Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Cu2O@COF core–shell catalyst for electrochemical CO2 reduction
Warisha
Tahir
a,
Eric
Liberra
b,
Islam E.
Khalil
a,
Prasenjit
Das
*ad,
Carsten
Prinz
c,
Franziska
Emmerling
 c,
Johannes
Schmidt
a,
Arno
Bergmann
c,
Johannes
Schmidt
a,
Arno
Bergmann
 b,
Beatriz Roldan
Cuenya
b,
Beatriz Roldan
Cuenya
 b and
Arne
Thomas
b and
Arne
Thomas
 *a
*a
aDepartment of Chemistry, Functional, Materials, Technische Universität Berlin, 10623 Berlin, Germany. E-mail: arne.thomas@tu-berlin.de
bDepartment of Interface Science, Fritz Haber Institute of the Max Planck Society, Faradayweg 4-6, D-14195 Berlin, Germany
cBAM Federal Institute for Materials Research and Testing, Richard-Willstätter-Str. 11, 12489 Berlin, Germany
dDepartment of Chemistry, Indian Institute of Technology Ropar, Punjab 140001, India
First published on 5th March 2026
Abstract
The electrochemical reduction of CO2 (CO2RR) to C2+ products offers a sustainable approach for carbon utilization and the production of valuable chemicals. Cu2O nanocubes (NCs) have emerged as effective electrocatalysts for the formation of C2+ products. In this study, we applied a coating of Cu2O NCs with robust covalent organic framework (COF) layers prepared by multicomponent reactions. Compact layers of COFs are formed, which, due to their porosity, allow full accessibility of the Cu2O catalytic surfaces to CO2 molecules. Furthermore, the thickness and chemical functionality of the COF layers can be continuously varied by changing the type and quantity of monomers used to cover the Cu2O NCs. Electrocatalytic measurements demonstrate that these hybrid core–shell Cu@COF systems remain active in CO2 reduction, achieving good current density and faradaic efficiency, while maintaining selectivity for the desired CO2 reduction products. Additionally, the COF shell provides remarkable stability to the system under electrochemical conditions.
Introduction
The extensive reliance on fossil fuel combustion to meet the global energy demand has increased the CO2 level in the atmosphere, leading to alarming environmental threats, such as global warming.1,2 Since the 1970s, many scientists have focused their attention to finding sustainable solutions for the capture and conversion of atmospheric CO2 to value-added products in order to mitigate the rising levels of CO2 in the atmosphere.3,4 Among various possible strategies, the electrochemical CO2 reduction reaction (CO2RR) can be an attractive, clean and cost effective approach to close the anthropogenic carbon cycle by converting carbon dioxide (CO2) back into carbon-based value-added fuels and chemicals. Manipulating electrochemical CO2 conversion to various valuable products such as C1 (formic acid, methane, carbon monoxide, methanol), C2 (ethanol, ethylene) or C3 products (propanol and propanal) can significantly alter the current trend of rising CO2 emission towards a closed sustainable cycle, leading to alleviation of climate change.5,6However, the most challenging task is finding an ideal electrocatalyst that exhibits high stability at low applied potential and high selectivity for C2+ products by suppressing, e.g., the parasitic hydrogen evolution reaction. Enormous amounts of effort have been made to design and develop an effective electrocatalyst for selective CO2 reduction into multi-carbon C2+ products.7 These C2+ products are not only essential feedstocks for chemical synthesis, but also provide a sustainable pathway to store renewable energy in chemical form. In this context, numerous transition metals have been examined for the CO2RR to C2+ products.8–11 However, high overpotentials, poor catalytic performance, low selectivity, high operational costs, and low durability are the main concerns yet to be addressed.
Among them, Cu2O nanocubes (NCs) have been described as one of the most promising electrocatalysts for obtaining C2+ products.12,13 The unique electronic properties, ability to stabilize critical intermediates and selectivity towards C2+ products make the Cu2O NCs a promising electrocatalyst for CO2 reduction.14–16
However, Cu2O nanoparticles are thermodynamically stable in air but eventually undergo degradation to copper(II) oxide in the presence of light and moisture.17 This facile surface degradation of Cu2O to CuO damages the surface of the electrocatalyst, resulting in a reduction of catalytic activity.18 Moreover, Cu2O deposited on electrodes also suffered from poor stability and surface passivation. In order to protect Cu2O NCs from surface oxidation degradation to enhance their catalytic performance, many approaches have been adopted, such as the formation of hybrid metal/Cu2O heterostructures,19,20 nanocomposites@Cu2O core/shell structures,21–23 and metal-doped Cu2O,24,25 which improved the stability towards surface degradation with selective conversion of CO2 to C2+ products.
Recently, it has been shown that coating Cu2O with organic molecules or polymers can alter its catalytic properties. Many studies have demonstrated that Cu2O with organic coatings, such as polyaniline (PANI),26 polypyrrole (PPy)27 and metal organic frameworks (MOFs),28 provide enhanced performances like improved selectivity, better electron transfer, higher CO2 adsorption, and suppressed hydrogen evolution, leading to a more efficient and selective CO2RR process.
Recently, covalent organic frameworks (COFs), have attracted considerable attention due to their tunable chemical structure and topology as well as high surface area and porosity.29,30 Because of these beneficial properties, COFs have already been described as promising materials for many applications such as photocatalysis,31–33 energy storage,34–36 gas adsorption,37,38 or biomedical application.39
In this regard, metal oxide/COF composites such as Fe3O4@COF,40–42 TiO2@COF,43 and NiFe2O4@COFs44 have also been developed and investigated for several applications. However, the formation of these composites often requires precise multistep synthetic protocols and the stability of the COF shell under harsh conditions is sometimes critical. Therefore, developing a simple and efficient one-pot strategy to synthesize well-defined metal oxide/COF composites with high durability remains a major but rewarding challenge.45,46
In this study, we use a core–shell system made by coating Cu2O NCs with a multicomponent COF via the Povarov reaction in a one-pot synthesis. The Cu2O NCs are then encapsulated with a compact COF shell. By changing the ratio of Cu2O NCs to COF monomers, the thickness of the COF shell could be continuously varied from 51 to 75 nm. The 2D COF layers grow parallel to the Cu2O surface, resulting in COF pores that are oriented perpendicular to the surface, thereby, making the catalyst surface and form open channels fully accessible to the reactants. Electrocatalytic tests ensured that the CO2 molecules can easily reach the catalytically active sites of Cu2O due to the high porosity of the COFs.
Results and discussion
Cu2O NCs with well defined (110) and (100) facets were synthesized via wet colloidal reduction of CuCl2 by L-ascorbic acid according to the previous literature with small modifications.47 The following properties were defined in advance for the COF selected for the coating: it should possess good crystallinity and high porosity to ensure access to the Cu2O surface. The COF must be furthermore very stable in order to remain a compact layer even under harsh electrocatalytic conditions. Finally, we hypothesized that Lewis-basic groups within the pores could be useful to enable a high absorption capability of CO2 and possibly even enrich CO2 on the catalytically active surface. We therefore decided for a pyridine-functionalized COF synthesized via the three-component Povarov reaction. Importantly, the pyridine-linked COF was selected due to the presence of pyridinic nitrogen sites, which are expected to play an active role in regulating the reaction pathway. These pyridinic N sites serve dual roles: (i) they can interact with CO2 molecules through Lewis acid–base and dipole interactions, which could facilitate CO2 accumulation near the catalytic interface, and (ii) they may coordinate with surface Cu species via N → Cu donor–acceptor interactions. Such coordination is proposed to stabilize Cu+ species at the interface and may favor the formation of Cu+/Cu0 interfacial sites, which are widely regarded as beneficial for CO2 activation and *CO2−/*COOH intermediate stabilization. Furthermore, the interfacial interaction between the Py-COF shell and the Cu2O core is expected to induce electronic coupling across the interface, thereby potentially modulating the local electronic structure of Cu and contributing to the observed enhancement in catalytic activity and stability. It was recently shown that this multicomponent reaction produces highly crystalline, high surface area and chemically very stable COFs, with a pore interior size that can be easily adjusted by the chosen vinyl component.48,49 For the synthesis, 2,4,6-Tris(4-aminophenyl) triazine (TAT), 4,4′,4″-trinitrilotribenzaldehyde (TBA) and 2-vinyl pyridine were reacted in a solvent mixture of o-dichlorobenzene/n-butanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and a catalytic amount of BF3·Et2O at 120 °C for 72 h to yield a yellow-colored COF with 86% yield.
1) and a catalytic amount of BF3·Et2O at 120 °C for 72 h to yield a yellow-colored COF with 86% yield.
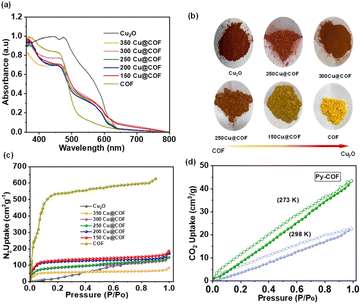
When adding the preformed Cu2O NCs to this mixture, a uniform COF layer with tuneable thickness was successfully grown on the Cu2O, resulting in well-defined core–shell structures. Using a fixed amount of COF monomers, we varied the amount of Cu2O from 150 to 350 mg (in increments of 50 mg) to achieve variation of the shell thickness. Samples were named xCu@COF NCs (where x = the amount in mg of Cu2O used). (SI Section 2: synthesis of Cu2O, COFs and xCu@COF electrocatalysts). The as-synthesized xCu@COF NCs show a color gradient from red to yellow with increasing shell thickness (vide supra).
The morphology and composition of Cu2O, COF and the core–shell xCu@COF NCs were examined by Field Emission Scanning Electron Microscopy (FESEM), High Resolution Transmission Electron Microscopy (HRTEM) and Energy-Dispersive X-ray Spectroscopy (EDX). Both HRTEM and FESEM images revealed well defined Cu2O NCs with particle size of ∼600 nm (Fig. 1a and S1). On the other hand, the pristine COF shows an irregular morphology in FESEM (Fig. S2). Using FESEM, a compact coating of the COF material with uniform thickness on the entire surface of the Cu2O NCs was observed (Fig. S3–S5). Elemental mapping confirms that the elements Cu and O are located in the core, while C and N elements are found in the shell (Fig. 1c), confirming the formation of a core–shell xCu@COF NC nanostructure. This is further proven by HRTEM measurements showing cubic cores surrounded by a compact and uniform bright shell formed from the COF with the average thickness varying from ∼51 ± 5 to ∼75 ± 2 nm (Fig. 1d and S6–S11). Besides the uniform coatings, some small spherical particles are seen on the COF layer under HRTEM. Elemental mapping rules out the possibility that these particles consist of copper oxide and it can be therefore assumed that these are residual COF particles which have first formed in the bulk before they reacted with the monomers of the growing COF layers. The TEM images at high magnification also reveal that the pores of the COFs are orientated exactly perpendicular to the Cu2O surface (Fig. S9 and S10), which is an important prerequisite to ensure unhindered mass transport to and from the catalytically active surface. Finally, HRTEM line scanning confirms that the core–shell structure consists of Cu2O as the core and the COF as the shell (Fig. S12).
The crystallinity of Cu2O, the pristine COF and the core–shell xCu@COF NCs was investigated by powder X-ray diffraction (PXRD) patterns. The Cu2O show reflections at 29.6°, 36.4° and 42.3°, which can be attributed to the (110), (111) and (200) facets, respectively, evidenced by JCPDS pattern (34-1354-Cu2O). For the pure COF, the reflections at 4.3 and 7.4° 2θ correspond to the (100) and (2–10) facets, and a broad diffraction at ∼22.3° 2θ suggests the formation of a 2D layered material stacking in the ‘c’ direction confirming the formation of a crystalline COF (Fig. 2a).50 All the core–shell xCu@COF structures showed both reflections of Cu2O and COF. The ratio of the reflections from Cu2O NCs and COF changes continuously with x and follows the expected trend.
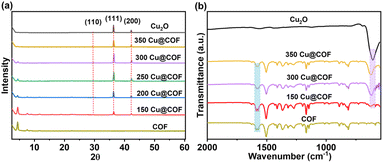 | ||
| Fig. 2 (a) PXRD patterns of Cu2O NCs, pristine COF and core–shell xCu@COF NCs. (b) FTIR spectra of Cu2O NCs, COF and core–shell xCu@COF NCs. | ||
To examine the chemical structure of the as-produced core–shell xCu@COF NCs, Fourier transform infrared (FTIR) spectroscopy measurements were carried out. The sharp peak at 603 cm−1 is assigned to the stretching vibrations of Cu–O, confirming the successful formation of Cu2O, while the strong peaks at 1580 cm−1 and 1624 cm−1 correspond to typical stretching vibrations of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C
C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, and thus can be attributed to the COF shell.51,52 Again, the FTIR spectra of the core–shell xCu@COF NC systems combine all the characteristic peaks of both Cu2O and COF, with changing ratios as the amount of core and shell varies (Fig. 2b).
N, and thus can be attributed to the COF shell.51,52 Again, the FTIR spectra of the core–shell xCu@COF NC systems combine all the characteristic peaks of both Cu2O and COF, with changing ratios as the amount of core and shell varies (Fig. 2b).
X-ray photoelectron energy spectroscopy (XPS) measurements were carried out to investigate the chemical state of the elements present in the xCu@COF electrocatalysts. The XPS survey spectrum of the optimized core–shell 300Cu@COF system confirms the presence of Cu, O, N, and C elements (Fig. S13). Compared to pure Cu2O NCs, where the peaks for Cu(I) (overlapping with those of metallic Cu) appear at around 932 eV and 952 eV, corresponding to the Cu 2p3/2 and Cu 2p1/2 levels, respectively, the core–shell xCu@COF electrocatalysts additionally showed shoulder peaks of Cu 2p3/2 and Cu 2p1/2 for Cu(II), typically appearing at 933 eV and 953 eV (Fig. S14). This shows that some amount of surface oxidation of the Cu2O NCs occurs during the formation of the COF shell. These shifts cause the splitting of the main peaks into two components, indicating the coexistence of both Cu(I) and Cu(II) on the material's surface. It should be noted that XPS might also detect some scattered Cu ions within the COF layer since the COF layer thickness is larger than the inelastic mean free path of the photoelectrons. Due to the presence of the Cu ions on the surface of the core–shell xCu@COF electrocatalysts, satellite peaks typically appear at around 940 eV.53 N 1s XPS measurements further validate the presence of quinoline, triazine, and pyridine nitrogen, as well as triphenyl amine groups of the COF from which the first three show a peak at 398.7 and the latter at 400.1 eV, respectively, while the peak near 402 eV corresponds to oxidized nitrogen species.54 With decreasing shell thickness, the overall intensity of N 1s XPS decreases (Fig. S15).
Solid-state ultraviolet-visible diffuse reflectance spectra (UV-vis DRS) were collected for the core–shell xCu@COF nanocubes and compared with those of pristine Cu2O nanocrystals and the isolated Py-COF. The Py-COF exhibits an absorption onset at approximately 520 nm, which is characteristic of an extended π-conjugated aromatic framework. In contrast, Cu2O nanocrystals show a broader absorption extending to longer wavelengths, with an onset around 640 nm (Fig. 3a). Upon formation of the core–shell architecture, the optical response of the xCu@COF composites reflects the combined contributions of both components, with the relative absorption intensity in the visible region increasing with increasing Cu2O content, consistent with the compositional trend. The gradual color evolution of the xCu@COF materials further supports the optical characteristics of the COF shell and the Cu2O core (Fig. 3b). Importantly, the visible-light absorption of the Py-COF is indicative of significant π-electron delocalization within the framework, which is expected to facilitate electronic coupling and interfacial charge transfer in the core–shell catalyst.
The formation of the Py-COF shell on Cu2O nanocrystals is proposed to occur via preferential heterogeneous nucleation at the Cu2O surface during COF formation. The Cu2O nanocrystals expose under-coordinated Cu and surface oxide sites, which can interact with the amine and aldehyde monomers and may lower the nucleation barrier for COF growth compared to a homogeneous nucleation in solution. As a result, COF crystallization is expected to be initiated at the Cu2O interface rather than in the bulk solution. Upon nucleation, the COF grows outward to form a conformal and continuous shell, yielding well-defined core–shell architectures with tuneable shell thickness, consistent with the TEM observations.
Nitrogen sorption isotherms were measured to determine the specific surface area of the pristine COF, Cu2O NCs and core–shell xCu@COF NC catalysts. The surface areas of the Cu2O NCs and pristine COF are 132 m2 g−1 and 1720 m2 g−1, respectively. The surface area of the core–shell xCu@COF NCs gradually increases with increasing amount of the COF (Fig. 3c), with a surface area of 386 m2 g−1 for the composite with the thinnest shell (350Cu@COF) to 414 m2 g−1 for an intermediate thickness shell (250Cu@COF) and 710 m2 g−1 for the thickest shell (150Cu@COF) (Table S16). Notably, the surface areas of the core–shell materials remain significantly lower than that of the pristine COF. Importantly, this reduction does not necessarily indicate a loss of porosity of the COF shell but rather reflects the common intrinsic characteristics of core–shell systems, in which the nonporous inorganic core contributes minimally to gas adsorption.
Solid-state CO2 adsorption measurements were performed to further elucidate the ability of the Py-COF shell to enrich CO2 at the catalyst interface. CO2 sorption isotherms recorded at 273 and 298 K reveal a clear temperature-dependent uptake behaviour of physisorption (Fig. 3d). Notably, the Py-COF exhibits measurable CO2 adsorption even at 298 K (23 cm3 g−1), demonstrating sufficient affinity for CO2 under conditions relevant to electrocatalytic operation, while at 273 K the amount absorbed was high, reaching 43 cm3 g−1. The corresponding isosteric heat of adsorption (Qst) for the Py-COF, derived from adsorption isotherms measured at 273 and 298 K, is shown in Fig. S17. The Py-COF exhibits a relatively high initial Qst, which decreases and stabilizes at lower values with increasing CO2 loading. These values show that the COFs have the ability to adsorb considerable amounts of CO2, while the adsorption enthalpies found are in an intermediate range to ensure accumulation but not too strong binding of CO2, enabling further utilization. This trend indicates the presence of stronger adsorption sites even at low CO2 uptake, followed predominantly by physisorption at higher CO2 coverage. These results provide direct experimental evidence that pyridine groups in the COF shell contribute to CO2 enrichment at the catalyst interface, supporting their role in enhancing CO2RR performance.
Thermogravimetric analysis (TGA) was carried out to investigate the thermal stability of the composites (Fig. S18). The pure Cu2O NCs are highly stable and no weight loss is observed until 800 °C under nitrogen gas. On the other hand, for the pristine COF, a major weight loss starts from 500 °C. The slight weight loss of ∼5 wt% from room temperature (RT) to 500 °C can be probably attributed to surface-adsorbed water or residual solvents from the synthesis. For the core–shell xCu@COF NCs, the first major weight loss starts even earlier, namely at around 440 °C in all cases, probably showing a catalytic effect of the metal oxide core on the decomposition of the COF. As the decomposition of the COFs occurs in a stepwise manner and is not yet completed at 800 °C, the weight ratio of the COF shell is difficult to determine. However, the remaining mass at 800 °C follows again the expected trend, and thus is lower when a higher amount of COF was originally deposited.
Electrochemical measurements
Having achieved a uniform coating of Cu2O NCs with crystalline and highly porous COFs with different shell thicknesses, we were interested to see (a) if the Cu2O surface is still accessible for charge carriers and reactants, and thus if the composites are also active in electrocatalytic reactions and (b) if the organic coating and its thickness influence the activity and product selectivity of the catalysts. The electrocatalytic performance of the xCu@COF electrocatalysts was examined by chronoamperometric (CA) measurements at −1.0 V vs. RHE for 1 h in an electrolyte of 0.1 M KHCO.The current density of all synthesized xCu@COF electrocatalysts are comparable to the pure Cu2O, indicating that the Cu2O surface remains fully accessible to reactants due to the high porosity of the COFs and that the π-conjugated framework of the COF is allowing electronic transport to the core. In addition to the activity, the selectivity of all electrocatalysts towards H2, C1 and C2+ products as compared via their faradaic efficiencies is also comparable or slightly lower than for the pure Cu2O (Fig. 4a). Methane formation appears to be promoted with all coated catalysts, but at the expense of the more desirable ethylene formation. In general, no clear monotonic trend in the formation of one or the other product with the COF shell thickness can be drawn. However, the shift towards methane over ethylene formation is the strongest for the thinnest and thickest COF overlayers over the Cu2O NCs. Following this finding, it is noteworthy that 300Cu@COF shows the highest FE towards C3 products, reaching 4.9%, 3.9% and 2.8% for n-propanol, propionaldehyde and allyl alcohols, respectively (Fig. 4b), formed at the expense of a lower FE for ethylene as observed compared to the pure Cu2O NCs. Our selectivity data suggest furthermore a link between a decreased methane FE and an increased C3 FE. The current density during the CO2RR of 300Cu@COF is highest among bare Cu2O NCs and the thinner and thickest core–shell xCu@COF electrocatalysts showing that the optimal thickness of the COF shell around the Cu2O core plays a crucial role in catalytic activity (Fig. S19).
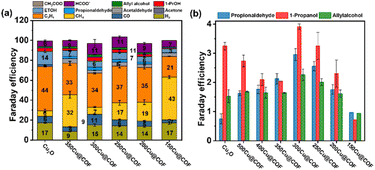 | ||
| Fig. 4 (a) faradaic efficiencies (FE, %) of H2, C1, and C2+ for Cu2O and xCu@COF electrocatalysts. (b) Selectivity of Cu2O and different xCu@COF electrocatalysts for C3 products. | ||
Besides, a clear potential-dependent selectivity behaviour was revealed by faradaic efficiencies and partial current densities as a function of applied potential (−0.8 to −1.3 V vs. RHE) together with linear sweep voltammetry (LSV) curves for the best-performing 300Cu@COF catalyst (Fig. S20a and b). At relatively positive potentials (−0.8 and −0.9 V), H2 evolution dominates with minor CO formation, indicating that CO2 reduction is kinetically limited and C–C coupling is negligible. Upon increasing the potential to −1.0 V, a significant shift in product distribution is observed, where C2H4 becomes the major carbon product and the overall C2+ selectivity reaches its maximum. When going towards more negative potentials (−1.1 and −1.3 V), the total current density increases; however, the selectivity shifts toward CH4 formation, indicating enhanced hydrogenation of *CO intermediates rather than C–C coupling. Therefore, −1.0 V vs. RHE represents the optimal potential, providing the best balance between catalytic activity and C2+ selectivity.
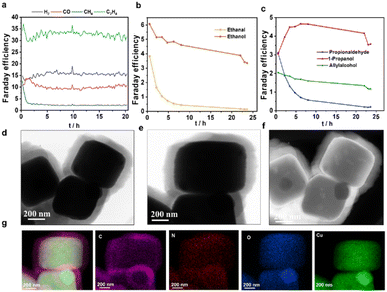
In consequence, the CO2RR stability of 300Cu@COF was tested for 24 h, and we found a stable production of ethylene up to 20 h and a significantly lower FE towards methane. In parallel, it can be seen that in the initial 8 h period, the FE of the most important liquid products decreased while the FE towards the C3 liquid product 1-propanol increased. This happens at the expense of 1-propanal, showing that the aldehyde formed is reduced to the alcohol in the course of the reaction (Fig. 5a–c). Thus, it can be concluded that the C2+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C1 product ratio increases during extended operation.
C1 product ratio increases during extended operation.
TEM images after a 24 h stability test showed that the core–shell 300Cu@COF electrocatalyst still retains its structure and morphology after the electrocatalytic reaction, finally proving the stable coating of COF on Cu2O (Fig. 5d–g). Consistently, SEM images of xCu@COF electrocatalysts show that the core–shell cubic morphology remained intact after a 24 h electrochemical test (Fig. S21–S23). It has to be noted that besides the xCu@COF, smaller Cu-based nanostructures formed, but their formation could not be quantified and thus linked to the determined CO2RR performance. However, the observed CO2RR behaviour, characterized by suppressed hydrogen evolution and stable C2+ product formation, is not typical of bare small Cu nanoparticles, suggesting the dominant catalytic activity arises from the confined Cu2O@Py-COF architecture rather than from newly formed tiny Cu particles. Overall, these results indicate that the Py-COF shell remains functionally intact and continues to regulate the catalytic interface during operation.
In addition, it is well established that Cu2O-based catalysts undergo partial or complete reduction to metallic Cu under cathodic CO2RR conditions.55 Accordingly, Cu2O in the present system should be regarded as a precatalyst rather than the static active phase. Post-reaction TEM and EDX analyses do not show a clear determination of the copper oxidation state; these observations suggest that any reduction-induced reconstruction does not occur with large aggregation of metallic Cu-particles. Importantly, the presence of the Py-COF shell is expected to influence this transformation by restricting large-scale particle aggregation and stabilizing interfacial Cu+ species. Such stabilized Cu+/Cu0 interfacial sites have been widely proposed as key contributors to enhanced CO2RR selectivity.56,57
Conclusion
In conclusion, core–shell xCu@COF nanocubes are applied as electrocatalysts for the CO2RR. The COF shells are formed by the multi-component Povarov reaction and provide the COF with high stability and the possibility of introducing various functionalities into the pores. In this case, the COF contains pyridine groups as Lewis bases in the pores and the shell thickness can be continuously varied. High resolution TEM measurements reveal that the COF layers grow parallel to the Cu2O surface, so that the pores of the COF are orientated perpendicular to it. This orientation enables very good accessibility of reactants to the metal oxide core, which is confirmed by the fact that the core–shell structure still shows high catalytic activity. Furthermore, the π-conjugated and fully aromatic framework of the Py-COF is expected to facilitate interfacial charge transfer between the Cu2O core and the electrolyte, thereby supporting sustained electrocatalytic activity. This is exemplified by using the xCu@COF structures as electrocatalysts for CO2 reduction where they show stable performance over 20 h.Author contributions
W. T., P. D. and A. T. in collaboration with A. B. and B. R. C. conceived the project. W. T. designed and synthesized the materials, interpreted the analytical data and wrote the original draft. E. L. conducted electrochemical tests. P. D. performed TEM images. All authors analyzed and discussed the results and jointly prepared the final manuscript.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, characterization results (TEM, SEM, XPS, gas sorption, TGA). See DOI: https://doi.org/10.1039/d5ta09283c.Acknowledgements
The authors are grateful to Germany's Excellence Strategy-EXC 2008-390540038-UniSysCat and the German Federal Ministry of Education and Research (Bundesministerium für Bildung and Forschung, BMBF) under grant no. 03EW0015B (CatLab) for financial support. W. T. thanks the Einstein Center of Catalysis/Berlin International Graduate School of Natural Sciences and Engineering. P. D. acknowledges the Alexander von Humboldt Foundation and Anusandhan National Research Foundation (ANRF) Ramanujan Fellowship (RJF/2025/000660), Govt. of India, for financial support. The authors extend special thanks to Hüseyin Küçükkeçeci, Christina Eichenauer and Maria Unterweger for their assistance.References
- A. Cherevotan, J. Raj, L. Dheer, S. Roy, S. Sarkar, R. Das and S. C. Peter, ACS Energy Lett., 2021, 6, 509–516 CrossRef CAS
.
- S. C. Peter, ACS Energy Lett., 2018, 3, 1557–1561 CrossRef CAS
.
- S. Karmakar, S. Barman, F. A. Rahimi and T. K. Maji, Energy Environ. Sci., 2021, 14, 2429–2440 RSC
.
- D. Gao, R. M. Arán-Ais, H. S. Jeon and B. Roldan Cuenya, Nat. Catal., 2019, 2, 198–210 CrossRef CAS
.
- M. Cai, Z. Wu, Z. Li, L. Wang, W. Sun, A. A. Tountas, C. Li, S. Wang, K. Feng, A. B. Xu and S. Tang, Nat. Energy, 2021, 6, 807–814 CrossRef CAS
.
- D. Bagchi, J. Raj, A. K. Singh, A. Cherevotan, S. Roy, K. S. Manoj and S. C. Peter, Adv. Mater., 2022, 34, 2109426 CrossRef CAS
.
- F. Franco, C. Rettenmaier, H. S. Jeon and B. R. Cuenya, Chem. Soc. Rev., 2020, 49, 6884–6946 RSC
.
- Y. Zhou, F. Che, M. Liu, C. Zou, Z. Liang, P. De Luna and E. H. Sargent, Nat. Chem., 2018, 10, 974–980 CrossRef CAS
.
- H. Xu, D. Rebollar, H. He, L. Chong, Y. Liu, C. Liu and T. Xu, Nat. Energy, 2020, 5, 623–632 CrossRef CAS
.
- K. Jiang, R. B. Sandberg, A. J. Akey, X. Liu, D. C. Bell, J. K. Nørskov and H. Wang, Nat. Catal., 2018, 1, 111–119 CrossRef CAS
.
- Z. Li, D. He, X. Yan, S. Dai, S. Younan, Z. Ke and J. Gu, Angew. Chem., Int. Ed., 2020, 59, 18572–18577 CrossRef CAS PubMed
.
- P. Grosse, A. Yoon, C. Rettenmaier, A. Herzog, S. W. Chee and B. Roldan Cuenya, Nat. Commun., 2021, 12, 6736 Search PubMed
.
- J. Timoshenko, A. Bergmann, C. Rettenmaier, A. Herzog, R. M. Arán-Ais, H. S. Jeon and F. T. Haase,
et al.
, Nat. Catal., 2022, 5, 259–267 CrossRef CAS
.
- C. Zhan, F. Dattila, C. Rettenmaier, A. Bergmann, S. Kühl, R. García-Muelas, N. López and B. Roldan Cuenya, ACS Catal., 2021, 11, 7694–7701 Search PubMed
.
- L. Xu, P. Trogadas and M.-O. Coppens, Adv. Energy Mater., 2023, 13, 2302974 Search PubMed
.
- C. Rettenmaier, A. Herzog, D. Casari, M. Rüscher, H. S. Jeon, D. Kordus and M. Lopez Luna,
et al.
, EES Catal., 2024, 2, 311–323 Search PubMed
.
- X. Wang, M. Chen, Y. He and J. Zhu, J. Alloys Compd., 2015, 628, 50–56 CrossRef CAS
.
- Y. Gao, Q. Wu, X. Liang, Z. Wang, Z. Zheng, P. Wang, Y. Liu, Y. Dai, M.-H. Whangbo and B. Huang, Adv. Sci., 2020, 7, 1902820 CrossRef CAS PubMed
.
- L. Wang, J. Ge, A. Wang, M. Deng, X. Wang, S. Bai, R. Li, J. Jiang, Q. Zhang, Y. Luo and Y. Xiong, Angew. Chem., Int. Ed., 2014, 53, 5107–5111 CrossRef CAS
.
- H. Jung, S. Y. Lee, C. W. Lee, M. K. Cho, D. H. Won, C. Kim, H.-S. Oh, B. K. Min and Y. J. Hwang, J. Am. Chem. Soc., 2019, 141, 4624–4633 CrossRef CAS PubMed
.
- Q. Zhang, P. Liu, T. Wang, Q. Liu and D. Wu, J. Mater. Chem. A, 2023, 11, 24823–24835 RSC
.
- M. Xia, L. Pan, Y. Liu, J. Gao, J. Li, M. Mensi, K. Sivula, S. M. Zakeeruddin, D. Ren and M. Grätzel, J. Am. Chem. Soc., 2023, 145, 27939–27949 CrossRef CAS
.
- S. Wang, W. Peng, H. Ma, X. Pan, Y. Shen, J. Zhao, J. Ye, Q. Wang, P. Xie, S. Zhang and X. Gao, ACS Catal., 2025, 15, 14127–14137 CrossRef CAS
.
- Y. Jia, Y. Ding, T. Song, Y. Xu, Y. Li, L. Duan, F. Li, L. Sun and K. Fan, Adv. Sci., 2023, 10, 2303726 CrossRef CAS PubMed
.
- Q. Wu, R. Du, P. Wang, G. I. N. Waterhouse, J. Li, Y. Qiu, K. Yan, Y. Zhao, W.-W. Zhao, H.-J. Tsai, M.-C. Chen, S.-F. Hung, X. Wang and G. Chen, ACS Nano, 2023, 17, 12884–12894 CrossRef CAS
.
- L. Zhang, D. Jing, L. Guo and X. Yao, ACS Sustain. Chem. Eng., 2014, 2, 1446–1452 CrossRef CAS
.
- N. Maruthi, M. Faisal, N. Raghavendra, B. P. Prasanna, S. R. Manohara and M. Revanasiddappa, Colloids Surf., A, 2021, 621, 126611 CrossRef CAS
.
- P. Asen and S. Shahrokhian, J. Phys. Chem. C, 2017, 121, 6508–6519 CrossRef CAS
.
- X. Yang, J. Cheng, X. Yang, Y. Xu, W. Sun and J. Zhou, Chem. Eng. J., 2022, 431, 134171 CrossRef CAS
.
- K. Geng, T. He, R. Liu, S. Dalapati, K. T. Tan, Z. Li, S. Tao, Y. Gong, Q. Jiang and D. Jiang, Chem. Rev., 2020, 120, 8814–8933 CrossRef CAS PubMed
.
- R. B. Lin and B. Chen, Joule, 2018, 2, 1030–1032 CrossRef
.
- I. E. Khalil, P. Das, H. Küçükkeçeci, V. Dippold, J. Rabeah, W. Tahir, J. Roeser, J. Schmidt and A. Thomas, Chem. Mater., 2024, 36, 8330–8337 CrossRef CAS
.
- Y. Zhao, L. Hao, A. Ozden, S. Liu, R. K. Miao, P. Ou, T. Alkayyali, S. Zhang, J. Ning, Y. Liang and Y. Xu, Nat. Synth., 2023, 2, 403–412 CAS
.
- M. Zhang, M. Lu, Z. L. Lang, J. Liu, M. Liu, J. N. Chang, L. Y. Li, L. J. Shang, M. Wang, S. L. Li and Y. Q. Lan, Angew. Chem., 2020, 132, 6562–6568 CrossRef
.
- S. Li, B. Kumbhakar, B. Mishra, J. Roeser, N. Chaoui, J. Schmidt, A. Thomas and P. Pachfule, ACS Appl. Energy Mater., 2023, 6, 9256–9263 CrossRef CAS
.
- L. M. Lanni, R. W. Tilford, M. Bharathy and J. J. Lavigne, J. Am. Chem. Soc., 2011, 133, 13975–13983 CrossRef CAS PubMed
.
- Y. Liu, Y. Zhu, S. B. Alahakoon and E. Egap, ACS Mater. Lett., 2020, 2, 1561–1566 CrossRef CAS
.
- C. J. Doonan, D. J. Tranchemontagne, T. G. Glover, J. R. Hunt and O. M. Yaghi, Nat. Chem., 2010, 2, 235–238 CrossRef CAS PubMed
.
- R. J. White, R. Luque, V. L. Budarin, J. H. Clark and D. J. Macquarrie, Chem. Soc. Rev., 2009, 38, 481–494 RSC
.
- S. Bhunia, K. A. Deo and A. K. Gaharwar, Adv. Funct. Mater., 2020, 30, 2002046 CrossRef CAS
.
- T. Sun, J. Xie, W. Guo, D.-S. Li and Q. Zhang, Adv. Energy Mater., 2020, 10, 1904199 CrossRef CAS
.
- V. Romero, S. P. Fernandes, L. Rodriguez-Lorenzo, Y. V. Kolen'ko, B. Espiña and L. M. Salonen, Nanoscale, 2019, 11, 6072–6079 RSC
.
- Y. Liao, J. Li and A. Thomas, ACS Macro Lett., 2017, 6, 1444–1450 CrossRef CAS PubMed
.
- C. C. Li, M. Y. Gao, X. J. Sun, H. L. Tang, H. Dong and F. M. Zhang, Appl. Catal., B, 2020, 266, 118586 CrossRef CAS
.
- X. Wang, H. Ji, F. Wang, X. Cui, Y. Liu, X. Du and X. Lu, Microchim. Acta, 2021, 188, 161 CrossRef CAS PubMed
.
- W. Tahir, Y. Wei, M. Wang, I. E. Khalil, P. Das, T. Wang, C. Cheng, S. Li and A. Thomas, J. Am. Chem. Soc., 2026, 148, 743–755 CrossRef CAS PubMed
.
- Y. Zhu, D. Zhu, Q. Yan, G. Gao, J. Xu, Y. Liu, S. B. Alahakoon, M. M. Rahman, P. M. Ajayan, E. Egap and R. Verduzco, Chem. Mater., 2021, 33, 6158–6165 CrossRef CAS
.
- Y. A. Wu, I. McNulty, C. Liu, K. C. Lau, Q. Liu, A. P. Paulikas, C.-J. Sun, Z. Cai, R.J. Guest, Y. Ren, V. Stamenkovic, L. A. Curtiss, Y. Liu and T. Rajh, Nat. Energy, 2019, 4, 957–968 CrossRef CAS
.
- P. Das, G. Chakraborty, J. Yang, J. Roeser, H. Küçükkeçeci, A. D. Nguyen, M. Schwarze, J. Gabriel, C. Penschke, S. Du, V. Weigelt, I. E. Khalil, J. Schmidt, P. Saalfrank, M. Oschatz, J. Rabeah, R. Schomäcker, F. Emmerling and A. Thomas, Adv. Energy Mater., 2026, 16, 2501193 CrossRef CAS
.
- P. Das, G. Chakraborty, N. Friese, J. Roeser, C. Prinz, F. Emmerling, J. Schmidt and A. Thomas, J. Am. Chem. Soc., 2024, 146, 17131–17139 CrossRef CAS PubMed
.
- P. Das, G. Chakraborty, J. Roeser, S. Vogl, J. Rabeah and A. Thomas, J. Am. Chem. Soc., 2023, 145, 2975–2984 CrossRef CAS PubMed
.
- Y. Liu, H. Tan, Y. Wei, M. Liu, J. Hong, W. Gao, S. Zhao, S. Zhang and S. Guo, ACS Nano, 2023, 17, 5994–6001 CrossRef CAS PubMed
.
- W. Su, S. Zhong and Y. Fan, Appl. Catal., B, 2024, 354, 124145 CrossRef CAS
.
- M. C. Biesinger, L. W. Lau, A. R. Gerson and R. S. C. Smart, Appl. Surf. Sci., 2010, 257, 887–898 CrossRef CAS
.
- C. Peng, P. Wei, X. Li, Y. Liu, Y. Cao, H. Wang, H. Yu, F. Peng, L. Zhang, B. Zhang and K. Lv, Nano Energy, 2018, 53, 97–107 CrossRef CAS
.
- L. Bai, F. Franco, J. Timoshenko, C. Rettenmaier, F. Scholten, H. S. Jeon, A. Yoon, M. Rüscher, A. Herzog, F. T. Haase and S. Kühl, J. Am. Chem. Soc., 2024, 146, 9665–9678 CrossRef CAS PubMed
.
- Z. Yan, P. Gao, Z. Li, Y. Zhang, C. Hu, D. Cao and D. Cheng, Small, 2025, 21, 2500950 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |