Tailoring the electronic structure of FeRu-NC for efficient electrochemical oxidative dehydrogenation of L-ascorbic acid
Bing
Liu†
a,
Haoming
Zhong†
a,
Hina
Gul
a,
Hao
Peng
ac,
Xiaolei
Guo
 *ab,
Yu-Long
Xie
*ab,
Yu-Long
Xie
 d,
Minhua
Ai
d,
Minhua
Ai
 ac,
Xiangwen
Zhang
ac,
Ji-Jun
Zou
ac,
Xiangwen
Zhang
ac,
Ji-Jun
Zou
 ac and
Lun
Pan
ac and
Lun
Pan
 *ac
*ac
aKey Laboratory for Green Chemical Technology of the Ministry of Education, School of Chemical Engineering and Technology, National Industry-Education Platform for Energy Storage, Tianjin University, Tianjin 300072, China. E-mail: panlun76@tju.edu.cn
bSchool of Chemical Engineering and Technology, Hebei University of Technology, Tianjin 300401, China. E-mail: xlguo@hebut.edu.cn
cTJU Binhai Industrial Research Institute Co., Ltd, Tianjin 300452, China
dSchool of Chemistry and Materials Science, Qinghai Minzu University, Xining 810007, China
First published on 29th January 2026
Abstract
Biomass electrooxidation coupled with hydrogen production offers a promising route to overcome the sluggish kinetics and high energy cost of conventional water electrolysis. The electrocatalytic oxidation of L-ascorbic acid (AA) to high-value dehydroascorbic acid (DHA) is particularly attractive but requires efficient catalysts to facilitate the challenging two-step dehydrogenation process. Here, we report an FeRu-NC catalyst with excellent L-ascorbic acid oxidation reaction (AAOR) catalytic activity and selectivity. The FeRu-NC catalyst achieves a significantly lower overpotential of 10 mV at 10 mA cm−2 and delivers a high current density of 200 mA cm−2 at only 0.68 V vs. RHE, while maintaining stable operation for over 80 h. DHA is produced with high yield (∼95%), selectivity (∼97%), and faradaic efficiency (FE) (∼95%). XPS and XAFS studies and in situ IR spectroscopy reveal that the introduction of trace Ru modulates the electronic structure of Fe single-atom sites, enhancing the adsorption of key intermediates and promoting the second dehydrogenation step, presenting high AAOR activity and high selectivity for DHA.
1. Introduction
Hydrogen energy has emerged as a highly promising clean and sustainable energy carrier due to its high energy density and environmental benefits.1–5 Water electrolysis, powered by renewable electricity, is an emerging economically and environmentally friendly route for hydrogen production.6–8 However, its widespread application is limited by the high reaction energy barrier and sluggish kinetics of the anodic oxygen evolution reaction (OER), which involves a complex four-electron transfer process.9–11 Therefore, developing alternative anode reactions with superior kinetic and thermodynamic favorability over the OER has become a research focus.Recently, replacing the OER with the electrooxidation of biomass derivatives (e.g., alcohols and aldehydes) has gained significant attention.12,13 This strategy not only effectively reduces the reaction energy barrier but also produces high-value chemicals instead of low-value oxygen.14–19 Among various biomass derivatives, L-ascorbic acid (AA, Scheme 1a) stands out due to its unique enediol structure, where a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond is flanked by two hydroxyl groups. Its oxidation product, L-dehydroascorbic acid (DHA, Scheme 1a), holds considerable commercial value in the pharmaceutical, cosmetic, and food industries.20–22 Mechanistically, the hydroxyl group distant from the carbonyl group is readily dehydrogenated due to a p–π conjugation effect, forming a stable intermediate HA−.23–25 However, this stabilization renders the subsequent dehydrogenation of the carbonyl-adjacent hydroxyl group more challenging due to the strong electron-withdrawing effect of the carbonyl group. Therefore, to accelerate the dehydrogenation of the carbonyl-adjacent hydroxyl group, regulating the adsorption between active sites and the reaction intermediate HA− was considered. This regulation can enhance the p–π conjugation effect between the C–O bonds in the enediol structure and increase the polarity of the carbonyl-adjacent hydroxyl group on the other side, thereby achieving highly efficient dehydrogenation.26 Previous research has demonstrated that Fe exhibits a superior adsorption capacity towards intermediates HA− compared to the other transition metals such as Cu, Co and Ni.27 Consequently, in order to optimize the adsorption of Fe and HA− with greater precision, it may be advisable to incorporate a second metal atom into Fe-based catalysts. This approach can modulate the electronic structure and coordination environment of the Fe active sites,28,29 thereby tuning the adsorption properties of the target intermediates, accelerating the reaction kinetics and enhancing the overall catalytic activity.30–33 Therefore, the design of electrocatalysts is of paramount importance.34
C bond is flanked by two hydroxyl groups. Its oxidation product, L-dehydroascorbic acid (DHA, Scheme 1a), holds considerable commercial value in the pharmaceutical, cosmetic, and food industries.20–22 Mechanistically, the hydroxyl group distant from the carbonyl group is readily dehydrogenated due to a p–π conjugation effect, forming a stable intermediate HA−.23–25 However, this stabilization renders the subsequent dehydrogenation of the carbonyl-adjacent hydroxyl group more challenging due to the strong electron-withdrawing effect of the carbonyl group. Therefore, to accelerate the dehydrogenation of the carbonyl-adjacent hydroxyl group, regulating the adsorption between active sites and the reaction intermediate HA− was considered. This regulation can enhance the p–π conjugation effect between the C–O bonds in the enediol structure and increase the polarity of the carbonyl-adjacent hydroxyl group on the other side, thereby achieving highly efficient dehydrogenation.26 Previous research has demonstrated that Fe exhibits a superior adsorption capacity towards intermediates HA− compared to the other transition metals such as Cu, Co and Ni.27 Consequently, in order to optimize the adsorption of Fe and HA− with greater precision, it may be advisable to incorporate a second metal atom into Fe-based catalysts. This approach can modulate the electronic structure and coordination environment of the Fe active sites,28,29 thereby tuning the adsorption properties of the target intermediates, accelerating the reaction kinetics and enhancing the overall catalytic activity.30–33 Therefore, the design of electrocatalysts is of paramount importance.34
Here, we synthesized bimetallic FeRu-NC single-atom catalysts for the AAOR via a two-step adsorption-pyrolysis method based on ZIF-8. The resulting FeRu-NC catalyst exhibits remarkable AAOR activity, achieving an ultralow overpotential of 10 mV at 10 mA cm−2, outperforming monometallic Fe-NC (71 mV) and Ru-NC (60 mV), and the noble metal catalysts RuO2 (56 mV) and 20% Pt/C (45 mV). More importantly, this advantage is maintained at high current densities: the overpotential is only 130 mV at 100 mA cm−2, and a current density of 200 mA cm−2 is attained at a low potential of 0.68 V (vs. RHE). The catalyst also demonstrates excellent operational stability at this high current density, maintaining its performance over 80 hours of continuous AAOR testing. Product analysis confirms that the electrooxidation of AA selectively generates DHA with high conversion (∼98%), high selectivity (∼97%), and impressive yield and FE (both ∼95%). This work demonstrates that rational electronic structure modulation of FeRu-NC effectively promotes the AAOR via optimized intermediate adsorption, offering a promising strategy for designing high-performance catalysts toward energy-efficient biomass valorization coupled with hydrogen production.
2. Experimental section
2.1 Chemicals and materials
Zinc nitrate hexahydrate (Zn(NO3)2·6H2O, 99%), ferrocene (Fe(C5H5)2, 98%), ruthenocene (Ru(C5H5)2, 98%), anhydrous sodium sulphate (Na2SO4, AR, ≥99.5%), o-phenylenediamine (OPDA, C6H8N2, 99.5%), copper sulfate (CuSO4, AR) and L-ascorbic acid (AA, C6H8O6, 99.99%) were purchased from Shanghai Aladdin Biochemical Technology Company. 2-Methylimidazole (2-MeIm, C4H6N2, 98%) was purchased from Hangzhou Chuangshi Biotechnology Co., Ltd. Methanol (CH3OH, HPLC), sulfuric acid (H2SO4, AR), and isopropanol (C3H8O, AR) were bought from Tianjin Jiangtian Chemical Technology Co., Ltd. Nafion solution (5.0 wt%) was purchased from DuPont Company. Deionized water (18.25 MΩ cm) was produced by an Ultrapure Water System (HYP-QX-UP). Ruthenium oxide and 20 wt% Pt/C were bought from Shanghai McLean Biochemical Technology Co., Ltd. All of the materials were used without any further purification.2.2 Synthesis of electrocatalysts
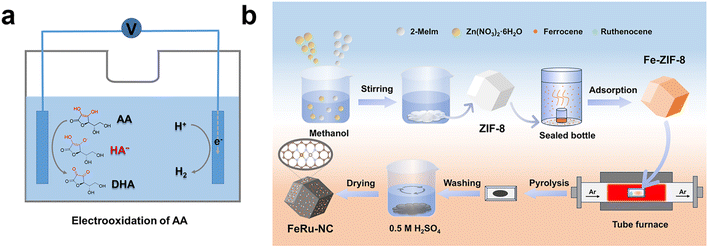
The FeRu-NC electrocatalysts were fabricated using a two-step adsorption-pyrolysis method, as illustrated in Scheme 1b. The synthesis commenced with the preparation of a ZIF-8 support. Specifically, Zn(NO3)2·6H2O (10 mmol) and 2-MeIm (40 mmol) were dissolved in methanol (80 mL) with vigorous stirring for 1 h and then stirring was continued at room temperature for 24 h. White ZIF-8 was collected by filtration, washed three times with methanol, and dried overnight at 60 °C under vacuum. Then, the obtained ZIF-8 was subjected to Fe incorporation via a vapor-phase infiltration process. The choice of ferrocene as an Fe source was motivated by its combination of a low sublimation temperature (∼100 °C) and a molecular diameter (∼4.5 Å) smaller than the pore aperture of ZIF-8 (∼6 Å), which allows ferrocene vapor to diffuse into the cavities, enabling high loading and dispersion of Fe single atoms.35 Specifically, 250 mg of ZIF-8 was loaded into a 100 mL sealed bottle, alongside a 5 mL glass bottle containing 50 mg of ferrocene, followed by heating at 155 °C for 9 h to obtain Fe-ZIF-8. The resulting Fe-ZIF-8 was subsequently subjected to a high-temperature pyrolysis process to construct the FeRu-NC single-atom catalyst. The Fe-ZIF-8 powder and ruthenocene were loaded in separate ceramic boats, positioned at the upstream and downstream locations, respectively, within a tube furnace. The furnace temperature was then raised from room temperature to 950 °C at a rate of 5 °C min−1 under an Ar atmosphere and then held at 950 °C for 3 h. During pyrolysis, the Zn species in the framework evaporated, generating abundant vacant sites. Concurrently, the sublimated ruthenocene vapor, carried by the Ar flow, uniformly infiltrated the pores of the Fe-ZIF-8. Meanwhile, the metallic Fe and Ru atoms were liberated from their metallocene precursors, enabling their coordination into the newly formed NC skeleton. After natural cooling to room temperature, the resulting product was stirred in a 0.5 M H2SO4 solution overnight to remove unstable species. The final FeRu-NC sample was then collected by filtration, washed repeatedly with methanol and deionized water, and dried at 60 °C under vacuum. For comparison, Fe-NC, Ru-NC, and NC catalysts were prepared using identical procedures without adding the Ru source, the Fe source, and both Fe and Ru sources, respectively.2.3 Catalyst characterization
The morphology of the catalyst was analyzed by Scanning Electron Microscope (SEM, HITACHI Regulus 8100) and Transmission Electron Microscope (TEM, JEOL JEM-F200) characterization. The crystal phase of the catalyst was analyzed by powder X-Ray Diffraction (XRD, Bruker AXS D8 FOCUS), Raman (HORIBA LabRAM HR Evolution), and X-ray Photoelectron Spectroscopy (XPS, Thermo K-Alpha+). The specific surface area and pore size distribution of the samples were evaluated by recording nitrogen adsorption–desorption isotherms using a BET Surface Area and Pore Size Analyzer (Micromeritics-ASAP 2460). Metal element content was tested by Inductively Coupled Plasma-Optical Emission Spectrometry (ICP-OES, Agilent ICP-OES 725 ES) and Inductively Coupled Plasma-Mass Spectrometry (ICP-MS, Agilent 7850). Product characterization and mechanism inference were analyzed using a Fourier Transform Infrared (FTIR) spectrometer (Bruker INVENIO S). The valence and coordination structures of the catalysts were analyzed by X-ray Absorption Fine Structure spectrometry (XAFS-500-A, Anhui Chuang Spectrum).2.4 Electrochemical measurement
The prepared catalyst (5 mg) was dispersed in a mixture of isopropanol (800 µL) and deionized water (180 µL), and the mixture was ultrasonicated for 1 h. Then, 20 µL of Nafion solution was added and then ultrasonicated for 0.5 h to obtain the catalyst ink. 10 µL of ink (0.25 mg cm−2) was taken using a pipette gun and dripped onto a glassy carbon electrode (0.5 cm in diameter), and the catalyst was allowed to dry naturally to obtain the working electrode. AA (17.6 g) and Na2SO4 (14.2 g) were dissolved in deionized water (100 mL), sonicated until completely dissolved, and transferred to a 100 mL electrolytic cell. AAOR electrochemical tests were all performed in 1 M Na2SO4 solution. A saturated calomel electrode and a Pt mesh electrode were used as the reference and counter electrodes, respectively. The electrochemical tests were performed at room temperature using a CHI660E electrochemical workstation. Linear sweep voltammetry (LSV) curves were obtained at a scan rate of 5 mV s−1 over the potential range of −0.1 to 1.0 V (vs. RHE) with 90% IR compensation (post-test correction). All RHE potentials were derived from the equation E (vs. RHE) = E (vs. SCE) + 0.0591 × pH + 0.2415. Electrochemical impedance (EIS) measurements were carried out at different potentials (0.4, 0.5, 0.6, and 0.8 V vs. RHE), and Nyquist plots were obtained by ZSimpWin fitting. A non-Faraday voltage window (0.07 to 0.37 V vs. RHE) was selected, and CV cycling was performed at scan rates of 20, 40, 60, 80, 100, and 120 mV s−1. Then, at the potential of 0.23 V (vs. RHE), take points at half the difference between the oxidation current density and the reduction current density, and the scan rate was plotted as a function of the corresponding current density. The function was linearly fitted to find the slope of the function to obtain the capacitance of the double layer (Cdl) to compare the electrochemically active area (ECSA). Finally, electrochemical stability was tested by chronopotentiometry (CP). The working electrode was prepared by depositing 130 µL of catalyst ink onto a 1 cm2 carbon paper substrate, yielding a catalyst loading of 0.65 mg cm−2. The measurements were conducted in a 100 mL electrolytic cell at room temperature, using a Pt mesh counter electrode and a saturated calomel reference electrode. A constant current density of 200 mA cm−2 was applied, and the electrolyte was replaced every 12 hours to maintain consistent reaction conditions.| ECSA = Cdl/Cs | (1) |
Moreover, the turnover frequency (TOF) was calculated to strengthen the intrinsic activity analysis. For the two-electron transfer process, the calculation formula is as follows:
| TOF = (j × A)/(2 × n × F) | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1) and n is the number of moles of the active materials.
485 C mol−1) and n is the number of moles of the active materials.
A constant potential test was conducted at various potentials. As the reaction proceeded, the current density decreased gradually and eventually approached zero. This indicated the completion of the electrochemical reaction. Then, the reactant (AA) and product (DHA) in the electrolyte were collected for analysis. AA was determined using a high-performance liquid chromatograph (HPLC, Agilent 1260) equipped with a C18 column (4.6 mm × 150 mm gasket packed GWS, 5 µm) and a UV-vis detector. The wavelength of the detector was set at 254 nm and the mobile phase was methanol–PBS buffer in a ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 at a flow rate of 0.5 mL min−1. Specifically, 10 µL of the electrolyte was removed during constant potential electrolysis and then diluted to 1 mL with deionized water and the resulting solution was analyzed by HPLC. DHA can be measured by its fluorescence emission due to the reaction with OPDA, whereas AA cannot. However, AA can be completely oxidized to DHA by CuSO4, allowing for indirect measurement.37 The product DHA was quantified using a fluorescence spectrophotometer (F-4600 FL Spectrophotometer), with an excitation wavelength of 369 nm and an emission wavelength of 430 nm. Electrolyte collected after complete electrooxidation of AA in a 20 mL electrolytic cell at different voltages was analyzed by fluorescence spectroscopy. For each sample, 10 µL of electrolyte was added to 10 mL of 0.08 mg mL−1 OPDA solution, reacted for 30 minutes, filtered, and then analyzed. A series of AA standard solutions of 0.5, 1.0, 1.5, 2.0 and 2.5 mg mL−1 were prepared. They were oxidized with 0.04 mg mL−1 CuSO4 for 15 min, followed by reaction with OPDA for 25 min. Subsequently, a standard calibration curve was plotted to conduct quantitative analysis.
98 at a flow rate of 0.5 mL min−1. Specifically, 10 µL of the electrolyte was removed during constant potential electrolysis and then diluted to 1 mL with deionized water and the resulting solution was analyzed by HPLC. DHA can be measured by its fluorescence emission due to the reaction with OPDA, whereas AA cannot. However, AA can be completely oxidized to DHA by CuSO4, allowing for indirect measurement.37 The product DHA was quantified using a fluorescence spectrophotometer (F-4600 FL Spectrophotometer), with an excitation wavelength of 369 nm and an emission wavelength of 430 nm. Electrolyte collected after complete electrooxidation of AA in a 20 mL electrolytic cell at different voltages was analyzed by fluorescence spectroscopy. For each sample, 10 µL of electrolyte was added to 10 mL of 0.08 mg mL−1 OPDA solution, reacted for 30 minutes, filtered, and then analyzed. A series of AA standard solutions of 0.5, 1.0, 1.5, 2.0 and 2.5 mg mL−1 were prepared. They were oxidized with 0.04 mg mL−1 CuSO4 for 15 min, followed by reaction with OPDA for 25 min. Subsequently, a standard calibration curve was plotted to conduct quantitative analysis.
The conversion of AA, the selectivity, the yield of DHA, and the FE were calculated using the following equations, respectively.
| AA conversion = [n(AA consumed)/n(initial AA)] × 100% | (3) |
| AA selectivity = [n(DHA formed)/n(AA consumed)] × 100% | (4) |
| DHA yield = [n(DHA formed)/n(initial AA)] × 100% | (5) |
| FE = [(m × n × F)/(I × t)] × 100% | (6) |
3. Results and discussion
3.1 Crystal and electronic structures of catalysts
As shown in the SEM and TEM images (Fig. 1a–d and S1), FeRu-NC, Fe-NC, Ru-NC, and NC all maintain a uniform dodecahedral morphology resembling that of ZIF-8 precursor, with particle sizes ranging from approximately 300 to 500 nm (Fig. S2). Notably, the framework of FeRu-NC and Fe-NC exhibits a slight degree of collapse in comparison to the pristine NC, which can be attributed to surface shrinkage induced by pyrolysis defects and metal reduction during the thermal treatment.38 TEM, high-resolution TEM (HRTEM) and scanning transmission electron microscopy (STEM) images (Fig. 1e–g) reveal that there are no discernible metal clusters or oxide particles detected within the carbon framework of the FeRu-NC catalyst, indicating that the metal species (Fe and Ru) are likely stabilized in the form of single atoms. This inference is further supported by EDS elemental mapping (Fig. 1h–k), which reveals uniform distribution of Fe, Ru, N, and C throughout the dodecahedral framework. The consistent results obtained from the control Fe-NC, Ru-NC and NC catalysts (Fig. S3–S5) confirm the general efficacy of our synthetic approach in fabricating M-NC catalysts for achieving atomic metal dispersion. Moreover, the actual metal loadings were quantitatively determined by ICP-OES to be 2.08 wt% for Fe and 0.16 wt% for Ru (Table S1), further corroborating the successful incorporation of both metals into the NC framework. | ||
| Fig. 1 Morphology and structure characterization. (a–d) SEM images of FeRu-NC, Fe-NC, Ru-NC and NC. (e) TEM, (f) HRTEM and (g) STEM images of FeRu-NC. (h–k) EDS mapping images of FeRu-NC. | ||
To further elucidate the phase composition of the catalysts, XRD analysis was performed. Initially, ferrocene is encapsulated into the ZIF-8 pores via thermal adsorption, wherein the resulting Fe-ZIF-8 composite exhibits an XRD pattern identical to that of pristine ZIF-8 (Fig. S6), confirming the preservation of the ZIF-8 framework. After pyrolysis, the XRD pattern of the FeRu-NC catalyst (Fig. 2a) displays only two broad peaks at approximately 26° and 44°, corresponding to the (002) and (101) planes of graphitic carbon.39 The absence of any discernible crystalline phases related to metals or metal oxides confirms the atomic dispersion of the metal species, which is consistent with the TEM and HRTEM observations.
 | ||
| Fig. 2 Chemical composition of FeRu-NC. (a) XRD patterns. (b) Raman spectra. (c) High-resolution XPS spectra of C 1s, (d) N 1s, (e) Fe 2p and (f) Ru 3p. | ||
The graphitic carbon structure was further probed by Raman spectroscopy (Fig. 2b). All samples show the characteristic D band (∼1330 cm−1, corresponding to the disordered carbon) and G band at (∼1575 cm−1, corresponding to the sp2-bonded graphitic carbon). The intensity ratio (ID/IG) is indicative of the graphitization degree.40 The ID/IG values for FeRu-NC (1.69) and Fe-NC (1.58) are lower than those for Ru-NC (2.87) and NC (2.72), suggesting that the incorporation of Fe promotes a higher degree of graphitization, which is beneficial for electrical conductivity and electrocatalytic activity. While metal doping can disrupt the ordered graphitic carbon structure,31,35 the slightly larger ID/IG ratio of FeRu-NC compared to Fe-NC indicates the presence of more structural defects. These defects may be more conducive to exposing active sites and enhancing catalytic activity. The porosity and specific surface area of the catalyst were analyzed by N2 adsorption–desorption (Fig. S7 and Table S2). The FeRu-NC catalyst possesses a large specific surface area of 659 m2 g−1 with an average pore size of 6.1 nm. In comparison, the NC sample exhibits the largest surface area of 1508 m2 g−1 and the smallest pore size of 3.1 nm. The observed reduction in surface area and increase in pore size originate from the framework collapse caused by Zn evaporation and metallocene decomposition during thermal treatment.
To elucidate the surface composition and chemical states, XPS spectroscopy was conducted (Fig. S8 and Table S3). The C 1s XPS spectra (Fig. 2c, S9a, S10a, and S11a) reveal that they have similar carbon structures, which aligns with the Raman results. The N 1s XPS spectra (Fig. 2d, S9b, S10b, S11b and Table S4) reveal the coexistence of nitrogen oxide, graphite, pyrrolyl nitrogen, and pyridine nitrogen, suggesting the presence of diverse nitrogen species. These different nitrogen configurations, including potential nitrogen defects, likely facilitate the anchoring of Fe and Ru atoms through coordination with available nitrogen lone pairs, thereby promoting metal stabilization and enhancing catalytic activity. Moreover, the pyridinic N peak shows a positive shift of 0.7 eV for FeRu-NC and 0.5 eV for Fe-NC, compared to that of NC (Fig. S12). In addition, the stronger shift (0.2 eV) of FeRu-NC than Fe-NC indicates that Ru doping makes the binding energy of N further enhanced, which may be due to the adsorption of N lone electron pairs by Ru, reducing the electron density around N and increasing the binding energy. Furthermore, the Fe 2p XPS spectra (Fig. 2e and S9c) indicate the presence of Fe2+ and Fe3+ states in both FeRu-NC and Fe-NC, with no metallic Fe(0) detected. Similarly, the Ru 3p XPS spectra (Fig. 2f and S10c) demonstrate that Ru primarily existed in the Ru4+ oxidation state in FeRu-NC and Ru-NC rather than the metal state. Compared to Fe-NC, a notable shift of approximately 0.2 eV towards higher binding energy is observed for the Fe 2p peaks in FeRu-NC. This shift can be attributed to the introduction of trace Ru, which possesses a higher electronegativity (χ ≈ 2.2) than Fe (χ ≈ 1.8). The more electronegative Ru attracts electron density from the coordinating nitrogen atoms, consequently reducing the electron density around the Fe centers and making them more electron-deficient. Consistent with this interpretation, quantitative analysis reveals a higher proportion of Fe3+ in FeRu-NC than Fe-NC (Table S5). Together, these results demonstrate that the electronic structure of the Fe sites is effectively modulated by the incorporation of trace Ru, a modification that is potentially favorable for boosting catalytic performance.
In order to accurately confirm the coordination number and electronic structure of the active Fe centers, X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) spectroscopy were performed at the Fe K absorption edge. The Fe K-edge XANES spectra (Fig. 3a) reveal the absence of metallic Fe. Furthermore, the absorption edge of FeRu-NC shifts to higher energy compared to that of Fe-NC, indicating a higher average valence state of Fe, which is consistent with the XPS results. In addition, a distinct front peak at approximately 7112 eV, a characteristic fingerprint of a square planar FeN4 structure, is observed.41 The appearance of this peak originated from an electric dipole-allowed jump of the 1s → 4pz orbitals accompanied by the ligand-to-metal charge transfer (LMCT) process, suggesting the predominant presence of a similar planar FeN4 structure in FeRu-NC.
The local coordination environment of Fe was further elucidated by the Fourier Transform EXAFS (FT-EXAFS) spectra of Fe in R-space (Fig. 3b). Both FeRu-NC and Fe-NC exhibit a dominant peak at approximately 1.5 Å, attributable to the Fe–N coordination peak. This result, combined with the XPS results, which clearly show a positive shift in the N binding energy, provides direct evidence for metal–nitrogen coordination. Furthermore, the absence of Fe–Fe coordination peaks (∼2.2 Å) and Fe–O coordination peaks (∼1.3 Å) rules out the presence of iron clusters and oxides in FeRu-NC and Fe-NC, consistent with XPS and XANES results. Quantitative analyses of Fe configurations are presented in Fig. 3c and Table S6, and the Fe K-edge FT-EXAFS spectrum of FeRu-NC fits well with the FeRuN6 structural model, wherein Fe and Ru atoms share two nitrogen atoms. In contrast, the Fe K-edge FT-EXAFS spectrum of Fe-NC is well-fitted with the FeN4 model (Fig. S13). These results collectively confirm that the introduced metal atoms are coordinated by nitrogen. To further reveal coordination information, Wavelet Transform (WT) analysis was performed on the EXAFS oscillations (Fig. 3d). The WT-EXAFS maximum intensity for both FeRu-NC and Fe-NC appears near k = 6.5 Å−1, while that for Fe foil occurs at 8.0 Å−1, further indicating their atomic dispersion. Moreover, a discernible shift in the k-value for FeRu-NC relative to that of Fe-NC provides direct evidence that Ru doping alters the local coordination environment and electronic structure of the Fe centers.
3.2 Electrochemical performance
In an acidic AA solution at pH = 2.3, the FeRu-NC catalyst exhibits excellent AAOR activity, achieving a current density of 10 mA cm−2 at a low overpotential of 10 mV. This performance significantly outperforms that of commercial 20% Pt/C (45 mV) and RuO2 (56 mV) catalysts (Fig. 4a, with 90% IR compensation). The uncompensated LSV is shown in Fig. S14. To confirm that the observed anodic current originates from the AAOR and not from the OER, a control experiment was conducted in an AA-free electrolyte (1 M Na2SO4, pH = 2.3). As shown in Fig. S15, the potential required to reach 10 mA cm−2 in this control experiment is nearly 1.6 V vs. RHE, which is approximately 1.1 V higher than the corresponding potential in the AA-containing solution, definitively attributing the low-potential current to the AAOR. The low operational potential of the AAOR represents a significant practical advantage. In contrast, anodic half-reactions involving the electrooxidation of most other biomass substrates typically necessitate much higher overpotentials, often resulting in a large working potential exceeding 1.23 V (Table S7).42–45 The substantially lower potential required in this work highlights the exceptional efficiency of the FeRu-NC-based system and its promising potential for practical applications, particularly in coupling with cathodic hydrogen evolution to reduce the overall energy input for hydrogen production.In order to elucidate the origin of this high performance, the catalytic properties of bimetallic FeRu-NC were compared with its monometallic counterparts, Fe-NC and Ru-NC. As shown in Fig. 4a, FeRu-NC requires a much lower overpotential to achieve a current density of 10 mA cm−2 than Fe-NC (71 mV) and Ru-NC (60 mV). At a higher current density of 100 mA cm−2, FeRu-NC maintains a substantial advantage, exhibiting an overpotential of only 130 mV, which is significantly lower than those of Fe-NC (470 mV), Ru-NC (410 mV), commercial Pt/C (230 mV), and RuO2 (490 mV) (Fig. S16). Moreover, the FeRu-NC catalyst achieves a high current density of 200 mA cm−2 at only 0.68 V (Table S8), substantially outperforming the commercial Pt/C catalyst (0.9 V). This demonstrates the potential of the FeRu-NC catalyst for practical industrial applications. The Tafel slope of FeRu-NC (79.9 mV dec−1) is smaller than that of Pt/C (96.8 mV dec−1), RuO2 (112.7 mV dec−1), Fe-NC (120.6 mV dec−1) and Ru-NC (103.4 mV dec−1), as shown in Fig. 4b, indicating faster AAOR reaction kinetics on the bimetallic catalyst. In order to assess the intrinsic activity, the Cdl and ECSA for FeRu-NC, Fe-NC and Ru-NC were calculated (Fig. 4c–f). It is found that FeRu-NC exhibits larger Cdl (22.2 mF cm−2) than Fe-NC (9.6 mF cm−2) and Ru-NC (16.2 mF cm−2), and the corresponding ECSA has the same trend (Fig. S17a). Furthermore, the LSV curves of normalized ECSA (Fig. S17b) show that FeRu-NC exhibits better jECSA than the other catalysts, proving its superior intrinsic catalytic activity. The intrinsic activity was further evaluated by comparing the TOF of FeRu-NC with that of Fe-NC (Fig. S18). It can be seen that FeRu-NC has the highest TOF (0.55 s−1) at an overpotential of 10 mV, better than Fe-NC (0.09 s−1), Ru-NC (0.11 s−1) commercial Pt/C (0.07 s−1) and RuO2 (0.009 s−1), which demonstrates that trace Ru doping significantly enhances the intrinsic activity of the FeRu-NC catalyst.
EIS was further employed to investigate the charge transfer kinetics. As shown in Fig. 4g and Table S9, the FeRu-NC catalyst exhibits the smallest charge transfer resistance among all synthesized and commercial catalysts at the same potential (0.6 V vs. RHE), indicating the most rapid charge transfer kinetics. This observation is consistent with its superior activity in the LSV results. To further probe the reaction kinetics of AAOR, EIS curves (Fig. S19) at different applied potentials were recorded. It reveals that in the low frequency region, the Nyquist plots show significant changes with increasing potentials and gradually change from a straight line to a semicircular curve when the potential exceeds 0.5 V (vs. RHE), indicating the presence of the AAOR process. As the potential increases, the semicircular curve of the AAOR becomes smaller, which indicates lower impedance of the AAOR and faster reaction kinetics. To further investigate the reaction kinetics of the AAOR, LSV measurements were performed in 1 M Na2SO4 electrolytes with varying AA concentrations (0 to 1.0 M), at a constant pH of 2.3 (adjusted with H2SO4). As shown in Fig. S20, the anodic current density of the FeRu-NC catalyst increased significantly with increasing AA concentration, demonstrating a pronounced concentration-dependent effect. This pronounced concentration dependence further corroborates that the measured current is directly linked to the AAOR. The long-term stability of catalysts under acidic conditions is a major challenge, especially for non-precious metal electrocatalysts. The durability of FeRu-NC for the AAOR was evaluated by chronopotentiometry (CP) at a constant current density of 200 mA cm−2 (Fig. 4h). The potential remained stable with no significant degradation over 80 hours, demonstrating excellent operational stability. Subsequently, the post-reaction catalyst was characterized in order to demonstrate the structural and compositional stability of the catalyst after a long-term AAOR process. The morphology of the catalyst characterized by SEM and TEM (Fig. S21 and S22) reveals that the distinctive dodecahedral morphology is well preserved after the long-term test, with metal species remaining highly dispersed and no signs of particle aggregation. The XRD pattern (Fig. S23) of the post-reaction catalyst shows no detectable peaks corresponding to metallic or oxide phases, confirming the preservation of stable active sites and the absence of phase changes. ICP-MS analysis of the post-reaction electrolyte (Fig. S24) reveals only minimal concentrations of leached metals, with concentrations of 1.64 µg L−1 for Fe and 0.02 µg L−1 for Ru. This provides direct evidence for the exceptional structural stability and minimal dissolution of the active components during electrocatalysis.
3.3 Product characterization of AA by electrocatalytic oxidation
Qualitative and quantitative analyses of the anodic AA oxidation products were carried out by HPLC, fluorescence spectroscopy and in situ IR spectroscopy. Chronoamperometric tests were conducted at different applied potentials to ensure the complete formation of the final oxidation product for subsequent analysis (Fig. S25). Then, the initial qualitative assessment by HPLC (Fig. S26) exhibits a prominent decrease in the peak intensity corresponding to AA (∼10 min), confirming the effective consumption of AA after the AAOR. For quantitative analysis and product identification, a fluorescence-based method was employed. This method is based on the selective reaction between DHA and OPDA to form a fluorescent derivative, whereas AA itself yields no detectable signal under identical conditions (Fig. S27a). To verify the selectivity of the electrooxidation process, a control experiment was performed in which AA was fully and selectively oxidized to DHA using CuSO4, a chemical oxidant known to convert AA to DHA with high specificity. The fluorescence intensity of the resulting DHA–OPDA derivative served as a reference, corresponding to complete conversion of AA to DHA. The nearly identical fluorescence intensity observed for the electrooxidized sample after the AAOR strongly indicates that the electrochemical reaction also converts AA to DHA with high selectivity and negligible generation of other by-products that could interfere with the assay. Based on this selective response, a calibration curve relating DHA concentration to fluorescence intensity was constructed (Fig. S27b and Table S10) for quantitative analysis.As shown in Fig. 5a, a DHA yield of 95% was achieved after 1.5 h of electrolysis at 1.2 V vs. RHE. As shown in Fig. 5b and Table S11, with increasing applied potential, the reaction time decreased markedly while maintaining near-complete conversion (∼98%), high selectivity (∼97%), and yields and FE exceeding 95%. These results collectively indicate that DHA is the primary product of AA electrooxidation, in agreement with in situ IR spectra (Fig. 6a). The emergence of a characteristic carbonyl peak at 1797 cm−1, which is ascribed to the product DHA, provides additional evidence for its formation.
3.4 Mechanism of the AA reaction by electrocatalytic oxidation
In the in situ IR spectra (Fig. 6), characteristic peaks corresponding to the bending and stretching vibrations of Fe–O bonds appear at 620 cm−1 and 3625 cm−1, respectively.46,47 As can be seen, the intensity of these Fe–O vibrational modes increases progressively with applied potential. Mechanistically, the emergence and enhancement of these signals can be attributed to the adsorption of dehydrogenated AA intermediates (e.g., HA−) onto the Fe active sites, forming Fe–O bonds. This bonding strengthens the conjugation within the enediol structure and increases the polarity of the remaining hydroxyl group, thereby facilitating further dehydrogenation steps. Notably, the Fe–O vibrational peaks are more pronounced in FeRu-NC than in Fe-NC. This indicates a stronger interaction between the Fe sites in FeRu-NC and the oxygen-containing intermediate (HA−), leading to more effective hybridization between Fe and O.Consequently, the Fe3+ centers in FeRu-NC exhibit a greater affinity for trapping the HA− intermediate, which is consistent with its enhanced catalytic activity. Integrating these observations with LSV, XPS and XAFS results, it can be concluded that Fe sites serve as the primary active centers during the AAOR process. The role of trace Ru is identified as a synergistic modulator of the FeN4 electronic structure, which elevates the high-spin Fe3+ content and fine-tunes intermediate adsorption energetics, ultimately boosting the catalytic activity.
4. Conclusion
In summary, an FeRu-NC electrocatalyst was successfully fabricated through a two-step adsorption-pyrolysis synthesis method based on ZIF-8. This bimetallic catalyst demonstrates prominent performance in anodic AA electrooxidation coupled with a cathodic hydrogen production process, achieving a remarkably low overpotential of 10 mV at 10 mA cm−2 and maintaining excellent operation stability for over 80 h at 200 mA cm−2. Furthermore, the FeRu-NC catalyst also maintains an overpotential of only 130 mV at 100 mA cm−2 and requires a low potential of 0.68 V (vs. RHE) to achieve a high current density of 200 mA cm−2, showing its potential in AAOR industrial applications. Product analysis confirms that AA is predominantly converted to DHA through electrooxidation with approximately 95% FE. XPS and XAFS studies reveal that the incorporation of trace Ru can modulate the electronic structure of the Fe active sites. Furthermore, in situ IR spectroscopy demonstrates that the introduction of Ru can regulate the chemical adsorption between Fe and reaction intermediates, facilitating further dehydrogenation, thus effectively enhancing catalytic activity. This work not only provides a feasible approach for designing high-performance AAOR catalysts via modulating the electronic structure of active sites but also contributes to the development of economical and efficient biomass valorization coupled with hydrogen production technologies.Conflicts of interest
There are no conflicts to declare.Data availability
Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request.The data supporting this article have been included as part of the manuscript and supplementary information (SI). Supplementary information: additional figures and experimental datasets for morphological and structural characterization, electrochemical performance tests, and product analysis. See DOI: https://doi.org/10.1039/d5ta10117d.
Acknowledgements
The authors appreciate the support from the National Natural Science Foundation of China (U24A20547, 22222808, and 22408272) and the Natural Science Foundation of Tianjin (25JCJQJC00120).References
- C. Tarhan and M. A. Cil, J. Energy Storage, 2021, 40, 102176 Search PubMed.
- T. T. Le, P. Sharma, B. J. Bora, V. D. Tran, T. H. Truong, H. C. Le and P. Q. P. Nguyen, Int. J. Hydrogen Energy, 2024, 54, 791–816 CrossRef CAS.
- B. E. Lebrouhi, J. J. Djoupo, B. Lamrani, K. Benabdelaziz and T. Kousksou, Int. J. Hydrogen Energy, 2022, 47, 7016–7048 Search PubMed.
- S. Sikiru, T. L. Oladosu, T. I. Amosa, J. O. Olutoki, M. N. M. Ansari, K. J. Abioye, Z. U. Rehman and H. Soleimani, Int. J. Hydrogen Energy, 2024, 56, 1152–1182 Search PubMed.
- F. Li, D. Liu, K. Sun, S. Yang, F. Peng, K. Zhang, G. Guo and Y. Si, Sustainability, 2024, 16, 1890 Search PubMed.
- J. Chi and H. Yu, Chin. J. Catal., 2018, 39, 390–394 Search PubMed.
- F. M. Sapountzi, J. M. Gracia, C. J. K. J. Weststrate, H. O. A. Fredriksson and J. W. H. Niemantsverdriet, Prog. Energy Combust. Sci., 2017, 58, 1–35 Search PubMed.
- S. R. Arsad, A. Z. Arsad, P. J. Ker, M. A. Hannan, S. G. H. Tang, S. M. Goh and T. M. I. Mahlia, Int. J. Hydrogen Energy, 2024, 60, 780–801 Search PubMed.
- N. T. Suen, S. F. Hung, Q. Quan, N. Zhang, Y. J. Xu and H. M. Chen, Chem. Soc. Rev., 2017, 46, 337–365 Search PubMed.
- K. Zhang and R. Zou, Small, 2021, 17, 2100129 Search PubMed.
- R. Qin, G. Chen, X. Feng, J. Weng and Y. Han, Adv. Sci., 2024, 11, 2309364 CrossRef CAS.
- J. Qi, Y. Xia, X. Meng, J. Li, S. Yang, H. Zou, Y. Ma, Y. Zhang, Y. Du, L. Zhang, Z. Lin and J. Qiu, Adv. Mater., 2025, 37, 2419058 CrossRef CAS.
- J. Li, X. Meng, X. Song, J. Qi, F. Liu, X. Xiao, Y. Du, G. Xu, Z. Jiang, S. Ye, S. Huang and J. Qiu, Adv. Funct. Mater., 2024, 34, 2316718 CrossRef CAS.
- H. Zhao, D. Lu, J. Wang, W. Tu, D. Wu, S. W. Koh, P. Gao, Z. J. Xu, S. Deng, Y. Zhou, B. You and H. Li, Nat. Commun., 2021, 12, 2008 CrossRef CAS PubMed.
- W. J. Liu, Z. Xu, D. Zhao, X. Q. Pan, H. C. Li, X. Hu, Z. Y. Fan, W. K. Wang, G. H. Zhao, S. Jin, G. W. Huber and H. Q. Yu, Nat. Commun., 2020, 11, 265 Search PubMed.
- L. Guo, X. Zhang, L. Gan, L. Pan, C. Shi, Z. F. Huang, X. Zhang and J. J. Zou, Adv. Sci., 2023, 10, 2205540 CrossRef CAS.
- H. Sun, L. Li, Y. Chen, H. Kim, X. Xu, D. Guan, Z. Hu, L. Zhang, Z. Shao and W. Jung, Appl. Catal., B, 2023, 325, 122388 CrossRef CAS.
- Z. Li, Y. Yan, S. M. Xu, H. Zhou, M. Xu, L. Ma, M. Shao, X. Kong, B. Wang, L. Zheng and H. Duan, Nat. Commun., 2022, 13, 147 CrossRef CAS.
- M. Amiri and S. Arshi, Electroanalysis, 2020, 32, 1391–1407 CrossRef CAS.
- M. Dosedel, E. Jirkovsky, K. Macakova, L. K. Krcmova, L. Javorska, J. Pourova, L. Mercolini, F. Remiao, L. Novakova and P. Mladenka, Nutrients, 2021, 13, 615 CrossRef CAS PubMed.
- J. M. Pullar, A. C. Carr and M. C. M. Vissers, Nutrients, 2017, 9, 866 CrossRef.
- H. S. Burdurlu, N. Koca and F. Karadeniz, J. Food Eng., 2006, 74, 211–216 CrossRef CAS.
- L. Huang, S. Tian, W. Zhao, K. Liu and J. Guo, Talanta, 2021, 222, 121645 CrossRef CAS.
- L. Stojanov and V. Mirceski, J. Electrochem. Soc., 2023, 170, 065504 CrossRef CAS.
- Y. Abe, S. Okada, H. Horii, S. Taniguchi and S. Yamane, J. Chem. Soc., Perkin Trans. 2, 1987, 715–720 RSC.
- C. Qiu, Q. Zhou, R. Gao, Y. Guo, J. Qin, D. Wang and Y. Song, Nanomaterials, 2023, 13, 2669 CrossRef CAS PubMed.
- Z. J. Chen, J. Dong, J. Wu, Q. Shao, N. Luo, M. Xu, Y. Sun, Y. Tang, J. Peng and H. M. Cheng, Nat. Commun., 2023, 14, 4210 CrossRef CAS PubMed.
- Q. Ling, Z. Zhao, Z. Li, K. Yan, C. Ding, P. Chen, Z. Sun, G. He, J. Lv and M. Zhang, J. Mater. Chem. A, 2023, 11, 2876–2888 RSC.
- L. Zhang, J. Yao, J. Zhang, W. He, Y. Li, L. Liang, C. Liu, H. Liu and Q. Hao, Catal. Sci. Technol., 2022, 12, 5435–5441 RSC.
- J. Cui, J. Min, H. Wang, J. Shui, L. Peng, Z. Kang, J. Liu, Q. Chen, S. Bai and Y. Liu, Ind. Chem. Mater., 2024, 2, 634–643 RSC.
- X. Zhou, S. Chu, Z. Jin, K. Hu, P. Liu, H. J. Qiu and X. Lin, ACS Mater. Lett., 2023, 5, 1656–1664 CrossRef CAS.
- L. Li, J. Zhu, F. Kong, Y. Wang, C. Kang, M. Xu, C. Du and G. Yin, Matter, 2024, 7, 1517–1532 CrossRef CAS.
- Q. Sun, X. Yue, L. Yu, F. Z. Li, Y. Zheng, M. T. Liu, J. Z. Peng, X. Hu, H. M. Chen, L. Li and J. Gu, J. Am. Chem. Soc., 2024, 146, 35295–35304 Search PubMed.
- Y. Du, X. Meng, Y. Ma, J. Qi, G. Xu, H. Zou and J. Qiu, Adv. Funct. Mater., 2024, 34, 2408013 Search PubMed.
- G. Ye, Q. He, S. Liu, K. Zhao, Y. Su, W. Zhu, R. Huang and Z. He, J. Mater. Chem. A, 2019, 7, 1658–16515 RSC.
- A. Karmakar and S. Kundu, Mater. Today Energy, 2023, 33, 101259 Search PubMed.
- S. Q. Gong, L. H. Zhao, F. Chen and F. Zhang, Res. Explor. Lab., 2012, 31, 19–21 CAS.
- J. Tian, Y. Zhu, X. Yao, L. Yang, C. Du, Z. Lv, M. Hou, S. Zhang, X. Ma and C. Cao, J. Mater. Chem. A, 2023, 11, 5288–5295 RSC.
- C. Liang, Y. Chen, M. Wu, K. Wang, W. Zhang, Y. Gan, H. Huang, J. Chen, Y. Xia, J. Zhang, S. Zheng and H. Pan, Nat. Commun., 2021, 12, 119 Search PubMed.
- E. Dervishi, Z. Ji, H. Htoon, M. Sykora and S. K. Doorn, Nanoscale, 2019, 11, 16571–16581 RSC.
- W. Liu, L. Zhang, X. Liu, X. Liu, X. Yang, S. Miao, W. Wang, A. Wang and T. Zhang, J. Am. Chem. Soc., 2017, 139, 10790–10798 CrossRef CAS PubMed.
- J. Li, R. Qiu, S. Zhang, L. Peng, Y. Dong, Y. Jiang, Y. Li, N. Fang, J. Yu, J. C. Dong, H. Zheng, L. Ding, J. Wan, I. Akpinar, J. Kuang, G. Chen, J. Ye, Y. Sun, L. Lin, S. Zheng, S. Yang, J. Li and J. F. Li, Adv. Mater., 2025, 37, 2417684 CrossRef CAS PubMed.
- R. Zhong, P. Wu, Q. Wang, X. Zhang, L. Du, Y. Liu, H. Yang, M. Gu, Z. C. Zhang, L. Huang and S. Ye, Green Chem., 2023, 25, 4674–4684 RSC.
- L. Zeng, Y. Chen, M. Sun, Q. Huang, K. Sun, J. Ma, J. Li, H. Tan, M. Li, Y. Pan, Y. Liu, M. Luo, B. Huang and S. Guo, J. Am. Chem. Soc., 2023, 145, 17577–17587 CrossRef CAS PubMed.
- S. Anantharaj, S. R. Ede, K. Karthick, S. S. Sankar, K. Sangeetha, P. E. Karthik and S. Kundu, Energy Environ. Sci., 2018, 11, 744–771 RSC.
- L. Z. Wang and Y. B. She, Spectrosc. Spectral Anal., 2009, 29, 694–697 CAS.
- S. Cui, X. D. Shen, B. L. Lin, G. D. Jiang and W. H. Zhang, J. Wuhan Univ. Technol., Mater. Sci. Ed., 2008, 23, 436–439 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |