Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Interfacial pathways in Na2B12H12/SiO2 nanocomposites boost the ionic conductivity and enable solid-state sodium batteries at room temperature
Jonas D. Hehn a,
Gabriele Masiaab,
Masoud Lazemi
a,
Gabriele Masiaab,
Masoud Lazemi a,
Juliette C. Verschoora,
Karan Kotalgia,
Savannah J. Turner
a,
Juliette C. Verschoora,
Karan Kotalgia,
Savannah J. Turner c,
Hlynur Gretarssond,
Martin Sundermann
c,
Hlynur Gretarssond,
Martin Sundermann de,
Fabrizio Murgiab,
Petra E. de Jongh
de,
Fabrizio Murgiab,
Petra E. de Jongh a and
Peter Ngene
a and
Peter Ngene *a
*a
aMaterials Chemistry and Catalysis, Debye Institute for Nanomaterials Science, Utrecht University, 3584 CG Utrecht, The Netherlands. E-mail: p.ngene@uu.nl
bDepartment of Chemical, Physical, Mathematical and Natural Sciences, University of Sassari, 07100 Sassari, Italy
cElectron Microscopy Centre, Utrecht University, 3584 CG Utrecht, The Netherlands
dDeutsches Elektronen-Synchrotron DESY, 22607 Hamburg, Germany
eMax Planck Institute for Chemical Physics of Solids, 01187 Dresden, Germany
First published on 11th February 2026
Abstract
All-solid-state sodium metal batteries are an attractive alternative to Li-ion batteries due to their high energy density, improved safety, and the lower price of sodium. A major bottleneck is the development of suitable solid electrolytes with sufficient ionic conductivity and electrochemical stability. Here, we report new all-solid-state sodium metal batteries based on sodium closo-dodecahydridoborate (Na2B12H12) electrolyte and both Prussian white (Na2MnFe(CN)6) and TiS2 cathode active materials. Although the pristine Na2B12H12 has a low ionic conductivity at room temperature, the conductivity increases by more than three orders of magnitude upon nanocomposite formation with mesoporous SiO2 via high-energy ball milling. The high ionic conductivity (5 × 10−4 S cm−1, 30 °C) and oxidative stability (3.9 V vs. Na+/Na) of Na2B12H12/SiO2 enable room-temperature battery operation with impressive capacity retention. Using a variety of techniques, including X-ray Raman scattering and electron energy loss spectroscopy, we show the presence of an interphase formed by an interface reaction between Na2B12H12 and SiO2. The observed interface effects are highly influenced by the morphology of the oxidic framework and the preparation conditions of the nanocomposite.
Introduction
Sustainable energy production relies on fluctuating factors such as sunshine or wind; therefore, reliable and secure solutions are essential for storing the generated electrical energy. The lithium-ion (Li-ion) battery technology is paving the way to address this global energy challenge.1 On the other hand, battery technologies based on geographically widespread, abundant and potentially cheaper elements offer attractive development prospects. Hence, sodium-ion (Na-ion) based battery chemistries are a promising alternative to Li-ion batteries.2,3 Although the high energy densities of state-of-the-art Li-ion batteries remain unchallenged, next-generation sodium batteries, such as those utilizing solid electrolytes (SEs), are a promising option. Certain SEs are compatible with metallic anodes, enabling capacities beyond the limits of commercially used anodes.4,5 Additionally, the replacement of the flammable liquid electrolytes with SEs would potentially improve the safety aspects of sodium batteries.5These promising features have led to enormous efforts in the scientific community to develop new SEs. Next to sodium superionic conductors (NASICON),6,7 β-alumina7,8 and sulfide-based7,9 solid-state Na-ion conductors, sodium hydridoborates have recently gained particular interest.10,11 Due to their good mechanical properties (softness) and superior reductive stability, excellent interface contact can be established with different classes of electrodes, including sodium metal anodes (Na metal).12 One example of this class of materials is Na2B12H12, which is non-flammable and has a low density (hence high energy content), due to its composition of only light elements.13–16 Following a temperature-induced polymorphic transition above 256 °C, ionic conductivities of ∼0.1 S cm−1 were reported.17,18 Approaches like nanostructuring17,19,20 or partial anion substitution21–23 aim to stabilize the high-temperature polymorph of these polyhedral-hydridoborate salts. Other strategies are the controlled oxidation24–26 or the confinement in oxidic scaffold structures,25,27,28 which leads to improved ionic conductivity potentially originating from the presence of boron oxygen bonds.
Building on these previous results, Su et al.25 recently demonstrated that the mechanochemical treatment of oxidized Na2B12H12 within a SiO2 scaffold further enhances its electrochemical properties, similar to previous findings on related lithium hydridoborates.29,30 This is interesting because various research groups24,28 have earlier reported that nanocomposite formation with SiO2 led to only a slight improvement in the ionic conductivity of sodium-based hydridoborates (e.g. NaBH4), unlike in Li2B12H12 and LiBH4, for which enhancements in Li-ion conductivity were observed.31–33 This raises the question whether the improved conductivity is primarily due to the partial oxidation of Na2B12H12 or true interface effects.
Thus, in this work, we investigate the ionic conductivity and electrochemical properties of non-oxidized Na2B12H12/SiO2 nanocomposites, and the influence of the oxide scaffold properties on the conductivity of the nanocomposite. We used ordered mesoporous and pyrogenic SiO2 with different surface-to-volume ratios to prepare Na2B12H12-based nanocomposites. Using a variety of characterization techniques, we show that interface reaction with SiO2 surface groups led to about four orders of magnitude increase in the Na-ion conductivity of the Na2B12H12. We show that both the morphology of the SiO2 and ball milling time have a profound impact on the overall ionic conductivity of the nanocomposite electrolytes. Furthermore, owing to the high stability window of the nanocomposite (3.9 V vs. Na+/Na), we prepared an all-solid-state battery (ASSB) using a Na metal anode, and a high-voltage Prussian white (PW) cathode. The ASSB shows impressive capacity retention (>95%) after 50 cycles at room temperature (RT). This demonstrates the efficacy of interface engineering as a tool to tune the ionic conductivity and electrochemical properties of SEs.
Experimental section
Synthesis
Na2B12H12 (>98%, KatChem) was dried at 200 °C for 16 h under reduced pressure (1 mbar) before use. Ordered mesoporous silica (SBA-15) or fumed SiO2 (Aerosil 90, 200, 300, 380 from Degussa, Evonik) with distinct different Brunauer–Emmett–Teller (BET) surface areas (Table 1) were dried for 16 h at 270 °C under dynamic vacuum (1 mbar) and subsequently combined with Na2B12H12. An overview of all sample compositions is provided in Table S1. All samples were handled and stored in an argon-filled glovebox (MBraun, Lab Star Glove Box, <1 ppm O2 and <1 ppm H2O). A Fritsch Pulverisette 7 ball mill with 20 mL tungsten carbide grinding bowls and 5 mm spheres was operated at 400 rpm to mechanochemically synthesize the nanocomposites in argon atmosphere. A discontinuous grinding process with 5-minute steps was performed, followed by a 5-minute break to prevent thermally induced decomposition. The ball-to-sample weight ratio was kept constant at 140![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and an absolute amount of 500 mg per batch was used.
1, and an absolute amount of 500 mg per batch was used.
| Pore volume (ml g−1) | SiO2 type | BET surface area (m2 g−1) | Equivalent primary particle size | |
|---|---|---|---|---|
| SiO2-A90 | 152 | Aerosil 90 | 70.2 | 85.5 nm |
| SiO2-A200 | 437 | Aerosil 200 | 191.2 | 31.4 nm |
| SiO2-A300 | 742 | Aerosil 300 | 271.5 | 22.1 nm |
| SiO2-A380 | 692 | Aerosil 380 | 349.2 | 17.2 nm |
| SiO2-SBA15 | 1207 | SBA-15 | 804.4 |
Structural characterization
Powder X-ray diffraction (XRD) patterns were recorded from 2θ = 10° to 60° under argon atmosphere in an airtight low background specimen holder on a Bruker D2 Phaser with Cobalt Kα1,2 radiation, λ = 1.79026 Å (30 kV and 10 mA). A TriStar II Plus gas-volumetric instrument (Micromeritics) was used for nitrogen physisorption measurements at 77 K. The BET surface area was calculated by approximating a BET isotherm to the dataset (0.05 < p/p0 < 0.25).34 Diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) samples were diluted with 97 wt% KBr (≥99%, Sigma-Aldrich), ground to a fine powder, and measured in a moisture-free chamber compatible with the Praying Mantis system (Harrick Scientific Products) installed in a Nicolet iS50 FT-IR instrument (Thermo Fisher Scientific). X-ray Raman scattering (XRS) measurements were carried out using an airtight sample holder, described elsewhere35 at beamline P01 of PETRA III, Deutsches Elektronen-Synchrotron (DESY), in Hamburg, Germany. Sample pellets were inserted into the holder and aligned at 18.3° with respect to the beam. Twelve spherically bent Si (220) analyzers centered around 2θ = 55° focus the scattered photons onto a Medipix-3 pixel detector with a resolution of 0.7 eV. In the given geometry, XRS operates basically at the dipole limit, and the resulting spectra will be analogous to the X-ray absorption spectra (XAS).30 The spectra were obtained by varying the incident beam energy while maintaining the analyzer energy fixed at about 9.7 keV. A Thermo Fisher Scientific Spectra 300 microscope was used for scanning transmission electron microscopy (STEM) at 300 kV combined with electron energy loss spectroscopy (EELS). Scanning electron microscopy (SEM) imaging was carried out on a Zeiss Evo 15 microscope operated at 15 kV. Elemental mapping was performed with energy dispersive X-ray spectroscopy (EDS).Electrochemical characterization
A Parstat MC multichannel potentiostat with PMC-1000 module (AMETEK) was used for the electrochemical investigation of the nanocomposites. Potentiostatic electrochemical impedance spectroscopy (EIS) was performed by applying 10 mV AC from 1 MHz to 1 Hz. A typical EIS cell was assembled by compacting 40–50 mg of SE at 187 MPa between two rods (A = 0.785 cm2), yielding pellets whose thickness (L = 200–400 µm) was obtained using a micrometer (Table S2).30 The cathode composite used in the PW-ASSB cells was made from Na2MnFe(CN)6 (MSE PRO, theoretical capacity 171 mAh g−1), SE and carbon (Ketjenblack EC600JD, AkzoNobel) in a 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (RT cell) or 7
1 (RT cell) or 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5
2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 (40 °C cell) weight ratio. The PW cathode composites (RT cell: 1.5 mg, 0.541 mAh cm−2; 40 °C cell: 1.2 mg, 0.505 mAh cm−2) were uniaxially pressed with 347 MPa onto either a 35 mg (RT) or a 45 mg (40 °C) SE pellet with a theoretical thickness of 0.80 and 1.03 mm, respectively. If TiS2 (99.9%, Sigma-Aldrich, theoretical capacity 239 mAh g−1) was used as the cathode active material, it was blended in a 7
0.5 (40 °C cell) weight ratio. The PW cathode composites (RT cell: 1.5 mg, 0.541 mAh cm−2; 40 °C cell: 1.2 mg, 0.505 mAh cm−2) were uniaxially pressed with 347 MPa onto either a 35 mg (RT) or a 45 mg (40 °C) SE pellet with a theoretical thickness of 0.80 and 1.03 mm, respectively. If TiS2 (99.9%, Sigma-Aldrich, theoretical capacity 239 mAh g−1) was used as the cathode active material, it was blended in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio with the SE. Subsequently, a freshly polished Na metal (99.9%, Sigma-Aldrich) disk and a copper current collector were added on the anode side. The three screws of the ASSB cells (A = 0.283 cm2) were tightened to a torque of 1.2 Nm, which corresponds to a stack pressure of 106 MPa. Before applying a current, the ASSB cells were allowed to rest and equilibrate to the corresponding temperature for at least three hours at open circuit voltage.
3 ratio with the SE. Subsequently, a freshly polished Na metal (99.9%, Sigma-Aldrich) disk and a copper current collector were added on the anode side. The three screws of the ASSB cells (A = 0.283 cm2) were tightened to a torque of 1.2 Nm, which corresponds to a stack pressure of 106 MPa. Before applying a current, the ASSB cells were allowed to rest and equilibrate to the corresponding temperature for at least three hours at open circuit voltage.
Results and discussion
Effect of preparation conditions and oxide properties on the nanocomposite conductivity
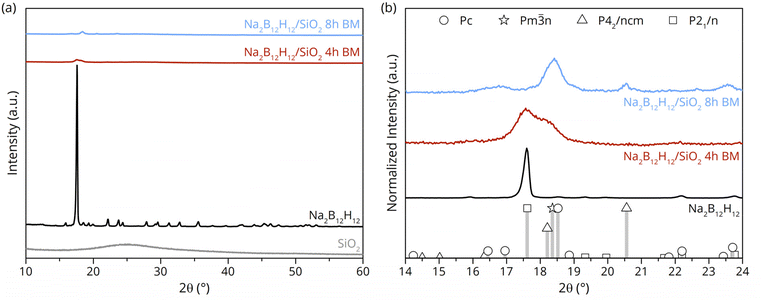
A series of Na2B12H12/SiO2 nanocomposites with different compositions and mechanochemical treatment (ball milling) durations was prepared using SiO2 scaffolds with different surface areas and morphologies in order to assess the effect of these parameters on the ionic conductivity. Fig. 1a compares the XRD patterns of the pristine Na2B12H12 and SiO2 with those of the nanocomposites ball milled for 4 h and 8 h. A clear change can be seen in the XRD pattern of the Na2B12H12-phase after the mechanochemical treatment with SiO2, whereby ball milling Na2B12H12 in the absence of silica only reduces the crystallite size (Fig. S1a). The broadening of the main diffraction feature of Na2B12H12 in the nanocomposite compared to the pristine material suggests that the mechanochemical treatment with varying SiO2 content results in nanostructuring and some degree of amorphization of Na2B12H12, as would be expected due to the mechanical impact (Fig. S1b).As seen from Fig. 1b, the normalized intensity diffractogram of the nanocomposite ball-milled for 4 h shows broadening of the main peak at 2θ = 17.6°. This is due to the co-presence of the low-temperature monoclinic and the high-temperature Na2B12H12 phases.17,36 After an extended treatment duration of up to 8 h, a complete phase transformation of the monoclinic Na2B12H12 phase is observed, as evidenced by the shift of the main diffraction peak at 2θ = 17.6° to the slightly higher angle of 2θ = 18.4° and the absence of a temperature-induced polymorphic transition (Fig. S2). These results demonstrate that mechanochemical treatment of Na2B12H12 with SiO2 leads to a loss of the long-range crystallinity and promotes the stabilization of the high-temperature Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) n phase at RT, alongside the phases Pc and P42/ncm.
n phase at RT, alongside the phases Pc and P42/ncm.
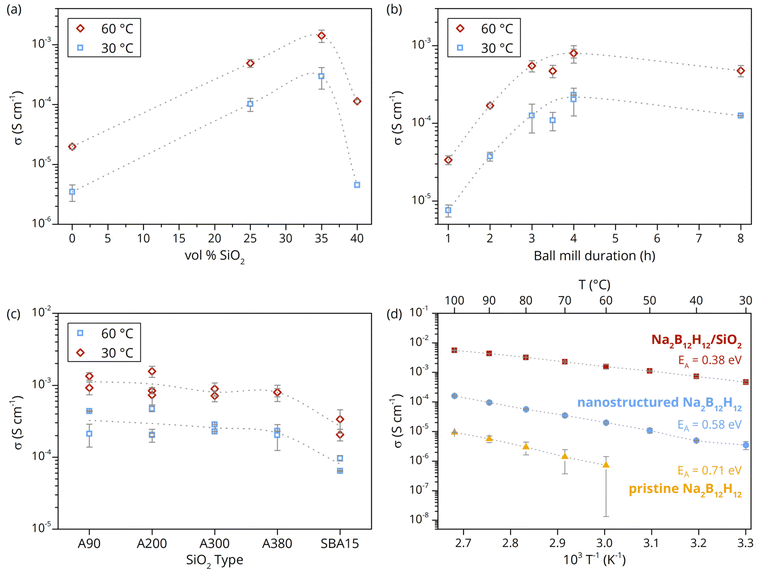
Fig. 2a compares the ionic conductivity of ball-milled Na2B12H12 to that of nanocomposites containing 25 to 40 vol% SiO2. The results show a profoundly higher ionic conductivity in the nanocomposites compared to that of nanostructured Na2B12H12. Interestingly, the increase in conductivity largely depends on the amount of SiO2, reaching a peak value (1.4 × 10−3 S cm−1 at 60 °C) at 35 vol% SiO2, after which the conductivity decreases with increasing SiO2 content. This demonstrates that nanocomposite formation with SiO2 increases the ionic conductivity of Na2B12H12 tremendously. Conversely, the decreased conductivity at high SiO2 content is attributed to the absence of good percolation pathways for a facile transport of the Na-ions between the insulating SiO2 particles.
Having established that nanocomposites made with 35 vol% SiO2 are ideal for ion transport between the silica particles, hence show maximum ionic conductivity, the effect of ball milling time was investigated. Fig. 2b shows the ionic conductivities at 60 °C of nanocomposites ball-milled for different durations. The conductivity increases with the duration of the mechanochemical treatment until 4 h, after which it decreases upon further milling. This is in line with the increasing proportion of the high-temperature phase with the milling time, as seen in Fig. 1. The slight decrease in conductivity after more than 4 h of ball milling, despite the increase in the concentration of the high-temperature Na2B12H12 phase, could be due to the severe distortion of the structure, as previously reported for NaCB11H12.19,20 This is also in line with the high degree of amorphization observed in the XRD pattern upon prolonged milling. Thus, while high defect concentrations improve ionic conductivity, too high defect concentrations are detrimental to the overall ionic motion.
Since the nanocomposites contain a significant proportion of SiO2, and the increased ionic conductivity is related to an interaction with SiO2, the influence of the SiO2 morphologies on the conductivity enhancement was investigated by using different types of silica (Table 1). For this purpose, Na2B12H12-based nanocomposites were prepared using either ordered mesoporous SiO2 (SBA-15) with a high surface area (800 m2 g−1) or various amorphous pyrogenic silicas with surface areas ranging from 70 to 350 m2 g−1. While the ionic conductivity of the nanocomposites with incorporated fumed silica showed only minor surface area dependency, the high surface area and different morphology of the ordered mesoporous silica seemed to have a negative impact (Fig. 2c). This is surprising because the ionic conductivity is expected to scale with the surface area of the nanocomposite, since the conductivity enhancement is related to interactions at the interface. This suggests that the interparticle pore volume between the nanosized primary fumed silica particles is advantageous for the mechanochemical synthesis approach, rather than the well-defined and narrow internal mesopores of SBA-15, which could be partly inaccessible, resulting in a higher void fraction and increased impedance. Indeed, SEM-EDS imaging and elemental mapping revealed a heterogeneous appearance independent of the silica type, whereby different sample domains were observed (Fig. S3 and S4). The microscopy images indicate that SiO2 aggregates are partly embedded in Na2B12H12, whereas bigger silica agglomerates are covered with Na2B12H12. The irregular mixing (heterogeneity) on the micrometer scale is consistent with the results of the conductivity measurements, which implies that the internal mesopores of SBA-15 cannot be completely filled via the mechanochemical nanocomposite preparation approach.
Based on the results discussed above, the use of 35 vol% SiO2-A200 and the 4 h mechanochemical treatment gives rise to nanocomposites with the best ionic conductivity. Fig. 2d illustrates the ionic conductivity of Na2B12H12 + 35 vol% SiO2-A200 at various temperatures compared to the pristine (or bulk-like) and mechanochemically processed Na2B12H12. The remarkable ∼3 orders of magnitude increased ionic conductivity in the nanocomposite is mostly due to the reduction of the activation energy for Na-ion hopping from 0.71 eV (pristine Na2B12H12) to 0.58 eV and 0.38 eV for the ball-milled (nanostructured) Na2B12H12 and Na2B12H12/SiO2 nanocomposite, respectively. It is also clear that only nanostructuring of Na2B12H12 already improves the ionic conductivity (1.9 × 10−5 S cm−1 at 60 °C) and reduces the activation energy for an ion-jump. Nevertheless, the presence of silica promotes the stabilization of the high-temperature polymorph, hence further facilitating the ionic motion and reducing the activation energy. This suggests that the addition of silica not only increases the concentration of the mobile ion carrier but also modifies the conduction mechanism, possibly originating from a chemically altered Na2B12H12 interphase compound that forms at the interface with the oxide-containing framework, as reported for Li2B12H12 and LiBH4.30,37 This will be discussed in detail in the next section.
Origin of the enhanced ionic conductivity in nanocomposites
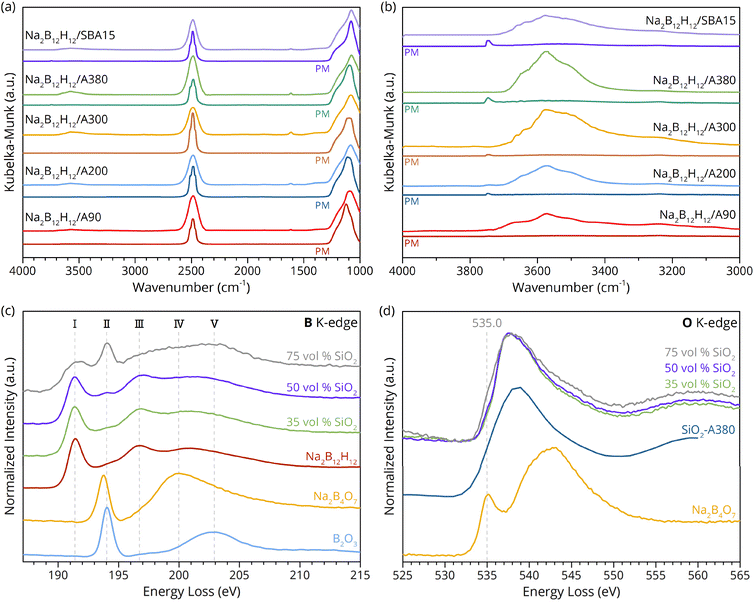
Vibrational spectroscopy is a good approach to investigate the origin of the enhanced ionic conductivity by probing the surface chemistry and the nature of bonds in the pristine compounds and the nanocomposites. Fig. 3 shows the diffuse reflectance infrared spectroscopy data of the investigated nanocomposites compared to their physical mixtures. The surface chemistry of a dried SiO2 framework is mainly dominated by absorption bands of isolated silanol groups (Si–OH) at 3750 cm−1 and features originating from inert siloxane groups (Si–O–Si) at low wavenumbers.38 The absence of the O–H bending mode at ∼1630 cm−1 and the overall weak intensity in the O–H vibration region (3300–3700 cm−1) in the infrared spectra of the physical mixtures before the mechanochemical treatment indicate effective water removal during the drying step of the SiO2, and a low degree of surface hydroxylation (Fig. 3a). The band assigned to free silanol groups is present in all physical mixtures, whereby the intensity scales with the surface area of the SiO2 scaffold, but vanishes in the associated nanocomposites. Conversely, several overlapping bands emerge in the O–H stretch vibration region of the nanocomposite spectra, and their intensities are proportional to the presence of free silanol groups in the corresponding physical mixtures (Fig. 3b). The observed disappearance of the silanol groups upon nanocomposite formation is consistent with previous studies.39,40 A plausible explanation would be that free silanol groups are reduced, i.e. to Si–H,41 or lose their freedom to move in the solid. In this process, the B12H122− anion could function as the reducing agent, hence being oxidized under the harsh synthesis conditions, and H2O may be formed, as suggested by the presence of the O–H bending and stretching modes.Further, all spectra in Fig. 3a contain the characteristic B–H vibration band of Na2B12H12 at ∼2500 cm−1.42 Noticeably, this peak is triple degenerate in the physical mixtures, whereas it has merged into a single broad feature for nanocomposites, indicating a distortion in the local chemical symmetry of the icosahedral boron cluster. Moreover, the shape of the asymmetric silanol bending mode at ∼1080 cm−1, slightly changed upon ball milling for all investigated samples. However, our results indicate that the quantity of free silanol groups prior to ball milling does not significantly affect the ionic conductivity; instead, the morphology of the framework plays a key role.
X-ray Raman scattering was employed to obtain further insight into the electronic structure of the nanocomposite and thus the conductivity enhancement mechanism. XRS experiments operated in the dipole limit allow for the investigation of light element K-edges, whereby results similar to XAS can be obtained. The advantage of XRS compared to XAS lies in its bulk sensitivity and the facilitated sample preparation, as the use of hard X-rays eliminates the need for measurements in high vacuum. Fig. 3c and d show the B and O K-edge spectra of Na2B12H12/SiO2 nanocomposites compared to reference compounds. The boron K-edge of the nanocomposite containing 35 vol% SiO2 appears similar to that of the reference Na2B12H12, since the overall signal is dominated by bulk-like Na2B12H12 far from the interface with the SiO2 (Fig. 3c). This is expected because the high fraction of Na2B12H12 in the 35 vol% SiO2 nanocomposite is essential to ensure a continuous Na-ion network in the SE, which is crucial for operation.
However, to exclusively probe the interphase layer a series of nanocomposites with higher SiO2 contents (50 and 75 vol%), and hence lower bulk like Na2B12H12, were investigated. Following this approach, only boron in close proximity to the oxidic scaffold material is probed, and changes can be unambiguously assigned to the interphase layer. Hence, the evolution of peak II, pronounced in the 50 and 75 vol% SiO2 nanocomposites, is attributed to a mechanochemically formed interphase layer. This evidence is consistent with previous reports, describing the formation of a tertiary compound in LiBH4 or Li2B12H12-based nanocomposites.29,35,43,44 Since a feature at 194 eV is characteristic for boron compounds with a trigonal planar geometry, such as found in Na2B4O7 or B2O3, the increasing peak intensity at 194 eV in the investigated nanocomposites implies similar moieties are formed during an interface reaction between Na2B12H12 and SiO2 (Fig. 3c).45–47
Furthermore, the plateau ranging from peak III to peak V in pristine Na2B12H12 flattens significantly in the nanocomposite samples, implying that the boron bonds of the interphase compound are still influenced by sodium, as can be seen in comparison to the reference spectra of Na2B4O7.
Moreover, examination of the oxygen K-edge indicated peak-shape variations depending on the oxidic scaffold fraction. When high SiO2 contents were used, an increasing influence of a feature at 535 eV is observed, which is associated with nonbridging oxygens (Fig. 3d).48 This suggests that nonbridging oxygen atoms react during the mechanochemical synthesis, which is in excellent agreement with the observed disappearance of the free silanol groups with DRIFTS.
Likewise, the Na K-edges were investigated and are given in Fig. S5. The edge-start of the Na2B12H12 reference has a peak maximum at 1073.8 eV with a shoulder located at 1073.2 eV. However, for the nanocomposites, the edge-start maximum clearly shifted towards a lower energy loss of 1073.4 eV. Moreover, the main-edge feature of the reference material at 1075.8 eV is becoming less pronounced in favor of a peak evolution at 1076.6 eV and 1080.5 eV. This suggests that the sodium ions are located in structurally disordered environments, such as in sodium silicate glasses, where Na-ions are present as only weakly bound network modifiers.49
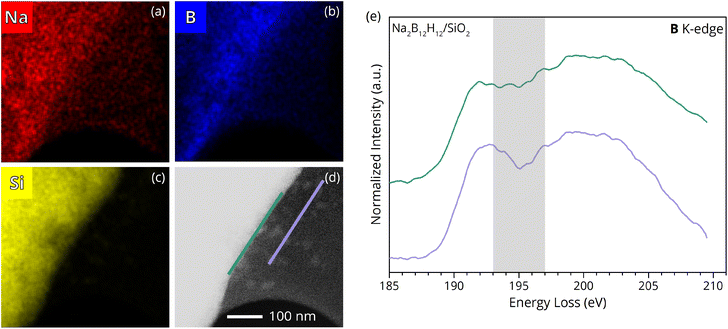
Since distinct changes in the boron, oxygen and sodium K-edge structure imply a chemical modification of B12H122− following the mechanochemical treatment, the elemental distribution at the Na2B12H12/SiO2 interface was mapped with STEM-EDS (Fig. 4a–d and S6). The obtained images illustrate nano proximity between the constituent compounds. However, a boron enrichment at the surface of the SiO2 agglomerate was observed, whereas sodium seems to be homogenously distributed over the SiO2 particle (Fig. S7). The uneven boron distribution suggests the formation of an interphase layer anchored on the surface of silica particles. This is in excellent agreement with the disappearance of the free silanol groups, which are mainly located at the surface of SiO2, pointing to a chemical reaction/interface formation with Na2B12H12, where B–O bonds are formed at the interface.
The boron electronic structure was further investigated utilizing the spatial resolution of STEM-EELS, which enables zooming in closer to the interface (Fig. 4e). Details about the EELS background removal are given in Fig. S8. The STEM-EELS investigation suggests a modification of the B12H122− anion in the nano proximity to the oxidic scaffold, which is consistent with the peak evolution at 194 eV observed with XRS measurements. However, the B K-edge structure in the vicinity of SiO2 particles shows minor deviations from the XRS spectra obtained for pristine Na2B12H12 (Fig. 3c), which can be attributed to plasmonic effects, the smaller sampling region, and the presence of small primary SiO2 particles visible as bright unfocused spots on the TEM grid in Fig. 4d.
Nevertheless, both the XRS and the STEM-EELS results reveal modifications of the electronic structure of the Na2B12H12 upon nanocomposite formation, whereby the B and Na (also O from the scaffold) are significantly altered at the interface. Together with the new vibrational modes and the disappearance of the oxide surface groups observed from the DRIFTS measurements, the existence of a boron-oxygen-rich, highly defective interphase at the Na2B12H12/SiO2 interface is highly likely.
Electrochemical properties and battery performance of the nanocomposites
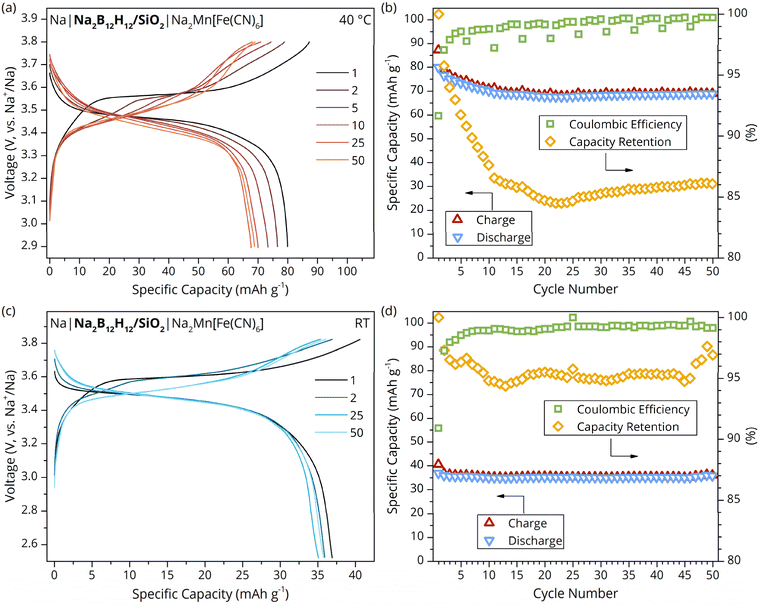
In view of the good ionic conductivity of the samples, the electrochemical properties and performance of the material as SE in batteries were investigated. The EIS and chronoamperometry results in Fig. S9 and S10 indicate that the measured conductivity is truly ionic in nature; hence, the nanocomposites might be suitable as SEs. Specifically, the Na-ion transference number was estimated in a symmetric cell configuration with non-blocking Na metal electrodes to be tNa+ = 0.97. The assigned value is in accordance with earlier reports,25 although the additional polarization impedance is less pronounced (Fig. S11). To evaluate the oxidative electrochemical stability limit and compatibility with the highly reducing Na metal anode, linear sweep voltammetry was conducted (Fig. S12). An oxidative event with an onset voltage of Eonset = 3.9 V vs. Na+/Na was found. Therefore, the oxidative stability limit is comparable to the value found for pristine Na2B12H12. EIS performed before and after the electrochemically induced oxidative event clearly illustrates an increase in impedance, suggesting a partial decomposition of the material (Fig. S13), possibly to larger BxHy clusters as has been recently suggested computationally.50 It is worth mentioning that the oxidative stability of Na2B12H12/SiO2 is enhanced compared to Li2B12H12-based nanocomposites, which builds on previous findings regarding related hydridoborates.29,30,51 The Na metal compatibility was tested in a symmetric cell configuration and exhibited stable polarization voltages for over 150 h, while alternating constant currents of 0.05 mA cm−2 were applied, which is in agreement with previous reports (Fig. S14).25 Furthermore, critical current density measurements, utilizing the various fumed silica scaffolds with high or lower surface area, showed a similar current density limit of 0.3 mA cm−2 (Fig. S15).Given the high oxidative stability and compatibility with Na metal, ASSBs based on Na2B12H12/SiO2 as SE were assembled in combination with a Na metal anode and PW (Na2Mn[Fe(CN)6]) or TiS2 as active cathode material. Cells were operated at different temperatures ranging from RT (∼20 °C) to 60 °C. The sodium manganese hexacyanoferrate cathode cell operated at 40 °C and C/10 showed nominal capacity fading during the first 10 cycles (Fig. 5a and b) before stabilizing at ∼85% with respect to the first discharge capacity. The coulombic efficiency gradually increased with the number of cycles, from an average of 98.7% for cycles 7–10 to 99.7% for cycles 46–50. EIS measurements were performed periodically (Fig. S16) resulting in reduced coulombic efficiency values in the subsequent cycle. In addition, a battery cell with TiS2 as cathode active material was operated at 60 °C, demonstrating the versatile suitability of the developed nanocomposite SE for a wide temperature range (Fig. S17).
The charge–discharge profile of the PW-ASSB operated at RT and C/20 is shown in Fig. 5c and d. It can be seen that the investigated Na2B12H12/SiO2 nanocomposite exhibits excellent properties as an SE, which is reflected in overall high coulombic efficiency values. Furthermore, a capacity retention of 96.9% was measured after 50 cycles, while fluctuations in room temperature are likely responsible for the recovery from 94.7% capacity retention after the 45th cycle. Nevertheless, the overall measured specific capacity is low compared to a PW-ASSB cell operated at 40 °C. The reduced capacity is attributed to increased internal impedance and lower electrotonic conductivity of PW in the cathode composite in a colder environment.52 Furthermore, an uneven distribution of electronically conductive carbon in the cathode composite mixture may have resulted in electronically isolated PW grains that consequently remain electrochemically inactive during battery cycling. While the cathode formulation requires further improvement to increase the charge/discharge capacity, this work clearly demonstrates the compatibility of Na2B12H12/SiO2 with Na metal anode and state-of-the-art high-voltage cathode materials (PW) at ambient conditions.
Conclusions
We have studied the effects of SiO2 on the ionic conductivity and electrochemical properties of Na2B12H12 in all-solid-state sodium batteries. Our work revealed that Na2B12H12/SiO2 nanocomposites exhibit more than 3 and 2 orders of magnitude higher ionic conductivity at room temperature compared to the pristine and ball-milled Na2B12H12, respectively. Structural and electronic characterization suggest that the enhanced ionic conductivity is related to interface reactions with surface groups of the SiO2 scaffold, leading to highly disordered Na2B12H12 with partially oxidized B12H122−. The morphology of the SiO2 scaffolds and preparation conditions for the nanocomposite were found to influence the ionic conductivity. Moreover, upon addition of SiO2, the electrochemical oxidative stability was retained at 3.9 V vs. Na+/Na. This results in a good cycling stability in combination with Na metal anode and both TiS2 and Prussian white cathodes between room temperature and 60 °C. Our work clearly demonstrates the efficacy of interface engineering in improving the ionic conductivity and electrochemical properties of sodium-based solid electrolytes, and their appealing properties for application in high-capacity all-solid-state sodium batteries.Author contributions
J. D. H. and P. N. conceived the experiments. J. D. H. and G. M. synthesized the samples, carried out electrochemical and structural measurements, and analyzed the data. J. D. H., M. L., J. C. V., M. S. and H. G. performed the XRS measurements. S. J. T. carried out the TEM-EELS measurements, and K. K. acquired the SEM images. J. D. H. and P. N. wrote the original manuscript. F. M., P. E. d. J. and P. N. supervised the research and reviewed the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data underlying this study are available at the Zenodo repository at: https://doi.org/10.5281/zenodo.17378151.Supplementary information (SI): additional structural, electronic, and electrochemical characterizations. See DOI: https://doi.org/10.1039/d5ta09128d.
Acknowledgements
Ramon van Maanen, Jan Willem de Rijk and Dennie Wezendonk are thanked for their technical assistance. The authors acknowledge the financial support from the Dutch Research Council (NWO). This publication is part of the ‘BatteryNL – Next Generation Batteries based on Understanding Materials Interfaces’ project (with project number NWA.1389.20.089) of the NWA research programme ‘Research on Routes by Consortia (ORC)’ funded by NWO. We acknowledge DESY (Hamburg, Germany), a member of the Helmholtz Association HGF, for the provision of experimental facilities and beamline P01, PETRA III. Beamtime was allocated for proposal I-20241256. PN and ML acknowledge funding from the European Union under the ERC Grant agreement no. 101171583 – Interfacial Ionics. For access to the TFS Spectra300 microscope at EM Centre Utrecht, we acknowledge the Netherlands Electron Microscopy Infrastructure (NEMI), project number 184.034.014, part of the National Roadmap and financed by NWO. GM and FM acknowledge funding from the European Union under the MSCA-IF Grant agreement no. 101069033 – STREAM and Next Generation EU, and the program DM 737/2021 fundings 2021–2022. Views and opinions expressed are, however, those of the author(s) only and do not necessarily reflect those of the European Union or the European Research Council Executive Agency (granting authority). Neither the European Union nor the granting authority can be held responsible for them.References
- D. M. Davies, M. G. Verde, O. Mnyshenko, Y. R. Chen, R. Rajeev, Y. S. Meng and G. Elliott, Nat. Energy, 2019, 4, 42–50 CrossRef.
- F. Duffner, N. Kronemeyer, J. Tübke, J. Leker, M. Winter and R. Schmuch, Nat. Energy, 2021, 6, 123–134 CrossRef CAS.
- P. K. Nayak, L. Yang, W. Brehm and P. Adelhelm, Angew. Chem., Int. Ed., 2018, 57, 102–120 CrossRef CAS PubMed.
- K. M. Abraham, ACS Energy Lett., 2020, 5, 3544–3547 CrossRef CAS.
- C. Bai, Y. Li, G. Xiao, J. Chen, S. Tan, P. Shi, T. Hou, M. Liu, Y.-B. He and F. Kang, Chem. Rev., 2025, 125, 6541–6608 CrossRef CAS PubMed.
- S. Chen, C. Wu, L. Shen, C. Zhu, Y. Huang, K. Xi, J. Maier and Y. Yu, Adv. Mater., 2017, 29, 1700431 Search PubMed.
- C. Zhao, L. Liu, X. Qi, Y. Lu, F. Wu, J. Zhao, Y. Yu, Y.-S. Hu and L. Chen, Adv. Energy Mater., 2018, 8, 1703012 Search PubMed.
- Y. Yung-Fang Yu and J. T. Kummer, J. Inorg. Nucl. Chem., 1967, 29, 2453–2475 CrossRef.
- A. Hayashi, K. Noi, A. Sakuda and M. Tatsumisago, Nat. Commun., 2012, 3, 856 CrossRef PubMed.
- Z. Lu, J.-X. Kang, P. Qiu and X. Chen, Batter. Supercaps, 2025, 8, e202400636 CrossRef CAS.
- M. Jin, D. Xu, Z. Su, R. Liu, M. Xiang, Y. Cheng, R. Wu and Y. Guo, Adv. Mater., 2025, e07809 CrossRef PubMed.
- F. Cuevas, M. B. Amdisen, M. Baricco, C. E. Buckley, Y. W. Cho, P. de Jongh, L. M. de Kort, J. B. Grinderslev, V. Gulino, B. C. Hauback, M. Heere, T. Humphries, T. R. Jensen, S. Kim, K. Kisu, Y.-S. Lee, H.-W. Li, R. Mohtadi, K. T. Møller, P. Ngene, D. Noréus, S.-I. Orimo, M. Paskevicius, M. Polanski, S. Sartori, L. N. Skov, M. H. Sørby, B. C. Wood, V. A. Yartys, M. Zhu and M. Latroche, Prog. Energy, 2022, 4, 032001 CrossRef CAS.
- J. Janek and W. G. Zeier, Nat. Energy, 2023, 8, 230–240 Search PubMed.
- L. M. de Kort, V. Gulino, P. E. de Jongh and P. Ngene, J. Alloys Compd., 2022, 901, 163474 Search PubMed.
- Q. Liu, X. Zhao, Q. Yang, L. Hou, D. Mu, G. Tan, L. Li, R. Chen and F. Wu, Adv. Mater. Technol., 2023, 8, 2200822 CrossRef CAS.
- T.-T. Le, M. Abbas, D. M. Dreistadt, T. Klassen and C. Pistidda, Chem. Eng. J., 2023, 473, 145315 Search PubMed.
- W. S. Tang, M. Matsuo, H. Wu, V. Stavila, A. Unemoto, S.-I. Orimo and T. J. Udovic, Energy Storage Mater., 2016, 4, 79–83 Search PubMed.
- T. J. Udovic, M. Matsuo, A. Unemoto, N. Verdal, V. Stavila, A. V. Skripov, J. J. Rush, H. Takamura and S.-I. Orimo, Chem. Commun., 2014, 50, 3750–3752 RSC.
- V. Gulino, A. Longo, L. M. de Kort, H. P. Rodenburg, F. Murgia, M. Brighi, R. Černý, C. J. Sahle, M. Sundermann, H. Gretarsson, F. de Groot and P. Ngene, Small Methods, 2024, 8, 2300833 CrossRef CAS PubMed.
- F. Murgia, M. Brighi, L. Piveteau, C. E. Avalos, V. Gulino, M. C. Nierstenhöfer, P. Ngene, P. de Jongh and R. Černý, ACS Appl. Mater. Interfaces, 2021, 13, 61346–61356 CrossRef CAS PubMed.
- M. Brighi, F. Murgia, Z. Łodziana, P. Schouwink, A. Wołczyk and R. Cerny, J. Power Sources, 2018, 404, 7–12 CrossRef CAS.
- M. Brighi, F. Murgia and R. Černý, Cell Rep. Phys. Sci., 2020, 1, 100217 CrossRef CAS.
- K. Yoshida, T. Sato, A. Unemoto, M. Matsuo, T. Ikeshoji, T. J. Udovic and S.-I. Orimo, Appl. Phys. Lett., 2017, 110, 103901 CrossRef.
- X. Luo, A. Rawal and K.-F. Aguey-Zinsou, Inorganics, 2021, 9, 2 CrossRef CAS.
- Z. Su, M. Jin, C. Hu, R. Liu, R. Wu, D. Xu and Y. Guo, ACS Appl. Energy Mater., 2025, 8, 5656–5663 CrossRef CAS.
- X. Luo, A. Rawal and K.-F. Aguey-Zinsou, J. Phys. Chem. C, 2024, 128, 14861–14870 CrossRef CAS.
- M. S. Andersson, V. Stavila, A. V. Skripov, M. Dimitrievska, M. T. Psurek, J. B. Leão, O. A. Babanova, R. V. Skoryunov, A. V. Soloninin, M. Karlsson and T. J. Udovic, J. Phys. Chem. C, 2021, 125, 16689–16699 CrossRef CAS PubMed.
- Y. Dou, H. A. Hansen, S.-M. Xu and D. Blanchard, Mater. Chem. Front., 2021, 5, 4989–4996 RSC.
- C. Zhou, H. Sun, Q. Wang, J. B. Grinderslev, D. Liu, Y. Yan and T. R. Jensen, J. Alloys Compd., 2023, 938, 168689 CrossRef CAS.
- J. D. Hehn, H. P. Rodenburg, M. Lazemi, J. C. Verschoor, M. Perxés Perich, M. Sundermann, H. Gretarsson, J. E. S. van der Hoeven, F. M. F. de Groot, P. E. de Jongh and P. Ngene, ACS Appl. Mater. Interfaces, 2025, 17, 33824–33833 CrossRef CAS PubMed.
- S. Das, P. Ngene, P. Norby, T. Vegge, P. E. de Jongh and D. Blanchard, J. Electrochem. Soc., 2016, 163, A2029 CrossRef CAS.
- M. H. W. Verkuijlen, P. Ngene, D. W. de Kort, C. Barré, A. Nale, E. R. H. van Eck, P. J. M. van Bentum, P. E. de Jongh and A. P. M. Kentgens, J. Phys. Chem. C, 2012, 116, 22169–22178 CrossRef CAS.
- L. M. de Kort, O. E. Brandt Corstius, V. Gulino, A. Gurinov, M. Baldus and P. Ngene, Adv. Funct. Mater., 2023, 33, 2209122 CrossRef CAS.
- S. Brunauer, P. H. Emmett and E. Teller, J. Am. Chem. Soc., 1938, 60, 309–319 CrossRef CAS.
- L. M. de Kort, M. Lazemi, A. Longo, V. Gulino, H. P. Rodenburg, D. Blanchard, C. Sahle, M. Sundermann, H. Gretarsson, A. M. J. van der Eerden, H. Elnaggar, F. M. F. de Groot and P. Ngene, Adv. Energy Mater., 2024, 14, 2303381 CrossRef CAS.
- N. Verdal, J.-H. Her, V. Stavila, A. V. Soloninin, O. A. Babanova, A. V. Skripov, T. J. Udovic and J. J. Rush, J. Solid State Chem., 2014, 212, 81–91 CrossRef CAS.
- V. Gulino, L. Barberis, P. Ngene, M. Baricco and P. E. de Jongh, ACS Appl. Energy Mater., 2020, 3, 4941–4948 CrossRef CAS.
- L. T. Zhuravlev, Colloids Surf., A, 2000, 173, 1–38 Search PubMed.
- P. Ngene, S. F. H. Lambregts, D. Blanchard, T. Vegge, M. Sharma, H. Hagemann and P. E. de Jongh, Phys. Chem. Chem. Phys., 2019, 21, 22456–22466 Search PubMed.
- L. M. de Kort, J. Harmel, P. E. de Jongh and P. Ngene, J. Mater. Chem. A, 2020, 8, 20687–20697 Search PubMed.
- S. F. H. Lambregts, L. M. de Kort, F. Winkelmann, M. Felderhoff, P. Ngene, E. R. H. van Eck and A. P. M. Kentgens, J. Phys. Chem. C, 2024, 128, 12186–12193 CrossRef CAS PubMed.
- E. L. Muetterties, R. E. Merrifield, H. C. Miller, W. H. Knoth and J. R. Downing, J. Am. Chem. Soc., 1962, 84, 2506–2508 CrossRef CAS.
- P. S. Miedema, P. Ngene, A. M. J. van der Eerden, D. Sokaras, T.-C. Weng, D. Nordlund, Y. S. Au and F. M. F. de Groot, Phys. Chem. Chem. Phys., 2014, 16, 22651–22658 RSC.
- Y. S. Choi, Y.-S. Lee, D.-J. Choi, K. H. Chae, K. H. Oh and Y. W. Cho, J. Phys. Chem. C, 2017, 121, 26209–26215 Search PubMed.
- M. E. Fleet and S. Muthupari, J. Non-Cryst. Solids, 1999, 255, 233–241 CrossRef CAS.
- M. E. Fleet and X. Liu, Phys. Chem. Miner., 2001, 28, 421–427 CrossRef CAS.
- G. S. Henderson, F. M. F. de Groot and B. J. A. Moulton, Rev. Mineral. Geochem., 2014, 78, 75–138 CrossRef CAS.
- F. Frati, M. O. J. Y. Hunault and F. M. F. de Groot, Chem. Rev., 2020, 120, 4056–4110 CrossRef CAS PubMed.
- S. De Wispelaere, D. Cabaret, C. Levelut, S. Rossano, A.-M. Flank, P. Parent and F. Farges, Chem. Geol., 2004, 213, 63–70 CrossRef CAS.
- R. Asakura, Z. Łodziana, R. Grissa, D. Rentsch, C. Battaglia and A. Remhof, ACS Appl. Energy Mater., 2025, 8, 9637–9645 CrossRef CAS.
- O. Tutusaus, H. Kuwata, M. J. Counihan and R. Mohtadi, Chem. Commun., 2023, 59, 4746–4749 RSC.
- R. Sun and Y. You, ACS Appl. Mater. Interfaces, 2023, 15, 44599–44606 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |