 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Approaching the reactivity of anions in battery electrolytes via conceptual density functional theory
Ervin
Rems
 ab,
Matej
Huš
ab,
Matej
Huš
 *cd and
Robert
Dominko
*cd and
Robert
Dominko
 *abe
*abe
aDepartment of Materials Chemistry, National Institute of Chemistry, Hajdrihova 19, SI-1001 Ljubljana, Slovenia. E-mail: robert.dominko@ki.si
bFaculty of Chemistry and Chemical Technology, University of Ljubljana, Večna pot 113, SI-1000 Ljubljana, Slovenia
cDepartment of Catalysis and Chemical Reaction Engineering, National Institute of Chemistry, Hajdrihova 19, SI-1001 Ljubljana, Slovenia. E-mail: matej.hus@ki.si
dAssociation for Technical Culture of Slovenia (ZOTKS), Zaloška 65, SI-1000 Ljubljana, Slovenia
eALISTORE - European Research Institute, FR CNRS 3104, 80039 Amiens Cedex, France
First published on 19th February 2026
Abstract
The practical deployment of batteries with superior energy and power densities is, among other factors, prohibited by the lack of suitable electrolyte formulations. While first-principles design promises accelerated discovery of practical battery electrolytes, its high computational cost limits the chemical space that can be analysed through screening. Herein, we show a computationally inexpensive approach, using descriptors within the framework of conceptual density functional theory, that can provide valuable insights into the reactivity of battery electrolyte components. By focusing on anions in liquid electrolytes for which abundant experimental data is available for benchmarking, we explore several descriptors against key experimental observables for a series of salts (LiPF6, LiBF4, LiTFSI, LiDFOB, LiTDI). We show that the descriptors evaluated for single species can provide insights consistent with those obtained from experiments or more complex models, while maintaining a favourable computational cost. This establishes conceptual density functional theory as a promising tool for high-throughput screening of electrolyte components (salts, solvents, diluents, additives).
1. Introduction
Lithium-ion batteries have revolutionized energy storage, enabling the rapid expansion of portable electronics and electric vehicles.1 However, for further electrification of society, an additional increase in the energy density of Li batteries is crucial, expanding their range of applications and optimizing the cost.2 Three main directions to increase Li battery energy density are (i) decreasing the battery mass by replacing a negative graphite electrode with metallic Li,3 (ii) increasing battery output voltage by employing high-voltage positive electrode materials,4 and (iii) the use of positive electrode materials with high specific capacity.5 A major challenge of these approaches is their incompatibility with conventional Li-ion battery electrolytes, mainly due to Li dendrite growth and electrolyte degradation.6 New electrolytes are needed for advancing application-relevant battery technologies, which can be proposed by systematic screening and rational design.7Leveraging in silico screening approaches for battery electrolyte discovery is a promising avenue to accelerate the search across the extensive chemical space of electrolyte components.8 Hitherto reported studies typically rely on density functional theory (DFT) calculations,8,9 classical molecular dynamics (MD) simulations,10,11 machine learning of experimental data,12,13 or a combination of these methods.14–16 However, these approaches come with important drawbacks: classical MD simulations depend on the availability of experimentally validated force–field parameters17 with limited transferability, reducing the predictive power for experimentally unexplored formulations. Similarly, the performance of machine learning models remains limited to analogous formulations, performing poorly for compositions that differ significantly from the training set.18,19 While DFT can be readily applied to new and experimentally unexplored chemical systems, the computational cost of quantum chemical calculations presents a practical bottleneck that severely limits the searchable chemical space. Indeed, advances in DFT-based approaches that would allow screening more diverse chemical spaces and, at the same time, yield experimentally relevant descriptors have been identified as a priority research direction in computational electrolyte design.20
As a promising remedy, we turn to conceptual DFT, which focuses on the extraction of chemically relevant concepts and principles from DFT.21,22 While conceptual DFT has long established itself as a standard methodology in theoretical organic chemistry,23 it has received surprisingly limited attention in the context of battery electrolytes,24–29 and has not been systematically compared to experimental observables across a range of chemical compositions.
Here, we show that descriptors within the framework of conceptual DFT can provide valuable insights into the (electro)chemical properties of battery electrolytes at a very moderate computational cost. As an example, we focus on anions as they strongly influence electrolyte structure, transport properties, and electrochemical behaviour.30–33 We assess the effect of anions on the electrochemical stability of the electrolyte, the mechanism of their oxidative/reductive anion decomposition, and the interaction of anions with nucleophiles/electrophiles, by focusing on electrolytes based on LiPF6, LiBF4, lithium bis(trifluoromethylsulfonyl)azanide (LiTFSI), lithium difluoro(oxalato)borate (LiDFOB) and lithium 2-trifluoromethyl-4,5-dicyanoimidazole (LiTDI) salts (structural formulae of the corresponding anions are shown in Fig. 1a). This set of anions was chosen to capture the chemical diversity (size, charge delocalization, functional groups) of battery electrolyte salts, while remaining small enough to allow direct comparison with previous experimental studies and costly high-level calculations rather than screen a vast theoretical configurational space. In essence, this is a conceptual study rather than a screening endeavor.
 | ||
Fig. 1 Stability of electrolyte components explored in this work. (a) Structural formulae of PF−6, BF−4, bis(trifluoromethanesulfonyl)azanide (TFSI−), difluoro(oxalato)borate (DFOB−), 4,5-dicyano-2-(trifluoromethyl)imidazole-1-ide (TDI−) salt anions, ethylene carbonate (EC) and ethyl methyl carbonate (EMC) solvent molecules, and H2O and HF molecules as common impurities. (b) Experimental reduction (left) and oxidation (right) cyclic voltammetry curves of various 1 M salt in EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EMC (3 EMC (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 wt%) solvent mixtures. (This panel is reprinted from Delp et al.48 with permission from Elsevier.). (c) Chemical hardness of the investigated anions, without accounting for solvent. (d) Stability windows of investigated anions, solvents, and impurities. Oxidation limit is determined as the ionization potential of the corresponding species, whereas reduction limit is determined as the electronic affinity of the species-Li+ cluster. 7 wt%) solvent mixtures. (This panel is reprinted from Delp et al.48 with permission from Elsevier.). (c) Chemical hardness of the investigated anions, without accounting for solvent. (d) Stability windows of investigated anions, solvents, and impurities. Oxidation limit is determined as the ionization potential of the corresponding species, whereas reduction limit is determined as the electronic affinity of the species-Li+ cluster. | ||
2. Computational method
First-principles calculations were performed at the DFT level, employing a long-range- and dispersion-corrected hybrid functional ωB97X-D.34 Molecular geometries were optimized within the def2-TZVPD basis set,35,36 while the def2-QZVPD basis set36,37 was employed for single-point calculations. The DFT calculations were performed using Gaussian 16 (ES64L-G16RevC.02).38 Basis sets were obtained through Basis Set Exchange (BSE).39 Pymatgen40 and Multiwfn41 were used to handle input/output files. Visual Molecular Dynamics (VMD)42 was used for visualization. The convergence criterion for geometry optimization was set using the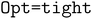 keyword to apply the following convergence thresholds: maximum force < 1.5 × 10−5Eha0−1, root mean square (RMS) force < 1.0 × 10−5Eha−10, maximum step < 6.0 × 10−5 Å, and RMS step < 4.0·10−5 Å. Vibrational analysis was performed to verify the minima of the potential energy surface through the absence of vibrational modes with imaginary frequencies. In models that include implicit solvation, the EC
keyword to apply the following convergence thresholds: maximum force < 1.5 × 10−5Eha0−1, root mean square (RMS) force < 1.0 × 10−5Eha−10, maximum step < 6.0 × 10−5 Å, and RMS step < 4.0·10−5 Å. Vibrational analysis was performed to verify the minima of the potential energy surface through the absence of vibrational modes with imaginary frequencies. In models that include implicit solvation, the EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EMC 3
EMC 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 blend (ε = 18.5)43 was mimicked with the solvent parameters of acetone (ε = 20.5), which is deemed an adequate approximation.44,45 Li+-anion association strength is reported as the opposite of the Li+-anion binding energy ΔE. In the calculation of bond dissociation energies, the geometries of all species were optimized.
7 blend (ε = 18.5)43 was mimicked with the solvent parameters of acetone (ε = 20.5), which is deemed an adequate approximation.44,45 Li+-anion association strength is reported as the opposite of the Li+-anion binding energy ΔE. In the calculation of bond dissociation energies, the geometries of all species were optimized.
3. Results and discussion
A key parameter limiting the output voltage of a battery cell is the electrochemical stability window (ESW) of the electrolyte,46i.e., the range in volts between its limits of oxidative and reductive decomposition. Therefore, we first focus on the effect of the Li salt by comparing the ESW of 1 M solutions of the five salts in ethylene carbonate (EC): ethyl methyl carbonate (EMC) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 weight ratio), a typical solvent blend and salt concentration used in commercial Li-ion batteries.47 Delp et al. experimentally investigated the ESWs of these systems by cyclic voltammetry (CV) measurements using a glassy carbon working electrode (Fig. 1b).48 Experimentally, ESWs of electrolytes (disregarding small irreversible peaks that likely originate from impurities) widen, as follows: LiTDI < LiDFOB < LiTFSI < LiBF4 < LiPF6.
7 weight ratio), a typical solvent blend and salt concentration used in commercial Li-ion batteries.47 Delp et al. experimentally investigated the ESWs of these systems by cyclic voltammetry (CV) measurements using a glassy carbon working electrode (Fig. 1b).48 Experimentally, ESWs of electrolytes (disregarding small irreversible peaks that likely originate from impurities) widen, as follows: LiTDI < LiDFOB < LiTFSI < LiBF4 < LiPF6.
We begin by exploring a simple descriptor of global anion stability, the absolute hardness η.49 It is defined as the second derivative of energy with respect to the number of electrons:
 | (1) |
The experimental determination of ESW is limited by the potentials at which reduction and oxidation of the electrolyte occur. While η provides a valuable approximation, it is more rigorous to address oxidation and reduction separately. To shed light on the species and effects limiting the ESW, we separately investigate the five anions, both solvent molecules, and the two common impurities: H2O and HF. Trace amounts of both impurities are known to heavily impact the electrolyte (electro)chemistry.54–56 Trace H2O is always present in organic solvents, while HF is formed via the hydrolysis of PF−6 anions.57 We approximate the stability window based on the vertical ionization potential of the species and the vertical electronic affinity of the Li+-species pair:
 | (2) |
 | (3) |
The stability windows in the gas phase are shown in Fig. 1d. The trend of anion IP+Li/Li agrees with the experimentally determined oxidative stability of the corresponding electrolytes. On the other hand, anion EA+Li/Li has values well below those of the solvent or the impurities. This suggests anion-limited oxidative stability and impurity- and solvent-limited reductive stability.
Thus, we focus on anion and solvent oxidation by comparing the vertical IP in the gas phase with the adiabatic IP in the implicit solvent (Fig. 2a). The anion ordering by the experimental oxidative stability (Fig. 1b) aligns well with the ordering of vertical IP in the gas phase. Still, these results cannot explain the comparable experimental oxidative stability of LiBF4 and LiPF6 electrolytes. Considering adiabatic IP in the implicit solvent, the IP of EMC and EC drops below those of BF−4 and PF−6, pointing to solvent-limited oxidative stability in the case of LiBF4 and LiPF6. This behavior agrees with an earlier report on reduced, solvent-dependent oxidative stability of systems with small difference between IP of anion and solvent60 – as for BF−4 and PF−6 in the present case.
Similarly, we compare EA (electron affinity) of Li+-solvent and Li+-impurity pairs in Fig. 2b EAs of Li+-anion pairs are not shown here as they are substantially lower (Fig. 1d). Despite significant quantitative differences between vertical (gas phase) and adiabatic (implicit solvent) EA, the EA increases, as follows: EMC ≈ EC < H2O < HF. The EC < H2O < HF ordering agrees with experimental reduction potentials.54,55 For LiPF6-based electrolytes, this suggests that anode passivation proceeds via sequential reduction of HF and H2O impurities, followed by solvent reduction. This could explain the extended reductive stability of the LiPF6-based electrolyte and agrees with the seminal work of Peled et al., proposing a LiF-dominated inner solid electrolyte interphase (SEI), Li2O/LiOH-dominated intermediate SEI, and organic outer SEI.61
Let us explore other salts beyond LiPF6. The EA for Li+-anion− species (Fig. 1d) cannot explain the differences in experimental reductive stability (Fig. 1b). Interestingly, the reductive stability does correlate with the anion association strength (Fig. 2c, SI Discussion 1 and Table S2): the stronger the ionic association, the stronger the reductive stability. We hypothesize this could be related to a higher population of contact ion pairs and ionic aggregates in electrolytes with strongly associating anions, given the tendency for ion-pair formation in EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EMC-based electrolytes.62 This explanation is analogous to the reasoning behind the improved reductive stability of super-concentrated electrolytes, i.e., the significant population of contact ion pairs and aggregates tends to shift the reduction from solvent molecules to anions,63 which have lower EA.Anions not only contribute to the electrolyte ESW, but can also modulate the composition of the SEI and cathode electrolyte interphase (CEI).30,32,64 A rational design of anion-derived interphases depends on the understanding of the underlying anion decomposition mechanisms. This motivates us to explore how the electronic structure of anions responds to the extraction and addition of electrons, mimicking oxidation and reduction, respectively. This response can be characterized via the Fukui function
EMC-based electrolytes.62 This explanation is analogous to the reasoning behind the improved reductive stability of super-concentrated electrolytes, i.e., the significant population of contact ion pairs and aggregates tends to shift the reduction from solvent molecules to anions,63 which have lower EA.Anions not only contribute to the electrolyte ESW, but can also modulate the composition of the SEI and cathode electrolyte interphase (CEI).30,32,64 A rational design of anion-derived interphases depends on the understanding of the underlying anion decomposition mechanisms. This motivates us to explore how the electronic structure of anions responds to the extraction and addition of electrons, mimicking oxidation and reduction, respectively. This response can be characterized via the Fukui function  :65
:65
 | (4) |
 is the electron density. Given the discontinuity of
is the electron density. Given the discontinuity of  with respect to N,66 it is meaningful to define the oxidative Fukui function
with respect to N,66 it is meaningful to define the oxidative Fukui function  and the reductive Fukui function
and the reductive Fukui function  , and approximate them via finite differences:
, and approximate them via finite differences: | (5) |
 | (6) |
 ,
,  and
and  are electron densities of the anion, neutral radical and dianion radical, respectively.
are electron densities of the anion, neutral radical and dianion radical, respectively.
The oxidative/reductive Fukui functions for DFOB−, TFSI−, and TDI− anions are illustrated in Fig. 3a (isosurface values are provided in Table S3, while in Fig. S1 we separately show positive and negative isosurfaces). The Fukui functions are evaluated in the IEF-PCM implicit solvent, except for f+ of DFOB−, which is evaluated in the gas phase (the corresponding IEF-PCM f+ is shown in Fig. S2). The Fukui functions of BF−4 and PF−6 are not reported due to the degeneracy of their frontier molecular orbitals (see SI Discussion 2).
We first focus on the electrochemical decomposition of DFOB−, an important additive in high-voltage Li-ion batteries.67 The oxidative Fukui function f− changes its sign along the B–O bond, suggesting its cleavage under oxidizing conditions. Indeed, the cleavage of the B–O bond has been proposed several times68–71 as the initial stage of oxidative DFOB− decomposition, leading to a favorable passivation of the positive electrode via CEI formation.72 The characterization of reductive decomposition is experimentally more challenging, as it competes with the solvent reduction,72 which is typically more favoured (vide supra). However, recent ab initio molecular dynamics (AIMD) simulations identified the C–O(cycle) and B–F cleavage as drivers of reductive decomposition of DFOB− in a polyethylene oxide matrix on Li(100).73 This is in line with the reductive Fukui function f+, which points to the reductive C–O(cycle) cleavage. Henceforth, we refer to a chemical bond along which the oxidative or reductive Fukui function changes its sign as the bond of the oxidative Fukui inversion or the bond of the reductive Fukui inversion.
In TFSI−, the bonds of oxidative and reductive Fukui inversion are S–C and N–S, respectively. We complement these results by evaluating the bond-breaking energetics upon electron attachment/removal (Table S4), which support C–S-cleavage-driven oxidative TFSI− decomposition and reductive decomposition via breaking of the N–S (Fig. 3b). Previously, the oxidative decomposition was attributed to the S–C cleavage in an organic battery electrolyte74 and to the N–S cleavage in WiS.75 On the other hand, studies on the reduction of TFSI−-containing ionic liquids76,77 and ether-based battery electrolyte78 suggest a reductive decomposition via the N– S cleavage, while the C– F cleavage was proposed for the LiTFSI WiS electrolytes.75,79 The oxidative/reductive Fukui inversions adequately point to breaking-prone chemical bonds, as evidenced from the agreement with the bond-breaking energetics and most previous reports.
Bond dissociation energies for TDI− suggest the formation of fluorine radicals and fluoride anions under oxidative and reductive conditions, respectively (Table S5). The corresponding Fukui functions also point to the possibility of C–F bond breaking in both cases. We note that Fukui functions could also point to ring opening, while bond dissociation energetics suggest that this mechanism is less favorable. Previous reports have also reported the decomposition of TDI−via the cleavage of the C–F bond.80,81
Noteworthily, Fukui function may fail to predict the correct product under kinetically controlled ambident reactivity.82 In anionic systems with significant delocalization error, analytical evaluation of the Fukui function was shown to perform better than the finite-difference approximation.83 Overall, while Fukui functions do provide important insights into species response to oxidative/reductive perturbation, we believe they should be interpreted with care and complemented by other methods.Another important aspect of electrolyte reactivity is their tendency for speciation with nucleophilic and electrophilic species. For example, a nucleophilic attack of H+ on EC triggers a chemical cascade that compromises all battery components.84 Keeping the focus on the three anions, we evaluate them using the dual descriptor (DD)  ,85 a metric that allows the identification of nucleophilic and electrophilic sites.86 We evaluate it as the difference between the corresponding Fukui functions (in the gas phase):
,85 a metric that allows the identification of nucleophilic and electrophilic sites.86 We evaluate it as the difference between the corresponding Fukui functions (in the gas phase):
 | (7) |
 indicates a (more) electrophilic region, and a (more) negative
indicates a (more) electrophilic region, and a (more) negative  indicates a (more) nucleophilic region. Positive (red) and negative (blue) DD isosurfaces are shown in Fig. 4a (isosurface values are provided in Table S6), indicating electrophilic and nucleophilic regions, respectively.
indicates a (more) nucleophilic region. Positive (red) and negative (blue) DD isosurfaces are shown in Fig. 4a (isosurface values are provided in Table S6), indicating electrophilic and nucleophilic regions, respectively.
 | ||
| Fig. 4 Local nucleophilicity and electrophilicity of DFOB−, TFSI−, and TDI anions identified via dual descriptor. (a) Positive (red) and negative (blue) isosurfaces of the dual descriptor. Colour code for atoms is the same as in Fig. 3a. (b) Structural formulae of anions with atoms color based on the value of the condensed dual descriptor. | ||
In DFOB−, the boron atom appears strongly electrophilic, in agreement with the non-redox reactivity87,88 of DFOB− with nucleophilic components of SEI, leading to a ring opening via attack on the boron atom. On the other hand, carbonyl oxygen atoms are the most nucleophilic, aligning with ring opening, induced by coordination of the Li+ electrophile to carbonyl oxygen atoms.89
The nitrogen and sulphur atoms are nucleophilic and electrophilic centres of the TFSI− anion, respectively. The speciation of the TFSI− anion has been investigated via AIMD simulations for WiS electrolytes, revealing that the proton attaches to the nitrogen of TFSI−, forming a superacid HTFSI,90 in agreement with DD. An AIMD simulation of nucleophilic OH− attack on TFSI− in the gas phase environment showed that OH− preferentially attacks the sulphur atom.91 However, simulations that explicitly include water solvent molecules indicated a preferential nucleophilic substitution at the carbon atom, driven by steric effects91 that DD cannot capture. On the other hand, a nucleophilic attack of OH− on the sulphur atom of [MgTFSI]+ contact ion pair, explicitly solvated by diglyme, was also observed by AIMD, in line with DD.92
In TDI−, the values of DD are rather low. The most electrophilic region appears to be the carbon atom of the cyano group, while the cyclic carbon atom, to which –CN is bonded, appears the most nucleophilic. This does not agree with the experimentally observed coordination of Li+ and the proton of H2O to the nitrogen atoms of TDI− (cyano or cyclic).93,94 We probe nucleophilicity of TDI− sites by evaluating energetics for a model nucleophile (H+), confirming the cyclic N atom as the most favourable site (Fig. S3). While DD successfully identified the most nucleophilic site it failed for the most electrophilic site, likely due to a low magnitude of DD and small variation in DD across the anion, possibly related to the strong chemical and thermal stability of LiTDI-containing electrolytes.93,95
Projection of DD values to specific atoms can be more useful, especially in the context of high-throughput approaches. We achieve this by partitioning the species charge into CM5 partial charges,96 obtaining an atom-specific condensed dual descriptor (CDD). CDDs are illustrated in Fig. 4b, with the corresponding numerical values provided in Fig. S4. CDDs match the real-space DDs well and allow a direct comparison of reactive centres between the various anions: B of DFOB− stands out as a very strong electrophile, while N of TFSI− and carbonyl O of DFOB− are moderate nucleophiles. All other atoms exhibit significantly weaker electrophilicity/nucleophilicity.
To assess its predictive power, CDD is tested against the binding energies of anions with representative model species: electrophile H+ and nucleophile OH− (Table 1 and Fig. S3). We observe a linear correlation between the CDD maximum and H+ binding energy, despite small energy differences between TFSI− and DFOB− (Fig. S4). While CDD fails to predict a slightly more favourable OH− binding to TDI− compared to TFSI−, presumably due to the proximity of a strongly nucleophilic N, the strong nucleophilicity of the B atom of DFOB− with large CDD indeed leads to much stronger OH− binding to DFOB− compared to both TDI− and TFSI−.
4. Conclusion
In conclusion, we have shown that descriptors from the framework of conceptual DFT can be used to understand the behavior of anions in battery electrolytes, including some important aspects of their (electro)chemical reactivity. While some descriptors are too simplistic to accurately predict all observables, they capture the key trends remarkably well, making them suitable for quick screening of the extensive chemical space, e.g., for the initial layers of the computational funnel approach.97 In addition to anions, the descriptors discussed in this work can be used to study solvent, diluent, and electrolyte additive molecules. More broadly, this framework could also be applied to other systems, where anions play a key role, such as electrolytes for Li-mediated nitrogen reduction reaction98 or electrochemical CO2 reduction.99 We believe this work will motivate the community to utilize the variety of tools that conceptual DFT offers and accelerate the design of advanced electrolytes for electrochemical energy storage/conversion.Author contributions
E. R. conceptualized the project, performed the calculations, and wrote the initial draft. E. R. and M. H. interpreted the data. M. H. supervised the project. R. D. secured funding and managed the project. All authors reviewed, edited, and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Cartesian coordinates of optimized molecular geometries. See DOI: https://doi.org/10.1039/d6ta00383d.Acknowledgements
The research was funded by the Slovenian Research and Innovation Agency (ARIS) through core funding P2-0423 (E.R., R.D.) and P2-0152 (M.H.), and grants GC-0004 and J2-70085 (M.H.). The research was co-funded under the HyBReED project, supported by the European Union – NextGenerationEU. The authors acknowledge the HPC RIVR consortium and EuroHPC JU for funding this research by providing computing resources of the HPC system Vega at the Institute of Information Science, Maribor, Slovenia. The authors thank Alessandra Serva and Svit Menart for helpful discussions.References
- M. Armand and J.-M. Tarascon, Building better batteries, Nature, 2008, 451, 652–657 CrossRef CAS PubMed.
- J. B. Goodenough and K.-S. Park, The Li-ion rechargeable battery: A perspective, J. Am. Chem. Soc., 2013, 135, 1167–1176 CrossRef CAS PubMed.
- L. Yang, N. M. Hagh, J. Roy, E. Macciomei, J. R. Klein, U. Janakiraman and M. E. Fortier, Review—challenges and opportunities in lithium metal battery technology, J. Electrochem. Soc., 2024, 171, 060504 CrossRef CAS.
- J. Xiang, Y. Wei, Y. Zhong, Y. Yang, H. Cheng, L. Yuan, H. Xu and Y. Huang, Building practical high-voltage cathode materials for lithium-ion batteries, Adv. Mater., 2022, 34, 2200912 CrossRef CAS PubMed.
- L. Wang, Z. Wu, J. Zou, P. Gao, X. Niu, H. Li and L. Chen, Li-free cathode materials for high energy density lithium batteries, Joule, 2019, 3, 2086–2102 CrossRef CAS.
- X. Fan and C. Wang, High-voltage liquid electrolytes for Li batteries: progress and perspectives, Chem. Soc. Rev., 2021, 50, 10486–10566 RSC.
- M. Li, C. Wang, Z. Chen, K. Xu and J. Lu, New concepts in electrolytes, Chem. Rev., 2020, 120, 6783–6819 CrossRef CAS PubMed.
- L. Cheng, R. S. Assary, X. Qu, A. Jain, S. P. Ong, N. N. Rajput, K. Persson and L. A. Curtiss, Accelerating electrolyte discovery for energy storage with high-throughput screening, J. Phys. Chem. Lett., 2015, 6, 283–291 CrossRef CAS PubMed.
- X. Qu, A. Jain, N. N. Rajput, L. Cheng, Y. Zhang, S. P. Ong, M. Brafman, E. Maginn, L. A. Curtiss and K. A. Persson, The electrolyte genome project: A big data approach in battery materials discovery, Comput. Mater. Sci., 2015, 103, 56–67 CrossRef CAS.
- T. Mendez-Morales, Z. Li and M. Salanne, Computational screening of the physical properties of water-in-salt electrolytes, Batter. Supercaps, 2021, 4, 646–652 CrossRef CAS.
- Y. Ko, M. A. Baird, X. Peng, T. Ogunfunmi, Y.-W. Byeon, L. M. Klivansky, H. Kim, M. C. Scott, J. Chen, A. J. D'Angelo, J. Chen, S. Sripad, V. Viswanathan and B. A. Helms, Omics-enabled understanding of electric aircraft battery electrolytes, Joule, 2024, 8, 2393–2411 CrossRef CAS.
- S. C. Kim, S. T. Oyakhire, C. Athanitis, J. Wang, Z. Zhang, W. Zhang, D. T. Boyle, M. S. Kim, Z. Yu, X. Gao, T. Sogade, E. Wu, J. Qin, Z. Bao, S. F. Bent and Y. Cui, Data-driven electrolyte design for lithium metal anodes, Proc. Natl. Acad. Sci. U. S. A, 2023, 120, e2214357120 CrossRef CAS PubMed.
- M. Zohair, V. Sharma, E. A. Soares, K. Nguyen, M. Giammona, L. Sundberg, A. Tek, E. A. V. Vital and Y.-H. La, Chemical foundation model-guided design of high ionic conductivity electrolyte formulations, npj Comput. Mater., 2025, 11, 283 CrossRef CAS.
- R. Atwi and N. N. Rajput, Guiding maps of solvents for lithium-sulfur batteries via a computational data-driven approach, Patterns, 2023, 4, 100799 CrossRef CAS PubMed.
- D. Lu, R. Li, M. M. Rahman, P. Yu, L. Lv, S. Yang, Y. Huang, C. Sun, S. Zhang, H. Zhang, J. Zhang, X. Xiao, T. Deng, L. Fan, L. Chen, J. Wang, E. Hu, C. Wang and X. Fan, Ligand-channel-enabled ultrafast Li-ion conduction, Nature, 2024, 627, 101–107 CrossRef CAS PubMed.
- R. Kumar, M. C. Vu, P. Ma and C. V. Amanchukwu, Electrolytomics: A unified big data approach for electrolyte design and discovery, Chem. Mater., 2025, 37, 2720–2734 CrossRef CAS.
- J. Ruža, P. Leon, K. Jun, J. Johnson, Y. Shao-Horn and R. Gómez-Bombarelli, Benchmarking classical molecular dynamics simulations for computational screening of lithium polymer electrolytes, Macromolecules, 2025, 58, 6732–6742 CrossRef.
- S. K. Kauwe, J. Graser, R. Murdock and T. D. Sparks, Can machine learning find extraordinary materials?, Comput. Mater. Sci., 2020, 174, 109498 CrossRef.
- S. Ju, J. You, G. Kim, Y. Park, H. An and S. Han, Application of pretrained universal machine-learning interatomic potential for physicochemical simulation of liquid electrolytes in Li-ion batteries, Digit. Discov., 2025, 4, 1544–1559 RSC.
- M. A. Makeev and N. N. Rajput, Computational screening of electrolyte materials: status quo and open problems, Curr. Opin. Chem. Eng., 2019, 23, 58–69 CrossRef.
- P. Geerlings, F. De Proft and W. Langenaeker, Conceptual density functional theory, Chem. Rev., 2003, 103, 1793–1874 CrossRef CAS PubMed.
- D. Chakraborty and P. K. Chattaraj, Conceptual density functional theory based electronic structure principles, Chem. Sci., 2021, 12, 6264–6279 RSC.
- L. R. Domingo, M. Ríos-Gutiérrez and P. Pérez, Applications of the conceptual density functional theory indices to organic chemistry reactivity, Molecules, 2016, 21, 748 CrossRef PubMed.
- E. Jónsson and P. Johansson, Electrochemical oxidation stability of anions for modern battery electrolytes: a CBS and DFT study, Phys. Chem. Chem. Phys., 2015, 17, 3697–3703 RSC.
- P. Jankowski, M. Poterała, N. Lindahl, W. Wieczorek and P. Johansson, Chemically soft solid electrolyte interphase forming additives for lithium-ion batteries, J. Mater. Chem. A, 2018, 6, 22609–22618 RSC.
- A. Kopač Lautar, A. Hagopian and J.-S. Filhol, Modeling interfacial electrochemistry: concepts and tools, Phys. Chem. Chem. Phys., 2020, 22, 10569–10580 RSC.
- A. Kopač Lautar, J. Bitenc, R. Dominko and J.-S. Filhol, Building Ab Initio interface Pourbaix diagrams to investigate electrolyte stability in the electrochemical double layer: Application to magnesium batteries, ACS Appl. Mater. Interfaces, 2021, 13, 8263–8273 CrossRef PubMed.
- L. H. B. Nguyen and J.-S. Filhol, Solvation–reduction coupling in Ca2+ electroactivity in glyme-based electrolytes: A first principles study, Adv. Energy Mater., 2023, 13, 2300311 CrossRef CAS.
- Y. Zhang, H. Zhuo, P. Lei, D. Tang, Q. Hu, X. Du, C.-J. Zheng, J.-L. Yang, Z.-Y. Gu, J. Zhao, S. Tao and X.-L. Wu, Fukui function-engineered gel electrolytes: Thermodynamic/kinetic-synergistic regulation for long-cycling zinc metal batteries, Adv. Mater., 2025, 37, 2508722 CrossRef CAS PubMed.
- S. Ilic, S. N. Lavan and J. G. Connell, Anion-derived contact ion pairing as a unifying principle for electrolyte design, Chem, 2024, 10, 2987–3007 CAS.
- X.-Q. Zhang, X. Chen, L.-P. Hou, B.-Q. Li, X.-B. Cheng, J.-Q. Huang and Q. Zhang, Regulating anions in the solvation sheath of lithium ions for stable lithium metal batteries, ACS Energy Lett., 2019, 4, 411–416 CrossRef CAS.
- Z. Huang, X. Li, Z. Chen, P. Li, X. Ji and C. Zhi, Anion chemistry in energy storage devices, Nat. Rev. Chem., 2023, 7, 616–631 CrossRef PubMed.
- E. Rems, T. Šlosar, S. Drvarič Talian, M. Huš, R. Dominko and A. Serva, Strongly vs. weakly associating anions: transport–structure relationship in LiTFSI–LiNO3 electrolytes, Phys. Chem. Chem. Phys., 2025, 27, 22082–22092 RSC.
- J.-D. Chai and M. Head-Gordon, Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections, Phys. Chem. Chem. Phys., 2008, 10, 6615–6620 RSC.
- F. Weigend and R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- D. Rappoport and F. Furche, Property-optimized gaussian basis sets for molecular response calculations, J. Chem. Phys., 2010, 133, 134105 CrossRef PubMed.
- F. Weigend, F. Furche and R. Ahlrichs, Gaussian basis sets of quadruple zeta valence quality for atoms H–Kr, J. Chem. Phys., 2003, 119, 12753–12762 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, and D. J. Fox, “Gaussian 16 Revision C.02,” , Gaussian, Inc., Wallingford CT, 2021. Search PubMed.
- B. P. Pritchard, D. Altarawy, B. Didier, T. D. Gibson and T. L. Windus, New basis set exchange: An open, up-to-date resource for the molecular sciences community, J. Chem. Inf. Model., 2019, 59, 4814–4820 CrossRef CAS PubMed.
- S. P. Ong, W. D. Richards, A. Jain, G. Hautier, M. Kocher, S. Cholia, D. Gunter, V. L. Chevrier, K. A. Persson and G. Ceder, Python materials genomics (pymatgen): A robust, open-source python library for materials analysis, Comput. Mater. Sci., 2013, 68, 314–319 Search PubMed.
- T. Lu, A comprehensive electron wavefunction analysis toolbox for chemists, Multiwfn, J. Chem. Phys., 2024, 161, 082503 Search PubMed.
- W. Humphrey, A. Dalke and K. Schulten, VMD: Visual molecular dynamics, J. Mol. Graph., 1996, 14, 33–38 CrossRef CAS PubMed.
- D. S. Hall, J. Self and J. R. Dahn, Dielectric constants for quantum chemistry and li-ion batteries: Solvent blends of ethylene carbonate and ethyl methyl carbonate, J. Phys. Chem. C, 2015, 119, 22322–22330 CrossRef CAS.
- Y. Okamoto, Ab Initio calculations of thermal decomposition mechanism of LiPF6-based electrolytes for lithium-ion batteries, J. Electrochem. Soc., 2013, 160, A404–A409 CrossRef CAS.
- T. Hou, K. D. Fong, J. Wang and K. A. Persson, The solvation structure, transport properties and reduction behavior of carbonate-based electrolytes of lithium-ion batteries, Chem. Sci., 2021, 12, 14740–14751 Search PubMed.
- A. Manthiram, An outlook on lithium ion battery technology, ACS Cent. Sci., 2017, 3, 1063–1069 CrossRef CAS PubMed.
- K. Xu, Electrolytes and interphases in Li-ion batteries and beyond, Chem. Rev., 2014, 114, 11503–11618 CrossRef CAS PubMed.
- S. A. Delp, O. Borodin, M. Olguin, C. G. Eisner, J. L. Allen and T. R. Jow, Importance of reduction and oxidation stability of high voltage electrolytes and additives, Electrochim. Acta, 2016, 209, 498–510 CrossRef CAS.
- R. G. Parr and R. G. Pearson, Absolute hardness: companion parameter to absolute electronegativity, J. Am. Chem. Soc., 1983, 105, 7512–7516 CrossRef CAS.
- R. G. Pearson, Hard and soft acids and bases, J. Am. Chem. Soc., 1963, 85, 3533–3539 CrossRef CAS.
- J. Tomasi, B. Mennucci and R. Cammi, Quantum mechanical continuum solvation models, Chem. Rev., 2005, 105, 2999–3094 CrossRef CAS PubMed.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions, J. Phys. Chem. B, 2009, 113, 6378–6396 CrossRef CAS PubMed.
- P. Jaramillo, P. Pérez, P. Fuentealba, S. Canuto and K. Coutinho, Solvent effects on global reactivity properties for neutral and charged systems using the sequential Monte Carlo quantum mechanics model, J. Phys. Chem. B, 2009, 113, 4314–4322 CrossRef CAS PubMed.
- D. Strmcnik, I. E. Castelli, J. G. Connell, D. Haering, M. Zorko, P. Martins, P. P. Lopes, B. Genorio, T. Østergaard, H. A. Gasteiger, F. Maglia, B. K. Antonopoulos, V. R. Stamenkovic, J. Rossmeisl and N. M. Markovic, Electrocatalytic transformation of HF impurity to H2 and LiF in lithium-ion batteries, Nat. Catal., 2018, 1, 255–262 CrossRef.
- M. Martins, D. Haering, J. G. Connell, H. Wan, K. L. Svane, B. Genorio, P. Farinazzo Bergamo Dias Martins, P. P. Lopes, B. Gould, F. Maglia, R. Jung, V. Stamenkovic, I. E. Castelli, N. M. Markovic, J. Rossmeisl and D. Strmcnik, Role of catalytic conversions of ethylene carbonate, water, and HF in forming the solid-electrolyte interphase of Li-ion batteries, ACS Catal., 2023, 13, 9289–9301 CrossRef CAS.
- E. W. C. Spotte-Smith, T. B. Petrocelli, H. D. Patel, S. M. Blau and K. A. Persson, Elementary decomposition mechanisms of lithium hexafluorophosphate in battery electrolytes and interphases, ACS Energy Lett., 2023, 8, 347–355 CrossRef CAS.
- T. Kawamura, S. Okada and J. ichi Yamaki, Decomposition reaction of LiPF6-based electrolytes for lithium ion cells, J. Power Sources, 2006, 156, 547–554 CrossRef CAS.
- S. Trasatti, The absolute electrode potential: an explanatory note (recommendations 1986), Pure Appl. Chem., 1986, 58, 955–966 CrossRef CAS.
- N. N. Rajput, X. Qu, N. Sa, A. K. Burrell and K. A. Persson, The coupling between stability and ion pair formation in magnesium electrolytes from first-principles quantum mechanics and classical molecular dynamics, J. Am. Chem. Soc., 2015, 137, 3411–3420 CrossRef CAS PubMed.
- E. R. Fadel, F. Faglioni, G. Samsonidze, N. Molinari, B. V. Merinov, W. A. Goddard III, J. C. Grossman, J. P. Mailoa and B. Kozinsky, Role of solvent-anion charge transfer in oxidative degradation of battery electrolytes, Nat. Commun., 2019, 10, 3360 CrossRef PubMed.
- E. Peled, D. Golodnitsky and G. Ardel, Advanced model for solid electrolyte interphase electrodes in liquid and polymer electrolytes, J. Electrochem. Soc., 1997, 144, L208 CrossRef CAS.
- S. A. Krachkovskiy, J. D. Bazak, S. Fraser, I. C. Halalay and G. R. Goward, Determination of mass transfer parameters and ionic association of LiPF6: Organic carbonates solutions, J. Electrochem. Soc., 2017, 164, A912 Search PubMed.
- O. Borodin, J. Self, K. A. Persson, C. Wang and K. Xu, Uncharted waters: Super-concentrated electrolytes, Joule, 2020, 4, 69–100 CrossRef CAS.
- H. Wang, Z. Yu, X. Kong, S. C. Kim, D. T. Boyle, J. Qin, Z. Bao and Y. Cui, Liquid electrolyte: The nexus of practical lithium metal batteries, Joule, 2022, 6, 588–616 CrossRef CAS.
- R. G. Parr and W. Yang, Density functional approach to the frontier-electron theory of chemical reactivity, J. Am. Chem. Soc., 1984, 106, 4049–4050 CrossRef CAS.
- J. P. Perdew, R. G. Parr, M. Levy and J. L. Balduz, Density-functional theory for fractional particle number: Derivative discontinuities of the energy, Phys. Rev. Lett., 1982, 49, 1691–1694 CrossRef CAS.
- H. Gao, F. Maglia, P. Lamp, K. Amine and Z. Chen, Mechanistic study of electrolyte additives to stabilize high-voltage cathode–electrolyte interface in lithium-ion batteries, ACS Appl. Mater. Interfaces, 2017, 9, 44542–44549 CrossRef CAS PubMed.
- B. Wang, J. Wang, L. Zhang, P. K. Chu, X.-F. Yu, R. He and S. Bian, Adsorptive shield derived cathode electrolyte interphase formation with impregnation on LiNi0.8Mn0.1Co0.1O2 cathode: A mechanism-guiding-experiment study, ACS Appl. Mater. Interfaces, 2024, 16, 50747–50756 CrossRef CAS.
- Y. Zhu, Y. Li, M. Bettge and D. P. Abraham, Positive electrode passivation by LiDFOB electrolyte additive in high-capacity lithium-ion cells, J. Electrochem. Soc., 2012, 159, A2109 CrossRef CAS.
- I. A. Shkrob, Y. Zhu, T. W. Marin and D. P. Abraham, Mechanistic insight into the protective action of bis(oxalato)borate and difluoro(oxalate)borate anions in li-ion batteries, J. Phys. Chem. C, 2013, 117, 23750–23756 CrossRef CAS.
- M. Mao, B. Huang, Q. Li, C. Wang, Y.-B. He and F. Kang, In-situ construction of hierarchical cathode electrolyte interphase for high performance LiNi0.8Co0.1Mn0.1O2/Li metal battery, Nano Energy, 2020, 78, 105282 CrossRef CAS.
- S. Jiao, X. Ren, R. Cao, M. H. Engelhard, Y. Liu, D. Hu, D. Mei, J. Zheng, W. Zhao, Q. Li, N. Liu, B. D. Adams, C. Ma, J. Liu, J.-G. Zhang and W. Xu, Stable cycling of high-voltage lithium metal batteries in ether electrolytes, Nat. Energy, 2018, 3, 739–746 CrossRef CAS.
- E. K. W. Andersson, L.-T. Wu, L. Bertoli, Y.-C. Weng, D. Friesen, K. Elbouazzaoui, S. Bloch, R. Ovsyannikov, E. Giangrisostomi, D. Brandell, J. Mindemark, J.-C. Jiang and M. Hahlin, Initial SEI formation in LiBOB-, LiDFOB- and LiBF4-containing PEO electrolytes, J. Mater. Chem. A, 2024, 12, 9184–9199 RSC.
- J. Holoubek, Q. Yan, H. Liu, E. J. Hopkins, Z. Wu, S. Yu, J. Luo, T. A. Pascal, Z. Chen and P. Liu, Oxidative stabilization of dilute ether electrolytes via anion modification, ACS Energy Lett., 2022, 7, 675–682 CrossRef CAS.
- M. Paillot, A. Wong, S. A. Denisov, P. Soudan, P. Poizot, B. Montigny, M. Mostafavi, M. Gauthier and S. Le Caër, Predicting degradation mechanisms in lithium bistriflimide “water-in-salt” electrolytes for aqueous batteries, ChemSusChem, 2023, 16, e202300692 CrossRef CAS PubMed.
- P. C. Howlett, E. I. Izgorodina, M. Forsyth and D. R. MacFarlane, Electrochemistry at negative potentials in bis(trifluoromethanesulfonyl)amide ionic liquids, Z. Phys. Chem., 2006, 220, 1483–1498 CrossRef CAS.
- K. Forster-Tonigold, F. Buchner, A. Groß and R. J. Behm, Reactive interactions between the ionic liquid BMP-TFSI and a Na surface, Batter. Supercaps, 2023, 6, e202300336 CrossRef CAS.
- J. Alvarado, M. A. Schroeder, T. P. Pollard, X. Wang, J. Z. Lee, M. Zhang, T. Wynn, M. Ding, O. Borodin, Y. S. Meng and K. Xu, Bisalt ether electrolytes: a pathway towards lithium metal batteries with Ni-rich cathodes, Energy Environ. Sci., 2019, 12, 780–794 RSC.
- H.-G. Steinrück, C. Cao, M. R. Lukatskaya, C. J. Takacs, G. Wan, D. G. Mackanic, Y. Tsao, J. Zhao, B. A. Helms, K. Xu, O. Borodin, J. F. Wishart and M. F. Toney, Interfacial speciation determines interfacial chemistry: X-ray-induced lithium fluoride formation from water-in-salt electrolytes on solid surfaces, Angew. Chem., Int. Ed., 2020, 59, 23180–23187 CrossRef.
- I. A. Shkrob, K. Z. Pupek, J. A. Gilbert, S. E. Trask and D. P. Abraham, Chemical stability of lithium 2-trifluoromethyl-4,5-dicyanoimidazolide, an electrolyte salt for Li-ion cells, J. Phys. Chem. C, 2016, 120, 28463–28471 CrossRef CAS.
- F. Lindgren, C. Xu, J. Maibach, A. M. Andersson, M. Marcinek, L. Niedzicki, T. Gustafsson, F. Björefors and K. Edström, A hard X-ray photoelectron spectroscopy study on the solid electrolyte interphase of a lithium 4,5-dicyano-2-(trifluoromethyl)imidazolide based electrolyte for Si-electrodes, J. Power Sources, 2016, 301, 105–112 CrossRef CAS.
- T. Stuyver, F. De Proft, P. Geerlings and S. Shaik, How do local reactivity descriptors shape the potential energy surface associated with chemical reactions? The valence bond delocalization perspective, J. Am. Chem. Soc., 2020, 142, 10102–10113 CrossRef CAS.
- B. Wang, P. Geerlings, S. Liu and F. De Proft, Extending the scope of conceptual density functional theory with second order analytical methodologies, J. Chem. Theory Comput., 2024, 20, 1169–1184 CrossRef CAS PubMed.
- S. Ilic, M. Martins, H. Liu, P. Farinazzo Bergamo Dias Martins, D. Haering, J. Yang, T. Hatsukade, B. Genorio, S. E. Weitzner, L. F. Wan, Z. Zhang, J. G. Connell, B. Key, J. Cabana and D. Strmcnik, An unwanted guest in the electrochemical oxidation of high-voltage Li-ion battery electrolytes: the life of highly reactive protons, Energy Environ. Sci., 2025, 18, 8303–8312 RSC.
- C. Morell, A. Grand and A. Toro-Labbé, New dual descriptor for chemical reactivity, J. Phys. Chem. A, 2005, 109, 205–212 CrossRef CAS PubMed.
- J. I. Martínez-Araya, Why is the dual descriptor a more accurate local reactivity descriptor than fukui functions?, J. Math. Chem., 2014, 53, 451–465 CrossRef.
- S. Shui Zhang, An unique lithium salt for the improved electrolyte of Li-ion battery, Electrochem. Commun., 2006, 8, 1423–1428 CrossRef.
- T. Schedlbauer, S. Krüger, R. Schmitz, R. Schmitz, C. Schreiner, H. Gores, S. Passerini and M. Winter, Lithium difluoro(oxalato)borate: A promising salt for lithium metal based secondary batteries?, Electrochim. Acta, 2013, 92, 102–107 CrossRef CAS.
- M. Xu, L. Zhou, L. Hao, L. Xing, W. Li and B. L. Lucht, Investigation and application of lithium difluoro(oxalate)borate (LiDFOB) as additive to improve the thermal stability of electrolyte for lithium-ion batteries, J. Power Sources, 2011, 196, 6794–6801 CrossRef CAS.
- K. Goloviznina, A. Serva and M. Salanne, Speciation of the proton in water-in-salt electrolytes, Faraday Discuss., 2024, 253, 478–492 RSC.
- A. France-Lanord, F. Pietrucci, A. M. Saitta, J.-M. Tarascon, A. Grimaud and M. Salanne, Chemical decomposition of the TFSI anion under aqueous basic conditions, PRX Energy, 2022, 1, 013005 CrossRef.
- Y. Yu, A. Baskin, C. Valero-Vidal, N. T. Hahn, Q. Liu, K. R. Zavadil, B. W. Eichhorn, D. Prendergast and E. J. Crumlin, Instability at the electrode/electrolyte interface induced by hard cation chelation and nucleophilic attack, Chem. Mater., 2017, 29, 8504–8512 CrossRef CAS.
- C. Xu, S. Renault, M. Ebadi, Z. Wang, E. Björklund, D. Guyomard, D. Brandell, K. Edström and T. Gustafsson, LiTDI: A highly efficient additive for electrolyte stabilization in lithium-ion batteries, Chem. Mater., 2017, 29, 2254–2263 CrossRef CAS.
- M. Dranka, P. Jankowski and G. Z. Żukowska, Snapshots of the hydrolysis of lithium 4,5-dicyanoimidazolate–glyme solvates. impact of water molecules on aggregation processes in lithium-ion battery electrolytes, J. Phys. Chem. C, 2018, 122, 3201–3210 CrossRef CAS.
- L. Niedzicki, E. Karpierz, A. Bitner, M. Kasprzyk, G. Zukowska, M. Marcinek and W. Wieczorek, Optimization of the lithium-ion cell electrolyte composition through the use of the LiTDI salt, Electrochim. Acta, 2014, 117, 224–229 CrossRef CAS.
- A. V. Marenich, S. V. Jerome, C. J. Cramer and D. G. Truhlar, Charge model 5: An extension of Hirshfeld population analysis for the accurate description of molecular interactions in gaseous and condensed phases, J. Chem. Theory Comput., 2012, 8, 527–541 CrossRef CAS PubMed.
- A. Benayad, D. Diddens, A. Heuer, A. N. Krishnamoorthy, M. Maiti, F. L. Cras, M. Legallais, F. Rahmanian, Y. Shin, H. Stein, M. Winter, C. Wölke, P. Yan and I. Cekic-Laskovic, High-throughput experimentation and computational freeway lanes for accelerated battery electrolyte and interface development research, Adv. Energy Mater., 2022, 12, 2102678 CrossRef CAS.
- X. Fu, S. Li, N. H. Deissler, J. B. V. Mygind, J. Kibsgaard and I. Chorkendorff, Effect of lithium salt on lithium-mediated ammonia synthesis, ACS Energy Lett., 2024, 9, 3790–3795 CrossRef CAS.
- K. Trapp, J. Yoo and M. R. Lukatskaya, The role of anions and additives in electrochemical CO2 reduction, Curr. Opin. Electrochem., 2025, 52, 101707 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |






