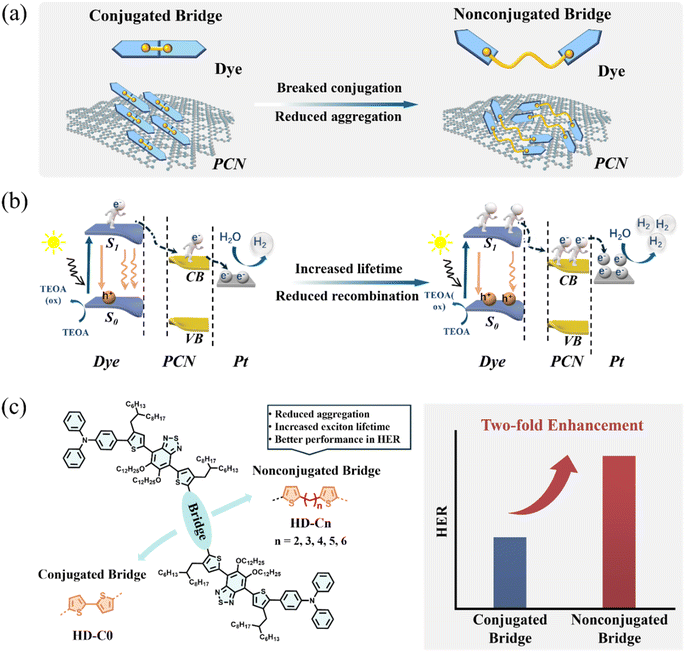
Modulating molecular aggregates via nonconjugated bridges for enhanced photocatalytic hydrogen evolution
Changzun Jianga,
Yanting Chena,
Wentao Yuana,
Qianqian Li*a and
Zhen Li *ab
*ab
aHubei Key Lab on Organic and Polymeric Opto-Electronic Materials, Department of Chemistry, Wuhan University, Wuhan 430072, China. E-mail: liqianqian@whu.edu.cn; lizhen@whu.edu.cn; Web: https://ligroup.whu.edu.cn/
bCollege of Chemistry and Chemical Engineering, Hubei University, Wuhan 430062, China
First published on 9th February 2026
Abstract
Dye/polymer carbon nitride (PCN) composite systems demonstrate considerable promise for photocatalytic hydrogen evolution (PHE), owing to their extensive spectral absorption range and enhanced charge separation efficiency. However, severe charge recombination, often arising from the inherent molecular structure of organic dyes and their consequent aggregation tendencies, remains a critical issue, thereby presenting a major challenge in modulating electronic processes. Herein, this study employs integrated experimental and theoretical approaches to elucidate the impact of the bridge mode (conjugated versus nonconjugated) between electron donor (D) and acceptor (A) moieties on the aggregation behavior, recombination processes, and photocatalytic performance of dyes. Key findings reveal that the nonconjugated bridge of alkyl chains can efficiently hinder non-radiative transitions and charge recombination by twisted molecular conformations with suppressed intermolecular π–π interactions, thereby facilitating charge carrier transport. Consequently, organic dyes with nonconjugated bridges exhibit superior exciton dissociation and charge transport, which results in a significantly enhanced HER of 859.47 µmol h−1, representing a more than two-fold increase over that of the conjugation-linked analogue dye (401.31 µmol h−1). This work establishes bridge engineering as a powerful molecular-level strategy to control aggregation and charge recombination in organic photosensitizers, opening new avenues for designing highly efficient photocatalytic materials.
Introduction
Photocatalytic hydrogen evolution (PHE), which harnesses solar energy to split water molecules, stands as a highly promising strategy for sustainable hydrogen fuel production.1–12 PHE was initiated by the landmark discovery of Fujishima and Honda in 1972, who demonstrated water splitting using titanium dioxide under ultraviolet light.13 Over the past few decades, numerous excellent photocatalysts have been developed, and the PHE efficiency has exceeded 900 mmol h−1 g−1.14–27 Among various PHE materials, PCN possesses notable characteristics, including a high specific surface area and abundant active sites, offering broad application potential.28–33 However, its wide bandgap restricts light absorption primarily to the ultraviolet region, which constitutes only about 4% of the solar spectrum.34–36 Moreover, the rapid recombination of photogenerated electron–hole pairs and the occurrence of backward reactions significantly limit its hydrogen evolution efficiency. Dye sensitization has emerged as a promising strategy to address these limitations by extending the excitation wavelength range into the visible light region, facilitating exciton dissociation, and improving charge carrier transport, thereby enhancing the overall photocatalytic hydrogen production performance of PCN.37–40In dye-sensitized PHE, the deposition behaviour of dye molecules on the catalyst surface critically determines the overall catalytic performance. Although substantial research has been devoted to this subject (Table S1), most studies have focused primarily on the anchoring modes of dye molecules, while their aggregation behaviour on catalyst surfaces has been rarely investigated. However, it is well established in PHE that improper molecular aggregation can induce significant charge recombination, thereby impeding exciton dissociation and charge carrier transport.41 Recently, Cooper and co-workers observed that (2,6-bis(4-cyanophenyl)-4-(9-phenyl-9H-carbazol-3-yl)pyridine-3,5-dicarbonitrile) (CNP) with different aggregate states exhibited contrasting PHE performance, which can be ascribed to the extent of excited state delocalization and the excited state dynamics in different aggregate states.42 Notably, both exciton dissociation and charge carrier mobility are key factors governing photocatalytic efficiency. Therefore, precise control over the aggregation state of dye molecules is essential for improving photocatalytic performance.
Herein, we synergistically regulate the electronic structure and aggregation behaviours of organic dyes through varying bridge modes (conjugated-bridge vs. nonconjugated-bridge) between D and A moieties. This approach effectively suppresses severe aggregation, minimizes charge recombination, and thereby extends the charge carrier lifetime.43,44 Compared to the control dye HD-C0 with a conjugated bridge, dyes with varied lengths of alkyl chains (n = 2–6) as non-conjugated bridges exhibit suppressed aggregation (Fig. 1a), reduced recombination (Fig. 1b), and prolonged exciton lifetime. Consequently, nonconjugated chain-linked dyes demonstrated enhancement in exciton lifetime and a significantly higher photocatalytic hydrogen evolution rate (HER) of 859.47 µmol h−1 was achieved (Fig. 1c) by dye HD-C4, a more than two-fold increase over that of dye HD-C0 (401.31 µmol h−1). Thus, the introduction of a non-conjugated bridge as a novel molecular design strategy for organic dyes effectively addresses the key challenges of aggregation and recombination at the dye/PCN interface, resulting in a performance leap for photocatalytic hydrogen production.
Results and discussion
Design and synthesis of organic dyes
Organic dyes possessed two units of D–A moieties with triphenylamine (TPA) (D) and benzothiadiazole (A), as the key units for light-harvesting and charge carrier transport. Their molecular conformations were adjusted by the different bridge modes between them, including conjugated and nonconjugated ones. The nonconjugated bridges were constructed by alkyl chains with different lengths, which can tune the molecular arrangement of D–A moieties on the dye/PCN interface. The D–A moieties were synthesized using the same methods as in our previous work, and both conjugated and nonconjugated bridge moieties were incorporated by Stille-coupling reactions. These target dyes demonstrate excellent solubility in common organic solvents due to their long, branched side chains, with their molecular structures being characterized by 1H NMR, 13C NMR, MALDI-TOF (MS), and HPLC (Fig. S1–S14).Photophysical properties of organic dyes
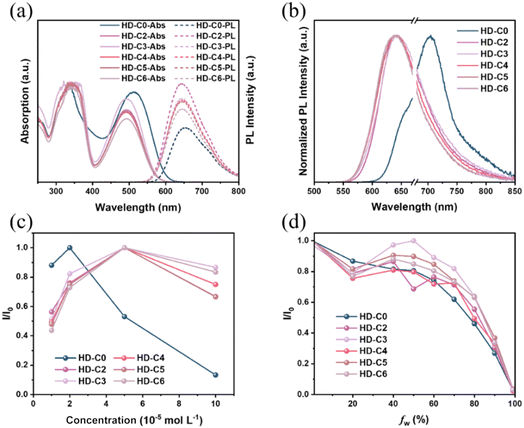
Through the incorporation of alkyl chains as the nonconjugated bridges, UV-Vis absorption spectra in diluted dichloromethane solution (concentration: 1 × 10−5 mol L−1) demonstrated slight blue-shifts with the maximum absorption wavelength changing from 512 nm (HD-C0) to 493 nm (HD-C2–HD-C6) (Fig. 2a). Accordingly, the molar extinction coefficients decreased from 8.5 × 104 L mol−1 cm−1 (HD-C0) to 5.4–7.1 × 104 L mol−1 cm−1 (HD-C2–HD-C6) (Fig. S15 and S16). Accordingly, the highest occupied molecular orbital (HOMO) energy levels (EHOMO) of dyes HD-C2–HD-C6 (around −5.05 eV) were slightly higher than that of HD-C0 (−5.09 eV), as detected by cyclic voltammetry (CV) (Fig. S17 and S18b). The energy gaps (Eg) were determined using the cutoff absorption in UV-Vis absorption (λonset) spectra (Fig. 2a) and calculated using the equation Eg = 1240/λonset. It was found that the Egs values of HD-C2–HD-C6 (from 2.18 eV to 2.19 eV) were higher than that of HD-C0 due to their blue-shifted absorption. Thus, the corresponding lowest unoccupied molecular orbital (LUMO) energy levels (ELUMO) of HD-C2–HD-C6 were calculated to be around −2.87 eV (calculated using the equation ELUMO = EHOMO + Eg), which were markedly higher than that of HD-C0 (−3.02 eV) (Table 1). These values are in good agreement with the UPS data (Fig. S19 and S20), thus validating their accuracy. Thus, upon dye sensitization, the dyes with nonconjugated bridges demonstrated larger energy offset with the substrate (PCN/Pt), thereby facilitating electron transfer.| Compound | λmax,absa (nm) | λonset,absa (nm) | Egb (eV) | EHOMOc (eV) | ELUMOd (eV) | λmax,PLe (nm) | λmax,PLf (nm) | PLQYg (%) |
|---|---|---|---|---|---|---|---|---|
| a The maximum absorption wavelengths and onset wavelengths of the absorption spectra of dyes in dichloromethane solution at a concentration of 1 × 10−5 mol L−1.b The optical band gaps of dyes as measured using the cutoff absorption in UV-Vis absorption spectra and calculated using the equation Eg = 1240/λonset.c HOMO energy levels as calculated by CV in dichloromethane solution at a concentration of 1 × 10−6 mol L−1.d LUMO energy levels as calculated by using ELUMO = EHOMO + Eg.e The maximum emission wavelengths of fluorescence spectra of dyes in dichloromethane solution at a concentration of 1 × 10−5 mol L−1.f The maximum emission wavelengths of fluorescence spectra of dyes in the solid state.g PLQY of dyes in the solid state. | ||||||||
| HD-C0 | 512 | 599 | 2.07 | −5.09 | −3.02 | 654 | 705 | 37.69 |
| HD-C2 | 492 | 568 | 2.18 | −5.06 | −2.88 | 643 | 638 | 56.53 |
| HD-C3 | 493 | 567 | 2.19 | −5.05 | −2.86 | 644 | 645 | 63.72 |
| HD-C4 | 492 | 568 | 2.18 | −5.05 | −2.87 | 643 | 639 | 60.87 |
| HD-C5 | 492 | 568 | 2.18 | −5.05 | −2.87 | 645 | 641 | 54.83 |
| HD-C6 | 493 | 567 | 2.19 | −5.04 | −2.85 | 644 | 639 | 61.28 |
Given that the dye-sensitized catalytic performance is closely related to the excited-state properties of organic dyes, photoluminescence properties were measured to characterize their excited-state behaviours. In diluted dichloromethane solutions (1 × 10−5 mol L−1) as an isolated state, dyes (HD-C2–HD-C6) with nonconjugated bridges exhibited a maximum emission wavelength of 642 nm, which is slightly blue-shifted (11 nm) compared to that of dye HD-C0 (653 nm). Meanwhile, dyes with nonconjugated bridges show much higher emission intensities than dyes with conjugated bridges under the same conditions (Fig. 2a). Theoretical calculations were conducted to elucidate their distinct photophysical properties (Fig. S21). Conformational optimization showed that dyes with nonconjugated bridges (HD-C2–HD-C6) demonstrated larger dihedral angles between the adjacent thiophene units (>33°) versus the HD-C0 analog (16.5°) (Fig. S22). Their variations in the conjugation effect throughout the whole molecule can be observed by the different distributions of HOMOs and LUMOs (Fig. S21). For all dyes, the LUMO is consistently located on the benzothiadiazole moiety, while a significant difference is observed in HOMO distribution between conjugated and non-conjugated systems. In the conjugated dye HD-C0, the HOMO is primarily localized on the central thiophene units. In contrast, for the non-conjugated dyes HD-C2–HD-C6, the HOMO is predominantly distributed over the stronger electron-donating triphenylamine moiety. We propose that this deliberate shift in HOMO distribution plays a crucial role; it elevates the HOMO energy via the stronger electron donor, thereby offsetting the energy-gap widening typically caused by conjugation interruption. Thus, it results in only minor differences in the photophysical properties between conjugated and non-conjugated bridging dyes in their dilute solution.
Upon aggregation in solid states, the dyes exhibited a pronounced red-shift in their UV-Vis DRS absorption spectra compared to those in dilute solution (Fig. S23). The most substantial redshift (78 nm) was observed for HD-C0. This value decreased with the introduction of longer alkyl chains, following the order: HD-C2 (71 nm) > HD-C3 (51 nm) ≈ HD-C4 (58 nm) ≈ HD-C5 (50 nm) ≈ HD-C6 (55 nm). While the solid-state emission of HD-2–HD-6 remained largely unchanged from their solution spectra (λmax ≈ 640 nm), the HD-C0 analogue displayed a substantially red-shifted emission at approximately 715 nm (Fig. 2b), highlighting the critical role of alkyl chains to suppress the severe aggregation in the solid states by twisted molecular conformations.
Moreover, dyes HD-2–HD-6 with nonconjugated bridges exhibited significantly longer lifetimes (3.76–6.57 ns) and higher PLQYs (54.83–63.72%, Fig. S18a) compared to HD-C0 with a conjugated bridge (1.70 ns and 37.69%, respectively). This consistent enhancement in both parameters collectively indicated that non-radiative transitions are substantially suppressed by the non-conjugated bridges. Such suppressed non-radiative decay is generally beneficial for exciton dissociation and charge carrier transport, thereby ultimately enhancing the HER.
The variations of aggregation behaviours of dyes bearing conjugated and nonconjugated bridges were further investigated by the emission properties under different conditions. First, the dependence of fluorescence intensity on dye concentrations was measured (Fig. 2c, S24 and S25). With the increased concentrations of solutions from 1 × 10−5 mol L−1 to 1 × 10−4 mol L−1 in dichloromethane solution, the emission intensities first increased and then decreased due to the concentration quenching effect. For dyes HD-C2–HD-C6, the inflection point is located at a concentration of 5 × 10−5 mol L−1, while that of dye HD-C0 is located at a lower concentration of 2 × 10−5 mol L−1, and the emission intensity decreased more rapidly with increasing concentrations compared to that of the dyes with nonconjugated bridges (Fig. 2c). Additionally, we performed concentration-dependent PLQY measurements in dichloromethane solution (Fig. S37). The nonconjugated dyes HD-C2–HD-C6 retained high PLQYs (89.03–92.91%) with relatively mild concentration quenching (>70% retained). In contrast, the conjugated dye HD-C0 not only started with a lower PLQY (41.83%), but also suffered from a drastic reduction, retaining only ∼20.34% at high concentrations. These results collectively corroborate that HD-C0 undergoes significant intermolecular aggregation at higher concentrations, which is consistent with the trends observed in fluorescence intensity measurements. This means that the traditional dye with conjugated bridges can easily aggregate with others due to strong π–π interaction forces and results in severe non-radiative recombination, which is consistent with its lower fluorescence lifetimes and PLQY in the solid state. In contrast, the dyes with nonconjugated bridges effectively avoided the aforementioned problems by suppressing severe aggregation. This can be further confirmed by the varied quenching effect by the addition of water as a poor solvent to the dilute tetrahydrofuran solutions (Fig. 2d and S26).45 A notable divergence emerged as the water fraction was raised from 20% to 40%: the emission intensities of dyes HD-C2–HD-C6 increased, while that of HD-C0 diminished. Upon further increasing the water fraction beyond 60%, HD-C0 exhibited a sharp decline in emission intensity, markedly faster than that of dyes with nonconjugated bridges. This accelerated quenching is consistent with more substantial aggregation behaviour in HD-C0. Thus, the incorporation of alkyl chains as nonconjugated bridges can efficiently suppress the severe aggregation of π-conjugated systems, as non-radiative recombination, benefiting the carrier transportation at the dye/PCN interface.
To complement the aggregate-level picture provided by excited-state measurements, we employed femtosecond transient absorption spectroscopy (fs-TAS) for assessment of the excited states (Fig. 3a–f). The excited-state dynamics of the dyes were characterized in dilute solution (3 × 10−5 mol L−1, λex: 450 nm). This concentration was chosen to be representative of the PCN loading conditions while retaining a strong signal, thus providing a baseline of their photophysical behaviour before being loaded onto PCN/Pt. The results reveal distinct excited-state behaviours between HD-C0 and HD-C2–HD-C6 with conjugated and nonconjugated bridges, respectively. As shown in Fig. 3g, the decay trace at 880 nm for dyes with nonconjugated bridges (HD-C2–HD-C6) exhibits a near-linear profile and does not fully recover to the baseline within the measured 5 ns, indicating rapid population of long-lived deep trap states following photoexcitation. In contrast, the dye with the conjugated bridge (HD-C0) displays biphasic exciton dynamics, with a short lifetime (τ1 = 9.86 ps) attributed to rapid exciton recombination, followed by a longer-lived component (τ2 > 1 ns). This suggests that the nonconjugated-bridge-strategy can effectively suppress fast recombination, thereby facilitating exciton dissociation and charge carrier migration to enhance PHE performance.
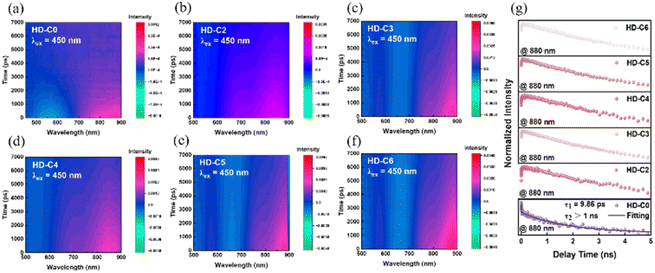 | ||
| Fig. 3 (a)–(f) Transient absorption (TA) kinetics of dyes in dilute dichloromethane solution (λex: 450 nm, concentration: 3 × 10−5 mol L−1). (g) Lifetime fitting curves of dyes at 880 nm. | ||
The characterization of dye/PCN/Pt systems
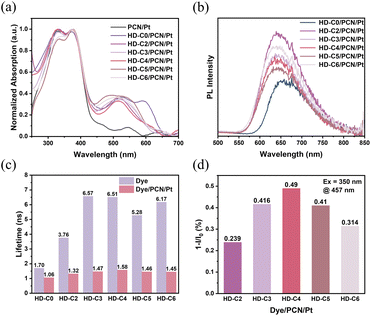
The preparation of the dye/PCN/Pt systems as photocatalysts was according to our previous work, and the concentration of dye loading is 2 wt%. Fourier Transform Infrared (FTIR) spectroscopy and XRD spectroscopy confirmed that the PCN/Pt framework remains intact during the subsequent doping of the dye onto the PCN/Pt surface (Fig. S29). The morphology of photocatalysts was characterized using a transmission electron microscope (TEM) and scanning electron microscope (SEM) (Fig. S30). Both of them showed lamellar morphology for these dye/PCN/Pt catalysts (Fig. 4d–i). Elemental mapping confirmed that Pt particles were uniformly deposited as small, well-dispersed particles on the PCN surface, and the crystalline structure of the Pt particles was further identified using a High-Resolution Transmission Electron Microscope (HRTEM), where their (111) plane had an interplanar spacing of 0.2 nm (Fig. S31). This indicates that the crystal structure of Pt remained intact following the doping of organic dyes. No distinct morphology for the dyes could be resolved, which may be attributed to their low concentration (Fig. S32).After being doped with dyes, the UV-Vis DRS absorption spectra of dye/PCN/Pt systems show obvious broadened absorption in the visible region relative to bare PCN/Pt powder (Fig. 4a). The maximum absorption wavelength of HD-C0/PCN/Pt is located at 600 nm, representing a red-shift of approximately 88 nm relative to that of HD-C0 in dilute solution (512 nm) (Fig. 2a and S23). In contrast, the absorption spectra of HD-C2 to HD-C6/PCN/Pt showed a maximum absorption wavelength of around 520 nm, with only a 27 nm red-shift compared to the corresponding dyes in solution. This indicates distinct aggregation behaviours at the dye/PCN interface. For dyes HD-C2 to HD-C6, which feature alkyl chains of varying lengths as non-conjugated bridges, their twisted conformations effectively suppress severe dye aggregation at the PCN/Pt interface, thereby favouring the HER.
Subsequently, the fluorescence spectra of dye/PCN/Pt were measured to characterize their excited states. Upon light irradiation (350 nm), both the fluorescence peaks of PCN/Pt (located at 457 nm) and the dyes (located at around 650 nm) are observed simultaneously. When 450 nm is used as the excitation wavelength, only the fluorescence peaks of the dyes are observed, due to the absence of visible-light-harvesting ability of PCN/Pt (Fig. S33a). Meanwhile, HD-C2–HD-C6/PCN/Pt systems demonstrated slightly blue-shifted emission spectra compared to that of HD-C0/PCN/Pt (Fig. 4b), which is consistent with the above-mentioned fluorescence emission characteristics of bare dyes. Compared to bare PCN/Pt, the fluorescence intensity of dye/PCN/Pt at 450 nm decreased largely, indicating the possible charge/energy transfer between dyes and PCN/Pt. As shown in Fig. 4d, the fluorescence quenching ratios followed the order: HD-C4 (49.0%) > HD-C3 (41.6%) > HD-C5 (41.0%) > HD-C6 (31.4%) > HD-C2 (23.9%). The superior quenching of HD-C4 suggests the most efficient electron transfer in the initial step of the photocatalytic cycle. We propose that a chain length of n = 4 may optimally facilitate electron coupling between organic dyes and PCN, promoting the key dynamic electron transfer process.
To probe the charge-transfer dynamics, we measured the fluorescence lifetimes of dyes anchored on PCN/Pt, revealing a marked contrast between those with conjugated and non-conjugated bridges (Fig. 4c). For HD-C0/PCN/Pt, the fluorescence lifetime decreased only slightly from 1.70 ns (HD-C0) to 1.06 ns (HD-C0/PCN/Pt), corresponding to a 38% reduction, which implies inefficient charge transport. In contrast, the HD-C2–HD-C6/PCN/Pt systems showed a pronounced shortening of lifetimes, declining from 3.76, 6.57, 6.51, 5.28, and 6.17 ns (pristine dyes) to 1.32, 1.47, 1.58, 1.46, and 1.45 ns (dye/PCN/Pt), respectively. This high quenching efficiency (65–76%) underscores the role of non-conjugated bridges in facilitating efficient charge transfer.
Photocatalytic hydrogen production properties
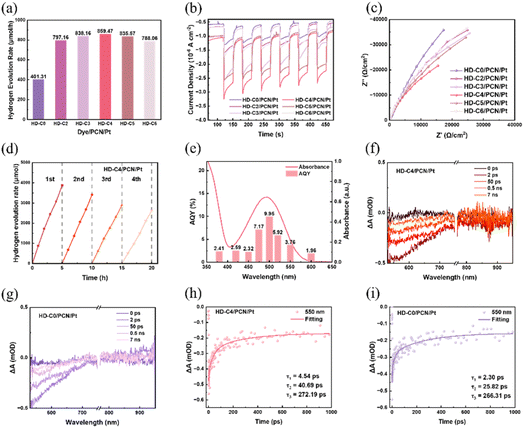
The hydrogen evolution activities of dye/PCN/Pt systems as photocatalysts were evaluated under visible-light irradiation (≥420 nm), with triethanolamine (TEOA) as a sacrificial agent. For bare PCN/Pt, it exhibits a low H2 generation of 23.34 µmol h−1. After dye loading, the HER performance of the photocatalyst is significantly improved, due to the enhanced visible-light absorption capacity and superior exciton dissociation efficiency (Fig. 5a). Among them, HD-C2–HD-C6/PCN/Pt systems exhibited significantly higher HER performance (>750 µmol h−1) than HD-C0/PCN/Pt one (401.31 µmol h−1), while HD-C4/PCN/Pt achieved the highest performance of 859.47 µmol h−1. This trend correlates directly with the dye aggregation behaviours at the PCN/Pt interface, as evidenced by photoluminescence quantum yields, lifetimes, and transient absorption measurements as mentioned above. HD-C0-featuring a conjugated bridge exhibits a stronger aggregation tendency than its non-conjugated counterparts. The pronounced aggregation of HD-C0 suggests significant non-radiative losses in the HD-C0/PCN/Pt composite, leading to low exciton dissociation efficiency and poor photocatalytic performance. In contrast, dyes with non-conjugated bridges show suppressed aggregation, which mitigates non-radiative recombination and thus enhances the overall catalytic efficiency. Among these, HD-C4/PCN/Pt achieved the highest fluorescence quenching efficiency of 49.0%, indicating the most efficient exciton transfer to PCN/Pt and accounting for its superior photocatalytic performance.To assess the efficiency of exciton dissociation, photocurrent measurements were conducted (Fig. 5b). HD-C4/PCN/Pt generated the highest photocurrent response among these photocatalysts, suggesting more efficient generation and separation of photoinduced charge carriers. It was further supported by electrochemical impedance spectroscopy (EIS), where HD-C4/PCN/Pt generated the lowest charge transfer resistance (Fig. 4c). This indicated superior charge transfer kinetics at the electrode–electrolyte interface of the HD-C4/PCN/Pt system. Subsequently, the apparent quantum yield (AQY) of HD-C4/PCN/Pt was tested under different monochromatic light irradiation (Fig. 5e). The corresponding trend was consistent with the absorption spectra of HD-C4, and the highest AQY was achieved up to 9.95% at 500 nm. The stability of the HD-C4/PCN/Pt catalyst was evaluated over four consecutive photocatalytic cycles (Fig. 4d). As a result, in the stability test of HD-C4/PCN/Pt, the maximum hydrogen evolution amount in one cycle was 3872.51 µmol and the minimum was 2621.40 µmol, representing about 67.7% of its initial activity. The decline in the hydrogen evolution rates may be due to the possible photodegradation of organic dyes under continuous light irradiation. Additionally, we tested the stability of HD-C0/PCN/Pt under identical conditions (Fig. S36); the hydrogen evolution decreased from 2119.32 µmol in the first cycle to 1457.78 µmol in the fourth cycle, corresponding to approximately 68.9% of the initial performance. The data demonstrate that the extent of activity loss is very similar between the two systems. Thus, the structural difference, whether the bridge is conjugated or non-conjugated, does not lead to a significant difference in their photocatalytic stability under the tested conditions.
To further investigate the influence of excited charge carriers in photocatalysts, femtosecond transient absorption spectroscopy (fs-TAS) was also performed with PCN/Pt, HD-C0/PCN/Pt, and HD-C4/PCN/Pt as the selected samples. To avoid potential alterations in the surface distribution of dyes caused by processing methods, such as ultrasonication, the samples were tested in their pristine powder form without any treatment, thereby ensuring that the results reflect the intrinsic properties of the catalysts. Fig. 5 displays the transient absorption (TA) maps (Fig. 5f and g) and representative decay profiles over delay times ranging from 2 ps to 7 ns (Fig. 5h and i) with 380 nm pump light as the excitation source. It is worth noting that the transient absorption spectra of PCN/Pt do not exhibit distinct features in the 500–700 nm range (Fig. S34). Thus, the signals observed in this region for HD-C0/PCN/Pt and HD-C4/PCN/Pt can be attributed to the presence of dye molecules. The kinetic traces monitored at 550 nm were fitted with a triple-exponential function. The decay kinetics analysis yielded three characteristic lifetimes (τ1, τ2, and τ3): the fast components of τ1 and τ2 are assigned to hot-electron cooling (to the conduction band minimum) and exciton annihilation, respectively, and the distinct, relatively long-lived τ3 component is related to charge population in shallow trap states.46 As shown in Fig. 5h, HD-C4/PCN/Pt demonstrated significantly increased τ1 (4.54 ps) and τ2 (40.69 ps) values, compared to those of HD-C0/PCN/Pt (Fig. 5i, τ1 = 2.30 ps and τ2 = 25.82 ps). A slower exciton decay implies more efficient exciton dissociation into free electrons, thereby allowing more active electrons to migrate to PCN/Pt and participate in photocatalytic reactions.23 Thus, HD-C4/PCN/Pt possessed an increased probability for the utilization of the free photoinduced electrons, which promotes greater involvement of electrons in photoreduction processes, ultimately contributing to the improved photocatalytic performance. HD-C4/PCN/Pt exhibited a similar τ3 value (272.19 ps) to HD-C0/PCN/Pt (266.31 ps), which reflects the uniform electron transfer to the PCN/Pt substrate, leading to consistent electron localization behaviour across both samples.
Conclusions
In this work, the bridge mode as the key factor in organic dyes has been well-modulated to improve the photocatalytic hydrogen evolution performance of dye/PCN/Pt systems. Compared to the conjugated bridge, the nonconjugated ones, which were acted by alkyl chains with adjustable lengths, exhibited a significant advantage. More than a two-fold increase in the HER has been achieved, which increased from 401.31 µmol h−1 (conjugation-linked dye) to 859.47 µmol h−1 (nonconjugation-linked dye). With the systematic investigation of molecular conformations, aggregation behaviours, and PHE performance, two key points can be concluded:(1) Owing to their elevated LUMO energy levels, non-conjugated dyes enable more efficient electron injection into PCN upon photoexcitation, thereby enhancing exciton dissociation and subsequent charge migration.
(2) The twisted molecular conformations of non-conjugated dyes effectively mitigate dye aggregation and the associated non-radiative recombination at the interface. This suppression leads to improved utilization of photoelectrons and prolonged charge carrier lifetimes.
By elucidating the critical role of nonconjugated bridges in promoting exciton dissociation and charge transport, this study offers an effective strategy for developing efficient organic dye-sensitized photocatalysts to boost the PHE activity.
Author contributions
Q. L. and Z. L. conceived the project. C. J. synthesized the organic compounds and performed all the measurements. Y. C. performed part of the photocatalytic test. W. Y. performed the DFT calculations. Q. L. and Z. L. discussed and revised the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information (SI). Supplementary information: data on NMR, mass spectrometry, photophysical properties, catalyst morphology, etc. See DOI: https://doi.org/10.1039/d5ta09516f.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22235006 and 22475157), the Foundation of Hubei Scientific Committee (2024AFA021 and 2024BAB014), and the Fundamental Research Funds for the Central Universities (2042025kf0009). The authors thank the Core Facility of Wuhan University for assistance with XPS, UPS, and SEM analysis. The authors would like to acknowledge the Center for Electron Microscopy and Core Research Facilities of the College of Chemistry and Molecular Sciences at Wuhan University for their substantial support of TEM work. The authors also thank Dr Guang OuYang from the Core Facility of Wuhan University for his assistance with TA. We are grateful to the National Supercomputing Center in Shenzhen and Beijing Parallel Technology Corporation for theoretical calculation support.Notes and references
- T. Banerjee, F. Podjaski, J. Kröger, B. P. Biswal and B. V. Lotsch, Nat. Rev. Mater., 2021, 6, 168–190 CrossRef CAS.
- X. Huang, W. Xie, T. Xu, W. Weng, T. Zhou and J. Guo, Angew. Chem., Int. Ed., 2025, 64, e202509095 CrossRef CAS.
- Q. Xu, L. Zhang, B. Cheng, J. Fan and J. Yu, Chem, 2020, 6, 1543–1559 CAS.
- C. Xiaobo, S. Shaohua, G. Liejin and S. M. Samuel, Chem. Rev., 2010, 110, 6503–6570 CrossRef.
- W. J. Youngblood, S.-H. A. Lee, K. Maeda and T. E. Mallouk, Acc. Chem. Res., 2009, 42, 1966–1973 CrossRef CAS PubMed.
- L. Hao, R. Shen, C. Qin, N. Li, H. Hu, G. Liang and X. Li, Sci. China Mater., 2024, 67, 504–513 CrossRef CAS.
- Y. Huang, B. Gao, Q. Huang, D. L. Ma, H. Wu and C. Qian, Aggregate, 2025, 6, e669 CrossRef CAS.
- X. A. Li, Z. Z. Liang, Y. C. Zhou, J. F. Huang, X. L. Wang, L. M. Xiao and J. M. Liu, Aggregate, 2024, 5, e442 CrossRef CAS.
- X. Jiang, H. Xu, W. Zou, X. Zhang, F. Jin, L. Sun, W. Ding, Y. Kan, Y. Sun and K. Gao, Sci. China Mater., 2025, 68, 4097–4106 CrossRef.
- M. Wang, Z. Wang, M. Shan, J. Wang, Z. Qiu, J. Song and Z. Li, Chem. Mater., 2023, 35, 5368–5377 CrossRef CAS.
- Q. Zhang, Y. Zhang, H. Shi, H. Zhang, J. Zhao, Z. Zheng, H. Yang and P. Yang, Aggregate, 2023, 5, e424 CrossRef.
- T. Zhang, P. Wang, S. Yue, F. Li, Z. Zhao and S. Zhan, Sci. China Chem., 2024, 67, 1161–1174 CrossRef CAS.
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 CrossRef CAS.
- S. Wu, Z. Zhang, Y. Lee, Y. Li, X. Tai, W. Si, S. Bai and Y. Lin, Angew. Chem., Int. Ed., 2025, 64, e202422779 CrossRef CAS PubMed.
- X. Wang, L. Chen, S. Y. Chong, M. A. Little, Y. Wu, W. H. Zhu, R. Clowes, Y. Yan, M. A. Zwijnenburg, R. S. Sprick and A. I. Cooper, Nat. Chem., 2018, 10, 1180–1189 CrossRef CAS.
- J. Kosco, F. Moruzzi, B. Willner and I. McCulloch, Adv. Energy Mater., 2020, 10, 2001935 Search PubMed.
- L. Stegbauer, K. Schwinghammer and B. V. Lotsch, Chem. Sci., 2014, 5, 2789–2793 RSC.
- Z. R. Tan, Y. Q. Xing, J. Z. Cheng, G. Zhang, Z. Q. Shen, Y. J. Zhang, G. Liao, L. Chen and S. Y. Liu, Chem. Sci., 2022, 13, 1725–1733 Search PubMed.
- K. Tang, J.-Y. Shao, J. Li, S. Li, J.-H. Tang, R. Duan, J. Yao and Y.-W. Zhong, Chem, 2024, 10, 1925–1939 Search PubMed.
- W. Zhang, M. Yu, T. Liu, M. Cong, X. Liu, H. Yang, Y. Bai, Q. Zhu, S. Zhang, H. Gu, X. Wu, Z. Zhang, Y. Wu, H. Tian, X. Li, W.-H. Zhu and A. I. Cooper, Nat. Synth., 2024, 3, 595–605 CrossRef CAS.
- Y. Guo, J. Sun, Z. Xu, L. He, W. Zhao, T. Guo, G. Wu, T. He, X. Cao, X. Bi, S. Ding, G. Long, H. Chen, Y. Liu and Z. Yao, Sci. China Chem., 2025, 68, 6682–6692 Search PubMed.
- Z.-W. Wang and G.-Y. Yang, Sci. China Chem., 2026, 69, 737–741 CrossRef.
- B. Fang, Z. Xing, F. Du, W. Kong, Z. Li and W. Zhou, J. Mater. Chem. A, 2022, 10, 16439–16447 RSC.
- Y. Feng, X. Fang, J. Zang, B. Song, C. Hu, X. Dong and Y. Ding, Sci. China Chem., 2025, 68, 772–780 CrossRef CAS.
- J. Wu, W. Xia, M. Lan, X.-J. Xing, J.-C. Hu, L. Huang, J. Liu, Y.-Y. Ren, H. Liu and F. Wang, J. Mater. Chem. A, 2020, 8, 21690–21699 Search PubMed.
- X. Xiong, L. Gui, L. Yang, J. Li, S. Xu, F. Yu, J. Qian, T. Mei and X. Wang, J. Mater. Chem. A, 2025, 13, 16839–16849 RSC.
- C. Yang, Z. Cheng, G. Divitini, C. Qian, B. Hou and Y. Liao, J. Mater. Chem. A, 2021, 9, 19894–19900 Search PubMed.
- G. Zhang, Z. A. Lan, L. Lin, S. Lin and X. Wang, Chem. Sci., 2016, 7, 3062–3066 RSC.
- X. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76–80 CrossRef CAS PubMed.
- W. Dai, R. Wang, Z. Chen, S. Deng, C. Huang, W. Luo and H. Chen, J. Mater. Chem. A, 2023, 11, 7584–7595 Search PubMed.
- R. Jia, Q. Gui, L. Sui, Y. Huang, H. Lu, H. Dong, S. Ma, Z. Gan, L. Dong and L. Yu, J. Mater. Chem. A, 2021, 9, 14768–14774 Search PubMed.
- S. Wang, D. Liu, C. Zhang, J. Huang, S. Zhao, K. Lu, B. Wang, H. Peng, Y. Si and M. Liu, Sci. China Mater., 2024, 67, 2957–2964 CrossRef CAS.
- X. Wu, H. Fan, W. Wang, L. Lei, X. Chang and L. Ma, J. Mater. Chem. A, 2022, 10, 17817–17826 Search PubMed.
- Q. Yao, P. Liu, F. Yang, Y. Zhu, Y. Pan, H. Xue, W. Mao and L. Chu, Sci. China Mater., 2024, 67, 3160–3167 Search PubMed.
- D. Zhao, Y. Yang, V. Binas and S. Shen, Sci. China Mater., 2024, 67, 1765–1779 CrossRef CAS.
- Y. Zou, S. Li, D. Zheng, J. Feng, S. Wang, Y. Hou and G. Zhang, Sci. China Chem., 2024, 67, 2215–2223 Search PubMed.
- Y. Chen, C. Jiang, S. Liu, W. Yuan, Q. Li and Z. Li, Angew. Chem., Int. Ed., 2025, 64, e202419850 CrossRef CAS PubMed.
- S. Liu, Q. Chen, Y. Chen, P. Lin, H. Zhuzhang, M. Han, Z.-A. Lan, X. Chen, X. Wang, Q. Li and Z. Li, J. Mater. Chem. A, 2023, 11, 14682–14689 RSC.
- L. Xu, B. Tian, T. Wang, Y. Yu, Y. Wu, J. Cui, Z. Cao, J. Wu, W. Zhang, Q. Zhang, J. Liu, Z. Li and Y. Tian, Energy Environ. Sci., 2022, 15, 5059–5068 RSC.
- S. Liu, P. Lin, M. Wu, Z.-A. Lan, H. Zhuzhang, M. Han, Y. Fan, X. Chen, X. Wang, Q. Li and Z. Li, Appl. Catal. B Environ., 2022, 309, 121257 CrossRef CAS.
- J. Mao, Q. Fan, Z. Yan, X. Chen, S. Zhao, Y. Lu, S. Li, W. Jiang, Z. Xu, Z. Wang and J. Wang, J. Am. Chem. Soc., 2025, 147, 12730–12739 CrossRef CAS PubMed.
- H. Yang, C. Li, T. Liu, T. Fellowes, S. Y. Chong, L. Catalano, M. Bahri, W. Zhang, Y. Xu, L. Liu, W. Zhao, A. M. Gardner, R. Clowes, N. D. Browning, X. Li, A. J. Cowan and A. I. Cooper, Nat. Nanotechnol., 2023, 18, 307–315 CrossRef CAS PubMed.
- Y. Gong, W. Zhang, Z. Liu, M. Fang, J. Yang, Y. Wang, M. Gao, J. Zhang, Q. H. Yang and Z. Li, Adv. Mater., 2024, 36, 2312486 CrossRef CAS PubMed.
- Z. Liu, Y. Tian, J. Yang, A. Li, Y. Wang, J. Ren, M. Fang, B. Z. Tang and Z. Li, Light Sci. Appl., 2022, 11, 142 CrossRef CAS.
- G. B. Bodedla, M. Imran, J. Zhao, X. Zhu and W. Y. Wong, Aggregate, 2023, 4, e364 Search PubMed.
- R. Shen, C. Huang, L. Hao, G. Liang, P. Zhang, Q. Yue and X. Li, Nat. Commun., 2025, 16, 2457 CrossRef CAS .
| This journal is © The Royal Society of Chemistry 2026 |