 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Polyamines with reactive CO2 diffusion for carbon capture: the obvious and the unexpected
Shweta Singh†
 ,
Taliehsadat Alebrahim†,
Narjes Esmaeili
,
Taliehsadat Alebrahim†,
Narjes Esmaeili ,
Yang Jiao* and
Haiqing Lin
,
Yang Jiao* and
Haiqing Lin *
*
Department of Chemical and Biological Engineering, University at Buffalo, The State University of New York, Buffalo, NY 14260, USA. E-mail: yjiao3@buffalo.edu; haiqingL@buffalo.edu
First published on 27th April 2026
Abstract
Polyamines have emerged as a leading materials platform for sorbents in direct air capture (DAC) of CO2 and membranes for post-combustion capture (PCC) due to their reactions with CO2. Such reactions compete with hydrogen bonding among amine groups, are sensitive to temperature and water content, and can self-limit the further diffusion and sorption of CO2 in polyamines. These unique characteristics give rise to interesting, often conflicting phenomena in DAC sorbents and PCC membranes, which have not been resolved at the molecular level. Herein, we highlight the imbalanced effects of substrates on amine efficiency for CO2 sorption, the complex interplay of temperature and time on pseudo-equilibrium CO2 sorption, and the intricate effects of water vapor on CO2 sorption and desorption in DAC. For PCC membranes, both facilitated and hindered CO2 transport are critically reviewed with an integrated experimental and simulation approach. We elucidate the role of CO2-reactive diffusion in both sorbent and membrane applications, providing cohesive guidance for designing polyamine-based systems to enhance CO2 capture performance.
1 Introduction
The use of abundant carbon-based energy sources (such as coal, natural gas, oil, and traditional biomass) has dramatically improved living conditions for our society, but it inevitably increases atmospheric CO2 concentrations, influencing the global climate. These carbon-based energy sources are inexpensive, account for 80–90% of global energy consumption, and are expected to continue playing a significant role in the energy landscape due to their well-established infrastructure and reliability. Therefore, mitigating CO2 emissions into the atmosphere has emerged as a grand challenge for our society.Various approaches to carbon capture have been explored, and two have attracted significant attention for their potential impacts.1 First, CO2 can be captured from the air (i.e., direct air capture, or DAC), thereby directly reducing atmospheric CO2 concentration.2–4 Such technologies can be used at any location and may be carbon-negative. To this end, solid sorbents are widely used for DAC, as they offer low energy input, low operating costs, and good scalability. Second, CO2 can be captured from flue gas at various point sources after combustion of carbon sources (i.e., post-combustion carbon capture, or PCC).5–7 For instance, flue gas streams from coal-fired power plants contain 10–13% CO2, 82% N2, and other components such as O2 and H2O. As an add-on approach, it does not disrupt existing industrial or power plants and may significantly reduce CO2 emissions, as these point sources account for ∼40% of CO2 emissions. To this end, membrane technology has emerged as one of the leading technologies due to its high energy efficiency, small footprint, and avoidance of chemical use and emission.5,8–10
Interestingly, polyamines have emerged as a leading materials platform for both DAC sorbents11–14 and PCC membranes.7,8,15,16 Amine groups are key to achieving high CO2 sorption capacity and selectivity over the major components of air (N2 and O2).17,18 Eqn (1) and (2) elucidate the reaction mechanisms for the reaction between primary and secondary amines and CO2. Under dry conditions, these amines react with CO2 to form ammonium carbamate.19,20 The maximum molar ratio of CO2/N can reach 0.5 and increase to 1.0 in the presence of moisture (forming bicarbonates), as shown in eqn (3). Eqn (4) also shows that tertiary amines can react with CO2 at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio in the presence of water vapor:
1 molar ratio in the presence of water vapor:
| CO2 + 2RNH2 ↔ RNH3+ + RNHCOO− | (1) |
| CO2 + 2R1R2NH ↔ R1R2NH2+ + R1R2NCOO− | (2) |
| CO2 + R1R2NH + H2O ↔ R1R2NH2+HCO3− | (3) |
| CO2 + R1R2R3N + H2O ↔ R1R2R3NH+HCO3− | (4) |
Separated review articles have been published on polyamines for DAC sorbents12,21,22 and PCC membranes.23 The effect of amine type and its structure (e.g., tetraethylenepentamine (TEPA), polyethylenimine (PEI), and diethylenetriamine (DETA)) on CO2 sorption performance has also been reviewed in the literature.21,22 In most cases, the affinity of amines for CO2 was exploited to enhance CO2 sorption and permeation, resulting in superior CO2 separation performance. However, unusual phenomena have been reported in the literature: CO2 sorption in polyamines increased with increasing temperature;13,24,25 their affinity hindered CO2 diffusion and reduced CO2 permeability, resulting in unexpectedly high N2/CO2 and H2/CO2 selectivity.26,27 These apparent contradictions in these two processes have not been resolved at the mechanistic level.
This paper highlights the unusual behaviors of polyamines for DAC sorbent and PCC membrane applications, underlying reactive CO2 diffusion and its impact on CO2 capture performance, and provides a cohesive framework for CO2 transport in both sorbents and membranes (Fig. 1). First, we critically review polyamines on a broad spectrum of substrates for DAC and underline how substrates affect reactive CO2 diffusion and sorption in these sorbents. The effect of water vapor, temperature, and time on CO2 capture capability is revealed. Second, polyamine-based membranes for PCC are discussed, and the facilitated and hindered transport mechanisms are elucidated and contrasted through modeling and experiments. Finally, the contradictory results are analyzed to emphasize CO2 reactive diffusion in polyamines, shedding light on the design of next-generation sorbents and membranes with enhanced carbon capture performance.
 | ||
| Fig. 1 Overview of polyamine-based sorbents for direct air capture (DAC) and membranes for post-combustion carbon capture (PCC: CO2/N2 separation). | ||
2 Polyamine-based sorbents for direct air capture
The key to DAC is high-performance sorbents with high CO2 sorption capacity, long-term durability, and low costs.12,28,29 Polyamines, such as polyethylenimine (PEI), have been widely used due to their strong interactions with CO2, and they are often incorporated into porous supports to enhance the accessibility of amines to dilute CO2 in air and to reduce pressure drops through the sorbents.22,30–32The morphology of the support materials used to incorporate or append polyamines for DAC applications plays a significant role in determining carbon capture performance.11,13 Support materials are selected based on key properties, including high surface area, appropriate pore size and pore structure, and good thermal and mechanical stability, as well as their interactions with polyamines. All these factors determine the distribution of polyamines (such as their layer thickness on the inner wall) and the structural characteristics of the resulting sorbents (such as pore size and porosity), thereby impacting CO2 diffusion to amine sites and CO2 sorption kinetics, including effects of diffusion resistance.19,33 Notably, various approaches have been adopted to reduce diffusion limitations in these sorbents and enhance CO2 accessibility to active amine sites, such as incorporating surfactants34 and low viscosity diluents like polyethylene glycol (PEG)35 and ionic liquids.36 Additionally, supports with bimodal porous structures have been shown to facilitate rapid CO2 diffusion while maintaining high amine loading.37
A variety of porous materials suitable for amine impregnation have been explored for DAC applications, including silica, zeolites, polymeric porous substrates, and newly emerging microporous materials such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs). Table 1 lists representative polyamine sorbents, based on various substrates and amine structures (primary, secondary, and tertiary amines), along with their DAC properties. In general, it is difficult to directly compare the capture performance of the sorbents due to the complex interplay between the supports' pore morphologies and interactions with polyamines, and there is no quantitative analysis or prediction of the effect of the porous support on DAC properties, in addition to CO2 binding strength and diffusion resistance. Therefore, instead of conducting a comprehensive review, we highlight representative sorbents with high CO2 sorption capacities, particularly their unusual behaviors, including the effects of temperature and water vapor on DAC performance. Detailed mechanisms are also provided in Section 4. Discussion and conclusion.
| Substrates | Polyamines | Surface area (m2 g−1) | Temp. (°C) | RH (%) | CO2 sorption (mmol g−1) | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| Types | Names | Typesa | Content (%) | |||||
| a Ph-3-ED: phenyl-3-ethylene diamine; Ph-3-PD: phenyl-3-propylenediamine; PPI: branched poly(propylenimine); AEAPTMS: N-(3-(trimethoxysilyl)propyl)ethane-1,2-diamine; APTES: 3-aminopropyltriethoxysilane; NFC: nanofibrillated cellulose; TREN: tris(2-aminoethyl)amine; TEPA: tetraethylenepentamine; TAPA: tris(3-aminopropyl)amine; TAEA: tris(2-aminoethyl)amine; SH-bPEI: sulfhydryl-grafted PEI; AEAPDMS: N-(2-aminoethyl)-3-aminopropylmethyldimethoxysilane; PIM-1: polymer of intrinsic microporosity; PF-15: fibers of amidoxime functionalized PIM-1. | ||||||||
| Silica | Silica | PEI | 47 | — | 25 | 0 | 2.4 | 38 |
| HS | TEPA | 70 | — | 30 | 0 | 5.2 | 39 | |
| H–SiO2 | PEI | ∼70 | — | 30 | 19 | 3.4 | 40 | |
| γ-Alumina | PEI | 48.1 | — | 25 | 0 | 1.7 | 41 | |
| γ-Al2O3 | TEPA | 20 | — | −20 | 0 | 0.81 | 42 | |
| PEI | 40 | — | 25 | 70 | 2 | |||
| SBA-15 | TEPA | — | 500–800 | 25 | 0 | 1.5–2.5 | 43 | |
| LPPI | 50 | 80 | 25 | 0 | 1.2 | 44 | ||
| PPI | 40 | — | 35 | 0 | 1.4 | 45 | ||
| PEI | 39.9 | 25 | 0 | 1.1 | 41 | |||
| Ph-3-ED | 60 | — | 35 | 0 | 1.9 | 46 | ||
| 35 | 30 | 2.9 | ||||||
| Ph-3-PD | 50 | 55 | 35 | 0 | 0.56 | |||
| AEAPTMS | — | 45 | 25 | 0 | 1.7 | 47 | ||
| V-B-SBA-15 | PEI | 20 | 600–900 | 25 | 40 | 2.5–3 | 48 | |
| Ti-SBA-15 | PEI | 31.8 | 209 | 25 | 0 | 0.64 | 49 | |
| Zr-SBA-15 | PEI | 34.7 | 230 | 25 | 0 | 0.85 | 49 | |
| Mg/Al-MMO | PEI | 67 | — | 25 | 0 | 2.3 | 50 | |
| TEPA | 67 | 25 | 0 | 3.0 | 51 | |||
| Titania-silsesquioxane aerogel | APTES | — | — | 30 | 0 | 1.6 | 52 | |
| Porous polymers | Solupor | PEI | 48 | — | 25 | 0 | 0.53 | 13 |
| PEI | 48 | — | 25 | 30 | 1.3 | |||
| PIM-1 (powder) | PEI | 21 | 220 | 35 | — | 0.23 | 53 | |
| PIM-1 (fibers) | PEI | 25 | 30 | 35 | — | 0.25 | ||
| PF-15 | TAEA | 21 | 8 | 25 | 50 | 1.0 | 54 | |
| NFC | AEAPDMS | 50 | 7.1 | 25 | 40 | 1.4 | 55 and 56 | |
| Ion exchange resin | Quaternary ammonium | — | 800 | 23 | 0.5 | 0.98 | 57 | |
| MOFs | Mg2(dobpdc) | MMEN | 12 | 70 | 25 | 0 | 2.1 | 58 |
| EN | 1253 | 25 | 0 | 2.8 | 59 | |||
| MIL-101(Cr) | TREN | 45 | — | 25 | 0 | 2.8 | 60 | |
| PEI | 45 | — | 25 | 0 | 1.1 | |||
| MIL101(Cr)/CA | TEPA | 10 | 1247 | −20 | 70 | 0.46 | 61 | |
| PEI | 10 | 1200 | −20 | 70 | 0.66 | |||
| MOF-808 | PA | — | — | 25 | 50 | 0.7 | 62 | |
| COFs | COF-999 | PEI | — | — | 25 | 75 | 2.1 | 63 |
| COF-609 | TAPA | — | — | 25 | 50 | 0.39 | 64 | |
| COF-709 | SH-bPEI | — | — | 25 | 75 | 1.2 | 65 | |
| PE-MCM-41 | Triamine | — | 367 | 25 | 0 | 0.98 | 66 | |
| Hybrid materials | SIPs | PEI | 49 | — | 25 | 0 | 3.7 | 67 |
| NPEI | 49 | — | 25 | 0 | 2.8 | 67 | ||
| PAN/OPZN | NIPEI | — | — | 25 | 0 | 1.7 | 68 | |
| Li-SX zeolite | — | — | >600 | 25 | 0 | 1.3 | 69 | |
2.1 Sorbents based on inorganic porous substrates
Porous silica supports, such as SBA-15, have been widely used to incorporate PEI for DAC applications. SBA-15 was further modified with hydrophobic vinyltrimethoxysilane (VTMS), as shown in Fig. 2a.48 The modified support was then acid-etched to remove boron and hydrolyze the remaining methoxy groups, followed by impregnation with 20 wt% PEI. The generated hydroxyl groups had a good affinity towards PEI. Additionally, a hydrophobic agent, HMDS (hexamethyldisilazane), was also used to modify SBA-15, enhancing the surface morphology.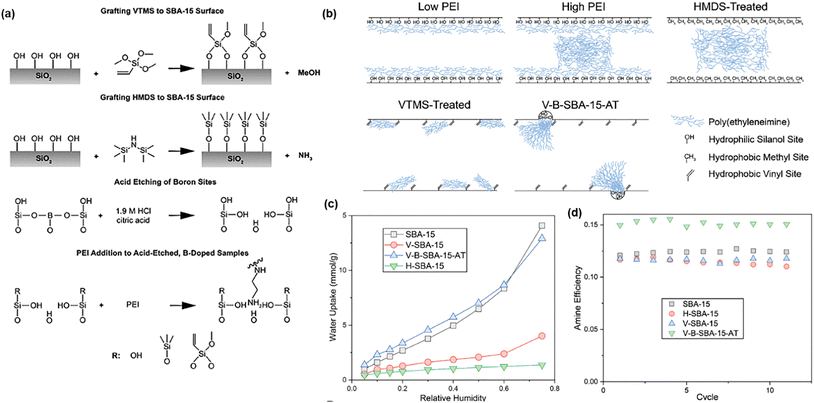 | ||
| Fig. 2 PEI incorporated into SBA-15 modified using VTMS and HMDS. (a) Reaction scheme showing surface functionalization, acid etching of boron sites, and subsequent PEI incorporation. (b) Schematic illustration of porous supports with different PEI loadings and surface treatments. (c) Water sorption isotherms at 30 °C. (d) Amine efficiency at 40% RH and 400 ppm CO2. Copyright 2024 Wiley.48 | ||
Fig. 2b illustrates how surface hydrophobicity affects PEI distribution on the support and amine efficiency of the sorbents. On untreated substrates, PEI showed strong affinity for the surface due to its strong hydrogen bonding with surface silanol groups; HMDS treatment reduced amine–silanol interactions, and the high PEI loading resulted in plug-like domains. By contrast, for VTMS-treated support (V-SBA-15), the vinyl groups exerted a caging effect, limiting PEI mobility and its interactions with silanol groups, thereby improving amine accessibility.
Fig. 2c shows water isotherms for the substrates alone to compare their hydrophobicity and better understand the distribution and interactions of PEI after impregnation. H-SBA-15 and V-SBA-15 exhibited low water uptake due to their hydrophobic surface, whereas the pristine SBA-15 and V-B-SBA-15 showed higher water uptake. Particularly, V-B-SBA-15 had silanol nests that promoted water condensation. When incorporating 20 wt% PEI, SBA-15, H-SBA-15, and V-SBA-15 showed similar amine efficiency with 400 ppm CO2 and 40% RH (Fig. 2d). By contrast, the V-B-SBA-15 showed 25% higher amine efficiency and good stability over 11 cycles. Additionally, H-SBA-15 showed degraded performance after 10 cycles due to its high hydrophobicity and the loss of PEI during thermal desorption.
2.2 Sorbents based on polymeric porous supports
Polymeric substrates have been used to prepare DAC sorbents due to their excellent processability, good handleability, and low costs, such as PIM-1 with microporosity and high surface area (>700 m2 g−1).54,70 Fig. 3a displays cellulose acetate hollow fibers containing MIL-101(Cr) (MIL-101(Cr)/CA), which were first spun via “dry-jet wet quench” process and then functionalized with polyamines such as tetraethylenepentamine (TEPA) and PEI via the wet impregnation method.61 The fibers were porous, resulting in low pressure drops. Fig. 3b displays that the obtained sorbent exhibited a working capacity of 0.9 mmol g−1 during a temperature swing operation between −20 and 60 °C. The samples did not degrade throughout the 10 sorption/desorption cycles. | ||
| Fig. 3 Sorbents based on polymeric porous supports. (a) SEM images of dense and hollow MIL-101(Cr)/CA fibers; (b) PEI10 MIL-101(Cr)/CA fibers for TPD cycles under dry 400 ppm CO2.61 Copyright 2023 Elsevier. (c) PEI-incorporated Solupor (SPEI). (d) Effect of testing time and temperature on CO2 sorption in the SPEI containing 48 mass% PEI (25k).13 (e) Schematic of CO2-reactive diffusion in PEI films. Copyright 2024 American Chemical Society. | ||
A commercial support, Solupor, made of high-density polyethylene (HDPE), was impregnated with branched PEI by wet impregnation (Fig. 3c).13,71 Fig. 3d displays the effect of temperature and testing time on the sorption performance of a sorbent containing 48% PEI. Interestingly, increasing the temperature from 25 to 50 °C increased CO2 pseudo-equilibrium sorption capacity by 36% from 0.53 to 0.72 mmol g−1 due to greater CO2 diffusivity, which can be attributed to non-equilibrium CO2 sorption caused by its self-restricted diffusion and pore blocking by PEI. As CO2 reacts with PEI, it cross-links the PEI surface and lowers CO2 diffusion (Fig. 3e). Increasing the temperature from 25 to 50 °C reduced the CO2 content on the PEI surface and accelerated CO2 diffusion into the films, enhancing CO2 sorption. On the other hand, further increasing the temperature to 65 °C reduced CO2 sorption capacity due to the dominant thermodynamic effect. The effect of diffusion-limited sorption has also been reported elsewhere.24,25,72 For instance, increasing the temperature from −20 to 25 °C increased the sorption capacity by 70% from 1.26 to 2.14 mmol g−1 in 50 mass% TEPA-modified MIL-101(Cr).73
2.3 Sorbents based on MOFs
MOFs have emerged as a highly attractive platform for CO2 capture due to their tunable pore structures, high surface areas, and thus high CO2 sorption capacity.74,75 Many MOFs containing open metal sites, such as MOF-74,76 exhibit remarkable CO2 uptake once guest molecules are removed from the framework. However, these materials often exhibit structural instability in the presence of water vapor, thereby reducing CO2 sorption. Amine functionalization has been employed not only to enhance structural robustness but also to substantially improve CO2 adsorption capacity through a moisture-assisted mechanism.Fig. 4 compares the DAC performance of sorbents based on MIL-101(Cr) and a conventional γ-Al2O3 impregnated with TEPA.11 Interestingly, MIL-101(Cr)-TEPA(30) showed weak chemisorption dominance (i.e., CO2 desorption occurred below 25 °C), while Al2O3-TEPA(20) showed strong chemisorption dominance (i.e., CO2 desorption occurred above 25 °C). Fig. 4a displays that MIL-101(Cr)_TEPA(30) mostly formed carbamic acid with weak binding energy due to the strong amine-support interaction, along with a small amount of ammonium carbamate ion pairs, while Al2O3_TEPA(20) only formed ammonium carbamate with strong binding energy (Fig. 4b), which can be ascribed to the weak amine-support interactions and amine clustering. Nevertheless, there are no quantitative models in the literature for how support properties affect CO2 binding strength under DAC conditions.11,42,77
 | ||
| Fig. 4 Comparison of TEPA-containing sorbents based on γ-Al2O3 and MIL-101(Cr). Reaction mechanism of CO2 with (a) γ-Al2O3_TEPA (with weak amine-support interaction) and (b) MIL-101(Cr)_TEPA (with strong amine-support interaction). Pseudo-equilibrium adsorption and amine efficiency of (c) MIL-101(Cr)_TEPA(30) and (d) γ-Al2O3_TEPA(20) at various temperatures under dry and humid (70% RH) conditions. Copyright 2023 American Chemical Society.11 | ||
Fig. 4c and d compares the effect of temperature and humidity on the sorption performance of MIL-101(Cr)_TEPA(30) and Al2O3-TEPA(20). Decreasing temperature increased the sorption capacity of MIL-101(Cr)_TEPA but decreased the capacity of Al2O3-TEPA, suggesting that the sorption in MIL-101(Cr)-TEPA was dominated by thermodynamic equilibrium, and the sorption in Al2O3-TEPA was limited by CO2 diffusion into the bulk TEPA.
Fig. 5 demonstrates superior DAC performance by pip2-Mg2(dobpdc) (pip2 = 1-(2-aminoehtly)piperidine).78 Mg2(dobpdc) containing diamines exhibited a maximum CO2 uptake at a molar ratio of 1 for CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) diamine; by contrast, pip2-Mg(dobpdc) exhibited a CO2
diamine; by contrast, pip2-Mg(dobpdc) exhibited a CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) diamine ratio of 1.5 due to the cooperative chemisorption and physisorption steps.
diamine ratio of 1.5 due to the cooperative chemisorption and physisorption steps.
 | ||
| Fig. 5 Superior DAC performance by pip2-Mg2(dobpdc). (a) (Left) CO2 adsorption mechanism, (mid) simulated structure, and (right) H4dobpdc and pip2 structures; (b) CO2 sorption isotherms at 25, 40, and 50 °C. (c) Temperature-swing adsorption–desorption cycle at atmospheric pressure. Copyright (2024) American Chemical Society.78 | ||
Fig. 5b shows pure CO2 adsorption isotherms with a two-step adsorption profile at 25, 40, and 50 °C, implying cooperative chemisorption and physisorption. At 25 and 40 °C, the first step of adsorption occurred at 50 and 150 mbar, yielding uptakes of 1.3 and 1.5 mmol g−1, respectively. The second step showed a doubling of sorption capacity compared to the first step, yielding total capacities of 4.9 and 4.6 mmol g−1, respectively. A similar increase in CO2 uptake was observed at 50 °C; however, the uptake value was lower than that at lower temperatures. Fig. 5c shows the stability of pip2-Mg2(dobpdc) over 500 cycles of adsorption–desorption, where the adsorption was conducted for 15 min with a dry CO2/N2 (60/40) mixture at 30 °C, and the desorption took place for 1 min under dry CO2 at 80 °C. The sorbent demonstrated a consistent sorption capacity of 3.8 mmol g−1.
2.4 Sorbents based on COFs
COFs have also been explored for CO2 capture due to their pore structure and tunability, combining the structural advantages of MOFs with enhanced chemical and thermal stability enabled by covalent bonding.79,80 Fig. 6a displays the synthesis of an amine-functionalized COF (COF-999), achieving high CO2 sorption capacity, fast kinetics, and low regeneration energy.63 Crystalline olefin-linked COF precursor (COF-999-N3) was first synthesized, and then the azide group was converted to an amine group via the Staudinger reaction. The obtained COF-999-NH2 was further treated with aziridine to produce polyamines in the pores, ultimately yielding COF-999. Fig. 6b shows that COF-999 exhibited CO2 uptake of 0.91 mmol g−1 with 400 ppm CO2. The CO2 sorption capacity increased with increasing RHs (Fig. 6c). The sorbent achieved a CO2 sorption capacity of 2.09 mmol g−1 under 75% RH at 25 °C. | ||
| Fig. 6 DAC sorbent based on COF-999. (a) Schematic diagram of the material synthesis; (b) CO2 sorption isotherm with 400 ppm CO2; (c) CO2 sorption uptake with RH values of 0%, 25%, 50% and 75% at 400 ppm CO2. Copyright 2024 Springer Nature.63 | ||
2.5 Sorbents based on hybrid liquid-nanoparticles
Hybrid materials have been developed for DAC sorbents using waterless CO2 solvents, such as ionic liquids (ILs), CO2-binding organic ligands, and liquid-like nanoparticle organic hybrid materials (NOHMs).67 Fig. 7a shows that PEI-functionalized NOHMs (NOHM-I-PEI) can be encapsulated in a polymer shell by UV curing and formed into thin films. The solvent impregnated polymers (SIP) particles were synthesized by grinding these films, and the CO2 capture mechanism of the NOHM-I-PEI-incorporated SIPs (NPEI-SIPs) is shown in Fig. 7a. | ||
| Fig. 7 CO2 capture in NPEI-SIPs containing 49 mass% NOHM-1-PEI. (a) Structure and sorption mechanism. (b) Comparison with pristine NOHM-I-PEI. (c) Effect of humidity on CO2 capture for 400 ppm CO2.67 Copyright 2021 Wiley. | ||
Fig. 7b compares the CO2 capture kinetics of the hybrid material (NPEI-SIPs) containing 49% NOHM-I-PEI with that of the pristine NOHM-I-PEI. The NPEI-SIPs exhibited CO2 sorption capacity of 5.55 mmol CO2 per g NOHM-I-PEI, much higher than that for NOHM-I-PEI (0.09 mmol CO2 per g). The base liquid exhibited high viscosity, resulting in very slow sorption kinetics; by contrast, the polymer matrix in the SIPs showed high CO2 diffusivity, leading to fast kinetics. Fig. 7c shows that introducing water vapor (80% RH) increased CO2 sorption capacity.
2.6 Moisture-driven DAC based on polyamine derivatives
Most polyamines are thermally regenerated after CO2 sorption, leading to high energy consumption, and they are susceptible to oxidative degradation at high temperatures.81,82 Recently, a novel approach of moisture-swing-based DAC has been developed with the sorbents regenerated at high RH and ambient temperature,83–86 as shown in Fig. 8. These processes are often based on polyamine derivatives, such as quaternary ammonium-containing polymers. Specifically, CO2 is captured under dry conditions and released upon exposure to higher RH levels, as shown in the equations in Fig. 8a. The hydroxide-functionalized quaternary ammonium groups react with CO2 under dry conditions to form bicarbonate (eqn (i)). When exposed to high RH levels, water drives the reverse reaction, thereby converting bicarbonate to carbonate and releasing CO2 (eqn (ii) and (iii)), enabling sorbent regeneration. | ||
| Fig. 8 Moisture-swing sorbents based on quaternary ammonium-functionalized graphene oxide (fGO). (a) Sorption and desorption mechanism, including three reactions, and (b) effect of humidity on CO2 sorption.83 Copyright 2024 Wiley. | ||
Fig. 8b displays the effect of RHs on CO2 sorption in an example moisture-swing sorbent (fGO), prepared by functionalizing graphene oxide (GO) with glycidyltrimethylammonium chloride.83 The fGO displayed the highest CO2 uptake at 20% RH, followed by a decline in capacity as RH increased to 100%, giving a moisture-swing working capacity of 2.3 mmol g−1. Moreover, the fGO sorbent exhibited excellent cycling stability, retaining performance for over 40 adsorption–desorption cycles, with only a 7.5% loss after 50 cycles.
3 Polyamines for membrane CO2 separation
3.1 Modeling of facilitated CO2 transport in polyamines
Gas transport in non-facilitated polymers is usually described using the solution-diffusion model, and gas permeability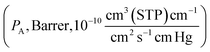 is expressed as:
is expressed as:| PA = SA × DA | (5) |
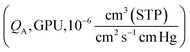 with a thickness (l, cm) is given by:
with a thickness (l, cm) is given by:| QA = PA/l | (6) |
Polyamines have been widely reported to facilitate CO2 transport in the presence of water through reversible reactions with amines.87,88 For example, in eqn (3), reagents and products can be simplified using the following abbreviations: A = CO2, B = R–NH2, C = HCO3−, and D = R–NH3+. Water solubility in polyamines is significantly larger than CO2 solubility, and therefore, it is considered a constant.89 The total amine concentration in polyamines (CT) can be defined as follows:
| CT = CB + CD | (7) |
The reaction equilibrium constant (Keq) can be defined as:
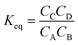 | (8) |
Assuming that the CO2 partial pressure in the permeate is negligible, CO2 flux (JCO2) can be calculated as follows:90
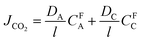 | (9) |
HCO3− concentration can be correlated to the CO2 concentration using eqn (7) and (8).
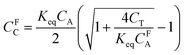 | (10) |
By using eqn (9) and (10), CO2 permeance (QCO2) can be written as:90
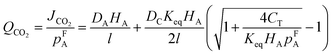 | (11) |
The physical properties parameters used in eqn (11) can be determined from CO2 sorption isotherms. Additionally, a lumped form of eqn (7) was used to investigate the effect of CO2 partial pressure on permeance, as follows.
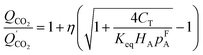 | (12) |
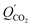 is the permeance due to the solution-diffusion mechanism, and η represents the contribution of the facilitated transport. This facilitated transport model was validated using experimental data. As an example, eqn (12) agrees well with experimental CO2 permeances as a function of CO2 partial pressure (0.1–100 kPa) and operating temperatures (57–77 °C).90
is the permeance due to the solution-diffusion mechanism, and η represents the contribution of the facilitated transport. This facilitated transport model was validated using experimental data. As an example, eqn (12) agrees well with experimental CO2 permeances as a function of CO2 partial pressure (0.1–100 kPa) and operating temperatures (57–77 °C).90
To better understand the complex mechanism of facilitated transport, the role of amine carriers in these membranes was examined at the molecular level using computational approaches, including density functional theory (DFT) calculations and molecular simulations.91–93 For example, DFT methods were utilized to investigate the amine-CO2 chemistry of poly(N-vinylformamide-co-vinylamine) (PNVF-co-VAm) with fixed carriers and two mobile carriers, piperazine glycinate (PZ-Gly) and 2-(1-piperazinyl)ethylamine sarcosinate (PZEA-Sar) (Fig. 9a).92 CO2 interacts with amino groups via two primary reaction pathways: carbamate pathway (eqn (1) and (2)) and bicarbonate pathway (eqn (3) and (4)), in which the bicarbonate pathway with equimolar stoichiometry provides a higher CO2 sorption capacity.
 | ||
| Fig. 9 DFT and MD calculation of facilitated CO2 diffusion in PZ-Gly and PZEA-Sar. (a) Chemical structures. (b) Relative electronic energy (ΔEcarbamate) of structures in the amine-CO2 reactions of carriers. Copyright 2020 American Chemical Society.92 | ||
Fig. 9b displays that PZEA-Sar and PZ-Gly exhibited lower relative electronic energy (ΔEcarbamate) values (−18.1 and −17.5 kcal mol−1, respectively), compared to VAm (−13.8 kcal mol−1) following the carbamate pathway, suggesting that CO2 has a higher reactivity towards the primary amino group on the mobile carrier than the fixed carrier. The simulation results confirmed that adding PZEA-Sar to PNVF-co-VAm increased CO2 permeance, with an increase greater than that observed with PZ-Gly. Furthermore, an MD simulation suggested that CO2 diffused faster through the bicarbonate state than through the carbamate state, since bicarbonate species can not only associate with protonated carriers but also hop between them.93 CO2 reaction intermediates (carbamate and bicarbonate species) were also confirmed using operando surface-enhanced Raman spectroscopy (SERS) in combination with in situ transmission FTIR spectroscopy.94
3.2 Polyamine-based membranes with facilitated CO2 transport
Amines can act as either fixed or mobile carriers within the polymer matrix. For fixed-carrier polyamines, the reactive amino groups are covalently bonded to the polymer backbone, in which CO2 molecules can hop across amine reactive sites down the concentration gradient.93,95,96 Alternatively, amine groups can be dispersed as small molecules within the polymer matrix, where they reversibly react with CO2 and diffuse through the membrane, thereby enhancing CO2 transport efficiency. Table 2 lists the representative polyamines and their CO2/N2 separation properties in water-saturated gas mixtures. In general, there is no correlation between the amine content and CO2/gas separation properties,97 while increasing water content typically increases CO2 permeance and CO2/gas selectivity.98,99| Polyaminesa | Amine loading (wt%) | Thickness (µm) | Temp. (°C) | Pressure (bar) | CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2 mixture N2 mixture |
QCO2 | PCO2 | CO2/N2 selectivity | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| a The matrix is displayed in brackets.b PG: piperazine glycinate. PNVF-co-VAm: poly(N-vinylformamide-co-vinylamine); PEGu: poly(ethylene guanidine); PZEA-Sar: 2-(1-piperazinyl)ethylamine sarcosinate; ProK: potassium prolinate; PMVAM: poly(N-methyl-N-vinylamine); HMMP: high-valence MMP; MMP: metal-induced ordered microporous polymers; AIBA-K: 2-aminoisobutyric acid potassium salt. | |||||||||
| PVAm | 1.2 | 25 | 2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
104 | 197 | 100 | ||
| PGb (PVAm) | 65 | 0.25 | 57 | 0.1 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 80 |
945 | 87 | 101 | |
| HMMP-1 (PVAm) | 0.1 | 0.18 | 45 | 2 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
1544 | 252 | 102 | |
| PZEA-Sar/PEGu (PNVF-co-VAm) | 0.17 | 67 | 4 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 80 |
2397 | 186 | 8 | ||
| ProK (PVA) | 40 | 0.5 | ≈23 | 2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
791 | 40 | 103 | |
| UiO-66-NH2 (aPEO) | 10 | 0.328 | 23 | 2.36 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
1400 | 76 | 6 | |
| MMP-3 | 0.05 | 25 | 2 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
3000 | 78 | 104 | ||
| am-PTFE AF | 0.01 | 25 | 1.2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
∼1000 | ∼1000 | 7 | ||
| TMC/DNMDAm/DGBAmE | 0.5 | 22 | 1.1 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
1613 | 138 | 105 | ||
| PMVAm | 12 | 102 | 1 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 80 |
6804 | 350 | 20 | ||
| Lupamin® 9095/AIBA-K/AF-MWNT (PVA) | 40 | 21 | 107 | 15 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 40 |
886 | 428 | 106 | |
| PAA-C3H7 (PVA) | 70 | 25 | 110 | 2 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 40 |
295 | 335 | 88 | |
| NH2–Co/ZIF-8 (PEO) | 5 | 300 | 25 | ∼0.1 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90 |
2916 | 47 | 107 | |
| Porphyrin (PSF) | 20 | ≈23 | 2–10 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
134 | 115 | 108 | ||
| NUS-8-NH2 (PIM-1) | 10 | 54 | 25 | 2 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 80 |
14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 638 638 |
29 | 109 | |
| UiO-66-NH2@IL (PIM-1) | 10 | ∼70 | 20 | 1 | 8283 | 23 | 110 | ||
Fig. 10 illustrates the structure, composition, and CO2/N2 separation performance of polyamine-based TFC membranes.8 Fig. 10a displays the configuration of membranes comprising a dense selective layer (∼170 nm) supported on a porous support (30 µm). The selective layer contained both PNVF-co-VAm and PEGu as the fixed layer and PZEA-Sar as the mobile layer, which together provided a balance between CO2 reactivity and structural integrity. Increasing the temperature from 67 to 77 °C increased CO2 permeance from 2500 to 4200 GPU, accompanied by a slight decline in CO2/N2 selectivity. Additionally, PEI-decorated multilayered montmorillonite (MMT) was incorporated into PVAm membranes, resulting in CO2 permeance of 217 GPU and CO2/N2 selectivity of 112.111
 | ||
| Fig. 10 Superior CO2/N2 separation performance in FTMs based on PNVF-co-VAm. (a) Schematic illustration and chemical composition, and (b) CO2/N2 separation performance of the scale-up membrane at 67 and 77 °C.8 Copyright 2024 Elsevier. | ||
Fig. 11a shows a hybrid-integrated (HI) membrane comprising a modified amine-rich surface layer and a polymeric support layer with superior CO2/N2 separation properties.7 Two commercial membranes, polydimethylsiloxane (PDMS) and polytetrafluoroethylene (PTFE AF), were selected as the support layer. The amine-functionalized PDMS and PTFE AF (am-PDMS and am-PTFE) exhibited lower CO2 permeability than their pristine membranes, and CO2 permeability decreased slightly with increasing pressure, consistent with the facilitated transport mechanism (Fig. 11b). By contrast, these membranes exhibited much higher mixed-gas CO2/N2 selectivity than their unmodified membranes (Fig. 11c). These membranes exhibited CO2 permeability >1000 Barrer and CO2/N2 selectivity >100, surpassing the 2019 upper bound.112
 | ||
| Fig. 11 Superior CO2/N2 separation properties in hybrid-integrated membranes of polymeric supports with amine-enriched surface. (a) Schematic. (b) CO2 permeability and (c) CO2/N2 selectivity as a function of pressure. (d) Comparison with other polymers. The solid gray and red lines represent the 2008 (ref. 113) and 2019 upper bound,112 respectively. Copyright 2022 AAAS.7 | ||
Polyamines can also be used to fabricate porous materials (metal-induced ordered microporous polymers, or MMPs) for CO2/N2 separation.104 PVAm or PEI were selected as the amine-rich polymer, while aceclofenac or 4-chloroisophthalic acid served as small organic linkers, and Cu(CH3COO)2 or Zn(NO3)2·6H2O as divalent metal ions (Fig. 12a). The Cl and carboxylate groups interacted with the polymer chains through dipolar interactions, forming the MMP structure (Fig. 12b). TFC membranes with a selective layer as thin as 50 nm on an mPSF support were fabricated over large areas (>100 cm2) (Fig. 12c and d). The membrane exhibited a CO2 permeance of 3000 GPU and CO2/N2 selectivity of 78 in the presence of saturated water vapor at 25 °C (Fig. 12e).
 | ||
| Fig. 12 Polyamine-derived MMP by the polymer-directed chemical synthesis (PDCS) strategy. (a) Schematic. (b) Independent frameworks of MMP-1. (c) Surface, (d) cross-section image, and (e) CO2/N2 separation performance of MMP-1/mPSf membrane. Filled and open points represent the mPSf and MMP/mPSf membrane, respectively. The feed gas was humid CO2/N2 mixed gas (v/v: 15/85) at 25 °C and 1–2 bar.104 Copyright 2019 Springer Nature. | ||
3.3 Polyamine-based membranes with hindered CO2 transport
Although a variety of amine-modified polymers exhibit improved CO2/N2 selectivity under humidified conditions, contradictory results have also been reported. For example, amine-functionalized polysulfone (PSF) was synthesized by introducing three basic substituents, namely arylamine, benzylamine, and phthalimide, leading to PSF-NH2, PSF-CH2–NH2, and PSF-CH2-imide, respectively (Fig. 13a).114 Only PSF-CH2–NH2 showed enhancement in CO2 solubility and CO2/CH4 solubility selectivity, and introducing arylamine and phthalimide diminished CO2/CH4 solubility selectivity (Fig. 13b and Table 3). For instance, PSF-NH2 (38%) showed CO2 solubility of 2.2 cm3 (STP) (cm−3 atm−1) and CO2/CH4 solubility selectivity of 2.8 at 35 °C, which were comparable to those of PSF (2.08 cm3 (STP) (cm−3 atm−1) and CO2/CH4 solubility selectivity of 3.1, respectively). | ||
| Fig. 13 Ineffective amine groups in amine-functionalized PSF. (a) Chemical structures. (b) Effect of polar group concentration on CO2/CH4 solubility selectivity. A = PSF, B = PSF-NH2 (16%), C = PSF-NH2 (51%), D = PSF-CH2-imide (51%), and E = PSF-NH2 (38%). Copyright 1996 American Chemical Society.115 | ||
| Polymers | FFV | SCO2 | SCO2/SCH4 | DCO2 | DCO2/DCH4 | PCO2(Barrer) | PCO2/PCH4 |
|---|---|---|---|---|---|---|---|
| PSF | 0.147 | 2.08 | 3.1 | 2.0 | 7.4 | 5.5 | 23 |
| PSF-NH2 (16%) | 0.134 | 1.90 | 3.2 | 1.1 | 7.9 | 2.7 | 24 |
| PSF-NH2 (38%) | 0.118 | 2.20 | 2.8 | 1.1 | 8.5 | 3.2 | 25 |
| PSF-CH2–NH2 (51%) | 0.125 | 3.04 | 4.7 | 0.5 | 3.5 | 1.95 | 18 |
| PSF-CH2 imide (51%) | 0.122 | 1.76 | 3.4 | 0.9 | 7.6 | 2.12 | 26 |
The amine functionalization decreased CO2 diffusivity and thus permeability, which can be attributed to reduced fractional free volume (FFV). All amine-functionalized PSFs showed similar CO2/CH4 selectivity (Table 3), suggesting that amines interact with CO2 with extremely low efficiency. Similarly, functionalization of PSF with tertiary amine groups (benzyldimethylamine or DMA) or quaternary ammonium groups did not enhance CO2 solubility or CO2/CH4 solubility selectivity.116
Fig. 14 presents another example of PEI-based hindered transport membranes (HTMs) with an unusual N2/CO2 separation factor.26 A mesoporous MCM-48 membrane was impregnated with PEI, and the obtained membrane exhibited unexpectedly high N2/CO2 selectivity (18–81) at 20 °C, even in the presence of 2.6% water vapor, and the selectivity increased with increasing CO2 concentration (or partial pressure) in the feed. By contrast, the membrane exhibited N2/CO2 separation factor of ∼1 at 90 °C (Fig. 14c), which is still unexpected, considering the high content of amine groups in the membranes.
 | ||
| Fig. 14 Unexpectedly high N2/CO2 separation factor in HTMs based on PEI-modified MCM-48. (a) Membrane structure. (b) Reaction. (c) Effect of CO2 partial pressure on N2/CO2 separation factor at 20 and 90 °C with 2.6% water vapor.26 Copyright 2007 American Chemical Society. | ||
PEI can be cross-linked with hexamethylene diisocyanate (HMDI) at a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mass ratio (Fig. 15a) and used to form TFC membranes with a 450 nm-selective layer (XLPEI30, Fig. 15b).27 Despite the high content of amine groups, the membrane exhibited H2 permeance of 5.5 GPU and H2/CO2 selectivity of 1100, the highest reported for known polymeric membranes. The membrane was further evaluated with various H2/CO2 mixtures at 9.0 atm and 70 °C (Fig. 15c and d). The mixed-gas H2 permeance was much lower than the pure-gas value (where CO2 partial pressure is 0 atm) because of the ionic cross-linking of the XLPEI30 by the sorbed CO2 during the mixed-gas permeation. Higher CO2 partial pressure increased CO2 sorption and cross-linking degree, decreased free volume, and decreased gas permeance, but retained mixed-gas H2/CO2 selectivity at 100.
3 mass ratio (Fig. 15a) and used to form TFC membranes with a 450 nm-selective layer (XLPEI30, Fig. 15b).27 Despite the high content of amine groups, the membrane exhibited H2 permeance of 5.5 GPU and H2/CO2 selectivity of 1100, the highest reported for known polymeric membranes. The membrane was further evaluated with various H2/CO2 mixtures at 9.0 atm and 70 °C (Fig. 15c and d). The mixed-gas H2 permeance was much lower than the pure-gas value (where CO2 partial pressure is 0 atm) because of the ionic cross-linking of the XLPEI30 by the sorbed CO2 during the mixed-gas permeation. Higher CO2 partial pressure increased CO2 sorption and cross-linking degree, decreased free volume, and decreased gas permeance, but retained mixed-gas H2/CO2 selectivity at 100.
 | ||
| Fig. 15 Superior H2/CO2 separation performance of HTMs of XLPEI30.27 (a) Synthesis of XLPEIs from PEI and HMDI. (b) Cross-sectional SEM image. (c) Mixed-gas permeance and (d) H2/CO2 selectivity as a function of the feed CO2 partial pressure at 70 °C and 9 atm. (e) Mixed-gas CO2 permeability as a function of CO2 adsorption energy (Eads(CO2)) in simulated nanoporous graphene membranes with pore diameter from 4.3 to 9.3 Å. The error bars were calculated from ten replicate runs. | ||
Water vapor content plays an important role in determining H2/CO2 selectivity. Increasing relative humidity at 70 °C from 25% to 100% decreased H2/CO2 selectivity from 10 to 0.14.27 To elucidate the hindered transport at the molecular level, CO2 transport through a nanoporous graphene membrane was simulated using MD simulations (Fig. 15e), with adjustable CO2 adsorption energies (Eads(CO2)) from −9.3 to −190 kJ mol−1 and pore diameters from 4.3 to 9.3 Å. CO2 permeability initially increased with increasing Eads(CO2) due to the enhanced affinity, but then decreased as the affinity became too strong for the sorbed CO2 to desorb and diffuse through the membrane. Consequently, CO2 permeability exhibited a volcano-shaped trend across all three pore sizes, with peaks at about −37 kJ mol−1, indicating the boundary between CO2-facilitated and hindered transport. Notably, this was only for dry conditions; when water vapor was introduced, it participated in the reaction between CO2 and amines, facilitating CO2 transport, decreasing H2/CO2 selectivity with increasing RH levels.
4 Discussion and conclusion
Polyamines with a strong affinity towards CO2 have emerged as one of the most promising classes of materials for CO2 capture, and their highly tunable molecular architecture has greatly expanded the design space for DAC sorbents and PCC membranes. As expected, polyamines react with CO2, resulting in high CO2 sorption capacity even at CO2 levels as low as 400 ppm, and introducing water vapor further enhances CO2 sorption capacity. However, several unusual, sometimes counterintuitive phenomena have been reported and are summarized below.1. The amine efficiency towards CO2 sorption depends sensitively on the substrates, and the measured pseudo-equilibrium CO2 sorption may unconventionally increase with increasing temperature, reflecting the importance of the CO2 diffusion and access of amine sites.
2. Polyamines can exhibit hindered CO2 diffusion, leading to much lower CO2 diffusivity than expected and unexpectedly high N2/CO2 or H2/CO2 selectivity. These results differ dramatically from those of FTMs, showing that CO2 transport can be governed by reactive diffusion rather than a solution-diffusion mechanism.
3. The presence of water vapor influences CO2 sorption and desorption, which has been harnessed to design moisture-swing sorbents and membranes for uphill CO2 permeation based on polyamine derivatives.84,85
The key to understanding these discrepancies lies in the role of reactive CO2 diffusion in polyamines.93,117 As CO2 diffuses through polyamines, it reacts with amines, forming ionic cross-links among polymer chains, increasing chain rigidity, reducing CO2 diffusivity, and shielding the remaining polyamines from CO2 access. However, no quantitative models are available to describe this behavior.13,27 Furthermore, the strong interaction between CO2 and amines may prevent the CO2 dissociation and dramatically reduce its overall diffusivity. Consequently, the reported CO2 sorption data are mostly at pseudo-equilibrium, and sorption is influenced by temperature thermodynamically and kinetically.
We expect that polyamines will remain a dominant materials platform for DAC sorbents and PCC membranes, and they may be optimized through integrated experimental and modeling studies. A key priority will be the creation of robust predictive frameworks that couple CO2 sorption thermodynamics with mass-transfer kinetics, providing fundamental understanding of CO2 reactive diffusion and facilitating scale-up from benchtop demonstration to commercial deployment.
Conflicts of interest
The authors declare no competing financial interests.Data availability
Data are available upon request to the corresponding author.Acknowledgements
We acknowledge the financial support of the U.S. Department of Energy National Energy Technology Laboratory (NETL # DE-FE0031960 and DE-SC0020730).References
- A. Ozden, M. Luo and Y. Lum, Chem. Eng. J., 2025, 519, 165535 CrossRef CAS.
- H. Akaya, S. Lamnini, H. Sehaqui and J. Jacquemin, ACS Appl. Mater. Interfaces, 2025, 17, 16380–16395 CrossRef CAS PubMed.
- H. E. Holmes, M. J. Realff and R. P. Lively, Nat. Chem. Eng., 2024, 1, 208–215 CrossRef CAS.
- H. Bouaboula, J. Chaouki, Y. Belmabkhout and A. Zaabout, Chem. Eng. J., 2024, 484, 149411 CrossRef CAS.
- T. Merkel, R. Baker, P. Hao, J. Kniep, Z. He, Y. Huang and W. Salim, J. Membr. Sci., 2026, 738, 124829 CrossRef CAS.
- G. Zhang, C. J. Shah, W. I. Lee, K. Kisslinger, N. Esmaeili, V. T. Bui, L. Zhu, C. Y. Nam and H. Lin, Adv. Funct. Mater., 2024, 34, 2404785 CrossRef CAS.
- M. Sandru, E. M. Sandru, W. F. Ingram, J. Deng, P. M. Stenstad, L. Deng and R. J. Spontak, Science, 2022, 376, 90–94 CrossRef CAS PubMed.
- Y. Yang, Y. Han, C. Zou, R. Pang, J. Hu, K. Chen and W. S. W. Ho, J. Membr. Sci., 2024, 696, 122520 CrossRef CAS.
- F. Attia, T. Tran, V. Bui, B. Valluri, E. Deng, G. Zhang, N. Esmaeili, L. Huang and H. Lin, Chem. Mater., 2024, 36, 9603–9612 CrossRef CAS.
- P. Ignatusha, H. Lin, N. Kapuscinsky, L. Scoles, W. Ma, B. Patarachao and N. Du, Membranes, 2024, 14, 30 CrossRef CAS PubMed.
- G. Rim, P. Priyadarshini, M. Song, Y. Wang, A. Bai, M. J. Realff, R. P. Lively and C. W. Jones, J. Am. Chem. Soc., 2023, 145, 7190–7204 CrossRef CAS PubMed.
- X. Shi, H. Xiao, H. Azarabadi, J. Song, X. Wu, X. Chen and K. S. Lackner, Angew. Chem., Int. Ed., 2020, 59, 6984–7006 CrossRef CAS PubMed.
- T. Tran, S. Singh, S. Cheng and H. Lin, ACS Appl. Mater. Interfaces, 2024, 16, 22715–22723 CrossRef CAS PubMed.
- Y. Jin, H. Lin, Y. Liu, H. An and J. Lee, Renewable Sustainable Energy Rev., 2025, 217, 115782 CrossRef CAS.
- S. Rao, Y. Han and W. W. Ho, Sep. Sci. Technol., 2023, 58, 1050–1071 CrossRef CAS.
- T. Tran, Y. Fu, D. Jiang and H. Lin, Macromolecules, 2022, 55, 9860–9867 CrossRef CAS.
- A. A. Hosseini and M. J. Lashaki, Sep. Purif. Technol., 2023, 325, 124580 CrossRef.
- J. Hack, N. Maeda and D. M. Meier, ACS Omega, 2022, 7, 39520–39530 CrossRef CAS PubMed.
- E. S. Sanz-Perez, C. R. Murdock, S. A. Didas and C. W. Jones, Chem. Rev., 2016, 116, 11840–11876 CrossRef CAS PubMed.
- Z. Tong and W. S. W. Ho, J. Membr. Sci., 2017, 543, 202–211 CrossRef CAS.
- X. Shi, Y. Lin and X. Chen, MRS Bull., 2022, 47, 405–415 CrossRef CAS.
- A. Sayari, Q. Liu and P. Mishra, ChemSusChem, 2016, 9, 2796–2803 CrossRef CAS PubMed.
- Y. Han and W. W. Ho, J. Membr. Sci., 2021, 628, 119244 CrossRef CAS.
- X. Wang, M. Fujii, X. Wang and C. Song, Ind. Eng. Chem., 2020, 59, 7267–7273 CrossRef CAS.
- X. Ma, X. Wang and C. Song, J. Am. Chem. Soc., 2009, 131, 5777–5783 CrossRef CAS PubMed.
- P. Kumar, S. Kim, J. Ida and V. V. Guliants, Ind. Eng. Chem., 2008, 47, 201–208 CrossRef CAS.
- L. Hu, A. J. Gottipalli, G. Zhang, K. Fung, T. Tran, N. Esmaeili, P. Zhang, Y. Ding, K. Shi and H. Lin, Sci. Adv., 2025, 11, eadz2830 CrossRef CAS PubMed.
- A. Sodiq, Y. Abdullatif, B. Aissa, A. Ostovar, N. Nassar, M. El-Naas and A. Amhamed, Environ. Technol. Innov., 2022, 29, 102991 CrossRef.
- L. Jiang, W. Liu, R. Wang, A. Gonzalez-Diaz, M. Rojas-Michaga, S. Michailos, M. Pourkashanian, X. Zhang and C. Font-Palma, Prog. Energy Combust. Sci., 2023, 95, 101069 CrossRef.
- A. Heydari-Gorji, Y. Yang and A. Sayari, Energy Fuels, 2011, 25, 4206–4210 CrossRef CAS.
- A. Holewinski, M. A. Sakwa-Novak and C. W. Jones, J. Am. Chem. Soc., 2015, 137, 11749–11759 CrossRef CAS PubMed.
- H. Peng, J. Zhang, J. Zhang, F. Zhong, P. Wu, K. Huang, J. Fan and F. Liu, Chem. Eng. J., 2019, 359, 1159–1165 CrossRef CAS.
- L. A. Darunte, K. S. Walton, D. S. Sholl and C. W. Jones, Curr. Opin. Chem. Eng., 2016, 12, 82–90 CrossRef.
- J. Wang, D. Long, H. Zhou, Q. Chen, X. Liu and L. Ling, Energy Environ. Sci., 2012, 5, 5742–5749 RSC.
- L. Wang, M. Al-Aufi, C. N. Pacheco, L. Xie and R. M. Rioux, ACS Sustain. Chem. Eng., 2019, 7, 14785–14795 CrossRef CAS.
- J. Zhang, H. Peng, Y. Liu, D. Tao, P. Wu, J. Fan and K. Huang, ACS Sustain. Chem. Eng., 2019, 7, 9369–9377 CrossRef CAS.
- T. Witoon, Mater. Chem. Phys., 2012, 137, 235–245 CrossRef CAS.
- S. Choi, M. L. Gray and C. W. Jones, ChemSusChem, 2011, 4, 628–635 CrossRef CAS PubMed.
- V. Kulkarni, D. Panda and S. K. Singh, Ind. Eng. Chem., 2023, 62, 3800–3811 CrossRef CAS.
- H. T. Kwon, M. A. Sakwa-Novak, S. H. Pang, A. R. Sujan, E. W. Ping and C. W. Jones, Chem. Mater., 2019, 31, 5229–5237 CrossRef CAS.
- W. Chaikittisilp, H.-J. Kim and C. W. Jones, Energy Fuels, 2011, 25, 5528–5537 CrossRef CAS.
- P. Priyadarshini, G. Rim, C. Rosu, M. Song and C. W. Jones, ACS Environ. Au, 2023, 3, 295–307 CrossRef CAS PubMed.
- A. Kumar, D. G. Madden, M. Lusi, K. Chen, E. A. Daniels, T. Curtin, J. J. Perry IV and M. J. Zaworotko, Angew. Chem., Int. Ed., 2015, 54, 14372–14377 CrossRef CAS PubMed.
- S. H. Pang, R. P. Lively and C. W. Jones, ChemSusChem, 2018, 11, 2628–2637 CrossRef CAS PubMed.
- M. L. Sarazen, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, ACS Sustain. Chem. Eng., 2019, 7, 7338–7345 CrossRef CAS.
- D. R. Kumar, C. Rosu, A. R. Sujan, M. A. Sakwa-Novak, E. W. Ping and C. W. Jones, ACS Sustain. Chem. Eng., 2020, 8, 10971–10982 CAS.
- S. Choi, J. H. Drese, P. M. Eisenberger and C. W. Jones, Environ. Sci. Technol., 2011, 45, 2420–2427 CrossRef CAS PubMed.
- A. Vallace, Z. S. Campbell, H. J. Moon, W. J. Koros, C. W. Jones and R. P. Lively, Small, 2024, 20, 2401422 CrossRef CAS.
- Y. Kuwahara, D. Kang, J. R. Copeland, P. Bollini, C. Sievers, T. Kamegawa, H. Yamashita and C. W. Jones, Chem.–Eur. J., 2012, 18, 16649–16664 CrossRef CAS PubMed.
- X. Zhu, T. Ge, F. Yang, M. Lyu, C. Chen, D. O'Hare and R. Wang, J. Mater. Chem. A, 2020, 8, 16421–16428 RSC.
- M. Zhao, J. Xiao, W. Gao and Q. Wang, J. Energy Chem., 2022, 68, 401–410 CrossRef CAS.
- Y. Kong, G. Jiang, Y. Wu, S. Cui and X. Shen, Chem. Eng. J., 2016, 306, 362–368 CrossRef CAS.
- S. H. Pang, M. L. Jue, J. Leisen, C. W. Jones and R. P. Lively, ACS Macro Lett., 2015, 4, 1415–1419 CrossRef CAS PubMed.
- A. K. Sekizkardes, V. A. Kusuma, J. T. Culp, P. Muldoon, J. Hoffman, J. A. Steckel and D. Hopkinson, J. Mater. Chem. A, 2023, 11, 11670–11674 RSC.
- C. Gebald, J. A. Wurzbacher, P. Tingaut, T. Zimmermann and A. Steinfeld, Environ. Sci. Technol., 2011, 45, 9101–9108 CrossRef CAS PubMed.
- C. Gebald, J. A. Wurzbacher, A. Borgschulte, T. Zimmermann and A. Steinfeld, Environ. Sci. Technol., 2014, 48, 2497–2504 CrossRef CAS PubMed.
- T. Wang, K. S. Lackner and A. Wright, Environ. Sci. Technol., 2011, 45, 6670–6675 CrossRef CAS PubMed.
- T. M. McDonald, W. R. Lee, J. A. Mason, B. M. Wiers, C. S. Hong and J. R. Long, J. Am. Chem. Soc., 2012, 134, 7056–7065 CrossRef CAS PubMed.
- W. R. Lee, S. Y. Hwang, D. W. Ryu, K. S. Lim, S. S. Han, D. Moon, J. Choi and C. S. Hong, Energy Environ. Sci., 2014, 7, 744–751 RSC.
- L. A. Darunte, A. D. Oetomo, K. S. Walton, D. S. Sholl and C. W. Jones, ACS Sustain. Chem. Eng., 2016, 4, 5761–5768 CrossRef CAS.
- M. Song, G. Rim, Y. Wang, I. Borne, C. W. Jones and R. P. Lively, Chem. Eng. J., 2023, 477, 147135 CrossRef CAS.
- O. I.-F. Chen, C. Liu, K. Wang, E. Borrego-Marin, H. Li, A. H. Alawadhi, J. A. Navarro and O. M. Yaghi, J. Am. Chem. Soc., 2024, 146, 2835–2844 CrossRef CAS PubMed.
- Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li and A. H. Alawadhi, Nature, 2024, 635, 96–101 CrossRef CAS PubMed.
- H. Lyu, H. Li, N. Hanikel, K. Wang and O. M. Yaghi, J. Am. Chem. Soc., 2022, 144, 12989–12995 CrossRef CAS PubMed.
- H. Li, Z. Zhou, T. Ma, K. Wang, H. Zhang, A. H. Alawadhi and O. M. Yaghi, J. Am. Chem. Soc., 2024, 146, 35486–35492 CrossRef CAS PubMed.
- Y. Belmabkhout, R. Serna-Guerrero and A. Sayari, Ind. Eng. Chem. Res., 2010, 49, 359–365 CrossRef CAS.
- G. Rim, T. G. Feric, T. Moore and A. H. A. Park, Adv. Funct. Mater., 2021, 31, 2010047 CrossRef CAS.
- K. D. Kersey, G. A. Lee, J. H. Xu, M. K. Kidder, A. H. A. Park and Y. L. Joo, Adv. Funct. Mater., 2023, 33, 2301649 CrossRef CAS.
- A. N. Stuckert and R. T. Yang, Environ. Sci. Technol., 2011, 45, 10257–10264 CrossRef CAS PubMed.
- K. Baamran, P. Muldoon, K. Damodaran, S. Banerjee, K.-J. Kim, J. A. Steckel and A. K. Sekizkardes, Sep. Purif. Technol., 2025, 133186 CrossRef CAS.
- T. Tran, S. Pan, X. Chen, X.-C. Lin, A. K. Blevins, Y. Ding and H. Lin, ACS Appl. Mater. Interfaces, 2020, 12, 49192–49199 CrossRef CAS PubMed.
- F. Rezaei, R. P. Lively, Y. Labreche, G. Chen, Y. Fan, W. J. Koros and C. W. Jones, ACS Appl. Mater. Interfaces, 2013, 5, 3921–3931 CrossRef CAS PubMed.
- G. Rim, F. Kong, M. Song, C. Rosu, P. Priyadarshini, R. P. Lively and C. W. Jones, JACS Au, 2022, 2, 380–393 CrossRef CAS PubMed.
- P. M. Bhatt, Y. Belmabkhout, A. Cadiau, K. Adil, O. Shekhah, A. Shkurenko, L. J. Barbour and M. Eddaoudi, J. Am. Chem. Soc., 2016, 138, 9301–9307 CrossRef CAS PubMed.
- O. Shekhah, Y. Belmabkhout, Z. Chen, V. Guillerm, A. Cairns, K. Adil and M. Eddaoudi, Nat. Commun., 2014, 5, 4228 CrossRef CAS PubMed.
- W. L. Queen, M. R. Hudson, E. D. Bloch, J. A. Mason, M. I. Gonzalez, J. S. Lee, D. Gygi, J. D. Howe, K. Lee, T. A. Darwish, M. James, V. K. Peterson, S. J. Teat, B. Smit, J. B. Neaton, J. R. Long and C. M. Brown, Chem. Sci., 2014, 5, 4569–4581 RSC.
- J. Hoffman, L. Proaño and C. W. Jones, ACS Appl. Polym. Mater., 2025, 7, 15671–15681 Search PubMed.
- Z. Zhu, H. Tsai, S. T. Parker, J. Lee, Y. Yabuuchi, H. Z. Jiang, Y. Wang, S. Xiong, A. C. Forse and B. Dinakar, J. Am. Chem. Soc., 2024, 146, 6072–6083 Search PubMed.
- W. Wei, Z. Wang, C. Xu, Z. Li, H. Bai and Z. Liu, Ind. Eng. Chem. Res., 2025, 64, 24276–24299 Search PubMed.
- H. Daglar, Z. Zhou, R. Zhu, P. Parihar, J. I. Siepmann, O. M. Yaghi and L. Gagliardi, J. Am. Chem. Soc., 2025, 148(1), 1614–1622 Search PubMed.
- M. Fayaz and A. Sayari, ACS Appl. Mater. Interfaces, 2017, 9, 43747–43754 Search PubMed.
- A. Sinha, L. A. Darunte, C. W. Jones, M. J. Realff and Y. Kawajiri, Ind. Eng. Chem., 2017, 56, 750–764 CrossRef CAS.
- I. Nicotera, A. Enotiadis and C. Simari, Small, 2024, 20, 2401303 CrossRef CAS PubMed.
- J. L. Wade, H. L. Marques, W. Wang, J. Flory and B. D. Freeman, J. Membr. Sci., 2023, 685, 121954 CrossRef CAS.
- I. S. Metcalfe, G. A. Mutch, E. I. Papaioannou, S. Tsochataridou, D. Neagu, D. J. L. Brett, F. Iacoviello, T. S. Miller, P. R. Shearing and P. A. Hunt, Nat. Energy, 2024, 9, 1074–1083 Search PubMed.
- T. Wang, K. S. Lackner and A. B. Wright, Phys. Chem. Chem. Phys., 2013, 15, 504–514 RSC.
- C. Wu, Q. Huang, Z. Xu, A. T. Sipra, N. Gao, L. P. d. S. Vandenberghe, S. Vieira, C. R. Soccol, R. Zhao, S. Deng, S. K. S. Boetcher, S. Lu, H. Shi, D. Zhao, Y. Xing, Y. Chen, J. Zhu, D. Feng, Y. Zhang, L. Deng, G. Hu, P. A. Webley, D. Liang, Z. Ba, A. Mlonka-Mędrala, A. Magdziarz, N. Miskolczi, S. Tomasek, S. S. Lam, S. Y. Foong, H. S. Ng, L. Jiang, X. Yan, Y. Liu, Y. Ji, H. Sun, Y. Zhang, H. Yang, X. Zhang, M. Sun, D. C. W. Tsang, J. Shang, C. Muller, M. Rekhtina, M. Krödel, A. H. Bork, F. Donat, L. Liu, X. Jin, W. Liu, S. Saqline, X. Wu, Y. Xu, A. L. Khan, Z. Ali, H. Lin, L. Hu, J. Huang, R. Singh, K. Wang, X. He, Z. Dai, S. Yi, A. Konist, M. H. S. Baqain, Y. Zhao, S. Sun, G. Chen, X. Tu, A. Weidenkaff, S. Kawi, K. H. Lim, C. Song, Q. Yang, Z. Zhao, X. Gao, X. Jiang, H. Ji, T. E. Akinola, A. Lawal, O. S. Otitoju, M. Wang, G. Zhang, L. Ma, B. C. Sempuga, X. Liu, E. Oko, M. Daramola, Z. Yu, S. Chen, G. Kang, Q. Li, L. Gao, L. Liu and H. Zhou, Carbon Capture Sci. Technol., 2024, 11, 100178 CAS.
- Y. Zhao and W. W. Ho, J. Membr. Sci., 2012, 415, 132–138 Search PubMed.
- J. S. Schultz, J. D. Goddard and S. R. Suchdeo, AIChE J., 1974, 20, 417–445 CrossRef CAS.
- Y. Han and W. S. W. Ho, Ind. Eng. Chem., 2020, 59, 5340–5350 Search PubMed.
- H. Xu, S. G. Pate and C. P. O'Brien, Chem. Eng. J., 2023, 460, 141728 CrossRef CAS.
- X. Deng, C. Zou, Y. Han, L. Lin and W. W. Ho, J. Phys. Chem. C, 2020, 124, 25322–25330 CrossRef CAS.
- C. Zou, X. Deng, Y. Han and L.-C. Lin, J. Phys. Chem. C, 2025, 129, 9550–9561 Search PubMed.
- H. Xu, S. G. Pate and C. P. O'Brien, J. Membr. Sci., 2024, 689, 122163 Search PubMed.
- Y. Zhao, Z. Wan, Z. Feng, D. Yang, Y. Zhang and F. Qu, Int. J. Rock Mech. Min. Sci., 2012, 52, 132–138 Search PubMed.
- E. Cussler, R. Aris and A. Bhown, J. Membr. Sci., 1989, 43, 149–164 Search PubMed.
- Z. Zhang, S. Rao, Y. Han, R. Pang and W. S. W. Ho, J. Membr. Sci., 2021, 638, 119696 Search PubMed.
- J. Zou and W. S. W. Ho, J. Membr. Sci., 2006, 286, 310–321 CrossRef CAS.
- R. Quinn and D. V. Laciak, J. Membr. Sci., 1997, 131, 49–60 CrossRef CAS.
- M. Sandru, T. Kim and M.-B. Hägg, Desalination, 2009, 240, 298–300 CrossRef CAS.
- Y. Chen and W. S. W. Ho, J. Membr. Sci., 2016, 514, 376–384 CrossRef CAS.
- Y. Yuan, Z. Qiao, J. Xu, J. Wang, S. Zhao, X. Cao, Z. Wang and M. D. Guiver, J. Membr. Sci., 2021, 620, 118923 CrossRef CAS.
- Z. Dai, J. Deng, L. Ansaloni, S. Janakiram and L. Deng, J. Membr. Sci., 2019, 578, 61–68 CrossRef CAS.
- Z. Qiao, S. Zhao, M. Sheng, J. Wang, S. Wang, Z. Wang, C. Zhong and M. D. Guiver, Nat. Mater., 2019, 18, 163–168 CrossRef CAS PubMed.
- S. Li, Z. Wang, X. Yu, J. Wang and S. Wang, Adv. Mater., 2012, 24, 3196–3200 CrossRef CAS PubMed.
- L. Ansaloni, Y. Zhao, B. T. Jung, K. Ramasubramanian, M. G. Baschetti and W. S. W. Ho, J. Membr. Sci., 2015, 490, 18–28 CrossRef CAS.
- S. Cong, Q. Shen, M. Shan, J. Wang, J. Liu and Y. Zhang, Chem. Eng. J., 2020, 383, 123137 CrossRef CAS.
- S. Saqib, S. Rafiq, N. Muhammad, A. L. Khan, A. Mukhtar, S. Ullah, M. H. Nawaz, F. Jamil, C. Zhang and V. Ashokkumar, J. Hazard. Mater., 2021, 411, 125155 CrossRef CAS PubMed.
- Y. Pu, Z. Yang, V. Wee, Z. Wu, Z. Jiang and D. Zhao, J. Membr. Sci., 2022, 641, 119912 CrossRef CAS.
- J. Lu, X. Zhang, L. Xu, G. Zhang, J. Zheng, Z. Tong, C. Shen and Q. Meng, Membranes, 2021, 11, 35 CrossRef CAS PubMed.
- X. Lu, J. Wang, Y. Wang and X. Zhang, Sep. Purif. Technol., 2025, 358, 130380 CrossRef CAS.
- B. Comesaña-Gándara, J. Chen, C. G. Bezzu, M. Carta, I. Rose, M.-C. Ferrari, E. Esposito, A. Fuoco, J. C. Jansen and N. B. McKeown, Energy Environ. Sci., 2019, 12, 2733–2740 RSC.
- L. M. Robeson, J. Membr. Sci., 2008, 320, 390–400 CrossRef CAS.
- K. Ghosal, R. Chern, B. Freeman, W. Daly and I. Negulescu, Macromolecules, 1996, 29, 4360–4369 CrossRef CAS.
- K. Ghosal, R. T. Chern, B. D. Freeman, W. H. Daly and I. I. Negulescu, Macromolecules, 1996, 29, 4360–4369 CrossRef CAS.
- L. Zhu, D. Tian, D. Shin, W. Jia, C. Bae and H. Lin, J. Polym. Sci., Part B: Polym. Phys., 2018, 56, 1239–1250 CrossRef CAS.
- Y. Long, J. Jiang, J. S. Smink, J. E. ten Elshof, W. Rohlfs, C. W. Visser and W. Brilman, Results Eng., 2025, 26, 104766 CrossRef CAS.
Footnote |
| † Singh and Alebrahim: equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |
