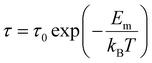Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correlating Li+ jump pathways and vibrational properties in lithium aluminum germanium phosphate glasses
Chang Gyu Baeka,
Young Hoon Rim*b,
Jae-Hyeon Koc and
Yong Suk Yang *a
*a
aDepartment of Nanoenergy Engineering, College of Nanoscience and Nanotechnology, Pusan National University, Busan 46241, Republic of Korea. E-mail: ysyang@pusan.ac.kr
bCollege of Liberal Arts, Semyung University, Jecheon, Chungbuk 27136, Republic of Korea. E-mail: yrim@semyung.ac.kr
cSchool of Semiconductor Display Technology, Nano Convergence Technology Center, Hallym University, Chuncheon-si, Gangwon-do 24252, Republic of Korea
First published on 10th February 2026
Abstract
Lithium aluminum germanium phosphate (LAGP) glass ceramics have attracted considerable attention as promising solid electrolytes for next-generation lithium batteries owing to their improved safety and thermal stability. Despite significant progress, fundamental questions regarding Li+ hopping processes, effective charge carrier concentration, and carrier mobility in glassy systems remain unresolved. In this work, we investigate the fundamental relationship between fast lithium-ion transport and the network structure in xLi2O–0.25Al2O3–1.5GeO2–1.5P2O5 (xLi2O-AGPO; 0.75 ≤ x ≤ 2.5) glasses. The dc conductivity increases by nearly three orders of magnitude, from 1.73 × 10−9 S cm−1 (x = 0.75) to 1.67 × 10−6 S cm−1 (x = 2.5) at 50 °C, while the dc activation energy decreases with increasing Li2O content. The effective charge carrier concentration is evaluated from the conductivity data, and the corresponding carrier mobility is estimated. The charge carrier concentration increases nearly proportionally with Li2O content, whereas the carrier mobility exhibits an exponential increase, similar to the trend observed for the dc conductivity. These results indicate that the exponential enhancement of dc conductivity with increasing x is primarily governed by the increase in carrier mobility. The facile migration of Li+ is attributed to fractal conduction pathways formed by non-bridging oxygen sites, which are generated through the depolymerization of the phosphate network.
1. Introduction
Li-ion batteries are increasingly in demand for applications ranging from portable electronic devices to electric vehicles and large-scale energy storage systems owing to their high energy density and long cycle life.1,2 Recently, solid electrolytes for all-solid-state batteries have emerged as key components for advanced electrochemical energy storage technologies because of their enhanced safety, excellent chemical and thermal stability, long cycle life, and high energy density. Among the various classes of oxide ion conductors, lithium-ion conductors are considered promising electrolyte candidates for these applications.3–5 In particular, NASICON-type glass-ceramic electrolytes with the composition Li1.5Al0.5Ge1.5(PO4)3 (hereafter referred to as LAGP) have attracted considerable attention, as they exhibit a high ionic conductivity of ∼10−4 S cm−1 at room temperature, along with superior electrochemical stability and a wide electrochemical window.6,7 The NASICON crystal structure is composed of PO4 tetrahedra that share corner oxygen atoms with MO6 octahedra (where M = Ge in LiGe2(PO4)3). Partial substitution of Al3+ for Ge4+ in LiGe2(PO4)3 results in the incorporation of additional Li+ ions at the 36f sites to maintain charge neutrality. Consequently, the Li+ concentration in the LAGP structure increases, leading to a significant enhancement of ionic conductivity from ∼10−8 to ∼10−4 S cm−1.8 Furthermore, it has been reported that ionic conductivity can be further improved by substituting cations with larger ionic radii than Ge4+ and Al3+, which effectively enlarges the diffusion channels for Li+ migration.9Li et al. reported in an early publication that the ionic conductivity of Li1+xGe2−xAlxP3O12 (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) C structure) reaches a maximum value of 3.5 × 10−5 S cm−1 at room temperature for the composition x = 0.5 among the investigated compositions in the range 0.1 ≤ x ≤ 0.6.10 Although LAGP is regarded as one of the most promising solid electrolytes for lithium-ion batteries, its practical application is still limited by relatively low bulk and grain-boundary ionic conductivities. To further enhance the ionic conductivity, Xu et al. conducted a systematic study on NASICON-type LAGP-xLi2O (x = 0.0–0.20) lithium-ion-conducting glass-ceramics. They demonstrated that the glass-ceramic sample with x = 0.05 exhibits the highest ionic conductivity, reaching 7.25 × 10−4 S cm−1 at room temperature.7
C structure) reaches a maximum value of 3.5 × 10−5 S cm−1 at room temperature for the composition x = 0.5 among the investigated compositions in the range 0.1 ≤ x ≤ 0.6.10 Although LAGP is regarded as one of the most promising solid electrolytes for lithium-ion batteries, its practical application is still limited by relatively low bulk and grain-boundary ionic conductivities. To further enhance the ionic conductivity, Xu et al. conducted a systematic study on NASICON-type LAGP-xLi2O (x = 0.0–0.20) lithium-ion-conducting glass-ceramics. They demonstrated that the glass-ceramic sample with x = 0.05 exhibits the highest ionic conductivity, reaching 7.25 × 10−4 S cm−1 at room temperature.7
Pershina et al. investigated the effects of Al2O3 addition on the microstructure, molecular structure, and ionic conductivity of LiGe2(PO4)3 glass-ceramics by synthesizing Li1+xAlxGe2−x(PO4)3 (0 ≤ x ≤ 0.65) compositions.11 They reported that the Li1.5Al0.5Ge1.5(PO4)3 composition (x = 0.5) exhibits the highest lithium-ion conductivity, reaching 5.03 × 10−4 S cm−1 at 25 °C. In recent years, most studies have focused on the synthesis of LAGP glass-ceramics via glass crystallization. However, reported data on the phase composition of these conductors and their corresponding ionic conductivities remain inconsistent. This inconsistency arises because crystallization parameters strongly influence lithium-ion mobility, the fraction of residual amorphous phases, and the formation of microstructural defects such as cracks and pores, all of which have a significant impact on the ionic conductivity of the materials.
However, the development of crystalline solid electrolytes for lithium-ion batteries inevitably faces several challenges, including high grain-boundary resistance and difficulties in achieving sufficient densification. In this regard, glass-based electrolytes offer distinct advantages over sintered polycrystalline materials. For example, glass systems provide broad compositional flexibility, enable processing at relatively low fabrication temperatures through the appropriate selection of glass formers, and allow for the formation of nanocrystalline constituents with isotropic physical properties within the disordered network structure.12–14
In ion-conducting glasses, the lack of direct experimental data on charge carrier concentration and mobility has led to differing interpretations of the conduction mechanism, with the glassy systems being alternatively treated as strong or weak electrolytes.15,16 Therefore, the independent determination of charge carrier concentration and mobility is of crucial importance for a comprehensive understanding of ionic conductivity mechanisms in glasses. Recently, Souquet et al. proposed a model in which ionic displacement arises from the migration of cationic pairs formed through partial dissociation, a process that is thermally activated.17 Using the Almond–West formalism, Hairetdinov et al. estimated the concentration of free charge carriers in fast-ion-conducting glasses by assuming strong electrolyte behavior.18 In addition, Marple et al. demonstrated that an atomistic interpretation of the crossover hopping frequency, ωh, in Jonscher's universal dielectric response provides a consistent description of ionic transport in glasses.19 In this framework, mobile ions are temporarily trapped in local cages and subsequently hop across percolation clusters. Nevertheless, the Almond–West approach has been criticized for being phenomenological rather than constituting a rigorous microscopic theory.
Bunde et al. proposed a dynamic structure model, in which the existence of percolative diffusion pathways leads to an exponential increase in ionic conductivity with increasing ion concentration.20 Sidebottom et al. argued that the anomalous-diffusion model of ionic transport in LiPO3 glass supports both the jump relaxation model and the Coulomb-interacting lattice model.21 However, it was later shown that the estimated diffusion length scale in lithium phosphate glass is too short to be comparable with the characteristic hopping distances in glassy systems.
By applying bond-valence techniques in conjunction with reverse Monte Carlo (RMC) simulations to mixed-alkali phosphate glasses, Swenson and Adams suggested that the two types of alkali ions are randomly mixed and migrate through distinct low-dimensional conduction pathways.22 Based on the RMC-produced structure model, Rim et al. derived Jonscher's universal conductivity expression by introducing fractional Klein-Kramers kinetic equations containing non-integer differential and integral operators, which lead to a modified fractional Rayleigh equation describing the time- and velocity-dependent dynamics of ion transport.23
In a previous study, Rim et al. reported that the addition of lithium oxide in xLi2O–2Fe2O3–3P2O5 glasses enhances the depolymerization of the iron phosphate network, accompanied by the formation of non-bridging oxygen (NBO) species through the conversion of Q2 units to Q1 and Q0 units.24 Here, the notation Qn represents the structural units of the glass network, where n denotes the number of bridging oxygens per tetrahedral unit. Alkali-ion transport in glassy systems occurs via a thermally activated hopping process under an applied electric field, where ions migrate between NBO sites through potential minima in the disordered network.25 Within the energy landscape of the glass network, the depth of the potential wells is determined by Coulomb interactions between the alkali ions and the surrounding network structure.26 At low alkali-ion concentrations, ion conduction is primarily governed by long-range Coulomb interactions between mobile ions, whereas at higher ion concentrations, structural modifications of the glass network play a dominant role in determining the conduction behavior.26 However, Rim et al. also reported that the relatively low ionic conductivity of the lithium silicate glasses arises from the limited participation of Li+ ions in transport through NBO sites.27 Consequently, elucidating the mechanism of fast ion transport in mixed aluminum–germanium–phosphate glasses remains a significant challenge, as the different charge valences of lithium, aluminum, and germanium ions interact distinctly with the surrounding phosphate network, leading to complex structural modification that critically influences ionic transport.
Polycrystalline, nanocrystalline, and glass-ceramic forms of Li1+xAlxGe2−xP3O12 (x = 0.5), which exhibit typical ionic conductivities on the order of ∼10−4 S cm−1, can be produced via glass-based processing routes. Such glass-derived approaches offer distinct advantages, as key microstructural parameters, including crystal size, degree of crystallinity, and the volume fraction of the crystalline and residual glassy phases, can be precisely tailored. Establishing the fundamental properties of the glassy state is of significant scientific importance because structurally disordered glasses exhibit electrical, thermal, mechanical, chemical, and optical properties that differ markedly from those of structurally ordered crystalline materials. To date, numerous studies have investigated Li1+xAlxGe2−xP3O12 in crystalline, glass-ceramic, and glassy forms; however, most of these investigations have been restricted to a substitution range of x ≈ 0.5.
In the present work, we aim to synthesize LAGP-based glasses over a broader range of Li2O contents, starting from x = 0.5 and systematically increasing x up to the compositional limit at which a glassy phase can be maintained. The fundamental physical properties of the resulting glasses are investigated with particular emphasis on elucidating the relationships among ionic conductivity, effective charge carrier concentration, and mobility as functions of Li2O content. In this study, lithium alumina germanium phosphate glasses with compositions xLi2O–0.25Al2O3–1.5GeO2–1.5P2O5 (xLi2O-AGPO), where 0.75 ≤ x ≤ 2.5, were prepared, and their electrical impedance and Raman spectroscopic properties were systematically examined. An anomalous enhancement of dc conductivity is observed in the xLi2O–0.25Al2O3–1.5GeO2–1.5P2O5 glass system. As x increases from 0.75 to 2.5, the dc conductivity increases by nearly three orders of magnitude, from 1.73 × 10−9 to 1.67 × 10−6 S cm−1. Such an exponential increase in dc conductivity with increasing Li2O content has rarely been reported in ionic glasses. The combined electrical and Raman spectroscopic results presented in this study provide valuable insights into the design of glassy and glass-ceramic electrolytes with enhanced ionic conductivity.
The processes of ionic conduction and relaxation in xLi2O-AGPO glasses are analyzed using Cole–Cole power-law representations. The effective concentration of mobile ionic charge carriers is evaluated, and the corresponding mobility is estimated from the dc conductivity. Furthermore, the origin of Li+ transport is revealed by correlating Li+ jump pathways with structural modifications of the glassy network. The present work provides a comprehensive understanding of ion dynamics in these glasses, offering valuable insights for both fundamental research and the development of high-performance electrochemical energy storage materials.
2. Experimental
AR-grade chemicals Li2CO3, Al2O3, GeO2, and NH4H2PO4 were used as starting materials to prepare lithium aluminum germanium phosphate glasses. The raw materials were thoroughly mixed for 1 h in an alumina bowl according to the molar ratio xLi2CO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25Al2O3
0.25Al2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5GeO2
1.5GeO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.0NH4H2PO4 (xLi2O-AGPO; x = 0.75, 1.0, 1.25, 1.5, 1.75, 2.0, 2.5). Each batch was transferred into a platinum crucible and pre-annealed at 700 °C for 1 h in an electric furnace to remove volatile species such as CO2 and NH3. After cooling to room temperature, the annealed samples were reground for an additional 1 h to ensure compositional homogeneity.
3.0NH4H2PO4 (xLi2O-AGPO; x = 0.75, 1.0, 1.25, 1.5, 1.75, 2.0, 2.5). Each batch was transferred into a platinum crucible and pre-annealed at 700 °C for 1 h in an electric furnace to remove volatile species such as CO2 and NH3. After cooling to room temperature, the annealed samples were reground for an additional 1 h to ensure compositional homogeneity.
The reground powders were subsequently melted in a platinum crucible at temperatures between 1200 and 1300 °C. The resulting melts were quenched by pressing between metal plates to obtain bulk glass samples. The quenched bulk glasses were transparent, with a typical thickness of approximately 0.06 cm.
To prepare thin glass samples suitable for impedance measurements, the bulk glasses were crushed, remelted at 1200–1300 °C, and rapidly quenched using a twin-roller technique. The thickness of the resulting glass sheets ranged from 5.8 × 10−3 to 7.7 × 10−3 cm, as measured using a digital micrometer with a precision of ± 5.0 × 10−5 cm.
The amorphous nature of the samples was confirmed by X-ray diffraction (XRD) using a Miniflex II diffractometer (Rigaku, Japan) with a CuKα radiation (λ = 1.5406 Å). XRD patterns were recorded over the 2θ range of 10–80° with a step width of 0.05° and a counting time of 3 s per step.
Complex impedance measurements were carried out using an impedance analyzer (4294A, Keysight, U.S.A.). Circular gold electrodes with a radius of 0.15 cm were deposited on both sides of each sample, and gold leads were attached using silver paste. To remove organic components from the silver paste, the samples were heat-treated at 300 °C for 10 min. Impedance spectra were collected over a frequency range of 100 Hz to 30 MHz. Electrical measurements were performed in a temperature-controlled furnace from room temperature to 240 °C with a heating rate of 2 °C min−1.
Raman spectra were obtained using a micro-Raman spectrometer (LabRAM HR Evolution, Horiba). Excitation was provided by a diode-pumped solid-state laser with a wavelength of 532 nm and an output power of approximately 9 mW. Raman measurements were conducted on small fragments of heat-treated samples. Spectra were recorded over the Raman shift range of 20–1400 cm−1 with an integration time of 10 s for each spectrum.
3. Results and discussion
3.1. Impedance analysis
Fig. 1 shows the typical XRD patterns of xLi2O-AGPO (x = 0.75, 1.0, 1.25, 1.5, 1.75, 2.0, 2.5) glasses at 50 °C. The XRD patterns exhibit a wide halo without crystalline sharp Bragg peaks in the range of 10–80°, which is characteristic of the amorphous state. The scanning process was repeated 8 times, and then the XRD data were taken as an average.The complex impedance spectra have been calculated using experimentally measured values of the dielectric constant and dielectric loss tangent as
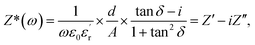 | (1) |
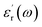 is the real part of the relative dielectric constant, ε0 is the vacuum permittivity, ω is the angular frequency, and
is the real part of the relative dielectric constant, ε0 is the vacuum permittivity, ω is the angular frequency, and 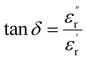 is the loss tangent factor. The loss tangent factor represents the ratio of the energy dissipated per radian in the dielectric to the energy stored. The electrode area is fixed at A = π(0.15)2 cm2 for all samples, while the sample thickness d ranges from 5.8 × 10−3 cm (x = 1.25) to 7.7 × 10−3 cm (x = 0.75).
is the loss tangent factor. The loss tangent factor represents the ratio of the energy dissipated per radian in the dielectric to the energy stored. The electrode area is fixed at A = π(0.15)2 cm2 for all samples, while the sample thickness d ranges from 5.8 × 10−3 cm (x = 1.25) to 7.7 × 10−3 cm (x = 0.75).
The complex Cole–Cole formalism is one method for understanding the electrical response to the applied field. In particular, it is useful to understand how ions move through a broad distribution of random energy barriers. The Cole–Cole impedance is expressed as follows:28
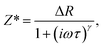 | (2) |
Fig. 2(a) represents the compositional dependence of complex impedance Cole–Cole spectra measured at 50 °C for several xLi2O-AGPO glasses. In the inset in Fig. 2(a), the equivalent R-CPE parallel circuit is shown. Based on the impedance expression
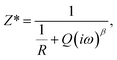 | (3) |
A depressed semicircle arc of the complex impedance spectra intersects the Z′ axis on the low-frequency side when the impedance is extrapolated with the Cole–Cole formula, as seen for x = 1.25 in Fig. 2(a). This real part of complex impedance is defined by Z0′ and it gives the dc conductivity from the Cole–Cole representation in eqn (2), that is, 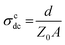 . The obtained value of the impedance at the intersection is Z0′ (x = 0.75) = 6.28 × 107 Ω at 50 °C, and it decreases to Z0′ (x = 2.5) = 6.32 × 104 Ω at the same temperature, indicating that the dc conductivity increases rapidly with increasing concentration of lithium content. Namely, the dc conductivity σcdc (x = 0.75) = 1.60 × 10−9 S cm−1 increases to σcdc (x = 2.5) = 1.58 × 10−6 S cm−1, showing that the conductivity increases exponentially with increasing x content.
. The obtained value of the impedance at the intersection is Z0′ (x = 0.75) = 6.28 × 107 Ω at 50 °C, and it decreases to Z0′ (x = 2.5) = 6.32 × 104 Ω at the same temperature, indicating that the dc conductivity increases rapidly with increasing concentration of lithium content. Namely, the dc conductivity σcdc (x = 0.75) = 1.60 × 10−9 S cm−1 increases to σcdc (x = 2.5) = 1.58 × 10−6 S cm−1, showing that the conductivity increases exponentially with increasing x content.
The calculated exponent γ in eqn (2) varies slightly from 0.81 (x = 0.75) to 0.78 (x = 2.5) at various temperatures. The result indicates that the relaxation time distribution increases with increasing x content in xLi2O-AGPO glasses.
Fig. 2(b) shows that the dc conductivity σcdc obtained from the Cole–Cole formula obeys the Arrhenius relationship such as 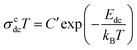 , where C′ is a constant and kB is the Boltzmann constant. The slope of the least-squares straight-line fits represents the value of the activation energies. The Arrhenius relationship indicates that the charge carriers contributing to dc conductivity are thermally activated. The obtained dc activation energy decreases from Ecdc = 0.68 eV (x = 0.75) to Ecdc = 0.46 eV (x = 2.5), indicating that the average heights of the energy barrier for ion movements decrease with increasing Li2O content.
, where C′ is a constant and kB is the Boltzmann constant. The slope of the least-squares straight-line fits represents the value of the activation energies. The Arrhenius relationship indicates that the charge carriers contributing to dc conductivity are thermally activated. The obtained dc activation energy decreases from Ecdc = 0.68 eV (x = 0.75) to Ecdc = 0.46 eV (x = 2.5), indicating that the average heights of the energy barrier for ion movements decrease with increasing Li2O content.
In Fig. 2(b), we also present the relaxation time τ, obtained from the impedance Cole–Cole plot as a function of 1000/T. The relaxation time is obtained with ωm, which denotes the frequency at which the imaginary part of the impedance, Z″ (dissipation), reaches its maximum in the impedance plane plot, satisfying the relationship ωmτ = 1.28 As seen in the figure, the relaxation time exponentially decreases from 1.5 × 10−3 s (x = 0.75) to 1.2 × 10−6 s (x = 2.5) at 50 °C with Li2O content. This indicates that the time requirement to move between average hopping distances becomes shorter with the increase of x content, i.e., ions move faster with larger x. The slope of ln τ versus 1/T decreases linearly with T. The relaxation time τ can be expressed as τ = τ0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(Em/kBT), where Em is the energy at the frequency, where the dissipation is maximum. The calculated Em energy of the xLi2O-AGPO glasses decreases from Em = 0.67 eV (x = 0.75) to Em = 0.37 eV (x = 2.5). The energy Em is often referred to as migration or motional energy inside the network, where low migration energies lead to high ionic mobility. Therefore, the exponentially increasing dc conductivity and the lowering of the energy barriers are the origin of the exponentially increasing mobility in the xLi2O-AGPO glasses.
exp(Em/kBT), where Em is the energy at the frequency, where the dissipation is maximum. The calculated Em energy of the xLi2O-AGPO glasses decreases from Em = 0.67 eV (x = 0.75) to Em = 0.37 eV (x = 2.5). The energy Em is often referred to as migration or motional energy inside the network, where low migration energies lead to high ionic mobility. Therefore, the exponentially increasing dc conductivity and the lowering of the energy barriers are the origin of the exponentially increasing mobility in the xLi2O-AGPO glasses.
3.2. Conductivity: charge carrier density and mobility
The frequency-dependent conductivity σ(ω) is calculated from the measured dielectric constant and dielectric loss tangent according to
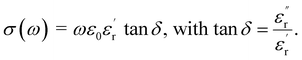 | (4) |
The conductivity spectra are analyzed using Jonscher's universal power law, separating the frequency-independent dc conductivity σdc and frequency-dependent ac conductivity σac contributions. The fitting procedure has been carried out using25
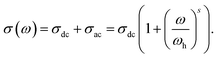 | (5) |
The nearly constant loss (NCL) term proportional to ω is not observed in the present xLi2O-AGPO glasses.30 It is noted that ωh is the crossover onset frequency from dc conductivity to ac conductivity, defined by σ(ωh) = 2σdc, and the exponent s is a dimensionless constant 0 ≤ s ≤ 1 for ionic glass.
The compositional dependence of the frequency-dependent conductivity, σ(ω), measured at 50 °C for various xLi2O-AGPO glass samples is presented in Fig. 3(a). The inset shows the conductivity spectra for the x = 0.75 sample measured at different temperatures. We have observed that the dc conductivity σdc = 1.73 × 10−9 S cm−1 for x = 0.75 increases to 1.67 × 10−6 S cm−1 for x = 2.5, exhibiting that σdc(x = 2.5)/σdc(x = 0.75) = 9.65 × 102.
 | ||
Fig. 3 Conductivity analysis; (a) frequency spectra of the real conductivity σ(ω) for xLi2O-AGPO (x = 0.75, 1.0, 1.25, 1.5, 1.75, 2.0, 2.5) glasses at 50 °C. The inset of the figure shows the conductivity spectra for x = 0.75 at various temperatures. (b) Reciprocal temperature dependence of the dc conductivity and the hopping frequency ωh obtained from the universal power law analysis in eqn (3). The solid lines indicate that ωh obeys the Arrhenius relationship  . . | ||
Meanwhile, the result in the inset shows that the dc conductivity σdc = 1.73 × 10−9 S cm−1 at 50 °C increases to 2.22 × 10−6 S cm−1 at 200 °C. The results indicate that the conductivity values become almost 103 times higher with increasing content of Li2O as well as increasing temperature. Consequently, the results from the power-law analysis are consistent with those obtained from the Cole–Cole representation.
The average value of exponent s in eqn (5) is 0.70 ± 0.02 for xLi2O-AGPO glasses, which are the obtained values from the fit at various temperatures and at several contents of Li2O. That is, the values of s were found to be independent of temperature as well as composition in xLi2O-AGPO glasses. As shown in Fig. 3(b), the reciprocal temperature dependence of ωh follows the Arrhenius relationship:
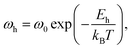 | (6) |
For the calculation of the charge carrier concentration nc and the mobility µ, we present the composition dependent dc conductivity σdc and hopping frequency ωh for the xLi2O-AGPO glasses in Table 1. The similarity of the activation energies calculated from the Cole–Cole representation and the universal power-law formalism implies that the activation energy is an appropriate parameter for describing the electrical relaxation and conduction mechanisms. It is emphasized that the ion transport mechanism is based on the hopping process of ions through the sub-transport in the multiple trapping percolation clusters with the low-dimensional conduction pathway.23
| x | σdc (50 °C) (S cm−1) | ωh (50 °C) (Hz) | nc (50 °C) (cm−3) | µ (50 °C) (cm2 s−1 V−1) |
|---|---|---|---|---|
| 0.75 | 1.73 × 10−9 | 5.12 × 103 | 3.91 × 1020 | 2.77 × 10−11 |
| 1.0 | 1.15 × 10−8 | 3.29 × 104 | 4.05 × 1020 | 1.77 × 10−10 |
| 1.25 | 2.47 × 10−8 | 6.78 × 104 | 4.22 × 1020 | 5.52 × 10−10 |
| 1.5 | 9.18 × 10−8 | 2.26 × 105 | 4.70 × 1020 | 1.22 × 10−9 |
| 1.75 | 3.34 × 10−7 | 7.70 × 105 | 5.02 × 1020 | 4.16 × 10−9 |
| 2.0 | 7.31 × 10−7 | 1.53 × 106 | 5.54 × 1020 | 8.25 × 10−9 |
| 2.5 | 1.67 × 10−6 | 3.33 × 106 | 5.81 × 1020 | 1.80 × 10−8 |
How many charge carriers contribute to the ionic conductivity and how much their mobility contributes are fundamental questions in glass systems. The frequency dependence of conductivity suggested by Jonscher in eqn (5) is sometimes referred to as the Almond–West (AW) formula. Almond and West identified the crossover frequency ωh with a hopping rate of the mobile ions in a solid. Using the values of ωh and σdc, which are obtained from the experiment and analysis, with the Nernst–Einstein relationship, the concentration of mobile ions nc (ref. 25) can be obtained as follows:
 | (7) |
In this expression, q is the charge of the mobile ion, λ denotes the elementary hopping distance, kB is the Boltzmann constant, and T is the temperature. The elementary hopping distance in the solid is 2–3 Å, and we have used a fixed value of 3 Å for all samples in calculating the carrier concentration.31
The carrier mobility µ can be calculated from the dc conductivity for solid materials, which is given by
| σdc = ncqµ. | (8) |
The use of the effective concentration of charge carriers nc in eqn (7) yields µ in eqn (8) and the results are summarized in Table 1.
The calculated charge carrier concentration exhibits a trend consistent with the variation in Li2O content, increasing from 3.91 × 1020 to 5.81 × 1020 cm−3 as 0.75 ≤ x ≤ 2.5. Meanwhile, when the Li2O content increases from x = 0.75 to 2.75, both the dc conductivity and the charge carrier mobility increase by approximately three orders of magnitude, from 1.73 × 10−9 to 1.67 × 10−6 S cm−1 and from 2.77 × 10−11 to 1.67 × 10−8 cm2 V−1 s−1, respectively.
The relationship between σdc, nc, and µ is presented in Fig. 4, where the calculated σdc from the measured data is plotted versus the x content. The inset in Fig. 4 shows that the calculated mobility µ is plotted versus the x content. Meanwhile, the calculated charge carrier concentration nc increases proportionally with the mole ratio of x in the xLi2O-AGPO glasses. The result indicates that the exponential increase in dc conductivity is caused by the mobility rather than by the charge carrier concentration.
The results show that the conducting channels are widely open for long-range cooperative hopping due to the increase in NBO sites because of the depolymerization of the network. As a consequence, the ionic dc conductivity increases exponentially, as does the mobility in xLi2O-AGPO glasses.
3.3. Relationship between Raman spectra and conductivity
Raman spectroscopy is a powerful tool for examining the phase composition of solid electrolytes and for probing local and cooperative structural changes. Fig. 5(a) presents the Raman spectra of xLi2O-AGPO glasses with x = 0.75, 1.5, and 2.5 measured at room temperature. The reduced spectra were deconvoluted using Gaussian functions to assign the vibrational modes. The baseline correction was performed using an exponential decay function to remove background contributions. For comparison, the spectra were also processed using a Bose–Einstein population factor, and no significant differences in the overall spectral features or trends were observed. The Raman spectra of xLi2O-AGPO glasses exhibit two characteristic spectral bands: (i) a low-frequency band below 800 cm−1, which is mainly associated with the bending modes of (P, Ge, Al)–O polyhedra, chains and ring units. In addition, the Boson peak arising from cooperative vibrational motion appears in the frequency range below ∼200 cm−1. (ii) A high-frequency band above 800 cm−1, which is attributed to the internal stretching modes of germanate and phosphate polyhedral units.Fig. 5(a) shows the Raman spectra, in which broad bands are observed at 69–103 (P1), 202–247 (P2), 331–349 (P3), 418 (P4), 467–498 (P5), 568–589 (P6), 655–670 (P7), 759–771 (P8), 864–890 (P9), 949–955 (P10), 1047–1054 (P11), 1120–1133 (P12), 1207–1227 (P13), and 1269–1280 cm−1 (P14). The relative intensities of the Raman bands for the xLi2O-AGPO glasses in Fig. 5(b) and the corresponding assignments of vibrational units are summarized in Table 2. Fig. 5(c) shows the composition dependence of the fractional intensity, for selected Raman bands (P7, P8, P11, and P14) in xLi2O-AGPO glasses.
| Peak no. | Peak frequency (cm−1) (relative area (%)) | Raman assignments [and references] | ||||||
|---|---|---|---|---|---|---|---|---|
| x = 0.75 | x = 1.0 | x = 1.25 | x = 1.5 | x = 1.75 | x = 2.0 | x = 2.5 | ||
| P1 | 93 (11.5) | 97 (11.3) | 103 (11.0) | 99 (10.9) | 100 (6.5) | 69 (5.7) | 69 (5.2) | Boson peak33–35 |
| P2 | 247 (10.8) | 240 (9.6) | 240 (8.5) | 245 (9.1) | 204 (6.9) | 202 (6.9) | 211 (7.5) | Ge–O–Ge bending (Q2) tetrahedra36 |
| P3 | 349 (12.6) | 344 (12.4) | 342 (12.0) | 341 (10.1) | 331 (14.5) | 333 (14.3) | 334 (10.9) | Ge deformation,37 O–P–O (Q2) bending38 |
| P4 | 418 (6.2) | 418 (5.5) | 418 (4.2) | 418 (4.4) | 418 (4.5) | 419 (6.1) | 419 (11.8) | Ge–O–Ge symmetric stretching in the GeO4 ring37 |
| P5 | 496 (17.6) | 498 (19.6) | 498 (21.0) | 498 (19.2) | 467 (13.3) | 479 (14.7) | 480 (16.1) | O–P–O bending (Q0),39,40 in-chain O–P–O stretching (Q2)41 |
| P6 | 581 (6.8) | 583 (5.5) | 585 (5.0) | 589 (8.1) | 568 (18.4) | 576 (12.9) | 576 (10.5) | Ge–O–P bending for GeO4 (ref. 42) |
| P7 | 655 (12.2) | 655 (12.6) | 655 (13.1) | 667 (10.5) | 670 (6.2) | 670 (6.7) | 670 (4.5) | P–O–P bending in chain 24, Ge-O-P bending for GeO6 (ref. 42) |
| P8 | 771 (1.4) | 770 (1.5) | 771 (1.2) | 770 (1.4) | 765 (2.0) | 763 (2.7) | 759 (4.2) | Ge–O–P stretching,37, P–O–P symmetric stretching (Q1)44 |
| P9 | 890 (0.3) | 890 (0.4) | 890 (0.5) | 890 (0.6) | 890 (0.7) | 890 (0.6) | 864 (0.6) | Ge–O vibration in GeO4 (ref. 45 and 46) |
| P10 | 950 (1.0) | 950 (1.0) | 949 (1.0) | 952 (1.1) | 950 (1.1) | 950 (1.3) | 955 (3.1) | O–P–O stretching (Q0),38 P–O–Ge4 stretching42 |
| P11 | 1050 (4.1) | 1050 (4.9) | 1050 (6.3) | 1054 (9.5) | 1050 (12.7) | 1050 (16.6) | 1047 (18.6) | PO2 symmetric stretching (Q1)50 |
| P12 | 1130 (8.6) | 1125 (9.3) | 1124 (9.7) | 1129 (9.9) | 1120 (8.1) | 1129 (7.6) | 1133 (4.6) | O–P–O symmetric stretching (Q2)51,52 |
| P13 | 1210 (3.6) | 1210 (3.4) | 1220 (4.6) | 1227 (4.3) | 1216 (5.1) | 1216 (3.7) | 1207 (2.6) | P–O symmetric stretching (Q1)53 |
| P14 | 1280 (3.3) | 1268 (3.0) | 1270 (1.6) | 1269 (0.9) | X | X | X | O–P–O asymmetric stretching (Q2),51 metaphosphate (PO3)− (Q2)54 |
The broad band observed below ∼200 cm−1 (P1) is assigned to the Boson peak, which is characteristic of disordered glassy systems.32–34 The band at 202–247 cm−1 (P2) is attributed to the Ge–O–Ge bending mode of Q2 tetrahedral units.35 The bands appearing between 331 and 349 cm−1 (P3) are associated with Ge deformation motions within the network as well as O–P–O bending vibrations related to Q2 phosphate units.36,37 The peak at 418 cm−1 (P4) originates from the symmetric stretching vibrations of bridging oxygens in six-membered GeO4 rings (Ge–O–Ge).36 The band in the range of 467–498 cm−1 (P5) is attributed to O–P–O bending vibrations in Q2 units, although contributions from O–P–O bending of Q0 units or translational modes of Ge4+ cations have also been reported in this region.38–40 The band centered around 580 cm−1 (P6) is assigned to Ge–O–P bending modes associated with GeO4 structural units.41 The bands near 655–670 cm−1 (P7) are mainly attributed to Ge–O–P bending modes involving GeO6 groups.41 According to Das et al., the increased oxygen crowding around GeO6 units hinders Ge–O–P bending, resulting in higher vibrational frequencies.41 This interpretation is consistent with the observed decrease in the relative intensity of P7 with increasing x content, as shown in Fig. 5(b). Additionally, the P7 band may also include contributions from P–O–P bending vibrations in phosphate chains.24 These observations are consistent with the NMR results reported by Schöder et al., which indicate an increase in Ge–O–P connectivity accompanied by a reduction in P–O–P linkages.42 The band in the range of 750–770 cm−1 (P8) is assigned to the symmetric stretching vibrations of P–O–P bridging oxygens between two phosphate tetrahedra, characteristic of Q1 species.43 A contribution from Ge–O–P stretching vibrations has also been reported near this frequency.36 The band near 880–890 cm−1 (P9) is attributed to Ge–O stretching vibrations in GeO4 tetrahedra.44,45 The shoulder observed around 944–955 cm−1 (P10) indicates the presence of isolated PO43− groups (Q0 units) and is associated with symmetric stretching vibrations of O–P–O non-bridging oxygens or P–O–Ge stretching modes.37,41,46 The band centered near 1050 cm−1 (P11) is assigned to P–O− valence vibrations in pyrophosphate groups (P2O74−), as well as PO2 stretching modes associated with non-bridging oxygens in Q1 tetrahedra.47–49 The intensity of this band increases systematically with increasing x content. The dominant band observed near 1120–1133 cm−1 (P12) is attributed to the symmetric stretching vibrations of non-bridging oxygens in Q2 phosphate units.50,51
The bands observed near 1207–1227 cm−1 (P13) are assigned to the symmetric stretching vibrations of P–O bonds in Q1 phosphate tetrahedra.52 The high-frequency shoulder appearing at 1269–1280 cm−1 (P14) is attributed to (PO3)− units associated with metaphosphate groups or to the asymmetric O–P–O stretching vibrations of Q2 tetrahedra.50,53 The presence of this band indicates small amounts of metaphosphate (Q2) linkages coexisting with the predominant pyrophosphate structure, consistent with an O/P ratio ≥3.5.
With increasing x content, the intensities of the bands at 1120–1133 cm−1 (P12) and 1269–1280 cm−1 (P14) progressively decrease, while those of the bands at ∼750–770 cm−1 (P8) and ∼1050 cm−1 (P11) increase. These changes indicate that the incorporation of Li+ ions into the xLi2O-AGPO glass system modifies the phosphate network through a transformation from metaphosphate (Q2) to pyrophosphate (Q1) linkages. The shift of the P–O–P vibrational modes toward lower wavenumbers, particularly for the P8 band, can be attributed to changes in the local bonding environment associated with depolymerization, where the mass contribution of phosphorus dominates relative to that of oxygen in O–P–O units. Overall, the addition of Li2O promotes the formation of pyrophosphate units via depolymerization of metaphosphate chains, leading to the generation of additional non-bridging oxygen (NBO) sites. Henderson and Amos reported that alkali germanophosphate glasses can be regarded as comprising separate germanate and phosphate subnetworks, with depolymerization occurring predominantly through the formation of Q2 and Q1 PO4 tetrahedra within the phosphate network rather than the germanate framework.50 The creation of NBO sites facilitates Li+ ion migration by providing favorable conduction pathways. Consistent with this interpretation, increasing Li/P ratios in xLi2O-AGPO glasses result in progressively depolymerized phosphate structures,52 which in turn lead to enhanced ionic conductivity with increasing x content.
For compositions with x ≥ 1.75, the relative intensity of the Ge–O–P bending mode associated with GeO4 structural units near 580 cm−1 (P6) initially increases and then decreases with further increasing x content. In contrast, the intensity of the band near 655–670 cm−1 (P7) for x ≥ 1.75 is lower than that observed for compositions with 0.75 ≤ x ≤ 1.5. Henderson and Amos proposed that the connectivity between phosphate and germanate components in alkali germanophosphate glasses is mediated through O–Li–O bridges, in which Li+ ions are coordinated by multiple oxygen atoms, including bridging oxygens (BOs) associated with GeO4 tetrahedra and non-bridging oxygens (NBOs) associated with PO4 tetrahedra.50 They further suggested that the formation of three-membered GeO4 rings provides suitable interstitial sites for accommodating alkali cations, with BOs associated with these rings acting as coordination sites. According to this model, Li+ ions simultaneously coordinate to NBOs of phosphate tetrahedra and BOs of germanate tetrahedra. The observed changes in the intensities of the P6 and P7 bands for x ≥ 1.75 therefore suggest a reduction in the number of suitable interstitial sites available for alkali cation accommodation with increasing x content in xLi2O-AGPO glasses. Although the formation of extended phosphate chains may promote long-range ion hopping pathways, the number of effectively accommodated Li+ ions gradually decreases at higher x values.
Pershina et al. suggested that the loosening of the glass network in Li2O–Al2O3–GeO2–SiO2–P2O5 glasses originates from an increase in modifier content (Li2O + Al2O3) accompanied by a decrease in glass formers (GeO2 + SiO2 + P2O5).54 It is well established that the incorporation of metal cations into phosphate glasses leads to the cleavage of P–O–P bonds and the formation of P–O–M (M = metal) linkages. Brow et al. reported that, with increasing Al content in Al(PO3)3-based systems, P–O–Li+ bonds are progressively replaced by P–O–Al bonds, accompanied by a reduction in P–O–P linkages.55 More recently, Schröder et al. demonstrated in Li1+xAlxGe2−x(PO4)3 (LAGP) glass-ceramic systems that the formation of P–O–Al and P–O–Ge linkages is favored over P–O–P and Ge–O–Al linkages.42 They further showed that the local substitution of Ge4+ by Al3+ leads to an expansion of the octahedral sites, with AlO6 polyhedra being noticeably larger than GeO6 units in the NASICON-type LiGe2(PO4)3 structure, resulting in an increase of the local P⋯Al distance by approximately 7% compared to the Ge⋯P distance. Similarly, Moguš-Milanković et al. reported that the addition of Al2O3 to alkali alumina phosphate glasses promotes the replacement of alkali metaphosphate units by aluminum metaphosphate, where Al(OP)6 units act as cross-linking centers between phosphate chains, leading to an increase in P–O–Al linkages and the formation of extended phosphate networks.49 Consistent with these observations, an increase in x content in xLi2O-AGPO glasses results in a progressive broadening of the strong Raman band at ∼1050 cm−1 (P11), indicating enhanced structural disorder. This trend is in good agreement with the disorder inferred from the Cole–Cole analysis. As the degree of structural disorder increases, the calculated Li+ mobility (µ) listed in Table 1 also increases systematically with increasing x content in xLi2O-AGPO glasses.
The evolution of the Raman spectra in xLi2O-AGPO glasses can be correlated with changes in the structural network as a function of the O/P ratio. Brow et al. reported that octahedrally coordinated Al species are predominant in phosphate glasses with O/P ratios below 3.5, corresponding to the pyrophosphate structural limit, whereas tetrahedrally coordinated Al becomes dominant at O/P ratios exceeding 3.5.55 For the xLi2O-AGPO glass with x = 2.5, the O/P ratio is expected to exceed 3.5, which is consistent with the pronounced changes observed in the Raman spectra, particularly for the bands at ∼418 cm−1 (P4) and 1047–1054 cm−1 (P11). As discussed above for glasses with higher x content, the cross-linking of phosphate chains leads to increased overlap in the distribution of potential barriers, resulting in a reduction of the effective potential well depths for Li+ ion migration. This evolution of the potential energy landscape implies a progressive lowering of the activation barriers associated with long-range ion transport. Accordingly, the dc activation energy decreases systematically from 0.68 ± 0.02 eV to 0.47 ± 0.03 eV as the x content increases from 0.75 to 2.5.
4. Conclusions
xLi2O–0.25Al2O3–1.5GeO2–1.5P2O5 (xLi2O-AGPO; x = 0.75–2.5) glasses were synthesized by the plate quenching method. Experimental and analysis results demonstrate that the dc ionic conductivity increases, while the dc activation energy decreases systematically with increasing Li2O content in the xLi2O-AGPO glasses. The calculated charge carrier concentration nc increases proportionally with the Li2O mole fraction x, whereas the ionic dc conductivity exhibits an exponential increase, primarily governed by the enhanced Li+ mobility. The improved Li+ transport is attributed to the formation of non-bridging oxygen (NBO) sites arising from depolymerization of the phosphate network. At higher Li2O contents, the formation of extended phosphate chains leads to a shallower potential energy landscape for Li+ migration, resulting in reduced activation energies. These results highlight the strong correlation between the glass network structure and ionic transport dynamics, providing important insights for the design of functional glassy solid electrolytes for electrochemical energy storage applications.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: complex impedance plane plots, conductivity and Raman spectra, and model fitting parameters. See DOI: https://doi.org/10.1039/d5ta09357k.Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. RS-2023-00276591, and RS-2021-NR062805). The work was also supported by the Ministry of Education (RS-2021-NR066344), Republic of Korea.References
- J. B. Goodenough and Y. Kim, Chem. Mater., 2019, 22, 587–603 CrossRef.
- M. Armand and J.-M. Tarascon, Nature, 2008, 451, 652–657 Search PubMed.
- Y. Kobayashi, S. Seki, A. Yamanaka, M. Miyashiro, Y. Mita and T. Iwahori, J. Power Sources, 2005, 146, 719–722 CrossRef CAS.
- J. Schwenzei, V. Thangadurai and W. Weppner, J. Power Sources, 2006, 154, 232–238 Search PubMed.
- L. He, Q. Sun, C. Chen, J. A. S. Oh, J. Sun, M. Li, W. Tu, H. Zhou, K. Zeng and L. Lu, ACS Appl. Mater. Interfaces, 2019, 11, 20895–20904 CrossRef CAS PubMed.
- Y. Zhang, H. Liu, Z. Xie, W. Qu, D. J. Freschi and J. Liu, Adv. Funct. Mater., 2023, 33, 2300973 CrossRef CAS.
- X. Xu, Z. Wen, X. Wu, X. Yang and Z. Gu, J. Am. Ceram. Soc., 2007, 90(9), 2802–2806 CrossRef CAS.
- P. H. Kuo and J. Du, J. Phys. Chem. C, 2019, 123, 27385–27398 CrossRef CAS.
- Y. Nikodimos, L. H. Abrha, H. H. Weldeyohannes, K. N. Shitaw, N. T. Temesgen, B. W. Olbasa, C.-J. Huang, S.-K. Jiang, C.-H. Wang, H.-S. Sheu, S.-H. Wu, W.-N. Su, C.-C. Yang and B. J. Hwang, J. Mater. Chem. A, 2020, 8, 26055–26065 RSC.
- S. Li, J. Cai and Z. Lin, Solids State Ion., 1988, 28–30, 1265–1270 Search PubMed.
- S. V. Pershina, B. D. Antonov, A. S. Farlenkov and E. G. Vovkotrub, J. Alloys Compd., 2020, 835, 155281 CrossRef CAS.
- C. G. Baek, H. W. Choi and Y. S. Yang, J. Alloys Compd., 2014, 615, 745–748 CrossRef CAS.
- M. C. Ersundu, A. E. Ersundu, M. T. Soltani and M. Baazouzi, Ceram. Int., 2017, 43, 491–497 CrossRef CAS.
- C. G. Baek, M. Kim, O. H. Kwon, H. W. Choi and Y. S. Yang, Cryst. Growth Des., 2017, 17, 5684–5690 CrossRef CAS.
- S. W. Martin, J. Am. Ceram. Soc., 1991, 74, 1767–1784 CrossRef CAS.
- D. Ravaine and J. L. Souquet, Phys. Chem. Glasses, 1977, 18, 27–31 CAS.
- J. L. Souquet, M. L. F. Nascimento and A. C. M. Rodrigues, J. Chem. Phys., 2010, 132, 034704 CrossRef PubMed.
- E. F. Hairetdinov, N. F. Uvarov, H. K. Patel and S. W. Martin, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 13259–13266 Search PubMed.
- M. A. T. Marple, H. Avila-Paredes, S. Kim and S. Sen, J. Chem. Phys., 2018, 148, 204507 Search PubMed.
- A. Bunde, M. D. Ingram and P. Maass, J. Non-Cryst. Solids, 1994, 172–174, 1222–1236 Search PubMed.
- D. L. Sidebottom, P. F. Green and R. K. Brow, Phys. Rev. B:Condens. Matter Mater. Phys., 1995, 51, 2770–2776 Search PubMed.
- J. Swenson and St. Adams, Phys. Rev. Lett., 2003, 90, 155507 CrossRef PubMed.
- Y. H. Rim, M. Kim, J. E. Kim and Y. S. Yang, N. J. Phys., 2013, 15, 023005 CrossRef CAS.
- C. G. Baek, Y. H. Rim and Y. S. Yang, J. Alloys Compd., 2022, 924, 166523 Search PubMed.
- J. C. Dyre, P. Maass, B. Roling and D. L. Sidebottom, Rep. Prog. Phys., 2009, 72, 046501 CrossRef.
- B. Roling, C. Martiny and S. Bruckner, Phys. Rev. B:Condens. Matter Mater. Phys., 2001, 63, 214203 CrossRef.
- Y. H. Rim, M. Kim, C. G. Baek and Y. S. Yang, J. Alloys Compd., 2020, 827, 154253 CrossRef CAS.
- E. Barsoukov and J. R. Macdonald, Impedance Spectroscopy, Third Ed, John Wiley & Sons Inc., New Jersey, 2017 Search PubMed.
- J. R. Macdonald, Solids State Ion., 1984, 13, 147–149 CrossRef CAS.
- C. León, A. Rivera, A. Várez, J. Sanz, J. Santamaria and K. L. Ngai, Phys. Rev. Lett., 2001, 86, 1279–1282 Search PubMed.
- P. Posch, S. Lunghammer, S. Berendts, S. Ganschow, G. J. Redhammer, A. Wilkening, M. Lerch, B. Gadermaier, D. Rettenwander and H. M. R. Wilkening, Energy Storage Mater., 2020, 24, 220–228 CrossRef.
- E. Courtens, M. Foret, B. Hehlen, B. Rufflé and R. Vacher, J. Phys.: Condens. Matter, 2003, 15, S1279–S1290 Search PubMed.
- N. S. Shcheblanov, L. Giacommazzi, M. E. Povarnitsyn, S. Kohara, L. Martin-Samos, G. Mountjoy, R. J. Newport, R. C. Haworth, N. Richard and N. Ollier, Phys. Rev. B, 2019, 100, 134309 Search PubMed.
- P. Tarte, A. Rulmont and C. Merckaert-Ansay, Spectrochim. Acta, Part A, 1986, 42, 1009–1016 Search PubMed.
- E. I. kamitsos, Y. D. Yiannopoulos, M. A. karakassides, G. D. Chryssikos and H. Jains, J. Phys. Chem., 1996, 100, 11755–11765 CrossRef CAS.
- S. Kumar, S. Murugavel and K. J. Rao, J. Phys. Chem. B, 2001, 105, 5862–5873 CrossRef CAS.
- B. W. Stuart, C. A. Grant, G. E. Stan, A. C. Popa, J. J. Titman and D. M. Grant, J. Mech. Behav. Biomed. Mater., 2018, 82, 371–382 CrossRef CAS PubMed.
- A. R. Zanatta, Results Phys., 2020, 19, 103500 CrossRef.
- A. Moguš-Milanković, A. Šantić, S. T. Reis, K. Furic and D. E. Day, J. Non-Cryst. Solids, 2004, 342, 97–109 CrossRef.
- S. Bano, A. R. Romero, T. Islam, D. M. Grant, I. Ahmed and T. Hussain, J. Therm. Spray Technol., 2021, 30, 1862–1874 Search PubMed.
- A. Das, P. S. R. Krishina, M. Goswami and M. Krishinan, J. Solid State Chem., 2019, 271, 74–80 Search PubMed.
- C. Schröder, J. Ren, A. Candida, M. Rodrigues and H. Eckert, J. Phys. Chem. C, 2014, 118, 9400–9411 CrossRef.
- G. Galleani, S. H. Santagneli, Y. Messaddeq, M. de Oliveira Jr and H. Eckert, Phys. Chem. Chem. Phys., 2017, 19, 21612–21624 RSC.
- J. F. Scott, Phys. Rev. B, 1970, 1, 3488–3493 CrossRef.
- T. P. Mernagh and L.-G. Liu, Phys. Chem. Miner., 1997, 24, 7–16 CrossRef CAS.
- A. Moguš-Milanković, A. Šantić, S. T. Reis, K. Furic and D. E. Day, J. Non-Cryst. Solids, 2005, 351, 3246–3258 CrossRef.
- Y. Kowada, M. Tatsumisago and T. Minami, J. Phys. Chem., 1989, 93, 2147–2151 CrossRef CAS.
- R. K. Brow, J. Non-Cryst. Solids, 2000, 263–264, 1–28 CrossRef.
- A. Moguš-Milanković, A. Šantić, A. Gajović and D. E. Day, J. Non-Cryst. Solids, 2001, 289, 204–213 CrossRef.
- G. S. Henderson and R. T. Amos, J. Non-Cryst. Solids, 2003, 328, 1–19 CrossRef CAS.
- P. Goj, M. Ciecińska, M. Szumera and P. Stoch, J. Therm. Anal. Calorim., 2020, 142, 203–209 CrossRef CAS.
- P. Stoch, A. Stoch, M. Ciecinska, I. Krakowiak and M. Sitarz, J. Non-Cryst. Solids, 2016, 450, 48–60 CrossRef CAS.
- X. Fang, C. S. Ray, A. Moguš-Milanković and D. E. Day, J. Non-Cryst. Solids, 2001, 283, 162–172 CrossRef CAS.
- S. V. Pershina, T. A. Kuznetsova, E. G. Vovkotrub and S. A. Belyakov, Membranes, 2022, 12, 1245 CrossRef CAS PubMed.
- R. K. Brow, R. J. Kirkpatrick and G. L. Turner, J. Am. Ceram. Soc., 1993, 76, 919–928 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |