Impedimetric sensing of C-reactive protein using a novel molecularly imprinted polymer coupled with bismuth-enhanced cobalt ferrite nanocomposites for cardiovascular risk assessment
Siddhima
Singh
a,
Astha
Singh
a,
Neelottma
Singh
*a and
Jay
Singh
 *b
*b
aSam Higginbottom University of Agriculture, Technology and Sciences, Prayagraj, 211007, Uttar Pradesh, India. E-mail: neelottma.singh@shiats.edu.in; Tel: +91-8299585369
bDepartment of Chemistry, Institute of Sciences, Banaras Hindu University, Varanasi, 221005, Uttar Pradesh, India. E-mail: jaysingh.chem@bhu.ac.in; Tel: +91-9871766453
First published on 3rd December 2025
Abstract
This study reports an approach of using a molecularly imprinted polymer (MIP) combined with Bi-doped cobalt ferrite (BixCoFe2O4) nanoparticles (NPs) for detecting C-reactive protein (CRP), a marker associated with cardiovascular diseases (CVDs). Sudden cardiac arrest is a growing concern in India, where CVDs have become the leading cause of mortality. MIPs have recently drawn increasing interest over time; consequently, the objective of this study is to engineer an MIP-based electrochemical sensor due to their reliability, ease of electrochemical control for template removal, and cavity renewal. MIPs are selective polymers that can bind target molecules and are synthesised using a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 of a novel functional monomer (4-nitrophenyl methacrylate), a template (CRP), and a crosslinker (EGDMA) via the bulk polymerisation method, along with BixCoFe2O4 NPs (Bi = 0.05, 0.10, 0.15, and 0.20 M). These NPs and MIPs were characterised using powder X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, field emission scanning electron microscopy (FE-SEM), high-resolution transmission electron microscopy (HR-TEM), energy dispersive X-ray (EDX) analysis, dynamic light scattering (DLS), ultraviolet-visible (UV) spectroscopy, X-ray photoelectron spectroscopy (XPS), atomic force microscopy (AFM), and the Brunauer–Emmett–Teller (BET) method. The synthesised NPs and MIP were fabricated onto an indium tin oxide (ITO) electrode using the electrophoretic deposition (EPD) process. Moreover, an electrochemical analysis was conducted using voltammetry and electrochemical impedance sensing (EIS) techniques for CRP detection across two linear ranges: a lower range of 0.5–80 ng mL−1 with a limit of detection (LOD) of 0.1609 ng mL−1 and a sensitivity of 0.740 Ω ng−1 cm−2, and a higher range of 90–400 ng mL−1, in which the LOD was 0.3262 ng mL−1, sensitivity was 0.0631 Ω ng−1 cm−2 and the response time of the fabricated sensor was observed to be 10 seconds.
20 of a novel functional monomer (4-nitrophenyl methacrylate), a template (CRP), and a crosslinker (EGDMA) via the bulk polymerisation method, along with BixCoFe2O4 NPs (Bi = 0.05, 0.10, 0.15, and 0.20 M). These NPs and MIPs were characterised using powder X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, field emission scanning electron microscopy (FE-SEM), high-resolution transmission electron microscopy (HR-TEM), energy dispersive X-ray (EDX) analysis, dynamic light scattering (DLS), ultraviolet-visible (UV) spectroscopy, X-ray photoelectron spectroscopy (XPS), atomic force microscopy (AFM), and the Brunauer–Emmett–Teller (BET) method. The synthesised NPs and MIP were fabricated onto an indium tin oxide (ITO) electrode using the electrophoretic deposition (EPD) process. Moreover, an electrochemical analysis was conducted using voltammetry and electrochemical impedance sensing (EIS) techniques for CRP detection across two linear ranges: a lower range of 0.5–80 ng mL−1 with a limit of detection (LOD) of 0.1609 ng mL−1 and a sensitivity of 0.740 Ω ng−1 cm−2, and a higher range of 90–400 ng mL−1, in which the LOD was 0.3262 ng mL−1, sensitivity was 0.0631 Ω ng−1 cm−2 and the response time of the fabricated sensor was observed to be 10 seconds.
1. Introduction
Molecularly imprinted polymers (MIPs) are artificial polymer matrices specifically engineered to incorporate recognition sites for target molecules, referred to as templates during the synthesis process, which enables selective binding. MIPs mimic the natural biological recognition mechanisms found in enzymes and antibodies, earning them the nickname “plastic antibodies” because of their unique capabilities. These polymers are useful in drug delivery, separation, sensing, and more. A wide range of materials, including inorganic ions, pharmaceuticals, proteins, nucleic acids, and others, can be utilized in the preparation of MIPs. Methacrylic and acrylic monomers are commonly used in MIP formation because they often result in polymers with strong binding and recognition capabilities.1 The primary advantage of these monomers is their increased flexibility in choosing the appropriate functionalities and polymerisation methods, such as bulk polymerisation and surface polymerization.2 Based on the lock-and-key model for identifying target molecules, these artificial receptors are widely used in sample preparation.3 Their stability, robustness, specificity, affordability, ease of preparation, and capacity for miniaturisation offer several advantages over natural biomolecules, making them viable alternatives to natural bioreceptors in sensor technology. Although MIP-based sensors exhibit inherent stability and selectivity, they often encounter challenges such as sluggish electron transfer and poor conductivity.4 These limitations can hinder the sensitivity and response time of the electrode. One effective method to address these limitations is to incorporate nanoparticles (NPs) into MIP matrices.5 NPs, such as metal–organic frameworks (MOFs), Fe2O4, gold NPs, and quantum dots, exhibit superior signal transduction properties, a vast surface area, and enhanced electrical conductivity. When these NPs are incorporated into MIPs, they increase the density of recognition sites, facilitate rapid electron transfer, and intensify electrochemical signals, thus enhancing the sensor's response times. In this research work, Bi-doped cobalt ferrite NPs (BixCoFe2O4) are being utilized. As CoFe2O4 is a type of spinel ferrite NP (MFe2O4) known for its unique physicochemical properties, it improves the sensitivity, selectivity, and stability of the functionalized electrode.6 Doping bismuth (Bi3+) into cobalt ferrite enhances the electrical conductivity of NPs, benefiting electrochemical sensors. Bi3+ doping also improves the structural and physical properties, as well as the exchange current density of the electrode.7 The synthesis of BixCoFe2O4 (NPs) was performed using a hydrothermal method,8 as it is an energy-efficient and lower-temperature method compared to traditional methods. These NPs/MIP/ITO electrodes are further utilized in the detection of C-reactive protein (CRP), a sensitive indicator of inflammation.9,10 The liver produces CRP in response to inflammatory stimuli, and this research work discusses the detection of CRP, a marker of inflammation related to cardiovascular diseases (CVDs).10 Cardiac arrest has become a growing concern in India, where CVDs are the major cause of mortality. Among the various inflammatory biomarkers, the acute-phase protein CRP stands out. CRP is a strong predictor of low, moderate, and high-risk groups for CVD events, indicated by CRP levels of <1, 1–3, and >3 mg L−1, respectively.11,12 Highly sensitive CRP (Hs-CRP) can even detect low levels of inflammation as part of heart disease.13 CRP is a useful tool in CVD detection for risk prediction, therapy, and prognosis monitoring. S. Resende et al. fabricated a MIP on a photonic PDMS substrate for optical detection of CRP with a linear range of 5–1215 ng mL−1 with a LOD of 3 ng mL−1.14 Subsequently, the present work discusses a noble MIP with NPs electrodeposited on the ITO electrode to monitor CRP for cardiovascular disease detection.2. Experimental methodology
2.1. Chemicals
The following chemicals were used in this study: Co(NO3)2·6H2O, Bi(NO3)3·5H2O, and Fe(NO3)3·9H2O, purchased from Merck Specialities Private Ltd, Mumbai, India. Polyvinyl alcohol (PVA) and 25% solution of ammonium hydroxide (NH4OH) were used to control the pH during synthesis and were obtained from Sigma. Methacrylic acid (MA), CAS no. 149780, was acquired from CDH; acetonitrile (ACN) from Fisher Scientific; azobisisobutyronitrile (AIBN) and ethylene glycol dimethacrylate (EGDMA) from Thermo Scientific, produced by Naveen Chemical Pvt Ltd, Prayagraj. The indium tin oxide (ITO) glass electrode was sourced from UltraNanobiotech. C-reactive protein (CRP), used as a biomarker, was manufactured by Photozyme. Trypsin is from porcine pancreas obtained from TCI Co., Ltd. Hemoglobin, ascorbic acid, uric acid, histamine, and cholesterol were obtained from Sigma. Human blood serum was collected from Pathkind Lab, Mahmoorganj, Varanasi, U.P., India. All solutions were prepared with deionised water (DI), and glassware was thoroughly cleaned before use.2.2. Synthesis of doped NPs (BixCoFe2O4)
BixCoFe2O4 nanoparticles with varying Bi concentrations (x = 0.05, 0.10, 0.15, and 0.20 M) were synthesized using a hydrothermal process15 because it minimizes agglomeration and allows for achieving a controlled morphology, enabling efficient in situ doping of ions. Initially, a single-phase precursor solution was prepared by dissolving 0.5 M Co(NO3)2·6H2O and 0.1 M Fe(NO3)3·9H2O in DI water. Then, various concentrations of Bi(NO3)3·9H2O, corresponding to the desired Bi doping levels, were added to the precursor and dissolved completely. The resulting solution was stirred continuously at 70 °C to promote in situ doping of Bi3+. After maintaining magnetic stirring for 5 minutes, 0.5 g of polyvinyl alcohol (PVA) was added as a surfactant to stabilize the NPs and control their size and shape. A 25% ammonia solution was added dropwise until the pH reached 8–9, while maintaining the temperature at 70 °C. This temperature control was crucial in preventing metal oxide precipitation and ensuring uniform particle formation and purity, as illustrated in Fig. 1. This step aided in partial reduction and uniform mixing of the reactants. The final solution was transferred into a Teflon-lined stainless-steel autoclave and heated hydrothermally at 180 °C for 24 hours.16 After cooling to room temperature, a precipitate formed, which was separated by centrifugation at 5000 rpm, washed repeatedly (5–6 times) with DI water, and rinsed with a mixture of DI water and ethanol to remove residual impurities. Finally, the cleaned precipitate was dried at 80 °C to produce the Bi-doped CoFe2O4 NPs.2.3. Elaboration of virtual library
The three-dimensional (3D) structure of CRP was obtained from the PubMed online database. A virtual library of 10 different monomers was selected and created based on their chemical features that might interact selectively with CRP, and was generated using the online Cactus (chemical identifier resolver) server. Each monomer structure was then converted into SMILES (simplified molecular input line entry system) and PDB (protein data bank) formats. These monomers, chosen for their potential selectivity toward the analyte, were prepared for molecular docking studies. Furthermore, using AutoDock Vina, virtual docking was performed between CRP and each ligand (i.e., the monomer). The binding energies from these simulations were analyzed to identify the monomer with the strongest affinity for CRP, which was then selected as the most suitable ligand for selective recognition of CRP.172.4. Synthesis of monomer (4-nitrophenyl methacrylate)
A functional monomer, 4-nitrophenyl methacrylate (NPMA), commonly used as a copolymer in conducting polymers, was synthesized through an esterification reaction. First, 4.4 g of P-nitrophenol was dissolved in 40 mL of double-distilled water in a round-bottom flask (RBF) placed on a magnetic stirrer maintained at 70 °C. While stirring continuously, 1 M NaOH was added dropwise over one hour. Next, 400 mL of toluene, a nonpolar solvent, was added to the reaction mixture. The RBF was subsequently tied to a reflux apparatus and heated at 140 °C for two hours using a heating mantle. After the mixture cooled to room temperature, methacrylic acid was added slowly while stirring constantly. A yellow precipitate began to form within an hour. The mixture was then refluxed again at 80 °C for two hours. Once completed, the mixture was transferred to a Petri dish and dried in a water bath. This process produced the 4-nitrophenyl methacrylate functional monomer, which was later used in further polymerisation reactions18 (detailed reaction of 4-nitrophenyl methacrylate is provided in the SI, Fig. S1).2.5. Procedure for MIP synthesis
The synthesis of MIP was achieved through a bulk polymerisation process using a ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 of the functional monomer, template molecule, and crosslinker. CRP served as the template, 4-nitophenyl methacrylate was used as the functional monomer, and EGDMA acted as the crosslinker.19 These components were dissolved in acetonitrile (ACN) to form a homogeneous solution, as shown in Fig. 1a. The mixture was purged with nitrogen under inert conditions for at least 10 minutes. AIBN was then added as the initiator, and polymerisation was conducted at a controlled temperature of 35 °C. The polymerisation proceeded for 48 hours (in an oil bath), resulting in a white polymer product. During the post-polymerisation process, the material was washed for 24 hours with a mixture of methanol and acetic acid solution at a ratio of 4
20 of the functional monomer, template molecule, and crosslinker. CRP served as the template, 4-nitophenyl methacrylate was used as the functional monomer, and EGDMA acted as the crosslinker.19 These components were dissolved in acetonitrile (ACN) to form a homogeneous solution, as shown in Fig. 1a. The mixture was purged with nitrogen under inert conditions for at least 10 minutes. AIBN was then added as the initiator, and polymerisation was conducted at a controlled temperature of 35 °C. The polymerisation proceeded for 48 hours (in an oil bath), resulting in a white polymer product. During the post-polymerisation process, the material was washed for 24 hours with a mixture of methanol and acetic acid solution at a ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to remove the template, followed by a final rinse with methanol to eliminate residual impurities.20 This method follows established MIP synthesis protocols, in which template removal via solvent extraction creates selective binding cavities.
1 to remove the template, followed by a final rinse with methanol to eliminate residual impurities.20 This method follows established MIP synthesis protocols, in which template removal via solvent extraction creates selective binding cavities.
2.6. Fabrication of the ITO electrode with synthesised NPs
Before fabricating the ITO electrode, the surface was functionalized to modify its surface area. The ITO electrode was prepared using the electrophoretic deposition (EPD) process, which allows for uniform electrocoating. A thin film of Bi(0.15)CoFe2O4 NPs was deposited onto the conductive surface of a functionalized ITO glass electrode using an electrophoresis unit (Genentix-GX300). For deposition, a suspension was prepared by dispersing 30 mg of Bi(0.15)CoFe2O4 in a solution of ethanol and DI water in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio.
4 ratio.
The mixture was then sonicated for 60 minutes to ensure homogeneity. During the EPD process, the functionalized ITO electrode served as the cathode, while a copper electrode acted as the anode, forming a two-electrode system. Both electrodes were immersed vertically in the prepared NP suspension, maintaining a 1 cm distance between them. When a voltage of 35 mV and a current of 40 µA were applied, a thin film quickly formed on the conductive surface of the ITO electrode within seconds. Afterwards, Bi(0.15)CoFe2O4-coated ITO was rinsed with DI water to clean off any excess or roughly attached particles. Similarly, a suspension solution was prepared with the MIP material for coating the Bi(0.15)CoFe2O4/ITO electrode via the EPD process. Finally, the fabricated electrode with the MIP was stored in a temperature-controlled environment for subsequent use.21
2.7. Preparation/formulation of standard stock
A stock solution for CRP sensing related to CVDs via the fabricated Bi(0.15)CoFe2O4 NPs/MIP ITO electrode was prepared in PBS (phosphate buffer solution), pH 6. This solution was used to dilute the 55 mg L−1 standard solution of CRP, creating an analytical range at lower concentrations (0.05–80 ng mL−1) and higher concentrations (90–400 ng mL−1), to facilitate electrochemical sensing through electrochemical impedance sensing (EIS).2.8. Optimization of the ITO electrode
The electrochemical reaction involves a three-electrode system, where the Bi(0.15)CoFe2O4/MIP/ITO electrode serves as the working electrode. Compared to a two-electrode system, this configuration offers a more precise and controlled environment for electrochemical analysis.22 The oxidation–reduction behaviour of the Bi(0.15)CoFe2O4/MIP/ITO electrode was investigated in a 50 mM PBS solution containing 5 mM Fe[(CN)6] and 0.9% NaCl. The electrode was developed using the EPD method onto ITO at a scan rate of 50 mV s−1. Redox reactions were investigated, and EIS studies were employed for investigating sensing performance, and CV was utilised for various analysis purposes, such as pH, the scan rate, response time, reusability, stability, and real sample analysis, over a potential range from −0.4 to 0.7 V. The characterization technique is discussed in detail in the SI, Section S1.3. Results and discussion
3.1. X-ray diffraction analysis
XRD patterns of BixCoFe2O4 with “x” values of 0.05, 0.10, 0.15, and 0.20 M are provided in the SI S2(a). The cobalt ferrite NPs obtained feature a single-phase face-centred cubic inverse spinel structure showing diffraction peaks at (220), (311), (400), (511), (440), and (622). Each diffraction peak corresponds perfectly to the JCPDS card no. 22-1086. When bismuth is substituted in modest amounts, the spinal structure remains unaffected, maintaining a single-phase cubic spinel structure.8 The intense peak obtained at hkl = 311 is located at an angle 2θ = 35.53.The crystal size is calculated through the Scherrer eqn (1)
 | (1) |
3.2. FTIR analysis
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) carbonyl stretching is detected around 1720–1740 cm−1. The peak between 1630 and 1680 cm−1 is attributed to C
O) carbonyl stretching is detected around 1720–1740 cm−1. The peak between 1630 and 1680 cm−1 is attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching, indicating methylated double bond stretching. The asymmetric stretching peak appears around 1520–1559 cm−1, and the symmetric peak appears around 1340–1389 cm−1, which is attributed to N
C stretching, indicating methylated double bond stretching. The asymmetric stretching peak appears around 1520–1559 cm−1, and the symmetric peak appears around 1340–1389 cm−1, which is attributed to N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, indicating the presence of a nitro group, and a peak around 900–700 cm−1 is attributed to C–C stretching.18
O stretching, indicating the presence of a nitro group, and a peak around 900–700 cm−1 is attributed to C–C stretching.18
3.3. 1H NMR spectrum of 4-nitrophenyl methacrylate
The 1H NMR spectrum of 4-nitrophenyl methacrylate is provided in the SI S4. The peak observed around 7–8 ppm is attributed to the aromatic protons on the benzene ring. The nitro group is strongly electron-withdrawing. Peaks around the 5.5–6.5 ppm region correspond to the vinyl protons (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2) of the methacrylate group, as these signals appear for the terminal alkene's protons. The peak around 2 ppm corresponds to the methyl group (CH3) attached to the methacrylate double bond. These peaks confirm the synthesis of 4-nitrophenyl methacrylate.18
CH2) of the methacrylate group, as these signals appear for the terminal alkene's protons. The peak around 2 ppm corresponds to the methyl group (CH3) attached to the methacrylate double bond. These peaks confirm the synthesis of 4-nitrophenyl methacrylate.18
3.4. UV analysis
The UV spectra of BixCoFe2O4 (x = 0.05, 0.10, 0.15, 0.20) are shown in Fig. 3(a). UV spectroscopy gives a piece of qualitative information about the different concentrations. In general, UV radiation excites electrons to higher energy levels, leading to the absorption of light.24Fig. 3(a) shows a plot of absorbance versus wavelength, showing the strong absorption in the region between 200 and 400 nm, and the absorption decreases as the wavelength increases. Furthermore, a Tauc plot is used to analyze a material's absorption spectra to determine its optical band gap, so a Tauc plot of BixCoFe2O4 with a band gap of 2.3–2.9 eV is shown in Fig. 3(b), showing the optical properties of the material. | ||
| Fig. 3 (a) UV spectra of Bi-doped cobalt ferrite, (b) the Tauc plot of modified NPs, (c) and (d) the BET image of synthesized MIP and NIP, and (e) and (f) the AFM image of Bi(0.15)CoFe2O4. | ||
3.5. BET analysis
The average pore size, surface area and pore volume of MIP are measured using the BET technique. Fig. 3(c) and (d) illustrate the BET analysis of MIP and NIP. In MIP, the specific surface area increased, confirming a rise in the porosity of MIP, along with a slight increase in pore radii. This change is attributed to the enhanced porous nature of MIP and the increase in the number of pores. In MIP, the pore volume decreases by only 0.005 units25 (Table S1 shows the pore size of the prepared MIP and NIP, provided in the SI).3.6. AFM analysis
AFM was used to analyse the surface morphology and topography of the modified material (Bi(0.15)CoFe2O4), with a 3D and 2D model as shown in Fig. 3(e and f).26 It shows an agglomerated nanostructure with an irregular grain size, indicating the polycrystalline nature of cobalt ferrite with a dopant like bismuth. The roughness value is 17.21 nm, suggesting a fine nanoscale texture, and this enhances the effective surface area. Furthermore, the measured film thickness is 0.52 µm, indicating a sufficiently thick film for robust electrode modification, and the lateral scan is recorded over 10 × 10 µm, showing the surface uniformity.3.7. XPS analysis
The element composition and chemical status of the synthesised Bi(0.15)CoFe2O4 NPs and CoFe2O4 (XPS of CoFe2O4 is provided in the SI, Fig. S5) were characterised by XPS. The Co 2p spectra of Bi(0.15)CoFe2O4 NPs and CoFe2O4 show Co2+ and Co3+ states, which cause slight variations in binding energy (BE) peaks around 778.7 eV and 792.9 eV in the Bi(0.15)CoFe2O4 spectrum [see Fig. 4(a)], and in the BE peaks around 781 eV and 796 eV in the CoFe2O4 spectrum [see Fig. S5(a)]. Furthermore, a clear spectrum of Bi 4f is depicted in Fig. 4(C), which is absent for CoFe2O4. The Bi 4f doublets are illustrated around 159 eV (4f7/2) and 164 eV (4f5/2), which confirms the Bi doping in cobalt ferrite. Slight variations in the shape and intensity of satellite peaks suggest that the environment of cobalt has been changed, which may be due to oxidation state distribution or changes in the cation. The XPS spectra of Fe 2p show the main 2p3/2 and 2p1/2 lines at about 706–720 eV. The 2p3/2 B.E. peak of spinel ferrite appears around 708 eV for Fe2+ ions in Bi(0.15)CoFe2O4 and at 711.5 eV for Fe3+ ions in CoFe2O4.27 Further minor changes in the shape of the peak suggest that Bi influences the Fe 2p electronic structure. The O 1s spectra of Bi(0.15)CoFe2O4 show a notable shift of peaks toward lower B.E. around 523.3 eV, 526.07 eV and 527.2 eV, compared to the CoFe2O4 spectral peaks around 527.80, 530.02 eV, and 530.57 eV, suggesting the modification of oxygen environments caused by Bi doping, which can introduce oxygen vacancies or alter the chemical shift. Thus, Bi3+ was successfully incorporated into cobalt ferrite, changing its surface chemistry and electronic structure significantly compared to the undoped material.283.8. Surface morphology and elemental mapping
The surface morphology of the synthesised Bi(0.15)CoFe2O4 NPs is shown in Fig. 5(a) and (b) at 1 µm and 300 nm resolution, respectively. This image illustrates that the distribution of the particles is non-uniform, and the particles are randomly shaped and are of different sizes. Observed agglomerations or clusters of NPs could potentially be interpreted due to the Bi(0.15)CoFe2O4 NPs’ magnetic interactions. Due to the overlapping and clumping of smaller nanoparticles, they also exhibit a catalytic morphology. The Bi doping causes a slight reduction in lattice parameters due to the replacement of Fe3+ ions by the larger Bi3+ ions, which can be inferred from microstructural changes seen in SEM.29 The morphological structures of MIP and NIP are recorded under a magnification of 500 nm resolution, as illustrated in Fig. 6(c), showing a clearer chain-like structure with a greater number of pores, resulting from the removal of CRP, which creates cavities suitable for CRP rebinding. Fig. 6(d) depicts a bulkier, agglomerated form of the particles with fewer pores, displaying a chain-like structure of the synthesised polymer without CRP. SEM images further confirm that the MIP and NIP have been successfully synthesised. [All the original images of FE-SEM used in the manuscript are provided in the SI as Fig. S11 and S12.] | ||
| Fig. 5 The SEM images of (a) and (b) Bi(0.15)CoFe2O4 at 1 µm and 300 nm resolution, (c) NIP, and (d) MIP. | ||
Elemental mapping of Bi(0.15)CoFe2O4 is given in the SI, Fig. S6. The figure demonstrates the presence of oxygen, bismuth, iron, and cobalt in different colours, confirming the synthesis of Bi(0.15)CoFe2O4.
3.9. TEM/HR-TEM, EDX and DLS analyses
HR-TEM is used to analyse the atomic structure of Bi(0.15)CoFe2O4, at different resolutions, and it is an important technique for determining the nanostructure of the synthesised NPs. Fig. 6(a) shows the HR-TEM image at 50 nm resolution, revealing the internal structure and aggregation state of the nanomaterial; meanwhile, Fig. 6(b) shows the higher-resolution image at 20 nm, and the inset histogram reveals that the average particle size of Bi(0.15)CoFe2O4 is 17 nm, with data evaluated by ImageJ software. Fig. 6(c) depicts the selected area electron diffraction (SAED) image, indicating the crystallinity of the NPs, which is validated by XRD data matching the hkl values with JCPDS card no. 22-1086. The identified planes are 111, 220, 311, 311, 400, 511, and 440. Fig. 6(d) shows the fringes at 10 nm, and Fig. 6(e) provides a zoomed-in image of 10 nm, clearly showing the fringes and allowing for the calculation of the d-spacing (the distance between lattice fringes) using ImageJ software, which is 2.9 nm with an hkl value of 220; this is further validated by the XRD data.8The EDX spectrum, shown in Fig. 6(f), displays the elemental composition of the synthesized material with tabulated data of elements, weight%, and atomic%. Elements like Bi have a weight% of 13.5 and an atomic% of 3.1; Co has a weight% of 23.8 and an atomic% of 19.3; Fe has a weight% of 51.3 and an atomic% of 43.9; and O has a weight% of 11.3 and an atomic% of 33.8, respectively [all the original images of HR-TEM used in the manuscript are provided in the SI in Fig. S13].
The particle size distribution was determined by using the DLS technique. The hydrodynamic diameter of the core, solvent layers, and any molecules adsorbed on the surface of the Bi(0.15)CoFe2O4 has been measured. The polydispersity index (PDI) is 0.613. The sample, considered to be highly polydisperse, reflects the hydrodynamic diameter of the nanoparticles. The DLS graph is provided in the SI, Fig. S7.
4. Electrochemical analysis
4.1. pH optimization
pH optimization is a crucial step in electrochemical analysis, as it has a significant effect on the stability of the electrode and the kinetics of electron transfer reactions. The buffer system is essential for maintaining pH stability and also affects the redox potential, which is critical for accurate and reproducible electrochemical measurements. To explore the effects of pH on the electrochemical performance of the Bi(0.15)CoFe2O4/MIP/ITO electrode, cyclic voltammetry was performed in phosphate-buffered saline (PBS) with ferri/ferrocyanide. At pH 6.5, the highest maximum current was recorded, as shown in Fig. 7(a), indicating that this pH is optimal for electrochemical activity. Therefore, all further electrochemical analyses of the Bi(0.15)CoFe2O4/MIP/ITO electrode were conducted at pH 6.5 to ensure the maximum sensitivity and stability.4.2. Comparative study of the modified electrode
To assess the impact of the synthesized material (BixCoFe2O4) with MIP and NIP on the bare ITO electrode, different techniques (CV, DPV, and EIS) were used for a comparative electrochemical study. This comparative study validates the fabrication process and optimizes the conditions for analyte detection.Firstly, the CV technique was used for a comparative study of the bare ITO electrode, and the current was noted to be 0.67 mA. After that, a CV study was conducted for different concentrations of BixCoFe2O4 (x = 0.05, 0.10, 0.15, and 0.20 M), and the current of Bi(0.15)CoFe2O4 was observed to be 1.160 mA, the highest among all the concentrations, as illustrated in Fig. 7(b). Furthermore, MIP is electrodeposited on the BixCoFe2O4/ITO surface, and a CV study was performed for the BixCoFe2O4/MIP/ITO electrode, where x = 0.05, 0.10, 0.15, and 0.20 M, which also followed a similar trend, and the current of the Bi(0.15)CoFe2O4/MIP/ITO electrode is the highest among all the concentrations, that is 0.886 mA. Hence, the Bi(0.15)CoFe2O4/MIP/ITO electrode was used in further studies, as shown in Fig. 7(c). Similarly, NIP is also electrodeposited on the BixCoFe2O4/ITO surface, which follows the same trend as the Bi(0.15)CoFe2O4/MIP/ITO electrode [CV curves are given in SI, Fig. S4(a)]. Subsequently, a CV study is performed for bare ITO, BixCoFe2O4/ITO, the Bi(0.15)CoFe2O4/MIP/ITO electrode, and the Bi(0.15)CoFe2O4/NIP/ITO electrode.
Furthermore, CV was used to evaluate the redox performance of the bare ITO, Bi(0.15)CoFe2O4/ITO, and Bi(0.15)CoFe2O4/MIP/ITO electrode, as shown in Fig. 7(d), towards 5 mM ferri/ferrocyanide in 0.9% NaCl. The scan rate was set at 50 mV s−1. The Randles–Sevcik equation is used to calculate the surface-active area.
| ip = 2.69 × 105n3/2AD1/2CV1/2 | (2) |
Subsequently, a DPV and EIS study conducted for a comparative analysis follows a similar trend, with the current flowing through the electrode in a pattern similar to that depicted in Fig. 7(e) and (f).
4.3. Scan rate and kinetic analysis of the Bi(0.15)CoFe2O4/MIP/ITO electrode
A kinetic study of the modified Bi(0.15)CoFe2O4/MIP/ITO electrode was conducted by adjusting the scan rate (10–100 mV s−1) during the CV analysis (Fig. 7(g)). The analyte–electrode interactions are influenced by the scan rate, which is a controlled parameter during the experiment and determines how rapidly the applied potential varies. As the scan rate increases, the diffusion layer becomes thinner, resulting in a higher current response.30 Furthermore, a shift in potential occurs, and the redox peak current response of the dispersed species varies linearly with the square root of the scan rate (v1/2) (Fig. 7(h)). As the scan rate increased, the oxidation and reduction peak potentials shifted toward more positive and negative values, respectively, confirming an electrochemical quasi-reversible redox process. The potential shift of the peaks is shown in Fig. 7(g), which similarly demonstrates a linear relationship between v1/2 and peak-to-peak separation (ΔEp = Epa − Epc).The Laviron model is used to calculate the charge transfer rate constant (ks) as represented in eqn (3) and (4).
| m = (RT/F)(Ks/nv) | (3) |
| Ks = mnFv/RT | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1); R is the gas constant (8.314 J K−1 mol); and T is the temperature (27 °C). The calculated charge transfer rate constant (ks) value of the Bi(0.15)CoFe2O4/MIP/ITO electrode is 0.7 cm s−1.
485 C mol−1); R is the gas constant (8.314 J K−1 mol); and T is the temperature (27 °C). The calculated charge transfer rate constant (ks) value of the Bi(0.15)CoFe2O4/MIP/ITO electrode is 0.7 cm s−1.
Subsequently, the surface concentration of the modified electrode is calculated using the Brown–Anson model eqn (5):
| γ = Ip × 4RTn2F2Av | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), and A is the modified electrode's surface area of 1.585 cm2. The calculated surface concentration is 1.59 × 10−8 mol cm−2.
485 C mol−1), and A is the modified electrode's surface area of 1.585 cm2. The calculated surface concentration is 1.59 × 10−8 mol cm−2.
The Randles–Sevick equation is used to calculate the diffusion coefficient (D) of the modified electrode:
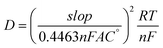 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), R is the gas constant (8.314 J K−1 mol), T is the temperature (27 °C), and C° is the concentration of [Fe(CN)6]3−/4− (5 mM). The diffusion concentration is calculated to be 1.22 × 10−4 cm2 s−1 [complete calculation of the scan rate is provided in SI, Table S2].
485 C mol−1), R is the gas constant (8.314 J K−1 mol), T is the temperature (27 °C), and C° is the concentration of [Fe(CN)6]3−/4− (5 mM). The diffusion concentration is calculated to be 1.22 × 10−4 cm2 s−1 [complete calculation of the scan rate is provided in SI, Table S2].
4.4. EIS sensing of the Bi(0.15)CoFe2O4/MIP/ITO electrode
Electrochemical impedance spectroscopy (EIS) is a key electrochemical technique used to measure the impedance of a circuit, expressed in ohms, a unit of resistance. Impedance consists of two components: a real part and an imaginary part. When the real component (Z′) is plotted along the X-axis and the imaginary component (Z″) along the Y-axis, the resulting graph is known as a Nyquist plot.31For EIS sensing, two different concentration ranges were considered: lower and higher concentrations, as shown in Fig. 8(a) and (b), respectively. The lower range spans from 0.5 to 80 ng mL−1, with a linear regression (R2) value of 0.9887, a sensitivity of 0.740 Ω ng−1 cm−2, and a limit of detection (LOD) of 0.16545 ng mL−1, respectively, which are detected via an impedimetric study (Fig. 8(c)). Conversely, at higher concentrations, 90–400 ng mL−1, a more gradual linear region appears, with an R2 of 0.98162, a sensitivity of 0.0631 Ω ng−1 cm−2, and a LOD of 0.3261 ng mL−1, respectively (Fig. 8(d)). The R2 values of both concentrations indicate excellent linear fits, suggesting that these can be used as a reliable calibration curve.
The electrochemical response of the sensor exhibited an inverse relationship with the C-reactive protein (CRP) concentration, where higher CRP levels caused a proportional decrease in the peak current response. The curve value indicates identical circuit components, and eqn (7) represents the Rseries, which is the overall series resistance.
| Rseries = Rs + Rct + Rd | (7) |
| Sensor | Probe | Technique | Linear range (ng mL−1) | Limit of detection (ng mL−1) | Reference |
|---|---|---|---|---|---|
| Note: PLA: polylactic acid; Dist-LFIA: distance-based lateral flow immunoassay; AuNPs: gold nanoparticles; and EIS: electrochemical impedance spectroscopy. | |||||
| PLA arrayed microneedle | Aptamer | Calorimetric assay | 0–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1 000 ng mL−1 |
100–500 ng mL−1 | 32 |
| AuNPs | Anti-CRP | Dist-LFIA | 0–200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1 000 ng mL−1 |
120 ng mL−1 | 33 |
| Microfluidic | Anti-CRP | Electrochemical sensor | 100–1000 ng mL−1 | 100 ng mL−1 | 34 |
| Chemiresistive CuO | Anti-CRP | Linear sweep voltammetry | 100–500 ng mL−1 | 49 ng mL−1 | 35 |
| Bi(0.15)CoFe2O4/MIP/ITO electrode | MIP | EIS | 0.5–80 ng ml−1 | 0.16 ng mL−1 | Present work |
| MIP | EIS | 90–400 ng mL−1 | 0.32 ng mL−1 | Present work | |
4.5. Response time
In electrochemical analysis, response time is an important parameter, as it gauges how quickly an analyte interacts with the modified electrode. A graph for response time is plotted as current (mA) vs. time (s), which is also referred to as a digital measuring parameter. A response time study of the Bi(0.15)CoFe2O4/MIP/ITO electrode was conducted over a range of 1–60 seconds at 50 mV s−1. During the study, the current was amplified from 1 to 30 seconds, after which a steady current was observed up to 60 seconds [CV curves are given in SI, Fig. S8(a)]. The maximum current of 0.84 mA is measured at 10 seconds (Fig. 7(i)), and RSD was also calculated to be 1.98% [complete calculation is given in SI, Table S3].4.6. Stability and reusability of the electrode
While keeping the Bi(0.15)CoFe2O4/MIP/ITO electrode dry at a temperature of 4 °C, its stability was examined, as stability and reusability of the electrode have been crucial aspects of electrochemical analysis. The CV technique was used to assess both stability and reusability at a scan rate of 50 mV s−1. [Corresponding CV curves are given in SI, Fig. S8(b).] The stability of the Bi(0.15)CoFe2O4/MIP/ITO electrode was investigated throughout 1 to 60 days in PBS with regular intervals of 5 days; after 30 days, activity of the electrode is 0.728 mA with an RSD of 1.65%, and after 60 days, the current response was 0.705 mA with an RSD of 4.28% (Fig. 8(e)) [calculated data are given in SI, Table S4]. To evaluate the reusability of the Bi(0.15)CoFe2O4/MIP/ITO electrode, repetitive CV signal response was measured. The current response was recorded for up to 15 scans; the current increased or remained similar up to the 10th scan (0.684 mA), and then gradually decreased after the 11th scan, with an RSD of 2.3% (Fig. 8(f)). [Corresponding CV curves are given in SI, Fig. S8(c).] Thus, the Bi(0.15)CoFe2O4/MIP/ITO electrode demonstrated outstanding reusability and sustained CRP sensing with MIP according to CVD analysis [calculated data are given in SI, Table S5].4.7. Interference and real sample study
The amperometric response of the Bi(0.15)CoFe2O4/MIP/ITO electrode was utilised to evaluate selectivity in the presence of common interferents such as ascorbic acid, cholesterol, glucose, histamine, uric acid, haemoglobin, and trypsin. The CV technique was employed in PBS (50 mM) at pH 6.5 and 0.9% NaCl solution, using [Fe(CN)6]3−/4− (5 mM) as a mediator. The scan rate was set at 50 mV s−1 in the potential range of 0.4 to −0.7 V. For the standard CRP (80 ng mL−1) [the corresponding CV curves are given in the SI, Fig. S9], a slight decrement in Ipa was observed as shown in Fig. 8(g). The RSD is approximately 13% and the recovery is 90% with the standard sample in the presence of interferents [detailed data are available in the SI, Table S6 (a–h)].As shown in Fig. 8(h), a real sample was also used to assess the standard CRP sample and confirm the performance of the Bi(0.15)CoFe2O4/MIP/ITO electrode. Blood serum samples with identical CRP concentrations were obtained: serum sample C-1 was 2.9 mg L−1, serum sample C-2 was 5.2 mg L−1, serum sample C-3 was 3.3 mg L−1, and serum sample C-4 was 6.7 mg L−1, and the samples were diluted in the analyte ranges for validation of the modified electrode, respectively. Furthermore, these serum samples were dissolved at pH 6.5 to validate the Bi(0.15)CoFe2O4/MIP/ITO electrode. The range of detection is 40–90 ng mL−1 for analytes and serum samples C-1, C-2, C-3, and C-4 [corresponding CV curves are given in the SI, Fig. S10]. The RSD is approximately 5%, and an average recovery of 95% is observed, indicating a minimal variation in the blood samples analysed using the Bi(0.15)CoFe2O4/MIP/ITO electrode for CRP with respect to CVD detection [detailed data are available in the SI, Table S7(a–d)].
5. Conclusion
In this study, a Bi(0.15)CoFe2O4/MIP/ITO electrode was effectively modified and characterised using advanced analytical techniques for CRP detection in CVDs. Initially, Bi(0.15)CoFe2O4 was synthesised via a hydrothermal method, which enhances the electrode's conductivity, increases the electron transfer rate on the surface, and provides a larger surface area. A MIP was synthesised through bulk polymerisation to achieve highly sensitive and selective CRP detection. Structural and morphological characterisation was performed on the synthesised nanoparticles and the MIP. The modified electrode operates within two concentration ranges: a lower range (0.5–90 ng mL−1) with a LOD of 0.1609 ng mL−1 and a sensitivity of 0.740 Ω ng−1 cm−2, and a higher range (100–400 ng mL−1) with a LOD of 0.3262 ng mL−1 and a sensitivity of 0.0631 Ω ng−1 cm−2. This sensor offers a rapid response time of 10 seconds. It shows great potential for use in portable devices and low-cost biomedical diagnostics.Author contributions
S. S.: data curation, investigation, visualisation, and writing – original draft. A. S.: data curation, data validation, and writing – review of the original draft. N. S.: conceptualisation, validation, project administration, supervision, and writing – review and editing of the original draft. J. S.: conceptualisation, validation, project administration, resources, supervision, and writing – review and editing of the original draft.Conflicts of interest
The authors declare no competing financial interest. This work received no specific grant from public, commercial, or not-for-profit funding agencies.Data availability
All the data associated with this work have been included as part of the supplementary information (SI). Supplementary information: characterisation techniques, DLS, and high-resolution images: SEM, HR-TEM, AFM, and FTIR. Interference study of different interferences towards the CRP, tabulation of the real blood serum samples studied: % recovery, determination of concentration, and % RSD towards the standard CRP, and CV graph of the fabricated Bi(0.15)CoFe2O4/MIP/ITO electrode towards CRP, and complete calculations with % recovery and %RSD of stability, response time, reusability, and the scan rate. See DOI: https://doi.org/10.1039/d5tb01698c.Acknowledgements
This work received no specific grant from public, commercial, or not-for-profit funding agencies. S. S. and A. S. are thankful to their affiliated institution for providing constant financial support. N. S. acknowledges her institute for providing constant support. J. S. acknowledges BHU for providing a additional seed grant and a BRIDGE grant under the MoE Govt. India, Institute of Eminence (IoE), under Dev. Scheme No. 6031 and 6031A, respectively.References
- L. Wang, M. Pagett and W. Zhang, Sens. Actuators, Rep., 2023, 5, 100153 CrossRef
.
- Y. Li, L. Luo, Y. Kong, Y. Li, Q. Wang, M. Wang, Y. Li, A. Davenport and B. Li, Biosens. Bioelectron., 2024, 249, 116018 CrossRef CAS PubMed
.
- T. Sajini and B. Mathew, Talanta Open, 2021, 4, 100072 CrossRef
.
- A. Al Faysal, A. Cetinkaya, T. Erdoğan, S. A. Ozkan and A. Gölcü, Talanta, 2025, 281, 126791 CrossRef CAS PubMed
.
- R. Ali and M. M. El-Wekil, J. Alloys Compd., 2023, 962, 171180 CrossRef CAS
.
- S. M. Ansari, A. Younis, Y. D. Kolekar and C. V. Ramana, Appl. Phys. Rev., 2025, 12, 021308 CAS
.
- S. Patil, S. Meti, P. S. Kanavi, R. F. Bhajantri, M. Anandalli, R. Mondal, S. Karmakar, M. Muhiuddin, M. R. Rahman, B. C. Kumar and B. G. Hegde, Mater. Sci. Eng. B, 2023, 296, 116570 CrossRef CAS
.
- R. Kalia, A. Chauhan, R. Jasrotia, M. F. Ijaz, K. M. Batoo and R. Verma, J. Mater. Sci.: Mater. Electron., 2025, 36, 483 CrossRef CAS
.
- A. Singh, S. Singh, N. Singh and J. Singh, ACS Appl. Bio Mater., 2025, 8, 5078–5089 CrossRef CAS PubMed
.
- R. Grewal, G. A. Ortega, F. Geng, S. Srinivasan and A. R. Rajabzadeh, Bioelectrochemistry, 2024, 159, 108743 CrossRef CAS PubMed
.
- S. Singh, A. Singh, S. Shaktawat, N. Singh and J. Singh, Langmuir, 2025, 41, 20567–20578 CrossRef CAS PubMed
.
- S. Balayan, N. Chauhan, V. Hooda, R. Chandra, W. Rosario and U. Jain, Talanta Open, 2024, 10, 100369 CrossRef
.
- S.-C. Chang, C.-H. Wang, K.-H. Cheng and G.-B. Lee, Sens. Actuators, B, 2025, 440, 137949 CrossRef CAS
.
- S. Resende, J. Fernandes, P. C. Sousa, C. Calaza, M. F. Frasco, P. P. Freitas, M. Goreti and F. Sales, Chem. Eng. J., 2024, 485, 149924 CrossRef CAS
.
- R. Priyadharsini, P. Dhamodharan, M. Venkateshwarlu and C. Manoharan, Solid State Sci., 2023, 137, 107123 CrossRef CAS
.
- A. N. Alqarni, M. A. Almessiere, S. Güner, M. Sertkol, S. E. Shirsath, N. Tashkandi and A. Baykal, Ceram. Int., 2022, 48, 5450–5458 CrossRef CAS
.
- P. C. Agu, C. A. Afiukwa, O. U. Orji, E. M. Ezeh, I. H. Ofoke, C. O. Ogbu, E. I. Ugwuja and P. M. Aja, Sci. Rep., 2023, 13, 13398 CrossRef CAS PubMed
.
- R. Soltanmoradi and M. H. Nasirtabrizi, Cumhuriyet Üniversitesi Fen Edebiyat Fakultesi Fen Bilimleri Dergisi, 2015, 36, 1664–1673 Search PubMed
.
- A. N. Hasanah and I. Susanti, Polymers, 2023, 15, 3401 CrossRef CAS PubMed
.
- S. Akgönüllü, S. Kılıç, C. Esen and A. Denizli, Polymers, 2023, 15, 629 CrossRef PubMed
.
- S. Shaktawat, R. Verma, K. R. Singh and J. Singh, Sustainable Food Technol., 2024, 2, 447–460 RSC
.
- M. Abbasi, A. R. Mahmoudi and K. Asadpour-Zeynali, Sens. BioSensing Res., 2024, 43, 100635 CrossRef
.
- K. M. Batoo, M. Hadi, A. Chauhan, R. Verma, M. Singh, O. M. Aldossary and G. K. Bhargava, Appl. Phys. A: Mater. Sci. Process., 2022, 128, 283 CrossRef CAS
.
- N. Dhanda, P. Thakur and A. Thakur, Mater. Today: Proc., 2023, 73, 237–240 CAS
.
- J. Dolai, H. Ali and N. R. Jana, New J. Chem., 2021, 45, 19088–19096 RSC
.
- A. A. H. El-Bassuony, R. S. Hafez, N. M. S. Matter and H. K. Abdelsalam, JOM, 2024, 76, 3945–3959 CrossRef CAS
.
- S. R. Naik and A. V. Salker, J. Mater. Chem., 2012, 22, 2740–2750 RSC
.
- V. S. Kirankumar and S. Sumathi, J. Mater. Sci.: Mater. Electron., 2018, 29, 8738–8746 CrossRef CAS
.
- M. Jamshaid, H. Khan, M. A. Nazir, M. A. Wattoo, K. Shahzad, M. Malik and A.-U. Rehman, Int. J. Environ. Anal. Chem., 2024, 104, 1186–1201 CrossRef CAS
.
- N. Elgrishi, K. J. Rountree, B. D. McCarthy, E. S. Rountree, T. T. Eisenhart and J. L. Dempsey, J. Chem. Educ., 2018, 95, 197–206 CrossRef CAS
.
- H. S. Magar, R. Y. A. Hassan and A. Mulchandani, Sensors, 2021, 21, 6578 CrossRef CAS PubMed
.
- G.-B. Lee, S.-C. Chang, C.-H. Wang and K.-H. Cheng, 2024, 5017909, Available at SSRN.
- S. Ruantip, U. Pimpitak, S. Puthong, S. Rengpipat, A. Yakoh, M. M. Sain, K. Komolpis and S. Chaiyo, Sens. Actuators, B, 2025, 434, 137602 CrossRef CAS
.
- Z.-X. Lai, C.-C. Wu and N.-T. Huang, Biosensors, 2022, 12, 1163 CrossRef CAS
.
- F. Bayansal and H. A. Çetinkara, J. Alloys Compd., 2022, 926, 166737 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |






