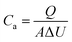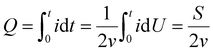A high-performance multifunctional three-electrode electrochromic device based on a Zn auxiliary electrode
Zinan
Zhao
 ,
Menghan
Tian
*,
Yue
Wang
,
Zelin
Lu
,
Menghao
Ma
,
Fan
Wang
and
Xiaolan
Zhong
,
Menghan
Tian
*,
Yue
Wang
,
Zelin
Lu
,
Menghao
Ma
,
Fan
Wang
and
Xiaolan
Zhong
 *
*
School of Physics, Beihang University, Beijing 100191, P. R. China. E-mail: zhongxl@buaa.edu.cn
First published on 5th February 2026
Abstract
Electrochromic materials exhibit electrically tunable optical properties and are widely applied in smart windows, optical switches, and photonic devices. However, the modulation amplitude of conventional electrochromic devices (ECDs) is fundamentally constrained by sluggish ion diffusion and incomplete redox reactions. Herein, we report a high-performance three-electrode electrochromic device (TRECD) incorporating a zinc (Zn) auxiliary electrode. The Zn auxiliary electrode can introduce additional Zn2+ into the device and participate in the electrochemical reaction, effectively enhancing the device performance. By optimizing the Li+/Zn2+ ratio in the electrolyte, the TRECD achieves substantially enhanced electrochemical and optical performance, including an 18-fold increase in areal capacitance, ultrafast response times (1.43 s for coloring and 2.72 s for bleaching), a record-high coloration efficiency of 603.37 cm2 C−1, and an exceptional optical modulation amplitude of 98.1%, reaching a minimum transmittance of 0.2%. This unprecedented contrast enables reversible switching between nearly full transparency and near-zero transmittance. Our results demonstrate that introducing an auxiliary Zn electrode and leveraging its coupled ion chemistry offers a powerful strategy for boosting the efficiency and functionality of electrochromic systems. This three-electrode structure opens new pathways for high-contrast optical switches, tunable filters, smart windows, and reconfigurable photonic devices.
1 Introduction
Electrochromic materials enable the reversible tuning of optical properties—such as transmittance, reflectance, and absorbance—through electrically driven ion insertion and extraction.1–3 Benefiting from advantages including bistability, wide modulation ranges, and low power consumption, electrochromic materials have been widely deployed in smart windows, optical filters, optical switches, and reconfigurable photonic systems. Their electrically tunable spectral response also provides an attractive approach for breaking the inherent limitations of traditional passive optical components, enabling dynamically adjustable plasmonic structures, tunable filters, and compact integrated photonic elements.4–8 These developments are crucial for advancing optical technologies toward intelligent, miniaturized, and multifunctional platforms.Despite significant progress, conventional electrochromic devices (ECDs) suffer from intrinsically slow ion diffusion kinetics and limited electrochemical activity, which constrain both the attainable optical modulation amplitude and the accessible spectral range.9–11 Numerous strategies have been proposed to enhance the reversibility and efficiency of ion–electron interactions in ECDs, including nanostructuring electrochromic films to shorten transport pathways,12 introducing mixed-ion electrolytes to diversify redox species,13 integrating hybrid pseudocapacitive materials,14–20 using a metal as the counter electrode to enhance the redox capability,1,21,22 the structural deformation induced by Jahn–Teller distortion leading to more excellent electrochromic properties,23 and employing shared-electrode or “rocking-chair” configurations.24–26 These approaches have led to notable improvements, with optical modulation amplitudes in optimized systems reaching approximately 50–80%. Nevertheless, achieving both high modulation amplitude and multispectral tunability simultaneously remains challenging, particularly for applications such as high-contrast optical switches and dynamic optical filters that require near-complete on/off optical states.
To overcome these limitations, we propose a three-electrode electrochromic device (TRECD) based on a zinc (Zn) auxiliary electrode. The Zn auxiliary electrode generates Zn2+ and participates in electrochemical reactions, significantly enhancing the device performance.
With the assistance of the Zn auxiliary electrode, the TRECD achieves an optical modulation amplitude as high as 98.1%, enabling a reversible transition from nearly full transparency to near-zero transmittance, which is superior to most reported ECDs (Fig. 1c).12–19,21,23–41 Additionally, the device exhibits an 18-fold enhancement in areal capacitance, fast coloring/bleaching times, and multicolor display capabilities. These results demonstrate that the synergistic action of the auxiliary Zn electrode and the three-electrode configuration provides an effective route to simultaneously boost the areal capacitance, switching speed, and optical tunability in electrochromic systems. This structural strategy offers a new design for next-generation high-performance electrochromic devices, facilitating broad applications in smart windows, reconfigurable optical filters, high-contrast optical switches, and dynamically programmable metasurfaces.
2 Results and discussion
2.1 The comparison of electrochromic performance between the TECD and the TRECD
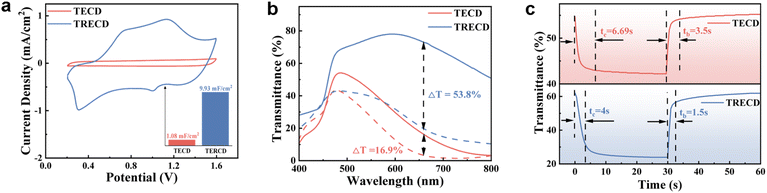
We prepared the traditional electrochromic device (TECD) by using polyaniline (PANI) as the positive electrode and tungsten oxide (WO3) as the negative electrode (Fig. 1a), and by inserting Zn as an auxiliary electrode between PANI and WO3 to construct a TRECD; the schematic diagram of the TRECD structure is shown in Fig. 1b. Zn is a common low-cost metal with high activity, strong reducibility, and volatile electronic properties, which is expected to effectively drive the completion of redox reactions. Fig. 2a shows the cyclic voltammetry (CV) curves of TECD and TRECD at a scan rate of 50 mV s−1. It can be seen that compared to the TECD, the CV curve envelope area of the TRECD is significantly larger. The areal capacitance of the devices is calculated based on the CV curves, which is defined as:where Q represents the inserted or extracted charge from the device, A represents the effective area of the device, and ΔU is the voltage window. The charge Q of the device is typically calculated from the CV curve as follows:
where S represents the area enclosed by the CV curve and v represents the voltage sweep rate. We can see from the calculation that the areal capacitance of the TECD is 1.08 mF cm−2, while that of the TRECD is 9.93 mF cm−2, representing a significant increase of more than nine times. The TRECD still maintains 8.07 mF cm−2 at a scan rate of 100 mV s−1, corresponding to a capacitance retention of 77.75%, indicating that the TRECD possesses excellent rate capability (Fig. S1).42Fig. 2b shows the transmittance curves of the TECD and TRECD in the colored and bleached states. The devices bleach under a voltage of 0.2 V and color under a voltage of 1.6 V. The optical modulation amplitude of the TECD is only 16.9%, without an obvious visual color change, whereas the optical modulation amplitude of the TRECD is 53.8%.
The coloration efficiency (CE) is a practical parameter that can be used to estimate the energy conversion efficiency of an electrochromic device. The ECDs with a higher CE mean that higher optical modulation can be achieved with less injected charge consumption. According to calculations (SI2), the CE of the TECD is 76.24 cm2 C−1 (Fig. S2a), while the TRECD is increased to 95.32 cm2 C−1 (Fig. S2b). The response time is an important parameter for evaluating the performance of electrochromic devices, defined as the time required for the optical change during the coloring or bleaching process to reach 90% of the maximum modulation amplitude. The coloring/bleaching times of the TECD are 6.69 s/3.5 s, while those of the TRECD are decreased to 4 s/1.5 s (Fig. 2c). As can be seen, compared with the TECD, the TRECD can provide higher areal capacitance, greater optical modulation amplitude, faster response time, and higher coloration efficiency, showing significant improvement in both electrochemical and electrochromic properties.
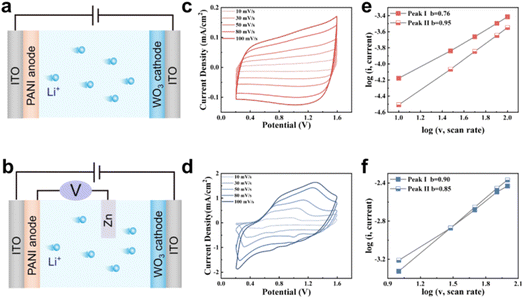
To further compare the performance of the TECD (Fig. 3a) and TRECD (Fig. 3b), we measured the CV curves of TECD and TRECD at different scan rates. The CV curves of the TECD are relatively smooth without obvious redox peaks, and the shape does not change with increasing scan rate (Fig. 3c). In contrast, the CV curves of the TRECD exhibit distinct redox peaks, and the peak current intensity increases continuously with increasing scan rate. Moreover, within the scan rate range of 10–80 mV s−1, the redox peak positions shift toward higher voltages as the scan rate increases, indicating that the TRECD shows favorable ion transport properties (Fig. 3d). When the scanning rate is increased to 100 mV s−1, the shape of the cathode peak changes significantly, and the growth rate of peak current slows down significantly. This indicates that the device exhibits a certain degree of electrochemical deactivation at high scan rates, leading to performance degradation, which may be due to irreversible damage to the surface structure of the electrochromic film caused by rapid ion insertion and extraction processes.43
To reveal the electrochemical kinetics of the device, we further investigated the redox peak currents of the TECD and TRECD at different scan rates. The peak current i in the CV curves follows a mathematical relationship with the scan rate:
| i = abv, |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i = b × log i = b × log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v + log v + log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a, a, |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i and log
i and log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v. When b = 0.5, the electrochemical reaction is mainly controlled by ion diffusion; when b = 1, the process is dominated by surface capacitive contributions; when b is between these values, the reaction is controlled by both factors.44–46 We fitted the linear relationship between log
v. When b = 0.5, the electrochemical reaction is mainly controlled by ion diffusion; when b = 1, the process is dominated by surface capacitive contributions; when b is between these values, the reaction is controlled by both factors.44–46 We fitted the linear relationship between log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i and log
i and log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v based on CV curves at different scan rates, as shown in Fig. 3e and f. It can be seen that the b-value for the cathode of the TECD is 0.95, while for the TRECD it is 0.85, indicating that the redox processes in both the TECD and TRECD are jointly controlled by ion diffusion and surface capacitive behavior. Both PANI and WO3 exhibit good compatibility with the Zn auxiliary electrode (Fig. S3). The reaction process of TECD is more influenced by capacitively controlled conditions compared with the TRECD.
v based on CV curves at different scan rates, as shown in Fig. 3e and f. It can be seen that the b-value for the cathode of the TECD is 0.95, while for the TRECD it is 0.85, indicating that the redox processes in both the TECD and TRECD are jointly controlled by ion diffusion and surface capacitive behavior. Both PANI and WO3 exhibit good compatibility with the Zn auxiliary electrode (Fig. S3). The reaction process of TECD is more influenced by capacitively controlled conditions compared with the TRECD.
To investigate the reaction mechanism of TRECD, we conducted XPS characterization on the WO3 electrode (Fig. S4). The upper plot of Fig. S4 shows the XPS spectrum of the WO3 electrode in its original state, the middle plot presents the XPS spectrum of the electrode in the colored state after 100 cycles of CV testing at the scan rate of 100 mV s−1, and the bottom plot presents the XPS spectrum in the bleached state. The test results indicate that no Zn element was detected in the WO3 film in its initial state; after 100 CV cycles, distinct Zn 2p characteristic peaks appeared in the spectrum of the WO3 film in the colored state, whereas no obvious peaks were detected in the bleached state. This phenomenon confirms that Zn2+ was dissolved from the Zn electrode and participated in the redox reaction. It can also be seen from the CV curve in Fig. 2a that the redox peaks observed at approximately 0.305 V/1.135 V correspond to the redox reactions of Zn2+.40 This may be attributed to the high chemical activity of Zn, which undergoes an electron loss process under the applied voltage and is oxidized to Zn2+. Therefore, the redox reaction of the TRECD originates from the combined action of Li+ in the electrolyte and Zn2+ dissolved from the Zn electrode. This mechanism significantly promotes more efficient ion injection and extraction processes.
2.2 The mechanism of the TRECD
To further investigate the role of Zn auxiliary electrodes in the redox reaction of TRECD, we designed two comparative devices: one with 0.1 M Zn(ClO4)2 added to the electrolyte of TECD, denoted as Zn-TECD, and the other maintaining the TRECD structure in which the Zn electrode is in an open-circuit state; the schematic diagram of the device structure is shown in Fig. 4a and b, respectively. As shown in Fig. 4c, the CV curves of Zn-TECD exhibit a modified shape compared to TECD, which can be attributed to the participation of Zn2+ from the electrolyte in the redox reactions. However, due to the high impedance of the organic solvent electrolyte, resulting in low ion diffusion rates, the CV curves' envelope area did not increase significantly compared to the TECD. Consequently, the areal capacitance of Zn-TECD showed no notable improvement, reaching only 2.37 mF cm−2 at a scan rate of 10 mV s−1. As shown in Fig. 4d and f, when the Zn electrode only exists as a structural component in the device in an open-ciruit state, both the CV curves' shape and areal capacitance are nearly the same as the TECD, indicating that the Zn electrode does not substantially affect the device performance under this configuration.Fig. 4g and h show the response speed and optical modulation amplitude of the two devices, respectively, which indicate no significant enhancement in either performance. Moreover, the Zn-TECD exhibits longer coloring/bleaching times, which may be due to the larger hydrated radius of Zn2+ compared to Li+. As a result, the ion insertion and extraction processes are more difficult, leading to a slower response speed. In terms of coloration efficiency, the Zn-TECD achieves a value of 67.53 cm2 C−1 (Fig. S5a), while the TRECD in which the Zn electrode is in an open-circuit state achieves a value of 74.94 cm2 C−1 (Fig. S5b). Compared to the coloration efficiency of the TECD of 76.24 cm2 C−1 (Fig. S2a), neither device demonstrates significant improvement. These results further demonstrate that simply introducing a Zn electrode or Zn2+ does not effectively improve the performance of electrochromic devices. It is only by utilizing the Zn electrode as an auxiliary electrode in a working state that the ion diffusion rate can be enhanced, promoting more complete insertion and extraction of ions and significantly improving the performance of the TRECD.
2.3 The performance optimization of the TRECD
We have previously demonstrated that during the reaction process of the TRECD, the Zn electrode undergoes oxidation to Zn2+. These Zn2+ subsequently participate in the reaction alongside Li+ to synergistically enhance the device performance. Therefore, doping with a certain concentration of Zn2+ is expected to further improve the performance of the TRECD. The electrochemical performance of TRECDs with different Zn2+ doping concentrations is shown in Fig. 5. The schematic diagram of the TRECD with Zn2+ doping in the electrolyte is shown in Fig. 5a. Fig. 5b shows the CV curves measured at a scan rate of 50 mV s−1. We can see that the concentration of Zn2+ increases, and the redox peak gradually shifts towards lower voltages, showing a polarization trend at the voltage boundary around 0.2 V. This phenomenon may be attributed to the larger hydrated radius of Zn2+. Excessive concentration of Zn2+ leads to increased resistance during ion insertion and extraction processes, thereby affecting the reaction kinetics. At a scan rate of 10 mV s−1, the areal capacitance of 0.1-TRECD reaches as high as 22.29 mF cm−2, while those of 0.3-TRECD and 0.5-TRECD are 11.67 mF cm−2 and 11.3 mF cm−2, respectively. Compared to Zn-TECD, the areal capacitance of 0.1-TRECD increases more than 9-fold, and it increases more than 8-fold compared to the TECD, representing a highly significant enhancement (Fig. 5c). As can be seen from Fig. 5d, the transmittance of 0.1-TRECD in the colored state at a wavelength of 633 nm is 1.6%, and in the bleached state it is 99.7%; the optical modulation amplitude of 0.1-TRCED is as high as 98.1%. The transmission spectra of TRECD with three concentrations of Zn2+ in the colored state show little difference, indicating that ions have achieved sufficient embedding during the reaction process. However, the transmittance of 0.5-TRCED significantly decreases in the bleached state, which may be due to the larger hydrated radius of Zn2+, making it difficult to completely remove them from the electrochromic film at high Zn2+ concentrations, resulting in a decrease in the transmittance of the bleached state. In addition, there has been a significant improvement in the coloration efficiency of 0.1-TRCED of 603.37 cm2 C−1 (Fig. 5e), which is nearly 8-fold higher than the coloration efficiency of 76.24 cm2 C−1 of the TECD (Fig. 5f). Benefiting from the rapid coloring/bleaching speed and excellent optical properties ranging from full transparency to near-zero transmittance in the visible light band, the 0.1-TRECD holds promising applications in smart windows. We present the photographic images of the 0.1 M TRECD under different voltages (Fig. S6), where the device exhibits coloration states ranging from fully transparent to yellow, light green, dark green, and ultimately to opaque deep blue, demonstrating excellent electrochromic performance. To expand the practical application scenarios, a large-area 0.1 M TRECD (5 cm × 8 cm) was fabricated for physical demonstration, showing significant differences between colored and bleached states, thereby validating its practical application value. It can be seen that the large-area TRECD exhibits significant differences in color and optical transmittance between the colored and bleached states (Fig. 5g).3 Conclusions
In summary, we have developed a high-performance electrochromic device by introducing a Zn auxiliary electrode. The Zn auxiliary electrode can provide additional Zn2+ to participate in the electrochemical reaction, and the synergistic effect of Zn2+ and Li+ significantly enhances the device performance. Compared with conventional two-electrode devices, the TRECD exhibits substantially enhanced electrochemical and optical performance, including an optical modulation amplitude of up to 98.1%, an areal capacitance of 22.29 mF cm−2, fast response times of 1.43 s for coloring and 2.72 s for bleaching, and an exceptionally high coloration efficiency of 603.37 cm2 C−1. Mechanistic analysis confirms that the performance enhancement originates from the synergistic effect of Zn2+ dissolution provided by the three-electrode structure, which facilitates rapid and efficient ion insertion/extraction. Furthermore, the device demonstrates excellent multicolor tunability and scalability, as evidenced by the fabrication of a large-area TRECD (5 cm × 8 cm) with uniform switching behavior. This work provides a new structural and mechanistic framework for designing next-generation electrochromic devices. The demonstrated high contrast and fast switching highlight the strong potential of this three-electrode strategy for applications in smart windows, adaptive optical filters, high-speed optical switches, photonic chips, and multifunctional optoelectronic systems.4 Experimental
4.1 Preparation of the polyaniline (PANI) electrode
The ITO glass substrates were sequentially subjected to ultrasonic cleaning in acetone, anhydrous ethanol, and deionized water for 15 minutes each to remove organic contaminants and surface impurities. After blow-drying with high-purity nitrogen, the PANI film was electrodeposited on the glass/ITO substrate through a three-electrode electrochemical cell. The resistance of the ITO was 7–10 ohms. The deposition electrolyte consisted of 0.5 M aniline monomer and 1 M sulfuric acid, which was freshly prepared and deaerated with nitrogen for 30 minutes prior to use. The reference electrode was Ag/AgCl, and a 2 cm × 2 cm platinum (Pt) sheet was used as the counter electrode. The PANI film was deposited for 400 s at 0.75 V using the potentiostatic technique. Then, all deposited PANI films were washed with deionized water and dried on a hot plate at 50 °C for 1 h. The thickness of the PANI film was 325 nm, as shown in Fig. S7a.4.2 Preparation of the tungsten oxide (WO3) electrode
The ITO glass substrates were sequentially subjected to ultrasonic cleaning in acetone, anhydrous ethanol, and deionized water for 15 minutes each to remove organic contaminants and surface impurities. After blow-drying with high-purity nitrogen, the WO3 film was electrodeposited on the glass/ITO substrate through a three-electrode electrochemical cell. The resistance of the ITO was 7–10 ohms. 125 mL of deionized water was measured using a graduated cylinder, and H2O2 and 0.8 mL of concentrated sulfuric acid were added with continuous stirring. After stirring for half an hour, the precursor solution of WO3 was obtained, which was freshly prepared and deaerated with nitrogen for 30 minutes prior to use. The reference electrode was Ag/AgCl, and a 2 cm × 2 cm Pt sheet was used as the counter electrode. The WO3 film was deposited for 900 s at −0.4 V using the potentiostatic technique. Then, all deposited WO3 films were washed with deionized water and dried on a hot plate at 50 °C for 1 h. The thickness of the WO3 film was 219 nm, as shown in Fig. S7b.4.3 Preparation of the TECD
Lithium perchlorate (LiClO4) was dissolved in propylene carbonate (PC) to prepare a 1 mol per L LiClO4/PC electrolyte solution. The prepared electrolyte was then uniformly mixed with a UV-curable adhesive at a volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode and WO3 as the negative electrode. The device was irradiated under ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the TECD. The active area of the device was 1.5 cm × 1.5 cm.
1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode and WO3 as the negative electrode. The device was irradiated under ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the TECD. The active area of the device was 1.5 cm × 1.5 cm.
4.4 Preparation of the TRECD
LiClO4 was dissolved in PC to prepare a 1 mol per L LiClO4/PC electrolyte solution. The prepared electrolyte was then uniformly mixed with a UV-curable adhesive at a volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode, WO3 as the negative electrode, and Zn as the auxiliary electrode. The Zn electrode used was a commercially purchased high-purity zinc plate with a Zn purity of ≥99.99%, and its effective area in the TRECD was 0.2 mm × 0.5 mm. The Zn was sandwiched between the positive and negative electrodes without making contact with them, and the distances between the Zn electrode and the PANI/WO3 electrodes were both 1 mm. The electrodes were then irradiated with ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the TRECD. The active area of the device was 1.5 cm × 1.5 cm.
1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode, WO3 as the negative electrode, and Zn as the auxiliary electrode. The Zn electrode used was a commercially purchased high-purity zinc plate with a Zn purity of ≥99.99%, and its effective area in the TRECD was 0.2 mm × 0.5 mm. The Zn was sandwiched between the positive and negative electrodes without making contact with them, and the distances between the Zn electrode and the PANI/WO3 electrodes were both 1 mm. The electrodes were then irradiated with ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the TRECD. The active area of the device was 1.5 cm × 1.5 cm.
4.5 Preparation of the Zn-TECD
LiClO4 and zinc perchlorate (Zn(ClO4)2) were dissolved in PC to prepare a 1 mol L−1 + 0.1 mol L−1 LiClO4 & Zn(ClO4)2/PC electrolyte solution. The prepared electrolyte was then uniformly mixed with a UV-curable adhesive at a volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode and WO3 as the negative electrode. The device was irradiated under ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the Zn-TECD. The active area of the device was 1.5 cm × 1.5 cm.
1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode and WO3 as the negative electrode. The device was irradiated under ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining the Zn-TECD. The active area of the device was 1.5 cm × 1.5 cm.
4.6 Preparation of TRECDs with different doping ratios
LiClO4 and Zn(ClO4)2 were dissolved in PC to prepare 1 mol L−1 + 0.1 mol L−1 (0.3 mol L−1 and 0.5 mol L−1) LiClO4 & Zn(ClO4)2/PC electrolyte solutions. The prepared electrolyte was then uniformly mixed with a UV-curable adhesive at a volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode, WO3 as the negative electrode, and Zn as the auxiliary electrode. The Zn was sandwiched between the positive and negative electrodes without making contact with them. The electrodes were then irradiated with ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining 0.1-TRECD, 0.3-TRECD, and 0.5-TRECD, respectively. The active area of the device was 1.5 cm × 1.5 cm.
1. The model of the UV-curable adhesive used was UV 7000. The device was assembled using this resulting electrolyte, with PANI as the positive electrode, WO3 as the negative electrode, and Zn as the auxiliary electrode. The Zn was sandwiched between the positive and negative electrodes without making contact with them. The electrodes were then irradiated with ultraviolet light for 15 minutes to cure the UV adhesive, thus obtaining 0.1-TRECD, 0.3-TRECD, and 0.5-TRECD, respectively. The active area of the device was 1.5 cm × 1.5 cm.
4.7 Electrochromic and electrochemical measurements
The voltammetry cycle (CV) was measured with an electrochemical workstation (CHI660E, Shanghai Chenhua), and the in situ transmittance of the sample was measured using an ultraviolet-visible spectrometer (TU-1810, PERSEE). The coloring/bleaching times were measured by a combination of an ultraviolet-visible spectrometer (TU-1810, PERSEE) and an electrochemical workstation (CHI660E, Shanghai Chenhua). X-ray photoelectron spectroscopy (XPS) was measured with an X-ray photoelectron spectrometer (Thermo Scientific K-Alpha, America), and the excitation source was Al Kα radiation (1486.6 eV) with a beam spot size of 400 µm.Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information: Fig. S1–S7 and further experimental details. See DOI: https://doi.org/10.1039/d5ta09759b.Acknowledgements
This work was supported by the National Natural Science Foundation of China (62505014 and U23A20481). We are grateful to the Atomic-Scale In Situ Fabrication Platform of the Analysis & Testing Center of Beihang University for the facilities and the scientific and technical assistance.Notes and references
- H. Li, W. Zhang and A. Y. Elezzabi, Adv. Mater., 2020, 32, 2003574 CrossRef CAS PubMed.
- Y. Wang, H. Nie, J. Han, Y. An, Y.-M. Zhang and S. X.-A. Zhang, Light: Sci. Appl., 2021, 10, 33 CrossRef CAS PubMed.
- J.-L. Wang, Y.-R. Lu, H.-H. Li, J.-W. Liu and S.-H. Yu, J. Am. Chem. Soc., 2017, 139, 9921–9926 CrossRef CAS PubMed.
- T. Xu, E. C. Walter, A. Agrawal, C. Bohn, J. Velmurugan, W. Zhu, H. J. Lezec and A. A. Talin, Nat. Commun., 2016, 7, 10479 CrossRef CAS PubMed.
- E. Hopmann and A. Y. Elezzabi, Nano Lett., 2020, 20, 1876–1882 CrossRef CAS PubMed.
- G. Kim, D. Kim, S. Ko, J.-H. Han, J. Kim, J. H. Ko, Y. M. Song and H.-H. Jeong, Microsyst. Nanoeng., 2024, 10, 22 Search PubMed.
- M. Tian, B. Liu, Z. Lu, Y. Wang, Z. Zheng, J. Song, X. Zhong and F. Wang, Light: Sci. Appl., 2024, 13, 278 Search PubMed.
- J. Peng, H. Yu, X. Jiang, P. Xu, Y. Li and H. Zhong, Adv. Funct. Mater., 2025, 2500993 Search PubMed.
- K. Wang, H. Wu, Y. Meng, Y. Zhang and Z. Wei, Energy Environ. Sci., 2012, 5, 8384–8389 RSC.
- Z. He, B. Gao, T. Li, J. Liao, B. Liu, X. Liu, C. Wang, Z. Feng and Z. Gu, ACS Sustain. Chem. Eng., 2018, 7, 1745–1752 CrossRef.
- M. Zhu, Y. Huang, Y. Huang, W. Meng, Q. Gong, G. Li and C. Zhi, J. Mater. Chem. A, 2015, 3, 21321–21327 RSC.
- S.-Z. Sheng, J.-L. Wang, B. Zhao, Z. He, X.-F. Feng, Q.-G. Shang, C. Chen, G. Pei, J. Zhou, J.-W. Liu and S.-H. Yu, Nat. Commun., 2023, 14, 3231 CrossRef CAS PubMed.
- H. Li, C. J. Firby and A. Y. Elezzabi, Joule, 2019, 3, 2268–2278 CrossRef CAS.
- R. Li, X. Ma, J. Li, J. Cao, H. Gao, T. Li, X. Zhang, L. Wang, Q. Zhang, G. Wang, C. Hou, Y. Li, T. Palacios, Y. Lin, H. Wang and X. Ling, Nat. Commun., 2021, 12, 1587 CrossRef CAS PubMed.
- Y. Su, Y. Wang, Z. Lu, M. Tian, F. Wang, M. Wang, X. Diao and X. Zhong, Chem. Eng. J., 2023, 456, 141075 CrossRef CAS.
- M. Tian, X. Liu, X. Diao and X. Zhong, Sol. Energy Mater. Sol. Cells, 2023, 253, 112239 CrossRef CAS.
- X.-A. Liu, J. Wang, D. Tang, Z. Tong, H. Ji and H.-Y. Qu, J. Mater. Chem. A, 2022, 10, 12643–12655 RSC.
- S. Cherumannil Karumuthil, M. K. Ganesha, I. Mondal, A. K. Singh and G. U. Kulkarni, J. Mater. Chem. A, 2022, 10, 23265–23273 RSC.
- M. K. Ganesha, H. Hakkeem, I. Mondal, A. K. Singh and G. U. Kulkarni, Small, 2024, 20, 2405467 CrossRef CAS PubMed.
- R. Roy, G. Mahendra and A. K. Singh, Chem. Eng. J., 2024, 500, 156870 CrossRef CAS.
- R. Roy, M. K. Ganesha, P. Dutta, D. Pal and A. K. Singh, ACS Appl. Energy Mater., 2023, 6, 11683–11693 CrossRef CAS.
- H. Li, L. McRae, C. J. Firby and A. Y. Elezzabi, Adv. Mater., 2019, 31, 1807065 CrossRef PubMed.
- R. Roy, R. Greeshma, P. Dutta, I. Mondal, R. Banerjee and A. K. Singh, ACS Appl. Mater. Interfaces, 2024, 16, 39539–39550 CrossRef CAS PubMed.
- Y. Luo, H. Jin, Y. Lu, Z. Zhu, S. Dai, L. Huang, X. Zhuang, K. Liu and L. Huang, ACS Energy Lett., 2022, 7, 1880–1887 CrossRef CAS.
- W. Zhang, H. Li and A. Y. Elezzabi, Adv. Funct. Mater., 2023, 33, 2300155 CrossRef CAS.
- G. Xu, W. Zhang, G. Zhu, H. Xia, H. Zhang, Q. Xie, P. Jin, H. Zhang, C. Yi, R. Zhang, L. Ji, T. Shui, N. Moloto, W. She and Z. Sun, Adv. Sci., 2024, 11, 2401948 CrossRef CAS PubMed.
- Z. Jia, Y. Sui, L. Qian, X. Ren, Y. Zhao, R. Yao, L. Wang, D. Chao and C. Yang, Nat. Commun., 2024, 15, 6110 CrossRef CAS PubMed.
- L. Zhang, D. Chao, P. Yang, L. Weber, J. Li, T. Kraus and H. J. Fan, Adv. Energy Mater., 2020, 10, 2000142 Search PubMed.
- W. C. Poh, A. L.-S. Eh, W. Wu, X. Guo and P. S. Lee, Adv. Mater., 2022, 34, 2206952 CrossRef CAS PubMed.
- Q. Wang, S. Cao, Q. Meng, K. Wang, T. Yang, J. Zhao and B. Zou, Mater. Horiz., 2023, 10, 960–966 Search PubMed.
- J. Feng, Y. Luo, X. Wang, G. Cai and R. Cao, Small, 2023, 19, 2304691 Search PubMed.
- G. Cai, P. Cui, W. Shi, S. Morris, S. N. Lou, J. Chen, J.-H. Ciou, V. K. Paidi, K.-S. Lee, S. Li and P. S. Lee, Adv. Sci., 2020, 7, 1903109 CrossRef CAS PubMed.
- Z. Wang, X. Jia, P. Zhang, Y. Liu, H. Qi, P. Zhang, U. Kaiser, S. Reineke, R. Dong and X. Feng, Adv. Mater., 2022, 34, 2106073 Search PubMed.
- W. Wu, H. Fang, H. Ma, L. Wu, W. Zhang and H. Wang, Nano-Micro Lett., 2020, 13, 20 Search PubMed.
- C. Deng, K. Zhang, L. Liu, Z. He, J. Huang, T. Wang, Y. Liu, X. He, K. Du and Y. Yi, J. Mater. Chem. A, 2022, 10, 17326–17337 Search PubMed.
- J. Wang, L. Zhang, L. Yu, Z. Jiao, H. Xie, X. W. Lou and X. Wei Sun, Nat. Commun., 2014, 5, 4921 CrossRef CAS PubMed.
- H. Li, W. Zhang and A. Y. Elezzabi, Adv. Mater., 2020, 32, 2003574 Search PubMed.
- Y. Ding, H. Sun, Z. Li, C. Jia, X. Ding, C. Li, J.-G. Wang and Z. Li, J. Mater. Chem. A, 2023, 11, 2868–2875 RSC.
- W. Zhang, H. Li, W. W. Yu and A. Y. Elezzabi, Light: Sci. Appl., 2020, 9, 121 Search PubMed.
- D. Ma, T. Yang, X. Feng, P. Wang, J. Huang, J. Wang and H. Li, Adv. Sci., 2024, 11, 2307223 Search PubMed.
- M. K. Ganesha, H. Hakkeem and A. K. Singh, Small, 2024, 20, 2403156 CrossRef CAS PubMed.
- Y. Wang, X. Zhong, X. Liu, Z. Lu, Y. Su, M. Wang and X. Diao, J. Mater. Chem. A, 2022, 10, 3944–3952 RSC.
- R. Zhang, Q. Zhou, S. Huang, Y. Zhang and R.-T. Wen, Nat. Commun., 2024, 15, 2294 CrossRef CAS PubMed.
- N. V. Talagaeva, E. V. Zolotukhina, P. A. Pisareva and M. A. Vorotyntsev, J. Solid State Electrochem., 2016, 20, 1235–1240 CrossRef CAS.
- N. Zhang, M. Jia, Y. Dong, Y. Wang, J. Xu, Y. Liu, L. Jiao and F. Cheng, Adv. Funct. Mater., 2019, 29, 1807331 CrossRef.
- D. Wang, L. Wang, G. Liang, H. Li, Z. Liu, Z. Tang, J. Liang and C. Zhi, ACS Nano, 2019, 13, 10643–10652 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |