 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Hydrogen defects in LaBi2O4X (X = Cl, Br, and I) Sillén oxyhalide phases and their impacts on ionic transport
ShinYoung
Kang
 a,
Andrew J. E.
Rowberg
a,
Andrew J. E.
Rowberg
 a,
Shenli
Zhang
a,
Shenli
Zhang
 ab and
Joel B.
Varley
ab and
Joel B.
Varley
 *a
*a
aMaterials Science Division and Laboratory for Energy Application for the Future (LEAF), Lawrence Livermore National Laboratory, Livermore, California 94550, USA. E-mail: varley2@llnl.gov
bDepartment of Chemical and Materials Engineering, San José State University, San Jose, California 95192, USA
First published on 4th March 2026
Abstract
Sillén oxyhalides have recently emerged as promising materials for both photocatalytic and ionic transport applications, yet the role of likely-ubiquitous hydrogen-related defects in these layered compounds remains largely unexplored. Here, we employ first-principles defect calculations to investigate incorporation energetics for hydrogen- and oxygen-related defects, as well as their migration barriers in LaBi2O4X (X = Cl, Br, I) phases. We find that hydrogen interstitials, particularly protonic species (Hi+), are readily accommodated within the open Bi–O layers. Protons compete with oxygen vacancy donors (VO2+) and charge-compensate with oxygen interstitial acceptors (Oi2−). By linking hydrogen defect formation to water- and oxygen-related redox equilibria, we reveal that VO2+ facilitates Hi+ incorporation, while Oi2− promotes interstitial hydroxide formation, establishing a direct connection between proton and oxide-ion transport. Calculated migration barriers indicate that ionic diffusion is confined to Bi–O layers with low barriers of 0.20–0.25 eV for Hi+ and 0.14–0.25 eV for VO2+, suggesting that the materials contain intrinsic pathways for mixed ionic conduction. These results provide a microscopic picture of hydrogen behavior in Sillén oxyhalides and point to design strategies for integrating protonic and oxide-ion transport in layered oxyhalide electrolytes. Band-edge alignment analysis shows that LaBi2O4I provides the optimal combination of hydrogen solubility, oxygen defect stability, and mixed ionic conductivity, highlighting its potential for low-temperature electrochemical and energy-conversion applications. Overall, this work establishes the defect-driven origin of hydrogen transport in Sillén oxyhalides and expands their applicability beyond photocatalysis to mixed ionic conduction and hydrogen electrochemistry.
Introduction
Sillén–Aurivillius compounds are layered materials consisting of alternating fluorite-like [M2O2]2+ slabs with perovskite-like or halide interlayers.1 This structural motif allows versatile chemical modification that can easily tune the electronic and optical properties, as well as flexible morphological regulations with variable number of interlayers during synthesis. Such tunability has highlighted Sillén–Aurivillius compounds as promising and versatile platforms for visible-light photocatalysts in water splitting.2–9 Among these, LaBi2O4Cl has been identified as a novel variant of the Sillén phases, containing triple fluorite-like slabs ([M3O4]+) charge-balanced by halide layers.10 While various Sillén compounds have been recognized for photocatalytic activity like Sillén–Aurivillius phases,11–13 LaBi2O4Cl-based oxychloride families have also emerged as fast oxygen-ion conductors: Yaguchi et al. demonstrated interstitialcy-driven oxygen transport, reporting conductivities of 10 mS cm−1 at 431 °C in Bi1.9Te0.1LuO4.05Cl and 20 mS cm−1 at 702 °C in LaBi1.9Te0.1O4.05Cl14,15—values that surpass state-of-the-art electrolytes such as yttria-stabilized zirconia (YSZ) and La0.8Sr0.2Ga0.83Mg0.17O2.815 (LSGM). The corresponding activation energies of 0.22–0.50 eV underline their excellent ionic transport properties. More recently, Meng et al. reported exceptionally fast vacancy-mediated oxygen conduction with a migration barrier as low as 0.1 eV.16Beyond their outstanding photocatalytic reactivity and oxygen conductivity, Sillén and Sillén–Aurivillius phases are also considered chemically stable in water. In many Sillén and Sillén–Aurivillius oxyhalides, the valence band edge (VBM) is dominated by oxygen 2p orbitals, which may effectively suppress the self-oxidation of halogen species, i.e., dissolution or volatilization in aqueous environments.5,13 This raises a critical and largely unexplored question: how do these compounds interact with water, and how do hydrogen-related point defects impact ionic transport?
Compared to established proton- and mixed-conducting oxides such as perovskites (e.g., BaZrO3, BaCeO3) or fluorites, Sillén-type oxyhalides offer a fundamentally distinct structural platform for hydrogen incorporation. Their layered architecture comprises open Bi–O planes separated by halide layers, creating low-coordination environments that possibly affect both the stability and diffusion barriers of hydrogen-related defects. Furthermore, the favorable permeability of oxygen species in Sillén oxyhalides14–16 suggests defect chemistries that naturally couple proton and oxide-ion transport with strong implications for understanding mixed ion conduction.
The ability of a material to function as an electrolyte or membrane for water-splitting and/or separation-related applications is largely dictated by its chemical stability in water/steam and the permeability of hydrogen defects. Collectively, these factors determine whether these families of compounds could serve as oxygen- or proton-conducting materials for electrochemical devices such as electrolyzer cells, fuel cells, and solid-state separation membranes, provided they satisfy other application-specific properties like controllable electronic conductivity and thermal stability.17–19 In this work, we use LaBi2O4X (X = Cl, Br, or I) oxyhalides as a case study for understanding the consequences of hydrogen incorporation, particularly in the context of ionic transport. Having already demonstrated excellent oxygen conduction in the case of LaBi2O4Cl, they provide an ideal model to investigate how chemistry influences water-material interactions and whether hydrogen-related defects can be stabilized and mobilized alongside oxygen defects. In this class of materials, halogen substitution provides an effective means of tuning the electronic structure, particularly the band gap and the relative energetics of the band edges. The VBM, which is mainly contributed by oxygen 2p orbitals, is sensitive to the interlayer spacing, itself governed by the choice of halogen species.2,5,13 Consequently, the coupled structural and electronic modifications induced by halogen substitution can have a profound impact on defect stability and migration kinetics. This study thus bridges the known oxygen-ion conduction with the unexplored potential of protonic transport, expanding the possible application space of Sillén-type oxyhalides beyond photocatalysis into electrochemical energy conversion.
Results and discussions
To assess the relative stability of hydrogen-involving defects in LaBi2O4X (X = Cl, Br, and I), we calculated the formation energies for a range of species: interstitials (Hi, Oi, and OHi), substituted species (HO and HX), and vacancies, including those coupled with hydrogen (VO, VX, Hi–VO, Hi–VX, and HO–VO). To calculate the defect formation energies, we adopt the conventional defect supercell approach20 and use the PBE exchange correlation functional, along with band edge corrections obtained with the HSE06 hybrid functional with spin–orbit corrections,21,22 as implemented in the VASP code.23 Additional tests to assess the relevance of charge localization for particular defects and charge states (e.g., VO0 and VO2+, Hi0, Hi+, and Bi5+ bipolarons) were performed with hybrid functionals and verified the qualitative defect behavior obtained with PBE. Oxygen- and halogen-rich environments were used for the choice of chemical potentials (see the SI for details). We evaluated three possible interstitial sites: within the La layer, within the halogen (X) layer, and within the Bi2O2 layer (Fig. 1(a)). Across all halide variants (X = Cl, Br, I), interstitial acceptors, such as Hi−, Oi2−, and OHi−, are most stable when located near La cations, while the Hi+ interstitial donor preferentially resides within the Bi–O layer to bond with oxygen anions. Representative lowest-energy configurations are illustrated in Fig. 1(b).The thermodynamic stability of defects is examined through the defect formation energies, Ef, plotted as a function of the Fermi energy in LaBi2O4Cl, LaBi2O4Br, and LaBi2O4I (Fig. 2). Note that chemical potentials of O, H, and X are chosen to reflect equilibrium with O2 gas, H2O gas, and solid La2O3 and Bi2O3 phases, consistent with synthesis conditions and those typical of low-temperature water-containing environments (See SI). In all three compounds, VO2+, Hi+, and Oi2− emerged as the most stable defects for Fermi levels spanning the band gaps; we indicate transitions between the most favorable defect species with black and yellow stars in Fig. 2. Specifically, VO2+ dominates near the VBM, Hi+ is preferred at intermediate Fermi levels, and Oi2− becomes most favorable near the conduction band minimum (CBM). Note that, however, the Fermi level regime where VO2+ is dominant over Hi+ is where Ef[VO2+] is negative, indicating that these oxyhalides are unstable in these conditions. Neutral Oi0 is more stable when paired with a lattice oxygen ion to form a dumbbell configuration, Oi0(db), than as an isolated Oi0. However, Oi2−(db) is never stabilized relative to either Oi0(db) or Oi0 within the band gap for any of the compounds studied. In LaBi2O4I, we observe that the formation energy of VI+ is comparable to that of Hi+, and thus VI+ and Hi+ will compete as the dominant donor defects. For all compounds, VO exclusively acts as a double donor for all Fermi levels within their band gaps, which was verified with independent hybrid functional calculations (see Fig. S5). For LaBi2O4Cl, Ef[VO2+] at the VBM is −1.71 eV, significantly lower than previously reported values,16 likely due to differences in correction schemes. For example, the modified band alignment (MBA)24 used in ref. 16 relies on the neutral state of the defect, which is ill-defined in these materials due to neutral VO spuriously populating the conduction band with electrons when calculated using PBE, which likely impacts the alignment corrections scheme used in this case. The range of Fermi levels where Hi+ is the most stable defect decreases from LaBi2O4Cl, LaBi2O4Br, to LaBi2O4I, which we later discuss in the context of relative band alignments. We give approximate Fermi-level positions expected from charge-neutrality for these defects, which is governed by the crossing of the Hi+ and Oi2− defects; these Fermi levels are denoted by yellow stars in Fig. 2. Note that in its ground-state configuration, the Hi+ defect forms a localized protonic species by binding to an oxygen atom in the Bi–O layer, with a relaxed O–H bond length of approximately 0.99 Å and no significant hydrogen bonding to neighboring oxygen atoms in all LaBi2O4X compounds. Moreover, proton decoration of the Oi2−(db) configuration leads to spontaneous relaxation toward the interstitial hydroxide species, OHi−, coupled with a lattice oxygen, reinforcing that hydrogen–oxygen defect interactions are more likely to manifest as hydroxide formation.
By contrast, defect complexes involving trapping of hydrogen defects in halogen or oxygen vacancies (HO and HX) are consistently high in energy (>2 eV), suggesting they are rare, at least under the considered conditions. We found that the Hi–VO complex recombines into HO in all compounds. Hydride ions, Hi−, in the La layer are also less stable than protons, Hi+, on the Bi2O2 layer by 6.0, 5.6, and 5.2 eV in –Cl, –Br, and –I compounds, respectively. In addition, we found that the interstitial hydroxide species, OHi−, essentially a complex between Oi2− and Hi+, has meaningful stability, especially for n-type conditions, with Ef[OHi−] = 0.93, 0.88, and 0.77 at the CBM for –Cl, –Br, and –I compounds, respectively. These results indicate that LaBi2O4X readily accommodates Hi+, Oi2−, and OHi−, consistent with the open structure of the Sillén phases.
The calculated Ef values further inform the reaction thermodynamics for proton formation. Table 1 summarizes three representative processes: (1) water-splitting (and proton-incorporation) aided by VO2+, (2) direct water-splitting into Hi+ and OHi−, and (3) proton-liberation from reactions between VO2+ and OHi−. Note that these reaction energies are independent of the choice of element chemical potentials. The proton formation aided by VO2+ is an exothermic reaction for LaBi2O4Cl but becomes less favorable in the –I analog (from −0.09 to 0.24 eV/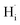 for –Cl and –I compounds). On the other hand, despite the low Ef of OHi− (<1 eV at the CBM), direct formation of Hi+ + OHi− from water is energetically prohibitive (>1.70 eV/Hi+) for all compounds. However, OHi− can react with VO2+ to form protons with low reaction energies from −1.88 to −1.34 eV/Hi+. It is worth noting that these calculated proton formation energies are valid only in the bulk. In other words, surface reactions and materials conditions—such as microstructure—significantly impact the defect formation energies, and hence reaction energetics, summarized here. Hydrogen incorporation from water is inherently a surface-mediated process involving adsorption and dissociation steps, which are not explicitly treated in this work. However, given the layered topology and open Bi–O planes of Sillén-type oxyhalides, bulk defect energetics provide a meaningful first-order description of hydrogen stability and transport once incorporation occurs. These results highlight the importance of oxygen defects as mediators for hydrogen incorporation, as the VO2+-aided water-splitting reaction is the most kinetically preferred route to generate Hi+, with LaBi2O4Cl providing the most favorable environment. The overall contributions of the reaction to proton solubility need more systematic studies that consider structural inhomogeneity, along with processing conditions.
for –Cl and –I compounds). On the other hand, despite the low Ef of OHi− (<1 eV at the CBM), direct formation of Hi+ + OHi− from water is energetically prohibitive (>1.70 eV/Hi+) for all compounds. However, OHi− can react with VO2+ to form protons with low reaction energies from −1.88 to −1.34 eV/Hi+. It is worth noting that these calculated proton formation energies are valid only in the bulk. In other words, surface reactions and materials conditions—such as microstructure—significantly impact the defect formation energies, and hence reaction energetics, summarized here. Hydrogen incorporation from water is inherently a surface-mediated process involving adsorption and dissociation steps, which are not explicitly treated in this work. However, given the layered topology and open Bi–O planes of Sillén-type oxyhalides, bulk defect energetics provide a meaningful first-order description of hydrogen stability and transport once incorporation occurs. These results highlight the importance of oxygen defects as mediators for hydrogen incorporation, as the VO2+-aided water-splitting reaction is the most kinetically preferred route to generate Hi+, with LaBi2O4Cl providing the most favorable environment. The overall contributions of the reaction to proton solubility need more systematic studies that consider structural inhomogeneity, along with processing conditions.
| Reaction | Reaction energies | X = Cl | X = Br | X = I | |
|---|---|---|---|---|---|
| a To avoid Oi2− migration to VO2+ and annihilation, the position of Oi2− was fixed. b The VO2+ – Oi2− configuration could not be stabilized in LaBi2O4I. | |||||
| Proton formation (eV/Hi+) | (1) VO2+ + H2O → 2Hi+ | −0.09 | −0.10 | 0.24 | |
| (2) H2O → Hi+ + OHi− | 1.78 | 1.85 | 1.86 | ||
| (3) VO2+ + OHi− → Hi+ | −1.88 | −1.76 | −1.34 | ||
| Hydrogen defect trapping (eV/Hi) | (4) Hi+ + Oi2− → OHi− | −0.71 | −0.84 | −0.90 | |
| (5) Hi− + VO2+ → HO+ | −1.25 | −0.38 | −0.90 | ||
| (6) Hi+ + VO0 → HO+ | −0.07 | 0.64 | −0.55 | ||
| (7) Hi+ + VO2+ + e− → HO+ + h+ | at VBM | 4.77 | 5.10 | 4.21 | |
| at CBM | 0.74 | 1.51 | 0.77 | ||
| (8) Hi+ + VX+ → [Hi − VX]2+ | 0.24 | 0.16 | 0.15 | ||
| (9) Hi+ + VX+ → HX2+ | 4.19 | 3.20 | 3.65 | ||
| Oxygen defect annihilation (eV/O) | (10) VO2+ + Oi2− → OO0 | −2.59 | −2.60 | −2.24 | |
| Oxygen frenkel pair formation energy (eV/O) | (11) VO2+ + Oi2− → VO2+ − Oi2− | −1.61a | −1.41a | N/Ab | |
To characterize hydrogen trapping at defects, the binding energies of hydrogen defects, Hi+ and Hi−, with low-energy oxygen defects, Oi2− and VO2+, are evaluated (Table 1). By convention, a negative binding energy indicates attraction and thus promotes hydrogen trapping at that site. Two reactions are consistently favorable across all halides: (i) a proton binding with an oxygen interstitial to form an interstitial hydroxide (reaction 4), and (ii) a hydride binding with an oxygen vacancy to form a substitutional HO+ defect (reaction 5). Combining reactions 1, 3, and 4 with the formation energies in Fig. 2, one can identify the optimal conditions to increase proton solubility in these materials. For example, shifting the Fermi level closer to the CBM (e.g. via donor doping) will stabilize oxygen interstitials over vacancies, consuming protons to form hydroxides (reaction 4) and thereby reducing bulk proton solubility. When the Fermi level is closer to the VBM (e.g. via acceptor doping), on the other hand, oxygen vacancies become dominant and promote the formation of protons (reaction 3). Both reactions are expected to influence oxygen conductivity by altering the relative concentrations of Oi2−, VO2+, and OHi−. Both VO2+ and Oi2− have been identified as primary carriers for oxygen diffusion in Sillén phases.14–16
Despite favorable reaction energies, we note that reactions 5 and 6 (involving hydride ions) are not expected to be relevant, since Hi− are never predicted to be favored over Hi+ (Fig. 2). Similarly, proton trapping in a halide ion vacancy, VX+, (reaction 9) is thermodynamically highly unfavorable with low reaction rates attributed to the high Ef[HX2+], while protons can easily bind with VX+ (reaction 8). The impact of VX+ on proton migration kinetics will be discussed in the following section. Additionally, we find that annihilation of VO2+ and Oi2− is substantially more favorable than Frenkel pair formation in all three oxyhalide systems, suggesting the importance of stoichiometry control and operation conditions for achieving high oxide conductivity.
To compare defect energetics and redox potentials across compounds, VBMs and CBMs of Sillén phases are aligned to the vacuum level using surface models (Fig. 3(a)). Additional details on the band-alignment procedure, including a comparison with bulk core-level alignment, are provided in the SI. Interestingly, the PBE and HSE06 functionals yielded consistent CBM positions for the same lattice constants, while HSE06 shifted the VBMs downward with respect to those from PBE, resulting in the band gap widening from 1.21 to 2.01 eV (X = Cl), from 1.08 to 1.80 eV (X = Br), and from 1.21 to 1.72 eV (X = I). The corresponding ionization potentials are 5.77, 5.56, and 5.31 eV in PBE, and 6.52, 6.23, and 5.81 eV in HSE06 for the –Cl, –Br, and –I phases, respectively. From these energies, we can directly compare the relative and absolute band edge positions across these materials, enabling predictions of charge transfer, interface behavior, and band alignments. For (photo)catalytic applications, this is useful for understanding how the band edges straddle important redox potentials; for instance, having the CBM above the standard hydrogen electrode (SHE) potential (0 V vs. SHE) can facilitate reductive electron transfer for the hydrogen evolution reaction (HER), and/or a VBM below the oxygen evolution reaction (OER) potential (+1.23 V vs. SHE) can facilitate oxidative hole transfer. For this particular example, this translates to electron affinities closer to the vacuum level than the SHE potential (−4.44 eV)25 and ionization potentials further away than the OER potential (−5.67 eV). Our band alignment in Fig. 3(a) shows that all VBMs computed in the HSE06 functional are positioned below the OER level, while only LaBi2O4I has a CBM positioned above the HER level. Interestingly, the BiOX (X = Cl, Br, I) family, whose layered structure consists of alternating [Bi2O2]2+ slabs and double halogen layers analogous to those in Sillén phases, has demonstrated promising photocatalytic activity owing to their well-aligned band edge positions.26,27
 | ||
| Fig. 3 (a) Variation of energy levels in LaBi2O4Cl, LaBi2O4Br, and LaBi2O4I, showing VBMs (horizontal green lines) and CBMs (horizontal blue lines) calculated using PBE (dashed lines) and HSE06 (solid lines) functionals. Yellow star markers represent the Fermi levels when the formation energy of proton interstitials donors, Hi+, equals that of Oi2− acceptors for our studied conditions, as in Fig. 2. Black star markers are for the equilibrium between Hi+ and VO2+ donors. Formation energies of Hi+ compared to (b) VO and (c) Oi. The vacuum level is set to zero in the y-axis of (a) and x-axis of (b and c). Gray dashed lines indicate the standard hydrogen electrode (SHE) and the oxygen evolution reaction (OER) potentials, which are −4.44 and −5.67 eV with respect to vacuum, respectively. | ||
Overall, the defect formation energies suggest that Hi+ and VO2+ are the competing dominant hydrogen and oxygen defects at EFermi closer to the VBM, while Hi+ and Oi2− (and VI+ in LaBi2O4I) are dominant at EFermi closer to the CBM. Assuming these water-related defects are the dominant charged species, which follows from their low formation energies in Fig. 2, charge neutrality should be determined by compensation between Hi+ and Oi2− (and VI+ in LaBi2O4I), leading to Fermi levels in the vicinity of the yellow stars in Fig. 2 and 3. This Fermi level value falls within the band gap for all of the studied oxyhalides but is close to the CBM, suggesting water-related defects may have larger indirect effects on ionic conduction by suppressing the concentrations of mobile VO2+ rather than strongly impacting the electronic conductivity. We note that this simple analysis does not account for other impurity or dopant effects that could shift the Fermi level if concentrations are comparable to those of the hydrogen- or oxygen-related defects.
To gain insight into the relative proton solubility in the oxyhalides at a common electrochemical reference, we replotted the defect formation energies as a function of Fermi level referenced to vacuum in Fig. 3(b and c). These results highlight how, for the same conditions, the proton formation energy in LaBi2O4I is 0.17 (0.21 eV) lower than that in LaBi2O4Cl (LaBi2O4Br). Interestingly, VO2+ formation is also more favorable in LaBi2O4I by 0.95 (0.84 eV) than in LaBi2O4Cl (LaBi2O4Br), suggesting that LaBi2O4I can incorporate much higher concentrations of Hi+ and VO2+ than the other two under identical conditions. On the other hand, the formation energy of Oi2− in LaBi2O4I is 0.61 (0.48 eV) higher than that in LaBi2O4Cl (LaBi2O4Br). Thus, at common absolute Fermi levels and conditions, the formation energies of Oi2− (VO2+) are positively (negatively) correlated with the halide electronegativity in the LaBi2O4X series, with LaBi2O4I having the lowest Ef[VO2+] and highest Ef[Oi2−], in addition to the lowest Ef[Hi+]. This suggests that LaBi2O4I is more effective than –Cl and –Br phases at promoting VO formation and suppressing Oi formation. Considering that VO is a primary carrier for oxygen conductivity in LaBi2O4Cl, LaBi2O4I would greatly benefit from increasing the solubility of mobile oxygen defects.
The overall favorable formation of Hi+ in these oxyhalides naturally raises the question of whether they are promising candidates as mixed ionic conductors. To address this question, we computed the migration barriers of hydrogen and oxygen defects using the Climbing Image Nudged Elastic Band (CI-NEB) calculations28 to fully assess their conductivity. Four migration pathways were considered for Hi+, namely, interlayer and intralayer pathways with and without a halogen vacancy. These pathways are illustrated in Fig. 1(b), and their barriers are summarized in Table 2. Hopping from one lowest-energy Bi–O layer site to another includes two-dimensional (2D) in-plane migration and three-dimensional (3D) out-of-plane migration (Path I and II). The 2D diffusion pathway exhibits low barriers of 0.20–0.25 eV, while 3D diffusion across the La layer shows moderate barriers of 0.65–0.78 eV in all compounds.
| Species | Migration pathways | X = Cl | X = Br | X = I |
|---|---|---|---|---|
| Hi+ | Path I: in-plane on Bi–O layer without VX | 0.24 | 0.25 | 0.20 |
| — | Path I′: in-plane on Bi–O layer with VX | 0.15 | 0.16 | 0.16 |
| — | Path II: between Bi–O layers | 0.78 | 0.72 | 0.65 |
| — | Path III: from Bi–O layer to X layer without VX | 1.79 (0.11) | 1.95 (0.22) | 2.01 (0.44) |
| — | Path IV: from Bi–O layer to X layer with VX | 2.02 (0.00) | 2.19 (0.00) | 2.11 (0.00) |
| OHi− | In-plane on La layer | 0.87 | 0.85 | 0.85 |
| Oi2− | In-plane on La layer | 0.81 | 0.86 | 0.94 |
| VO2+ | In-plane on Bi–O layer | 0.14 | 0.18 | 0.25 |
These results suggest that these Sillén phases could serve as fast proton conducting materials dominated by 2D conductivity at low temperatures, while the quasi-3D diffusion mechanism could be activated at higher temperatures, thereby fully utilizing the interconnected network of La–O polyhedra. Interestingly, the migration barriers show little variation across the halide series, despite the increase in the lattice constants and the hopping distances from –Cl to –I. Due to the high site energy on the halogen layer, migration barriers there are higher than 1.7 eV (Path III), inhibiting the full 3D diffusion beyond the Bi–O layer. We confirmed that halogen vacancies do not help access the full 3D diffusion (Path IV). However, notably, halogen vacancies can further accelerate the 2D conductivity by lowering the Bi–O in-plane migration barriers by 0.04–0.09 eV (Path I′). We found that the VX+ induces slight structural distortions on the adjacent Bi–O plane, leading to lower the H migration barrier. Considering the sensitive stability competition between Hi+ and VX+, especially in LaBi2O4I (Fig. 2), the beneficial impact of VX+ on hydrogen transport is encouraging.
As Hi+ easily reacts with Oi2− to form OHi− (reaction 3 in Table 1), we also computed the migration barriers of OHi−. The barriers range from 0.85–0.87 eV and can be activated at high temperatures. The migration barriers of Oi2− and VO2+ are 0.81–0.94 eV and 0.14–0.25 eV, respectively, in good agreement with recent reports.16 In contrast to Hi+, the migration barriers of OHi−, Oi2−, and VO2+ do show a stronger dependence on the halide, increasing from –Cl to –I. We attribute this to the stronger Bi–O bond in materials with smaller lattice constants, which aids the migration of oxygen-related defects by reducing their hopping distance, while more rigid Bi–O bonds may hinder the rotation of O–H bonds and proton hopping.
Overall, these results establish that LaBi2O4X Sillén phases can host both oxygen and hydrogen defects, with distinct trends across halide chemistry. LaBi2O4Cl shows the most favorable oxygen vacancy-assisted proton generation, while LaBi2O4I combines favorable proton transport with enhanced defect solubility for both hydrogen and oxygen, making it a promising candidate for mixed ionic conduction. Both LaBi2O4Cl and LaBi2O4I are reported to be thermodynamically stable within the quaternary La–Bi–O–X phase diagrams,29 and their successful synthesis has been experimentally demonstrated.10,16,30 Interestingly, LaBi2O4I has been theoretically predicted to exhibit promising optoelectronic and thermoelectric performance, attributed to its strong optical response in the visible–ultraviolet range and favorable band topology.31 Taken together, these results suggest that LaBi2O4I offers an attractive combination of bulk hydrogen solubility, transport properties, and intrinsic stability, while recognizing that the optimal halogen composition under realistic operating conditions will depend on surface insertion kinetics and chemical stability. More broadly, this work delineates a clear design space in which halogen chemistry can be leveraged to balance hydrogen incorporation and transport for targeted electrochemical applications as summarized in Fig. 4.
Conclusions
In summary, we have described water-related defects in LaBi2O4X (X = Cl, Br, I) Sillén phases. Our first-principles defect calculations reveal that these materials can readily incorporate hydrogen defects, with proton solubility inversely proportional to the halide size/electronegativity at common electrochemical reference energies. Protons have low migration barriers (0.20–0.25 eV) along the Bi–O layers that enable facile 2D conductivity, with barriers largely independent of the halide species. While oxygen vacancy-assisted water splitting is the most plausible route for proton generation, oxygen interstitials can also bind with protons to form interstitial hydroxide species. Thus, proton solubility and transport are intimately connected with water and oxygen atmospheres, and controlled defect engineering will be essential to maximize proton solubility. Notably, while proton transport is largely insensitive to the halide but becomes slightly easier with increasing molar volume from Cl to I, migration of oxygen-related defects shows the opposite trend, with VO2+ migration remaining comparably facile with barriers on the order of 0.14–0.25 eV. These findings suggest that, while all of these LaBi2O4X oxyhalides may serve as mixed ionic conductors, LaBi2O4Cl is most favorable for vacancy-assisted proton generation based on bulk reaction energetics, whereas LaBi2O4I offers a favorable combination of bulk proton solubility and mobility once hydrogen is incorporated. Which halogen composition is preferred will depend on the specific operating regime, including the relative importance of surface incorporation kinetics, environmental conditions, and chemical stability. Further investigations of stability, synthesizability, and electrochemical performance under operating conditions are needed to evaluate this material for ionic transport and photocatalytic applications. Overall, this work expands the potential of Sillén oxyhalides beyond their established photocatalytic applications, highlighting their promise as electrolytes or membranes in low-temperature electrochemical devices.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: DFT settings, summary of structural and electronic properties of bulk LaBi2O4X, reference chemical potentials, and energy level alignment approaches. See DOI: https://doi.org/10.1039/d5ta09720g.Acknowledgements
This work was performed under the auspices of the U.S. Department of Energy (DOE) by the Lawrence Livermore National Laboratory under Contract No. DE-AC52-07NA27344 and supported the Office of Energy Efficiency and Renewable Energy (EERE), and the Hydrogen and Fuel Cell Technologies Office (HFTO). Computing support was provided by the Lawrence Livermore National Laboratory Institutional Computing Grand Challenge program and resources sponsored by the Department of Energy's Office of Critical Minerals and Energy Innovation, located at the National Laboratory of the Rockies.References
- O. K. Moune-Minn, L. Albert and P. Caro, Synthesis and spectroscopic properties of the sillen phases, J. Less-Common Met., 1983, 93(1), 143–150, DOI:10.1016/0022-5088(83)90459-9.
- J. Chen, Y. Gu, S. Xu, Y. Zhang, Z. Zhang, L. Shi, Z. Mu, C. Zhou, J. Zhang and Q. Zhang, Band Gap Engineering in Quadruple-Layered Sillén–Aurivillius Perovskite Oxychlorides Bi7Fe2Ti2O17X (X = Cl, Br, I) for Enhanced Photocatalytic Performance, Catalysts, 2023, 13, 751 CrossRef CAS.
- V. Werner, U. Aschauer, G. J. Redhammer, J. Schoiber, G. A. Zickler and S. Pokrant, Synthesis and Structure of the Double-Layered Sillén–Aurivillius Perovskite Oxychloride La2.1Bi2.9Ti2O11Cl as a Potential Photocatalyst for Stable Visible Light Solar Water Splitting, Inorg. Chem., 2023, 62(17), 6649–6660, DOI:10.1021/acs.inorgchem.3c00116.
- M. Ogawa, H. Suzuki, K. Ogawa, H. Ubukata, O. Tomita, A. Nakada, S. Nozawa, A. Saeki, H. Kageyama and R. Abe, Flux Synthesis of Sillén–Aurivillius Oxyiodide for Visible Light Water Splitting Photocatalysis, Chem. Mater., 2025, 37(3), 1123–1131, DOI:10.1021/acs.chemmater.4c02946.
- K. Ogawa, H. Suzuki, A. Walsh and R. Abe, Orbital Engineering in Sillén–Aurivillius Phase Bismuth Oxyiodide Photocatalysts through Interlayer Interactions, Chem. Mater., 2023, 35(14), 5532–5540, DOI:10.1021/acs.chemmater.3c00932.
- J. Yang, Y. Xue, C. Han, X. Zhang, K. Sa, J. Jia, H. Ye and Y. Liang, In situ construction of a Sillén–Aurivillius layered perovskite-based 0D/2D homologous Schottky junction for efficient piezo-photocatalytic activity, Catal. Sci. Technol., 2023, 13(18), 5313–5325, 10.1039/D3CY00876B.
- D. Ozaki, H. Suzuki, K. Ogawa, R. Sakamoto, Y. Inaguma, K. Nakashima, O. Tomita, H. Kageyama and R. Abe, Synthesis, band structure and photocatalytic properties of Sillén–Aurivillius oxychlorides BaBi5Ti3O14Cl, Ba2Bi5Ti4O17Cl and Ba3Bi5Ti5O20Cl with triple-, quadruple- and quintuple-perovskite layers, J. Mater. Chem. A, 2021, 9(13), 8332–8340, 10.1039/D0TA12550D.
- D. Ozaki, H. Suzuki, O. Tomita, Y. Inaguma, K. Nakashima, H. Kageyama and R. Abe, A new lead-free Sillén–Aurivillius oxychloride Bi5SrTi3O14Cl with triple-perovskite layers for photocatalytic water splitting under visible light, J. Photochem. Photobiol., A, 2021, 408, 113095, DOI:10.1016/j.jphotochem.2020.113095.
- S. Xu, Y. Zhang, Y. Wang, J. Chen, C. Zhou, Z. Mu, Z. Zhang, J. Zhang, J. Wang and Q. Zhang, Coupled ferroelectric polarization in novelty Sillén–Aurivillius BaBi4TiNbO11Cl material for photocatalysis, J. Alloys Compd., 2023, 952, 169932, DOI:10.1016/j.jallcom.2023.169932.
- C. J. Milne, P. Lightfoot, J. D. Jorgensen and S. Short, Synthesis and structure of Bi2LaO4Cl: a novel variant of the sillen phases, J. Mater. Chem., 1995, 5(9), 1419–1421, 10.1039/JM9950501419.
- W. Dong, T. Xie, Z. Wu, H. Peng, H. Ren, F. Meng and H. Lin, Oxygen-vacancy-rich BiOCl materials with ultrahigh photocatalytic efficiency by etching bismuth glass, RSC Adv., 2021, 11(61), 38894–38906, 10.1039/D1RA06961F.
- Y. Shi, J. Li, C. Mao, S. Liu, X. Wang, X. Liu, S. Zhao, X. Liu, Y. Huang and L. Zhang, Van Der Waals gap-rich BiOCl atomic layers realizing efficient, pure-water CO2-to-CO photocatalysis, Nat. Commun., 2021, 12(1), 5923, DOI:10.1038/s41467-021-26219-6.
- H. Suzuki, H. Kunioku, M. Higashi, O. Tomita, D. Kato, H. Kageyama and R. Abe, Lead Bismuth Oxyhalides PbBiO2X (X = Cl, Br) as Visible-Light-Responsive Photocatalysts for Water Oxidation: Role of Lone-Pair Electrons in Valence Band Engineering, Chem. Mater., 2018, 30(17), 5862–5869, DOI:10.1021/acs.chemmater.8b01385.
- H. Yaguchi, D. Morikawa, T. Saito, K. Tsuda and M. Yashima, High Oxide-Ion Conductivity through the Interstitial Oxygen Site in Sillén Oxychlorides, Adv. Funct. Mater., 2023, 33(27), 2214082, DOI:10.1002/adfm.202214082.
- N. Ueno, H. Yaguchi, K. Fujii and M. Yashima, High Conductivity and Diffusion Mechanism of Oxide Ions in Triple Fluorite-Like Layers of Oxyhalides, J. Am. Chem. Soc., 2024, 146(16), 11235–11244, DOI:10.1021/jacs.4c00265.
- J. Meng, M. S. Sheikh, L. E. Schultz, W. O. Nachlas, J. Liu, M. P. Polak, R. Jacobs and D. Morgan, Ultrafast Oxygen Conduction in Sillén Oxychlorides, Chem. Mater., 2025, 37(9), 3147–3157, DOI:10.1021/acs.chemmater.4c03163.
- A. J. E. Rowberg, M. Li, T. Ogitsu and J. B. Varley, Incorporation of protons and hydroxide species in BaZrO3 and BaCeO3, Mater. Adv., 2023, 4(23), 6233–6243, 10.1039/D3MA00308F.
- A. J. E. Rowberg, M. Li, T. Ogitsu and J. B. Varley, Polarons and electrical leakage in ${\mathrm{BaZrO}}_{3}$ and ${\mathrm{BaCeO}}_{3}$, Phys. Rev. Mater., 2023, 7(1), 015402, DOI:10.1103/PhysRevMaterials.7.015402.
- S. Zhang, A. J. E. Rowberg, T. Ogitsu, T. A. Pham and J. B. Varley, Electron-Phonon Renormalization in the Proton-Conducting Electrolyte $\mathrm{Ba}\mathrm{Zr}\mathrm{O}$${}_{3}$ and Its Implications for High-Temperature Electrolysis, PRX Energy, 2025, 4(1), 013013, DOI:10.1103/PRXEnergy.4.013013.
- C. Freysoldt, B. Grabowski, T. Hickel, J. Neugebauer, G. Kresse, A. Janotti and C. G. Van de Walle, First-principles calculations for point defects in solids, Rev. Mod. Phys., 2014, 86(1), 253–305, DOI:10.1103/RevModPhys.86.253.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. B:condens. Matter Mater. Phys, 1996, 77, 3865, DOI:10.1103/PhysRevLett.77.3865.
- A. V. Krukau, O. A. Vydrov, A. F. Izmaylov and G. E. Scuseria, Influence of the exchange screening parameter on the performance of screened hybrid functionals, J. Chem. Phys., 2006, 125(22) DOI:10.1063/1.2404663.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B:condens. Matter Mater. Phys, 1999, 59(3), 1758–1775, DOI:10.1103/PhysRevB.59.1758.
- M. P. Polak, R. Kudrawiec, R. Jacobs, I. Szlufarska and D. Morgan, Modified band alignment method to obtain hybrid functional accuracy from standard DFT: Application to defects in highly mismatched III-V:Bi alloys, Phys. Rev. Mater., 2021, 5(12), 124601, DOI:10.1103/PhysRevMaterials.5.124601.
- S. Trasatti, The absolute electrode potential: an explanatory note (Recommendations 1986), Pure Appl. Chem., 1986, 58(7), 955–966, DOI:10.1351/pac198658070955.
- Z. Ali, J. Ma, M. Hong and R. Sun, Review: applications of the functional photocatalysts BiOX (X = Cl, Br, I) for clean energy, the environment, and future photobiorefineries, J. Mater. Chem. A, 2023, 11(7), 3297–3314, 10.1039/D2TA09877F.
- A. Hussain, J. Hou, M. Tahir, S. S. Ali, Z. U. Rehman, M. Bilal, T. Zhang, Q. Dou and X. Wang, Recent advances in BiOX-based photocatalysts to enhanced efficiency for energy and environment applications, Catal. Rev., 2024, 66(1), 119–173, DOI:10.1080/01614940.2022.2041836.
- G. Henkelman, A. Arnaldsson and H. Jonsson, A fast and robust algorithm for Bader decomposition of charge density, Comput. Mater. Sci., 2006, 36(3), 354–360, DOI:10.1016/j.commatsci.2005.04.010.
- A. Jain, S. P. Ong, G. Hautier, W. Chen, W. D. Richards, S. Dacek, S. Cholia, D. Gunter, D. Skinner and G. Ceder, et al., Commentary: The Materials Project: A materials genome approach to accelerating materials innovation, APL Mater., 2013, 1(1), 011002, DOI:10.1063/1.4812323.
- M. Schmidt, H. Oppermann, C. Hennig, R. W. Henn, E. Gmelin, N. Söger and M. Binnewies, Untersuchungen zu Bismutseltenerdoxidhalogeniden der Zusammensetzung Bi2SEO4X (X = Cl, Br, I), Z. Anorg. Allg. Chem., 2000, 626(1), 125–135, DOI:10.1002/(SICI)1521-3749(200001)626:1<125::AID-ZAAC125>3.0.CO;2-S.
- R. K. Joshi, S. R. Bhandari and M. P. Ghimire, Structural stability, electronic, optical, and thermoelectric properties of layered perovskite Bi2LaO4I, RSC Adv., 2022, 12(37), 24156–24162, 10.1039/D2RA03859E.
| This journal is © The Royal Society of Chemistry 2026 |



