 Open Access Article
Open Access ArticleElucidating the structure–performance relationship in single-particle NCM cathodes via controlled precursor synthesis
Soonhyun Hong†
a,
Heesang Lee† a,
Jahun Kooa,
Wonchan Hwangb,
Ji Hwan Kimbcd,
Yung-Eun Sung
a,
Jahun Kooa,
Wonchan Hwangb,
Ji Hwan Kimbcd,
Yung-Eun Sung cd,
Jungjin Park
cd,
Jungjin Park *b,
Young-Sang Yu*ef and
Chunjoong Kim*a
*b,
Young-Sang Yu*ef and
Chunjoong Kim*a
aDepartment of Materials Science and Engineering, Chungnam National University, 99 Daehak-ro, Yuseong-gu, Daejeon 34134, Republic of Korea. E-mail: ckim0218@cnu.ac.kr
bEnergy Storage Research Center, Clean Energy Research Division, Korea Institute of Science and Technology (KIST), 5 Hwarang-ro, Seongbuk-gu, Seoul 02792, Republic of Korea. E-mail: jpark716@kist.re.kr
cSchool of Chemical and Biological Engineering, Seoul National University, Seoul 08826, Republic of Korea
dCenter for Nanoparticle Research, Institute for Basic Science (IBS), Seoul 08826, Republic of Korea
eDepartment of Physics, Chungbuk National University, 1 Chungdae-ro, Seowon-gu, Cheongju 28644, Republic of Korea. E-mail: youngsang@chungbuk.ac.kr
fAdvanced Energy Research Institute, Chungbuk National University, 1 Chungdae-ro, Seowon-gu, Cheongju 28644, Republic of Korea
First published on 13th March 2026
Abstract
While high-energy-density Ni-rich layered oxides, LiNi1−x−yCoxMnyO2 (Ni > 0.8), are of significant attention as next-generation cathode materials, their practical application is limited by intergranular cracking and mechanical degradation arising from anisotropic lattice strain. The single-particle cathode strategy has emerged as a promising solution, effectively suppressing intergranular cracking by eliminating boundaries between primary particles. However, the fundamental relationships between single-particle size, internal microstructure, and electrochemical performance remain poorly understood. Here, we present a molten-salt synthesis strategy to produce LiNi0.92Co0.03Mn0.05O2 single-particle cathodes with tunable particle and crystallite sizes via precursor morphology control. Systematic analysis reveals clear correlations between precursor shape, final particle microstructure, and rate capability. This work aims to establish the causal link between precursor morphology, final particle microstructure, and electrochemical performance, thereby providing a core design principle for developing high-stability and -power single-particle cathode materials.
1. Introduction
Recently, the blueprint for net-zero carbon emissions has been announced as part of a global effort against climate change.1 Based on the comprehensive carbon emission reduction plan, electric vehicles (EVs) are projected to occupy approximately 77% of the global automobile market by 2030.2 Accordingly, the demand for high-performance lithium-ion batteries (LIBs) that can power EVs is rapidly increasing. The most promising candidate is still the layered oxide with a high nickel concentration (Ni content >80 at% in the composition of the transition metals), Ni-rich LiNi1−x−yCoxMnyO2 (Ni-rich NCM), which is derived from the first commercialized cathode material, LiCoO2.3,4 Despite its high theoretical capacity (>200 mA h g−1), the Ni-rich NCM cathode material exhibits intrinsic limitations, including fast capacity fade and safety concerns.5,6 The origin of capacity fading in commonly used Ni-rich NCM aggregates is the significant volume change of primary particles, which gradually leads to intergranular cracking during repeated cycling.7,8 The formation and propagation of cracks deteriorate mechanical integrity, ultimately resulting in the loss of electrical contacts.9 In addition, the cracks keep exposing a fresh surface or interface to the electrolyte, followed by the formation of electrochemically inactive phases.10,11 Therefore, the emergence of such inactive phases leads to continuous capacity decay as well as sluggish Li+ transport.12,13 Therefore, the emergence of such inactive phases leads to continuous capacity decay as well as sluggish Li+ transport. Such surface accompanied by oxygen loss driven cation migration, forming surface-densified Ni-rich phases that increase interfacial resistance and accelerate performance decay.14Among various strategies such as transition metal (TM) gradient,15 doping,16 and surface coating,17 Ni-rich single-particle NCM (hereafter, SP-NCM) materials stand out as a promising solution. The SP-NCM reveals great potential to enhance mechanical integrity and cycle performance by minimizing particle cracking, gas evolution, and electrolyte reactivity, despite certain handling issues during the manufacturing process.18–20 Furthermore, the Ni-rich SP-NCM cathode materials are readily applicable to all-solid-state batteries due to their curtailed interfacial contact with the electrolyte.21 However, deliberate control over particle size and morphology in SP-NCM systems remains challenging, since most synthetic routes rely on complex multi-step processes and high-temperature calcination steps.22–24
Herein, we firstly propose a rational synthesis protocol that combines coprecipitation of transition-metal precursors with a subsequent molten-salt reaction to synthesize SP-NCM with tunable particle and domain sizes. During the coprecipitation reaction, transition-metal hydroxide, TM(OH)2 (TM = Ni, Co, Mn), was prepared, with the thickness of primary particles manipulated through fine control of the reaction variables.25–27 The following molten-salt synthesis renders the calcination and sintering of SP-NCM with various particles and domain sizes from the TM hydroxide precursor.28 Systematic analyses confirmed a strong correlation between the precursor morphology and the resulting single-particle size, as well as a clear dependence of electrochemical performance on both particle and domain sizes. We believe that our work, therefore, provides a fundamental understanding of the structure–performance relationship in SP-NCMs and establishes a generalizable synthesis framework for designing high-performance single-particle cathode materials.
2. Experimental section
2.1. Synthesis of Ni-rich precursor
Ni0.92Co0.03Mn0.05(OH)2 precursors were synthesized using the coprecipitation method. A 1.5 M aqueous solution with a molar ratio of Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co
Co![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mn = 0.92
Mn = 0.92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.03
0.03![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05 was prepared using NiSO4·6H2O, CoSO4·7H2O, and MnSO4·H2O, respectively. The mixed solution was pumped into a 5 L continuous stirred-tank reactor at a rate of 0.12 mol h−1 under a nitrogen atmosphere. Concurrently, aqueous solutions of 4.0 M NaOH and 14.8 M NH3·H2O were introduced to regulate pH (i.e., molar concentration of hydroxyl anions) and concentration of chelating agent, respectively. The pH values of the solution were controlled from 11.2 to 12.1 by adjusting the NaOH feed rate, while the ammonia concentration in the reactor was varied from 0.5 M to 5.0 M. The reaction temperature was maintained at ∼48 °C using a hot water circulation system, and the stirrer was operated at 800 rpm. The reaction was allowed to proceed for 20 hours, yielding spherical Ni0.92Co0.03Mn0.05(OH)2 precursor. The resulting precipitates were filtered using a vacuum filter system and washed with a slightly lower pH NaOH solution and distilled water to remove residual sodium ions. The filtered precipitate was then dried at 80 °C in the convection oven overnight.
0.05 was prepared using NiSO4·6H2O, CoSO4·7H2O, and MnSO4·H2O, respectively. The mixed solution was pumped into a 5 L continuous stirred-tank reactor at a rate of 0.12 mol h−1 under a nitrogen atmosphere. Concurrently, aqueous solutions of 4.0 M NaOH and 14.8 M NH3·H2O were introduced to regulate pH (i.e., molar concentration of hydroxyl anions) and concentration of chelating agent, respectively. The pH values of the solution were controlled from 11.2 to 12.1 by adjusting the NaOH feed rate, while the ammonia concentration in the reactor was varied from 0.5 M to 5.0 M. The reaction temperature was maintained at ∼48 °C using a hot water circulation system, and the stirrer was operated at 800 rpm. The reaction was allowed to proceed for 20 hours, yielding spherical Ni0.92Co0.03Mn0.05(OH)2 precursor. The resulting precipitates were filtered using a vacuum filter system and washed with a slightly lower pH NaOH solution and distilled water to remove residual sodium ions. The filtered precipitate was then dried at 80 °C in the convection oven overnight.
2.2. Synthesis of Ni-rich NCM
For the synthesis of single-particle LiNi0.92Co0.03Mn0.05O2 (SP-NCM), three representative Ni0.92Co0.03Mn0.05(OH)2 precursors with distinct morphologies were selected. The corresponding coprecipitation conditions were as follows: 1.0 M NH3 at a pH of 11.2, 2.0 M NH3 at a pH of 12.1, and 5.0 M NH3 at a pH of 12.1. For the following molten-salt reaction, the precursor, LiOH·H2O, and Li2SO4·H2O were thoroughly mixed with a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25, respectively. The homogenous mixture was then held at 850 °C for 10 hours under an oxygen atmosphere and slowly cooled to 250 °C. The ramping and cooling rates were set to be 10 °C min−1 and 2 °C min−1, respectively. Then, the material was allowed to cool naturally from 250 °C to room temperature (RT). The resulting composite was grounded, washed with distilled water, and centrifuged several times (for 5 min at 5000 rpm each time) to remove residual Li salts. The washed material was dried at 80 °C overnight, heated at 700 °C for 6 hours in oxygen to remove residual water, and then ground once again. The final products are designated as SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, corresponding to precursor synthesis conditions of 1.0 M NH3 at a pH 11.2, 2.0 M NH3 at a pH 12.1, and 5.0 M NH3 at a pH 12.1 respectively.
0.25, respectively. The homogenous mixture was then held at 850 °C for 10 hours under an oxygen atmosphere and slowly cooled to 250 °C. The ramping and cooling rates were set to be 10 °C min−1 and 2 °C min−1, respectively. Then, the material was allowed to cool naturally from 250 °C to room temperature (RT). The resulting composite was grounded, washed with distilled water, and centrifuged several times (for 5 min at 5000 rpm each time) to remove residual Li salts. The washed material was dried at 80 °C overnight, heated at 700 °C for 6 hours in oxygen to remove residual water, and then ground once again. The final products are designated as SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, corresponding to precursor synthesis conditions of 1.0 M NH3 at a pH 11.2, 2.0 M NH3 at a pH 12.1, and 5.0 M NH3 at a pH 12.1 respectively.
2.3. Structural characterizations
The crystallographic structures of the samples were analyzed using X-ray diffractometer (XRD, D8 ADVANCE, Bruker), operated at 40 kV and 40 mA with Cu Kα radiation (λavg = 1.5406 Å). The XRD peaks were measured over a range of 10° to 70° at a scan rate of 2° min−1 and a scan step of 0.02°. The surface morphology of the prepared samples was examined using scanning electron microscopy (SEM, CLARA, TESCAN) operated at an acceleration voltage of 10 kV. To evaluate the primary particle thickness of the precursors, more than eighty individual primary particles within each aggregated precursor were measured. Likewise, the particle sizes of the SP-NCM samples were estimated from major and minor axes of over fifty individual particles, obtained from SEM images and analyzed using ImageJ software.292.4. Measurement of the electrochemical performances
The 80 µm-thick working electrode was prepared on 20 µm-thick aluminum foil by casting a slurry, which consists of 80% active material, 10% binder (polyvinylidene fluoride, PVDF), and 10% carbon black (DENKA BLACK) in NMP (N-methyl-2-pyrrolidone) solvent. The electrode was dried under a vacuum at 120 °C overnight to evaporate the solvent and then calendared to 50% of the active material thickness. The cathode mass loading was controlled to approximately 3 mg cm−2 for most electrodes. Circular electrodes with a 14 mm diameter were punched and accurately weighed. CR2032 coin-type half-cells were assembled using the prepared working electrode as the cathode and a Li metal foil as the anode. Coin cells were assembled using 100 µl of electrolyte and a 25 µm-thick PE monolayer microporous separator. The electrolyte consisted of 1 M LiPF6 dissolved in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC), and ethyl–methyl carbonate (EMC) (volume ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, Donghwa electrolyte). All cell assembly was performed in a high-purity argon-filled glovebox with O2 and moisture levels below 0.1 ppm. Electrochemical tests were conducted using a WonATech WBCS 3000L system. Galvanostatic charge–discharge cycling was performed with a potential range of 2.7–4.3 V (vs. Li+/Li). The first two cycles were carried out at 0.1C (assuming 1C = 200 mA g−1) at room temperature, and followed by cycling at 1C for additional electrochemical tests. For the long-term cycling stability test at 1C, discharge was conducted at 1C, whereas charge was carried out using a constant–current/constant–voltage (CC/CV) protocol (CC at 1C to 4.3 V followed by a CV hold at 4.3 V until the current decreased to 0.05C). For the rate capability test, both charge and discharge were performed under the CC mode at each current density within 2.7–4.3 V. Rate capability was evaluated by varying current from 20 mA g−1 to 2000 mA g−1 within the same potential window, recording five electrochemical measurements at each current rate. To further examine Li+ diffusion kinetics, the galvanostatic intermittent titration technique (GITT) was performed over 2.7–4.3 V during five cycles.
1, Donghwa electrolyte). All cell assembly was performed in a high-purity argon-filled glovebox with O2 and moisture levels below 0.1 ppm. Electrochemical tests were conducted using a WonATech WBCS 3000L system. Galvanostatic charge–discharge cycling was performed with a potential range of 2.7–4.3 V (vs. Li+/Li). The first two cycles were carried out at 0.1C (assuming 1C = 200 mA g−1) at room temperature, and followed by cycling at 1C for additional electrochemical tests. For the long-term cycling stability test at 1C, discharge was conducted at 1C, whereas charge was carried out using a constant–current/constant–voltage (CC/CV) protocol (CC at 1C to 4.3 V followed by a CV hold at 4.3 V until the current decreased to 0.05C). For the rate capability test, both charge and discharge were performed under the CC mode at each current density within 2.7–4.3 V. Rate capability was evaluated by varying current from 20 mA g−1 to 2000 mA g−1 within the same potential window, recording five electrochemical measurements at each current rate. To further examine Li+ diffusion kinetics, the galvanostatic intermittent titration technique (GITT) was performed over 2.7–4.3 V during five cycles.
3. Results and discussion
The morphological evolution of Ni0.92Co0.03Mn0.05(OH)2 was systematically explored by adjusting concentrations of OH− and chelating agent (CA), NH3 (see the Experimental section for methodological details). Since the concentrations of the hydroxyl anion and the CA are main factors governing the formation of atomically homogeneous precursors,30 both pH and NH3 concentrations during the coprecipitation reaction were carefully manipulated to ensure desirable morphologies with homogeneous composition of Ni, Co, and Mn ions in Ni0.92Co0.03Mn0.05(OH)2 without formation of individual metal hydroxide phases such as Ni(OH)2, Co(OH)2, and Mn(OH)2.31 The addition of ammonia retards the precipitation reaction between TM cations and hydroxyl anions. As a result, TM ions undergo two competitive reactions: (1) coprecipitation with hydroxyl anions to form TM(OH)2, and (2) complexation with NH3 to yield soluble TM(NH3)n2+ species (where n ranges from 4 to 8). The delicate balance between these two reactions determines the nucleation and growth dynamics of the precursors, ultimately governing their morphology and compositional uniformity.Firstly, the NH3 concentration was systematically varied (0.5 M, 1.0 M, 1.5 M, 2.0 M, 2.5 M, and 5.0 M) during the coprecipitation reaction while maintaining a constant pH of 12.1. In addition to participating in the complexation reaction with TM cations, NH3 strongly coordinates to TM cations, particularly on the (001) planes of TM(OH)2.32 Consequently, increasing the NH3 concentration impedes vertical crystal growth along the c-axis, favoring lateral expansion within the ab plane. Therefore, plate-like primary particles predominantly form, as shown in Fig. 1a. With increasing CA concentration from 0.5 M to 2.0 M, the primary particles become progressively thinner, indicating the enhanced formation of plate-like morphology. However, at concentrations above 2.0 M, the TM(OH)2 plates tend to be agglomerated to minimize the surface energy due to the thermodynamic instability of excessively thin plate-like primary particles.33
Next, the effect of OH− concentration on precursor morphology was investigated by varying pH values (11.2, 11.5, 11.8, and 12.1) during the coprecipitation reaction under the constant ammonia concentration (1.0 M). As the pH increased from 11.2 to 12.1, the primary particles became thicker along the c-axis, which can be attributed to the enhanced reactivity of hydroxyl ions. Under higher OH− concentration, the coprecipitation of TM(OH)2 is favored over the complexation reaction between TM cations and NH3, leading to the suppression of (001)-plane coordination by NH3 on the TM(OH)2 surface (Fig. 1b). A summary of the primary particle thickness as a function of NH3 and OH− concentrations is presented in Table 1 and Fig. 1c. The results clearly indicate that both hydroxyl anion and NH3 concentrations significantly influence the precursor morphology. Thicknesses of primary particles were varied by nearly two orders of magnitude, ranging from approximately 10.4 nm to 1.0 µm, depending on the pH and CA concentrations. The thinnest and thickest primary particles could be obtained from the precursors synthesized at 1.0 M NH3 at a pH 11.2 and 5.0 M NH3 at a pH 12.1, respectively. It should be noted that thicker primary particles exhibited stronger aggregation tendency due to their lower anisotropy in the three-dimensional space, resulting in a noticeable difference in the bulk density of the aggregated precursors, as summarized in Fig. S1.
| NH3 conc.b [M] | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 | 5.0 |
| Ave. thick. [nm] | 264.0 | 217.7 | 156.4 | 117.8 | 170.4 | 1010.4 |
Fig. 2a and b present the powder X-ray diffraction (XRD) patterns of the Ni-rich Ni0.92Co0.03Mn0.05(OH)2 precursors prepared with various CA and hydroxyl concentrations, respectively. All samples exhibit an identical crystal structure, the layered double hydroxide structure with β-Ni(OH)2, which belongs to the space group P3m1.34 The prominent peaks at 2θ of ∼19.15°, ∼33.07°, ∼38.44°, and ∼51.96° are indexed to the (001), (100), (101), and (102) planes, respectively.35 The relative crystal growth between the (hk0) basal plane and (00l) edge plane can be gauged by the intensity ratios of R(100)/(001), where Rx/y represents the ratio between the peak intensities of the x and y planes. Since the (100) diffraction is intrinsically weak due to the low atomic density of transition metals in that plane, R(101)/(001) and R(102)/(001) are also calculated to provide more confident indicators to elucidate the preferential growth of hydroxides.36 As depicted in Fig. 2c, increasing the pH led to a systematic decrease in R(100)/(001), R(101)/(001), and R(102)/(001), indicating more preferential growth (or stacking) along c-axis over the planar growth along the ab plane. Meanwhile, planar growth along the ab plane is most dominant from the precursors synthesized at the 2.0 M NH3 concentration, as evidenced by the highest R(100)/(001), R(101)/(001), and R(102)/(001). The inverse trend in diffraction intensity ratios with increasing pH is highly consistent with the microscopic observations of the primary particles (Fig. 1a). A detailed growth mechanism of the Ni0.92Co0.03Mn0.05(OH)2 precursors is beyond the scope of this work and will be addressed in detail in a future study.
Among the Ni0.92Co0.03Mn0.05(OH)2 precursors, three representative precursors exhibiting distinct primary-particle morphology but comparable aggregate sizes (precursors prepared under 1.0 M NH3 with a pH of 11.2, 2.0 M NH3 with a pH of 12.1, and 5.0 M NH3 with a pH of 12.1) were selected to be calcined with LiOH·H2O to produce single-particle LiNi0.92Co0.03Mn0.05O2 (SP-NCM) as described in the Experimental section. As shown in Table S1, ICP-(AES) confirms that all representative Ni-rich precursors maintain the intended transition-metal stoichiometry (target composition: Ni 0.92, Co 0.03, Mn 0.05). The measured TM fractions show only minor deviations from the target, indicating that varying the synthesis conditions does not induce composition drift and that incomplete complexation is unlikely. N2 adsorption (BET) measurements were conducted for the same precursors, and the specific surface area and total pore volume are summarized in Table S2. These data are provided to support the discussion on surface-related electrochemical behavior.
As depicted in Fig. 3, the average particle sizes of the resulting SP-NCMs were 1.169 (mean) ± 0.394 (standard deviation) µm, 1.668 ± 0.515 µm, and 4.485 ± 1.052 µm, corresponding to SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively. The precursor, with its more porous structure composed of thinner primary particles, led to the formation of the smaller SP-NCM particles after the molten-salt reaction, owing to its lower bulk density and higher reactivity during calcination.
In general, the exposed crystal facets of SP-NCMs are highly influenced by the oxygen solubility during the calcination procedure.37,38 Although all samples were subjected to identical environments during the molten-salt reaction, well-defined truncated octahedron crystal facets were revealed only on the surface of SP-2.0/12.1. In contrast, SP-1.0/11.2 and SP-5.0/12.1 exhibited relatively smooth, featureless surface without prominent crystalline faceting. The surfaces of SP-2.0/12.1 are enclosed by (001), (012), and (104) facets, as predicted by experimental observation and density-functional theory (DFT) calculations (Fig. S2).39,40 The dominantly exposed crystal facets play a crucial role in determining the electrochemical stability, oxygen release, thermal decomposition, crystallographic reconstructions from layered to spinel or rock-salt, metal segregation, etc.41,42 Among them, the (104) family facet is known to be most stable during electrochemical operations, whereas the (012) surface tends to be more reactive than the (001) surface, contributing to a higher initial discharge capacity but lower cycling stability.43 In the case of SP-2.0/12.1, the predominant exposure of the (012) and (001) facets likely governs its distinctive electrochemical characteristics.
Fig. 3b presents the XRD patterns of SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1. All samples revealed an identical hexagonal α-NaFeO2 type structure with the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group without any other notable impurity phases, confirming the successful formation of the layered structure and the reliability of the synthesis process. Additionally, Rietveld refinement was performed to quantify the lattice parameters and the degree of Li/Ni cation mixing for the three samples (Fig. S3). The refined lattice parameters (a, c, and V) are nearly identical across the samples, whereas the Li/Ni cation mixing is ∼4% with a small but measurable variation of approximately ±0.5%. The overall domain sizes of the SP-NCM cathodes were estimated from the (003) peaks using the Scherrer equations, yielding coherent-crystalline sizes of 278.9, 240.5, and 226.3 nm for SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively (Fig. 3c). Interestingly, an inverse correlation between particle and Scherrer-based coherent domain size (003) sizes was observed; SP-1.0/11.2 exhibits the largest domain size despite having the smallest particle size. This phenomenon can be attributed to the relatively higher degree of recrystallization, promoted by the more porous precursor structure at the aggregate level. Conversely, the densely aggregated precursor, composed of thicker primary particles, produced the largest SP-NCM particle (SP-5.0/12.1) but exhibited the smallest domain size due to restricted recrystallization during the high-temperature molten-salt process. The inverse relationship between particle and domain sizes is a unique feature of the single-particle NCMs synthesized via our molten-salt route. To directly verify the internal domain structure corresponding to the XRD-based grain/domain-size trend, cross-sectional EBSD analysis was additionally conducted, confirming that SP-1.0/11.2 has a larger internal domain size while SP-2.0/12.1 and SP-5.0/12.1 show similarly smaller domains (Fig. S4 and S5). Furthermore, the intensity ratio between the (003) and (104) reflections, I(003)/I(104), was identified as a structural quality indicator, given its sensitivity to cation mixing between Li+ and M cations.44,45 The measured I(003)/I(104) ratios were 2.03, 3.43, and 2.18 in SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively. In general, the I(003)/I(104) value exceeding 2.0 indicate a well-ordered layered structure, confirming that all SP-NCM samples possess high crystallinity with minimal cation disorder. Notably, SP-2.0/12.1 exhibited the highest I(003)/I(104) ratio, suggesting the lowest degree of cation mixing. It should be noted, however, that structural morphology can also significantly affect the I(003)/I(104). The well-grown truncated octahedron with dominant (001) facets in SP-2.0/12.1 likely contributes to its enhanced (003) peak intensity and correspondingly higher I(003)/I(104).
m space group without any other notable impurity phases, confirming the successful formation of the layered structure and the reliability of the synthesis process. Additionally, Rietveld refinement was performed to quantify the lattice parameters and the degree of Li/Ni cation mixing for the three samples (Fig. S3). The refined lattice parameters (a, c, and V) are nearly identical across the samples, whereas the Li/Ni cation mixing is ∼4% with a small but measurable variation of approximately ±0.5%. The overall domain sizes of the SP-NCM cathodes were estimated from the (003) peaks using the Scherrer equations, yielding coherent-crystalline sizes of 278.9, 240.5, and 226.3 nm for SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively (Fig. 3c). Interestingly, an inverse correlation between particle and Scherrer-based coherent domain size (003) sizes was observed; SP-1.0/11.2 exhibits the largest domain size despite having the smallest particle size. This phenomenon can be attributed to the relatively higher degree of recrystallization, promoted by the more porous precursor structure at the aggregate level. Conversely, the densely aggregated precursor, composed of thicker primary particles, produced the largest SP-NCM particle (SP-5.0/12.1) but exhibited the smallest domain size due to restricted recrystallization during the high-temperature molten-salt process. The inverse relationship between particle and domain sizes is a unique feature of the single-particle NCMs synthesized via our molten-salt route. To directly verify the internal domain structure corresponding to the XRD-based grain/domain-size trend, cross-sectional EBSD analysis was additionally conducted, confirming that SP-1.0/11.2 has a larger internal domain size while SP-2.0/12.1 and SP-5.0/12.1 show similarly smaller domains (Fig. S4 and S5). Furthermore, the intensity ratio between the (003) and (104) reflections, I(003)/I(104), was identified as a structural quality indicator, given its sensitivity to cation mixing between Li+ and M cations.44,45 The measured I(003)/I(104) ratios were 2.03, 3.43, and 2.18 in SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively. In general, the I(003)/I(104) value exceeding 2.0 indicate a well-ordered layered structure, confirming that all SP-NCM samples possess high crystallinity with minimal cation disorder. Notably, SP-2.0/12.1 exhibited the highest I(003)/I(104) ratio, suggesting the lowest degree of cation mixing. It should be noted, however, that structural morphology can also significantly affect the I(003)/I(104). The well-grown truncated octahedron with dominant (001) facets in SP-2.0/12.1 likely contributes to its enhanced (003) peak intensity and correspondingly higher I(003)/I(104).
The electrochemical responses of the SP-NCM cathodes are investigated, as shown in Fig. 4. The smaller particles (SP-1.0/11.2 and SP-2.0/12.1) exhibited higher utilization of Li+ during the initial cycle at a rate of 0.1C (assuming 1C = 200 mA g−1). As shown in Fig. 4a, the initial discharge capacities of SP-1.0/11.2 and SP-2.0/12.1 were nearly the same, ∼192 mA h g−1, whereas SP-5.0/12.1 delivered a slightly lower initial discharge capacity of ∼184 mA h g−1. The initial cycle coulombic efficiencies (CEs) were 83.9%, 86.0%, and 82.7% for SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively. The highest quality of the layered structure in SP-2.0/12.1, as evidenced by the highest I(003)/(104) ratio, accounts for its highest CE. The cycling retention at a rate of 1C after two formation cycles showed that SP-1.0/11.2 and SP-2.0/12.1 maintained higher discharge capacities (∼179 mA h g−1) than SP-5.0/12.1 (∼165 mA h g−1), as shown in Fig. 4b. Again, SP-2.0/12.1 exhibited the highest CE among the samples even at the 1C rate, with initial CEs of 96.3%, 99.7%, and 96.3% for SP-1.0/11.2, SP-2.0/12.1, and SP-5.0/12.1, respectively. SP-2.0/12.1 also showed the highest capacity retention of ∼83.5% after 200 cycles, which is far more stable compared with commercial aggregate-level cathode structures. Its impressive cycling durability and rate performance are attributed to the high mechanical integrity of the single-particle structure, which effectively suppresses intergranular cracking. The remaining capacity fading is mainly caused by surface parasitic reaction and irreversible phase transformation during Li+ (de)intercalation, as reported in the literature.
As shown in Fig. 4d–f, the structural stability of the SP-NCMs during cycling was evaluated by analyzing the differential capacity curves (dQ/dV vs. V). The corresponding cyclic voltammetry (CV) profiles at different scan rates are provided in Fig. S6. All samples showed four characteristic redox peaks, corresponding to one second-order transition within the rhombohedral phase (H1) and three first-order transitions: H1 to monoclinic phase (M), M to hexagonal phase (H2), and H2 to the hexagonal phase (H3). For Ni-rich layered oxides (Ni > 0.9), the H2–H3 phase transition is often irreversible, owing to anisotropic lattice changes that induce mechanical strain within the particles, ultimately leading to a deterioration in cycling performances.46 Therefore, the loss of the peak corresponding to the H2 to H3 transition is directly related to the irreversible loss of capacity.47 As seen in Fig. 4d and f, the anodic peaks corresponding to the H2–H3 transition nearly disappear after 100 cycles for SP-1.0/11.2 and SP-5.0/12.1, indicating severe structural degradation. On the other hand, the H2–H3 phase transition is well preserved in SP-2.0/12.1, indicating the reversible Li (de)intercalation upon repeated cycling, which is related to the excellent cycle retention of SP-2.0/12.1.
Interestingly, contrary to the general expectation that a faster electrochemical response can be achieved in the smaller particles due to the larger reactive surface area to volume ratio and the shorter Li-ion diffusion path,48 smallest and largest SP-NCM particles showed the best rate performance at the low and high current density, respectively as shown in Fig. 4c. This discrepancy suggests that the rate-limiting mechanism for Li+ transport within SP-NCMs may vary with the applied current density. At lower cycling rates, the surface reaction kinetics likely dominate Li+ (de)intercalation behavior, as evidenced by the smaller particles. This hypothesis is supported by the fact that the smallest particles with a larger surface-to-volume ratio exhibited the highest capacity retention at those lower rates. Conversely, at higher current densities, bulk diffusion becomes the primary rate-determining process. In this regime, the smaller domain size within the largest particles (SP-5.0/12.1) provides shorter diffusion pathways and enhanced Li+ ionic conductivity within the bulk, resulting in a faster electrochemical response.
To further examine the kinetic contributions, the scan-rate dependence of peak current was analyzed using ip = aνb. The fitted b-value is close to 1 in the low scan-rate region but gradually approaches 1/2 at higher scan rates, indicating a transition from a surface-controlled (capacitive-like) regime to a diffusion-controlled (solid-state diffusion limited) regime. Notably, SP-2.0/12.1 maintains the b ≈ 1 behavior up to a relatively higher scan-rate (current) region, consistent with its reduced characteristic diffusion length due to the smaller particle size and internal domain size. The transition point shifts depending on the characteristic diffusion length scale, consistent with the combined influence of particle size and internal domain size (SI, Fig. S7).
To further examine the electrochemical kinetics of Li+ transport during cycling, the lithium-ion diffusion coefficients (DLi+) were derived using the galvanostatic intermittent titration technique (GITT), as originally proposed by Weppner and Huggins.49 The GITT measurements were carried out with a voltage range of 2.7–4.3 V over five cycles. The corresponding time–voltage profiles obtained during GITT experiments are presented in Fig. 5a and b. The lithium-ion diffusion coefficient was calculated using the following equation.50
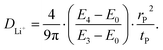 | (1) |
The parameters, DLi+ (cm2 s−1), tp (s), and rp (cm), represent the lithium-ion diffusion coefficient, pulse time, and radius of particles, respectively, as illustrated in Fig. 5a. Upon initiation of the charging process, DLi+ value decreased by approximately fourfold during the second-order H1 phase transition. Subsequently, the diffusion coefficient partially recovered and remained relatively stable throughout the continued charging process. A further ∼50% drop in DLi+ was observed near ∼4.1 V, corresponding to the H2–H3 transition, where significant volume changes occur (Fig. 5c). The variation in DLi+ during discharge has a symmetrical trend to that observed during charge, suggesting Li+ intercalation and deintercalation follow the same reaction pathway. The highest average DLi+ value (5.80 × 10−12 cm2 s−1) was observed for the largest SP particle, SP-5.0/12.1. The enhanced Li+ diffusion coefficient in the largest SP-NCM particles can be ascribed to their smallest domain size, which facilitates shorter diffusion paths within the bulk. Because the GITT expression explicitly depends on the characteristic length scale (rp2), we additionally compared a normalized metric, D/rp2, to mitigate geometric effects associated with particle size (SI, Fig. S8). The normalized values show only small differences across samples, suggesting broadly similar intrinsic diffusivity. Notably, SP-2.0/12.1 exhibits a slightly higher D/rp2, indicating marginally more favorable effective transport kinetics. Importantly, despite its larger particle size, SP-5.0/12.1 shows a comparable D/rp2 relative to the other samples, suggesting that the internal domain size can strongly influence the effective diffusion length and compensate for particle-size effects. Overall, these results indicate that the effective Li+ transport is influenced by the combined contribution of particle size and internal domain size (i.e., the effective diffusion length) rather than particle size alone. Synthetically, the normalized D/rp2 analysis and the domain-size characterization suggest that the effective Li+ transport is governed by the characteristic diffusion length determined by both particle size and internal domain size. Notably, SP-5.0/12.1 shows a comparable D/rp2 despite its larger particle size, supporting that internal domain size can compensate for particle-size effects by shortening the effective diffusion length. Moreover, the lithium-ion diffusion coefficients remained stable up to the fifth cycle (Fig. S9), demonstrating the kinetic robustness of the SP-NCM cathodes during prolonged operation.
The physical and chemical stability of SP-NCM cathode particles was studied by postmortem analysis. In conventional polycrystal NCMs with aggregated secondary structure, repeated anisotropic change along the c- and a-axis typically results in the formation of intergranular cracks during prolonged cycles.51–53 However, SP-NCM cathode particles show neither visible inter- nor intragranular fractures even after 200 cycles, as evidenced by cross-sectional SEM (Fig. 6a). As shown in Fig. 6b, the chemical stability of the SP-NCM cathode was further evaluated by Ni 2p X-ray photoelectron spectroscopy (XPS).54–57 The quantitative analysis reveals that the Ni3+ fraction follows the order of SP-2.0/12.1 > SP-1.0/11.2 > SP-5.0/12.1, which is perfectly aligned with the long-term cycling stability results presented in Fig. 4b.
This stability trend is governed by the synergistic effect of particle size and crystalline domain size. SP-2.0/12.1 represents an optimized balance that minimizes the specific surface area while maintaining high crystalline integrity, resulting in the highest Ni3+ ratio and superior chemical stability. In contrast, SP-5.0/12.1, despite having the largest particle size, possesses the smallest crystalline domain size, leading to a high density of internal grain boundaries that intersect the particle surface. These boundaries act as active sites for surface reduction (Ni2+ formation) and localized phase transitions. Crucially, even without macroscopic mechanical fractures, this domain boundary-mediated chemical degradation ultimately accelerates capacity fading. Conversely, SP-1.0/11.2 maintains a large domain size but suffers from an excessively high specific surface area, which promotes greater electrolyte contact and unavoidable surface reduction.58,59 These findings collectively clarify that the superior performance of SP-2.0/12.1 is due to the successful decoupling and control of particle and domain size effects, ensuring enhanced structural and electrochemical robustness.
4. Conclusion
In summary, single-particle NCM cathodes were successfully synthesized through a two-step scalable process combining controlled coprecipitation and a subsequent molten-salt reaction. By precisely tuning the precursor morphology through ammonia concentration and pH, we enabled systematic control over both the final particle size and crystalline domain size in the synthesized cathode materials. This study demonstrates that the crystalline domain size governs the rate performance, while the specific surface area and the density of internal domain boundaries, determined by the particle size and domain size, primarily dictate the chemical and electrochemical stability. These insights establish a fundamental understanding of how to optimize rate capability and structural durability in single-particle Ni-rich NCM cathodes, offering a practical route to mitigate surface-driven degradation while retaining high performance. The findings presented here are expected to guide the design of next-generation layered oxide cathode materials with enhanced kinetic and structural stability for advanced lithium-ion batteries.Author contributions
Soonhyun Hong: writing – original draft, visualization, validation, software, methodology, investigation, formal analysis, data curation, conceptualization. Heesang Lee: writing – original draft, visualization, software, methodology, investigation, formal analysis, data curation. Jahun Koo: visualization, software, methodology, investigation, formal analysis, data curation. Wonchan Hwang: visualization, software, methodology, investigation, formal analysis, data curation. Ji Hwan Kim: visualization, software, methodology, investigation, formal analysis, data curation. Jungjin Park: writing – review & editing, validation, supervision, project administration, funding acquisition, conceptualization. Young-Sang Yu: writing – review & editing, validation, supervision, project administration, funding acquisition, conceptualization. Chunjoong Kim: writing – review & editing, validation, supervision, project administration, funding acquisition, conceptualization.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental procedures, additional SEM images with EBSD domain maps, XRD with Rietveld refinement and electrochemical impedance spectroscopy (EIS) data. See DOI: https://doi.org/10.1039/d5ta10228f.Acknowledgements
S. H. and H. L. contributed equally to this work. C. K. was supported through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (RS-2023-NR077186) and the Ministry of Education (RS-2021-NR060128). Y.-S. Y. was supported by the Commercialization Promotion Agency for R&D Outcomes (COMPA) grant funded by the Korean Government (Ministry of Science and ICT, Grant No. RS-2023-00304768). This work was supported by the KIST Institutional Program (No. 26E0321 and No. 26E0324).References
- S. Fankhauser, S. M. Smith, M. Allen, K. Axelsson, T. Hale, C. Hepburn, J. M. Kendall, R. Khosla, J. Lezaun, E. Mitchell-Larson, M. Obersteiner, L. Rajamani, R. Rickaby, N. Seddon and T. Wetzer, Nat. Clim. Change, 2022, 12, 15–21 CrossRef.
- H. E. Melin, The lithium-ion battery end-of-life market – A baseline study, World Economic Forum, 2018 Search PubMed.
- N. Nitta, F. X. Wu, J. T. Lee and G. Yushin, Mater. Today, 2015, 18, 252–264 CrossRef CAS.
- K. Mizushima, P. C. Jones, P. J. Wiseman and J. Goodenough, Solid State Ionics, 1981, 3–4, 171–174 CrossRef CAS.
- S. Watanabe, M. Kinoshita, T. Hosokawa, K. Morigaki and K. Nakura, J. Power Sources, 2014, 260, 50–56 CrossRef CAS.
- X. Wu, K. Song, X. Zhang, N. Hu, L. Li, W. Li, L. Zhang and H. Zhang, Front. Energy Res., 2019, 7, 65 CrossRef.
- L. K. Willenberg, P. Dechent, G. Fuchs, D. U. Sauer and E. Figgemeier, Sustainability, 2020, 12, 557 CrossRef CAS.
- J. Li, A. R. Cameron, H. Y. Li, S. Glazier, D. J. Xiong, M. Chatzidakis, J. Allen, G. A. Botton and J. R. Dahn, J. Electrochem. Soc., 2017, 164, A1534–A1544 CrossRef CAS.
- H.-H. Ryu, B. Namkoong, J.-H. Kim, I. Belharouak, C. S. Yoon and Y.-K. Sun, ACS Energy Lett., 2021, 6, 2726–2734 CrossRef CAS.
- L. De Biasi, A. Schiele, M. Roca-Ayats, G. Garcia, T. Brezesinski, P. Hartmann and J. Janek, ChemSusChem, 2019, 12, 2240–2250 CrossRef CAS.
- J. Zhang, J. Zhang, X. Ou, C. Wang, C. Peng and B. Zhang, ACS Appl. Mater. Interfaces, 2019, 11, 15507–15516 CrossRef CAS PubMed.
- K.-J. Park, J.-Y. Hwang, H.-H. Ryu, F. Maglia, S.-J. Kim, P. Lamp, C. S. Yoon and Y.-K. Sun, ACS Energy Lett., 2019, 4, 1394–1400 CrossRef CAS.
- H. Li, A. Liu, N. Zhang, Y. Wang, S. Yin, H. Wu and J. R. Dahn, Chem. Mater., 2019, 31, 7574–7583 CrossRef CAS.
- P. Xiao, T. Shi, W. Huang and G. Ceder, ACS Energy Lett., 2019, 4, 811 CrossRef CAS.
- G.-T. Park, H.-H. Ryu, T.-C. Noh, G.-C. Kang and Y.-K. Sun, Mater. Today, 2022, 52, 9–18 CrossRef CAS.
- Y. Liu, X. Fan, B. Luo, Z. Zhao, J. Shen, Z. Liu, Z. Xiao, B. Zhang, J. Zhang and L. Ming, J. Colloid Interface Sci., 2021, 604, 776–784 CrossRef CAS.
- K. Park, D. J. Ham, S. Y. Park, J. Jang, D.-H. Yeon, S. Moon and S. J. Ahn, RSC Adv., 2020, 10, 26756–26764 RSC.
- L. Zheng, J. C. Bennett and M. N. Obrovac, J. Electrochem. Soc., 2020, 167, 130536 CrossRef CAS.
- J. E. Harlow, X. Ma, J. Li, E. Logan, Y. Liu, N. Zhang, L. Ma, S. L. Glazier, M. M. E. Cormier and M. Genovese, J. Electrochem. Soc., 2019, 166, A3031–A3044 CrossRef CAS.
- J. Cheng, X. Ma, X. Sun, F. Lian, X. Liu, L. Lu and D. Xia, Energy Storage Mater., 2025, 79, 104332 CrossRef.
- Y. Han, S. H. Jung, H. Kwak, S. Jun, H. H. Kwak, J. H. Lee, S. T. Hong and Y. S. Jung, Adv. Energy Mater., 2021, 11, 2100126 CrossRef CAS.
- A. Chen, K. Wang, J. Li, Q. Mao, Z. Xiao, D. Zhu, G. Wang, P. Liao and J. He, Front. Energy Res., 2020, 8, 593009 CrossRef.
- J. Li, H. Li, W. Stone, R. Weber, S. Hy and J. R. Dahn, J. Electrochem. Soc., 2017, 164, A3529 CrossRef CAS.
- H. Li, J. Li, N. Zaker, N. Zhang, G. A. Botton and J. R. Dahn, J. Electrochem. Soc., 2019, 166, A1956 CrossRef CAS.
- M. H. Lee, Y. J. Kang, S. T. Myung and Y. K. Sun, Electrochim. Acta, 2004, 50, 939–948 CrossRef CAS.
- L. Liang, K. Du, Z. Peng, Y. Cao, J. Duan, J. Jiang and G. Hu, Electrochim. Acta, 2014, 130, 82–89 CrossRef CAS.
- C. Deng, L. Liu, W. Zhou, K. Sun and D. Sun, Electrochim. Acta, 2008, 53, 2441–2447 CrossRef CAS.
- F. Lv, Y. Zhang, M. Wu and Y. Gu, Small, 2022, 18, 2201946 CrossRef CAS.
- C. A. Schneider, W. S. Rasband and K. W. Eliceiri, Nat. Methods, 2012, 9, 671–675 CrossRef CAS PubMed.
- F. Peng, D. Mu, R. Li, Y. Liu, Y. Ji, C. Dai and F. Ding, RSC Adv., 2019, 9, 21922–21930 RSC.
- K.-M. Nam, H.-J. Kim, D.-H. Kang, Y.-S. Kim and S.-W. Song, Green Chem., 2015, 17, 1127–1135 RSC.
- Y. Yang, S. Xu, M. Xie, Y. He, G. Huang and Y. Yang, J. Alloys Compd., 2015, 619, 846–853 CrossRef CAS.
- P. L. Guzzo, A. A. A. Tino and J. B. Santos, Powder Technol., 2015, 284, 122–129 CrossRef CAS.
- J. Huang, P. Xu, D. Cao, X. Zhou, S. Yang, Y. Li and G. Wang, J. Power Sources, 2014, 246, 371–376 CrossRef CAS.
- R. Wang, J. Lang, Y. Liu, Z. Lin and X. Yan, NPG Asia Mater., 2015, 7, e183 CrossRef CAS.
- L. Qiu, M. Zhang, Y. Ming, Y. Song, C. Xu, Z. Wu, Q. Xu, T. Chen, G. Wang and Y. Liu, Chem. Eng. Sci., 2021, 233, 116337 CrossRef CAS.
- Z. Wu, Y. Zhou, C. Hai, J. Zeng, X. Ren, Y. Sun, Y. Shen, X. Li, S. Dong and G. Zhang, Ceram. Int., 2022, 48, 17279–17288 CrossRef CAS.
- J. Langdon and A. Manthiram, Energy Storage Mater., 2021, 37, 143–160 CrossRef.
- T. Wang, K. Ren, M. He, W. Dong, W. Xiao, H. Pan, J. Yang, Y. Yang, P. Liu and Z. Cao, Front. Chem., 2020, 8, 747 CrossRef CAS PubMed.
- J. C. Garcia, J. Bareño, J. Yan, G. Chen, A. Hauser, J. R. Croy and H. Iddir, J. Phys. Chem. C, 2017, 121, 8290–8299 CrossRef CAS.
- S. Sharifi-Asl, F. A. Soto, A. Nie, Y. Yuan, H. Asayesh-Ardakani, T. Foroozan, V. Yurkiv, B. Song, F. Mashayek and R. F. Klie, Nano Lett., 2017, 17, 2165–2171 CrossRef CAS PubMed.
- P. Yan, J. Zheng, J. Zheng, Z. Wang, G. Teng, S. Kuppan, J. Xiao, G. Chen, F. Pan and J.-G. Zhang, Adv. Energy Mater., 2016, 6, 1502455 CrossRef.
- J. Zhu and G. Chen, J. Mater. Chem. A, 2019, 7, 5463–5474 RSC.
- H. J. Kim, Y. Park, Y. Kwon, J. Shin, Y.-H. Kim, H.-S. Ahn, R. Yazami and J. W. Choi, Energy Environ. Sci., 2020, 13, 286–296 RSC.
- S. Sloop, L. Crandon, M. Allen, K. Koetje, L. Reed, L. Gaines and W. Sirisaksoontorn, Sustain. Mater. Technol., 2020, 25, e00152 CAS.
- J. Lai, J. Zhang, Z. Li, Y. Xiao, W. Hua, Z. Wu, Y. Chen, Y. Zhong and W. Xiang, Chem. Commun., 2020, 56, 4886–4889 RSC.
- J. Xu, E. Hu, D. Nordlund, A. Mehta, S. N. Ehrlich, X. Q. Yang and W. Tong, ACS Appl. Mater. Interfaces, 2016, 8, 31677–31683 CrossRef CAS.
- B. Kang and G. Ceder, Nature, 2009, 458, 190–193 CrossRef CAS.
- W. Weppner and R. A. Huggins, J. Electrochem. Soc., 1977, 124, 1569 CrossRef CAS.
- A. Nickol, T. Schied, C. Heubner, M. Schneider, A. Michaelis, M. Bobeth and G. Cuniberti, J. Electrochem. Soc., 2020, 167, 090546 CrossRef CAS.
- F. Zhang, S. Lou, S. Li, Z. Yu, Q. Liu, A. Dai, C. Cao, M. F. Toney, M. Ge and X. Xiao, Nat. Commun., 2020, 11, 3050 CrossRef CAS.
- J. Li and A. Manthiram, Adv. Energy Mater., 2019, 9, 1902731 CrossRef CAS.
- J. Li, L. E. Downie, L. Ma, W. Qiu and J. R. Dahn, J. Electrochem. Soc., 2015, 162, A1401 CrossRef CAS.
- Y. Wang, S. Tao, H. Lin, S. Han, W. Zhong, Y. Xie, J. Hu and S. Yang, RSC Adv., 2020, 10, 33475–33482 RSC.
- Y. S. Chen, J. F. Kang, B. Chen, B. Gao, L. F. Liu, X. Y. Liu, Y. Y. Wang, L. Wu and H. Y. Yu, J. Phys. D: Appl. Phys., 2012, 45, 065303 CrossRef.
- N. S. Mcintyre, T. C. Chan and C. Chen, Oxid. Met., 1990, 33, 457–479 CrossRef CAS.
- R. Hang, X. Huang, L. Tian, Z. He and B. Tang, Electrochim. Acta, 2012, 70, 382–393 CrossRef CAS.
- J. Kim, H. Cha, H. Lee, P. Oh and J. Cho, Batter. Supercaps, 2020, 3, 309–322 CrossRef CAS.
- Y. Kim, ACS Appl. Mater. Interfaces, 2012, 4, 2329 CrossRef CAS.
Footnote |
| † S. H. and H. L. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |






