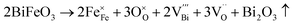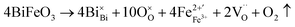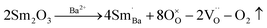Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A bismuth ferrite-based lead-free piezoelectric ceramic surpassing PZT-8 in thermal stability
Xiaodong Yana,
Chaoyang Shib,
Jianjun Wuc,
Kevin Chen*d and
Zhengbao Yang *a
*a
aDepartment of Mechanical and Aerospace Engineering, The Hong Kong University of Science and Technology, Clear Water Bay, Hong Kong, China. E-mail: zbyang@ust.hk
bSchool of Mechanical Engineering, Tianjin University, Tianjin 300072, China
cSichuan Energy Investment Tianfu Clean Energy Research Institute, Chengdu 610041, China
dDepartment of Electronic and Computer Engineering, The Hong Kong University of Science and Technology, Clear Water Bay, Hong Kong, China. E-mail: eekjchen@ust.hk
First published on 20th April 2026
Abstract
Piezoelectric materials are essential for electronics but suffer from thermal instability, especially lead-free alternatives to lead zirconate titanate (PZT). The absence of quantitative thermal stability metrics has hindered progress. In this work, we introduce a new index, denoted as “Sd”, which accounts for both the operating temperature range and the central operating temperature, allowing for a comprehensive assessment. Applying this criterion, we found that the commercial PZT-4 material exhibits the highest thermal stability among the ceramics tested, confirming its reputation as the industry standard. Notably, we developed a new lead-free piezoelectric ceramic based on bismuth ferrite that achieved a stable piezoelectric coefficient at 38–247 °C; the corresponding Sd value (173) exceeds that (163) of commercial PZT-8 (20–232 °C). Further analysis revealed that reducing the concentration of point defects improved its electrical homogeneity and reduced conductivity, leading to enhanced thermal stability. This work provides a new evaluation framework that can accelerate the development of thermally stable lead-free piezoelectric materials. The implications of this research extend beyond piezoelectrics, as the Sd index could find broader application in the characterization of other functional oxides.
1. Introduction
Piezoelectric materials can achieve mutual conversion between mechanical energy and electrical energy and are key components in sensors, transducers, and actuators.1–3 With the increasing requirement for piezoelectric devices in extreme environments, such as aerospace and deep petroleum exploration,4 large piezoelectricity with good temperature stability has become an urgent need. Due to its excellent performance,5–7 PZT dominates the market for more than half a century. Unfortunately, PZT is now facing worldwide legislative restrictions due to its toxicity, and this has triggered a worldwide search for Pb-free substitutes.7–12Currently, the main lead-free piezoelectric materials include BaTiO3 (BT)-based, (K, Na)NbO3 (KNN)-based, (Na0.5Bi0.5)TiO3 (NBT)-based and BiFeO3 (BF)-based perovskite systems. Among them, the BiFeO3-based ferroelectrics, such as bismuth ferrite–barium titanate (BiFeO3–BaTiO3, BF–BT), possess high Curie temperature and strong spontaneous polarization,13–17 making them potential alternatives to lead-containing piezoelectrics. However, high dielectric loss and high conductivity severely hinder the stability of their piezoelectric properties.13,15 It was reported that the small-signal piezoelectric coefficient presents a large fluctuation (as high as 50% from 25 °C to 150 °C),18 and the large-signal piezoelectric coefficient remains stable below 150 °C or even lower due to leakage.19–21
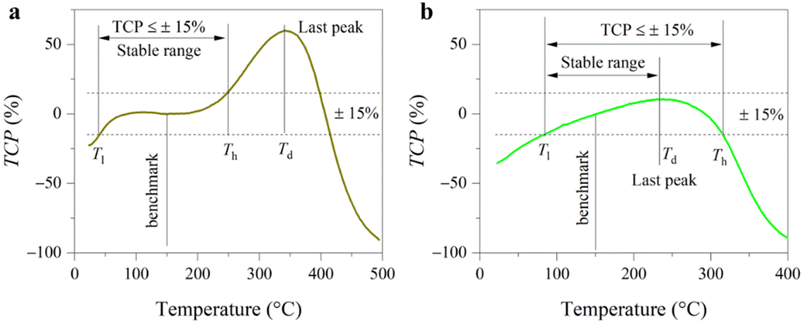
In addition, there is no international standard to evaluate the temperature stability of piezoelectricity. By referring to the evaluation method of the stability of capacitors,22–24 we know that the temperature coefficient of piezoelectricity (TCP) can be defined usually by the formula as follow:
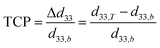 | (1) |
Here, based on the redefinition of the temperature-stable range, we propose a new criterion for evaluating the stability of piezoelectricity. Specifically, this criterion emphasizes that both high operating temperature and wide temperature range are crucial for temperature-stability (a new stability index) of piezoelectric coefficient d33. We calculated the index value of different piezoceramics and found that the highest value of up to 224 belongs to PZT-4, a well-known piezoceramic with the widest temperature range and high operating temperature. In contrast, within the lead-free piezoceramic category, we did not identify any materials that match the performance of their lead-based counterparts. To address this gap, we designed a new lead-free ceramic (Sm doped 0.7BiFeO3–0.3BaTiO3, composition near the morphotropic phase boundary15) with an index value of 173 (temperature-stable range of 38–247 °C), much higher than that of its undoped counterpart (152), and even comparable to that (163) of PZT-8 (20–232 °C). We further analyzed the origins of the temperature-stable piezoelectricity in the doped BiFeO3–BaTiO3 ceramics. Our results indicate that a significantly reduced concentration of point defects enhances electrical homogeneity and decreases conductivity, contributing to improved thermal stability.
2. Results and discussion
2.1 The new metric of temperature stability of piezoelectricity
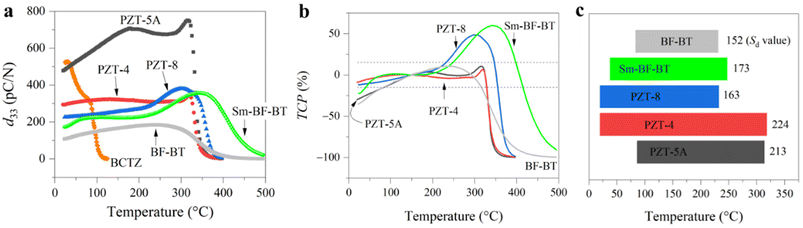
We first selected classical materials including commercial PZTs and lead-free ones to verify reliability. Fig. 2a shows the piezoelectric coefficients of BF–BT, Sm-doped BF–BT, (Ba, Ca) (Ti, Zr)O3 (BCTZ)27 and commercial PZTs at various temperatures. For the BCTZ ceramic, d33 is approximately 500 pC N−1 at room temperature, but it is extremely sensitive to temperature due to low Curie temperature and polymorphic phase transition.28,29 For the PZT family, their piezoelectric coefficients show no obvious decay up to nearly 300 °C. Among them, PZT-5A, a soft piezoelectric material, shows the highest d33 value and a large fluctuation with temperature. In comparison, hard PZTs (PZT-4 and PZT-8) show more stable d33 values. Interestingly, although the d33 value of Sm-BF–BT at room temperature is slightly lower than that of hard PZT-8, it is more resistant to high temperatures. We calculate the temperature coefficient of piezoelectricity (TCP) according to formula (1). In view that the high temperature piezoelectric devices need to work at 300 °C or above,4 the benchmark temperature was set to 150 °C. Therefore, the temperature-stable range of the piezoelectric coefficient is identified preliminary based on TCP ≤ ±15%,22 as shown in Fig. 2b. Apart from the TCP conditions, we considered the permanent decay of the piezoelectric coefficient at de-polarization temperature (Td). For PZT-4, PZT-5A and BF–BT, because Td is lower than the high temperature boundary (Th) determined by TCP conditions, the former is determined as the upper limit temperature (Tmax) of the temperature-stable range. Fig. 2c shows the stable range of d33 of lead-free BF–BT, Sm-BF–BT and PZTs. It can be found that PZT-4 has the widest temperature range (20–318 °C). PZT-8 (20–232 °C) is more stable than PZT-5A (86–314 °C) at below 200 °C, but it is inferior to the latter at higher temperature, even not as stable as lead-free Sm-BF–BT (38–247 °C). Obviously, this comparison is still not intuitive enough.To quantify this difference, we propose a new index to evaluate the stability of piezoelectric coefficient d33 (Sd) as follows:
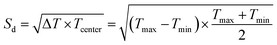 | (2) |
According to formula (2), we calculated the Sd value of lead-free BF–BT, Sm-BF–BT and PZTs, as marked in Fig. 2c. The highest Sd value was obtained in PZT-4, thanks to its wide temperature range and high upper limit temperature, which is consistent with previous cognition that PZT-4 is one of the most stable piezoelectric materials.30 Due to a low upper-limit temperature, the Sd value of PZT-8 is much lower than those of PZT-4 and PZT-5A. In addition, we find that the Sd value (173) of Sm doped BF–BT (temperature range of 38–247 °C) is higher than that (152) of its undoped counterpart, and even comparable to that (163) of PZT-8 (temperature range of 20–232 °C). We further check the influence of the threshold (Fig. S1) and benchmark temperature (Fig. S2) on the temperature-stable range and Sd value. The results indicate that only slight differences arise when the benchmark temperature is changed to 100 °C. In other cases, the prerequisites have no significant influence on the comparative results of Sd.
2.2 The enhanced and temperature-stable piezoelectricity in Sm-doped BF–BT ceramics
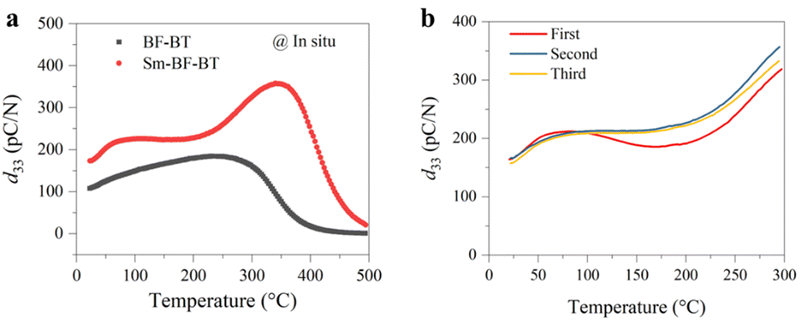
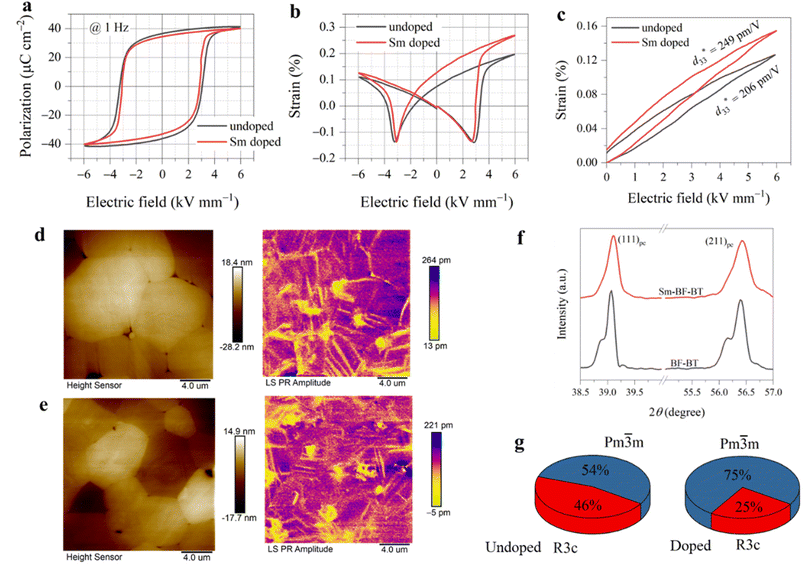
We next investigate the piezoelectricity and its temperature stability of the Sm-doped BF–BT ceramic. Here, the optimal doping content was determined through a preliminary experiment, in which 0.7BiFeO3–0.3Ba1−xSmxTiO3 (x from 0.00 to 0.04) ceramics were prepared and electrical properties were measured (Fig. S3). The result shows that 0.7BiFeO3–0.3Ba0.99Sm0.01TiO3 has the highest d33 value and dielectric permittivity at room temperature. Fig. 3a shows piezoelectric coefficients of undoped and Sm-doped BF–BT ceramics at various temperatures. On the one hand, at room temperature, d33 of the doped sample is about 175 pC N−1, which is much higher than that of its undoped counterpart. On the other hand, the d33 of the undoped sample starts to fall once the temperature approaches 230 °C; in comparison, d33 of the doped one shows no obvious decay until the temperature is near 340 °C, indicating better thermal stability. We further assess the reliability of piezoelectricity of the Sm-BF–BT ceramic. d33 of the poled sample is measured three times during the heating process from 20 to 300 °C, as shown in Fig. 3b. In the measurement, the sample was cooled to 20 °C before the next test starts. We can see that d33 under the second heating presents a small difference compared to that under the first measurement, and there is almost no difference between the third and second measurements, particularly in the temperature below 200 °C. These results suggest that the Sm-doped BF–BT ceramic has a good thermal cycling stability.We not only improved the piezoelectricity at room temperature but also enhanced the temperature stability by doping. First, we explored the mechanism behind the improvement of piezoelectric properties. Fig. 4a shows the polarization–electric field (P–E) hysteresis loop of the poled samples. After doping, the P–E loop shrinks, accompanied by a decrease in coercive field and remnant polarization, but it still exhibits a high remnant polarization of 34 µC cm−2. Fig. 4b shows their strain–electric field (S–E) loops. The asymmetric shape is because the sample was pre-poled with a DC field before the measurement, causing the alignment of defect dipoles.5,21 It is noted that the direction of defect dipoles is unchanged after the first electric treatment, but they can be stretched or contracted. The amplitude of strain is determined by the behaviors of defect dipoles and spontaneous polarization.5 The larger strain under the positive electric field is due to the initially applied electric field being the same as the polarization direction of the sample. In this case, the defect dipoles are stretched and spontaneous polarization is aligned along the applied field. Under the negative electric field, the defect dipoles are contracted and spontaneous polarization is also aligned along the applied field. Despite that, the P–E loop is almost symmetric because the effect of defect dipoles and spontaneous polarization on measured polarization is the same regardless of the direction of the applied electric field.5 From Fig. 4b, we can also observe a decrease in coercive field and a significant increase in strain for the doped sample. We further measured the strain under a unipolar electric field. As shown in Fig. 4c, the doped sample exhibited a larger unipolar strain, corresponding to an increase in the piezoelectric strain coefficient from 206 to 249 pm V−1. The macroscopic electrical properties are closely related to the microstructure and domain structure of the crystals.2,31 We observed the domain structure of the samples using piezoresponse force microscopy, as shown in Fig. 4d and e. Both samples exhibited clear ferroelectric domain morphology, and regular lamellar/strip domains dominate.32 Interestingly, the domain size significantly decreased, and the domain density increased after doping. This may be related to the reduced grain size (Fig. 4e and S4) and altered crystal structure.33,34
Fig. 4f and S5 show the X-ray diffraction patterns of the samples. Both undoped and doped samples exhibit a pure perovskite phase. After doping, the diffraction peaks shift towards higher angles, indicating a decrease in lattice volume. This is due to the fact that Sm3+ (1.24 Å) has a smaller radius compared to Ba2+ (1.61 Å).35 Additionally, it can be observed that the split diffraction peaks become less pronounced after doping. It has been reported that 0.7BF–0.3BT exhibits a crystal structure with coexisting rhombohedral R3c and pseudo-cubic Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phases.36,37 We performed fitting of the patterns using a mixed-phase of R3c and Pm
m phases.36,37 We performed fitting of the patterns using a mixed-phase of R3c and Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m. It can be observed that the content of the R3c phase significantly decreases after doping (Fig. 4g and Table S1), indicating an enhanced symmetry of the crystal structure. Previous reports have indicated that differences in valence and ionic radii lead to discontinuities in the crystal structure and polarization,2 increasing the interfacial energy of the system. Competition between the bulk and interfacial energy may lead to a flattened free-energy profile,38 consistent with the enhanced symmetry of the crystal structure and reduced domain size. In addition, the higher electronegativity of Sm3+ compared to Ba2+ increases the degree of local covalent bonding, and the hybridization of ionic and covalent bonds can also promote piezoelectricity.11
m. It can be observed that the content of the R3c phase significantly decreases after doping (Fig. 4g and Table S1), indicating an enhanced symmetry of the crystal structure. Previous reports have indicated that differences in valence and ionic radii lead to discontinuities in the crystal structure and polarization,2 increasing the interfacial energy of the system. Competition between the bulk and interfacial energy may lead to a flattened free-energy profile,38 consistent with the enhanced symmetry of the crystal structure and reduced domain size. In addition, the higher electronegativity of Sm3+ compared to Ba2+ increases the degree of local covalent bonding, and the hybridization of ionic and covalent bonds can also promote piezoelectricity.11
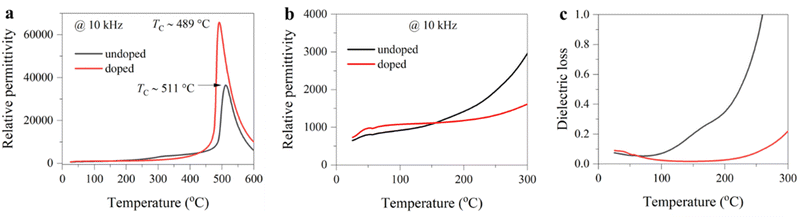
Second, we investigate the origin of enhanced thermal stability. It is well known that the Curie temperature of a ferroelectric is a critical factor affecting the stability of piezoelectricity.30 Fig. 5a and S6 present the temperature-dependent dielectric permittivity and loss of undoped and doped BF–BT ceramics. We can see that the undoped sample presents a high Curie temperature of ∼510 °C. After doping, the Curie temperature decreases to ∼490 °C. Generally, a higher Curie temperature corresponds to a higher thermal depolarization temperature and thus greater stability of the piezoelectric performance.30 However, in our work, the doped sample exhibits the higher operating temperature and enhanced piezoelectric stability (Fig. 3a). First, the temperature stability of electrical properties depends on the lattice symmetry. A ferroelectric with low lattice symmetry presents a large fluctuation with the temperature due to the fragmentation of long-range ferroelectric order. For ferroelectrics with high symmetry, the change is usually smooth.39 In BFO-based systems, there were multiple phase transitions from room temperature to the Curie temperature.40 After doping, the lattice symmetry increases, and phase transition becomes less abrupt; thus, the stability of dielectric permittivity and piezoelectricity is highly improved. As shown in Fig. S6, we can find that a small hump near 300 °C disappeared after Sm doping. Second, the point defect, such as oxygen vacancies, is also a vital factor that affects the temperature stability. By comparing the dielectric permittivity and loss at a lower temperature range (Fig. 5b and c), we can see that the undoped sample exhibits extremely unstable dielectric properties, particularly in terms of dielectric loss, where an obvious rise occurs at temperatures near 100 °C. Dielectric loss originates from electrically active regions such as point defects.41 For BF-based materials, bismuth volatilization, reduction of iron ions, and self-decomposition of BF can lead to the generation of oxygen vacancies.42 In contrast, the doped sample shows significantly enhanced stability in dielectric properties, with a dielectric loss below 0.1 in the temperature range from room temperature to 250 °C.
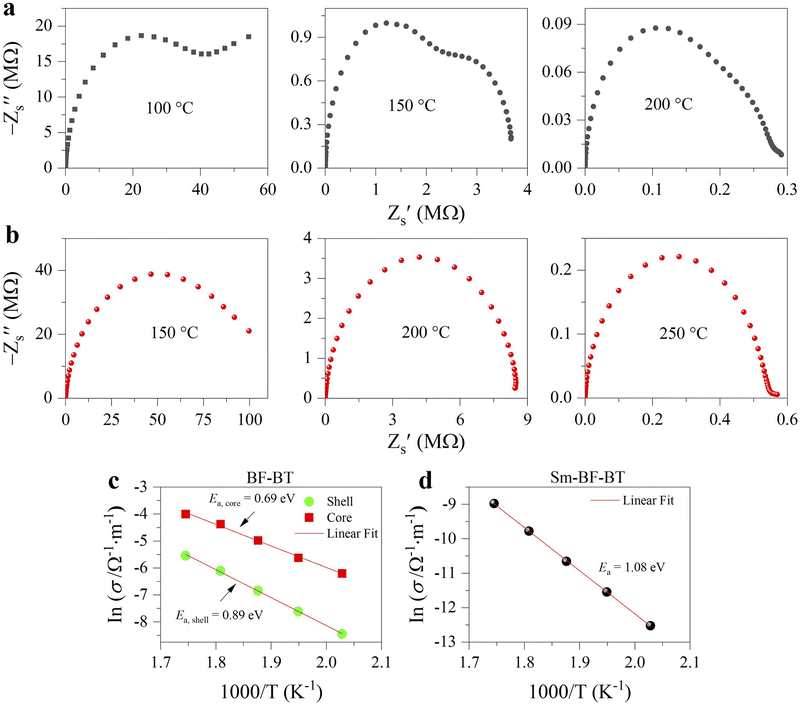
To further identify the point defects, we measured the impedance spectra of the samples at elevated temperatures. Fig. 6a displays the impedance complex plane plots of the undoped sample. Interestingly, we observe two semicircular arcs, indicating that the impedance of the sample is composed of two different electrical responses,43 namely grain and grain boundary, in the order of decreasing frequency. In contrast, in the doped sample, only a single arc can be observed in this temperature range, as shown in Fig. 6b. This electrical response implies that the resistance of grains in the doped sample is much larger than that in the undoped sample; therefore, the resistance of grains and grain boundaries becomes comparable. Previous reports pointed out the presence of a core–shell microstructure in BF–BT-based ceramics,44 consisting of an electrically active core and a resistive shell. As shown in Fig. S7, we can observe dark shells and bright cores within individual grains, representing Ba, Ti rich and Bi, Fe rich regions, respectively. The electrically active cores may be attributed to an abundance of defects.44 Additionally, we find that the resistance (the intersection points of the arc and the X-axis) of the doped sample increases by an order of magnitude than its undoped counterpart at the same temperature. To further understand the defect dynamics, quantitative analysis was conducted. The measured impedance spectra were fitted according to an equivalent circuit. For the undoped sample, a circuit consisting of two parallel resistance (R) and constant phase (Q) elements connected in series (Fig. S8) was adopted,45 while only a parallel R and Q element was required in the doped sample due to the single arc. The fitted curves are displayed in Fig. S9. Based on a previous report,45 the temperature dependence of the conductivities of undoped and doped ceramics was calculated and is presented in Fig. 6c and d. The conductivities of two specimens roughly show an Arrhenius-type behavior.46,47 It can be observed that the conductivity of the Sm doped sample is significantly lower than that of the undoped sample (core and shell). Moreover, corresponding activation energies (Ea) derived from the Arrhenius equation46 are given in Fig. 6c and d. For the undoped sample, core and shell activation energies are 0.69 eV and 0.89 eV, respectively. In comparison, the activation energy (1.08 eV) for the Sm doped sample is much higher than those of the undoped ones. The increased Ea further proves a decrease in oxygen vacancy concentration.48
In the BF-based system, both the reduction of Fe3+ and volatilization of Bi3+ lead to a decrease in positive charge.42 To maintain the electrical neutrality, extra oxygen vacancies are generated. The defect chemistry can be represented as follows:
These oxygen and cation vacancies are coupled to form defect dipoles, which increase the conductivity of the ceramic and generate leakage currents, especially at high temperatures or under strong electric fields. With the introduction of Sm3+ ions at the Ba site, compensating positive charges mitigate the generation of oxygen vacancies, as shown in the following formula:42w
After Sm doping, the main defect dipoles 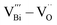 and
and 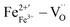 in the perovskite ABO3 structure were changed to
in the perovskite ABO3 structure were changed to 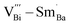 and
and 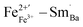 . Among them,
. Among them, 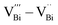 and
and 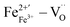 are along the 〈110〉 and 〈001〉 directions,49 and the defect dipole reorientation needs only 1 step of
are along the 〈110〉 and 〈001〉 directions,49 and the defect dipole reorientation needs only 1 step of 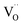 migration.5 In comparison, both
migration.5 In comparison, both 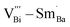 and
and 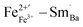 are along the 〈111〉 direction, which is consistent with the directions of ferroelectric polarization in the rhombohedral phase.3
are along the 〈111〉 direction, which is consistent with the directions of ferroelectric polarization in the rhombohedral phase.3 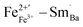 cannot move due to the absence of ion exchange channels, and the
cannot move due to the absence of ion exchange channels, and the 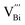 in defect dipole
in defect dipole 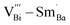 needs 2 steps of migration to realize reorientation, thus costing much more energy. Therefore, it is more difficult for Sm-doped BF–BT to depolarize, which will contribute to improved thermal stability at high temperature.
needs 2 steps of migration to realize reorientation, thus costing much more energy. Therefore, it is more difficult for Sm-doped BF–BT to depolarize, which will contribute to improved thermal stability at high temperature.
To investigate the impact of electrical homogeneity on the poling process and piezoelectric properties, we developed two physical models (Fig. S11 and Table S2). The first model consists of a conducting core and an insulating shell, simulating the undoped sample, while the second model features a core with conductivity similar to that of the shell, representing the doped sample. Using COMSOL 6.1 (Physics of current), we analyzed the polarization distribution (Fig. 7). Our results show that in electrically inhomogeneous dielectrics, the conducting core exhibits minimal polarization (Fig. 7a), as the electric field is primarily concentrated in regions with higher resistivity (Fig. S11). In contrast, electrically homogeneous dielectrics display more uniform polarization distribution in entire regions (Fig. 7b). Furthermore, the high-temperature ferroelectric measurements support these findings. As shown in Fig. S12, the undoped sample begins to exhibit leakage at 50 °C, whereas the doped sample remains stable until 150 °C. The significant leakage in the undoped sample leads to insufficient alignment of ferroelectric domains, which is reflected in the impedance and phase spectrum at the resonance frequency (Fig. S13).50 Notably, the maximum phase angle of the doped sample is significantly higher than that of the undoped sample. Our results clearly demonstrate that stable piezoelectricity at high temperatures is attributed to enhanced electrical homogeneity and reduced conductivity.
3. Conclusion
We introduce a novel metric to quantitatively assess the temperature stability of piezoelectricity, where exceptional temperature stability is characterized by a broad temperature range and high operating temperature concurrently. This metric has been validated for both commercial PZTs and lead-free piezoelectric materials. Although the temperature stability of lead-free Sm-doped BF–BT ceramics is inferior to that of commercial PZT-4 and PZT-5A, it surpasses that of commercial PZT-8. Notably, the enhanced temperature stability in the Sm-doped BF–BT ceramic, compared to its undoped counterpart, is attributed to the suppression of electrical inhomogeneity resulting from a reduction in defect concentration. This work is poised to serve as a paradigm for the design of high-temperature piezoelectrics and pave the way for the development of high-temperature electronic devices.4. Materials and methods
4.1 Material preparation
The 0.7BiFeO3–0.3Ba1−xSmxTiO3 (x = 0, 0.01, 0.02, 0.03, 0.04) ceramics were prepared by a conventional solid-state reaction. The raw powders, Bi2O3, Fe2O3, BaCO3, TiO2 and Sm2O3, were dried and then weighed according to the stoichiometric ratio. The powders were mixed by ball milling in ethanol. The dried mixture was calcined at 800 °C for 6 h and then was refined by ball milling in ethanol. The 5% PVA solution was added into the dried powder. The powders were formed into pellets by uniaxial pressing of ∼200 MPa, followed by binder burn-out at 600 °C. The samples were sintered at 980–1020 °C for 3 h. All sintered ceramics were annealed at 800 °C for 2 h in an air atmosphere, followed by an air-quenching process before the temperature drops. Subsequently, the surfaces of the samples were ground. Silver electrodes were prepared by magnetron sputtering (Q150T ES Plus, Quorum Technologies), followed by annealing at 400 °C. The samples were poled under an electric field of 4.5–5 kV mm−1 at 80–120 °C for 30 min. After aging for 24 h, the electrical properties were measured.The commercial PZTs (PZT-5A, PZT-4 and PZT-8) are purchased from Zhuhai Jiaming Electronic Technology Co., Ltd.
4.2 Material characterization
The bulk densities of the ceramics were measured using a density meter (XS104, Mettler Toledo, Switzerland). The crystal structures of the powder samples were examined with X-ray diffraction (D8 advance, Bruker). The lattice parameters and phase content were analyzed with Rietveld refinement software (Topas 3.0). The morphology and grain size of the sample were observed using a scanning electron microscope (Quanta 450, FEI). To observe the ferroelectric domain, the electrodes of the poled sample were ground off and the surfaces were polished. The ferroelectric domains were identified with an atomic force microscope (Dimension Icon with ScanAsyst, Bruker) in an optimized vertical domain mode. In the measurement, a conductive probe (Asyelec.02-R2, Asylum research) is utilized. Chemical states of oxygen were detected by X-ray photoelectron spectroscopy (Axis Supra+, Kratos Analytical).The capacitance and loss of the poled sample were tested with an inductance capacitance resistance meter (E4980A, Agilent Technologies) at temperatures of 20–600 °C with a heating rate of 3 °C min−1. The ferroelectric polarization and electric-field induced strain were measured using a ferroelectric test system (CPE1801, PolyK Technologies) equipped with a fotonic sensor (MTI2100, MTI Instruments). The high-temperature impedance in the frequency range of 10−2 Hz to 107 Hz was measured using a broadband impedance spectrometer (Concept 40, Novocontrol Technologies) under a small alternating voltage of 1 VRMS, and the impedance spectra were fitted using commercial software (ZSimpWin). The piezoelectric charge coefficients at various temperatures were tested using an in situ quasi-static d33 meter (TZFD-600, Harbin Julang Technology), with a heating rate of 3 °C min−1. The impedances and phase angles of poled samples near the resonance frequency were measured using an ENA network analyzer (E5061B, Keysight).
Conflicts of interest
There are no conflicts to declare.Data availability
All the data have been presented in the manuscript and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta09690a.Acknowledgements
The research was supported by the National Natural Science Foundation of China (Project No. T2522031), the Hong Kong Research Grants Council (General Research Fund Project No. 16214025), and the Innovation and Technology Fund (Project No. ITS/232/24) of the Innovation and Technology Commission, the Government of the Hong Kong Special Administrative Region. We thank Prof. Dragan Damjanovic for revising the manuscript.References
- G. H. Haertling, J. Am. Ceram. Soc., 1999, 82, 797–818 CrossRef CAS.
- F. Li, D. B. Lin, Z. B. Chen, Z. X. Cheng, J. L. Wang, C. C. Li, Z. Xu, Q. W. Huang, X. Z. Liao, L. Q. Chen, T. R. Shrout and S. J. Zhang, Nat. Mater., 2018, 17, 349–354 CrossRef CAS PubMed.
- X. Ren, Nat. Mater., 2004, 3, 91–94 CrossRef CAS PubMed.
- Y. Dong, K. Zou, R. Liang and Z. Zhou, Prog. Mater. Sci., 2023, 132, 101026 CrossRef CAS.
- G. Huangfu, K. Zeng, B. Wang, J. Wang, Z. Fu, F. Xu, S. Zhang, H. Luo, D. Viehland and Y. Guo, Science, 2022, 378, 1125–1130 CrossRef PubMed.
- J. Rödel, K. G. Webber, R. Dittmer, W. Jo, M. Kimura and D. Damjanovic, J. Eur. Ceram. Soc., 2015, 35, 1659–1681 CrossRef.
- J. Wu, D. Xiao and J. Zhu, Chem. Rev., 2015, 115, 2559–2595 CrossRef CAS PubMed.
- S. Kim, R. Miyauchi, Y. Sato, H. Nam, I. Fujii, S. Ueno, Y. Kuroiwa and S. Wada, Adv. Mater., 2023, 35, 2208717 CrossRef CAS PubMed.
- M.-H. Zhang, C. Shen, C. Zhao, M. Dai, F.-Z. Yao, B. Wu, J. Ma, H. Nan, D. Wang, Q. Yuan, L. L. da Silva, L. Fulanović, A. Schökel, P. Liu, H. Zhang, J.-F. Li, N. Zhang, K. Wang, J. Rödel and M. Hinterstein, Nat. Commun., 2022, 13, 3434 CrossRef CAS PubMed.
- G. Wang, T. Hu, W. Zhu, Z. Lu, A. Kleppe, M. Diaz Lopez, A. Feteira, D. C. Sinclair, Z. Fu, H. Huang, D. Wang and I. M. Reaney, Phys. Rev. Lett., 2023, 130, 076801 CrossRef CAS PubMed.
- Y. Saito, H. Takao, T. Tani, T. Nonoyama, K. Takatori, T. Homma, T. Nagaya and M. Nakamura, Nature, 2004, 432, 84–87 CrossRef CAS PubMed.
- M. Waqar, H. Wu, J. S. Chen, K. Yao and J. Wang, Adv. Mater., 2022, 34, 2106845 CrossRef CAS PubMed.
- J. Wu, Z. Fan, D. Xiao, J. Zhu and J. Wang, Prog. Mater. Sci., 2016, 84, 335–402 CrossRef CAS.
- J. Wu, J. Appl. Phys., 2020, 127, 190901 CrossRef CAS.
- D. Wang, G. Wang, S. Murakami, Z. Fan, A. Feteira, D. Zhou, S. Sun, Q. Zhao and I. M. Reaney, J. Adv. Dielectr., 2018, 08, 1830004 CrossRef CAS.
- Z. Shen, J. Guo, X. Gao, W. Xuan, J. Zhang, D. Wang, J. Cheng, S. Zhang and J. Chen, J. Materiomics, 2025, 11, 100946 CrossRef.
- H. Li, J. Zhao, Y. Li, L. Chen, X. Chen, H. Qin, H. Zhou, P. Li, J. Guo and D. Wang, ACS Appl. Mater. Interfaces, 2024, 16, 9078–9087 CrossRef CAS PubMed.
- M. H. Lee, D. J. Kim, J. S. Park, S. W. Kim, T. K. Song, M.-H. Kim, W.-J. Kim, D. Do and I.-K. Jeong, Adv. Mater., 2015, 27, 6976–6982 CrossRef CAS PubMed.
- J. Chen, J. E. Daniels, J. Jian, Z. Cheng, J. Cheng, J. Wang, Q. Gu and S. Zhang, Acta Mater., 2020, 197, 1–9 CrossRef CAS.
- G. Wang, Z. Fan, S. Murakami, Z. Lu, D. A. Hall, D. C. Sinclair, A. Feteira, X. Tan, J. L. Jones, A. K. Kleppe, D. Wang and I. M. Reaney, J. Mater. Chem. A, 2019, 7, 21254–21263 RSC.
- J. Lin, F. Lv, Z. Hong, B. Liu, Y. Wu and Y. Huang, Adv. Funct. Mater., 2024, 34, 2313879 CrossRef CAS.
- W. Jia, Y. Hou, M. Zheng, Y. Xu, M. Zhu, K. Yang, H. Cheng, S. Sun and J. Xing, IET Nanodielectr., 2018, 1, 3–16 CrossRef.
- W. Jia, Y. Hou, M. Zheng, Y. Xu, X. Yu, M. Zhu, K. Yang, H. Cheng, S. Sun and J. Xing, J. Am. Ceram. Soc., 2018, 101, 3468–3479 CrossRef CAS.
- American National Standard, Ceramic Dielectric Capacitors Classes I, II, III, and IV. Part 1, Characteristics and requirements, Electronic Components, Assemblies & Materials Association, EIA, Arlington, VA, 2002 Search PubMed.
- J.-S. Zhou, K. Wang, F.-Z. Yao, T. Zheng, J. Wu, D. Xiao, J. Zhu and J.-F. Li, J. Mater. Chem. C, 2015, 3, 8780–8787 RSC.
- T. Zheng, H. J. Wu, Y. Yuan, X. Lv, Q. Li, T. L. Men, C. Zhao, D. Q. Xiao, J. G. Wu, K. Wang, J. F. Li, Y. L. Gu, J. Zhu and S. J. Pennycook, Energy Environ. Sci., 2017, 10, 528–537 RSC.
- X. D. Yan, M. P. Zheng, X. Gao, M. K. Zhu and Y. D. Hou, Acta Mater., 2020, 187, 29–40 CrossRef CAS.
- W. F. Liu and X. B. Ren, Phys. Rev. Lett., 2009, 103, 257602 CrossRef PubMed.
- D. Xue, Y. Zhou, H. Bao, C. Zhou, J. Gao and X. Ren, J. Appl. Phys., 2011, 109, 054110 CrossRef.
- M. Fang, S. Rajput, Z. Dai, Y. Ji, Y. Hao and X. Ren, Acta Mater., 2019, 169, 155–161 CrossRef CAS.
- C. R. Qiu, B. Wang, N. Zhang, S. J. Zhang, J. F. Liu, D. Walker, Y. Wang, H. Tian, T. R. Shrout, Z. Xu, L. Q. Chen and F. Li, Nature, 2020, 577, 350–354 CrossRef CAS PubMed.
- B. Xun, A. Song, J. Yu, Y. Yin, J. F. Li and B. P. Zhang, ACS Appl. Mater. Interfaces, 2021, 13, 4192–4202 CrossRef CAS PubMed.
- P. Zheng, J. L. Zhang, Y. Q. Tan and C. L. Wang, Acta Mater., 2012, 60, 5022–5030 CrossRef CAS.
- D. Ghosh, A. Sakata, J. Carter, P. A. Thomas, H. Han, J. C. Nino and J. L. Jones, Adv. Funct. Mater., 2014, 24, 885–896 CrossRef CAS.
- R. D. Shannon, Acta Crystallogr., 1976, 32, 751–767 CrossRef.
- M. M. Kumar, A. Srinivas and S. V. Suryanarayana, J. Appl. Phys., 2000, 87, 855–862 CrossRef CAS.
- S. Kim, G. P. Khanal, H.-W. Nam, I. Fujii, S. Ueno, C. Moriyoshi, Y. Kuroiwa and S. Wada, J. Appl. Phys., 2017, 122, 164105 CrossRef.
- Y. Nahas, A. Akbarzadeh, S. Prokhorenko, S. Prosandeev, R. Walter, I. Kornev, J. Iniguez and L. Bellaiche, Nat. Commun., 2017, 8, 15944 CrossRef CAS PubMed.
- F.-Z. Yao, K. Wang, W. Jo, K. G. Webber, T. P. Comyn, J.-X. Ding, B. Xu, L.-Q. Cheng, M.-P. Zheng, Y.-D. Hou and J.-F. Li, Adv. Funct. Mater., 2016, 26, 1217–1224 CrossRef CAS.
- D. V. Karpinsky, M. V. Silibin, S. V. Trukhanov, A. V. Trukhanov, A. L. Zhaludkevich, S. I. Latushka, D. V. Zhaludkevich, V. A. Khomchenko, D. O. Alikin, A. S. Abramov, T. Maniecki, W. Maniukiewicz, M. Wolff, V. Heitmann and A. L. Kholkin, Nanomaterials, 2020, 10, 801 CrossRef CAS PubMed.
- G. Liu, S. Zhang, W. Jiang and W. Cao, Mater. Sci. Eng. R, 2015, 89, 1–48 CrossRef PubMed.
- M. Habib, P. Ahmad, F. Akram, I. Kebaili, A. Rahman, I. U. Din, M. Javid Iqbal, M.-H. Kim, S. Lee, M. Uddin Khandaker, H. Goo Yeo, A. Karoui and T. K. Song, Ceram. Int., 2022, 48, 26608–26617 CrossRef CAS.
- N. Hirose and A. R. West, J. Am. Ceram. Soc., 1996, 79, 1633–1641 CrossRef CAS.
- G. Wang, J. Li, X. Zhang, Z. Fan, F. Yang, A. Feteira, D. Zhou, D. C. Sinclair, T. Ma, X. Tan, D. Wang and I. M. Reaney, Energy Environ. Sci., 2019, 12, 582–588 RSC.
- X. Guo and Z. Zhang, Acta Mater., 2003, 51, 2539–2547 CrossRef CAS.
- F. D. Morrison, D. C. Sinclair and A. R. West, J. Am. Ceram. Soc., 2001, 84, 531–538 CrossRef CAS.
- Y. Deng, J. Dai, Z. Fan, W. Yue, F. Huang, A. A. Mohamad, A. Evcin, Y. Tabak, G. Abdurakhmanov, M. Abu Abdeen and D. Wang, Ceram. Int., 2025, 51, 55067–55072 CrossRef CAS.
- O. Raymond, R. Font, N. Suarez-Almodovar, J. Portelles and J. M. Siqueiros, J. Appl. Phys., 2005, 97, 084107 CrossRef.
- Z. Zhao, Y. Lv, Y. Dai and S. Zhang, Acta Mater., 2020, 200, 35–41 CrossRef CAS.
- B.-W. Xun, N. Wang, B.-P. Zhang, X.-Y. Chen, Y.-Q. Zheng, W.-S. Jin, R. Mao and K. Liang, Ceram. Int., 2019, 45, 24382–24391 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |