Open Access Article
Open Access ArticleIn vacuo XPS study: controlled ALD growth of Al2O3 on metallic lithium enabled by plasma pretreatment
Tippi Verhelle a,
Lowie Henderick
a,
Lowie Henderick a,
Saeed Yaribcd,
Siebe Coessens
a,
Saeed Yaribcd,
Siebe Coessens a,
Matthias M. Minjauwa,
Louis De Taeye
a,
Matthias M. Minjauwa,
Louis De Taeye ce,
Philippe M. Vereeckence,
Jolien Dendooven
ce,
Philippe M. Vereeckence,
Jolien Dendooven a,
Mohammadhosein Safari
a,
Mohammadhosein Safari bcd and
Christophe Detavernier*a
bcd and
Christophe Detavernier*a
aDepartment of Solid State Sciences, Ghent University, Krijgslaan 285 S1, 9000 Gent, Belgium. E-mail: Christophe.Detavernier@Ugent.be
bInstitute for Materials Research (IMO-imomec), UHasselt, Martelarenlaan 42, 3500, Hasselt, Belgium
cEnergyville, Thor Park 8320, 3600, Genk, Belgium
dImec Division IMOMEC, 3590, Diepenbeek, Belgium
eImec, Kapeldreef 75, Leuven, 3001, Belgium
First published on 24th April 2026
Abstract
Direct atomic layer deposition (ALD) of protective coatings on metallic lithium is a promising strategy to suppress unwanted side reactions in next-generation batteries. Although several studies report ALD coatings on lithium, with the majority depositing Al2O3, inconsistencies in growth behavior are present. Also in this work, the deposition of Al2O3 using trimethylaluminium (TMA) and O2 plasma on as-received metallic lithium results in irreproducible and non-ALD-like growth. This highlights the need for a fundamental understanding of how the initial surface state governs subsequent ALD reactions. In this work, plasma pretreatments are used to systematically control the lithium surface and in vacuo X-ray photoelectron spectroscopy (XPS) is used during the TMA/O2 plasma ALD process in order to understand its growth behavior on metallic lithium. The as-received lithium surface consists of Li2CO3, LiOH and Li2O, with composition evolving over time, making it an unreliable starting surface for ALD. Argon plasma pretreatment efficiently removes carbon contamination, producing a Li2O-terminated surface, while O2 plasma results in a mixed LiOH/Li2O surface. Self-limiting, ALD-like growth of (lithiated) Al2O3 is observed only on the LiOH/Li2O surface, enabled by reactive hydroxyl groups that facilitate TMA adsorption. In contrast, TMA exposure on the Li2O surface results in immediate precursor decomposition, likely due to a porous nature of the Li2O layer allowing direct interaction between TMA and the bulk lithium underneath. These results show that controlling and clearly defining the initial surface state is essential for achieving reproducible ALD growth on metallic lithium and may help explain the inconsistencies reported in earlier studies.
1 Introduction
The sustainable electrification of our society is more than ever at the center of global discussions. Lithium-ion batteries (LIBs) can play an essential role in enabling this transition. Due to their high energy density and long cycle life, they are widely being used for energy storage in electrical vehicles and large-scale energy storage systems.1 However, as demand for increasing performance continues to grow, state-of-the-art LIBs are approaching their energy density limits, driving the search for next-generation battery chemistries.2 Metallic lithium has long been considered as the ‘holy grail’ of anode materials due to its high theoretical specific capacity (3860 mA h g−1) and low electrochemical potential (−3.04 V vs. standard hydrogen electrode).1 Combining metallic lithium with current or next-generation cathode materials could result in higher energy density batteries.2 Although promising in theory, the practical implementation of lithium metal anodes remains a significant challenge.Upon contact with the electrolyte, a solid-electrolyte interphase (SEI) is formed on the lithium metal surface due to electrolyte reduction. The SEI continues to grow during cycling, leading to progressive loss of active lithium.2 Due to the SEI's complex and inhomogeneous composition,2–6 properties such as ionic conductivity are also non-uniform across the surface, resulting in preferential ion transport through pathways of least resistance.4,7 As a result, lithium-ion deposition happens at localized ‘hot spots’,5,8 initializing the formation of dendritic structures.5,8,9 Once nucleated, these dendrites grow in size and shape and induce stress upon the SEI, resulting in fractures and exposure of fresh lithium, thereby triggering additional parasitic reactions and repeated SEI formation.4,6,7,10 During discharge, dendrites are not completely stripped due to the thick and uneven SEI,4 resulting in electrically isolated lithium, referred to as dead lithium.4,6,10 Clearly, this behavior of continuous loss of electrolyte and lithium severely degrades the performance of the lithium metal battery. In severe cases, dendrites can grow into needle-like structures capable of piercing the separator, posing a serious risk for short-circuits and thermal runaway.2,6,7
This problem highlights the critical role of the inhomogeneity of the SEI's composition in the battery performance.9 To address this issue, different strategies aim to passivate the lithium surface with a homogeneous and stable artificial SEI layer with uniform interfacial properties to improve cycle lifetime and safety of the lithium metal battery.2,5,6 One approach is solution-based and involves engineering the liquid electrolyte by modifying salts, solvents or adding additives. Although a protective SEI can be formed, the resulting SEI composition is dictated by uncontrolled decomposition products.6 Vacuum techniques such as magnetron sputtering and atomic layer deposition (ALD), offer more control over composition and thickness.6 ALD, which relies on self-limiting surface reactions by sequentially introducing individual precursor gases, enables the growth of conformal coatings with precise atomic-scale thickness control.
In 2015, Kozen et al.11 demonstrated the direct deposition of Al2O3 on Li metal foil using TMA and O2 plasma, reporting that a 14 nm film could delay atmospheric and organic solvent corrosion, leading to an overall improved discharge capacity retention in Li–S batteries. Shortly thereafter, Kazyak et al.12 showed that even a limited number of ALD cycles (e.g. 30 cycles of TMA–H2O) could already significantly extend the cycle lifetime and suppress dendrite formation. While these pioneering studies highlighted the potential of ALD coatings for Li metal protection, growth mechanisms were not studied. Thickness was reported on a conventional Si wafer,11 however, TMA or H2O do not necessarily react in self-limiting ways with the Li surface. The importance of differentiating between layer growth on silicon versus lithium was highlighted by Chen et al.13 They reported that the initial TMA and H2O reactions on Li do not saturate, leading to more deposited material than expected, when compared to growth on Si. Although promising results could be obtained, with the abovementioned contributions forming a promising starting point for optimized lithium surfaces, general progress has been slow with only few studies further exploring ALD on Li metal.14–23 Furthermore, it is often not specified whether the used lithium foil has undergone any pretreatment step before ALD. Some mention using the lithium foil as-received, while others mechanically scraped the foil.22 As-received lithium foil contains a native surface layer, with contaminants such as Li2CO3, LiOH and Li2O,15,24,25 even under inert storage, with composition evolving over time. Previous results highlight that ALD surface enhancement for metallic lithium could be very promising, but more effort is needed in order to fully understand how native surface species interact with ALD precursors, such as TMA, and how further ALD growth is influenced.
In this work, we address this knowledge gap by studying the initial interaction between TMA and plasma pretreated lithium surfaces. Instead of starting from as-received foil, the lithium surface composition is tailored through Ar and O2 plasma pretreatments performed in the same high vacuum (HV) chamber as the subsequent ALD process. These plasma pretreatments (partially) remove the native surface layer and produce a defined and reproducible starting surface in a more controlled manner. For comparison, the reaction of TMA on as-received and mechanically treated lithium foils is also investigated. Scanning electron microscopy (SEM) reveals that highly non-reproducible and localized growth is obtained for the as-received and mechanically treated foil. In vacuo X-ray photoelectron spectroscopy (XPS) and SEM results indicate that Ar plasma pretreatment promotes TMA decomposition, while after an O2 plasma pretreatment step, controlled deposition of (lithiated) Al2O3 is possible. These findings demonstrate that precursor reactivity and growth evolution depend on the initial lithium surface. Establishing a controlled and reproducible starting surface is therefore essential for understanding and controlling ALD processes on Li foil.
2 Experimental section
A custom-built cluster tool, schematically shown in Fig. 1, was used for in vacuo XPS characterization. This system includes a pump-type ALD reactor (base pressure: 10−7 mbar) directly connected to a Theta Probe XPS (base pressure: 10−9 mbar). An Ar-filled glovebox (H2O/O2 < 0.5 ppm) is integrated into this system and is connected to the ALD reactor through an UHV transfer line (10−9 mbar), allowing sample handling in an inert environment. Lithium samples (250 µm, Cambridge Energy Solutions Ltd) are treated in the ALD setup and then transferred to the XPS chamber in vacuum (10−7 mbar) and in under two minutes, minimizing contamination risk. For each XPS measurement, a fresh piece of lithium foil was treated and analyzed, avoiding repeated transfers and further reducing the risk of contamination. Each piece was separately loaded into the reactor where the full pretreatment and ALD sequence was performed and stopped at the step of interest. To ensure reproducibility and account for foil-to-foil variability, selected measurements were repeated up to three times, each on a new piece of lithium foil.Plasma pretreatments were conducted in the ALD reactor, which was equipped with a plasma column (diameter: 63 mm) on top, above the substrate, ensuring line-of-sight, without substrate bias. Plasmas of Ar (≥99.999%) and O2 (99.5%) were generated by a remote inductively coupled plasma source of 13.56 MHz operated at 200 W, with each gas maintained at a pressure of 1 × 10−3 mbar. The influence of plasma pressure is elucidated in the SI, Fig. S1, showing the importance of maintaining a low pressure to ensure surface etching. Subsequent ALD treatments involved trimethylaluminium (TMA, Al(CH3)3, 97%, Volatec, 10−2 mbar) in combination with O2 plasma (200 W, 5 × 10−3 mbar), with the first TMA dose pulsed immediately after the plasma pretreatment, with only 15 s of pumping in between. The ALD cycle consisted of a 60 s TMA pulse, 30 s pumping, 30 s O2 plasma exposure and 30 s pumping. Substrate temperature during treatments was maintained at 100 °C, unless mentioned otherwise.
XPS measurements were performed using a Theta Probe XPS system with a monochromatic Al Kα source (15 kV, 70 W) focused to a 0.3 mm diameter spot and incident at 30° relative to the surface normal. The concentric hemispherical analyzer accepted electrons over a wide angular range (20°–80° relative to the surface normal) and the input lens was positioned at 50°. High-resolution spectra were acquired using a pass energy of 50 eV and a step size of 0.1 eV, while survey spectra were collected at 200 eV pass energy and 0.5 eV step size. Charge neutralization was not used. Quantification and peak fitting were carried out using CasaXPS,26 employing a U 2 Tougaard background and LA(50) line shapes. Binding energy referencing could not be performed based on the adventitious hydrocarbon C 1s core level as the C 1s signal was not always present, hence the O 1s Li2O peak was used and positioned at 531.2 eV.27 A minor Li2CO3 contribution in the C 1s signal, when present, served as additional consistency check. Reference samples of Al2O3 were also prepared on Si substrates to provide an internal reference for the Al2O3 O 1s peak. Peak fitting of Li2CO3, LiOH and Li2O was done based on binding energy separations determined in the work of Wood et al.27 and were constrained within narrow intervals. The O 1s FWHM values were kept consistent across spectra.
Surface morphology was characterized using scanning electron microscopy (JEOL JSM-IT800, accelerating voltage of 10 kV) with a secondary-electron detector. Elemental analysis was performed by energy-dispersive X-ray spectroscopy (EDX, OXFORD X4 Xplore 30) at a working distance of 10 mm. Treated lithium foils were transferred to the SEM without air exposure, making use of the Environmental Cell Specimen Holder.28
3 Results and discussion
3.1 Starting surface
A pretreatment step is often not mentioned in other work, implying that the lithium foil is used as-received. It is well-known that commercial lithium foil develops a native surface layer during manufacturing and glovebox storage,15,24,25 which we also observe in our in vacuo XPS measurements, shown in Fig. 2(a). The C 1s spectrum shows a dominant Li2CO3 peak at 293 eV, along with adventitious carbon at 288.2 eV, shifted from the common 285 eV due to charging, as reported by Wood et al.27 The Li2CO3 signal is also visible in the Li 1s (at 58.1 eV) and in the O 1s (at 534.8 eV) spectra. An additional peak at 56.2 eV for Li 1s and 531.2 for O 1s is present, corresponding to Li2O.27 A minor feature around 55 eV in the Li 1s region indicates the presence of Li0, suggesting that the native layer is either sufficiently thin or locally discontinuous to allow detection of the underlying lithium metal. SEM imaging of this surface, Fig. 2(b), shows a relatively smooth morphology. However, the composition of the native layer is highly dependent on how the lithium foil has been manufactured and on storage time in the glovebox.25 Depending on how the foil was prepassivated, other contaminants, such as P and F, could also play a role.29 Furthermore, by leaving the foil inside the glovebox for a couple of weeks, the fraction of carbonate at the surface increases and generally, a thicker native layer is obtained.25 This makes the comparison between different studies more difficult, as often the state of the initial surface is not reported7 and it is unclear how this influences ALD growth. To make sure that depositions are always performed in the same manner, a pretreatment step, resulting in a reproducible surface, should be applied.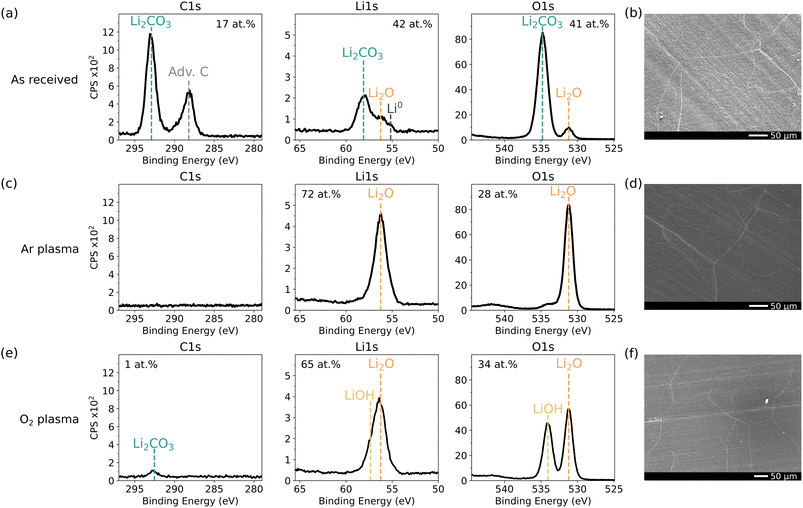 | ||
Fig. 2 In vacuo XPS spectra of C 1s, Li 1s and O 1s and SEM images of (a and b) as-received, (c and d) Ar* and, (e and f)  treated lithium foil. Survey spectra can be found in Fig. S2. treated lithium foil. Survey spectra can be found in Fig. S2. | ||
A common pretreatment is mechanical treatment, i.e. scraping the foil to remove the native layer, creating a more reactive lithium surface. While straightforward, this approach is time sensitive since the scraping can only be performed in the glovebox, where the freshly exposed surface reacts with residual gases, meaning its state at deposition strongly depends on the delay between scraping and transfer.
To gain better control over the surface composition, we carried out a plasma pretreatment step in the same vacuum chamber as the deposition. Similar plasma treatments have been used in our previous work30 to generate distinct surface chemistries. In this work, we solely focus on Ar and O2 plasma, from now on denoted as Ar* and  . Ar* treatment, shown in Fig. 2(c), fully removes carbon contamination and produces a Li2O surface, as observed in the Li 1s and O 1s spectra. From our own experience and reported in literature,24,27,31 obtaining a pure metallic Li surface by ion bombardment remains challenging. This can be attributed to the extreme reactivity of Li, which readily forms Li2O via trace H2O from the reactor or Ar gas impurities. Even in UHV, continuous sputtering is needed to sustain the metallic nature.27 Furthermore, Li2CO3 decomposes into Li2O under ion bombardment, releasing CO2 gas:24,27
. Ar* treatment, shown in Fig. 2(c), fully removes carbon contamination and produces a Li2O surface, as observed in the Li 1s and O 1s spectra. From our own experience and reported in literature,24,27,31 obtaining a pure metallic Li surface by ion bombardment remains challenging. This can be attributed to the extreme reactivity of Li, which readily forms Li2O via trace H2O from the reactor or Ar gas impurities. Even in UHV, continuous sputtering is needed to sustain the metallic nature.27 Furthermore, Li2CO3 decomposes into Li2O under ion bombardment, releasing CO2 gas:24,27
| Li2CO3 → Li2O + CO2(g) |
In practice, both mechanisms likely contribute to the Li2O surface.  treatment (Fig. 2(e)) similarly removes most carbon contamination, although a small amount of C (1 at%) in the form of Li2CO3 remains, potentially due to incomplete etching or CO2 redeposition.32,33 In addition to Li2O, the O 1s spectrum shows an additional component at 534 eV, which we attribute to LiOH. Distinguishing LiOH from Li2O2 is difficult due to overlapping binding energies and identical Li
treatment (Fig. 2(e)) similarly removes most carbon contamination, although a small amount of C (1 at%) in the form of Li2CO3 remains, potentially due to incomplete etching or CO2 redeposition.32,33 In addition to Li2O, the O 1s spectrum shows an additional component at 534 eV, which we attribute to LiOH. Distinguishing LiOH from Li2O2 is difficult due to overlapping binding energies and identical Li![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O ratios, as noted by Wood et al.27 This feature likely also arises from residual background species interacting with the plasma or incomplete etching. Both plasma treatments maintain a smooth surface morphology observed by SEM (Fig. 2(d and f)).
O ratios, as noted by Wood et al.27 This feature likely also arises from residual background species interacting with the plasma or incomplete etching. Both plasma treatments maintain a smooth surface morphology observed by SEM (Fig. 2(d and f)).
To obtain additional information on layer thicknesses, angle-resolved XPS (ARXPS) measurements were performed on the as-received, Ar* and  pretreated surfaces (Fig. S3). ARXPS was preferred over sputter depth profiling, as ion bombardment can significantly alter the surface chemistry.25,27 For as-received lithium, the Li0 and Li2O signals decrease with increasing surface sensitivity, confirming that these species are located beneath the Li2CO3 layer, consistent with previous reports.25 For both Ar* and
pretreated surfaces (Fig. S3). ARXPS was preferred over sputter depth profiling, as ion bombardment can significantly alter the surface chemistry.25,27 For as-received lithium, the Li0 and Li2O signals decrease with increasing surface sensitivity, confirming that these species are located beneath the Li2CO3 layer, consistent with previous reports.25 For both Ar* and  treated lithium, a weak Li0 signal remains detectable at low emission angles, indicating that metallic lithium is still accessible beneath the oxide/hydroxide layers, which are therefore relatively thin (within the XPS probing depth of ∼ 5–10 nm). Furthermore, ARXPS shows that the LiOH contribution becomes more pronounced at higher emission angles, indicating LiOH-enrichment near the outermost surface.
treated lithium, a weak Li0 signal remains detectable at low emission angles, indicating that metallic lithium is still accessible beneath the oxide/hydroxide layers, which are therefore relatively thin (within the XPS probing depth of ∼ 5–10 nm). Furthermore, ARXPS shows that the LiOH contribution becomes more pronounced at higher emission angles, indicating LiOH-enrichment near the outermost surface.
3.2 TMA exposure on as-received metallic lithium
Although several reports on ALD of Al2O3 on as-received lithium exist,11,13,15,19,20,23 the initial TMA exposure and further ALD growth has not been discussed in much detail. In this work, we exposed as-received lithium foils to different cycles of TMA- , with a TMA pulse of 60 s and
, with a TMA pulse of 60 s and  exposure of 30 s. Each of these foils were then investigated with SEM.
exposure of 30 s. Each of these foils were then investigated with SEM.
After exposing a lithium foil to 60 s of TMA, isolated black spots were visible on the foil. SEM imaging of these black spots, Fig. 3(a and b) showed that these were dispersed particles, already a few micrometers in size, mainly nucleating around the grain boundaries. EDX measurements on these particles, exhibited a strong Al signal compared to the surrounding smooth surface where little to no Al was detected (Fig. S4), indicating extensive reactions between TMA and the surface, unlike conventional ALD growth on a Si wafer.
 | ||
Fig. 3 SEM images of (a and b) lithium foil without any pretreatment after a 60 s TMA pulse, (c and d) after 5 cycles TMA –  , (e and f) after 10 cycles and (g and h) after 100 cycles. , (e and f) after 10 cycles and (g and h) after 100 cycles. | ||
With more ALD cycles, the density of the black regions increased and SEM images (Fig. 3(c–f)) show that the structures are merging together. The deposited material appeared porous, consisting of densely packed pillar-like structures. After 100 cycles, the foil was completely covered in black spots and SEM (Fig. 3(g and h)) shows an exceptionally thick layer of ∼10 µm, far exceeding the 15–20 nm typically obtained on Si under similar ALD conditions.
Notably, this outcome was not consistently reproducible. With identical process conditions and the same batch of as-received lithium foil, the foil sometimes remained metallic without black spots, with only very sparse particles visible in SEM. We believe that the way the initial TMA reaction happens, strongly dictates the subsequent growth mode. When extensive nucleation occurs in the first cycle, growth proceeds more uncontrolled, whereas fewer nucleation sites may result in a more ALD-like growth. Furthermore, this is in contrast with the results reported by Kang et al.,20,23 where they demonstrated that longer TMA pulses (5–30 s) result in blackening of the foil due to the formation of a Li–Al–C layer, several micrometers thick after only 20 s of TMA exposure. The reason for this inconsistent behavior remains unclear, but it could be related to variation in the morphology, microstructure (e.g. increased presence of defects at grain boundaries4,15) and thickness of the native surface layer and/or the sample surface history (i.e. the contaminant exposure accumulation since production), which highlights the importance of controlling the starting surface.
3.3 TMA exposure on an Ar plasma pretreated lithium surface
In order to obtain consistent results, the surface should be as reproducible as possible. This can be achieved through a pretreatment step, such as mechanical scraping of the lithium foil or plasma pretreatments.Even though mechanical scraping is an effective technique to remove the native layer, the freshly exposed surface is rapidly recontaminated. As an example, we scratched the top half of a piece of Li foil and left it in the glovebox for 10 minutes before scratching the other half. The foil was then transferred to the ALD reactor in under one minute and exposed to 30 s of TMA. Fig. 4(a) illustrates the effect of recontamination as the freshly scratched half turned completely black, whereas the ‘aged’ half remained relatively shiny. Since this surface cannot be controlled in a reproducible manner, mechanical scraping as a pretreatment will not be further discussed.
In order to limit the risk of recontamination, pretreatment steps should be performed in the same vacuum chamber as the ALD process. In this work, we use plasma treatments inside the ALD reactor to ensure reproducible starting surfaces. For the Li2O starting surface, obtained after Ar* treatment, a 30 s TMA exposure leads to a similar outcome as the scratched foil, rapidly coloring the surface visibly black (Fig. 4(b)), again pointing at substantial reactions between TMA and lithium. SEM imaging after just 2 s of TMA shows pronounced morphological changes (Fig. 4(c)). The surface is densely covered with spherical particles, with growth into pillar-like structures occurring simultaneously at certain places. This morphology is similar to TMA exposure on as-received lithium foil, however, in this case, the particles appear more rapidly and they are smaller and more densely packed. Already from 5 s of TMA, the surface appears completely black and Fig. 4(d and e) shows that the surface is completely covered with these pillars, however, forming a porous morphology visible from the voids in between. Extended exposure of TMA results in continuous growth as a cross-sectional SEM image after 30 s TMA shows a thickness of ∼2 µm Fig. 4(f and g).
XPS analysis of Ar* treated lithium exposed to 30 s of TMA at a substrate temperature of 100 °C, reveals significant incorporation of Al (18 at%) and C (46 at%), with only a small amount of oxygen. High-resolution scans are shown in Fig. 5 for different substrate temperatures. The Al 2p spectrum shows two distinct peaks, at 74.6 eV and 72.4 eV. Given the low O content, an Al–O bond is less likely and is found in our case at higher binding energies (i.e. 76.6 eV,34,35 see discussion below). Both the Al 2p component at 74.6 eV and the C 1s peak at 285.6 eV decrease proportionally in signal intensity with increasing substrate temperature, maintaining a constant C/Al atomic ratio of ∼3.7. This correlated behavior indicates that these signals are chemically linked and therefore, we have labeled these peaks as Al–C, even though an exact match in literature was not found.35 In parallel, the Al 2p peak at 72.4 eV, which is close to the expected metallic Al binding energy,35 increases in intensity with increasing substrate temperature. A similar trend is observed for the Li 1s signal at 54.8 eV, which corresponds to metallic Li.27 Furthermore, a small peak is visible around 58.7 eV, which we have tentatively assigned to Li–C bond. These results indicate that TMA exposure leads to an Al–C rich surface at lower temperatures, gradually shifting towards a more metallic character with increasing substrate temperature. The reason for this is unclear, but it could be related to more mobile Li atoms at higher temperatures as the substrate temperature is nearing the lithium melting point of 180 °C.
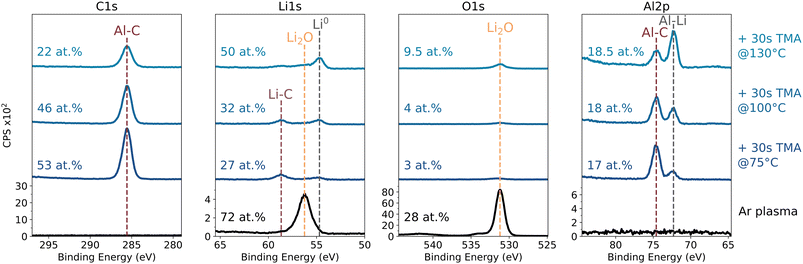 | ||
| Fig. 5 In vacuo XPS of an Ar* treated Li foil exposed to a 30 s dose of TMA at temperatures of 75, 100 and 130 °C. Survey spectra are available in Fig. S5. | ||
At a substrate temperature of 100 °C, the surface composition consisted out of 46 at% C, 32 at% Li, 18 at% Al and 4 at% O, which appears similar to the Li–Al–C layer reported by Kang et al.,20,23 with a composition of 30 at% C, 40 at% Li, 20 at% Al and 10 at% O, as determined by XPS.
While XPS provides surface sensitive information (5–10 nm), SEM reveals that the layer quickly grows into the micrometer range. In order to get an idea about the bulk composition, an EDX linescan was performed across a cross-sectional SEM image. Fig. S6 shows that while the outer surface is indeed C-rich, the C content in the bulk decreases. In contrast, the Al signal also decreases with depth, but remains substantial throughout the cross-section, indicating significant Al incorporation beyond the surface layer, likely pointing to a more Al–Li character.
The above results indicate that after an Ar* pretreatment, TMA decomposes onto the surface and conventional ALD growth cannot be obtained. A straightforward chemical pathway between TMA and Li2O that could support continuous growth is not easily thought of. The O2− anions in Li2O are strongly bound to Li+ and lack available protons for ligand exchange, making direct reaction with TMA unfavorable. We therefore hypothesize that the Li2O surface layer contains defects or voids that allow interaction with the underlying metallic lithium, an interpretation that is consistent with its low Pilling-Bedworth ratio (PBR)36 of 0.57. A PBR below unity indicates that the oxide is unlikely to fully passivate the metal surface, as molar volume mismatch between the metal and the oxide induces cracks or voids, exposing the metallic lithium underneath.25,36,37 Such exposed Li can act as a powerful reducing agent capable of donating electrons to the TMA precursor and thereby promoting reductive decomposition and potentially transmetalation of the methyl-ligands to form Li–C species. Concurrent formation and diffusion of elemental Al into the bulk can lead to Li–Al alloying, explaining the continuous growth and uncontrolled film formation.23 Even though the exact decomposition mechanism is not fully understood, our results suggest that access to metallic Li beneath the Li2O layer likely plays a dominant role in the observed film growth. Detailed calculation and interpretation of the PBR is incorporated in the SI.
3.4 TMA exposure on an O2 plasma pretreated lithium surface
If the uncontrolled reactivity originates from exposed metallic lithium beneath a defective or non-passivating Li2O layer, then forming a more passivating surface layer should suppress direct electron transfer and enable self-limiting ALD surface reactions. plasma pretreatment was therefore explored, as it produces not only Li2O, but also a LiOH component, as shown in Fig. 2. LiOH has a PBR value of 1.26 (SI), which is above unity and therefore consistent with formation of a continuous, passivating layer.
plasma pretreatment was therefore explored, as it produces not only Li2O, but also a LiOH component, as shown in Fig. 2. LiOH has a PBR value of 1.26 (SI), which is above unity and therefore consistent with formation of a continuous, passivating layer.
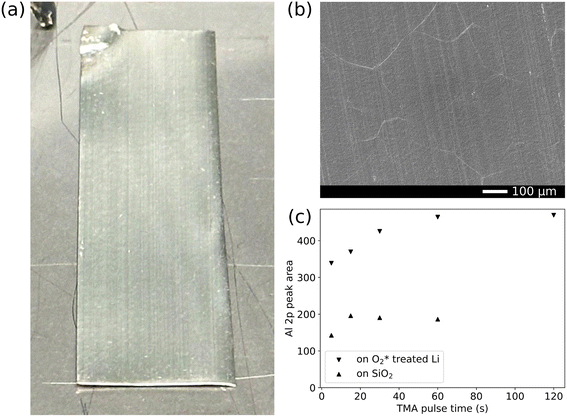
In contrast to the Ar* pretreatment, the surface of an  treated lithium foil exposed to TMA remained visually metallic, no black spots were visible, even after 120 s of TMA exposure (Fig. 6(a)). With SEM, a smooth surface remained (Fig. 6(b)) and a low Al signal was measured with EDX.
treated lithium foil exposed to TMA remained visually metallic, no black spots were visible, even after 120 s of TMA exposure (Fig. 6(a)). With SEM, a smooth surface remained (Fig. 6(b)) and a low Al signal was measured with EDX.
From in vacuo XPS measurements, we were able to determine the saturation behavior of TMA and  during the first ALD cycle. Fig. 6(c) and S7 show that saturated ALD-like growth is observed for TMA after 60 s of exposure. The data points in Fig. 6(c) were obtained from the peak areas of high-resolution Al 2p scans in Fig. S7. The Al 2p peak after 60 s is relatively small, well below the maximum intensity expected from the photoelectron information depth, as shown in Fig. 7, indicating that the observed saturation arises from self-limiting surface reactions rather than signal attenuation due to layer thickness. Although saturating, the measured Al 2p peak area for TMA exposure on Li is higher than that on Si, suggesting more Al material is incorporated after a single pulse and hinting at a non-conventional reaction mechanism.
during the first ALD cycle. Fig. 6(c) and S7 show that saturated ALD-like growth is observed for TMA after 60 s of exposure. The data points in Fig. 6(c) were obtained from the peak areas of high-resolution Al 2p scans in Fig. S7. The Al 2p peak after 60 s is relatively small, well below the maximum intensity expected from the photoelectron information depth, as shown in Fig. 7, indicating that the observed saturation arises from self-limiting surface reactions rather than signal attenuation due to layer thickness. Although saturating, the measured Al 2p peak area for TMA exposure on Li is higher than that on Si, suggesting more Al material is incorporated after a single pulse and hinting at a non-conventional reaction mechanism.
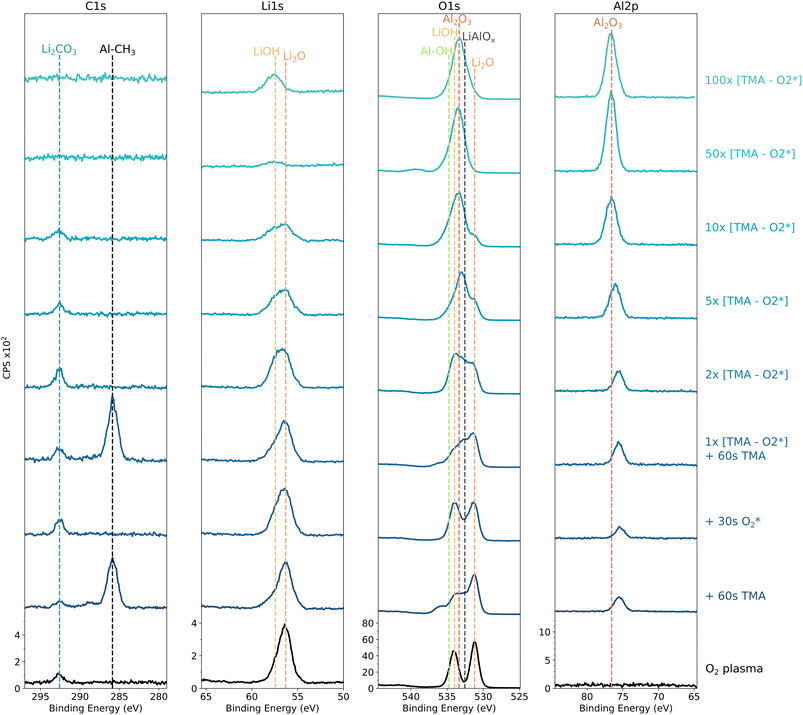
After exposing the  plasma treated lithium to 60 s of TMA, the LiOH (534 eV) and Li2O (531.2 eV) components in the O 1s spectrum decrease in intensity, indicating partial consumption of the surface –OH groups by TMA (Fig. 7). Expected from this reaction is an Al–CH3 terminated surface,34 which is confirmed by the presence of an Al 2p peak at 75.5 eV and the C 1s peak at 285.9 eV. Furthermore, a third peak appears in the O 1s spectrum at ∼ 532.6 eV, likely due to Al–O bonds that have formed on the surface. However, the binding energy does not exactly correspond with reported Al2O3 binding energy values.34,38 The O 1s component at 532.6 eV is therefore more likely related to an LiAlOx layer, which has been suggested by others,13,19,22 as it is found at lower binding energies than Al2O3, however, without consistent peak labeling through the different reports. A small shoulder is observed in the O 1s spectrum at higher binding energies (536 eV) and is further discussed in Fig. S8. The initial reaction between TMA and the
plasma treated lithium to 60 s of TMA, the LiOH (534 eV) and Li2O (531.2 eV) components in the O 1s spectrum decrease in intensity, indicating partial consumption of the surface –OH groups by TMA (Fig. 7). Expected from this reaction is an Al–CH3 terminated surface,34 which is confirmed by the presence of an Al 2p peak at 75.5 eV and the C 1s peak at 285.9 eV. Furthermore, a third peak appears in the O 1s spectrum at ∼ 532.6 eV, likely due to Al–O bonds that have formed on the surface. However, the binding energy does not exactly correspond with reported Al2O3 binding energy values.34,38 The O 1s component at 532.6 eV is therefore more likely related to an LiAlOx layer, which has been suggested by others,13,19,22 as it is found at lower binding energies than Al2O3, however, without consistent peak labeling through the different reports. A small shoulder is observed in the O 1s spectrum at higher binding energies (536 eV) and is further discussed in Fig. S8. The initial reaction between TMA and the  pretreated surface was also investigated at substrate temperatures of 75 and 130 °C. However, no significant differences in spectra were observed and therefore, the substrate temperature was kept at 100 °C to study further growth.
pretreated surface was also investigated at substrate temperatures of 75 and 130 °C. However, no significant differences in spectra were observed and therefore, the substrate temperature was kept at 100 °C to study further growth.
To complete the first ALD cycle, the surface was exposed to  . High-resolution XPS scans in Fig. S7 show that
. High-resolution XPS scans in Fig. S7 show that  saturation is reached after 30 s for all spectra. Therefore, saturated conditions of 60 s TMA and 30 s
saturation is reached after 30 s for all spectra. Therefore, saturated conditions of 60 s TMA and 30 s  are used for further depositions. After the 30 s
are used for further depositions. After the 30 s  exposure, an increase in LiOH O 1s signal is observed, also clear from broadening of the Li 1s peak due to the LiOH component at 57.4 eV, as shown in Fig. 7. Furthermore,
exposure, an increase in LiOH O 1s signal is observed, also clear from broadening of the Li 1s peak due to the LiOH component at 57.4 eV, as shown in Fig. 7. Furthermore,  efficiently removes the methyl ligands through combustion, however, a small amount of Li2CO3 is detected, probably from limited CO2 redeposition.32,33
efficiently removes the methyl ligands through combustion, however, a small amount of Li2CO3 is detected, probably from limited CO2 redeposition.32,33
After the second cycle, the Li2O signal further decreases while the LiOH intensity remains nearly unchanged, indicating that either the lithium is able to diffuse through the thin surface layer to interact with the energetic plasma or that the thin layer is not yet continuous. After 5–10 cycles, these components become smaller. Furthermore, the Al 2p peak progressively shifts to higher binding energies, eventually remaining at the peak position of 76.6 eV after 10 cycles. A similar evolution is observed for the O 1s spectra, where the main peak shifts to 533.4 eV. The binding energy difference between these Al 2p and O 1s components (456.8 eV) closely matches reported values for Al2O3,34,38 confirming the formation of an Al2O3 surface layer and confirming that the initial O 1s peak at 532.6 eV could be related to LiAlOx.
Although the Li 1s signal decreases with increasing ALD cycle, the signal reappears after 100 cycles, implying that lithium is able to diffuse through relatively thick Al2O3 films over extended timescales. Another reason could be due to cracking of the layer or pinhole formation, however, this was not obvious from SEM images (Fig. S9). Compared to 50 cycles, the O 1s peak broadens, suggesting a mixture of lithiated and non-lithiated Al2O3. Even though Kozen et al.11 reported no Li 1s signal after ‘14 nm’ of Al2O3, more recent work shows a clear lithium signal for more than 100 ALD cycles.19,22
From these XPS results, it is clear that a (lithiated) Al2O3 thin film can be grown in a much more controlled way when O2 plasma is used for pretreatment. SEM imaging (Fig. S9) shows that after 50 and 100 cycles of TMA- , the surface remains smooth and without change in morphology. Furthermore, EDX measurements confirm that the Al signal is comparable to what is expected on Si (Fig. S10). Even though more material is getting incorporated in the initial cycle, this excess uptake is hypothesized to be associated with the formation of a thin LiAlOx interlayer, after which the process transitions to a stable, ALD-like growth of Al2O3.13
, the surface remains smooth and without change in morphology. Furthermore, EDX measurements confirm that the Al signal is comparable to what is expected on Si (Fig. S10). Even though more material is getting incorporated in the initial cycle, this excess uptake is hypothesized to be associated with the formation of a thin LiAlOx interlayer, after which the process transitions to a stable, ALD-like growth of Al2O3.13
3.5 Importance of the LiOH component
The above findings reveal a clear difference between the two plasma pretreatments. While TMA exposure on an Ar* pretreated surface leads to uncontrolled TMA decomposition, clear from the continuous growth and substantial Al and C uptake, the pretreatment results in a more stable surface that supports ALD-like Al2O3 growth. The main compositional difference between these two surfaces is the presence of LiOH, which is only observed after
pretreatment results in a more stable surface that supports ALD-like Al2O3 growth. The main compositional difference between these two surfaces is the presence of LiOH, which is only observed after  treatment. To determine whether this LiOH component indeed governs the surface reactivity towards TMA, rather than other plasma-related effects, an Ar* treated surface was dosed with 30 s H2O at a pressure of 10−3 mbar, forming LiOH according to:
treatment. To determine whether this LiOH component indeed governs the surface reactivity towards TMA, rather than other plasma-related effects, an Ar* treated surface was dosed with 30 s H2O at a pressure of 10−3 mbar, forming LiOH according to:| Li2O + H2O → 2LiOH |
This should result in a similar surface composition to that obtained after an  pretreatment (i.e. LiOH/Li2O). As shown in Fig. 8, the Li2O surface is indeed partially converted to LiOH after H2O exposure, clear from the additional O 1s peak at 534 eV and the broadening of the Li 1s peak. When this surface is subsequently exposed to TMA, the resulting XPS spectra are very similar to TMA exposure after an
pretreatment (i.e. LiOH/Li2O). As shown in Fig. 8, the Li2O surface is indeed partially converted to LiOH after H2O exposure, clear from the additional O 1s peak at 534 eV and the broadening of the Li 1s peak. When this surface is subsequently exposed to TMA, the resulting XPS spectra are very similar to TMA exposure after an  treatment. A self-limiting reaction occurs, with Al–CH3, Li2O and LiAlOx components observed at the surface.
treatment. A self-limiting reaction occurs, with Al–CH3, Li2O and LiAlOx components observed at the surface.
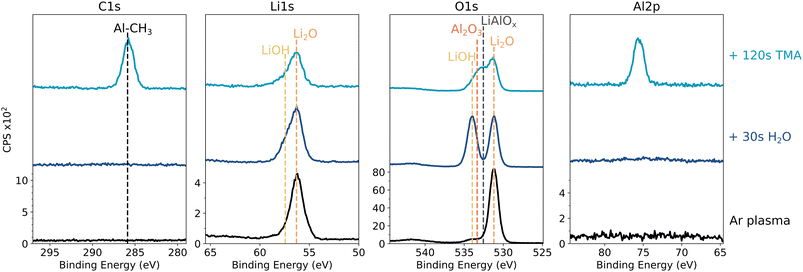 | ||
| Fig. 8 (a) In vacuo XPS of an Ar* treated Li foil exposed to 30 s of H2O and 120 s of TMA sequentially. Survey spectra can be found in Fig. S11. | ||
Visually, the foil remained shiny without black spots and SEM imaging revealed no noticeable morphological changes (Fig. 9).
 | ||
| Fig. 9 (a) Visual and (b) SEM image of an Ar* treated Li foil exposed to 30 s of H2O and 120 s of TMA sequentially. | ||
This additional result demonstrates that the presence of LiOH on the surface is critical for enabling ALD-like reactivity with TMA. Based on peak fitting of the O 1s signal, a LiOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Li2O ratio of approximately 30
Li2O ratio of approximately 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 is already sufficient to suppress TMA decomposition (Fig. S12). Analogous to surface Si–OH groups on a conventional Si wafer,39 the LiOH groups act as Lewis bases that interact with the electron-deficient Al center of TMA. This results in a ligand exchange in which a proton from LiOH is transferred to a methyl group of TMA, releasing methane as a byproduct and forming (Li-)O–Al bonds.
70 is already sufficient to suppress TMA decomposition (Fig. S12). Analogous to surface Si–OH groups on a conventional Si wafer,39 the LiOH groups act as Lewis bases that interact with the electron-deficient Al center of TMA. This results in a ligand exchange in which a proton from LiOH is transferred to a methyl group of TMA, releasing methane as a byproduct and forming (Li-)O–Al bonds.
Although H2O dosing on an Ar*-treated lithium can lead to self-limiting reactions with TMA, the reaction of H2O with metallic lithium itself is not self-limiting (Fig. S13). Continued reactions occur during H2O exposure,13,25 which makes reproducibility of the initial surface state more complicated. An  pretreatment is therefore able to provide a more controlled approach to obtain a LiOH surface and achieve subsequent ALD-like growth.
pretreatment is therefore able to provide a more controlled approach to obtain a LiOH surface and achieve subsequent ALD-like growth.
Using the Pilling–Bedworth ratios of the different components present at the surface after pretreatment, it is possible to explain the difference in reactivity towards the TMA precursor. Since LiOH and Li2CO3 exhibit PBR values of 1.26 and 1.35, respectively, it is expected that both materials are therefore able to form a continuous, passivating layer. On the contrary, the Li2O surface layer has a lower PBR value of 0.57, which is consistent with the possibility that the layer contains potential cracks or voids at the nanoscale, resulting in uncontrolled TMA decomposition due to direct interaction between TMA and lithium. However, the formation of a LiOH-terminated surface by an  pretreatment makes self-limiting ALD-like growth possible.
pretreatment makes self-limiting ALD-like growth possible.
Furthermore, this could clarify the discrepancy observed between our results and those reported by Kang et al.20,23 for TMA exposure on as-received lithium foil. In our case, the native oxide layer was largely converted into Li2CO3, with only a minor Li2O contribution remaining. Although not discussed by Kang et al.,20,23 their lithium foil may have contained a higher fraction of unconverted Li2O at the surface, thus exhibiting a different reactivity toward TMA.
4 Outlook
Although the main focus of this work is understanding how the lithium surface governs the initial TMA dosing, our findings have potential implications for the protection of reactive metal anodes.Al2O3 coatings deposited by ALD have repeatedly been shown to improve cycling stability of lithium metal anodes.11–13,19,22 However, in many cases, the initial lithium surface state was not controlled or explicitly reported. Since lithium plating and stripping are highly sensitive to the interfacial chemistry and its properties, variations in the initial surface state may contribute to differences in reported electrochemical performance. Our results imply that TMA exposure on as-received lithium can induce partial decomposition rather than homogeneous self-limiting reactions. When shorter precursor pulses, i.e. microdoses, are used, as done so in previous reports, decomposition may occur only locally and for a very short time, resulting in smaller nucleation sites. As a consequence, the decomposition is less obvious due to the minimal exposure time. This is supported by the results of Chen et al.,13,23 where non-self-limiting microdoses of TMA on untreated lithium resulted in significantly higher uptake of deposited material compared to the eventual steady-state growth. As a result, continuous TMA-H2O/  cycles may sustain growth at these sites or gradually cover them, eventually yielding an apparent uniform Al2O3 film, even though the initial growth mechanism deviated from ideal ALD behavior.
cycles may sustain growth at these sites or gradually cover them, eventually yielding an apparent uniform Al2O3 film, even though the initial growth mechanism deviated from ideal ALD behavior.
At the same time, previous work by Kang et al.20,23 has shown that TMA decomposition forming a Li–Al–C layer can also result in promising cycling stability, provided the TMA exposure time is limited in extent. The improved cycling stability was attributed to the formation of a multilayered interphase with distinct chemical and structural properties. However, the observed decomposition behavior depended strongly on the freshness of the lithium surface, as the reaction was suppressed when a thicker native layer was present.
The present work demonstrates that the reaction pathway can be deliberately selected through plasma pretreatment. By employing  , self-limiting, layer-by-layer growth of (lithiated) Al2O3 can be achieved already from the first cycle. In contrast, Ar* pretreatment enables controlled removal of the native layer and reproducible initiation of the Li–Al–C formation. By providing well-defined starting surfaces and the ability to control the reaction pathway, the obtained results may lead to improved consistency of electrochemical performance and further optimization of protective coatings for highly reactive metal anodes.
, self-limiting, layer-by-layer growth of (lithiated) Al2O3 can be achieved already from the first cycle. In contrast, Ar* pretreatment enables controlled removal of the native layer and reproducible initiation of the Li–Al–C formation. By providing well-defined starting surfaces and the ability to control the reaction pathway, the obtained results may lead to improved consistency of electrochemical performance and further optimization of protective coatings for highly reactive metal anodes.
5 Conclusion
In this work, a well-known ALD process, TMA and O2 plasma, was directly deposited on metallic lithium and the growth mechanism was studied in detail for the first time. Deposition on as-received lithium, as commonly reported in ALD literature, was found to result in uncontrolled growth. The film thickness after 100 cycles not only exceeded that obtained during conventional ALD on SiO2, but it was also not consistently reproduced. To obtain more control over the deposition, plasma pretreatments in vacuum were applied to establish well-defined and reproducible starting surfaces. After Ar plasma treatment, the native surface layer was completely removed, however, immediate formation of Li2O is inevitable due to the extreme reactivity of lithium, even under UHV conditions. Subsequent exposure to TMA resulted in continuous precursor decomposition. After only 5 s of TMA dosing, the lithium foil became completely black and a thick Li–Al–C layer consisting of densely packed pillars was formed. After 30 s of exposure, the decomposition layer reached a thickness of approximately 2 µm. Based on the Pilling–Bedworth ratio for Li2O, it is hypothesized that the oxide layer formed after Ar plasma is porous, thereby allowing the lithium underneath to react with incoming TMA and driving continuous growth. When O2 plasma was used, a surface consisting of both LiOH and Li2O was obtained. The foil remained smooth and shiny even after extended TMA exposure, indicating that a more ALD-like, self-limiting growth regime was achieved. The LiOH layer is proposed to moderate lithium's reactivity by providing well-defined sites for reaction with TMA. Self-limiting behavior was observed for TMA and O2 plasma, although longer precursor dosing times were needed compared to saturation on SiO2. Initial exposure resulted in the formation of a LiAlOx layer, but with increasing number of ALD cycles, a shift in XPS signal to (lithiated) Al2O3 was observed. Overall, the results presented in this work demonstrate that careful control of the initial lithium surface is essential for achieving reproducible ALD behavior. These findings underline the importance of critically considering the underlying reaction mechanisms, which differ greatly from these on conventional SiO2 substrates. Reporting and standardizing starting surface conditions is strongly encouraged, as it has a significant influence on film growth and could facilitate future research in this field.Conflicts of interest
There are no conflicts to declare.Data availability
All data needed for interpretation of this work is available in the manuscript and supplementary information (SI). Supplementary information: XPS spectra showing the influence of pressure; XPS survey spectra of the initial surface states; Angle-resolved XPS spectra for as-received, Ar* and O2* treated lithium foil; EDX spectra of TMA exposure on as-received lithium foil; XPS survey spectra of TMA exposure on Ar* treated lithium foil at different temperatures; EDX linescan of TMA exposure on Ar* treated lithium foil; XPS spectra of saturation behavior of TMA and O2 plasma steps on O2 plasma treated lithium foil; XPS survey spectra of TMA- cycles on
cycles on  treated Li and table with corresponding atomic percentages; SEM image of 100 TMA-
treated Li and table with corresponding atomic percentages; SEM image of 100 TMA- cycles on
cycles on  treated surface; EDX spectra of 100 TMA-
treated surface; EDX spectra of 100 TMA- cycles on
cycles on  treated lithium compared to Si; XPS survey spectra of H2O and TMA exposure on an Ar* treated lithium foil; XPS spectra of other controlled hydroxylation experiments; XPS spectra of different H2O exposure times on Ar* treated lithium foil with corresponding survey spectra; Pilling–Bedworth ratio calculations. See DOI: https://doi.org/10.1039/d6ta00751a.
treated lithium compared to Si; XPS survey spectra of H2O and TMA exposure on an Ar* treated lithium foil; XPS spectra of other controlled hydroxylation experiments; XPS spectra of different H2O exposure times on Ar* treated lithium foil with corresponding survey spectra; Pilling–Bedworth ratio calculations. See DOI: https://doi.org/10.1039/d6ta00751a.
Acknowledgements
This work was supported by the GOA project from the Special Research Fund - UGent (01G02124) and VLAIO (Vlaams Agentschap Innoveren & Ondernemen) within the SBO project MESH-BAT (HBC.2023.0669). The authors acknowledge the financial support from Fonds Wetenschappelijk Onderzoek - Vlaanderen (FWO) for the FWO medium-sized research infrastructure project L-SCAN I003222N and for providing Tippi Verhelle with an SB grant (1SH9024N) and Lowie Henderick with a junior postdoctoral fellowship (1254324N). Siebe Coessens holds a grant (HBC.2022.0157) which is supported by VLAIO. Furthermore, a special thank you goes out to Geert Rampelberg and Stefaan Broekaert for their technical assistance and their eye for detail while developing/building the reactor used in this work and Olivier Janssens for his assistance during SEM operation.References
- J.-M. Tarascon and M. Armand, Issues and challenges facing rechargeable lithium batteries, Nature, 2001, 414, 359–367 CrossRef CAS PubMed.
- J. Liu, et al., Pathways for practical high-energy long-cycling lithium metal batteries, Nat. Energy, 2019, 4, 180–186 CrossRef CAS.
- D. Aurbach, Review of selected electrode–solution interactions which determine the performance of Li and Li ion batteries, J. Power Sources, 2000, 89, 206–218 CrossRef CAS.
- A. J. Sanchez and N. P. Dasgupta, Lithium Metal Anodes: Advancing our Mechanistic Understanding of Cycling Phenomena in Liquid and Solid Electrolytes, J. Am. Chem. Soc., 2024, 146, 4282–4300 CrossRef CAS PubMed , PMID: 38335271..
- Z. Han, C. Zhang, Q. Lin, Y. Zhang, Y. Deng, J. Han, D. Wu, F. Kang, Q.-H. Yang and W. Lv, A Protective Layer for Lithium Metal Anode: Why and How, Small Methods, 2021, 5, 2001035 CrossRef CAS PubMed.
- Q. Wang, B. Liu, Y. Shen, J. Wu, Z. Zhao, C. Zhong and W. Hu, Confronting the Challenges in Lithium Anodes for Lithium Metal Batteries, Advanced Science, 2021, 8, 2101111 CrossRef CAS PubMed.
- K. N. Wood, M. Noked and N. P. Dasgupta, Lithium Metal Anodes: Toward an Improved Understanding of Coupled Morphological, Electrochemical, and Mechanical Behavior, ACS Energy Lett., 2017, 2, 664–672 CrossRef CAS.
- A. J. Sanchez, E. Kazyak, Y. Chen, K.-H. Chen, E. R. Pattison and N. P. Dasgupta, Plan-View Operando Video Microscopy of Li Metal Anodes: Identifying the Coupled Relationships among Nucleation, Morphology, and Reversibility, ACS Energy Lett., 2020, 5, 994–1004 CrossRef CAS.
- K. N. Wood, E. Kazyak, A. F. Chadwick, K.-H. Chen, J.-G. Zhang, K. Thornton and N. P. Dasgupta, Dendrites and Pits: Untangling the Complex Behavior of Lithium Metal Anodes through Operando Video Microscopy, ACS Cent. Sci., 2016, 2, 790–801 CrossRef CAS PubMed , PMID: 27924307..
- K.-H. Chen, K. N. Wood, E. Kazyak, W. S. LePage, A. L. Davis, A. J. Sanchez and N. P. Dasgupta, Dead lithium: mass transport effects on voltage, capacity, and failure of lithium metal anodes, J. Mater. Chem. A, 2017, 5, 11671–11681 RSC.
- A. C. Kozen, C.-F. Lin, A. J. Pearse, M. A. Schroeder, X. Han, L. Hu, S.-B. Lee, G. W. Rubloff and M. Noked, Next-Generation Lithium Metal Anode Engineering via Atomic Layer Deposition, ACS Nano, 2015, 9, 5884–5892 CrossRef CAS PubMed , PMID: 25970127..
- E. Kazyak, K. N. Wood and N. P. Dasgupta, Improved Cycle Life and Stability of Lithium Metal Anodes through Ultrathin Atomic Layer Deposition Surface Treatments, Chem. Mater., 2015, 27, 6457–6462 CrossRef CAS.
- L. Chen, J. G. Connell, A. Nie, Z. Huang, K. R. Zavadil, K. C. Klavetter, Y. Yuan, S. Sharifi-Asl, R. Shahbazian-Yassar, J. A. Libera, A. U. Mane and J. W. Elam, Lithium metal protected by atomic layer deposition metal oxide for high performance anodes, J. Mater. Chem. A, 2017, 5, 12297–12309 RSC.
- Y. Cao, X. Meng and J. W. Elam, Atomic Layer Deposition of LixAlyS Solid-State Electrolytes for Stabilizing Lithium-Metal Anodes, ChemElectroChem, 2016, 3, 858–863 CrossRef CAS.
- C.-F. Lin, A. C. Kozen, M. Noked, C. Liu and G. W. Rubloff, ALD Protection of Li-Metal Anode Surfaces – Quantifying and Preventing Chemical and Electrochemical Corrosion in Organic Solvent, Adv. Mater. Interfaces, 2016, 3, 1600426 CrossRef.
- L. Chen, K.-S. Chen, X. Chen, G. Ramirez, Z. Huang, N. R. Geise, H.-G. Steinrück, B. L. Fisher, R. Shahbazian-Yassar, M. F. Toney, M. C. Hersam and J. W. Elam, Novel ALD Chemistry Enabled Low-Temperature Synthesis of Lithium Fluoride Coatings for Durable Lithium Anodes, ACS Appl. Mater. Interfaces, 2018, 10, 26972–26981 CrossRef CAS PubMed , PMID: 29986134..
- P. K. Alaboina, S. Rodrigues, M. Rottmayer and S.-J. Cho, In Situ Dendrite Suppression Study of Nanolayer Encapsulated Li Metal Enabled by Zirconia Atomic Layer Deposition, ACS Appl. Mater. Interfaces, 2018, 10, 32801–32808 CrossRef CAS PubMed , PMID: 30157373..
- M. Wang, X. Cheng, T. Cao, J. Niu, R. Wu, X. Liu and Y. Zhang, Constructing ultrathin TiO2 protection layers via atomic layer deposition for stable lithium metal anode cycling, J. Alloys Compd., 2021, 865, 158748 CrossRef CAS.
- E. Jin, K. Tantratian, C. Zhao, A. Codirenzi, L. V. Goncharova, C. Wang, F. Yang, Y. Wang, P. Pirayesh, J. Guo, L. Chen, X. Sun and Y. Zhao, Ionic Conductive and Highly-Stable Interface for Alkali Metal Anodes, Small, 2022, 18, 2203045 CrossRef CAS PubMed.
- D. Kang, J. W. Elam and A. U. Mane, Formation of lithium-metal-carbon protecting layer and removal of lithium carbonate on lithium metal, US Patent, 2022 Search PubMed.
- B. Zhao, J. Li, M. Guillaume, V. Cremers, L. Henderick, J. Dendooven and C. Detavernier, Atomic layer deposition of yttrium oxide as a protective coating for lithium metal anodes, Dalton Trans., 2023, 52, 7302–7310 RSC.
- S. Pakseresht, V. Miikkulainen, F. A. Obrezkov, A. W. M. Al-Ogaili, J. Lahtinen and T. Kallio, Atomic layer deposition for protecting lithium metal anodes to high-voltage battery applications, J. Power Sources, 2025, 653, 237731 CrossRef CAS.
- K. Park, Y. L. Kim, J. G. Connell, C. Liu, H. Kim, S. Kim, C. T. Nguyen, A. U. Mane, D. Kang and J. W. Elam, Chemical Vapor Transformation of Lithium Metal: Mechanism and Enhanced Stability, ACS Appl. Mater. Interfaces, 2025, 17, 67763–67775 CrossRef CAS PubMed , PMID: 41342298..
- S.-K. Otto, Y. Moryson, T. Krauskopf, K. Peppler, J. Sann, J. Janek and A. Henss, In-Depth Characterization of Lithium-Metal Surfaces with XPS and ToF-SIMS: Toward Better Understanding of the Passivation Layer, Chem. Mater., 2021, 33, 859–867 CrossRef CAS.
- S.-K. Otto, T. Fuchs, Y. Moryson, C. Lerch, B. Mogwitz, J. Sann, J. Janek and A. Henss, Storage of Lithium Metal: The Role of the Native Passivation Layer for the Anode Interface Resistance in Solid State Batteries, ACS Appl. Energy Mater., 2021, 4, 12798–12807 CrossRef CAS.
- N. Fairley, V. Fernandez, M. Richard-Plouet, C. Guillot-Deudon, J. Walton, E. Smith, D. Flahaut, M. Greiner, M. Biesinger, S. Tougaard, D. Morgan and J. Baltrusaitis, Systematic and collaborative approach to problem solving using X-ray photoelectron spectroscopy, Appl. Surf. Sci. Adv., 2021, 5, 100112 CrossRef.
- K. N. Wood and G. Teeter, XPS on Li-Battery-Related Compounds: Analysis of Inorganic SEI Phases and a Methodology for Charge Correction, ACS Appl. Energy Mater., 2018, 1, 4493–4504 CrossRef CAS.
- JEOL Environmental Airlock system. https://www.jeolusa.com/RESOURCES/Electron-Optics/Documents-Downloads/environmental-airlock-for-jsm-it210-series-sem.
- U. Wietelmann, Surface-passivated lithium metal and method for the production thereof, US Pat., 2013 Search PubMed.
- B. Zhao, J. Li, M. Guillaume, J. Dendooven and C. Detavernier, In vacuo XPS investigation of surface engineering for lithium metal anodes with plasma treatment, J. Energy Chem., 2022, 66, 295–305 CrossRef CAS.
- F. She, A. Gao, P. Jiang, Y. Zhou, X. Zhang, M. Yang, L. Gong, J. Chen, X. Lu and F. Xie, Exploring the stability of lithium metal surface by X-ray photoelectron spectroscopy, Vacuum, 2023, 211, 111893 CrossRef CAS.
- V. R. Rai, V. Vandalon and S. Agarwal, Surface Reaction Mechanisms during Ozone and Oxygen Plasma Assisted Atomic Layer Deposition of Aluminum Oxide, Langmuir, 2010, 26, 13732–13735 CrossRef CAS PubMed , PMID: 20806961..
- N. Hornsveld, W. Kessels and M. Creatore, Mass Spectrometry Study of Li2CO3 Film Growth by Thermal and Plasma-Assisted Atomic Layer Deposition, J. Phys. Chem. C, 2019, 123, 4109–4115 CrossRef CAS.
- H.-E. Nieminen, M. Chundak, M. J. Heikkilä, P. R. Kärkkäinen, M. Vehkamäki, M. Putkonen and M. Ritala, In vacuo cluster tool for studying reaction mechanisms in atomic layer deposition and atomic layer etching processes, J. Vac. Sci. Technol., A, 2023, 41, 022401 CrossRef CAS.
- C. Hinnen, D. Imbert, J. Siffre and P. Marcus, An in situ XPS study of sputter-deposited aluminium thin films on graphite, Appl. Surf. Sci., 1994, 78, 219–231 CrossRef CAS.
- N. Pilling, The oxidation of metals at high temperature, J. Inst. Met., 1923, 29, 529–582 Search PubMed.
- Y. Qi, J. Liu, M. Feng, K. Tantratian, L. Chen, X. Xiao and A. K. Sachdev, From the Passivation Layer on Aluminum to Lithium Anode in Batteries, Metall. Mater. Trans. A, 2025, 56, 429–438 CrossRef CAS.
- O. Renault, L. G. Gosset, D. Rouchon and A. Ermolieff, Angle-resolved x-ray photoelectron spectroscopy of ultrathin Al2O3 films grown by atomic layer deposition, J. Vac. Sci. Technol., A, 2002, 20, 1867–1876 CrossRef CAS.
- R. L. Puurunen, Surface chemistry of atomic layer deposition: A case study for the trimethylaluminum/water process, J. Appl. Phys., 2005, 97, 121301 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |