Open Access Article
Open Access ArticleWater-soluble acylhydrazone macrocycles as potent reversal agents for cisatracurium-induced neuromuscular blockade
Yongfei
Yin†
c,
Yangyang
Ge†
b,
Qian
Li†
a,
Yi
Chen
a,
Siyuan
Zhou
a,
He
Deng
a,
Shigui
Chen
 *a,
Yun
Lin
*b and
Lu
Wang
*a,
Yun
Lin
*b and
Lu
Wang
 *a
*a
aThe Institute for Advanced Studies, Hubei Key Lab on Organic and Polymeric Opto-Electronic Materials, Wuhan University, 299 Bayi Road, Wuhan, Hubei 430072, China. E-mail: sgchen@whu.edu.cn; wanglu-027@whu.edu.cn
bDepartment of Anesthesiology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Jiefang Road 1277, Wuhan, Hubei 430022, China. E-mail: franklinyun@hust.edu.cn
cBiomass Oligosaccharides Engineering Technology Research Center of Anhui Province, School of Chemistry and Material Engineering, Fuyang Normal University, 100 Qinghe West Road, Fuyang, Anhui 236000, China
First published on 13th March 2026
Abstract
Cisatracurium is a widely used non-depolarizing neuromuscular blocking agent during anesthesia; however, residual neuromuscular blockade post-surgery can lead to severe respiratory complications, highlighting the urgent need for effective reversal agents. In this study, we designed and synthesized a highly water-soluble acylhydrazone macrocycle featuring an electron-rich cavity as an efficient supramolecular reversal agent for cisatracurium. The macrocycle binds cisatracurium through electrostatic and hydrophobic interactions, exhibiting a binding constant of up to 105 M−1. Cytotoxicity assays confirmed its excellent biocompatibility, and in vivo studies in cisatracurium-anesthetized mice demonstrated that the macrocycle effectively reverses cisatracurium-induced neuromuscular blockade. This work provides a new strategy for developing water-soluble acylhydrazone macrocycles as potential supramolecular reversal agents for neuromuscular blockers.
Introduction
In clinical anesthesia practice, neuromuscular blocking agents (NMBAs) are among the most frequently administered drugs prior to surgery.1 Intravenous administration of NMBAs induces skeletal muscle relaxation by blocking neuromuscular transmission, thereby reducing muscle reflex-related interference during surgical manipulation, improving operative conditions, facilitating tracheal intubation, and providing better visualization for procedures involving the thoracic or abdominal cavities. Rapid and complete reversal of neuromuscular block at the end of surgery is essential for the timely recovery of spontaneous respiration and for reducing the risk of residual neuromuscular block (RNMB), making it a critical component of anesthetic management.2Traditional reversal agents are primarily acetylcholinesterase inhibitors, such as neostigmine and edrophonium.3–5 These agents inhibit the breakdown of acetylcholine, increasing its concentration at the neuromuscular junction and enhancing its ability to outcompete NMBAs for binding to nicotinic acetylcholine receptors (nAChRs), thereby reversing neuromuscular block. However, their reversal efficacy is limited, and they are often associated with adverse effects including bradycardia and bronchospasm.6 In recent years, with the rapid progress of supramolecular chemistry,7–10 “supramolecular reversal agents” based on host–guest interactions have emerged as a promising strategy for NMBA reversal.11–14 Macrocyclic hosts such as cyclodextrins, pillararenes,15–21 cucurbiturils,22–26 and calixarenes27,28 have been explored for the design of agents capable of selectively binding and clearing NMBAs. Among them, the γ-cyclodextrin derivative Sugammadex represents a major clinical breakthrough,29 efficiently reversing aminosteroid NMBAs such as rocuronium (Roc), vecuronium (Vec), and pancuronium (Pan) through inclusion complexation. Nevertheless, the cavity size of cyclodextrins limits their ability to encapsulate bulkier neuromuscular blockers. As a result, Sugammadex shows poor efficacy against larger and more rigid NMBAs such as cisatracurium (Cis) and other benzylisoquinolinium compounds.30 Notably, Cis is widely used in intensive care units and is considered the NMBA of choice for patients at high risk of acute respiratory distress syndrome (ARDS), due to its organ-independent metabolism and minimal histamine release.31 Therefore, there is an urgent clinical need for a supramolecular antagonist capable of rapidly and efficiently reversing Cis-induced neuromuscular block.
Hydrogen-bonded aromatic amide/hydrazone macrocycles constitute an emerging class of cyclic compounds constructed from consecutive intramolecular hydrogen bonds and aromatic units.32–34 Owing to their high conformational rigidity, tunable structural features, readily functionalizable molecular frameworks, electron-rich cavities, and versatile host–guest recognition capabilities, these macrocycles have been extensively explored and applied in diverse research areas, including supramolecular vesicles,35 artificial ion channels,36,37 gels,38–40 supramolecular catalysis,41–43 and artificial molecular machines.44 Importantly, these properties also suggest the potential of aromatic amide/hydrazone macrocycles as supramolecular antagonists for NMBAs, yet, to the best of our knowledge, no such application has been reported to date.
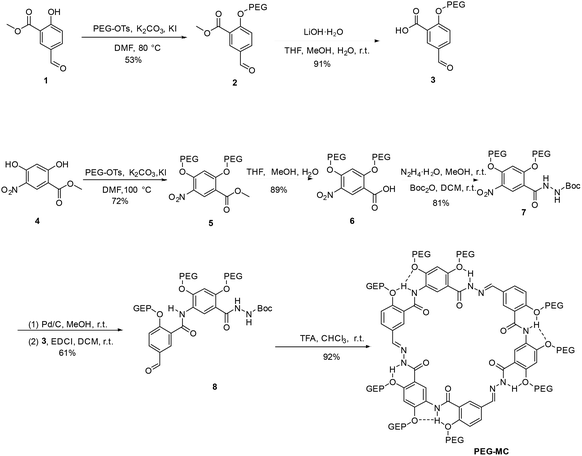
Herein, we report the design and synthesis of a water-soluble hydrazone-based macrocyclic compound, PEG-MC, featuring a large electron-rich cavity and a rigid framework. The compound selectively binds the bulky Cis molecule via host–guest recognition, exhibiting a high binding constant of 5.58 × 105 M−1 in aqueous solution. In vivo, PEG-MC markedly accelerates recovery from Cis-induced neuromuscular blockade (NMB), shortening the time to spontaneous respiration from 78.8 s to 51.5 s. These results establish PEG-MC as a highly effective supramolecular antagonist and exemplify the potential of rational macrocyclic design for developing next-generation reversal agents for neuromuscular blockers (Scheme 1).
Experimental section
Cell culture
293 T cells were incubated in complete culture medium comprising DMEM supplemented with 10% FBS, 100 IU mL−1 penicillin, and 100 mg mL−1 streptomycin, and then cultured at 37 °C in a humidified atmosphere with 5% CO2. The attached cells were routinely passaged with a 0.25% trypsin-EDTA solution after reaching 80% confluency. The medium was replaced every 3 days.Cytotoxicity experiments
To evaluate the cytotoxicity of PEG-MC, an MTT cell proliferation and cytotoxicity assay kit (Beyotime Biotechnology) was used for toxicity studies. Following cell digestion and counting, 5000 cells per well were seeded in a 96-well plate. After a 2-day culture period, PEG-MC was added to six wells designated for each drug concentration. Following a 24 h incubation period, 10 µL of MTT solution (0.5 mg mL−1) was added to each well for a 4 h incubation. Subsequently, 100 µL of formazan solution was added to dissolve the dark blue formazan crystals. The absorbance was measured at 570 nm using a microplate reader. The cell viability rate is presented as a percentage ratio between the absorbance of the treated group and that of the control group.Respiratory monitoring
C57BL/6 mice (8 weeks old; body weight 24.0–26.0 g) were used in this study. All drugs were administered via tail vein injection. The animals were randomly assigned to two groups: (1) the Cis group, in which Cis was prepared as an aqueous solution at a concentration of 1.0 mg mL−1 and administered at an injection volume of 1 mL; and (2) the Cis + PEG-MC premix group, in which PEG-MC was fully dissolved in the Cis aqueous solution to obtain a formulation containing 1.0 mg mL−1Cis and 2.13 mg mL−1PEG-MC, with an injection volume of 1 mL. All formulations were stored at 4 °C prior to use. The entire experimental procedure was video-recorded to enable continuous monitoring of diaphragmatic movements. The time interval between the cessation of diaphragmatic movement and the first observable reappearance of movement was measured and defined as the spontaneous breathing recovery time (s). Animals were excluded from the analysis if drug extravasation or injection failure occurred during tail vein administration. All animals received care in compliance with the guidelines outlined in the Guide for the Care and Use of Laboratory Animals. All of the animal experiments were approved by the Institutional Animal Care and Use Committee of Huazhong University of Science and Technology ([2023] IACUC Number: 4887).Statistical analysis
The experimental data were expressed as mean ± standard error (mean ± SEM) using GraphPad and Origin SPSS software was used for statistical analysis. Comparison between the two groups was performed using a paired t-test, and P < 0.05 was considered statistically significant (****: P < 0.0001; ***: P < 0.001; **: P < 0.01; and *: P < 0.05).Results and discussion
Host–guest interactions between Cis and C8-MC
To verify the potential of the acylhydrazone macrocycle as a supramolecular antagonist for cis-atracurium Cis, a lipophilic acylhydrazone macrocycle bearing an octyl chain (C8-MC) was designed and synthesized. First, the host–guest interactions between C8-MC and Cis in the organic phase were investigated by 1H NMR titration experiments in a CDCl3/DMSO-d6 (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) solvent system (Fig. 1a and Fig. S1). The results showed that with the gradual increase of the Cis concentration, significant changes in both the peak shape and chemical shift were observed for the characteristic proton signals of C8-MC and Cis, indicating the occurrence of distinct host–guest interactions between them. Furthermore, the chemical shift variations (Δδ = δobsd − δfree) of the characteristic protons in Cis were analyzed after mixing C8-MC and Cis at a 1
1, v/v) solvent system (Fig. 1a and Fig. S1). The results showed that with the gradual increase of the Cis concentration, significant changes in both the peak shape and chemical shift were observed for the characteristic proton signals of C8-MC and Cis, indicating the occurrence of distinct host–guest interactions between them. Furthermore, the chemical shift variations (Δδ = δobsd − δfree) of the characteristic protons in Cis were analyzed after mixing C8-MC and Cis at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio (Fig. 1b). It was found that the signals of Ha and Hc protons on the quaternary ammonium head group of Cis shifted downfield due to deshielding effects, while the signals of Hd, He, and Hf protons on the alkyl chain shifted upfield because of shielding effects. These observations suggest that the Ha and Hc protons of Cis are located deep inside the cavity of C8-MC, whereas Hd, He, and Hf are positioned near the rim of the cavity. In addition, the continuous variation method (Job's plot) confirmed a 1
1 molar ratio (Fig. 1b). It was found that the signals of Ha and Hc protons on the quaternary ammonium head group of Cis shifted downfield due to deshielding effects, while the signals of Hd, He, and Hf protons on the alkyl chain shifted upfield because of shielding effects. These observations suggest that the Ha and Hc protons of Cis are located deep inside the cavity of C8-MC, whereas Hd, He, and Hf are positioned near the rim of the cavity. In addition, the continuous variation method (Job's plot) confirmed a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding stoichiometry between C8-MC and Cis. The association constant (Ka) was determined to be 4.40 × 103 M−1 by nonlinear curve fitting (Fig. S2).
1 binding stoichiometry between C8-MC and Cis. The association constant (Ka) was determined to be 4.40 × 103 M−1 by nonlinear curve fitting (Fig. S2).
Density functional theory (DFT) calculations were performed to provide deeper insight into the host–guest interactions and the binding mechanism between C8-MC and Cis by analyzing the structural characteristics of the complex. To simplify the computational model, a macrocycle with an identical framework (MC) was employed as a representative analog of C8-MC (Fig. 1c). The optimized geometry revealed that the originally symmetric structure of Cis became significantly
distorted upon complexation and penetrated through the cavity of MC (Fig. 1d and Fig. S3), indicating a pronounced host–guest interaction. Moreover, the Ha and Hc protons of Cis were located deep within the cavity of MC, which is consistent with the results obtained from the 1H NMR titration experiments. Further electronic structure analysis was conducted by calculating the highest occupied molecular orbital (HOMO), lowest unoccupied molecular orbital (LUMO) (Fig. S4), and electrostatic potential (ESP) distributions of the MC⊃Cis complex (Fig. S5). The ESP mapping showed that the cavity of MC possessed a strongly negative potential, whereas the quaternary ammonium head of Cis exhibited an evident positive potential. This electrostatic complementarity facilitates the formation of a stable complex through ion–dipole and other noncovalent interactions. In addition, both the HOMO and LUMO distributions of the host and guest molecules exhibited noticeable changes after complexation, further suggesting electronic interactions between them.
To visualize the noncovalent interactions (NCIs) between MC and Cis, Multiwfn and VMD software were employed, and the interaction patterns were analyzed using the independent gradient model (IGM) approach. The resulting NCI plots clearly illustrated multiple ion–dipole interactions between MC and Cis (Fig. 1e), confirming that the host–guest interaction is primarily driven by ion–dipole and other noncovalent forces.
Host–guest interactions between Cis and PEG-MC
Acylhydrazone macrocycles (MC) were demonstrated to accommodate the relatively large anesthetic molecule Cis. To further improve the water solubility of the aromatic acylhydrazone macrocycle and enable its application in more complex biological systems, nine polyethylene glycol (PEG) chains were introduced onto the macrocyclic backbone, leading to the successful synthesis of the water-soluble PEG-functionalized acylhydrazone macrocycle (PEG-MC) (Scheme 2).Subsequently, the host–guest interactions between PEG-MC and Cis in aqueous solution were investigated using 1H NMR spectroscopy. Due to pronounced aggregation of PEG-MC in water, the 1H NMR spectrum of the macrocycle was complex, and the assignment of its protons was challenging (Fig. S6). Nevertheless, upon gradual addition of Cis to a D2O solution of PEG-MC, significant changes were observed in the characteristic proton signals of Cis (Fig. S7). Normalized chemical shift changes (Δδ = δobsd − δbound) were plotted to analyze the proton shift trends of Cis as its concentration increased in the PEG-MC solution (Fig. S8). The results revealed that the chemical shifts of the protons on the alkyl chain of Cis (Hd, He, Hf) changed most significantly (Δδ = −0.025, −0.106, −0.052 ppm), whereas the aromatic proton Ha exhibited a minimal shift (Δδ = −0.003 ppm). Notably, the pronounced shift of He suggests that the alkyl chain of Cis is deeply embedded within the electron-rich cavity of PEG-MC, while the aromatic ring remains positioned near the cavity periphery.
The host–guest interactions and assembly mechanism of PEG-MC with Cis in aqueous solution were similarly examined through DFT calculations and structural optimizations. To simplify the computational model, MC was used as a representative. The optimized structure of the MC⊃Cis complex revealed that the macrocyclic backbone of MC exhibits slight bending and is positioned near the quaternary ammonium head of Cis (Fig. S9), indicating a pronounced host–guest interaction. Notably, the He proton of Cis is located deep within the cavity of MC, whereas the Ha proton resides near the cavity periphery, consistent with the previously obtained 1H NMR titration results.
Further electronic structure analysis was conducted by examining the HOMO, LUMO, and ESP distributions. In the free state, the HOMO and LUMO of MC are mainly localized on the benzene rings of the macrocyclic backbone, while those of Cis are concentrated on its aromatic head. Upon complexation, significant changes in both the HOMO and LUMO distributions were observed (Fig. S10), further confirming electronic interactions between the host and guest. The ESP mapping showed that the cavity of MC is strongly negative, whereas the quaternary ammonium head of Cis exhibits pronounced positive potential (Fig. S11), facilitating stable complex formation through electrostatic and other noncovalent interactions. Additionally, charge transfer from Cis to MC results in an increase in local positive charge density on MC, suggesting the presence of cation–π interactions. Finally, noncovalent interaction (NCI) analysis revealed that in aqueous solution (Fig. S12), Cis penetrates the cavity of MC, and multiple noncovalent interactions exist between the quaternary ammonium head of Cis and the macrocyclic backbone of MC, leading to the formation of a stable 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest complex. Overall, the host–guest association between MC and Cis is primarily driven by cation–π and hydrophobic interactions.
1 host–guest complex. Overall, the host–guest association between MC and Cis is primarily driven by cation–π and hydrophobic interactions.
The host–guest interactions between PEG-MC and Cis in aqueous solution were further investigated using UV-visible spectroscopy (UV-vis) and isothermal titration calorimetry (ITC) experiments. As shown in Fig. 2a, upon gradual addition of Cis to the PEG-MC aqueous solution, the characteristic absorption band of the acylhydrazone macrocycle at 315 nm decreased markedly in intensity. A distinct inflection point was observed at 1.0 equivalent of Cis (Fig. 2b), indicating a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest stoichiometry between PEG-MC and Cis. Nonlinear least-squares fitting afforded an association constant (Ka) of 5.98 × 105 M−1 (Fig. S13). Consistently, ITC measurements confirmed the 1
1 host–guest stoichiometry between PEG-MC and Cis. Nonlinear least-squares fitting afforded an association constant (Ka) of 5.98 × 105 M−1 (Fig. S13). Consistently, ITC measurements confirmed the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation between PEG-MC and Cis, with a binding constant of 5.58 × 105 M−1, in good agreement with the UV–vis titration results. The corresponding thermodynamic parameters were determined as ΔG = −7.87 kcal mol−1, ΔH = −0.530 kcal mol−1, and ΔS = −2.46 × 10−2 kcal mol−1 K−1. These results suggest that the formation of the Cis⊂PEG-MC complex is primarily enthalpy-driven and exhibits high binding stability. Moreover, the pronounced exothermic peaks observed in the ITC thermograms imply that a certain degree of aggregation occurs during the complexation process.45 To verify this hypothesis, the self-assembly behavior of Cis⊂PEG-MC was further characterized by dynamic light scattering (DLS) and transmission electron microscopy (TEM). DLS analysis revealed that PEG-MC alone exhibited a hydrodynamic diameter (Dh) centered at approximately 531 nm with a polydispersity index (PDI) of 0.2, whereas the addition of an equimolar amount of Cis increased Dh to about 955 nm with a PDI of 0.4 (Fig. S15). To further evaluate the stability of PEG-MC in aqueous media, its particle size distribution was monitored over time. After storage at room temperature for 10 days, the Dh of PEG-MC increased slightly to approximately 610 nm, with a PDI of 0.38 (Fig. S16). Although small increases in particle size and dispersity were observed, no significant large-scale aggregation or precipitation occurred, indicating that the system exhibits good stability. TEM images showed that PEG-MC self-assembled into spherical nanoparticles with an average diameter of ∼451 nm, while Cis⊂PEG-MC formed larger spherical nanoparticles with an average diameter of ∼1106 nm (Fig. S17). Taken together, these findings demonstrate that the host–guest interactions between Cis and PEG-MC significantly promote further aggregation and hierarchical self-assembly of PEG-MC, leading to the formation of larger supramolecular nanoparticles in aqueous solution.
1 complexation between PEG-MC and Cis, with a binding constant of 5.58 × 105 M−1, in good agreement with the UV–vis titration results. The corresponding thermodynamic parameters were determined as ΔG = −7.87 kcal mol−1, ΔH = −0.530 kcal mol−1, and ΔS = −2.46 × 10−2 kcal mol−1 K−1. These results suggest that the formation of the Cis⊂PEG-MC complex is primarily enthalpy-driven and exhibits high binding stability. Moreover, the pronounced exothermic peaks observed in the ITC thermograms imply that a certain degree of aggregation occurs during the complexation process.45 To verify this hypothesis, the self-assembly behavior of Cis⊂PEG-MC was further characterized by dynamic light scattering (DLS) and transmission electron microscopy (TEM). DLS analysis revealed that PEG-MC alone exhibited a hydrodynamic diameter (Dh) centered at approximately 531 nm with a polydispersity index (PDI) of 0.2, whereas the addition of an equimolar amount of Cis increased Dh to about 955 nm with a PDI of 0.4 (Fig. S15). To further evaluate the stability of PEG-MC in aqueous media, its particle size distribution was monitored over time. After storage at room temperature for 10 days, the Dh of PEG-MC increased slightly to approximately 610 nm, with a PDI of 0.38 (Fig. S16). Although small increases in particle size and dispersity were observed, no significant large-scale aggregation or precipitation occurred, indicating that the system exhibits good stability. TEM images showed that PEG-MC self-assembled into spherical nanoparticles with an average diameter of ∼451 nm, while Cis⊂PEG-MC formed larger spherical nanoparticles with an average diameter of ∼1106 nm (Fig. S17). Taken together, these findings demonstrate that the host–guest interactions between Cis and PEG-MC significantly promote further aggregation and hierarchical self-assembly of PEG-MC, leading to the formation of larger supramolecular nanoparticles in aqueous solution.
Experiment for reversal of Cisin vivo
Given the strong binding affinity of PEG-MC toward Cis, we further investigated its ability to reverse neuromuscular blockade in vivo. Prior to the animal studies, the cytotoxicity of PEG-MC was assessed using the MTT assay in human embryonic kidney 293T cells. The results showed that PEG-MC exhibited negligible cytotoxicity, likely owing to the presence of eight hydrophilic PEG chains. Even at a relatively high concentration of 80 µM, the cell viability remained above 80% (Fig. S18), indicating that PEG-MC possesses good biocompatibility.Subsequently, rats (n = 4) were anesthetized with isoflurane and tracheally intubated for mechanical ventilation and respiratory monitoring to assess the neuromuscular blockade reversal effect of PEG-MC. The results showed that the recovery time of spontaneous respiration in the control group was 78.8 seconds, while intravenous injection of PEG-MC significantly reduced the recovery time to 51.5 seconds (Fig. 3a). These results indicate that PEG-MC can strongly bind free Cis molecules in vivo, thereby competing with the binding sites of nAChRs on the postsynaptic membrane and effectively reversing the neuromuscular transmission blockade caused by Cis (Fig. 3b). PEG-MC reduces the effective concentration of Cis in the body, thereby weakening or even eliminating its inhibitory effect on synaptic transmission and promoting the rapid recovery of spontaneous respiration in anesthetized animals. In summary, PEG-MC exhibited excellent biocompatibility and remarkable in vivo efficacy in reversing Cis-induced neuromuscular blockade, demonstrating its potential as a supramolecular antidote for Cis.
Conclusions
In conclusion, we successfully synthesized the water-soluble hydrazone macrocycle PEG-MC. Detailed characterization demonstrated that PEG-MC forms a stable 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 pseudorotaxane-type complex with Cis, featuring a strong host–guest interaction with an association constant on the order of 105 M−1. In vitro studies confirmed its excellent biocompatibility, and in vivo experiments further revealed that PEG-MC effectively reverses Cis-induced neuromuscular blockade in mice, shortening the recovery time of spontaneous respiration from 78.8 s to 51.5 s. Collectively, these findings validate PEG-MC as a promising candidate for clinically reversing Cis and highlight the broader potential of supramolecular therapeutic strategies in advancing biomedical interventions, particularly in anesthesia and the reversal of neuromuscular blockade.
1 pseudorotaxane-type complex with Cis, featuring a strong host–guest interaction with an association constant on the order of 105 M−1. In vitro studies confirmed its excellent biocompatibility, and in vivo experiments further revealed that PEG-MC effectively reverses Cis-induced neuromuscular blockade in mice, shortening the recovery time of spontaneous respiration from 78.8 s to 51.5 s. Collectively, these findings validate PEG-MC as a promising candidate for clinically reversing Cis and highlight the broader potential of supramolecular therapeutic strategies in advancing biomedical interventions, particularly in anesthesia and the reversal of neuromuscular blockade.
Author contributions
Y. F. Yin: formal analysis, investigation, data curation, and writing of the original draft. Y. Y Ge: investigation and data curation. Q. Li: investigation, resources, and data curation. Y. Chen: compound synthesis, investigation, and data curation. S. Y. Zhou: compound synthesis, investigation, resources, and data curation. H. Deng: compound synthesis and investigation. S. G. Chen: conceptualization, writing – review and editing, supervision, project administration, and funding acquisition. Y. Lin: conceptualization, writing – review and editing, supervision, project administration, and funding acquisition. L. Wang: conceptualization, writing – review and editing, supervision, project administration, and funding acquisition.Conflicts of interest
The authors declare no conflict of interest.Data availability
All relevant data are within the manuscript and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tb02867a.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22371218, 21702153, 52270070 and 21801194) and the Youth Program of Educational Commission of Anhui Province of China (2025AHGXZK40705). We thank the support of the Core Facility of Wuhan University and the Large-scale Instrument and Equipment Sharing Foundation of Wuhan University.Notes and references
- J. M. Hunter and M. Blobner, Br. J. Anaesth., 2024, 132, 461–465 CrossRef CAS PubMed.
- C. G. Stäuble and M. Blobner, Curr. Opin. Anaesthesiol., 2020, 33, 490–498 CrossRef PubMed.
- N. J. Harper, E. G. Bradshaw and T. E. Healy, Br. J. Anaesth., 1984, 56, 1089–1094 CrossRef CAS PubMed.
- J. L. D. Thomsen, A. K. Staehr-Rye, O. Mathiesen, D. Hägi-Pedersen and M. R. Gätke, Anaesthesia, 2020, 75, 1164–1172 CrossRef CAS PubMed.
- M. Sanfilippo, V. Vilardi, G. Fierro, G. Rosa, P. Pelaia and A. Gasparetto, Acta Anaesthesiol. Scand., 1988, 32, 437–440 CrossRef CAS PubMed.
- H.-C. Chang, S.-Y. Liu, M.-J. Lee, S.-O. Lee and C.-S. Wong, J. Formosan Med. Assoc., 2022, 121, 2639–2643 CrossRef CAS PubMed.
- D. B. Amabilino and P. A. Gale, Chem. Soc. Rev., 2017, 46, 2376–2377 RSC.
- I. Alfonso, Chem. Commun., 2024, 60, 9692–9703 RSC.
- A. A. H. Laporte and J. N. H. Reek, Chem. Rev., 2025, 125, 7223–7274 CrossRef CAS PubMed.
- C. Liu, Y. Jin, J. Li, Z. Wang, J. Wang and W. Tian, Chin. J. Chem., 2025, 43, 2053–2068 CrossRef CAS.
- Z. Wang, H. Yin and R. Wang, J. Med. Chem., 2025, 68, 6955–6957 CrossRef CAS PubMed.
- C.-L. Deng, S. L. Murkli and L. D. Isaacs, Chem. Soc. Rev., 2020, 49, 7516–7532 RSC.
- J. M. Adam, D. J. Bennett, A. Bom, J. K. Clark, H. Feilden, E. J. Hutchinson, R. Palin, A. Prosser, D. C. Rees, G. M. Rosair, D. Stevenson, G. J. Tarver and M.-Q. Zhang, J. Med. Chem., 2002, 45, 1806–1816 CrossRef CAS PubMed.
- H. Yin, X. Zhang, J. Wei, S. Lu, D. Bardelang and R. Wang, Theranostics, 2021, 11, 1513–1526 CrossRef CAS PubMed.
- W. Zhang, E. A. Bazan-Bergamino, A. P. Doan, X. Zhang and L. Isaacs, Chem. Commun., 2024, 60, 4350–4353 RSC.
- Q. Zhao, J. Zhu, Y. Chen, H. Dong, S. Zhou, Y. Yin, Q. Cai, S. Chen, C. Chen and L. Wang, J. Hazard. Mater., 2024, 469, 133875 CrossRef CAS PubMed.
- Z. Fang, Z. Zhang, R. Wang, S. Li, S. Lin, Y. Zhou, J. Chen, C. Li and Q. Meng, J. Med. Chem., 2024, 67, 21568–21576 CrossRef CAS PubMed.
- X. Zhang, Q. Cheng, L. Li, L. Shangguan, C. Li, S. Li, F. Huang, J. Zhang and R. Wang, Theranostics, 2019, 9, 3107–3121 CrossRef CAS PubMed.
- D. N. Shurpik, O. A. Mostovaya, D. A. Sevastyanov, O. A. Lenina, A. S. Sapunova, A. D. Voloshina, K. A. Petrov, I. V. Kovyazina, P. J. Cragg and I. I. Stoikov, Org. Biomol. Chem., 2019, 17, 9951–9959 RSC.
- Y. Chai, L. Chen, Y. Zhang, L. Zhao, Z. Meng, J. Chen, C. Li and Q. Meng, Chin. Chem. Lett., 2022, 33, 3003–3006 CrossRef CAS.
- M. Li, Y. Cheng, R. Fei, D. Xia, Z. Zhang, S. Qi and J. Du, Chem. Commun., 2025, 61, 5982–5985 RSC.
- D. Ma, B. Zhang, U. Hoffmann, M. G. Sundrup, M. Eikermann and L. Isaacs, Angew. Chem., Int. Ed., 2012, 51, 11358–11362 CrossRef CAS PubMed.
- J. Yang, Y. Wu, Y.-Y. Liu, S.-B. Yu, K. Feng, H. Wang, W. Zhou, D. Ma, G. Zhao, J. Zhang, D.-W. Zhang and Z.-T. Li, J. Med. Chem., 2025, 68, 7031–7043 CrossRef CAS PubMed.
- H.-K. Liu, F. Lin, S.-B. Yu, Y. Wu, S. Lu, Y.-Y. Liu, Q.-Y. Qi, J. Cao, W. Zhou, X. Li, H. Wang, D.-W. Zhang, Z.-T. Li and D. Ma, J. Med. Chem., 2022, 65, 16893–16901 CrossRef CAS PubMed.
- D. H. Macartney, Future Med. Chem., 2013, 5, 2075–2089 CrossRef CAS PubMed.
- Y. Wu, J. Yang, S.-Y. Zhuang, S.-B. Yu, Y. Zong, Y.-Y. Liu, G. Wu, Q.-Y. Qi, H. Wang, J. Tian, W. Zhou, D. Ma, D.-W. Zhang and Z.-T. Li, J. Med. Chem., 2024, 67, 2176–2187 CrossRef CAS PubMed.
- A. J. Selinger, N. A. Cavallin, A. Yanai, I. Birol and F. Hof, Angew. Chem., Int. Ed., 2022, 61, e202113235 CrossRef CAS PubMed.
- M. K. Abd El-Rahman and A. M. Mahmoud, RSC Adv., 2015, 5, 62469–62476 RSC.
- A. Bom, M. Bradley, K. Cameron, J. K. Clark, J. van Egmond, H. Feilden, E. J. MacLean, A. W. Muir, R. Palin, D. C. Rees and M.-Q. Zhang, Angew. Chem., Int. Ed., 2002, 41, 265–270 CrossRef CAS.
- H. D. de Boer, J. van Egmond, F. van de Pol, A. Bom and L. H. Booij, Br. J. Anaesth., 2006, 96, 473–479 CrossRef CAS PubMed.
- V. C. Roy, S. Mehta and R. Bala, Turk. J. Anaesthesiol. Reanim., 2025, 53, 293–300 Search PubMed.
- Q. Li, H. Deng, W. Zeng, Q. Zhao, T. Zhan, S. Chen and L. Wang, Chem. – Asian J., 2025, 20, e202500289 CrossRef CAS PubMed.
- L.-L. Wang, Y.-K. Tu, A. Valkonen, K. Rissanen and W. Jiang, Chin. J. Chem., 2019, 37, 892–896 CrossRef CAS.
- W. Zeng, Q. Li, H. Deng, K.-D. Zhang, M. Zhang, Y. Yin, S. Chen and L. Wang, Chin. Chem. Lett., 2026 DOI:10.1016/j.cclet.2026.112526.
- H. Deng, G. Gong, S. Lv, Y. Chen, Q. Zhao, S. Liu, S. Chen and L. Wang, Org. Chem. Front., 2023, 10, 317–326 RSC.
- P. Saha and N. Madhavan, Org. Lett., 2020, 22, 5104–5108 CrossRef CAS PubMed.
- P. Xin, S. Tan, Y. Wang, Y. Sun, Y. Wang, Y. Xu and C.-P. Chen, Chem. Commun., 2017, 53, 625–628 RSC.
- C. Ren, S. Xu, J. Xu, H. Chen and H. Zeng, Org. Lett., 2011, 13, 3840–3843 CrossRef CAS PubMed.
- Y. He, M. Xu, R. Gao, X. Li, F. Li, X. Wu, D. Xu, H. Zeng and L. Yuan, Angew. Chem., Int. Ed., 2014, 53, 11834–11839 CrossRef CAS PubMed.
- Q. Li, S. Lv, F. Xu, H. Deng, W. Zeng, J. Mao, Y.-X. Xu, S. Chen and L. Wang, ACS Macro Lett., 2025, 14, 1622–1630 CrossRef CAS PubMed.
- Z. Yang, K. Fu, W. Yu, A. Jia, X. Chen, Y. Cai, X. Li, W. Feng and L. Yuan, Chin. Chem. Lett., 2025, 36, 110842 CrossRef CAS.
- Z. Ye, M. Lian, Z. Yang, Y. Fu, Z. Wang, Y. Mu, S. Ji, H.-L. Zhang, L. Yuan, Z. Chi and Y. Huo, Adv. Opt. Mater., 2023, 11, 2202521 CrossRef CAS.
- X.-N. Xu, L. Wang and Z.-T. Li, Chem. Commun., 2009, 6634–6636 RSC.
- J. Zhu, X.-Z. Wang, Y.-Q. Chen, X.-K. Jiang, X.-Z. Chen and Z.-T. Li, J. Org. Chem., 2004, 69, 6221–6227 CrossRef CAS PubMed.
- P. F. Garrido, P. Rodríguez-Dafonte, L. García-Río and Á. Piñeiro, Langmuir, 2021, 37, 11781–11792 CrossRef PubMed.
Footnote |
| † Equally contributed to this work. |
| This journal is © The Royal Society of Chemistry 2026 |