Open Access Article
Open Access ArticleNanocomposites with tunable dielectric properties: BaTiO3 loaded cellulose nanocrystal-liquid crystalline polymers
Patrick K. Njengaa,
Ning Guo b,
Haotian Longc,
Dennis M. Ndayab,
Akram H. Alhadainy
b,
Haotian Longc,
Dennis M. Ndayab,
Akram H. Alhadainy bd,
Chinedum O. Osuji
bd,
Chinedum O. Osuji c,
Yang Cao
c,
Yang Cao be and
Rajeswari M. Kasi
be and
Rajeswari M. Kasi *ab
*ab
aDepartment of Chemistry, University of Connecticut, Storrs, CT 06269, USA. E-mail: rajeswari.kasi@uconn.edu; Tel: +1 (860)-486-4713
bInstitute of Materials Science, University of Connecticut, Storrs, CT 06269, USA
cChemical and Biomolecular Engineering, University of Pennsylvania, PA 19104-639, USA
dDepartment of Biomedical Engineering, University of Connecticut, Storrs, CT 06269, USA
eDepartment of Electrical Engineering, University of Connecticut, Storrs, CT 06269, USA
First published on 29th April 2026
Abstract
Dielectric nanocomposites are emerging as next-generation energy storage materials due to their applicability in advanced electronics, renewable energy systems, and electric vehicles. In this work, we report the rational molecular design, synthesis, and structure–property investigation of a BaTiO3/cellulose nanocrystal (CNC)-liquid crystal polymer (LCP) nanocomposite with enhanced dielectric performance. The hybrid platform integrates BaTiO3 nanoparticles dispersed within a cyanobiphenyl-based polyacrylate matrix (PAACB12-r-PAA), followed by in situ interlocking with CNCs. This biomaterial-supported anisotropic system, exploits both the permanent dipole of smectic cyanobiphenyl liquid crystalline mesogens and ordering of CNC-LC materials to induce hierarchical self-assembly and promote strong dielectric responses. Films are fabricated through hot-pressing at the liquid crystalline transition temperature (TLC), followed by rapid quenching to lock in the alignment. We systematically investigate the influence of processing parameters such as BaTiO3 concentration, LCP alignment, and thermal treatment on dielectric behavior. Temperature-controlled small-angle X-ray scattering (TSAXS), broadband dielectric spectroscopy, and microscopy are employed to correlate mesophase orientation, dispersion quality, and molecular interactions with the dielectric constant, breakdown strength, energy density, and dielectric loss. Our findings reveal that the liquid crystalline matrix not only enhances BaTiO3 dispersion and interfacial polarization but also facilitates structural ordering that improves the composite's dielectric performance. The permanent dipoles within the LCP matrix further augment polarization via Ti4+ displacement from the O2− octahedra in BaTiO3, offering tunable dielectric enhancement. This work establishes a general strategy for designing multifunctional dielectric nanocomposites by integrating mesogen alignment, nanofiller anisotropy, and optimized processing to achieve tunable dielectric performance for advanced electronic applications.
Introduction
Dielectric nanocomposites have attracted significant interest as next-generation energy storage materials due to their broad applications in advanced electronics, renewable energy systems, and electric vehicles.1–3 Among these, ceramic–polymer nanocomposites, especially those combining barium titanate (BaTiO3) with functional polymer matrices, stand out for their tunable electrical, mechanical, and structural properties.4–6 One such promising platform integrates BaTiO3 with liquid crystal polymers (LCPs), which offer molecular-level ordering, anisotropic dielectric responses, and field-induced alignment.7–10BaTiO3 is an energy storage ceramic with a high dielectric constant and reversible polarization, making it ideal for capacitors, sensors, and transducers.11–13 However, its use in practical applications is hindered by brittleness, high density, temperature sensitivity, low dielectric strength, and poor processability.14–17 Embedding BaTiO3 within flexible polymeric matrices is hypothesized to improve the mechanical robustness and processing characteristics, while retaining or even enhancing dielectric performance.6,18–20
Organic dielectric polymers have been employed as dielectric materials due to their low-temperature and facile processability, impressive flexibility, corrosion resistance, excellent insulation properties, lightweight nature, and good thermal stability.14,21,22 In this category, biodegradable cellulose nanocrystals have gained increased interest due to their non-toxicity, renewability, high aspect ratio, adaptable surface chemistry, liquid crystallinity, and stiffness, making them a potential replacement for most synthetic dielectric polymers, which pose challenges to the environment.11,23–25 Du et al. demonstrated the natural dielectric behavior of cellulosic biopolymers in A4 paper form, where the paper is adapted as an energy storage material with excellent wettability.26–28 In a similar study, Jose and coworkers reported the temperature-dependent (30 °C to 100 °C) dielectric properties of CNC paper over a frequency range of 1 Hz to 10 MHz. Here, the authors established a temperature trend of the dielectric properties, proving the potential of CNCs as an environmentally benign energy storage material in flexible electronics.29
Other liquid crystalline polymers (LCPs), such as smectic cyanobiphenyl, present distinct advantages, such as a well-defined processing window governed by their liquid crystalline transition temperature (TLC), intrinsic anisotropy, and stimuli-responsive molecular alignment, all of which can be tailored to enhance dielectric performance.30–32 Interlocking these anisotropic smectic LC molecules with chiral nematic CNCs modifies the dielectric performance of the system.32–34 The electric field-induced alignment of these polar structures enhances local polarization by facilitating Ti4+ displacement from the O2− octahedra in BaTiO3, thereby augmenting dipole formation and energy storage.35–37
While the dielectric properties of LCPs have been extensively studied, the use of broadband dielectric spectroscopy to investigate the effects of nanoparticles on the dynamics of LCPs with mesomorphic phases has received limited attention. In this study, we present dielectric spectroscopy results of a novel BaTiO3/CNC-LCP nanocomposite. The incorporation of BaTiO3 nanoparticles not only significantly enhances the dielectric constant but also suppresses the librational fluctuations of the 12-spacer cyanobiphenyl mesogen around the long molecular axis. Because BaTiO3 does not establish a wetted interface with CNC-i-PAACB12-r-PAA, the nanoparticles act more as steric obstacles within the long-range ordered structure (whose formation is also facilitated by BaTiO3) to hinder the motion of the mesogen. This constrained movement results in a slight increase in the liquid–crystal transition temperature, TLC1, of the nanocomposite. These findings provide new insight into how inorganic fillers modulate the dielectric response of LCP-based nanocomposites.
Experimental
Materials
Barium titanate, BaTiO3 (cubic), 4-dimethylamino pyridine, DMAP (99.0%), potassium carbonate (99%), acrylic acid (99%) and azobisisobutyronitrile, AIBN (98%), are purchased from Sigma Aldrich. 12-Bromo-1-dodecanol (95%) is provided by Lab Seeker Chemicals, while 4-cyano-4′-hydroxyphenyl is obtained from TCI America. Cytiva Life Sciences supplied standard-grade Whatman filter paper, while dimethyl formamide (99%) is purchased from Across Organics. 1-Ethyl-3-(3-dimethyl aminopropyl) carbodiimide hydrochloride, EDC·HCl (98.0%) and 1-butyl-3-methylimidazolium acetate (95%) are acquired from Oakwood Chemicals. Thermo Fisher Scientific supplied dichloromethane (98%) and dimethyl sulfoxide (99.0%).Synthesis of BaTiO3/PAACB12-r-PAA
BaTiO3 nanoparticles and PAACB12-r-PAA were homogenously dispersed in THF by ultrasonication at room temperature for 1 h. The sonicated sample is dried over nitrogen and subsequently vacuum-dried to form BaTiO3/PAACB12-r-PAA, as outlined in Scheme 1 below. | ||
| Scheme 1 Synthetic scheme for the formation of BaTiO3/CNC-i-PAACB12-r-PAA nanocomposites for dielectric studies. | ||
Preparation of BaTiO3/CNC-i-PAACB12-r-PAA nanocomposites
BaTiO3/PAACB12-r-PAA is then interlocked in situ with CNCs via double interlocking chemistry, as shown in Scheme 2. The reactants (Table 1) are transferred to a clean, dry 50 mL round-bottomed flask equipped with a magnetic stir bar. 5 mL THF is added, and the flask is sealed with a rubber septum. The reaction mixture is purged with argon for 3 minutes and stirred in an oil bath at 0 °C for 2 hours. The temperature gradually increased to 30 °C, and the reaction was allowed to continue for a further 6 hours. The crude reaction mixture is then cooled and carefully transferred into a clean, dry scintillation vial and ultrasonicated at 25 °C for 1 hour for optimal dispersion. After complete sonication, the sample is vacuum-dried at room temperature overnight. Free-standing films were then prepared by hot-pressing the nanocomposite at the corresponding TLC (for 1 hour), followed by cold-air quenching to induce hierarchical self-assembly. This enables phase alignment, essential for high-performance dielectric materials.38–40 The synthetic scheme for the formation of BaTiO3/CNC-i-PAACB12-r-PAA nanocomposite films is shown in Scheme 1.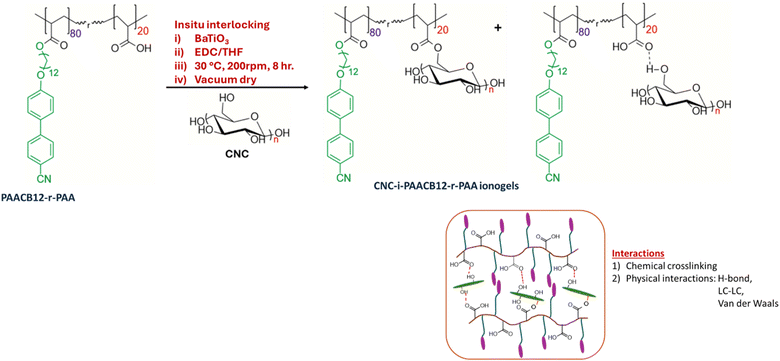 | ||
| Scheme 2 Synthetic pathway showing dual network BaTiO3/CNC-i-PAACB12-r-PAA nanocomposite gels using 40 wt% EDC in THF. | ||
| Nanocomposite | PAACB12-r-PAA (mg) | CNC (mg) | BaTiO3 (mg) | EDC (mg) | THF (mL) |
|---|---|---|---|---|---|
| CNC-i-PAACB12-r-PAA | 80 | 20 | 0 | 40 | 1 |
| BaTiO3 (10 wt%)/CNC-i-PAACB12-r-PAA | 80 | 20 | 41.6 | 40 | 1 |
| BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA | 80 | 20 | 83.2 | 40 | 1 |
| BaTiO3 (40 wt%)/CNC-i-PAACB12-r-PAA | 80 | 20 | 166.4 | 40 | 1 |
Results and discussion
The system studied is a hybrid LCP consisting of an amphiphilic random copolymer, PAACB12-r-PAA, with relative degrees of polymerization of 80 for PAACB12 and 20 for PAA, as shown in Scheme 1. The backbone has cyanobiphenyl mesogens attached at the ends as side chains to a polyacrylate backbone, while the other unit contains free COOH groups that interlock/crosslink with CNCs.Structural identification of the copolymer is performed using 1H NMR, assessing the chemical shifts of the respective protons, while that of the hybrid LCP and the BaTiO3-loaded nanocomposites is achieved by identifying the functional groups of the material using Fourier Transform Infrared (FTIR) spectroscopy.
Thermal analysis of the hybrid LCP and the nanocomposites is performed to understand thermal stability and phase transition behavior. Thermal gravimetric analysis (TGA) enables us to understand the decomposition profile of the material using the decomposition temperatures. The higher the decomposition temperature (Td), the more thermally stable the material. TGA studies also inform us about the interaction of the hybrid LCP with BaTiO3 and provide the range of temperatures over which DSC studies can be conducted. Differential Scanning Calorimetry (DSC) studies provide information on the phase transitions exhibited by the materials, such as melting temperatures (Tm), glass transition temperature (Tg), crystallization temperatures (Tc), liquid crystal transition temperatures (TLC), and isotropic temperatures. We can leverage DSC studies to investigate the interactions between CNCs and the copolymer in the formation of the hybrid LCP and the interaction of BaTiO3 with the hybrid LCP when preparing the nanocomposites.
X-ray techniques, such as X-ray Diffraction (XRD) and Small-Angle X-ray Scattering (SAXS), can be employed to investigate the microstructure and self-assembly behavior of BaTiO3-loaded CNC-cyanobiphenyl LCP hybrid nanocomposites. XRD scattering patterns reveal the crystallite sizes of BaTiO3, CNC-i-PAACB12-r-PAA, and BaTiO3/CNC-i-PAACB12-r-PAA nanocomposites. We can also assess the interfacial interactions of BaTiO3 with the hybrid LCP (CNC-i-PAACB12-r-PAA) by monitoring changes in crystallinity symbolized by any shift or broadening in diffraction peaks. On the other hand, SAXS studies give information regarding the hierarchical ordering and periodicity of the hybrid LCP and the BaTiO3-loaded nanocomposites. This helps us decipher the resultant mesophase during self-assembly. CNCs can also serve as alignment agents for PAACB12-r-PAA, which promotes ordered domains that enhance dielectric anisotropy.
Structure identification
The fundamental step in characterizing CNC-LCP nanocomposites is to conduct structural identification of the copolymer by assessing the chemical shifts of the respective protons in Nuclear Magnetic Resonance (NMR) spectroscopy, while that of the hybrid LCP and the BaTiO3-loaded nanocomposites is achieved by identifying the functional groups of the material using Fourier Transform Infrared (FTIR) spectroscopy. Careful evaluation of these functional groups gives us information on dominant interactions between CNCs, LCPs, and BaTiO3, which can then be correlated with the dielectric performance of the resultant nanocomposites.Successful formation of the cyanobiphenyl mesogen, acrylate monomer, and LC copolymer is characterized by 1H NMR. For the AACB12 monomer, we observe a proton signal peak at 4.0 ppm that serves as the connection peak. In contrast, the signal peak between 6.0 and 6.5 ppm shows the presence of a double bond in the acrylate monomer, a polymerization site. Biphenyl protons appear at 7.1 ppm, 7.6 ppm, and 8.0 ppm, respectively. These peaks confirm the successful grafting of the CB12 mesogen onto the acrylate backbone, as shown in Fig. S2. The 1H NMR spectrum for the copolymer shows the disappearance of the peaks at 6.0–6.5 ppm, indicating complete polymerization. We also observe an acidic proton signal at 12.0 ppm, which further confirms the success of copolymerization as seen in Fig. S3.
Formation of the hybrid LCP and BaTiO3/CNC-i-PAACB12-r-PAA nanocomposite is further confirmed by FTIR studies. For instance, we observe a strong C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching band at 2235.2 cm−1, C
N stretching band at 2235.2 cm−1, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch at 1712.2 cm−1, O–H at 3339.6 cm−1, and C–O at 1287.3 cm−1 in the nanocomposite, as shown in Fig. 1 below. We monitor the surface modification of BaTiO3 nanoparticles by comparing FTIR spectra of pure and modified BaTiO3. FTIR spectra of neat BaTiO3 show a prominent Ti–O bond stretching along the polar axis at 502.15 cm−1, which corresponds to the symmetric stretching vibration of TiO6 octahedra. This peak is important for monitoring any changes in the crystal structure after surface functionalization of BaTiO3. The peak at 1435 cm−1 is considered an artefact corresponding to BaCO3 impurities. BaTiO3/CNC-i-PAACB12-r-PAA shows additional asymmetric vibrations at around 600 cm−1, indicating distortion of the crystal symmetry of BaTiO3. This distortion results from the interplay of the polar axis of BaTiO3 with the permanent dipole in the LCP. Further studies on perturbation of the crystal structure after functionalization are performed using thermal and microstructural analyses, discussed in the next section below (Table 2).
O stretch at 1712.2 cm−1, O–H at 3339.6 cm−1, and C–O at 1287.3 cm−1 in the nanocomposite, as shown in Fig. 1 below. We monitor the surface modification of BaTiO3 nanoparticles by comparing FTIR spectra of pure and modified BaTiO3. FTIR spectra of neat BaTiO3 show a prominent Ti–O bond stretching along the polar axis at 502.15 cm−1, which corresponds to the symmetric stretching vibration of TiO6 octahedra. This peak is important for monitoring any changes in the crystal structure after surface functionalization of BaTiO3. The peak at 1435 cm−1 is considered an artefact corresponding to BaCO3 impurities. BaTiO3/CNC-i-PAACB12-r-PAA shows additional asymmetric vibrations at around 600 cm−1, indicating distortion of the crystal symmetry of BaTiO3. This distortion results from the interplay of the polar axis of BaTiO3 with the permanent dipole in the LCP. Further studies on perturbation of the crystal structure after functionalization are performed using thermal and microstructural analyses, discussed in the next section below (Table 2).
 | ||
| Fig. 1 Stacked FTIR results of copolymers, hybrid LCPs, and BaTiO3/CNC-i-PAACB12-r-PAA nanocomposites. | ||
| Functionality | Wavenumbers (cm−1) |
|---|---|
C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N N |
2229.53 (cyanobiphenyl LC) |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
1601.40 |
| C–O | 1288.56 |
| O–H | 3333.91, 3497.62 |
| C–H | 2849.60, 2917.50 |
| Ti–O | 502.15 (perovskite) |
Thermal analysis
To understand the thermal behavior of the materials, we conduct thermal analysis using thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC).41,42 TGA gives us information concerning the decomposition behavior of the material.42,43 We establish a linear relationship between thermal stability and BaTiO3 concentration in the nanocomposite, as confirmed by the increase in onset thermal decomposition temperature (Td). All the materials are reported to be thermally stable within the range of DSC measurements. The TGA results for these materials are shown in Table S1 and Fig. 2 below.Thermal transitions for the PAACB12-r-PAA copolymer, CNC-i-PAACB12-r-PAA, and BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA are probed by DSC as shown in Fig. 3. Samples are heated at a constant heating rate of 10 °C min−1 to 150 °C. The 1st heating cycle of each sample run is assumed to represent the artifact and thermal history present in the sample. The first cooling and second heating cycles are analyzed and reported as shown in Fig. 3. The PAACB12-r-PAA copolymer shows a prominent peak at 84.34 °C from the 1st cooling cycle and 97.99 °C in the 2nd heating cycle. The copolymer appears to be highly crystalline, thus masking the mobile nature of the CB12 mesogen. The 1st cooling DSC profile for CNC-i-PAACB12-r-PAA shows two distinct transitions: TLC at 40.75 °C with an enthalpy requirement of 3.79 J g−1 and a TC at 81.13 °C. Similar observations are made in 10 wt% nanocomposites with a TLC at 42.26 °C and a TC at 78.53 °C. Similar observations are made during the 2nd heating cycle. Neat CNC and BaTiO3 samples show no presence of thermal transitions, as shown in Fig. S4.
 | ||
| Fig. 3 1st cooling and 2nd heating cycles DSC thermograms for the copolymer, hybrid LCP and BaTiO3 (10 wt%)/CNC-i-(PAACB12-r-PAA) nanocomposite. | ||
To gain insight into self-assembly and resultant mesomorphic structures, further studies of the hybrid LCP and BaTiO3/CNC-i-PAACB12-r-PAA nanocomposites are conducted by small-angle X-ray scattering studies (SAXS).
Microstructure
We investigate the microstructural changes resulting from the integration of cubic BaTiO3 into the CNC-i-PAACB12-r-PAA matrix using X-ray diffraction (XRD) studies. Fig. 4 shows the XRD profile for BaTiO3 (40 wt%)/CNC-i-PAACB12-r-PAA, where the material shows biphasic behavior with the diffraction peaks traceable to PDF 79-2263, corresponding to cubic BaTiO3 nanoparticles. Bragg's reflections 1, 2, 5, 6, and 8 are traceable to the CB12 mesogen and 3, 4, 7, and 9 are pertinent to CNCs. Peak fitting is performed using Origin software, where the obtained full width at half maximum (FWHM) helps us calculate the line broadening in radians. After obtaining the line broadening, we use the Scherrer equation to calculate the crystallite size of BaTiO3 using the prominent peak, represented by the lattice parameters (110). The calculation of the crystallite size of BaTiO3 is shown below.where D – crystallite size (nm), K – Scherrer constant (∼0.94 for spherical particles with a cubic symmetry), λ – the X-ray wavelength of Cu K-alpha (0.15406 nm), β – full width at half maximum, FWHM (gives line broadening in radians), and θ – Bragg angle.
This calculated crystallite size agrees with the observed size in TEM studies, as shown in Fig. S5 and S6.
Presence of polymorphism
Room-temperature and temperature-resolved SAXS measurements of the LC composites with different barium titanate loadings were employed to investigate the nanoscale structures present in the materials. For 0, 5, and 20 wt% BaTiO3, the room-temperature SAXS profiles (Fig. 5) all exhibit two superposed families of reflections arising from layer-like structures. The first series exhibits a primary Bragg peak at qA (qA = 0.141 Å−1) followed by a higher-order peak at 3qA. We interpret this as consistent with a layer-like structure, or lamellar order, in which the scattering at 2qA is strongly suppressed due to form-factor effects associated with a symmetric electron-density profile. The d-spacing of this structure is calculated as dA = 2π/qA = 2π/0.141 Å−1 ≈ 4.46 nm. This dimension is roughly twice the length of a single cyanobiphenyl (CB12) mesogenic side chain (∼2.5 nm from previous work),44 which strongly suggests the formation of a smectic bilayer. In such a structure, the repeating unit is not a single layer of mesogens but a pair of layers. The second series exhibits scattering maxima with peak locations in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio with the d-spacing dB = 2π/qB = 2π/0.300 Å−1 ≈ 2.09 nm (qB = 0.300 Å−1), which is consistent with the dimensions of a single layer of the sidechain mesogen. The presence of two distinct sets of periodic reflections, with non-commensurate fundamental q-values, indicates nanoscale phase coexistence, implying that the as-cast material is not a single, homogeneous mesophase but is instead a polymorphic system containing at least two different, well-ordered lamellar structures.
2 ratio with the d-spacing dB = 2π/qB = 2π/0.300 Å−1 ≈ 2.09 nm (qB = 0.300 Å−1), which is consistent with the dimensions of a single layer of the sidechain mesogen. The presence of two distinct sets of periodic reflections, with non-commensurate fundamental q-values, indicates nanoscale phase coexistence, implying that the as-cast material is not a single, homogeneous mesophase but is instead a polymorphic system containing at least two different, well-ordered lamellar structures.
 | ||
| Fig. 5 Room temperature small-angle X-ray scattering (SAXS) results for the hybrid LCP and nanocomposites. | ||
The temperature-controlled SAXS data, including the first heating and cooling cycles, show that qB, qC, and qD appear and disappear simultaneously, suggesting that they originate from the same structure (Fig. 6). However, qC and qD are not readily indexed as higher-order lamellar reflections, and we therefore attribute them to additional electron-density correlations, likely associated with CNC-rich domains in the system. Upon heating the sample above 90 °C, the structure evolves from the bilayer structure (qA, 3qA) to the monolayer-included structure (qB, qC, and qD), which corresponds to the TLC2 transition observed in the DSC data. During the subsequent cooling cycle, the monolayer-based structure persists and the bilayer reflections do not re-emerge, even at room temperature, for both 0 and 20 wt% BaTiO3. The bilayer structure is therefore identified as a kinetically trapped, metastable state, which could be formed during the process of hot pressing followed by rapid quench, while the other structures are in the thermodynamically stable state. Both exist simultaneously for the as-cast sample, so both series of peaks are shown in the RT SAXS data. Notably, varying the BaTiO3 loading from 0 to 20 wt% does not fundamentally alter this polymorphic behavior or the nature of the thermal transition, indicating a robust self-assembly of the LCP matrix that successfully incorporates the nanoparticles without significant structural disruption.
 | ||
| Fig. 6 Temperature resolved small-angle X-ray scattering (TSAXS) results for the hybrid LCP and nanocomposites. | ||
Electronic structure analysis
The absorption intensity of the hybrid LCP and BaTiO3/CNC-i-PA-ACB12-r-PAA nanocomposites is observed to increase sharply with the BaTiO3 loading ratio, as shown in Fig. 7. Nanocomposites with a 40 wt% BaTiO3 loading ratio had a maximum band gap energy of ∼4.1 eV, which is about 20% more compared to the neat LCP. All the materials are non-conducting since their bandgap energy is higher than 3.0 eV. We also observe a distinct absorption peak at 275 nm, which corresponds to the π–π stacking resulting from cyanobiphenyl. At BaTiO3 loading ratios of 20 wt% and 40 wt%, we observe a hypsochromic effect where the absorption peak appears blue-shifted from 375 nm relative to the 10 wt% nanocomposite and hybrid LCP. This proves that the BaTiO3 concentration is vital when modifying the electrical behavior of dielectric nanocomposites. We therefore need to have a systematic balance between the intricate interplay between BaTiO3, CB12, and CNCs. These bandgap energies are calculated from the λmax using the equation below, and the results are summarized in Table 3.where Eg is the bandgap energy, h – Planck's constant, c – speed of light (∼3.0 × 108 m s−1), and λ – wavelength (nm).
| Nanocomposite | Eg (eV) |
|---|---|
| CNC-i-PAACB12-r-PAA | 3.4 |
| BaTiO3 (10 wt%)/CNC-i-PAACB12-r-PAA | 3.6 |
| BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA | 3.8 |
| BaTiO3 (40 wt%)/CNC-i-PAACB12-r-PAA | 4.1 |
We observe that the LCP type has a significant impact on the electrical behavior of the nanocomposites. BaTiO3/CNC nanocomposites appear more blue-shifted relative to BaTiO3/PAACB12-r-PAA and BaTiO3/CNC-i-PAACB12-r-PAA. Introduction of BaTiO3 in CNC-i-PAACB12-r-PAA increases the absorption intensity of the material while impeding π–π stacking resulting from CB12 at 275 nm, as shown in Fig. 8.
Morphology
To understand the size of BaTiO3 nanoparticles, we utilized transmission electron microscopy (TEM) studies.45 Neat BaTiO3 NPs were observed to have a quasi-spherical morphology, with an average particle size of about 200 nm as measured from a TEM micrograph (Fig. S6). This agreed with the calculated crystallite size calculated from XRD studies using the Scherrer equation. This shows that the particle size of the BaTiO3 remained unchanged.On the other hand, SEM studies offered complementary morphological insights into the nanoscale structural information obtained from TEM and X-ray studies.45 SEM samples are sputtered with a 5 nm layer of 80/20 AuPd before the test. At 10 K magnification, SEM micrographs for BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA nanocomposites annealed at different LC transition temperatures (Fig. S7 and S8) showed a smooth CNC-LCP matrix with distributed particulate features, pertinent to the BaTiO3 nanoparticles. We observe minor agglomeration clusters of about 100 nm in isolated regions and a layered texture, consistent with the smectic cyanobiphenyl polymer as confirmed by the SAXS scattering results. This layered texture also exhibited a fibrillated texture associated with CNC alignment. The partially agglomerated clusters appeared sparsely distributed, while most of the BaTiO3 is embedded along the CNC-rich domains. This aligns with the SAXS evidence of smectic self-assembly of CNC-LCP nanocomposites, where CNCs provide the nucleation site for BaTiO3 and act as a rigid template that influences LC molecular alignment. This templated dispersion minimizes random aggregation, resulting in a long-range order of the BaTiO3 and mesogens, as observed in the room-temperature SAXS profile (Fig. 5). We note that bulk-level homogeneity cannot be confirmed without cross-sectional electron microscopy. However, the SEM results revealed the absence of interfacial voids between BaTiO3 and the LCP, which correlates with the small domain analysis conducted by X-ray diffraction (XRD) studies, where the persistence of CNC and LC structural peaks after BaTiO3 integration showed a retained crystalline order of the individual moieties. The inclusion of BaTiO3 stabilized the thermal behavior of the nanocomposites, as confirmed by TGA studies (Fig. 2).
Dielectric performance
To investigate the impact of BaTiO3 on the dielectric properties, broadband dielectric spectroscopy of both the CNC-i-PAACB12-r-PAA polymer matrix and BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA nanocomposites is performed. As shown in Fig. 9(a)–(d), the dielectric constant (ε) and loss of the polymer matrix and nanocomposite become significantly large at relatively high temperatures (>0 °C) and low frequencies, indicating considerable contributions from electrode polarization (EP) and D.C. conductivity. At 10 kHz, the ε of CNC-i-PAACB12-r-PAA is 5.9 at −30 °C and 8.1 at 30 °C, whereas the nanocomposite reaches 8.2 and 10.8, potentially due to the Maxwell–Wagner-Sillars polarization at the BaTiO3–polymer interface.The effect of BaTiO3 on relaxation dynamics is further examined via the imaginary part of the electrical modulus, M*(ω) = 1/ε*(ω). As illustrated in Fig. 9(e)–(h), both the polymer matrix and the nanocomposite display two loss peaks in their imaginary M*. The loss peaks appearing in the very low frequency range, where ε of both materials significantly increases, are EP-induced relaxation processes. The low-frequency EP loss peak of the nanocomposite overlaps more with its high-frequency peak compared to the polymer matrix (Fig. 9(e)–(h)), which is attributed to the increased D.C. conductivity due to the incorporation of BaTiO3, thus reducing the time constant of the EP relaxation process.46,47 The high-frequency peak, observed in the imaginary M* of both materials (not in the nanocomposite alone), is associated with the dielectric response of the polymer matrix rather than the BaTiO3-polymer interfacial polarization. This is further supported by the fact that the high-frequency loss peak is still observed in PAACB12-r-PAA without CNC interlocking (Fig. S9). Since the dielectric spectroscopy measurement is conducted at TLC1, the observed relaxation process should be attributed to the β process due to the librational fluctuations of the mesogen (12-spacer cyanobiphenyl) around the long molecular axis (carboxyl groups are largely fixed due to hydrogen bonding with CNCs). Arrhenius plots in Fig. 9(g) demonstrate the temperature-dependent relaxation rates (1/τpeak) of this relaxation process for both materials. The relaxation process of the nanocomposite possesses a higher activation energy and a slower relaxation rate compared to the polymer matrix, implying a suppressed relaxation process due to the presence of BaTiO3 nanoparticles. A traditional reason for restricted relaxation in nanocomposite polymers is the attraction at the interface between inorganic nanoparticles and the polymer matrix.48 However, in the LCP nanocomposite, BaTiO3 and CNC-i-PAACB12-r-PAA do not form a classical thermodynamically wetted interface. In contrast, the presence of interfacial adhesion, mediated by dipolar and surface interactions, strengthens the proposed mechanism: restricted mesogen mobility arises from interfacial anchoring and ordering constraints imposed by the BaTiO3 surface, rather than from a fully wetted or chemically bonded interface.
Therefore, BaTiO3 nanoparticles primarily function as a steric hindrance while facilitating the formation of a long-range ordered structure, thereby reducing the mobility of the mesogen. Due to this restricted movement of the mesogen, the liquid–crystal transition temperature (TLC1) of the nanocomposite is also slightly increased.
Due to the exceptionally high dielectric constant of BaTiO3(20 wt%)/CNC-i-PAACB12-r-PAA nanocomposites, which exceeds that of state-of-the-art commercial capacitor-grade film (BOPP) by more than four-fold, a discharge energy density of 0.1 J cm−3 has already been achieved at only ∼40 MV m−1, Fig. 10. Given the relatively large sample thickness, electrical breakdown did not occur even after the maximum voltage of our power supply (10 kV) was applied, which limits the experimental evaluation of discharge energy density at higher electric fields. However, based on theoretical estimation using dielectric constant, the discharge energy density of BaTiO3(20 wt%)/CNC-i-PAACB12-r-PAA nanocomposites can reach approximately 2.2 J cm−3 at 200 MV m−1 (the typical design field of BOPP), assuming a 70% charge–discharge efficiency. In comparison, BOPP delivers a 0.39 J cm−3 at 200 MV m−1.49 The D–E loop of BaTiO3(20 wt%)/CNC-i-PAACB12-r-PAA nanocomposites and CNC-i-PAACB12-r-PAA are presented in Fig. 10.
The energy storage behavior of dielectric materials can be described by the following equations. As illustrated in the D-E loop in Fig. 11, the charged energy density corresponds to the total area of Area I and Area II, whereas the discharged energy density is represented by Area I alone. Therefore, minimizing Area II, which reflects energy loss, is essential for achieving high charge–discharge efficiency and maximizing discharge energy density.
| Ue = ∫EdD = AreaI |
| Uloss = AreaII |
where Ue: discharged energy density, Uloss: loss, E: electric field, D: electric displacement, AreaI: area of Area I, AreaII: area of Area II, and η: charge–discharge efficiency.
Conclusion
We successfully designed a novel hybrid energy storage material comprising BaTiO3-loaded CNC/LCP nanocomposites and conducted a comprehensive structure–property evaluation, including mesophase behavior. The correlation between self-assembled mesostructures and dielectric performance was systematically established. Compression molding of BaTiO3-loaded CNC/LCP nanocomposite films promoted the ordering of liquid crystals (LCs) around the BaTiO3 nanoparticles, enabling long-range organization without the need for external field-induced alignment. LC–LC interactions were found to dominate the system, leading to mesostructures consistent with the smectic mesophase associated with the CB12 mesogen.Broadband dielectric spectroscopy performed on CNC-i-PAACB12-r-PAA and BaTiO3 (20 wt%)/CNC-i-PAACB12-r-PAA samples revealed a significant increase in the dielectric constant (ε) and loss at temperatures above 0 °C and low frequencies, attributed to electrode polarization (EP) and DC conductivity. These effects diminished above 10 kHz, consistent with Kramers–Kronig analysis. The ε values increased from 5.9 to 8.1 (−30 °C to 30 °C) for the CNC-i-PAACB12-r-PAA hybrid polymer and from 8.2 to 10.8 for the BaTiO3 (20 wt%) nanocomposite. This enhancement is attributed to Maxwell–Wagner–Sillars polarization at the BaTiO3–polymer interface. Overall, the dielectric response is strongly governed by LCP molecular alignment and interfacial polarization effects. Variations in relative permittivity and loss tangent further highlight the role of CNC interlocking in promoting uniform morphology and retaining structural integrity of the films.
Relaxation dynamics, analyzed via the imaginary part of the electrical modulus (M*(ω)), revealed two distinct loss peaks in both systems. The low-frequency peak, associated with EP relaxation, overlapped with the high-frequency peak in the nanocomposite due to enhanced DC conductivity from BaTiO3, resulting in reduced EP relaxation time. The high-frequency peak originated from the polymer matrix dielectric response, as confirmed by its presence in PAACB12-r-PAA without CNC interlocking. The relaxation mechanism corresponds to mesogen (CB12) β-process fluctuations, with Arrhenius analysis indicating clear temperature dependence. Compared to the polymer matrix, the nanocomposite exhibited higher activation energy, a slower relaxation rate, and an increased liquid–crystal transition temperature (TLC), consistent with restricted side-chain mobility induced by BaTiO3 nanoparticles, in agreement with the DSC results.
Overall, the incorporation of BaTiO3 into CNC-based cyanobiphenyl LCP nanocomposites enhances domain ordering and interfacial polarization, leading to improved dielectric performance. While the observed morphology suggests good structural integrity upon filler incorporation, quantitative evaluation of mechanical properties such as toughness and brittleness requires further investigation. Nevertheless, the synergistic integration of LCP ordering, CNC reinforcement, and BaTiO3 highlights the strong potential of this nanocomposite system for next-generation energy storage applications, particularly capacitors with tunable dielectric properties enabled by controlled molecular design and self-assembly.
Conflicts of interest
The authors declare no conflict of interest.Data availability
All the data reported in this manuscript are available in tables and figures both in the main manuscript and the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ta00808a.Acknowledgements
P. K. N. and the authors remain indebted to Dr Sun Xuanhao of the Bioscience Electron Microscopy Laboratory and Dr Lichun Zhang of the Center for Advanced Microscopy and Materials Analysis (CAMMA) at the Innovation Partnership Building (IPB) for their assistance. R. M. K. is grateful for support from NSF through DMR-1507045 and the University of Connecticut Office of the Vice President for Research (OVPR) Research Excellence Program. N. G. and Y. C. would like to acknowledge the support of the Office of Naval Research (N0014-23-1-2062). C. O. and H. L. acknowledge NSF support through DMR 2223705. We also acknowledge the central instrumentation facilities within the Institute of Materials Science and the Department of Chemistry, including instrumentation in the laboratory of Prof. Steven Suib, and the small-angle X-ray scattering facilities at the University of Pennsylvania. Finally, we thank Prof. Menka Jain and her students for valuable discussions.References
- M. Yang, M. Guo, E. Xu, W. Ren, D. Wang, S. Li, S. Zhang, C.-W. Nan and Y. Shen, Polymer nanocomposite dielectrics for capacitive energy storage, Nat. Nanotechnol., 2024, 19(5), 588–603 CrossRef CAS PubMed.
- F. Faheem, M. Arsalan and M. E. Khan, Recent developments of nanocomposites in energy-related applications, Nanocomposites-Advanced Materials for Energy and Environmental Aspects, 2023, pp. 111–127 Search PubMed.
- Q.-K. Feng, S.-L. Zhong, J.-Y. Pei, Y. Zhao, D.-L. Zhang, D.-F. Liu, Y.-X. Zhang and Z.-M. Dang, Recent progress and future prospects on all-organic polymer dielectrics for energy storage capacitors, Chem. Rev., 2021, 122(3), 3820–3878 CrossRef PubMed.
- D. Vignesh and E. Rout, Multipurpose Polymer Dielectric Materials and Composites for Advanced Energy Applications, in Metal Oxide-Based High-K Dielectrics, CRC Press, 2025, pp 289–332 Search PubMed.
- Y. Wang, M. Yao, R. Ma, Q. Yuan, D. Yang, B. Cui, C. Ma, M. Liu and D. Hu, Design strategy of barium titanate/polyvinylidene fluoride-based nanocomposite films for high energy storage, J. Mater. Chem. A, 2020, 8(3), 884–917 RSC.
- L. You, B. Liu, H. Hua, H. Jiang, C. Yin and F. Wen, Energy storage performance of polymer-based dielectric composites with two-dimensional fillers, Nanomaterials, 2023, 13(21), 2842 CrossRef CAS PubMed.
- J. Chen, X. Liu, Y. Tian, W. Zhu, C. Yan, Y. Shi, L. B. Kong, H. J. Qi and K. Zhou, 3D-Printed anisotropic polymer materials for functional applications, Adv. Mater., 2022, 34(5), 2102877 CrossRef CAS.
- M. M. Mhatre, A. Katariya-Jain, M. H. Saeed and R. Deshmukh, Effects of multifunctional thiol monomers and BaTiO3 nanoparticles on electro-optical and dielectric properties of polymer dispersed liquid crystal films, J. Mol. Liq., 2024, 413, 125945 CrossRef CAS.
- W. Wu, X. Sun, D. Wang, H. Qian, B. Wang, X. Wang, X. Rong, X. Zhang and G. Wu, The effect of high dielectric BaTiO 3 nanoparticle dimensions on the dielectric properties and electro-optical performance of polymer dispersed liquid crystal films, J. Mater. Chem. C, 2024, 12(20), 7386–7397 RSC.
- D. Zhang, C. Ma, X. Zhou, S. Chen, H. Luo, C. R. Bowen and K. Zhou, High performance capacitors using BaTiO3 nanowires engineered by rigid liquid-crystalline polymers, J. Mater. Chem. C, 2017, 121(37), 20075–20083 CAS.
- Y. B. Adediji, A. M. Adeyinka, D. I. Yahya and O. V. Mbelu, A review of energy storage applications of lead-free BaTiO3-based dielectric ceramic capacitors, Energy, Ecol. Environ., 2023, 8(5), 401–419 CrossRef CAS.
- D. Hu, Z. Pan, X. Tan, F. Yang, J. Ding, X. Zhang, P. Li, J. Liu, J. Zhai and H. Pan, Optimization the energy density and efficiency of BaTiO3-based ceramics for capacitor applications, Chem. Eng. J., 2021, 409, 127375 CrossRef CAS.
- C. Deng, Y. Zhang, D. Yang, H. Zhang and M. Zhu, Recent progress on barium titanate-based ferroelectrics for sensor applications, Adv. Sens. Res., 2024, 3(6), 2300168 CrossRef CAS.
- Prateek, V. K. Thakur and R. K. Gupta, Recent progress on ferroelectric polymer-based nanocomposites for high energy density capacitors: synthesis, dielectric properties, and future aspects, Chem. Rev., 2016, 116(7), 4260–4317 CrossRef CAS PubMed.
- S. Wang, C. Yang, X. Li, H. Jia, S. Liu, X. Liu, T. Minari and Q. Sun, Polymer-based dielectrics with high permittivity and low dielectric loss for flexible electronics, J. Mater. Chem. C, 2022, 10(16), 6196–6221 RSC.
- H. Tao, M. Yan, L. Zhang, Z. Zou, B. Han, H. Dong, B. Zhu and X. Li, Progress in preparation, processing, and application of high dielectric polypropylene matrix composite materials, Polym. Compos., 2024, 45(6), 4819–4838 CrossRef CAS.
- G. Zhang, Q. Li, E. Allahyarov, Y. Li and L. Zhu, Challenges and opportunities of polymer nanodielectrics for capacitive energy storage, ACS Appl. Mater. Interfaces, 2021, 13(32), 37939–37960 CrossRef CAS PubMed.
- Y. Jin, N. Xia and R. A. Gerhardt, Enhanced dielectric properties of polymer matrix composites with BaTiO3 and MWCNT hybrid fillers using simple phase separation, Nano Energy, 2016, 30, 407–416 CrossRef CAS.
- A. Bouhamed, N. Jöhrmann, S. Naifar, B. Böhm, O. Hellwig, B. Wunderle and O. Kanoun, Collaborative filler network for enhancing the performance of BaTiO3/PDMS flexible piezoelectric polymer composite nanogenerators, Sensors, 2022, 22(11), 4181 CrossRef CAS PubMed.
- J. Su and J. Zhang, Recent development on modification of synthesized barium titanate (BaTiO3) and polymer/BaTiO3 dielectric composites, J. Mater. Sci.: Mater. Electron., 2019, 30(3), 1957–1975 CrossRef CAS.
- S. Guo, X. Duan, M. Xie, K. C. Aw and Q. Xue, Composites, fabrication and application of polyvinylidene fluoride for flexible electromechanical devices: A review, Micromachines, 2020, 11(12), 1076 CrossRef PubMed.
- L. Li, L. Han, H. Hu and R. Zhang, A review on polymers and their composites for flexible electronics, Mater. Adv., 2023, 4(3), 726–746 RSC.
- X. Hu, A. K. An and S. S. Chopra, Life cycle assessment of the polyvinylidene fluoride polymer with applications in various emerging technologies, ACS Sustain. Chem. Eng., 2022, 10(18), 5708–5718 CrossRef CAS.
- B. Améduri and H. Hori, Recycling and the end of life assessment of fluoropolymers: recent developments, challenges and future trends, Chem. Soc. Rev., 2023, 52(13), 4208–4247 RSC.
- B. Ameduri, From vinylidene fluoride (VDF) to the applications of VDF-containing polymers and copolymers: recent developments and future trends, Chem. Rev., 2009, 109(12), 6632–6686 CrossRef CAS PubMed.
- D. Ji, M. Zhang, H. Sun, B. Yuan, C. Ma, Z. He, Y. Ni and S. Song, Enhanced mechanical and dielectric properties of lignocellulosic composite papers with biomimetic multilayered structure and multiple hydrogen-bonding interactions, Int. J. Biol. Macromol., 2024, 281, 136247 CrossRef CAS PubMed.
- Z. Du, N. Wang and J. Du, Recent Progress in Cellulose-Based Conductive Hydrogels, Polymers, 2025, 17(8), 1089 CrossRef CAS PubMed.
- G. Du, J. Wang, Y. Liu, J. Yuan, T. Liu, C. Cai, B. Luo, S. Zhu, Z. Wei and S. Wang, Fabrication of advanced cellulosic triboelectric materials via dielectric modulation, Advanced Science, 2023, 10(15), 2206243 CrossRef CAS PubMed.
- J. Jose, J. A. Salam and V. Thomas, Effect of temperature and frequency on the dielectric behaviour of cellulose nanocrystals, Ind. Crops Prod., 2024, 222, 119428 CrossRef CAS.
- L. Zhou, S. Liu, X. Miao, P. Xie, N. Sun, Z. Xu, T. Zhong, L. Zhang and Y. Shen, Advancements and applications of liquid crystal/polymer composite films, ACS Mater. Lett., 2023, 5(10), 2760–2775 CrossRef CAS.
- J. Guardia, J. A. Reina, M. Giamberini and X. Montane, An up-to-date overview of liquid crystals and liquid crystal polymers for different applications: a review, Polymers, 2024, 16(16), 2293 CrossRef CAS PubMed.
- F. K. Masese, P. K. Njenga, D. M. Ndaya and R. M. Kasi, Recent advances and opportunities for cellulose nanocrystal-based liquid crystalline polymer hybrids and composite materials, Macromolecules, 2023, 56(17), 6567–6588 CrossRef CAS.
- S. Jia, B. Yang, J. Du, Y. Xie, L. Yu, Y. Zhang, T. Tao, W. Tang and J. Gong, Uncovering the Recent Progress of CNC-Derived Chirality Nanomaterials: Structure and Functions, Small, 2024, 20(35), 2401664 CrossRef CAS PubMed.
- A. Abbasi Moud, Chiral liquid crystalline properties of cellulose nanocrystals: fundamentals and applications, ACS Omega, 2022, 7(35), 30673–30699 CrossRef CAS PubMed.
- R. K. Mishra, A study of control mechanisms in micro and nano system-enhanced polymer nanocomposites under mechanical and electrical stimuli, An Experimental and Computational Investigation, Cranfield University, 2023 Search PubMed.
- B. S. Sahoo, N. K. Nath, R. Parida, B. N. Parida and N. C. Nayak, Dielectric Behaviour and Energy Storage Performance of La-Doped GdFeO3 Nanoparticle Filled PVDF Composite, Ceram. Int., 2025, 54244–54254 CrossRef CAS.
- R. Guo, H. Luo, M. Yan, X. Zhou, K. Zhou and D. Zhang, Significantly enhanced breakdown strength and energy density in sandwich-structured nanocomposites with low-level BaTiO3 nanowires, Nano Energy, 2021, 79, 105412 CrossRef CAS.
- Q. Luo, Y. Liu, G. Zhou and X. Xu, A new strategy to improve the dielectric properties of cellulose nanocrystals (CNCs): Surface modification of small molecules, Carbohydr. Polym., 2024, 324, 121451 CrossRef CAS.
- A. Babaei-Ghazvini and B. Acharya, The effects of aspect ratio of cellulose nanocrystals on the properties of all CNC films: Tunicate and wood CNCs, Carbohydr. Polym. Technol. Appl., 2023, 5, 100311 CAS.
- A. Durairaj, M. Maruthapandi, A. Saravanan, J. H. Luong and A. Gedanken, Cellulose nanocrystals (CNC)-based functional materials for supercapacitor applications, Nanomaterials, 2022, 12(11), 1828 CrossRef CAS PubMed.
- N. F. A. Zainal, J. M. Saiter, S. I. A. Halim, R. Lucas and C. H. Chan, Thermal analysis: basic concept of differential scanning calorimetry and thermogravimetry for beginners, Chem. Teach. Int., 2021, 3(2), 59–75 CrossRef.
- D. Lőrinczy, A. Bata, Z. Lábas, F. Shafiei, H. Szabó, J. Cifra and L. G. Nöt, Thermal analysis of tendon, cartilage and bone samples from shoulder joint replacement patients using differential scanning calorimetry (DSC) and thermogravimetry (DTA/TG), J. Therm. Anal. Calorim., 2024, 149(20), 11471–11483 CrossRef.
- C. Bracht, T. Preitschopf, M. Schöneich, T. Neff, C. Kallfaß and M. Finze, Influence of Crucible Types on Thermal Stability Analysis of Li-Ion Battery Components by Thermogravimetric Analysis–Differential Scanning Calorimetry, Energy Technol., 2024, 12(8), 2400365 CrossRef CAS.
- F. K. Masese, D. Ndaya, C.-H. Liu, N. Eddy, M. D. Morales-Acosta, M.-P. Nieh and R. M. Kasi, Self-assembled materials from cellulose nanocrystals conjugated with a thermotropic liquid crystalline moiety, Soft Matter, 2022, 18(42), 8165–8174 RSC.
- T. Yu, X. Ji, Z. Fu, B. Xia, S. Jiang and M. Gao, Morphology, structure, and defect design of BaTiO3-based ceramics for optimizing energy storage performance, J. Alloys Compd., 2025, 1026, 180220 CrossRef CAS.
- F. Kremer and A. Schönhals, Broadband Dielectric Spectroscopy, Springer Science & Business Media, 2002 Search PubMed.
- W. H. Woodward Broadband dielectric spectroscopy—A practical guide, in Broadband Dielectric Spectroscopy: A Modern Analytical Technique, ACS Publications, 2021, pp 3–59 Search PubMed.
- S. K. Sharma, J. Prakash, J. Bahadur, M. Sahu, S. Mazumder and P. K. Pujari, Free volume and lamellar structure of poly vinyl alcohol-nanosized BaTiO3 composite: positron annihilation and small angle X-ray scattering study, Eur. Polym. J., 2016, 84, 100–110 CrossRef CAS.
- Z. Dai, Z. Bao, S. Ding, C. Liu, H. Sun, H. Wang, X. Zhou, Y. Wang, Y. Yin and X. Li, Scalable polyimide-poly (amic acid) copolymer based nanocomposites for high-temperature capacitive energy storage, Adv. Mater., 2022, 34(5), 2101976 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |