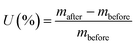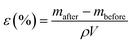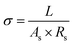Open Access Article
Open Access ArticleMolecularly engineered Li compensation agent-integrated separator enabling regeneration of degraded LiFePO4
Fujun
Tao†
 a,
Zeyi
Yao†
a,
Zeyi
Yao†
 a,
Jiahui
Hou
a,
Jiahui
Hou
 a,
Zexin
Wang
a,
Zhenzhen
Yang
a,
Zexin
Wang
a,
Zhenzhen
Yang
 b and
Yan
Wang
b and
Yan
Wang
 *a
*a
aDepartment of Material Science and Engineering, Worcester Polytechnic Institute, 100 Institute Rd, Worcester, MA 01609, USA. E-mail: yanwang@wpi.edu
bChemical Sciences and Engineering Division, Argonne National Laboratory, 9700 S Cass Ave, Lemont, IL 60439, USA
First published on 6th February 2026
Abstract
Lithium replenishment separators (LRSs) integrating pre-lithiation agents can regenerate degraded lithium cathodes via facile reassembly with a fresh anode and the LRS. A persistent challenge is the formation of gas or solid residues during pre-lithiation. To address this, for the first time, we develop an LRS based on a molecularly engineered dilithium salt of tetrafluorohydroquinone, which compensates for lithium loss while generating decomposition products that dissolve in the electrolyte as a favorable additive, without forming gas or solid residues, thus offering a green route for lithium compensation. A pristine LiFePO4‖graphite full cell with the LRS exhibits 9.3% higher overall capacity than a polypropylene separator (PPS) cell after 50 cycles at 0.5C, and the degraded LiFePO4‖graphite full cell incorporating this LRS achieves a 44.9% higher capacity than the PPS-based cell after 200 cycles at 0.5C. Our LRS demonstrates strong potential for high-performance lithium-ion batteries and spent battery regeneration.
1. Introduction
The escalating demand for electric vehicles and portable electronics underscores the urgent need to develop lithium-ion batteries that offer both high energy density and prolonged cycle life.1–4 The formation of a solid electrolyte interphase (SEI) and the related side reactions on the graphite anode consume 5–20% of lithium from the cathode during the first cycle, leading to substantial initial irreversible capacity loss and decreased lifetime and energy density of lithium-ion batteries.5–7 To address this challenge, pre-lithiation,8 which is strategically engineered to offset the initial lithium loss in lithium-ion batteries, represents a conceptually distinct approach from conventional electrode or material pretreatment. This strategy involves the controlled incorporation of additional electrochemically active lithium through designed agents, functionalized materials, or optimized processing before cycling.9 To date, various pre-lithiation routes have been explored, including chemical10–12 and electrochemical prelithiation,13,14 direct contact15 or the external short circuit strategy,16 as well as the use of pre-lithiation additives in cathodes,17–19 anodes,20 separators,21,22 or electrolytes.23Using pre-lithiation additives has emerged as a promising strategy to mitigate initial irreversible capacity loss in lithium-ion batteries, owing to their facile and safe implementation with a minimal amount of high-capacity agents. In cathode pre-lithiation, the additive undergoes oxidation during charging to release Li+, which is subsequently incorporated into the anode, thereby compensating for lithium consumption during the initial cycle. Representative additives include ternary LixMyOz compounds (M = Ni, Co, Fe, etc.) such as Li6CoO4,24 Li5FeO4,17 and Li2NiO2,19 binary lithium compounds (e.g., Li2O225 and Li3N26), and lithium-containing salts such as Li2C4O4,22 Li2C2O4,18 and Li2C3O5.27 Nevertheless, the decomposition of these compounds frequently generates gaseous byproducts, which impede charge transport, compromise cathode integrity, and degrade electrochemical performance. To suppress gas evolution, conversion-type nanocomposites such as M/Li2O,5 M/LiF,7 and M/Li2S6,21 (M = Ni, Co, Fe, etc.) have been developed. However, these materials introduce substantial pre-lithiation-derived solid residues, which reduce practical energy density and limit their applicability. For example, the decomposition of Co/Li2O results in the formation of Co3O4 as a solid residue.5 In addition, inorganic lithium compounds, including Li2S,28 Li2Se,29 and Li3P30 have been explored to suppress gas evolution during de-lithiation; nevertheless, the resulting solid residues of S,28 Se,29 or P30 in the cathode similarly limit their practical deployment.
Inspired by cathode pre-lithiation, an in situ lithium compensation strategy based on a LRS has recently emerged as a promising approach,21,31 owing to its simplicity and operational advantages. Unlike cathode pre-lithiation, which may induce material compatibility issues, processing challenges, and pore formation in the electrode, the LRS physically separates the pre-lithiation agent from the cathode, preserving the electrode's structural integrity after its decomposition.32 In addition, the conductive LRS can function as a secondary current collector,32 enhancing electron transport and improving the utilization of active materials, thereby boosting the electrochemical performance of lithium-ion batteries32 or sodium-ion batteries.32,33 Moreover, the LRS can be directly applied to regenerate degraded cathodes, such as LiFePO4 (LFP).34–36 Specifically, degraded cathodes harvested from spent lithium-ion batteries can be reassembled with the LRS and a fresh anode. Unlike conventional recycling approaches,37,38 which require separation of active materials from the aluminum current collector, this strategy allows direct reassembly of the degraded cathode, bypassing the separation step. Upon initial charging to a predetermined voltage, the prelithiation agent in the LRS decomposes, restoring the cell capacity relative to the spent battery. This approach reduces recycling costs while providing a practical and environmentally friendly method for cathode regeneration in spent lithium-ion batteries.34–36
While significant efforts have been devoted to developing novel pre-lithiation agents and methods to compensate for the active lithium loss, the gas production22,32,34–36 and solid residues5–7,21,28–30 arising from the decomposition of the pre-lithiation agents inhibit the practical application. In this work, we first report an LRS that produces neither gas nor solid residues during the pre-lithiation process, prepared with a molecularly engineered pre-lithiation agent, lithium 2,3,5,6-tetrafluorobenzene-1,4-bis(olate) (LTFBB), directly coated on a commercial PPS. Using a pristine LFP electrode, the LFP‖Gr full cell with the LRS exhibits a 9.3% higher overall capacity than the cell with a PPS after 50 cycles at 0.5C. For degraded LFP (D-LFP) batteries, the D-LFP‖Gr full cell incorporating the LRS achieves a 44.9% higher overall capacity compared to the PPS-based cell after 200 cycles at 0.5C. Furthermore, the decomposition product tetrafluoro-1,4-benzoquinone (TFBQ) of LTFBB dissolves in the electrolyte without generating gas or solid residues, avoiding common side effects associated with conventional designs. This LRS suppresses gas and solid residue formation during the pre-lithiation process, offering new opportunities in this field and demonstrating potential for industrial application.
2. Results and discussion
2.1 Direct regeneration strategy for degraded LFP using an LRS
The LRS-enabled regeneration of degraded LFP and its operational mechanism are schematically depicted in Fig. 1. The lithium compensation agent is coated onto one side of the commercial PPS to fabricate the LRS. Subsequently, the degraded LFP electrode is reassembled into a regenerated battery with a fresh graphite anode, simultaneously replacing the commercial PPS with the LRS (Fig. 1a). During the initial charging process, the high-capacity lithium compensation agent decomposes over a specific voltage range, releasing additional Li+ ions that can be inserted into the lattice of the LFP cathode during discharge (Fig. 1b). Consequently, after the initial cycle, the degraded LFP can theoretically be restored, allowing the regenerated Li-ion battery to recover its original capacity. | ||
| Fig. 1 Schematic illustration of (a) the direct regeneration strategy for degraded LFP batteries using the LRS and (b) Li+ intercalation into the LFP cathode during discharge. | ||
2.2 Synthesis and characterization of LTFBB
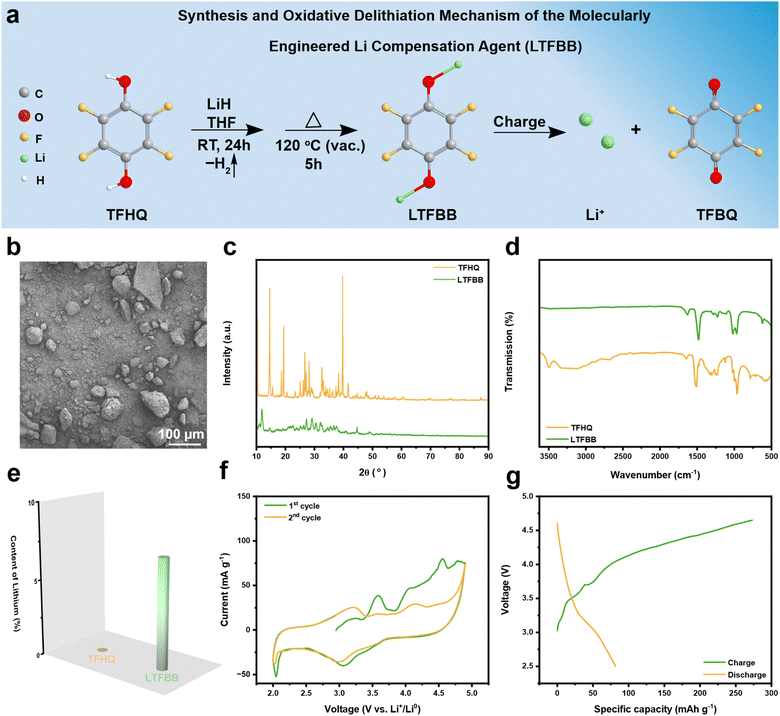
In a previous study,39 a 4-fluoro-1,2-dihydroxybenzene lithium salt, based on a catechol (1,2-dihydroxybenzene) scaffold, was employed as a multifunctional Li-compensation molecule. Fluorination of the catechol backbone facilitated the formation of a robust cathode/anode electrode interfaces (CEI/SEI) on the electrode surface, contributing to stable long-term cycling performance.39 Catechol is known to be more prone to oxidation than hydroquinone under ambient conditions.Here, an air-stable hydroquinone (1,4-dihydroxybenzene) scaffold is employed to construct a molecularly engineered lithium compensation agent. As shown in Fig. 2a, the dilithium salt of tetrafluorohydroquinone (TFHQ) is synthesized via LiH-mediated deprotonation of TFHQ in tetrahydrofuran (THF) at room temperature over 24 h, yielding para-diolate C6F4(OLi)2 (LTFBB; Fig. S1). This reaction can be interpreted in terms of Lewis acid–base theory,39–41 with LiH acting as a Lewis base by donating electron pairs and the hydroxyl groups in TFHQ serving as Lewis acids by accepting electrons through their oxygen atoms. Deprotonation generates two alkoxide intermediates (–OLi) and facilitates coordination of additional lithium ions, producing LTFBB, a stabilized dilithium salt in which both Li+ ions are effectively coordinated. Each LTFBB unit provides two lithium ions, while the four fluorine substituents exert strong inductive electron-withdrawing effects, finely tuning delithiation and oxidation potentials and enhancing chemical stability.42–44 Upon charging, LTFBB is oxidized to release Li+, compensating for active lithium loss and sustaining capacity during prolonged cycling. Furthermore, as reported in previous studies,39–41 the –OLi intermediates are converted into quinone through oxidation. Consequently, LTFBB is converted into TFBQ, which dissolves in the electrolyte and acts as a stabilizing additive.45,46
The as-prepared LTFBB exhibited a hierarchical micro–nano structure, in which the micron-sized secondary particles observed in Fig. 2b are assembled from both nano- and micron-sized primary particles (Fig. S1b). As shown in Fig. 2c, the position and intensity of the characteristic peaks in the powder X-ray diffraction (XRD) patterns changed significantly between TFHQ and LTFBB, indicating distinct crystal structures and confirming the formation of a new compound. The Fourier Transform Infrared (FTIR) spectra of LTFBB and TFHQ are shown in Fig. 2d. The disappearance of peaks in the 3000–3500 cm−1 range after the transformation from TFHQ to LTFBB suggests that the hydrogen atoms of the hydroxyl groups were replaced by lithium.39,41 Inductively Coupled Plasma-Optical Emission Spectroscopy (ICP-OES) was used to determine the Li content of TFHQ and LTFBB. As shown in Fig. 2e, LTFBB exhibits a Li content of 7.14% (Table S1), approaching the theoretical value of 7.2% (Table S2). Moreover, the Energy-Dispersive X-ray Spectroscopy (EDS) results in Fig. S2 show an F![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O weight ratio of 2.15, which is close to the theoretical ratio of 2.38, indicating that the as-synthesized LTFBB is essentially of high purity. X-ray photoelectron spectroscopy (XPS) elemental analysis was used to identify TFHQ and LTFBB. Furthermore, the Li 1s signal is observed for LTFBB in the XPS spectrum, whereas no corresponding signal is detected in the TFHQ spectrum (Fig. S3a). For LTFBB, a distinct and symmetric Li–O peak appears at approximately 56.4 eV (Fig. S3a and b), consistent with those reported for other lithium-containing organic compounds.39,40
O weight ratio of 2.15, which is close to the theoretical ratio of 2.38, indicating that the as-synthesized LTFBB is essentially of high purity. X-ray photoelectron spectroscopy (XPS) elemental analysis was used to identify TFHQ and LTFBB. Furthermore, the Li 1s signal is observed for LTFBB in the XPS spectrum, whereas no corresponding signal is detected in the TFHQ spectrum (Fig. S3a). For LTFBB, a distinct and symmetric Li–O peak appears at approximately 56.4 eV (Fig. S3a and b), consistent with those reported for other lithium-containing organic compounds.39,40
The electrochemical performance of LTFBB was evaluated by cyclic voltammetry (CV). Material conductivity plays a critical role in determining electrochemical behavior. For instance, highly conductive GeP5 (ref. 47) delivers exceptional reversible capacity, whereas poorly conductive Si48 exhibits low utilization. For LTFBB, its low conductivity is expected to hinder delithiation. To investigate this effect, three conductive additives, namely Super C65, Super C65 & mesoporous carbon (CMK), and carbon black-ketjenblack EC-600JD (KB), were employed to assess their impact on the delithiation potential. As shown in Fig. S4a, the CV profile of LTFBB with Super C65 exhibits two pronounced anodic peaks at approximately 3.7 and 4.64 V, extending to about 4.75 V, corresponding to Li+ extraction. A weak cathodic peak suggests that lithium removal is largely irreversible. In the second cycle, both anodic and cathodic features persist but are markedly diminished, indicating that most capacity loss occurs in the first cycle. Galvanostatic charge–discharge measurements corroborate this observation, revealing a charge-specific capacity of 200.3 mA h g−1 at 4.75 V, while the capacity is only 57.6 mA h g−1 at 4.3 V (Fig. S4b). This is significantly higher than the typical operating voltages of commercial cathodes such as LFP and lithium nickel cobalt manganese oxide (NCM), which are below 4.3 V.18
To enhance conductivity, LTFBB was combined with CMK in THF under an argon atmosphere to produce an LTFBB & CMK composite, which was further mixed with Super C65 for electrode preparation. The corresponding CV curve (Fig. S5a) shows a slight shift of the main anodic peak from 4.64 to approximately 4.59 V, with capacities of 261.5 and 68.5 mA h g−1 at 4.75 and 4.3 V (Fig. S5b), respectively, which are slightly higher than those obtained with Super C65 alone. When KB was used, the CV curve (Fig. 2f) displayed a reduced anodic peak at about 4.5 V, extending to approximately 4.65 V. The corresponding charge–discharge profiles (Fig. 2g) deliver a charge capacity of 273.2 mA h g−1 at 4.65 V, which is close to the theoretical specific capacity of 276.3 mA h g−1, and a charge capacity of 150.3 mA h g−1 at 4.3 V, with a corresponding discharge capacity of approximately 81.8 mA h g−1. The irreversible primary capacity of LTFBB demonstrates its suitability as a pre-lithiation additive. Compared with Super C65 and Super C65 & CMK, Fig. S6 demonstrates that KB as a conductive additive delivers the highest charge capacity at 4.3 V, indicating its strong potential for application in commercial cathode systems. Our findings are consistent with the previous report.18 Using KB as the conductive additive enables the prelithiation agent to exhibit a higher capacity and a lower delithiation potential.18 This improvement arises from the large specific surface area (∼1180 m2 g−1) of KB,18 which allows it to embed with LTFBB and form a more efficient electronically conductive network. Moreover, the high intrinsic conductivity of KB further facilitates charge transfer. Impressively, LTFBB retains 92.2% of its capacity (i.e., 251.8 mA h g−1) after 24 h of exposure to air at 50% relative humidity (RH) (Fig. S7), indicative of its high air stability. To further investigate the decomposition of LTFBB, we used ICP to determine the Li content of the LTFBB electrode after charging to 4.3 V. As shown in Fig. S8 and Table S3, about 3.12% of Li content remains in LTFBB, indicating that ∼56.3% of Li+ was extracted from the electrode and that LTFBB decomposed during charging. These results demonstrate that LTFBB can serve as an effective pre-lithiation agent.
2.3 Characterization and electrochemical performance of the LRS
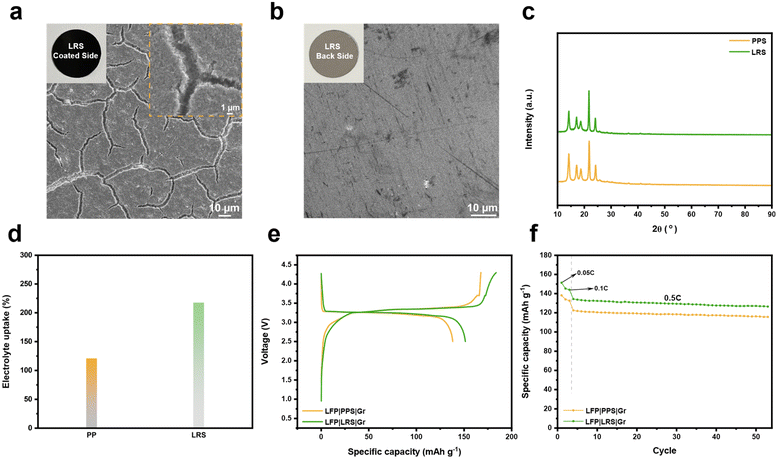
To verify the feasibility of using LTFBB as a pre-lithiation agent and to enable a facile pre-lithiation process, we coated it onto a commercial PPS as a functional layer to prepare the LRS, which served as an additional lithium source in the batteries. As shown in the insets of Fig. 3a, b and S9, the two sides of the LRS exhibit distinct appearances. The lithium-replenishment layer (LTFBB-coated side, the inset of Fig. 3a) appears black due to the uniform distribution of LTFBB and KB, whereas the opposite side (back side, the inset of Fig. 3b) retains the original smooth appearance of the separator. SEM imaging reveals multiple cracks on the coated side (Fig. 3a), originating from the high porosity of the KB conductive carbon network. The corresponding inset (Fig. 3a) further displays a coarse, granular morphology resulting from the LTFBB–KB coating. In contrast, the backside retains the original smooth surface of the separator, with no penetration of coating particles detected at the microscopic level (Fig. 3b), confirming that the separator's insulating properties remain unaffected. The XRD patterns (Fig. 3c) of the PPS and LRS in the 10–90° range and the zoomed region (Fig. S10) from 25° to 40° show no appreciable differences, as the peaks corresponding to the LTFBB loading are likely obscured by those of the PPS. XPS analysis was then performed to examine the chemical state of LTFBB after coating onto the PPS. A distinct Li–O peak at ∼56.4 eV (Fig. S11) indicates that the local chemical environment of lithium is largely preserved during the coating process. The wettability of the PPS and LRS toward the LP57 electrolyte is shown in Fig. S12. The contact angles of the PPS and LRS are 59 ± 1° and 18 ± 1°, respectively. The significantly lower contact angle of the LRS indicates its superior electrolyte wettability. Fig. 3d presents the electrolyte absorption behavior of the LRS. After being fully immersed in LP57 electrolyte, the LRS exhibits markedly enhanced electrolyte uptake capacity compared with the PPS. This enhancement is attributed to the lithium compensation agent coating with high specific surface area KB on the PPS, enabling the LRS to achieve an electrolyte uptake of ∼217%, significantly higher than the ∼121% observed for the PPS. Furthermore, the LRS exhibited a higher porosity of 79.5% compared to 45% for the PPS (Fig. S13). The increased electrolyte uptake and porosity may enhance ion transport. As shown in Fig. S13, the LRS shows a higher ionic conductivity of 1.23 mS cm−1, compared to 0.55 mS cm−1 for the PPS.Active lithium loss during the initial cycle substantially reduces the energy density of lithium-ion batteries. To address this issue, a LRS was employed as an external lithium source to compensate for the initial lithium loss, providing a straightforward strategy for lithium replenishment.21,22,32 The electrochemical performance of the LRS was evaluated in full cells comprising fresh LFP as the cathode and graphite as the anode. Fig. S14 presents the initial charge–discharge profiles of pristine LFP‖Gr full cells with the PPS or LRS loaded with ∼0.15 mg cm−2 LTFBB. The LRS-equipped cell exhibits higher initial charge and discharge capacities than the conventional PPS cell (Fig. S14a). Specifically, the PPS cell delivers a charge capacity of 167.7 mA h g−1 and a discharge capacity of 138.1 mA h g−1, whereas the LRS-containing cell achieves a charge capacity of 176.5 mA h g−1 and a discharge capacity of 146.3 mA h g−1, corresponding to increases of 8.8 and 8.1 mA h g−1, respectively. This improvement is ascribed to the effective compensation of initial lithium loss by LTFBB. The LRS cell further demonstrates enhanced cycling stability, retaining 6% higher capacity than the PPS cell after 50 cycles at 0.5C (Fig. S14b). Charge–discharge profiles (Fig. S14c and d) collected at 0.05C, 0.1C, and 0.5C throughout cycling indicate that the capacity enhancement of the LRS persists across all tested current rates. Upon increasing the LTFBB loading on the LRS to ∼0.3 mg cm−2, the benefits of the LRS are further amplified. The corresponding full cell delivers a charge capacity of 183.8 mA h g−1 and a discharge capacity of 151.3 mA h g−1, representing improvements of 16.1 and 13.2 mA h g−1 relative to the PPS cell (Fig. 3e). The LRS-equipped cell also exhibits superior cycling performance, retaining 9.3% higher capacity than the PPS cell after 50 cycles at 0.5C (Fig. 3f). Compared with the PPS (Fig. S14d), charge–discharge profiles collected at 0.05C, 0.1C, and 0.5C during the 50th cycle (Fig. S15) indicate that the LRS consistently enhances both charge and discharge capacities across all tested current rates. The coulombic efficiency (Fig. S16) remains nearly unchanged for both the PPS cell and LRS cells, suggesting that the capacity improvement in the LRS cell mainly originates from the additional lithium supplied to the cell. Furthermore, the XRD patterns (Fig. S17a) of the pristine LRS and cycled LRS remain nearly unchanged. Moreover, the LRS retains an identical morphology before (Fig. 3a) and after cycling (Fig. S17b), demonstrating that the functionalization preserves its structural integrity upon delithiation.
2.4 Electrochemical evaluation of the regeneration strategy using the LRS
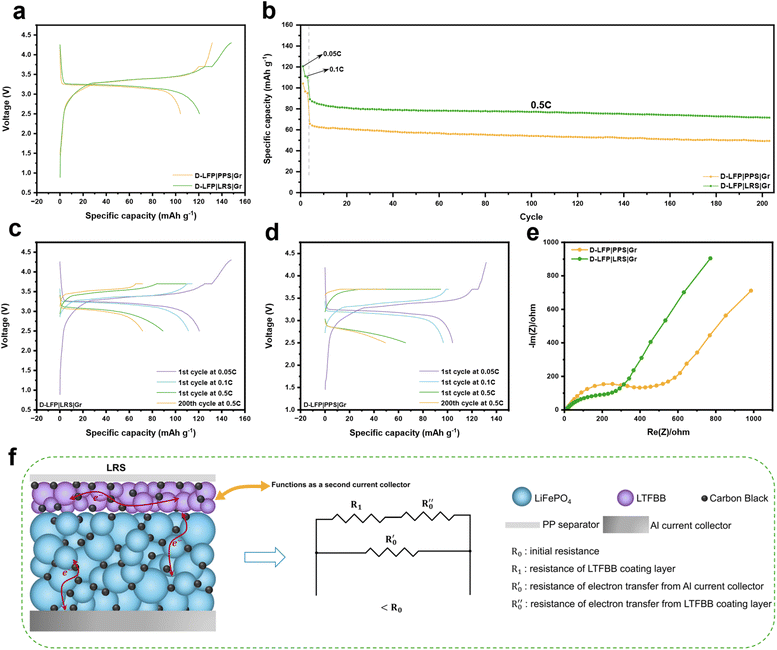
The promising capacity-enhancing effect of the LRS in fresh LFP‖Gr full cells motivated us to investigate its impact on D-LFP electrodes. We examined the electrochemical performance of full cells based on a D-LFP cathode and fresh graphite anode with or without the LRS. As shown in Fig. 4a, the initial charge capacity of the D-LFP‖Gr full cell is only 131.6 mA h g−1 and the corresponding discharge capacity is 104.5 mA h g−1 at 0.05C, likely due to lithium loss, oxidation of the LFP electrode, or other degradation mechanisms. In contrast, the regenerated cell incorporating the LRS with ∼0.4 mg cm−2 LTFBB achieves a significantly higher initial charge capacity of 148.2 mA h g−1, attributed to Li+ compensation from the decomposition of LTFBB. The additional lithium supplied by LTFBB restores the discharge capacity of D-LFP to 120.6 mA h g−1. After 200 cycles at 0.5C (Fig. 4b), the regenerated cell exhibits a 44.9% capacity improvement compared to the cell with the conventional PPS. Specifically, the LRS-based regenerated cell reaches 71.6 mA h g−1, whereas the PPS-based cell delivers 49.4 mA h g−1. Furthermore, the LRS-based cell (Fig. 4c) retains higher charge/discharge capacities than the PPS-based cell (Fig. 4d) across increasing rates of 0.05C, 0.1C, and 0.5C. Electrochemical impedance spectra (EIS) of LRS and PPS cells after formation (Fig. 4e and S18) reveal that the LRS cell has lower resistance, possibly due to enhanced electron transfer facilitated by the conductive separator acting as a secondary current collector (Fig. 4f) compared with the PPS (Fig. S19). This LRS could promote more efficient electron transport and active material utilization, leading to reduced resistance and improved capacity recovery.32Increasing the LTFBB loading on the LRS to ∼0.65 mg cm−2 (Fig. S20) increases the charge and discharge capacities to 160.7 and 126.7 mA h g−1, respectively. After 100 cycles at 0.5C, the regenerated cell shows a 51% capacity improvement over the PPS-based cell. Further increasing the LTFBB loading to ∼1 mg cm−2 (Fig. S21) boosts the charge and discharge capacities to 172.4 and 131.3 mA h g−1, respectively. However, excessive LTFBB loading on the LRS may bring adverse effects to the battery, causing faster capacity fade at 0.5C. As shown in Fig. S22a, the PPS clearly exhibits its internal pores. In comparison, the main part (Fig. S22b and c) of the LTFBB coating on the LRS with ∼1.0 mg cm−2 LTFBB loading not only covers the LRS pores, but the cracked regions (Fig. S22d) of the LRS also cover the pores of the PPS. With increasing LTFBB loading (i.e., thicker coating), more nano- to micro-sized particles accumulate on the separator, which increases the ion transport path and likely causes the capacity decay observed in the LRS at high LTFBB loadings. Besides, the ionic conductivity of the LRS (Fig. S23) decreases from 1.703 mS cm−1 for ∼0.4 mg cm−2 LTFBB to 1.108 mS cm−1 for ∼1.0 mg cm−2 LTFBB, reflecting the hindered ion transport caused by the thicker LTFBB coating.
Although the D-LFP cell could not be fully restored to its ideal capacity due to the limited LTFBB capacity in the voltage window (≤4.3 V), restricted LTFBB loading, and the inability of Li+ to intercalate into degraded LFP electrodes, the results demonstrate the effectiveness of in situ electrochemical regeneration for D-LFP electrodes. The capacity fading of D-LFP may arise from cycling-induced micro-cracks in larger LFP grains,49,50 loss of crystallinity and the active material,49,51 and carbon aggregation and agglomeration.52–54 As shown in Fig. S24, the (002) plane of conductive carbon is observed in the XRD pattern of the degraded LFP electrode, whereas it is absent in fresh LFP powder and fresh LFP electrodes. This carbon peak likely originates from carbon aggregation, which further leads to a reduction in the effective active material and consequently inhibits the capacity recovery of the D-LFP electrodes during the prelithiation process. This strategy provides a promising route for directly regenerating degraded electrodes via the LRS by incorporating a molecularly engineered Li compensation agent with high theoretical specific capacity, which merits further exploration.
2.5 Electrochemical evaluation of the byproduct of the LRS
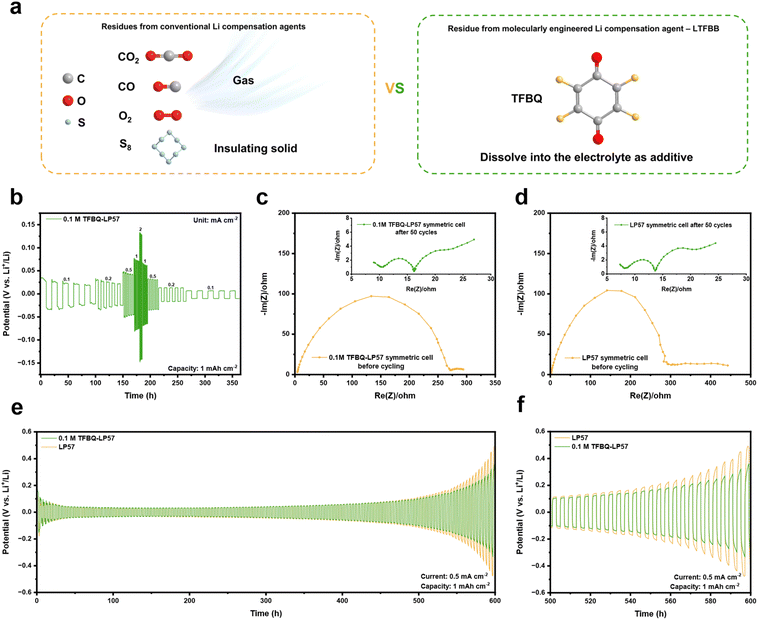
Compared with previously reported conventional LRSs, which often generate gaseous products (e.g., CO2,34 CO,32 and O2 (ref. 36)) or solid residues (e.g., S8)28 during cycling (Fig. 5a), the developed LRS based on LTFBB operates without gas evolution or solid by-products. Notably, the reaction by-product TFBQ is highly soluble in ether- and ester-based electrolytes.45 On the anode of the LFP‖Gr full cell, similar to the previous report,39 the by-product TFBQ participates in the redox reaction, during which the C–F bonds are cleaved, and the released F− combines with Li+ to form LiF while generating C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O functionalities.45 As shown in Fig. S25a and b, the F 1s XPS spectrum reveals a higher LiF content in the LRS-cycled cell (∼98.8%) compared with the PPS-cycled cell (∼73.7%). This increased LiF content contributes to reinforcing the SEI and mitigating lithium dendrite formation.39,45,55 Furthermore, increased C
O functionalities.45 As shown in Fig. S25a and b, the F 1s XPS spectrum reveals a higher LiF content in the LRS-cycled cell (∼98.8%) compared with the PPS-cycled cell (∼73.7%). This increased LiF content contributes to reinforcing the SEI and mitigating lithium dendrite formation.39,45,55 Furthermore, increased C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups are observed in the LRS-cycled cell, as indicated by the C 1s (Fig. S25c and d) XPS spectra (∼19.2% vs. ∼14.3%). These C
O groups are observed in the LRS-cycled cell, as indicated by the C 1s (Fig. S25c and d) XPS spectra (∼19.2% vs. ∼14.3%). These C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups enhance ionic conduction at the electrolyte–electrode interface, while their higher bond energy helps suppress electrolyte oxidation.39
O groups enhance ionic conduction at the electrolyte–electrode interface, while their higher bond energy helps suppress electrolyte oxidation.39
Furthermore, lithium metal batteries offer ultrahigh energy density owing to the high capacity and low redox potential of lithium metal anodes, but their practical application is hindered by severe lithium dendrite growth and poor cycling stability.56 In our previous work,45 TFBQ showed high solubility in 1,3-dioxolane (DOL)/1,2-dimethoxyethane (DME) with lithium bis(trifuoromethanesulfonyl)imide (LiTFSI) electrolyte and improved cell stability and cycle life in lithium metal batteries by suppressing lithium dendrite formation on the anode. Moreover, TFBQ is also well soluble in the LP57 electrolyte (Fig. S26), which is the base electrolyte employed in this work. As shown in Table S4, the concentration of TFBQ generated during the LTFBB decomposition in the electrolyte was ∼7.46, 14.9, 19.9, 32.7, and 50.1 mM for the LRS with LTFBB loadings of ∼0.15, 0.3, 0.4, 0.65, and 1 mg cm−2, respectively. And following our previous work,45 0.1 M TFBQ was added to LP57 to prepare the 0.1 M TFBQ–LP57 electrolyte. As shown in Fig. 5b, the Li–Li symmetrical cell with 0.1 M TFBQ–LP57 exhibits potentials of ∼22/25/46/75/138/64/34/16/10 mV at current densities of 0.1/0.2/0.5/1/2/1/0.5/0.2/0.1 mA cm−2, respectively. In comparison, the cell with LP57 shows lower polarization, with potentials of ∼22/23/37/56/92/48/28/14/9 mV under the same conditions (Fig. S27), indicating that the presence of TFBQ slightly increases overpotential. The initial interfacial resistance of the cell with 0.1 M TFBQ–LP57 after 24 h of rest is relatively large (Fig. 5c) but decreases to ∼20 Ω after 50 cycles (inset of Fig. 5c and S28a), which is comparable to that of the LP57 cell (inset of Fig. 5d and S28b). At a current density of 0.5 mA cm−2 and capacity of 1 mA h cm−2, the Li–Li symmetrical cell with 0.1 M TFBQ–LP57 exhibits a higher potential than the LP57 cell during the first 50 cycles (Fig. 5e), attributed to the additive and the incomplete formation of the F-rich protective layer on the Li surface. After 500 h, the TFBQ-containing cell clearly exhibits a lower potential than the LP57 cell (Fig. 5f), suggesting that the F-rich protective layer effectively suppresses dendrite growth. A similar trend is observed at a higher current density of 2 mA cm−2 (Fig. S29a and b). Notably, from 50 h to 80 h, the cell with 0.1 M TFBQ–LP57 consistently retains a lower overpotential than the LP57 cell. Although TFBQ slightly increases polarization across 0.1–2 mA cm−2, its long-term effect is beneficial, as the cell demonstrates reduced overpotential after 50 h of cycling at 2 mA cm−2. This finding demonstrates a promising approach to utilize molecularly engineered LTFBB as a lithium compensation agent for mitigating active lithium loss in lithium-ion batteries without generating gas or solid byproducts, with residual TFBQ concurrently serving as a stabilizing electrolyte additive.
3. Conclusions
A functional lithium replenishment separator incorporating a molecularly engineered organic lithium compensation agent, LTFBB, has been developed to regenerate D-LFP. LTFBB was synthesized via a simple chemical reaction using LiH and TFHQ. The areal mass loading of LTFBB on the PPS can be precisely adjusted, enabling controlled Li+ release to replenish the capacity of LFP batteries. Consequently, LTFBB exhibited significant effects in compensating for the initial lithium loss when incorporated into a pristine LFP full cell. The pristine LFP full cell with the LRS shows a 9.3% higher capacity than the LFP full cell with the PPS. Impressively, the D-LFP‖Gr full cell incorporating the LRS achieves a 44.9% higher overall capacity than the PPS-based cell after 200 cycles at 0.5C. This as-made molecularly engineered LRS provides a promising and simple regeneration strategy for lithium battery recovery, broadening the horizons of the existing industrial battery recycling process. It is promising to anticipate that increased research efforts on this molecularly engineered strategy-based LRS, which does not produce gaseous products or solid residues in the battery, will contribute to advancing the regeneration of degraded lithium-ion batteries.4. Methods
4.1 Materials
Tetrafluorohydroquinone (TFHQ), tetrafluoro-1,4-benzoquinone (TFBQ), anhydrous tetrahydrofuran (THF), mesoporous carbon (CMK), N-methyl-2-pyrrolidone (NMP) and isopropyl alcohol (IPA) were purchased from Sigma-Aldrich Corporation. Lithium hydride (LiH) was obtained from Thermo Fisher Scientific. Carbon black-ketjenblack EC-600JD (KB) was provided by the Fuel Cell Store. Super C65 conductive carbon black and LA133 were sourced from MSE supplies. The PP separator (PPS) was procured from Celgard LLC. The LiFePO4, Gr powder, and polyvinylidene fluoride (PVDF) were acquired from MTI Corporation. The degraded LFP electrode, with a loading of approximately 6 mg cm−2, was supplied by MTI Corporation. Its capacity had degraded to less than 80% of the original capacity and it had been exposed to air for an extended period. All chemicals and solvents were used as received without any further purification.4.2 Lithium salt synthesis
The lithium salt was synthesized following a previously reported method.39–41 All preparations were conducted under an argon atmosphere at ambient temperature. 1 mmol of TFHQ was dissolved in 2 mL of anhydrous THF in a glass vial and stirred for 10 minutes to ensure complete dissolution. Subsequently, 2 mmol LiH was introduced to the transparent solution, and the resulting mixture was stirred continuously for 24 hours. The resulting product, lithium 2,3,5,6-tetrafluorobenzene-1,4-bis(olate) (LTFBB), was obtained after being dried under vacuum at 120 °C for 5 hours in an oven.4.3 Preparation of the LTFBB electrode and LRS
The LTFBB electrodes for cyclic voltammetry (CV) and capacity testing were prepared by mixing LTFBB, conductive carbon, and PVDF binder in a weight ratio of 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, using NMP as the solvent. The conductive carbons used were Super C65, a Super C65 & CMK mixture, and KB, respectively. For LTFBB electrodes containing a mixture of Super C65 and CMK conductive carbons, LTFBB and CMK were first mixed in THF inside a glovebox to form the LTFBB & CMK composite, which was then dried in a vacuum oven at 55 °C. This composite was subsequently mixed with Super C65 and PVDF to obtain a final composition of 60% LTFBB, 15% CMK, 15% Super C65, and 10% PVDF. The resulting slurry was cast onto aluminum foil and dried under vacuum at 55 °C for 12 h. The LTFBB electrodes had an areal loading of approximately 1 mg cm−2 with a diameter of ∼12 mm. The LRS was fabricated by mixing 60 wt% LTFBB, 30 wt% KB, and 10 wt% PVDF in NMP to obtain a homogeneous slurry. The slurry was cast onto the PPS using a doctor blade and subsequently dried in a vacuum oven at 55 °C for 12 h. The LTFBB loading was adjusted by varying the coating thickness with the doctor blade. After drying, the LRS was punched into 16 mm-diameter disks for full-cell assembly. Finally, by adjusting the doctor-blade gap to increase the thickness of the LTFBB coating, lithium replenishment separators (LRSs) with different areal loadings of LTFBB (i.e., ∼0.15, 0.3, 0.4, 0.65, and 1 mg cm−2) were prepared.
1, using NMP as the solvent. The conductive carbons used were Super C65, a Super C65 & CMK mixture, and KB, respectively. For LTFBB electrodes containing a mixture of Super C65 and CMK conductive carbons, LTFBB and CMK were first mixed in THF inside a glovebox to form the LTFBB & CMK composite, which was then dried in a vacuum oven at 55 °C. This composite was subsequently mixed with Super C65 and PVDF to obtain a final composition of 60% LTFBB, 15% CMK, 15% Super C65, and 10% PVDF. The resulting slurry was cast onto aluminum foil and dried under vacuum at 55 °C for 12 h. The LTFBB electrodes had an areal loading of approximately 1 mg cm−2 with a diameter of ∼12 mm. The LRS was fabricated by mixing 60 wt% LTFBB, 30 wt% KB, and 10 wt% PVDF in NMP to obtain a homogeneous slurry. The slurry was cast onto the PPS using a doctor blade and subsequently dried in a vacuum oven at 55 °C for 12 h. The LTFBB loading was adjusted by varying the coating thickness with the doctor blade. After drying, the LRS was punched into 16 mm-diameter disks for full-cell assembly. Finally, by adjusting the doctor-blade gap to increase the thickness of the LTFBB coating, lithium replenishment separators (LRSs) with different areal loadings of LTFBB (i.e., ∼0.15, 0.3, 0.4, 0.65, and 1 mg cm−2) were prepared.
4.4 Cell assembly
The half and full cells were assembled into 2032-type coin cells in an argon-filled glovebox (H2O and O2 < 0.01 ppm). The LTFBB half cells, used for capacity testing, comprised an LTFBB cathode, a PPS, electrolyte, and a lithium metal anode. For the LFP‖Gr full cells, LFP electrodes were prepared by mixing LFP, Super C65 conductive carbon, and PVDF binder in a weight ratio of 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 using NMP as the solvent. Graphite electrodes were fabricated by blending graphite powder, Super C65 conductive carbon, and LA133 binder in a weight ratio of 94
5 using NMP as the solvent. Graphite electrodes were fabricated by blending graphite powder, Super C65 conductive carbon, and LA133 binder in a weight ratio of 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. The slurries were cast onto aluminum foil (LFP) and copper foil (graphite) and then dried under vacuum at 120 °C for 12 h. The LFP electrodes had an areal loading of approximately 5 mg cm−2 with a diameter of ∼12 mm, while the graphite electrodes had a loading of approximately 2.7 mg cm−2 and a diameter of ∼14 mm. The negative-to-positive (N/P) capacity ratio was ∼1.1. The electrolyte (LP57) comprised 1 M lithium hexafluorophosphate (LiPF6) dissolved in a 3
4. The slurries were cast onto aluminum foil (LFP) and copper foil (graphite) and then dried under vacuum at 120 °C for 12 h. The LFP electrodes had an areal loading of approximately 5 mg cm−2 with a diameter of ∼12 mm, while the graphite electrodes had a loading of approximately 2.7 mg cm−2 and a diameter of ∼14 mm. The negative-to-positive (N/P) capacity ratio was ∼1.1. The electrolyte (LP57) comprised 1 M lithium hexafluorophosphate (LiPF6) dissolved in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 (v/v) mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC), with 70 µL added per coin cell. Cells were assembled in an argon-filled glove box with moisture and oxygen levels maintained below 0.01 ppm. For the LRS-based full cell, the as-prepared LRS was oriented with its coated side facing the LFP cathode. For the Li–Li symmetrical cells, 0.1 M TFBQ–LP57 and pristine LP57 were used as the electrolytes, with 50 µL added to each coin cell.
7 (v/v) mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC), with 70 µL added per coin cell. Cells were assembled in an argon-filled glove box with moisture and oxygen levels maintained below 0.01 ppm. For the LRS-based full cell, the as-prepared LRS was oriented with its coated side facing the LFP cathode. For the Li–Li symmetrical cells, 0.1 M TFBQ–LP57 and pristine LP57 were used as the electrolytes, with 50 µL added to each coin cell.
4.5 Material characterization
Morphological characterization and Energy-Dispersive X-ray Spectroscopy (EDS) analyses were performed using a JEOL 7000F scanning electron microscope. Crystal structures of the LTFBB precursor, LTFBB, and LRS were analyzed by X-ray diffraction (Bruker D8 Advance) with Cu Kα radiation in Bragg–Brentano geometry over a 2θ range of 10° to 90°. Fourier transform infrared spectra were obtained with a Bruker Vertex 70 FTIR spectrometer, and X-ray photoelectron spectroscopy (XPS) was performed using an ULVAC-PHI PHI 5000 VersaProbe II system. The Inductively Coupled Plasma-Optical Emission Spectroscopy (ICP-OES) analyses were performed using a PerkinElmer Optima 8000 CROSS FLOW. ICP samples were prepared by either dissolving the materials directly in a 2.0 wt% nitric acid aqueous solution or by dissolving them in aqua regia (a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 v/v mixture of concentrated nitric and hydrochloric acids) followed by dilution with 2.0 wt% nitric acid. The contact angle was measured using an optical contact angle goniometer (DataPhysics Instruments/OCA11).
3 v/v mixture of concentrated nitric and hydrochloric acids) followed by dilution with 2.0 wt% nitric acid. The contact angle was measured using an optical contact angle goniometer (DataPhysics Instruments/OCA11).
4.6 Calculation methodology
The electrolyte uptake (U, %) of the separator was determined by weighing the separator before and after immersion in the electrolyte for 24 h and calculated using the following equation:32where mbefore and mafter are the weights of the separator before and after immersion in the electrolyte, respectively.
The porosity of the separator was measured using IPA absorption analysis,57 which involved determining the dry and wet weights of the separator samples before and after soaking in IPA. The separator samples were soaked in IPA for 1 h. The porosity (ε, %) was calculated according to the following equation:21,58
The ionic conductivity (σ) of the separator in the electrolyte was measured by electrochemical impedance spectroscopy (EIS) using a stainless-steel symmetric cell. The ionic conductivity was calculated according to the following equation:22,32
4.7 Electrochemical characterization
Electrochemical tests were performed using a LAND battery testing system at 25 °C. All cells were rested for 24 h prior to testing. The LFP‖Gr full cell was first charged to 3.7 V at 0.05C (1C = 154 mA h g−1 for fresh LFP and 1C = 150 mA h g−1 for degraded LFP), followed by a constant voltage-constant current (CVC) step, and then charged to 4.3 V at a lower current density of 0.02C to decompose the LTFBB. Capacity testing of the LTFBB electrode was carried out using the same procedure. Rate performance of Li–Li symmetrical cells was evaluated at 0.1, 0.2, 0.5, 1, and 2 mA cm−2, with a fixed capacity of 1 mA h cm−2 at each current density, and 0.5 and 2 mA cm−2 were selected for cycling tests. CV and electrochemical impedance spectroscopy (EIS) measurements were conducted on an electrochemical analyzer (Bio-Logic SAS VMP3). EIS measurements of LFP‖Gr full cells and Li–Li symmetrical cells were conducted over frequency ranges of 100 kHz to 0.1 Hz and 100 kHz to 0.01 Hz, respectively, with a 10 mV voltage amplitude.Author contributions
F. Tao and Z. Yao conducted the experiments and wrote the manuscript. J. Hou and Z. Wang performed the ICP measurements. Z. Yang carried out the XPS characterization. Y. Wang supervised this research project.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: a photo and the SEM image of LTFBB; EDS spectrum of LTFBB; XPS spectra of TFHQ and LTFBB; CV curves of the LTFBB electrode with Super C65 and Super C65 & CMK, respectively; galvanostatic charge–discharge profiles of the LTFBB electrode with Super C65, Super C65 & CMK and KB as the conductive carbon, respectively; photos of the LRS; XRD patterns of the PPS and LRS over the region from 25° to 40°; XPS spectrum of the LRS; electrochemical performance of LFP‖Gr full cells with LRS ∼ 0.15 mg cm−2 LTFBB and PPS; charge–discharge profiles of the full cells with LRS ∼ 0.3 mg cm−2 LTFBB at various cycles; SEM image of the LRS after cycling; EIS spectra for D-LFP‖Gr full cells with the LRS or PPS after formation; schematic illustration of the effect of the PPS on resistance; electrochemical performance of D-LFP‖Gr full cells with LRS ∼ 0.65 mg cm−2 LTFBB and PPS; electrochemical performance of D-LFP‖Gr full cells with LRS ∼ 1 mg cm−2 LTFBB and PPS; photos of 0.1 M TFBQ–LP57 electrolyte and LP57 at different viewing angles; galvanostatic rate performance of LP57 symmetrical cells; EIS spectra of 0.1 M TFBQ–LP57 and LP57 symmetrical cells, before and after the first 50 cycles; galvanostatic cycling performance of LP57 and 0.1 M TFBQ–LP57 symmetrical cells at 2 mA cm−2/1 mA h cm−2; calculation of Li content of LTFBB; theoretical atomic (C, O, F, and Li) weight of LTFBB; calculation of Li content of LTFBB after decomposition and the decomposition percentage of LTFBB during charging to 4.3 V. See DOI: https://doi.org/10.1039/d5ta09041e.Acknowledgements
The authors would like to acknowledge the financial support from the U. S. Army Research Laboratory through their award # W911NF-22-2-0040. Z. Y. acknowledges the support from the Post Test Facility of Argonne National Laboratory, supported by the U. S. DOE, Vehicle Technologies office, under contract number DE-AC02-06CH11357.References
- J. M. Tarascon and M. Armand, Nature, 2001, 414, 359–367 CrossRef CAS PubMed.
- M. Armand and J. M. Tarascon, Nature, 2008, 451, 652–657 CrossRef CAS PubMed.
- X. Chen, W. Li, C. Luo, H. Zhang, C. Gao, C. Sun, R. Wu, Y. Gong, P. Mu, Z. Lv and G. Cui, Adv. Mater., 2025, 37, e04360 CrossRef CAS PubMed.
- M. Li, H. Liu, Z. Cheng, J. He, H. Li, L. Zhang, T. Liu, X. Wang, P. Wang, Z. Liu and G. Cui, Adv. Energy Mater., 2025, 15, 2405766 CrossRef CAS.
- Y. Sun, H.-W. Lee, Z. W. Seh, N. Liu, J. Sun, Y. Li and Y. Cui, Nat. Energy, 2016, 1, 15008 CrossRef CAS.
- Y. Sun, H.-W. Lee, Z. W. Seh, G. Zheng, J. Sun, Y. Li and Y. Cui, Adv. Energy Mater., 2016, 6, 1600154 CrossRef.
- Y. Sun, H.-W. Lee, G. Zheng, Z. W. Seh, J. Sun, Y. Li and Y. Cui, Nano Lett., 2016, 16, 1497–1501 CrossRef CAS.
- J. Sun, L. Huang, G. Xu, S. Dong, C. Wang and G. Cui, Mater. Today, 2022, 58, 110–118 CrossRef CAS.
- R. Zhan, X. Wang, Z. Chen, Z. W. Seh, L. Wang and Y. Sun, Adv. Energy Mater., 2021, 11, 2101565 CrossRef CAS.
- J. Choi, H. Jeong, J. Jang, A. R. Jeon, I. Kang, M. Kwon, J. Hong and M. Lee, J. Am. Chem. Soc., 2021, 143, 9169–9176 CrossRef CAS.
- Y. L. Cheah, V. Aravindan and S. Madhavi, J. Electrochem. Soc., 2013, 160, A1016 CrossRef CAS.
- Z. Liu, S. Ma, X. Mu, R. Li, G. Yin and P. Zuo, ACS Appl. Mater. Interfaces, 2021, 13, 11985–11994 CrossRef CAS PubMed.
- P. Vanaphuti, L. Su and A. Manthiram, Small Methods, 2024, 8, 2301159 CrossRef CAS.
- M. Cao, Z. Liu, X. Zhang, L. Yang, S. Xu, S. Weng, S. Zhang, X. Li, Y. Li, T. Liu, Y. Gao, X. Wang, Z. Wang and L. Chen, Adv. Funct. Mater., 2023, 33, 2210032 CrossRef CAS.
- X.-Y. Yue, Y.-X. Yao, J. Zhang, S.-Y. Yang, Z. Li, C. Yan and Q. Zhang, Adv. Mater., 2022, 34, 2110337 CrossRef CAS.
- C. Li, H. Du, Y. Kang, Y. Zhao, Y. Tian, J. Wozny, J. Lu, T. Li, N. Tavajohi, M. Huang, B. Lan, F. Kang and B. Li, Next Sustain., 2023, 1, 100008 CrossRef.
- B. Zhu, W. Zhang, Q. Wang, Y. Lai, J. Zheng, N. Wen and Z. Zhang, Adv. Funct. Mater., 2024, 34, 2315010 CrossRef CAS.
- G. Huang, J. Liang, X. Zhong, H. Liang, C. Cui, C. Zeng, S. Wang, M. Liao, Y. Shen, T. Zhai, Y. Ma and H. Li, Nano Res., 2023, 16, 3872–3878 CrossRef CAS.
- H. Park, T. Yoon, Y.-U. Kim, J. H. Ryu and S. M. Oh, Electrochim. Acta, 2013, 108, 591–595 CrossRef CAS.
- B. Huang, T. Huang, L. Wan and A. Yu, ACS Sustain. Chem. Eng., 2021, 9, 648–657 CrossRef CAS.
- Z. Rao, J. Wu, B. He, W. Chen, H. Wang, Q. Fu and Y. Huang, ACS Appl. Mater. Interfaces, 2021, 13, 38194–38201 Search PubMed.
- G. Lin, T. Meng, Y. Peng, P. Li and X. Hu, Small Methods, 2025, 9, 2401133 CrossRef CAS.
- S. Chen, G. Wu, H. Jiang, J. Wang, T. Chen, C. Han, W. Wang, R. Yang, J. Zhao, Z. Tang, X. Gong, C. Li, M. Zhu, K. Zhang, Y. Xu, Y. Wang, Z. Hu, P. Chen, B. Wang, K. Zhang, Y. Xia, H. Peng and Y. Gao, Nature, 2025, 638, 676–683 CrossRef CAS PubMed.
- K. Jun, L. Kaufman, W. Jung, B. Park, C. Jo, T. Yoo, D. Lee, B. Lee, B. D. McCloskey, H. Kim and G. Ceder, Adv. Energy Mater., 2023, 13, 2301132 CrossRef CAS.
- Y. Bie, J. Yang, J. Wang, J. Zhou and Y. Nuli, Chem. Commun., 2017, 53, 8324–8327 RSC.
- Y. Sun, Y. Li, J. Sun, Y. Li, A. Pei and Y. Cui, Energy Storage Mater., 2017, 6, 119–124 Search PubMed.
- Q. Nie, W. Wan, Y. Mei, G. Qu, Z. Liu, C. Tie, J. Zhu, C. Ji, J. Chen, C. Wang and Y. Huang, Adv. Funct. Mater., 2025, 35, 2422034 Search PubMed.
- Y. Zhan, H. Yu, L. Ben, Y. Chen and X. Huang, Electrochim. Acta, 2017, 255, 212–219 CrossRef CAS.
- Y. Fu, Y. Xie, L. Zeng and Z. Shi, Surf. Interfaces, 2023, 36, 102610 Search PubMed.
- X. Wang, C. Liu, S. Zhang, H. Wang, R. Wang, Y. Li and J. Sun, ACS Appl. Energy Mater., 2021, 4, 5246–5254 CrossRef CAS.
- Q. Meng, M. Fan, X. Chang, H. Li, W.-P. Wang, Y.-H. Zhu, J. Wan, Y. Zhao, F. Wang, R. Wen, S. Xin and Y.-G. Guo, Adv. Energy Mater., 2023, 13, 2300507 CrossRef CAS.
- G. Liu, W. Wan, Q. Nie, C. Zhang, X. Chen, W. Lin, X. Wei, Y. Huang, J. Li and C. Wang, Energy Environ. Sci., 2024, 17, 1163–1174 RSC.
- L. Du, G. Xu, Z. Zhang, X. Du, G. Kang, Y.-H. Zhang, B. Xie, X. Zhuang, J. Wang, T. Dong, C. Li, Q. Zhang, J. Ju, H. Zhang, S. Dong, G. Zhou and G. Cui, ACS Energy Lett., 2025, 10, 3941–3951 Search PubMed.
- M. Fan, Q. Meng, X. Chang, C.-F. Gu, X.-H. Meng, Y.-X. Yin, H. Li, L.-J. Wan and Y.-G. Guo, Adv. Energy Mater., 2022, 12, 2103630 CrossRef CAS.
- X. Chang, M. Fan, B. Yuan, W.-H. He, C.-F. Gu, C. Li, Q. Meng and Y.-G. Guo, Angew. Chem., Int. Ed., 2024, 63, e202406557 CrossRef CAS PubMed.
- N. Yao, F. Liu, A. Shao, R. Xue, Q. Jia, Y. Liu, H. Wang, X. Wang, Y. Zhang, M. Zhang, Z. Wang, Y. Li, J. Tang, X. Tang and Y. Ma, Adv. Mater., 2024, 36, 2408268 CrossRef CAS.
- M. K. Tran, M.-T. F. Rodrigues, K. Kato, G. Babu and P. M. Ajayan, Nat. Energy, 2019, 4, 339–345 CrossRef CAS.
- M. Shan, C. Dang, K. Meng, Y. Cao, X. Zhu, J. Zhang, G. Xu and M. Zhu, Mater. Today, 2024, 73, 130–150 CrossRef CAS.
- W. Wu, A. Wang, Q. Zhan, Z. Hu, W. Tang, L. Zhang and J. Luo, Small, 2023, 19, 2301737 CrossRef CAS PubMed.
- D. Wang, Z. Zhang, B. Hong and Y. Lai, Chem. Commun., 2019, 55, 10737–10739 RSC.
- P. Jeżowski, O. Crosnier, E. Deunf, P. Poizot, F. Béguin and T. Brousse, Nat. Mater., 2018, 17, 167–173 Search PubMed.
- T. Siodła, W. P. Ozimiński, M. Hoffmann, H. Koroniak and T. M. Krygowski, J. Org. Chem., 2014, 79, 7321–7331 Search PubMed.
- J. T. Gozdalik, A. Adamczyk-Woźniak and A. Sporzyński, Pure Appl. Chem., 2018, 90, 677–702 CrossRef CAS.
- Q. Guo, H. Xu, X. Chu, X. Huang, M. Yu and X. Feng, Chem. Soc. Rev., 2025, 54, 4035–4086 RSC.
- Z. Yao, J. Fu, Y. Liu, J. Hou, P. Vanaphuti, X. Ma, R. Zhang, Z. Yang and Y. Wang, Sustain. Energy Fuels, 2021, 5, 5574–5580 RSC.
- Y. Hu, L. Liu, J. Zhao, D. Zhang, J. Shen, F. Li, Y. Yang, Z. Liu, W. He, W. Zhao and J. Liu, Batteries, 2023, 9, 322 CrossRef.
- W. Li, H. Li, Z. Lu, L. Gan, L. Ke, T. Zhai and H. Zhou, Energy Environ. Sci., 2015, 8, 3629–3636 RSC.
- W.-R. Liu, Z.-Z. Guo, W.-S. Young, D.-T. Shieh, H.-C. Wu, M.-H. Yang and N.-L. Wu, J. Power Sources, 2005, 140, 139–144 CrossRef CAS.
- P. Liu, J. Wang, J. Hicks-Garner, E. Sherman, S. Soukiazian, M. Verbrugge, H. Tataria, J. Musser and P. Finamore, J. Electrochem. Soc., 2010, 157, A499 CrossRef CAS.
- J. Wang and X. Sun, Energy Environ. Sci., 2015, 8, 1110–1138 RSC.
- M. R. Roberts, A. Madsen, C. Nicklin, J. Rawle, M. G. Palmer, J. R. Owen and A. L. Hector, J. Phys. Chem. C, 2014, 118, 6548–6557 CrossRef CAS PubMed.
- D.-T. Ngo, R. Scipioni, S. B. Simonsen, P. S. Jørgensen and S. H. Jensen, Int. J. Energy Res., 2016, 40, 2022–2032 CrossRef CAS.
- G. Nichols, S. Byard, M. J. Bloxham, J. Botterill, N. J. Dawson, A. Dennis, V. Diart, N. C. North and J. D. Sherwood, J. Pharm. Sci., 2002, 91, 2103–2109 CrossRef CAS PubMed.
- R. Scipioni, P. S. Jørgensen, D.-T. Ngo, S. B. Simonsen, Z. Liu, K. J. Yakal-Kremski, H. Wang, J. Hjelm, P. Norby, S. A. Barnett and S. H. Jensen, J. Power Sources, 2016, 307, 259–269 CrossRef CAS.
- J. Tan, J. Matz, P. Dong, J. Shen and M. Ye, Adv. Energy Mater., 2021, 11, 2100046 Search PubMed.
- Y. Wang, S. Zhang, Z. Chen, H. Zhang, F. Tian, J. Wang, Y. Zhu, G. Yang, Z. Li and G. Cui, Adv. Mater., 2025, e14210 Search PubMed.
- A. Hussain, M. M. Mohamed, M. Faheem, Y. P. Hardianto, M. O. Aijaz, M. R. Karim and M. A. Aziz, J. Polym. Sci., 2025, 63, 3824–3836 CrossRef CAS.
- H. Widiyandari, O. A. Putra, A. Purwanto and Z. Abidin, Mater. Today: Proc., 2021, 44, 3245–3248 Search PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |