Open Access Article
Open Access ArticleChain-length-selective adsorption governs diffusion-limited dendrite growth mode in battery electrodes
Aakriti
Aggarwal
a,
Wenrui
Chai
b,
Graeme
Henkelman
 b and
Kent J. X.
Zheng
b and
Kent J. X.
Zheng
 *acd
*acd
aDepartment of Chemical Engineering, University of Texas at Austin, Austin, Texas 78712, USA. E-mail: kentz@utexas.edu
bDepartment of Chemistry, University of Texas, Austin, Texas 78712, USA
cTexas Materials Institute, University of Texas at Austin, Austin, Texas 78712, USA
dBard Center for Electrochemistry, University of Texas at Austin, Austin, Texas 78712, USA
First published on 4th March 2026
Abstract
Growth of classical dendrites in metal anodes represents a far-from-equilibrium, diffusion-limited crystallization process that critically impacts the safety and performance of energy storage systems. Zn—a promising aqueous anode material—exhibits two distinct modes: continuous growth (CG), where a few crystallographically guided dendrites propagate rapidly, and independent nucleation (IN), where numerous smaller, randomly oriented dendrites form with slower propagation. Here we show that ethylene glycol-derived additives, HO–(CH2CH2O)n–H, can reliably shift growth from CG to IN. Using operando tools including optical visualization, electrochemical impedance spectroscopy, and quartz crystal microbalance, we resolve the kinetic and interfacial origins of this transition. The effect is highly chain-length dependent: PEG-400 (n ≈ 9) achieves the most efficient CG → IN shift with a threshold of 0.001 wt% (10 ppm), while shorter or longer chains require ∼1 wt%, and no transition occurs for n ≤ 3 at all. Experimental measurements and quantum-chemical analysis reveals that the transition arises from two coupled processes: adsorption of PEG molecules at the electrode surface, and coordination with Zn adatoms and Zn2+ cations, which destabilize continuous growth pathways and favor repeated independent nucleation. Importantly, the shift from CG to IN reduces dendrite propagation velocity by a factor of 6. These findings establish molecular-level design rules for engineering task-specific organic molecules to control far-from-equilibrium crystallization, with direct implications for high-power batteries and electrochemical synthesis.
Crystallization under far-from-equilibrium conditions, driven by steep gradients in temperature or concentration, remains a fundamental scientific challenge with broad industrial relevance.1–3 Such interfacial instabilities often give rise to dendritic growth, characterized by highly ramified, self-similar fractal geometries. In metallurgy, aggressive formation of coarse thermal dendrites are well known to degrade the mechanical properties of solidified alloys.4,5 In batteries, dendrites form under fast-charging conditions when rapid ion reduction (M(solution)n+ + ne− → M(solid)) depletes local ion concentration, establishing sharp gradients that trigger self-reinforcing propagation into the bulk electrolyte.6–8 This behavior is commonly rationalized by the relative rates of ion transport and electrochemical reaction: when the reaction kinetics (exchange current density, j0) greatly exceeds the transport-limits (jlim), the electrode surface experiences severe ion depletion and dendrite initiation.9,10 It is further revealed that the crystallographic anisotropy of the depositing metal strongly influences dendrite evolution.3,11,12 For high-anisotropy metals such as hexagonal close-packed (HCP) Zn, once a dendrite nucleates it propagates rapidly along lattice-preferred directions, producing highly localized filaments that accelerate short-circuit failure.11
Organic species have long been recognized as effective modulators of electrodeposition.13–15 Among them, ethylene-glycol-derived (EG) molecules—ranging from short oligomers to long-chain polymers—are widely reported to enhance the interfacial stability of metal deposition in aqueous electrolytes through surface adsorption.8,16–19 While surface adsorption is well established in the literature, the precise mechanisms by which it governs the electrodeposition process—particularly, in the mass-transport-limited dendritic regime—remain an open area of investigation; with few exceptions,8,20 most prior reports have focused on the influence of EG-derived molecules at electrodeposition rates well below the limiting current density, where classical dendritic growth is not expected. Several mechanisms have been proposed, including suppression of electrokinetics—as evidenced by significant reduction in j0—that slows ion consumption8,21,22 and modulation of viscoelastic properties that dampen electroconvection.23,24 This motivated our work to specifically probe the impact of trace organic species on electrodeposition in the diffusion-limited regime, opening new opportunities for enabling ultrafast charging. Our recent results reveal that PEG-400 can fundamentally alter the geometry of Zn dendrites formed under classical mass-diffusion-limited conditions.11 In the absence of PEG-400, Zn growth is dominated by a small number of colossal dendrites that, once nucleated, undergo continuous growth (Mode I: CG) along crystallographically preferred directions. This highly localized tip growth produces extremely rapid propagation rates (∼6 × 101 µm s−1) and is particularly detrimental to stability. By contrast, in the presence of 1 wt% PEG-400, growth shifts to independent nucleation (Mode II: IN), characterized by a large number of smaller dendrites forming without orientational correlation. This morphology remarkably slows the effective dendrite-front propagation speed, by a factor of 4. Together, these earlier observations highlight the complex role of EG-derived organics in aqueous electrodeposition and their profound influence on morphological evolution under far-from-equilibrium conditions.
Motivated by the decisive role of a small amount (1 wt%) of PEG-400 in shifting Zn dendrite growth from Mode I: CG to Mode II: IN, this work employs operando optical visualization, electrochemical impedance spectroscopy (EIS), quartz crystal microbalance (QCM), and nuclear magnetic resonance (NMR) to elucidate the governing mechanisms of ethylene-glycol-derived additives [HO–(CH2CH2O)n–H] in shifting the dendritic growth mode. Operando microscopy establishes the primary trends: the CG → IN transition is absent for short chains (n ≤ 3), begins to appear at n ≥ 4 with a concentration threshold Ct of ∼1 wt%, and reaches maximum efficiency at n ≈ 9 (PEG-400), where the threshold is as low as 0.001 wt%! For longer chains (n > 9), the transition remains possible but requires progressively higher concentrations. The threshold concentration (Ct) is defined as the highest measured polymer additive concentration at which independent nucleation (IN) growth mode is observed before transition to continuous growth (CG) mode upon dilution. QCM measurements reveal that adsorbed mass on the Zn surface increases monotonically with chain length, explaining why short oligomers cannot induce the transition. However, this trend cannot account for the reduced efficiency observed for very long chains. NMR measurements provide complementary insight, showing the largest salt-induced peak shift at n ≈ 9, indicative of the strongest PEG–Zn2+ interaction. Together, these results suggest that the growth-mode transition requires two conditions: (i) effective adsorption of the additive on the electrode and (ii) strong interaction with Zn species. This dual criterion explains the exceptional efficiency of PEG-400. Density functional theory (DFT) calculations further support this model, showing that adsorbed species stabilize surface adatoms, suggesting suppression of continuous tip growth and promoting independent nucleation. These findings demonstrate how molecular-level physicochemical interactions dictate macroscale morphological evolution under far-from-equilibrium conditions, offering design rules for task-specific molecular engineering in electrochemical deposition and energy storage.
Results and discussion
Operando mapping of critical transitions in dendrite growth modes
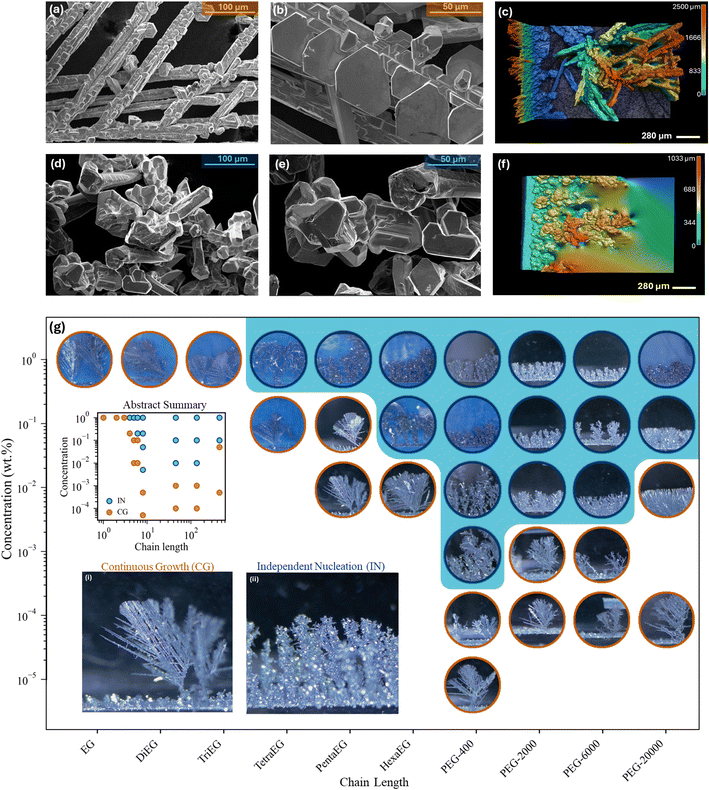
Fig. 1 presents the dendrite morphological modes in the presence and absence of EG-derived species in the electrolyte, characterized by operando optical visualization, SEM, and laser profilometry. Details of the experimental setup and electrochemical protocol are provided in Fig. S1 and S2.25 In concentrated ZnCl2 aqueous electrolyte without EG-derived species, dendritic growth is dominated by a small number of primary dendrites with parallel branches and correlated six-fold geometrical features, indicative of continuous growth along fast-propagating directions (e.g., [100]Zn) defined by the HCP lattice (Fig. 1a–c; Mode I: CG). By contrast, the addition of EG-derived species shifts the dendrite growth into a fundamentally different regime, producing a large number of independently nucleated Zn particles without orientational correlation (Fig. 1d–f; Mode II: IN). See Fig. S3 for detailed visualizations of the transition and Fig. S4 for a more quantitative definition of the CG and IN growth modes.Most importantly, operando visualization reveals a critical dependence of the CG → IN transition on both the chain length and concentration of EG-derived species (Fig. 1g). When the chain length n < 4, no CG → IN transition is observed even at concentrations as high as 20 wt% (Fig. S5), indicating that EG-derived species with n = 1, 2, or 3 have no effect on Zn dendrite growth mode. See also Video S1 for CG growth mode observed in 1 wt% EG. By contrast, when n ≥ 4, the CG → IN transition becomes apparent even at 1 wt% additive concentration. Notably, the lowest threshold concentration (Ct) required to induce the transition occurs at n ≈ 9 (PEG-400); only 0.001 wt% (10 ppm) is sufficient to drive the CG → IN transition! See Video S2 for operando visualization. For longer chains (n > 9), Ct increases, but the CG → IN transition remains robust in all situations at 1 wt%.
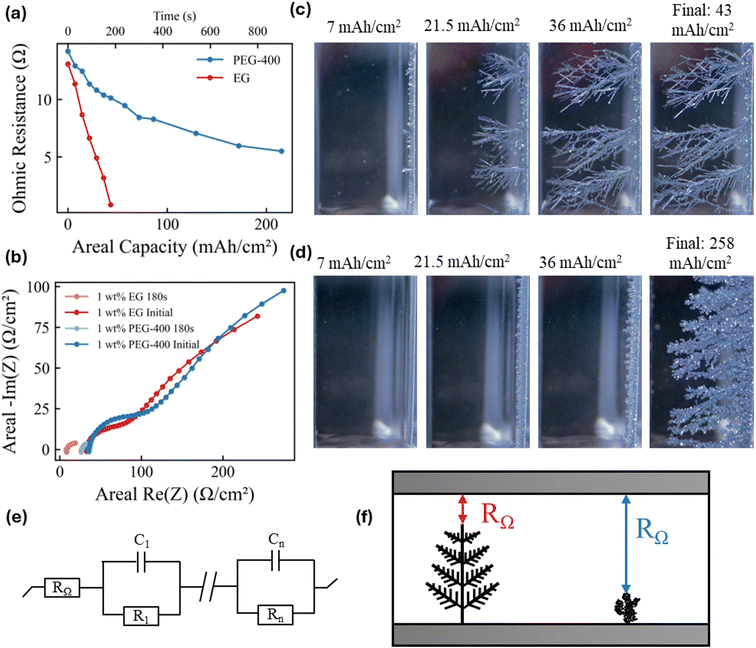
These two distinct dendrite growth modes lead to fundamentally different electrochemical behavior. As shown in Fig. 2a, the ohmic resistance RΩ decreases much more rapidly with increasing areal deposition capacity (Q) in Mode I: CG (with 1 wt% EG, n = 1) than in Mode II: IN (with 1 wt% PEG-400), as evidenced by the Nyquist plots from the EIS measurements (Fig. 2b and S8). Operando optical visualization of two growth modes (Fig. 2c and d) offers a clear explanation for this observed difference in the shape of the RΩ(Q) curve. The dendrite front propagation rate in Mode I: CG toward the counter electrode is 6 times higher than in Mode II: IN. This morphological evolution reduces the inter-electrode distance and therefore significantly lowers the measured system-level RΩ, eventually leading to complete shorting of the cell (Fig. 2e and f). The steeper the RΩ(Q) curve, the more efficiently the dendrite penetrates the inter-electrode gap (Fig. S9). In a given system, the slope dRΩ/dQ can thus be viewed as a quantitative measure of the dendrite's shorting tendency. To generalize this comparison across different cells, RΩ can further be normalized and expressed as the percent change in RΩ per unit areal capacity. This value in the Mode I: CG system (2.18) is 7 times larger than that in Mode II: IN (0.28). We attribute this stark contrast in the shape of RΩ(Q) to the fact that dendrites in Mode I: CG preferentially extend along a few fast-growing crystallographic directions, resulting in highly localized but concerted and therefore rapid propagation. Conversely, the numerous independent Zn crystals in Mode II: IN lack orientational correlation and thus propagate toward the counter electrode in a less concerted manner. Taken together, the results in Fig. 1 and 2 demonstrate that the CG → IN transition, which profoundly affects electrochemical behavior and cell stability, is strongly governed by the molecular-level chemistry of the EG-derived species. Mechanistic insights into how these species drive the transition will be essential for guiding the rational design of electrolytes for fast-charging battery systems.
Understanding interfacial dynamics driving dendrite mode transition
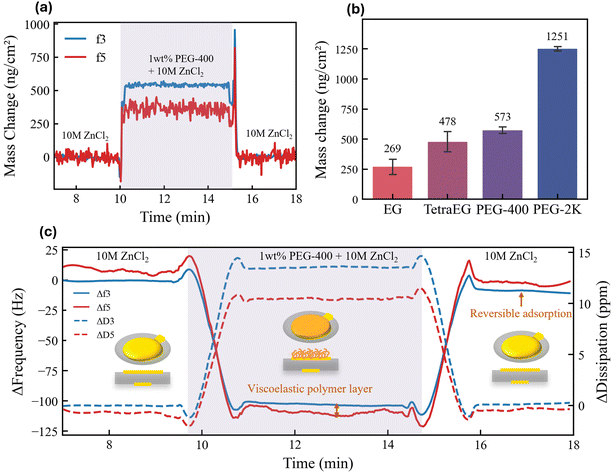
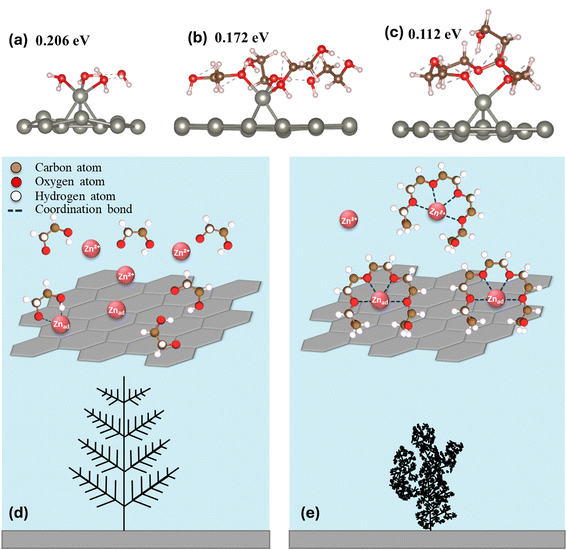
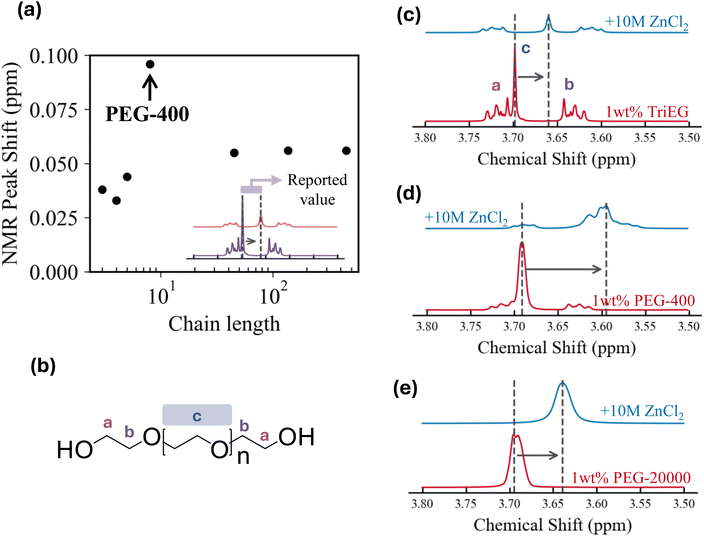
Motivated by the observation that this critical transition occurs even at ppm-level concentrations of PEG-400, we hypothesize that the phenomenon is highly interfacial in nature, rather than arising from changes in bulk electrolyte properties such as conductivity or solvation structure. This hypothesis is consistent with earlier studies showing that PEG species readily adsorb on metallic electrode surfaces such as Zn,26 Cu,27 and Au.28 The possibility of bulk electrolyte properties being responsible for the CG → IN transition was explored and eliminated as discussed in Fig. S11. QCM-D measurements were performed to gain quantitative insight into the surface adsorption of EG species (Fig. S12–S15). Fig. 3a shows the mass change of the electrode when a 1 wt% PEG-400 electrolyte is flowed into the cell.29 The return of mass change to zero after a baseline PEG-free electrolyte is re-introduced indicates that the adsorption is reversible. The difference in mass change between f3 and f5, along with a non-zero dissipation value indicates the adsorption layer is viscoelastic, refer to Note S1 for a further discussion regarding the analysis and limitations.30 A clear positive correlation is observed between polymer chain length and the mass adsorbed on the sensor surface (Fig. 3b). This means that longer-chain polymers adsorb more extensively on the electrode surface, occupying a greater interfacial volume (Fig. S16).31We now consider how adsorption can promote independent nucleation and drive the CG → IN transition. As a point of departure, the intrinsic continuous growth mode observed without any EG-derived species can be rationalized by classical nucleation theory. The formation of new crystals requires creating particles with high specific surface area, which imposes a higher kinetic barrier than simply extending pre-existing crystals. DFT calculations provide atomistic insight into the interaction between adsorbed EG-derived species and the growing Zn metal surface. The DFT calculations are intended to capture qualitative, comparative trends among water, ethylene glycol, and low-molecular weight PEGs, rather than to provide quantitatively accurate surface or binding energies. Fig. 4a–c shows the proposed interaction framework whereby water, ethylene glycol and PEG molecules all interact strongly with the Zn surfaces through their oxygen atom. The adsorbed molecules are likely to bind with Zn adatoms on the surface, stabilizing these otherwise highly active growth sites and thereby “passivating” the surfaces of existing crystals. This adatom passivation could make continuous growth less energetically favorable and shift the balance between continuous growth and independent nucleation, rendering the latter comparatively more favorable (Fig. 4d and e). The binding strength appears to increase with chain length, suggesting stronger adatom passivation and a greater tendency toward independent nucleation (Fig. S17). The absence of CG → IN transition in systems with n ≤ 3 can be understood in terms of weaker adatom passivation; when passivation is insufficient, continuous growth dominates over independent nucleation.
While we can now rationalize the absence of the CG → IN transition at short chain lengths, it is less straightforward why the threshold concentration Ct reaches its minimum at n ≈ 9 with an average molecular weight Mw of 400. QCM-D measurements indicate that large amounts of EG-derived species adsorb on the electrode surface. This apparent discrepancy suggests that the “efficiency” of promoting the CG → IN transition decreases when the polymer chains become too long. DFT simulations of long-chain molecules are technically challenging due to the complexity of chain conformations and the large number of atoms required in the unit cell. It is well established that polymer chains can exhibit entanglement,32 in which they overlap and interpenetrate in space once they exceed a critical chain length, defined by the entanglement molecular weight (Me). When chains entangle with one another, many repeat units are consumed in chain–chain overlap rather than chain–metal surface interaction, weakening the effective polymer–metal binding. Interestingly, the Me of PEG has been reported to be on the order of 103 g mol−1.33 We therefore theorize that the reduced efficiency of promoting CG → IN transition could arise from chain entanglement as the length increases.
1H-NMR measurements provide a complementary perspective on how adsorbed EG-derived species influence dendrite growth modes. We recorded the 1H-NMR spectra of 1 wt% EG-derived species dissolved in D2O, both before and after adding ZnCl2, to probe how interactions with ionic species alter the spectra (refer to Fig. S18 and S19 for alternative methods attempted). Notably, the magnitude of the peak shift reaches its maximum for PEG-400 (Fig. 5a) and decreases as the chain length increases. In particular, we tracked the 1H-NMR signal corresponding to the proton labeled “c,” which is bound to the backbone carbon atom (Fig. 5b). Representative spectra are shown on the right (Fig. 5c–e). The physical significance of the peak shift can be understood by considering its origin. A downfield (rightward) shift indicates increased shielding of the observed protons upon salt addition, which corresponds to an increase in local electron density.34 This is consistent with hydrated Zn2+ ions coordinating with the electron-donating ether oxygens of the polymer backbone. To maintain charge neutrality, chloride anions would also be associated with the polymer vicinity.35–37 A proposed molecular interaction scheme is illustrated in Fig. S20.38 Therefore, the trend of peak shifts shown in Fig. 5a suggests that the strongest electronic interaction between Zn2+ cations and the EG-derived species is achieved with PEG-400. It is however important to note that the Zn2+ cations discussed here are distinct from the charge-neutral Zn0 adatoms considered in the DFT calculations.
The role of the interaction between Zn2+ cations and EG-derived species can be speculated in the context of mass transport. The adsorbed polymer layer acts as a viscoelastic interphase covering the electrode surface. During electrodeposition, Zn2+ ions must migrate from the bulk electrolyte, across this interphase, to reach the crystal surface. Stronger Zn2+–polymer interactions make this process kinetically less favorable, as each cation must dissociate from one –(CH2CH2O)– site before moving to the next, thereby slowing ion transport through the layer. An analogous negative correlation between interaction strength—for example, as reflected by the binding energy of the diffusing species to the substrate or medium—and diffusivity has been reported in a variety of systems.39 When this interaction is maximized with PEG-400, ion transport across the adsorption layer to existing crystals is significantly hindered, making independent nucleation more favorable than continuous growth and rendering PEG-400 the most effective among EG-derived species in driving the CG → IN transition. This explanation is highly consistent with the trends observed in the QCM and NMR data; however, the presence of chain entanglement under the low polymer concentrations, highly saline conditions, and surface-confined environment used here cannot be experimentally confirmed and should therefore be regarded as a plausible but speculative mechanistic hypothesis. In summary, based on QCM and NMR results, the reduced efficiency of the CG → IN transition observed with longer chains could be attributed to possible chain entanglement and their diminished effectiveness as Zn2+ transport barriers to existing crystals, compared with PEG-400.
Understanding impact on cell cycling
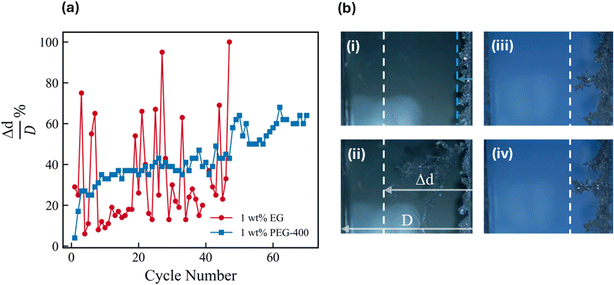
In order to understand the impact of growth morphology on cell cycling, the same operando cell set-up was used to study behavior over multiple plating and stripping cycles. The progression of the dendritic growth front towards the counter electrode was tracked over cycles as an additional metric to understand propensity of a growth morphology towards shorting behavior. Fig. 6a shows that growth Mode I: CG (1 wt% EG representative system) results in erratic and uneven dendrite growth, with the growth front advancing irregularly towards the counter electrode in consecutive cycles. This sporadic propagation suggests a high risk of an early short-circuit, as isolated dendrites can rapidly bridge the gap between electrodes.40 In this mode, a short could potentially occur after only a few cycles due to the unpredictable leaps of the dendritic front. In contrast to this, with the growth Mode II: IN (1 wt% PEG-400 representative system) dendrite growth progresses in a uniform and controlled manner, with the growth front advancing at a nearly linear rate over successive cycles. Consequently, the propensity for dendrites to reach the counter electrode unpredictably is greatly reduced, translating to a lower short-circuit risk. Therefore, despite the uniform potential signature during cycling shown in Fig. S21, the operando visualization as demonstrated by Fig. 6b reveals improved predictability and cycle life with growth Mode II over growth Mode I. The erratic, unstable dendrite propagation observed in Mode I points to the heightened vulnerability of such morphologies under fast-charging conditions, where local overpotentials and concentration gradients are exacerbated often leading to dendritic growth. These insights are particularly valuable for the design of aqueous Zn batteries, where dendrite formation remains one of the key bottlenecks limiting practical charging rates and energy densities.40Conclusion
The primary discovery of this work is that even trace amounts of EG-derived species (as low as 0.001 wt%, 10 ppm) can decisively govern the interfacial dynamics of diffusion-limited electrochemical crystallization. Adsorbed EG-derived species passivate Zn adatoms on existing crystals and create an energy barrier for Zn2+ transport through the viscoelastic interphase formed on the electrode surface. This interfacial dynamic suppresses continuous dendrite growth and instead promotes independent nucleation of new crystals with orientational correlation, driving the observed CG → IN transition. The transition is accompanied by a sixfold reduction in dendrite propagation rate, significantly improving electrochemical stability. We further show that the efficiency of this phenomenon is highly sensitive to polymer chain length, with PEG-400 (n ≈ 9) achieving the strongest effect. Reduced efficiency at shorter or longer chain lengths arises from weaker adsorption or chain entanglement, leaving continuous growth more favorable. These findings open new directions, including electrochemical QCM-D under applied bias to quantify adsorption and molecular dynamics simulations to probe transport across the viscoelastic layer. Overall, the reported interfacial adsorption–induced CG → IN transition offers important implications for molecular-level design of electrolyte components for fast-charging electrochemical energy storage systems.Author contributions
A. A. performed the electrochemical experiments and analyses. W. C. and G. H. conducted the DFT calculation. K. Z. directed the project.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included in the main text and supplementary information (SI) file. Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta09175f.Acknowledgements
The authors acknowledge financial support from the National Science Foundation (NSF) Materials Research Science and Engineering Center (MRSEC, DMR-2308817) and the University of Texas Science and Technology Acquisition and Retention (STARs) Fund. K. Z. gratefully acknowledges the membership of Allen J. Bard Center for Electrochemistry. The authors gratefully acknowledge the Allen J. Bard Center for Electrochemistry, University of Texas at Austin, for providing research support for this work through the Bard CEC Instrumentation Facility maintained under grant no. H-F-0037 from the Welch Foundation. The authors thank the Texas Materials Institute for assisting in the physical characterization of the samples in this work. The authors acknowledge the Texas Advanced Computing Center (TACC) at The University of Texas at Austin for providing computational resources that have contributed to the research results reported within this paper. The authors also thank Prof. Venkat Ganesan, Prof. Venkat R. Subramanian, and Prof. Charles Buddie Mullins for many useful discussions.References
- R. Trivedi and W. Kurz, Dendritic growth, Int. Mater. Rev., 1994, 39(2), 49–74 CrossRef CAS.
- W. Kurz and D. J. Fisher, Dendrite growth at the limit of stability: tip radius and spacing, Acta Metall., 1981, 29(1), 11–20 CrossRef CAS.
- D. Barkey, F. Oberholtzer and Q. Wu, Kinetic anisotropy and dendritic growth in electrochemical deposition, Phys. Rev. Lett., 1995, 75(16), 2980 CrossRef CAS PubMed.
- B. P. Reis, R. P. França, J. A. Spim, A. Garcia, E. M. Da Costa and C. A. Santos, The effects of dendritic arm spacing (as-cast) and aging time (solution heat-treated) of Al–Cu alloy on hardness, J. Alloys Compd., 2013, 549, 324–335 CrossRef CAS.
- W. R. Osorio, P. R. Goulart, A. Garcia, G. A. Santos and C. M. Neto, Effect of dendritic arm spacing on mechanical properties and corrosion resistance of Al 9 Wt Pct Si and Zn 27 Wt Pct Al alloys, Metall. Trans. A, 2006, 37(8), 2525–2538 CrossRef.
- J. Zheng, J. Yin, D. Zhang, G. Li, D. C. Bock, T. Tang, Q. Zhao, X. Liu, A. Warren and Y. Deng, Spontaneous and field-induced crystallographic reorientation of metal electrodeposits at battery anodes, Sci. Adv., 2020, 6(25), eabb1122 CrossRef CAS PubMed.
- P. Bai, J. Li, F. R. Brushett and M. Z. Bazant, Transition of lithium growth mechanisms in liquid electrolytes, Energy Environ. Sci., 2016, 9(10), 3221–3229 RSC.
- S. J. Banik and R. Akolkar, Suppressing dendrite growth during zinc electrodeposition by PEG-200 additive, J. Electrochem. Soc., 2013, 160(11), D519–D523 CrossRef CAS.
- D. A. Cogswell, Quantitative phase-field modeling of dendritic electrodeposition, Phys. Rev. E, 2015, 92(1), 011301 CrossRef PubMed.
- Y. Liu, X. Xu, M. Sadd, O. O. Kapitanova, V. A. Krivchenko, J. Ban, J. Wang, X. Jiao, Z. Song and J. Song, Insight into the critical role of exchange current density on electrodeposition behavior of lithium metal, Advanced Science, 2021, 8(5), 2003301 CrossRef CAS.
- W.-Y. Jao, D. Sheyfer, J.-S. Micha, A. Aggarwal, H. Higdon, C.-C. Hu and J. X. K. Zheng, Dendritic Growth in Battery Anodes: on the Hidden Role of Crystallographic Anisotropy at Propagating Electrochemical Interfaces, 2025 Search PubMed.
- W. Shao and G. Zangari, Dendritic growth and morphology selection in copper electrodeposition from acidic sulfate solutions containing chlorides, J. Phys. Chem. C, 2009, 113(23), 10097–10102 CrossRef CAS.
- M. A. Pasquale, L. M. Gassa and A. J. Arvia, Copper electrodeposition from an acidic plating bath containing accelerating and inhibiting organic additives, Electrochim. Acta, 2008, 53(20), 5891–5904 CrossRef CAS.
- J. L. Ortiz-Aparicio, Y. Meas, G. Trejo, R. Ortega, T. W. Chapman and E. Chainet, Effects of organic additives on zinc electrodeposition from alkaline electrolytes, J. Appl. Electrochem., 2013, 43(3), 289–300 CrossRef CAS.
- M. Paunovic and M. Schlesinger, Fundamentals of Electrochemical Deposition, John Wiley & Sons, 2006 Search PubMed.
- S. K. Kim, D. Josell and T. P. Moffat, Electrodeposition of Cu in the PEI-PEG-Cl-SPS additive system: reduction of overfill bump formation during superfilling, J. Electrochem. Soc., 2006, 153(9), C616 CrossRef CAS.
- M. Tan and J. N. Harb, Additive behavior during copper electrodeposition in solutions containing Cl−, PEG, and SPS, J. Electrochem. Soc., 2003, 150(6), C420 CrossRef CAS.
- M. Kang and A. A. Gewirth, Influence of additives on copper electrodeposition on physical vapor deposited (PVD) copper substrates, J. Electrochem. Soc., 2003, 150(6), C426 CrossRef CAS.
- J. J. Kelly, C. Tian and A. C. West, Leveling and microstructural effects of additives for copper electrodeposition, J. Electrochem. Soc., 1999, 146(7), 2540 CrossRef CAS.
- S. J. Banik and R. Akolkar, Suppressing dendritic growth during alkaline zinc electrodeposition using polyethylenimine additive, Electrochim. Acta, 2015, 179, 475–481 CrossRef CAS.
- K. R. Hebert, Role of chloride ions in suppression of copper electrodeposition by polyethylene glycol, J. Electrochem. Soc., 2005, 152(5), C283 CrossRef CAS.
- V. D. Jović and B. M. Jović, Copper electrodeposition from a copper acid baths in the presence of PEG and NaCl, J. Serb. Chem. Soc., 2001, 66(11–12), 935–952 CrossRef.
- S. Wei, Z. Cheng, P. Nath, M. D. Tikekar, G. Li and L. A. Archer, Stabilizing electrochemical interfaces in viscoelastic liquid electrolytes, Sci. Adv., 2018, 4(3), eaao6243 CrossRef PubMed.
- A. Warren, D. Zhang, S. Choudhury and L. A. Archer, Electrokinetics in Viscoelastic Liquid Electrolytes above the Diffusion Limit, Macromolecules, 2019 Search PubMed.
- Y. Jeon, Y. Wu, Y. Zhang, C. Hwang, H.-W. Lee, H.-K. Song and N. Liu, In situ visualization of zinc plating in gel polymer electrolyte, Electrochim. Acta, 2021, 391, 138877 CrossRef CAS.
- A. Mitha, A. Z. Yazdi, M. Ahmed and P. Chen, Surface adsorption of polyethylene glycol to suppress dendrite formation on zinc anodes in rechargeable aqueous batteries, ChemElectroChem, 2018, 5(17), 2409–2418 CrossRef CAS.
- G. S. Lindsay, D. Rohde, C. Wendeln, G. Vazhenin and A. A. Gewirth, Using SERS to Evaluate Additive Kinetics During Reverse Pulse Plating of Cu, J. Electrochem. Soc., 2025, 172(4), 042501 CrossRef CAS.
- M. L. Walker, L. J. Richter and T. P. Moffat, In situ ellipsometric study of PEG/Cl− coadsorption on Cu, Ag, and Au, J. Electrochem. Soc., 2005, 152(6), C403 CrossRef CAS.
- D. Johannsmann, A. Langhoff and C. Leppin, Studying soft interfaces with shear waves: Principles and applications of the quartz crystal microbalance (QCM), Sensors, 2021, 21(10), 3490 CrossRef CAS PubMed.
- A. D. Easley, T. Ma, C. I. Eneh, J. Yun, R. M. Thakur and J. L. Lutkenhaus, A practical guide to quartz crystal microbalance with dissipation monitoring of thin polymer films, J. Polym. Sci., 2022, 60(7), 1090–1107 CrossRef CAS.
- J. J. Kelly and A. C. West, Copper deposition in the presence of polyethylene glycol: I. Quartz crystal microbalance study, J. Electrochem. Soc., 1998, 145(10), 3472 CrossRef CAS.
- R. P. Wool, Polymer entanglements, Macromolecules, 1993, 26(7), 1564–1569 CrossRef CAS.
- S. Y. Kim, H. W. Meyer, K. Saalwächter and C. F. Zukoski, Polymer dynamics in PEG-silica nanocomposites: effects of polymer molecular weight, temperature and solvent dilution, Macromolecules, 2012, 45(10), 4225–4237 CrossRef CAS.
- H. Günther, NMR Spectroscopy: Basic Principles, Concepts and Applications in Chemistry, John Wiley & Sons, 2013 Search PubMed.
- G.-K. Liu, S. Zou, D. Josell, L. J. Richter and T. P. Moffat, SEIRAS study of chloride-mediated polyether adsorption on Cu, J. Phys. Chem. C, 2018, 122(38), 21933–21951 CrossRef CAS.
- Z. V. Feng, X. Li and A. A. Gewirth, Inhibition due to the interaction of polyethylene glycol, chloride, and copper in plating baths: a surface-enhanced Raman study, J. Phys. Chem. B, 2003, 107(35), 9415–9423 CrossRef CAS.
- Y. Wu, Z. Mao, T.-W. Jiang, X.-Y. Ma, L. Zheng, K. Jiang and W.-B. Cai, Revisiting the polyethylene glycol-chloride adsorption structure on Cu electrode in sulfuric acid solution by wide-frequency ATR-SEIRAS, J. Phys. Chem. C, 2023, 127(44), 21695–21703 CrossRef CAS.
- S. Jin, J. Yin, X. Gao, A. Sharma, P. Chen, S. Hong, Q. Zhao, J. Zheng, Y. Deng and Y. L. Joo, Production of fast-charge Zn-based aqueous batteries via interfacial adsorption of ion-oligomer complexes, Nat. Commun., 2022, 13(1), 2283 CrossRef CAS PubMed.
- C. D. Geary, I. Zudans, A. V. Goponenko, S. A. Asher and S. G. Weber, Electrochemical investigation of Pb2+ binding and transport through a polymerized crystalline colloidal array hydrogel containing benzo-18-crown-6, Anal. Chem., 2005, 77(1), 185–192 CrossRef CAS PubMed.
- Y.-y. Zuo, K.-l. Wang, P.-c. Pei, M.-h. Wei, X.-t. Liu, Y. Xiao and P.-f. Zhang, Zinc dendrite growth and inhibition strategies, Mater. Today Energy, 2021, 20, 100692 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |