 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Lithium coordination disorder controlling ionic conductivity in mixed-halide borate glasses
Yohei Onodera *a,
Yasuyuki Takimotob,
Hiroyuki Hijiyac,
Noriyoshi Kayabac,
Masamichi Tanidad,
Kazutaka Ikeda
*a,
Yasuyuki Takimotob,
Hiroyuki Hijiyac,
Noriyoshi Kayabac,
Masamichi Tanidad,
Kazutaka Ikeda e and
Shinji Kohara
e and
Shinji Kohara a
a
aCenter for Basic Research on Materials, National Institute for Materials Science, Sengen 1-2-1, Tsukuba, Ibaraki 305-0047, Japan. E-mail: ONODERA.Yohei@nims.go.jp
bInnovative Technology Laboratories, AGC, Inc., 1-1 Suehiro-cho, Tsurumi-ku, Yokohama, Kanagawa 230-0045, Japan
cMaterials Integration Laboratories, AGC, Inc., 1-1 Suehiro-cho, Tsurumi-ku, Yokohama, Kanagawa 230-0045, Japan
dElectronic Materials General Division, Electronics Company, AGC Inc., 1-5-1 Marunouchi, Chiyoda-Ku, Tokyo 100-8405, Japan
eNeutron Industrial Application Promotion Center, Comprehensive Research Organization for Science and Society, 162-1 Shirakata, Tokai, Naka, Ibaraki 319-1106, Japan
First published on 1st April 2026
Abstract
Understanding the structure–property relationships in glasses remains challenging owing to their structural complexity, especially in multicomponent practical glasses. In this work, neutron diffraction with isotopic substitution was employed to investigate lithium coordination environments in halide (chloride and/or bromide)-doped borate glasses, which exhibit variations in lithium-ion conductivity associated with anion mixing. Lithium-specific neutron pair distribution function analysis reveals that the conductivity enhancement in oxide–halide mixed glasses originates from the breaking of one short Li–O bond and the formation of two longer Li–halogen bonds. The formation of LiO3Cl2 and LiO3Br2 units is observed in chloride- and bromide-doped glasses, respectively, whereas a glass containing equal amounts of chloride and bromide exhibits LiO3ClBr units in addition to LiO3Cl2 and LiO3Br2 units. These results suggest that the coexistence of multiple lithium–oxygen–halogen polyhedral units hinders lithium-ion migration, leading to reduced ionic conductivity in halide–halide mixed glasses. This work provides new insights into the structure–property relationships in lithium-ion conducting glasses through lithium-specific structural analysis.
1. Introduction
Glasses with high ionic conductivity have attracted considerable attention since the discovery of superionic conducting glasses by Minami and coworkers in 1977.1,2 In recent years, there has been an increase in research focusing on the practical utilization of ionic conducting glasses as solid electrolytes for lithium-ion batteries, particularly for portable devices and electric vehicles.3,4 The substitution of a flammable organic electrolyte in conventional lithium-ion batteries with a solid electrolyte has the potential to enhance battery safety and improve energy and power densities. Consequently, candidate materials must exhibit high ionic conductivity when utilized as solid electrolytes. A comprehensive understanding of lithium-ion conduction mechanisms in solid electrolytes is essential for the development of new materials with high ionic conductivity. The mechanism of lithium-ion conduction in crystalline ionic conductors has been elucidated through crystal structure analysis,5,6 which has accelerated materials development. In contrast, the absence of long-range order in glasses makes analytical approaches developed for crystalline materials inapplicable. Therefore, structural studies of glasses require a combination of techniques, including diffraction, spectroscopy, and computer simulations, to elucidate the mechanism underlying ionic conductivity.Numerous studies have been conducted on ionic conducting oxide and sulfide glasses to date. In oxide glasses, borate-7 and phosphate-8 based glasses are known to exhibit lithium-ion conductivity. Recently, ternary lithium borovanadate (Li2O–V2O5–B2O3) glasses have been reported to exhibit an ionic conductivity of the order of 10−4 S cm−1 at room temperature, representing the highest level of ionic conductivity among oxide glasses.9 Structural studies have shown that lithium incorporation significantly modifies the glass network. For example, 10B nuclear magnetic resonance (NMR) spectroscopy revealed that the structural units in binary Li2O–B2O3 glasses transform from boroxol, diborate, and tetraborate units to metaborate, pyroborate, orthoborate, and loose BO4 tetrahedral units with increasing Li2O content, creating non-bridging oxygens.10 An increase in the number of non-bridging oxygens with increasing Li2O content has also been observed in Li2O–V2O5–B2O3 glasses.9 Similar depolymerization of the network consisting of PO4 tetrahedra has also been observed in Li2O–P2O5 glasses based on 31P magic angle spinning (MAS) NMR measurements.11 On the other hand, X-ray and neutron diffraction studies demonstrated that lithium ions are tetrahedrally coordinated by four oxygen atoms in borate and phosphate glasses.12–14 Moreover, ionic conduction pathways in these glasses have been visualized using bond valence (BV) analysis of structure models generated by reverse Monte Carlo (RMC) modelling based on diffraction data.15–18 In sulfide glasses, the development of Li7P3S11 glass ceramic obtained by partial crystallization of 70Li2S–30P2S5 glass, which exhibits an ionic conductivity exceeding 10−3 S cm−1 comparable to that of organic liquid electrolytes, has significantly stimulated research in the Li2S–P2S5 system.19 Spectroscopic studies using Raman19 and 31P MAS NMR20,21 techniques revealed that the fundamental structural units in 70Li2S–30P2S5 glass and Li7P3S11 glass ceramic are thiophosphate species such as P2S74− and PS43−. Structural analyses combining diffraction experiments and RMC modelling have indicated that fourfold coordination around lithium ions plays an important role in ionic conduction22 and have enabled visualization of conduction pathways23 through BV analysis.15–18 Recent studies combining RMC modelling and first-principles calculations have further clarified the structural and dynamical features associated with lithium-ion migration in Li2S–P2S5 glasses.24–26
Consequently, lithium-ion-conducting glasses have been extensively investigated in response to growing interest in all-solid-state lithium-ion batteries. However, the intricate interplay between the lithium coordination environment and the conduction mechanism in glasses remains incompletely understood. Understanding this relationship is crucial for the rational design of glassy solid electrolytes with improved ionic conductivity. Beyond studies focused directly on ionic conduction, it is well recognized that the physical and functional properties of multicomponent glasses can be systematically tuned through compositional variation. Variations in cation and anion species have been shown to modify optical, dielectric, and mechanical properties in a variety of multicomponent oxide glasses, including rare-earth-doped lead–silicate27 and lead–borate28 glasses, lithium fluoroborophosphate glasses,29 CuO-doped silicate30 and aluminosilicate31 glasses, and halide-substituted borophosphate glasses.32 These findings highlight that compositional diversity strongly influences both local structures and macroscopic properties in glasses. In particular, glasses containing mixed anions have attracted attention because combining different anion species can significantly influence ionic conductivity.33,34 Nevertheless, only a limited number of structural studies have been reported for such mixed-anion glasses.
One major challenge arises from the difficulty of extracting lithium-related structural information from the disordered atomic arrangements of multicomponent glasses, even when neutron diffraction, which is sensitive to light elements, is employed. Lithium exhibits a negative neutron coherent scattering length (b = −1.90 fm),35 and atomic correlations between lithium and other atoms appear as negative peaks in the pair distribution function (PDF) obtained by neutron diffraction. However, in multicomponent glasses, these negative peaks often overlap with other positive peaks, making quantitative structural analysis extremely difficult. Therefore, an element-specific experimental approach is required to determine the lithium coordination environment and to clarify its role in ionic conduction.
In this study, we investigated five compositions of 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses, in which single Cl, single Br, and mixtures of Cl and Br in the ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 1
3, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 3
1, and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were adopted. To clarify the effect of the addition and mixing of halide anions on ionic conductivity, the structures of the glasses were examined using a combination of high-energy X-ray and neutron diffraction measurements. Specifically, in this study, we employed the neutron diffraction with isotopic substitution (NDIS) technique36–38 to investigate lithium-specific atomic correlations. The NDIS technique utilizes the difference in neutron coherent scattering lengths between isotopes and allows us to obtain element-specific structural information. Indeed, structural studies with a particular focus on lithium using NDIS have been reported for Li2O–2SiO2,39 Li2O–2B2O3,40 and LiAlSiO4 glasses.41 In this study, two glasses with different lithium isotopic enrichments were prepared, enabling lithium-specific structural data to be obtained from the difference between neutron diffraction data. Consequently, the interatomic distances and coordination numbers around lithium cations were determined by lithium-specific PDF analysis employing the NDIS data. The coordination numbers for non-lithium-related correlations were also determined using neutron diffraction data of nullLi-enriched glasses, where the coherent neutron scattering length of lithium is adjusted to zero. In addition, the structural parameters obtained were verified through their application to the reproduction of high-energy X-ray diffraction data. Thus, a cutting-edge structural analysis, founded upon element-specific quantum beam measurements, has been meticulously executed. In this work, we investigate the relationship between the lithium coordination environments and ionic conductivity in 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses using lithium-specific structural information obtained by our novel structural analysis.
1 were adopted. To clarify the effect of the addition and mixing of halide anions on ionic conductivity, the structures of the glasses were examined using a combination of high-energy X-ray and neutron diffraction measurements. Specifically, in this study, we employed the neutron diffraction with isotopic substitution (NDIS) technique36–38 to investigate lithium-specific atomic correlations. The NDIS technique utilizes the difference in neutron coherent scattering lengths between isotopes and allows us to obtain element-specific structural information. Indeed, structural studies with a particular focus on lithium using NDIS have been reported for Li2O–2SiO2,39 Li2O–2B2O3,40 and LiAlSiO4 glasses.41 In this study, two glasses with different lithium isotopic enrichments were prepared, enabling lithium-specific structural data to be obtained from the difference between neutron diffraction data. Consequently, the interatomic distances and coordination numbers around lithium cations were determined by lithium-specific PDF analysis employing the NDIS data. The coordination numbers for non-lithium-related correlations were also determined using neutron diffraction data of nullLi-enriched glasses, where the coherent neutron scattering length of lithium is adjusted to zero. In addition, the structural parameters obtained were verified through their application to the reproduction of high-energy X-ray diffraction data. Thus, a cutting-edge structural analysis, founded upon element-specific quantum beam measurements, has been meticulously executed. In this work, we investigate the relationship between the lithium coordination environments and ionic conductivity in 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses using lithium-specific structural information obtained by our novel structural analysis.
2. Experimental
2.1 Preparation of glasses
The 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx (x = 0, 0.25, 0.5, 0.75, and 1) glasses were prepared from mixtures of reagent-grade Li2CO3, H311BO3, LiCl, and LiBr. The mixtures were melted at 900–1000 °C for 15 min, and each melt was then quenched by the twin-roller method. In this study, two types of glasses with different lithium isotope ratios were prepared: natLi-enriched 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses (natLi glasses) and nullLi-enriched 1/3Li2O–1/3B2O3–1/3LiCl1–xBrx glasses (nullLi glasses). For the preparation of the nullLi glasses, a mixture of natLi2CO3 and 6Li2CO3 was used to achieve the nullLi composition (6Li![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7Li = 0.5265
7Li = 0.5265![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.4735). For comparison, the 0.5Li2O–0.5B2O3 (LiBO2) glass was also prepared in a similar manner to the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses. The densities of the glasses were measured by helium gas pycnometry using a AccuPyc II 1340 gas pycnometer (Micrometrics, USA).
0.4735). For comparison, the 0.5Li2O–0.5B2O3 (LiBO2) glass was also prepared in a similar manner to the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses. The densities of the glasses were measured by helium gas pycnometry using a AccuPyc II 1340 gas pycnometer (Micrometrics, USA).
2.2 Conductivity measurements
The electrical conductivities of the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses were measured at room temperature by the ac four-probe method using a Solartron SI 1287 frequency response analyser (Solartron Analytical, UK). Au electrodes with a diameter of 4 mm were formed on both sides of the glass-flake samples by vacuum deposition. The applied voltage was 50 mV, and the frequency range was from 1 Hz to 1 MHz. All measurements were carried out under a dry-air atmosphere. The electrical conductivities of the glasses were determined from the resistance values obtained at the real-axis intercept of the Nyquist plots.2.3 NMR measurements
11B MAS NMR experiments were conducted on the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glass samples at room temperature using an ECA600 spectrometer (JEOL, Japan). The experiments were conducted with a 11B Larmor frequency of 192.55 MHz with a spin rate of 20 kHz and a pulse delay of 3 s. The 11B chemical shifts were estimated with respect to an external saturated boric acid solution (19.6 ppm). The obtained spectra were subsequently deconvoluted into two components.2.4 Diffraction measurements
The high-energy X-ray diffraction experiments were conducted at beamline BL04B2 at SPring-8 (Hyogo, Japan) using a diffractometer dedicated to disordered materials.42 Crushed glass samples were loaded into SiO2 glass capillaries. The incident X-ray energy was 61.23 keV. The diffraction patterns of the glasses were measured at room temperature in transmission geometry. The intensity of the incident X-rays was monitored in an Ar-filled ionization chamber, and the scattered X-rays were detected using four CdTe detectors and three Ge detectors. A vacuum chamber was used to suppress air scattering around the sample. The raw data were corrected for polarization, absorption, and background, and the contribution of Compton scattering was subtracted using standard data analysis software.43The neutron diffraction measurements were conducted on the high-intensity total diffractometer, NOVA,44 installed at beamline BL21 of the Materials and Life Science Experimental Facility (MLF) at the J-PARC spallation neutron source (Ibaraki, Japan). The crushed glass samples were loaded into vanadium–nickel (V–Ni) null alloy cells with an outer diameter of 6.0 mm and a thickness of 0.1 mm. The wavelength range of the incident neutron beam was 0.12 < λ < 8.3 Å. Measurements were performed for the samples contained in the V–Ni cell, the empty V–Ni cell, the empty instrument, and a vanadium standard for normalization at room temperature. The observed scattering intensities for the samples were corrected for the instrument background and for attenuation by the sample and cell. Subsequently, the corrected intensities were normalized using the incident beam profile and further corrected for multiple and incoherent scattering.
The fully corrected X-ray and neutron diffraction data were normalized to give the Faber–Ziman total structure factor S(Q):45
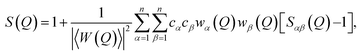 | (1) |
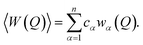 | (2) |
Total correlation functions, T(r), were obtained by a Fourier transform of S(Q) with a Lorch function, M(Q).46
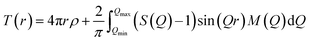 | (3) |
To obtain lithium-specific structural information, the NDIS was employed. The NDIS technique allows element-specific structural information to be obtained by utilizing the difference in coherent neutron scattering lengths between isotopes. In the present study, two glasses with identical chemical compositions but different lithium isotopic ratios (natLi and nullLi glasses) were prepared. Because only the neutron scattering length of lithium differs between the two samples, taking the difference between the diffraction data extract structural correlations involving lithium atoms. The differential intensity ΔLiI(Q) between the scattering intensities of the natLi and nullLi glasses is expressed as
| ΔLiIcoh(Q) = ΔLi[〈b2〉 − 〈b〉2] + ΔLi[〈b2〉]ΔLiS(Q), | (4) |
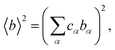 | (5) |
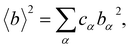 | (6) |
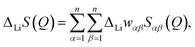 | (7) |
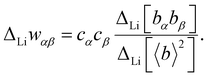 | (8) |
Compared with the total structure factors S(Q) obtained from X-ray and neutron diffraction, ΔLiS(Q) significantly enhances the contributions from Li-related Sαβ(Q) while suppressing contributions from the other partials. Accordingly, ΔLiS(Q) for the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses can be expressed as
| ΔLiS(Q) = ΔLiwLiLiSLiLi(Q) + 2ΔLiwLiBSLiB(Q) + 2ΔLiwLiOSLiO(Q) + 2ΔLiwLiClSLiCl(Q) + 2ΔLiwLiBrSLiBr(Q), | (9) |
To obtain detailed structural information on the short range, S(Q) and T(r) were analysed using the pair function method proposed by Mozzi and Warren.47 The pair function method is useful for analysing real-space functions from which the structural parameters such as the interatomic distance and coordination number can be determined. Utilizing the pair function formalism, the calculated total correlation function Tcalc(r) was obtained using the following equation for the interatomic distance rα–β and coordination number Nα–β of the α–β pair:
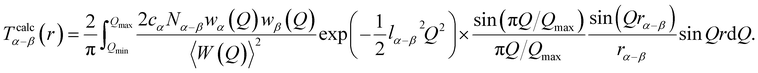 | (10) |
The term lα–β is a convergence factor representing the static and thermal disorders of the α–β correlation. The calculated differential total correlation function ΔLiTcalc(r) was also obtained using the following equation for the interatomic distance rLi–β and coordination number NLi–β of the Li–β pair:
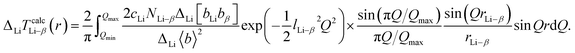 | (11) |
The variations in rα–β (rLi–β) and Nα–β (NLi–β) obtained from the fitting T(r) and ΔLiT(r) were estimated to be ±0.02 Å and ±0.3, respectively.
3. Results and discussion
3.1 Glass composition and density
The chemical compositions of the obtained 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses are listed in Table 1. The glasses are designated by the ratio of Cl to Br, e.g., Cl50Br50 refers to a glass containing an equal mixture of Cl and Br (1/3Li2O–1/3B2O3–1/3LiCl0.5Br0.5 glass). The densities of the 1/3Li2O–1/3B2O3–1/3LiCl1–xBrx glasses are summarized in Table 2. The density increases monotonically with the degree of Br substitution. The average number densities ρ of the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses, calculated from these densities, are also listed in Table 2.| Glass sample | Element (at%) | ||||
|---|---|---|---|---|---|
| Li | B | O | Cl | Br | |
| Br100 | 31.6 | 18.9 | 39.1 | 0 | 10.4 |
| Cl25Br75 | 30.6 | 19.6 | 39.7 | 2.5 | 7.5 |
| Cl50Br50 | 30.1 | 20.3 | 41.3 | 4.3 | 4.0 |
| Cl75Br25 | 28.0 | 21.5 | 42.0 | 6.7 | 1.8 |
| Cl100 | 29.4 | 20.5 | 40.8 | 9.3 | 0 |
| Glass sample | Density (g cm−3) | Number density (Å−3) |
|---|---|---|
| Br100 | 2.47 | 0.0790 |
| Cl25Br75 | 2.38 | 0.0817 |
| Cl50Br50 | 2.28 | 0.0874 |
| Cl75Br25 | 2.18 | 0.0884 |
| Cl100 | 2.06 | 0.0880 |
3.2 Conductivity
The electrical conductivities of the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses range from 2.7 × 10−6 to 6.6 × 10−6 S cm−1 at room temperature (Fig. 1). These values are approximately one order of magnitude higher than those of LiBO2 glass (∼10−7 S cm−1),48–50 indicating that the addition of halide anions enhances the lithium-ion conductivity. Similar behaviour has been reported in other glass systems, including Li2O–B2O3–LiX (X = F, Cl, Br, I),7 LiPO3–LiX (X = Cl, Br, I),51 and Li2S–B2S3–LiI.52It is well established that the mixing of different types of anion, known as the mixed-anion effect, enhances lithium-ion conductivity in various glass systems.33,34 Indeed, the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses exhibit enhanced conductivity in comparison with LiBO2 glass, attributable to the mixing of oxide and halide anions. In contrast, the Cl25Br75, Cl50Br50, and Cl75Br25 glasses exhibit a substantial decrease in conductivity compared with the Cl100 and Br100 glasses, as shown in Fig. 1. In particular, the Cl50Br50 glass exhibits the lowest conductivity among the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses, suggesting that the mixing of halide anions leads to a reduction in conductivity. Therefore, the variation in ionic conductivity for the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses can be interpreted in two stages: (i) enhancement of ionic conductivity induced by the mixing of oxide and halide anions, and (ii) reduction of ionic conductivity caused by the mixing of halide (chloride and bromide) anions. A comparable phenomenon is observed in silicate glasses exhibiting the mixed alkali effect, where alkali mixing leads to substantial reductions in conductivity, viscosity, and dielectric constant.53–55 In our previous study, a combination of PDF analysis with structural modelling revealed that potassium ions were trapped within highly coordinated potassium–oxygen polyhedra, forming a correlated pair arrangement with sodium–oxygen polyhedra in a silicate glass with the mixed alkali effect.55 Therefore, the variation in ionic conductivity shown in Fig. 1 is closely related to changes in the lithium coordination environment in the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses. To elucidate the structural origin of this behaviour, the local atomistic structure around lithium ions was examined by combining high-energy X-ray diffraction and NDIS.
3.3 Coordination number analyses using diffraction data
 | ||
| Fig. 2 X-ray diffraction data of LiBO2 glasses in (a) Q space and (b) real space. Red solid curves represent experimental data, and black broken curves represent calculated data. | ||
| B–O (I) | B–O (II) | Li–O (I) | Li–O (II) | O–O | B–B | Li–B | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| rB–O (Å) | NB–O | rB–O (Å) | NB–O | rLi–O (Å) | NLi–O | rLi–O (Å) | NLi–O | rO–O (Å) | NO–O | rB–B (Å) | NB–B | rLi–B (Å) | NLi–B | |
| LiBO2 glass (this study) | 1.42 | 2.75 | 1.63 | 0.45 | 1.98 | 3.2 | 2.35 | 0.9 | 2.42 | 4.4 | 2.42 | 2.4 | 2.70 | 4.0 |
| LiBO2 glass59 | 1.42 | 3.00 | 1.63 | 0.25 | 1.98 | 2.9 | 2.35 | 1.2 | 2.41 | 4.4 | 2.42 | 2.4 | 2.70 | 4.0 |
| B–O (I) | B–O (II) | O–O | B–B | |||||
|---|---|---|---|---|---|---|---|---|
| rB–O (Å) | NB–O | rB–O (Å) | NB–O | rO–O (Å) | NO–O | rB–B (Å) | NB–B | |
| Br100 | 1.40 | 3.0 | 1.52 | 0.4 | 2.41 | 4.0 | 2.42 | 2.4 |
| Cl25Br75 | 1.40 | 3.0 | 1.52 | 0.4 | 2.41 | 4.0 | 2.42 | 2.4 |
| Cl50Br50 | 1.40 | 3.0 | 1.52 | 0.4 | 2.41 | 4.0 | 2.41 | 2.4 |
| Cl75Br25 | 1.40 | 3.0 | 1.52 | 0.4 | 2.41 | 4.0 | 2.42 | 2.4 |
| Cl100 | 1.40 | 3.0 | 1.52 | 0.4 | 2.41 | 4.0 | 2.42 | 2.4 |
3.4 Lithium-specific coordination number analyses using NDIS data
To obtain Li-specific structural information, ΔLiS(Q) for the Cl100, Cl50Br50, and Br100 glasses were analysed (Fig. 5(a)). These data were obtained by taking the difference between the neutron diffraction data of the natLi and nullLi glasses. A negative peak is observed at Q ∼2 Å−1 in all the glasses. Majérus et al. reported that a similar negative peak originates from Li-centred intermediate-range ordering in Li2O–2B2O3 glass.40 However, in the present 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses, the peak appears at a higher Q position and exhibits an asymmetric shape, suggesting the formation of a Li-centred intermediate-range structure with a different characteristic length scale. The differential total correlation functions, ΔLiT(r), obtained from the Fourier transforms of ΔLiS(Q) are shown in Fig. 5(b). Three broad peaks arising from Li-related correlations are observed at approximately 1.8–3.0 Å in all the glasses. The first peak at approximately 2.0 Å corresponds to the Li–O correlation, consistent with the negative peak observed in the neutron T(r) data for the natLi glasses (Fig. 4(b)). The second peak at approximately 2.4 Å is evident in the Cl100 and Cl50Br50 glasses and is mainly attributed to the Li–Cl correlation, in agreement with the Li–Cl distance (2.45 Å) reported for molten LiCl.64 In the Br100 glass, the second peak appears as a shoulder peak at approximately 2.5–2.7 Å and is assigned to the Li–Br correlation, consistent with the Li–Br distance (2.68 Å) in molten LiBr.64 The third peak mainly originates from the Li–B correlation associated with Li–O–B linkages, as discussed in the X-ray diffraction results for the LiBO2 glasses.The Li–O, Li–Cl, Li–Br, and Li–B coordination numbers were determined by fitting calculated ΔLiS(Q) and ΔLiT(r) to the experimental data using eqn (11). Two Li–O pair functions were adopted based on the results for the LiBO2 glasses. As shown in Fig. 5(a), the calculated ΔLiS(Q) reproduces the experimental data well for Q > 2 Å−1, and good agreement between the calculated and experimental ΔLiT(r) is also obtained (Fig. 5(b)). The derived structural parameters are summarized in Table 5. The Li–O coordination number is 3.1–3.2 in all the glasses, indicating that the tetrahedral coordination around lithium cations collapses in the halide-containing glasses. Lithium cations are threefold coordinated by oxygen atoms, with two oxygen atoms at approximately 1.95–1.96 Å and one additional oxygen atom at approximately 2.30–2.36 Å, indicating the breaking of one short Li–O bond upon halide addition. The Li–Cl coordination number decreases from 1.8 in the Cl100 glass to 1.1 in the Cl50Br50 (mixed-halide) glass. Similarly, the Li–Br coordination number decreases from 1.9 (Br100) to 1.0 (Cl50Br50). The Li–B coordination number is approximately 3.0 in all the glasses. This behaviour is consistent with the results observed in the LiBO2 glasses, where the Li–B coordination number was almost identical to that of Li–O, suggesting the presence of Li–O–B linkages. To further validate these results, high-energy X-ray diffraction data were analysed based on the NDIS results using the pair-function analysis (eqn (10)). The calculated X-ray S(Q) and T(r) (Fig. S3(a) and (b)) reproduce the experimental data well. The structural parameters for lithium-related correlations derived from the X-ray diffraction data (Tables S3 and S4) agree with those obtained from the NDIS data (Table 5). Additionally, the structural parameters for the B–O, O–O, and B–B correlations also agree with those obtained from the neutron diffraction data of the nullLi glasses (Table 4). These results demonstrate that reliable coordination numbers in the 1/3Li2O–1/3B2O3–1/3LiCl1–xBrx glasses were obtained through an element-specific PDF analysis combining NDIS and high-energy X-ray diffraction data.
| Li–O (I) | Li–O (II) | Li–Cl | Li–Br | Li–B | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| rLi–O (Å) | NLi–O | rLi–O (Å) | NLi–O | rLi–Cl (Å) | NLi–Cl | rLi–Br (Å) | NLi–Br | rLi–B (Å) | NLi–B | |
| Br100 | 1.95 | 2.3 | 2.36 | 0.8 | — | — | 2.64 | 1.9 | 2.95 | 3.0 |
| Cl50Br50 | 1.96 | 2.3 | 2.33 | 0.9 | 2.45 | 1.1 | 2.64 | 1.0 | 2.91 | 3.1 |
| Cl100 | 1.96 | 2.3 | 2.30 | 0.8 | 2.46 | 1.8 | — | — | 2.85 | 3.1 |
3.5 Origin of the improvement in ionic conductivity by mixing of oxide and halide anions
The ionic conductivity of the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses (∼10−6 S cm−1) is approximately one order of magnitude higher than that of LiBO2 glass (∼10−7 S cm−1).48–50 This enhancement is attributed to the mixing of oxide and halide anions. To clarify the lithium environment responsible for this behaviour, representative lithium–anion polyhedra in LiBO2 and the 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses were visualized based on the coordination number analyses (Fig. 6(a–d)).In LiBO2 glasses, lithium cations are tetrahedrally coordinated by four oxygen atoms: three at 1.98 Å and one at 2.35 Å (Li–O coordination number is 4.1, Table 3) (Fig. 6(a)). Similar tetrahedrally coordinated lithium sites have been reported in binary oxide39 and sulfide22,65 glasses, where asymmetric Li–O and Li–S correlation peaks indicate distorted tetrahedra. Upon incorporation of halide anions, the Li–O coordination number decreases from 4.1 to 3.1–3.2 (Table 5). The number of shorter Li–O bonds (1.95–1.98 Å) decreases from 2.9–3.2 to 2.3, whereas the longer Li–O bonds (2.30–2.36 Å) remain nearly unchanged (0.8–1.2). Simultaneously, lithium–halogen coordination numbers of 1.8, 2.1 (1.1 + 1.0), and 1.9 were obtained for the Cl100, Cl50Br50, and Br100 glasses, respectively. These results indicate that lithium cations occupy fivefold-coordinated sites in the halide-containing glasses, denoted as LiO3X2 (X = Cl and/or Br) (Fig. 6(b–d)).
The LiO3Cl2 (Fig. 6(b)) and LiO3Br2 (Fig. 6(d)) units are characteristic of the Cl100 and Br100 glasses, respectively, whereas LiO3ClBr units (Fig. 6(c)) are additionally present in the mixed-halide Cl50Br50 glass. Similar decreases in Li–O coordination number and the formation of Li–Cl bonds with increasing LiCl content have been reported for (LiCl)x(Li2O–2B2O3)1−x glasses by neutron diffraction with the aid of RMC modelling.66 The change in the lithium coordination environments in ternary and quaternary glasses has been observed exclusively through simulation studies; however, the present study directly reveals such changes using Li-specific PDF analysis with NDIS data. The change in lithium coordination numbers plays a pivotal role in enhancing ionic conductivity upon oxide–halide mixing. Incorporation of halide anions replaces one short Li–O bond (1.95–1.98 Å) with two longer Li–halogen bonds (2.45–2.46 Å for Li–Cl; 2.64 Å for Li–Br). According to bond-strength–bond-distance relationships,67,68 these longer Li–halogen bonds are weaker than Li–O bonds. This is also reflected in bond valence calculations, which relate bond strength to bond length.69,70 For example, when compared in crystalline lithium oxide (Li2O) and halides (LiCl and LiBr), the bond valence decreases from 0.236 for Li2O (Li–O distance: 2.00 Å) to 0.170 for LiCl (Li–Cl distance: 2.565 Å) and 0.141 for LiBr (Li–Br distance: 2.745 Å). Similarly, the single bond strengths calculated from the dissociation energies show that Li–Cl (115 kJ mol−1) and Li–Br (104 kJ mol−1)71 are weaker than Li–O (150 kJ mol−1).72 Consequently, replacing one short Li–O bond with two longer Li–halogen bonds reduces the local constraints on lithium cations, facilitating lithium-ion migration and enhancing ionic conductivity in the 1/3Li2O–1/3B2O3–1/3LiCl1–xBrx glasses. This interpretation is also consistent with the lower melting points of LiCl (605 °C) and LiBr (552 °C) compared with Li2O (1570 °C), suggesting that lithium cations bonded to halogens require less thermal energy to become mobile than those bonded to oxygens, despite LiCl or LiBr exhibiting Li–Cl/Br coordination numbers of 6 (higher than the Li–O coordination number of 4 in Li2O). The specific lithium coordination environments identified in the glasses (Fig. 6(b–d)) are rarely observed in crystalline materials, as the rigid glass network can sustain such metastable structural units.55,73–75 These unique local structures can provide useful insights into the structural design of glasses with high ionic conductivity.
3.6 Cause of the reduction in ionic conductivity upon mixing of halide anions
Lithium-ion conduction in glasses occurs through pathways formed by interconnected lithium-centred polyhedra.5,14–17,22,23,66 Therefore, understanding the connectivity of the lithium–oxygen–halogen polyhedra (Fig. 6(b–d)) is essential for elucidating the reduction in ionic conductivity in the 1/3Li2O–1/3B2O3–1/3LiCl1–xBrx glasses caused by halide–halide mixing.Schematic representations of clusters of LiO3X2 units in the Cl100, Cl50Br50, and Br100 glasses are shown in Fig. 6(e–g). In these figures, the network consisting of BO3 triangles and BO4 tetrahedra is omitted. In the Cl100 and Br100 glasses, lithium cations predominantly occupy LiO3Cl2 (Fig. 6(e)) or LiO3Br2 (Fig. 6(g)) polyhedra, forming continuous conduction pathways through fivefold-coordinated sites consisting of three oxide and two halide anions. In contrast, the Cl50Br50 glass contains LiO3Cl2, LiO3Br2, and LiO3ClBr units (Fig. 6(f)). The halide-mixed LiO3ClBr polyhedra create an intermediate local environment with slightly different lithium–halide bond lengths compared with the LiO3Cl2 and LiO3Br2 units. This structural diversity introduces a mismatch in the occupation energies of the lithium sites, which disrupts lithium-ion migration between different polyhedra. Molecular dynamics simulations of mixed alkali metasilicate glasses have demonstrated that alkali cations migrate along independent pathways and that the ionic conduction among the sites occupied by other types of alkali cations is restricted by energy mismatches.76,77 Similarly, in the Cl50Br50 glass, the variation in lithium coordination environments leads to a partial inhibition of lithium-ion conduction.
The mixed-halogen effect observed in this study differs from the classical mixed-alkali effect, where conductivity can decrease by several orders of magnitude.53–55 In the Cl50Br50 glass, the reduction in conductivity is only about half an order of magnitude (Fig. 1), because the substituted halide anions do not act as charge carriers (in contrast to the mixed-alkali effect, where the alkali cations functioning as carriers themselves are substituted). Therefore, the energy mismatch between lithium sites in mixed-halide glasses is smaller than that between different alkali cations in mixed-alkali glasses.
Traditionally, the mixed-anion effect has been associated with the combination of anions with different valences (e.g., oxide and halide), which improves ionic conductivity in glasses.7,33,34,51,52 However, this study demonstrates that mixing monovalent halides can reduce ionic conductivity. Li-specific PDF analysis combining NDIS data enabled the determination of the lithium-related intermediate-range structure formed by interconnected LiO3X2 units. This configuration was identified as the origin of the reduced lithium-ion conductivity in the mixed-halide Cl50Br50 glass.
Anion mixing has a long history in glass science for tuning material properties, and recent studies have extended this strategy to crystalline materials.78 Well-designed crystalline mixed-anion compounds exhibit unique coordination environments within long-range ordered atomic arrangements and enhanced physicochemical properties, such as pure hydride (H−) conduction in La2−x−ySrx+yLiH1−x+yO3−y oxyhydrides,79 correlated disorder in SrMO2N (M = Nb, Ta) perovskite oxynitrides,80 a significant spin–orbit interaction effect in bulk BiTeI,81 and exceptionally high lithium-ion conductivity in Li9.54Si1.74P1.44S11.7Cl0.3 solid electrolyte.82 Conversely, in glasses, the compositional and structural flexibility allows diverse lithium environments to form, as demonstrated in this study. Although designing new glasses with tailored structures and properties is still challenging, the insights from element-specific quantum-beam analyses, as demonstrated in this study, pave the way for future glass design.
4. Conclusions
In this study, we investigated the lithium-cation environment in 1/3Li2O–1/3B2O3–1/3LiCl1−xBrx glasses, which exhibit variations in ionic conductivity due to anion mixing. We found that oxide–halide mixing enhances ionic conductivity by an order of magnitude compared with LiBO2 glass, whereas halide–halide mixing reduces ionic conductivity to nearly half of the value observed prior to mixing. Lithium-specific coordination number analysis based on neutron diffraction with isotopic substitution for natLi- and nullLi-enriched glasses revealed that the conductivity enhancement in oxide–halide mixed glasses arises from the breaking of short lithium–oxygen bonds and the formation of longer lithium–halogen bonds, which weaken constraints on lithium mobility. In a halide–halide mixed glass, analysis of lithium–anion coordination numbers showed the formation of three distinct fivefold lithium–oxygen–halogen polyhedral units. The coexistence of these units forms a polyhedral network that hinders lithium-ion migration due to mismatched site occupation energies. These findings provide new insights into the structure–property relationships of lithium-ion conducting glasses and offer guidance for designing glassy electrolytes with improved ionic conductivity.Author contributions
Y. O. and Y. T. designed the study. The samples were prepared by H. H. and N. K. Electrical conductivity measurement was performed by N. K. The high-energy X-ray diffraction experiment was conducted by Y. O., Y. T., and S. K. The neutron diffraction experiment was conducted by Y. O., Y. T., and K. I. The obtained data were analysed by Y. O., Y. T., H. H., N. K., M. T. and S. K. Y. O. wrote the manuscript with input from all the authors.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
The data that support the findings of this study are presented in the paper and the supplementary information (SI) file and are available from the corresponding author upon reasonable request. Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta10123a.Acknowledgements
This work was partially supported by JSPS Grant-in-Aid for Transformative Research Areas (A) “Hyper-Ordered Structures Science” [grant numbers 20H05878 (to S. K.) and 20H05881 (to Y. O. and S. K.)]. The synchrotron radiation experiments were performed at BL04B2 of SPring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI) (Proposal number 2019B1233). The neutron experiments at the Materials and Life Science Facility of J-PARC were performed under a user program (Proposal number 2019B0082).References
- T. Minami, Y. Takuma and M. Tanaka, J. Electrochem. Soc., 1977, 124, 1659–1662 CrossRef CAS.
- T. Minami, H. Nambu and M. Tanaka, J. Am. Ceram. Soc., 1977, 60, 467–469 CrossRef CAS.
- Z. Wang, S. Luo, X. Zhang, S. Guo, P. Li and S. Yan, J. Non-Cryst. Solids, 2023, 619, 122581 CrossRef CAS.
- Y. Daiko, A. Sakuda, T. Honma and A. Hayashi, J. Ceram. Soc. Jpn., 2022, 130, 552–557 CrossRef CAS.
- N. Kamaya, K. Honma, Y. Yamakawa, M. Hirayama, R. Kanno, M. Yonemura, T. Kamiyama, Y. Kato, S. Hama, K. Kawamoto and A. Mitsui, Nat. Mater., 2011, 10, 682–686 CrossRef CAS PubMed.
- Y. Li, S. Song, H. Kim, K. Nomoto, H. Kim, X. Sun, S. Hori, K. Suzuki, N. Matsui, M. Hirayama, T. Mizoguchi, T. Saito, T. Kamiyama and R. Kanno, Science, 2023, 381, 50–53 CrossRef CAS PubMed.
- A. Levasseur, J. C. Brethous, J. M. Reau, P. Hagenmuller and M. Couzi, Solid State Ionics, 1980, 1, 177–186 CrossRef CAS.
- S. W. Martin and C. A. Angell, J. Non-Cryst. Solids, 1986, 83, 185–207 CrossRef CAS.
- M. G. Moustafa, K. M. A. Saron, M. Saad, M. S. Alqahtani, A. Qasem and A. S. Hassanien, Solid State Sciences, 2023, 141, 107212 CrossRef CAS.
- S. A. Feller, W. J. Dell and P. J. Bray, J. Non-Cryst. Solids, 1982, 51, 21–30 CrossRef CAS.
- T. M. Alam and R. K. Brow, J. Non-Cryst. Solids, 1998, 223, 1–20 CrossRef CAS.
- Y. Waseda, E. Matsubara, K. Sugiyama, I. K. Suh, T. Kawazoe, O. Kasu, M. Ashizuka and E. Ishida, Sci. Rep. Res. Inst., Tohoku Univ., Ser. A, 1990, 35, 19–33 CAS.
- J. Swenson, L. Börjesson and W. S. Howells, Phys. Rev. B, 1995, 52, 9310–9319 CrossRef CAS PubMed.
- J. Swenson, A. Matic, L. Börjesson and W. S. Howells, Solid State Ionics, 2000, 136–137, 1055–1060 CrossRef CAS.
- J. Swenson and S. Adams, Phys. Rev. Lett., 2003, 90, 155507 CrossRef PubMed.
- A. Hall, S. Adams and J. Swenson, Ionics, 2004, 10, 396–404 CrossRef CAS.
- S. Adams and J. Swenson, J. Phys.: Condens. Matter, 2005, 17, S87–S101 CrossRef CAS.
- A. Hall, S. Adams and J. Swenson, Phys. Rev. B, 2006, 74, 174205 CrossRef.
- F. Mizuno, A. Hayashi, K. Tadanaga and M. Tatsumisago, Solid State Ionics, 2006, 177, 2721–2725 CrossRef CAS.
- Y. Seino, M. Nakagawa, M. Senga, H. Higuchi, K. Takada and T. Sasaki, J. Mater. Chem. A, 2015, 3, 2756–2761 RSC.
- M. Murakami, K. Shimoda, S. Shiotani, A. Mitsui, K. Ohara, Y. Onodera, H. Arai, Y. Uchimoto and Z. Ogumi, J. Phys. Chem. C, 2015, 119, 24248–24254 CrossRef CAS.
- Y. Onodera, K. Mori, T. Otomo, M. Sugiyama and T. Fukunaga, J. Phys. Soc. Jpn., 2012, 81, 044802 CrossRef.
- K. Mori, T. Ichida, K. Iwase, T. Otomo, S. Kohara, H. Arai, Y. Uchimoto, Z. Ogumi, Y. Onodera and T. Fukunaga, Chem. Phys. Lett., 2013, 584, 113–118 CrossRef CAS.
- K. Ohara, A. Mitsui, M. Mori, Y. Onodera, S. Shiotani, Y. Koyama, Y. Orikasa, M. Murakami, K. Shimoda, K. Mori, T. Fukunaga, H. Arai, Y. Uchimoto and Z. Ogumi, Sci. Rep., 2016, 6, 21302 CrossRef CAS PubMed.
- T. Ohkubo, K. Ohara and E. Tsuchida, ACS Appl. Mater. Interfaces, 2020, 12, 25736–25747 CrossRef CAS PubMed.
- H. Yamada, K. Ohara, S. Hiroi, A. Sakuda, K. Ikeda, T. Ohkubo, K. Nakata, H. Tsukasaki, H. Nakajima, L. Temleitner, L. Pusztai, S. Ariga, A. Matsuo, J. Ding, T. Nakano, T. Kimura, R. Kobayashi, T. Usuki, S. Tahara, K. Amezawa, Y. Tateyama, S. Mori and A. Hayashi, Energy Environ. Mater., 2024, 7, e12612 CrossRef CAS.
- R. K. Guntu, Mater. Sci. Eng. B, 2020, 262, 114784 CrossRef CAS.
- J. Budida, C. S. Rao, N. R. Chand, R. K. Guntu and N. K. Mohan, J. Alloy Compd., 2025, 1037, 182433 CrossRef CAS.
- G. R. Kumar, M. K. Rao, T. Srikumar, M. C. Rao, V. R. Kumar, N. Veeraiah and C. S. Rao, J. Alloy Compd., 2018, 752, 179–190 CrossRef.
- R. K. Guntu, V. Venkatramu, C. S. Rao and V. R. Kumar, Opt. Mater., 2021, 113, 110876 CrossRef CAS.
- R. K. Guntu, Ceram. Int., 2025, 51, 16524–16538 CrossRef CAS.
- P. Zhang, C. Calahoo, J. Cao, A. Duval and L. Wondraczek, Glass Europe, 2025, 3, 125–146 CrossRef.
- B. Carette, M. Ribes and J. L. Souquet, Solid State Ionics, 1983, 9–10, 735–738 CrossRef CAS.
- M. Tatsumisago, N. Machida and T. Minami, J. Ceram. Assoc. Jpn., 1987, 95, 197–201 CrossRef CAS PubMed.
- V. F. Sears, Neutron Scattering, Part A, in Methods of Experimental Physics, ed. K. Sköld and D. L. Price, Academic Press, 1986, vol. 23, pp. 521–550 Search PubMed.
- S. Biggin and J. E. Enderby, J. Phys. C: Solid State Phys., 1981, 14, 3129–3136 CrossRef CAS.
- I. Petri, P. S. Salmon and H. E. Fischer, Phys. Rev. Lett., 2000, 84, 2413–2416 CrossRef CAS PubMed.
- P. S. Salmon and I. Petri, J. Phys.: Condens. Matter, 2003, 15, S1509–S1528 CrossRef CAS.
- J. Zhao, P. H. Gaskell, M. M. Cluckie and A. K. Soper, J. Non-Cryst. Solids, 1998, 232–234, 721–727 CrossRef CAS.
- O. Majérus, L. Cormier, G. Calas and A. K. Soper, Phy. B, 2004, 350, 258–261 CrossRef.
- L. Cormier, P. H. Gaskell, G. Calas, J. Zhao and A. K. Soper, Phys. Rev. B, 1998, 57, R8067–R8070 CrossRef CAS.
- K. Ohara, Y. Onodera, M. Murakami and S. Kohara, J. Phys.: Condens. Matter, 2021, 33, 383001 CrossRef CAS PubMed.
- S. Kohara, M. Itou, K. Suzuya, Y. Inamura, Y. Sakurai, Y. Ohishi and M. Takata, J. Phys.: Condens. Matter, 2007, 19, 506101 CrossRef.
- T. Otomo, K. Suzuya, M. Misawa, N. Kaneko, H. Ohshita, T. Fukunaga, K. Itoh, K. Mori, M. Sugiyama, M. Kameda, Y. T. Yamaguchi, K. Yoshida, Y. Kawakita, K. Maruyama, S. Shamoto, S. Takeda, S. Saitoh, S. Muto, J. Suzuki, I. Ino, H. Shimizu, T. Kamiyama, S. Ikeda, Y. Yasu, K. Nakayoshi, H. Senda, S. Uno and M. Tanaka, KENS Rep., 2011, 17, 28–36 Search PubMed.
- T. E. Faber and J. M. Ziman, Phil. Mag., 1965, 11, 153–173 CrossRef CAS.
- E. Lorch, J. Phys. C: Solid State Phys., 1969, 2, 229–237 CrossRef.
- R. L. Mozzi and B. E. Warren, J. Appl. Cryst., 1969, 2, 164–172 CrossRef CAS.
- M. Tatsumisago, K. Yoneda, N. Machida and T. Minami, J. Non-Cryst. Solids, 1987, 95–96, 857–864 CrossRef CAS.
- N. Tsuda, M. Tanida and T. Miyajima, AGC Res. Rep., 2018, 68, 8–12 Search PubMed.
- V. Montouillout, H. Fan, L. del Campo, S. Ory, A. Rakhmatullin, F. Fayon and M. Malki, J. Non-Cryst. Solids, 2018, 484, 57–64 CrossRef CAS.
- S. W. Martin, J. Am. Ceram. Soc., 1991, 74, 1767–1783 CrossRef CAS.
- H. Wada, M. Menetrier, A. Levasseur and P. Hagenmuller, Mater. Res. Bull., 1983, 18, 189–193 CrossRef CAS.
- J. O. Isard, J. Non-Cryst. Solids, 1969, 1, 235–261 CrossRef CAS.
- M. Tomozawa and V. McGahay, J. Non-Cryst. Solids, 1991, 128, 48–56 CrossRef CAS.
- Y. Onodera, Y. Takimoto, H. Hijiya, T. Taniguchi, S. Urata, S. Inaba, S. Fujita, I. Obayashi, Y. Hiraoka and S. Kohara, NPG Asia Mater., 2019, 11, 75 CrossRef CAS.
- K. Suzuya, Y. Yoneda, S. Kohara and N. Umesaki, Phys. Chem. Glasses, 2000, 41, 282–285 CAS.
- R. L. Mozzi and B. E. Warren, J. Appl. Cryst., 1970, 3, 251–257 CrossRef CAS.
- A. C. Hannon, D. I. Grimley, R. A. Hulme, A. C. Wright and R. N. Sinclair, J. Non-Cryst. Solids, 1994, 177, 299–316 CrossRef CAS.
- O. L. G. Alderman, C. J. Benmore and J. K. R. Weber, Appl. Phys. Lett., 2020, 117, 131901 CrossRef CAS.
- M. Tatsumisago, M. Takahashi, T. Minami, M. Tanaka, N. Umesaki and N. Iwamoto, J. Ceram. Assoc. Jpn., 1986, 94, 464–469 CrossRef CAS PubMed.
- D. Ravaine, J. Non-Cryst. Solids, 1980, 38–39, 353–358 CrossRef CAS.
- M. Atoji and W. N. Lipscomb, J. Chem. Phys., 1957, 27, 195 CrossRef CAS.
- A. Filipponi and P. D. Angelo, J. Chem. Phys., 1998, 109, 5356–5362 CrossRef CAS.
- H. A. Levy, P. A. Agron, M. A. Bredig and M. D. Danford, Ann. N. Y. Acad. Sci., 1960, 79, 762–780 CrossRef CAS.
- K. Itoh, M. Sonobe, M. Sugiyama, K. Mori and T. Fukunaga, J. Non-Cryst. Solids, 2008, 354, 150–154 CrossRef CAS.
- J. Swenson, L. Börjesson and W. S. Howells, Phys. Rev. B, 1998, 57, 13514–13526 CrossRef CAS.
- I. D. Brown and R. D. Shannon, Acta. Cryst., 1973, A29, 266–282 CrossRef.
- R. D. Shannon, Acta. Cryst., 1976, A32, 751–767 CrossRef CAS.
- I. D. Brown and D. Altermatt, Acta Cryst., 1985, B41, 244–247 CAS.
- I. D. Brown, Chem. Rev., 2009, 109, 6858–6919 CrossRef CAS PubMed.
- P. Cortona, Phys. Rev. B, 1992, 46, 2008–2014 CrossRef CAS PubMed.
- K.-H. Sun, J. Am. Ceram. Soc., 1947, 30, 277–281 CrossRef CAS.
- S. Kohara, K. Suzuya, K. Takeuchi, C. -K Loong, M. Grimsditch, J. K. R. Weber, J. A. Tangeman and T. S. Key, Science, 2004, 303, 1649–1652 CrossRef CAS PubMed.
- Y. Onodera, S. Kohara, H. Masai, A. Koreeda, S. Okumura and T. Ohkubo, Nat. Commun., 2017, 8, 15449 CrossRef CAS PubMed.
- T. Aoyagi, S. Kohara, T. Naito, Y. Onodera, M. Kodama, T. Onodera, D. Takamatsu, S. Tahara, O. Sakata, T. Miyake, K. Suzuya, K. Ohara, T. Usuki, Y. Hayashi and H. Takizawa, Sci. Rep., 2020, 10, 7178 CrossRef CAS PubMed.
- J. Habasaki, I. Okada and Y. Hiwatari, J. Non-Cryst. Solids, 1995, 183, 12–21 CrossRef CAS.
- J. Habasaki, I. Okada and Y. Hiwatari, J. Non-Cryst. Solids, 1996, 208, 181–190 CrossRef CAS.
- H. Kageyama, K. Hayashi, K. Maeda, J. P. Attfield, Z. Hiroi, J. M. Rondinelli and K. R. Poeppelmeier, Nat. Commun., 2018, 9, 772 CrossRef PubMed.
- G. Kobayashi, Y. Hinuma, S. Matsuoka, A. Watanabe, M. Iqbal, M. Hirayama, M. Yonemura, T. Kamiyama, I. Tanaka and R. Kanno, Science, 2016, 351, 1314–1317 CrossRef CAS PubMed.
- M. Yang, J. Oró-Solé, J. A. Rodgers, A. B. Jorge, A. Fuertes and J. P. Attfield, Nat. Chem., 2011, 3, 47–52 CrossRef CAS PubMed.
- K. Ishizuka, M. S. Bahramy, H. Murakawa, M. Sakano, T. Shimojima, T. Sonobe, K. Koizumi, S. Shin, H. Miyahara, A. Kimura, K. Miyamoto, T. Okuda, H. Namatame, M. Taniguchi, R. Arita, N. Nagaosa, K. Kobayashi, Y. Murakami, R. Kumai, Y. Kaneko, Y. Onose and Y. Tokura, Nat. Mater., 2011, 10, 521–526 CrossRef PubMed.
- Y. Kato, S. Hori, T. Saito, K. Suzuki, M. Hirayama, A. Mitsui, M. Yonemura, H. Iba and R. Kanno, Nat. Energy, 2016, 1, 16030 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





