Ce–La ratio-controlled structural transitions in Ni/Ce–La oxide catalysts for direct decomposition of methane
Jae-Rang
Youn
a,
Min-Jae
Kim
ab,
Ki Cheol
Kim
ac,
Mincheol
Kim
a,
Taesung
Jung
a,
Kang-Seok
Go
ad,
Sang Goo
Jeon
a,
Jinghua
Guo
 b and
Woohyun
Kim
b and
Woohyun
Kim
 *a
*a
aKorea Institute of Energy Research, 152 Gajeong-ro, Yuseong-gu, Daejeon 34129, Republic of Korea. E-mail: wkim@kier.re.kr; Fax: +82-42-860-3739; Tel: +82-42-860-3421
bAdvanced Light Source, Lawrence Berkeley National Laboratory, Berkeley, CA 94720, USA
cGraduate School of Energy Science and Technology, Chungnam National University, 99 Daehak-ro, Yuseong-gu, Daejeon 34134, Republic of Korea
dKorea National University of Science and Technology, 217 Gajeong-ro, Yuseong-gu, Daejeon 34113, Republic of Korea
First published on 20th February 2026
Abstract
Ce–La mixed oxides represent widely applicable catalyst supports owing to their controllable crystal structures, oxygen storage capacity, and electronic properties. In this study, Ni/Ce1−xLax catalysts were systematically synthesized to investigate how Ce–La composition determines the lattice structure, redox behavior, and the resulting catalytic performance. Structural analyses by XRD and H2-TPR revealed that moderate La incorporation (x ≤ 0.3) expands the CeO2 fluorite lattice and enhances oxygen vacancy formation, thereby stabilizing highly dispersed Ni0 nanoparticles. XPS and soft XAS confirmed that this composition maximizes the Ce3+ fraction and optimizes the electronic interaction between Ni and the support. By contrast, excessive La content (x ≤ 0.5) induces a structural transition toward perovskite phases (LaNiO3 and La2NiO4) and secondary La-based oxides (La2O2CO3 and La(OH)3), which embed Ni within the lattice and reduce its accessibility as active sites. These structural differences critically influence the quality of carbon products formed via methane decomposition. The optimally doped Ni/Ce0.7La0.3 catalyst exhibited remarkable performance, producing CO2-free hydrogen together with highly crystalline CNTs featuring narrow diameters, high graphitization, and strong π* transitions.
1. Introduction
Supported metal catalysts have attracted considerable attention due to their unique catalytic properties in heterogeneous reactions. While extensive research studies have focused on the dispersion, oxidation state, and stability of the active metal species, equal emphasis has been placed on the characteristics of the support itself.1,2 Such studies demonstrate that the types of support materials, the incorporation of multiple cations, and even the morphology of the support strongly influence catalytic activity, selectivity, and stability.3 These findings collectively indicate that the support does not play a passive role; rather it actively governs metal–support interactions (MSI), redox behavior, and the stabilization of active sites. Moreover, the support often serves as storage for lattice oxygen and a source of defect sites that participate in redox cycling and reactant activation, influencing intrinsic performance and long-term stability of the catalysts.4,5 Accordingly, they can dramatically change the adsorption–desorption behavior of key intermediates, thereby determining the dominant reaction pathway. In other words, the support does not merely help the active metal; it actively tailors the whole catalyst system, participating in catalytic reactions and modulating both activity and selectivity together with the active metal species.Amongst various lanthanide oxides, ceria (CeO2) has been considered as one of the most promising and effective support materials in supported catalysts, owing to its ability to enhance metal dispersion, promote redox cycling, and strengthen metal–support interactions.6 Based on these attributes, CeO2-supported catalysts have been widely applied in thermo-catalytic reactions such as steam reforming, dry reforming of methane, the water-gas shift reaction, and selective catalytic reduction.6–8 To further improve oxygen ion conductivity and facilitate rapid oxygen transport, CeO2 has frequently been combined with trivalent lanthanide dopants, such as gadolinium (Gd), samarium (Sm), and lanthanum (La).9–11 Despite forming a trivalent oxide, lanthanum is known to exhibit high solubility (up to ca. 50 mol%) in the fluorite lattice of ceria. Ce–La mixed oxides can therefore retain a disordered fluorite phase or undergo structural transitions toward perovskite-like arrangements depending on the Ce/La ratio.12 Such transformations are not merely compositional. They constitute structural engineering strategies that modify lattice constants, oxygen vacancy concentrations, and MSI at a fundamental level. Nevertheless, the mechanistic role of Ce–La structural transitions in governing physicochemical properties of active metals remains insufficiently understood.
Catalytic decomposition of methane (CDM) provides a unique pathway for the co-production of CO2-free hydrogen and graphitic carbon nanomaterials such as carbon nanotubes (CNTs). Unlike non-catalytic thermal decomposition, which requires extremely high temperatures and typically yields low-quality carbon, the catalytic route enables the reaction to proceed under milder conditions with enhanced selectivity toward crystalline carbon structures.13–15 A primary requirement for catalysts is to maintain stable activity under harsh operating conditions: elevated reaction temperature and continuous carbon deposition on the catalyst surface.16 These factors often lead to particle sintering, encapsulation by carbon layers, and ultimately rapid deactivation of conventional catalysts. To address these challenges, Ni has been the most widely employed active metal because of its high intrinsic activity for C–H bond cleavage, favorable selectivity toward CNT growth, and cost-effectiveness compared to noble metals.13,15 However, the performance of Ni catalysts critically depends on the characteristics of the support to stabilize Ni nanoparticles and tune their electronic structure. The support not only influences metal dispersion and reducibility but also determines the extent to which carbon deposition can be mitigated during long-term operation.4 In this context, lanthanide oxides such as CeO2 and La2O3 have attracted significant attention. When employed as supports or dopants, these oxides exhibit enhanced redox properties, facilitate the generation and migration of oxygen vacancies, and strengthen metal–support interactions.17 These features are particularly promising for improving the stability and performance of Ni-based catalysts, as demonstrated in this study. Thus, employing Ce–La-based mixed oxides as catalyst supports represents a promising approach for the efficient co-production of hydrogen and CNTs via the methane decomposition reaction. A series of Ce–La mixed oxides with varying Ce/La ratios were synthesized by a co-precipitation method, followed by the addition of Ni through the incipient wetness impregnation technique. The catalyst samples were characterized by a suite of X-ray and electron-microscopy techniques, and their catalytic performance was evaluated to establish an integrated, multidimensional correlation between catalytic methane decomposition (CMD) and the physicochemical and structural properties of the supports.
2. Experimental
2.1 Catalyst preparation
In this study, a Ce1−xLax support material was synthesized using a co-precipitation method. First, cerium and lanthanum nitrate hexahydrates were dissolved in distilled water (2.0 L). The weight ratios of Ce to La were adjusted to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 0.9
0, 0.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, 0.7
0.1, 0.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.3, 0.5
0.3, 0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, 0.3
0.5, 0.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.7, 0.1
0.7, 0.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.9, and 0
0.9, and 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. A precipitating solution containing 15 wt% potassium carbonate was added dropwise until the pH reached 10.1. The resulting slurry was aged at room temperature for 4 h, and the precipitate was thoroughly washed several times to remove any impurities. After washing, it was dried at 110 °C for 24 h and calcined in air at 600 °C for 4 h.
1. A precipitating solution containing 15 wt% potassium carbonate was added dropwise until the pH reached 10.1. The resulting slurry was aged at room temperature for 4 h, and the precipitate was thoroughly washed several times to remove any impurities. After washing, it was dried at 110 °C for 24 h and calcined in air at 600 °C for 4 h.
Ni, using nickel nitrate hexahydrate as a precursor, was loaded onto the Ce1−xLax support via wet impregnation. The loading amount of active metals was fixed at 10 wt%, and after impregnation, the samples were dried at 110 °C for 24 h and then calcined in air at 600 °C for 4 h. The catalysts are denoted as Ce1−xLax, where x represents the weight fraction of Ce.
2.2 Catalyst characterization
The specific surface area, total pore volume, and N2 adsorption–desorption isotherms of the powdered samples were determined via N2 adsorption at −196 °C using the Brunauer–Emmett–Teller (BET) method with a BELSORP-max apparatus (BEL Japan, Inc., Japan). Powder XRD analysis was conducted using a D/MAX-2500 diffractometer (Rigaku, Japan) with Cu-Kα radiation over a 2θ range of 10–90° to investigate the crystal structures of the as-prepared and reduced catalysts. H2-TPR analysis was performed using an Autochem II 2920 analyzer (Micromeritics, USA). The surface electronic states and elemental compositions of the reduced samples were characterized by XPS. Measurements were conducted using an ESCALAB Mark II spectrometer (Vacuum Generators, UK) equipped with Al-Kα radiation (hν = 1486.6 eV) at a constant pass energy of 50 eV. The binding energies were calibrated with reference to the C 1s peak at 285.0 eV. Quantitative analysis was performed via peak deconvolution and integration of the fitted peak areas to determine the relative elemental concentrations. Soft X-ray absorption spectroscopy (XAS) experiments were conducted at beamline 7.3.1, Advanced Light Source, Lawrence Berkeley National Laboratory (USA). Data acquisition was performed in the total fluorescence yield (TFY) mode, providing a probing depth of approximately 100 nm. All spectra were normalized to the beam flux, which was measured using an upstream gold mesh. The photon energies in the C K-edge and Ni L3-edge spectra were calibrated using a standard sample with a resolution of 0.2 eV. CO2-TPD analysis was conducted using an Autochem II 2920 analyzer (Micromeritics, USA) to evaluate the basicity of the prepared catalysts. Before the measurements, the catalyst (∼100 mg) was loaded into a quartz U-tube reactor and reduced under a 10 vol% H2/Ar flow at 500 °C for 1 h. After cooling to 50 °C, the sample was exposed to a 10 vol% CO2/He stream for 1 h to allow adsorption. The physically adsorbed CO2 was subsequently removed by purging with pure He. The sample was then heated from 50 to 900 °C at a ramping rate of 5 °C min−1. The number of basic sites (mmol g−1) was quantified by integrating the desorption peaks.CH4-TPD analysis was performed using a BELCAT-II instrument (BEL Japan Inc., Japan) to investigate the CH4 adsorption and desorption characteristics of the reduced catalysts. For the analysis, the catalyst (∼100 mg) was loaded into a quartz tubular reactor and reduced under a 10 vol% H2/N2 flow at 500 °C for 2 h. After cooling to 50 °C, the sample was exposed to a 5 vol% CH4/N2 gas mixture for 1 h to enable methane adsorption, followed by a 30 min N2 purge to remove physically adsorbed species. The TPD measurements were conducted by heating the sample from 50 to 900 °C at a rate of 5 °C min−1 under a continuous N2 flow. During the desorption process, the evolution of CH4 (m/z = 16), CO2 (m/z = 44), and CO (m/z = 28) was monitored in real-time using a quadrupole mass spectrometer to evaluate CH4 decomposition behavior.
The morphological characteristics of the carbon materials grown on the catalysts were examined by field-emission scanning electron microscopy (FE-SEM) using a Regulus 8220 microscope (HITACHI, Japan). Microstructural features and elemental distribution were examined using transmission electron microscopy (TEM) and energy-dispersive X-ray spectroscopy (EDX) mapping, performed on a F200 microscope (JEOL, Japan) operated at 200 kV. Raman spectroscopy was performed to evaluate the crystallinity of the carbon materials using an NRS-5100 spectrometer (JASCO Co., Japan) equipped with a 532 nm excitation source from an Nd-YAG laser. The spectra were recorded in the wavenumber range of 1000–4000 cm−1. Thermogravimetric (TG) analysis was conducted using an SDT-Q600 thermal analyzer (TA Instruments, USA) under a controlled air atmosphere. The samples were heated from room temperature to 1000 °C at a constant heating rate of 10 °C min−1, and the corresponding weight changes were continuously monitored to assess the thermal stability and compositional characteristics until a steady state was achieved.
2.3 Catalyst performance test
A thermogravimetric (TG) analyzer and tubular chemical vapor deposition (CVD) reactor were employed to conduct the catalytic reactions. The TG analyzer (Fig. 1(a)) was particularly useful for methane decomposition experiments, as it enabled quantitative analysis of the catalytic decomposition behavior, carbon yield, and temperature-dependent reaction trends using a sensitive balance. A catalyst sample (5 mg) was placed in a crucible mounted on an analytical balance and pretreated under a 10% H2/N2 flow at 500 °C for 1 h for reduction. Subsequently, the temperature was increased to 600 °C at a rate of 10 °C min−1 and held for 1 h under specific reaction conditions. The reactant gas, a mixture of 50% CH4 in N2, was introduced into the catalytic reactor at a weight hourly space velocity (WHSV) of 360 L gcat−1 h−1 based on CH4 gas. After the reaction, the carbon yield was estimated by monitoring the weight change of the crucible containing the reaction products. The methane decomposition rate (RCH4,TG) for each catalyst was evaluated using eqn (1), where mi is the initial mass of the catalyst, Δm is the change in the mass of sample during the reaction and Mc is the molecular weight of carbon. | ||
| Fig. 1 Schematic of the experimental setup: (a) thermogravimetric analyzer and (b) tubular chemical vapor deposition reactor. | ||
A tubular CVD reactor (Fig. 1(b)) was used for large-scale carbon production and analysis of the reaction gases, with the temperature of the Pt crucible positioned at the center of the reactor controlled by infrared radiation. The catalyst (50 mg) was placed in the crucible of the reactor, reduced under a 10% H2/N2 flow at 500 °C for 1 h, and then the reaction gas (50% CH4/N2) was introduced at 600 °C. This reaction gas was supplied to the catalytic reactor at a WHSV of 36 L gcat−1 h−1 based on CH4. The compositions of the reaction gases were analyzed in real-time using an AO2000 analyzer (ABB, UK) equipped with a non-dispersive infrared gas sensor (Uras 26) and thermal conductivity detector (Caldos 27), enabling continuous monitoring. Following the reaction, the crucible containing the products was weighed directly using an analytical balance, and the carbon yield (Cyield) was calculated using eqn (2). The methane decomposition rate (RCH4,CVD)and hydrogen production rate (RH2,CVD) in the tubular CVD reactor were given by eqn (3) and (4), respectively.
In eqn (3) and (4), FCH4,in and FCH4,out refer to the inlet and outlet flow rates of methane, respectively. FCH4,out was determined as Ftotal,out × PCH4,out, where Ftotal,out was calculated from FN2,in/PN2,out. Eqn (4) provides the calculation formula based on the methane decomposition reaction (CH4 (g) → C (s) + 2H2 (g)), where no considerable side reactions are detected.
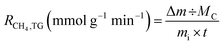 | (1) |
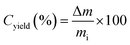 | (2) |
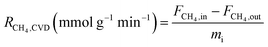 | (3) |
| RH2,CVD = RCH4,CVD × 2 | (4) |
3. Results and discussion
3.1 Structural and textural properties of Ni/Ce1−x-Lax catalysts
Powder XRD provides the structural framework against which subsequent spectroscopic and catalytic results are interpreted. XRD analysis was performed to determine the crystal structure of the catalyst before and after reduction. The analysis identifies the crystallographic phases of the Ce–La mixed-oxide supports and Ni-containing species. It also monitors La-induced lattice expansion via systematic 2θ shifts of fluorite reflections, and reveals potential composition-dependent transitions toward perovskite-like phases as the Ce/La ratio varies.
Fig. 2(a) shows the XRD patterns of the Ni/Ce1−xLax (x = 0–1.0) catalysts calcined at 600 °C. The Ni/Ce1.0, Ni/Ce0.9La0.1, and Ni/Ce0.7La0.3 samples exhibit diffraction peaks corresponding to the CeO2 phase (JCPDS 34-0394), along with peaks attributed to the NiO phase (JCPDS 47-1049, denoted by  ). A shift of the CeO2 diffraction peaks toward lower angles is observed with increasing La concentration up to 30%, which is attributed to the incorporation of La3+ into the CeO2 lattice.12 When the La content exceeds 50%, peak positions and relative intensities change markedly, indicating the emergence of mixed oxides such as LaNiO3 (
). A shift of the CeO2 diffraction peaks toward lower angles is observed with increasing La concentration up to 30%, which is attributed to the incorporation of La3+ into the CeO2 lattice.12 When the La content exceeds 50%, peak positions and relative intensities change markedly, indicating the emergence of mixed oxides such as LaNiO3 ( ) and La2NiO4 (
) and La2NiO4 ( ) and a structural transition of the catalyst from the fluorite to the perovskite phase.17,18 In the perovskite structures, Ni3+ is incorporated into the crystal lattice. Accordingly, no distinct diffraction peaks corresponding to NiO are observed in the La-rich catalysts. Unlike the NiO species supported on fluorites, this structure is less likely to participate directly in the reaction.19 Characteristic peaks corresponding to La2O2CO3 (
) and a structural transition of the catalyst from the fluorite to the perovskite phase.17,18 In the perovskite structures, Ni3+ is incorporated into the crystal lattice. Accordingly, no distinct diffraction peaks corresponding to NiO are observed in the La-rich catalysts. Unlike the NiO species supported on fluorites, this structure is less likely to participate directly in the reaction.19 Characteristic peaks corresponding to La2O2CO3 ( ) were also detected for the Ni/Ce0.1La0.9 and Ni/La1.0 catalysts. This phase likely formed from the reaction between La and carbonate precursors, indicating incomplete decomposition of the carbonate precursor.20
) were also detected for the Ni/Ce0.1La0.9 and Ni/La1.0 catalysts. This phase likely formed from the reaction between La and carbonate precursors, indicating incomplete decomposition of the carbonate precursor.20
Fig. 2(b) shows the XRD patterns of the catalysts after reduction at 500 °C. For the CeO2-dominant catalysts (Ce 0.5–1.0), the fluorite-type CeO2 phase remains unchanged, which is consistent with those observed in the as-synthesized samples. The presence of metallic Ni peaks ( ) indicates the successful reduction of Ni species.21 In contrast, for Ni/Ce0.1La0.9 and Ni/La1.0, additional diffraction peaks appeared, which can be attributed to La2NiO4 (
) indicates the successful reduction of Ni species.21 In contrast, for Ni/Ce0.1La0.9 and Ni/La1.0, additional diffraction peaks appeared, which can be attributed to La2NiO4 ( ) and La(OH)3 (
) and La(OH)3 ( ). Since these phases are absent prior to reduction, their appearance suggests structural decomposition and phase segregation during H2 treatment. The appearance of these La-containing phases indicates structural instability, implying that uniform active species were not successfully formed.17,19 Furthermore, incomplete reduction under the applied conditions likely led to the formation of secondary phases such as La2O3, La(OH)3, and La2NiO4.20 These changes reflect a structural transformation from a CeO2-type fluorite structure toward complex La-containing oxide phases as the La content increases.
). Since these phases are absent prior to reduction, their appearance suggests structural decomposition and phase segregation during H2 treatment. The appearance of these La-containing phases indicates structural instability, implying that uniform active species were not successfully formed.17,19 Furthermore, incomplete reduction under the applied conditions likely led to the formation of secondary phases such as La2O3, La(OH)3, and La2NiO4.20 These changes reflect a structural transformation from a CeO2-type fluorite structure toward complex La-containing oxide phases as the La content increases.
The structural transformation of the lattice observed with the XRD analyses is further supported by the following TEM-based analyses of the reduced catalyst samples. Fig. 3(a) shows that high Ce content catalysts exhibit well-defined, spherical Ni nanoparticles on the surface. However, as the La content increases above 0.5, the reduced Ni nanoparticles are no longer distinguishable in the TEM images, and the EDS maps confirm that Ni is uniformly distributed alongside Ce and La within the catalyst particles. This observation supports the hypothesis that, in La-rich environments, Ni is incorporated into perovskite-like structures (LaNiO3 and La2NiO4) within the oxide matrix, which inhibits the segregation of reduced Ni on the surface.
 | ||
| Fig. 3 (a) TEM-EDS elemental mapping images of Ni, Ce, La, and O species and (b) atomistic structural models of LaNiO3, La2NiO4, and Ni/CeO2. | ||
Fig. 3(b) shows atomic models illustrating the structural differences in Ni incorporation depending on the Ce/La ratio. In La-rich environments, the formation of perovskite-type structures such as LaNiO3 and La2NiO4 is thermodynamically favored, with Ni atoms structurally embedded within the La–O lattice, as represented by the left and middle models in Fig. 3(b).18,19 This configuration effectively stabilizes Ni within the oxide framework, restricting its reduction and subsequent segregation into metallic nanoparticles on the surface. In contrast, the right model in Fig. 3(b), representing the high Ce content condition, shows Ni species segregated on the CeO2 surface as metallic nanoparticles, rather than being incorporated into the oxide lattice.6 This structural arrangement facilitates the reduction of Ni2+ to metallic Ni0, resulting in the formation of well-defined Ni nanoparticles observed in the TEM images, which is consistent with the clear metallic Ni diffraction peaks in the XRD patterns after reduction.
Thus, the combined XRD and TEM-EDS results strongly suggest that the Ce–La mixed oxide system effectively tunes the reducibility and distribution of Ni species by controlling the structural evolution of the catalyst, thereby modulating the metal–support interactions and catalytic performance during methane decomposition. This structural transition was accompanied by a notable increase in the particle size and decrease in the BET surface area, as summarized in Table 1. The perovskite-type oxides typically form dense, highly crystalline structures with low surface areas and limited porosity.18,19 Moreover, the Ni species incorporated into these lattice frameworks are less likely to be exposed on the surface as active sites. These structural changes are likely to limit the catalytic activity by reducing both surface accessibility and Ni dispersion.22
| Sample | CH4 conversion ratea [mmol g−1 min−1] | Relative standard deviationa [%] | BET surface area [m2 g−1] | Metal particle sizeb [nm] | Metal dispersionb [%] | Ni loadingc [wt%] | Ce/La ratioc [wt%] |
|---|---|---|---|---|---|---|---|
| a Measured by TG analysis (Fig. 8(a)). b Estimated by CO chemisorption. c Measured by ICP analysis. | |||||||
| Ni/Ce1.0 | 2.59 | 1.81 | 39.5 | 35.6 | 2.84 | 10.4 | 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| Ni/Ce0.9La0.1 | 3.66 | 1.25 | 15.0 | 41.3 | 2.45 | 11.6 | 0.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
| Ni/Ce0.7La0.3 | 11.97 | 0.39 | 14.1 | 45.6 | 2.22 | 11.0 | 0.69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.31 0.31 |
| Ni/Ce0.5La0.5 | 8.24 | 0.44 | 9.3 | 55.9 | 1.72 | 10.7 | 0.48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.52 0.52 |
| Ni/Ce0.3La0.7 | 3.82 | 0.83 | 5.5 | 92.3 | 1.09 | 11.8 | 0.31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.70 0.70 |
| Ni/Ce0.1La0.9 | 4.22 | 0.89 | 5.5 | 108.5 | 0.93 | 11.8 | 0.89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.19 0.19 |
| Ni/La1.0 | 1.99 | 1.06 | 7.0 | 73.0 | 1.39 | 11.2 | 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
3.2 Physicochemical properties influenced by the Ce–La effect
The reduction characteristics of the calcined catalysts were analyzed using H2-TPR, as shown in Fig. 4. Prior to discussing the reduction behavior, ICP analysis confirmed that the Ni loading and Ce/La weight ratios of all catalysts were consistent with the nominal compositions, indicating that unintended compositional deviation did not influence the H2-TPR profiles. All samples exhibit reduction peaks corresponding to the stepwise reduction of the Ni species and associated structural transitions. For Ni/Ce1.0, a distinct reduction peak is observed at 220 °C, corresponding to the reduction of surface NiO species weakly interacting with the CeO2 support, while a second peak at 302 °C is ascribed to the reduction of bulk NiO species more strongly interacting with the support. A broad peak above 700 °C is associated with the Ce4+ → Ce3+ reduction.25,26 | ||
| Fig. 4 Redox properties of Ni/Ce1−xLax catalysts characterized by H2 temperature-programmed reduction. | ||
As the La content increases to x = 0.3 (Ni/Ce0.7La0.3), the main NiO reduction peak gradually shifts to higher temperatures, indicating that balanced La incorporation strengthens the metal–support interaction, as discussed in the previous section. Such a strong interaction facilitates the maintenance of highly dispersed and reducible Ni species during reduction.27 Alternatively, for La-rich samples (x ≥ 0.5), the reduction peaks shift back to lower temperatures, and broad reduction features appear over a wide temperature range, especially in the Ni/Ce0.1La0.9 and Ni/La1.0 catalysts. This profile is attributed to the formation of various La-based oxide phases, as confirmed by XRD. These phases tend to contain weakly bound or phase-separated Ni species.18 Specifically, Ni/La1.0 shows three well-defined peaks at 344, 464, and 601 °C, which are assigned to the stepwise reduction of LaNiO3 → La2NiO4 → La2O3 + Ni0, respectively.18,20 The presence of La2O2CO3, which typically decomposes in the temperature range of 400–600 °C, may also contribute to these reduction steps.21 Overall, the H2-TPR profiles reveal that the La content influences the reduction behavior of NiO species. At lower La contents, the growth of the low-temperature reduction peak indicates facilitated reduction of surface NiO, consistent with improved oxygen mobility and vacancy formation. With further La incorporation, however, the reduction features progressively shift toward the medium- and high-temperature regions, reflecting stronger Ni–O–Ce/La interactions and partial stabilization of bulk NiO.12,25,27 This trend explains that excessive La doping disrupts the fluorite lattice through excessive lattice expansion and structural distortion, which gradually undermines the stability of the metal–support interactions and deteriorates the overall structural integrity of the mixed oxide matrix.28
XPS analysis was conducted to investigate the surface composition and oxidation states of the catalysts, offering insight into the electronic structure and metal–support interactions that govern their catalytic properties. Therefore, X-ray photoelectron spectroscopy (XPS) was carried out to determine the oxidation states of Ce in the Ni/Ce1−xLax catalysts. The relative Ce3+/Ce4+ ratio, as derived from the Ce 3d spectra (Fig. 5(a)), serves as a key descriptor of oxygen vacancy formation and provides complementary evidence for the trends observed in the reduction behavior. The Ce3+ species are labeled as v0, v′, u0, and u′, whereas the Ce4+ species are labeled as v, v″, v‴, u, u″, and u‴.29–31 The relative Ce3+ concentration, summarized in Table 2, increases with the La content up to x = 0.3, reaching a maximum of 43.7%. This indicates that moderate La doping enhances the formation of Ce3+ species, which are often associated with the presence of oxygen vacancies. These oxygen vacancies facilitate improved redox behavior and can promote the activation of CH4 molecules.32 Furthermore, a slight shift in the binding energies of the Ce 3d peaks is observed upon the addition of La. This suggests an electronic structure modification around the Ce–O environment, possibly due to lattice distortion or electron redistribution induced by La3+ substitution.31,33 The La 3d spectra were also examined to investigate the electronic structure of the La species within the Ce–La mixed oxide system. As shown in Fig. 5(b), all La-containing samples exhibit characteristic La 3d5/2 and La 3d3/2 spin–orbit doublets, corresponding to La3+ in an oxide environment.32,34 As the La ratio decreases, the La 3d peaks of the Ce–La mixed oxides shift slightly to lower binding energies, indicating that the La3+ species are not present as phase-separated La2O3 domains but are instead incorporated into the CeO2 lattice, forming a more homogeneous mixed oxide phase.12,35
 | ||
| Fig. 5 X-ray photoelectron spectroscopy and Raman spectroscopy of Ni/Ce1−xLax catalysts: (a) Ce 3d, (b) La 3d, and (c) O 1s XPS spectra, and (d) Raman spectra. | ||
| Sample | Relative concentration [%] | Binding energy [eV] | |||||
|---|---|---|---|---|---|---|---|
| Ce 3d | O 1s | La 3d5/2 | |||||
| Ce3+ | Ce4+ | O2− | O−/O2− | CO32−/OH− | OH− | ||
| Ni/Ce1.0 | 29.5 | 70.5 | 67.6 | 25.0 | — | 7.4 | — |
| Ni/Ce0.9La0.1 | 31.5 | 68.5 | 41.7 | 19.5 | 19.7 | 19.2 | 832.7 |
| Ni/Ce0.7La0.3 | 43.7 | 56.3 | 29.4 | 32.5 | 20.8 | 17.3 | 832.4 |
| Ni/Ce0.5La0.5 | 36.1 | 63.9 | 39.7 | 22.4 | 23.8 | 14.2 | 833.0 |
| Ni/Ce0.3La0.7 | 32.1 | 67.9 | 17.1 | 10.0 | 55.2 | 17.7 | 834.1 |
| Ni/Ce0.1La0.9 | 33.0 | 67.0 | 10.1 | 25.4 | 34.6 | 29.9 | 834.2 |
| Ni/La1.0 | — | — | 20.8 | 18.0 | 37.5 | 23.7 | 833.8 |
The O 1s XPS spectra of the catalysts were analyzed to examine the surface oxygen chemical states, as shown in Fig. 5(c). The O 1s XPS spectra of the as-prepared catalysts were deconvoluted into four primary components corresponding to lattice oxygen (O2−, ∼528.5 eV), surface oxygen species (O−/O2−, ∼530.1 eV), carbonate or hydroxyl groups (CO32−/OH−, ∼531.2 eV), and surface hydroxyls (OH−, ∼532.5 eV).36,37 As summarized in Table 2, the relative content of lattice oxygen (O2−) is highest in Ni/Ce1.0 and progressively decreases as the La content increases, reaching as low as 10.1% for Ni/Ce0.1La0.9. This decrease explains a disruption of the CeO2 fluorite lattice due to La incorporation, which may weaken the Ce–O bond and alter oxygen mobility.12 In contrast, the content of surface oxygen species and hydroxyls (O−/O2−, CO32−/OH−, and OH−) increased significantly with La ratio. Among all the samples, Ni/Ce0.7La0.3 and Ni/Ce0.5La0.5 showed a higher ratio of surface oxygen species (O−/O2−) compared with those of the others, which is consistent with their enhanced CH4 decomposition activity. These species are generally regarded as highly active and closely related to the presence of oxygen vacancies, which is also supported by the increased Ce3+ concentration observed in the Ce 3d spectra.36,37 As the La content increases (x ≥ 0.5), the contributions of CO32− and OH− species also increase, consistent with the appearance of La-based crystalline phases in the XRD patterns of La2O2CO3, La(OH)3, and La2NiO4 (Fig. 2). The presence of such secondary phases suggests structural heterogeneity and phase segregation, which may lead to the accumulation of loosely bound carbonate or hydroxyl groups on the catalyst surface.19,21,36 Consequently, excessive La incorporation disrupts the formation of a uniform mixed-oxide lattice and reduces the availability of catalytically active oxygen species. In such cases, even a high ratio of surface oxygen species does not necessarily enhance CH4 conversion. Indeed, these results highlight that balanced La incorporation is crucial for maximizing active surface oxygen species while minimizing phase separation and the accumulation of inactive surface adsorbates.
While XPS provides valuable information on the surface redox state of cerium, its surface-sensitive nature limits insight into bulk defect structures and lattice disorder. Therefore, Raman spectroscopy was subsequently employed to probe vacancy-related lattice perturbations and structural modifications within the CeO2-based framework. The Raman spectra of the reduced catalysts, as shown in Fig. 5(d), exhibit several characteristic bands that can be assigned to ceria- and lanthanum-related lattice vibrations. For Ce-containing samples, a pronounced Raman band centered at approximately 461 cm−1 is observed, which corresponds to the F2g symmetric stretching vibration of the Ce–O bond in the fluorite CeO2 lattice. This mode is a signature of the well-ordered fluorite structure and is commonly used as a reference for evaluating structural disorder in ceria-based materials.29 Moreover, samples dominated by the CeO2 fluorite lattice exhibit a broad band near 561 cm−1. This feature is generally attributed to defect-induced Raman modes associated with oxygen vacancies as well as local lattice distortions, which are generated by the partial reduction of Ce4+ to Ce3+ within the CeO2 framework.29 It should be recognized that the Ce3+/Ce4+ ratio obtained from XPS and the Raman defect-band intensity represent complementary descriptors of defect-related surface reduction and bulk lattice disorder, respectively, and that a strictly linear correlation between the two is not necessarily required. The relative contribution of this defect-related band was evaluated using the integrated area ratio with respect to the F2g mode, providing an indirect descriptor of vacancy-related lattice disorder. Based on this analysis, the defect-band/F2g area ratio increases sequentially in the order of Ni/Ce1.0 (0.3739) < Ni/Ce0.9La0.1 (0.4718) < Ni/Ce0.7La0.3 (0.6476) < Ni/Ce0.5La0.5 (1.0842), indicating progressively enhanced defect-related lattice perturbation upon La incorporation into the CeO2 lattice. Notably, although XPS Ce 3d analysis reveals the highest surface Ce3+ fraction for the Ni/Ce0.7La0.3 catalyst, the Raman-derived defect index reaches its maximum for Ni/Ce0.5La0.5. This discrepancy can be rationalized by considering that the Raman band at ∼561 cm−1 reflects not only oxygen-vacancy formation but also lattice disorder and structural perturbation. At higher La contents, charge compensation induced by La3+ substitution leads to significant lattice distortion and partial disruption of the fluorite CeO2 framework, thereby amplifying the defect-related Raman response even without a proportional increase in Ce3+ concentration.5,20 Consequently, the elevated defect index observed for Ni/Ce0.5La0.5 is attributed to enhanced lattice disorder arising from La-induced structural modification rather than a simple increase in oxygen-vacancy concentration alone. For La-rich catalysts, additional Raman bands located at approximately 349 and 459 cm−1 become prominent.49 These bands are assigned to La–O lattice vibrations characteristic of the hexagonal La2O3 phase, indicating a gradual loss of fluorite-type CeO2 dominance and the emergence of La-based oxide structures. In this compositional regime, the Raman response is primarily governed by structural transformation and phase heterogeneity rather than ceria-related oxygen-vacancy defects, rendering the conventional defect-band/F2g analysis less applicable. Nevertheless, the emergence of Raman bands associated with La–O lattice vibrations indicates an increased contribution of La-based oxide structures, accompanied by a diminished relative contribution of ceria-related defect features.
To elucidate the electronic structure of Ni within the bulk phase of Ni/Ce1−xLax catalysts, soft XAS was performed at the Ni L3-edge in the total fluorescence yield mode (Fig. 6). Unlike XPS, which primarily probes the surface region, soft XAS enables the investigation of unoccupied electronic states and provides element-specific information about the local coordination and oxidation state of Ni in the bulk region. The main spectral feature corresponds to the Ni L3-edge transition (2p3/2 → 3d) at 852.6 eV.14 In this analysis, an additional peak was consistently observed at approximately ∼851 eV, which was attributed to the La M4-edge, in agreement with previous studies on La-containing oxide systems.38 This La M4-edge feature gradually increased with increasing La content in the Ce–La supports, indicating the presence of La within the support lattice. However, the spectral overlap between the La M4-edge and Ni L3-edges complicates the direct interpretation of the Ni electronic state, particularly in samples with higher La content.38 In order to quantify the oxidation state of the Ni species, linear combination fitting was employed using the reference spectra of Ni foil (Ni0), NiO (Ni2+), and Li(NiMnCo)O2 (Ni3+). This approach enables the effective deconvolution of the Ni L3-edge spectra despite interference from the La M4-edge, allowing the relative concentration of Ni0, Ni2+, and Ni3+ species to be accurately determined in the series of catalysts.38 The analysis reveals that the relative content of metallic Ni increased with the La content, reaching a maximum of 81% for the Ni/Ce0.7La0.3 catalyst. However, further increases in the La content led to higher contents of Ni2+ and Ni3+ species, which were attributed to the formation of perovskite-type structures that stabilize oxidized Ni within the lattice. These results align with the XRD and TEM findings, indicating that excessive La promotes the self-incorporation of Ni into the perovskite framework, thereby reducing metallic Ni on the catalyst surface.
 | ||
| Fig. 6 Ni L3-edge soft X-ray absorption spectroscopy and linear combination fitting results for Ni/Ce1−xLax catalysts. | ||
CO2-TPD is a well-established technique for determining the total basicity of solid catalysts (Fig. 7 and Table 3). All samples exhibit three distinct desorption regions, corresponding to weak (50–150 °C), medium (150–500 °C), and strong (>500 °C) basic sites.39–41 Among the various types of basic sites, medium basic sites are especially noteworthy because of their association with surface oxygen species capable of facilitating CH4 activation via H2 abstraction and stabilizing active CHx intermediates.41 Notably, catalysts with higher CH4 decomposition activities are associated with pronounced contributions from medium basic sites.39 This observation implies that adjusting the Ce/La ratio facilitates the formation of metal-oxygen pairs (Mn+–O2−), which are characteristic of moderately basic sites. These sites serve as bifunctional centers capable of donating and accepting electrons during CH4 activation. This enables C–H bond weakening and electron-density redistribution, collectively reducing the activation energy required for bond cleavage.40,41 By comparison, samples with excessive La content (x ≥ 0.7) exhibited a marked increase in strong basic sites, which are typically associated with low-mobility O2− species and La-rich domains (e.g., La2O3).37 Excessively strong basic sites may increase activation energy barriers or impede the desorption of surface species, potentially resulting in surface poisoning or the accumulation of carbonaceous deposits, such as amorphous carbon.39
| Sample | Weak basic sites | Medium basic sites | Strong basic sites | Ratio of medium basic sites |
|---|---|---|---|---|
| CO2 uptake [µmol g−1] | CO2 uptake [µmol g−1] | CO2 uptake [µmol g−1] | ||
| Ni/Ce1.0 | 109.9 | 188.5 | 162.7 | 0.41 |
| Ni/Ce0.9La0.1 | 34.7 | 526.1 | 395.4 | 0.55 |
| Ni/Ce0.7La0.3 | 38.5 | 717.2 | 157.4 | 0.78 |
| Ni/Ce0.5La0.5 | 36.9 | 221.7 | 68.6 | 0.67 |
| Ni/Ce0.3La0.7 | 23.6 | 394.8 | 725.7 | 0.34 |
| Ni/Ce0.1La0.9 | 14.0 | 315.7 | 1234.0 | 0.20 |
| Ni/La1.0 | 38.6 | 162.9 | 1509.6 | 0.46 |
3.3 Catalytic performance of the Ni/Ce1−x-Lax catalysts
Fig. 8(a) presents the carbon yield and CH4 conversion rates of the prepared catalysts supported on Ce–La mixed oxides to investigate the effect of the Ce/La ratio under standard reaction conditions. The reaction was conducted at 600 °C for 1 h with a WHSV of 360 L g−1 h−1 using the TG analyzer. Among the catalysts, Ni/Ce0.7La0.3 exhibits the highest carbon yield of 811.4 wt%, which is nearly five times that of the Ni/Ce1.0 catalyst. The activity order of the samples is as follows: Ni/Ce0.7La0.3 > Ni/Ce0.5La0.5 > Ni/Ce0.9La0.1 ≈ Ni/Ce0.3La0.7 ≈ Ni/Ce0.1La0.9 > Ni/Ce1.0 ≈ Ni/La1.0 (Fig. 8(a) and Table 1). The hydrogen production rates are not monotonic with Ce content, indicating that catalytic performance is governed by the Ce![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) La ratio rather than by the absolute Ce concentration alone. The superior performance of Ni/Ce0.7La0.3 is associated with its balanced reduction behavior and stable metal–support interaction. The balance between Ce and La is crucial because an excessive proportion of La in the support tends to induce a perovskite-like structure, which is less likely to participate directly in the catalytic reaction, as confirmed by XRD and H2-TPR analyses. In addition, the CO2-TPD results indicate that the dominance of medium-strength basic sites contributes to its high catalytic activity. Previous theoretical studies have shown that such medium-strength sites maintain more favorable C–H activation barriers (64–91 kJ mol−1) compared to strong acid-base pairs, which increase the barrier to 94–126 kJ mol−1.40,41 Therefore, medium basic sites in the form of metal-oxygen pairs play a crucial role in the initial activation of CH4 and are considered one of the key factors in lowering the barrier for C–H bond dissociation.
La ratio rather than by the absolute Ce concentration alone. The superior performance of Ni/Ce0.7La0.3 is associated with its balanced reduction behavior and stable metal–support interaction. The balance between Ce and La is crucial because an excessive proportion of La in the support tends to induce a perovskite-like structure, which is less likely to participate directly in the catalytic reaction, as confirmed by XRD and H2-TPR analyses. In addition, the CO2-TPD results indicate that the dominance of medium-strength basic sites contributes to its high catalytic activity. Previous theoretical studies have shown that such medium-strength sites maintain more favorable C–H activation barriers (64–91 kJ mol−1) compared to strong acid-base pairs, which increase the barrier to 94–126 kJ mol−1.40,41 Therefore, medium basic sites in the form of metal-oxygen pairs play a crucial role in the initial activation of CH4 and are considered one of the key factors in lowering the barrier for C–H bond dissociation.
To further clarify the impact of reaction temperature, additional catalytic tests were conducted between 500 and 800 °C in a tubular CVD reactor (WHSV = 36 L g−1 h−1). As shown in Fig. 8(b), the highest activity is achieved at 600 °C. At temperatures above 700 °C, catalytic deactivation occurs, likely due to sintering of Ni active sites or coverage by amorphous carbon, both of which inhibit catalytic function. Based on these findings, 600 °C is selected as the optimal temperature for long-term testing.
Following the initial activity tests, long-term experiments were performed to evaluate the lifespan of the catalysts. Rapid carbon deposition on the catalyst surface is widely recognized as a major cause of catalyst deactivation during the catalytic decomposition of methane. Fig. 8(c and d) shows the long-term activity profiles of Ni/Ce1−xLax catalysts at 600 °C, evaluated using a tubular CVD reactor. Most catalysts reach their peak activities within 45 min. The Ni/Ce0.7La0.3 and Ni/Ce0.5La0.5 catalysts exhibit remarkable catalytic stability, maintaining high H2 production rates of 16.5 and 14.8 mmol g−1 min−1, respectively, for approximately 300 min without noticeable decline. Notably, these catalysts sustain H2 production rates comparable to those observed during the initial 30 min of the reaction, demonstrating their excellent long-term stability under CH4 decomposition conditions. Ni/Ce0.9La0.1 also demonstrates a high initial activity and an extended catalytic lifetime; however, its H2 production rate gradually decreases after 140 min. In the case of Ni/Ce0.3La0.7, Ni/Ce0.1La0.9, and Ni/La1.0 catalysts, despite achieving high initial H2 production rates of 12.9, 12.1, and 11.3 mmol g−1 min−1, respectively, rapid activity decline is observed within 120 min of the reaction. This is associated with the weakened metal–support interactions and the reduced structural stability observed in the physicochemical characterization studies. In addition, the catalytic performance is also related to the Ni particle size. In general, smaller Ni particles exhibit higher initial activity due to their larger surface area and greater number of exposed active sites.21,23 As shown in Table 1, when the La content in the catalyst exceeds 50%, the Ni particle size increases significantly. Consequently, exceeding the optimal particle size range reduces the exposure of active sites, which can lead to a decline in catalytic performance and durability.24
The CO and CO2 formation profiles are presented in Fig. 8(e). Although CO and CO2 were simultaneously plotted for comparative understanding, their concentrations were consistently below approximately 0.02% throughout the reaction, indicating that the catalytic pathway was dominated by direct CH4 → C + 2H2 routes. Accordingly, the CH4 decomposition rate profiles included in Fig. 8(c) validate the reliability of the performance comparison originally presented using hydrogen production in Fig. 8(d).
Fig. 8(f) presents the Arrhenius plots for the Ni/Ce1.0, Ni/Ce0.7La0.3, and Ni/La1.0 catalysts based on the CH4 decomposition rates measured at different temperatures (Fig. 7(b)). Arrhenius plots provide further evidence for the role of the Ce/La ratio in modulating CH4 activation. Ni/Ce0.7La0.3 exhibited the lowest apparent activation energy (62.9 kJ mol−1 K−1), indicating that partial La substitution enhances the ease of CH4 dissociation, likely through synergistic modifications of surface basicity and oxygen mobility. Conversely, Ni/La1.0 showed the highest activation energy (129.0 kJ mol−1 K−1), consistent with excessive lattice distortion and destabilized Ni–support interactions at high La contents. Ni/Ce1.0 displayed an intermediate value of 114.1 kJ mol−1 K−1. Taken together, these results demonstrate that the catalytic performance trends directly reflect the structure–property relationships derived from Ce/La optimization, with partial La incorporation providing an optimal balance of lattice stability, oxygen vacancy formation, and Ni–support interactions.
It should be noted that the remarkable activity and durability of Ni/Ce0.7La0.3 can be attributed to its optimized physicochemical properties, including an increased Ce3+ fraction, balanced reducibility of Ni species without excessive incorporation into the perovskite-type lattice and the effect of the basic site. Their synergetic features promote the catalytic performance.
To more rigorously evaluate the long-term stability requirement, an extended methane decomposition experiment was conducted using the Ni/Ce0.7La0.3 catalyst beyond the initially evaluated 300 min (more than 600 min). As shown in Fig. S2, the catalyst maintains a high and stable H2 production rate of approximately 16 mmol g−1 min−1 for nearly 5 h. After this period, a gradual decline in activity is observed, indicating the onset of catalyst deactivation under the fixed reaction conditions employed in this study. Although the reaction could not be sustained up to 10 h with preserved activity, the extended test clearly demonstrates the superior durability of Ni/Ce0.7La0.3 compared to other compositions investigated in this work. It should be emphasized that all experiments were conducted under identical reaction parameters to ensure a fair comparison, without adjusting gas composition, pressure, or space velocity. While deactivation becomes evident when the reaction time is extended past 5 h under these conditions, further optimization of operating parameters is expected to extend the catalyst lifetime. This aspect will be a primary issue for the practical application of the catalyst.
3.4 Characterization of carbon materials
To directly elucidate the structural nature of the carbon materials formed during methane decomposition, high-resolution transmission electron microscopy (HRTEM) analysis was performed prior to further morphological and spectroscopic characterization. As shown in Fig. 9(a), the carbon product exhibits a well-defined hollow tubular morphology composed of multiple concentric graphitic layers, which is a characteristic feature of multi-walled carbon nanotubes (MWCNTs). While the formation of concentric graphitic layers confirms the presence of CNT structures, the HRTEM analysis further reveals that La-rich compositions (La ≥ 0.7) tend to exhibit additional amorphous carbon domains adjacent to the tubular carbon. This suggests that excessive La incorporation promotes less ordered carbon deposition, leading to the coexistence of CNTs and amorphous carbon species. This direct structural evidence confirms that CNTs constitute the dominant carbon nanostructure formed under the present reaction conditions. Based on this confirmation, complementary analyses including SEM, diameter distribution, Raman spectroscopy, C K-edge XAS, and TG analysis were subsequently employed to further evaluate the morphology, graphitization degree, and thermal stability of the CNT products. | ||
| Fig. 9 (a) Transmission electron microscopy and (b) scanning electron microscopy images and (c) corresponding CNT diameter distribution graphs for catalysts after CH4 decomposition. | ||
The physicochemical properties of the carbon materials formed on the catalyst surface depend significantly on the catalyst characteristics. Fig. 9(b and c) shows the FE-SEM images of the carbon materials produced on different Ce1−xLax-supported catalysts after CH4 decomposition, along with the corresponding CNT diameter distributions. All catalysts led to the formation of CNTs, but the morphology and diameter distribution varied significantly depending on the catalyst composition. Ni/Ce1.0 and Ni/Ce0.9La0.1 generated relatively thin and tangled CNTs with moderate uniformity. The Ni/Ce0.7La0.3 catalyst exhibited the narrowest diameter distribution and smallest average diameter (24.5 nm), indicating more controlled CNT nucleation and growth. Ni/Ce0.5La0.5 formed slightly thicker carbon nanomaterials than Ni/Ce0.7La0.3 but maintained a relatively well-aligned structure with an average diameter of 34.6 nm. However, catalysts from Ni/Ce0.3La0.7 to Ni/La1.0 produced thicker and more irregular CNTs, with average diameters ranging from 41.5 to 48.3 nm, accompanied by disordered structures and particle encapsulation. Strong CH4 adsorption can lead to the excessive accumulation of carbonaceous species on the surface, which may hinder proper CNT nucleation and promote the growth of amorphous carbon over filamentous carbon structures.13,24,42 These morphological trends indicate that excessively strong CH4 adsorption can lead to undesirable carbon accumulation, whereas a balanced adsorption-activation behavior is essential for the growth of high-quality CNTs.
TG analysis is useful for both qualitative and quantitative investigation of carbon products formed on catalysts. A mixture of different carbon species can result in a stepwise weight loss curve during TG analysis. Moreover, the temperature at which weight loss occurs can help identify the types of carbon materials present, such as amorphous carbon, single-walled CNTs, and multi-walled CNTs.43,44 As shown in Fig. 10, all catalysts exhibited remarkable weight loss in the range of 500–700 °C, corresponding to the oxidation of carbon products formed during CH4 decomposition. Most samples showed a single sharp oxidation step, which is indicative of more homogeneous and structured carbon, likely consisting predominantly of well-formed CNTs with minimal amorphous content. Interestingly, the Ni/Ce0.1La0.9 and Ni/La1.0 catalysts displayed a two-step weight loss pattern, suggesting the presence of carbon species with different structural characteristics. The initial weight loss at lower temperatures was attributed to the oxidation of amorphous or poorly ordered carbon, which is less thermally stable.44 The subsequent gradual weight loss at higher temperatures corresponds to the oxidation of more graphitized, crystalline CNT structures, which resist oxidation owing to their higher structural order.43 This stepwise oxidation behavior implies that amorphous carbon and CNTs coexist on these catalysts.
Following thermal analysis, Raman spectroscopy and carbon K-edge soft XAS were performed, to evaluate the structural ordering and chemical purity of the CNTs formed on the catalyst surface. As shown in Fig. 11(a) and summarized in Table 4, the ID/IG ratios obtained from the Raman spectra ranged from 0.79 and 1.10. The La-based catalysts, such as Ni/Ce0.3La0.7, Ni/Ce0.1La0.9, and Ni/La1.0, showed relatively low ID/IG ratios (0.93–1.00), indicating a high degree of graphitization.45,46 In contrast, Ni/Ce0.7La0.3 exhibited a slightly higher ID/IG ratio of 0.98, despite producing the most uniform and well-ordered CNTs, according to SEM (Fig. 10(b)).
 | ||
| Fig. 11 (a) Raman and (b) C K-edge soft X-ray absorption spectra of carbon materials formed on Ni/Ce1−xLax catalysts. | ||
| Sample | I D/IG ratio of CNTs | Oxidation temperature [°C] |
|---|---|---|
| Ni/Ce1.0 | 0.92 | 612 |
| Ni/Ce0.9La0.1 | 1.10 | 605 |
| Ni/Ce0.7La0.3 | 0.98 | 655 |
| Ni/Ce0.5La0.5 | 1.00 | 644 |
| Ni/Ce0.3La0.7 | 0.79 | 637 |
| Ni/Ce0.1La0.9 | 0.93 | 336 and 632 |
| Ni/La1.0 | 0.93 | 350, 606, and 717 |
To provide a more detailed understanding of the electronic structure and bonding characteristics of the carbon materials generated during the reaction, carbon K-edge near-edge X-ray absorption fine structure spectroscopy was performed. The resulting spectra are displayed in Fig. 11(b). All samples exhibited clear resonances associated with π* and σ* transitions, offering valuable insights into the hybridization states within the carbon framework. The π* resonance at 285 eV, indicative of transitions to unoccupied π* orbitals of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds, is characteristic of the sp2-hybridized carbon structures typical of CNTs.47,48 Meanwhile, the σ* resonance was observed in the approximate range of 291–292 eV, corresponding to transitions to unoccupied σ* states of C–C single bonds, reflecting sp3-hybridization within the carbon matrix.47,48 In samples with higher Ce content, the π* peak appeared relatively broad and of lower intensity, suggesting greater presence of defects and amorphous carbon species.47,48 In contrast, increasing the La content in the catalyst resulted in a sharper and more intense π* resonance, indicating the development of well-ordered sp2-hybridized carbon networks and high structural alignment within the nanotube walls.47 Moreover, with increasing La content, a distinct peak at 288 eV associated with C–H related resonances became evident, implying the presence of surface-bound functional groups likely formed during the CNT growth process.47 The concurrent presence of a pronounced π* resonance and C–H feature at 288 eV indicates that the CNTs preserve their high crystallinity and graphitic purity while also exhibiting a certain level of surface functionalization.
C double bonds, is characteristic of the sp2-hybridized carbon structures typical of CNTs.47,48 Meanwhile, the σ* resonance was observed in the approximate range of 291–292 eV, corresponding to transitions to unoccupied σ* states of C–C single bonds, reflecting sp3-hybridization within the carbon matrix.47,48 In samples with higher Ce content, the π* peak appeared relatively broad and of lower intensity, suggesting greater presence of defects and amorphous carbon species.47,48 In contrast, increasing the La content in the catalyst resulted in a sharper and more intense π* resonance, indicating the development of well-ordered sp2-hybridized carbon networks and high structural alignment within the nanotube walls.47 Moreover, with increasing La content, a distinct peak at 288 eV associated with C–H related resonances became evident, implying the presence of surface-bound functional groups likely formed during the CNT growth process.47 The concurrent presence of a pronounced π* resonance and C–H feature at 288 eV indicates that the CNTs preserve their high crystallinity and graphitic purity while also exhibiting a certain level of surface functionalization.
Overall, the above analyses suggest that a low ID/IG ratio alone does not ensure the purity of the CNTs, as it can be affected by the presence of graphitized but hydrogenated carbon species.
4. Conclusion
In this study, a series of Ni/Ce1−xLax catalysts was synthesized and evaluated for catalytic methane decomposition to simultaneously produce hydrogen and CNTs. XRD and XPS analyses confirm that the partial substitution of La3+ into the CeO2 lattice induced lattice distortion and electronic coupling without phase segregation at appropriate La contents. In contrast, excessive La incorporation (x ≥ 0.5) yields La-based perovskite-type phases, such as LaNiO3 and La2NiO4, as well as La2O2CO3 and La(OH)3, which significantly alter the catalyst structure. These secondary phases immobilize Ni within the lattice as Ni3+ species, thereby suppressing its reduction to active Ni0. The shift in reducibility and appearance of strong basic sites are associated with poor CH4 activation and rapid deactivation. This structural transition from the fluorite to perovskite phases is a key factor contributing to the decreased activity and carbon yield of the La-based catalysts.The performance of CH4 decomposition highly depends on the Ce/La ratio of the support. Ni/Ce0.7La0.3 exhibited the highest carbon yield (811.4 wt%) and sustained hydrogen production at 600 °C over extended reaction times, reflecting its favorable reduction behavior and optimized Ni–support interactions. However, catalysts with excessive La content show weakened reducibility and poor long-term stability, consistent with the structural instability and strong basic sites inferred from physicochemical characterization. The apparent activation energies confirm this trend, with Ni/Ce0.7La0.3 showing the lowest barrier for CH4 activation, whereas Ni/La1.0 required substantially higher energy input.
Carbon characterization clarifies the structure–performance relationship. Ni/Ce1.0, Ni/Ce0.9La0.1, Ni/Ce0.7La0.3, and Ni/Ce0.5La0.5 catalysts facilitated the growth of uniform CNTs with narrow diameter distributions (average 24.5–39.1 nm), whereas catalysts with excessive La content predominantly produced amorphous and disordered carbon structures. Although some La-based catalysts exhibited low ID/IG ratios, soft XAS analysis confirmed the presence of hydrogenated carbon species, suggesting that a low ID/IG ratio does not necessarily reflect high-purity CNTs.
These results emphasize the critical role of precisely controlling La incorporation to suppress unfavorable phase transitions while retaining the structural and electronic features necessary for efficient methane activation and CNT growth. More importantly, this work advances the understanding of structure-property-performance relationships in Ce–La mixed oxides and presents a rational design strategy for developing high-performance Ni-based catalysts, thereby enabling efficient and CO2-free co-production of hydrogen and carbon nanomaterials. Such carbon nanostructures are also highly promising for applications in nanoscale devices and lithium-ion batteries, further broadening the impact of this work.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Acknowledgements
This work was conducted under framework of the research and development program of the Korea Institute of Energy Research (C5-2402). This research was also supported by the National Research Council of Science & Technology(NST) grant by the Korea government (MSIT) (No. GTL25101-000).References
- D. Leybo, U. J. Etim, M. Monai, S. R. Bare, Z. Zhong and C. Vogt, Metal-support interactions in metal oxide-supported atomic, cluster, and nanoparticle catalysis, Chem. Soc. Rev., 2024, 53(21), 10450–10490, 10.1039/d4cs00527a.
- J. L. Snider, V. Streibel, M. A. Hubert, T. S. Choksi, E. Valle and D. C. Upham, et al., Revealing the Synergy between Oxide and Alloy Phases on the Performance of Bimetallic In-Pd Catalysts for CO2 Hydrogenation to Methanol, ACS Catal., 2019, 9(4), 3399–3412, DOI:10.1021/acscatal.8b04848.
- M. Grabchenko, G. Pantaleo, F. Puleo, T. S. Kharlamova, V. I. Zaikovskii and O. Vodyankina, et al., Design of Ni-based catalysts supported over binary La-Ce oxides: Influence of La/Ce ratio on the catalytic performances in DRM, Catal. Today, 2021, 382, 71–81, DOI:10.1016/j.cattod.2021.07.012.
- K. Sutthiumporn and S. Kawi, Promotional effect of alkaline earth over Ni-La2O3 catalyst for CO2 reforming of CH4: Role of surface oxygen species on H2 production and carbon suppression, Int. J. Hydrogen Energy, 2011, 36(22), 14435–14446, DOI:10.1016/j.ijhydene.2011.08.022.
- A. Gupta, U. V. Waghmare and M. S. Hegde, Correlation of oxygen storage capacity and structural distortion in transition-metal-, noble-metal-, and rare-earth-ion-substituted CeO2 from first principles calculation, Chem. Mater., 2010, 22(18), 5184–5198, DOI:10.1021/cm101145d.
- M. J. Kim, J. R. Youn, H. J. Kim, M. W. Seo, D. Lee and K. S. Go, et al., Effect of surface properties controlled by Ce addition on CO2 methanation over Ni/Ce/Al2O3 catalyst, Int. J. Hydrogen Energy, 2020, 45(46), 24595–24603, DOI:10.1016/j.ijhydene.2020.06.144.
- M. Greluk, M. Rotko and S. Turczyniak-Surdacka, Enhanced catalytic performance of La2O3 promoted Co/CeO2 and Ni/CeO2 catalysts for effective hydrogen production by ethanol steam reforming: La2O3 promoted Co(Ni)/CeO2 catalysts in SRE, Renew. Energy, 2020, 155, 378–395, DOI:10.1016/j.renene.2020.03.117.
- I. Luisetto, S. Tuti, C. Romano, M. Boaro, E. Di Bartolomeo and J. K. Kesavan, Dry reforming of methane over Ni supported on doped CeO2 : New insight on the role of dopants for CO2 activation, J. CO2 Util., 2019, 30, 63–78, DOI:10.1016/j.jcou.2019.01.006.
- P. Vinchhi, A. Ray, K. Mallik and R. Pati, Gd-doped ceria with extraordinary oxygen-ion conductivity for low temperature solid oxide fuel cells, Sci. Rep., 2024, 14(1), 1–13, DOI:10.1038/s41598-024-59030-6.
- X. Hou, Q. Lu and X. Wang, Enhanced catalytic properties of La-doped CeO2 nanopowders synthesized by hydrolyzing and oxidizing Ce46La5C49 alloys, J. Sci. Adv. Mater. Devices, 2017, 2(1), 41–44, DOI:10.1016/j.jsamd.2017.02.006.
- Y. Xu, L. Gao, Q. Hou, P. Wu, Y. Zhou and Z. Ding, Enhanced Oxygen Storage Capacity of Porous CeO2 by Rare Earth Doping, Molecules, 2023, 28(16), 6005, DOI:10.3390/molecules28166005.
- R. Zhou, M. Mohamedali, Y. Ren, Q. Lu and N. Mahinpey, La-Ce binary oxide catalysts for low-temperature dry reforming, Int. J. Hydrogen Energy, 2023, 48(89), 34766–34782, DOI:10.1016/j.ijhydene.2023.05.281.
- S. Takenaka, S. Kobayashi, H. Ogihara and K. Otsuka, Ni/SiO2 catalyst effective for methane decomposition into hydrogen and carbon nanofiber, J. Catal., 2003, 217(1), 79–87, DOI:10.1016/S0021-9517(02)00185-9.
- J. R. Youn, M. J. Kim, K. C. Kim, M. Kim, T. Jung and K. S. Go, et al., Highly efficient Co-added Ni/CeO2 catalyst for co-production of hydrogen and carbon nanotubes by methane decomposition, Fuel Process. Technol., 2024, 263, 108130, DOI:10.1016/j.fuproc.2024.108130.
- A. Rastegarpanah, F. Meshkani and M. Rezaei, COx-free hydrogen and carbon nanofibers production by thermocatalytic decomposition of methane over mesoporous MgO·Al2O3 nanopowder-supported nickel catalysts, Fuel Process. Technol., 2017, 167, 250–262, DOI:10.1016/j.fuproc.2017.07.010.
- S. Afzal, A. Prakash, P. Littlewood, H. Choudhury, Z. K. Ghouri and S. Mansour, et al., Catalyst Deactivation by Carbon Deposition: The Remarkable Case of Nickel Confined by Atomic Layer Deposition, ChemCatChem, 2021, 13(13), 2988–3000, DOI:10.1002/cctc.202100109.
- Y. Xu, J. Qiao, W. Sun, Z. Wang and K. Sun, Enhancement of CO2 activation and coke-resistant ability on Ni/CeO2 catalyst with La doping for dry reforming of methane, Int. J. Hydrogen Energy, 2024, 88, 1451–1462, DOI:10.1016/j.ijhydene.2024.07.027.
- M. F. Bekheet, P. D. K. Nezhad, N. Bonmassar, L. Schlicker, A. Gili and S. Praetz, et al., Steering the Methane Dry Reforming Reactivity of Ni/La2O3 Catalysts by Controlled in Situ Decomposition of Doped La2NiO4 Precursor Structures, ACS Catal., 2021, 11(1), 43–59, DOI:10.1021/acscatal.0c04290.
- A. Bhaskaran, S. A. Singh, P. D. Costa and S. Roy, Unveiling the catalytic behaviour of LaNiO3 and La2NiO4 for dry reforming of methane, Int. J. Hydrogen Energy, 2024, 68, 623–634, DOI:10.1016/j.ijhydene.2024.04.295.
- R. Pereñíguez, V. M. González-DelaCruz, J. P. Holgado and A. Caballero, Synthesis and characterization of a LaNiO3 perovskite as precursor for methane reforming reactions catalysts, Appl. Catal., B, 2010, 93(3–4), 346–353, DOI:10.1016/j.apcatb.2009.09.040.
- B. M. Faroldi, J. F. Múnera and L. M. Cornaglia, In situ characterization of phase transformation and reactivity of high surface area lanthanum-based Ru catalysts for the combined reforming of methane, Appl. Catal., B, 2014, 150–151, 126–137, DOI:10.1016/j.apcatb.2013.12.005.
- S. Takenaka, E. Kato, Y. Tomikubo and K. Otsuka, Structural change of Ni species during the methane decomposition and the subsequent gasification of deposited carbon with CO2 over supported Ni catalysts, J. Catal., 2003, 219(1), 176–185, DOI:10.1016/S0021-9517(03)00152-0.
- P. G. Lustemberg, Z. Mao, A. Salcedo, B. Irigoyen, M. V. G. Pirovano and C. T. Campbell, Nature of the Active Sites on Ni/CeO2 Catalysts for Methane Conversions, ACS Catal., 2021, 11(16), 10604–10613, DOI:10.1021/acscatal.1c02154.
- W. Liang, H. Yan, C. Chen, D. Lin, K. Tan and X. Feng, et al., Revealing the effect of nickel particle size on carbon formation type in the methane decomposition reaction, Catalysts, 2020, 10(8), 1–20, DOI:10.3390/catal10080890.
- S. Chen, L. Higgins, I. Giarnieri, P. Benito and A. M. Beale, A detailed characterization study of Ni/CeO2 catalysts identifies Ni availability as the primary factor affecting CO2 methanation performance, J. Catal., 2024, 439, 115778, DOI:10.1016/j.jcat.2024.115778.
- J. Lucas, N. S. P. Naveen, M. J. Janik, K. Alexopoulos, G. Noh and D. Aireddy, et al., Improved Selectivity and Stability in Methane Dry Reforming by Atomic Layer Deposition on Ni-CeO2-ZrO2/Al2O3 Catalysts, ACS Catal., 2024, 14(12), 9115–9133, DOI:10.1021/acscatal.4c02019.
- M. Greluk, M. Rotko and S. Turczyniak-Surdacka, Enhanced catalytic performance of La2O3 promoted Co/CeO2 and Ni/CeO2 catalysts for effective hydrogen production by ethanol steam reforming: La2O3 promoted Co(Ni)/CeO2 catalysts in SRE, Renew. Energy, 2020, 155, 378–395, DOI:10.1016/j.renene.2020.03.117.
- M. Y. Dogan, H. Arbag, H. M. Tasdemir, N. Yasyerli and S. Yasyerli, Effect of ceria content in Ni–Ce–Al catalyst on catalytic performance and carbon/coke formation in dry reforming of CH4, Int. J. Hydrogen Energy, 2023, 48(60), 23013–23030, DOI:10.1016/j.ijhydene.2023.04.011.
- B. Choudhury and A. Choudhury, Ce3+ and oxygen vacancy mediated tuning of structural and optical properties of CeO2 nanoparticles, Mater. Chem. Phys., 2012, 131(3), 666–671, DOI:10.1016/j.matchemphys.2011.10.032.
- W. Ma, T. Mashimo, S. Tamura, M. Tokuda, S. Yoda and M. Tsushida, et al., Cerium oxide (CeO2-x) nanoparticles with high Ce3+ proportion synthesized by pulsed plasma in liquid, Ceram. Int., 2020, 46(17), 26502–26510, DOI:10.1016/j.ceramint.2020.07.093.
- S. G. Peera and S. W. Kim, Rare Earth Ce/CeO2 Electrocatalysts: Role of High Electronic Spin State of Ce and Ce3+/Ce4+ Redox Couple on Oxygen Reduction Reaction, Nanomaterials, 2025, 15(8), 600, DOI:10.3390/nano15080600.
- P. R. L. Keating, D. O. Scanlon and G. W. Watson, The nature of oxygen states on the surfaces of CeO2 and La-doped CeO2, Chem. Phys. Lett., 2014, 608, 239–243, DOI:10.1016/j.cplett.2014.05.094.
- N. Shehata, K. Meehan, M. Hudait and N. Jain, Control of oxygen vacancies and Ce+3 concentrations in doped ceria nanoparticles via the selection of lanthanide element, J. Nanopart. Res., 2012, 14(10), 1–10, DOI:10.1007/s11051-012-1173-1.
- J. Zhang, H. Wong, D. Yu, K. Kakushima and H. Iwai, X-ray photoelectron spectroscopy study of high-k CeO2/La2O3 stacked dielectrics, AIP Adv., 2014, 4(11), 117117, DOI:10.1063/1.4902017.
- N. Shehata, K. Meehan, M. Hudait and N. Jain, Control of oxygen vacancies and Ce+3 concentrations in doped ceria nanoparticles via the selection of lanthanide element, J. Nanopart. Res., 2012, 14(10), 1–10, DOI:10.1007/s11051-012-1173-1.
- Y. Yu, Y. M. Gan, C. Huang, Z. H. Lu, X. Wang and R. Zhang, et al., Ni/La2O3 and Ni/MgO–La2O3 catalysts for the decomposition of NH3 into hydrogen, Int. J. Hydrogen Energy, 2020, 45(33), 16528–16539, DOI:10.1016/j.ijhydene.2020.04.127.
- K. Sutthiumporn and S. Kawi, Promotional effect of alkaline earth over Ni-La2O3 catalyst for CO2 reforming of CH4: Role of surface oxygen species on H2 production and carbon suppression, Int. J. Hydrogen Energy, 2011, 36(22), 14435–14446, DOI:10.1016/j.ijhydene.2011.08.022.
- M. Hepting, D. Li, C. J. Jia, H. Lu, E. Paris and Y. Tseng, et al., Electronic structure of the parent compound of superconducting infinite-layer nickelates, Nat. Mater., 2020, 19(4), 381–385, DOI:10.1038/s41563-019-0585-z.
- A. Figueiredo, M. A. Soria, L. M. Madeira and C. Rocha, Preparation and Characterization of Materials for Low- to Intermediate-Temperature CO2 Adsorption, Processes, 2024, 12(11), 2403, DOI:10.3390/pr12112403.
- A. Misol, I. Giarnieri, F. Ospitali, A. Ballarini, J. Jiménez-Jiménez and E. Rodríguez-Castellón, et al., CO2 hydrogenation over Ru hydrotalcite-derived catalysts, Catal. Today, 2024, 425, 3–11, DOI:10.1016/j.cattod.2023.114362.
- Y. R. Dias and O. W. Perez-Lopez, CO2 methanation over Ni-Al LDH-derived catalyst with variable Ni/Al ratio, J. CO2 Util., 2023, 68, 102381, DOI:10.1016/j.jcou.2022.102381.
- L. Dong, Y. Du, J. Li, H. Wang, Y. Yang and S. Li, et al., The effect of CH4 decomposition temperature on the property of deposited carbon over Ni/SiO2 catalyst, Int. J. Hydrogen Energy, 2015, 40(31), 9670–9676, DOI:10.1016/j.ijhydene.2015.06.005.
- E. Mansfield, A. Kar, C. M. Wang and A. N. Chiaramonti, Statistical sampling of carbon nanotube populations by thermogravimetric analysis, Anal. Bioanal. Chem., 2013, 405(25), 8207–8213, DOI:10.1007/s00216-013-7221-6.
- A. Buzarovska, V. Stefov, M. Najdoski and G. Bogoeva-Gaceva, Thermal analysis of multi-walled carbon nanotubes material obtained by catalytic pyrolysis of polyethylene, Maced. J. Chem. Chem. Eng., 2015, 34(2), 373–379, DOI:10.20450/mjcce.2015.620.
- B. J. Kim, J. P. Kim and J. S. Park, Effects of Al interlayer coating and thermal treatment on electron emission characteristics of carbon nanotubes deposited by electrophoretic method, Nanoscale Res. Lett., 2014, 9(1), 1–6, DOI:10.1186/1556-276X-9-236.
- C. Zhou, R. Senegor, Z. Baron, Y. Chen, S. Raju and A. A. Vyas, et al., Synthesis and interface characterization of CNTs on graphene, Nanotechnology, 2016, 28(5) DOI:10.1088/1361-6528/28/5/054007.
- N. H. Tran, M. A. Wilson, A. S. Milev, J. R. Bartlett, R. N. Lamb and D. Martin, et al., Photoemission and absorption spectroscopy of carbon nanotube interfacial interaction, Adv. Colloid Interface Sci., 2009, 145(1–2), 23–41, DOI:10.1016/j.cis.2008.07.006.
- V. Shmatko, D. Leontyeva, N. Nevzorova, N. Smirnova, M. Brzhezinskaya and G. Yalovega, Interaction between NiOx and MWCNT in NiOx/MWCNTs composite: XANES and XPS study, J. Electron Spectrosc. Relat. Phenom., 2017, 220, 76–80, DOI:10.1016/j.elspec.2017.03.016.
- J. Xiong, H. Yu, Y. Wei, C. Xie, K. Lai and Z. Zhao, et al., Metal Ions (Li, Mg, Zn, Ce) Doped into La2O3 Nanorod for Boosting Catalytic Oxidative Coupling of Methane, Catalysts, 2022, 12, 713, DOI:10.3390/catal12070713.
| This journal is © The Royal Society of Chemistry 2026 |




