Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Injectable oligomer-cross-linked chitosan hydrogels for biomedical applications
Iram
Maqsood
 *ab,
Hafiz Awais
Nawaz
ac,
Ketpat
Vejjasilpa
*ab,
Hafiz Awais
Nawaz
ac,
Ketpat
Vejjasilpa
 a,
Caroline
Kohn-Polster
a,
Jan
Krieghoff
a,
Caroline
Kohn-Polster
a,
Jan
Krieghoff
 a,
Alexandra
Springwald
a,
Michaela
Schulz-Siegmund
a and
Michael C.
Hacker
a,
Alexandra
Springwald
a,
Michaela
Schulz-Siegmund
a and
Michael C.
Hacker
 *ad
*ad
aInstitute of Pharmacy, Pharmaceutical Technology, Faculty of Medicine at Leipzig University, 04317 Leipzig, Germany
bDepartment of Pharmaceutics, School of Pharmacy, University of Maryland, Baltimore, Maryland 21201, USA. E-mail: imaqsood@rx.umaryland.edu; Tel: +1(443)794-3120
cInstitute of Pharmaceutical Sciences (IPS), University of Veterinary & Animal Sciences (UVAS), Abdul Qadir Jillani road, Lahore, Pakistan
dHeinrich Heine University Düsseldorf, Faculty of Mathematics and Natural Sciences, Institute of Pharmaceutics and Biopharmaceutics, 40225 Duesseldorf, Germany. E-mail: michael.hacker@hhu.de; Tel: +49 211 8114385
First published on 9th March 2026
Abstract
Injectable chitosan hydrogels, designed to emulate the extracellular matrix (ECM) in tissue engineering, are conventionally formed through physical gelation. This study aims to enhance stability and broaden the range of gel properties by adopting a covalent cross-linking approach. To achieve this, a series of hydrophilic oligomeric oligomers were synthesized, incorporating acryloyl morpholine (AMo) and reactive maleic anhydride (MA) in varying ratios, both with and without the hydrophobic comonomer pentaerythritol diacrylate monostearate (PEDAS). These oligomers, characterized by low molecular weight (Mn < 5000 Da) and differing anhydride content, were rheologically assessed for their ability to cross-link chitosan under physiological conditions. The resulting injectable oligomer-cross-linked chitosan hydrogels (iCsgel) exhibited substantial elastic strength (with an estimated E of up to 28 kPa). Furthermore, oligomer-cross-linking facilitated the production of chitosan-based hydrogels with customizable mechanical properties, controllable swelling behavior, and regulated degradation kinetics. Importantly, cell–laden iCsgel demonstrated excellent cytocompatibility and supported cell proliferation. As a proof-of-concept, some oligomers were partially modified with a fluorescent dye before the cross-linking process, resulting in decorated iCsgel. In summary, the established injectable oligomer-cross-linked chitosan hydrogels represent a promising platform with tunable material properties, making them well-suited for applications in tissue engineering and various biomedical fields, including bioprinting.
Introduction
Injectable hydrogels play a pivotal role in biomedical applications due to their minimally invasive administration, high water content, porous structure, and the ability to support live cells, facilitating the transport of nutrients and oxygen.1,2 Macromolecules that form in situ gels can yield hydrogels that closely replicate the microarchitecture, mechanical properties, and functionalities of the extracellular matrix (ECM).3 Chitosan, characterized by its innate antibacterial properties, bioactivity, biocompatibility, biodegradability, and osteoconductivity, is a promising natural biomaterial for cartilage and bone tissue engineering.4–6 Chitosan can be in vivo hydrolysed by lysozyme, with degradation rates dependent on its molecular weight, and the resulting degradation products are known to be non-toxic, non-carcinogenic, and non-immunogenic.7,8Current chitosan cross-linking strategies remain limited by the intrinsic drawbacks of both physical and chemical approaches. Physical cross-linking methods (e.g., sodium tripolyphosphate or β-glycerophosphate) typically produce mechanically weak and structurally heterogeneous networks that are sensitive to environmental pH and ionic strength, resulting in limited long-term stability and uncontrolled drug release profiles.9–13 In contrast, chemical cross-linking strategies often rely on reactive agents such as glutaraldehyde, formaldehyde, glyoxal, or epoxy compounds,14–17 which can compromise cytocompatibility due to residual reactivity and potential toxicity.18,19 Moreover, these agents may reduce the availability of functional groups and provide limited control over gelation kinetics. Although genipin offers improved biocompatibility, it exhibits comparatively slow reaction kinetics and may interact undesirably with encapsulated therapeutics.16,20,21 Collectively, these constraints restrict the mechanical robustness, cytocompatibility, and long-term performance of injectable chitosan hydrogels for biomedical and tissue engineering applications.22,23
Our group has previously developed reactive maleic anhydride-containing ter-oligomers capable of efficiently cross-linking amine-bearing biomacromolecules for the fabrication of hydrogels and microstructured scaffolds in regenerative applications.24,25 Upon copolymerization, maleic anhydride forms pendant succinic anhydride units along the oligomer backbone, which exhibit high reactivity toward nucleophilic amines.26,27 Nucleophilic ring opening of the anhydride yields stable amide bonds while simultaneously generating a carboxylate group, thereby converting cationic amines into negatively charged moieties and contributing to network stabilization.28
Compared to aldehyde- or genipin-mediated cross-linking, anhydride chemistry enables rapid and controllable amine–anhydride conjugation under mild aqueous conditions. Importantly, anhydrides are transiently reactive and are converted into stable amide linkages without persistent cross-linker species, thereby minimizing residual reactivity and improving cytocompatibility. These features render anhydride-based systems particularly attractive for injectable, cell-compatible biomaterials.29,30
To address the need for injectable delivery, cytocompatibility, precise mechanical attributes, and controlled degradation, improvements were required to enhance the hydrophilicity of anhydride-containing oligomers. The objective of this study was to design a cytocompatible, hydrophilic oligomeric macromer incorporating reactive anhydride groups capable of covalently cross-linking chitosan under physiological conditions to meet injectability requirements, enabling cell encapsulation upon gelation. The ultimate aim is the application of injectable, cell–laden, oligomer-cross-linked chitosan hydrogels for regenerative purposes. A material design based on established free radical polymerization chemistry was employed to control the physico-chemical properties of the oligomeric oligomers by varying the comonomer type and ratio. Hydrophilic oligomeric oligomers were synthesized from acryloyl morpholine (AMo) to enhance hydrophilicity, maleic anhydride (MA) to introduce reactivity toward macromolecule amines, and pentaerythritol diacrylate monostearate (PEDAS) to enhance cohesion between polymeric chains and cell-material interaction, thereby strengthening the resulting hydrogel. These hydrophilic oligomeric oligomers are denoted as oPMoMA-x {oligo(PEDAS-co-AMo-co-MA)}, where x represents the MA molarity relative to one mole of PEDAS. Additionally, PEDAS-free oligomers were synthesized and designated as oMoMA-x {oligo(AMo-co-MA)}.
In our previous studies, we developed hydrophobic anhydride-containing oligomeric macromers for cross-linking gelatine in two-component hydrogel systems, establishing a modular and adaptable platform chemistry. These oligomers incorporated maleic anhydride to provide reactive anhydride functionalities, PEDAS to enhance mechanical cohesion and bio-interactivity, and a third comonomer to tailor physicochemical properties while suppressing maleic anhydride homopolymerisation. The first generation employed N-isopropylacrylamide (NiPAAm) to introduce thermo-responsive behaviour, yielding oPNMA-x {oligo(PEDAS-co-NiPAAm-co-MA)}.24 In the second generation, NiPAAm was replaced by diacetone acrylamide (DAAm), introducing carbonyl functionalities to expand bioconjugation potential and producing oPDMA-x {oligo(PEDAS-co-DAAm-co-MA)}.25
In contrast to these earlier hydrophobic systems designed for gelatine-based matrices, the present study focuses on highly hydrophilic, low-molecular-weight oligomers specifically optimized for rapid injectable cross-linking of chitosan under physiological conditions. These platforms enable the creation of diverse gel compositions that differ in chemical makeup, peptide content, and mechanical attributes, with some being readily processable, forming stable monolithic gels. These anhydride group-bearing oligomers have been successfully applied to create hydrogels with various materials, including PEG–amines,24 different gelatine fractions,25,28,31 gelatine microparticles,31 tubular conduits32,33 and peripheral nerve regeneration.34
In current study, several hydrophilic oligomers, namely, oPMoMA-x and oMoMA-x with varying anhydride content were synthesized and characterized. The anhydride content and molecular weight per anhydride group determine the hydrogel cross-linking density, allowing for tuneable properties within a suitable range. Injectable covalently cross-linked chitosan hydrogel (iCsgel) was fabricated from these novel, low molecular weight hydrophilic oligomers upon reaction with chitosan. The resulting hydrogels were characterized concerning their composition, microarchitecture, porosity, and mechanical strength. Importantly, the cytocompatibility of cell–laden iCsgel was extensively investigated. The effect of oligomeric oligomer chemistry and cross-linking ratio on swelling and degradation properties of hydrogels was examined in comparison to a physically assembled chitosan gel.
In our two-component hydrogels, bioconjugation with amine-bearing molecules is easily achievable by decorating these molecules on the oligomer in solution, maintaining a defined stoichiometric ratio for amide linkage conjugation while retaining anhydrides for subsequent hydrogel formation. A similar approach was applied to injectable chitosan-based gel formulations using the fluorescent dye 6-aminofluorescein (6-AF) as a model substance.
Materials and methods
Acryloyl morpholine (4-acryloyl morpholine, AMo), pentaerythritol diacrylate monostearate (PEDAS), 2,2′-azobis(2-methylpropionitrile) (azobisisobutyronitrile, AIBN), 6-aminofluorescein (6-AF), 2,4,6-trinitrobenzenesulfonic acid (TNBS), Dulbecco's modified Eagle's medium (DMEM) low glucose (with and without phenol red), dimethyl sulfone (DMS), chitosan (low molecular weight, Mw 50–190 kDa, degree of deacetylation: 75–85%) were purchased from Sigma-Aldrich (Seelze, Germany). Tetrahydrofuran (THF) was obtained from VWR BDH Prolabo (Darmstadt, Germany), refluxed over sodium and potassium, and freshly distilled before use. Deuterated chloroform (CDCl3) was obtained from Armar Chemicals (Leipzig, Germany). Diethyl ether, dichloromethane, β-glycerol phosphate (β-GP), phosphate buffered saline (PBS) and hydrochloric acid (HCl) 37% were purchased from AppliChem (Darmstadt, Germany). Foetal bovine serum (FBS), penicillin/streptomycin (P/S), and trypsin were purchased from PAA Laboratories (Pasching, Austria). AlamarBlueTM reagent, ethidium homodimer-1 (EthD-1), and calcein-acetoxymethyl (calcein-AM) were obtained from Invitrogen (Darmstadt, Germany). WST-8 assay reagent (Rotitest® Vital) was provided by Carl Roth GmbH (Karlsruhe, Germany). Anhydrous dimethyl sulfoxide (DMSO) was obtained from Life Technologies (Darmstadt, Germany). Maleic anhydride (MA) and 1,2-propanediol (+99% propylene glycol (PG)) were obtained from Acros Organics (Geel, Belgium). Glucose monohydrate and 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI) was purchased from Merck (Darmstadt, Germany). Deionized, ultrapure water as used for all experiments was produced by a Milli-Q® purifier system (Millipore Elix 10, Schwalbach, Germany).Oligomer synthesis
Novel oligomers, referred to as oPMoMA-x {oligo(pentaerythritol diacrylate monostearate) (PEDAS)-co-acryloyl morpholine (AMo)-co-maleic anhydride (MA)}, were synthesized via free radical copolymerization of the comonomers PEDAS, AMo, and MA (see Fig. 1A).24 The molar ratio of PEDAS to AMo + MA in the synthesis feed was kept at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, while the MA reaction feed relative to PEDAS varied between 2 and 10. The reaction involved dissolving the appropriate amounts of comonomers in dried THF and initiating copolymerization with AIBN (2% n/n) at 60 °C under a nitrogen atmosphere. After 18 hours, the synthesized polymer was concentrated and precipitated with diethyl ether. The resulting polymer was vacuum-dried under ambient conditions and subjected to chemical characterization.
20, while the MA reaction feed relative to PEDAS varied between 2 and 10. The reaction involved dissolving the appropriate amounts of comonomers in dried THF and initiating copolymerization with AIBN (2% n/n) at 60 °C under a nitrogen atmosphere. After 18 hours, the synthesized polymer was concentrated and precipitated with diethyl ether. The resulting polymer was vacuum-dried under ambient conditions and subjected to chemical characterization.
Similarly, hydrophilic oligomers without PEDAS {oligo(AMo-co-MA)} (referred to as oMoMA-x)(see Fig. S1A), as well as previously established hydrophobic oligomers, oPNMA-x {oligo (PEDAS-co-N-isopropylacrylamide(NiPAAm)-co-MA)}24,31 and oPDMA-x {oligo(PEDAS-co-diacetone acrylamide(DAAm)-co-MA)},25 were synthesized through free radical copolymerization of the monomers. PEDAS-free hydrophilic oligomer (named as) oMoMA-x have been synthesized by free radical copolymerization of the comonomers AMo and MA. Briefly, the co-monomer combination of 20 co-monomers was kept constant, out of which x represent MA in oMoMA. The co-monomers were dissolved in dried THF, and free-radical copolymerization was initiated with AIBN (2% n/n) at 60 °C under an inert atmosphere. The resulting oligomers were concentrated and precipitated with diethyl ether to remove THF completely and were vacuum-dried before further chemical characterization.24
Chemical characterization
Verification of chemical composition of oligomers was accomplished through 1H NMR and 13C NMR spectra in CDCl3, while polymerization confirmation was achieved via FTIR-ATR. Molecular weight distributions were determined using a SECurity GPC system relative to polystyrene standards as previously described. The obtained molecular weight characteristics were rounded to the nearest multiple of 50. Anhydride content was determined via conductometric titration, and fractions of intact anhydrides were calculated using Brown-Fujimori titrations as detailed in previous studies.24Chitosan pre-processing and cross-linking
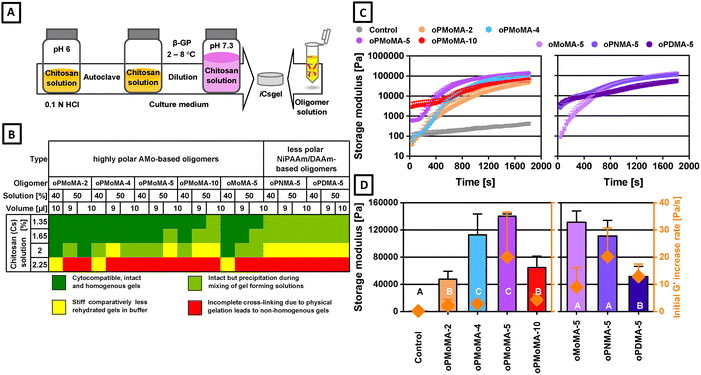
A concentrated chitosan solution (3.3% in 0.1 N HCl) was autoclaved and subsequently diluted with Dulbecco's modified Eagle's medium (DMEM) low glucose culture medium (resulting chitosan concentration: 1.65%) under aseptic conditions. The pH was adjusted to a physiological value (pH 7.3) using a sterile 0.03 M β-glycerophosphate (β-GP) solution in culture medium at 2–8 °C under vigorous stirring and aseptic conditions (see Fig. 2A). To characterize the cross-linking reaction between the novel hydrophilic oligomers (oPMoMA-x) and chitosan at physiological temperature, rheological measurements were carried out. A thermostated oscillating rheometer equipped with a steel cone geometry at 37 °C was used. The cross-linking reaction was initiated by mixing the pH-adjusted chitosan solution with the oPMoMA-x solution (40%) in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (v/v) mixture of DMSO and propylene glycol. The solutions of chitosan and oligomer were individually dispensed onto the rheometer plate before the gap was closed, and the measurement was started. Cross-linking commenced immediately upon contact of the solutions. Mixing was supported by an initial 720° rotation of the upper geometry within 10 seconds. A time sweep experiment was conducted to analyse storage modulus (G′), loss modulus (G″), and complex viscosity (η*) at a frequency of 1 Hz for over 30 minutes. Control time sweep experiments were also performed using a blank chitosan solvent to evaluate the chemical cross-linking ability of the oligomers via reaction with available free amines of chitosan, forming amide bonds. Rheological measurements were also conducted to investigate viscoelastic behaviour differences between pristine and autoclaved chitosan solutions at 20 and 32 °C.
3 (v/v) mixture of DMSO and propylene glycol. The solutions of chitosan and oligomer were individually dispensed onto the rheometer plate before the gap was closed, and the measurement was started. Cross-linking commenced immediately upon contact of the solutions. Mixing was supported by an initial 720° rotation of the upper geometry within 10 seconds. A time sweep experiment was conducted to analyse storage modulus (G′), loss modulus (G″), and complex viscosity (η*) at a frequency of 1 Hz for over 30 minutes. Control time sweep experiments were also performed using a blank chitosan solvent to evaluate the chemical cross-linking ability of the oligomers via reaction with available free amines of chitosan, forming amide bonds. Rheological measurements were also conducted to investigate viscoelastic behaviour differences between pristine and autoclaved chitosan solutions at 20 and 32 °C.
Injectable oligomer-cross-linked chitosan hydrogel (iCsgel) fabrication
ICsgel were fabricated in situ by cross-linking pH-adjusted chitosan (Cs) solution (1.65%) with anhydride-containing oligomers (oPMoMA-x). L929 mouse fibroblast cells (Cell Lines Service, Eppelheim, Germany) at passage 39–41 were aseptically dispersed at a density of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells/50 µL of chitosan solution and the dispersion was subsequently cross-linked with oligomer solution (40% in DMSO and PG (7
000 cells/50 µL of chitosan solution and the dispersion was subsequently cross-linked with oligomer solution (40% in DMSO and PG (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 v/v)) to fabricate cell–laden iCsgel. Blank (cell-free) iCsgel and physically gelled (gelation by pH adjustment without addition of oligomer) chitosan gels were fabricated as control materials. The resulting gels were incubated (37 °C, 5% CO2) in culture medium or PBS for cytocompatibility experiments or to determine physicochemical properties. While the culture medium or PBS was replaced every 48 hours.
3 v/v)) to fabricate cell–laden iCsgel. Blank (cell-free) iCsgel and physically gelled (gelation by pH adjustment without addition of oligomer) chitosan gels were fabricated as control materials. The resulting gels were incubated (37 °C, 5% CO2) in culture medium or PBS for cytocompatibility experiments or to determine physicochemical properties. While the culture medium or PBS was replaced every 48 hours.
iCsgel characterization
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), pH 8.5). Under ambient conditions in dark, freshly prepared 0.5% TNBS solution was added and samples were incubated at 40 °C for 4 h. Afterwards, 18 mL of 6 N HCl were added to stop the reaction and stabilize the coloured reaction products and samples were incubated at 60 °C for another 1.5 h successively. The gels were transferred to 96 well plate, mechanically destroyed and 100 µL of respective reaction mixture was added to each well to quantify the cross-linking degree by measuring the formation of an orange-coloured conjugate using a plate reader at 346 nm against blank samples, which were prepared following the same procedure with the addition of HCl solution prior to TNBS solution addition.
1), pH 8.5). Under ambient conditions in dark, freshly prepared 0.5% TNBS solution was added and samples were incubated at 40 °C for 4 h. Afterwards, 18 mL of 6 N HCl were added to stop the reaction and stabilize the coloured reaction products and samples were incubated at 60 °C for another 1.5 h successively. The gels were transferred to 96 well plate, mechanically destroyed and 100 µL of respective reaction mixture was added to each well to quantify the cross-linking degree by measuring the formation of an orange-coloured conjugate using a plate reader at 346 nm against blank samples, which were prepared following the same procedure with the addition of HCl solution prior to TNBS solution addition.
Results and discussion
Synthesis and characterization of reactive, cytocompatible, hydrophilic oligomeric oligomers
We sought to develop cytocompatible, highly hydrophilic oligomeric oligomers for the purpose of creating injectable chitosan hydrogels. To achieve this, we employed acryloyl morpholine (AMo) as a hydrophilic comonomer and co-polymerized it with maleic anhydride (MA). We synthesized these hydrophilic oligomers with (Fig. 1) and without (Fig. S1) pentaerythritol diacrylate monostearate (PEDAS), that has been shown to improve hydrogel storage modulus, using free radical polymerization (Table 1 and Table S1) according to a previously established strategy.25,31 The anhydride groups in the oligomers can later be used for covalently cross-linking nucleophilic amine-containing polymers like chitosan. AMo was chosen due to its non-ionic nature, solubility of its homopolymer in polar solvents, terminal unsaturation, and ability to form hydrogen bonds.36 The inclusion of PEDAS, which features a long stearate chain, facilitates interactions with proteins within the hydrogel matrix and enhances hydrogel storage modulus through maintaining a hydrophilic–lipophilic balance.| Oligomer type | Oligomer composition | Molecular weight (GPC) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Reaction feed (molar ratio) | AMo content [1H NMR] | MA content [Titration] | M n [Da] | M w [Da] | Đ M | ||||||
| PEDAS | AMo | MA | [AMo]a per PEDAS | AMob [wt%] | [wt%] | Per PEDAS | Intact MAc [%] | ||||
| a Molar AMo content per mol of PEDAS. b Calculated total AMo weight [wt%] as determined by 1H NMR analysis. c Anhydride intactness (fractions of chemically intact anhydride) and total anhydride content of oligomers expressed as weight percent [wt%] calculated from acid–base titrations. Number average molecular weight (Mn), weight average molecular weight (Mw), and molecular weight dispersity (ĐM = Mw/Mn). | |||||||||||
| oPMoMA-2 | 1 | 18 | 2 | 18.69 | 86.6 | 5.7 | 1.89 | 97.2 | 4378 ± 68 | 14924 ± 147 | 3.41 ± 0.02 |
| oPMoMA-4 | 1 | 16 | 4 | 17.79 | 78.5 | 12 | 3.87 | 96.2 | 3108 ± 18 | 8917 ± 37 | 2.87 ± 0.02 |
| oPMoMA-5 | 1 | 15 | 5 | 16.84 | 76.2 | 13.4 | 4.25 | 93.4 | 2976 ± 12 | 8861 ± 11 | 2.98 ± 0.02 |
| oPMoMA-10 | 1 | 10 | 10 | 13.80 | 66.1 | 20.5 | 6.07 | 93.8 | 1524 ± 20 | 3379 ± 58 | 2.22 ± 0.02 |
Proton NMR (1H NMR) data allowed for qualitative and quantitative of comonomer composition assessment according to previously published methodologies that use the fatty acid chain as internal reference. NMR spectroscopy analysis showed that the proton signals of the morpholine ring of AMo increased with the AMo feed (Fig. 1C). Determining the MA content from NMR was, however, challenging due to signal overlap with AMo. The use of dimethyl sulfone as an internal reference allowed us to quantify the AMo content in the absence of PEDAS (Fig. S1C). Carbon (13C) NMR analysis further confirmed the successful incorporation of AMo, PEDAS, and MA in the oligomers (Fig. 1D and Fig. S1D).
The FTIR spectra exhibited characteristic peaks corresponding to the presence of cyclic anhydride, indicating the successful copolymerization of MA with other comonomers (Fig. 1E). The spectral data confirmed the co-polymerized MA-AMo structures (Fig. 1E and Fig. S1E).
The MA content in the oligomers was determined through acid–base titrations, further indicating efficient MA copolymerization. The oligomers demonstrated good control of MA content (Fig. S2A). The obtained GPC data of oligomers complemented the NMR results and demonstrated the adjustability of AMo and MA contents in small increments. Molecular weights (Mn) of hydrophilic oligomers ranged between 1550 and 4350 Da of oPMoMA-x and 1150 and 2050 Da of oMoMA-x respectively as revealed by GPC and an indirect correlation with MA feed (Fig. S2B and C) together with direct correlation with AMo feed (Fig. S2D and E) was found (Table 1 and Table S1). Hence, oligomers with adjustable MA contents and average molecular weights (Mn) below 5000 Da, which enable high cross-linking degrees and adaptable material engineering, could be synthesized following our previously established protocols.
In summary, the co-polymerization of PEDAS, AMo, and MA produced oligomers with low molecular weights (Mn < 5000 Da), aligning with previous findings. These low molecular weights are advantageous for creating versatile, cross-linked hydrogels that ensure renal eliminability after hydrogel degradation. As biological building blocks, we previously used gelatine or gelatinous peptides. In this study, we intended to fabricate chitosan hydrogels with tuneable properties using these adjustable, low molecular weight oligomers. We consider these hydrophilic oligomers with anhydride functionalities better cytocompatible as compared to the commonly used cross-linking agent glutaraldehyde. In contrast to aldehydes, anhydrides rapidly lose their residual chemical reactivity in aqueous environments, making them suitable for injectable hydrogel formulations. Additionally, the physico-chemical properties of these two-component hydrogels will depend on both the biological component, chitosan, and the cross-linking oligomer. We are especially interested to tailor the mechanical properties through the choice of oligomer.
Cross-linking reaction with chitosan
A low molecular weight chitosan (50–190 kDa), obtained by alkaline deacetylation of chitin to a high deacetylation degree (∼80%), was selected for its biocompatibility, antibacterial properties, and biodegradability. The initial chitosan solution (3.3%) was subsequently diluted to a desired concentration (1.65%) with culture medium under aseptic conditions. The pH of the solution was adjusted to the physiological range using a sterile β-glycerophosphate solution (0.03 M) at low temperatures (2–8 °C) and vigorous stirring (Fig. 2A). Beta-glycerophosphate (β-GP) has been extensively used with chitosan to form physical hydrogels at 37 °C through ionic interactions under certain critical conditions, such as a concentration of more than 2% to achieve chain entanglement.12 Due to the free diffusibility of β-GP, it is considered a proton acceptor and not a divalent electrostatic cross-linker of chitosan amine groups.37 The phosphate base can also serve as a source of organic phosphate for bone mineralization and for osteogenic differentiation.38 Later, the above-mentioned pH adjusted chitosan solution was cross-linked with oligomer solution to fabricate iCsgel (Fig. 2A).Steam sterilization-induced reduction in chitosan solution viscosity motivated a rheological evaluation using a cone plate setup. Rheological characterization involved moduli and viscosity assessments obtained during oscillatory time and frequency tests at 20 °C and 32 °C (Fig. S3). A temperature of 32 °C instead of 37 °C was selected to analyse effects of cross-linking prior to any possible additive effect of physical gelation of neutralized chitosan at physiological temperature.
The observed minor viscosity decrease observed post sterilization is attributed to biopolymer hydrolytic depolymerization.39,40 Initially, modulus and viscosity of pristine chitosan exceeded that of autoclaved chitosan. Over time, autoclaved chitosan manifested heightened values towards the experiment's conclusion, suggesting more homogenous distribution over the measurement gap due to the initially reduced viscosity. Both solutions exhibited a sharp decline in complex viscosity with rising frequency at 20 °C and 32 °C, while moduli displayed a plateau, indicating shear thinning and possibly thixotropic behaviour41 (Fig. S3A–D). Overall, whether autoclaved or pristine, chitosan solutions exhibited more fluid-like behaviour at 32 °C, attributable to enhanced interactions among chitosan, β-GP, and water over time.42
The cross-linking reaction between anhydride-bearing hydrophilic oligomeric oligomers and chitosan was evaluated using oscillation rheology. Different molar ratios of chitosan and oligomer solutions were used to monitor the reaction rheologically (Fig. 2). Chitosan's total free amino groups were calculated, and it was found to have significantly more amino groups compared to gelatinous peptide used in previous studies (Table S2).43 This is one of the reasons why oligomer-cross-linked chitosan gels exhibited higher storage moduli as oligomer-cross-linked gelatinous peptide gels presented in previous studies. A heatmap was constructed of all investigated iCsgel using both components chitosan solution (100 µL) and oligomer in different concentration and volume indicating hydrogel quality based on formulation processability and efficiency of cross-linking (Fig. 2B). Hydrogel characteristics were influenced by the cross-linker type, solution concentration (40–50%), and chitosan content (1.35–2.25%), resulting in stable and uniform gels. ICsgel exhibited transparency at low MA (≤5), whereas MA-10 formulations showed increased turbidity and slight shrinkage. Increased macromer hydrophobicity correlated with reduced transparency and more pronounced precipitation. The formation of transient polyelectrolyte complexes between chitosan and anhydride-containing oligomers likely contributed to the observed precipitates. Based on these preliminary evaluations, formulations containing 40% oligomer and 1.65% chitosan were selected for subsequent investigations.
The optimized chitosan solution (1.65 w/v%) cross-linking was evaluated with a set of previously established hydrophobic oligomers (oPNMA-5 and oPDMA-5) in addition to hydrophilic oligomers (oPMoMA-x and oMoMA-x) (Fig. 2 and Fig. S4). The rheological data revealed an increase in dynamic moduli, indicating the formation of cross-linked elastic hydrogels with all tested oligomers (Fig. 2). The effect of the cross-linking group ratio ([MA]/[NH2]) on the reaction kinetics was examined (Fig. 2C). The cross-linking reaction progressed more rapidly for oPMoMA-4 and -5 ([MA]/[NH2] ratios of 0.59 and 0.64, respectively) as compared to other hydrophilic and hydrophobic oligomers. Hydrophobic oligomers showed higher initial storage moduli, possibly due to initial precipitation induced during mixing of hydrogel-forming solutions. Also, the hydrophobic and hydrophilic oligomers of higher anhydride content (MA = 10) demonstrated flatter gelation kinetic curves (Fig. S5). The control formulation without oligomers at 37 °C exhibited only a slight increase in moduli, likely due to transient physical gelation and increased chain entanglement. The rate of modulus increase was calculated from the reaction mixtures as a measure of the cross-linking reaction kinetics (Fig. 2D, right y-axis). Obviously, the rate increase was affected by relative anhydride content and molecular weight of oligomers. The rate was highest with the oPMoMA-5 oligomer, which provided the best balance of anhydride group density and chain mobility until viscosity increase. Network formation continued until the end of the oscillatory time test (30 minutes) and one could not determine time to modulus plateau from the measurements. All investigated oligomers induced rapid gelation, and the exact point of gelation (as crossover of G′ and G″) was often quicker than the first values could be recorded.
The complex viscosities obtained during the cross-linking reactions indicated efficient network formation, with the hydrophilic oligomers containing MA-4 and MA-5 found to be the most optimal for hydrogel formation (Fig. 3). The low molecular weight oligomers served as efficient cross-linkers for chitosan, resulting in stable network formation. Overall, the study successfully demonstrated the efficient cross-linking of chitosan with hydrophilic and hydrophobic oligomeric anhydride-containing oligomers.
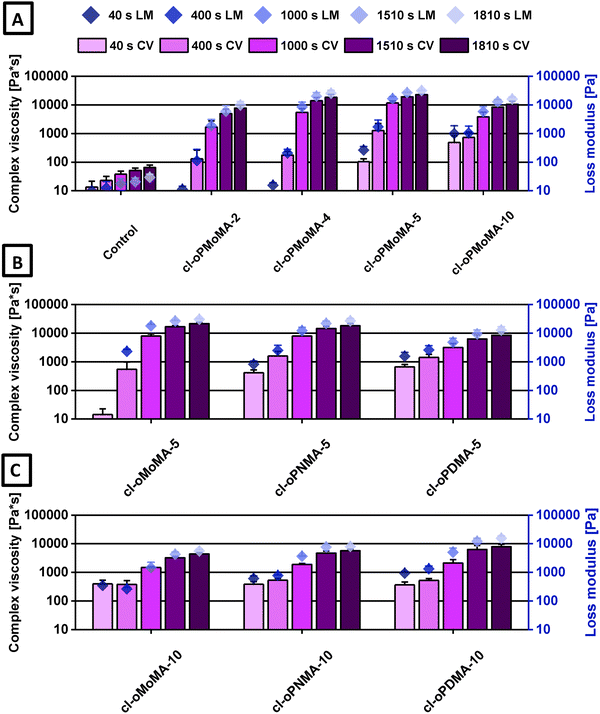 | ||
| Fig. 3 Complex viscosities (η*) (columns left y-axis) and loss modulus (G″) (diamonds, right y-axis) determined at different time points from rheological data as a measure of gelation kinetics after initiation of cross-linking reaction with various oligomers: (A) oPMoMA-x with different MA feeds in comparison to control, (B) different MA-5 oligomers, (C) different MA-10 oligomers. The corresponding storage moduli can be found in Fig. 2. Columns (complex viscosity) and data points (loss modulus) with error bars represent means ± standard deviations (n = 4). | ||
Injectable chitosan hydrogels (iCsgel) formation and characterization
Injectable chitosan hydrogels (iCsgel) were fabricated by in situ cross-linking of autoclaved pH-adjusted chitosan solutions with anhydride-containing oligomers by mixing 70 µL of 1.65% chitosan and 7 µL of 40% oligomer (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio). The resulting hydrogels were homogeneous, stable, and transparent, with transparency decreasing as the oligomer's maleic anhydride (MA) content increased (Fig. 4).
1 ratio). The resulting hydrogels were homogeneous, stable, and transparent, with transparency decreasing as the oligomer's maleic anhydride (MA) content increased (Fig. 4).
To evaluate their mechanical properties, the storage moduli (G′) of equilibrium-swollen hydrogels in culture medium at 37 °C were measured using frequency sweep oscillation rheology. An increase in hydrogel stiffness was observed with higher MA content (Fig. 4C). The apparent discrepancy in storage modulus for MA-10 gels between time-sweep (Fig. 2C) and frequency-sweep (Fig. 4C) measurements arises from differences in network maturation time. Rapid initial gelation constrained further modulus evolution within the 30-minute time-sweep experiment, whereas 24-hour equilibration in culture medium permitted continued amine–anhydride conjugation and equilibrium swelling. Under these conditions, the higher anhydride content of oPMoMA-10 yielded a greater cross-link density and consequently a higher equilibrium storage modulus compared to oPMoMA-4 and -5 cross-linked gels.
The storage moduli values for iCsgel cross-linked with oPMoMA-x oligomers ranged from 3.4 to 9.5 kPa. These values are in the range of mechanical moduli of soft tissues.44 Gel modulus increased in the order: oMoMA < oPMoMA < oPNMA < oPDMA regarding the oligomer type of the iCsgel composition.
Cross-linking density, expressed as 30–80% amine conversion, was quantified using a TNBS assay, revealing a significant increase with higher MA content (Fig. 4C, right y-axis). The best formulations had around 60% cross-linking degree. Physicochemical properties, such as water content, dry weight, and leachables, were assessed (Fig. S6). ICsgel with lower MA content absorbed more water than those cross-linked with MA-10 oligomers but less than non-cross-linked chitosan gels. There was an inverse correlation between water content, dry weight, and cross-linking degree, as observed in previous research.31 Furthermore, hydrogels with higher MA content had fewer leachables (Fig. S6).
For tissue engineering application, injectable hydrogels should be porous materials with open and interconnected networks to allow cell migration, cell proliferation, nutrient delivery and waste elimination.45 The porosity assessment of iCsgel cross-linked with oPMoMA-x was conducted by liquid displacement method and the results are summarized in Fig. 5.35 All tested chitosan hydrogels exhibited porosity in the range of 80–90%, irrespective of cross-linking degree and elastic strength (Fig. 5B). The highly porous, interconnected and homogenous microstructure of lyophilized iCsgel was also confirmed by scanning electron microscopy without considerable differences between the investigated formulations (Fig. 5C).46 For tissue regeneration applications appropriate pore size, porosity and water uptake ability are favourable attributes to provide some space to embedded cells, later cell adhesion, and proliferation.47–49 An interesting phenomenon was that no direct correlation was found between the obtained porosity of iCsgel and equilibrium water content which agreed with previous investigations.35
FTIR-ATR analysis of iCsgel demonstrated significant changes compared to pristine chitosan spectrum (Fig. S7). Initially, a weak ionic interaction between chitosan and β-GP was evidenced by slight shifts in the –NH3+ band (∼1550–1650 cm−1) and phosphate stretching bands (∼900–1150 cm−1) in iCsgel spectra. Further changes included a reduction in the broad absorption peak representing hydroxyl and amino groups, as well as the disappearance of the characteristic amide I peak of chitosan at 1650 cm−1, replaced by a new peak at 1628 cm−1. Additionally, the band at 1548 cm−1, associated with the amino group of chitosan, vanished. The FTIR spectrum of cross-linked iCsgel displayed signals from both chitosan and oligomeric oligomers, with the absence of peaks assigned to the amino group of chitosan and the anhydride of the oligomer (1777 cm−1 and 1850 cm−1). These observations indicated the successful amide formation (bands at 1628 cm−1, 1445 cm−1, 1235 cm−1 and 573 cm−1 representing –O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–N [Amide I], primary amide [Amide VI], (NH) + (–O
C–N [Amide I], primary amide [Amide VI], (NH) + (–O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–N) [Amide III] and (C–C) + (–O
C–N) [Amide III] and (C–C) + (–O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–N) [Amide IV]) from anhydrides and amines yielding chemically cross-linked chitosan-based hydrogel.
C–N) [Amide IV]) from anhydrides and amines yielding chemically cross-linked chitosan-based hydrogel.
Fluorescent labelling of iCsgel via cross-linking with derivatized oPMoMA-x was accomplished by covalently attaching 6-AF to the oligomer backbone through amide bond formation (Fig. 6A). The successful integration of the fluorescent probe was confirmed by 1H NMR analysis (Fig. S8). Stereomicroscopic images showed a yellow coloration in the fluorescently labelled iCsgel, while the control iCsgel remained colourless. Fluorescence micrographs demonstrated a significant increase in fluorescence intensity in the labelled gel compared to the control (Fig. 6B–E). This increased fluorescence was attributed to the incorporation of fluorescently labelled oligomers. Surprisingly, oPMoMA-10+6-AF cross-linked iCsgel exhibited significantly higher fluorescence intensities among other derivatized (+6-AF) iCsgel, which confirmed the hypothesis of increasing fluorescence intensity with increasing amide bond formation (Fig. 6F). The fluorescence intensity increased with higher concentrations of derivatized oligomers as evident from labelled iCsgel cross-linked with oPMoMA-5 modified with different MAeq of 6-AF (Fig. 6G). These findings open new possibilities for designing biomimetic hydrogels via derivatization with bioactive molecules.
Cell culture
In tissue regeneration, cell adhesion, proliferation, and differentiation are crucial for tissue development and are considered of fundamental importance for biomedical applications.50 In this study, we assessed the cytocompatibility of cell–laden iCsgel, which is prerequisite for further application in tissue regeneration and biomedical therapies. Initially, we encapsulated L929 mouse fibroblast cells in the iCsgel and examined their behaviour over time (Fig. 7).51 Confocal microscopy images revealed good cell viability, migration, and proliferation within the gel matrix. Initially, the cells had a rounded shape, but by day 7, increased cell spreading was observed (Fig. 7B). Cellularity and cell shape was supported by scanning electron microscopy (Fig. 7C). Overall, injectable formulations have shown good interaction with encapsulated cells, which can be attributed to the intrinsic biocompatibility of chitosan.52 Quantitative analysis of metabolic activity of the cells using the AlamarBlueTM assay supported this data and displayed cell viability and proliferation with cultivation time (Fig. 7D). The analysed colorimetric changes after day 7 were significantly different from day 1 and 353, though no significant difference was revealed among iCsgel cross-linked with oPMoMA of different AM feeds. Osmometric measurements showed that osmolarity was not a limiting factor for cell viability within the gel (Fig. 7E).Taken into consideration that encapsulated cells in oligomer-cross-linked chitosan hydrogels are not directly accessible to dye resulting in heterogeneous dye distribution and inconsistent measurement, a consecutive AlamarBlueTM assay was also conducted. A significant increase in fluorescence values was revealed in gel cross-linked with MA-4- and MA-5-type oligomers at day 3, however, in the gel cross-linked with oPMoMA-10, cellularity decreased due to the gels’ high cross-linking density and rigid nature54 (Fig. 7F). Regarding the analytical methodology, it is important to observe that gels without cells also showed significant fluorescence levels, suggesting a redox reaction between the material and dye, even when the material is cross-linked. In literature, the role of poly(ethylene-alt-maleic anhydride) as reduction catalyst has been discussed.55 Also, reduction of resazurin to resorufin has been reported to occur by different means than viable cells, such as photo-reduction with several types of amines upon irradiation and plasmon-mediated reduction by hydroxylamine and gold nanoparticles.56,57 Hence, we deduce that polymers containing polymerized maleic anhydride have the potential to reduce resazurin to resorufin even when the anhydrides have been amino- or hydrolysed.
We conducted additional experiments to encapsulate cells within gels cross-linked by different oligomers containing the same anhydride feed, here MA-5-type oligomers. Hydrophilic oligomers, oPMoMA-5 and oMoMA-5, cross-linked gels showed good cytocompatibility and increased cell numbers over time, while hydrophobic oligomer, oPNMA-5 and oPDMA-5, cross-linked gels led to significant cell death and inhibited proliferation (Fig. S9A).58,59 The inhibition of cell proliferation in hydrophobic oligomers cross-linked iCsgel was also confirmed by AlamarBlueTM and WST-8 assay, which showed almost negligible dye conversion for iCsgel cross-linked with hydrophobic oligomers but very good cell viability and proliferation in cell–laden iCsgel cross-linked with hydrophilic oligomers (oPMoMA and oMoMA) (Fig. S9B, left image). The blank cross-linked iCsgel (without cells) reduced yellow WST-8 to orange WST-8 formazan, therefore, data was corrected by subtracting the absorption values of control gel samples (Fig. S9B, right image). The cytocompatibility experiments clearly highlight the potential of cell–laden-hydrophilic oligomer cross-linked iCsgel in tissue engineering by facilitating viable cell encapsulation and survival.
Degradation study
The in vitro degradation of injectable chitosan hydrogels (iCsgel) cross-linked with various hydrophilic oligomers (oPMoMA-x) was conducted in PBS at 37 °C and the results were compared to non-cross-linked chitosan physical gels (Fig. 8A). The oligomer cross-linked chitosan hydrogels remained stable throughout the degradation study of 42 days, showing only slight opaqueness and shrinkage, likely due to the accumulation of buffer salts. The mass loss in the cross-linked gels was minimal, with oPMoMA-5 and oPMoMA-10 cross-linked gels being the most stable (Fig. 8B). Overall, mass loss correlated inversely with the anhydride content of the oligomer and cross-linking degree. These findings of a slow rate of degradation of cross-linked chitosan hydrogels are in general accordance with data reported for other covalently cross-linked chitosan formulations.7,60,61 It, however, was also shown that in vitro degradation rate can be accelerated by adding enzymes such as lysozyme to the buffer.62 For a more detailed insight in the hydrolytic degradation profiles of iCsgel, leachables (Fig. 8C), swelling ratio (Fig. 8D) and pH of incubation media (Fig. 8E) were monitored. Congruent with mass loss, leachables and swelling ratio from the oligomer cross-linked chitosan hydrogels were significantly less compared to non-cross-linked chitosan control gels, and the pH of the incubation media remained stable over time.Overall, our study demonstrated that these hydrophilic oligomer-cross-linked injectable chitosan hydrogels have controlled and adjustable hydrolytic degradation properties that correlate with the degree of cross-linking. In summary, the iCsgel platform appears promising to provide tailored hydrogels for the formulation of tissue engineering scaffolds suitable for cell and drug delivery applications.
Conclusion
In the present study, we have synthesized a set of hydrophilic oligomeric oligomers of low molecular weight with reactive and easily derivatizable anhydride groups that were subsequently used to covalently cross-link chitosan. Rheology revealed prompt reaction between reactive hydrophilic oligomers and chitosan. The injectable gels exhibited increased mechanical strength with increasing anhydride content of the oligomers. Generally, iCsgel exhibited a homogeneous 3D porous structure and uniform in vitro degradation at a physiologically relevant rate. Moreover, iCsgel showed tuneable mechanical, swelling, and degradative properties together with excellent cytocompatibility and supported cell proliferation. In summary, the investigated hydrophilic reactive oligomer cross-linked injectable chitosan hydrogel platform is highly diverse and allows for fabrication of in situ tissue engineering scaffolds as well as matrices for cells and drug delivery, all with precise characteristics.Author contributions
The defined roles of all the authors who contributed to the manuscript are in the following way: Iram Maqsood: investigation, methodology, writing, editing, and review of paper. Hafiz Awais Nawaz: investigation (particularly related to oligomers synthesis and characterization). Ketpat Vejjasilpa: some investigation related to cell culture. Caroline Kohn-Polster: some investigation related to cell culture. Jan Krieghoff: investigation related to oligomers synthesis. Alexandra Springwald: investigation related to oligomers synthesis. Michaela Schulz-Siegmund: resources, supervision, review of manuscript. Michael C. Hacker: conceptualization, funding acquisition, project administration, supervision, writing – editing and review.Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article has been included in supplementary information (SI). Supplementary information: these include detailed chemical characterizations of hydrophilic oligomeric oligomers, oMoMA-x (Fig. S1), their compositions and chemical properties (Table S1), and analyses of anhydride content and oligomer-molecular weight correlations (Fig. S2). Rheological assessments of pristine chitosan solutions before and after autoclaving are presented (Fig. S3). Reaction conditions of chitosan solution and oligomer solution, calculation of cross-linking group ratio and corresponding storage moduli are described (Table S2). Chemical illustration of injectable oligomer-cross-linked chitosan hydrogel (iCsgel) fabrication from chitosan and reactive anhydride-containing oligomer (Fig. S4). Time-sweep rheological profiles of cross-linking reactions between chitosan and various MA-10-containing oligomers are shown (Fig. S5), supported by instrument details and kinetic data. Physico-chemical properties of iCsgel formulations, including water content and leachables, are compared to control gels (Fig. S6), stacked FTIR spectra of oPMoMA-x cross-linked chitosan hydrogel in comparison to pristine chitosan and β-GP (Fig. S7), representative 1H NMR spectra confirming successful covalent attachment of 6-aminofluorescein (6-AF) to oPMoMA-x oligomers (Fig. S8) and cytocompatibility of encapsulated L929 fibroblast cells within iCsgel cross-linked with MA-5 containing oligomers is assessed through imaging and viability assays (Fig. S9). Raw data supporting all analyses can be found in the SI (Tables S3–S9). See DOI: https://doi.org/10.1039/d5tb02401c.Acknowledgements
Financial support by the German Research Council (DFG SFB, Projektnummer 59307082/Transregio 67, project A1) as well as the personal funding from Higher Education Commission (HEC-90% overseas scholarship), Pakistan, and the German Academic Exchange Services (DAAD) to Iram Maqsood (Phase II, Batch VI) is gratefully acknowledged. We are grateful to Jörg Lenzner (Faculty of Physics and Earth Science, Leipzig University) for scanning electron microscopic (SEM) imaging. We also acknowledge Lisa Franz (Institute of Pharmacy, Pharmaceutical/Medicinal Chemistry, Leipzig University) for providing access to a Fourier transform infrared spectrophotometer (FTIR-ATR). We also thank Lothar Hennig (Department of Organic Chemistry, Leipzig University) for NMR analysis of oligomers.References
- F. Y. Hsieh, L. Tao, Y. Wei and S. H. Hsu, A novel biodegradable self-healing hydrogel to induce blood capillary formation, NPG Asia Mater., 2017, 9(3), 1–11, DOI:10.1038/am.2017.23.
- J. W. Seo, et al., Injectable hydrogel derived from chitosan with tunable mechanical properties via hybrid-crosslinking system, Carbohydr. Polym., 2021, 251, 117036, DOI:10.1016/j.carbpol.2020.117036.
- C. D. Hoemann, J. Sun, A. Légaré, M. D. McKee and M. D. Buschmann, Tissue engineering of cartilage using an injectable and adhesive chitosan-based cell-delivery vehicle, Osteoarthr. Cartil., 2005, 13(4), 318–329, DOI:10.1016/j.joca.2004.12.001.
- M. Fujita, et al., Vascularization in vivo caused by the controlled release of fibroblast growth factor-2 from an injectable chitosan/non-anticoagulant heparin hydrogel, Biomaterials, 2004, 25(4), 699–706, DOI:10.1016/S0142-9612(03)00557-X.
- M. Rafat, et al., PEG-stabilized carbodiimide crosslinked collagen-chitosan hydrogels for corneal tissue engineering, Biomaterials, 2008, 29(29), 3960–3972, DOI:10.1016/j.biomaterials.2008.06.017.
- Y. Zhang, L. Tao, S. Li and Y. Wei, Synthesis of multiresponsive and dynamic chitosan-based hydrogels for controlled release of bioactive molecules, Biomacromolecules, 2011, 12(8), 2894–2901, DOI:10.1021/bm200423f.
- F. L. Mi, Y. C. Tan, H. F. Liang and H. W. Sung, In vivo biocompatibility and degradability of a novel injectable-chitosan-based implant, Biomaterials, 2002, 23(1), 181–191, DOI:10.1016/S0142-9612(01)00094-1.
- F. Mushtaq, et al., Injectable Chitosan-Methoxy Polyethylene Glycol Hybrid Hydrogel Untangling the Wound Healing Behavior: In Vitro and In Vivo Evaluation, ACS Omega, 2024, 9(2), 2145–2160, DOI:10.1021/acsomega.3c04346.
- M. Biernat, et al., Effect of Selected Crosslinking and Stabilization Methods on the Properties of Porous Chitosan Composites Dedicated for Medical Applications, Polymers, 2023, 15(11), 2507, DOI:10.3390/polym15112507.
- M. M. Ribeiro, M. Simões, C. Vitorino and F. Mascarenhas-Melo, Physical crosslinking of hydrogels: the potential of dynamic and reversible bonds in burn care, Coord. Chem. Rev., 2025, 542, 216868, DOI:10.1016/j.ccr.2025.216868.
- Z. Huang, et al., Food Hydrocolloids Crosslinking strategies and functionalization modification approaches for chitosan-based hydrogels in food preservation applications: a review, Food Hydrocolloids, 2026, 175, 112475, DOI:10.1016/j.foodhyd.2026.112475.
- P. Roughley, C. Hoemann, E. DesRosiers, F. Mwale, J. Antoniou and M. Alini, The potential of chitosan-based gels containing intervertebral disc cells for nucleus pulposus supplementation, Biomaterials, 2006, 27(3), 388–396, DOI:10.1016/j.biomaterials.2005.06.037.
- A. Chenite, et al., Novel injectable neutral solutions of chitosan form biodegradable gels in situ, Biomaterials, 2000, 21(21), 2155–2161, DOI:10.1016/S0142-9612(00)00116-2.
- J. Berger, M. Reist, J. M. Mayer, O. Felt, N. A. Peppas and R. Gurny, Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications, Eur. J. Pharm. Biopharm., 2004, 57(1), 19–34, DOI:10.1016/S0939-6411(03)00161-9.
- M. N. V. R. Kumar, R. A. A. Muzzarelli, C. Muzzarelli, H. Sashiwa and A. J. Domb, Chitosan chemistry and pharmaceutical perspectives, Chem. Rev., 2004, 104(12), 6017–6084, DOI:10.1021/cr030441b.
- N. Bhattarai, J. Gunn and M. Zhang, Chitosan-based hydrogels for controlled, localized drug delivery, Adv. Drug Delivery Rev., 2010, 62(1), 83–99, DOI:10.1016/j.addr.2009.07.019.
- N. Murata-Kamiya, H. Kaji and H. Kasai, Types of mutations induced by glyoxal, a major oxidative DNA-damage product, in Salmonella typhimurium, Mutat. Res., Fundam. Mol. Mech. Mutagen., 1997, 377(1), 13–16, DOI:10.1016/S0027-5107(97)00016-X.
- P. Sapuła, K. Bialik-Wąs and K. Malarz, Are Natural Compounds a Promising Alternative to Synthetic Cross-Linking Agents in the Preparation of Hydrogels?, Pharmaceutics, 2023, 15(1), 253, DOI:10.3390/pharmaceutics15010253.
- V. A. Reyna-urrutia, V. Mata-haro and J. V. Cauich-rodriguez, Effect of two crosslinking methods on the physicochemical and biological properties of the collagen-chitosan scaffolds, Eur. Polym. J., 2019, 117, 424–433, DOI:10.1016/j.eurpolymj.2019.05.010.
- N. Bhattarai, H. R. Ramay, J. Gunn, F. A. Matsen and M. Zhang, PEG-grafted chitosan as an injectable thermosensitive hydrogel for sustained protein release, J. Controlled Release, 2005, 103(3), 609–624, DOI:10.1016/j.jconrel.2004.12.019.
- A. C. Alavarse, E. C. G. Frachini, R. L. C. G. da Silva, V. H. Lima, A. Shavandi and D. F. S. Petri, Crosslinkers for polysaccharides and proteins: synthesis conditions, mechanisms, and crosslinking efficiency, a review, Int. J. Biol. Macromol., 2022, 202, 558–596, DOI:10.1016/j.ijbiomac.2022.01.029.
- K. Wegrzynowska-Drzymalska, et al., Crosslinking of Chitosan with Dialdehyde Chitosan as a New Approach for Biomedical Applications, Materials, 2020, 13(15), 1–27, DOI:10.3390/ma13153413.
- E. Szymańska and K. Winnicka, Stability of Chitosan—A Challenge for Pharmaceutical and Biomedical Applications, Mar. Drugs, 2015, 13(4), 1819–1846, DOI:10.3390/md13041819.
- T. Loth, R. Hennig, C. Kascholke, R. Hötzel and M. C. Hacker, Reactive and stimuli-responsive maleic anhydride containing macromers – Multi-functional cross-linkers and building blocks for hydrogel fabrication, React. Funct. Polym., 2013, 73(11), 1480–1492, DOI:10.1016/j.reactfunctpolym.2013.08.002.
- C. Kascholke, et al., Dual-Functional Hydrazide-Reactive and Anhydride-Containing Oligomeric Hydrogel Building Blocks, Biomacromolecules, 2017, 18(3), 683–694, DOI:10.1021/acs.biomac.6b01355.
- M. Carpintero, I. Marcet, C. Cortizo and P. Guerrero, Food Hydrocolloids Chitosan modification with octenyl succinic anhydride (OSA): effect of the degree of substitution on the structural, mechanical and barrier properties in the synthetized bioplastics, Food Hydrocolloids, 2026, 171, 111838, DOI:10.1016/j.foodhyd.2025.111838.
- M. Karimi, F. Ahadi, N. Esmati, M. Fan, L. Han and C. Y. Li, Heterogeneous polymerization via two-step crosslinking for tunable microribbon hydrogels, Biofabrication, 2026, 18(1), 15016, DOI:10.1088/1758-5090/ae235a.
- H. A. Nawaz, et al., Injectable oligomer-cross-linked gelatine hydrogels via anhydride-amine-conjugation, J. Mater. Chem. B, 2021, 9(9), 2295–2307, 10.1039/d0tb02861d.
- N. N. Shah, N. Soni and R. S. Singhal, Modification of proteins and polysaccharides using dodecenyl succinic anhydride: synthesis, properties and applications—A review, Int. J. Biol. Macromol., 2018, 107, 2224–2233, DOI:10.1016/j.ijbiomac.2017.10.099.
- N. Reddy, R. Reddy and Q. Jiang, Crosslinking biopolymers for biomedical applications, Trends Biotechnol., 2015, 33(6), 362–369, DOI:10.1016/j.tibtech.2015.03.008.
- T. Loth, R. Hötzel, C. Kascholke, U. Anderegg, M. Schulz-Siegmund and M. C. Hacker, Gelatin-based biomaterial engineering with anhydride-containing oligomeric cross-linkers, Biomacromolecules, 2014, 15(6), 2104–2118, DOI:10.1021/bm500241y.
- C. Kohn, et al., Dual-component collagenous peptide/reactive oligomer hydrogels as potential nerve guidance materials-from characterization to functionalization, Biomater. Sci., 2016, 4(11), 1605–1621, 10.1039/c6bm00397d.
- C. Kohn-Polster, et al., Dual-component gelatinous peptide/reactive oligomer formulations as conduit material and luminal filler for peripheral nerve regeneration, Int. J. Mol. Sci., 2017, 18(5), 1–29, DOI:10.3390/ijms18051104.
- C. Kohn-Polster, et al., Functionalization of Two-Component Gelatinous Peptide/Reactive Oligomer Hydrogels with Small Molecular Amines for Enhanced Cellular Interaction, Int. J. Mol. Sci., 2025, 26(11), 1–22, DOI:10.3390/ijms26115316.
- H. Qin, J. Wang, T. Wang, X. Gao, Q. Wan and X. Pei, Preparation and Characterization of Chitosan/β-Glycerophosphate Thermal-Sensitive Hydrogel Reinforced by Graphene Oxide, Front. Chem., 2018, 6, 565, DOI:10.3389/fchem.2018.00565.
- F. Eeckman, A. J. Moës and K. Amighi, Synthesis and characterization of thermosensitive copolymers for oral controlled drug delivery, Eur. Polym. J., 2004, 40(4), 873–881, DOI:10.1016/j.eurpolymj.2003.11.010.
- D. Filion and M. D. Buschmann, Chitosan-glycerol-phosphate (GP) gels release freely diffusible GP and possess titratable fixed charge, Carbohydr. Polym., 2013, 98(1), 813–819, DOI:10.1016/j.carbpol.2013.06.055.
- R. J. Krawetz, et al., Collagen I Scaffolds Cross-Linked with Beta-Glycerol Phosphate Induce Osteogenic Differentiation of Embryonic Stem Cells In Vitro and Regulate Their Tumorigenic Potential In Vivo, Tissue Eng., Part A, 2011, 18(9–10), 1014–1024, DOI:10.1089/ten.tea.2011.0174.
- H. K. No, J. W. Nah and S. P. Meyers, Effect of time/temperature treatment parameters on depolymerization of chitosan, J. Appl. Polym. Sci., 2003, 87(12), 1890–1894, DOI:10.1002/app.11628.
- A. San Juan, et al., Degradation of chitosan-based materials after different sterilization treatments, IOP Conf. Ser.: Mater. Sci. Eng., 2012, 31, 012007, DOI:10.1088/1757-899X/31/1/012007.
- M. A. Torres, M. M. Beppu, C. C. Santana and E. J. Arruda, Viscous and Viscoelastic Properties of Chitosan Solutions and Gels, Braz. J. Food Technol., 2006, 9(2), 101–108 CAS.
- E. A. El-hefian and A. H. Yahaya, Rheological study of chitosan and its blends: an overview, Maejo Int. J. Sci. Technol., 2010, 4(02), 210–220 CAS.
- J. E. Eastoe, The amino acid composition of mammalian collagen and gelatin, Biochem. J., 1955, 61(4), 589–600 CrossRef CAS PubMed.
- S. Yang, K.-F. Leong, Z. Du and C.-K. Chua, The Design of Scaffolds for Use in Tissue Engineering. Part I. Traditional Factors, Tissue Eng., 2001, 7(6), 679–689, DOI:10.1089/107632701753337645.
- S. Partap, A. Muthutantri, I. U. Rehman, G. R. Davis and J. A. Darr, Preparation and characterisation of controlled porosity alginate hydrogels made via a simultaneous micelle templating and internal gelation process, J. Mater. Sci., 2007, 42(10), 3502–3507, DOI:10.1007/s10853-007-1533-x.
- N. Annabi, et al., Controlling the porosity and microarchitecture of hydrogels for tissue engineering, Tissue Eng., Part B, 2010, 16(4), 371–383, DOI:10.1089/ten.teb.2009.0639.
- F. Mirahmadi, M. Tafazzoli-Shadpour, M. A. Shokrgozar and S. Bonakdar, Enhanced mechanical properties of thermosensitive chitosan hydrogel by silk fibers for cartilage tissue engineering, Mater. Sci. Eng., C, 2013, 33(8), 4786–4794, DOI:10.1016/j.msec.2013.07.043.
- I. F. Amaral, et al., Fibronectin-mediated endothelialisation of chitosan porous matrices, Biomaterials, 2009, 30(29), 5465–5475, DOI:10.1016/j.biomaterials.2009.06.056.
- D. Depan, B. Girase, J. S. Shah and R. D. K. Misra, Structure-process-property relationship of the polar graphene oxide-mediated cellular response and stimulated growth of osteoblasts on hybrid chitosan network structure nanocomposite scaffolds, Acta Biomater., 2011, 7(9), 3432–3445, DOI:10.1016/j.actbio.2011.05.019.
- L. Ma, et al., Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering, Biomaterials, 2003, 24(26), 4833–4841, DOI:10.1016/S0142-9612(03)00374-0.
- J. S. Mao, et al., A preliminary study on chitosan and gelatin polyelectrolyte complex cytocompatibility by cell cycle and apoptosis analysis, Biomaterials, 2004, 25(18), 3973–3981, DOI:10.1016/j.biomaterials.2003.10.080.
- S. Ladet, L. David and A. Domard, Multi-membrane hydrogels, Nature, 2008, 452(7183), 76–79, DOI:10.1038/nature06619.
- C. M. Hwang, et al., Fabrication of three-dimensional porous cell–laden hydrogel for tissue engineering, Biofabrication, 2010, 2(3), 035003, DOI:10.1088/1758-5082/2/3/035003.
- L. Xu, C. Wang, Y. Cui, A. Li, Y. Qiao and D. Qiu, Conjoined-network rendered stiff and tough hydrogels from biogenic molecules, Sci. Adv., 2019, 5(2), eaau3442, DOI:10.1126/sciadv.aau3442.
- T. F. Bodoza, F. Ntuli, D. F. Chauke and E. Muzenda, The effect of poly(ethylene-alt-maleic anhydride) on the reduction crystallisation behaviour of nickel powder, Can. J. Chem. Eng., 2013, 91(5), 822–829, DOI:10.1002/cjce.21753.
- C. J. B. Alejo, C. Fasciani, M. Grenier, J. C. Netto-Ferreira and J. C. Scaiano, Reduction of resazurin to resorufin catalyzed by gold nanoparticles: dramatic reaction acceleration by laser or LED plasmon excitation, Catal. Sci. Technol., 2011, 1(8), 1506–1511, 10.1039/c1cy00236h.
- G. V. Porcal, C. M. Previtali and S. G. Bertolotti, Photophysics of the phenoxazine dyes resazurin and resorufin in direct and reverse micelles, Dyes Pigm., 2009, 80(2), 206–211, DOI:10.1016/j.dyepig.2008.05.014.
- H. Vihola, A. Laukkanen, L. Valtola, H. Tenhu and J. Hirvonen, Cytotoxicity of thermosensitive polymers poly(N-isopropylacrylamide), poly(N-vinylcaprolactam) and amphiphilically modified poly(N-vinylcaprolactam), Biomaterials, 2005, 26(16), 3055–3064, DOI:10.1016/j.biomaterials.2004.09.008.
- D. Cody, A. Casey, I. Naydenova and E. Mihaylova, A Comparative Cytotoxic Evaluation of Acrylamide and Diacetone Acrylamide to Investigate Their Suitability for Holographic Photopolymer Formulations, Int. J. Polym. Sci., 2013, 2013, 1–6, DOI:10.1155/2013/564319.
- T. Jiang, et al., Chitosan-poly(lactide-co-glycolide) microsphere-based scaffolds for bone tissue engineering: in vitro degradation and in vivo bone regeneration studies, Acta Biomater., 2010, 6(9), 3457–3470, DOI:10.1016/j.actbio.2010.03.023.
- Y. Hong, H. Song, Y. Gong, Z. Mao, C. Gao and J. Shen, Covalently crosslinked chitosan hydrogel: properties of in vitro degradation and chondrocyte encapsulation, Acta Biomater., 2007, 3(1), 23–31, DOI:10.1016/j.actbio.2006.06.007.
- G. Lu, et al., Controlling the degradation of covalently cross-linked carboxymethyl chitosan utilizing bimodal molecular weight distribution, J. Biomater. Appl., 2009, 23(5), 435–451, DOI:10.1177/0885328208091661.
| This journal is © The Royal Society of Chemistry 2026 |