DOI:
10.1039/D5TA10343F
(Paper)
J. Mater. Chem. A, 2026, Advance Article
Carbon tetrafluoride – sulphur hexafluoride adsorption and separation with zirconium metal–organic frameworks decorated with fluorinated thiazolium salts
Received
18th December 2025
, Accepted 24th February 2026
First published on 24th February 2026
Abstract
The 8-connected fully-carbocyclic zirconium MOF [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2] [Zr_TCPB, H4TCPB = 1,2,4,5-tetrakis(4-carboxyphenyl)benzene] has been post-synthetically decorated with the two F-containing thiazolium salts 3-(4-carboxy-2-fluorobenzyl)thiazol-3-ium bromide [(HPhTzF)Br] and 3-(4-carboxy-2-(trifluoromethyl)benzyl)thiazol-3-ium bromide [(HPhTzCF3)Br] via Solvent-Assisted Ligand Incorporation (SALI). After salt anchorage to the metal nodes through their carboxylic function and concomitant water elimination (condensation), two mixed-linker MOFs of general formulae [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br (Zr_TCPB_F) and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br (Zr_TCPB_CF3) have been obtained. They have been fully characterized in the solid-state and exploited as functional materials for SF6 and CF4 adsorption. The introduction of an F-containing moiety in the pristine MOF improves the fluorinated gas thermodynamic affinity, as witnessed by the increased SF6 and CF4 adsorption enthalpies (Qst) of Zr_TCPB_F and Zr_TCPB_CF3 in comparison to those of bare Zr_TCPB. Intriguingly, the replacement of a simple fluoro (–F) substituent with a trifluoromethyl (–CF3) group on the thiazolium salt switches the MOF preferential adsorption from SF6 to CF4. This behavior is unprecedented to our knowledge, opening new horizons in the discrimination of fluorinated polluting gases in the field of air treatment and cleaning. Both MOFs are selective toward SF6 in SF6/CF4 equimolar binary mixtures, because of the higher SF6 polarizability (due to the much larger and more diffuse central sulfur atom) that translates into stronger dispersion (van der Waals) interactions with the MOFs. The best performance has been found for Zr_TCPB_CF3 (with SSF6/CF4 = 9). Therefore, both materials can be exploited in fluorinated gas separation as further demonstrated by periodic DFT calculations carried out on model systems.
1 Introduction
Air pollution, driven by a variety of human activities, continues to pose a major threat to both environmental health and climate stability. Among the lesser-known but highly impactful pollutants are fluorinated greenhouse gases, that contribute approximately 2% of the global greenhouse gas emissions.1,2 The rapidly increasing demand for refrigeration and cooling services, particularly in developing countries, threatens to increase fluorinated gas emissions considerably over the next few decades. Two important members of this family are carbon tetrafluoride (CF4) and sulfur hexafluoride (SF6). These gases [although present in much lower environmental concentrations in the order of parts per trillion (ppt) compared to carbon dioxide (CO2)] have an extremely high global warming potential (GWP) because, for a given amount of mass, they trap substantially more heat than CO2 and can persist in the atmosphere for hundreds to thousands of years, contributing to the greenhouse effect.3 CF4 is primarily used in the semiconductor industry, where it is employed as a plasma etching gas during the production of microchips. It is also used in the production of aluminum and as a refrigerant in certain niche applications. Its chemical inertness and stability make it an ideal compound for these high-tech processes, but this also means that once released, CF4 persists in the atmosphere for centuries.4–6 SF6 is widely used as an insulating gas in high-voltage electrical equipment, such as circuit breakers, switchgear, and transformers, due to its excellent dielectric properties. It is also used in particle accelerators and medical devices like MRI machines. The chemical stability and insulating qualities of SF6 make it invaluable in these applications, ensuring reliable and efficient operation of equipment that powers critical infrastructure. However, the use of SF6 in these industries has raised concerns due to its extremely high GWP, which is over 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times greater than CO2 on a per molecule basis.7–9 Small atmospheric concentrations of these two gases can have large effects on global temperatures. Thus, controlling and mitigating their release into the environment has become a critical objective for reducing the overall carbon footprint and achieving international climate goals, including those set out in the Paris Agreement.10 Metal–organic frameworks (MOFs), with their unique structural characteristics, have emerged as promising candidates for addressing this issue due to their exceptional surface area, tunable porosity, and chemical versatility.11–14 MOFs are typically composed of metal clusters linked by organic ligands, allowing for the precise design of pore structures that can selectively adsorb and separate specific gases. This adaptability makes MOFs a compelling solution for the selective capture of gases such as fluorinated compounds, which require highly specialized materials for effective separation. Given the strong C–F and S–F bonds in CF4 and SF6, their adsorption requires MOFs with tailored pore environments and strong host-guest interactions.15–18 MOFs containing open metal sites, fluorophilic functional groups, or optimized pore dimensions have demonstrated enhanced uptake of these gases. Additionally, post-synthetic modifications and functionalized ligands can further improve adsorption performance.19–25 In recent years, the incorporation of fluorine into the structure of MOFs has opened new avenues for enhancing their performance in gas separation applications. Fluorine, with its strong electronegativity, can contribute to the stabilization of metal centers and introduce additional functionalities to the material. Fluorinated MOFs in particular offer advantages, such as increased chemical stability and enhanced interactions with fluorinated gases. These unique properties enable these materials to act as highly efficient adsorbents, capable of selectively capturing and separating fluorinated compounds from complex gas mixtures.26–28 In the semiconductor industry, CF4 and SF6 mixtures are used in plasma etching processes for the fabrication of integrated circuits. These mixtures allow precise control of the etching, thanks to their chemical properties and the ability to form reactive plasmas. After use, exhaust gases may contain reactive or incompletely dissociated residues. Separating CF4 from SF6 allows the recovery and reuse of gases, the reduction of the load on scrubber systems and the improvement of process efficiency. SF6/CF4 mixtures are also exploited in gas-insulated switchgears in low-temperature environments, such as arctic regions. The addition of CF4 to SF6 prevents condensation of SF6 at temperatures below 233 K, maintaining insulating properties and ensuring reliable operation. During maintenance or emptying of the system, it is necessary to separate the two components to optimize recycling, to avoid contamination and to restore the original mixture in a controlled manner. Given the growing concerns over the environmental impact of fluorinated gases, the development of fluorine-containing MOFs for their selective adsorption/separation has the potential to play a key role in mitigating their release into the atmosphere. The introduction of fluorinated linkers in a MOF can be accomplished either via direct synthesis under solvothermal conditions or via post-synthetic modification through the reaction known as Solvent-Assisted Ligand Incorporation (SALI).29–32 Recent work of our group has exploited the latter approach for the construction of mixed-linker MOFs (MIXMOFs) decorated with thiazole-based organic groups for assorted applications, spanning from CO2 capture to luminescence sensing of wastewater contaminants.33–36 Following this research interest and with the fluorinated gas adsorption/separation application in mind, we have prepared two new F-containing monocarboxylic thiazolium salts (HPhTzF)Br and (HPhTzCF3)Br (Scheme 1). Subsequently, we have decorated the 8-connected zirconium MOF [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2] (H4TCPB = 1,2,4,5-tetrakis(4-carboxyphenyl)benzene, Scheme 1, Zr_TCPB] with these salts using SALI. The as-obtained MIXMOFs after water elimination with minimal formulae [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br (Zr_TCPB_F) and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br (Zr_TCPB_CF3) have been fully characterized in the solid-state and exploited as functional materials for SF6 and CF4 adsorption.
000 times greater than CO2 on a per molecule basis.7–9 Small atmospheric concentrations of these two gases can have large effects on global temperatures. Thus, controlling and mitigating their release into the environment has become a critical objective for reducing the overall carbon footprint and achieving international climate goals, including those set out in the Paris Agreement.10 Metal–organic frameworks (MOFs), with their unique structural characteristics, have emerged as promising candidates for addressing this issue due to their exceptional surface area, tunable porosity, and chemical versatility.11–14 MOFs are typically composed of metal clusters linked by organic ligands, allowing for the precise design of pore structures that can selectively adsorb and separate specific gases. This adaptability makes MOFs a compelling solution for the selective capture of gases such as fluorinated compounds, which require highly specialized materials for effective separation. Given the strong C–F and S–F bonds in CF4 and SF6, their adsorption requires MOFs with tailored pore environments and strong host-guest interactions.15–18 MOFs containing open metal sites, fluorophilic functional groups, or optimized pore dimensions have demonstrated enhanced uptake of these gases. Additionally, post-synthetic modifications and functionalized ligands can further improve adsorption performance.19–25 In recent years, the incorporation of fluorine into the structure of MOFs has opened new avenues for enhancing their performance in gas separation applications. Fluorine, with its strong electronegativity, can contribute to the stabilization of metal centers and introduce additional functionalities to the material. Fluorinated MOFs in particular offer advantages, such as increased chemical stability and enhanced interactions with fluorinated gases. These unique properties enable these materials to act as highly efficient adsorbents, capable of selectively capturing and separating fluorinated compounds from complex gas mixtures.26–28 In the semiconductor industry, CF4 and SF6 mixtures are used in plasma etching processes for the fabrication of integrated circuits. These mixtures allow precise control of the etching, thanks to their chemical properties and the ability to form reactive plasmas. After use, exhaust gases may contain reactive or incompletely dissociated residues. Separating CF4 from SF6 allows the recovery and reuse of gases, the reduction of the load on scrubber systems and the improvement of process efficiency. SF6/CF4 mixtures are also exploited in gas-insulated switchgears in low-temperature environments, such as arctic regions. The addition of CF4 to SF6 prevents condensation of SF6 at temperatures below 233 K, maintaining insulating properties and ensuring reliable operation. During maintenance or emptying of the system, it is necessary to separate the two components to optimize recycling, to avoid contamination and to restore the original mixture in a controlled manner. Given the growing concerns over the environmental impact of fluorinated gases, the development of fluorine-containing MOFs for their selective adsorption/separation has the potential to play a key role in mitigating their release into the atmosphere. The introduction of fluorinated linkers in a MOF can be accomplished either via direct synthesis under solvothermal conditions or via post-synthetic modification through the reaction known as Solvent-Assisted Ligand Incorporation (SALI).29–32 Recent work of our group has exploited the latter approach for the construction of mixed-linker MOFs (MIXMOFs) decorated with thiazole-based organic groups for assorted applications, spanning from CO2 capture to luminescence sensing of wastewater contaminants.33–36 Following this research interest and with the fluorinated gas adsorption/separation application in mind, we have prepared two new F-containing monocarboxylic thiazolium salts (HPhTzF)Br and (HPhTzCF3)Br (Scheme 1). Subsequently, we have decorated the 8-connected zirconium MOF [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2] (H4TCPB = 1,2,4,5-tetrakis(4-carboxyphenyl)benzene, Scheme 1, Zr_TCPB] with these salts using SALI. The as-obtained MIXMOFs after water elimination with minimal formulae [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br (Zr_TCPB_F) and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br (Zr_TCPB_CF3) have been fully characterized in the solid-state and exploited as functional materials for SF6 and CF4 adsorption.
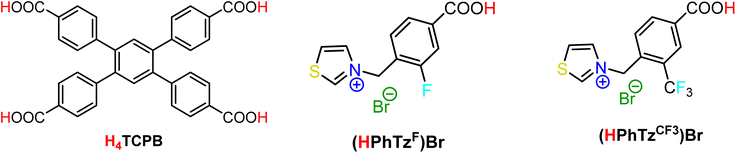 |
| | Scheme 1 The linkers used in this study for the construction of the ZrIV MIXMOFs Zr_TCPB_F and Zr_TCPB_CF3: 1,2,4,5-tetrakis(4-carboxyphenyl)benzene (H4TCPB), 3-(4-carboxy-2-fluorobenzyl)thiazol-3-ium bromide [(HPhTzF)Br] and 3-(4-carboxy-2-(trifluoromethyl)benzyl)thiazol-3-ium bromide [(HPhTzCF3)Br]. | |
2 Experimental section
2.1 Materials and methods
Zirconium chloride (ZrCl4, Sigma Aldrich), formic acid (HCOOH, Sigma Aldrich), N,N-dimethylformamide (DMF, Sigma Aldrich) and H2TCPB (Sigma Aldrich) were purchased from commercial vendors and used as received. Zr_TCPB was prepared according to the synthesis reported by Farha et al. in 2019.37 The preparation and characterization of [(HPhTzF)Br] and [(HPhTzCF3)Br] is reported in the SI (Fig. S1, S2, Tables S1 and S2). NMR spectra were recorded on BRUKER AVANCE 400/600 MHz spectrometers, with chemical shifts (δ) reported in parts per million (ppm) downfield of tetramethylsilane (TMS, 1H, 13C) or trichlorofluoromethane (CFCl3, 19F) and calibrated against the residual protiated solvent resonance. FT-IR spectra (KBr pellets) were recorded on a PerkinElmer Spectrum BX Series FTIR spectrometer, in the 4000–400 cm−1 range, with a 2 cm−1 resolution. Thermogravimetric analyses (TG-DTG) were performed under a N2 flow (100 mL min−1) at a heating rate of 5 K min−1 with an EXSTAR Thermo Gravimetric Analyzer Seiko 6200. The latter was coupled with a ThermoStarTM GSD 301T mass spectrometer for mass analysis of the volatile species. The elemental analyses were performed using a Thermo FlashEA 1112 Series CHNS–O elemental analyzer with an accepted tolerance of ±2% on carbon (C), hydrogen (H), nitrogen (N) and sulfur (S). X-ray fluorescence (XRF) qualitative analyses were performed under ambient conditions on powdered samples of Zr_TCPB_F and Zr_TCPB_CF3 using a PANalytical MINIPAL 2 instrument equipped with a sealed X-ray tube having a Cr anode. The generator was set at 30 kV and 3 µA. The nature and purity of all the batches employed for the functional characterization were assessed through powder X-ray diffraction (PXRD). PXRD qualitative measurements were carried out in the 2.0–50.0° 2θ region with a Panalytical X’PERT PRO diffractometer equipped with a Ni filter in the diffracted beam, a PIXcel© solid state detector and a sealed X-ray tube (Cu Kα, λ = 1.5418 Å). Slits were used on both the incident (Soller slits aperture: 0.25°; divergence slit aperture: 0.5°) and the diffracted (anti-scatter slit aperture: 7.5 mm) beam. The generator was operated at 40 kV and 40 mA.
2.2 Synthesis of Zr_TCPB_F
(HPhTzF)Br (FW = 318.16, 0.081 g, 0.25 mmol, 5 eq.) was dissolved in 45 mL of anhydrous MeCN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (9
DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_F was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 73% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzF)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_F, C81H53FNO32Zr6BrS (MW = 2230.6 g mol−1): C 43.6, H 2.4, N 0.6, S 1.4. Elemental analysis found (%): C, 43.8; H, 2.9; N, 0.7; S, 1.5. IR bands (KBr pellet, cm−1, Fig. S3): 1695(br), 1606(s), 1545(br), 1420(vs), 1183(w), 1146(w), 1108(w), 1018(w), 1004(w), 861(w), 782(m), 740(w), 716(m), 660(br), 582(w), 476(br).
1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_F was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 73% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzF)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_F, C81H53FNO32Zr6BrS (MW = 2230.6 g mol−1): C 43.6, H 2.4, N 0.6, S 1.4. Elemental analysis found (%): C, 43.8; H, 2.9; N, 0.7; S, 1.5. IR bands (KBr pellet, cm−1, Fig. S3): 1695(br), 1606(s), 1545(br), 1420(vs), 1183(w), 1146(w), 1108(w), 1018(w), 1004(w), 861(w), 782(m), 740(w), 716(m), 660(br), 582(w), 476(br).
2.3. Synthesis of Zr_TCPB_CF3
(HPhTzCF3)Br (FW = 368.17, 0.094 g, 0.26 mmol, 5 eq.) was dissolved in 45 mL of anhydrous MeCN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (9
DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_CF3 was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 75% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzCF3)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_CF3, C82H53F3NO32Zr6BrS (MW = 2280.6 g mol−1): C 43.2, H 2.3, N 0.6, S 1.4. Elemental analysis found (%): C, 43.4; H, 2.6; N, 0.6; S, 1.7. IR bands (KBr pellet, cm−1, Fig. S4): 1701(br), 1602(s), 1543(br), 1414(vs), 1308(m), 1288(w), 1122(br), 1054(w), 1020(w), 861(w), 782(m), 714(m), 662(br), 584(br), 478(br).
1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_CF3 was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 75% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzCF3)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_CF3, C82H53F3NO32Zr6BrS (MW = 2280.6 g mol−1): C 43.2, H 2.3, N 0.6, S 1.4. Elemental analysis found (%): C, 43.4; H, 2.6; N, 0.6; S, 1.7. IR bands (KBr pellet, cm−1, Fig. S4): 1701(br), 1602(s), 1543(br), 1414(vs), 1308(m), 1288(w), 1122(br), 1054(w), 1020(w), 861(w), 782(m), 714(m), 662(br), 584(br), 478(br).
2.4. Textural properties assessment through N2 adsorption. CF4 and SF6 adsorption isotherms
Due to the thermal instability of the thiazolium salts when encapsulated in Zr_TCPB (see the TGA-MS results reported in Fig. S9 and S10), the powdered samples (ca. 40 mg) of Zr_TCPB and Zr_TCPB_F/CF3 were activated at T = 423 K and 298 K respectively, under high vacuum (10−6 Torr) for 24 h before the measurement. The Brunauer–Emmett–Teller (BET) specific surface area, pore size distribution and pore volume (Vtot, Vmicro) were estimated by volumetric adsorption with an ASAP 2020 Micromeritics® instrument, using N2 as adsorbate at T = 77 K. For the BET specific surface area calculation, the 0.01–0.1 p/p0 pressure range of the isotherm was used to fit the data. Within this range, all the Rouquerol consistency criteria38,39 are satisfied. The material (micro)porosity was determined from the N2 adsorption isotherm using a NLDFT method (Tarazona approximation) and assuming a cylindrical pore shape (typical of metal oxides). SF6 and CF4 adsorption isotherms were recorded at T = 273 K, 298 K and 323 K at a maximum pressure of 1.2 bar, while the N2 adsorption isotherms used for the selectivity calculations were collected at T = 298 K at a maximum pressure of 1.2 bar. The isosteric heat of adsorption (Qst) values of both gases were calculated from the three isotherms at the lowest comparable coverage according to the differential form of the Clausius–Clapeyron equation:40,41| |
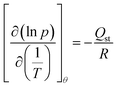 | (1) |
where R is the gas constant (8.314 J K−1 mol−1). The IAST A/B adsorption selectivity (A, B = SF6, CF4, N2) of binary mixtures at a total pressure of 1 bar42 and at T = 298 K was determined as the ratio of the adsorbed molar fractions of the two gases divided by the ratio of the gas phase initial molar fractions:43| |
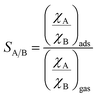 | (2) |
The (χA)ads and (χB)ads values were derived from the application of the free software pyIAST (https://github.com/CorySimon/pyIAST) to the experimental single-component isotherms collected at the chosen temperature. The initial composition (%) selected for the calculation was [50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50] for the [SF6
50] for the [SF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CF4] pair and [5
CF4] pair and [5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95] for the [SF6
95] for the [SF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2] and [CF4
N2] and [CF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2] pairs (the latter chosen to have compositions closer to the real atmospheric relative abundances). Various models (Quadratic, BET, Henry) were employed for the SF6, CF4 and N2 isotherms fitting, choosing the option that gives the lowest root-mean-square error (RMSE) on the experimental points (see Table S3 for the list of the fitting parameters). For a detailed explanation of these models and the related parameters, see the pyIAST webpage and documentation.
N2] pairs (the latter chosen to have compositions closer to the real atmospheric relative abundances). Various models (Quadratic, BET, Henry) were employed for the SF6, CF4 and N2 isotherms fitting, choosing the option that gives the lowest root-mean-square error (RMSE) on the experimental points (see Table S3 for the list of the fitting parameters). For a detailed explanation of these models and the related parameters, see the pyIAST webpage and documentation.
2.5. Computational details
Periodic DFT calculations were performed at the PBEsol0-3c level of theory,44 developed by some of us in the CRYSTAL code.45 PBEsol0-3c is a cost-effective composite method that has been shown to accurately predict structural features46 and adsorption properties33,47–49 of MOFs. It is based on the global hybrid PBEsol0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50,51
50,51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exchange–correlation functional combined with a double-zeta quality basis set for solid-state systems, and augmented with the D3
exchange–correlation functional combined with a double-zeta quality basis set for solid-state systems, and augmented with the D3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 52 and gCP53 corrections to account for van der Waals interactions and to remove the basis set superposition error (BSSE), respectively. For the numerical integration of the exchange-correlation term, 75 radial points and 974 angular points (XLGRID) in a Lebedev scheme were adopted. The SCF convergence was set to 10−7 during geometry optimization. The Pack–Monkhorst/Gilat shrinking factors for the diagonalization of the Kohn–Sham matrix in reciprocal space were set to 2. The truncation criteria (TOLINTEG) for the bielectronic integrals (Coulomb and exchange series) were set to 7 7 7 7 25. The JMol54 software was used to plot the electrostatic potential (ESP) maps.
52 and gCP53 corrections to account for van der Waals interactions and to remove the basis set superposition error (BSSE), respectively. For the numerical integration of the exchange-correlation term, 75 radial points and 974 angular points (XLGRID) in a Lebedev scheme were adopted. The SCF convergence was set to 10−7 during geometry optimization. The Pack–Monkhorst/Gilat shrinking factors for the diagonalization of the Kohn–Sham matrix in reciprocal space were set to 2. The truncation criteria (TOLINTEG) for the bielectronic integrals (Coulomb and exchange series) were set to 7 7 7 7 25. The JMol54 software was used to plot the electrostatic potential (ESP) maps.
3 Results and discussion
3.1 Synthesis and characterization of Zr_TCPB_F and Zr_TCPB_CF3
With the aim of preparing functional MOFs with fluorinated groups through extra linker insertion, we chose to start from the 8-coordinated zirconium MOF built with the commercially available tetradentate linker 1,2,4,5-tetrakis(4-carboxyphenyl)benzene: Zr_TCPB. This MOF has already been reported in the literature by the groups of Stock (with the label Zr-CAU-24) and Farha (called NU-903) in 2016![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 55 and 2019,37 respectively. It is a porous framework with rhombic channels of approximately 5.3 × 10.5 Å, suitable for hosting additional linkers of small–medium size. With this in mind and following the same approach used in the past for the synthesis of MOF linkers through N-quaternization of thiazole rings,35,36 we designed two new monocarboxylic fluorinated linkers using the F-containing molecules 4-(bromomethyl)-3-fluorobenzoic acid and 4-(bromomethyl)-3-(trifluoromethyl)benzoic acid (see the SI). The molecular size of [(HPhTzF)Br] (8.8 Å) and [(HPhTzCF3)Br] (9.0 Å) estimated from their crystal structures perfectly fits with the channel dimensions of Zr_TCPB. Thus, the MIXMOFs Zr_TCPB_F and Zr_TCPB_CF3 preparation was attempted through a post-synthetic SALI functionalization, i.e. a condensation reaction between the –COOH group of the thiazolium salts and the OH/H2O groups on the defects of the 8-coordinated [Zr6] metallic nodes. The pristine MOF was suspended in an anhydrous MeCN/DMSO mixture containing the dissolved salts and kept at T = 353 K for 24 h under gentle stirring. Subsequent filtration and washing with hot MeCN/acetone led to the successful obtainement of the pure products. Zr_TCPB_F and Zr_TCPB_CF3 have been thoroughly characterized in the solid state. The IR spectroscopic analysis confirms the extra-ligand insertion (Fig. S3 and S4), highlighting some typical bands of (HPhTzF)Br/(HPhTzCF3)Br at 1695/1701 cm−1 [ν(COO)], 1545/1543 cm−1 [ν(C
55 and 2019,37 respectively. It is a porous framework with rhombic channels of approximately 5.3 × 10.5 Å, suitable for hosting additional linkers of small–medium size. With this in mind and following the same approach used in the past for the synthesis of MOF linkers through N-quaternization of thiazole rings,35,36 we designed two new monocarboxylic fluorinated linkers using the F-containing molecules 4-(bromomethyl)-3-fluorobenzoic acid and 4-(bromomethyl)-3-(trifluoromethyl)benzoic acid (see the SI). The molecular size of [(HPhTzF)Br] (8.8 Å) and [(HPhTzCF3)Br] (9.0 Å) estimated from their crystal structures perfectly fits with the channel dimensions of Zr_TCPB. Thus, the MIXMOFs Zr_TCPB_F and Zr_TCPB_CF3 preparation was attempted through a post-synthetic SALI functionalization, i.e. a condensation reaction between the –COOH group of the thiazolium salts and the OH/H2O groups on the defects of the 8-coordinated [Zr6] metallic nodes. The pristine MOF was suspended in an anhydrous MeCN/DMSO mixture containing the dissolved salts and kept at T = 353 K for 24 h under gentle stirring. Subsequent filtration and washing with hot MeCN/acetone led to the successful obtainement of the pure products. Zr_TCPB_F and Zr_TCPB_CF3 have been thoroughly characterized in the solid state. The IR spectroscopic analysis confirms the extra-ligand insertion (Fig. S3 and S4), highlighting some typical bands of (HPhTzF)Br/(HPhTzCF3)Br at 1695/1701 cm−1 [ν(COO)], 1545/1543 cm−1 [ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)], 1420/1414 cm−1 [δ(CH/CH2)], 1183/1308 cm−1 [ν(Ar–F)/ν(CF3)] and 861/860 [γ(C–H)] respectively, besides those of TCPB4− at 1605/1601 cm−1 [ν(COO)]. The XRF qualitative analysis (Fig. S5 and S6) highlighted the presence of sulphur and bromine, confirming the successful SALI functionalization and revealing that the (HPhTzF)Br and (HPhTzCF3)Br ligands are incorporated within the MOF in their deprotonated form; the bromide anion is then necessary to balance the overall framework charge. Powder X-ray diffraction (PXRD, Fig. 1a) confirms that the parent crystallographic symmetry (orthorhombic, space group Cmmm)37 and network structural motif remain unaltered after functionalization. Differences in diffraction peaks relative intensities are due to changes in the electron density distribution introduced by the thiazolium salts within the unit cell. The extra-ligand is very likely to replace one OH/H2O couple on the metal node and not a bridging formate, given the favourite entropic factor associated to this coordination mode and the higher acidity of formic acid (pKa = 3.75) compared to benzoic acid (pKa = 4.19) that prevents a formate/benzoate exchange. This hypothesis is also supported by the presence of formic acid (m/z = 45 a.m.u.) in the mass spectra of the volatile byproducts forming during the MOFs thermal decomposition (vide infra). Therefore, this model structure has also been taken as starting guess for the DFT analysis (§3.4). To further support our structural hypothesis, a comparison between the experimental PXRD pattern and a “computational” pattern derived from the DFT-optimized model structures of Zr_TCPB_F and Zr_TCPB_CF3 has been made (Fig. S7 and S8). The matching between experimental and theoretical pattern is quite satisfactory. In the latter, there are more peaks coming from the lower crystal symmetry of the DFT model in comparison with that of the real crystal, because in the model there is only a finite portion of the lattice. This symmetry decrease generates additional peaks that are absent in the real sample. Since both (HPhTzF)Br and (HPhTzCF3)Br are unstable under strongly acidic or basic conditions (but –F/–CF3 signals remain detectable), linker quantification was carried out through combined 1H and 19F NMR spectroscopy of the digested solid in acidic solution (D2SO4). See the SI and Fig. S9–S12 for details. 19F signals are integrated against the 1H NMR signals of the H4TCPB ligand using 2,6-difluorobenzoic acid an internal standard.56 The multinuclear NMR analysis accounts for one thiazolium salt molecule per minimal formula, in line with the estimation coming from the S-elemental analysis (Experimental section). Consequently, based on the ligands relative stoichiometric ratio the MOF minimal formulae can be written as [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br. Thermogravimetric analysis (TGA, Fig. S13a and S14a) showed that the thermal stability of both MIXMOFs is lower than that of parent Zr_TCPB (Tdec = 773/798 vs. 820 K for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively). An initial weight loss of ca. 18–20 wt% (in line with the stoichiometric 1
C)], 1420/1414 cm−1 [δ(CH/CH2)], 1183/1308 cm−1 [ν(Ar–F)/ν(CF3)] and 861/860 [γ(C–H)] respectively, besides those of TCPB4− at 1605/1601 cm−1 [ν(COO)]. The XRF qualitative analysis (Fig. S5 and S6) highlighted the presence of sulphur and bromine, confirming the successful SALI functionalization and revealing that the (HPhTzF)Br and (HPhTzCF3)Br ligands are incorporated within the MOF in their deprotonated form; the bromide anion is then necessary to balance the overall framework charge. Powder X-ray diffraction (PXRD, Fig. 1a) confirms that the parent crystallographic symmetry (orthorhombic, space group Cmmm)37 and network structural motif remain unaltered after functionalization. Differences in diffraction peaks relative intensities are due to changes in the electron density distribution introduced by the thiazolium salts within the unit cell. The extra-ligand is very likely to replace one OH/H2O couple on the metal node and not a bridging formate, given the favourite entropic factor associated to this coordination mode and the higher acidity of formic acid (pKa = 3.75) compared to benzoic acid (pKa = 4.19) that prevents a formate/benzoate exchange. This hypothesis is also supported by the presence of formic acid (m/z = 45 a.m.u.) in the mass spectra of the volatile byproducts forming during the MOFs thermal decomposition (vide infra). Therefore, this model structure has also been taken as starting guess for the DFT analysis (§3.4). To further support our structural hypothesis, a comparison between the experimental PXRD pattern and a “computational” pattern derived from the DFT-optimized model structures of Zr_TCPB_F and Zr_TCPB_CF3 has been made (Fig. S7 and S8). The matching between experimental and theoretical pattern is quite satisfactory. In the latter, there are more peaks coming from the lower crystal symmetry of the DFT model in comparison with that of the real crystal, because in the model there is only a finite portion of the lattice. This symmetry decrease generates additional peaks that are absent in the real sample. Since both (HPhTzF)Br and (HPhTzCF3)Br are unstable under strongly acidic or basic conditions (but –F/–CF3 signals remain detectable), linker quantification was carried out through combined 1H and 19F NMR spectroscopy of the digested solid in acidic solution (D2SO4). See the SI and Fig. S9–S12 for details. 19F signals are integrated against the 1H NMR signals of the H4TCPB ligand using 2,6-difluorobenzoic acid an internal standard.56 The multinuclear NMR analysis accounts for one thiazolium salt molecule per minimal formula, in line with the estimation coming from the S-elemental analysis (Experimental section). Consequently, based on the ligands relative stoichiometric ratio the MOF minimal formulae can be written as [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br. Thermogravimetric analysis (TGA, Fig. S13a and S14a) showed that the thermal stability of both MIXMOFs is lower than that of parent Zr_TCPB (Tdec = 773/798 vs. 820 K for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively). An initial weight loss of ca. 18–20 wt% (in line with the stoichiometric 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 [Zr6]
1 [Zr6]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) thiazolium salt ratio) can be reasonably ascribed to loss of the salt and formic acid. Indeed, the DTG peaks found in this range fall at T = 429 K (salt) and T = 543 K (HCOOH). The former temperature value is close to that found for the decomposition of the isolated (HPhTzF)Br and (HPhTzCF3)Br, occurring at T = 495 and 530 K, respectively (Fig. S15). Further proof of evidence is provided by the MS analysis of the volatiles (Fig. S13b and S14b), where two peaks at m/z = 85 and 46 a.m.u. typical of thiazole and formic acid appear in the same temperature range. MOF decomposition is witnessed by the presence in the MS spectra of the volatiles of the peak at m/z = 78 a.m.u. typical of phenyl rings coming from TCPB.
thiazolium salt ratio) can be reasonably ascribed to loss of the salt and formic acid. Indeed, the DTG peaks found in this range fall at T = 429 K (salt) and T = 543 K (HCOOH). The former temperature value is close to that found for the decomposition of the isolated (HPhTzF)Br and (HPhTzCF3)Br, occurring at T = 495 and 530 K, respectively (Fig. S15). Further proof of evidence is provided by the MS analysis of the volatiles (Fig. S13b and S14b), where two peaks at m/z = 85 and 46 a.m.u. typical of thiazole and formic acid appear in the same temperature range. MOF decomposition is witnessed by the presence in the MS spectra of the volatiles of the peak at m/z = 78 a.m.u. typical of phenyl rings coming from TCPB.
 |
| | Fig. 1 (a) PXRD patterns (2–50° 2θ region) of Zr_TCPB, Zr_TCPB_F and Zr_TCPB_CF3 at comparison. (b) N2 isotherms measured at T = 77 K on thermally activated Zr_TCPB, Zr_TCPB_F and Zr_TCPB_CF3 at comparison. Empty symbols denote the desorption branch. | |
3.2. Textural properties assessment. CF4 and SF6 adsorption isotherms
The porosity of Zr_TCPB_F/Zr_TCPB_CF3 was evaluated through volumetric N2 adsorption at T = 77 K on pre-activated samples (Fig. 1b). The isotherm shape of Type I is the same as that of Zr_TCPB, typical of microporous materials. The BET surface area (1141/1126 vs. 1406 m2 g−1 for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively) as well as the total pore volume (0.53/0.46 vs. 1.60 cm3 g−1 for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively) are lower than that of pristine Zr_TCPB. This is in line with a partial micropore filling by the added thiazolium salts and of the successful SALI functionalization. Additional proof of evidence comes from the DFT pore size distribution (Fig. S16): the two main micropore sizes centered at w = 13.5 and 14.5 Å present in pristine Zr_TCPB partially disappear after the extra-linker inclusion in Zr_TCPB_F (main micropore diameter w = 13.5 Å only) and Zr_TCPB_CF3 (main micropore diameter w = 14.7 Å only). The BET area values found in our samples are comparable to those reported for similar MOFs in the literature bearing variably substituted TCPB linkers combined with [Zr6] metal nodes, such as NU-1008 (linker TCPB-Br2; BET area = 1370 m2 g−1),57 NU-600 (linker TCPB-Br2; BET area = 1490 m2 g−1), NU-603 (linkers TCPB-Br2 and BDC; BET area = 1300 m2 g−1), NU-604 (linkers TCPB-Br2 and BPDC; BET area = 1300 m2 g−1), NU-605 (linkers TCPB-Br2, BDC and BPDC; BET area = 1280 m2 g−1),58 NU-904 (linker TCPB-NO2; BET area = 1410 m2 g−1).37 The activated materials have been tested in SF6 and CF4 adsorption at pmax = 1.2 bar and at variable temperatures: T = 273, 298 and 323 K (Fig. S17 and S18). Both Zr_TCPB_F and Zr_TCPB_CF3 showed an increased affinity for both gases if compared with their parent MOF Zr_TCPB. The total SF6 uptake at pSF6 = 1 bar and T = 298 K is 32.8 wt% (2.2 mmol g−1) and 37.1 wt% (2.5 mmol g−1) for Zr_TCPB_F and Zr_TCPB_CF3, respectively (Fig. 2a). The absolute gas uptake at ambient temperature is proportional to the material surface area and it is much higher than that found for other Zr-based MOFs of the literature with smaller pore size like UiO-66 (1.6 mmol g−1) and UiO-66-Br2 (0.9 mmol g−1),59 but it is lower than that of UiO-67 (4.0 mmol g−1).60 In terms of SF6 isosteric heat of adsorption at zero coverage (Qst), the thiazolium-functionalized MOFs are featured by a higher Qst value than that found for its parent analogue (27.5/27.0 vs. 24.9 kJ mol−1 for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively, Table 1, Fig. S19 and S20). This is a proof of evidence that the SALI functionalization is beneficial for the improvement of the material thermodynamic affinity for sulfur hexafluoride. SF6 adsorption at low pressures can be greatly improved by incorporating a fluorinated functional group with a high polarizability into the pores of the adsorbent; the introduction of a polar molecule like a fluorinated thiazolium salt into the MOF channels strengthens the MOF–SF6 interaction. However, in comparison with other Zr-based MOFs of the literature (Table S4), the absolute Qst (SF6) values of Zr_TCPB_F and Zr_TCPB_CF3 are lower than those of the UiO-66-X (X = NH2, NO2, Cl, Br, Br, I) family (between 32 and 45 kJ mol−1)59 but they are higher than that of UiO-67 (20 kJ mol−1).60 This may be due to the stronger host-guest interaction occurring in smaller pores. Widening the horizon to non-Zr-based MOFs, the values found in our samples are comparable to those of MIL-100(Fe) (2.7 mmol g−1) or DUT-9 (2.1 mmol g−1), but they are lower than that of HKUST-1 with CuII open metal sites (4.0 mmol g−1).25 As for CF4, the total gas uptake at pCF4 = 1 bar and T = 298 K is 6.5 wt% (0.7 mmol g−1) and 4.1 wt% (0.5 mmol g−1) for Zr_TCPB_F and Zr_TCPB_CF3, respectively (Fig. 2b). The quantities adsorbed are smaller than those of SF6 under the same temperature and pressure conditions, and they are again proportional to the surface areas. The values found in our samples are comparable to those found for the UiO-66-X (X = NH2, NO2, Cl, Br, Br, I) family (between 0.5 and 0.7 mmol g−1, Table S4).59 If we consider other non-Zr-based MOFs, the amount of CF4 adsorbed by Zr_TCPB_F in particular is comparable to that of HKUST-1 (1.0 mmol g−1),25 despite the absence of open metal sites in the former. As observed for SF6, the CF4 isosteric heats of adsorption at zero coverage of both Zr_TCPB_F and Zr_TCPB_CF3 are higher than that of bare Zr_TCPB (22.1/30.1 vs. 21.9 kJ mol−1 for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively, Table 1, Fig. S19 and S20), showing again the beneficial effect coming from the inclusion of the fluorinated linker in the pores. Interestingly, a simple modification of the fluorinated group in the thiazolium salt induces a switch in the thermodynamic affinity for the two gases: while the fluoro substituent in Zr_TCPB_F gives a material where Qst (SF6) > Qst (CF4), the opposite holds for the trifluoromethyl group in Zr_TCPB_CF3, where Qst (SF6) < Qst (CF4). Therefore, these MOFs may represent “smart materials” for the discrimination of fluorinated polluting gases, opening new horizons in the field of air treatment and cleaning. In order to validate the suitability of these materials for practical use in real life contexts, long-term cycling stability tests were carried out with Zr_TCPB_F and SF6 at ambient temperature (Fig. S21). The amount of gas adsorbed is constant after four repeated adsorption/desorption cycles on the same batch (briefly re-activated under high vacuum for 2 h each time before the next cycle), confirming the MOF stability under these conditions. To shed further light on the MOFs adsorption behavior, IAST selectivity (SSF6/CF4) data for [SF6/CF4] binary equimolar mixtures at T = 298 K were estimated; the results are summarized in Table 2. Despite their different Qst values, all the three samples examined in this study show a preferential adsorption for sulfur hexafluoride. Albeit it is difficult to provide a comprehensive explanation of this phenomenon, the reason may stem from the different
polarizability of the two gases. Both CF4 and SF6 are globally nonpolar, but for slightly different reasons. C–F and S–F are both highly polarized bonds due to the high electronegativity of fluorine. The tetrahedral/octahedral molecular symmetry causes these dipoles to cancel each other out. However, SF6 has a higher polarizability (due to the much larger and more diffuse central sulfur atom) that translates into stronger dispersion (van der Waals) interactions with the MOFs. On the other hand, CF4 is less polarizable, more volatile and less easily adsorbed. The analysis of the SF6/CF4 adsorption selectivity and the possibility to discriminate between SF6 and CF4 through tailored (fluorinated) MOFs is unprecedented to our knowledge, and even more unexpected given the identical C–F and S–F bond polarization (based on the elements electronegativity: 2.5 and 4.0 for both C/S and F, respectively) and taking into account the considerations outlined in the previous paragraph. SSF6/CF4 reaches its maximum value (9) in Zr_TCPB_CF3. While the presence of the fluoro-substituent does not modify the selectivity significantly, a better improvement is observed upon introduction of the trifluoromethyl group. As for the selectivity towards N2, all MOFs show a preferential adsorption for the fluorinated gases (for SF6 in particular), given the extremely low amount of N2 adsorbed at ambient temperature (Fig. S22) even from highly nitrogen-enriched mixtures as that taken into account for the selectivity calculation [95%(N2): 5%(fluorinated gas)], more representative of the real atmospheric composition (the current concentration of CF4 and SF6 in the atmosphere is 90–100 ppt and 12 ppt, respectively).
 |
| | Fig. 2 Collective (a) SF6 and (b) CF4 adsorption isotherms at T = 298 K for Zr_TCPB, Zr_TCPB_F and Zr_TCPB_CF3 in comparison. The corresponding graphs for T = 273 K and T = 323 K are reported in Fig. S17 and S18, respectively. | |
Table 1 Main adsorption data for the MOFs in this study
| Sample |
BET area [m2 g−1] |
Qst (CF4) [kJ mol−1] |
CF4 quantity adsorbed at T = 298 K, p = 1 bar [mmol g−1] |
Qst (SF6) [kJ mol−1] |
SF6 quantity adsorbed at T = 298 K, p = 1 bar [mmol g−1] |
| Zr_TCPB |
1406 |
21.9 |
0.54 (4.7 wt%) |
24.9 |
3.04 (44.4 wt%) |
| Zr_TCPB_F |
1141 |
22.1 |
0.73 (6.5 wt%) |
27.5 |
2.24 (32.8 wt%) |
| Zr_TCPB_CF3 |
1126 |
30.1 |
0.47 (4.1 wt%) |
27.0 |
2.54 (37.1 wt%) |
Table 2 IAST SF6/CF4 adsorption selectivity data of binary gas mixtures [50%:50%], SF6/N2 and CF4/N2 adsorption selectivity data of binary gas mixtures [5%:95%] at ptot = 1 bar and T = 298 K for Zr_TCPB, Zr_TCPB_F and Zr_TCPB_CF3
| MOF |
SSF6/CF4 |
SSF6/N2 |
SCF4/N2 |
| Zr_TCPB |
6 |
25 |
4 |
| Zr_TCPB_F |
5 |
42 |
7 |
| Zr_TCPB_CF3 |
9 |
38 |
3 |
3.3. Dynamic breakthrough experiments with binary SF6/CF4 mixtures
Given the promising separation potential, the breakthrough experiment was further implemented at ambient conditions with the binary SF6/CF4 gaseous blend (volume ratio 50/50) that mimics the real typical industrial composition. At T = 298 K and ptot = 1 bar, the SF6/CF4 mixture was fed at a flow rate of 2 mL min−1 into adsorption columns. The actual separation performance of the adsorbent for the two components is evaluated by monitoring the relative breakthrough times of SF6 and CF4. Experimental results demonstrate that both Zr_TCPB_F and Zr_TCPB_CF3 achieve effective separation of SF6 from CF4. As displayed in Fig. 3, CF4 elutes immediately from the breakthrough column at 12.5 min and 10 min for Zr_TCPB_F and Zr_TCPB_CF3, respectively. In stark contrast, SF6 exhibits a noticeable breakthrough delay with its elution detected at 25 min for both MOFs, with an experimentally measured residence time difference between the two gases of 12.5 min and 15 min for Zr_TCPB_F and Zr_TCPB_CF3, respectively.
 |
| | Fig. 3 Breakthrough curves for a binary SF6/CF4 (50/50, v/v) mixture: (a) Zr_TCPB_F and (b) Zr_TCPB_CF3. | |
The separation factors obtained from the breakthrough experiments (Fig. S23 and eqn S3) are 6.66 for Zr_TCPB_F and 11.40 for Zr_TCPB_CF3, while the calculated IAST SF6/CF4 adsorption selectivity equals 5 and 9, respectively (Table 2). Although the experimental separation factors slightly exceed the calculated IAST selectivity values, the overall trend holds true, with Zr_TCPB_CF3 exhibiting superior separation efficiency. Furthermore, the high purity SF6 productivity of Zr_TCPB_CF3 calculated from single separation breakthrough curves is about 1.61 mmol g−1, larger than the 1.53 mmol g−1 found for Zr_TCPB_F (eqn S1 and S2). This evidence shows that multiple and complex supramolecular interactions are at work when the two MOFs bind SF6 or CF4.
3.4. DFT calculation of the electronic structure of Zr_TCPB_F/CF3 and their CF4/SF6 adducts
To build a realistic computational model of Zr_TCPB, the initial guess structure was taken from the CIF file deposited in the Cambridge Structural Database (CSD) with RefCode IZIBUQ.55 The Zr atoms of the Inorganic Building Unit (IBU) have been saturated with OH and H2O to ensure the electroneutrality of the model system. For the Zr_TCPB_F and Zr_TCPB_CF3 models, the thiazolium salts were covalently bound to the inorganic nodes, by removing one H2O molecule and a OH− group from the IBU, and a proton (H+) from the (HPhTzF)Br and (HPhTzCF3)Br salts, respectively. First, the atomic positions and lattice parameters of the empty framework models were fully relaxed. Then, to gain insight into the host-guest interactions and to identify the primary CF4 and SF6 adsorption sites in the MOFs, the gas molecules were manually docked into the framework pores by maximizing the interactions between host and guest through the complementarity of electrostatic potentials (Fig. S24–S27). In Zr_TCPB, CF4 and SF6 form hydrogen bonds with OH groups and H2O molecules of the [Zr6(µ3-O)4(µ3-OH)4]12+ node and engage in F⋯H–C interactions with the TCPB4− linker (Fig. S28 and S29). The same kind of hydrogen bonds were found in Zr_TCPB_F and Zr_TCPB_CF3 where the host-guest assemblies are further characterized by the formation of F⋯H hydrogen bonds and electrostatic interactions with the thiazolium salts, as shown by the optimized structures depicted in Fig. 4 and 5 for Zr_TCPB_F and in Fig. 6 and 7 for Zr_TCPB_CF3, respectively. Additionally, CF4 and SF6 tend to form weak sigma–hole (σ–hole) interactions with the Lewis base groups (H2O and OH) of the IBU through the polarized central atom. Similar weak interactions were previously reported for the selective adsorption of fluorinated gases in different MOF materials.22,61–63 Overall, the computed adsorption energies (Tables S5 and S6) are in reasonable agreement with the experimental findings, especially for Zr_TCPB and Zr_TCPB_CF3, while larger deviations are observed for Zr_TCPB_F. This discrepancy can be attributed to the presence of defects, amorphization, or other structural changes present in the real samples that were not accounted for in our “ideal” computational models. Given the limited thermal stability of the thiazolium salts [(HPhTzF)Br] and [(HPhTzCF3)Br], the activation of Zr_TCPB_F and Zr_TCPB_CF3 cannot be performed at high temperature. Consequently, some residual solvent may still be present after activation, blocking the gas access to the high-energy adsorption site (presumably close to the metallic node).
 |
| | Fig. 4 Optimized geometry of [CF4@Zr_TCPB_F] (portion of the crystal structure only, for the sake of clarity). Main gas-framework distances reported (Å). | |
 |
| | Fig. 5 Optimized geometry of [SF6@Zr_TCPB_F] (portion of the crystal structure only, for the sake of clarity). Main gas-framework distances reported (Å). | |
 |
| | Fig. 6 Optimized geometry of [CF4@Zr_TCPB_CF3] (portion of the crystal structure only, for the sake of clarity). Main gas-framework distances reported (Å). | |
 |
| | Fig. 7 Optimized geometry of [SF6@Zr_TCPB_CF3] (portion of the crystal structure only, for the sake of clarity). Main gas-framework distances reported (Å). | |
4 Conclusions
Following the research interest of our groups in the exploitation of MOFs for gas mixture adsorption and separation, we have prepared two new mixed-linker MOFs Zr_TCPB_F and Zr_TCPB_CF3 via SALI starting from the 8-connected zirconium MOF Zr_TCPB and decorating it with two fluorinated monocarboxylic thiazolium salts. The solids have been fully characterized in the solid-state and tested as functional materials for SF6 and CF4 adsorption. The presence of the F-containing dangling group in the MOFs pores is beneficial for the improvement of their chemical interaction with fluorinated gases, as witnessed by the higher SF6 and CF4 adsorption enthalpies of Zr_TCPB_F and Zr_TCPB_CF3 if compared with those of their parent material Zr_TCPB. More importantly, the (unprecedented) possibility to switch the MOF preferential adsorption from SF6 to CF4 through a simple replacement of the fluoro substituent with a trifluoromethyl group on the thiazolium salt opens promising perspectives for a selective adsorption of fluorinated gases with very similar physico-chemical nature (non-polar molecules) and bond polarity. Consequently, the prepared materials may find application in the field of atmospheric pollution treatment and air cleaning from dangerous gases to mitigate the greenhouse effect and the ozone layer depletion.
Author contributions
G. P., G. B., Z. F., T. Y.: investigation, formal analysis; B. C., G. G., G. T., H. H.: validation; L. D.: investigation, formal analysis, methodology, software, writing – original draft; A. R.: conceptualization, funding acquisition, project administration, supervision, writing – original draft.
Conflicts of interest
The authors have no conflicts of interest to declare.
Data availability
CCDC 2448941 (HPhTzF)Br and 2448942 (HPhTzCF3)Br contain the supplementary crystallographic data for this paper.64a,b
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthetic procedures and characterization (including crystal structure solution from single-crystal X-ray diffraction) of (HPhTzF)Br and (HPhTzCF3)Br, IR, XRF and TG-MS spectra of Zr_TCPB_F and Zr_TCPB_CF3, comparison between experimental and DFT-simulated PXRD patterns of Zr_TCPB_F and Zr_TCPB_CF3, linkers quantification through 1H and 19F NMR spectroscopy of the digested samples, breakthrough experiments details and methods, Clausius–Clapeyron data fitting for Qst calculations, long-term stability tests on repeated adsorption/desorption cycles, additional computational details: electrostatic potential maps, interaction energies, optimized geometries of [CF4@Zr_TCPB] and [SF6@Zr_TCPB]. See DOI: https://doi.org/10.1039/d5ta10343f.
Acknowledgements
A. R. and G. B. would like to acknowledge the Italian Ministry of University and Research (MUR) and the European Union (Next Generation EU) for funding this research activity through the PRIN 2022 project LUMIMOF (2022A3XNWJ) “Wastewater treatment and monitoring with luminescent mixed-linker Metal–Organic Frameworks as chemical sensors and adsorbents of contaminants of emerging concern”. A. R. is also grateful to Prof. Simona Galli and Ms. Anna Mauri (Università dell’Insubria, Como, Italy) for help with the XRF data collection and for fruitful crystallographic discussions. L. D. and B. C. gratefully acknowledge the support from the Project CH4.0 under the MUR program “Dipartimenti di Eccellenza 2023–2027” (CUP: D13C22003520001).
References
- D. J. Sheldon and M. R. Crimmin, Chem. Soc. Rev., 2022, 51, 4977–4995 RSC.
- https://www.ipcc.ch/sr15/chapter/chapter-2/, last access: December 2025.
- https://www.epa.gov/ghgemissions/fluorinated-gas-emissions, last access: December 2025.
- J. Kim, R. Thompson, H. Park, S. Bogle, J. Mühle, M.-K. Park, Y. Kim, C. M. Harth, P. K. Salameh, R. Schmidt, D. Ottinger, S. Park and R. F. Weiss, J. Geophys. Res.:Atmos., 2021, 126, e2021JD034888 CrossRef CAS PubMed.
- D. Say, A. J. Manning, L. M. Western, D. Young, A. Wisher, M. Rigby, S. Reimann, M. K. Vollmer, M. Maione, J. Arduini, P. B. Krummel, J. Mühle, C. M. Harth, B. Evans, R. F. Weiss, R. G. Prinn and S. O'Doherty, Atmos. Chem. Phys., 2021, 21, 2149–2164 CrossRef CAS.
- R. H. Harrison and D. R. Douslin, J. Chem. Eng. Data, 1966, 11, 383–388 Search PubMed.
- C. T. Dervos and P. Vassiliou, J. Air Waste Manage. Assoc., 2000, 50, 137–141 CrossRef CAS PubMed.
- H. C. Miller, L. S. Verdelli and J. F. Gall, Ind. Eng. Chem., 1951, 43, 1126–1129 CrossRef CAS.
- W. C. Schumb and E. L. Gamble, J. Am. Chem. Soc., 1930, 52, 4302–4308 CrossRef CAS.
- https://www.un.org/en/climatechange/paris-agreement, last access: December 2025.
- O. M. Yaghi, M. J. Kalmutzki and C. S. Diercks, Introduction to Reticular Chemistry: Metal–Organic Frameworks and Covalent Organic Frameworks, Wiley-VCH Verlag GmbH & Co. KGaA, 2019 Search PubMed.
- H. García and S. Navalón, Metal–Organic Frameworks: Applications in Separations and Catalysis, ed. H. García and S. Navalón, Wiley‐VCH Verlag GmbH & Co. KGaA, 2018 Search PubMed.
- S. Kaskel, The Chemistry of Metal–Organic Frameworks: Synthesis, Characterization, and Applications, ed. S. Kaskel, Wiley‐VCH Verlag GmbH & Co. KGaA, 2016 Search PubMed.
- D. Farrusseng, Metal–Organic Frameworks: Applications from Catalysis to Gas Storage, Wiley-VCH Verlag, Weinheim, 2011 Search PubMed.
- X. Sun, L. Zhou, J. Chen, Z. Jia, Z. Zhao and Z. Zhao, J. Mater. Chem. A, 2025, 13, 2360–2377 Search PubMed.
- E. Jo, S. Yang, D. W. Kim and D. W. Kang, Coord. Chem. Rev., 2024, 515, 215958 Search PubMed.
- H. Demir and S. Keskin, Mol. Syst. Des. Eng., 2022, 7, 1707–1721 Search PubMed.
- D. K. J. A. Wanigarathna, J. Gao and B. Liu, Mater. Adv., 2020, 1, 310–320 Search PubMed.
- G. Xu, T. Ke, R. Fan, K. Tan, W. Zhang, B. Su, Z. Zhang, Z. Bao, Q. Ren and Q. Yang, Adv. Sci., 2025, 12, 2411083 Search PubMed.
- J. Zhu, J. Hu, H. Xiao, L. Yang, M. Yang, S. Wang, J. Zhang and H. Xing, Sep. Purif. Technol., 2024, 331, 125614 Search PubMed.
- Z. Wan, T. Yan, M. Chang, M. Yang and D. Liu, Sep. Purif. Technol., 2023, 306, 122617 CrossRef CAS.
- S.-M. Wang, H.-L. Lan, G.-W. Guan and Q.-Y. Yang, ACS Appl. Mater. Interfaces, 2022, 14, 40072–40081 CrossRef CAS PubMed.
- M. Chang, T. Yan, Y. Wei, J.-X. Wang, D. Liu and J.-F. Chen, Chem. Mater., 2022, 34, 9134–9143 CrossRef CAS.
- T.-H. Chen, I. Popov, W. Kaveevivitchai, Y.-C. Chuang, Y.-S. Chen, A. J. Jacobson and O. Š. Miljanić, Angew. Chem., Int. Ed., 2015, 54, 13902–13906 CrossRef CAS PubMed.
- I. Senkovska, E. Barea, J. A. R. Navarro and S. Kaskel, Microporous Mesoporous Mater., 2012, 156, 115–120 CrossRef CAS.
- S.-M. Wang, P. Duan and Q.-Y. Yang, Coord. Chem. Rev., 2025, 525, 216339 CrossRef CAS.
- D. Morelli Venturi and F. Costantino, RSC Adv., 2023, 13, 29215–29230 RSC.
- A. Ebadi Amooghin, H. Sanaeepur, R. Luque, H. Garcia and B. Chen, Chem. Soc. Rev., 2022, 51, 7427–7508 Search PubMed.
- T. Islamoglu, S. Goswami, Z. Li, A. J. Howarth, O. K. Farha and J. T. Hupp, Acc. Chem. Res., 2017, 50, 805–813 CrossRef CAS PubMed.
- P. Deria, W. Bury, I. Hod, C.-W. Kung, O. Karagiaridi, J. T. Hupp and O. K. Farha, Inorg. Chem., 2015, 54, 2185–2192 Search PubMed.
- P. Deria, W. Bury, J. T. Hupp and O. K. Farha, Chem. Commun., 2014, 50, 1965–1968 RSC.
- P. Deria, J. E. Mondloch, O. Karagiaridi, W. Bury, J. T. Hupp and O. K. Farha, Chem. Soc. Rev., 2014, 43, 5896–5912 Search PubMed.
- G. Provinciali, A. L. Capodilupo, A. Mauri, S. Galli, L. Donà, B. Civalleri, G. Tuci, G. Giambastiani, C. Piccirillo and A. Rossin, ACS EST Water, 2024, 4, 2339–2351 Search PubMed.
- G. Mercuri, M. Moroni, S. Galli, C. Piccirillo, A.-L. Capodilupo, G. Tuci, G. Giambastiani and A. Rossin, Inorg. Chem. Front., 2022, 9, 90–102 RSC.
- G. Mercuri, M. Moroni, S. Galli, G. Tuci, G. Giambastiani, T. Yan, D. Liu and A. Rossin, ACS Appl. Mater. Interfaces, 2021, 13, 58982–58993 CrossRef CAS PubMed.
- L. Luconi, G. Mercuri, T. Islamoglu, A. Fermi, G. Bergamini, G. Giambastiani and A. Rossin, J. Mater. Chem. C, 2020, 8, 7492–7500 RSC.
- J. Lyu, X. Zhang, K.-i. Otake, X. Wang, P. Li, Z. Li, Z. Chen, Y. Zhang, M. C. Wasson, Y. Yang, P. Bai, X. Guo, T. Islamoglu and O. K. Farha, Chem. Sci., 2019, 10, 1186–1192 RSC.
- D. A. Gómez-Gualdrón, P. Z. Moghadam, J. T. Hupp, O. K. Farha and R. Q. Snurr, J. Am. Chem. Soc., 2016, 138, 215–224 Search PubMed.
- J. Rouquerol, P. Llewellyn and F. Rouquerol, in Studies in Surface Science and Catalysis, ed. P. L. Llewellyn, F. Rodriquez-Reinoso, J. Rouquerol and N. Seaton, Elsevier, Amsterdam, 2007, vol. 160, p. 49 Search PubMed.
- X. Zhu, C. Tian, G. M. Veith, C. W. Abney, J. Dehaudt and S. Dai, J. Am. Chem. Soc., 2016, 138, 11497–11500 Search PubMed.
- X. Zhu, S. M. Mahurin, S.-H. An, C.-L. Do-Thanh, C. Tian, Y. Li, L. W. Gill, E. W. Hagaman, Z. Bian, J.-H. Zhou, J. Hu, H. Liu and S. Dai, Chem. Commun., 2014, 50, 7933–7936 RSC.
- The total pressure value of 1 atm has been arbitrarily chosen for the sake of simplicity in the IAST calculations, since the SA/B values are only dependent from the mixture composition but not from its total pressure.
- J. Schell, N. Casas, R. Pini and M. Mazzotti, Adsorption, 2012, 18, 49–65 CrossRef CAS.
- L. Doná, J. G. Brandenburg and B. Civalleri, J. Chem. Phys., 2019, 151, 121101 CrossRef PubMed.
- A. Erba, J. K. Desmarais, S. Casassa, B. Civalleri, L. Donà, I. J. Bush, B. Searle, L. Maschio, L. Edith-Daga, A. Cossard, C. Ribaldone, E. Ascrizzi, N. L. Marana, J.-P. Flament and B. Kirtman, J. Chem. Theory Comput., 2023, 19, 6891–6932 CrossRef CAS PubMed.
- L. Donà, J. G. Brandenburg and B. Civalleri, J. Chem. Phys., 2022, 156, 094706 CrossRef PubMed.
- M. Pugliesi, M. Cavallo, C. Atzori, B. Garetto, E. Borfecchia, L. Donà, B. Civalleri, G. Tuci, G. Giambastiani, S. Galli, F. Bonino and A. Rossin, Adv. Funct. Mater., 2024, 34, 2403017 CrossRef CAS.
- T. Xiong, Y. Zhang, L. Donà, M. Gutiérrez, A. F. Möslein, A. S. Babal, N. Amin, B. Civalleri and J.-C. Tan, ACS Appl. Nano Mater., 2021, 4, 10321–10333 CrossRef CAS.
- B. E. Souza, L. Donà, K. Titov, P. Bruzzese, Z. Zeng, Y. Zhang, A. S. Babal, A. F. Möslein, M. D. Frogley, M. Wolna, G. Cinque, B. Civalleri and J.-C. Tan, ACS Appl. Mater. Interfaces, 2020, 12, 5147–5156 Search PubMed.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 Search PubMed.
- C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158–6170 Search PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- H. Kruse and S. Grimme, J. Chem. Phys., 2012, 136 Search PubMed.
- R. M. Hanson and X.-J. Lu, Nucleic Acids Res., 2017, 45, W528–W533 Search PubMed.
- M. Lammert, H. Reinsch, C. A. Murray, M. T. Wharmby, H. Terraschke and N. Stock, Dalton Trans., 2016, 45, 18822–18826 RSC.
- P. Deria, J. E. Mondloch, E. Tylianakis, P. Ghosh, W. Bury, R. Q. Snurr, J. T. Hupp and O. K. Farha, J. Am. Chem. Soc., 2013, 135, 16801–16804 Search PubMed.
- Y. Chen, S. Ahn, M. R. Mian, X. Wang, Q. Ma, F. A. Son, L. Yang, K. Ma, X. Zhang, J. M. Notestein and O. K. Farha, J. Am. Chem. Soc., 2022, 144, 3554–3563 Search PubMed.
- Y. Chen, K. B. Idrees, M. R. Mian, F. A. Son, C. Zhang, X. Wang and O. K. Farha, J. Am. Chem. Soc., 2023, 145, 3055–3063 Search PubMed.
- M.-B. Kim, K.-M. Kim, T.-H. Kim, T.-U. Yoon, E.-J. Kim, J.-H. Kim and Y.-S. Bae, Chem. Eng. J., 2018, 339, 223–229 Search PubMed.
- M.-B. Kim, T.-H. Kim, T.-U. Yoon, J. H. Kang, J.-H. Kim and Y.-S. Bae, J. Ind. Eng. Chem., 2020, 84, 179–184 Search PubMed.
- X. Zhang, Y.-L. Zhao, X.-Y. Li, X. Bai, Q. Chen and J.-R. Li, J. Am. Chem. Soc., 2024, 146, 19303–19309 Search PubMed.
- M. Zhang, L. Liu, Q. Li, H. Gong and Y. Chen, Ind. Eng. Chem. Res., 2023, 62, 7103–7113 Search PubMed.
- S.-M. Wang, X.-T. Mu, H.-R. Liu, S.-T. Zheng and Q.-Y. Yang, Angew. Chem., Int. Ed., 2022, 61, e202207066 CrossRef CAS PubMed.
-
(a) CCDC 2448941: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6b3b;
(b) CCDC 2448942: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6b4c.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article a,
Giulio Bicchierai
a,
Giulio Bicchierai a,
Zhi Fangb,
Tongan Yan
a,
Zhi Fangb,
Tongan Yan *c,
Lorenzo Donà
*c,
Lorenzo Donà *d,
Bartolomeo Civalleri
*d,
Bartolomeo Civalleri d,
Hongliang Huang
d,
Hongliang Huang c,
Giuliano Giambastiani
c,
Giuliano Giambastiani efa,
Giulia Tuci
efa,
Giulia Tuci af and
Andrea Rossin
af and
Andrea Rossin *af
*af
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times greater than CO2 on a per molecule basis.7–9 Small atmospheric concentrations of these two gases can have large effects on global temperatures. Thus, controlling and mitigating their release into the environment has become a critical objective for reducing the overall carbon footprint and achieving international climate goals, including those set out in the Paris Agreement.10 Metal–organic frameworks (MOFs), with their unique structural characteristics, have emerged as promising candidates for addressing this issue due to their exceptional surface area, tunable porosity, and chemical versatility.11–14 MOFs are typically composed of metal clusters linked by organic ligands, allowing for the precise design of pore structures that can selectively adsorb and separate specific gases. This adaptability makes MOFs a compelling solution for the selective capture of gases such as fluorinated compounds, which require highly specialized materials for effective separation. Given the strong C–F and S–F bonds in CF4 and SF6, their adsorption requires MOFs with tailored pore environments and strong host-guest interactions.15–18 MOFs containing open metal sites, fluorophilic functional groups, or optimized pore dimensions have demonstrated enhanced uptake of these gases. Additionally, post-synthetic modifications and functionalized ligands can further improve adsorption performance.19–25 In recent years, the incorporation of fluorine into the structure of MOFs has opened new avenues for enhancing their performance in gas separation applications. Fluorine, with its strong electronegativity, can contribute to the stabilization of metal centers and introduce additional functionalities to the material. Fluorinated MOFs in particular offer advantages, such as increased chemical stability and enhanced interactions with fluorinated gases. These unique properties enable these materials to act as highly efficient adsorbents, capable of selectively capturing and separating fluorinated compounds from complex gas mixtures.26–28 In the semiconductor industry, CF4 and SF6 mixtures are used in plasma etching processes for the fabrication of integrated circuits. These mixtures allow precise control of the etching, thanks to their chemical properties and the ability to form reactive plasmas. After use, exhaust gases may contain reactive or incompletely dissociated residues. Separating CF4 from SF6 allows the recovery and reuse of gases, the reduction of the load on scrubber systems and the improvement of process efficiency. SF6/CF4 mixtures are also exploited in gas-insulated switchgears in low-temperature environments, such as arctic regions. The addition of CF4 to SF6 prevents condensation of SF6 at temperatures below 233 K, maintaining insulating properties and ensuring reliable operation. During maintenance or emptying of the system, it is necessary to separate the two components to optimize recycling, to avoid contamination and to restore the original mixture in a controlled manner. Given the growing concerns over the environmental impact of fluorinated gases, the development of fluorine-containing MOFs for their selective adsorption/separation has the potential to play a key role in mitigating their release into the atmosphere. The introduction of fluorinated linkers in a MOF can be accomplished either via direct synthesis under solvothermal conditions or via post-synthetic modification through the reaction known as Solvent-Assisted Ligand Incorporation (SALI).29–32 Recent work of our group has exploited the latter approach for the construction of mixed-linker MOFs (MIXMOFs) decorated with thiazole-based organic groups for assorted applications, spanning from CO2 capture to luminescence sensing of wastewater contaminants.33–36 Following this research interest and with the fluorinated gas adsorption/separation application in mind, we have prepared two new F-containing monocarboxylic thiazolium salts (HPhTzF)Br and (HPhTzCF3)Br (Scheme 1). Subsequently, we have decorated the 8-connected zirconium MOF [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2] (H4TCPB = 1,2,4,5-tetrakis(4-carboxyphenyl)benzene, Scheme 1, Zr_TCPB] with these salts using SALI. The as-obtained MIXMOFs after water elimination with minimal formulae [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br (Zr_TCPB_F) and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br (Zr_TCPB_CF3) have been fully characterized in the solid-state and exploited as functional materials for SF6 and CF4 adsorption.
000 times greater than CO2 on a per molecule basis.7–9 Small atmospheric concentrations of these two gases can have large effects on global temperatures. Thus, controlling and mitigating their release into the environment has become a critical objective for reducing the overall carbon footprint and achieving international climate goals, including those set out in the Paris Agreement.10 Metal–organic frameworks (MOFs), with their unique structural characteristics, have emerged as promising candidates for addressing this issue due to their exceptional surface area, tunable porosity, and chemical versatility.11–14 MOFs are typically composed of metal clusters linked by organic ligands, allowing for the precise design of pore structures that can selectively adsorb and separate specific gases. This adaptability makes MOFs a compelling solution for the selective capture of gases such as fluorinated compounds, which require highly specialized materials for effective separation. Given the strong C–F and S–F bonds in CF4 and SF6, their adsorption requires MOFs with tailored pore environments and strong host-guest interactions.15–18 MOFs containing open metal sites, fluorophilic functional groups, or optimized pore dimensions have demonstrated enhanced uptake of these gases. Additionally, post-synthetic modifications and functionalized ligands can further improve adsorption performance.19–25 In recent years, the incorporation of fluorine into the structure of MOFs has opened new avenues for enhancing their performance in gas separation applications. Fluorine, with its strong electronegativity, can contribute to the stabilization of metal centers and introduce additional functionalities to the material. Fluorinated MOFs in particular offer advantages, such as increased chemical stability and enhanced interactions with fluorinated gases. These unique properties enable these materials to act as highly efficient adsorbents, capable of selectively capturing and separating fluorinated compounds from complex gas mixtures.26–28 In the semiconductor industry, CF4 and SF6 mixtures are used in plasma etching processes for the fabrication of integrated circuits. These mixtures allow precise control of the etching, thanks to their chemical properties and the ability to form reactive plasmas. After use, exhaust gases may contain reactive or incompletely dissociated residues. Separating CF4 from SF6 allows the recovery and reuse of gases, the reduction of the load on scrubber systems and the improvement of process efficiency. SF6/CF4 mixtures are also exploited in gas-insulated switchgears in low-temperature environments, such as arctic regions. The addition of CF4 to SF6 prevents condensation of SF6 at temperatures below 233 K, maintaining insulating properties and ensuring reliable operation. During maintenance or emptying of the system, it is necessary to separate the two components to optimize recycling, to avoid contamination and to restore the original mixture in a controlled manner. Given the growing concerns over the environmental impact of fluorinated gases, the development of fluorine-containing MOFs for their selective adsorption/separation has the potential to play a key role in mitigating their release into the atmosphere. The introduction of fluorinated linkers in a MOF can be accomplished either via direct synthesis under solvothermal conditions or via post-synthetic modification through the reaction known as Solvent-Assisted Ligand Incorporation (SALI).29–32 Recent work of our group has exploited the latter approach for the construction of mixed-linker MOFs (MIXMOFs) decorated with thiazole-based organic groups for assorted applications, spanning from CO2 capture to luminescence sensing of wastewater contaminants.33–36 Following this research interest and with the fluorinated gas adsorption/separation application in mind, we have prepared two new F-containing monocarboxylic thiazolium salts (HPhTzF)Br and (HPhTzCF3)Br (Scheme 1). Subsequently, we have decorated the 8-connected zirconium MOF [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2] (H4TCPB = 1,2,4,5-tetrakis(4-carboxyphenyl)benzene, Scheme 1, Zr_TCPB] with these salts using SALI. The as-obtained MIXMOFs after water elimination with minimal formulae [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br (Zr_TCPB_F) and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br (Zr_TCPB_CF3) have been fully characterized in the solid-state and exploited as functional materials for SF6 and CF4 adsorption.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (9
DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_F was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 73% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzF)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_F, C81H53FNO32Zr6BrS (MW = 2230.6 g mol−1): C 43.6, H 2.4, N 0.6, S 1.4. Elemental analysis found (%): C, 43.8; H, 2.9; N, 0.7; S, 1.5. IR bands (KBr pellet, cm−1, Fig. S3): 1695(br), 1606(s), 1545(br), 1420(vs), 1183(w), 1146(w), 1108(w), 1018(w), 1004(w), 861(w), 782(m), 740(w), 716(m), 660(br), 582(w), 476(br).
1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_F was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 73% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzF)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_F, C81H53FNO32Zr6BrS (MW = 2230.6 g mol−1): C 43.6, H 2.4, N 0.6, S 1.4. Elemental analysis found (%): C, 43.8; H, 2.9; N, 0.7; S, 1.5. IR bands (KBr pellet, cm−1, Fig. S3): 1695(br), 1606(s), 1545(br), 1420(vs), 1183(w), 1146(w), 1108(w), 1018(w), 1004(w), 861(w), 782(m), 740(w), 716(m), 660(br), 582(w), 476(br).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (9
DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_CF3 was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 75% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzCF3)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_CF3, C82H53F3NO32Zr6BrS (MW = 2280.6 g mol−1): C 43.2, H 2.3, N 0.6, S 1.4. Elemental analysis found (%): C, 43.4; H, 2.6; N, 0.6; S, 1.7. IR bands (KBr pellet, cm−1, Fig. S4): 1701(br), 1602(s), 1543(br), 1414(vs), 1308(m), 1288(w), 1122(br), 1054(w), 1020(w), 861(w), 782(m), 714(m), 662(br), 584(br), 478(br).
1). Zr_TCPB [Zr6O4(OH)6(H2O)2(HCOO)2(TCPB)2, FW = 1948.47, 0.01 g, 0.05 mmol] was added and dispersed in the previous solution under stirring. The mixture was heated at 353 K for 24 h. After cooling, the microcrystalline off-white powder of Zr_TCPB_CF3 was collected, washed with hot MeCN (3 × 10 mL) and acetone (3 × 10 mL) and finally dried under a nitrogen stream at room temperature. Yield: 75% (based on the minimal formula [Zr6O4(OH)5(H2O)(PhTzCF3)(HCOO)2(TCPB)2]Br). Elemental analysis calcd. (%) for Zr_TCPB_CF3, C82H53F3NO32Zr6BrS (MW = 2280.6 g mol−1): C 43.2, H 2.3, N 0.6, S 1.4. Elemental analysis found (%): C, 43.4; H, 2.6; N, 0.6; S, 1.7. IR bands (KBr pellet, cm−1, Fig. S4): 1701(br), 1602(s), 1543(br), 1414(vs), 1308(m), 1288(w), 1122(br), 1054(w), 1020(w), 861(w), 782(m), 714(m), 662(br), 584(br), 478(br).
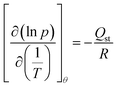

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50] for the [SF6
50] for the [SF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CF4] pair and [5
CF4] pair and [5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95] for the [SF6
95] for the [SF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2] and [CF4
N2] and [CF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2] pairs (the latter chosen to have compositions closer to the real atmospheric relative abundances). Various models (Quadratic, BET, Henry) were employed for the SF6, CF4 and N2 isotherms fitting, choosing the option that gives the lowest root-mean-square error (RMSE) on the experimental points (see Table S3 for the list of the fitting parameters). For a detailed explanation of these models and the related parameters, see the pyIAST webpage and documentation.
N2] pairs (the latter chosen to have compositions closer to the real atmospheric relative abundances). Various models (Quadratic, BET, Henry) were employed for the SF6, CF4 and N2 isotherms fitting, choosing the option that gives the lowest root-mean-square error (RMSE) on the experimental points (see Table S3 for the list of the fitting parameters). For a detailed explanation of these models and the related parameters, see the pyIAST webpage and documentation.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50,51
50,51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exchange–correlation functional combined with a double-zeta quality basis set for solid-state systems, and augmented with the D3
exchange–correlation functional combined with a double-zeta quality basis set for solid-state systems, and augmented with the D3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 52 and gCP53 corrections to account for van der Waals interactions and to remove the basis set superposition error (BSSE), respectively. For the numerical integration of the exchange-correlation term, 75 radial points and 974 angular points (XLGRID) in a Lebedev scheme were adopted. The SCF convergence was set to 10−7 during geometry optimization. The Pack–Monkhorst/Gilat shrinking factors for the diagonalization of the Kohn–Sham matrix in reciprocal space were set to 2. The truncation criteria (TOLINTEG) for the bielectronic integrals (Coulomb and exchange series) were set to 7 7 7 7 25. The JMol54 software was used to plot the electrostatic potential (ESP) maps.
52 and gCP53 corrections to account for van der Waals interactions and to remove the basis set superposition error (BSSE), respectively. For the numerical integration of the exchange-correlation term, 75 radial points and 974 angular points (XLGRID) in a Lebedev scheme were adopted. The SCF convergence was set to 10−7 during geometry optimization. The Pack–Monkhorst/Gilat shrinking factors for the diagonalization of the Kohn–Sham matrix in reciprocal space were set to 2. The truncation criteria (TOLINTEG) for the bielectronic integrals (Coulomb and exchange series) were set to 7 7 7 7 25. The JMol54 software was used to plot the electrostatic potential (ESP) maps.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 55 and 2019,37 respectively. It is a porous framework with rhombic channels of approximately 5.3 × 10.5 Å, suitable for hosting additional linkers of small–medium size. With this in mind and following the same approach used in the past for the synthesis of MOF linkers through N-quaternization of thiazole rings,35,36 we designed two new monocarboxylic fluorinated linkers using the F-containing molecules 4-(bromomethyl)-3-fluorobenzoic acid and 4-(bromomethyl)-3-(trifluoromethyl)benzoic acid (see the SI). The molecular size of [(HPhTzF)Br] (8.8 Å) and [(HPhTzCF3)Br] (9.0 Å) estimated from their crystal structures perfectly fits with the channel dimensions of Zr_TCPB. Thus, the MIXMOFs Zr_TCPB_F and Zr_TCPB_CF3 preparation was attempted through a post-synthetic SALI functionalization, i.e. a condensation reaction between the –COOH group of the thiazolium salts and the OH/H2O groups on the defects of the 8-coordinated [Zr6] metallic nodes. The pristine MOF was suspended in an anhydrous MeCN/DMSO mixture containing the dissolved salts and kept at T = 353 K for 24 h under gentle stirring. Subsequent filtration and washing with hot MeCN/acetone led to the successful obtainement of the pure products. Zr_TCPB_F and Zr_TCPB_CF3 have been thoroughly characterized in the solid state. The IR spectroscopic analysis confirms the extra-ligand insertion (Fig. S3 and S4), highlighting some typical bands of (HPhTzF)Br/(HPhTzCF3)Br at 1695/1701 cm−1 [ν(COO)], 1545/1543 cm−1 [ν(C
55 and 2019,37 respectively. It is a porous framework with rhombic channels of approximately 5.3 × 10.5 Å, suitable for hosting additional linkers of small–medium size. With this in mind and following the same approach used in the past for the synthesis of MOF linkers through N-quaternization of thiazole rings,35,36 we designed two new monocarboxylic fluorinated linkers using the F-containing molecules 4-(bromomethyl)-3-fluorobenzoic acid and 4-(bromomethyl)-3-(trifluoromethyl)benzoic acid (see the SI). The molecular size of [(HPhTzF)Br] (8.8 Å) and [(HPhTzCF3)Br] (9.0 Å) estimated from their crystal structures perfectly fits with the channel dimensions of Zr_TCPB. Thus, the MIXMOFs Zr_TCPB_F and Zr_TCPB_CF3 preparation was attempted through a post-synthetic SALI functionalization, i.e. a condensation reaction between the –COOH group of the thiazolium salts and the OH/H2O groups on the defects of the 8-coordinated [Zr6] metallic nodes. The pristine MOF was suspended in an anhydrous MeCN/DMSO mixture containing the dissolved salts and kept at T = 353 K for 24 h under gentle stirring. Subsequent filtration and washing with hot MeCN/acetone led to the successful obtainement of the pure products. Zr_TCPB_F and Zr_TCPB_CF3 have been thoroughly characterized in the solid state. The IR spectroscopic analysis confirms the extra-ligand insertion (Fig. S3 and S4), highlighting some typical bands of (HPhTzF)Br/(HPhTzCF3)Br at 1695/1701 cm−1 [ν(COO)], 1545/1543 cm−1 [ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)], 1420/1414 cm−1 [δ(CH/CH2)], 1183/1308 cm−1 [ν(Ar–F)/ν(CF3)] and 861/860 [γ(C–H)] respectively, besides those of TCPB4− at 1605/1601 cm−1 [ν(COO)]. The XRF qualitative analysis (Fig. S5 and S6) highlighted the presence of sulphur and bromine, confirming the successful SALI functionalization and revealing that the (HPhTzF)Br and (HPhTzCF3)Br ligands are incorporated within the MOF in their deprotonated form; the bromide anion is then necessary to balance the overall framework charge. Powder X-ray diffraction (PXRD, Fig. 1a) confirms that the parent crystallographic symmetry (orthorhombic, space group Cmmm)37 and network structural motif remain unaltered after functionalization. Differences in diffraction peaks relative intensities are due to changes in the electron density distribution introduced by the thiazolium salts within the unit cell. The extra-ligand is very likely to replace one OH/H2O couple on the metal node and not a bridging formate, given the favourite entropic factor associated to this coordination mode and the higher acidity of formic acid (pKa = 3.75) compared to benzoic acid (pKa = 4.19) that prevents a formate/benzoate exchange. This hypothesis is also supported by the presence of formic acid (m/z = 45 a.m.u.) in the mass spectra of the volatile byproducts forming during the MOFs thermal decomposition (vide infra). Therefore, this model structure has also been taken as starting guess for the DFT analysis (§3.4). To further support our structural hypothesis, a comparison between the experimental PXRD pattern and a “computational” pattern derived from the DFT-optimized model structures of Zr_TCPB_F and Zr_TCPB_CF3 has been made (Fig. S7 and S8). The matching between experimental and theoretical pattern is quite satisfactory. In the latter, there are more peaks coming from the lower crystal symmetry of the DFT model in comparison with that of the real crystal, because in the model there is only a finite portion of the lattice. This symmetry decrease generates additional peaks that are absent in the real sample. Since both (HPhTzF)Br and (HPhTzCF3)Br are unstable under strongly acidic or basic conditions (but –F/–CF3 signals remain detectable), linker quantification was carried out through combined 1H and 19F NMR spectroscopy of the digested solid in acidic solution (D2SO4). See the SI and Fig. S9–S12 for details. 19F signals are integrated against the 1H NMR signals of the H4TCPB ligand using 2,6-difluorobenzoic acid an internal standard.56 The multinuclear NMR analysis accounts for one thiazolium salt molecule per minimal formula, in line with the estimation coming from the S-elemental analysis (Experimental section). Consequently, based on the ligands relative stoichiometric ratio the MOF minimal formulae can be written as [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br. Thermogravimetric analysis (TGA, Fig. S13a and S14a) showed that the thermal stability of both MIXMOFs is lower than that of parent Zr_TCPB (Tdec = 773/798 vs. 820 K for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively). An initial weight loss of ca. 18–20 wt% (in line with the stoichiometric 1
C)], 1420/1414 cm−1 [δ(CH/CH2)], 1183/1308 cm−1 [ν(Ar–F)/ν(CF3)] and 861/860 [γ(C–H)] respectively, besides those of TCPB4− at 1605/1601 cm−1 [ν(COO)]. The XRF qualitative analysis (Fig. S5 and S6) highlighted the presence of sulphur and bromine, confirming the successful SALI functionalization and revealing that the (HPhTzF)Br and (HPhTzCF3)Br ligands are incorporated within the MOF in their deprotonated form; the bromide anion is then necessary to balance the overall framework charge. Powder X-ray diffraction (PXRD, Fig. 1a) confirms that the parent crystallographic symmetry (orthorhombic, space group Cmmm)37 and network structural motif remain unaltered after functionalization. Differences in diffraction peaks relative intensities are due to changes in the electron density distribution introduced by the thiazolium salts within the unit cell. The extra-ligand is very likely to replace one OH/H2O couple on the metal node and not a bridging formate, given the favourite entropic factor associated to this coordination mode and the higher acidity of formic acid (pKa = 3.75) compared to benzoic acid (pKa = 4.19) that prevents a formate/benzoate exchange. This hypothesis is also supported by the presence of formic acid (m/z = 45 a.m.u.) in the mass spectra of the volatile byproducts forming during the MOFs thermal decomposition (vide infra). Therefore, this model structure has also been taken as starting guess for the DFT analysis (§3.4). To further support our structural hypothesis, a comparison between the experimental PXRD pattern and a “computational” pattern derived from the DFT-optimized model structures of Zr_TCPB_F and Zr_TCPB_CF3 has been made (Fig. S7 and S8). The matching between experimental and theoretical pattern is quite satisfactory. In the latter, there are more peaks coming from the lower crystal symmetry of the DFT model in comparison with that of the real crystal, because in the model there is only a finite portion of the lattice. This symmetry decrease generates additional peaks that are absent in the real sample. Since both (HPhTzF)Br and (HPhTzCF3)Br are unstable under strongly acidic or basic conditions (but –F/–CF3 signals remain detectable), linker quantification was carried out through combined 1H and 19F NMR spectroscopy of the digested solid in acidic solution (D2SO4). See the SI and Fig. S9–S12 for details. 19F signals are integrated against the 1H NMR signals of the H4TCPB ligand using 2,6-difluorobenzoic acid an internal standard.56 The multinuclear NMR analysis accounts for one thiazolium salt molecule per minimal formula, in line with the estimation coming from the S-elemental analysis (Experimental section). Consequently, based on the ligands relative stoichiometric ratio the MOF minimal formulae can be written as [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzF)]Br and [Zr6O4(OH)5(H2O)(HCOO)2(TCPB)2(PhTzCF3)]Br. Thermogravimetric analysis (TGA, Fig. S13a and S14a) showed that the thermal stability of both MIXMOFs is lower than that of parent Zr_TCPB (Tdec = 773/798 vs. 820 K for Zr_TCPB_F/Zr_TCPB_CF3 vs. Zr_TCPB, respectively). An initial weight loss of ca. 18–20 wt% (in line with the stoichiometric 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 [Zr6]
1 [Zr6]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) thiazolium salt ratio) can be reasonably ascribed to loss of the salt and formic acid. Indeed, the DTG peaks found in this range fall at T = 429 K (salt) and T = 543 K (HCOOH). The former temperature value is close to that found for the decomposition of the isolated (HPhTzF)Br and (HPhTzCF3)Br, occurring at T = 495 and 530 K, respectively (Fig. S15). Further proof of evidence is provided by the MS analysis of the volatiles (Fig. S13b and S14b), where two peaks at m/z = 85 and 46 a.m.u. typical of thiazole and formic acid appear in the same temperature range. MOF decomposition is witnessed by the presence in the MS spectra of the volatiles of the peak at m/z = 78 a.m.u. typical of phenyl rings coming from TCPB.
thiazolium salt ratio) can be reasonably ascribed to loss of the salt and formic acid. Indeed, the DTG peaks found in this range fall at T = 429 K (salt) and T = 543 K (HCOOH). The former temperature value is close to that found for the decomposition of the isolated (HPhTzF)Br and (HPhTzCF3)Br, occurring at T = 495 and 530 K, respectively (Fig. S15). Further proof of evidence is provided by the MS analysis of the volatiles (Fig. S13b and S14b), where two peaks at m/z = 85 and 46 a.m.u. typical of thiazole and formic acid appear in the same temperature range. MOF decomposition is witnessed by the presence in the MS spectra of the volatiles of the peak at m/z = 78 a.m.u. typical of phenyl rings coming from TCPB.








