Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Digital and experimental design of CO2-responsive polymers based on acrylamide monomers for carbon capture
Emil
Pashayev
 and
Prokopios
Georgopanos
and
Prokopios
Georgopanos
 *
*
Helmholtz-Zentrum Hereon, Institute of Membrane Research, Max-Planck-Str. 1, 21502 Geesthacht, Germany. E-mail: prokopios.georgopanos@hereon.de
First published on 23rd February 2026
Abstract
This study provides a comprehensive evaluation of the CO2 capture performance of the CO2-responsive homopolymer poly(N-[3-(dimethylamino)propyl]-acrylamide) (PDMAPAm) and its diblock copolymer poly(N-[3-(dimethylamino)propyl]-acrylamide)-b-poly(methyl methacrylate) (PDMAPAm-b-PMMA), with a particular emphasis on their integration into membrane adsorbers for direct air capture (DAC) applications. A central novelty of this work is the development of a pioneering unified kinetic model that, for the first time, couples polymerization reaction kinetics with CO2 adsorption kinetics. This model enables the digital predictive design of polymer materials whose CO2 uptake is expressed directly as a function of polymer properties. By systematically varying the molar mass of the amine-functional PDMAPAm block and the poly(methyl methacrylate) (PMMA) content, this study identifies the optimal polymer properties that balance high CO2 uptake with favorable processing characteristics. Adsorption experiments conducted under dry conditions revealed a physisorption-dominated mechanism, where CO2 primarily interacts with tertiary amine and carbonyl functional groups. Notably, the unified model predicted a target molar mass of 18.5 kDa for the homopolymer of PDMAPAm and identified the optimal synthesis ratio of RAFT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) initiator
initiator![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) monomer. This model also accurately forecasts the CO2-uptake capacity of the PDMAPAm homopolymer to be 0.05 mmol g−1 and that of its corresponding diblock copolymer (PDMAPAm-b-PMMA) to be approximately 0.1 mmol g−1 under dry ambient conditions.
monomer. This model also accurately forecasts the CO2-uptake capacity of the PDMAPAm homopolymer to be 0.05 mmol g−1 and that of its corresponding diblock copolymer (PDMAPAm-b-PMMA) to be approximately 0.1 mmol g−1 under dry ambient conditions.
Introduction
Carbon capture technology is a key research field currently being developed for application1 in various industries such as power generation,2 chemical production,3 and metal production plants4 to reduce CO2 emissions. In addition, due to the insufficient progress in industrial decarbonization, attention has shifted toward capturing atmospheric CO2, even at a relatively low concentration, through an emerging approach known as direct air capture (DAC).As mentioned before, DAC is designed to capture carbon dioxide (CO2) directly from the atmosphere, addressing the urgent need to mitigate climate change.5–10 This process involves the use of adsorbents, which are materials that can selectively capture and hold CO2 molecules from the air.6,11,12 The DAC process typically involves drawing ambient air into a system where it passes over an adsorbent material.10,13 The CO2 molecules are captured on the adsorbent surface through physical or chemical interactions.10,13,14 This phenomenon is called adsorption. When the adsorbent becomes saturated with CO2, it undergoes a regeneration process, which is called desorption, often involving a temperature or pressure or electric swing, to release the captured CO2 for storage or utilization.15–17
The development and optimization of adsorbents for DAC are crucial for improving the efficiency, energy costs and scalability of this technology.6,18 By enhancing the CO2 adsorption capacity and stability of these materials, researchers aim to make DAC a viable solution for large-scale carbon removal, contributing to global efforts to combat climate change.11,12 Various adsorbent materials are utilized in DAC technologies to capture CO2 directly from the atmosphere.11,19–22 Among the various adsorbents, materials such as silica, alumina, zeolite 13X, and amine-functionalized adsorbents,23,24 including polyethyleneimine (PEI), tetraethylenepentamine (TEPA), and diblock copolymers,25,26 have shown significant promise. Recent studies have demonstrated that amine-functionalized adsorbents exhibit high CO2 adsorption capacities and excellent cyclic stability, making them suitable for repeated use in DAC systems.27 In particular, amine-containing polymers, such as polyethyleneimine (PEI) and tetraethylenepentamine (TEPA), have emerged as particularly promising adsorbents for DAC applications.11,28–31
Especially, amine-containing diblock copolymers exhibit an excellent performance in capturing CO2, considering that the amine group has a strong affinity for CO2.25,26 In particular, diblock copolymers with amine-containing groups such as poly(2-(dimethylamino)ethyl methacrylate) (PDMAEMA),32–35 poly(N,N-dimethylallylamine) (PDMAAm),36–39 poly(2-aminoethyl methacrylate) (PAEM),40,41 and poly(N-[3-(dimethylamino)propyl]-acrylamide) (PDMAPAm)42 hold great potential to be used as adsorbents for DAC due to their high CO2 reactivity, high CO2 selectivity,32,42,43 high porosity,44–48 and scalability.42,49–51 These polymers display advantageous properties ideal for CO2 capture technologies, featuring a stable, relatively high, and swift CO2 adsorption capacity, as well as a low desorption temperature.51–53 The presence of an amine-containing group as a block on the surface and within the pore walls of diblock copolymers, together with their tunable properties via simulation, enables them to exhibit CO2-responsive behavior. This allows for adjustable pore sizes and chemical surface interactions during CO2 adsorption and desorption.
Unlike conventional amine-functionalized adsorbents such as primary or secondary amine-grafted silica, amine-impregnated porous supports and amine-modified carbons, which often suffer from oxidative degradation, pore blockage, amine leaching, and poor cyclic stability under humid or elevated-temperature DAC conditions,54,55 CO2-responsive diblock copolymers have potential to mitigate many of these issues. Because their amine groups are covalently built into the polymer backbone rather than physically dispersed or surface-grafted, these materials exhibit enhanced structural integrity and improved resistance to chemical and thermal degradation.56 Moreover, the microphase separation inherent to diblock copolymers allows precise control over their domain morphology, facilitating the design of ordered nanoscale pathways for gas diffusion and providing tunable sorption–desorption kinetics, which are features not readily achievable in conventional amine-loaded sorbents.57 These distinct advantages justify the exploration of CO2-responsive diblock copolymers as next-generation adsorbents and highlight the rationale behind the material design adopted in this study. Apart from this, CO2-responsive polymers offer significant potential for use in polymer–filler mixed-matrix membrane adsorbers, owing to their tunable affinity toward CO2. Consequently, understanding their interactions with various fillers is critical. In previous publications,51,58 PDMAPAm and PDMAPAm-b-PMMA polymers were coated onto PDMS (gutter layer) to prepare a thin-film composite membrane, which showed stable permeability and selectivity values for several cycles of time-lag measurements for different gases such as CO2, O2, N2, and H2. The interaction between the PDMAPAm chains and the underlying PDMS gutter layer can be attributed to a combination of physical adsorption, hydrogen bonding, and interfacial entanglement, all of which contribute to the stability and performance of the composite membrane.59 Apart from PDMS, as fillers MOFs are also expected to show the strongest and most specific interactions with PDMAPAm-based polymers, primarily because these polymers contain amide and amine functional groups capable of coordinating directly with the open metal sites present in many CO2-selective MOFs, forming strong Lewis acid–base or hydrogen-bonding interactions at the polymer–filler interface.60 This coordination not only enhances interfacial adhesion but also increases the density of COs-affinitive sites, enabling synergistic sorption-driven transport, which is highly compatible with the chemical nature of PDMAPAm.61
A detailed understanding of the CO2 transport behavior in PDMAPAm- and PDMAPAm-b-PMMA-coated membranes as selective layers and its dependence on membrane morphology has been established in our previous studies.51,58 These studies demonstrated that the CO2/N2 permeability selectivity of the membranes can reach values as high as 10–15, indicating that the polymers exhibit pronounced CO2-responsive behavior.51,58 The dense structure of these polymers was confirmed through SEM imaging, which reveals a continuous and nonporous selective layer, in which gas transport is dominated by polymer–gas interactions rather than convective flow.51 In these dense polymer membranes, gas permeation follows the solution–diffusion mechanism, wherein gas molecules dissolve in the polymer matrix and subsequently diffuse through it.62
Recent reports on CO2-responsive polymers have established key structure–property relationships that guide contemporary material design strategies.63–65 Moreover, several studies have developed reaction kinetic models of RAFT polymerization to simulate polymer synthesis, enabling optimization of the polymer properties50,66 and polymerization rate50,67,68 and providing predictive tools for process upscaling.67 Furthermore, some modeling efforts have focused on CO2-adsorption isotherms in amine-based membrane adsorbers, offering valuable insights into their adsorption mechanisms and sorbent performance.69–72 However, to date, no study has established a correlation between polymerization kinetics and the CO2-adsorption behavior of the resulting polymer material. Building on this gap in the state of the art, we explicitly articulate the working hypothesis of the present study that CO2 uptake by the PDMAPAm homopolymer and PDMAPAm-b-PMMA diblock copolymer can be rationally optimized by establishing a unified kinetic model that couples polymerization kinetics with CO2 adsorption kinetics, thereby enabling predictive control over the polymer properties and its corresponding CO2-capture performance.
Experimental
Material and methods
N-[3-(Dimethylamino)propyl]acrylamide (stabilized with MEHQ) (DMAPAm, >98.0%, TSI, Zwijndrecht, Belgium) was firstly dissolved in 1,4 dioxane (≥99.8%, Merck, Darmstadt, Germany) and percolated through a column of basic aluminum oxide (>98%, Sigma-Aldrich) prior to use to remove the inhibitor. Then, 2,2′-azobis(2-methylpropionitrile) (AIBN, 98%, Sigma-Aldrich, Taufkirchen, Germany, stored at 4 °C) and 4-cyano-4-[(dodecylsulfanylthiocarbonyl)-sulfanyl]pentanoic acid (CDTPA, 97%, Sigma-Aldrich, Taufkirchen, Germany, stored at 4 °C) were added to the monomer and solvent mixture. Methyl methacrylate (99%, stabilized with ≤30 ppm MEHQ, Sigma-Aldrich, Steinheim, Germany) was also freshly percolated using the same procedure for the same reason. Nitrogen was purchased from Air Liquide (Hamburg, Germany, 99.999% purity). The polymer was precipitated in n-hexane (99%, Sigma-Aldrich, Taufkirchen, Germany). Proton nuclear magnetic resonance (1H NMR) measurements were carried out in deuterated chloroform-d1 (CDCl3, 99.8% containing 0.03% (v/v) TMS, Sigma-Aldrich, Taufkirchen, Germany, stored at 4 °C). Gel permeation chromatography (GPC) measurements were carried out in N,N-dimethyl acetamide (DMAc, ≥99.9%, Sigma-Aldrich, Taufkirchen, Germany).Synthesis of PDMAPAm
For the synthesis of PDMAPAm via reversible addition–fragmentation chain transfer (RAFT) polymerization in a small lab flask, CDTPA (18.0 mg, 0.04 mmol, 0.7 eq.), AIBN (10.0 mg, 0.06 mmol, 1 eq.) and percolated DMAPAm (2000 mg, 13.00 mmol, 200 eq.) were dissolved in 9 mL of 1,4-dioxane (20 wt% monomer content). The mixture in the flasks was degassed with nitrogen in a cold-water bath at 15 °C for 20 minutes. Following this, polymerization was conducted in a 20 mL glass flask using a thermo-shaker set at 70 °C and 250 rpm for 4 hours. The reaction was terminated by exposing the reaction medium to air in an ice bath (0 °C). Samples for (1H) NMR and GPC were taken to determine the monomer conversion (90%) and molecular weight of the synthesized polymer, respectively. The final product was precipitated from n-hexane, filtered, and dried overnight in a vacuum oven at 35 °C under 1 mbar.Synthesis of PDMAPAm-b-PMMA
To synthesize the PDMAPAm-b-PMMA diblock copolymer, dried PDMAPAm (1000 mg, 0.03 mmol, 2 eq.), which served as the macro-RAFT/macro-stabilizer agent, MMA (480 mg, 4.8 mmol, 165 eq.), and AIBN (5 mg, 0.03 mmol, 1 eq.) were dissolved in 6 mL of 1,4-dioxane. The mixture was degassed with nitrogen in a cold-water bath at 15 °C for 20 minutes. Then polymerization was conducted in a 10 mL glass flask using a thermo-shaker at 70 °C and 250 rpm for 24 hours. The reaction was terminated by exposing the medium to air in an ice bath (0 °C). Samples for 1H NMR and GPC were taken to determine the MMA conversion (92%) and the molecular weight of the synthesized diblock copolymer, respectively. The polymer was precipitated in n-hexane and dried in a vacuum oven at 35 °C and 1 mbar for 24 hours (see the detailed synthesis protocol in Tables S1 and S2 and properties of both the homopolymer and diblock copolymer in Table S3 and Fig. S1 in the SI).The synthesis protocol for PMMA70 can be found in a previous publication,49 while the polymer properties are given in Table S4 in the SI.
Characterization
Gas permeation chromatography (GPC)
The apparent molecular weight distributions of the PDMAPAm homopolymers (macro-RAFT agents) and PDMAPAm-b-PMMA diblock copolymers were measured by GPC at 50 °C using DMAc with added lithium chloride (0.1 M) as the eluent. A Waters 717 Plus instrument (Waters, Milford, MA, USA) equipped with PSS GRAM columns (PSS GmbH, Mainz, Germany) [GRAM pre-column (dimensions of 8–50 nm) and two GRAM columns of different porosity (3000 and 1000)] with dimensions of 8 × 300 mm and a particle size of 10 µm was used. The samples were measured at a flow rate of 1 mL min−1 using a VWR-Hitachi 2130 pump (VWR Hitachi, Darmstadt, Germany) and a VWR-Hitachi L2490 RI (refractive index) detector (VWR Hitachi, Darmstadt, Germany). The GPC was calibrated with narrow PMMA standards and data were analyzed using the PSS WinGPC UniChrom software (PSS GmbH, Mainz, Germany).Proton nuclear magnetic resonance (1H NMR)
1H NMR spectroscopy experiments were performed using a Bruker AV500 spectrometer (Bruker, Rheinstetten, Germany) and a Spinsolve Carbon 80 Ultra MultiX benchtop spectrometer (80 MHz, Magritek GmbH, Aachen, Germany). Spectra were recorded with the Bruker AV500 spectrometer by applying a 10 ms 90° pulse at a sample temperature of 298 K. 16 scans were recorded with a relaxation delay of 3 s. Similarly, the experiments with the Spinsolve Carbon 80 Ultra MultiX spectrometer were performed using the Proton+ protocol with 4 scans, an acquisition time of 6.4 s, a repetition time of 1 min, and a pulse angle of 90°. The sample concentration was 20 g L−1, and CDCl3 was employed as the solvent. The 1H NMR spectra were analyzed with the MestReNova 15.1 software (Mestrelab Research, Santiago de Compostela, Spain). 1,3,6-Trioxane was used as an internal standard. DMAPAm conversion from macro-RAFT synthesis was estimated by comparing the integration of the double-bond proton areas in the spectra of the sample before and after the reaction, yielding a conversion of 92%. Using the same method,51 the conversion of methyl methacrylate in the second block synthesis, also measured in CDCl3, was calculated from the decrease in the integral of the monomer peaks, which was 91%.High-pressure differential scanning calorimetry (DSC)
The change in the glass transition temperature of the PDMAPAm homopolymer was examined using high-pressure differential scanning calorimetry in CO2- and N2-containing environments at different pressures. A High-Pressure DSC 1 from Mettler Toledo was used to investigate the in situ thermal transitions of PDMAPAm. The measurements were conducted at 1, 3, 5, and 7 bar in a CO2-containing environment, whereas the measurements under nitrogen were performed only at a pressure of 1 bar. The temperature program for the HP-DSC measurements consists of two steps. Firstly, the sample was heated from 30 °C to 120 °C, held at 120 °C for 5 minutes to establish an isotherm, and then cooled back to 20 °C. In the second step, the glass transition temperature (Tg) of the sample at each pressure is determined by heating the sample to 20 °C for 2 hours, saturating it with CO2, and then increasing the temperature to 60 °C at 2 °C min−1. When pressure was not applied, the temperature increase rate was set to 10 °C min−1.Magnetic suspension balance
The gravimetric sorption method was employed to study the sorption of dry carbon dioxide using a Rubotherm magnetic suspension balance (Rubotherm Präzisionsmesstechnik GmbH, Bochum, now TA Instruments). The magnetic suspension balance enables precise measurement of the changes in force and mass on samples within controlled environments (such as pressure and temperature). These measurements facilitate the accurate determination of transport and state quantities (such as sorption and diffusion) and enable the investigation of chemical reactions (such as corrosion, coating, and drying). The holder remains suspended by a permanent magnet. The system allows for the measurement of weight changes in the holder and sample under varying pressure and temperature conditions. These changes can be linked to the specific gas uptake.73–75 During adsorption experiments, the gas is introduced stepwise at controlled pressure and temperature, and the balance records the increase in sample weight until equilibrium is reached in each step. For desorption, the pressure is gradually reduced, and the decrease in weight is monitored to determine the release of gas from the material. The graph of specific gas uptake at a constant temperature versus pressure is known as the sorption isotherm of the gas for the material. To minimize disturbances caused by non-ideal behavior, it is preferable to consider the real gas density rather than the ideal gas density for the evaluation of the results.73 A detailed description is provided in the literature.73Results and discussion
For the synthesis of the PDMAPAm-b-PMMA diblock copolymer, the block lengths were intentionally selected to balance two essential requirements: (1) maintaining a high fraction of CO2-responsive tertiary amine groups to ensure strong gas–polymer interactions and (2) incorporating a PMMA block of sufficient size to provide the mechanical integrity necessary for membrane formation.The reaction conditions and composition used for synthesizing PDMAPAm and PDMAPAm-b-PMMA were selected based on established RAFT polymerization principles and prior publications on the RAFT polymerization of amine-containing monomers.76–78 Specifically, the monomer-to-RAFT-agent and initiator ratios were chosen to ensure controlled radical polymerization with a narrow dispersity, following the common approach of maintaining an initiator-to-monomer ratio (AIBN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMAPAm) of approximately 1
DMAPAm) of approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 (ref. 51, 76 and 78) to achieve moderate polymerization rates and high end-group fidelity.67 High end-group fidelity is very important for further chain growth with MMA during the synthesis of diblock copolymers.79
200 (ref. 51, 76 and 78) to achieve moderate polymerization rates and high end-group fidelity.67 High end-group fidelity is very important for further chain growth with MMA during the synthesis of diblock copolymers.79
Our synthesis strategy began with the preparation of the PDMAPAm homopolymer, followed by chain extension with incrementally increasing PMMA block lengths. This stepwise approach allowed us to identify the range in which the diblock copolymer remained both mechanically stable and highly CO2-responsive.
After that, the synthesized polymers, PDMAPAm and its diblock copolymer, PDMAPAm-b-PMMA, were systematically characterized to evaluate their interactions with CO2 under dry conditions. Comparative analyses were conducted using PDMAPAm homopolymers with two distinct molar masses, as well as PDMAPAm-b-PMMA copolymers featuring a constant PDMAPAm block and varying PMMA block lengths. To accurately assess CO2 adsorption behavior, the gas was treated as a real gas, and its density was calculated using the real gas equation of state.
Mechanism of dry CO2 adsorption
Generally, the developed amine-based homopolymer, PDMAPAm, and diblock copolymer, PDMAPAm-b-PMMA, as shown in Fig. 1, can undergo physisorption when they interact with dry CO2.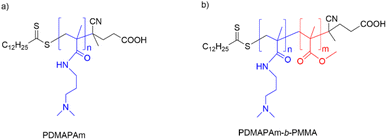 | ||
| Fig. 1 Chemical structure of the RAFT-polymerized (a) homopolymer PDMAPAm and (b) diblock copolymer PDMAPAm-b-PMMA. | ||
Dry CO2 physisorption mechanism on PDMAPAm
As represented in Fig. 1(a), PDMAPAm has amide groups (consisting of carbonyl and secondary amine groups) and tertiary amine groups in its structure. When the PDMAPAm powder is exposed to dry CO2, it is expected that its amide and tertiary amine groups can show affinity for CO2.80,81 Since the concentration of RAFT end groups is less than the concentration of functional groups on monomeric units, the interaction of the RAFT end groups with dry CO2 is neglected.Polyamides feature polar interaction sites, specifically (–NH) and (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) groups. A strong Lewis acid–base interaction occurs between CO2 and the oxygen of the amide carbonyl. Polyamide-based materials such as melamine-based porous organic amide polymers capture 0.1 mmol per g CO2 under dry conditions at P of 1 bar and T of 25 °C.82 As for the tertiary amine group, it interacts with dry CO2 only via one of the van der Waals interaction types,65,66 which may be achieved due to the temporary dipoles created by fluctuations in the electron density over the CO2 molecule and polarization of the tertiary amine group.83
O) groups. A strong Lewis acid–base interaction occurs between CO2 and the oxygen of the amide carbonyl. Polyamide-based materials such as melamine-based porous organic amide polymers capture 0.1 mmol per g CO2 under dry conditions at P of 1 bar and T of 25 °C.82 As for the tertiary amine group, it interacts with dry CO2 only via one of the van der Waals interaction types,65,66 which may be achieved due to the temporary dipoles created by fluctuations in the electron density over the CO2 molecule and polarization of the tertiary amine group.83
Dry CO2 physisorption mechanism on PMMA
Besides PDMAPAm, the PDMAPAm-b-PMMA diblock copolymer has a PMMA block in it structure, as shown in Fig. 1(b). Poly(methyl methacrylate) (PMMA) was thoroughly examined by Wissinger84,85 because of its swelling and adsorption characteristics. These analyses demonstrate its outstanding adsorption behavior across various CO2 pressures (0.1–10 MPa) and good swelling abilities,84,86–88 indicating the strong interaction between PMMA and carbon dioxide compared to other polymers.89,90 The interaction between the polymer side chains (containing methyl ester groups) and CO2 was examined using various techniques, including ab initio calculations, FT-IR spectroscopy, and Raman spectroscopy. These analyses demonstrated that the carbonyl group participates in a Lewis acid–base interaction with CO2,91–97 which means that the attachment of CO2 to PMMA is based on the interaction between the lone pairs of oxygen in the carbonyl groups of the polymer and the dipole moments of carbon dioxide.91–97 In the context of CO2 and PMMA, the oxygen in the carbonyl group acts as a Lewis base, donating an electron pair to the CO2 molecule, which acts as a Lewis acid.Dry CO2 adsorption measurement
Several adsorption measurements were conducted to analyse the dry CO2 capture performance of the synthesized CO2-responsive polymer in a gravimetric adsorption device, as presented in Fig. 2.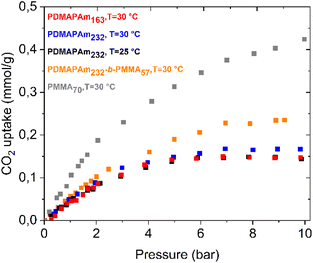 | ||
| Fig. 2 CO2 adsorption curves of PDMAPAm at different temperatures and molar masses compared with PMMA and PDMAPAm-b-PMMA. | ||
Fig. 2 illustrates the CO2 uptake capacity of the PMMA homopolymer, PDMAPAm homopolymer, and PDMAPAm-b-PMMA diblock copolymer at varying PMMA block contents, adsorption temperatures and molar masses. This is clear from the curves that both the homopolymer and diblock copolymer adsorb dry CO2 and the CO2 uptake capacity changes with the molar mass of the homopolymer, temperature and block content of the diblock copolymer. Another important point is that the CO2 uptake of the homopolymers increases up to 6 bar, and then becomes constant, whereas a plateau appears at a higher pressure. As shown by the results for dry CO2 adsorption, PDMAPAm captures 0.05 mmol g−1 under dry conditions at atmospheric pressure and 25 °C. This uptake capacity is very close to the CO2 capture capacity of tertiary-amine-based polymer98,99 and poly(amide)-based adsorbents, in which the interaction between CO2 and the tertiary amine groups occurs via van der Waals forces98–100 and the interactions between CO2 and –NH or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups occur based on Lewis acid–base interactions,82 respectively. This indicates that CO2 adsorption under dry conditions also occurred via identical interactions because PDMAPAm has both functional groups in its structure. Based on DFT studies,101,102 tertiary amine has stronger interaction energy with CO2, which is up to −16 kcal mol−1,80,103,104 than amide, which changes between −5 and −10 kcal mol−1.81 Due to the stronger interaction energy between tertiary amines and dry CO2 compared to amide groups, CO2 molecules are more likely to be retained near the tertiary amine sites. This leads to a higher local concentration and enhanced uptake of CO2. Therefore, in this study, it is assumed that the CO2 adsorption capacity of the PDMAPAm polymer, whose monomeric unit contains both a tertiary amine and an amide group, primarily arises from the interaction between dry CO2 and its tertiary amine functionality.
O groups occur based on Lewis acid–base interactions,82 respectively. This indicates that CO2 adsorption under dry conditions also occurred via identical interactions because PDMAPAm has both functional groups in its structure. Based on DFT studies,101,102 tertiary amine has stronger interaction energy with CO2, which is up to −16 kcal mol−1,80,103,104 than amide, which changes between −5 and −10 kcal mol−1.81 Due to the stronger interaction energy between tertiary amines and dry CO2 compared to amide groups, CO2 molecules are more likely to be retained near the tertiary amine sites. This leads to a higher local concentration and enhanced uptake of CO2. Therefore, in this study, it is assumed that the CO2 adsorption capacity of the PDMAPAm polymer, whose monomeric unit contains both a tertiary amine and an amide group, primarily arises from the interaction between dry CO2 and its tertiary amine functionality.
Effect of molar mass on CO2 capture
As is clear in Fig. 2, the CO2 capture capacity of the PDMAPAm homopolymer increases very slightly as its molar mass increases. This trend can be attributed to the higher number of amine groups per mole in the polymer with a greater molar mass, which are the primary sites for CO2 interaction, as reported previously.51 Additionally, longer chain lengths may provide more free volume for CO2 molecules.105,106 However, the increase remains small because under dry conditions, the lone pair electrons on the nitrogen of the tertiary amine are sterically hindered by two methyl groups, limiting their interaction with CO2.94–97 Consequently, even as the number of amine groups increases, steric hindrance restricts their accessibility, resulting in only a minor improvement in uptake capacity.Effect of PMMA content on CO2 capture
Furthermore, Fig. 2 shows that the CO2 adsorption capacity of the polymer increases with an increasing PMMA block portion in the diblock copolymer.This is also clearly seen in the CO2 adsorption capacity of PMMA as a homopolymer. The PMMA block can physically interact with CO2 and this interaction is primarily through physical adsorption, where CO2 molecules diffuse into the polymer matrix.107 The CO2 uptake capacity of the PMMA homopolymer reaches 0.1 mmol g−1 at 1 bar and 30 °C, which is very close to the value mentioned in the literature.107 As mentioned before, these interactions between CO2 and PMMA are described as Lewis acid–base interactions involving the transfer of electron pairs, resulting in strong, stable bonds.108 Higher pressure reduces the free volume, facilitating electron-pair transfer and strengthening the interaction.108 This results in a more efficient and higher CO2 uptake capacity. However, the interactions between PDMAPAm and CO2 are due to temporary dipoles created by fluctuations in electron density,83 and while an increasing pressure brings the molecules closer together, enhancing these transient dipoles, the interactions remain weaker and less specific compared to Lewis acid–base interactions.83 Therefore, the CO2 uptake capacity increases more significantly with pressure through Lewis acid–base interactions due to their stronger and more stable nature.
DFT findings show that the interaction energy between CO2 and the tertiary amine groups in PDMAPAm65 is three-times more than the interaction energy between CO2 and the oxygen of the carbonyl group in PMMA under dry conditions. This could be attributed to the fact that PDMAPAm has a greater CO2 uptake capacity than PMMA;109 however, this is not the case. Under dry conditions, the free electrons of the nitrogen in the tertiary amine groups of PDMAPAm are hindered by two methyl groups, and thus, the electrons of the tertiary amine groups cannot interact easily with CO2.54,110–112 However, the free electrons of the oxygen atoms of the carbonyl groups in the PMMA block are easily accessible to CO2, which indicates that these electrons are not hindered and the ester group provides very attractive interaction sites for CO2 molecules.113,114 This could explain the increase in the CO2 uptake capacity of the diblock copolymer with a PMMA block.
Effect of temperature on CO2 capture
Another consideration is that Fig. 2 explains the effect of adsorption temperature on the CO2 uptake capacity of the PDMAPAm homopolymer. The adsorption measurements with PDMAPAm were conducted at 25 °C and 30 °C because this interval reflects the typical ambient conditions at locations where DAC units are most likely to operate.115,116 It can be clearly seen that a higher temperature leads to a higher CO2 uptake capacity of the polymer, which was also mentioned in other publications,117 but the effect of temperature is not significantly high.Apart from this, 30 °C is very close to the glass transition temperature of the PDMAPAm257 polymer at 1 bar if it is exposed to CO2 (see Fig. 7). Therefore, above this temperature, it is possible that the CO2 adsorption capacity of PDMAPAm will not increase any further.
Simulation of dry CO2 physisorption model
To understand the kinetic mechanism of CO2 adsorption on the polymers, the CO2 adsorption isotherms of both the homopolymer and diblock copolymer were fit to two different adsorption models, the Langmuir and Freundlich models. The Langmuir and the Freundlich isotherm models were selected for fitting the CO2 adsorption data due to their widespread applicability and theoretical relevance. These models have been extensively validated in the literature for similar systems.14,118–122Fig. 3 shows the results of the fitting of the experimental CO2 uptake values at different pressures to the Langmuir and Freundlich isotherm models.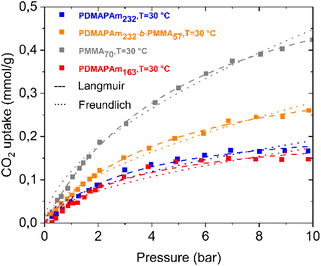 | ||
| Fig. 3 Kinetic model fitting for CO2 physisorption on the PDMAPAm and PMMA homopolymers and the PDMAPAm-b-PMMA diblock copolymer. | ||
It can be clearly seen from the fitting that the isotherms of both PDMAPAm and PDMAPAm-b-PMMA have a better fit with the Langmuir model than the Freundlich model. In particular, up to 7 bar, there is a good overlap between the simulated and experimental CO2 adsorption isotherms for both the homopolymer and diblock copolymer, but the model deviates from the data thereafter. The reason for the deviation is that the glass transition temperature of the PDMAPAm polymer decreases as it is exposed to dry CO2 at higher pressure, as shown in Fig. 7. Alternatively, when the PDMAPAm polymer is exposed to dry CO2 at this temperature and above 5 bar, it is no longer in a glassy state. Therefore, the adsorption no longer follows the Langmuir model.
The Langmuir model indicates that monolayer adsorption occurs at homogeneous sites on both PDMAPAm and PDMAPAm-b-PMMA.14 This model posits that the solid surface has a specific number of active sites, each capable of adsorbing only one adsorbate molecule (CO2). Equilibrium is reached when all these active sites are occupied by CO2 molecules, forming a monomolecular layer.123,124 Essentially, the Langmuir isotherm describes a gradual progression towards maximum adsorption, resulting in a monomolecular layer that completely covers the solid surface of both PDMAPAm and PDMAPAm-b-PMMA. The Langmuir model also assumes that the CO2 molecules adsorbed on the surface of the polymer material do not interact with each other, and the heat of adsorption is constant and independent of the degree of surface coverage.125,126
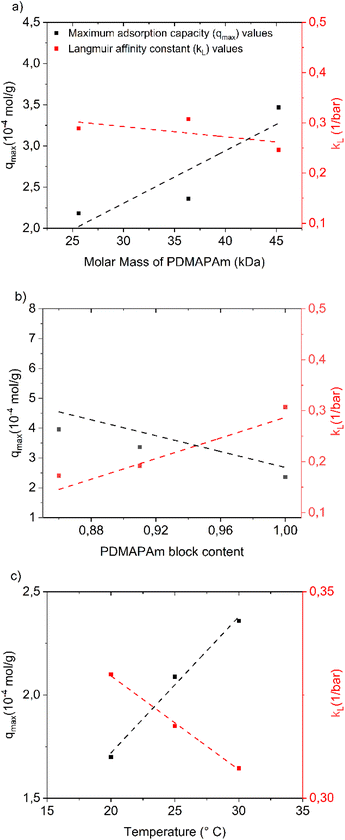
Moreover, from the fitting, the kinetic parameters of the Langmuir model, which are the equilibrium coefficient (kL) and the maximum CO2 capture capacity of the polymer (qmax), were estimated. With a variation in the molar mass of PDMAPAm, temperature, and the PMMA block content, the changes in the kinetic parameters of the Langmuir model were analysed, as shown in Fig. 4(a)–(c), respectively.
It can clearly be seen in Fig. 4 that as the molar mass of PDMAPAm, PMMA content, and adsorption temperature increase, qmax rises and kL decreases simultaneously. Particularly, in Fig. 4(b), the effect of PMMA block content on both kinetic parameters is larger than the effect of the molar mass of PDMAPAm in Fig. 4(a) and the adsorption temperature in Fig. 4(c). The mild dependence of adsorption on temperature and molar mass is consistent with the dominant solution–diffusion mechanism in dense polymer matrices, where gas solubility and free volume govern sorption, as described in the classical membrane-science literature.59,127 As shown in Fig. 4(b), the copolymers with larger hydrophobic PMMA domains exhibit higher adsorption capacities (qmax). This behavior is consistent with the role of PMMA in enhancing the polymer packing density and microphase-separated domain ordering, which promotes more accessible sorption pathways.128,129 This is supported by studies showing that PMMA blocks act as apolar segments driving the formation of well-defined, compact microphase-separated structures in block copolymers, where PMMA-rich domains increase structural ordering and influence transport pathways.128,130,131 At the same time, DFT results show that CO2 interacts with the tertiary amine groups65 in PDMAPAm about three-times more strongly than with the carbonyl oxygen109 in PMMA under dry conditions. Regardless, PDMAPAm does not exhibit higher CO2 uptake. This is likely because the nitrogen lone pair in the tertiary amine is sterically hindered by two methyl groups, limiting its ability to bind CO2.61,110–112 In contrast, the carbonyl oxygen in PMMA is more accessible, providing attractive interaction sites for CO2. This accessibility may explain why adding a PMMA block increases the CO2 uptake in the diblock copolymer.113,114
These effects of the polymer properties (molar mass of PDMAPAm and PMMA block content) and adsorption process variables (such as temperature and pressure in this case) on the kinetic parameters of the adsorption model can be used as an important bridge to combine polymer properties (which are mainly controlled by the polymerization kinetics42,50,67) with the adsorption behavior of the polymer.
Correlations between the surface and structural features of membrane materials and their performance arise from how the polymer morphology, free volume, and segmental mobility govern the gas diffusion pathways.132 Surface heterogeneity and the distribution of functional groups strongly affect sorption behavior by modulating the gas–polymer interactions on the molecular scale.132 Variations in chain packing density and local rigidity further determine the balance between diffusivity and solubility, thereby linking structural features directly to permeability and adsorption capacity.133 In block-copolymer-based or nanostructured membrane adsorbers, microphase-separated domains create selective transport channels, highlighting the importance of tailoring their structural architecture to enhance both uptake and selectivity.134
Desorption of CO2
In addition to evaluating the CO2 adsorption behavior of the PDMAPAm homopolymer and the PDMAPAm-b-PMMA diblock copolymer, the desorption process was investigated under dry conditions, and the corresponding results are presented in Fig. 5.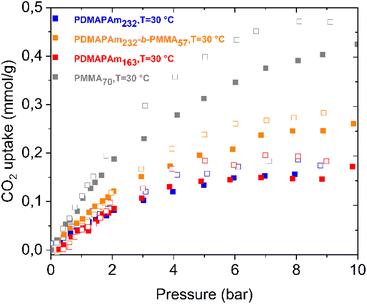 | ||
| Fig. 5 CO2 adsorption–desorption isotherms of PDMAPAm homopolymers with different molar masses, PMMA, and the PDMAPAm-b-PMMA diblock copolymer. | ||
Fig. 5 illustrates the CO2 adsorption and desorption isotherms of the PDMAPAm homopolymer and PDMAPAm-b-PMMA diblock copolymer. It can be clearly seen that the copolymer adsorbs more CO2 than the homopolymer, as mentioned before in the previous section. Additionally, hysteresis is observed between the adsorption and desorption isotherms of both the homopolymer and diblock copolymer. The isotherms of polymers were measured in powder form, and the desorption isotherms were higher than the sorption isotherms for both PDMAPAm and PDMAPAm-b-PMMA, indicating a notable hysteresis effect in the sorption-desorption process. This phenomenon can be attributed to either the trapping effect caused by the interaction of CO2 with the polymer or structural rearrangements. The hysteresis effect suggests that the sorbent/sorbate system is in a metastable state, and as pressure decreases, the gas is not released as readily as expected based on the thermodynamic equilibrium values.82 In detail, PMMA exhibits greater CO2 sorption hysteresis than PDMAPAm due to its glassy nature, which can lead to irreversible structural rearrangements and physical aging during desorption. The dual-mode sorption behavior and higher free volume in PMMA contribute to CO2 entrapment,135,136 whereas the more flexible and less glassy PDMAPAm allows for more reversible adsorption–desorption cycles.51,58
Cycling experiment
Since the polymers can also potentially be applied for long-term repeated adsorption–desorption processes in direct air capture technologies, it is very important to check their stability after several cycles of adsorption–desorption.In this part, five cycles of dry CO2 adsorption measurements were conducted at 1 bar and 30 °C for 1 h, whereas desorption was also performed for 1 h at 30 °C but under vacuum (0.001 bar). The multi-cycle performance of PDMAPAm232 and PDMAPAm232-b-PMMA57 is illustrated in Fig. 6(a) and (b), respectively.
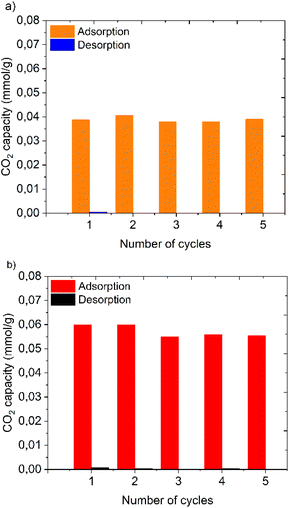 | ||
| Fig. 6 Change in CO2 uptake capacity of the polymer with the number of cycles. Adsorption–desorption cycling experiment on (a) PDMAPAm232 and (b) PDMAPAm232-b-PMMA57 at 1 bar and 30 °C. | ||
Fig. 6 shows that both PDMAPAm232 and PDMAPAm232-b-PMMA57 exhibited a stable CO2 uptake performance with very small fluctuations. The most crucial point is that both polymers maintained almost stable CO2 uptake capacities with the number of cycles. This might be related to the fact that both polymers interact with dry CO2 physically and do not form a chemical bond during adsorption, and therefore, the desorption process was performed at the same temperature as adsorption but at a pressure close to zero bar. This does not compromise the stability of the polymers, allowing them to be used for a long time after several cycles of adsorption–desorption.
Another important point is that both PDMAPAm and PDMAPAm-b-PMMA show very small hysteresis in the adsorption–desorption cycles, and the hysteresis fluctuates in each cycle. As mentioned before, the reason for the hysteresis is not due to the formation of chemical bonds but may be because the dry CO2, which was adsorbed, chose a different pathway during desorption than during adsorption.
High-pressure DSC
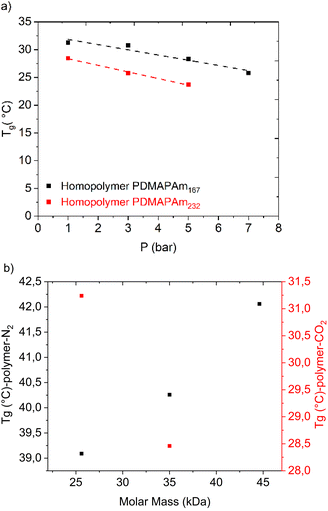
Furthermore, DSC analysis under N2 at 1 bar, as well as high-pressure DSC analysis with CO2 supplied at varying pressures, were conducted to monitor the change in the glass transition temperature of PDMAPAm. These measurements were performed with respect to both the molar mass of the homopolymer and the CO2 exposure pressure, and the results are presented in Fig. 7.Fig. 7(a) shows the glass transition temperatures (Tg) of PDMAPAm167 and PDMAPAm232 exposed to CO2 at different pressures, whereas Fig. 7(b) compares the glass transition temperatures of CO2-exposed PDMAPAm with that of N2-exposed PDMAPAm at different molar masses. It can be clearly seen that the Tg of the CO2-exposed polymer is less than the Tg of the N2-exposed polymer.
It is also crucial to note that the Tg of the N2-exposed PDMAPAm increases with the molar mass of the polymer, whereas the trend is reciprocal for the CO2-exposed PDMAPAm at 1 bar. As the molar mass of the polymer increases, the Tg of the N2-exposed polymer increases, whereas the Tg of the CO2-exposed polymer decreases, which means that the polymer interacts with CO2 differently than with N2. When PDMAPAm is exposed to dry CO2, the CO2 molecule physically interacts with the tertiary amine and amide groups of the polymer and attaches to them, and this interaction induces a plasticizing effect. These interactions reduce the intermolecular forces and increase the free volume within the polymer matrix, thereby enhancing the chain mobility and lowering the Tg.54,104 Another point is that PDMAPAm with a higher molar mass interacts with CO2 more. This trend totally agrees with the increasing trend of CO2 capture capacity with the molar mass of the polymer, as also mentioned before and demonstrated in Fig. 2. Another consideration is that the slope of the trend line of Tg change with pressure for the CO2-exposed polymer is almost the same as that for both homopolymers at different molar masses in the range of 1 bar to 7 bar.
Combination of kinetics for polymer synthesis with kinetics of physisorption (unified model)
To combine the polymerization kinetics67 with adsorption kinetics, the following case study is applied: the aim was to synthesize a homopolymer, PDMAPAm163, and its corresponding diblock copolymer, PDMAPAm163-b-PMMA30, and to simulate the adsorption behaviour of both the PDMAPAm163 homopolymer and PDMAPAm163-b-PMMA30 diblock copolymer considering physisorption phenomena.The kinetic model developed for the RAFT polymerization of dimethylaminopropylacrylamide (DMAPAm) is applied here to simulate the optimal synthesis recipe. The simulation showed that the optimal recipe for homopolymer (PDMAPAm) synthesis is RAFT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) initiator
initiator![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) monomer ratio of 1.2
monomer ratio of 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
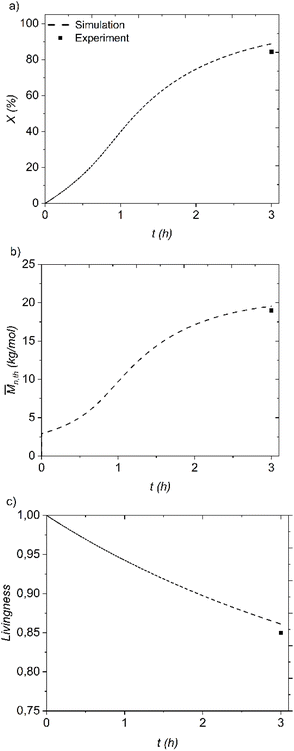
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 and at the same time, the model predicted the evolution of DMAPAm monomer conversion, number average molar mass and livingness of PDMAPAm for further chain extension with MMA to synthesize the PDMAPAm-b-PMMA diblock copolymer. The experiments for the synthesis of the homopolymer and diblock copolymer, PDMAPAm-b-PMMA, were conducted using the predicted optimal recipe to test whether the kinetic model for polymer synthesis is correct. The comparison between the simulation and experimental results is shown in Fig. 8, and the molar mass characterization results for PDMAPAm and PDMAPAm-b-PMMA are shown in Table 1.
200 and at the same time, the model predicted the evolution of DMAPAm monomer conversion, number average molar mass and livingness of PDMAPAm for further chain extension with MMA to synthesize the PDMAPAm-b-PMMA diblock copolymer. The experiments for the synthesis of the homopolymer and diblock copolymer, PDMAPAm-b-PMMA, were conducted using the predicted optimal recipe to test whether the kinetic model for polymer synthesis is correct. The comparison between the simulation and experimental results is shown in Fig. 8, and the molar mass characterization results for PDMAPAm and PDMAPAm-b-PMMA are shown in Table 1.
| Polymer |
![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) n,app (kDa)
n,app (kDa) |
![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) w,app (kDa)
w,app (kDa) |
Đ |
|---|---|---|---|
| PDMAPAm163 | 18.5 | 25.6 | 1.38 |
| PDMAPAm163-b-PMMA30 | 20.4 | 29.3 | 1.44 |
As can be clearly seen in Fig. 8 and Table 1, the kinetic model predicts the DMAPAm conversion, number-average molar mass, and livingness of PDMAPAm163 quite well, as evidenced by the good agreement between the simulated and experimental data.
The dry CO2 physisorption isotherm of the PDMAPAm163-b-PMMA30 polymer was simulated using the results from Fig. 4. Interpolation (also assuming that this adsorption follows the Langmuir physisorption model) can be applied to determine the kinetic parameters of adsorption, such as qmax and kL, and the CO2 adsorption isotherm of PDMAPAm-b-PMMA can be predicted. To prove whether the prediction by both Langmuir models is right, the dry CO2 adsorption isotherms of the PDMAPAm-b-PMMA diblock copolymer and PDMAPAm homopolymer were experimentally measured and compared with the simulated adsorption isotherm shown in Fig. 9.
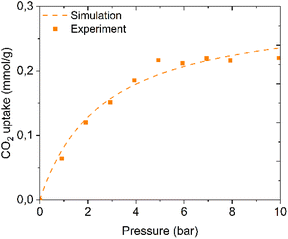 | ||
| Fig. 9 Comparison of the kinetic model prediction with the experimental results for dry CO2 adsorption isotherm on the PDMAPAm163-b-PMMA30 diblock copolymer. | ||
It is clear from both curves that the dry CO2 adsorption model accurately predicts the uptake capacity of the polymer, indicating that the physisorption model works correctly. In the dry CO2 adsorption case, there is a small deviation in the simulated isotherm from the experimental result, but it starts from a pressure of 5 bar, which is not as relevant to direct air capture because these polymers are planned to be applied at atmospheric pressure in DAC.
While the laboratory methodology developed in this study involves detailed kinetic modeling, polymer synthesis optimization, and membrane-adsorber characterization, the underlying workflow is highly scalable because each step aligns with established industrial practices in membrane manufacturing and polymer processing.137 The unified kinetic model offers a predictive design tool that reduces development time at the commercial scale by minimizing experimental trial-and-error during material selection and process optimization.138 Furthermore, the scalability of RAFT polymerization and membrane-fabrication techniques such as phase inversion or coating ensures that the materials described herein can be produced at industrially relevant throughputs.139 The ability to tailor the polymer composition digitally before synthesis contributes to significant cost savings by reducing raw-material waste, shortening development cycles, and enabling more sustainable manufacturing routes.140 Overall, the methodology supports commercial deployment by coupling material-level optimization with process efficiency, and its potential economic impact is aligned with global incentives for carbon-capture technologies and emerging climate-policy frameworks.141
Conclusion
This study provides a comprehensive investigation into the CO2 capture behavior of the CO2-responsive homopolymer PDMAPAm and its diblock copolymer PDMAPAm-b-PMMA, with a focus on their applicability as membrane adsorbers for direct air capture (DAC) technologies. Under dry conditions, CO2 was found to undergo physisorption, interacting primarily with the tertiary amine groups of PDMAPAm and the carbonyl functionalities of PMMA. This behavior was validated through gravimetric adsorption measurements, with the uptake capacities benchmarked against the existing literature.Beyond mechanistic characterization, this study explored the kinetics of CO2 adsorption and polymerization, incorporating physisorption pathways and polymerization kinetics into a unified model. This kinetic framework was integrated with polymerization kinetics to enable the rational design and optimization of the polymer properties. By varying the molar mass of the PDMAPAm block and the relative PMMA content, the influence of polymer composition on CO2 uptake was systematically assessed. This approach allowed for the identification of the most effective polymer architecture for DAC applications, balancing adsorption capacity with material property. Notably, the unified model predicted a target molar mass of 18.5 kDa for the PDMAPAm163 homopolymer and identified an optimal synthesis recipe of RAFT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) initiator
initiator![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) monomer ratio of 1.2
monomer ratio of 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200, corresponding to a polymerization time of 3 hours. The model also accurately forecasts the CO2-uptake capacity of the PDMAPAm163 homopolymer to be 0.05 mmol g−1 and that of the corresponding diblock copolymer (PDMAPAm163-b-PMMA30) to be approximately 0.1 mmol g−1 under dry ambient conditions.
200, corresponding to a polymerization time of 3 hours. The model also accurately forecasts the CO2-uptake capacity of the PDMAPAm163 homopolymer to be 0.05 mmol g−1 and that of the corresponding diblock copolymer (PDMAPAm163-b-PMMA30) to be approximately 0.1 mmol g−1 under dry ambient conditions.
Additionally, the impact of key process variables including temperature and pressure on CO2 capture performance was thoroughly examined. The optimal operating conditions were determined, providing valuable guidance for the deployment of these materials in real-world DAC systems.
In summary, this work not only advances the understanding of CO2-responsive polymer adsorption mechanisms but also establishes a robust framework for optimizing polymer design and process conditions. The findings underscore the promise of PDMAPAm-based materials in addressing the urgent need for effective DAC solutions and contribute to the broader efforts to mitigate atmospheric CO2 levels through innovative materials science.
Looking ahead, adsorption-based carbon removal will increasingly rely on materials that combine high CO2 uptake, low regeneration energy, and long-term cyclability under realistic atmospheric conditions. For PDMAPAm-based polymer membrane adsorbers, future research should focus on evaluating their performance under humid and variable-temperature environments, where the adsorption pathways or cooperative effects may further enhance their uptake. Integrating these materials into scalable membrane-adsorber modules, supported by techno-economic analysis and accelerated aging studies, will be essential to assess their practical viability in DAC operations. In parallel, expanding the unified kinetic model to include humidity-dependent equilibria, transport limitations, and multicomponent gas effects would provide a more predictive framework for guiding polymer design. Together, these efforts will help bridge the gap between laboratory-scale materials development and deployable DAC technologies.
Conflicts of interest
The authors declare no conflict of interest.Data availability
All data supporting this study are provided in the supplementary information (SI) and previously published datasets. Supplementary information: includes details such as the synthesis procedure, structural characterization, theoretical calculations, experimental methods, and spectral analyses. For access to the datasets, refer to DOI: https://www.doi.org/10.5281/zenodo.17959928. See DOI: https://doi.org/10.1039/d5ta10366e.Acknowledgements
This work was performed within the collaborative project DACStorE – Direct Air Capture and Storage (KA2-HSC-12), funded by the Initiative and Networking Fund of the Helmholtz Association. The authors specially thank Silvio Neumann for NMR and DSC experiments, Maren Brinkmann for the GPC measurements and Dr. Jelena Lillepaerg for assistance with the gravimetric sorption experiments. Dr Felix Kandelhard is gratefully acknowledged for discussions on the polymerization reaction kinetics. Scientific discussions with Prof. Dr Volker Abetz are also greatly acknowledged.References
- M. Bui, C. S. Adjiman, A. Bardow, E. J. Anthony, A. Boston, S. Brown, P. S. Fennell, S. Fuss, A. Galindo and L. A. Hackett, Energy Environ. Sci., 2018, 11, 1062–1176 RSC.
- C. M. White, B. R. Strazisar, E. J. Granite, J. S. Hoffman and H. W. Pennline, J. Air Waste Manage. Assoc., 2003, 53, 645–715 CrossRef CAS PubMed.
- P. C. Psarras, S. Comello, P. Bains, P. Charoensawadpong, S. Reichelstein and J. Wilcox, Environ. Sci. Technol., 2017, 51, 11440–11449 CrossRef CAS PubMed.
- X. Wang and C. Song, Front. Energy Res., 2020, 8, 560849 CrossRef.
- M. Erans, E. S. Sanz-Pérez, D. P. Hanak, Z. Clulow, D. M. Reiner and G. A. Mutch, Energy Environ. Sci., 2022, 15, 1360–1405 RSC.
- X. Zhu, W. Xie, J. Wu, Y. Miao, C. Xiang, C. Chen, B. Ge, Z. Gan, F. Yang and M. Zhang, Chem. Soc. Rev., 2022, 51, 6574–6651 RSC.
- L. Küng, S. Aeschlimann, C. Charalambous, F. McIlwaine, J. Young, N. Shannon, K. Strassel, C. N. Maesano, R. Kahsar and D. Pike, Energy Environ. Sci., 2023, 16, 4280–4304 RSC.
- J. Kotowicz, K. Niesporek and O. Baszczeńska, Energies, 2025, 18, 496 CrossRef CAS.
- P. Webb, H. Muslemani, M. Fulton and N. Curson, Scaling Direct Air Capture (DAC): A Moonshot or the Sky's the Limit?, OIES Paper, CM, 2023 Search PubMed.
- Y. Zhang, J. Feng, L. Li, S. Zhao, C. Wu, Z. Huang and H. Lin, Front. Energy, 2025, 1–13 Search PubMed.
- D. Panda, V. Kulkarni and S. K. Singh, React. Chem. Eng., 2023, 8, 10–40 RSC.
- J. Young, F. Mcilwaine, B. Smit, S. Garcia and M. Van der Spek, Chem. Eng. J., 2023, 456, 141035 CrossRef CAS.
- U. I. Premadasa, N. Kumar, D. Stamberga, V. Bocharova, J. T. Damron, T. Li, S. Roy, Y.-Z. Ma, V. S. Bryantsev and B. Doughty, J. Chem. Phys., 2024, 161, 164707 CrossRef CAS PubMed.
- J. Serafin and B. Dziejarski, Microporous Mesoporous Mater., 2023, 354, 112513 CrossRef CAS.
- S. Deutz and A. Bardow, Nat. Energy, 2021, 6, 203–213 CrossRef CAS.
- A. Ward, M. M. Papathanasiou and R. Pini, Adsorption, 2024, 30, 1829–1848 CrossRef CAS.
- K. An, K. Li, C.-M. Yang, J. Brechtl and K. Nawaz, J. CO2 Util., 2023, 76, 102587 CrossRef CAS.
- M. Soukri, P. Sitaula, D. O'Nolan, M. G. Izenson and S. D. Phillips, Development of Advanced Solid Sorbents for Direct Air Capture, RTI International, Research Triangle Park, NC (United States), 2023 Search PubMed.
- C. Zhang, J. Fang, X. Xu, M. Zhang, Z. Han and J. Liao, Ann. N. Y. Acad. Sci., 2025, 1552, 388–400 CrossRef CAS PubMed.
- D. Li, S. Ning, L. Yu, F. Jiang, D. Zhao, S. Zhang, M. Liao, Q. Meng, Q. Fang and H. Kang, Adv. Mater., 2025, 2501622 CrossRef CAS PubMed.
- G. Zhao, H. Gao, Z. Qu, H. Fan and H. Meng, Nat. Commun., 2023, 14, 7624 CrossRef CAS PubMed.
- X. Ju, X. Feng, X. Duan, B. Cui, G. Huang, Y. Chen, X. Tong, Z. Yang, P. Guo and S. Xu, Sep. Purif. Technol., 2025, 136443 Search PubMed.
- F. Yan, H. Chen, T. Chi, J. Lu, X. Shen, F. Xie, P. Wang and Z. Zhang, Chem. Eng. J., 2025, 166450 CrossRef CAS.
- X. Li, J. Yu, H. He, Z. Peng, J. Pu, L. Zheng, A. Chen, J. Qian, F. Huang and X. Gu, Langmuir, 2025, 41, 29180–29195 CrossRef CAS PubMed.
- L. B. Hamdy, C. Goel, J. A. Rudd, A. R. Barron and E. Andreoli, Mater. Adv., 2021, 2, 5843–5880 RSC.
- R. B. Said, J. M. Kolle, K. Essalah, B. Tangour and A. Sayari, ACS Omega, 2020, 5, 26125–26133 CrossRef CAS PubMed.
- R. Zhang and J. Zhou, Clean Energy Sci. Technol., 2024, 2, 126 CrossRef.
- E. Wang, R. Navik, Y. Miao, Q. Gao, D. Izikowitz, L. Chen and J. Li, Cell Rep. Phys. Sci., 2024, 101791 CrossRef.
- Z. Wan, R. Hunt, C. White, J. Gillbanks, J. Czapla, G. Xiao, S. Surin and C. Wood, ChemSusChem, 2024, e202400212 CrossRef CAS PubMed.
- S. H. Pang, R. P. Lively and C. W. Jones, ChemSusChem, 2018, 11, 2628–2637 CrossRef CAS PubMed.
- K. Li, J. Jiang, F. Yan, S. Tian and X. Chen, Appl. Energy, 2014, 136, 750–755 CrossRef CAS.
- C. Zhang, J. Zhou, X. Ye, Z. Li and Y. Wang, J. Membr. Sci., 2022, 641, 119928 CrossRef CAS.
- M.-S. Lebrun-Chopra, The Development of Novel Bio-Based CO2-Responsive Polymers for Binder Applications, Queen's University, Canada, 2021 Search PubMed.
- Y.-T. Shieh, Y.-T. Lin and C.-C. Cheng, Carbohydr. Polym., 2017, 170, 281–288 CrossRef CAS PubMed.
- K. Bauri, A. Pan, U. Haldar, A. Narayanan and P. De, J. Polym. Sci., Part A: Polym. Chem., 2016, 54, 2794–2803 CrossRef CAS.
- A. Riabtseva, S. N. Ellis, P. Champagne, P. G. Jessop and M. F. Cunningham, Ind. Eng. Chem. Res., 2021, 60, 9807–9816 CrossRef CAS.
- M. F. Cunningham and P. G. Jessop, Macromol. React. Eng., 2022, 16, 2200031 CrossRef CAS.
- M. F. Cunningham and P. G. Jessop, Chem. Commun., 2023, 59, 13272–13288 RSC.
- M. F. Cunningham and P. G. Jessop, Eur. Polym. J., 2016, 76, 208–215 CrossRef CAS.
- W. Ji, D. Panus, R. N. Palumbo, R. Tang and C. Wang, Biomacromolecules, 2011, 12, 4373–4385 CrossRef CAS PubMed.
- S. Demirci, S. D. Sutekin, O. Guven and N. Sahiner, Int. J. Hydrogen Energy, 2023, 48, 23002–23012 CrossRef CAS.
- E. Pashayev, F. Kandelhard and P. Georgopanos, Macromol. React. Eng., 2023, 17, 2200068 CrossRef CAS.
- R. Du, A. Chakma and X. Feng, J. Membr. Sci., 2007, 290, 19–28 CrossRef CAS.
- Z. Zhou, T. Liu, A. U. Khan and G. Liu, Sci. Adv., 2019, 5, eaau6852 CrossRef PubMed.
- A. Esmeraldo Paiva, J. F. Baez Vasquez, A. Selkirk, N. Prochukhan, F. GL Medeiros Borsagli and M. Morris, ACS Appl. Mater. Interfaces, 2022, 14, 35265–35275 CrossRef CAS PubMed.
- K. Nieswandt, P. Georgopanos and V. Abetz, Polym. Chem., 2021, 12, 2210–2221 RSC.
- R. Shevate, M. Kumar, M. Karunakaran, C. Canlas and K.-V. Peinemann, J. Mater. Chem. A, 2018, 6, 4337–4345 RSC.
- L. Tsaur and U. B. Wiesner, Polymers, 2023, 15, 2020 CrossRef CAS PubMed.
- F. Kandelhard, K. Schuldt, J. Schymura, P. Georgopanos and V. Abetz, Macromol. React. Eng., 2021, 15, 2000058 CrossRef CAS.
- F. Kandelhard, E. Pashayev, J. Schymura and P. Georgopanos, Ind. Eng. Chem. Res., 2023, 62, 8696–8708 CrossRef CAS.
- E. Pashayev and P. Georgopanos, Macromol. Mater. Eng., 2025, 310, 2400290 CrossRef CAS.
- T. Tiainen, J. K. Mannisto, H. Tenhu and S. Hietala, Langmuir, 2021, 38, 5197–5208 CrossRef PubMed.
- S. Lin and P. Theato, Macromol. Rapid Commun., 2013, 34, 1118–1133 CrossRef CAS PubMed.
- M. Zhao, L. Huang, Y. Gao, Z. Wang, S. Liang, X. Zhu, Q. Wang, H. He and D. O'Hare, Nano-Micro Lett., 2025, 17, 1–19 CAS.
- J. Y. Bae, S. G. Jang, J. Cho and M. Kang, Int. J. Mol. Sci., 2025, 26, 4313 CrossRef CAS PubMed.
- B. Zhang, C. Zheng, F. S. Bates and T. P. Lodge, ACS Appl. Polym. Mater., 2023, 5, 2223–2229 CrossRef CAS.
- D. J. Lohse and N. Hadjichristidis, Curr. Opin. Colloid Interface Sci., 1997, 2, 171–176 CrossRef CAS.
- E. Pashayev and P. Georgopanos, Chem. Ing. Tech., 2025, 97, 703–709 CrossRef CAS.
- R. W. Baker, Membrane Technology and Applications, John Wiley & Sons, 2023 Search PubMed.
- Y. Chen, K.-J. Lu, S. Japip and T.-S. Chung, Environ. Sci. Technol., 2020, 54, 12713–12722 CrossRef CAS PubMed.
- K. Sumida, D. L. Rogow, J. A. Mason, T. M. McDonald, E. D. Bloch, Z. R. Herm, T.-H. Bae and J. R. Long, Chem. Rev., 2012, 112, 724–781 CrossRef CAS PubMed.
- G. Firpo, E. Angeli, P. Guida, D. Pezzuoli, D. Repetto, L. Repetto and U. Valbusa, Polymers, 2019, 11, 910 CrossRef PubMed.
- J. Zhou, M. Deissenroth-Uhrig and M. Gallei, Adv. Funct. Mater., 2025, e20959 Search PubMed.
- A. K. Sekizkardes, P. Wang, J. Hoffman, S. Budhathoki and D. Hopkinson, Mater. Adv., 2022, 3, 6668–6686 Search PubMed.
- H. Liu, H. Yin and Y. Feng, Des. Monomers Polym., 2017, 20, 363–367 CrossRef CAS PubMed.
- A. L. Krasovskiy, E. Auyeung, B. D. Stubbert, A. V. Davis, J. Hou and C. H. Cummins, Process for preparing olefin-acrylate block copolymers, WO2021222694A1, 2023.
- E. Pashayev and P. Georgopanos, Polymers, 2025, 17, 1115 CrossRef CAS PubMed.
- G. Desmet, N. Rybel and P. Steenberge, Macromol. Rapid Commun., 2017, 39, 1700403 CrossRef PubMed.
- A. Spiteri, B. Megerle, A. Calbry-Muzyka, N. Casas and J. A. Wurzbacher, Method for capture of carbon dioxide from ambient air and corresponding adsorber structures with a plurality of parallel surfaces, WO2021239747A1, 2023.
- S. M. Lalji, M. Ayubi, S. I. Ali, S. u. Rahman and M. Mustafa, Discov. Chem., 2024, 1, 71 CrossRef.
- Q. Grossmann, P. A. Saenz-Cavazos, N. Ferru, D. R. Williams and M. Mazzotti, Ind. Eng. Chem. Res., 2025, 64, 7165–7175 CrossRef CAS PubMed.
- R. P. S. Gautam and S. Sahoo, Sādhanā, 2023, 48, 27 CrossRef CAS.
- M. O. Prache, Institutt for Kjemisk Prosessteknologi, 2012 Search PubMed.
- J. Hwang, H. Azzan, R. Pini and C. Petit, J. Chem. Eng. Data, 2022, 67, 1674–1686 CrossRef CAS.
- X. Yang, R. Kleinrahm, M. O. McLinden and M. Richter, Int. J. Thermophys., 2023, 44, 169 CrossRef CAS.
- Q. Li, X. He, Y. Cui, P. Shi, S. Li and W. Zhang, Polym. Chem., 2015, 6, 70–78 RSC.
- Q.-l. Li, L. Li, H.-s. Wang, R. Wang, W. Wang, Y.-j. Jiang, Q. Tian and J.-p. Liu, Chin. J. Polym. Sci., 2017, 35, 66–77 CrossRef CAS.
- Q. Li, L. Li, W. Wang, X. Zhang, S. Li, Q. Tian and J. Liu, RSC Adv., 2016, 6, 45305–45314 RSC.
- F. Monemian and A. Kargari, J. Polym. Environ., 2025, 33, 4921–4941 CrossRef CAS.
- E. Orestes, C. M. Ronconi and J. W. de Mesquita Carneiro, Phys. Chem. Chem. Phys., 2014, 16, 17213–17219 RSC.
- M. Haris, A. Aziz, M. Sohail and W. Sardar, Environ. Sci. Pollut. Res., 2025, 1–19 Search PubMed.
- S. Zulfiqar, M. I. Sarwar and C. T. Yavuz, RSC Adv., 2014, 4, 52263–52269 RSC.
- M. N. Khan, Y. van Ingen, T. Boruah, A. McLauchlan, T. Wirth and R. L. Melen, Chem. Sci., 2023, 14, 13661–13695 RSC.
- R. Wissinger and M. Paulaitis, J. Polym. Sci., Part B:Polym. Phys., 1987, 25, 2497–2510 CrossRef CAS.
- J. Kim, K. H. Kim, Y. Ryu and S. W. Cha, Polymers, 2022, 14, 596 CrossRef CAS PubMed.
- R. G. Wissinger and M. E. Paulaitis, Ind. Eng. Chem. Res., 1991, 30, 842–851 CrossRef CAS.
- J. H. Aubert, J. Supercrit. Fluids, 1998, 11, 163–172 CrossRef CAS.
- D. Liu, H. Li, M. S. Noon and D. L. Tomasko, Macromolecules, 2005, 38, 4416–4424 CrossRef CAS.
- K. F. Webb and A. S. Teja, Fluid Phase Equilib., 1999, 158, 1029–1034 CrossRef.
- E. Kiran, J. A. Sarver and J. C. Hassler, J. Supercrit. Fluids, 2022, 185, 105378 CrossRef CAS.
- J. Fried and W. Li, J. Appl. Polym. Sci., 1990, 41, 1123–1131 CrossRef CAS.
- M. R. Nelson and R. F. Borkman, J. Phys. Chem. A, 1998, 102, 7860–7863 CrossRef CAS.
- Y.-T. Shieh and K.-H. Liu, J. Supercrit. Fluids, 2003, 25, 261–268 CrossRef CAS.
- M. Takahashi, Y. Yamamoto, A. V. Nawaby and Y. P. Handa, J. Polym. Sci., Part B:Polym. Phys., 2003, 41, 2214–2217 CrossRef CAS.
- Y.-T. Shieh and K.-H. Liu, J. Polym. Res., 2002, 9, 107–113 CrossRef CAS.
- E. Ricci, M. G. De Angelis and M. Minelli, Chem. Eng. J., 2022, 435, 135013 CrossRef CAS.
- M. A. Blatchford, P. Raveendran and S. L. Wallen, J. Phys. Chem. A, 2003, 107, 10311–10323 CrossRef CAS.
- Y. G. Ko, S. S. Shin and U. S. Choi, J. Colloid Interface Sci., 2011, 361, 594–602 CrossRef CAS PubMed.
- P. Bollini, S. Choi, J. H. Drese and C. W. Jones, Energy Fuels, 2011, 25, 2416–2425 CrossRef CAS.
- K. Inkong, P. Rangsunvigit, T. Chaisuwan and S. Kulprathipanja, Chem. Eng. Trans., 2016, 52, 103–108 Search PubMed.
- C. Pornjariyawatch, V. Jitchum, K. Assawatwikrai, P. Leepakorn, M. Probst, B. Boekfa, T. Maihom and J. Limtrakul, ChemPhysChem, 2025, 26, e202400754 CrossRef PubMed.
- W. Jiang, Y. Lin, C. Sun, Y. Sun and Y. Zhu, Molecules, 2024, 29, 4618 CrossRef CAS PubMed.
- X. Sun, X. Shen, H. Wang, F. Yan, J. Hua, G. Li and Z. Zhang, Nat. Commun., 2024, 15, 5068 CrossRef CAS PubMed.
- J. Hack, N. Maeda and D. M. Meier, ACS Omega, 2022, 7, 39520–39530 CrossRef CAS PubMed.
- S. Lin, K. R. Storme, Y.-C. M. Wu, F. M. Benedetti, T. M. Swager and Z. P. Smith, J. Membr. Sci., 2023, 668, 121194 CrossRef CAS.
- F. M. Benedetti, Y.-C. M. Wu, S. Lin, Y. He, E. Flear, K. R. Storme, C. Liu, Y. Zhao, T. M. Swager and Z. P. Smith, JACS Au, 2022, 2, 1610–1615 CrossRef CAS PubMed.
- M. Ciulla, V. Canale, R. Wolicki, S. Pilato, P. Bruni, S. Ferrari, G. Siani, A. Fontana and P. Di Profio, Separations, 2023, 10, 505 CrossRef CAS.
- C. Li, D. Lu and C. Wu, Phys. Chem. Chem. Phys., 2020, 22, 11354–11361 RSC.
- T. Xu, Z. Wang, T. Liu, F. Wang and Y. Jing, J. Am. Chem. Soc., 2025, 147, 26969–26979 CrossRef CAS PubMed.
- R. Navik, E. Wang, X. Ding, K. Qiu and J. Li, Environ. Chem. Lett., 2024, 22, 1791–1830 CrossRef CAS.
- P. Singh, J. P. Niederer and G. F. Versteeg, Chem. Eng. Res. Des., 2009, 87, 135–144 CrossRef CAS.
- X. Deng, Y. Han, L.-C. Lin and W. W. Ho, Sep. Purif. Technol., 2022, 299, 121601 CrossRef CAS.
- S. Kilic, S. Michalik, Y. Wang, J. K. Johnson, R. M. Enick and E. J. Beckman, Macromolecules, 2007, 40, 1332–1341 CrossRef CAS.
- S. Kilic, S. Michalik, Y. Wang, J. Johnson, R. Enick and E. Beckman, Ind. Eng. Chem. Res., 2003, 42, 6415–6424 CrossRef CAS.
- H. Schellevis, J. De La Combé and D. Brilman, Energy Adv., 2024, 3, 1678–1687 RSC.
- X. Cai, M. A. Coletti, D. S. Sholl and M. R. Allen-Dumas, JACS Au, 2024, 4, 1883–1891 CrossRef CAS PubMed.
- A. A. Al-Absi, A. Domin, M. Mohamedali, A. M. Benneker and N. Mahinpey, Fuel, 2023, 333, 126401 CrossRef CAS.
- M. Mabuza, K. Premlall and M. O. Daramola, Int. J. Coal Sci. Technol., 2022, 9, 45 CrossRef CAS.
- B. Dziejarski and A. Kisiela-Czajka, Mach., Technol., Mater., 2021, 15, 299–302 CAS.
- M. Jiang, J. Wang, L. Li, K. Pan and B. Cao, RSC Adv., 2013, 3, 20625–20632 RSC.
- S. Fan, J. Chen, C. Fan, G. Chen, S. Liu, H. Zhou, R. Liu, Y. Zhang, H. Hu and Z. Huang, J. Hazard. Mater., 2021, 416, 126225 CrossRef CAS PubMed.
- J. Qu, L. Chang, M. Liu, B. Cao, M. Li, Q. Yang and W. Gong, Polymers, 2022, 14, 4187 CrossRef CAS PubMed.
- I. Langmuir, J. Am. Chem. Soc., 1916, 38, 2221–2295 CrossRef CAS.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- S. Kundu and A. K. Gupta, Chem. Eng. J., 2006, 122, 93–106 CrossRef CAS.
- K. Vijayaraghavan, T. Padmesh, K. Palanivelu and M. Velan, J. Hazard. Mater., 2006, 133, 304–308 CrossRef CAS PubMed.
- D. R. Paul, Polymeric Gas Separation Membranes, CRC press, 2018 Search PubMed.
- G. K. V. Saraiva, V. V. de Souza, L. C. de Oliveira, M. L. d. C. Noronha, J. C. Masini, H. Chaimovich, R. K. Salinas, F. H. Florenzano and I. M. Cuccovia, Colloid Polym. Sci., 2019, 297, 557–569 CrossRef CAS.
- X. Han, N. Gong, X. Si, C. Jiang, J. Xu, S. Sun, P. Zhan and J. Mu, J. Polym. Res., 2023, 30, 223 CrossRef CAS.
- F. Li, M. Schellekens, J. de Bont, R. Peters, A. Overbeek, F. A. Leermakers and R. Tuinier, Macromolecules, 2015, 48, 1194–1203 CrossRef CAS.
- A. Khaydarov, I. W. Hamley, T. M. Legge and S. Perrier, Eur. Polym. J., 2007, 43, 789–796 CrossRef CAS.
- M. A. Otmi, F. Willmore and J. Sampath, Macromolecules, 2023, 56, 9042–9053 CrossRef CAS.
- A. F. Ismail, T. D. Kusworo, A. Mustafa and H. Hasbulla, Understanding the Solution-Diffusion Mechanism in Gas Separation Membrane for Engineering Students, 2005 Search PubMed.
- T. Corrado and R. Guo, Mol. Syst. Des. Eng., 2020, 5, 22–48 RSC.
- R. K. Arya, D. Thapliyal, J. Sharma and G. D. Verros, Coatings, 2021, 11, 1049 CrossRef CAS.
- N. Berrahou, A. Mokaddem, B. Doumi, S. Hiadsi, N. Beldjoudi and A. Boutaous, Polym. Bull., 2016, 73, 3007–3017 CrossRef CAS.
- S. H. Alkandari and B. Castro-Dominguez, Front. Membr. Sci. Technol., 2024, 3, 1390599 CrossRef.
- M. Gaulke, V. Guschin, S. Knapp, S. Pappert and W. Eckl, Microporous Mesoporous Mater., 2016, 233, 39–43 CrossRef CAS.
- S. Perrier, Macromolecules, 2017, 50, 7433–7447 CrossRef CAS.
- U. Han, T. Kang, J. Im and J. Hong, Chem. Commun., 2022, 58, 10953–10956 RSC.
- M. Ho, V. Barahimi and E. Croiset, The State of Direct Air Capture Technology and Industry, Waterloo Climate Institute, University of Waterloo, Waterloo, Ontario, Canada, 2025 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |