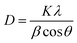A controlled growth strategy for CAU-17 MOF thin films via two-step synthesis
Muhammad Zain
Azeem
 a,
Khaled
Hassanein
a,
Khaled
Hassanein
 *b,
Badriah
Sultan
c,
Abduljabar
Alsayoud
a,
Syed B.
Qadri
d and
Qasem A.
Drmosh
*ab
*b,
Badriah
Sultan
c,
Abduljabar
Alsayoud
a,
Syed B.
Qadri
d and
Qasem A.
Drmosh
*ab
aDepartment of Materials Science and Engineering, King Fahd University of Petroleum & Minerals, Dhahran, 31261, Saudi Arabia. E-mail: drmosh@kfupm.edu.sa
bInterdisciplinary Research Center for Hydrogen Technologies and Carbon Management, King Fahd University of Petroleum & Minerals (KFUPM), Dhahran, 31261, Saudi Arabia. E-mail: khaled000s@yahoo.com
cDepartment of Physics, College of Science, King Khalid University, P.O. Box 9004, Abha, 61413, Saudi Arabia
dNaval Research Laboratory, Washington, DC 20375, USA
First published on 27th October 2025
Abstract
Sputtering-assisted synthesis is being investigated for the first time as a novel approach for the fabrication of CAU-17 MOF thin films. This method enables the production of MOF thin films without the risk of forming impurities or requiring harsh conditions, unlike conventional powder-based techniques. In this work, the synthesis of CAU-17 was achieved directly on different substrates via a two-step sequential process. Initially, a thin film of bismuth oxide was deposited onto the substrate using reactive DC magnetron sputtering. This oxide layer was subsequently transformed into a CAU-17 MOF through coordination with an organic linker under controlled conditions. The synthesized material was characterized using several advanced characterization techniques. The influence of synthesis conditions, such as growth temperature and formation on the surface of various support materials, was also investigated. This approach paves the way for advanced fabrication of MOF thin films with potential uses in sensing, catalysis, and other cutting-edge technologies.
Introduction
Metal–organic frameworks (MOFs) are a class of porous crystalline materials that exhibit permanent porosity.1,2 They were first conceptualized by Dr Omar Yaghi in 1999 with the discovery of MOF-5, commonly known as IRMOF-1.3 It is a hybrid material composed of organic and inorganic components.4,5 The organic component, known as a linker, acts as a connector between the inorganic units, which are referred to as metal nodes.6,7 These materials are renowned for their structural diversity, tunable porous nature, and extremely large surface area.8–10Recently, bismuth-based MOFs have gained the attention of researchers as promising materials for advanced nanoscience and technology applications. One of the main reasons for this is the safe and non-hazardous nature of bismuth. Additionally, it is one of the Earth's most abundant elements, which makes it easily accessible and affordable at an industrial level.11–13 To date, a limited number of Bi-based MOFs have been synthesized and characterized, and among them, only a few exhibit permanent porosity, i.e., the family of CAU (CAU-7, CAU-17, and CAU-35), SU-101, and NOTT-220. In this classification of Bi-MOFs, so far, only CAU-17 has been synthesized cheaply using trimesic acid.14 This MOF was first reported by Inge et al. in 201615 and was subsequently widely found to have emerging applications in several areas, such as electrochemical CO2 reduction to formate/formic acid, electrochemical sensors for detecting heavy metal ions, and the development of advanced batteries with higher conductivity and stability.16–21
Interestingly, the CAU-17 MOF has a more complex structure than many other MOFs. In terms of its topology, 54 unique nodes with 135 edges have been reported. Its structure consists of infinite linear chains of Bi-oxo rods formed by edge-sharing helical BiO9 polyhedra.22 In CAU-17, many framework species take part in asymmetric units, i.e., 9 Bi3+ cations, 9 BTC3− anions, and 9 coordination water units, where each 9 Bi3+ ions connect with 8 O atoms of the BTC3− and 1 O atom from coordinated water molecules.23 Each Bi-cation shares 4 O anions with adjacent cations, forming helical/spiral rods. These rods develop joints with BTC3− anions, resulting in an intricate 3D framework of CAU-17.15 In short, its crystal structure consists of left and right helical/spiral edge-sharing BiO9 polyhedra linked with BTC3− ions, forming channels of hexagonal, rectangular, and triangular shapes.24
One unique fact about CAU-17 is that it is highly sensitive to synthesis conditions. A minor change in synthesis parameters, such as temperature, time, or solvent, can result in a completely different structure. Nguyen et al. reported a study in which the impact of the solvent on CAU-17 crystallization was investigated.25 They found that using DMF, methanol, or even a mixture of DMF and methanol as the solvent led to crystallization into different structures. Köppen et al. determined the optimal synthesis temperature for CAU-17.24 In their work, they employed a microwave-assisted approach and identified 120 °C as the optimal condition for preparing the CAU-17 structure. They also noted that synthesizing above or below this temperature resulted in the formation of unknown impurities. Dong et al. investigated the influence of the synthesis technique on CAU-17 morphology.26 In that study, flower- and rod-like morphologies of CAU-17 with distinct crystalline structures were obtained using sonication and conventional solvothermal methods. Similar behavior was also reported by Huang et al. and Lee et al., who observed almond-flake-like and sheet-like morphologies of CAU-17.19,27
Until now, only three techniques have been reported for the synthesis of CAU-17, to the best of our knowledge: solvothermal, microwave-assisted, and sonochemical techniques. The major drawback of these methods is the formation of impurities and secondary phases during CAU-17 formation. For instance, previous reports have shown that solvothermal and microwave-assisted routes often resulted in the formation of unknown impurities, primarily due to the addition of bismuth(III) nitrate during the synthesis process.24,25 Another significant challenge is the unintentional formation of a Bi(btc) MOF instead of the desired CAU-17. Although both Bi-MOFs are synthesized using the same organic linker and chemical constituents, the key distinction between these two structures lies in their morphology: the Bi(btc) MOF is denser and less porous, whereas CAU-17 features a highly porous framework, which is crucial for applications requiring high surface area. It is well established that a dense phase occurs when CAU-17 crystallizes too rapidly or during prolonged synthesis times.20,25,28 Additionally, powder-based MOFs have previously demonstrated many challenges, such as poor bonding with the substrate, processability, and mechanical strength for advanced applications.29–31
The transition from powder-based technology to thin films is an attractive choice to overcome the above-stated problem, as it offers more precise control over the synthesis conditions and results in the formation of high-quality thin films with improved crystalline nature and a higher purity level.32 Furthermore, the direct synthesis of MOFs from metal oxides is considered a simple and cost-effective approach with strong adhesion between the substrate and MOF, along with admirable structural stability.33,34
The primary advantage of using magnetron sputtering for the synthesis of CAU-17 thin films is the use of a metallic target as a source of metal ions. This approach offers unique benefits over metal salts, as it effectively overcomes challenges related to the formation of unknown impurities or undesirable secondary phases. Additionally, this technique is greener, cost-effective, and scalable, in contrast to the powder-based technique previously employed for CAU-17 synthesis.35,36 Other advantages of this technique include strong adhesion strength, synthesis under mild conditions, and the formation of a uniform coating with great control over thickness.37,38 Aside from that, it has great potential for synthesizing a wide range of MOF thin films by modifying the organic linker, metal target, and synthesis conditions.
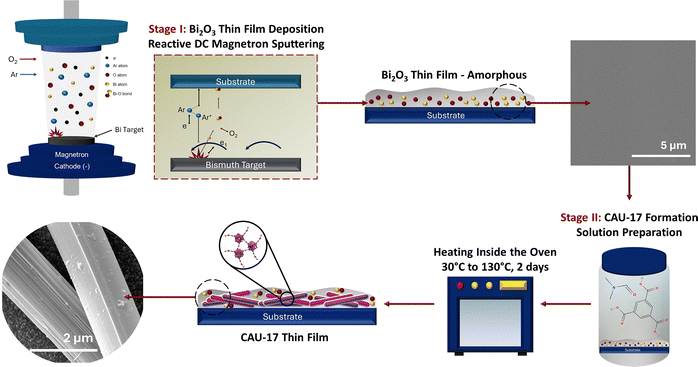
In this work, the CAU-17 MOF thin film was synthesized directly on the substrate surface via a two-step sequential process. Initially, a thin film of bismuth oxide was deposited onto the substrate using reactive DC magnetron sputtering. This oxide layer was subsequently transformed into CAU-17 through coordination with the organic linker under controlled conditions. Furthermore, the study systematically examined the formation of the CAU-17 thin film on various substrate materials and assessed the effect of synthesis temperature on the crystallization behavior and structural properties of the resulting CAU-17 MOF thin films.
Experimental work
Materials
A 99.99% pure bismuth target (2″ diameter and 0.125″ thickness bonded on Cu back plate) was purchased from Semiconductor Wafer Inc. 1,3,5-Benzenetricarboxylic acid (H3BTC) was purchased from Thermos Scientific. Ethyl alcohol (C2H6, absolute) was purchased from DUKSAN Pure Chemicals. Dimethylformamide (C3H7NO, ≥99.5%) was purchased from Fisher Scientific. All chemicals in this work were used as received without any purification. The solution of 0.01 M HCl was prepared by diluting the analytical grade HCl with distilled water.Characterization techniques
X-ray diffraction (XRD) analysis was performed on a Rigaku Mini X-ray diffractometer operating at 30 kV and 15 mA with a Cu Kα wavelength of 1.54 Å. The morphology of thin films was observed by field emission scanning electron microscopy (FESEM, Tescan Lyra 3) operating at 20 kV coupled with energy-dispersive X-ray spectroscopy (EDX). Raman spectroscopy was performed using a DXR Raman microscope (Thermo Fisher, Wisconsin, USA). The analysis was performed using a 10× confocal microscope objective equipped with 1200 lines per mm diffraction. The system was operated at a laser power and wavelength of 3 mW and 455 nm, respectively. Raman signals were acquired in 5 accumulations of 50 seconds from 400 to 2000 cm−1. FT-IR analysis was performed on a PerkinElmer Spectrum 3. The analysis was recorded between the wavelengths of 400 and 4000 cm−1 with a spectral resolution of 4 cm−1. The surface composition of the thin films was examined using the X-ray photoelectron spectroscopy technique (XPS, Thermo Scientific, ESCALAB 250Xi).Bi2O3 thin film deposition
The sputtered Bi2O3 thin films were synthesized on a quartz substrate using a DC reactive sputtering approach. Before deposition, the substrates were ultrasonicated in acetone, ethanol, and DI water separately for 10 min and then dried on a hotplate (100 ± 5 °C). After cleaning, the substrates were mounted on the substrate holder, and the deposition chamber was evacuated to a base pressure of 4.14 × 10−5 Torr. The bismuth target of 99.99% purity was sputtered by DC magnetron sputtering (NSC–4000 sputter coater) by using argon (Ar) and oxygen (O2) as sputtering gases. Bi2O3 thin films were deposited onto the quartz substrate with a cathode power of 50 W for 15 min at room temperature at an Ar![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2 flow rate (sccm) of 35
O2 flow rate (sccm) of 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35. During synthesis, the substrate table was rotated to achieve uniform thickness. Similarly, the same deposition conditions were used for the synthesis of Bi2O3 thin films on the FTO substrate and carbon paper.
35. During synthesis, the substrate table was rotated to achieve uniform thickness. Similarly, the same deposition conditions were used for the synthesis of Bi2O3 thin films on the FTO substrate and carbon paper.
Sputtered Bi2O3 conversion into the CAU-17 thin film
The conversion of the sputtered amorphous Bi2O3 thin film into the CAU-17 thin film is achieved by immersing the coated substrate into the reaction medium that consists of 5 mL of DMF solution, 100 mg of H3BTC, and 1 mL of 0.01 M HCl in a glass vial. Afterwards, the solution was sonicated until it became transparent, and then heated at 105 °C for 2 days. The synthesized CAU-17 thin film was then cleaned multiple times with an ethanol solution and dried in an oven (100 °C ± 10 °C) until the solvent had fully evaporated, as part of the solvent exchange process. For investigating the influence of temperature on CAU-17 formation, fresh samples were prepared in a wider range of temperatures (30 °C, 80 °C, 105 °C, 120 °C, and 130 °C) for 2 days. The complete synthesis procedure of the CAU-17 thin film is illustrated in Scheme 1. For the synthesis of CAU-17 on the surface of the FTO substrate and carbon paper coated with amorphous Bi2O3, a similar solution was prepared. The conversion of the Bi2O3-coated FTO substrate into CAU-17 occurs at 130 °C for 2 days, while for the carbon paper-coated Bi2O3 thin film, it takes place at 105 °C for 2 days.Results and discussion
Formation of CAU-17 MOF thin films
XRD analysis of CAU-17 MOF thin films was performed to confirm their structure, phase purity, and crystalline nature. XRD spectra of the sputtered Bi2O3 thin film and the CAU-17 thin film are shown in Fig. 1. As can be observed, no crystalline peaks were observed in the diffraction pattern of the as-deposited sputtered Bi2O3 thin film, indicating the amorphous nature of the thin film. In contrast, the XRD pattern of the CAU-17 thin film displayed several crystalline peaks. Among them the most intense peaks of CAU-17 were determined at 8.35°, 10.49°, 12.68°, 16.51°, 24.75°, and 33.13°. The average crystallite size of CAU-17 was about 62 nm, calculated by applying Scherrer's equation to the most intense (hkl) peak:where D is the crystallite size, β is the full width at half maximum (FWHM) measured in radians, λ is the wavelength of incident X-ray (0.15406 nm), K is the shape factor (0.9), and θ represents the X-ray diffraction angle.39 The XRD pattern of CAU-17 displayed distinct peaks in comparison to the reported simulated pattern of CAU-17.15 The main reason for obtaining a different diffraction pattern of CAU-17 is due to its high sensitivity towards synthesis conditions and preparation approaches.
 | ||
| Fig. 1 XRD pattern of the as-deposited amorphous Bi2O3 thin film via reactive DC magnetron sputtering and the CAU-17 thin film prepared at 105 °C for 2 days. | ||
FESEM results revealed that the surface of the Bi2O3 thin film transformed into CAU-17 nanorods with homogenous distribution, as shown in Fig. 2(a and b). The surface morphology of the CAU-17 thin film was also consistent with that reported in the literature.14,40,41 The width of the nanorods was determined by using the open-source image-processing software ImageJ (1.41v) developed by the NIH. The width of the nanorods was measured by considering 50 nanorods chosen randomly from the sample. The mean calculated width of the CAU-17 nanorods was 3.719 µm.
 | ||
| Fig. 2 FESEM images of the (a) as-deposited Bi2O3 thin film and (b) CAU-17 thin film, along with (c) EDX elemental mapping of the CAU-17 thin film prepared at 105 °C for 2 days. | ||
EDX elemental mapping of CAU-17 nanorod thin films, shown in Fig. 2(c), confirms the existence of bismuth (Bi), carbon (C), and oxygen (O). The homogenous distribution of these elements throughout the nanorods represents the well-formed structure of CAU-17. Additionally, it was also observed that the nanorod morphology consists of a high bismuth content, indicating a nearly complete transformation of the Bi2O3 thin film into CAU-17. To further confirm whether the synthesized material belongs to CAU-17 or not, additional spectroscopy techniques were employed.
To confirm the molecular structure of the CAU-17 thin film, Raman spectroscopy was used. The Raman spectra of H3BTC and CAU-17 are shown in Fig. 3(a), which exhibits multiple peaks corresponding to their respective functional groups. The Raman spectrum of the CAU-17 MOF thin film was highly consistent with the reported literature.42 The CAU-17 spectrum showed the characteristic peaks of H3BTC within the range of 700–2000 cm−1 and Bi–O-related bonds at 50–600 cm−1.14 The characteristic bands at 425.73 and 490.33 cm−1 belong to the Bi–O bond stretching modes. The stretching and bending modes of C–H bonds were observed at 731.39, 838.42, and 1205.79 cm−1. The C–O–O symmetric and asymmetric stretching vibration modes were determined at 1448.78 and 1554.85 cm−1, respectively.43 The peaks at 1003.30, 1133.48, and 1608.84 cm−1 represent the symmetric stretching of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds from the aromatic ring of H3BTC.43–45 The band at 1358.14 was assigned to the in-plane vibration of the C–O bond.46 The presence of C–O–O and Bi–O bonds in the CAU-17 spectrum indicates the existence of required functional groups and confirms metal–linker coordination.
C bonds from the aromatic ring of H3BTC.43–45 The band at 1358.14 was assigned to the in-plane vibration of the C–O bond.46 The presence of C–O–O and Bi–O bonds in the CAU-17 spectrum indicates the existence of required functional groups and confirms metal–linker coordination.
 | ||
| Fig. 3 (a) Raman spectra of H3BTC and the CAU-17 MOF thin film and (b) FT-IR spectra of H3BTC and the CAU-17 MOF thin film. | ||
FT-IR spectroscopy was applied to determine the existence of intended functional groups in the structure of the CAU-17 thin film. Fig. 3(b) shows the FT-IR results of H3BTC and the CAU-17 thin film. The FT-IR spectrum of H3BTC shows the characteristic peaks of O–H stretching, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, and O–H bending groups at 3000–2500 cm−1, 1763–1569 cm−1, and 996–764 cm−1, respectively.26 In the FT-IR spectrum of CAU-17, both characteristic peaks of the Bi–O and carboxylate groups were observed. The new peaks appeared within the range of 500 to 700 cm−1, corresponding to the Bi–O group, indicating the coordination of the COOH group of the organic linker with the Bi3+ ions through electrostatic interaction.21 The characteristic peaks at 536 and 720 cm−1 correspond to Bi–O stretching.47 The carboxylate groups were observed at 1352 and 1432 cm−1, attributed to the asymmetric vibration of the –COOH group.26 The disappearance of the characteristic peaks of the H3BTC linker, along with the appearance of new bands corresponding to the coordination between the linker and Bi3+ ions, confirms the successful formation of the CAU-17 thin film.
O stretching, and O–H bending groups at 3000–2500 cm−1, 1763–1569 cm−1, and 996–764 cm−1, respectively.26 In the FT-IR spectrum of CAU-17, both characteristic peaks of the Bi–O and carboxylate groups were observed. The new peaks appeared within the range of 500 to 700 cm−1, corresponding to the Bi–O group, indicating the coordination of the COOH group of the organic linker with the Bi3+ ions through electrostatic interaction.21 The characteristic peaks at 536 and 720 cm−1 correspond to Bi–O stretching.47 The carboxylate groups were observed at 1352 and 1432 cm−1, attributed to the asymmetric vibration of the –COOH group.26 The disappearance of the characteristic peaks of the H3BTC linker, along with the appearance of new bands corresponding to the coordination between the linker and Bi3+ ions, confirms the successful formation of the CAU-17 thin film.
XPS analysis was performed to determine the surface composition, chemical states, and functional groups of the CAU-17 MOF thin film. Fig. 4(a) shows the survey scan that confirms the existence of the main constituents of CAU-17, i.e., Bi, C, and O elements in the synthesized thin film, consistent with the aforementioned EDX mapping results. The presence of Sn is due to the synthesis of the CAU-17 thin film on the surface of the FTO substrate, while the existence of N at ∼399 eV corresponds to N–C bonding, which occurs due to the entrapment of a trace amount of DMF within the pores of CAU-17.48,49 For the correction of binding energies, the C 1s peak of carbon (BE = 284.6 eV) was used as a reference.50
 | ||
| Fig. 4 XPS analysis of the CAU-17 thin film: (a) survey scan; high-resolution spectra of (b) Bi 4f, (c) O 1s, and (d) C 1s. | ||
The high-resolution Bi 4f spectrum of CAU-17 is shown in Fig. 4(b), which consists of two binding energy peaks at 158.08 eV and 163.18 eV corresponding to Bi3+ 4f7/2 and Bi3+ 4f5/2, respectively.26,51 These peaks confirm the existence of Bi3+ ions in the CAU-17 thin film.52 The O 1s spectrum displayed three peaks (Fig. 4(c)), where the intense peak at 530.08 eV corresponds to Bi–O bonding, while the low intensity peaks at 531.68 eV and 532.98 eV belong to the O–H bond in H2O and O in the bismuth-oxo clusters of the CAU-17 thin film, respectively.16,26,53 The C 1s spectrum of CAU-17, shown in Fig. 4(d), consists of three peaks at 284.38, 286.28, and 287.68 eV corresponding to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–C, and O–C
C, C–C, and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O of the H3BTC linker in the CAU-17 framework, respectively.26,54,55 These findings were also consistent with the aforementioned Raman and FT-IR spectra.
O of the H3BTC linker in the CAU-17 framework, respectively.26,54,55 These findings were also consistent with the aforementioned Raman and FT-IR spectra.
By considering the spectra of Bi 4f and O 1s, and also the structure of CAU-17 {9 Bi3+, 9 BTC3− and 9 H2O}, it was observed that the Bi3+ ions are coordinated with 9 O atoms in the prepared CAU-17 thin film.16,23 In this coordination system, 8 O atoms belong to the BTC3− ligands and 1 O atom from water molecules, resulting in the formation of a porous framework of CAU-17.21
Influence of synthesis temperature
Many researchers previously confirmed that temperature plays a major role in the synthesis of MOFs and has a great influence on their structure and morphology.56–58 Additionally, temperature significantly influences thermodynamics, reaction kinetics, and coordination chemistry of MOFs.59 The influence of synthesis temperature on CAU-17 formation was investigated by heating at 30 °C, 55 °C, 80 °C, 105 °C, and 130 °C, and the corresponding XRD results are depicted in Fig. 5. The synthesis of CAU-17 at 30 °C displayed two weak XRD signals at 8.66° and 12.98°, indicating a poor crystalline nature and an early stage of nucleation. Increasing the synthesis temperature to 55 °C results in improved peak intensity; however, the CAU-17 thin film still exhibits low crystallinity, displaying a sharp peak at 8.58° along with a weak signal at 10.69°, 13.15°, 25.08°, and 33.46°. The main reason behind this is that Bi3+ ions belong to the group of metal ions that possess high valence. It has previously been reported that synthesizing MOFs using trivalent-metal ions at room temperature is challenging and usually requires external energy for their formation.60,61 Due to this reason, the synthesized CAU-17 at a lower temperature displayed weak signals. Further increasing the temperature from 80 °C to 130 °C displayed the formation of several XRD peaks, suggesting a strong temperature-dependent formation of the CAU-17 MOF thin film. In this temperature range, all three spectra of CAU-17 were highly consistent with each other and displayed the most intense peaks of CAU-17 at 8.35°, 10.49°, 12.68°, 16.51°, 24.75°, and 33.13°.The FESEM images of CAU-17 thin films fabricated at different temperatures are shown in Fig. 6, which reveals that temperature has a significant impact on the formation of CAU-17 morphology. An increase in the temperature from 30 °C to 130 °C resulted in the transformation of small nanorods into larger nanorods. Fig. 6(f) shows the influence of temperature on the CAU-17 nanorods’ width, measured using ImageJ software. For the samples prepared at 30 to 80 °C, individual nanorods were analyzed. However, for samples synthesized at higher temperatures (105 °C to 130 °C), only a single nanorod was considered, where multiple measurements were taken for improving accuracy. With the increase in temperature from 30 °C to 130 °C, the width of nanorods was found to increase from 0.15 to 6.86 µm. Moreover, it was also observed that the growth of CAU-17 nanorods was random and non-directional.
The synthesis of CAU-17 at 30 °C displayed the formation of multiple nanorods, as shown in Fig. 6(a). Additionally, it was also observed that multiple nanorods arranged themselves in one specific crystallographic direction. In the intermediate temperature range of 55 to 80 °C, it was observed that multiple nanorods grew along the same direction, which further fused into large single nanorods at higher temperatures, as observed in Fig. 6(b–e). Such growth behavior belongs to “oriented attachment,” conceptualized by Penn and Banfield et al.62 According to this model, the same crystallographic-oriented nanoparticles (i.e., one-, two-, and three-dimensional) undergo fusion to form a single crystal.63 This process consists of three stages: (1) self-organization of adjacent nanoparticles towards a specific crystallographic direction; (2) the fusion of nanoparticles and formation of a smooth and continuous interface; and (3) maintaining nanoparticle alignment during the entire growth process.64,65 Similarly, the same trend was observed in the CAU-17 nanorod formation, as evidenced by the FESEM image shown in Fig. 6(a), where smaller nanorods aligned themselves towards the specific crystallographic direction at a relatively low temperature. The interaction between nanorods takes place via van der Waals forces, and due to their thermal energy, they try to arrange themselves towards a specific crystallographic direction to find the lowest energy configuration.63 As the thermal energy of the material increases with temperature, the nanorods align more quickly and gain more freedom to form larger-sized nanorods at higher temperatures (80 °C to 130 °C). Herein, the oriented attachment growth mechanism takes place due to the in situ growth of CAU-17 on the quartz substrate.
Effect of the substrate on CAU-17 formation
Previously, it was determined that the formation of MOFs directly on the substrate surface plays an important role in their growth along a specific direction and strongly affects their crystal structure and orientation.66,67 Falcaro et al. reported direct synthesis of a MOF from the metal–hydroxide surface by reacting it with an organic linker. The resultant MOF formation takes place via heteroepitaxial growth, displaying directional growth along different crystallographic axes.68 Similarly, Scheurle et al. emphasized the crystal orientation of MOF-74 via vapor-assisted conversion, synthesized using different metal ions, on different substrates, where different degrees of order were achieved depending on the substrate nature.69In this investigation, the formation of CAU-17 on the surface of different substrates was studied. Fig. 7(a) confirms the synthesis of CAU-17 on the FTO surface. The sharp intense peaks of CAU-17 emerged at 8.64°, 10.8°, 13°, 16.84°, 25.08°, and 33.48° while peaks at 27°, 34.16°, 38.2°, 51.92°, 54.96°, and 61.96° belong to the FTO substrate. The surface morphology of CAU-17 on the FTO surface is displayed in Fig. 7(b), showing the formation of well-defined nanorods. The XRD results of CAU-17 on the surface of carbon paper revealed four peaks at 8.31°, 10.5°, 12.7°, and 33.18°, while peaks at 18.22°, 26.64°, 42.38°, 44.64°, and 54.68° correspond to carbon paper, as shown in Fig. 7(c). The CAU-17 surface morphology on the carbon paper substrate is displayed in Fig. 7(d), showing the formation of nanorods. On both substrates, the synthesis of CAU-17 showed the formation of randomly oriented nanorods.
Proposed mechanism for CAU-17 thin film growth
The formation of Bi2O3 takes place via reactive DC magnetron sputtering through the following reaction (eqn (1)–(3)).| e− + Ar(g) → 2e− + Ar+ | (1) |
| Ar+ + Bi(s) → Bi0 + e− | (2) |
| 4Bi0 + 3O2 → 2Bi2O3 | (3) |
Firstly, the ionization of Ar ions takes place, which results in the formation of Ar+ ions. These ionized Ar+ ions further bombarded the surface of the pure Bi target, leading to the ejection of Bi0 atoms from the surface of the target. These atoms further interact with the reactive O atoms in the sputtering chamber and result in the formation of Bi2O3 by consolidating on the surface of the substrate. In this study, XRD analysis confirmed that the sputtered Bi2O3 thin film deposited on different substrates is amorphous in nature. This observation could be attributed to the deposition of Bi2O3 thin films at room temperature, low working gas pressures, and low deposition power.70 Additionally, Ibrahim et al.,71 and Hernández et al.,72 reported that under these conditions, DC reactive sputtering typically led to the formation of an amorphous Bi2O3 thin film.
The formation of CAU-17 involves three primary reactions: (1) dissolution of Bi3+ ions; (2) deprotonation of H3BTC; and (3) coordination reaction between Bi3+ ions and deprotonated H3BTC, as illustrated in Fig. 8. In this study, CAU-17 {Bi(BTC)(DMF)} is formed in the solution of DMF and H3BTC, where pH was ranged between 4.80 and 7.48 with the addition of 0.01 M HCl. Prior studies also revealed that the addition of HCl results in a speed-up of the rate of the reaction during MOF synthesis and ensures phase-purity and reproducibility of results.73,74 In the present work, the dissolution of Bi2O3 was carried out in a slightly acidic medium, as shown in eqn (4).
| Bi2O3 + 6H+ → 2Bi3+ + 3H2O | (4) |
In the synthesis of CAU-17, H3BTC plays the role of a structure-directing agent and acts as a hard base because of its carboxyl groups (–COOH) that bind with Bi3+ ions, forming a stable structure.27 The DMF solution functioned as a deprotonation agent.75 CAU-17 is formed when the deprotonation of H3BTC takes place in the DMF solution.76 The –COOH group of H3BTC lost its three protons (H+), resulting in the formation of a negatively charged carboxylate ion (–COO−).76,77 H3BTC is a type of multifunctional ligand that consist of a benzene ring with three carboxylate groups at 1,3, and 5 positions. It is known for its ability to construct the coordination architecture with metal ions through adopting different coordination modes due to its fully or partially deprotonation ability.78 Depending on the pH of the solution, H3BTC can be either fully or partially deprotonated.79 Typically, the partial deprotonation of H3BTC takes place at a pH of 5, and full deprotonation takes place at a pH of 7 and 9.80
The deprotonation of H3BTC in this investigation occurs in a two-step reaction (eqn (5) and (6)). Initially, partial deprotonation of H3BTC leads to the formation of HBTC2− ions, followed by full deprotonation that results in the formation of BTC3− ions.
| H3BTC + 2e− → HBTC2− + H2 | (5) |
| HBTC2− → BTC3− + H+ | (6) |
| Bi3+ + BTC3− → [Bi(BTC)](CAU-17) | (7) |
The charge difference of Bi3+ and BTC3− ions encourages the formation of the CAU-17 framework, where Bi3+ serves as a central metal ion that coordinates with the –COO− group of deprotonated H3BTC to form a coordination bond (eqn (7)).77
Conclusions
Herein, we reported a novel two-step sequential synthesis approach of the CAU-17 MOF thin film based on the controlled transformation of the Bi2O3 thin film into CAU-17. By employing advanced materials characterization techniques, the main constituent elements and functional groups in the synthesized thin film were successfully confirmed. This method enables the synthesis of CAU-17 thin films under relatively mild conditions without any risk of forming secondary phases and impurities, which was unachievable with previous powder-based techniques. This work also highlights the formation of CAU-17 at temperatures above and below 120 °C, which previous studies considered as an ideal temperature for its synthesis. The formation of CAU-17 on different substrates further increases its application prospects in fields like sensing, catalysis, and other cutting-edge technologies.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data that support the findings of this study are available from the corresponding author, Khaled Hassanein, upon reasonable request.Acknowledgements
The authors would like to acknowledge the support provided by the H2 Future Consortium at King Fahd University of Petroleum & Minerals (KFUPM) through the project H2FC2402.References
- J. M. Zamaro, N. C. Pérez, E. E. Miró, C. Casado, B. Seoane, C. Téllez and J. Coronas, Chem. Eng. J., 2012, 195–196, 180–187 CrossRef CAS
.
- K. S. Lin, A. K. Adhikari, C. N. Ku, C. L. Chiang and H. Kuo, Int. J. Hydrogen Energy, 2012, 37, 13865–13871 CrossRef CAS
.
- H. Furukawa, K. E. Cordova, M. O’Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef PubMed
.
- X. X. Yang, N. Li, C. Li, Z. Bin Jin, Z. Z. Ma, Z. G. Gu and J. Zhang, J. Am. Chem. Soc., 2024, 146, 16213–16221 CrossRef CAS PubMed
.
- N. Baig, I. Abdulazeez, B. Salhi, H. A. Asmaly, A. I. Osman and K. H. Ahmed, Chem. Eng. J., 2025, 504, 158628 CrossRef CAS
.
- M. Safaei, M. M. Foroughi, N. Ebrahimpoor, S. Jahani, A. Omidi and M. Khatami, TrAC, Trends Anal. Chem., 2019, 118, 401–425 CrossRef CAS
.
- R. Zhai, Y. Zhu, L. Chang, Z. Gu and J. Zhang, Jiegou Huaxue, 2022, 41, 2209074–2209079 CAS
.
- I. Ahmed, G. Lee, H. J. Lee and S. H. Jhung, Chem. Eng. J., 2024, 488, 151022 CrossRef CAS
.
- Z. Z. Ma, Q. H. Li, Z. Wang, Z. G. Gu and J. Zhang, Nat. Commun., 2022, 13, 6347 CrossRef CAS
.
- L. M. Chang, Q. H. Li, P. Weidler, Z. G. Gu, C. Wöll and J. Zhang, CCS, Chemistry, 2022, 4, 3472–3481 CAS
.
- J. Pang, Q. Han, W. Liu, Z. Shen, X. Wang and J. Zhu, Appl. Surf. Sci., 2017, 422, 283–294 CrossRef CAS
.
- V. M. Alexander, R. P. Bhat and S. D. Samant, Tetrahedron Lett., 2005, 46, 6957–6959 CrossRef CAS
.
- Y. Wang, H. Sun, Z. Yang, Y. Zhu and Y. Xia, Carbon Neutralization, 2024, 3, 737–767 CrossRef CAS
.
- Y. Ying, B. Khezri, J. Kosina and M. Pumera, ChemSusChem, 2021, 14, 3402–3412 CrossRef CAS
.
- A. K. Inge, M. Köppen, J. Su, M. Feyand, H. Xu, X. Zou, M. O’Keeffe and N. Stock, J. Am. Chem. Soc., 2016, 138, 1970–1976 CrossRef CAS PubMed
.
- F. Li, G. H. Gu, C. Choi, P. Kolla, S. Hong, T. S. Wu, Y. L. Soo, J. Masa, S. Mukerjee, Y. Jung, J. Qiu and Z. Sun, Appl. Catal., B, 2020, 277, 119241 CrossRef CAS
.
- L. Liu, K. Yao, J. Fu, Y. Huang, N. Li and H. Liang, Colloids Surf., A, 2022, 633, 127840 CrossRef CAS
.
- S. Li, W. Shan, T. Rao, Y. Qi, Y. Xiong, Z. Lou, H. Yu, J. Cui and X. Feng, Microchem. J., 2023, 191, 108861 CrossRef CAS
.
- D. Lee, K. S. Oh, Y. Lee, J. Jin, S. Y. Lee, Y. Jho and J. H. Park, Chem. Eng. J., 2024, 496, 153825 CrossRef CAS
.
- S. Frank, E. Svensson Grape, E. D. Bøjesen, R. Larsen, P. Lamagni, J. Catalano, A. K. Inge and N. Lock, J. Mater. Chem. A, 2021, 9, 26298–26310 RSC
.
- Q. Huang, X. Sha, R. Yang, H. Li and J. Peng, ACS Appl. Mater. Interfaces, 2024, 16, 13882–13892 CrossRef CAS PubMed
.
- M. Köppen, V. Meyer, J. Ångström, A. K. Inge and N. Stock, Cryst. Growth Des., 2018, 18, 4060–4067 CrossRef
.
- Q. X. Wang and G. Li, Inorg. Chem. Front., 2021, 8, 572–589 RSC
.
- M. Köppen, A. Dhakshinamoorthy, A. K. Inge, O. Cheung, J. Ångström, P. Mayer and N. Stock, Eur. J. Inorg. Chem., 2018, 3496–3503 CrossRef
.
- V. H. Nguyen, L. Van Tan, T. Lee and T. D. Nguyen, Sustainable Chem. Pharm., 2021, 20, 100385 CrossRef CAS
.
- S. Dong, L. Wang, W. Lou, Y. Shi, Z. Cao, Y. Zhang and J. Sun, Ultrason. Sonochem., 2022, 91, 106223 CrossRef CAS
.
- R. Huang, Z. Zhou, X. Lan, F. K. Tang, T. Cheng, H. Sun, K. Cham-Fai Leung, X. Li and L. Jin, Mater. Today Bio, 2023, 18, 100507 CrossRef CAS
.
- H. Yang, Y. Zhao, Y. Guo, B. Wu, Y. Ying, Z. Sofer and S. Wang, Small, 2025, 20, 2307484 CrossRef
.
- D. Chakraborty, A. Yurdusen, G. Mouchaham, F. Nouar and C. Serre, Adv. Funct. Mater., 2023, 34, 2309089 CrossRef
.
- D. Bazer-Bachi, L. Assié, V. Lecocq, B. Harbuzaru and V. Falk, Powder Technol., 2014, 255, 52–59 CrossRef CAS
.
- B. Huang, Y. Li and W. Zeng, Chemosensors, 2021, 9, 226 CrossRef CAS
.
- A. Baptista, F. Silva, J. Porteiro, J. Míguez and G. Pinto, Coatings, 2018, 8, 402 CrossRef
.
- Y. Wang, H. Chen, X. Wang, B. Meng, N. Yang, X. Tan and S. Liu, Ind. Eng. Chem. Res., 2020, 59, 15576–15585 CrossRef CAS
.
- X. Liu, M. Ding, P. Ma, C. Duan and J. Yao, Sep. Purif. Technol., 2022, 295, 121336 CrossRef CAS
.
- J. Liang, Q. Liu, T. Li, Y. Luo, S. Lu, X. Shi, F. Zhang, A. M. Asiri and X. Sun, Green Chem., 2021, 23, 2834–2867 RSC
.
- P. Borowski and J. Myśliwiec, Coatings, 2025, 15, 922 CrossRef CAS
.
- F. Wang, H. Zhao, J. Liang, T. Li, Y. Luo, S. Lu, X. Shi, B. Zheng, J. Du and X. Sun, J. Mater. Chem. A, 2020, 8, 20260–20285 RSC
.
- X. Tian, Z. Hu, C. Jia, H. Wang and X. Wei, J. Environ. Chem. Eng., 2023, 11, 111516 CrossRef CAS
.
- A. Monshi, M. R. Foroughi and M. R. Monshi, World J. Nano Sci. Eng., 2012, 02, 154–160 CrossRef
.
- P. Lunca Popa, S. Sønderby, S. Kerdsongpanya, J. Lu, N. Bonanos and P. Eklund, J. Appl. Phys., 2013, 113, 046101 CrossRef
.
- B. Zhang, S. Cao, Y. Wu, P. Zhai, Z. Li, Y. Zhang, Z. Fan, C. Wang, X. Zhang, J. Hou and L. Sun, ChemElectroChem, 2021, 8, 880–886 CrossRef CAS
.
- H. A. Le Pham, D. T. Nguyen, V. C. Nguyen and T. Ky Vo, Inorg. Chem. Commun., 2024, 159, 111822 CrossRef
.
- S. A. Siddiqui, A. Prado-Roller and H. Shiozawa, Mater. Adv., 2022, 3, 224–231 RSC
.
- W. Moloto, P. Mbule, E. Nxumalo and B. Ntsendwana, J. Photochem. Photobiol., A, 2021, 407, 113063 CrossRef CAS
.
- H. Sun, X. Han, K. Liu, B. Shen, J. Liu, D. Wu and X. Shi, Ind. Eng. Chem. Res., 2017, 56, 9541–9550 CrossRef CAS
.
- L. Yaqoob, T. Noor, N. Iqbal, H. Nasir, M. Sohail, N. Zaman and M. Usman, Renew, Energy, 2020, 156, 1040–1054 CAS
.
- S. R. Zhu, M. K. Wu, W. N. Zhao, P. F. Liu, F. Y. Yi, G. C. Li, K. Tao and L. Han, Cryst. Growth Des., 2017, 17, 2309–2313 CrossRef CAS
.
- F. Khosravi, M. Gholinejad, J. M. Sansano and R. Luque, Appl. Organomet. Chem., 2022, 36, 6749 CrossRef
.
- S. Karimi, M. Gholinejad, R. Khezri, J. M. Sansano, C. Nájera and M. Yus, RSC Adv., 2023, 13, 8101–8113 RSC
.
- S. Han, R. A. Ciufo, M. L. Meyerson, B. K. Keitz and C. Buddie Mullins, J. Mater. Chem. A, 2019, 7, 19396–19406 RSC
.
- J. Sun, W. Zheng, S. Lyu, F. He, B. Yang, Z. Li, L. Lei and Y. Hou, Chin. Chem. Lett., 2020, 31, 1415–1421 CrossRef CAS
.
- P. Zhang, Y. Huang, Y. Rao, M. Chen, X. Li, W. Ho, S. Lee and J. Cao, Chem. Eng. J., 2021, 406, 126910 CrossRef CAS
.
- L. Zhao, Z. Pan, L. Cai, S. Wang, B. Lu, S. Lv, Y. Qiu and G. Wang, Desalination, 2024, 576, 117360 CrossRef CAS
.
- M. Stawowy, P. Jagódka, K. Matus, B. Samojeden, J. Silvestre-Albero, J. Trawczyński and A. Łamacz, Catalysts, 2020, 10, 108 CrossRef CAS
.
- C. Fan, H. Dong, Y. Liang, J. Yang, G. Tang, W. Zhang and Y. Cao, J. Cleaner Prod., 2019, 208, 353–362 CrossRef CAS
.
- W. Xuan, R. Ramachandran, C. Zhao and F. Wang, J. Solid State Electrochem., 2018, 22, 3873–3881 CrossRef CAS
.
- G. Zahn, P. Zerner, J. Lippke, F. L. Kempf, S. Lilienthal, C. A. Schröder, A. M. Schneider and P. Behrens, CrystEngComm, 2014, 16, 9198–9207 RSC
.
- P. Mahata, A. Sundaresan and S. Natarajan, Chem. Commun., 2007, 4471–4473 RSC
.
- Y. X. Sun and W. Y. Sun, Chin. Chem. Lett., 2014, 25, 823–828 CrossRef CAS
.
- P. Zhang, X. Kang, L. Tao, L. Zheng, J. Xiang, R. Duan, J. Li, P. Chen, X. Xing, G. Mo, Z. Wu and B. Han, CCS Chem., 2023, 5, 1462–1469 CrossRef CAS
.
- S. Dai, F. Nouar, S. Zhang, A. Tissot and C. Serre, Angew. Chem., Int. Ed., 2021, 60, 4282–4288 CrossRef CAS
.
- R. L. Penn and J. F. Banfield, Geochim. Cosmochim. Acta, 1999, 63, 1549–1557 CrossRef CAS
.
- Q. Zhang, S. J. Liu and S. H. Yu, J. Mater. Chem., 2009, 19, 191–207 RSC
.
- F. Cheng, L. Lian, L. Li, J. Rao, C. Li, T. Qi, Z. Zhang, J. Zhang and Y. Gao, Adv. Sci., 2019, 6, 1802202 CrossRef
.
- K. Wen and W. He, Nanotechnology, 2015, 26, 382001 CrossRef
.
- J. Park, H. R. Moon and J. Y. Kim, Mater. Chem. Front., 2023, 7, 5545–5560 RSC
.
- M. M. Sabzehmeidani, S. Gafari, S. jamali and M. Kazemzad, Appl. Mater. Today, 2024, 38, 102153 CrossRef
.
- P. Falcaro, K. Okada, T. Hara, K. Ikigaki, Y. Tokudome, A. W. Thornton, A. J. Hill, T. Williams, C. Doonan and M. Takahashi, Nat. Mater., 2017, 16, 342–348 CrossRef CAS PubMed
.
- P. I. Scheurle, A. Mähringer, A. Biewald, A. Hartschuh, T. Bein and D. D. Medina, Chem. Mater., 2021, 33, 5896–5904 CrossRef CAS
.
- K. Wang, Z. Cheng, G. Wu and Q. Qin, ChemCatChem, 2024, 16, 202400415 CrossRef
.
- S. Ibrahim, P. Bonnet, M. Sarakha, C. Caperaa, G. Monier and A. Bousquet, Mater. Chem. Phys., 2020, 243, 122580 CrossRef CAS
.
- G. Orozco-Hernández, J. Olaya-Flórez, C. Pineda-Vargas, J. E. Alfonso and E. Restrepo-Parra, Surf. Interfaces, 2020, 21, 100627 CrossRef
.
- F. Bigdeli, M. N. A. Fetzer, B. Nis, A. Morsali and C. Janiak, J. Mater. Chem. A, 2023, 11, 22105–22131 RSC
.
- R. S. Forgan, Chem. Sci., 2020, 11, 4546–4562 RSC
.
- R. Ediati, M. A. Setyani, D. O. Sulistiono, E. Santoso, D. Hartanto and M. M. Al Bakri Abdullah, J. Water Process Eng., 2021, 39, 101670 CrossRef
.
- Y. Devi, I. Ang, F. E. Soetaredjo, S. P. Santoso, W. Irawaty, M. Yuliana, A. E. Angkawijaya, S. B. Hartono, P. L. Tran-Nguyen, S. Ismadji and Y. H. Ju, J. Mater. Sci., 2020, 55, 13785–13798 CrossRef CAS
.
- J. Bai, J. H. Jia, Y. Wang, C. C. Yang and Q. Jiang, Nano-Micro Lett., 2025, 17, 60 CrossRef
.
- T. Zelenka, M. Baláž, M. Férová, P. Diko, J. Bednarčík, A. Királyová, Ľ. Zauška, R. Bureš, P. Sharda, N. Király, A. Badač, J. Vyhlídalová, M. Želinská and M. Almáši, Sci. Rep., 2024, 14, 15386 CrossRef CAS PubMed
.
- Y. F. Zhou, B. Y. Lou, D. Q. Yuan, Y. Q. Xu, F. L. Jiang and M. C. Hong, Inorg. Chim. Acta, 2005, 358, 3057–3064 CrossRef CAS
.
- R. Seetharaj, P. V. Vandana, P. Arya and S. Mathew, Arabian J. Chem., 2019, 12, 295–315 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |