 Open Access Article
Open Access ArticleProbing electrochemical phenomena in commercial pouch cells using a multimodal magnetic resonance approach
Konstantin Romanenko *a and
Nikolai I. Avdievichb
*a and
Nikolai I. Avdievichb
aUniversité Paris-Saclay, CEA, CNRS, NIMBE, Gif-sur-Yvette, 91191, France. E-mail: konstantin.romanenko@cea.fr
bMax-Planck Institute for Biological Cybernetics, Tübingen, 72076, Germany
First published on 30th April 2026
Abstract
Further advances in portable electrochemical energy storage will increasingly depend on multifunctional in situ diagnostic tools capable of probing materials at the molecular level under realistic operating conditions. This capability is essential for achieving an optimal balance between energy density, cycle life, and safety. The increasing energy density of battery cells reinforces the need for rapid, reliable, and non-destructive diagnostics, including evaluation of state-of-health (SoH) and state-of-charge (SoC). Nuclear magnetic resonance (NMR) spectroscopy and imaging (MRI) are highly sensitive to the environment and dynamics of key electrochemical elements, but most common industrial cell designs present significant obstacles: (a) interrogated nuclear spins are shielded from the radio-frequency (RF) field by conductive metallic structures such as casings and jelly roll sheets of the current collectors, and (b) magnetism from bulk metallic components causes misregistration artefacts and reduces resolution and signal-to-noise ratio. Surface-scan MRI and plug-and-play (PnP) NMR are two distinct in situ modalities designed to address these challenges. In this work, we combine the PnP RF adapter and surface-scan MRI sensor into a single multimodal device suitable for a wide range of academic and industrial applications, including SoH analysis and recycling. We illustrate the capabilities of this approach through complementary NMR and MRI experiments, highlighting diverse lithium environments and their transformations during electrochemical cycling in commercial LiCoO2‖graphite pouch cells.
1 Introduction
Traditional electrochemical techniques, such as galvanostatic cycling, cyclic voltammetry (CV), and electrochemical impedance spectroscopy (EIS), rely on several integral indicators, such as voltage, capacity, impedance, and their derivatives. These commonly used methods lack molecular-level detail and spatial resolution, often leading to inconclusive or inconsistent results when investigating complex multiphase, interfacial electrochemical phenomena and subtle degradation or mechanical defects. To gain deeper insight into electrochemical systems, the academic community has explored a suite of advanced in situ and operando characterization techniques,1–8 including X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), atomic force microscopy (AFM), Auger electron spectroscopy (AES), computed tomography (CT), ultrasonic wave imaging, magnetometry, and magnetic resonance (MR) methods such as nuclear magnetic resonance (NMR) spectroscopy and magnetic resonance imaging (MRI). Among these, state-of-the-art MR techniques stand out for their exceptional sensitivity to electrochemical phenomena and material properties, including structure, morphology, magnetism, ionic transport, and molecular-scale dynamics.8–45Unlike diffraction-based approaches, which primarily detect heavier atomic species, MR techniques excel in probing nuclei throughout the periodic table, provided they have a non-zero spin. This makes MR particularly relevant for battery research, as many key electrochemical elements in electrodes and electrolytes possess NMR-active isotopes, including 7Li, 23Na, 1H, 19F, 31P, 55Mn, and 59Co. The battery industry has prioritized Li- and Na-ion-based electrochemical systems, and these, along with other chemical elements, represent some of the most sensitive NMR probe species. MR experiments span a wide range of contrast mechanisms operating across multiple length and time scales, enabling discrimination of signals based on molecular structure, dynamics, and magnetic properties.
A long-standing obstacle in applying NMR to commercial battery cells has been the attenuation of radio-frequency (RF) signals by metal casings and tightly wound ‘jelly roll’ current-collector foils. Such architectures are some of the most energy-efficient designs, widely adopted in pouch, prismatic, and cylindrical cells. These conductive elements act as effective RF shields, preventing the excitation RF field (B1) from penetrating the interior of the cell, where the nuclei of interest reside. As a result, NMR signal detection becomes effectively impossible. This limitation has largely restricted the state-of-the-art electrochemical NMR to flat parallel-electrode configurations in ‘coin’ or Swagelok-type cells not fully encased in conductive materials.7–29,31–35 Sufficient RF penetration can be achieved via the skimming mechanism by aligning the electrodes with the direction of the B1 field. Although the simplicity of such model cells is key to informative in situ NMR experiments, optimistic outcomes of such tests often do not translate directly to practical battery prototypes.
In commercial cells, non-uniform spatial distributions of current density and SoC arise from factors such as the large area of current collectors, tab positioning, and finite electrode thickness. This introduces ionic concentration gradients and associated kinetic phenomena that occur in electrodes of resting and operating batteries.44,46–53 Solid-state diffusion, phase transitions, SEI formation, and metal plating can modulate battery performance. These processes lead to density gradients, volumetric deformation, particle fracturing, increased impedance or loss of electrical contact, and accelerated capacity fade.54–58
A further methodological challenge arises from the magnetism of battery terminals, extended tabs, steel casings, and paramagnetic electrode59–61 materials. These elements generate strong local magnetic-field gradients (∼100 s G cm−1)39 that affect the quality of MR experiments.62–64
The first notable in situ MR studies of commercial battery prototypes emerged around 2019.38–41,43–45 The challenges described above can now be largely addressed using plug-and-play (PnP) NMR30 and surface-scan MRI41–45 methods, two conceptually independent analytical approaches. The former technique30 provides a quantitative description of the chemical environments of electrochemically active elements (e.g., Li, Na, Co, etc.) in conventional NMR terms. The current collectors of the cell (e.g., aluminum and copper sheets of the cathode and anode, respectively) form a distributed conductive structure whose frequency-dependent impedance at NMR frequencies enables capacitive RF coupling and a high-fill-factor resonant detection volume. The active electromaterials confined between the current collectors serve as an NMR sample. Ion intercalation and transfer between cathode and anode can be directly observed, along with metal plating, electrolyte degradation, and solid–electrolyte interphase (SEI) formation. In addition, NMR lineshapes exhibit anisotropic properties due to bulk magnetic susceptibility (BMS) effects associated with paramagnetic electrode coatings.14,15,30
Surface-scan MRI41 represents an optimal inside-out MRI38 approach, employing rapid, distortion-free imaging protocols.39–41 A recent advancement yielding dramatic gains in sensitivity has been achieved through the development of unilateral RF sensors based on a parallel-plate architecture combined with solid-state detection media.45 By mapping magnetic field (B0) perturbations near the cell surface using purely phase-encoded two-dimensional (2D) k-space sampling, surface-scan MRI enables interrogation of magnetic susceptibility and current-density distributions within the cell. This principle allows the detection of various defect types as well as solid-state processes in electrodes, such as magnetic phase transitions, ion transport, and metal plating.41–45
In this work, we combine the surface-scan MRI sensor45 and PnP NMR adapter30 in a single multimodal device. Such a composite probe enables a wide range of experimental protocols to be efficiently executed across battery states (SoH, SoC) and operating modes, without repetitive or time-consuming hardware manipulations and associated standard procedures (e.g., shimming, temperature equilibration). Importantly, the coherent acquisition of complementary in situ NMR and MRI metrics unlocks their predictive potential.
2 Results and discussion
2.1 Multimodal probe architectures
Fig. 1 presents a schematic of the multimodal MR probe, which integrates surface-scan MRI (blue) and PnP NMR (black) RF sub-circuits. The two sub-circuits, operating at the Larmor frequencies of the proton and an X nucleus, respectively, are positioned on opposite sides of a printed circuit board. Photographs of a multimodal probe prototype compatible with a 7 T vertical magnet with an 89 mm bore illustrate the detailed arrangement of the RF components and the battery cell (Fig. 2). | ||
| Fig. 2 Photographs of the front (a) and back (b) sides of the multimodal RF probe. Visible components include the working surface (P) of the parallel-plate resonator (∼50 µm copper foil with a 1 mm silicone detection medium); variable tuning (CT-mri) and fixed matching (CM-mri) capacitors; fixed ceramic capacitors (CP) located between copper sheets above and below the silicone layer; terminals of the PnP adapter (T1,2) and of the electrochemical cycling circuit (T3,4); variable tuning (CT-nmr and fixed matching (CM-nmr) capacitors of the PnP NMR circuit; shielded cable traps65,66 (Fnmr, Fmri) resonant at the X- and 1H frequencies; and 50 Ω coaxial cables (RFmri, RFnmr). (c) A pouch cell – OnePlus7 (b) placed on the resonator's working surface (P) with its tabs connected to the PnP nmr terminals (T1,2). The cell is secured by plastic bars A1–A3 and screws S1–S3; A1 ensures firm metal-to-metal contact between the battery tabs and the terminals. (d) The multimodal probe with the mounted battery cell installed on the composite probe body compatible with a 7 T vertical-bore magnet (89 mm i.d.). | ||
The MRI sub-circuit consists of a parallel-plate resonator, tuning capacitors (CP, CT-mri), and a matching capacitor (CM-mri). The homogeneity and magnitude of the B1 field are controlled by the distribution of the capacitors (CP) along the top and bottom edges of the parallel plates.45 The resonator volume contains a 1 mm thick silicone layer, which provides the 1H signal used for magnetic field mapping.
The NMR sub-circuit consists of a tuning capacitor (CT-nmr), two matching capacitors (CM-mri), the reactive and resistive elements of the battery cell (CB, LB, RB), and the loop inductance (LE), which is part of the PnP NMR adapter (Fig. 1). The resonance frequency, ν0, of the NMR sub-circuit is:
| ν0 = (2π)−1 (L·C)−0.5 | (1) |
Determined by the total capacitance C:
| C = CB CT-nmr (CB + CT-nmr)−1 | (2) |
And inductance L:
| L = LE + LB | (3) |
The resonance frequency can be controlled by adjusting CT-nmr and LE. Note that
| LB = LBE + LBI | (4) |
| CT-nmr << CB | (5a) |
and
| C ≈ CT-nmr | (5b) |
Conditions (5a) and (5b) ensure that the PnP NMR sub-circuit does not require any adjustments specific to different battery cells. In low-frequency applications (i.e., low-γ or low-B0 experiments), the LE value (proportional to the loop area) can be increased to satisfy these conditions. Finally, to cancel a common mode produced on the outer surface of the cable shield, two double-tuned cable traps65,66 were introduced at all inputs of the probe, see Fig. 2.
Based on the same principle, the PnP adapter can be designed as a multi-frequency probe, either as a standalone tool or as part of the multimodal probe, enabling multi-dimensional NMR experiments and facilitating the analysis of complex NMR lineshapes. The selection of Larmor frequencies depends on the specific chemistries of the cathode, anode, and electrolyte. Materials used in commercial battery cells may contain combinations of the following NMR-active isotopes: 6,7Li, 23Na, 31P, 59Co, 55Mn, 51V, 63,65Cu, 27Al, 19F and 1H. For studies involving low-sensitivity isotopes (e.g., 6Li, 17O, 2H, and 13C), isotopic enrichment of electrode and electrolyte materials can be considered.
Fig. 3 shows a principal electronic schematic of a two-frequency PnP NMR circuit, provided with tuning and matching capacitors (CT1,2 CM1,2) and an LC trap. The concept of double-tuning the NMR probe, achieved by introducing an LC trap in series with the resonance loop, was first proposed by Schnall et al.67 To eliminate cross-talk between (i.e., decouple) the 1H- and X-terminals of the probe, additional band-stop filters (LC(X) and LC(1H), respectively) were introduced into the 1H- and X–RF lines. In this study, we used a multimodal probe (7Li NMR–1H MRI) to analyze the cell under normal conditions (0 < SoC < 100%) and overcharge conditions (100 < SoC < 142%), the latter of which leads to metal plating. A prototype two-frequency PnP 7Li–6Li NMR adapter was designed and experimentally validated.
2.2 Plug-and-play NMR
In our previous work, PnP 7Li NMR spectra of commercial pouch cells, based on the LiCoO2 (LCO) cathode and graphite anode, revealed complex lineshapes reminiscent of a first-order quadrupolar triplet at x ≈ 0.5 (SoC, 100%).30 Indeed, in model cells, different stages of graphite lithiation manifested as superimposed 7Li NMR triplets.16–18 In the commercial cells used in this work (OnePlus7 model), quadrupolar multiplets arising from lithiated graphite (LixC6) overlap with the broad 7Li NMR signals originating from the cathode. The analysis of such data can be simplified using a two-frequency (7Li, 6Li) PnP NMR adapter (Fig. 3). For demonstration purposes, one battery cell was overcharged to plate a detectable amount of Li metal on the anode. An overcharge of 1.6 Ah (ΔSoC ≈ 43%) was applied at a rate of 0.25C. 6Li and 7Li NMR spectra of this cell are shown in Fig. 4. The Li-metal signal is present in both spectra and can serve as an internal NMR reference (at ca. 269 ppm). A triplet-like spectral envelope is apparent in the 7Li NMR data, with the central transition at ca. 40 ppm and an apparent quadrupolar splitting of ca. 20.5 kHz. The 6Li NMR line at ca. 40 ppm appears effectively as a singlet due to its small quadrupolar constant.In situ 7Li NMR spectra of the OnePlus7 cell are shown in Fig. 5a as a 1D stack for a series of SoC values. The initially fully charged cell was discharged in increments of 2.5% SoC. Although an accurate decomposition of static NMR spectra can be challenging, the positions of relatively narrow anode (LixC6) signals can be identified, along with several broad components of the cathode in the low-field region (Fig. 5b).
The lineshape decomposition algorithm developed in this work (Section S1, SI) relies on well-resolved components of the quadrupolar triplets corresponding to stages 1 (LiC6), 2 (LiC12), and 2L (LiC18) of lithiated graphite (Fig. 5).16–18,68,69 A low-field shoulder that emerges upon discharging is attributed to lithiation of the LCO cathode. At SoC > 10%, the cathode contribution to the spectra can be reliably approximated by a superposition of three Lorentzian lines. These Li environments likely reflect variations in the structural and magnetic properties of the LixCoO2 coating21,70 at both intra- and inter-grain levels. Below 20% SoC, the cathode and residual anode resonances begin to merge into a featureless signal. A comparison between experimental and simulated NMR spectra is shown in Fig. 6. Examples of the lineshape decomposition analysis are presented in Fig. 7.
7Li NMR parameters, such as integrated signal intensities (Ai), Knight (δK) and chemical (δ) shifts, linewidths (Δν1/2), and first-order quadrupolar splitting (νQ), are sensitive to structural and magnetic phase transformations, as well as to morphological changes in electrode materials (Fig. 8).
Integrated intensities (Ai) of 7Li NMR lines associated with the cathode (Fig. 8a) and anode (Fig. 8b) are the most relevant metrics for SoC quantification, as they provide intercalant concentrations in both electrodes. NMR measurements reveal that the lithium content in each electrode changes systematically by approximately one order of magnitude across the SoC range from 10% to 100%. In contrast, electrochemical cycling data frequently display “flat” voltage regions, in which voltage-based SoC estimation becomes unreliable.
Knight shifts and linewidths constitute another set of sensitive metrics for SoC and SoH. In the cathode, δK values span an approximately 100 ppm range for each component (Fig. 8c), whereas the corresponding inverse linewidths (Δν1/2−1 ≈ 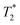 ) range from 100 to 450 µs (Fig. 8d).
) range from 100 to 450 µs (Fig. 8d).
Quadrupolar 7Li NMR lineshapes of LixC6 provide unique insights into anode structure and function. The SoC can be inferred from the relative contributions of each triplet. Our data reveal that three distinct LixC6 environments (1, 2, and 2L) coexist over the SoC range of approximately 50–100%. These observations are likely a consequence of the size distribution of graphite crystallites, leading to heterogeneous electrochemical behavior.
The populations of the LixC6 phases and their individual NMR parameters (δK, Δν1/2−1 of central and quadrupolar transitions, and νQ) vary with SoC in a complex manner, see Fig. 8e–h. At SoC < ca. 45%, a systematic decrease in δK may signal the emergence of 3L (LiC27) mixed with other low-density phases 4L (LiC36) and 1′. This interpretation is further supported by abrupt changes in νQ and Δν1/2 of both the central and quadrupolar transitions. Accurate identification of low-density phases from in situ 7Li NMR data remains challenging due to their low concentrations. A relatively narrow signal from the electrolyte (LiPF6) is detectable at SoC > 10%. Its chemical shift is influenced by the SoC-dependent magnetic susceptibility of the cell, making it a sensitive metric for assessing SoC in the 10–50% range (Fig. 8i).
Following the Li metal plating process with PnP 7Li NMR reveals resonance lines within a characteristic Knight shift range (245–270 ppm; Fig. 9a). The main line at 270 ppm and a high-field shoulder at ca. 248 ppm emerge at SoC > 110% and gradually increase with the level of overcharge. The Knight shift can vary14 depending on the metallic cluster morphology, orientation, and conductivity influenced by the interface chemistry. The integrated area of these signals as a function of SoC is shown in Fig. 9b. It should be noted that 7Li NMR detects only metallic lithium within the near-surface region defined by the RF skin depth. This NMR signal is modulated by local orientations of the metal faces with respect to the B1-field direction.11,31 Starting at a SoC of ∼115%, the amount of NMR-visible Li metal increases approximately linearly with SoC. Because RF penetration is limited in metallic Li, this linear growth likely reflects an increase in NMR-accessible surface area of plated Li; thicker deposits beyond the skin depth contribute little to the observed signal.
 | ||
| Fig. 9 (a) PnP 7Li NMR spectra of a OnePlus7 pouch cell at different stages of overcharge. (b) Integrated NMR intensities associated with metallic lithium deposition as a function of SoC. | ||
2.3 Surface-scan MRI
The surface-scan MRI method has demonstrated a unique capacity to investigate intercalation-dependent magnetism, SoC, solid-state phase transitions, and ion transport in electrodes, while also exhibiting high sensitivity to mechanical defects and currents in operating batteries.40–45 A commercial battery cell (pouch, prismatic, or cylindrical) placed in a uniform polarizing magnetic field (B0) generates characteristic magnetic-field perturbations (ΔB), enabling the prediction of the magnetic susceptibility distribution within electrode materials:38,39,44,45| ΔB ∝ χ(SoC) | (6) |
To facilitate the analysis of SoC-dependent MRI data (BSoC), magnetic field components that do not vary with SoC should be removed by subtraction of a suitable reference map, ΔBr:
| BSoC = ΔB(SoC) − ΔBr | (7) |
| ΔBr = ΔB(SoC = 100%) | (8) |
In general, the choice of the reference map, ΔBr, is context-dependent and should reflect the specific contrast mechanism of interest. For BSoC mapping, the objective is to isolate the magnetic field contribution arising from reversible electrochemical processes while suppressing static or geometry-driven components that are independent of SoC (e.g., dipolar fields from current collectors, tabs, and other metallic elements).
For a given “healthy” battery, ΔBr is therefore defined as the field map acquired at a well-defined reference state, typically SoC = 100%. All subsequent maps are expressed relative to this baseline, i.e., ΔB(SoC)–ΔBr, ensuring that the resulting contrast predominantly reflects changes in the active materials. This normalization minimizes contributions from invariant structural features and improves comparability across measurements.
The robustness of this approach relies on the reproducibility of the magnetic field maps. Our MRI methodology is inherently based on spatial pattern recognition: cells of identical design and electrochemical state are expected to produce field maps that are indistinguishable within an experimental uncertainty ε. In this work, repeated surface-scan MRI measurements on the same cell yield ΔB maps reproducible within ε ≈ 0.2 ppm, which defines the practical sensitivity limit of the method. This reproducibility enables a quantitative criterion for degradation assessment. Specifically, deviations between reference-state maps acquired at different stages of cycling can be evaluated as:
| ε = ΔB(SoC = 100%, cycle 0) − ΔB(SoC = 100%, cycle N) | (9) |
If ε remains within the experimental uncertainty (ε ≤ 0.2 ppm), the cell can be considered magnetically and structurally stable, indicating negligible degradation. Conversely, systematic deviations beyond this threshold reflect irreversible changes in magnetic susceptibility distributions, which may arise from processes such as material degradation, loss of active lithium, or mechanical deformation.
Finally, differences observed between nominally identical cells at the same SoC can be attributed to variations in manufacturing or assembly (e.g., coating inhomogeneity, electrode misalignment, or tab geometry). This sensitivity to spatial heterogeneity provides a direct basis for non-destructive quality control and defect identification in industrial battery cells.
Fig. 10 shows four representative examples of BSoC maps of an LCO-based pouch cell (Table S1, SI) at several SoCs (0, 10.6, 100 and 142.6% overcharge). Histograms of BSoC distributions and corresponding means, <BSoC>, and standard deviations, σ, are shown in Fig. 11a, b and c, respectively. Due to the strong magnetic susceptibility contrast between LCO phases O3-R2 (0.58 < x < 0.75) and O3-R1 (0.94 < x < 1),59,60 the BSoC data are expected to be particularly informative in the two-phase region 0.75 < x < 0.94 (12% < SoC < 50%).44 The statistical analysis of BSoC maps (Fig. 11) is consistent with these expectations and indicates several characteristic SoC regions: (A) 0–5% (O3-R1 dominated); (B) 5–40% (O3-R2 → O3-R1 – transition, mixed phase); (C) 40–82% (O3-R2 dominated); (D) 82–100% (O3-M – dominated); (E) 100–140% (overcharge – metal plating).
 | ||
| Fig. 10 (a–d) Examples of BSoC maps of a OnePlus7 pouch cell at SoC values of 0%, 10.6%, 100%, and 142.6% (overcharge), respectively. | ||
The MRI data suggest that <BSoC> and σ can be good metrics of SoC in the intervals 0–40% and 82–130%, Fig. 11b and c. Outside these regions, the parameters plateau, thus leaving a gap (40–82%) in the SoC analysis. Note, these conclusions are specific to characteristic magnetic phases of LCO. PnP NMR, a complementary approach, allows accurate predictions of SoCs within the range from 10 to 100% (Fig. 5, 6 and 8). Thus, combining our in situ MRI and NMR data enables accurate analysis of intercalation processes and metal plating in the SoC range from 0 to 140%.
Upon overcharging, the LCO cathode becomes excessively delithiated, which destabilizes both its crystal structure and interfacial chemistry. Progressive delithiation increases the Co4+ fraction and associated paramagnetism, leading to enhanced susceptibility variations and NMR line broadening. In terms of MRI experiments, overcharging resulted in a 1 ppm decrease in <BSoC>, indicating an overall increase in paramagnetism of the cathode (Fig. 11b). A 1.5 ppm increase in σ (Fig. 11c) suggests a greater magnetic heterogeneity that can be linked to SoH degradation and correlates with the metal plating observed with PnP NMR.
Note that BSoC maps describe magnetic field patterns that can occur due to spatial variations in (a) concentrations of intercalant ions and oxidation states in the electrode coating; (b) current density resulting from common geometrical constraints in battery designs; (c) properties of electrode coating such as the thickness, impurity content, type and amount of conductive and electrolyte additives, and the quality of electronic contact between active particles and the current collector.
3 Conclusions
This work demonstrates the potential of a highly versatile platform for the assessment of SoC and SoH in state-of-the-art pouch cells. Surface-scan MRI and PnP NMR, integrated within a single multimodal device, enable a broad range of complementary quantitative metrics that cannot be achieved by standalone NMR or MRI techniques. The optimal choice of MR observables inherently depends on cathode and anode chemistries. However, the widespread use of lithium- and sodium-based systems containing highly NMR-sensitive isotopes (7Li and 23Na) makes MR-based analytics broadly applicable.Within the PnP NMR framework, the current collectors act as an efficient RF probe due to the high fill factor of the dense electrode architecture. PnP NMR preserves the core capabilities of conventional NMR spectroscopy while enabling high-throughput measurements. We demonstrated strong sensitivity of PnP NMR to SoC, carbon anode intercalation chemistry, and, in particular, metal plating. In many cases, cathode analysis is also feasible, although severe paramagnetic line broadening in some materials may require extended protocols based on variable carrier frequency. The PnP NMR experiments enable clear discrimination of graphite staging and identification of multiple lithium environments in the lithium cobalt oxide (LCO) cathode. Notably, the data reveal that in LCO cells, the anode is not fully lithiated at a voltage-defined SoC of 100%, with the LiC6 phase accounting for only ∼30–40% of the graphite. This observation highlights the limitations of conventional voltage-based metrics and demonstrates the added value of NMR methodology.
We note that the key findings of this work are rooted in broadly applicable physical processes. Graphite remains the dominant anode material in Li-ion batteries, and the ability to resolve its lithiation stages (LixC6) is therefore intrinsically general. The PnP NMR approach directly tracks the evolution of these phases during operation, independent of the specific cathode chemistry.
The diverse intercalation-dependent magnetic phase behaviors of layered transition metal oxides and polyanion systems are critical for achieving strong surface-scan MRI contrast. Surface-scan MRI complements PnP NMR by circumventing linewidth limitations and providing spatially resolved maps of magnetic field perturbations. This enables non-invasive visualization of SoC heterogeneity and magnetic phase transitions within intact industrial cells. Beyond the susceptibility-based imaging, operando surface-scan MRI can also detect metal plating through analysis of current density distributions. Although more complex to implement, these measurements further extend the scope of the multimodal approach.
From the MRI perspective, LCO represents a demanding and informative test system. Its continuous and multi-phase magnetic susceptibility evolution across lithiation states provides a stringent benchmark for surface-scan MRI. In contrast, simpler biphasic systems such as lithium iron phosphate (LFP) are expected to produce more straightforward MRI responses. Successful application to LCO therefore supports the broader applicability of the method to a wide range of battery chemistries.
Overall, the multimodal MR approach provides a powerful and scalable platform for fundamental studies and practical battery analysis applications, including quality control, safety diagnostics (e.g., metal plating and mechanical defects), and recycling.
4 Methods
4.1 NMR and MRI experiments
Experiments were performed using a Bruker Avance NEO console and a 7 T vertical-bore magnet (I.D. 154 mm) equipped with a triaxial gradient set (79 G cm−1 at 200 A). Multimodal RF probes were constructed from 1.5 mm thick copper-plated PCBs, copper foil, non-magnetic variable capacitors (Johanson, Model 5641; 1.0–30 pF; 250 V, USA), fixed capacitors (Exxelia, 4–27 pF; 500 V), and 50 Ω coaxial cables with BNC connectors. The parallel-plate resonator consisted of a 1 mm thick silicone sheet (65 × 60 mm) sandwiched between two copper foil sheets. Twenty-two capacitors (CP ∼24 pF each) were evenly distributed along two opposite edges of the resonator (Fig. 2).PnP 7Li NMR experiments were performed at a carrier frequency of 116.64 MHz using a single-pulse sequence: 5 µs excitation RF pulse, 6.5 µs pre-acquisition delay, spectral width of 500 kHz, 1024 complex data points, and 1 s magnetization recovery delay. The B1 magnitude estimated from nutation experiments was ∼10 kHz (π/2 pulse duration ≈ 25 µs) at 1000 W transmitter power. Each spectrum was acquired with 500 signal averages, yielding a total measurement time of ∼9 min. Chemical shifts were referenced to an external 1 M LiCl solution (0 ppm). The battery management system (BMS) circuitry connecting the cell tabs was removed to improve RF transmission through the current collectors. This modification does not alter the intrinsic electrochemical behavior of the cell. Specifically, the capacity and voltage window remain unchanged, as they are determined by the electrode materials and electrolyte chemistry. The rate capability may be marginally influenced by differences in external wiring; however, this does not impact the conclusions drawn in this work. We further note that pouch cells without integrated BMS are widely used in both research and industrial contexts (e.g., bare cells supplied for module integration and laboratory testing). The PnP NMR methodology is therefore directly applicable to a broad class of commercially relevant systems.
Surface-scan MRI experiments employed a 2-dimensional centric-scan SPRITE protocol41,43–45 implemented in the ParaVision 360 environment. Nonselective, low-flip-angle RF pulses (carrier frequency: 300.356 MHz) were followed by a pre-acquisition phase encoding period TP0 (58 µs) and acquisition of FID data under the SPRITE gradient. Each RF pulse had a duration of 1 µs (flip angle ∼π/18) at 100 W transmitter power. Gradient switching and stabilization intervals were each 0.5 ms. Eight complex points (32 µs) were acquired at time TPk after each excitation pulse:
| TPk = TP0 + (k − 1) SW−1, k = 1 − 8 | (10) |
| ΔB(r) = γ−1 Δφ(r, TP) ΔTP−1 | (11) |
4.2 Electrochemical cycling
Electrochemical experiments were conducted using a BTS-4008-5V6A-S1 instrument (NEWARE, Hong Kong, China) at 20 °C. Battery cells (Section S2, SI) were cycled galvanostatically at approximately C/8, within the factory-specified voltage cutoff limits of 2.75–4.4 V for the OnePlus7 cells. Overcharging procedures were conducted outside the NMR magnet in a controlled laboratory environment with appropriate safety measures in place, including operation within an exhaust fume hood and the availability of a fire extinguisher.Author contributions
K. R. conceived the study, developed the experimental protocols, performed the NMR and MRI experiments, and conducted data analysis. The multimodal probes were developed through close collaboration between N. A. and K. R. Both authors contributed to the preparation and revision of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study are available from the corresponding author upon reasonable request.Supplementary information (SI): details of the NMR lineshape analysis and specifications of the commercial pouch cells used in this study. See DOI: https://doi.org/10.1039/d6ta02247b.
Acknowledgements
K. R. acknowledges financial support from the Agence Nationale de la Recherche (ANR), Project No. ANR-25-CE50-0032-01. N. A. thanks the Deutsche Forschungsgemeinschaft (DFG) for support under Project No. 568670018.References
- A. M. Tripathi, W.-N. Su and B. J. Hwang, Chem. Soc. Rev., 2018, 47, 736–851 RSC.
- D. Liu, Z. Shadike, R. Lin, K. Qian, H. Li, K. Li, S. Wang, Q. Yu, M. Liu, S. Ganapathy, X. Qin, Q. Yang, M. Wagemaker, F. Kang, X. Yang and B. Li, Adv. Mater., 2019, 31, 1800933 Search PubMed.
- Y. Tian, C. Lin, H. Li, J. Du and R. Xiong, Appl. Energy, 2021, 300, 117386 CrossRef CAS.
- S. Lee, S. Park, W. Lee, J. Seok, J. Kim, J. Kim and W. Yoon, Carbon Energy, 2024, 6, 12 CrossRef.
- Z. Deng, et al., Joule, 2020, 4, 2017–2029 CrossRef CAS.
- Y. Hu, et al., Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 10667–10672 CrossRef CAS PubMed.
- C. P. Grey and J. M. Tarascon, Nat. Mater., 2016, 16, 45–56 CrossRef CAS PubMed.
- O. Pecher, J. Carretero-González, K. J. Griffith and C. P. Grey, Chem. Mater., 2017, 29, 213–242 CrossRef CAS.
- X. Liu, Z. Liang, Y. Xiang, M. Lin, Q. Li, Z. Liu, G. Zhong, R. Fu and Y. Yang, Adv. Mater., 2021, 33, 2105207 Search PubMed.
- A. L. Freytag, A. D. Pauric, S. A. Krachkovskiy and G. R. Goward, J. Am. Chem. Soc., 2019, 141, 13758–13761 CrossRef CAS PubMed.
- E. G. Sorte, N. A. Banek, M. J. Wagner, T. M. Alam and Y. J. Tong, Chemelectrochem, 2018, 5, 2336–2340 CrossRef CAS.
- S. Benders, M. Mohammadi, C. A. Klug and A. Jerschow, Sci. Rep., 2020, 10, 1–11 Search PubMed.
- B. J. Walder, M. S. Conradi, J. J. Borchardt, L. C. Merrill, E. J. Deichmann, T. M. Anderson, T. M. Alam and K. L. Harrison, Sci. Adv., 2021, 7, eabf048 Search PubMed.
- N. M. Trease, L. Zhou, H. J. Chang, B. Y. Zhu and C. P. Grey, Solid State Nucl. Magn. Reson., 2012, 42, 62–70 CrossRef CAS PubMed.
- L. Zhou, M. Leskes, A. J. Ilott, N. M. Trease and C. P. Grey, J. Magn. Reson., 2013, 234, 44–57 CrossRef CAS PubMed.
- M. Letellier, F. Chevallier and M. Morcrette, Carbon, 2007, 45, 1025–1034 CrossRef CAS.
- F. Chevallier, F. Poli, B. Montigny and M. Letellier, Carbon, 2013, 61, 140–153 CrossRef CAS.
- S. A. Krachkovskiy, et al., J. Electrochem. Soc., 2020, 167, 130514 CrossRef CAS.
- B. Key, et al., J. Am. Chem. Soc., 2009, 131, 9239–9249 CrossRef CAS PubMed.
- F. Poli, J. S. Kshetrimayum, L. Monconduit and M. Letellier, Electrochem. Commun., 2011, 13, 1293–1295 CrossRef CAS.
- K. Shimoda, Electrochim. Acta, 2013, 108, 343–349 CrossRef CAS.
- M. Menetrier, I. Saadoune, S. Levasseur and C. Delmas, J. Mater. Chem., 1999, 9, 1135–1140 RSC.
- A. B. Gunnarsdóttir, C. V. Amanchukwu, S. Menkin and C. P. Grey, J. Am. Chem. Soc., 2020, 142, 20814–20827 CrossRef PubMed.
- K. Märker and C. P. Grey, J. Am. Chem. Soc., 2020, 142, 17447–17456 CrossRef PubMed.
- M. Liu, S. Ganapathy and M. Wagemaker, Acc. Chem. Res., 2022, 55, 333–344 CrossRef CAS PubMed.
- D. Columbus, et al., J. Am. Chem. Soc., 2022, 144, 9836–9844 CrossRef CAS PubMed.
- J. M. Stratford, P. K. Allan, O. Pecher, P. A. Chater and C. P. Grey, Chem. Commun., 2016, 52, 12430–12433 RSC.
- J. M. Stratford, et al., J. Am. Chem. Soc., 2021, 143, 14274–14286 CrossRef CAS PubMed.
- K. Gotoh, Batter. Supercaps, 2021, 4, 1267–1278 CrossRef CAS.
- K. Romanenko, N. Avdievich and E. Foy, J. Am. Chem. Soc., 2024, 146, 29407–29416 CrossRef CAS PubMed.
- S. Chandrashekar, et al., Nat. Mater., 2012, 11, 311–315 CrossRef CAS PubMed.
- A. J. Ilott, M. Mohammadi, H. J. Chang, C. P. Grey and A. Jerschow, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, 10779–10784 CrossRef CAS PubMed.
- P. M. Bayley, N. M. Trease and C. P. Grey, J. Am. Chem. Soc., 2016, 138, 1955–1961 CrossRef CAS PubMed.
- Y. Xiang, et al., Nat. Nanotechnol., 2020, 15, 883–890 CrossRef CAS PubMed.
- J. D. Bazak, J. P. Allen, S. A. Krachkovskiy and G. R. Goward, J. Electrochem. Soc., 2020, 167, 140518 CrossRef CAS.
- K. Romanenko, M. Forsyth and L. A. O'Dell, J. Magn. Reson., 2014, 248, 96–104 CrossRef CAS PubMed.
- K. Romanenko, L. Jin, P. Howlett and M. Forsyth, Chem. Mater., 2016, 28, 2844–2851 CrossRef CAS.
- A. J. Ilott, M. Mohammadi, C. M. Schauerman, M. J. Ganter and A. Jerschow, Nat. Commun., 2018, 9, 1–9 CAS.
- K. Romanenko and A. Jerschow, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 18783–18789 CrossRef CAS PubMed.
- K. Romanenko, P. W. Kuchel and A. Jerschow, Chem. Mater., 2020, 32, 2107–2113 CrossRef CAS.
- K. Romanenko and A. Jerschow, J. Mater. Chem. A, 2021, 9, 21078–21084 RSC.
- K. Romanenko and A. Jerschow, J. Magn. Reson. Open, 2022, 10–11, 100061 CrossRef CAS.
- K. Romanenko and N. Avdievich, J. Phys. Chem. C, 2022, 127, 85–91 CrossRef.
- K. Romanenko, et al., Chem. Mater., 2023, 35, 9789–9798 CrossRef CAS.
- K. Romanenko and N. Avdievich, J. Magn. Reson. Open, 2024, 18, 100141 CrossRef CAS.
- A. Fernando, M. Kuipers, G. Angenendt, K.-P. Kairies and M. Dubarry, Measurement: Energy, 2024, 3, 100013 Search PubMed.
- K. P. C. Yao, J. S. Okasinski, K. Kalaga, I. A. Shkrob and D. P. Abraham, Energy Environ. Sci., 2019, 12, 656–665 RSC.
- Z. Chen, D. L. Danilov, R.-A. Eichel and P. H. L. Notten, Energy Storage Mater., 2022, 48, 475–486 CrossRef.
- J. I. G. Dawkins, M. Z. Ghavidel, D. Chhin, I. Beaulieu, M. S. Hossain, R. Feng, J. Mauzeroll and S. B. Schougaard, Anal. Chem., 2020, 92, 10908–10912 CrossRef CAS PubMed.
- B. Pelletier-Villeneuve, B. Krueger, J. R. Adsetts, M.-C. Ricard and B. Schougaard, ECS Electrochem., 2025, 1, 1972–1976 CAS.
- G. A. Giffin, Nat. Commun., 2022, 13, 1–9 Search PubMed.
- D. Takamatsu, A. Yoneyama, Y. Asari and T. Hirano, J. Am. Chem. Soc., 2018, 140, 1608–1611 CrossRef CAS PubMed.
- B.-G. Chae, S. Y. Park, J. H. Song, E. Lee and W. S. Jeon, Nat. Commun., 2021, 12, 1–9 Search PubMed.
- S. Zhang, npj Comput. Mater., 2017, 3, 1–10 Search PubMed.
- F. Pistorio, D. Clerici, F. Mocera and A. Somà, Energies, 2022, 15, 9168 CrossRef CAS.
- J. S. Edge, S. O'Kane, R. Prosser, N. D. Kirkaldy, A. N. Patel, A. Hales, A. Ghosh, W. Ai, J. Chen and J. Yang, Phys. Chem. Chem. Phys., 2021, 23, 8200–8221 RSC.
- A. Lundkvist, P.-L. Larsson and E. Olsson, Powder Technol., 2025, 466, 121417 CrossRef CAS.
- J. S. Menye and M.-B. Camara, Energies, 2025, 18, 342 CrossRef CAS.
- J. T. Hertz, et al., Phys. Rev. B, 2008, 77, 075119 CrossRef.
- T. Motohashi, et al., Phys. Rev. B, 2009, 80, 165114 CrossRef.
- Y. Mekonnen, A. Sundararajan and A. I. Sarwat, SoutheastCon 2016, 2016, 1–6 Search PubMed.
- J. R. Reichenbach, R. Venkatesan, D. A. Yablonskiy, M. R. Thompson, S. Lai and E. M. Haacke, J. Magn. Reson. Imag., 1997, 7, 266–279 CrossRef CAS PubMed.
- M. J. Graves and D. G. Mitchell, J. Magn. Reson. Imag., 2013, 38, 269–287 CrossRef PubMed.
- M. A. Bernstein, K. F. King and X. J. Zhou, Handbook of MRI Pulse Sequences, Elsevier, Oxford, 2004 Search PubMed.
- J. J. Seeber and A. Menon, Concepts Magn. Reson. Part B, 2004, 21B, 26–31 CrossRef.
- N. I. Avdievich and H. P. Hetherington, J. Magn. Reson., 2007, 186, 341–346 CrossRef CAS PubMed.
- M. D. Schnall, V. Harihara Subramanian, J. S. Leigh and B. Chance, J. Magn. Reson., 1985, 65, 122–129 CAS.
- J. R. Dahn, Phys. Rev. B, 1991, 44, 9170–9177 CrossRef CAS PubMed.
- T. Ohzuku, Y. Iwakoshi and K. Sawai, J. Electrochem. Soc., 1993, 140, 2490–2498 CrossRef CAS.
- M. C. Tucker, J. A. Reimer, E. J. Cairns, S. Choi and A. Manthiram, J. Phys. Chem. B, 2002, 106, 3842–3847 CrossRef CAS.
- M. Halse, J. Rioux, S. Romanzetti, J. Kaffanke, B. MacMillan, I. Mastikhin, N. J. Shah, E. Aubanel and B. J. Balcom, J. Magn. Reson., 2004, 169, 102–117 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |








