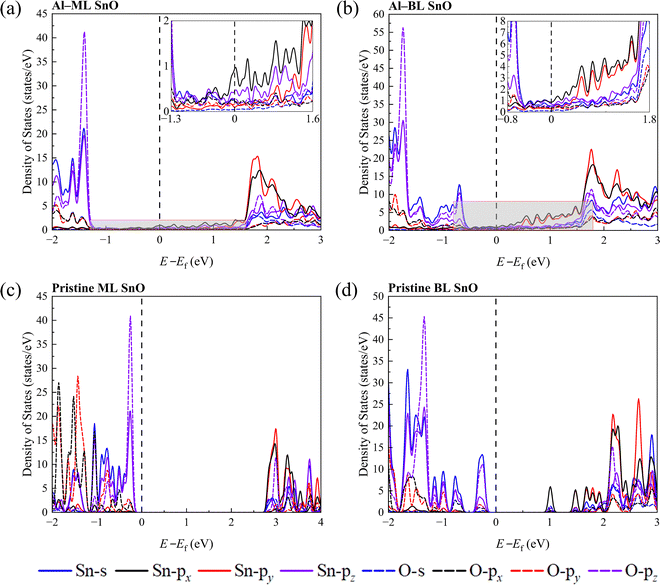
The states in the band gap are predominantly Sn p states, as shown in the insets of Fig. 3a and b. This phenomenon indicates orbital hybridisation between the Sn and interfacial metal atoms such that the states of Sn are extended into the band gap region. In comparison, the BL SnO heterostructures (Al–BL SnO and Cu–BL SnO) exhibit significantly more Sn p states in the band gap region than their ML counterparts, owing to the greater number of interfacial Sn atoms in the BL than the ML.
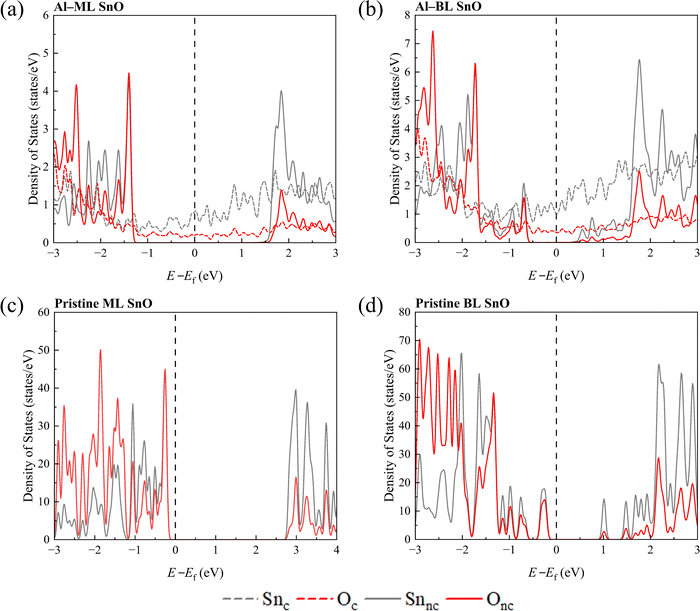
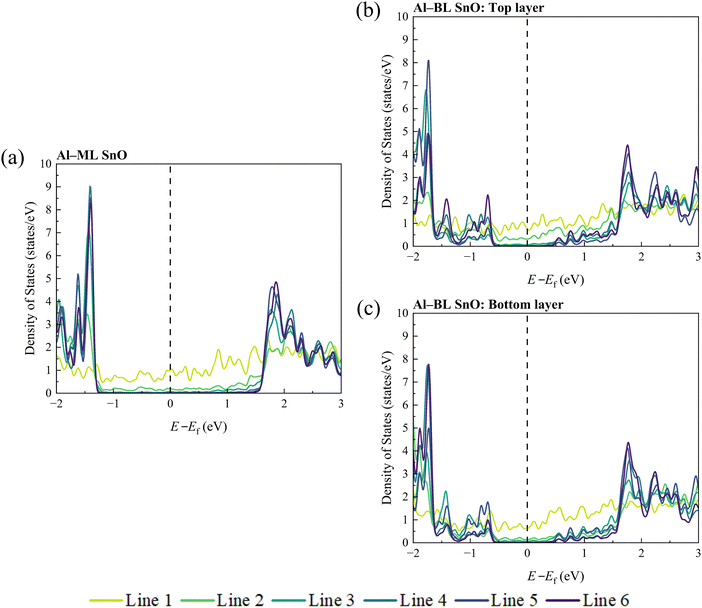
Next, the LDOS of Lines 1 to 6 of SnO is plotted to examine the penetration depth of metallic wave functions into it, as shown in Fig. 5 and Fig. S6. It appears that the metallic wave function penetrates up to Line 4 of SnO for all the ML and BL heterostructures, which is represented by the absence of states in the band gap region from Line 5 onwards. This observation suggests that the region spanning Lines 1 to 4 becomes metallised while that corresponding to Lines 5 and 6 retains its semiconducting nature for all the heterostructures. Generally, the number of states contributed to the band gap region gradually reduces as the atoms are located farther from the interface. The calculated penetration depths range from 0.57 to 0.63 nm, as listed in Table 1, resulting in at least a 10% reduction in the effective channel length for a sub-10 nm FET, considering the metallisation depths on both the left and right contacted edges of SnO.
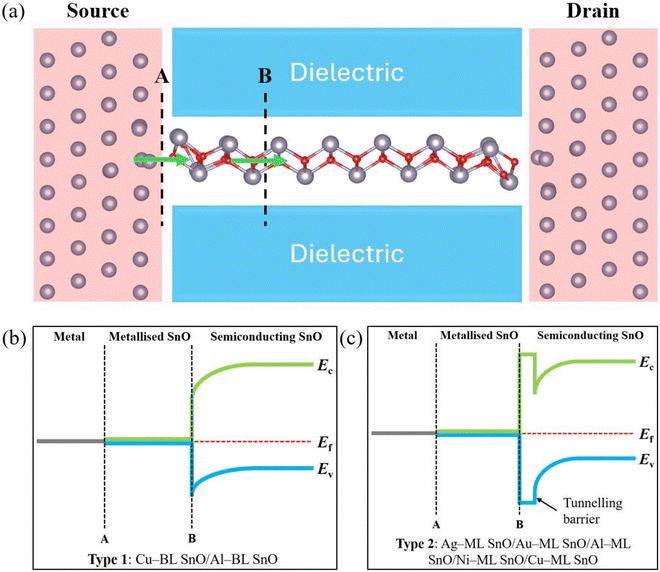
The metallisation of SnO gives rise to two different contact interfaces in an EC transistor along the charge carrier injection path, as shown in Fig. 6a. Interface A refers to the interface between the metal electrode and the metallised SnO region. Interface B refers to the interface between the metallised SnO region and the semiconducting SnO region. At interface A, no SB is present because of the closed band gap resulting from metallisation, thus facilitating charge carrier injection. An SB can emerge near interface B, depending on the work functions of the two sides, as this interface can be regarded as an M–S junction. Additionally, as discussed earlier in Section 3.2.1, a TB is present within the SnO layer in Ag–ML SnO, Al–ML SnO, Au–ML SnO, Ni–ML SnO, and Cu–ML SnO. Considering the location of the TB (as shown in the insets of Fig. 1 and Fig. S2) and the metallisation depth in SnO, it can be inferred that the TB and SB are likely to arise in approximately the same region of the SnO layer. This phenomenon results in two distinct types of band alignment for the EC transistors investigated in this study, Type 1 and Type 2, as illustrated in Fig. 6b and c. Type 1 is where only an SB is present, applicable to Al–BL SnO and Cu–BL SnO. Type 2 is where both a TB and an SB are present, applicable to Ag–ML SnO, Al–ML SnO, Au–ML SnO, Cu–ML SnO, and Ni–ML SnO. The coexistence of the SB and TB could hinder the efficiency of charge carrier injection and therefore give rise to higher electrical resistance in that region.
The metallisation in an EC configuration differs from that in a TC configuration, particularly in the case of a multilayered channel. In a TC configuration, metallisation occurs only in the semiconductor layers in the vicinity of the metal electrode, while the layers farther from the electrode may remain as pristine semiconductors38 because of the decaying nature of the metallic wave function. This phenomenon can result in the presence of an SB between the metallised and semiconducting layers. Additionally, a TB is also likely to occur between the semiconducting layers held together by vdW interactions.39 In comparison, for the case of an EC configuration, metallisation occurs across the edges of all the semiconductor layers as each layer is in direct contact with the bulk metal electrode.40 As demonstrated in this work, in some cases, only an SB is present between the metallised and semiconducting regions of the channel, e.g., in Al–BL SnO and Cu–BL SnO. Moreover, the metallisation of SnO eliminates the SB at the metal contact interface, reducing the effective channel length of the EC transistor. This phenomenon can reduce the carrier transition time within the SnO layer and thus improve the electrical performance.
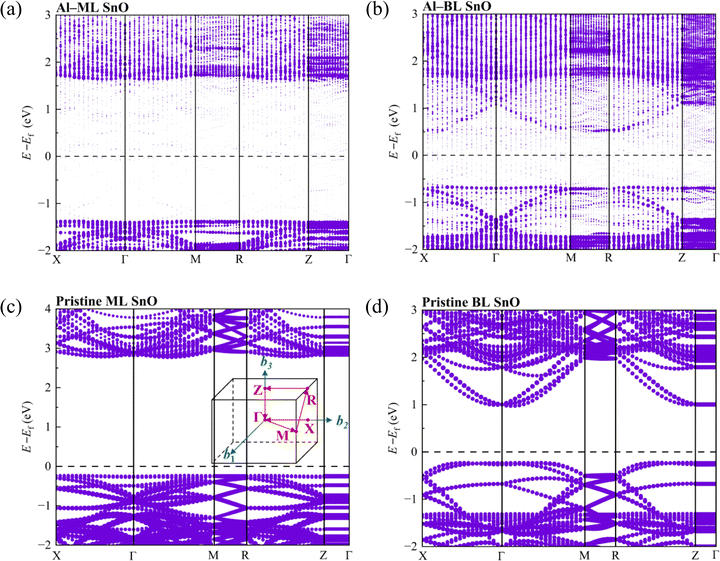
Despite the metallisation, the valence band maximum (VBM) and the conduction band minimum (CBM) of SnO could still be identified because of the distinct difference in their weights and those of the band gap states.15,41 The calculated band gaps (CBM – VBM) for pristine ML and BL SnO are 3.01 eV and 1.20 eV, respectively, in agreement with theoretical studies in the literature at the GGA level.15,22,42 When a metal with a lower work function contacts a semiconductor with a higher work function, charge transfer occurs because of the disparity in their work functions. This transfer causes the Fermi level of the metal to shift downwards and that of the semiconductor to shift upwards, eventually aligning the Fermi levels and establishing thermal equilibrium. The SBHs are estimated based on the energy difference between the Fermi level and the VBM or CBM for the hole SBH and electron SBH, respectively. The calculated SBHs at the GGA level are listed in Table 1. The calculated hole SBHs for ML and BL heterostructures range from 0.77 to 1.37 eV and from 0.41 to 0.66 eV, respectively.
SnO in the ML heterostructures and in Cu–BL SnO maintains a p-type characteristic despite the significant upward shift of the Fermi level. This behaviour can be attributed to the partial FLP effect on SnO, pinning the Fermi level near the VBM. In contrast, Al–BL SnO exhibits an n-type characteristic with an electron SBH of 0.49 eV and a hole SBH of 0.66 eV.
| Φe,ideal = Wm − χs, | (3) |
| Φh,ideal = Eg + χs − Wm, | (4) |
The primary factors contributing to FLP in this study are (1) the formation of MIGS, caused by the penetration of metallic wave functions into semiconducting SnO37 as suggested by Fig. 5 and Fig. S6; (2) the presence of an interface dipole as shown in Fig. 2 and Fig. S3, resulting from charge redistribution at the interface, which leads to a shift in the electronic energy levels;45 and (3) the emergence of band gap states due to the weakening of bonds between Sn and O atoms at the edge. This bond weakening is driven by strong interfacial interactions that distort the structure near the interface, as shown in Fig. 1 and Fig. S2, causing the Sn and O states to extend into the band gap region. This phenomenon was also observed in an MoS2 TC configuration.41
The FLP factor quantifies the extent to which the Fermi level of the semiconductor is pinned to a certain energy level with respect to changes in the work function of the contacting metal. It is mathematically defined as S = |dΦh/dWm|. When S = 1, there will be no FLP effect, and the Schottky–Mott rule is fully applicable; when S = 0, the FLP effect will be the strongest, and the Fermi level will be pinned to the charge neutrality level regardless of the work function of the contacting metal electrode. It is recognised that GGA generally underestimates the band gap and consequently the SBHs owing to the lack of quasiparticle and excitonic effect description in the functional,22 and to obtain an accurate band gap value, hybrid functionals such as HSE0646 are required. However, calculations based on hybrid functionals require extensive computational resources, especially for large material systems involving hundreds of atoms. Therefore, GGA was still employed in this work, and the general trend of GGA-calculated band gaps should still hold.
In this study, the ML SnO heterostructures are partially pinned with S = 0.48, as indicated in Fig. 8a. We have benchmarked the FLP factor obtained in this study against those from other theoretical studies of 2D semiconductors in the literature, covering both TC and EC configurations, as shown in Fig. 8b. Only theoretical studies were considered for comparison, as the FLP factor of a given material may differ between theoretical predictions and experimental results. This discrepancy arises because the contact properties in experimentally fabricated devices are highly sensitive to the fabrication methods and specific experimental conditions. Moreover, 2D materials typically contain defects, which can introduce defect-induced gap states, further influencing the FLP.47 Therefore, a direct comparison between theoretical and experimental studies would not be appropriate in this context.
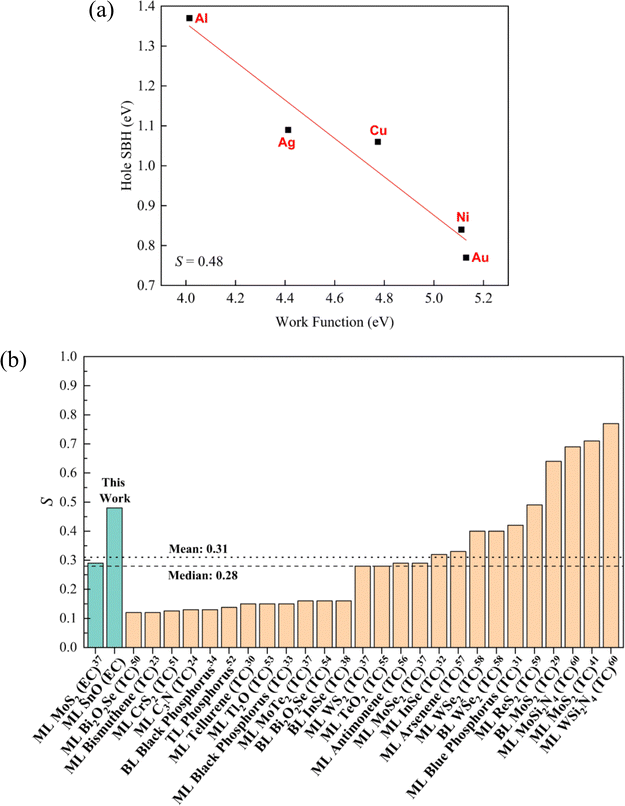 | ||
| Fig. 8 (a) FLP factor S of SnO ECs. (b) Comparison of the FLP factor calculated in this work with values reported in different theoretical studies (both TC and EC) in the literature.23,24,29–34,37,38,41,50–60 The cyan bars represent EC studies, and the orange bars represent TC studies. TL stands for trilayer. To the best of the authors' knowledge, the FLP factor of only one theoretically analysed EC has been reported. | ||
The ML SnO EC heterostructures in this study have a weak FLP effect as compared to other TCs and have the a weaker FLP effect than the theoretically analysed EC, as shown by the relatively low mean (0.31) and median (0.28) of all the values in the cited studies. This comparison suggests that ML SnO ECs have a better tunability of the SBH than most of the other 2D semiconductors through the appropriate selection of metal electrodes.
Because of the high computational cost, only a limited number of metal species (Al and Cu) were modelled for the BL SnO ECs in this study. Therefore, the FLP factor for the BL SnO heterostructures will not be reliable and is not addressed here. However, it can be anticipated that under ideal conditions, specifically in the absence of defects and impurities, the FLP factor in BL SnO ECs would exhibit only a minor deviation from that observed in the ML SnO ECs in this study. This conclusion can be drawn from a similar amount of structural distortion, surface dipole, and band gap states being introduced at the edge of BL SnO as compared to its ML counterpart, each corresponding to the three origins of FLP discussed earlier and as seen in Fig. 1, 2, 5 and Fig. S2, S3, and S6.
The FLP effect in each layer may be consistent in the EC configuration, unlike in the TC configuration. In TC configurations, only the surface layer of the semiconductor directly interacts with the bulk metal electrode, which brings about different contact conditions to each layer. Wang et al. demonstrated that the MIGS are confined to the MoS2 layers near the interface, leading to strong FLP at those layers, while layers further away from the interface remain largely de-pinned.48
In contrast, the EC configuration in this study is expected to have comparable FLP factors across each layer. This inference arises from the observed similarity in responses between the top and bottom layers, as shown in Fig. 5b, c and Fig. S2e, f, S3e, f and S6f, g. In particular, the structural distortion, surface dipole, and band gap states at the edge appear consistent between the top and bottom layers of the BL SnO EC heterostructures, suggesting that each SnO layer experiences similar contact conditions.
Under experimental conditions, the top and bottom layers may be exposed to different environments, so the FLP effect in the EC configuration is likely to deviate from one layer to another. This phenomenon has been observed in a study by Choi et al., where the FLP effect varies from layer to layer because of inhomogeneous charge distribution.49
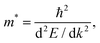 | (5) |
The equation suggests that a larger curvature (dispersive band) results in a smaller effective mass of charge carriers, which corresponds to better carrier mobility. Dispersive band features are observed along the high-symmetry paths X–Γ, Γ–M, and R–Z in the first Brillouin zone for Al–BL SnO and Cu–BL SnO. The X–Γ and R–Z paths in the first Brillouin zone correspond to the in-plane direction of SnO in real space, as depicted in the inset of Fig. 7c. This phenomenon suggests that the charge carriers exhibit a small effective mass primarily in the transport direction, which is beneficial for efficient carrier transport in EC FETs.
BL SnO in contact with Al and Cu exhibits similar band dispersion to pristine BL SnO, as shown in Fig. 7b, d and Fig. S7f, together with the well-preserved semiconducting behaviour observed in the non-contacting region, as discussed in Section 3.2.4. This behaviour suggests that the dispersive band characteristics mainly stem from the inherent properties of SnO, which are attributed to its layer-dependent nature. Zhou et al. have shown that the band structure became more dispersive (smaller effective mass of charge carriers) as the number of SnO layers increases, owing to the interlayer coupling between the Sn atoms.22 This statement suggests that the favourable inherent properties of SnO are well preserved in EC configurations despite the strong interaction between the metal electrode and SnO.
4. Conclusion
The electronic performance of 2D SnO ECs with various metal electrodes (Ag, Al, Au, Cu, and Ni) has been examined via the theoretical means of first-principles calculations. The edge of SnO is heavily metallised as represented by the absence of a band gap, regardless of the contact metal, owing to the strong orbital hybridisation with the interfacial metal atoms. As a result of the metal-like behaviour at the edge of SnO, an SB may arise within the SnO layer instead of at the contacting interface. The semiconducting properties of SnO remain well preserved in the channel region despite strong interactions at the SnO edge, indicating that its remarkable intrinsic characteristics, such as high charge mobility, are still retained. The FLP effect of the SnO EC is weak compared to those of the EC and TCs reported in previous theoretical studies. Nevertheless, its FLP factor of 0.48 pins the Fermi level near the VBM, resulting in p-type doping for all the metal contacts except for the case of Al–BL SnO. The obtained data and analysis in this work provide strong theoretical guidance for the design of SnO-based EC devices.Author contributions
Binhao Wang and Yujia Tian: conceptualisation, methodology, validation, formal analysis, writing – original draft, writing – review & editing, and visualisation. Devesh R. Kripalani: conceptualisation and writing – review & editing. Swee Lee Gan: conceptualisation and writing – review & editing. Ming Xue: conceptualisation, writing – review & editing, and supervision. Kun Zhou: conceptualisation, methodology, resources, writing – review & editing, supervision, project administration, and funding acquisition. The manuscript was written through contributions of all the authors.Conflicts of interest
The authors declare no competing financial interest.Data availability
All relevant data supporting the findings of this work are available within the main article and the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc03215f.Additional data are available from the corresponding author upon reasonable request.
Acknowledgements
This study was supported by the Economic Development Board, Singapore and Infineon Technologies Asia Pacific Pte. Ltd. through the Industrial Postgraduate Programme with Nanyang Technological University (NTU). The computational calculations for this study were partially conducted using the resources of the National Supercomputing Centre, Singapore and the High-Performance Computing Centre, NTU.References
- I. Ferain, C. A. Colinge and J.-P. Colinge, Nature, 2011, 479, 310–316 CrossRef CAS PubMed.
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, Y. Zhang, S. V. Dubonos, I. V. Grigorieva and A. A. Firsov, Science, 2004, 306, 666–669 CrossRef CAS PubMed.
- A. Gupta, T. Sakthivel and S. Seal, Prog. Mater. Sci., 2015, 73, 44–126 CrossRef CAS.
- X. Huang, C. Liu and P. Zhou, npj 2D Mater. Appl., 2022, 6, 51 CrossRef CAS.
- K. Zhou, G. Shang, H. Hsu, S. Han, V. A. L. Roy and Y. Zhou, Adv. Mater., 2023, 35, 2207774 CrossRef CAS PubMed.
- W. Zhou and N. Umezawa, Phys. Chem. Chem. Phys., 2015, 17, 17816–17820 RSC.
- Y. Hu, J. Hwang, Y. Lee, P. Conlin, D. G. Schlom, S. Datta and K. Cho, J. Appl. Phys., 2019, 126, 185701 CrossRef.
- C. W. Shih, A. Chin, C. F. Lu and W. F. Su, Sci. Rep., 2018, 8, 889 CrossRef PubMed.
- K. J. Saji, K. Tian, M. Snure and A. Tiwari, Adv. Electron. Mater., 2016, 2, 1500453 CrossRef.
- X. Liu, M. S. Choi, E. Hwang, W. J. Yoo and J. Sun, Adv. Mater., 2022, 34, 2108425 CrossRef CAS.
- A. Hassan, M. A. Nazir, Y. Shen, Y. Guo, W. Kang and Q. Wang, ACS Appl. Mater. Interfaces, 2022, 14, 2177–2184 CrossRef CAS.
- A. Hassan, Y. Guo, U. Younis, A. Mehmood, X. Tian and Q. Wang, Phys. Chem. Chem. Phys., 2024, 26, 11014–11022 RSC.
- M. Poljak, M. Matic and A. Zeljko, IEEE Electron Device Lett., 2021, 42, 1240–1243 CAS.
- M. S. Choi, N. Ali, T. D. Ngo, H. Choi, B. Oh, H. Yang and W. J. Yoo, Adv. Mater., 2022, 34, 2202408 CrossRef CAS PubMed.
- Y. Tian, D. R. Kripalani, M. Xue and K. Zhou, 2D Mater., 2023, 10, 045015 CrossRef CAS.
- D. R. Kripalani, P.-P. Sun, P. Lin, M. Xue and K. Zhou, Appl. Surf. Sci., 2021, 538, 147988 CrossRef CAS.
- J.-M. Zhang, F. Ma and K.-W. Xu, Appl. Surf. Sci., 2004, 229, 34–42 CrossRef CAS.
- D. R. Kripalani, P.-P. Sun, P. Lin, M. Xue and K. Zhou, Phys. Rev. B, 2019, 100, 214112 CrossRef CAS.
- Y. Tian, D. R. Kripalani, M. Xue, S. Li and K. Zhou, Phys. Rev. B, 2022, 105, 035308 CrossRef CAS.
- Z. Zhang, Y. Zhao and G. Ouyang, J. Phys. Chem. C, 2017, 121, 19296–19304 CrossRef CAS.
- M. Farmanbar and G. Brocks, Phys. Rev. B, 2016, 93, 085304 CrossRef.
- L. Wanzhong, S. Jian and D. Chong, Phys. Chem. Chem. Phys., 2022, 24, 7611–7616 RSC.
- Y. Guo, F. Pan, M. Ye, X. Sun, Y. Wang, J. Li, X. Zhang, H. Zhang, Y. Pan, Z. Song, J. Yang and J. Lu, ACS Appl. Mater. Interfaces, 2017, 9, 23128–23140 CrossRef CAS.
- W. Song, J. Dai, F. Zou, Y. Niu, Y. Cong, Q. Li and Y. Pan, RSC Adv., 2024, 14, 3820–3833 RSC.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- S. Grimme, J. Comput. Chem., 2006, 27, 1787–1799 CrossRef CAS PubMed.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- H. Zhong, R. Quhe, Y. Wang, Z. Ni, M. Ye, Z. Song, Y. Pan, J. Yang, L. Yang, M. Lei, J. Shi and J. Lu, Sci. Rep., 2016, 6, 21786 CrossRef CAS PubMed.
- J. Yan, X. Zhang, Y. Pan, J. Li, B. Shi, S. Liu, J. Yang, Z. Song, H. Zhang, M. Ye, R. Quhe, Y. Wang, J. Yang, F. Pan and J. Lu, J. Mater. Chem. C, 2018, 6, 6153–6163 RSC.
- J. Li, X. Sun, C. Xu, X. Zhang, Y. Pan, M. Ye, Z. Song, R. Quhe, Y. Wang, H. Zhang, Y. Guo, J. Yang, F. Pan and J. Lu, Nano Res., 2018, 11, 1834–1849 CrossRef CAS.
- B. Shi, Y. Wang, J. Li, X. Zhang, J. Yan, S. Liu, J. Yang, Y. Pan, H. Zhang, J. Yang, F. Pan and J. Lu, Phys. Chem. Chem. Phys., 2018, 20, 24641–24651 RSC.
- Y. Pan, Y. Wang, M. Ye, R. Quhe, H. Zhong, Z. Song, X. Peng, D. Yu, J. Yang, J. Shi and J. Lu, Chem. Mater., 2016, 28, 2100–2109 CrossRef CAS.
- Y. Pan, Y. Dan, Y. Wang, M. Ye, H. Zhang, R. Quhe, X. Zhang, J. Li, W. Guo, L. Yang and J. Lu, ACS Appl. Mater. Interfaces, 2017, 9, 12694–12705 CrossRef CAS PubMed.
- D. Çakır and F. M. Peeters, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 89, 245403 CrossRef.
- Y. Meng, Y. Xu, J. Zhang, J. Sun, G. Zhang and J. Leng, Phys. Lett. A, 2022, 456, 128535 CrossRef CAS.
- Y. Guo, D. Liu and J. Robertson, ACS Appl. Mater. Interfaces, 2015, 7, 25709–25715 CrossRef CAS PubMed.
- L. Xu, Y. Pan, S. Liu, B. Shi, L. Xu, J. Yang, J. Yan, H. Pang, X. Zhang, C. Yang, J. Yang, Y. Wang, Z. Zhang and J. Lu, ACS Appl. Nano Mater., 2019, 2, 6898–6908 CrossRef CAS.
- D. S. Schulman, A. J. Arnold and S. Das, Chem. Soc. Rev., 2018, 47, 3037–3058 RSC.
- J. Kang, W. Liu and K. Banerjee, Appl. Phys. Lett., 2014, 104, 093106 CrossRef.
- C. Gong, L. Colombo, R. M. Wallace and K. Cho, Nano Lett., 2014, 14, 1714–1720 CrossRef CAS PubMed.
- Y. Tian, D. Kripalani, M. Xue, S. L. Gan and K. Zhou, in 2024 IEEE International Symposium on the Physical and Failure Analysis of Integrated Circuits (IPFA), IEEE, Singapore, Singapore, 2024, pp. 1–6 Search PubMed.
- R. T. Tung, Appl. Phys. Rev., 2014, 1, 011304 Search PubMed.
- K. Parto, A. Pal, T. Chavan, K. Agashiwala, C.-H. Yeh, W. Cao and K. Banerjee, Phys. Rev. Appl., 2021, 15, 064068 CrossRef CAS.
- B. H. Moon, G. H. Han, H. Kim, H. Choi, J. J. Bae, J. Kim, Y. Jin, H. Y. Jeong, M.-K. Joo, Y. H. Lee and S. C. Lim, ACS Appl. Mater. Interfaces, 2017, 9, 11240–11246 CrossRef CAS PubMed.
- A. V. Krukau, O. A. Vydrov, A. F. Izmaylov and G. E. Scuseria, J. Chem. Phys., 2006, 125, 224106 CrossRef PubMed.
- R.-S. Chen, G. Ding, Y. Zhou and S.-T. Han, J. Mater. Chem. C, 2021, 9, 11407–11427 RSC.
- Q. Wang, Y. Shao, P. Gong and X. Shi, J. Mater. Chem. C, 2020, 8, 3113–3119 RSC.
- H. Choi, B. H. Moon, J. H. Kim, S. J. Yun, G. H. Han, S. Lee, H. Z. Gul and Y. H. Lee, ACS Nano, 2019, 13, 13169–13175 CrossRef CAS PubMed.
- S. Liu, L. Xu, Y. Pan, J. Yang, J. Li, X. Zhang, L. Xu, H. Pang, J. Yan, B. Shi, X. Sun, H. Zhang, L. Xu, J. Yang, Z. Zhang, F. Pan and J. Lu, Adv. Theory Simul., 2019, 2, 1800178 CrossRef.
- M. R. Habib, S. Wang, S. M. Obaidulla, Y. Khan, X. Pi and M. Xu, Phys. Status Solidi B, 2019, 256, 1800597 CrossRef.
- X. Zhang, Y. Pan, M. Ye, R. Quhe, Y. Wang, Y. Guo, H. Zhang, Y. Dan, Z. Song, J. Li, J. Yang, W. Guo and J. Lu, Nano Res., 2018, 11, 707–721 CrossRef CAS.
- Y. Pan, J. Dai, Z. Liu, M. Wu, H. Hu and J. Lu, J. Mater. Sci., 2020, 55, 11439–11450 CrossRef CAS.
- L. Xu, S. Liu, J. Yang, B. Shi, Y. Pan, X. Zhang, H. Li, J. Yan, J. Li, L. Xu, J. Yang and J. Lu, J. Phys. Chem. C, 2019, 123, 8923–8931 CrossRef CAS.
- X. Zhang, L. Feng, H. Li, Y. Liu, P. Liu, X. Zheng, M. Qu, X. Wang and J. He, Mater. Today Nano, 2023, 24, 100392 CrossRef CAS.
- H. Zhang, J. Xiong, M. Ye, J. Li, X. Zhang, R. Quhe, Z. Song, J. Yang, Q. Zhang, B. Shi, J. Yan, W. Guo, J. Robertson, Y. Wang, F. Pan and J. Lu, Phys. Rev. Appl., 2019, 11, 064001 CrossRef CAS.
- Y. Wang, M. Ye, M. Weng, J. Li, X. Zhang, H. Zhang, Y. Guo, Y. Pan, L. Xiao, J. Liu, F. Pan and J. Lu, ACS Appl. Mater. Interfaces, 2017, 9, 29273–29284 CrossRef CAS PubMed.
- Y. Wang, R. X. Yang, R. Quhe, H. Zhong, L. Cong, M. Ye, Z. Ni, Z. Song, J. Yang, J. Shi, J. Li and J. Lu, Nanoscale, 2016, 8, 1179–1191 RSC.
- N. Gao, S. Zhou, N. Liu, Y. Bai and J. Zhao, J. Mater. Chem. C, 2018, 6, 6764–6770 RSC.
- Q. Wang, L. Cao, S.-J. Liang, W. Wu, G. Wang, C. H. Lee, W. L. Ong, H. Y. Yang, L. K. Ang, S. A. Yang and Y. S. Ang, Npj 2D Mater. Appl., 2021, 5, 71 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |