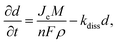Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Toward unified interphase engineering: the solid-electrolyte interphase in batteries and supercapacitors
Mehedi
Hasan
 *a,
Ishtiaq
Murshed
b,
Khayrul
Islam
*a,
Ishtiaq
Murshed
b,
Khayrul
Islam
 *c and
A. K. M.
Masud
d
*c and
A. K. M.
Masud
d
aDepartment of Industrial and Production Engineering, Bangladesh University of Engineering and Technology, Dhaka–1000, Bangladesh. E-mail: mehedihasanbuet29@gmail.com
bDepartment of Chemical Engineering, Bangladesh University of Engineering and Technology, Dhaka–1000, Bangladesh
cComputational Engineering Department, Lawrence Livermore National Laboratory, Livermore, CA 94550, USA
dDepartment of Industrial and Production Engineering, Bangladesh University of Engineering and Technology, Dhaka, Bangladesh
First published on 1st April 2026
Abstract
The development of next-generation electrochemical energy storage requires devices that synergistically combine the high energy density of batteries with the exceptional power capability and cycle life of supercapacitors, yet fundamental understanding of the interfacial phenomena governing performance across these platforms remains fragmented. While the solid-electrolyte interphase (SEI), a nanometer-scale passivation layer formed by electrolyte decomposition, has been extensively characterized in battery systems, analogous interfacial films in supercapacitors have received limited systematic investigation despite mounting experimental evidence for their existence and functional significance. This review advances the thesis that SEI formation constitutes a general electrochemical phenomenon arising whenever applied potentials drive electrode Fermi levels into electrolyte molecular orbitals beyond thermodynamic stability limits, independent of whether charge storage proceeds via faradaic redox or non-faradaic electrostatic mechanisms. Distinctions between battery SEIs (10–100 nm thickness, quasi-static evolution) and supercapacitor interphases (1–10 nm, dynamic reconstruction) reflect operational boundary conditions (potential range, cycling frequency, and ion flux) rather than fundamental mechanistic divergence. Quantitative analysis across reported studies indicates that interphase ionic resistivity (or fitted RSEI) generally increases as the interphase becomes more inorganic-rich (e.g., LiF/Li2CO3-dominated), although the reported ranges depend strongly on morphology/porosity, thickness, and the EIS fitting model and protocol Critically, rationally engineered interphases achieved through electrolyte additive optimization, atomic-layer-deposited protective coatings, or electrode surface functionalization substantially suppress parasitic leakage currents, maintain capacitance retention above 95% through tens of thousands of cycles, and enable stable operation exceeding 3.0 V in organic and ionic-liquid electrolytes. By establishing shared mechanistic principles and facilitating systematic knowledge transfer between battery and supercapacitor research communities, this unified framework enables predictive interphase engineering strategies that deliver battery-level energy density, capacitor-level power capability, and ultralong operational lifetimes for electrified transportation, renewable grid integration, and sustainable infrastructure.
1 Introduction
The accelerating global transition toward electrification and renewable energy integration has driven the relentless pursuit of advanced electrochemical energy-storage technologies.1–3 From smartphones and laptops to electric vehicles and grid-scale buffering, the demand for reliable, efficient, and sustainable energy-storage devices continues to rise.4,5 At the core of this technological revolution lie two complementary families of systems: rechargeable batteries and supercapacitors, each occupying a distinct yet overlapping domain of the energy–power landscape.6,7 Batteries, exemplified by lithium-ion and sodium-ion chemistries, deliver high energy density (100–300 Wh kg−1) through bulk faradaic redox reactions, while supercapacitors, or electrochemical capacitors, provide rapid charge–discharge capability (up to 10 kW kg−1) and ultralong cycle life (exceeding 106 cycles) through surface-controlled charge accumulation.8–10 The synergistic coexistence of these devices forms the backbone of modern electrochemical energy storage, and the quest to merge their advantages has sparked immense scientific and industrial interest (Fig. 1 and 2).11,12 | ||
| Fig. 1 Graphical abstract illustrating the unified interphase engineering framework bridging (a) super capacitors and (b) batteries. | ||
The concept of an interfacial passivation layer in electrochemical systems dates back to the pioneering work of Emanuel Peled in 1979, who first described the solid-electrolyte interphase (SEI) on alkali-metal electrodes.13 Peled observed that a spontaneously formed, nanometer-scale film (composed of electrolyte decomposition products) electronically insulates the electrode while permitting selective ion transport, thereby preventing continuous electrolyte breakdown.14 This discovery revolutionized battery science, establishing the SEI as an indispensable component governing cycle life, coulombic efficiency, and safety in lithium-ion, sodium-ion, and lithium–metal batteries.15–17
Over the ensuing decades, intensive research effort has been directed toward elucidating SEI formation mechanisms, chemical composition, morphology, and dynamic evolution in rechargeable batteries.18–20 Techniques such as X-ray photoelectron spectroscopy (XPS), cryogenic transmission electron microscopy (cryo-TEM), and operando electrochemical quartz-crystal microbalance (EQCM) have revealed that battery SEIs are typically 10–100 nm thick, composed of inorganic salts (e.g., LiF, Li2CO3, Li2O) in the inner region and polymeric organic species (e.g., polycarbonates, alkoxides) toward the outer surface.21–23 These layers evolve slowly over thousands of cycles and must endure significant mechanical stress due to volumetric changes in active materials during lithiation/delithiation.24
In contrast, the interfacial chemistry of supercapacitors has long been assumed to be purely non-faradaic, dominated by electrostatic ion adsorption at the electric double layer (EDL) without chemical bond formation or electrolyte decomposition.25,26 However, recent high-resolution spectroscopic and microscopic investigations have challenged this assumption, revealing the presence of ultrathin (1–10 nm), dynamic SEI-like films on activated-carbon, graphene, and MXene electrodes cycled at potentials above 2.5–3.0 V in organic, ionic-liquid, and even aqueous “water-in-salt” electrolytes.27–30 These films share compositional similarities with battery SEIs (including inorganic fluorides, carbonates, and polymeric species) but exhibit greater structural reversibility, elasticity, and continuous reconstruction under rapid charge–discharge cycling.31–34
Despite mounting experimental evidence for SEI formation in supercapacitors, the phenomenon remains poorly understood and lacks a unified theoretical framework. Several critical questions remain unanswered: (i) What are the thermodynamic and kinetic conditions that trigger SEI nucleation in non-faradaic systems? (ii) How do the composition, thickness, and morphology of supercapacitor SEI-like interphases differ from those in batteries, and what governs these differences? (iii) Can mechanistic insights and engineering strategies developed for battery SEIs (such as electrolyte additive design, artificial interphase coatings, and controlled formation protocols) be systematically translated to supercapacitors? (iv) How does the dynamic, self-healing nature of supercapacitor SEI-like interphases influence long-term cycling stability, self-discharge, and power performance? (Fig. 3).
 | ||
| Fig. 3 Historical development and evolving structural/mechanistic understanding of the solid electrolyte interphase (SEI) on negative electrodes in lithium-based batteries. The left panel presents a timeline of key milestones in SEI research: (a) the early observation of a passivation layer on lithium by Dey in the 1970s,35 (b) the formal introduction of the SEI concept by Peled in 1979,13 (c) identification of SEI chemistry, including Li2CO3, by Nazri and Muller and Aurbach et al.,36 (d) confirmation of SEI formation on graphite in 1990,37 (e) development of the multicomponent, multilayer SEI concept and equivalent-circuit interpretation by Peled,38 (f) mechanistic description of SEI formation initiated by electrolyte reduction from Aurbach et al.,39 (g) quantum-chemical calculations of SEI-forming reactions,40 (h) continuum modeling of SEI growth by Christensen et al.,41 (i) artificial SEI design by atomic layer deposition (ALD) on graphite, demonstrated by Jung et al.,42 (j) the two-layer/two-mechanism diffusion model proposed by Shi et al.,43 (k) direct visualization of the 3D/multilayer SEI by in situ electrochemical AFM from Cresce et al.,44 and (l) ALD-protected lithium metal reported by Kozen et al.45 The right panel summarizes representative models and consequences of this evolution, including the Aurbach multilayer SEI model (Reproduced from ref. 36 with permission from Elsevier, copyright 2014), the Peled mosaic model (Reproduced from ref. 38 with permission from IOP Publishing, Copyright 1997), the two-layer diffusion mechanism across organic and inorganic domains (Reproduced from ref. 43 with persmission from American Chemical Society, Copyright 2012), and the beneficial effect of ALD-derived artificial SEI layers on electrochemical durability (Reproduced from ref. 45 with persmission from American Chemical Society, Copyright 2015). | ||
To keep the Introduction focused on the unified framework developed in this review, we summarize the historical context of SEI briefly. Since early observations of passivation on alkali-metal electrodes and Peled's formal SEI concept,13 decades of battery research have established how electrolyte decomposition produces ionically conductive yet electronically insulating interphases,46 and how such layers govern efficiency, impedance growth, and lifetime.46,47 Here we leverage that mature battery baseline primarily to motivate and interpret supercapacitor-relevant interphase evidence, rather than re-review battery history in detail.
Current literature on supercapacitor interphases is fragmented across materials science, electrochemistry, and device engineering, with limited cross-referencing to the extensive body of battery SEI research.48–50 Moreover, the terminology is inconsistent: some authors refer to “interfacial films,” “surface layers,” or “electrolyte decomposition products” without explicitly invoking the SEI concept.51,52 This conceptual disconnect has hindered the development of predictive models, unified characterization protocols, and transferable design principles that could accelerate supercapacitor performance optimization.53
This review advances a cross-platform interphase framework in which electrolyte-driven passivation is treated as a shared interfacial response, while explicitly accounting for the device-specific boundary conditions that differentiate batteries from supercapacitors (e.g., pore-scale fields, rapid cycling, and defect-localized reactions).54–56 Rather than re-stating battery SEI fundamentals, we focus on translating battery-derived concepts into supercapacitor-specific evidence, metrics, and actionable design rules across EDLC, pseudocapacitor, and hybrid systems. The distinctions between battery SEIs and supercapacitor SEI-like interphases (thickness, composition, dynamics, and mechanical properties) reflect differences in operating conditions (potential range, cycling frequency, ion flux) rather than fundamental differences in formation mechanisms.16,20
Throughout this review, we use interphase as the umbrella term for electrode–electrolyte films and near-surface regions. We reserve SEI for the classical solid–electrolyte interphase in batteries. For supercapacitors, where films are often ultrathin, dynamic, and condition-dependent, we use SEI-like interphase (or SEI-like film) to denote decomposition-derived passivation layers. When discussing cross-platform similarities, we write interphase or SEI/SEI-like interphase explicitly to avoid ambiguity. Concretely, this framework is operationalized through: (i) a descriptor-to-metric mapping linking interphase signatures to leakage/self-discharge, ESR evolution, and capacitance retention; (ii) actionable design rules identifying when interphase formation is beneficial versus detrimental under realistic voltage windows and protocols; and (iii) an integrative summary table/decision map harmonizing evidence across EDLC, pseudocapacitor, and hybrid architectures for direct cross-study comparison.
To avoid ambiguity, we define what we mean by descriptor unification and quantitative analysis. Here, the unified descriptor set spans (i) driving-force descriptors (electrolyte stability window, electrode energetics/Fermi-level alignment, and local EDL/pore field), (ii) solvation/transport descriptors (ΔGsolv/desolvation propensity and effective ionic/electronic resistivity of the interphase), and (iii) interphase state variables (thickness/coverage, porosity, and inorganic/organic fraction) that map onto supercapacitor metrics (leakage/self-discharge, ESR evolution, and capacitance retention). The quantitative analysis in this review refers to synthesizing reported cross-study correlations among these descriptors and metrics, rather than proposing a single universal predictive equation.
The scope of applicability is the EDLC/pseudocapacitor/hybrid systems surveyed here in aqueous (including water-in-salt), organic, and ionic-liquid electrolytes operated within practical voltage windows. Outside these regimes (e.g., severe gas evolution, conversion-dominated reactions, strongly redox-active electrolytes, or extreme conditions), descriptor-to-metric relationships may change; we therefore treat such cases as boundary conditions rather than claiming full universality.
This review synthesizes fundamental principles, experimental characterization, computational modeling, and engineering strategies for SEI control across batteries and supercapacitors, progressing systematically from atomic-scale mechanisms to device-level performance optimization.
This review synthesizes knowledge from multiple research communities (electrochemistry, materials science, battery engineering, and supercapacitor technology) to construct a unified understanding of SEI phenomena. Our literature search encompassed peer-reviewed journal articles, conference proceedings, and authoritative reviews published between 1979 (Peled's seminal SEI discovery) and 2025. Primary databases included Web of Science, Scopus, PubMed, Google Scholar, and specialized electrochemical databases. Search terms combined “solid electrolyte interphase,” “SEI,” “supercapacitor,” “electrochemical capacitor,” “EDLC,” “pseudocapacitor,” “interfacial film,” “passivation layer,” and “electrolyte decomposition” with Boolean operators to capture both battery-focused and capacitor-focused literature.
Inclusion criteria prioritized: (i) experimental studies employing advanced characterization techniques (XPS, cryo-TEM, operando spectroscopy) to probe interfacial chemistry; (ii) computational investigations using ab initio methods, molecular dynamics, or machine learning to elucidate SEI mechanisms; (iii) engineering studies demonstrating electrolyte additive effects, surface modifications, or artificial interphase strategies; and (iv) review articles synthesizing battery SEI knowledge with potential applicability to supercapacitors. We excluded purely device-performance studies lacking mechanistic insight into interfacial processes and preliminary conference abstracts without peer-reviewed follow-up.
Cross-referencing was performed to identify seminal works cited across both battery and supercapacitor communities, revealing key conceptual bridges (e.g., water-in-salt electrolytes, film-forming additives, operando EQCM) that enable knowledge transfer between domains. Over 400 references are integrated into this review, organized thematically to guide readers from fundamental principles through cutting-edge applications.
Establishing SEI formation as a common phenomenon has profound implications for next-generation energy-storage device design. First, it enables predictive engineering: understanding how electrolyte composition, electrode surface chemistry, and operating conditions determine interphase properties allows rational design of SEIs with tailored thickness, ionic conductivity, and mechanical resilience.57,58 Second, it facilitates cross-platform innovation: strategies proven successful in batteries (such as FEC or VC additives that form uniform fluorinated SEIs59,60) can be systematically adapted to stabilize high-voltage supercapacitors. Conversely, the highly dynamic, self-healing SEIs observed in ionic-liquid capacitors offer insights for designing adaptive interfaces in next-generation battery chemistries.61,62
Third, unified SEI frameworks accelerate multiscale modeling integration. Atomistic simulations (DFT, reactive MD) capture early-stage nucleation and chemical composition; continuum transport models describe long-term growth and impedance evolution; and machine-learning surrogates bridge these scales to predict device-level performance from molecular descriptors.63–65 Such hierarchical workflows, validated against operando experimental data, will enable in silico optimization of electrolyte formulations and electrode architectures before costly laboratory synthesis.66,67
Fourth, recognizing the dynamic nature of supercapacitor SEI-like interphases (thinner, more elastic, continuously reconstructing) informs durability and reliability engineering. Unlike thick, quasi-static battery SEIs that crack under mechanical stress, supercapacitor interphases must tolerate millions of rapid charge–discharge cycles without catastrophic failure.24,31 Designing cross-linked binder networks, self-healing polymer matrices, and resilient artificial coatings becomes paramount for achieving ultralong cycle life.68,69
Finally, this unified perspective supports sustainability and circular-economy goals. By understanding which SEI components are environmentally benign, biodegradable, or recyclable, researchers can design electrolytes and electrode surface treatments that minimize environmental footprint while maintaining performance.70,71 Moreover, predictive SEI models reduce trial-and-error experimentation, lowering resource consumption and accelerating time-to-market for next-generation supercapacitors powering electric vehicles, grid storage, and portable electronics.53
2 Fundamentals of the solid-electrolyte interphase (SEI) in batteries and supercapacitors
The concept of the solid-electrolyte interphase (SEI) originated in battery science, where electrolyte reduction produces a self-passivating layer that stabilizes electrode–electrolyte contact during cycling. Recent evidence suggests that analogous, albeit thinner and more dynamic, SEI-like films can also arise in supercapacitors under extreme potentials, high surface reactivity, or defect-rich conditions. Understanding these parallels allows cross-fertilization of ideas: mechanistic principles from batteries clarify interphase chemistry in capacitors, while the reversible, elastic behavior of capacitor interfaces offers new insight into SEI adaptability in batteries.2.1 Capacitive charge storage fundamentals
Supercapacitors (also known as electrochemical capacitors) store energy through fast and reversible interfacial processes that bridge the gap between conventional capacitors and batteries. Depending on the dominant charge-storage mechanism, they are broadly classified as electric double-layer capacitors (EDLCs) and pseudocapacitors. In EDLCs, charge storage is non-faradaic and arises from electrostatic ion adsorption at the electrode–electrolyte interface. When an external potential is applied, oppositely charged ions accumulate near the electrode surface, forming an electric double layer (EDL) that acts as a nanoscale capacitor. The total stored charge depends on the accessible surface area of the electrode, the dielectric constant of the electrolyte, and the effective thickness of the EDL.72,73 This mechanism provides excellent power density and long cycle life, as it does not involve structural changes or phase transformations in the electrode material.Pseudocapacitors, by contrast, store charge through fast surface or near-surface faradaic reactions that involve electron transfer between the electrode and electrolyte species. Transition-metal oxides (e.g., MnO2, RuO2) and conducting polymers (e.g., polyaniline, polypyrrole) are typical pseudocapacitive materials.74,75 The capacitance arises from reversible redox, intercalation, or adsorption processes that occur over a continuum of potentials rather than discrete battery-like plateaus. Because these reactions are surface-confined, pseudocapacitors combine the high energy densities of batteries with the power performance of EDLCs.9,76,77 Hybrid or asymmetric supercapacitors integrate both mechanisms by pairing an EDLC-type electrode with a pseudocapacitive one, thereby expanding the voltage window and enhancing overall energy storage.78 The interfacial charge distribution that governs these processes also defines the potential gradients that initiate electrolyte decomposition and SEI nucleation in both batteries and capacitors. To avoid ambiguity throughout this review, we distinguish three interfacial regimes in supercapacitors: (i) EDL adsorption (non-faradaic, electrostatic ion accumulation; typically rectangular (cyclic voltammograms) CVs and minimal persistent resistance drift),79 (ii) reversible pseudocapacitive surface redox (fast faradaic but reversible surface/near-surface electron transfer; broad redox features that remain stable under cycling within a safe window),80 and (iii) irreversible SEI-like films (electrolyte decomposition or side reactions producing new surface species and progressive performance drift).81,82 Importantly, (ii) and (iii) can coexist on pseudocapacitive electrodes;83 therefore, assignments should be supported by coupled electrochemical metrics (leakage/self-discharge, ESR evolution, retention) and chemistry-resolved diagnostics (e.g., XPS/ToF-SIMS/FTIR) rather than by CV shape alone.79,81
2.2 Interfacial electric field distribution and ion dynamics
The electric double layer governs the microscopic origin of capacitance in supercapacitors. At the electrode–electrolyte interface, ions rearrange in response to the surface potential, producing a space-charge region characterized by the Stern–Gouy–Chapman model.84–86 The inner Helmholtz plane (IHP) consists of specifically adsorbed, partially desolvated ions in direct contact with the electrode surface, while the outer Helmholtz plane (OHP) contains solvated ions whose centers are separated by one solvent-molecule thickness.72,87 Beyond these layers lies the diffuse region, where ion distribution follows a Boltzmann potential profile. The differential capacitance depends on the local dielectric constant, ion concentration, and potential-dependent surface charge density.72 In nanoporous electrodes, confinement effects alter ion packing and solvation structure, giving rise to non-classical phenomena such as ion desolvation, crowding, and overscreening.88 These effects strongly influence voltage-dependent capacitance and energy density.Spectroscopic, scattering, and molecular-dynamics studies reveal that the EDL is not a simple continuum but a dynamically fluctuating interfacial region with heterogeneous ion orientations and time-dependent correlations.89 The interplay of ion–ion and ion–solvent interactions determines the local permittivity and thus the capacitance. In pseudocapacitive materials, surface redox reactions further modify the interfacial electric field. These same electrostatic gradients set the stage for SEI formation once the local potential exceeds the electrolyte stability limit, linking double-layer physics with interphase chemistry across both energy-storage platforms.
2.3 Origin and function of the solid-electrolyte interphase
The electrode–electrolyte interface is a chemically complex region where ion adsorption, charge transfer, and molecular decomposition occur simultaneously. While EDL formation governs the fast and reversible electrostatic storage that defines supercapacitors, parasitic reactions at the same interface can lead to the emergence of a thin, passivating SEI. Much of the mechanistic framework for interpreting such interphases derives from battery research, where SEI formation is intrinsic and indispensable. In supercapacitors, similar reactions occur only under overstress or at defect-rich sites, producing a more transient and reversible film.SEI-like films emerge when local interfacial conditions drive electrolyte reactions beyond the stability window, producing a thin electronically blocking but ion-permeable interphase.13 For batteries this interphase is intentionally formed and comparatively persistent,46 whereas in supercapacitors it is typically ultrathin and condition-dependent (e.g., high-voltage operation, defects, and rapid cycling),82 so its impact is better discussed using supercapacitor-specific observables such as leakage current, ESR evolution, and capacitance retention.
2.4 Compositional and structural distinctions across energy storage platforms
Bridging SEI behavior across these two systems clarifies how shared electrochemical principles yield different interphase dynamics. In rechargeable batteries, the SEI is essential for long-term operation: it passivates the anode against continuous electrolyte decomposition while permitting Li+ or Na+ transport. Battery SEIs are typically tens of nanometers thick, composed of inorganic salts (LiF, Li2O, Li2CO3) in the inner region and polymeric organic components (polycarbonates, alkoxides) toward the outer surface.15,90 Once formed, these layers remain largely static and evolve slowly over many cycles, occasionally thickening due to secondary reactions.In contrast, supercapacitor SEI-like interphases are thinner (often below 5–10 nm) and more dynamic because charge storage occurs primarily through non-faradaic or surface-confined pseudocapacitive processes.91 Interfacial potential fluctuations during rapid charging and discharging continually perturb the equilibrium structure, leading to reversible rearrangements or partial dissolution. Consequently, the SEI-like interphase in supercapacitors behaves as a quasi–elastic interface that expands and contracts without catastrophic failure.92 Furthermore, since the operating voltage of supercapacitors (1–3 V) is lower than typical battery cutoff voltages, electrolyte decomposition is limited, and SEI formation depends strongly on surface defects, residual moisture, and specific ion–surface interactions.15
From a compositional standpoint, organic-electrolyte supercapacitors often yield SEIs containing solvent fragments (acetonitrile or propylene carbonate) mixed with anion-derived fluorinated or borate species (BF4−, PF6−).19,93 In aqueous systems, decomposition of water forms hydroxide or oxide layers that are less stable and reconstruct during cycling, whereas ionic-liquid-based supercapacitors generate fluoride- or sulfur-enriched SEIs from anion cleavage.93 These dynamic interphases, although thinner, exhibit higher heterogeneity, influencing self-discharge and capacitance retention.
The mechanical demands placed upon the SEI diverge considerably between the two device classes. Battery interphases must accommodate substantial volumetric expansion inherent to high-capacity anodes such as lithium metal and silicon, whereas carbonaceous and oxide electrodes characteristic of supercapacitors undergo negligible dimensional change during operation. This mechanical quiescence reduces interfacial stress yet simultaneously heightens sensitivity to potential-induced polarization and adsorption heterogeneity.94 Collectively, these observations underscore that SEI chemistry is governed by generalizable electrochemical principles, namely electrolyte breakdown, ion-migration-limited growth, and self-passivation, while device-specific boundary conditions encompassing potential range, ion identity, and surface reactivity ultimately dictate interphase thickness, elasticity, and reversibility (Table 1).
| Feature | Battery SEI | Supercapacitor SEI-like film |
|---|---|---|
| Temporal behavior | Quasi-static after initial formation | Dynamic, continuously reconstructed under cycling |
| Primary function | Prevent further decomposition, enable ion intercalation | Modulate leakage current, stabilize double layer |
| Mechanical response | Rigid, stress-sensitive (must accommodate volume change) | Quasi-elastic, self-healing (minimal dimensional change) |
| Failure mode | Cracking from expansion/contraction | Resistive thickening from cumulative voltage stress |
2.5 Governing parameters in interphase evolution
The formation, composition, and stability of SEI layers depend on electrolyte formulation, operating voltage, temperature, and electrode surface characteristics.Overall, the physicochemical environment determines whether the SEI acts as a beneficial stabilizing layer or a resistive barrier. Integrating these parameters into unified electrochemical models will enable predictive interphase engineering across both batteries and supercapacitors. The following section (Section 3) examines the microscopic mechanisms that drive SEI nucleation, growth, and reconstruction, establishing the molecular basis for the interphase behaviors described above.
3 Microscopic mechanisms of SEI formation and evolution
SEI formation is a universal widely observed electrochemical phenomenon arising whenever the applied potential drives an electrolyte beyond its thermodynamic stability window.15,106,107 In both batteries and supercapacitors, the interphase originates from coupled electron-transfer, ion-migration, and molecular-reorganization events that spontaneously self-assemble at the electrode–electrolyte boundary.108 The distinction lies in scale rather than mechanism: battery SEIs are thick and quasi-static, whereas capacitor SEIs are ultrathin, dynamic, and continuously reconstructed under cycling.16,20 Understanding their shared microscopic origins is essential for cross-platform interphase engineering.3.1 Electron transfer and electrolyte reduction criteria
SEI nucleation begins when the electrode Fermi level intersects the lowest unoccupied molecular orbital (LUMO) of the electrolyte, enabling electron tunneling into solvated molecules and initiating bond cleavage.109,110 The same criterion governs electrolyte reduction in both Li-ion batteries and carbon-based supercapacitors, although the effective overpotential and reaction timescales differ.98Table 2 summarizes the principal thermodynamic parallels.| Aspect | Batteries | Supercapacitors |
|---|---|---|
| Driving potential | Electrolyte reduction at anode potentials (<1 V vs. Li/Li+) | Localized overpotential or defect-site fields (1–3 V window) |
| Film thickness | 10–100 nm, diffusion-limited growth | 1–10 nm, field-driven reconstruction |
| Dominant species | LiF, Li2CO3, Li2O | Carbonates, nitriles, fluorides |
| Growth mode | Self-limiting, inorganic barrier | Dynamic, elastic dielectric |
Electron injection into solvent or anion orbitals initiates decomposition of carbonate (EC, PC) or nitrile (ACN) molecules.111 Regardless of electrolyte type, the resulting fragments (carbonates, alkoxides, and nitrile oligomers) polymerize or condense on the surface, establishing the initial interphase.109,112 Anion reduction (BF4−, PF6−, TFSI−) produces inorganic fluorides and borates in both systems, yielding the dense, electronically insulating inner region that terminates further electron leakage.113 Thus, SEI nucleation follows the same thermodynamic sequence in batteries and capacitors, differing only in potential range and material identity.
3.2 Atomistic decomposition and fragment condensation
At the atomic scale, SEI formation proceeds through multiple, overlapping routes: solvent reduction, salt decomposition, and radical polymerization.114 In supercapacitors, decomposition pathways must be discussed in the context of electrolyte class and voltage window,115 because the dominant fragments differ strongly between (i) organic salts (e.g., TEABF4 in ACN/PC),82 (ii) ionic liquids (e.g., TFSI−-based RTILs), and116(iii) highly concentrated aqueous electrolytes (water-in-salt).117–119 In organic EDLCs, anion-derived fragments (BFxOy/C–F) and solvent-derived oligomers can form ultrathin films that suppress charge leakage; in RTILs, F/S-containing fragments frequently dominate; in water-in-salt, anion-derived inorganic-rich layers emerge as the key passivation motif. Therefore, supercapacitor SEI-like interphase chemistry should be summarized as electrolyte-specific evidence rather than as a battery analogy.In ionic-liquid and water-in-salt electrolytes, coordinated water or cation fragments yield inorganic fluorides or hydroxides that emulate the protective SEIs of batteries.120 Hence, solvent and anion decomposition chemistry is fundamentally transferable between the two device classes.
3.3 Heterogeneous interphase structure and charge selectivity
Once decomposition products accumulate, they organize into a bilayered morphology: a dense, inorganic inner layer and a porous, polymeric outer matrix.121 In batteries, LiF/Li2CO3 layers dominate the inner region.122 On the other hand, in capacitors, C–F/BFxOy and M–O–F domains play the same mechanical and electronic role.123 The outer polymeric zone, composed of polycarbonate, nitrile, or ether species, acts as an ion-permeable, elastically compliant dielectric. This architecture self-regulates via potential-dependent current tunneling: when the film reaches a critical thickness d, the tunneling current density| J ∝ exp(−βd) |
3.4 Desolvation and ion migration through passivating films
Ion solvation governs both the onset of electrolyte reduction and the transport properties of the resulting SEI. At the interface, strong electric fields induce partial desolvation, exposing bare ions that readily accept or donate electrons.124 In batteries, Li+ desolvation triggers EC reduction. In supercapacitors, desolvation must be framed through pore confinement and rate:125 ion desolvation and re-solvation occur repetitively at high frequency,126 and the interphase must remain thin enough to avoid blocking micropores that govern capacitance.125 Accordingly, evidence should be interpreted using measurable outputs (specifically, capacitance retention, ESR evolution, and self-discharge) rather than only molecular descriptors.127 When a film becomes too resistive or blocks pore entrances, capacitance decreases and ESR rises; when it is thin and electronically insulating, leakage decreases without sacrificing rate capability.127,128The degree of solvation controls the SEI composition: tightly bound solvates produce dense, inorganic-rich layers, while weakly solvated ions form thinner, more permeable films.124,129–131 This relationship can be expressed by the correlation between solvation energy (ΔGsolv) and effective ionic conductivity (σion):
3.5 Dynamic reconstruction under electrochemical stress
The SEI continually evolves under electrochemical and thermal stimuli. Elevated temperature accelerates decomposition and diffusion, densifying the film in both systems;135 low temperature slows polymerization, yielding porous, resistive layers. During potential cycling, elastic compression/relaxation and ongoing side-reaction turnover can drive reversible interphase restructuring, as observed by operando EQCM and cryo-TEM in both batteries and capacitors.23 In supercapacitors, the relevant SEI-like interphase is typically ultrathin (often ∼1–10 nm) and can exhibit two superimposed behaviors: (i) a baseline thickness/mass drift during early conditioning or prolonged voltage holds, and (ii) cycle-resolved fluctuations that track the charge–discharge period. Practically, the reconstruction timescale spans seconds to minutes under dynamic cycling (set by the cycling period), whereas net growth/compaction is most evident during minutes-to-hours voltage holds, consistent with diffusion-reaction control.These oscillations follow diffusion-reaction kinetics similar to logarithmic growth laws:136
| d(t) = kt1/2, |
3.6 SEI-mediated modulation of capacitance and impedance
The SEI critically influences electrochemical response by mediating ion accessibility and dielectric behavior. In both devices, its thickness and ionic conductivity determine the distance between ionic and electronic charge centers, thereby affecting differential capacitance and internal resistance.137,138 Nyquist plots of porous electrodes often display a mid-frequency arc or depressed semicircle, but this feature is non-unique.139,140 It may reflect interphase film response, contact/constriction effects, and/or distributed pore transport and charging (transmission-line behavior).141–143 For this reason, we interpret mid-frequency impedance features in supercapacitors using a porous-electrode-aware framework (Section 8.2) and reserve the term “interphase resistance” for cases where the fitted film element co-varies with independent interphase diagnostics (chemistry/thickness from XPS/ToF-SIMS/cryo-TEM, or mass uptake from EQCM)142,144 and shows consistent trends with voltage window and cycling protocol.119A thin, ion-permeable SEI minimizes leakage and stabilizes coulombic efficiency, whereas a thick, resistive one increases relaxation times and lowers power density.145,146
Spatial heterogeneity of the SEI also modifies local permittivity: inorganic-rich domains (εr ≈ 5–10) focus electric fields and promote ion layering, while polymeric regions (εr ≈ 20–40) smooth potential gradients. This heterogeneity generates two characteristic time constants in impedance spectra: a fast component from electric-double-layer charging and a slower one from ion migration through the SEI.147,148 In both batteries and capacitors, these coupled processes define the dynamic bottleneck for high-rate performance.
Furthermore, reversible redox rearrangements within the organic fraction of the SEI can contribute to pseudocapacitive charge storage. Such behavior, once noted in polymer-rich battery SEIs, equally appears in stabilized capacitor interfaces, where the SEI-like interphase acts as an active, charge-mediating dielectric rather than a passive barrier.149,150
3.7 Device-class evidence and metrics for SEI-like interphases in supercapacitors (EDLC, pseudocapacitive, and hybrid)
To meet the supercapacitor community's needs, SEI-like interphases should be summarized by device class and verified by both interfacial signatures and device-level metrics.151,152 In contrast to batteries, where SEI formation is intrinsic, supercapacitor interphases are typically triggered near the electrolyte stability boundary at defect-rich surfaces, or under aggressive voltage holds.153,154 Therefore, evidence must be reported with explicit operating details (electrolyte, voltage window, electrode type) and quantified outcomes (leakage current, self-discharge, ESR, capacitance retention).153,155In EDLCs (activated carbon, graphene, porous carbons), SEI-like films are most consistently reported in high-voltage organic and ionic-liquid electrolytes, particularly under voltage holds or extended cycling near the upper cut-off.155 Key evidence includes: (i) emergence of F-containing and carbonate/nitrile-derived surface species (XPS/ToF-SIMS),153 (ii) nm-scale coverage detectable by cryo-TEM/AFM in selected systems,156 and (iii) correlated suppression of leakage current and slower self-discharge when stable films form.157 However, excessive film growth or pore-mouth blockage causes measurable capacitance loss and ESR increase, indicating that thin and dynamic interphases are beneficial whereas thick and blocking interphases are detrimental.158,159
For pseudocapacitive electrodes (metal oxides, MXenes, conducting polymers), the interphase must be interpreted together with surface redox: some surface reactions are reversible and contribute to capacitance, while others produce irreversible films that progressively increase resistance.160 Evidence should therefore separate: (i) reversible surface chemistry (stable redox-active states) from (ii) irreversible electrolyte decomposition products (passivating layers).161Operando EIS/EQCM combined with post-mortem XPS is particularly valuable here, because it can distinguish mass changes from adsorption versus irreversible film accumulation.162 In aqueous systems, local pH shifts and proton-coupled processes can dominate, whereas in organic/IL electrolytes, fluorinated fragments and polymeric residues may accumulate at oxide/carbon interfaces.163
Hybrid capacitors (asymmetric devices, lithium-ion capacitors, battery-capacitor hybrids) operate in regimes where one electrode experiences more battery-like polarization.164 As a result, interphase formation in hybrid (EDLC + faradaic) cells can be strongly electrode-selective, governed by each electrode's individual stability limits, and can dominate aging even when the full device response remains largely capacitive; therefore, evidence should name the electrode that forms the dominant interphase and correlate it to ESR/impedance rise, Coulombic-efficiency/residual-current drift, and any electrode-to-electrode charge-balance (capacitance) mismatch that develops over cycling.31
3.8 Unified framework for interphase formation
Fig. 4 and 5 schematically summarizes SEI formation across energy-storage systems, outlining five common stages: (1) electron injection, (2) solvent/anion decomposition, (3) inorganic nucleation, (4) polymeric condensation, and (5) mechanical relaxation.165,166 Differences between batteries and capacitors arise primarily from potential magnitude and cycling frequency, not from reaction identity. | ||
| Fig. 4 Schematic overview of SEI formation across energy-storage systems, showing three common stages: (1) electron injection and inorganic nucleation. Reproduced from ref. 132 with permission from WILEY, Copyright 2021 (2) solvent/anion decomposition. Reproduced from ref. 133 with permission from IOP Publishing, Copyright 2018. and (3) mechanical relaxation. Reproduced from ref. 134 with permission from WILEY, Copyright 2025. | ||
 | ||
| Fig. 5 Schematic illustration of polymer-condensation-driven interphase growth as part of the broader interphase-formation framework across energy-storage systems. (a) Formation of SEI via electrolyte decomposition. (b) Design of a polymer–inorganic SEI using the RPC precursor. Reproduced from ref. 134 with permission from Nature Publishing Group, Copyright 2019. | ||
Across all electrochemical devices, the SEI embodies the same self-organization of matter under electric fields and chemical stress.16,167 Recognizing this universality generality transforms the SEI from a device-specific artifact into a general design principle linking molecular chemistry to macroscopic performance. Having established the fundamental mechanisms of SEI formation, we now turn to the experimental methodologies that enable direct observation and characterization of these interphases in Section 4, followed by computational modeling frameworks in Sections 6 and 7 that predict and simulate SEI evolution across multiple scales.
4 Experimental characterization of the SEI
Understanding the chemistry, morphology, and dynamics of the solid-electrolyte interphase (SEI) requires a combination of ex situ structural analysis and in situ or operando spectroscopy.168 Most techniques now used to probe SEI evolution were first developed in battery research, where stable interphases are intrinsic to operation, and have since been adapted to investigate the thinner, more dynamic SEI-like films in supercapacitors.22,169,170 Recognizing this shared experimental heritage enables a unified interpretation of interfacial chemistry across both systems and provides critical validation for atomistic simulations and continuum models discussed in Section 6.23,1714.1 Ex Situ techniques for interphase characterization
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and O–C
O, and O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O functionalities arising from solvent oxidation, while F 1s and B 1s peaks confirm BF4− or PF6− decomposition.174 Quantitative peak-shift analysis further provides information on local potential drops and the electronic insulation conferred by the SEI layer. Thus, XPS offers a common compositional framework for comparing SEI chemistry across batteries and capacitors.
O functionalities arising from solvent oxidation, while F 1s and B 1s peaks confirm BF4− or PF6− decomposition.174 Quantitative peak-shift analysis further provides information on local potential drops and the electronic insulation conferred by the SEI layer. Thus, XPS offers a common compositional framework for comparing SEI chemistry across batteries and capacitors.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), ∼1100 cm−1 (C–O–C), and carbonate stretches signal solvent decomposition and polymerization.172 In battery studies, these signatures verified polymeric carbonates and alkoxides; analogous features in supercapacitors indicate nitrile- or carbonate-based oligomers formed from acetonitrile or propylene carbonate breakdown. Mapping FTIR intensity across electrodes reveals heterogeneity linked to potential gradients and pore accessibility.175,176
O), ∼1100 cm−1 (C–O–C), and carbonate stretches signal solvent decomposition and polymerization.172 In battery studies, these signatures verified polymeric carbonates and alkoxides; analogous features in supercapacitors indicate nitrile- or carbonate-based oligomers formed from acetonitrile or propylene carbonate breakdown. Mapping FTIR intensity across electrodes reveals heterogeneity linked to potential gradients and pore accessibility.175,176
4.2 In situ monitoring of SEI formation and evolution
Electrochemical impedance spectroscopy (EIS) provides a frequency-resolved fingerprint of interfacial and porous–electrode processes, but in porous supercapacitor electrodes the mid-frequency semicircle is not uniquely attributable to an SEI.181 Similar semicircles (or depressed arcs) can arise from a convolution of (i) interphase film response (Rf‖ CPEf), (ii) contact/constriction resistance within the current collector-electrode network, and (iii) distributed pore transport and surface charging captured by transmission-line behavior.181 Accordingly, interphase resistance should be assigned only when (a) a film-like time constant is consistently observed across conditions and (b) complementary evidence (e.g., XPS/ToF-SIMS thickness/chemistry trends, EQCM mass uptake) co-varies with the fitted film element.151 In this review we therefore treat EIS-derived “RSEI” as an operational metric that requires a porous-electrode-aware fitting framework rather than a one-to-one identifier of SEI formation.181
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N or S
N or S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching modes trace reversible ion solvation or irreversible decomposition leading to SEI formation.182 These techniques, originally applied to monitor electrolyte degradation in Li-ion cells, now enable spatially resolved mapping of interfacial heterogeneity in capacitors.
O stretching modes trace reversible ion solvation or irreversible decomposition leading to SEI formation.182 These techniques, originally applied to monitor electrolyte degradation in Li-ion cells, now enable spatially resolved mapping of interfacial heterogeneity in capacitors.
 | ||
| Fig. 6 Recent advances in the mechanistic understanding of Li deposition and SEI evolution using advanced characterization and theoretical modeling methods. (a) Characterization techniques for probing Li plating and stripping behavior include magnetic resonance imaging (MRI), synchrotron X-ray computed tomography (SXCT), cryo-TEM, SEM, AFM-ETEM, optical microscopy (OM), liquid-cell TEM, cryo-FIB, and thermogravimetric analysis/GC-based methods where applicable. (b) Analytical methods for SEI characterization include cryo-TEM, AFM, SIMS, NMR, and cryo-STEM tomography. (c) Theoretical and computational approaches include Monte Carlo (MC), molecular dynamics (MD), finite-element modeling (FEM), density functional theory (DFT), and smoothed-particle hydrodynamics (SPH) for investigating diffusion barriers, Li-deposition behavior, and SEI structure. Reproduced from ref. 185 with permission from Wiley, copyright 2022. | ||
4.3 Cross-technique validation and data synthesis
Correlating chemical and electrochemical data is crucial for understanding SEI function in any electrochemical system. In both batteries and capacitors, combining XPS-derived composition with EIS-extracted RSEI values links specific motifs (fluorides, carbonates, phosphates) to fitted interphase resistivity/impedance (e.g., RSEI or Rint) with explicitly traceable quantitative sources.19 FTIR and Raman features associated with polymeric carbonates often coincide with higher leakage currents or reduced capacitance, indicating partial electronic conductivity through conjugated species. EQCM mass–change profiles synchronized with cyclic-voltammetry (CV) peaks directly reveal the competition between capacitive ion adsorption and interphase growth.186 These cross-correlations provide quantitative descriptors, such as “interphase efficiency” or “ionic utilization ratio,” that unify SEI analysis across technologies.Multimodal platforms now integrate spectroscopy, microscopy, and electrochemistry in real time. Operando Raman/EIS coupling simultaneously tracks vibrational and impedance signatures, while synchrotron-based soft X-ray absorption spectroscopy (sXAS) resolves element-specific oxidation states.187 These hybrid systems establish feedback loops between experiment and simulation, enabling parameter calibration for molecular-dynamics and DFT models and strengthening the link between mechanistic insight and performance metrics.
4.4 Comparative studies across electrode and electrolyte systems
A major barrier to unifying supercapacitor interphase literature is that many studies report surface chemistry without connecting it to device-level consequences, or report performance without identifying interphase chemistry.188 To enable actionable comparison across EDLC, pseudocapacitor, and hybrid systems, supercapacitor interphase evidence should be summarized in a standardized, metrics-linked format.188,189For transition-metal-oxide electrodes (e.g., MnO2, NiO, V2O5), XPS and FTIR reveal hybrid SEIs combining metal–oxygen species and organic fragments.192 Such interphases facilitate proton or cation insertion in both batteries and capacitors, though excessive growth hinders diffusion and causes capacitance fading.
Ionic-liquid-based systems exhibit inorganic-rich SEIs dominated by F- and S-containing fragments (TFSI− or PF6− derivatives).61 Cryo-TEM and ToF-SIMS confirm dense coverage that stabilizes both Li–metal and carbon interfaces. In solid or gel polymer electrolytes, FTIR and NMR reveal polymer-electrode cross-linking and hydrogen-bond networks controlling interfacial ion mobility.169,193 These case studies underscore that SEI chemistry reflects shared electrochemical principles, modified by electrode material, electrolyte chemistry, and potential range, rather than device category.
4.5 Sources of uncertainty and inter-laboratory variability
Despite progress, SEI characterization faces common limitations in both research domains. Ultrathin, reactive films challenge sample preparation and air-free transfer.194Ex situ methods capture static snapshots, whereas in situ techniques often trade spatial resolution for time resolution.195 Discrepancies among laboratories arise from differences in cell geometry, electrolyte purity, and reference calibration.196 The adoption of glovebox-integrated, cryogenic, and statistically replicated protocols now improves reproducibility. Moreover, coupling experimental outputs with simulation benchmarks, including radial distribution functions and predicted binding energies, provides cross-validation and minimizes interpretational ambiguity (Table 3).| Technique | Battery application | Supercapacitor adaptation | Insight bridged |
|---|---|---|---|
| XPS | Depth profiling of LiF/Li2CO3 SEI | Detection of fluorinated/nitrile films | Composition and oxidation states |
| FTIR/Raman | Organic carbonate identification | Nitrile/carbonate vibrations | Functional-group evolution |
| Cryo-TEM | SEI morphology on Li metal | Dynamic reconstruction in IL-based capacitors | Structural reversibility |
| EQCM/EIS | Mass and impedance evolution | Film growth vs. ion adsorption | Kinetic coupling |
| AFM/EC-AFM | Nanomechanical mapping | Interphase elasticity and adhesion | Mechanical response |
4.6 Summary
The migration of SEI-characterization methods from batteries to supercapacitors has unified interphase science across energy-storage technologies. Techniques once reserved for thick, inorganic-rich films now resolve nanometer-thin, reversible SEIs in capacitors. Establishing standardized cross-system protocols, including consistent potential referencing, cryogenic handling, and quantitative multimodal analysis, will enable reproducible comparison and accelerate the integration of experimental data with computational modeling.The experimental insights presented in this section provide the empirical foundation for the materials and interface engineering strategies discussed in Section 5, the multiscale modeling frameworks in Section 6, and the machine-learning approaches in Section 7 that translate observed interphase properties into predictive design tools.
5 Materials and interface engineering strategies
Performance and durability in both batteries and supercapacitors are governed by the interplay among electrodes, electrolytes, separators, and fabrication protocols. Although their energy-storage mechanisms differ, the fundamental challenge is the same: controlling interfacial reactions to form a stable, ion-conductive, and electronically insulating solid-electrolyte interphase (SEI).197,198 This section consolidates materials selection, electrolyte design, surface modification, artificial interphase strategies, and mechanical stabilization approaches that have traditionally evolved in battery science but are now being systematically translated to supercapacitors, establishing a coherent framework for cross-platform SEI engineering.The organization follows a logical progression: electrode materials and their surface engineering (Section 5.1), electrolyte chemistry and additive design (Section 5.2), architectural design principles (Section 5.3), manufacturing and structure–property relationships (Section 5.4), and actionable design rules (Section 5.5). Each subsection highlights how insights from batteries inform capacitor design, and vice versa.
5.1 Active material and interface modification approaches
The electrode acts as both the electronic conductor and the reactive template on which electrolyte decomposition initiates. In batteries, electrode surfaces catalyze reduction of solvent molecules and salts, forming SEI layers that regulate ion transport. A similar, though thinner and more dynamic, interphase develops in supercapacitors under high potentials.199,200 Understanding this shared interfacial chemistry requires analyzing structure, composition, and surface functionality of the electrode, as well as deliberate surface modification strategies.To improve cross-material comparison, Table 4 summarizes how carbon electrodes, redox-active metal oxides, and MXenes differ in their dominant interphase/SEI-like behavior, practical stability limits, and the typical voltage windows used in supercapacitor operation.
| Electrode family | Primary stability limitation | Practical stabilization levers | Ref. |
|---|---|---|---|
| a Windows are representative ranges commonly used in the supercapacitor literature and should be interpreted as practical operating windows rather than universal limits; the stable window depends on electrolyte chemistry, electrode microstructure/defects, and testing protocol (rate vs. hold). | |||
| Porous carbons (AC, CNT, graphene, CDC) | Voltage-hold aging and local hot spots → pore blocking and ESR drift; oxidative decomposition at the positive electrode near Vmax | Limit time at high V (avoid long holds); control defect density/functional groups; heteroatom doping to suppress parasitic reactions; conformal ultrathin coatings to homogenize fields | 31, 216 and 217 |
| Transition-metal oxides (e.g., MnO2, NiO, V2O5) | Dissolution/surface reconstruction and irreversible film thickening → loss of active sites, rising resistance, fading capacitance | Electrolyte/pH control (in aqueous); surface coatings (carbon/ALD oxides) to suppress dissolution; protocol control near onset potentials; engineered porosity to reduce local flux hot spots | 218–223 |
| MXenes (e.g., Ti3C2Tx) | Termination evolution/oxidation and resistive film growth under aggressive potentials; electrolyte-specific decomposition products accumulate with time/holds | Control termination chemistry and processing; select electrolyte to minimize oxidation; apply protective ultrathin coatings if ESR drift dominates; avoid sustained overpotential during holds | 224–229 |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) improve wettability and ion accessibility but can catalyze side reactions if uncontrolled; nitrogen or fluorine doping, by contrast, lowers surface free energy, anchors inorganic fragments, and suppresses solvent decomposition.230,231 In both batteries and capacitors, heteroatom doping modifies the electronic density of states near the Fermi level, shifting the onset potential for electrolyte reduction and enabling more benign, self-passivating SEI evolution.
O) improve wettability and ion accessibility but can catalyze side reactions if uncontrolled; nitrogen or fluorine doping, by contrast, lowers surface free energy, anchors inorganic fragments, and suppresses solvent decomposition.230,231 In both batteries and capacitors, heteroatom doping modifies the electronic density of states near the Fermi level, shifting the onset potential for electrolyte reduction and enabling more benign, self-passivating SEI evolution.
Conformal coatings, including atomic layer deposited (ALD) oxides, molecular-layer-deposited (MLD) polymers, polydopamine (PDA), and graphene-oxide (GO) films, pre-form artificial buffer layers that moderate the first contact between electrode and electrolyte.19,232 Thin (<5 nm) alumina or titania ALD coatings, proven to stabilize Li metal and Si anodes, have direct analogues in supercapacitors where they homogenize the interfacial field, prevent localized ion crowding, and mechanically reinforce porous structures.233 Recent advances in nanoscale electrode architecture, such as hierarchical porosity, vertically aligned nanotubes, and core–shell heterostructures, enable simultaneous control of ion transport and SEI uniformity.
Surface topology and curvature also influence SEI homogeneity. Hierarchically porous or scaffolded electrodes distribute ionic flux uniformly, reducing hot spots that trigger uncontrolled film growth.232 For flexible or solid-state configurations, the combination of engineered surface morphology and compliant gel electrolytes produces solid–solid interfaces with enhanced mechanical tolerance.198
5.2 Electrolyte formulation for controlled interphase formation
Electrolyte formulation is the second axis of SEI control. The electrolyte acts as both reactant and architect of the SEI, supplying the molecular precursors from which the interphase self-assembles.98,234 Regardless of whether the system stores charge through ion adsorption or faradaic reactions, electrolyte decomposition initiates interphase formation. Concepts first proven in battery electrolytes are now being systematically adapted to capacitors, revealing consistent mechanisms governing SEI chemistry.When effective, film-forming additives promote early formation of thin, electronically blocking yet ion-permeable interphases that suppress continuous electrolyte degradation; when ineffective, they can accelerate resistive thickening and pore blocking, increasing ESR and degrading power performance. Similarly, corrosion inhibitors or pH regulators in aqueous systems can stabilize surface chemistry and mitigate parasitic reactions, analogous to redox-mediated additives in batteries.242 Machine-learning-guided screening of additive/solvent/electrode combinations is emerging as a shared optimization tool across both research domains,243 but candidate additives should be down-selected using the actionable criteria summarized in Section 5.5 (design rules for leakage/ESR/retention trade-offs).
5.3 Architectural design principles
Device architecture transforms material-level control into macroscopic performance. Symmetric cells with identical electrodes mirror the balanced configurations of dual-ion batteries, whereas asymmetric and hybrid designs emulate full cells by coupling capacitive and faradaic electrodes.246 The same interfacial concerns, including contact resistance, electrolyte wetting, and mechanical adhesion, govern both technologies. Optimization therefore relies on common engineering metrics: minimized IR drop, uniform current distribution, and stable SEI adhesion.5.4 Manufacturing and structure–property relationships
Electrode fabrication directly impacts SEI nucleation and evolution. Active materials (carbon, oxide, or polymer) are typically mixed with conductive additives (carbon black, CNTs) and binders such as PVDF or PTFE.25,247 The slurry is cast onto metallic current collectors (Al, Ni, stainless steel) and processed by doctor-blade or drop-casting, then dried and compressed for uniform thickness. Binder-free architectures such as graphene foams, carbon cloths, and vertically aligned CNT arrays minimize inactive mass and enhance conductivity.248 These structural motifs, analogous to 3D current collectors in batteries, promote homogeneous SEI formation and mechanical stability.5.5 Actionable design rules for supercapacitor interphases (EDLC/pseudocapacitor/hybrid)
The supercapacitor literature increasingly confirms that SEI-like interphases can be either beneficial (suppressing leakage/self-discharge) or detrimental (raising ESR and driving capacitance fade), depending on voltage stress, electrolyte chemistry, and electrode surface functionality.169 To translate mechanistic understanding into practice, this section summarizes actionable design rules that connect (i) operating conditions, (ii) interphase signatures, and (iii) device-level metrics. These rules are intended to be applied together with the evidence-to-metric mapping framework introduced in Section 4 (metrics-linked reporting of interphase claims).Thus, good interphase ⇒ leakage/self-discharge ↓ (or stable) and ESR stable and retention stable; whereas bad interphase ⇒ ESR ↑ persistently and/or retention ↓ (often with pore blocking or loss of accessible redox sites), even if surface films are detected (Fig. 7).
| System | Typical voltage window | Interphase nature | Dominant mechanism | Beneficial vs. detrimental outcomes | Actionable design rule | Key ref. |
|---|---|---|---|---|---|---|
| Battery (Li-ion/Li-metal) | ∼2.5–4.5 V (cell), anode at low potential | Thicker, relatively persistent (10–100 nm), slowly evolving | Electrolyte reduction at negative electrode; self-limiting passivation (inner inorganic/outer organic) | Beneficial: passivation, high CE, safety; detrimental: Impedance rise, Li loss, cracking under strain | Form SEI via controlled formation; use additives/coatings to minimize resistive growth and mechanical failure | 14, 20, 273 and 274 |
| EDLC (organic; AC/graphene) | ∼2.5–3.0 V (typ.) | Ultrathin, dynamic/patchy; pore- and defect-localized | High-field decomposition near Vmax, especially at defects/micropores; partial reconstruction | Beneficial: leakage/self-discharge suppression; detrimental: pore blocking, ESR rise, capacitance loss | Operate below practical Vcrit; if high V is required, use brief conditioning + additives only if leakage drops without ESR drift | 28, 29 and 190 |
| EDLC (ionic liquid/high-voltage) | ∼3.0–3.5 V (typ.) | Thin but more inorganic-rich; dynamic under rapid cycling | Anion/salt-driven decomposition (F/S-containing fragments); film formation under holds | Beneficial: stabilizes high V; detrimental: resistive thickening under long holds | Avoid long potentiostatic holds; choose salts/additives that form ion-permeable films; validate by leakage + ESR + rate-capacitance | 27, 61, 275–278 |
| Pseudocapacitor (metal oxides; aqueous) | ∼0–1 V (electrode-dependent; device ∼1–2 V) | Highly condition-dependent; coupled to surface redox and local pH | Reversible surface redox vs. irreversible side reactions (dissolution/deposition/film) | Beneficial: stable redox capacitance; detrimental: active-site blocking, dissolution, ESR growth | Separate reversible redox from irreversible film growth (operando EIS/EQCM); tune pH regulators/coatings to preserve redox while suppressing parasitics | 191, 192 and 218 |
| Pseudocapacitor (MXene/polymers) | ∼0–1.2 V (electrode-dependent) | Interphase tied to terminations/solvation; can be reversible or aging-driven | Termination chemistry + solvent/salt reactions; polymer over-oxidation (for CPs) | Beneficial: stable pseudocapacitance; detrimental: resistive film/termination loss/over-oxidation | Control surface terminations; limit V to avoid irreversible chemistry; use ultrathin ion-permeable coatings if ESR drift dominates | 26, 33, 279 and 280 |
| Hybrid (LIC/battery-capacitor) | Device ∼3.8 V (typ), electrode-selective polarization | Asymmetric: battery-like SEI on one electrode + capacitor film on the other | One electrode experiences battery-like overpotential; imbalance drives aging | Beneficial: stabilized anode; detrimental: ESR drift, capacity imbalance, rapid fade if overpolarized | Mass-balance to avoid sustained overpolarization; monitor electrode potentials (3-electrode) where possible; accept additives only if both leakage and ESR improve | 11, 12, 281 and 282 |
5.6 Summary
Material and interface engineering strategies that stabilize SEIs in batteries, including controlled surface reactivity, electrolyte additive tuning, nanostructured architecture, artificial coatings, and mechanical stabilization, are directly translatable to supercapacitors. The convergence of electrode and electrolyte design principles across these technologies demonstrates that SEI formation follows universal shared chemical and mechanical rules rather than device-specific phenomena. By integrating experimental workflows (Section 4) and computational models (Sections 6 and 7), researchers can derive cross-platform descriptors linking composition, morphology, and transport. These descriptors provide the foundation for connecting SEI microstructure to macroscopic device performance, as discussed in Section 8.6 Multiscale modeling and simulation of SEI formation
Understanding the solid-electrolyte interphase (SEI) requires bridging phenomena that span more than ten orders of magnitude in time and length. Bond cleavage and charge transfer occur at the quantum level, while film growth and mechanical evolution unfold across nanometers to micrometers and over thousands of cycles. Modeling frameworks must therefore integrate electronic-structure theory, molecular dynamics (MD), continuum transport, and device-scale formalisms within a coherent hierarchy.212,283 Although the materials differ (Li metal anodes in batteries versus carbon electrodes in supercapacitors), the governing processes of electron tunneling, ion migration, and interphase reconstruction follow remarkably similar multiscale couplings.284,285 This section reviews computational approaches that link the microscopic mechanisms described in Section 3 with the experimental observables from Section 4, establishing predictive frameworks for SEI design.6.1 Atomistic simulation methods
MD provides the fundamental atomistic perspective on SEI structure and dynamics. Atoms are treated as classical particles whose trajectories obey Newton's equations, evaluated through empirical or reactive interatomic potentials.286–288 In both batteries and supercapacitors, MD captures ion adsorption, desolvation, and transport near polarized surfaces, thereby resolving the molecular origins of electric-double-layer formation and early electrolyte decomposition.289–291 Analyses of velocity-autocorrelation and residence-time functions yield diffusion coefficients and relaxation constants that can be directly compared with electrochemical impedance or cyclic-voltammetry measurements. These correlations position MD as the molecular bridge linking interfacial structure with experimentally measurable capacitance and resistance.292 The integrated atomistic-electronic workflow connecting AIMD/ReaxFF decomposition pathways to experimental impedance signatures is summarized in Fig. 9. | ||
| Fig. 8 Schematic overview of SEI-layer formation obtained from ReaxFF molecular-dynamics simulation at 300 K, showing the initial system configuration and the evolution of interphase species during early-stage electrolyte decomposition at the electrode interface. Reproduced from ref. 62 with permission from WILEY, Copyright 2023. | ||
 | ||
| Fig. 9 (1) Atomistic modeling workflow: (a) AIMD/ReaxFF simulations capturing electrolyte decomposition under varying electron-to-atom (E/A) ratios; (b) structural evolution of SEI precursor species; and (c) GENIP-based ab initio DFT + Green's-function framework used to evaluate interfacial electronic properties between the SEI and metal surface. Reproduced from ref. 303 with permission from Nature Publishing Group, Copyright 2023. (2) The first type of initial bond breaking mechanism of FEC leading to the formation of SEI. Reproduced from ref. 304 with permission from IOP Publishing, copyright 2020. (1) Electrochemical impedance spectra (EIS) of fresh vs. aged cells, highlighting characteristic frequency regimes associated with SEI resistance and charge transfer processes at the porous electrode (CT at PE). Reproduced from ref. 305 with permission from MDPI, Copyright 2018 under CC BY 4.0 (http://creativecommons.org/licenses/by/4.0/). | ||
6.2 Electrostatic treatment of polarized interfaces
Capturing realistic electrode polarization is central to simulating SEI nucleation. Constant-charge (CCM) and constant-potential (CPM) models remain the two canonical approaches. CCM assigns fixed atomic charges and thus reproduces qualitative ion layering but underestimates capacitance under strong electric fields. CPM dynamically adjusts atomic charges to maintain an imposed electrode potential, reproducing metallic screening and local charge redistribution.293,294 Recent extensions, including generalized (GCPM), heteroatomic (HCPM), and moment-tensor (mCPM) formulations, incorporate DFT-derived electronegativity and hardness, enabling accurate simulation of doped carbons, MXenes, and Li metal.295,296 Such self-consistent polarization models now delineate the spatial onset of electron tunneling and electrolyte reduction, providing mechanistic routes to SEI nucleation in both capacitors and batteries.2966.3 Chemical reactivity in force-field frameworks
The reliability of any MD study ultimately rests on its interatomic potential. Fixed-charge force fields such as OPLS, AMBER, and COMPASS are computationally efficient but non-reactive. Polarizable models, including induced-dipole and Drude-oscillator schemes, introduce electronic response, while charge-equilibration (QEq) and fluctuating-charge (FQ) approaches extend this to metallic polarization.297,298 Reactive potentials such as ReaxFF explicitly describe bond formation and scission, thereby allowing simulation of electrolyte decomposition and SEI film growth.299 In Li-ion systems, ReaxFF has clarified ethylene carbonate reduction to Li2CO3, whereas in supercapacitors it reproduces acetonitrile and ionic-liquid fragmentation that yields CN- or F-containing interphases.111,300 These studies consistently reveal heterogeneous nucleation, characterized by initial anchoring of inorganic fragments on the electrode followed by polymeric condensation. A representative ReaxFF simulation of SEI nucleation and early film formation is illustrated in Fig. 8, showing the molecular evolution captured during electrolyte decomposition.Machine-learning (ML) potentials such as DeepMD, NequIP, and SpookyNet offer near-ab initio accuracy with classical scalability.301 Trained on DFT trajectories for representative battery (LiPF6/EC) and capacitor (TEABF4/ACN) chemistries, these models reproduce many-body polarization, reactive events, and charge redistribution across diverse interfaces. Integration with active-learning and coarse-graining protocols has extended the accessible time window from nanoseconds to milliseconds, allowing direct simulation of interphase evolution under operational conditions.302
6.4 Mesoscale and macroscopic film evolution models
At larger scales, continuum and kinetic models treat the SEI as an effective mixed ionic-electronic conductor. Classical battery formulations by Peled and Newman describe diffusion-limited thickening with d ∝ t1/2,306 while supercapacitor adaptations incorporate the rapid charge–discharge cycles and mechanical elasticity of thin interphases. The temporal evolution of film thickness can be expressed aswhere Je is electron flux, M and ρ are the molar mass and density of the SEI, and kdiss captures dissolution or reconstruction. This formalism unifies the diffusion-limited growth observed in batteries with the dynamic breathing and partial reconstruction characteristic of supercapacitor interphases.307 Parameterization from MD or experimental data, such as ionic conductivity, reaction rate constants, and porosity, enables direct prediction of macroscopic ESR and capacitance decay.
6.5 Cross-scale coupling strategies
Multiscale coupling integrates information across five hierarchical tiers: (1) ab initio MD or DFT to resolve bond breaking and electronic structure; (2) reactive or ML-enhanced MD to describe nanometer-scale nucleation; (3) phase-field or coarse-grained dynamics for mesoscale morphology; (4) continuum transport for effective conductivity and mechanical response; and (5) device-level porous-electrode or equivalent-circuit models for performance metrics. Cross-scale demonstrations have revealed consistent scaling trends laws linking SEI thickness, ionic resistance, and energy efficiency in both Li-ion cells and ionic-liquid supercapacitors. Remaining challenges include rigorous parameter transfer, uncertainty propagation, and maintaining consistent boundary conditions for potential, solvation, and geometry across scales.212,308,3096.6 Model benchmarking and future directions
Reliable SEI modeling requires consistent methodologies and quantitative validation. While battery modeling benefits from established community benchmarks, comparable datasets for capacitors are scarce.310,311 Developing shared protocols encompassing standard boundary conditions, force-field benchmarks, and open reaction databases would enable direct cross-comparison and accelerate transferability of results. Operando XPS, Raman, and cryo-TEM provide indispensable validation for both technologies, while ML surrogates trained on combined battery-capacitor datasets may expose common descriptors such as local electric-field intensity, surface work function, and solvation number.312 Outstanding challenges include bridging the temporal gap between nanosecond simulations and hour-scale degradation, expanding reaction libraries beyond carbonates to nitriles and ionic liquids, and quantifying mechanical coupling between SEI formation and electrode deformation.313,314 Progress in these directions, anchored by interoperable datasets and open-source workflows, will transform SEI modeling from qualitative visualization to quantitative prediction, ultimately enabling rational interphase design that combines the energy density of batteries with the power capability of supercapacitors.The multiscale simulation frameworks established in this section provide the theoretical foundation for the machine-learning approaches discussed in Section 7, where data-driven methods accelerate prediction, enable inverse design, and bridge the gap between atomistic accuracy and device-scale applicability.
7 Machine learning and data-driven SEI design
Machine learning (ML) has become a transformative tool for understanding and designing solid-electrolyte interphases (SEIs) across all electrochemical energy-storage systems. Whether in lithium-ion batteries or high-power supercapacitors, the SEI emerges from coupled electrochemical and chemical processes that span multiple scales—from bond breaking and radical formation to film growth, ion transport, and mechanical reconstruction. Because these processes are complex and interdependent, traditional modeling alone cannot capture their full hierarchy. Data-driven approaches, trained on both simulation and experimental results, now provide an adaptive framework that connects quantum accuracy with device-scale predictability. This section highlights how machine-learning interatomic potentials, spectroscopic analytics, and hybrid multiscale workflows jointly accelerate SEI discovery and design in both batteries and supercapacitors.7.1 Machine learning-enhanced potentials for atomistic simulation
Machine-learning interatomic potentials (MLIPs) bridge the gap between first-principles fidelity and large-scale molecular dynamics (MD) simulation applicable to complex interfacial chemistry.315,316 Traditional classical force fields, while efficient, cannot capture bond formation, charge polarization, or reactive dynamics essential for SEI evolution. MLIPs trained on density-functional theory (DFT) data incorporate these phenomena at near-ab initio accuracy but at orders-of-magnitude lower computational cost.315–317 This paradigm has already proven valuable in both LiPF6/EC battery electrolytes and TEABF4/ACN capacitor electrolytes, indicating that the same ML frameworks can learn transferable interfacial chemistry across different devices.318A typical MLIP workflow begins with the generation of a representative dataset encompassing bulk electrolyte configurations, electrode surfaces (including doped carbons, Li metal, MXenes, and defects), solvation shells, and early decomposition products.319,320 Neural-network or graph-neural-network architectures—such as Deep Potential (DeepMD-kit),321 NequIP,322 Allegro,323 and SpookyNet—predict energies and forces from atomic configurations while enforcing rotational and translational equivariance (E(3) symmetry).324 Such architectures capture the diverse bonding motifs present in SEIs: inorganic fluorides, organic carbonates, nitrile polymers, and metal–oxygen–fluorine species. Their data efficiency enables accurate modeling even when trained on relatively small, chemically diverse DFT datasets.66,325
When applied to SEI formation, MLIPs reproduce both the reductive decomposition of solvents and the nucleation of inorganic fragments. For example, the same potential can learn EC → Li2CO3 reduction pathways relevant to batteries and ACN → poly(CN) polymerization seen in capacitors.67,320 Because MLIPs combine reactivity with scalability, they capture film nucleation, cross-linking, and mechanical densification—phenomena inaccessible to fixed-charge MD. They also allow microsecond-scale trajectories and nanometer-scale system sizes, bridging the gap between ab initio MD and continuum models.67
Nevertheless, successful deployment demands broad and physically balanced training sets. Datasets must cover the full configuration space—bulk, interface, radical intermediates, and decomposed fragments—to avoid extrapolation artifacts. In both batteries and capacitors, charge transfer and polarization are key; MLIPs must include charge-equilibration schemes or explicit electrostatics.326 Transferability across potential windows and electrode chemistries remains a challenge: models trained solely on bulk or low-bias conditions can fail under extreme potentials unless continually retrained. Active learning, uncertainty quantification, and on-the-fly data generation mitigate these issues by automatically sampling configurations where prediction confidence is low.327,328
Thus, MLIPs offer a unified platform: they translate electronic-structure fidelity to mesoscale simulations, enabling predictive studies of SEI formation, growth, and failure across all electrochemical systems.
7.2 Data-driven interpretation of experimental characterization
As the SEI increasingly defines the performance envelope of both batteries and supercapacitors, the ability to predict its chemistry from experimental observations becomes critical.15 ML-based analysis of spectroscopic and imaging data now provides this capability, transforming large, heterogeneous datasets into quantitative chemical maps.329High-dimensional data acquired from X-ray photoelectron spectroscopy (XPS), time-of-flight secondary-ion mass spectrometry (ToF-SIMS), Raman and infrared spectroscopy, or hyperspectral electron microscopy contain the full chemical fingerprint of interphases but are notoriously difficult to deconvolute.330 Traditional chemometric techniques—principal-component analysis, multivariate curve resolution—offer limited linear separation and cannot capture nonlinear correlations between spectral features and local chemistry.331 In contrast, modern ML methods such as convolutional neural networks (CNNs), autoencoders, and graph-based architectures excel at uncovering hidden relationships among peaks, textures, and morphologies.332
In SEI analysis, supervised networks are trained on curated datasets linking spectra or images to ground-truth composition obtained via XPS deconvolution or ex situ chemical analysis. For example, CNNs applied to ToF-SIMS maps can distinguish inorganic-rich from polymer-rich domains; recurrent networks analyzing operando Raman time-series can detect transition points from stable to degrading interphases. Cross-domain learning between battery and capacitor datasets—where both share core chemical species (C–O, C–F, LiF, BFxOy)—improves generalization and reduces data requirements.
The benefits are multifold. First, ML accelerates screening of electrode/electrolyte combinations by predicting probable SEI compositions from rapid spectral snapshots.67 Second, data-derived composition vectors can serve as direct inputs for continuum or transport models, eliminating assumptions about generic SEI chemistry.333 Third, inverse modeling enables performance-to-chemistry inference: given capacitance fade or impedance growth, ML can estimate likely compositional shifts and identify underlying degradation pathways.334
To ensure reliability, datasets must include diverse electrode materials (graphite, doped carbons, MXenes, Li metal), electrolyte families (carbonates, nitriles, ionic liquids), and cycling histories.335–337 Label accuracy and balance are critical; semi-supervised learning, transfer learning, and data augmentation help address data scarcity and imbalance. Interpretability tools—such as attention maps and SHAP analyses—link spectral features to specific bonds or chemical groups, ensuring that models retain physical meaning.338 Uncertainty quantification should accompany all predictions, particularly when models guide design decisions or automated optimization.
As large, labelled spectral and imaging repositories become available, ML-based SEI composition prediction will evolve from a diagnostic tool to a predictive engine guiding electrolyte and electrode design in both battery and capacitor technologies (Fig. 10).339
7.3 Computational design of optimized interphases
Machine learning now acts as the connective tissue linking atomistic simulations, materials discovery, and device-level performance modeling. In both Li-ion batteries and supercapacitors, the SEI evolves through reaction-diffusion-mechanical processes spanning orders of magnitude in time and space.340 ML frameworks integrate information from these scales to predict, optimize, and ultimately design interphases with tailored functionality. To make this mapping explicit, we provide one illustrative, end-to-end example (inputs → model targets → interphase properties → device metrics) using a supercapacitor-relevant configuration.Illustrative ML mapping example (supercapacitor EDLC case). Consider an activated carbon EDLC operated near the upper stability limit in an organic electrolyte (e.g., TEABF4/acetonitrile) where ultrathin SEI-like films may form and alter leakage and impedance.341 An ML pipeline that links chemistry to performance can be defined with a transparent input–output structure:• Inputs (descriptors) provided to the model. (i) Electrolyte descriptors: solvent reduction/oxidation tendency (e.g., computed/estimated redox potentials), donor number/polarity, viscosity, dielectric constant; salt identity and anion chemistry; additive identity and concentration.342,343 (ii) Electrode descriptors: surface functional groups/heteroatom content (XPS-derived O/C, N/C), defect density proxy (Raman ID/IG), and pore-size regime (micro/mesopore fraction).344 (iii) Operating protocol: Vmax (or cell window), presence/absence of voltage holds, temperature, and current density (or scan rate).344
• Training labels/targets (what the model learns to predict). Two linked target layers are used: (i) interphase properties (chemistry/structure): film thickness (EQCM mass-to-thickness proxy or microscopy), inorganic/organic fraction (e.g., F-containing species, carbonates, nitrile-derived oligomers from XPS/ToF-SIMS), and an effective ionic/electronic resistivity proxy (from impedance fitting under a stated circuit hypothesis).345 (ii) Device metrics: leakage current or self-discharge rate, ESR evolution from EIS, and capacitance retention over cycling/holds (as recommended in the evidence-to-metric reporting framework (Fig. 11)).346
• Model output and interpretation. Given candidate electrolyte/additive choices and a protocol near Vmax, the trained model outputs: (i) a predicted interphase state vector {d, finorg, ρion/elec} and (ii) predicted metric trends {Δleakage, ΔESR, ΔC} under the specified protocol.347 A beneficial prediction corresponds to leakage/self-discharge reduction without persistent ESR growth and with stable retention; a detrimental prediction corresponds to ESR drift and/or retention loss consistent with pore blocking or resistive thickening.254
• Decision/use case (actionable). The pipeline is then used to rank candidate electrolyte/additive/protocol combinations by a stability objective, e.g., minimize leakage and ESR drift subject to a retention constraint. Importantly, this example is illustrative: the review does not claim a single universal model, but clarifies how reported descriptors (electrolyte/electrode/protocol) can be mapped to measurable interphase properties and supercapacitor-relevant metrics in a reproducible ML workflow.
Together, these approaches constitute an ML-driven multiscale paradigm where data, physics, and experiment converge. By linking microscopic descriptors of SEI chemistry (bond energies, solvation motifs) to macroscopic metrics (resistance, capacitance retention), ML transforms SEI research from descriptive observation to predictive and generative design, unifying battery and capacitor interphase engineering.
7.4 Infrastructure for reproducible SEI research
The foundation of machine-learning progress is high-quality, interoperable data. While the battery community has begun establishing open datasets for electrolyte decomposition and SEI characterization, comparable resources for supercapacitors remain scarce. A unified benchmark repository—spanning both technologies—would dramatically accelerate cross-fertilization of modeling approaches.352,353An ideal dataset would record electrolyte composition (solvent, salt, additive, concentration), electrode characteristics (surface area, functionalization, defect density), cycling conditions (voltage range, current density, cycle number), measured SEI properties (thickness, composition from XPS/ToF-SIMS, ionic/electronic resistance), and device-level performance (capacitance retention, self-discharge, ESR). Consistent metadata—temperature, humidity, geometry—must accompany every entry. Such structured datasets would allow ML models trained on one class of devices to transfer learning to another, enabling joint benchmarking and discovery of shared interphase descriptors.19
Recent community initiatives314,354 advocate standardized protocols and open-source repositories for interphase modeling. Adapting these practices to the supercapacitor domain—and linking them with battery databases—will create the first truly interoperable SEI dataset ecosystem. This infrastructure will enable reproducibility, quantitative benchmarking, and generative design workflows where algorithms autonomously propose optimal electrolyte/electrode combinations for specific stability, conductivity, or mechanical targets.
7.5 Emerging paradigms in interphase discovery
Machine learning and data-driven modeling now stand at the frontier of SEI research.186 Their integration with physical theory (Section 6) and experimental validation (Section 4) promises a future in which interphase chemistry is not merely characterized but computationally designed. By unifying datasets, algorithms, and modeling protocols across batteries and supercapacitors, the community can establish a shared predictive foundation—accelerating discovery of robust, adaptive SEIs that deliver both the high energy of batteries and the high power of capacitors.355,356The modeling and machine-learning frameworks established in Sections 6 and 7 provide the theoretical and computational tools for SEI prediction and optimization. The materials and interface engineering strategies consolidated in Section 5 show how deliberate control of electrode surfaces, electrolyte chemistry, artificial coatings, and mechanical stabilization enables targeted SEI design in real devices.
8 Connecting SEI microstructure to device performance
The architecture and performance of supercapacitors are governed not only by the individual components (electrodes, electrolyte, and separator) but by the interplay of their interfaces, most notably the electrode–electrolyte interphase. In batteries this corresponds to the classical SEI, whereas in supercapacitors it is often an SEI-like interphase. This section explores how the microscopic features of these interphases, encompassing composition, thickness, ionic resistance, uniformity, and morphology, translate into macroscopic device parameters such as capacitance, leakage current, equivalent series resistance (ESR), self-discharge, and cycle life.21,357,358 We then examine how these interfacial attributes may be incorporated into device-level models (equivalent circuits, porous-electrode frameworks) to enable predictive links between materials design and performance.359,3608.1 Microstructural determinants of electrochemical metrics
The solid-electrolyte interphase (SEI) exerts a decisive influence on the electrochemical performance of supercapacitors by bridging nanoscale interfacial chemistry with macroscopic device behavior. Because the SEI occupies the immediate region between the electrode surface and the electrolyte, its thickness, composition, and uniformity directly regulate ion accessibility, charge–transfer pathways, and parasitic reactions.357,361 A thin, ion-permeable SEI enables rapid ion adsorption and desorption, preserving high specific capacitance and low internal resistance. In contrast, a thick or defective SEI impedes ion migration, reduces accessible surface area, and induces localized ionic starvation, manifesting as lower capacitance and higher equivalent-series resistance (ESR). Experimental aging studies consistently link capacitance fading and ESR growth to electrolyte decomposition, pore blockage, and interfacial degradation.21,357,358Beyond capacitance, the SEI critically modulates leakage current and self-discharge. Leakage current in a charged supercapacitor partly arises from unintended faradaic reactions at the electrode–electrolyte boundary. A stable SEI that is electronically insulating yet ionically conductive suppresses these parasitic currents and minimizes self-discharge, whereas a heterogeneous or discontinuous interphase promotes ongoing electrolyte breakdown and charge loss.16,362 From an electrical standpoint, both ionic and electronic resistances contribute to ESR: ion transport through the SEI film and continuity of the conductive network across the electrode/SEI–electrolyte junction determine overall device impedance. As the SEI thickens or becomes more tortuous, ESR rises, diminishing rate capability and power output.146,359
These mechanistic correlations translate directly into measurable device-level outcomes, but meaningful comparison across studies requires that each claim be reported together with (i) electrolyte identity, (ii) cell configuration, and (iii) voltage window/protocol. For example, CNT-based electrodes protected by an ultrathin ALD-derived Al2O3 interlayer show reduced leakage current and improved capacitance retention relative to uncoated controls (cell: symmetric two-electrode; electrolyte: 1 M TEABF4/ACN; window: 0–4 V; protocol: galvanostatic cycling + voltage-hold aging).159,363,364 Likewise, fluoroethylene carbonate (FEC) as an electrolyte additive promotes a thinner, LiF-enriched interphase that suppresses parasitic reactions and improves capacitance retention (cell: symmetric two-electrode; electrolyte: 1 M LiPF6/EC:DMC + 5 wt% FEC; window: 0–2.7 V; protocol: galvanostatic cycling).365,366 In pseudocapacitive hybrid electrodes (e.g., MnO2/graphene), controlled voltage protocols and electrolyte selection can suppress ESR growth and dissolution (cell: two-electrode asymmetric; electrolyte: 0.5 M Na2SO4 (aq); window: 0–1.8 V; rate: 1 A g−1).189,216 In these cases, post-cycling surface analysis (XPS/ToF-SIMS/FTIR) is most informative when explicitly tied to the above operating conditions and to quantitative metric drift (leakage/self-discharge, ESR evolution, retention).367,368 Accordingly, throughout this review we report supercapacitor interphase evidence in a minimally comparable format: electrolyte + voltage window/protocol + cell configuration + at least one metric (leakage/self-discharge, ESR drift, retention).
Collectively, these observations establish a coherent interphase-to-performance link: optimized SEI formation through artificial coatings, electrolyte or additive design, and mechanical stabilization enhances ion accessibility, suppresses leakage and ESR growth, and extends cycle life. Treating the SEI as a designed component rather than a passive by-product thus enables supercapacitors that combine high energy and power densities with exceptional long-term reliability (Fig. 12).369,370
 | ||
| Fig. 12 (1) Schematic overview linking the physical Li/electrolyte system with KMC-based SEI growth modeling and continuum-scale electrochemical transport. Reproduced from ref. 371 with permission from Nature Publishing Group, Copyright 2013. (2) Equivalent circuit models for the SEI analysis a metal foil equivalent circuit. Reproduced from ref. 145 with permission from IOP Publishing, copyright 2024. (3) SEI-thickness-distribution (a) and corresponding capacity fade including the cycle number (b) Reproduced from ref. 372 with permission from IOP Publishing, copyright 2017. | ||
8.2 Embedding interphase physics in device models
To translate interphase-level understanding into device-level predictive capability, modeling frameworks must embed SEI properties explicitly. Two modelling paradigms dominate: equivalent circuit modelling and porous-electrode (continuum) modelling.Equivalent circuit models represent the supercapacitor as an ideal capacitor in series and parallel with resistive and reactive elements: typically capacitance (C), equivalent series resistance (ESR), leakage resistance (Rleak), and sometimes a distributed transmission-line component to represent pore impedance.359 Within this simplified framework, the SEI may be represented as an additional resistive and capacitive layer: a series resistance and a parallel leakage path reflecting ion transport and parasitic reactions respectively. As the interphase evolves (thickness increases, transport resistance grows, composition changes), the corresponding circuit parameters change.359,373 For example, ESR rises, leakage resistance lowers, and effective capacitance is reduced. Careful calibration of equivalent-circuit elements to EIS data can link impedance evolution to interfacial degradation, but for porous electrodes the mapping is not one-to-one.374,375 Mid-frequency arcs can originate from interphase films (Rf‖CPEf), electronic/contact limitations, or pore transport/surface charging that is better represented by a transmission-line model (TLM).376 Therefore, we recommend reporting EIS-derived interphase metrics only within an explicit interpretation framework: (i) fit at least one porous-electrode-aware circuit (TLM-based) in addition to a lumped circuit; (ii) demonstrate parameter robustness across state-of-charge/voltage window and electrode thickness; and (iii) corroborate the assigned interphase element using independent interphase evidence (XPS/ToF-SIMS/cryo-TEM thickness/chemistry or EQCM mass evolution). Under these constraints, trends in fitted film resistance/capacitance can be used as quantitative indicators of interphase growth, pore blockage, or contact degradation rather than as a unique SEI “signature.”
Porous-electrode models build on more physically realistic descriptions of ionic and electronic transport within electrode architectures. They couple mass transport in electrolyte, ion adsorption at electrode surfaces (double-layer or pseudocapacitive behaviour), electron conduction in the solid phase, and interfacial kinetics.359 In such frameworks, the SEI can be modelled as an interfacial film with defined thickness, ionic conductivity, porosity, and reaction kinetics.360 For example, ionic diffusion through pore networks is slowed by an SEI film that reduces pore diameter or increases tortuosity; thereby, the effective ionic diffusivity and active surface area are reduced.359 Recent reviews of transport in charged porous media highlight how film layers and pore constrictions modulate ion accessibility and charge dynamics. By embedding SEI parameters into porous electrode models (for instance, adding an ionic resistance layer, reducing pore cross-section, or adjusting double-layer capacitance per unit area), one obtains predictive time-dependent performance simulations: charge–discharge curves, rate capability envelopes, EIS Nyquist spectra evolution, and cycle life prediction. These models enable bridging between atomic-scale SEI properties (thickness, composition, ionic transport) and system metrics (capacitance retention, ESR drift, self-discharge). Moreover, multi-physics modelling (coupling electrochemical, thermal and mechanical submodels) can incorporate temperature effects on SEI conductivity, mechanical cracking of the film, and consequent performance degradation (Fig. 13–15).212,380
 | ||
| Fig. 14 Representative porous-electrode transmission-line schematics used to describe distributed impedance in porous systems: (a) a detailed pore model including electrolyte resistance external to the pore (Re), solution resistance inside the pore (Rs,idx), metal resistance (Rm,idx), and distributed local impedance elements (Z1/dx, Z2/dx). Reproduced from ref. 381 with permission from IOP Publishing, copyright 2009; and (b) a simplified ladder-network representation highlighting distributed ionic/electronic pathways and interfacial contributions, including pore resistance (rp), series resistance (rs), pore capacitance (Cp), and terminal impedance terms Zelectron(D, K) and Zgas. Reproduced from ref. 382 with permission from the Korean Ceramic Society, copyright 2016, under CC BY-NC 4.0 (http://creativecommons.org/licenses/by-nc/4.0/). These schematics illustrate how porous-electrode architectures naturally generate distributed impedance responses, motivating porous-electrode-aware interpretation of Nyquist features. | ||
 | ||
Fig. 15 Representative impedance analyses showing that Nyquist features in porous electrochemical systems arise from multiple overlapping processes rather than from a unique SEI resistance. Panels (a) and (b) illustrate ideal and depressed semicircles, highlighting the influence of electrode polarization and interfacial heterogeneity on arc shape. Reproduced from ref. 383 with permission from De Gruyter, copyright 2021, under CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/). Panels (c) and (d) show the linear relationship between  and ω−0.5 before and after cycling, indicating diffusion-related impedance evolution. Panels (e) and (f) present Nyquist plots before cycling and after 50 cycles with corresponding equivalent-circuit fits, showing that cycling alters the combined contributions of bulk transport, charge transfer, interfacial response, and diffusion. Reproduced from ref. 382 with permission from the Korean Ceramic Society, copyright 2016, under CC BY-NC 4.0 (http://creativecommons.org/licenses/by-nc/4.0/). Together, these panels emphasize that impedance changes must be interpreted with physically justified circuit models and supporting interphase evidence. and ω−0.5 before and after cycling, indicating diffusion-related impedance evolution. Panels (e) and (f) present Nyquist plots before cycling and after 50 cycles with corresponding equivalent-circuit fits, showing that cycling alters the combined contributions of bulk transport, charge transfer, interfacial response, and diffusion. Reproduced from ref. 382 with permission from the Korean Ceramic Society, copyright 2016, under CC BY-NC 4.0 (http://creativecommons.org/licenses/by-nc/4.0/). Together, these panels emphasize that impedance changes must be interpreted with physically justified circuit models and supporting interphase evidence. | ||
8.3 Synergistic experimental-computational platforms
In modern supercapacitor research, the interphase between electrode and electrolyte no longer remains a mere passive layer but has evolved into a controllable interface through the convergence of in situ/operando measurement techniques, molecular dynamics (MD) modelling, and machine learning (ML)-based data interpretation.384 This closed-loop approach begins with high-temporal-resolution experimental measurements such as electrochemical impedance spectroscopy (EIS), electrochemical quartz-crystal microbalance (EQCM), operando Raman or X-ray reflectivity, and cryo-TEM imaging, all of which provide real-time signatures of the solid-electrolyte interphase (SEI) formation, ion transport changes and morphological evolution under cycling.171,385 These experimental datasets feed into ML algorithms that rapidly interpret spectral, imaging and impedance features, infer SEI metrics (e.g., thickness, ionic resistance, porosity, heterogeneity) and compare them to predictive models derived from MD. The ML output then informs MD (or ML-surrogate) simulations under the updated interphase state, applied voltage protocols and electrolyte conditions.62,386 Simultaneously, device-level performance metrics, including capacitance drift, leakage current increase, and equivalent series resistance (ESR) growth, are monitored to validate or recalibrate the model, thereby creating a feedback loop: experiment → ML insight → simulation → updated experiment.For supercapacitors, where high rate cycling, thin films and rapid interphase evolution are typical, this feedback loop enables real-time interphase management. For example, when impedance growth accelerates unexpectedly, the ML model can signal that the SEI ionic resistance has reached a threshold, prompting an adjusted cycling protocol (e.g., reduced voltage hold, rest period) or trigger an additive injection. The MD component provides mechanistic insight—how desolvation barriers, pore blockage or ion-film interaction evolve with film composition—thus strengthening the predictive fidelity of the workflow.387 This proactive framework transforms the SEI from a passive, post-mortem phenomenon into an actively managed interface.
The power of this integrated loop extends to accelerated lifetime prediction: early-cycle fingerprints of SEI growth (e.g., rapid increase in ionic resistance or specific Raman shifts) are correlated by ML with long-term cycle performance, enabling the simulation of interphase evolution under projected usage conditions. This anticipatory capability is especially valuable in high-power supercapacitors, where ensuring interphase stability over thousands of fast cycles is a prerequisite. By tying simulation parameters to actual device conditions (ion concentration, pore wetting state, morphological change) rather than idealised models, the feedback approach significantly reduces the gap between laboratory insight and real-world device behaviour.388
Nevertheless, successful deployment of such in situ/ML/MD feedback loops demands rigorous data synchronisation (experiment vs. simulation time scales), transparency of ML decision-making (avoiding black-box predictions), and sufficiently large and diverse training datasets linking SEI signatures to performance across cycling regimes and chemistries. In spite of these challenges, the closed-loop paradigm stands as a transformative route toward predictive, real-time SEI control and device-level interphase optimisation in next-generation supercapacitors.389
8.4 Interphase engineering for advanced device configurations
Hybrid supercapacitors, which combine characteristics of both batteries (high energy) and capacitors (high power), alongside solid-state supercapacitor architectures impose distinctive demands on the solid-electrolyte interphase (SEI).390 In battery-capacitor hybrids (such as Li-ion or K-ion insertion anode paired with a capacitive cathode), the SEI must tolerate high current density, large ion flux, and broad voltage swings while enabling rapid ion exchange typical of capacitors. In such systems, the negative electrode often undergoes faradaic insertion and SEI formation similar to battery anodes.391,392 Studies on non-aqueous potassium-ion hybrid supercapacitors reveal that formation protocols influence SEI composition (e.g., KF content) and cycling stability, demonstrating the critical role of the interphase in hybrid performance.393,394 Meanwhile, in solid-state supercapacitors (polymer or ceramic electrolytes), the interphase becomes a “solid–solid” boundary layer that must maintain mechanical and ionic continuity under compression, bending or thermal cycling. Here, the SEI must be ionically conductive, electronically insulating, and mechanically robust, often serving simultaneously as the electrolyte interface and protective interphase.284From a modelling and engineering perspective, these hybrid and solid-state systems demand tailored SEI strategies. For hybrids, simulation must incorporate ion insertion kinetics, interphase growth under dynamic flux, and coupling between faradaic and capacitive processes.395 For solid-state systems, interphase modelling must include mechanical deformation, adhesion, stress propagation, and film continuity under mechanical load.284 The engineering implications are significant: electrodes must be designed to tolerate mechanical strain and rapid flux, while electrolytes must be formulated to produce interphases that resist delamination, preserve ion pathways and mitigate interface degradation.396 Accordingly, SEI design in hybrid supercapacitors demands a holistic view integrating insertion chemistry, interphase transport, mechanical resilience and high-rate operation, enabling devices that deliver both high energy and high power with durability.390
8.5 Design for sustainability and end-of-life recovery
Beyond immediate performance improvements, the engineering of the SEI must address long-term sustainability and recyclability of supercapacitor technologies.70 The interphase layer, frequently composed of decomposition products, polymer fragments or engineered coatings, influences device lifecycle, end-of-life behaviour and material recovery. From a materials-circularity standpoint, SEI design must consider environmentally benign solvents, salts and additives, low-toxicity coatings, and film formulations that facilitate safe disposal or regeneration. For instance, avoiding high-toxicity fluorinated additives or unstable polymer residues may reduce environmental footprint and simplify recycling workflows.397Recycling supercapacitors often requires predictable interphase chemistry: electrodes coated with stable but removable or convertible layers allow more efficient separation of electrode, current collector and electrolyte during end-of-life processing. Engineered SEIs that deposit uniform inorganic salt layers (rather than uncontrolled polymeric debris) enable more consistent recycling behaviour and material recovery. Moreover, the reduction of self-discharge and leakage current through optimized SEI design extends device lifetime and thereby reduces waste associated with early replacement.
From a manufacturing and lifecycle perspective, SEI engineering also influences formation times (with energy savings), cell safety (reduced leakage), and module durability (fewer replacements).71 By integrating sustainability considerations into interphase design—selecting benign chemistries, designing films that facilitate separation or recovery, and minimising long-term degradation—researchers align high performance with environmental and economic viability.186,398 Ultimately, treating the SEI not only as a performance filter but as a sustainability fulcrum ensures that next-generation supercapacitors deliver high energy, high power and high longevity while embodying circular-economy principles.
8.6 Summary
This section has demonstrated how SEI microstructure—composition, thickness, ionic resistance, and morphology—directly determines macroscopic device metrics including capacitance, leakage current, ESR, and cycle life. By embedding interphase parameters into equivalent-circuit and porous-electrode models, and by establishing in situ feedback loops that couple MD, ML, and experiment, researchers can now predict and control device performance from first principles. The insights from Sections 2 through 8—spanning fundamental electrochemistry, microscopic mechanisms, experimental characterization, multiscale modeling, machine learning, materials engineering, and device integration—collectively establish a comprehensive framework for SEI science in supercapacitors. Section 9 builds on this foundation to identify strategic future directions that will transform SEI engineering from an empirical art to a predictive science.9 Future directions and opportunities
As the field of solid-electrolyte interphase (SEI) engineering for supercapacitors matures, several overarching opportunities emerge that promise to elevate interphase design from empirical trial-and-error to predictive, data-driven, and sustainable engineering. We highlight five strategic frontiers: unification of interphase theory across batteries and capacitors; artificial-intelligence (AI)-accelerated multiscale simulation for real-time interface evolution; open databases for SEI chemistries and degradation pathways; coupling quantum-electronic transport simulations with interfacial models; and design of sustainable electrolytes and eco-friendly SEI layers for next-generation energy storage.9.1 Unified theory across energy-storage modalities
Historically, SEI research has primarily focused on rechargeable batteries, especially lithium-ion systems, where the SEI at the anode plays a critical role in capacity retention and safety.134,399 In contrast, SEI phenomena in supercapacitors have received only limited attention, despite the fact that electrode–electrolyte interphases in high-power capacitive devices face analogous challenges (e.g., electrolyte decomposition, ion-transport impediment, interphase mechanical fatigue). A unifying theoretical framework that spans both battery and supercapacitor interphases would facilitate cross-pollination of mechanistic insights—such as electron tunnelling, ion–solvent reorganisation, interphase growth kinetics and mechanical failure modes.19,28Such convergence requires systematic comparison of interphase chemistries, growth pathways, transport resistances and mechanical behaviours under the distinct operating regimes of batteries (slow charge, deep insertion, large volumetric changes) and capacitors (fast charge/discharge, high ion flux, minimal diffusion depth). By leveraging common descriptors (e.g., electron tunnelling barrier, ion-film diffusivity, film fracture toughness), researchers can develop shared modelling tools, standardised measurement protocols and unified degradation metrics.
9.2 Machine-learning-enhanced multiscale modeling
A second frontier lies in the deployment of artificial intelligence (AI) to accelerate multiscale simulations that capture SEI formation and evolution in real time. Recent advances in physics-informed machine-learning workflows have shown that neural-network potentials and surrogate models can bridge atomic resolution to device timescales by several orders of magnitude.400,401 For SEI in supercapacitors, a coherent pipeline might begin with density-functional theory (DFT) calculations of solvent/salt reduction barriers, feed into molecular dynamics (MD) or reactive-force-field simulations of early film nucleation, and then leverage ML surrogates to predict film growth, ionic transport and impedance evolution over thousands of rapid charge/discharge cycles.Incorporating AI into this loop enables adaptive simulations: based on evolving interphase properties (thickness, porosity, composition) the ML model adjusts boundary conditions or cycling protocols and predicts future behaviour before experimental validation.402 Such “closed-loop modelling” is particularly powerful in supercapacitors, where fast kinetics and high-rate protocols dominate and where limited time exists for long-term ageing experiments.
9.3 Community data repositories and standards
The third strategic opportunity is the creation of shared, open databases linking electrolyte formulation, electrode surface chemistry, SEI composition and performance degradation metrics.354 Currently, data in interphase research are fragmented—individual studies report specific electrode/electrolyte pairs and SEI analyses, but lack standard formats, metadata or cross-system comparability. Establishing a comprehensive database would enable machine-learning models to learn from a broader chemical space, quantify trends, identify outliers and propose new interphase chemistries.A meaningful SEI database could include parameters such as solvent identity, salt species and concentration, additive content, electrode material and surface functionalisation, SEI thickness/composition (XPS, ToF-SIMS), ionic resistance and capacitance fade across cycling.354 This repository would support benchmarking of simulation models, facilitate transfer learning across systems, and catalyse inverse design of interphases.
9.4 First-principles treatment of interfacial charge transfer
While much of SEI modelling emphasises ion transport, film growth kinetics and mass-transport limitations, a crucial—but less explored—axis is electron transport through the interphase.403 Recent quantum-transport studies reveal that heterogeneous interfaces (e.g., LiF/Li2CO3 combinations) severely influence electron tunnelling barriers and defect-mediated leakage paths.404 For supercapacitors operating at high voltages and fast charge/discharge rates, even minor electron conduction through the interphase may trigger continuous electrolyte breakdown, self-discharge or power loss.Thus, coupling quantum-mechanical simulations (NEGF, DFT) of interphase materials with MD and continuum models offers a holistic picture where electron tunnelling, ion migration, solvent breakdown and film evolution all interplay.404 Integrating these quantum calculations into multiscale modelling enables designers to propose SEIs that are not only ionically conductive but also electronically insulating—minimising parasitic reactions and enhancing cycle life.
9.5 Green chemistry and eco-friendly electrolytes
Finally, the longevity, recyclability and environmental footprint of energy storage systems depend heavily on the interphase. While performance has often dominated research priorities, future supercapacitors must align with circular-economy principles. This means deploying electrolytes that are non-toxic, flame-retardant and low-cost, and engineering interphases (including SEI-like films where relevant) that are stable but amenable to safe end-of-life processing.68,405Sustainable SEI design involves selecting solvents and salts whose decomposition products are benign, coatings and additives that avoid critical raw-material scarcity, and interphases that enable recycling of electrode materials without extensive chemical breakdown. For instance, replacing fluorine-rich additives with alternative film-formers or employing bio-derived polymer interphases could reduce environmental burden while maintaining performance.68 Formation protocols that minimise energy consumption, reduce gas evolution and limit parasitic side-products will further improve life-cycle impact.
By embracing these five strategic directions—unified theory, AI-driven simulation, open data, quantum-informed modelling and sustainability—SEI engineering for supercapacitors can transition from incremental improvements to a generative, predictive design paradigm. This transformation will underpin the development of high-energy, high-power, durable and environmentally compatible supercapacitor technologies.
10 Concluding remarks and outlook
The scientific understanding of SEIs has advanced substantially over the past decade, driven by parallel progress in interface-sensitive experimental techniques and increasingly sophisticated computational methods. While traditional electrochemical measurements and ex situ surface analyses laid the foundation of SEI research, they are now complemented by operando spectroscopy, cryogenic microscopy, and multiscale simulation frameworks capable of resolving interfacial dynamics with atomic precision. Together, these developments have transformed interphase science from a phenomenological concept into a quantifiable, chemically heterogeneous interphase governing performance in both battery SEIs and supercapacitor SEI-like interphases.In this review, we have focused on the microscopic to mesoscopic mechanisms underlying SEI formation, evolution, and transport, emphasizing the roles of electronic-structure theory, reactive and machine-learning molecular dynamics, continuum models, and device-scale formulations. These approaches, adapted over the years to capture electron transfer, solvation breakdown, and polarization at complex electrode–electrolyte interfaces, now provide access to structural, chemical, and transport properties that were previously inaccessible through experiments alone.
Comparison with experiments provides a critical benchmark for validating multiscale SEI models. In most cases, simulations reproduce the qualitative trends observed in operando XPS, impedance spectroscopy, and cryo-TEM, lending confidence to the underlying assumptions regarding reduction pathways, ion transport, and interphase morphology. Quantitative agreement is increasingly attainable, particularly when simulation parameters are calibrated against ab initio energetics and experimentally measured reaction barriers, though such accuracy often comes at the cost of extensive sampling and high computational demand. The primary benefit of these computational approaches lies in their ability to resolve nanoscale structure, electron redistribution, and decomposition kinetics that remain invisible to most experimental probes. In batteries and supercapacitors alike, these insights have clarified the origins of inorganic–organic layering, elucidated the polarization-controlled onset of electrolyte reduction, and revealed how local electric fields guide SEI nucleation. Moreover, simulations have begun to suggest rational strategies for interface engineering, such as tuning solvent composition, optimizing surface terminations, and exploiting heterogeneous polarization, to stabilize thin, ionically conductive interphases. Although many of these design principles remain challenging to implement experimentally, they provide mechanistic routes for future improvements in interphase stability and device lifetime.
Despite these successes, significant challenges remain for achieving a fully predictive description of SEI formation and evolution across electrochemical technologies. In both battery and supercapacitor systems, the accessible time and length scales of atomistic simulations remain far smaller than those associated with long-term interphase growth, mechanical reconstruction, or electrolyte aging. Bridging these gaps will require tighter coupling between reactive MD, ML-augmented potentials, and continuum models capable of capturing ion transport, stress generation, and dissolution/redeposition dynamics. Methodologically, two major obstacles must be addressed. First, current force fields and ML potentials, while increasingly accurate, still struggle to represent the full chemical diversity of real electrolytes, particularly when multiple solvents, salts, or radical intermediates participate simultaneously in reduction processes. Extending these models to incorporate more explicit electronic descriptors and rare–event pathways will be essential for capturing system-specific reactivity. Second, transferring parameters and boundary conditions across scales remains nontrivial: polarization, solvation, and porosity must be represented consistently from the quantum level to the porous–electrode scale. These issues, combined with the computational cost of reactive simulations and the memory demands of high-resolution mesoscale models, continue to limit the direct simulation of realistic electrode architectures. Overcoming these bottlenecks is therefore central to advancing SEI modeling in the coming years.
In the case of MD-based approaches, substantial progress has been made in representing electrode surfaces and interfacial chemistry, yet important limitations persist. More explicit treatment of electronic structure, particularly localized charge transfer, image-charge effects, and tunneling, would markedly improve predictions of reduction onset and interphase composition, while incorporating bond flexibility and mechanical compliance in electrode models would enable the study of stress-driven SEI cracking and reconstruction during cycling. On the electrolyte side, models have evolved from fixed-charge representations to polarizable and reactive descriptions, yet coupling self-consistent solvent polarization with dynamically fluctuating electrode charges remains technically challenging and is not widely available in mainstream MD engines. Finally, although supercapacitor electrolytes undergo fewer faradaic reactions than battery systems, rare decomposition events can still influence long-term stability, but modeling these pathways lies beyond the reach of most classical and semiclassical methods. Ongoing efforts in battery SEI modeling — particularly multistate reaction networks, ML-accelerated rare-event sampling, and electronically informed reactive potentials—provide a valuable foundation, and adapting these advances to capacitor chemistries will be essential for capturing degradation and interphase evolution in high-voltage ionic-liquid and organic systems.
However, the most difficult challenges arise when extending SEI modeling to the broader family of emerging electrode and electrolyte chemistries used in both batteries and supercapacitors. While this review concentrated on carbonaceous systems and conventional carbonate or acetonitrile-based electrolytes, next-generation interfaces increasingly involve multicomponent materials whose reactive behavior and electronic structure differ substantially from the paradigms commonly studied. High-voltage spinel oxides, doped carbons, MXenes, and redox-active 2D materials introduce heteroatom-dependent surface states, variable metallicity, and nonuniform polarization, all of which complicate the description of electron transfer and electrolyte decomposition. Similarly, advanced electrolytes such as ionic liquids, water-in-salt solutions, fluorinated additives, and hybrid organic–inorganic systems exhibit complex solvation and reduction pathways that current empirical and reactive potentials do not always capture reliably. Properly representing these systems will require refined models capable of differentiating site-specific charge response, accommodating element-dependent hardness, and resolving situations in which only a subset of atomic species participate in metallic screening. Incorporating these chemical descriptors into constant-potential, reactive, and machine-learning based frameworks remains a demanding task, but doing so is essential for accurately predicting SEI composition and stability across the increasingly diverse set of materials used in modern electrochemical energy-storage devices.
Even greater complexity emerges when considering SEI formation on transition-metal oxides, alloying hosts, and other redox-active surfaces where proton transfer, surface reconstruction, and multielectron redox processes strongly influence interphase chemistry. In aqueous or hybrid electrolytes, these mechanisms couple tightly to solvent reorganization and acid–base equilibria, placing them outside the reliable domain of classical MD and most reactive force fields. Capturing such effects requires more advanced methods, most notably DFT-based ab initio molecular dynamics or finite-field electronic-structure calculations capable of resolving charge transfer, interfacial polarization, and proton-coupled electron transfer at each time step. Initial demonstrations of these approaches have successfully described early reduction events and potential-dependent solvation structure, but their applicability remains limited by system size: ab initio MD can typically simulate only a few hundred atoms, far smaller than the extended interfaces and nanostructured electrodes relevant for practical SEI studies. Machine-learned interatomic potentials trained on large DFT datasets offer a promising route to bridge this gap, providing near-DFT accuracy with classical scalability. However, current models remain at an early stage for complex electrolytes and multicomponent electrode surfaces, and substantial development is still required to ensure transferability across diverse chemistries and operating conditions.
Recent experimental advances have likewise expanded the landscape of SEI-relevant electrolytes. Highly concentrated formulations such as water-in-salt and solvent-in-salt systems introduce new interfacial chemistries, exhibiting nanostructured solvation environments and nonclassical reduction pathways that remain poorly understood at the molecular scale. Their complex organization near charged interfaces, together with experimentally observed coupling to pore topology and surface functionality, presents an open challenge for simulation and demands models capable of capturing collective structuring, fluctuating ion–solvent clusters, and voltage-dependent solvation breakdown. Emerging chemistries such as ionic liquids, fluorinated additives, redox-active ionic liquids, and hybrid organic-inorganic electrolytes offer routes to engineer more robust, self-limiting SEIs, yet their redox behavior requires methods that can treat reactive events and multiple electronic states with comparable accuracy. Conventional MD lacks this capability, motivating the development of electronically informed reactive potentials and nonadiabatic approaches such as surface-hopping to resolve coupled electron–ion dynamics. Taken together, these directions indicate that SEI research across batteries and supercapacitors will remain a vibrant field in the coming decade, with substantial opportunities for advancing microscopic simulation methods, expanding chemical realism, and establishing predictive models that unify interphase behavior across diverse electrochemical systems.
Key challenges and opportunities for future SEI research:
• Extending reactive and ML-certified force fields to cover the full chemical complexity of modern electrolytes and electrode surfaces (mixed solvents, ionic liquids, doped carbons, 2D materials, etc.).
• Building robust multiscale coupling pipelines that consistently preserve solvation, polarization, and porosity across electronic, atomistic, mesoscale, and device levels.
• Implementing constant-potential, electronically informed reactive simulations that are computationally tractable for realistic electrode architectures and long-time evolution.
• Developing open, interoperable datasets and benchmark protocols covering both battery and supercapacitor chemistries, to enable transfer learning, model validation, and community-wide comparison.
Future Roadmap: Short-, Mid-, and Long-Term Priorities for Interphase Engineering To maximize impact and accelerate translation from mechanistic insight to device-level reliability, we outline practical research priorities across three time horizons.
Short-term priorities (0–2 years): establish comparability and causal evidence. (i) Standardize reporting of interphase claims using a minimum set of operating details (electrolyte identity, voltage window, protocol/holds, cell configuration) together with at least one metric from each category: leakage/self-discharge, ESR evolution (EIS), and capacitance retention. (ii) Cross-validate characterization by routinely pairing surface chemistry (XPS/ToF-SIMS/FTIR) with operando electrochemical signatures (EIS/EQCM) to distinguish reversible pseudocapacitance from irreversible film growth. (iii) Define practical onset thresholds (e.g., Vcrit). for common electrolyte classes and electrode families under realistic cycling and hold protocols, enabling voltage-window selection as a design constraint rather than a performance target.
Mid-term priorities (2–5 years): engineer controllable, durable SEI-like interphases. (i) Protocol-guided formation (conditioning/stepwise ramping) and additive/coating strategies should be optimized against metric-first objectives (lower leakage and stable ESR, not chemistry alone). (ii) Porous-electrode-aware impedance interpretation (transmission-line models, DRT analysis) should become routine so that fitted interphase parameters are physically meaningful and comparable across laboratories. (iii) Materials-by-design interphases: develop ultrathin, ion-permeable, mechanically resilient coatings and binder networks that suppress defect-localized decomposition while preserving fast ion access and pseudocapacitive redox sites.
Long-term priorities (5–10 years): predictive, closed-loop interphase design across platforms. (i) Multiscale, validated digital twins integrating atomistic chemistry, mesoscale porous transport, and device-level degradation should enable quantitative lifetime prediction under realistic duty cycles (including holds and temperature variations). (ii) Data-driven inverse design: closed-loop workflows combining high-throughput experiments/simulations with ML should optimize electrolyte/electrode/protocol combinations toward targeted interphase state vectors (thickness, inorganic/organic fraction, effective resistivity) linked to stability metrics. (iii) Sustainable interphase engineering: prioritize electrolytes/additives and coatings with improved environmental compatibility and recyclability, while maintaining passivation efficacy and long-term safety.
Collectively, these priorities convert the unified interphase perspective into a practical roadmap for achieving high-voltage, long-life supercapacitors and transferable interface-control principles across next-generation electrochemical energy-storage technologies.In summary, while the road ahead is challenging, the convergence of advanced experiments, physics-based modeling, and machine learning offers strong prospects for a predictive, technology-agnostic SEI science — one capable of steering future interface design for both high-energy and high-power electrochemical devices.
Conflicts of interest
There are no conflicts of declare.Data availability
Data sharing is not applicable to this article as no new data were generated or analysed.References
- K. Detka and K. Górecki, Selected technologies of electrochemical energy storage—a review, Energies, 2023, 16, 5034 CrossRef CAS.
- G. Navarro, J. Torres, M. Blanco, J. Nájera, M. Santos-Herran and M. Lafoz, Present and future of supercapacitor technology applied to powertrains, renewable generation and grid connection applications, Energies, 2021, 14, 3060 CrossRef CAS.
- T. Ramachandran, R. K. Raji and M. Rezeq, From lab to market: the future of zinc–air batteries powered by MOF/MXene hybrids, J. Mater. Chem. A, 2025, 13, 12855–12890 RSC.
- M. S. Whittingham, Ultimate limits to intercalation reactions for lithium batteries, Chem. Rev., 2014, 114, 11414–11443 CrossRef CAS.
- J. B. Goodenough and K.-S. Park, The Li-ion rechargeable battery: a perspective, J. Am. Chem. Soc., 2013, 135, 1167–1176 CrossRef CAS PubMed.
- I. Beyers, A. Bensmann and R. Hanke-Rauschenbach, Ragone plots revisited: A review of methodology and application across energy storage technologies, J. Energy Storage, 2023, 73, 109097 CrossRef.
- A. Burke, Ultracapacitors: why, how, and where is the technology, J. Power Sources, 2000, 91, 37–50 CrossRef CAS.
- J. Ahn, M. M. Doeff and G. Chen, Strategies and Perspectives on Enhancing Energy Density in Lithium-Ion Batteries, ECS Meet. Abstr., 2024, 245, 509 CrossRef.
- Y. Lin, S. Zhang, L. Guan and J. Tao, Prospect of Ni-related metal oxides for high-performance supercapacitor electrodes, J. Mater. Sci., 2021, 56, 1897–1918 CrossRef CAS.
- C. Choi, D. S. Ashby, D. M. Butts, R. H. DeBlock, Q. Wei, J. Lau and B. Dunn, Achieving high energy density and high power density with pseudocapacitive materials, Nat. Rev. Mater., 2020, 5, 5–19 CrossRef.
- S. Li, Z. Luo, L. Li, J. Hu, G. Zou, H. Hou and X. Ji, Recent progress on electrolyte additives for stable lithium metal anode, Energy Storage Mater., 2020, 32, 306–319 CrossRef.
- Y. Wang, Y. Song and Y. Xia, Electrochemical capacitors: mechanism, materials, systems, characterization and applications, Chem. Soc. Rev., 2016, 45, 5925–5950 RSC.
- E. Peled, The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems—the solid electrolyte interphase model, J. Electrochem. Soc., 1979, 126, 2047 CrossRef CAS.
- E. Peled and S. Menkin, SEI: past, present and future, J. Electrochem. Soc., 2017, 164, A1703 CrossRef CAS.
- H. Adenusi, G. A. Chass, S. Passerini, K. V. Tian and G. Chen, Lithium batteries and the solid electrolyte interphase (SEI)—progress and outlook, Adv. Energy Mater., 2023, 13, 2203307 CrossRef CAS.
- S. J. An, J. Li, C. Daniel, D. Mohanty, S. Nagpure and D. L. Wood, The state of understanding of the lithium-ion-battery graphite solid electrolyte interphase (SEI) and its relationship to formation cycling, Carbon, 2016, 105, 52–76 CrossRef CAS.
- H. Wu, H. Jia, C. Wang, J.-G. Zhang and W. Xu, Recent progress in understanding solid electrolyte interphase on lithium metal anodes, Adv. Energy Mater., 2021, 11, 2003092 CrossRef CAS.
- X.-B. Cheng, R. Zhang, C.-Z. Zhao, F. Wei, J.-G. Zhang and Q. Zhang, A review of solid electrolyte interphases on lithium metal anode, Adv. Sci., 2016, 3, 1500213 CrossRef PubMed.
- J. Tan, L. Ma, Z. Li, Y. Wang, M. Ye and J. Shen, Structural insights into solid electrolyte interphase (SEI) on lithium metal anode: From design strategies to the stability evaluation, Mater. Today, 2023, 69, 287–332 CrossRef CAS.
- A. Wang, S. Kadam, H. Li, S. Shi and Y. Qi, Review on modeling of the anode solid electrolyte interphase (SEI) for lithium-ion batteries, npj Comput. Mater., 2018, 4, 15 CrossRef.
- J. E. Owejan, J. P. Owejan, S. C. DeCaluwe and J. A. Dura, Solid electrolyte interphase in Li-ion batteries: evolving structures measured in situ by neutron reflectometry, Chem. Mater., 2012, 24, 2133–2140 CrossRef CAS.
- W. Huang, P. M. Attia, H. Wang, S. E. Renfrew, N. Jin, S. Das, Z. Zhang, D. T. Boyle, Y. Li and M. Z. Bazant, others Evolution of the solid–electrolyte interphase on carbonaceous anodes visualized by atomic-resolution cryogenic electron microscopy, Nano Lett., 2019, 19, 5140–5148 CrossRef CAS PubMed.
- P. G. Kitz, M. J. Lacey, P. Novák and E. J. Berg, Operando EQCM-D with simultaneous in situ EIS: new insights into interphase formation in Li ion batteries, Anal. Chem., 2018, 91, 2296–2303 CrossRef PubMed.
- J. D. McBrayer, C. A. Apblett, K. L. Harrison, K. R. Fenton and S. D. Minteer, Mechanical studies of the solid electrolyte interphase on anodes in lithium and lithium ion batteries, Nanotechnology, 2021, 32, 502005 CrossRef CAS PubMed.
- P. Simon and Y. Gogotsi, Materials for electrochemical capacitors, Nat. Mater., 2008, 7, 845–854 CrossRef CAS PubMed.
- M. Salanne, B. Rotenberg, K. Naoi, K. Kaneko, P.-L. Taberna, C. P. Grey, B. Dunn and P. Simon, Efficient storage mechanisms for building better supercapacitors, Nat. Energy, 2016, 1, 1–10 Search PubMed.
- E. Frackowiak, Electrode materials with pseudocapacitive properties, Supercapacitors: Materials, Systems, and Applications, 2013, pp. 207–237 Search PubMed.
- T. Quan, E. Hark, Y. Xu, I. Ahmet, C. Hohn, S. Mei and Y. Lu, Unveiling the formation of solid electrolyte interphase and its temperature dependence in “water-in-salt” supercapacitors, ACS Appl. Mater. Interfaces, 2021, 13, 3979–3990 CrossRef CAS PubMed.
- M.-E. Yvenat, B. Chavillon, E. Mayousse, E. De Vito, A. Boulineau, F. Perdu and P. Azaïs, Study of the influence of the formation protocol on the SEI layer formed at the graphite electrode surface of a non-aqueous potassium-ion hybrid supercapacitor (KIC) through STEM and XPS analyses, Sustainable Energy & Fuels, 2023, 7, 4150–4159 RSC.
- T. Ramachandran, F. Hamed, Y. A. Kumar, R. K. Raji and H. Hegazy, Multifunctional covalent-organic frameworks (COFs)-2D MXenes composites for diverse applications, J. Energy Storage, 2023, 73, 109299 CrossRef.
- E. Pameté, L. Köps, F. A. Kreth, S. Pohlmann, A. Varzi, T. Brousse, A. Balducci and V. Presser, The many deaths of supercapacitors: degradation, aging, and performance fading, Adv. Energy Mater., 2023, 13, 2301008 CrossRef.
- G. S. Hwang, Fundamental Understanding of Faradaic and Non-Faradaic Processes in Carbon-Based Supercapacitors, ECS Meet. Abstr., 2019, 235, 644 CrossRef.
- C. Ding, T. Liu, X. Yan, L. Huang, S. Ryu, J. Lan, Y. Yu, W.-H. Zhong and X. Yang, An ultra-microporous carbon material boosting integrated capacitance for cellulose-based supercapacitors, Nano-Micro Lett., 2020, 12, 63 CrossRef CAS PubMed.
- A. M. Bogale, T. Ramachandran, M. E. Suk, B. B. Badassa, M. M. Solomon, J. He, A. Yusuf, R. K. Raji, B. A. Zenebe and N. K. Amare, others Boosted charge storage in symmetric supercapacitors using Zn-Co/MgCo2O4 hybrid nanosheets, J. Phys. Chem. Solids, 2025, 113079 Search PubMed.
- A. Dey, Film formation on lithium anode in propylene carbonate, J. Electrochem. Soc., 1970, 117 Search PubMed.
- V. A. Agubra and J. W. Fergus, The formation and stability of the solid electrolyte interface on the graphite anode, J. Power Sources, 2014, 268, 153–162 CrossRef CAS.
- R. Fong, U. Von Sacken and J. R. Dahn, Studies of lithium intercalation into carbons using nonaqueous electrochemical cells, J. Electrochem. Soc., 1990, 137, 2009 CrossRef CAS.
- E. Peled, D. Golodnitsky and G. Ardel, Advanced model for solid electrolyte interphase electrodes in liquid and polymer electrolytes, J. Electrochem. Soc., 1997, 144, L208 CrossRef CAS.
- D. Aurbach, B. Markovsky, M. Levi, E. Levi, A. Schechter, M. Moshkovich and Y. Cohen, New insights into the interactions between electrode materials and electrolyte solutions for advanced nonaqueous batteries, J. Power Sources, 1999, 81, 95–111 CrossRef.
- Y. Wang, S. Nakamura, M. Ue and P. B. Balbuena, Theoretical studies to understand surface chemistry on carbon anodes for lithium-ion batteries: reduction mechanisms of ethylene carbonate, J. Am. Chem. Soc., 2001, 123, 11708–11718 CrossRef CAS PubMed.
- J. Christensen and J. Newman, A mathematical model for the lithium-ion negative electrode solid electrolyte interphase, J. Electrochem. Soc., 2004, 151, A1977 CrossRef CAS.
- Y. S. Jung; A. S. Cavanagh; L. A. Riley; S.-H. Kang; A. C. Dillon; M. D. Groner; S. M. George; S.-H. LeeUltrathin Direct Atomic Layer Deposition on Composite Electrodes for Highly Durable and Safe Li-Ion Batteries. 2010, Search PubMed.
- S. Shi, P. Lu, Z. Liu, Y. Qi, L. G. Hector Jr, H. Li and S. J. Harris, Direct calculation of Li-ion transport in the solid electrolyte interphase, J. Am. Chem. Soc., 2012, 134, 15476–15487 CrossRef CAS PubMed.
- A. v. Cresce, S. M. Russell, D. R. Baker, K. J. Gaskell and K. Xu, In situ and quantitative characterization of solid electrolyte interphases, Nano Lett., 2014, 14, 1405–1412 CrossRef CAS PubMed.
- A. C. Kozen, C.-F. Lin, A. J. Pearse, M. A. Schroeder, X. Han, L. Hu, S.-B. Lee, G. W. Rubloff and M. Noked, Next-generation lithium metal anode engineering via atomic layer deposition, ACS Nano, 2015, 9, 5884–5892 CrossRef CAS PubMed.
- E. Peled and H. Yamin, Solid Electrolyte Interphase (SEI) Electrodes. Part 1. The Kinetics of Lithium in LiAlCl4-SOCl2, Isr. J. Chem., 1979, 18, 131–135 CrossRef CAS.
- E. Peled, Film forming reaction at the lithium/electrolyte interface, J. Power Sources, 1983, 9, 253–266 CrossRef CAS.
- J. M. Lim, Y. S. Jang, H. V. T. Nguyen, J. S. Kim, Y. Yoon, B. J. Park, D. H. Seo, K.-K. Lee, Z. Han and K. K. Ostrikov, others Advances in high-voltage supercapacitors for energy storage systems: materials and electrolyte tailoring to implementation, Nanoscale Adv., 2023, 5, 615–626 RSC.
- M. Pathak, D. Bhatt, R. C. Bhatt, B. S. Bohra, G. Tatrari, S. Rana, M. C. Arya and N. G. Sahoo, High energy density supercapacitors: an overview of efficient electrode materials, electrolytes, design, and fabrication, Chem. Rec., 2024, 24, e202300236 CrossRef CAS PubMed.
- A. Patel, S. K. Patel, R. Singh and R. Patel, Review on recent advancements in the role of electrolytes and electrode materials on supercapacitor performances, Discover Nano, 2024, 19, 188 CrossRef CAS PubMed.
- R. Yuan, Y. Dong, R. Hou, S. Zhang and H. Song, Influencing factors and suppressing strategies of the self-discharge for carbon electrode materials in supercapacitors, J. Electrochem. Soc., 2022, 169, 030504 CrossRef CAS.
- D. Xu, X. Zhang, K. Zhang, Y. Han, X. Sun, Y. Xu, C. Li, K. Wang, X. Zhang and Y. Ma, Recent advances in high-voltage lithium-ion capacitors, J. Solid State Electrochem., 2025, 1–27 Search PubMed.
- Y. Yao, X. Rui, R. Bai, Y. Ouyang, G. Li, Y. Zhao, Y.-H. Zhu, M. Zhao, B.-Q. Li and X. Zhang, others Roadmap for Next-Generation Electrochemical Energy Storage Technologies: Secondary Batteries and Supercapacitors, ACS Nano, 2025, 19, 30568–30687 CrossRef CAS PubMed.
- X. Liu, D. Lyu, C. Merlet, M. J. Leesmith, X. Hua, Z. Xu, C. P. Grey and A. C. Forse, Structural disorder determines capacitance in nanoporous carbons, Science, 2024, 384, 321–325 CrossRef CAS PubMed.
- K. Mazloomian, H. Lancaster, C. Howard, P. Shearing and T. Miller, Supercapacitor degradation: Understanding mechanisms of cycling-induced deterioration and failure of a pseudocapacitor, Batteries Supercaps, 2023, 6(8), e202300214 CrossRef CAS.
- Y. Wu, G. Ge, S. Wang, L. Xiong and Z. He, Formation mechanisms of solid electrolyte interphase and its influence on lithium battery performance, Mater. Today Energy, 2025, 102124 CrossRef CAS.
- H. Wan, J. Xu and C. Wang, Designing electrolytes and interphases for high-energy lithium batteries, Nat. Rev. Chem., 2024, 8, 30–44 CrossRef CAS PubMed.
- X. Jiang, H. Zhang, Y. Qu, Z. Wang, Y. Xie, W. Zhang, H. Hu and Z. He, Engineering electrolyte strong-weak coupling effect toward wide-temperature supercapacitor, Energy Storage Mater., 2024, 68, 103374 CrossRef.
- Y. B. Yohannes, S. D. Lin, N.-L. Wu and B.-J. Hwang, SEI grown on MCMB-electrode with fluoroethylene carbonate and vinylene carbonate additives as probed by in situ DRIFTS, J. Electrochem. Soc., 2019, 166, A2741 CrossRef CAS.
- P. G. Kitz, M. J. Lacey, P. Novák and E. J. Berg, Operando investigation of the solid electrolyte interphase mechanical and transport properties formed from vinylene carbonate and fluoroethylene carbonate, J. Power Sources, 2020, 477, 228567 CrossRef CAS.
- P. Jankowski, W. Wieczorek and P. Johansson, Functional ionic liquids: cationic SEI-formers for lithium batteries, Energy Storage Mater., 2019, 20, 108–117 CrossRef.
- M. Y. Yang, S. V. Zybin, T. Das, B. V. Merinov, W. A. Goddard, E. K. Mok, H. J. Hah, H. E. Han, Y. C. Choi and S. H. Kim, Characterization of the solid electrolyte interphase at the Li metal–ionic liquid interface, Adv. Energy Mater., 2023, 13, 2202949 CrossRef CAS.
- F. Du, H. Wang, J. Chen, H. Huang, T. Ye, S. Duan, R. Zhang, J. Liu and H. Hu, Atomistic Insights into Fluorinated/Chlorinated Ether-Based LHCEs on Lithium Metal: Decomposition Mechanisms via Multiscale Simulations, J. Phys. Chem. B, 2025, 129, 8919–8932 CrossRef CAS PubMed.
- B. Kim, H. Shin, J. Choi and M. Cho, Multiscale modeling of load transfer characteristics in crosslinked epoxy nanocomposites, Mech. Adv. Mater. Struct., 2022, 29, 4768–4778 CrossRef.
- Y. You, D. Zhang, F. Wu, X. Cao, Y. Sun, Z.-Z. Zhu and S. Wu, Principal component analysis enables the design of deep learning potential precisely capturing LLZO phase transitions, npj Comput. Mater., 2024, 10, 57 CrossRef CAS.
- J. Chen, X. Fan, Q. Li, H. Yang, M. R. Khoshi, Y. Xu, S. Hwang, L. Chen, X. Ji and C. Yang, others Electrolyte design for LiF-rich solid–electrolyte interfaces to enable high-performance microsized alloy anodes for batteries, Nat. Energy, 2020, 5, 386–397 CrossRef CAS.
- X. Liu, V. Koverga, H. T. Nguyen, A. T. Ngo and T. Li, Exploring solvation structure and transport behavior for rational design of advanced electrolytes for next generation of lithium batteries, Applied Physics Reviews, 2024, 11, 021307 CrossRef CAS.
- Z. Ju, X. Tao, Y. Wang, Q. Yang, T. Liu, J. Nai, W. Zhang, S. Chen, Y. Liu and H. Tian, others A self-healing Li-crosslinked elastomer promotes a highly robust and conductive solid–electrolyte interphase, Energy Environ. Sci., 2024, 17, 4703–4713 RSC.
- Y. Liu, D. Lin, P. Y. Yuen, K. Liu, J. Xie, R. H. Dauskardt and Y. Cui, An artificial solid electrolyte interphase with high Li-ion conductivity, mechanical strength, and flexibility for stable lithium metal anodes, Adv. Mater., 2016, 29, 1605531 CrossRef PubMed.
- B. Ng, P. T. Coman, W. E. Mustain and R. E. White, Non-destructive parameter extraction for a reduced order lumped electrochemical-thermal model for simulating Li-ion full-cells, J. Power Sources, 2020, 445, 227296 CrossRef CAS.
- Z. Huang, J.-C. Lai, S.-L. Liao, Z. Yu, Y. Chen, W. Yu, H. Gong, X. Gao, Y. Yang and J. Qin, others A salt-philic, solvent-phobic interfacial coating design for lithium metal electrodes, Nat. Energy, 2023, 8, 577–585 CrossRef CAS.
- S. Sikiru, J. O. Olutoki, S. A. Musa, L. O. Afolabi and M. M. Mahat, The projection role of surface charge density in electric double-layer capacitance for enhanced energy storage, J. Energy Storage, 2025, 138, 118728 CrossRef CAS.
- S. SHIRAISHI and Y. HATAKEYAMA, Electrode Carbon Material for Electric Double Layer Capacitors, Vac. Surf. Sci., 2019, 62, 703–708 CrossRef CAS.
- T. Ramachandran, F. Hamed, R. K. Raji, S. M. Majhi, D. Barik, Y. A. Kumar, R. M. Jauhar, M. Pachamuthu, L. Vijayalakshmi and S. Ansar, Enhancing asymmetric supercapacitor performance with NiCo2O4–NiO hybrid electrode fabrication, J. Phys. Chem. Solids, 2023, 180, 111467 CrossRef CAS.
- T. Ramachandran, A.-H. I. Mourad, R. K. Raji, R. Krishnapriya, N. Cherupurakal, A. Subhan and Y. Al-Douri, KOH mediated hydrothermally synthesized hexagonal-CoMn2O4 for energy storage supercapacitor applications, Int. J. Energy Res., 2022, 46, 16823–16838 CrossRef CAS.
- B. U. Tale, K. R. Nemade and P. V. Tekade, Novel graphene based MnO2/polyaniline nanohybrid material for efficient supercapacitor application, J. Porous Mater., 2024, 31, 2053–2065 CrossRef CAS.
- R.-J. Zhu, J. Liu, C. Hua, H.-Y. Pan, Y.-J. Cao and M. Li, Preparation of vanadium-based electrode materials and their research progress in solid-state flexible supercapacitors, Rare Met., 2024, 43, 431–454 CrossRef CAS.
- T. Ramachandran, M. Pachamuthu, R. Raji and F. R. M. S. Raj, A Layered Fe–SnO2/MXene Nanohybrid with 1225.6 F/g Capacitance and Long-Term Cycle Durability, Mater. Chem. Phys., 2025, 131860 Search PubMed.
- B. E. Conway, Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications, pringer Science & Business Media, 2013 Search PubMed.
- J. Zheng, P. Cygan and T. Jow, Hydrous ruthenium oxide as an electrode material for electrochemical capacitors, J. Electrochem. Soc., 1995, 142, 2699 CrossRef CAS.
- J. Phojaroen, P. Wuamprakhon, T. Sangsanit, K. Santiyuk, K. Homlamai, N. Anansuksawat, W. Tejangkura, R. Songthan and M. Sawangphruk, Investigation of Electrolyte Decomposition Byproducts in Gas and Liquid Phases Due to Water Impurities in Large-Scale Acetonitrile-Based Supercapacitors, Batteries Supercaps, 2025, 8, e202400738 CrossRef CAS.
- R. Songthan, J. Phojaroen, T. Sangsanit, P. Wuamprakhon, W. Tejangkura and M. Sawangphruk, Correlation Between Electrolyte Degradation Products and Overcharging Voltages in Supercapacitors, J. Electrochem. Soc., 2025, 172, 040509 CrossRef CAS.
- K. Mazloomian, H. J. Lancaster, C. A. Howard, P. R. Shearing and T. S. Miller, Supercapacitor degradation: understanding mechanisms of cycling-induced deterioration and failure of a pseudocapacitor, Rare Met., 2023, 6, e202300214 CAS.
- O. Stern, Zur theorie der elektrolytischen doppelschicht, Z. Phys. Chem., 1924, 30, 508–516 CAS.
- M. Gouy, Sur la constitution de la charge électrique à la surface d’un électrolyte, J. Phys. Theor. Appl., 1910, 9, 457–468 CrossRef.
- D. L. L. I. Chapman, A contribution to the theory of electrocapillarity, London, Edinburgh Dublin Philos. Mag. J. Sci., 1913, 25, 475–481 CrossRef.
- T. Dong, T. Qin, W. Zhang, Y. Zhang, Z. Feng, Y. Gao, Z. Pan, Z. Xia, Y. Wang and C. Yang, others Nature of the electric double layer to modulate the electrochemical behaviors of Fe2O3 electrode, Acta Mater., 2024, 263, 119500 CrossRef CAS.
- K. Ge, H. Shao, Z. Lin, P.-L. Taberna and P. Simon, Advanced characterization of confined electrochemical interfaces in electrochemical capacitors, Nat. Nanotechnol., 2025, 20, 196–208 CrossRef CAS PubMed.
- J. Tan, Z. Li, M. Ye and J. Shen, Nanoconfined space: Revisiting the charge storage mechanism of electric double layer capacitors, ACS Appl. Mater. Interfaces, 2022, 14, 37259–37269 CrossRef CAS PubMed.
- J. Grill and J. Popovic-Neuber, Long term porosity of solid electrolyte interphase on model silicon anodes with liquid battery electrolytes, Commun. Chem., 2024, 7, 297 CrossRef CAS PubMed.
- R. Akhter and S. S. Maktedar, MXenes: A comprehensive review of synthesis, properties, and progress in supercapacitor applications, J. Materiomics, 2023, 9, 1196–1241 CrossRef.
- S. Zhao, G. Li, B. Zhang, T. Li, M. Luo, B. Sun, G. Wang and S. Guo, Technological roadmap for potassium-ion hybrid capacitors, Joule, 2024, 8, 922–943 CrossRef CAS.
- Z. Huang, X. Li, Z. Chen, P. Li, X. Ji and C. Zhi, Anion chemistry in energy storage devices, Nat. Rev. Chem., 2023, 7, 616–631 CrossRef PubMed.
- R. He, L. Zhou, R. Tenent and M. Zhou, Basics of the scanning electrochemical microscope and its application in the characterization of lithium-ion batteries: a brief review, Mater. Chem. Front., 2023, 7, 662–678 RSC.
- J. Chen, J. Vatamanu, L. Xing, O. Borodin, H. Chen, X. Guan, X. Liu, K. Xu and W. Li, Improving electrochemical stability and low-temperature performance with water/acetonitrile hybrid electrolytes, Adv. Energy Mater., 2020, 10, 1902654 CrossRef CAS.
- Z. T. Gossage, N. Ito, T. Hosaka, R. Tatara and S. Komaba, Understanding the Development and Properties of SEI in Concentrated Aqueous Electrolytes Via Scanning Electrochemical Microscopy, ECS Meet. Abstr., 2023, 244, 2900 CrossRef.
- F. Wang and J. Cheng, Unraveling the origin of reductive stability of super-concentrated electrolytes from first principles and unsupervised machine learning, Chem. Sci., 2022, 13, 11570–11576 RSC.
- S. Ko, T. Obukata, T. Shimada, N. Takenaka, M. Nakayama, A. Yamada and Y. Yamada, Electrode potential influences the reversibility of lithium-metal anodes, Nat. Energy, 2022, 7, 1217–1224 CrossRef CAS.
- K.-H. Ko, K. Kim, Y. Kim, S. Han, J. Park, H. Park, J. Yang, B. Kim, J. Park and K. Kang, Degradation Mechanism Induced by Depth-Dependent Inhomogeneity in Thick High-Areal-Capacity Graphite Electrode, Small, 2025, 21, 2410795 CrossRef CAS PubMed.
- Z. Chen, D. L. Danilov, L. H. Raijmakers, K. Chayambuka, M. Jiang, L. Zhou, J. Zhou, R.-A. Eichel and P. H. Notten, Overpotential analysis of graphite-based Li-ion batteries seen from a porous electrode modeling perspective, J. Power Sources, 2021, 509, 230345 CrossRef CAS.
- J. G. Lee, J. Kim, T. J. Lee, J. B. Lee, H.-s. Kim, J. Soon, J. H. Ryu and S. M. Oh, Damage of Solid Electrolyte Interphase (SEI) Layer By the Mechanical Stress Induced By Volume Change of Si Electrode, ECS Meet. Abstr., 2016, 1140 CrossRef.
- R. D. Deshpande and D. M. Bernardi, Modeling solid-electrolyte interphase (SEI) fracture: coupled mechanical/chemical degradation of the lithium ion battery, J. Electrochem. Soc., 2017, 164, A461 CrossRef CAS.
- M. Pavone, F. Fasulo and A. B. Munoz Garcia, Reactivity of VC Electrolyte at Li-Metal Electrode: New Insights on SEI Initial Formation from Density Functional Embedding Theory, ECS Meet. Abstr., 2022, 242, 486 CrossRef.
- G. Zhang, X. Wei, X. Wang, S. Chen, J. Zhu and H. Dai, Evolution mechanism and non-destructive assessment of thermal safety for lithium-ion batteries during the whole lifecycle, Nano Energy, 2024, 126, 109621 CrossRef CAS.
- P. Barai and V. Srinivasan, Impact of Solid Electrolyte Interphase (SEI) Stiffness on Lithium Dendrite Growth, ECS Meet. Abstr., 2019, 235, 100 CrossRef.
- L. Suo, D. Oh, Y. Lin, Z. Zhuo, O. Borodin, T. Gao, F. Wang, A. Kushima, Z. Wang and H.-C. Kim, others How solid-electrolyte interphase forms in aqueous electrolytes, J. Am. Chem. Soc., 2017, 139, 18670–18680 CrossRef CAS PubMed.
- E. W. C. Spotte-Smith, R. L. Kam, D. Barter, J. Self, X. Xie, T. Hou, S. Dwaraknath, S. M. Blau and K. A. Persson, Towards a Mechanistic Explanation for Solid Electrolyte Interphase Formation in Lithium-Ion Batteries, ECS Meet. Abstr., 2022, 241, 2268 CrossRef.
- J. Ding, J. Lian, X. Li, J. Huang, K. Sun, H. Yang, L. Gong and P. Tan, Quantitative understanding of coupled electron-ion transfer at the silicon-electrolyte interphase of lithium-ion batteries, Chem. Eng. J., 2024, 500, 157109 CrossRef CAS.
- B. Li, Y. Chao, M. Li, Y. Xiao, R. Li, K. Yang, X. Cui, G. Xu, L. Li and C. Yang, others A review of solid electrolyte interphase (SEI) and dendrite formation in lithium batteries, Electrochem. Energy Rev., 2023, 6, 7 CrossRef CAS.
- M. C. Madhusudhanan, S. A. Kumar, S. Nair, N. Srinivasan, M. Buragohain and S. Kunnikuruvan, Revisiting the Relation Between the Stability of the LUMO of the Electrolytes and the Kinetics of Solid Electrolyte Interface Formation in Lithium-and Post-Lithium-ion Batteries, Rare Met., 2023, 6, e202200430 CAS.
- Y. Wang, Y. Liu, Y. Tu and Q. Wang, Reductive decomposition of solvents and additives toward solid-electrolyte interphase formation in lithium-ion battery, J. Phys. Chem. C, 2020, 124, 9099–9108 CrossRef CAS.
- S. M. Russell, A. v. Cresce and K. Xu, Electrolyte-Electrode Interactions and Interphases, Electrochemical Society Meeting Abstracts imlb2016, 2016, pp 303 Search PubMed.
- E. K. Andersson, L.-T. Wu, L. Bertoli, Y.-C. Weng, D. Friesen, K. Elbouazzaoui, S. Bloch, R. Ovsyannikov, E. Giangrisostomi and D. Brandell, others Initial SEI formation in LiBOB-, LiDFOB-and LiBF 4-containing PEO electrolytes, J. Mater. Chem. A, 2024, 12, 9184–9199 RSC.
- S. Banerjee and S. S. Yamijala, Formation and evolution of the solid electrolyte interphase at calcium surfaces, ACS Appl. Energy Mater., 2025, 8, 5936–5947 CrossRef CAS.
- P. Wuamprakhon, J. Phojaroen, T. Sangsanit, K. Santiyuk, K. Homlamai, W. Tejangkura and M. Sawangphruk, Unveiling a Novel Decomposition Pathway in Propylene Carbonate-Based Supercapacitors: Insights from a Jelly Roll Configuration Study, ChemSusChem, 2024, 17, e202400053 CrossRef CAS PubMed.
- B. Karamanova, L. Soserov, E. Lefterova, T. Stankulov and A. Stoyanova, Influence of Acetonitrile on the Electrochemical Behavior of Ionic Liquid-Based Supercapacitors, Batteries, 2024, 10, 266 CrossRef CAS.
- M. M. Amaral, V. Y. Yukuhiro, R. Vicentini, A. C. Peterlevitz, L. M. Da Silva, P. Fernandez and H. Zanin, Direct observation of the CO2 formation and C–H consumption of carbon electrode in an aqueous neutral electrolyte supercapacitor by in-situ FTIR and Raman, J. Energy Chem., 2022, 71, 488–496 CrossRef CAS.
- Y. Yang, J. Li, J. Shi, F. Pan, S. Wu, D. Zhang, Y. Zhu and W. Zhang, Expanding the electrochemical stable window of aqueous-based electrolytes via competitive solvation induced by aprotic solvents, J. Power Sources, 2025, 644, 236925 CrossRef CAS.
- R. Venancio, R. Vicentini, D. A. Corrêa, A. N. Miranda, A. C. Queiroz, F. T. Degasperi, L. J. Siqueira, L. M. Da Silva and H. Zanin, others Combining electrochemical, molecular simulation and operando techniques to investigate the stability of electrodes and organic electrolytes used in EDLCs, Energy Storage Mater., 2023, 62, 102943 CrossRef.
- J. Sun, L. A. O'Dell, M. Armand, P. C. Howlett and M. Forsyth, Anion-derived solid-electrolyte interphase enables long life Na-ion batteries using superconcentrated ionic liquid electrolytes, ACS Energy Lett., 2021, 6, 2481–2490 CrossRef CAS.
- C. Yan, L.-L. Jiang, Y.-X. Yao, Y. Lu, J.-Q. Huang and Q. Zhang, Nucleation and growth mechanism of anion-derived solid electrolyte interphase in rechargeable batteries, Angew. Chem., 2021, 133, 8602–8606 CrossRef.
- L. Wang, J. Guo, Q. Qi, X. Li, Y. Ge, H. Li, Y. Chao, J. Du and X. Cui, Revisiting dipole-induced fluorinated-anion decomposition reaction for promoting a LiF-rich interphase in lithium-metal batteries, Nano-Micro Lett., 2025, 17, 111 CrossRef CAS PubMed.
- H. Wu, J. Qu, X. Yan, S. Zhang, X. Wang, J. Liang, N. Zhang, B. Dai, J. Yue and T. Pang, others Revealing the Underlying Role of Li2CO3 in Enhancing Performance of Oxyhalide-Based Solid-State Batteries, Adv. Mater., 2025, 2502067 CrossRef CAS PubMed.
- J.-A. Lee, C. Song, S. H. Han, B. Kim and N.-S. Choi, Designing Anode-Electrolyte Interfaces for Low-Temperature Lithium-and Sodium-Ion Batteries: Challenges and Strategies, ACS Energy Lett., 2025, 10, 5474–5484 CrossRef CAS.
- C. Prehal, C. Koczwara, N. Jäckel, A. Schreiber, M. Burian, H. Amenitsch, M. A. Hartmann, V. Presser and O. Paris, Quantification of ion confinement and desolvation in nanoporous carbon supercapacitors with modelling and in situ X-ray scattering, Nat. Energy, 2017, 2, 16215 CrossRef CAS.
- A. C. Forse, J. M. Griffin, C. Merlet, J. Carretero-Gonzalez, A.-R. O. Raji, N. M. Trease and C. P. Grey, Direct observation of ion dynamics in supercapacitor electrodes using in situ diffusion NMR spectroscopy, Nat. Energy, 2017, 2, 16216 CrossRef.
- T. Kress, X. Liu and A. C. Forse, Pore network tortuosity controls fast charging in supercapacitors, Nat. Mater., 2025, 1–7 Search PubMed.
- M. E. Şahin, F. Blaabjerg and A. Sangwongwanich, A comprehensive review on supercapacitor applications and developments, Energies, 2022, 15, 674 CrossRef.
- H. Guo, M. Elmanzalawy, P. Sivakumar and S. Fleischmann, Unifying electrolyte formulation and electrode nanoconfinement design to enable new ion–solvent cointercalation chemistries, Energy Environ. Sci., 2024, 17, 2100–2116 RSC.
- R. Subramanian, Electroinitiated polymerization on electrodes. Electric Phenomena in Polymer Science, 2005, pp. 33–58 Search PubMed.
- G.-X. Li, V. Koverga, A. Nguyen, R. Kou, M. Ncube, H. Jiang, K. Wang, M. Liao, H. Guo and J. Chen, others Enhancing lithium-metal battery longevity through minimized coordinating diluent, Nat. Energy, 2024, 9, 817–827 CrossRef CAS.
- M. Nojabaee, D. Kopljar, N. Wagner and K. A. Friedrich, Understanding the Nature of Solid-Electrolyte Interphase on Lithium Metal in Liquid Electrolytes: A Review on Growth, Properties, and Application-Related Challenges, Rare Met., 2021, 4, 909–922 CAS.
- J.-L. Li, Y.-N. Wang, S.-Y. Sun, Z. Zheng, Y. Gao, P. Shi, Y.-J. Zhao, X. Li, Q. Li and X.-Q. Zhang, others Understanding and regulating the mechanical stability of solid electrolyte interphase in batteries, Adv. Energy Mater., 2025, 15, 2403845 CrossRef CAS.
- Y. Gao, Z. Yan, J. L. Gray, X. He, D. Wang, T. Chen, Q. Huang, Y. C. Li, H. Wang and S. H. Kim, others Polymer–inorganic solid–electrolyte interphase for stable lithium metal batteries under lean electrolyte conditions, Nat. Mater., 2019, 18, 384–389 CrossRef CAS PubMed.
- J. M. Asl, Z. Shafieizadeh, M. Anaraki and J. Sabbaghzadeh, Inspecting The Effect Of Thermal Stress On The Interdiffusion Of Metal Multilayer Produced By Electron-Beam Gun Evaporation, AIP Conf. Proc., 2010, 67–70 CrossRef CAS.
- D. Peak and J. Corbett, Diffusion-controlled reaction kinetics, Phys. Rev. B, 1972, 5, 1226 CrossRef.
- A. S. Kulathuvayal and Y. Su, Ionic transport through the solid electrolyte interphase in lithium-ion batteries: A review from first-principles perspectives, ACS Appl. Energy Mater., 2023, 6, 5628–5645 CrossRef CAS.
- Y. Chu, Y. Shen, F. Guo, X. Zhao, Q. Dong, Q. Zhang, W. Li, H. Chen, Z. Luo and L. Chen, Advanced characterizations of solid electrolyte interphases in lithium-ion batteries, Electrochem. Energy Rev., 2020, 3, 187–219 CrossRef.
- A. Syugaev, R. Zonov, K. Mikheev, A. Maratkanova and G. Mikheev, Electrochemical impedance of laser-induced graphene: Frequency response of porous structure, J. Phys. Chem. Solids, 2023, 181, 111533 CrossRef CAS.
- H. Zhu, Z. Li, C. Li, H. Jia, H. Fang, L. Qiao, P. Lv and X. Li, Near-in-situ electrochemical impedance spectroscopy analysis based on lithium iron phosphate electrode, Electrochim. Acta, 2023, 464, 142919 CrossRef CAS.
- M. Levi and D. Aurbach, Impedance spectra of porous, composite intercalation electrodes: The origin of the low-frequency semicircles, J. Power Sources, 2005, 146, 727–731 CrossRef CAS.
- F. La Mantia, J. Vetter and P. Novák, Impedance spectroscopy on porous materials: A general model and application to graphite electrodes of lithium-ion batteries, Electrochim. Acta, 2008, 53, 4109–4121 CrossRef CAS.
- A. Plis, P. Połczyński and R. Jurczakowski, Equivalent circuit for assessing pore size distribution in porous electrodes by EIS, Electrochem. Commun., 2024, 164, 107716 CrossRef CAS.
- L. Xu, Y. Xiao, Z.-X. Yu, Y. Yang, C. Yan and J.-Q. Huang, Revisiting the Electrochemical Impedance Spectroscopy of Porous Electrodes in Li-ion Batteries by Employing Reference Electrode, Angew. Chem., Int. Ed., 2024, 63, e202406054 CrossRef CAS PubMed.
- R. Morasch, H. A. Gasteiger and B. Suthar, Li-ion battery material impedance analysis ii: graphite and solid electrolyte interphase kinetics, J. Electrochem. Soc., 2024, 171, 050548 CrossRef CAS.
- S. D. Talian, S. Brutti, M. A. Navarra, J. Moškon and M. Gaberscek, Impedance spectroscopy applied to lithium battery materials: Good practices in measurements and analyses, Energy Storage Mater., 2024, 69, 103413 CrossRef.
- Z. Sun, J. Yang, H. Xu, C. Jiang, Y. Niu, X. Lian, Y. Liu, R. Su, D. Liu and Y. Long, others Enabling an inorganic-rich interface via cationic surfactant for high-performance lithium metal batteries, Nano-Micro Lett., 2024, 16, 141 CrossRef CAS PubMed.
- Z. Chen, J. Pan, W. Huang, K. Shi, Z. Yang, H. Wu, S. Wei, G. Jiang, W. Zou and R. Zhang, others Heterogeneity-Segment Charge-Induced-Coupling Catalysis of Component-Selective-Type Covalent Organic Frameworks Interface toward Stabilizing Lithium Metal Anode, ACS Nano, 2025, 19, 13160–13174 CrossRef CAS PubMed.
- C. Xu, J. Mu, T. Zhou, S. Tian, P. Gao, G. Yin, J. Zhou and F. Li, Surface Redox Pseudocapacitance Boosting Vanadium Nitride for High-Power and Ultra-Stable Potassium-Ion Capacitors, Adv. Funct. Mater., 2022, 32, 2206501 CrossRef CAS.
- M. Hao, S. Weng, C. Zhong, Y. Li and X. Wang, Structure and evolution of solid electrolyte interphase (SEI) at the electrode-electrolyte interface, Mater. Today Energy, 2025, 101998 CrossRef CAS.
- C. Groher, D. M. Cupid, A. Mautner, E. Rosenberg and J. Kahr, Operando GC/MS for the investigation of different decomposition pathways during solid electrolyte interphase (SEI) formation with SEI forming additives, J. Power Sources, 2024, 605, 234481 CrossRef CAS.
- Y. Liu, K. Dong, T. Lv, Z. Chen, S. Cao, F. Zheng and T. Chen, Constructing bilayer heterogeneous organogel electrolytes of Lewis acidic/basic polymers to suppress self-discharge of supercapacitors, Chem. Eng. J., 2024, 479, 147716 CrossRef CAS.
- Y. Chen, W. Wu, S. Gonzalez-Munoz, L. Forcieri, C. Wells, S. P. Jarvis, F. Wu, R. Young, A. Dey and M. Isaacs, others Nanoarchitecture factors of solid electrolyte interphase formation via 3D nano-rheology microscopy and surface force-distance spectroscopy, Nat. Commun., 2023, 14, 1321 CrossRef CAS PubMed.
- Y. Liang, N. Song, M. Zhang, X. An, K. Song, W. Chen, J. Feng, S. Xiong and B. Xi, Robust Interfacial Chemistry Induced by B-Doping Enables Rapid, Stable Sodium Storage, Adv. Energy Mater., 2023, 13, 2302825 CrossRef CAS.
- J. Chen, X. Rao, F. Zhang, Y. Ein-Eli and D. Q. Tan, Unveiling Activated Carbon Degradation in Supercapacitor Using Liquid Cell Transmission Electron Microscopy, ACS Appl. Energy Mater., 2024, 7, 9797–9805 CrossRef CAS.
- K. Machida, S. Suematsu and K. Tamamitsu, Polyfluorene/carbon nanocomposite for electrochemical capacitors, Electrochemistry, 2007, 75, 601–603 CrossRef CAS.
- S. Kholimatussadiah, C.-L. Hsu, S.-W. Ke, T.-C. Chou, Y.-F. Wu, R. Yakimova, A. Kumatani, K.-H. Chen, L.-C. Chen and H.-Y. Du, In-situ observation of hydrogen nanobubbles formation on graphene surface by AFM-SECM, Electrochim. Acta, 2024, 493, 144425 CrossRef CAS.
- P. Ruch, D. Cericola, A. Foelske-Schmitz, R. Kötz and A. Wokaun, Aging of electrochemical double layer capacitors with acetonitrile-based electrolyte at elevated voltages, Electrochim. Acta, 2010, 55, 4412–4420 CrossRef CAS.
- D. Gandla, G. Song, C. Wu, Y. Ein-Eli and D. Q. Tan, Atomic layer deposition (ALD) of alumina over activated carbon electrodes enabling a stable 4 V supercapacitor operation, ChemistryOpen, 2021, 10, 402–407 CrossRef CAS PubMed.
- S. Fleischmann, J. B. Mitchell, R. Wang, C. Zhan, D.-e. Jiang, V. Presser and V. Augustyn, Pseudocapacitance: from fundamental understanding to high power energy storage materials, Chem. Rev., 2020, 120, 6738–6782 CrossRef CAS PubMed.
- S. Rondinini, A. Minguzzi, E. Achilli, C. Locatelli, G. Agostini, S. Pascarelli, G. Spinolo, A. Vertova and P. Ghigna, The dynamics of pseudocapacitive phenomena studied by Energy Dispersive X-Ray Absorption Spectroscopy on hydrous iridium oxide electrodes in alkaline media, Electrochim. Acta, 2016, 212, 247–253 CrossRef CAS.
- B. R. P. Yip, C. Chen, Y. Jiang, D. Ohayon, G. C. Bazan and X. Wang, Aqueous asymmetric pseudocapacitor featuring high areal energy and power using conjugated polyelectrolytes and Ti3C2T x MXene, Nat. Commun., 2025, 16, 7984 CrossRef CAS PubMed.
- A. Slesinski, S. Sroka, K. Fic, E. Frackowiak and J. Menzel, Operando monitoring of local pH value changes at the carbon electrode surface in neutral sulfate-based aqueous electrochemical capacitors, ACS Appl. Mater. Interfaces, 2022, 14, 37782–37792 CrossRef CAS PubMed.
- K. Palanisamy, S. Daboss, J. Romer, D. Schäfer, M. Rohnke, J. K. Flowers, S. Fuchs, H. S. Stein, M. Fichtner and C. Kranz, Microscopic and spectroscopic analysis of the solid electrolyte interphase at hard carbon composite anodes in 1 M NaPF6/diglyme, Rare Met., 2024, 7, e202300482 CAS.
- Y. Aoki, M. Oda, S. Kojima, Y. Yamaga, T. Ishihama, T. Nagashima, T. Doi and M. Inaba, Effective approach by computational chemical prediction and experimental verification to elucidate SEI formation mechanism in LiPF6-, LiFSI-, and LiBF4-containing electrolyte solutions, J. Phys. Chem. C, 2022, 127, 69–77 CrossRef.
- Y. Li, F. Bai, C. Li, Y. Wang and T. Li, Understanding the inorganic-rich feature of anion-derived solid electrolyte interphase, Adv. Energy Mater., 2024, 14, 2304414 CrossRef CAS.
- Q. Wu, M. T. McDowell and Y. Qi, Effect of the electric double layer (EDL) in multicomponent electrolyte reduction and solid electrolyte interphase (SEI) formation in lithium batteries, J. Am. Chem. Soc., 2023, 145, 2473–2484 CrossRef CAS PubMed.
- T. Ramachandran, R. K. Raji, S. Palanisamy, N. Renuka and K. Karuppasamy, The role of in situ and operando techniques in unraveling local electrochemical supercapacitor phenomena, J. Ind. Eng. Chem., 2025, 145, 144–168 CrossRef CAS.
- F. Capone, J. Sottmann, V. Meunier, L. P. Ramírez, A. Grimaud, A. Iadecola, M. Scardamaglia, J.-P. Rueff and R. Dedryvère, Operando observation of the dynamic SEI formation on a carbonaceous electrode by near-ambient pressure XPS, Energy Environ. Sci., 2024, 17, 1509–1519 RSC.
- B. Freitas, W. G. Nunes, C. G. Real, C. B. Rodella, G. Doubek, L. M. da Silva, E. H. Thaines, L. A. Pocrifka, R. G. Freitas and H. Zanin, Combining in situ electrochemistry, operando XRD & Raman spectroscopy, and density functional theory to investigate the fundamentals of Li 2 CO 3 formation in supercapacitors, J. Mater. Chem. A, 2023, 11, 20636–20650 RSC.
- D. Diddens, W. A. Appiah, Y. Mabrouk, A. Heuer, T. Vegge and A. Bhowmik, Modeling the solid electrolyte interphase: Machine learning as a game changer?, Adv. Mater. Interfaces, 2022, 9, 2101734 CrossRef CAS.
- A. G. Shard and M. A. Baker, Practical guides for x-ray photoelectron spectroscopy: Use of argon ion beams for sputter depth profiling and cleaning, J. Vac. Sci. Technol., A, 2024, 42, 050801 CrossRef CAS.
- S. Yakovlev, J. Zekonyte, C.-H. Solterbeck and M. Es-Souni, Interfacial effects on the electrical properties of multiferroic BiFeO3/Pt/Si thin film heterostructures, Thin Solid Films, 2005, 493, 24–29 CrossRef CAS.
- A. Andersson, D. Abraham, R. Haasch, S. MacLaren, J. Liu and K. Amine, Surface characterization of electrodes from high power lithium-ion batteries, J. Electrochem. Soc., 2002, 149, A1358 CrossRef CAS.
- L.-S. Chang, Y.-C. Lin, C.-Y. Su, H.-C. Wu and J.-P. Pan, Effect of C60 ion sputtering on the compositional depth profiling in XPS for Li (Ni, Co, Mn) O2 electrodes, Appl. Surf. Sci., 2011, 258, 1279–1281 CrossRef CAS.
- K. Ridier, D. Aureau, B. Bérini, Y. Dumont, N. Keller, J. Vigneron, A. Etcheberry and A. Fouchet, Enhanced depth profiling of perovskite oxide: low defect levels induced in SrTiO3 by Argon cluster sputtering, J. Phys. Chem. C, 2016, 120, 21358–21363 CrossRef CAS.
- Z. D. Schultz, J. M. Marr and H. Wang, Tip enhanced Raman scattering: plasmonic enhancements for nanoscale chemical analysis, Nanophotonics, 2014, 3, 91–104 CrossRef CAS.
- T. Schmid, L. Opilik, C. Blum and R. Zenobi, Nanoscale chemical imaging using tip-enhanced Raman spectroscopy: a critical review, Angew. Chem., Int. Ed., 2013, 52, 5940–5954 CrossRef CAS PubMed.
- K. Vilkevičius, I. Ignatjev, A. Selskis, G. Niaura and E. Stankevičius, Tuning SERS performance through the laser-induced morphology changes of gold nanostructures, Appl. Surf. Sci., 2024, 660, 160003 CrossRef.
- R. Qin, Y. Wang, Q. Zhao, K. Yang and F. Pan, EQCM for in-depth study of metal anodes for electrochemical energy storage, Chin. J. Struct. Chem., 2020, 39, 605–614 CAS.
- S. Daboss, T. Philipp, K. Palanisamy, J. Flowers, H. Stein and C. Kranz, Characterization of the solid/electrolyte interphase at hard carbon anodes via scanning (electrochemical) probe microscopy, Electrochim. Acta, 2023, 453, 142345 CrossRef CAS.
- M. Zhou, Y. Liao, L. Li, R. Xiong, G. Shen, Y. Chen, T. Huang, M. Li, H. Zhou and Y. Zhang, Unraveling the heterogeneity of solid electrolyte interphase kinetically affecting lithium electrodeposition on lithium metal anode, J. Energy Chem., 2023, 85, 181–190 CrossRef CAS.
- Q. Zhang, C. Zhou, M. Li, Y. Zhu, X. Wei, S. Shen, Z. Ji, G. Luo, Y. Cheng and X. Yang, others Revealing Structural Insights of Solid Electrolyte Interphase in High-Concentrated Non-Flammable Electrolyte for Li Metal Batteries by Cryo-TEM, Small, 2023, 19, 2300849 CrossRef CAS PubMed.
- S. Izawa, K. Nakano, K. Suzuki, Y. Chen, T. Kikitsu, D. Hashizume, T. Koganezawa, T.-Q. Nguyen and K. Tajima, Crystallization and polymorphism of organic semiconductor in thin film induced by surface segregated monolayers, Sci. Rep., 2018, 8, 481 CrossRef PubMed.
- Y. Xu, K. Dong, Y. Jie, P. Adelhelm, Y. Chen, L. Xu, P. Yu, J. Kim, Z. Kochovski and Z. Yu, others Promoting mechanistic understanding of lithium deposition and solid-electrolyte interphase (SEI) formation using advanced characterization and simulation methods: recent progress, limitations, and future perspectives, Adv. Energy Mater., 2022, 12, 2200398 CrossRef.
- S. Chen, Z. Gong, P. Zhao, Y. Zhang, B. Cheng, J. Hou, J. Song, X. Ding, J. Sun and J. Shi, others Design of nanostructure in solid electrolyte interphase for enhancing the mechanical durability of lithium metal anode by deep-learning approach, Energy Storage Mater., 2024, 65, 103096 CrossRef.
- Y. Fan, X. Wang, G. Bo, X. Xu, K. W. See, B. Johannessen and W. K. Pang, Operando Synchrotron X-Ray Absorption Spectroscopy: A Key Tool for Cathode Material Studies in Next-Generation Batteries, Advanced Science, 2025, 12, 2414480 CrossRef CAS PubMed.
- J. W. Gittins, Y. Chen, S. Arnold, V. Augustyn, A. Balducci, T. Brousse, E. Frackowiak, P. Gómez-Romero, A. Kanwade and L. Köps, others Interlaboratory study assessing the analysis of supercapacitor electrochemistry data, J. Power Sources, 2023, 585, 233637 CrossRef CAS.
- J. P. A. dos Santos, F. C. Rufino, J. I. Y. Ota, R. C. Fernandes, R. Vicentini, C. J. Pagan, L. M. Da Silva and H. Zanin, Best practices for electrochemical characterization of supercapacitors, J. Energy Chem., 2023, 80, 265–283 CrossRef.
- C. Shen, S. Wang, Y. Jin and W.-Q. Han, In situ AFM imaging of solid electrolyte interfaces on HOPG with ethylene carbonate and fluoroethylene carbonate-based electrolytes, ACS Appl. Mater. Interfaces, 2015, 7, 25441–25447 CrossRef CAS PubMed.
- H. Zhang, D. Wang and C. Shen, In-situ EC-AFM and ex-situ XPS characterization to investigate the mechanism of SEI formation in highly concentrated aqueous electrolyte for Li-ion batteries, Appl. Surf. Sci., 2020, 507, 145059 CrossRef CAS.
- Y. Liang, M. Burton, B. Jagger, H. Guo, J. Ihli and M. Pasta, In situ XPS investigation of the SEI formed on LGPS and LAGP with metallic lithium, Chem. Commun., 2024, 60, 12597–12600 RSC.
- R. Guo, D. Wang, L. Zuin and B. M. Gallant, Reactivity and Evolution of Ionic Solid-Electrolyte-Interphases in Battery Electrolytes, ECS Meet. Abstr., 2021, 239, 149 CrossRef.
- G. C. Mohanty, C. C. Gowda, P. Gakhad, S. Das, M. Sanjay, S. Chowdhury, K. Biswas, A. Singh and C. S. Tiwary, Iron-cobalt-nickel-copper-zinc (FeCoNiCuZn) high entropy alloy as positive electrode for high specific capacitance supercapacitor, Electrochim. Acta, 2023, 470, 143272 CrossRef CAS.
- X. Guoguang, W. Min, W. Jian, L. Hongzhen, W. Yang, W. Yang and Z. Yuegang, In-situ Electrochemical Characterization Methodology, Chin. J. Catal., 2019, 40, 187 Search PubMed.
- R. Yuan, H. Jiao, H. Zhu, D. Fang and S. Jiao, In situ characterization techniques and methodologies for high-temperature electrochemistry, Chem, 2023, 9, 2481–2508 CAS.
- Y. Zhou, M. Su, X. Yu, Y. Zhang, J.-G. Wang, X. Ren, R. Cao, W. Xu, D. R. Baer and Y. Du, others Real-time mass spectrometric characterization of the solid–electrolyte interphase of a lithium-ion battery, Nat. Nanotechnol., 2020, 15, 224–230 CrossRef CAS PubMed.
- W. Zhang, V. Koverga, S. Liu, J. Zhou, J. Wang, P. Bai, S. Tan, N. K. Dandu, Z. Wang and F. Chen, others Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries, Nat. Energy, 2024, 9, 386–400 CrossRef CAS.
- C.-z. Yang, Y. Lou, J. Zhang, X. Xie and B. Xia, Materials and Working Mechanisms of Secondary Batteries, Springer, 2023, pp 207–226 Search PubMed.
- L. T. Hess, N. P. Nguyen, A. H. Dee, A. K. Gupta, Z. Kwon and S. Yue, How Surface Functionalization Controls Confined Electrolyte Structure and Dynamics at Graphene Interfaces, J. Phys. Chem. B, 2025, 10864–10872 CrossRef CAS PubMed.
- M. Sk, P. Pradhan, B. Patra and A. Guria, Green biomass derived porous carbon materials for electrical double-layer capacitors (EDLCs), Mater. Today Chem., 2023, 30, 101582 CrossRef CAS.
- K. Kraiwattanawong, A review on the development of a porous carbon-based as modeling materials for electric double layer capacitors, Arabian J. Chem., 2022, 15, 103625 CrossRef CAS.
- Y. Kado, Y. Soneda, H. Hatori and M. Kodama, Advanced carbon electrode for electrochemical capacitors, J. Solid State Electrochem., 2019, 23, 1061–1081 CrossRef CAS.
- Y. Wang, H. Zhao, L. Li and B. An, Minimizing the confinement effect of micropores to optimize the capacitive performance of carbon materials, J. Alloys Compd., 2025, 1010, 177228 CrossRef CAS.
- M. Li, C. Ma, X. Cai, K. Yue, J. Yue, Y. Wang, J. Luo, H. Yuan, J. Nai and S. Zou, others Structural composite solid electrolyte interphases on lithium metal anodes induced by inorganic/organic activators, Mater. Today Energy, 2024, 46, 101734 CrossRef CAS.
- S. Shaheen Shah, S. Oladepo, M. Ali Ehsan, W. Iali, A. Alenaizan, M. Nahid Siddiqui, M. Oyama, A.-R. Al-Betar and M. A. Aziz, Recent progress in polyaniline and its composites for supercapacitors, Chem. Rec., 2024, 24, e202300105 CrossRef CAS PubMed.
- J. Pan, C. Li, Y. Peng, L. Wang, B. Li, G. Zheng and M. Song, Application of transition metal (Ni, Co and Zn) oxides based electrode materials for ion-batteries and supercapacitors, Int. J. Electrochem. Sci., 2023, 18, 100233 CrossRef CAS.
- M. Marr, J. Kuhn, C. Metcalfe, J. Harris and O. Kesler, Electrochemical performance of solid oxide fuel cells having electrolytes made by suspension and solution precursor plasma spraying, J. Power Sources, 2014, 245, 398–405 CrossRef CAS.
- C.-Y. Tseng, Y.-S. Ye, J. Joseph, K.-Y. Kao, J. Rick, S.-L. Huang and B.-J. Hwang, Tuning transport properties by manipulating the phase segregation of tetramethyldisiloxane segments in modified polyimide electrolytes, J. Power Sources, 2011, 196, 3470–3478 CrossRef CAS.
- L. Yu, J. Li, N. Ahmad, X. He, G. Wan, R. Liu, X. Ma, J. Liang, Z. Jiang and G. Zhang, Recent progress on carbon materials for emerging zinc-ion hybrid capacitors, J. Mater. Chem. A, 2024, 12, 9400–9420 RSC.
- Y. Huang, J. Zhou, N. Gao, Z. Yin, H. Zhou, X. Yang and Y. Kuang, Synthesis of 3D reduced graphene oxide/unzipped carbon nanotubes/polyaniline composite for high-performance supercapacitors, Electrochim. Acta, 2018, 269, 649–656 CrossRef CAS.
- D. Bedrov, D. Dong and M. Ebrahiminia, Multiscale Modeling of Formation and Properties of Solid-Electrolyte Interphases in Li-Ion Batteries, ECS Meet. Abstr., 2020, 237, 2754 CrossRef.
- Y. Wang, J. Zhang, X. Zhao, Z. Ren, M. Tang, R. Han, G. Feng, B. Li, D. Zhou and F. Kang, Electric Field-Guided Ion Orchestration for Multi-Chemistry Zinc Metal Batteries, Adv. Mater., 2025, e11163 CrossRef CAS PubMed.
- Y. Chen, Y. Shi, G. Song, B. Yang and H. Pang, Compositing MXene with organic coordination frameworks (MOFs, COFs, and HOFs) for enhanced electrochemical performance, Next Mater., 2025, 6, 100323 CrossRef CAS.
- T. Naren, R. Jiang, Q. Gu, G.-c. Kuang, L. Chen and Q. Zhang, Fluorinated organic compounds as promising materials to protect lithium metal anode: a review, Mater. Today Energy, 2024, 40, 101512 CrossRef CAS.
- P. Kurzweil, J. Schottenbauer and C. Schell, Past, present and future of electrochemical capacitors: Pseudocapacitance, aging mechanisms and service life estimation, J. Energy Storage, 2021, 35, 102311 CrossRef.
- N. Ma, D. Yang, S. Riaz, L. Wang and K. Wang, Aging mechanism and models of supercapacitors: A review, Technologies, 2023, 11, 38 CrossRef.
- S. Mahala, K. Khosravinia and A. Kiani, Unwanted degradation in pseudocapacitors: Challenges and opportunities, J. Energy Storage, 2023, 67, 107558 CrossRef.
- B. Nishchith, S. Ashoka, M. P. Bhat, M. D. Kurkuri, S. Acharya, R. Kumar and Y. Kalegowda, Reversible surface reconstruction of Na3NiCO3PO4: A battery type electrode for pseudocapacitor applications, J. Power Sources, 2022, 520, 230903 CrossRef CAS.
- Y. Huang, C. Yang, B. Deng, C. Wang, Q. Li, C. D. V. Thibault, K. Huang, K. Huo and H. Wu, Nanostructured pseudocapacitors with pH-tunable electrolyte for electrochromic smart windows, Nano Energy, 2019, 66, 104200 CrossRef CAS.
- J. Ahn, Y. Song, Y. J. Kim, D. Nam, T. Kim, K. Kwak, C. H. Kwon, Y. Ko, S. J. Lee and J. Cho, Redox-active ligand-mediated assembly for high-performance transition metal oxide nanoparticle-based pseudocapacitors, Chem. Eng. J., 2023, 455, 140742 CrossRef CAS.
- Z.-H. He, J.-F. Gao and L.-B. Kong, Electrolyte effect on electrochemical behaviors of manganese fluoride material for aqueous asymmetric and symmetric supercapacitors, Rare Met., 2024, 43, 1048–1061 CrossRef CAS.
- S. Dang, Z. Wang, W. Jia, Y. Cao and J. Zhang, Facile synthesis of rod-like nickel-cobalt oxide nanostructure for supercapacitor with excellent cycling stability, Mater. Res. Bull., 2019, 116, 117–125 CrossRef CAS.
- S. Gong, F. Zhao, H. Xu, M. Li, J. Qi, H. Wang, Z. Wang, X. Fan, C. Li and J. Liu, Iodine-functionalized titanium carbide MXene with ultra-stable pseudocapacitor performance, J. Colloid Interface Sci., 2022, 615, 643–649 CrossRef CAS PubMed.
- J. Kumar, R. A. Soomro, B. Fan, J. Tan, N. Sun and B. Xu, Butanedioic acid unlock shelf-stable Ti3C2Tx (MXene) dispersions and their electrochemical performance in supercapacitor, J. Alloys Compd., 2024, 1009, 176749 CrossRef CAS.
- K. Kawai, M. Fujita, R. Iizuka, A. Yamada and M. Okubo, Influence of surface termination groups on electrochemical charge storage of MXene electrodes, 2D Materials, 2023, 10, 014012 CrossRef CAS.
- N. Badawi, M. Bhuyan, M. Luqman, R. S. Alshareef, M. R. Hatshan, A. Al-Warthan and S. F. Adil, MXenes the future of solid-state supercapacitors: Status, challenges, prospects, and applications, Arabian J. Chem., 2024, 17, 105866 CrossRef CAS.
- W. Bai, Z. Yong, S. Wang, X. Wang, C. Li, F. Pan, D. Liang, Y. Cui and Z. Wang, Polyaniline-MXene composite electrode with excellent electrochemical properties for all-solid flexible supercapacitors, J. Energy Storage, 2023, 71, 108053 CrossRef.
- N. Anjum, A. A. Noman, M. M. Rahman, D. Sen, R. A. Lazenby and O. I. Okoli, Electrochemical impedance analysis of Ti3C2Tx MXene for pseudocapacitive charge storage, J. Compos. Sci., 2025, 9, 139 CrossRef CAS.
- R. Singh, M. S. Samuel, M. Ravikumar, S. Ethiraj and M. Kumar, Graphene materials in pollution trace detection and environmental improvement, Environ. Res., 2024, 243, 117830 CrossRef CAS.
- J. Wang, Y. Li, S. Jin, X. Zheng, J. Yuan, D. Kong, H. Hu, X. Feng and D. Chen, Structural design and solid electrolyte interphase modulation of SiOC-based anodes via N-doping for fast-charging lithium-ion batteries, Chem. Eng. J., 2025, 164661 CrossRef CAS.
- X. Liu, J. Sun, Y. Ran, X. Liu, J. Chai, Z. Liu, P. Zheng and Y. Zheng, Surface engineering for enhanced cycling performance of nanosilicon-based anodes in lithium-ion batteries, J. Power Sources, 2025, 654, 237893 CrossRef CAS.
- N. Zheng, C. Yang, Q. Dong, S. Hu, Q. Tang, J. Ou, H. Wang, J. Wang, H. Zhang and J. Ge, others Coupled design of surface chemistry and spatial topology enabling lithiophilic host for stable Li metal batteries, Chem. Eng. J., 2025, 165412 CrossRef CAS.
- R. Jamil, S. Loomba, M. Kar, G. E. Collis, D. S. Silvester and N. Mahmood, Metal anodes meet ionic liquids: An interfacial perspective, Applied Physics Reviews, 2024, 11, 011307 CrossRef CAS.
- S. Azmi, A. Klimek and E. Frackowiak, Why electrochemical capacitor electrolytes should not be ignored?, Electrochim. Acta, 2023, 452, 142347 CrossRef CAS.
- P. Krishnan and V. Biju, Effect of electrolyte concentration on the electrochemical performance of RGO-Na2SO4 supercapacitor, Mater. Today: Proc., 2022, 54, 958–962 CAS.
- Y. Zhang, Elucidating Solvation Structures and Their Impact on SEI Formation at Electrode-Electrolyte Interfaces, ECS Meet. Abstr., 2022, 241, 1146 CrossRef.
- B. Zhu, X. Shi, T. Zheng, J. Xiong, Y.-J. Cheng and Y. Xia, Usefulness of uselessness: Teamwork of wide temperature electrolyte enables LFP/Li cells from-40 °C to 140 °C, Electrochim. Acta, 2022, 425, 140698 CrossRef CAS.
- D. Chai, Y. Zhu, C. Guan, T. Zhang, S. Tang, H. Zhu, X. Li and Y. Fu, Achieving stable interphases toward lithium metal batteries by a dilute and anion-rich electrolyte, Energy Storage Mater., 2023, 62, 102957 CrossRef.
- S. Zhang, Z. Bo, H. Yang, J. Yang, L. Duan, J. Yan and K. Cen, Insights into the effects of solvent properties in graphene based electric double-layer capacitors with organic electrolytes, J. Power Sources, 2016, 334, 162–169 CrossRef CAS.
- S. Sayah, A. Ghosh, M. Baazizi, R. Amine, M. Dahbi, Y. Amine, F. Ghamouss and K. Amine, How do super concentrated electrolytes push the Li-ion batteries and supercapacitors beyond their thermodynamic and electrochemical limits?, Nano Energy, 2022, 98, 107336 CrossRef CAS.
- W. Wang, S. Chen, X. Liao, R. Huang, F. Wang, J. Chen, Y. Wang, F. Wang and H. Wang, Regulating interfacial reaction through electrolyte chemistry enables gradient interphase for low-temperature zinc metal batteries, Nat. Commun., 2023, 14, 5443 CrossRef CAS.
- E. Rahmanian, A. Sajedi-Moghaddam, M. T. Hoveizavi and S. H. Aboutalebi, Electrolyte Hydration Energy as a Universal Descriptor for Ion-Specific Capacitance: Insights from Interpretable Machine Learning, Adv. Powder Mater., 2025, 100361 Search PubMed.
- L. Kong, M. Cheng, H. Huang, J. Pang, S. Liu, Y. Xu and X.-H. Bu, Metal-organic frameworks for advanced aqueous ion batteries and supercapacitors, EnergyChem, 2022, 4, 100090 CrossRef CAS.
- X. Li, Y. Zhang, J. Chen, Y. Wang, Z. Cheng, X. Chen and M. Guo, A cellulose-based interpenetrating network hydrogel electrolyte for flexible solid-state supercapacitors, Cellulose, 2023, 30, 2399–2412 CrossRef CAS.
- Y. Shao, M. F. El-Kady, J. Sun, Y. Li, Q. Zhang, M. Zhu, H. Wang, B. Dunn and R. B. Kaner, Design and mechanisms of asymmetric supercapacitors, Chem. Rev., 2018, 118, 9233–9280 CrossRef CAS PubMed.
- G. Wang, L. Zhang and J. Zhang, A review of electrode materials for electrochemical supercapacitors, Chem. Soc. Rev., 2012, 41, 797–828 RSC.
- N. Zhang, M. Wang, Y. Quan, X. Li, X. Hu, J. Yan, Y. Wang, M. Sun and S. Li, A review of binder-free electrodes for advanced supercapacitors, J. Ind. Eng. Chem., 2025, 141, 1–31 CrossRef CAS.
- J. Lefebvre, F. Galli, C. L. Bianchi, G. S. Patience and D. C. Boffito, Experimental methods in chemical engineering: X-ray photoelectron spectroscopy-XPS, Can. J. Chem. Eng., 2019, 97, 2588–2593 CrossRef CAS.
- L. Matuana, J. Balatinecz, R. Sodhi and C. Park, Surface characterization of esterified cellulosic fibers by XPS and FTIR spectroscopy, Wood Sci. Technol., 2001, 35, 191–201 CrossRef CAS.
- K. Liu, C. Yu, W. Guo, L. Ni, J. Yu, Y. Xie, Z. Wang, Y. Ren and J. Qiu, Recent research advances of self-discharge in supercapacitors: Mechanisms and suppressing strategies, J. Energy Chem., 2021, 58, 94–109 CrossRef CAS.
- I. E. Rauda, V. Augustyn, B. Dunn and S. H. Tolbert, Enhancing pseudocapacitive charge storage in polymer templated mesoporous materials, Acc. Chem. Res., 2013, 46, 1113–1124 CrossRef CAS.
- T. Tevi and A. Takshi, Modeling and simulation study of the self-discharge in supercapacitors in presence of a blocking layer, J. Power Sources, 2015, 273, 857–862 CrossRef CAS.
- W. Li, X. Yang, Z. Chen, T. Lv, X. Wang and J. Qiu, Synthesis and structure regulation of armor-wearing biomass-based porous carbon: Suppression the leakage current and self-discharge of supercapacitors, Carbon, 2022, 196, 136–145 CrossRef CAS.
- J. Yao, M. Shi, W. Li, Q. Han, M. Wu, W. Yang, E. Wang and X. Lu, Fluorinated Ether-Based Electrolyte for Supercapacitors with Increased Working Voltage and Suppressed Self-discharge, ChemElectroChem, 2022, 9, e202200223 CrossRef CAS.
- M. Liu, M. Xia, R. Qi, Q. Ma, M. Zhao, Z. Zhang and X. Lu, Lyotropic liquid crystal as an electrolyte additive for suppressing self-discharge of supercapacitors, ChemElectroChem, 2019, 6, 2531–2535 CrossRef CAS.
- S. Hamedi, T. Ghanbari, E. Moshksar and Z. Hosseini, Time-varying model of self-discharge in a double layer supercapacitor with blocking layer, J. Energy Storage, 2021, 40, 102730 CrossRef.
- R. Tang, Studies on electrochemical degradation reactions in supercapacitors, TANSO, 2021, 2021, 127–128 CrossRef.
- X. Chen, Y. Wu and R. Holze, Ag (e) ing and degradation of supercapacitors: Causes, mechanisms, models and countermeasures, Molecules, 2023, 28, 5028 CrossRef CAS PubMed.
- M. Thomas, S. Veleva, B. Karamanova, A. Brigandì, N. Rey-Raap, A. Arenillas, A. Stoyanova and F. Lufrano, Highly stable and reliable asymmetric solid-state supercapacitors with low self-discharge rates, Sustainable Mater. Technol., 2023, 38, e00770 CrossRef CAS.
- G. Liu, X. Li, C. Li, Q. Zheng, Y. Wang, R. Xiao, F. Huang, H. Tian, C. Wang and X. Chen, others Efficient Fabrication of Disordered Graphene with Improved Ion Accessibility, Ion Conductivity, and Density for High-Performance Compact Capacitive Energy Storage, Advanced Science, 2024, 11, 2405155 CrossRef CAS.
- K. Sarkar and M. A. Talukder, Effects of Edge Atoms and Channel Width on Charge Storage in Nanoporous Carbon Supercapacitors, arXiv, 2026, preprint, arXiv:2601.01601, DOI:10.48550/arXiv.2601.01601.
- Y. Li, X. Fan, M. Zhang, L. Cui and T. Jiao, Enhanced electrochemical performance of the activated carbon electrodes with a facile and in-situ phosphoric acid modification, J. Energy Storage, 2019, 24, 100744 CrossRef.
- I. Hussain, K. Singh, M. N. Naseer, M. Al Mahmud, M. K. Aslam, P. Rosaiah, A. Riza, H. Shahzad, A. Ali and W. U. Arifeen, others MXenes in aqueous electrolytes: integrating computational and experimental insights, J. Ind. Eng. Chem., 2026 Search PubMed , in press.
- K. Shevchuk, K. Matthews, R. Wang and Y. Gogotsi, In Situ Electrochemical Raman Spectroscopy of MXenes in Confined Electrolytes, ECS Meet. Abstr., 2023, 244, 2668 CrossRef.
- L. Zhang, Y. Cai, R. Fang, Y. Wang and S. Xu, Construction of fully coated polypyrrole oxygen-barrier film based on MXene nanosheets for high reliability capacitive deionization, Sep. Purif. Technol., 2024, 337, 126362 CrossRef CAS.
- X. Li, Z. Huang, C. E. Shuck, G. Liang, Y. Gogotsi and C. Zhi, MXene chemistry, electrochemistry and energy storage applications, Nat. Rev. Chem., 2022, 6, 389–404 CrossRef PubMed.
- M. Wu, S. Xie, C. Yang, X. Xue, H. Li, C. Zhang and J. Xu, An experimental study on the failure behaviors of hybrid supercapacitors under overdischarge conditions, Ionics, 2024, 30, 1061–1074 CrossRef CAS.
- L. Jin, X. Guo, C. Shen, N. Qin, J. Zheng, Q. Wu, C. Zhang and J. P. Zheng, A universal matching approach for high power-density and high cycling-stability lithium ion capacitor, J. Power Sources, 2019, 441, 227211 CrossRef CAS.
- O. G. Rojas and W. Sugimoto, Enhanced cycle life and capacity retention of dual electrolyte Li-ion capacitor through optimization of the solid electrolyte, J. Power Sources Adv., 2025, 33, 100179 CrossRef CAS.
- V. Vanpeene, I. Martens, M. Mirolo, A. Benayad, L. Daniel, S. Geniès, Y. Maletin, N. Stryzhakova, S. Zelinskyi and S. Chernukhin, others Quantifying degradation mechanisms in a high-performance parallel hybrid lithium-ion supercapacitor induced by long term cycling at high current rates, J. Electrochem. Soc., 2023, 170, 060517 CrossRef CAS.
- Y. Qian, B. Wu, Y. Li, Z. Pan, J. Tian, N. Lin and Y. Qian, Pressure-dependent self-template pyrolysis modulates the porosity and surface chemical configuration of carbon for potassium ion hybrid capacitors, Chem. Eng. J., 2023, 451, 138579 CrossRef CAS.
- C. H. Lee, J. A. Dura, A. LeBar and S. C. DeCaluwe, Direct, operando observation of the bilayer solid electrolyte interphase structure: Electrolyte reduction on a non-intercalating electrode, J. Power Sources, 2019, 412, 725–735 CrossRef CAS.
- D. Qiao, X. Wei, J. Zhu, G. Zhang, S. Yang, X. Wang, B. Jiang, X. Lai, Y. Zheng and H. Dai, Mechanism of battery expansion failure due to excess solid electrolyte interphase growth in lithium-ion batteries, ETransportation, 2025, 100450 CrossRef.
- Q. Zhang, Y. Wang and B. Wei, Distinct Self-Discharge Processes via Manipulating Electrode Pore Size of Carbon-Based Electrochemical Capacitors, Adv. Energy Mater., 2023, 13, 2301860 CrossRef CAS.
- B. Uralcan and I. B. Uralcan, Origin of enhanced performance in nanoporous electrical double layer capacitors: insights on micropore structure and electrolyte composition from molecular simulations, ACS Appl. Mater. Interfaces, 2022, 14, 16800–16808 CrossRef CAS.
- W. Fang, Z. Wen, L. Chen, Z. Qin, J. Li, Z. Zheng, Z. Weng, G. Wu, N. Zhang and X. Liu, others Constructing inorganic-rich solid electrolyte interphase via abundant anionic solvation sheath in commercial carbonate electrolytes, Nano Energy, 2022, 104, 107881 CrossRef CAS.
- M. C. Santos, G. G. Silva, R. Santamaria, P. F. Ortega and R. L. Lavall, Discussion on operational voltage and efficiencies of ionic-liquid-based electrochemical capacitors, J. Phys. Chem. C, 2019, 123, 8541–8549 CrossRef CAS.
- M. Hu, L. Chen, Y. Jing, Y. Zhu, J. Dai, A. Meng, C. Sun, J. Jia and Z. Li, Intensifying electrochemical activity of Ti3C2Tx MXene via customized interlayer structure and surface chemistry, Molecules, 2023, 28, 5776 CrossRef CAS PubMed.
- X. Jiang, X. Chu, X. Zhang, Y. Xie, T. Yang, J. Huang, W. Li, W. Deng, H. Zhang and W. Yang, Surplus Charge Injection Enables High-Voltage Stable 2d Polyaniline Supercapacitors, Electrochim. Acta, 2023, 445, 142052 CrossRef CAS.
- A. Parejo-Tovar, F. Béguin and P. Ratajczak, Comprehensive potentiodynamic analysis of electrodes performance in hybrid capacitors, Electrochem. Commun., 2023, 147, 107436 CrossRef CAS.
- Q. Abbas, H. Fitzek, V. Pavlenko and B. Gollas, Towards an optimized hybrid electrochemical capacitor in iodide based aqueous redox-electrolyte: Shift of equilibrium potential by electrodes mass-balancing, Electrochim. Acta, 2020, 337, 135785 CrossRef CAS.
- W. I. Choi, M. S. Park, Y. Shim, H. S. Lee and J. Shin, Atomistic Simulation of Solid Electrolyte Interphase (SEI) Using Reactive Force Field, ECS Meet. Abstr., 2016, 230, 572 CrossRef.
- Z. Zhang and W.-Q. Han, From liquid to solid-state lithium metal batteries: fundamental issues and recent developments, Nano-Micro Lett., 2024, 16, 24 CrossRef CAS PubMed.
- Y. Wang, J. Hong and F. Hao, Mechanical and Li diffusion properties of interface systems in the solid electrolyte interphase, JOM, 2024, 76, 1153–1161 CrossRef CAS.
- L. Zeng, J. Peng, J. Zhang, X. Tan, X. Ji, S. Li and G. Feng, Molecular dynamics simulations of electrochemical interfaces, J. Chem. Phys., 2023, 159, 091001 CrossRef CAS PubMed.
- L. Yu, X. Chen, N. Yao, Y.-C. Gao and Q. Zhang, Constant-potential molecular dynamics simulation and its application in rechargeable batteries, J. Mater. Chem. A, 2023, 11, 11078–11088 RSC.
- O. M. Magnussen and A. Groß, Toward an atomic-scale understanding of electrochemical interface structure and dynamics, J. Am. Chem. Soc., 2019, 141, 4777–4790 CrossRef CAS PubMed.
- Y. He, J. Deng, K. Wang, Q. Gou, H. Luo, Z. Luogu, Z. Chen, K. Wen, Y. Zheng and M. Li, B io-inspired ion channels for suppressing interfacial parasitic reactions and enabling low-energy ion desolvation in aqueous supercapacitors, Chem. Sci., 2025, 16757–16769 RSC.
- C. Pean, B. Daffos, B. Rotenberg, P. Levitz, M. Haefele, P.-L. Taberna, P. Simon and M. Salanne, Confinement, desolvation, and electrosorption effects on the diffusion of ions in nanoporous carbon electrodes, J. Am. Chem. Soc., 2015, 137, 12627–12632 CrossRef CAS PubMed.
- X. Tan, M. Chen, J. Zhang, S. Li, H. Zhang, L. Yang, T. Sun, X. Qian and G. Feng, Decoding electrochemical processes of lithium-ion batteries by classical molecular dynamics simulations, Adv. Energy Mater., 2024, 14, 2400564 CrossRef CAS.
- P. M. Singer, D. Asthagiri, W. G. Chapman and G. J. Hirasaki, Molecular dynamics simulations of NMR relaxation and diffusion of bulk hydrocarbons and water, J. Magn. Reson., 2017, 277, 15–24 CrossRef CAS PubMed.
- J. Yang, Z. Bo, H. Yang, H. Qi, J. Kong, J. Yan and K. Cen, Reliability of Constant Charge Method for Molecular Dynamics Simulations on EDLCs in Nanometer and Sub-Nanometer Spaces, ChemElectroChem, 2017, 4, 2486–2493 CrossRef CAS.
- L. Zeng, X. Tan, X. Ji, S. Li, J. Zhang, J. Peng, S. Bi and G. Feng, Constant charge method or constant potential method: Which is better for molecular modeling of electrical double layers?, J. Energy Chem., 2024, 94, 54–60 CrossRef CAS.
- A. Chaudhari, K. Agrawal and A. J. Logsdail, Machine learning generalised DFT+ U projectors in a numerical atom-centred orbital framework, Digital Discovery, 2025, 3701–3727 RSC.
- W.-Q. Li, G. Wu, J. M. Arce-Ramos, Y. H. Lau and M.-F. Ng, Enabling accurate modelling of materials for a solid electrolyte interphase in lithium-ion batteries using effective machine learning interatomic potentials, Mater. Horiz., 2025, 10770–10781 RSC.
- J. Dai, X. Li, L. Zhao and H. Sun, Enthalpies of mixing predicted using molecular dynamics simulations and OPLS force field, Fluid Phase Equilib., 2010, 289, 156–165 CrossRef CAS.
- S. Riniker, Fixed-charge atomistic force fields for molecular dynamics simulations in the condensed phase: An overview, J. Chem. Inf. Model., 2018, 58, 565–578 CrossRef CAS PubMed.
- Y. Han, D. Jiang, J. Zhang, W. Li, Z. Gan and J. Gu, Development, applications and challenges of ReaxFF reactive force field in molecular simulations, Front. Chem. Sci. Eng., 2016, 10, 16–38 CrossRef CAS.
- M. J. Hossain, G. Pawar, B. Liaw, K. L. Gering, E. J. Dufek and A. C. Van Duin, Lithium-electrolyte solvation and reaction in the electrolyte of a lithium ion battery: A ReaxFF reactive force field study, J. Chem. Phys., 2020, 152, 184301 CrossRef CAS PubMed.
- O. T. Unke, S. Chmiela, M. Gastegger, K. T. Schütt, H. E. Sauceda and K.-R. Müller, SpookyNet: Learning force fields with electronic degrees of freedom and nonlocal effects, Nat. Commun., 2021, 12, 7273 CrossRef CAS PubMed.
- M. O. Wilson and D. M. Huang, Anisotropic molecular coarse-graining by force and torque matching with neural networks, J. Chem. Phys., 2023, 159, 024110 CrossRef CAS PubMed.
- Y. Xu, H. Jia, P. Gao, D. E. Galvez-Aranda, S. P. Beltran, X. Cao, P. M. Le, J. Liu, M. H. Engelhard and S. Li, others Direct in situ measurements of electrical properties of solid–electrolyte interphase on lithium metal anodes, Nat. Energy, 2023, 8, 1345–1354 CrossRef CAS.
- Y. Zhang, D. Krishnamurthy and V. Viswanathan, Engineering solid electrolyte interphase composition by assessing decomposition pathways of fluorinated organic solvents in lithium metal batteries, J. Electrochem. Soc., 2020, 167, 070554 CrossRef.
- V. J. Ovejas and A. Cuadras, Impedance characterization of an LCO-NMC/graphite cell: Ohmic conduction, SEI transport and charge-transfer phenomenon, Batteries, 2018, 4, 43 CrossRef.
- K. Vishweswariah, N. G. Ningappa, M. D. Bouguern, A. Kumar MR, M. B. Armand and K. Zaghib, Evaluation and Characterization of SEI Composition in Lithium Metal and Anode-Free Lithium Batteries, Adv. Energy Mater., 2025, 2501883 CrossRef CAS.
- P. Ramadass, B. Haran, R. White and B. N. Popov, Capacity fade of Sony 18650 cells cycled at elevated temperatures: Part I. Cycling performance, J. Power Sources, 2002, 112, 606–613 CrossRef CAS.
- J. J. Gabriel, N. H. Paulson, T. C. Duong, F. Tavazza, C. A. Becker, S. Chaudhuri and M. Stan, Uncertainty quantification in atomistic modeling of metals and its effect on mesoscale and continuum modeling: A review, Jom, 2021, 73, 149–163 CrossRef PubMed.
- F. Schomburg, B. Heidrich, S. Wennemar, R. Drees, T. Roth, M. Kurrat, H. Heimes, A. Jossen, M. Winter and J. Y. Cheong, others Lithium-ion battery cell formation: status and future directions towards a knowledge-based process design, Energy Environ. Sci., 2024, 17, 2686–2733 RSC.
- T. Zhang, H. Sun, C. Yin, Y. H. Jung, S. Min, Y. Zhang, C. Zhang, Q. Chen, K. J. Lee and Q. Chi, Recent progress in polymer dielectric energy storage: from film fabrication and modification to capacitor performance and application, Prog. Mater. Sci., 2023, 140, 101207 CrossRef CAS.
- R. Weber, M. Genovese, A. Louli, S. Hames, C. Martin, I. G. Hill and J. Dahn, Long cycle life and dendrite-free lithium morphology in anode-free lithium pouch cells enabled by a dual-salt liquid electrolyte, Nat. Energy, 2019, 4, 683–689 CrossRef CAS.
- T. He, X. Kang, F. Wang, J. Zhang, T. Zhang and F. Ran, Capacitive contribution matters in facilitating high power battery materials toward fast-charging alkali metal ion batteries, Mater. Sci. Eng., R, 2023, 154, 100737 CrossRef.
- P. J. Weddle, E. W. C. Spotte-Smith, A. Verma, H. D. Patel, K. Fink, B. J. T. de Villers, M. C. Schulze, S. M. Blau, K. A. Smith and K. A. Persson, others Continuum-level modeling of Li-ion battery SEI by upscaling atomistically informed reaction mechanisms, Electrochim. Acta, 2023, 468, 143121 CrossRef CAS.
- S. C. DeCaluwe, Open software for chemical and electrochemical modeling: Opportunities and challenges, Electrochem. Soc. Interface, 2019, 28, 47 CrossRef CAS.
- P. Hou, Y. Tian and X. Meng, Improving Molecular-Dynamics Simulations for Solid–Liquid Interfaces with Machine-Learning Interatomic Potentials, Chem. - Eur. J., 2024, 30, e202401373 CrossRef CAS PubMed.
- Q. Chen, S. Wang and C. Ling, Redefining atomistic simulations of all-solid-state batteries through machine learning interatomic potentials, J. Energy Chem., 2025, 666–687 Search PubMed.
- M. Bin Jassar, C. Michel, S. Abada, T. De Bruin, S. Tant, C. Nieto-Draghi and S. N. Steinmann, A Perspective on the Molecular Modeling of Electrolyte Decomposition Reactions for Solid Electrolyte Interphase Growth in Lithium-Ion Batteries, Adv. Funct. Mater., 2024, 34, 2313188 CrossRef CAS.
- S. Rocken and J. Zavadlav, Enhancing machine learning potentials through transfer learning across chemical elements, J. Chem. Inf. Model., 2025, 65, 7406–7414 CrossRef PubMed.
- V. G. Fletcher, A. P. Bartók and L. B. Pártay, Optimal Autonomous MLIP Dataset Building, arXiv, 2025, preprint, arXiv:2508.08864, DOI:10.48550/arXiv.2508.08864.
- L.-T. Wu, B. J. Hwang and J.-C. Jiang, Combined machine learning and computational protocols to predict electrolyte behavior and SEI formation in Li-metal batteries, Chem. Eng. J., 2025, 163801 CrossRef CAS.
- H. Wang, L. Zhang and J. Han, others DeePMD-kit: A deep learning package for many-body potential energy representation and molecular dynamics, Comput. Phys. Commun., 2018, 228, 178–184 CrossRef CAS.
- S. Gramatte, V. Turlo and O. Politano, Do we really need machine learning interatomic potentials for modeling amorphous metal oxides? Case study on amorphous alumina by recycling an existing ab initio database, Modell. Simul. Mater. Sci. Eng., 2024, 32, 045010 CrossRef.
- T. Maxson, A. Soyemi, X. Zhang, B. W. Chen and T. Szilvási, MS25: materials science-focused benchmark data set for machine learning interatomic potentials, J. Chem. Inf. Model., 2025, 65, 8097–8112 CrossRef CAS PubMed.
- V. Bihani, S. Mannan, U. Pratiush, T. Du, Z. Chen, S. Miret, M. Micoulaut, M. M. Smedskjaer, S. Ranu and N. A. Krishnan, EGraFFBench: evaluation of equivariant graph neural network force fields for atomistic simulations, Digital Discovery, 2024, 3, 759–768 RSC.
- A. Siddiqui and N. D. Hine, Machine-learned interatomic potentials for transition metal dichalcogenide Mo1- x W x S2- 2 y Se2 y alloys, npj Comput. Mater., 2024, 10, 169 CrossRef CAS.
- J. Chen and T. J. Martínez, QTPIE: Charge transfer with polarization current equalization. A fluctuating charge model with correct asymptotics, Chem. Phys. Lett., 2007, 438, 315–320 CrossRef CAS.
- V. Zaverkin, D. Holzmüller, H. Christiansen, F. Errica, F. Alesiani, M. Takamoto, M. Niepert and J. Kästner, Uncertainty-biased molecular dynamics for learning uniformly accurate interatomic potentials, npj Comput. Mater., 2024, 10, 83 CrossRef.
- J. Wang, D. Yu, X. Sun, H. Wang and J. Li, Anodes for low-temperature rechargeable batteries, Escience, 2024, 4, 100252 CrossRef.
- Q. Sun, Y. Xiang, Y. Liu, L. Xu, T. Leng, Y. Ye, A. Fortunelli, W. A. Goddard III and T. Cheng, Machine learning predicts the x-ray photoelectron spectroscopy of the solid electrolyte interface of lithium metal battery, Mater. Today Phys., 2022, 13, 8047–8054 CAS.
- B. Tyler, Interpretation of TOF-SIMS images: multivariate and univariate approaches to image de-noising, image segmentation and compound identification, Appl. Surf. Sci., 2003, 203, 825–831 CrossRef.
- S. Chatterjee, B. Singh, A. Diwan, Z. R. Lee, M. H. Engelhard, J. Terry, H. D. Tolley, N. B. Gallagher and M. R. Linford, A perspective on two chemometrics tools: PCA and MCR, and introduction of a new one: Pattern recognition entropy (PRE), as applied to XPS and ToF-SIMS depth profiles of organic and inorganic materials, Appl. Surf. Sci., 2018, 433, 994–1017 CrossRef CAS.
- M. Bertz, M. Yanagisawa and T. Homma, Deep Learning Combined with Surface-Enhanced Raman Spectroscopy for Chemical Sensing and Recognition, ECS Meet. Abstr., 2021, 239, 1311 CrossRef.
- X. Xu, X. Han, L. Lu, F. Wang, M. Yang, X. Liu, Y. Wu, S. Tang, Y. Hou and J. Hou, others Challenges and opportunities toward long-life lithium-ion batteries, J. Power Sources, 2024, 603, 234445 CrossRef CAS.
- B. Zhu, K. Kretschmer, N. Schlüter and D. Schröder, Combined Machine Learning and Electrochemical Impedance Spectroscopy to Diagnose and Predict the State-of-Health of Commercial SMD Solid-State Batteries, ECS Meet. Abstr., 2023, 243, 2469 CrossRef.
- Y. Zheng, W. Zhang, F. Liu, Q. Liang, W. Li, X. Zhou, N. Yue and W. Zheng, Additive-rejuvenated anions (De) intercalation into graphite cathode enables optimum dual-ion battery, Energy Storage Mater., 2024, 67, 103326 CrossRef.
- I. A. Soomro, M. N. Lakhan, A. Hanan, H. Almujibah, A. Hussain, A. H. Pato, M. Ahmed, I. A. Chandio, S. A. Memon and M. Umer, others 2D MXenes as electrode materials for metal-sulfur batteries: A review, Mater. Today Phys., 2024, 45, 101453 CrossRef CAS.
- J. Wang, M. Wang, F. Chen, Y. Li, L. Zhang, Y. Zhao and C. Chen, In-situ construction of lithiophilic interphase in vertical micro-channels of 3D copper current collector for high performance lithium-metal batteries, Energy Storage Mater., 2021, 34, 22–27 CrossRef.
- J. Xiao, Q. Dai, X. Shen, X. Xie, J. Dai, J. Lam and K.-W. Kwok, Semi-supervised domain adaptation on graphs with contrastive learning and minimax entropy, Neurocomputing, 2024, 580, 127469 CrossRef.
- M.-F. Ng, Y. Sun and Z. W. Seh, Machine learning-inspired battery material innovation, Energy Adv., 2023, 2, 449–464 RSC.
- A. Verma, P. Weddle, A. M. Colclasure, J. Atkins and S. C. DeCaluwe, Multiscale Modeling of Solid Electrolyte Interphase Growth in Silicon Anodes, ECS Meet. Abstr., 2021, 240, 382 CrossRef.
- M. Arvani, J. Keskinen, D. Lupo and M. Honkanen, Current collectors for low resistance aqueous flexible printed supercapacitors, J. Energy Storage, 2020, 29, 101384 CrossRef.
- S. C. Kim, S. T. Oyakhire, C. Athanitis, J. Wang, Z. Zhang, W. Zhang, D. T. Boyle, M. S. Kim, Z. Yu, X. Gao, T. Sogade, E. Wu, J. Qin, Z. Bao, S. F. Bent and Y. Cui, Data-driven electrolyte design for lithium metal anodes, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2214357120 CrossRef CAS PubMed.
- S. Manna, S. S. Manna and B. Pathak, Integrated supervised and unsupervised machine learning approach to map the electrochemical windows over 4500 solvents for battery applications, ACS Appl. Mater. Interfaces, 2024, 16, 42138–42152 CrossRef CAS PubMed.
- B. Reddy, P. Narayana, A. Maurya, U. M. R. Paturi, J. Sung, H.-J. Ahn, K. Cho and N. Reddy, Modeling capacitance of carbon-based supercapacitors by artificial neural networks, J. Energy Storage, 2023, 72, 108537 CrossRef.
- Y. Chai, W. Jia, Z. Hu, S. Jin, H. Jin, H. Ju, X. Yan, H. Ji and L.-J. Wan, Monitoring the mechanical properties of the solid electrolyte interphase (SEI) using electrochemical quartz crystal microbalance with dissipation, Chin. Chem. Lett., 2021, 32, 1139–1143 CrossRef CAS.
- L. Liu, C. Liu, M.-Y. Wang, B. Li, K. Wang, X.-Q. Fan, L.-Y. Wang, H.-Q. Wang, S.-L. Hu and X.-G. Diao, Anti-self-discharge ultrathin all-inorganic electrochromic asymmetric supercapacitors enabling intelligent and effective energy storage: L. Liu et al, Rare Met., 2023, 42, 2957–2971 CrossRef CAS.
- W. Guo, Y. Li, Z. Sun, S. B. Vilsen and D. I. Stroe, A digital twin to quantitatively understand aging mechanisms coupled effects of NMC battery using dynamic aging profiles, Energy Storage Mater., 2023, 63, 102965 CrossRef.
- H. You, X. Wang, J. Zhu, B. Jiang, G. Han, X. Wei and H. Dai, Investigation of lithium-ion battery nonlinear degradation by experiments and model-based simulation, Energy Storage Mater., 2024, 65, 103083 CrossRef.
- X. Wang, Y. Jin, S. Schmitt and M. Olhofer, Recent advances in Bayesian optimization, ACM Computing Surveys, 2023, vol. 55, pp. 1–36 Search PubMed.
- H. Li, J. Hao and S.-Z. Qiao, AI-Driven Electrolyte Additive Selection to Boost Aqueous Zn-Ion Batteries Stability, Adv. Mater., 2024, 36, 2411991 CrossRef CAS PubMed.
- P. Ma, R. Kumar, K.-H. Wang and C. V. Amanchukwu, Active learning accelerates electrolyte solvent screening for anode-free lithium metal batteries, Nat. Commun., 2025, 16, 8396 CrossRef CAS PubMed.
- P. Yan, M. Fischer, H. Martin, C. Wölke, A. N. Krishnamoorthy, I. Cekic-Laskovic, D. Diddens, M. Winter and A. Heuer, Non-aqueous battery electrolytes: high-throughput experimentation and machine learning-aided optimization of ionic conductivity, J. Mater. Chem. A, 2024, 12, 19123–19136 RSC.
- Q. A. Sial, U. Safder, S. Iqbal and R. B. Ali, Advancement in supercapacitors for IoT applications by using machine learning: current trends and future technology, Sustainability, 2024, 16, 1516 CrossRef CAS.
- D. Rajagopal, A. Koeppe, M. Esmaeilpour, M. Selzer, W. Wenzel, H. Stein and B. Nestler, Data-Driven Virtual Material Analysis and Synthesis for Solid Electrolyte Interphases, Adv. Energy Mater., 2023, 13, 2301985 CrossRef CAS.
- M. G. Nam, H. J. Song, J. K. Koo, G. H. Choi, Y. S. Kim, H. J. Kim, C.-S. Shin, Y. Kim, J. H. Nah and Y.-J. Kim, others Standardized cycle life assessment of batteries using extremely lean electrolytic testing conditions, Commun. Mater., 2024, 5, 29 CrossRef CAS.
- X. Zhang, Y. Wang, P. Li and Y. Shang, Physics-based modeling and simulation of the evolution of solid electrolyte interphase film in lithium-ion batteries, J. Energy Storage, 2026, 141, 119329 CrossRef CAS.
- P. E. Lokhande, U. S. Chavan and A. Pandey, Materials and fabrication methods for electrochemical supercapacitors: overview, Electrochem. Energy Rev., 2020, 3, 155–186 CrossRef CAS.
- T. L. Kulova and A. M. Skundin, Electrode/electrolyte interphases of sodium-ion batteries, Energies, 2022, 15, 8615 CrossRef CAS.
- D. Peyrow Hedayati, G. Singh, M. Kucher, T. D. Keene and R. Böhm, Physicochemical Modeling of Electrochemical Impedance in Solid-State Supercapacitors, Materials, 2023, 16, 1232 CrossRef CAS PubMed.
- F. Single, B. Horstmann and A. Latz, Theory of impedance spectroscopy for lithium batteries, J. Phys. Chem. C, 2019, 123, 27327–27343 CrossRef CAS.
- L. M. Housel, A. Abraham, G. D. Renderos, K. J. Takeuchi, E. S. Takeuchi and A. C. Marschilok, Surface electrolyte interphase control on magnetite, Fe3O4, electrodes: impact on electrochemistry, MRS Adv., 2018, 3, 581–586 CrossRef CAS.
- Y. Liu, K. Dong, T. Lv, Z. Chen, S. Cao, Q. Lu, F. Zheng and T. Chen, Revealing the mechanism of bilayer heterogeneous polyelectrolytes to suppress the self-discharge of symmetric supercapacitors, Small Struct., 2023, 4, 2300046 CrossRef CAS.
- K. Crompton, M. Hladky, H. H. Park, S. Prokes, C. Love and B. Landi, Lithium-ion cycling performance of multi-walled carbon nanotube electrodes and current collectors coated with nanometer scale Al2O3 by atomic layer deposition, Electrochim. Acta, 2018, 292, 628–638 CrossRef CAS.
- D. Gandla, G. Song, C. Wu, Y. Ein-Eli and D. Q. Tan, Cover Feature: Atomic Layer Deposition (ALD) of Alumina over Activated Carbon Electrodes Enabling a Stable 4 V Supercapacitor Operation (4/2021), ChemistryOpen, 2021, 10, 388 CrossRef CAS.
- C. Zheng, J. Wu, L. Zhang and H. Wang, Vital roles of fluoroethylene carbonate in electrochemical energy storage devices: a review, J. Mater. Chem. C, 2023, 11, 344–363 RSC.
- J. Lee, J.-Y. Jeong, J. Ha, Y.-T. Kim and J. Choi, Understanding solid electrolyte interface formation on graphite and silicon anodes in lithium-ion batteries: Exploring the role of fluoroethylene carbonate, Electrochem. Commun., 2024, 163, 107708 CrossRef CAS.
- B. Babu, Self-discharge in rechargeable electrochemical energy storage devices, Energy Storage Mater., 2024, 67, 103261 CrossRef.
- P. Przygocki, P. Ratajczak and F. Béguin, Quantification of the Charge Consuming Phenomena under High-Voltage Hold of Carbon/Carbon Supercapacitors by Coupling Operando and Post-Mortem Analyses, Angew. Chem., 2019, 131, 18137–18145 CrossRef.
- N. U. H. L. Ali, P. Pazhamalai, A. Sathyaseelan, R. Swaminathan and S.-J. Kim, Sodium polyacrylate hydrogel electrolyte: Flexible rechargeable supercapacitor using thermally reduced graphene oxide nanosheets, J. Power Sources, 2025, 625, 235648 CrossRef CAS.
- Y. Cao, H. Zhang, Y. Zhang, Z. Yang, D. Liu, H. Fu, Y. Zhang, M. Liu and Q. Li, Epitaxial nanofiber separator enabling folding-resistant coaxial fiber-supercapacitor module, Energy Storage Mater., 2022, 49, 102–110 CrossRef.
- J. Wagner-Henke, D. Kuai, M. Gerasimov, F. Röder, P. B. Balbuena and U. Krewer, Knowledge-driven design of solid-electrolyte interphases on lithium metal via multiscale modelling, Nat. Commun., 2023, 14, 6823 CrossRef CAS PubMed.
- A. Tahmasbi, T. Kadyk and M. Eikerling, Statistical physics-based model of solid electrolyte interphase growth in lithium ion batteries, J. Electrochem. Soc., 2017, 164, A1307 CrossRef CAS.
- E. Locorotondc, L. Pugi, L. Berzi, M. Pierini, S. Scavuzzc, A. Ferraris, A. G. Airale and M. Carello, Modeling and simulation of constant phase element for battery electrochemical impedance spectroscopy, 2019 IEEE 5th International Forum on Research and Technology for Society and Industry (RTSI), 2019, pp 225–230 Search PubMed.
- J. Moškon and M. Gaberšček, Transmission line models for evaluation of impedance response of insertion battery electrodes and cells, J. Power Sources Adv., 2021, 7, 100047 CrossRef.
- U. Tröltzsch and O. Kanoun, Generalization of transmission line models for deriving the impedance of diffusion and porous media, Electrochim. Acta, 2012, 75, 347–356 CrossRef.
- M. Schalenbach, V. Selmert, A. Kretzschmar, L. Raijmakers, Y. E. Durmus, H. Tempel and R.-A. Eichel, How microstructures, oxide layers, and charge transfer reactions influence double layer capacitances. Part 1: impedance spectroscopy and cyclic voltammetry to estimate electrochemically active surface areas (ECSAs), Phys. Chem. Chem. Phys., 2024, 26, 14288–14304 RSC.
- M. Pech-Canul and P. Castro, Corrosion measurements of steel reinforcement in concrete exposed to a tropical marine atmosphere, Cem. Concr. Res., 2002, 32, 491–498 CrossRef CAS.
- M. Itagaki, S. Suzuki, I. Shitanda, K. Watanabe and H. Nakazawa, Impedance analysis on electric double layer capacitor with transmission line model, J. Power Sources, 2007, 164, 415–424 CrossRef CAS.
- J. Lin, W. Hu, J. Yang, L. Pan, X. Xia, Y. Wei, Z. Gong and Y. Yang, Revisiting High-Frequency Impedance in Li-Ion Batteries: Decoupling Solid Electrolyte Interphase Resistance from Pore Impedance, Mater. Today Phys., 2025, 16, 7490–7497 CAS.
- S. Perez Beltran and P. B. Balbuena, Unraveling the Dynamics of Solid-Electrolyte Interphase (SEI) Formation on Lithium Metal: Insights from Multiscale Modeling, ECS Meet. Abstr., 2024, 245, 157 CrossRef.
- D. D. Macdonald, Why electrochemical impedance spectroscopy is the ultimate tool in mechanistic analysis, ECS Trans., 2009, 19(20), 55–79 CrossRef.
- E.-C. Shin, P.-A. Ahn, H.-H. Seo and J.-S. Lee, others Application of a general gas electrode model to Ni-YSZ symmetric cells: Humidity and current collector effects, J. Korean Ceram. Soc., 2016, 53, 511–520 CrossRef CAS.
- S. I. Abdul Halim, C. H. Chan and J. Apotheker, Basics of teaching electrochemical impedance spectroscopy of electrolytes for ion-rechargeable batteries–part 1: a good practice on estimation of bulk resistance of solid polymer electrolytes, Chem. Teach. Int., 2021, 3, 105–115 CrossRef.
- X. Wang, M. Salari, D.-e. Jiang, J. Chapman Varela, B. Anasori, D. J. Wesolowski, S. Dai, M. W. Grinstaff and Y. Gogotsi, Electrode material–ionic liquid coupling for electrochemical energy storage, Nat. Rev. Mater., 2020, 5, 787–808 CrossRef CAS.
- S. Xiang, L. Zhu, L. Fu, M. Wang, X. Zhang, Y. Tang, D. Sun and H. Wang, Cryogenic and in situ characterization techniques for electrode interphase analysis, eScience, 2025, 5, 100291 CrossRef.
- D. E. Galvez-Aranda, C. Vicharra, L. Selis, F. Franco-Gallo, T. M. Gamero and J. M. Seminario, Molecular Dynamics Study of Interphase Solid Electrolyte/Electrode in Li-Ion Batteries, ECS Meet. Abstr., 2018, 450 CrossRef.
- R. Li, L. Bao, L. Chen, C. Zha, J. Dong, N. Qi, R. Tang, Y. Lu, M. Wang and R. Huang, others Accelerated aging of lithium-ion batteries: bridging battery aging analysis and operational lifetime prediction, Sci. Bull., 2023, 68, 3055–3079 CrossRef CAS PubMed.
- Z. Sheng, X. Lin, H. Wei, Y. Zhang, Z. Tian, C. Wang, D. Xu and Y. Wang, Green synthesis of nitrogen-doped hierarchical porous carbon nanosheets derived from polyvinyl chloride towards high-performance supercapacitor, J. Power Sources, 2021, 515, 230629 CrossRef CAS.
- S. Liu, Z. Liu, X. Shen, X. Wang, S.-C. Liao, R. Yu, Z. Wang, Z. Hu, C.-T. Chen and X. Yu, others Li–Ti cation mixing enhanced structural and performance stability of Li-rich layered oxide, Adv. Energy Mater., 2019, 9, 1901530 CrossRef.
- J. Jin, X. Geng, Q. Chen and T.-L. Ren, A better Zn-ion storage device: recent progress for Zn-ion hybrid supercapacitors, Nano-Micro Lett., 2022, 14, 64 CrossRef CAS PubMed.
- J. Liang, A. Rawal, M. Yu, K. Xiao, H. Liu, Y. Jiang, A. Lennon and D.-W. Wang, Low-potential solid-solid interfacial charging on layered polyaniline anode for high voltage pseudocapacitive intercalation Li-ion supercapacitors, Nano Energy, 2023, 105, 108010 CrossRef CAS.
- Z. Jia, S. Hou, J. Peng, X. Wu, W. Tang, W. Sun, S. Lv, X. Yuan, L. Liu and Y. Wu, Recent advances in aqueous and non-aqueous alkali metal hybrid ion capacitors, J. Mater. Chem. A, 2024, 12, 17835–17895 RSC.
- D. Majumdar, M. Mandal and S. K. Bhattacharya, Journey from supercapacitors to supercapatteries: recent advancements in electrochemical energy storage systems, Emergent Mater., 2020, 3, 347–367 CrossRef CAS.
- L. Xia, B. Tang, J. Wei and Z. Zhou, Recent advances in alkali metal-ion hybrid supercapacitors, Rare Met., 2021, 4, 1108–1121 CAS.
- B. A. Ali and N. K. Allam, A first-principles roadmap and limits to design efficient supercapacitor electrode materials, Phys. Chem. Chem. Phys., 2019, 21, 17494–17511 RSC.
- L. Jia, J. Zhu, X. Zhang, B. Guo, Y. Du and X. Zhuang, Li–solid electrolyte interfaces/interphases in all-solid-state Li batteries, Electrochem. Energy Rev., 2024, 7, 12 CrossRef CAS.
- G. Capson-Tojo, R. Moscoviz, S. Astals, Á. Robles and J.-P. Steyer, Unraveling the literature chaos around free ammonia inhibition in anaerobic digestion, Renewable Sustainable Energy Rev., 2020, 117, 109487 CrossRef CAS.
- X. Zhang, Z. Guo, X. Li, Q. Liu, H. Hu, F. Li, Q. Huang, L. Zhang, Y. Tang and J. Huang, Cryo-ultramicrotomy enables TEM characterization of global lithium/polymer interfaces, Energy Environ. Sci., 2024, 17, 1436–1447 RSC.
- J. Tan, J. Matz, P. Dong, J. Shen and M. Ye, A growing appreciation for the role of LiF in the solid electrolyte interphase, Adv. Energy Mater., 2021, 11, 2100046 CrossRef CAS.
- A. M. Miksch, T. Morawietz, J. Kästner, A. Urban and N. Artrith, Strategies for the construction of machine-learning potentials for accurate and efficient atomic-scale simulations, Nucl. Eng. Technol., 2021, 2, 031001 Search PubMed.
- F. Antonello, J. Buongiorno and E. Zio, Physics informed neural networks for surrogate modeling of accidental scenarios in nuclear power plants, Nuclear Engineering and Technology, 2023, 55, 3409–3416 CrossRef CAS.
- R. Batra, L. Song and R. Ramprasad, Emerging materials intelligence ecosystems propelled by machine learning, Nat. Rev. Mater., 2021, 6, 655–678 CrossRef.
- N. Takenaka, A. Bouibes, Y. Yamada, M. Nagaoka and A. Yamada, Frontiers in theoretical analysis of solid electrolyte interphase formation mechanism, Adv. Mater., 2021, 33, 2100574 CrossRef CAS PubMed.
- X. Zhou, R. Gao, Z. Hu, W. Zhou, Y. Kwok and G. Chen, Unveiling Electron Transport Properties of Lithium Fluoride/Lithium Oxide Interfaces in Solid Electrolyte Interphase on Lithium Metal Anodes, J. Power Sources, 2025, 237946 CrossRef CAS.
- R. Lin, Y. Jin, Y. Li, M. Fu, Y. Gong, L. Lei, Y. Zhang, J. Xu and Y. Xiong, Decoupling Interfacial Stability and Ion Transport in Solid Polymer Electrolyte by Tailored Ligand Chemistry for Lithium Metal Battery, Adv. Funct. Mater., 2025, 35, 2421880 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |