 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Conformational isomerism tunes barocaloric potential of MIL-53(Ga)-fum frameworks
C. C.
Hong
 a,
W. L. W.
Leung
b,
C.
Wilson
a,
W. L. W.
Leung
b,
C.
Wilson
 b,
M. J.
Liddle
b,
R. S.
Forgan
b,
M. J.
Liddle
b,
R. S.
Forgan
 b,
J. J. B.
Levinsky
b,
J. J. B.
Levinsky
 a,
A. S. J. S.
Mey
a,
A. S. J. S.
Mey
 a and
C. L.
Hobday
a and
C. L.
Hobday
 *a
*a
aEaStCHEM, School of Chemistry and Centre for Science at Extreme Conditions, University of Edinburgh, Edinburgh, UK. E-mail: claire.hobday@ed.ac.uk
bSchool of Chemistry, University of Glasgow, Glasgow, UK. E-mail: ross.forgan@glasgow.ac.uk
First published on 17th March 2026
Abstract
With the negative implications of current refrigerants on the environment, there is a necessity for greener alternatives. Solid-state caloric materials exhibit large entropy and temperature changes that avoid liquid-vapour transitions of current refrigerants. Through guest adsorption, flexible metal-organic frameworks (MOFs) exhibit large entropy changes under small applied pressures, making them promising candidates. Though effective, flexible MOFs lack long-term mechanical stability owing to the mechanical stress induced during operation. Herein, we highlight and categorise the previously unexplored conformational isomerism of the MIL-53(X)-fum frameworks, which originates from the relative orientations of the fumarate linkers, and explore the effect on structural flexibility and barocaloric potential. Using single crystal X-ray diffraction (SCXRD) and ab initio molecular dynamics (AIMD), we reveal the third and final possible conformational isomer, MIL-53(X)-fum(c), to complement the two previously reported (MIL-53(X)-fum(a) and MIL-53(X)-fum(b)), and investigate the effects of the isomerism on the framework properties. We uncover the isomerically unique mechanical stability of MIL-53(Ga)-fum(c) through simulated potential energy surfaces and find that the vacated open phase has exceptional thermal stability of up to 650 K. Through grand canonical Monte Carlo (GCMC) simulations, the reversible adsorption-driven entropy changes under 0.4 MPa of applied pressure were calculated to be ΔS = 210(30) J K−1 kg−1 and 150(30) J K−1 kg−1 for the adsorption of H2O and CO2, respectively. The results of our studies suggest that MIL-53(Ga)-fum(c) can exhibit a caloric response that is competitive with current commercial refrigerants. Furthermore, we outline the significance of exploring conformational isomerism in MOFs to increase the space in which materials discovery can occur.
Introduction
The continuous increase of the average global temperature makes the effects of climate change apparent and results in an increased demand for refrigeration in everyday life.1 This is reflected by the projected growth of the global refrigerator market size from $74.74 billion to $120.01 billion between 2023 to 2032.2 Currently, refrigeration is primarily achieved by the vapour compression cycle of gaseous refrigerants such as hydrofluorocarbons (HFCs), shown in Fig. 1a.3 HFCs have large global warming potentials (GWP) and, therefore, contribute significantly to climate change.1,4 Hence, there is an urgent need to identify sustainable alternatives.A promising way of addressing this issue is to use a solid-state caloric approach which eliminates the need for a gas-liquid phase change. The caloric effect is defined as the induced temperature change under an externally applied field.5 The application of magnetic fields, electric fields, uniaxial stress, or hydrostatic pressure gives rise to the magnetocaloric, electrocaloric, elastocaloric, and barocaloric effects, in turn.6 An example of a solid-state cooling cycle based on the barocaloric effect is shown in Fig. 1b. For calorics to be viable for refrigeration applications, their performance must be comparable or better than the HFCs. The performance of a caloric material is quantified by the isothermal entropy change (ΔSiso), adiabatic temperature change (ΔTad), and the field required to drive the transition, for example, the change in hydrostatic pressure (ΔP).7 A commonly used HFC, R-134a (1,1,1,2-tetrafluoroethane), exhibits an ΔSiso = ∼520 J kg−1 K−1 for ΔP = 1.5 MPa.8,9 Specific interest was garnered towards barocaloric materials when Li et al. and Lloveras et al. demonstrated that neopentyl glycol (NPG) exhibits pressure-driven entropy changes on par with R-134a.10,11
There are three main challenges associated with barocalorics. (1) The working pressure (ΔP) required to drive the phase transition is typically large. For practical applications, the ΔP required should ideally be lower than 30 MPa as set out by the Henry Royce Institute.12 (2) The isothermal entropy change (ΔSiso) must be comparable to, or improve on, that of current refrigerant gases. (3) The mechanical stress induced during cyclic operation by repeated large volume changes (ΔV) can lead to mechanical failure. This is commonly observed in temperature- or pressure-induced structural phase transitions which give rise to the formation of defects that alter the properties of crystalline materials.13–15 While a variety of material systems such as molecular and ionic plastic crystals,16,17 spin-crossover compounds,18 inorganic salts,19 and two-dimensional perovskites20 have been reported to exhibit colossal barocaloric effects, the ΔP values required to drive such responses are still preventively high. Metal-organic frameworks (MOFs) are a class of caloric materials which have demonstrated the ability to operate at pressures in the same magnitude as that of R-134a.8,21
MOFs are a class of porous, crystalline materials consisting of metal nodes that are coordinated by organic linkers, which extend to create a framework that is highly modular in structure and properties.22–24 Their versatility and tuneable properties mean these materials have found applications across various fields such as gas storage,25–28 drug delivery,29 molecular sieves for mixture separations,30,31 substrate specific catalysis,32,33 and many more.34–36 Many MOFs have intrinsically flexible topologies, exhibiting significant structural changes in response to external stimuli such as temperature, pressure, and the presence of pore-bound guests.37,38 This feature has been coined “breathing” and is generally attributed to several factors such as inherent ligand flexibility, metal polarization, and degrees of freedom within the framework topology.39–41 These easily accessible, large amplitude volume changes and gas adsorption processes make MOFs potential candidates as caloric materials. The working mechanism is similar to barocalorics but instead of non-penetrating hydrostatic pressure, penetrating gaseous pressure is applied which leads to gas adsorption and drives the phase transition, hence the term “breathing-calorics”.8 An example of this adsorption-driven cooling cycle is shown in Fig. 1c.
In 2022, García–Ben et al. reported an ΔSiso = 311 J K−1 kg−1 by the application of 1.6 MPa of carbon dioxide pressure to the flexible MOF, MIL-53(Al) (MIL = Matériaux de l'Institut Lavoisier). The structure of MIL-53(Al) is characterised by one-dimensional [Al(RCO2)2(OH)]n chains that are bridged by µ2-OH units and 1,4-benzenedicarboxylate (BDC) linkers.8 Induced by temperature changes or by the adsorption and desorption of carbon dioxide molecules, MIL-53(Al) undergoes “open-to closed-pore” structural phase transitions with ΔV = 50%.42,43 With such a large ΔV, the problem with mechanical failure remains. Following this, carbon dioxide adsorption in MOF-508b ([Zn2(BDC)2(bpy)], bpy = 4,4′-bipyridine) was studied.21 MOF-508b exhibits a two-step structural phase transition with ΔV1 = 14.4% and ΔV2 = 3.8% that is accompanied by a total ΔSiso = 438.0 J K−1 kg−1. Despite the smaller overall ΔV, the ΔSiso was larger than that of MIL-53(Al). Pressure-gradient differential scanning calorimetric (PGDSC) studies by Feldmann et al. showed that the ΔSiso from the ΔV was endothermic in nature as opposed to the exothermic ΔSiso that arises from the corresponding adsorption event.44 Thus, it was concluded that minimising the ΔV could not only reduce mechanical failure, but also increase the overall ΔSiso, that is primarily due to gas adsorption. In this work, we therefore explore rigid MOFs, which show minimal volume change (ΔV ≈ 0%) during adsorption–desorption cycles, as potential refrigerants. While previous studies have mainly addressed the caloric effects of flexible MOFs or the gas capture properties of rigid MOFs,21,26,29,45,46 our work connects these two domains.
MIL-53(X)-fum (X = Al, Ga, In, and fum = fumarate) is an isoreticular analogue to the archetypal MIL-53(X) framework with the fumarate dianion, derived from (2E)-but-2-enedioic acid, replacing BDC as the organic linker. The structure of the Al homologue, commercialised by BASF as Basolite A520, was not reported until 2015 despite significant early investigations into its adsorption capabilities.47 The crystal structure of hydrated MIL-53(Al)-fum was obtained by a combination of analyses, including solid-state NMR spectroscopy, powder X-ray diffraction, and molecular simulations.48 The Ga homologue was reported soon after.49 The powder X-ray diffractogram was said to strongly resemble that of Basolite A520 and thus, density functional theory (DFT) optimisation was used to derive the structure of MIL-53(Ga)-fum from that of the Al homologue, yielding the hydrated and anhydrous MIL-53(Ga)-fum framework structures. This study of the mechanical response of MIL-53(Ga)-fum concluded that it shows promise as a “shock absorber” for mechanical energy storage. Around the same time, a separate study reported the syntheses of the Ga and In homologues, including a crystal structure of hydrated MIL-53(Ga)-fum determined by Rietveld refinement which showed subtle differences to the previous MIL-53(Ga)-fum structure.50
A key missing element from the studies conducted on these homologues is the consideration of the inherent asymmetric nature of the fumarate linker. Despite the isoreticular nature of the various reported MIL-53(X)-fum frameworks, we note distinct variances in terms of the fumarate linker orientations. We denote these different structures as conformational isomers. Our analyses of the structures of MIL-53(Ga)-fum, studied by Ramaswamy et al., and the Al Basolite A520 have determined they comprise the same isomer.47,49 On the other hand, the MIL-53(Ga)-fum solved by Zhang et al. exists as a different conformational isomer.50 In this work, we have synthesised and experimentally characterised what we believe to be the first confirmed single crystal structure of a third isomer of MIL-53(X)-fum. While this isomer has yet to be reported for fumarate-based frameworks, we have identified this conformational isomer in the structure of the related MOF, MIL-53(Al)-mes (mes = mesaconate, a mono-methylated derivative of fumarate), which was obtained using Rietveld refinement by Stassin et al.51 Notably, conformational isomers of MOFs based on linker orientation have been known to exhibit distinctive properties. Examples include DUT-8, TRUMOF-1, and IRMOF-3-AMPh which showed different closing mechanisms, mechanical properties, and elastic stability, respectively.52–54 Hence, the identification and understanding of the properties of these conformational isomers are significant.
In a previous study by S. Ehrling et al. a simple schematic to describe linker-based disorders was conceived, where each porous environment was represented as a jigsaw piece and the edges represented the orientation of the linker.55 This cannot be translated to the MIL-53(X)-fum series due to the one-dimensional chains of the MIL-53 framework structure (see Fig. 2a); the four linkers making up the pore are not all coplanar to each other (see Fig. 2b).56 The similarities and distinctive differences between the three isomeric structures are exemplified in Section 2 of the SI, highlighting the difficulties in discerning between the structures and the need for a simplified presentation. We have therefore devised a scheme based on a vector diagram to differentiate between the conformational isomers (see Fig. 2c, d, and 2e). The colour of the arrows encode information on which fumarate linkers are coplanar, i.e., connect the same two metal ions on the one-dimensional chain secondary building unit (SBU). The direction of the arrow indicates the orientation of the linker, travelling upwards or downwards in connecting two chain SBUs. The different conformational isomers (Fig. 2c–e) will be denoted as MIL-53(X)-fum(a/b/c), hereafter.
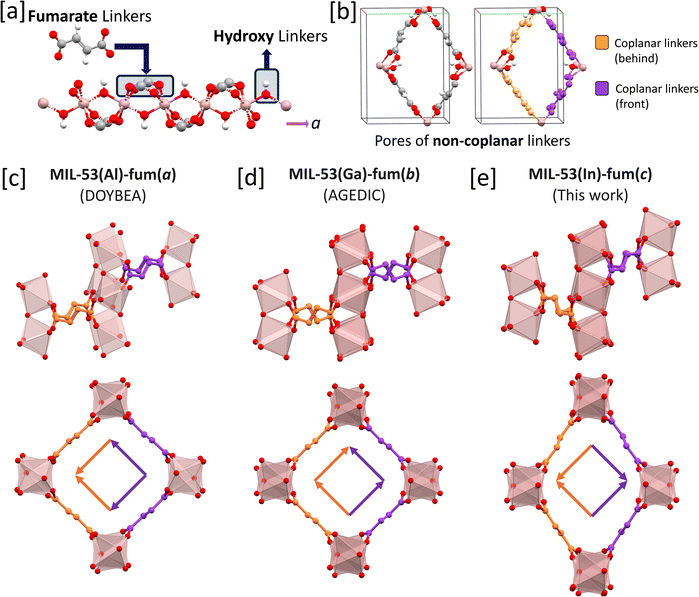 | ||
Fig. 2 Non-coplanar nature and classification scheme of the MIL-53(X)-fum frameworks. (a) The one-dimensional chains of metal oxide nodes along the a-direction with the oxygens from either the fumarate or hydroxyl linkers. In this representation, the colours: red, white, grey, and pink denote the elements: oxygen, hydrogen, carbon, and metal, respectively. (b) The porous environment is shown by observing the structure on the bc-plane. In this plane, it is noted that the four linkers around a pore are not all coplanar but rather, form coplanar pairs, as highlighted by the orange and purple colours. (c)–(e) Crystal structures and vector diagrams showing the three possible local structures based on fumarate disorder where (c) is MIL-53(Al)-fum(a) (DOYBEA),47 (d) is MIL-53(Ga)-fum(b) (AGEDIC),49 and (e) is MIL-53(In)-fum(c) (from this work). The first row shows the side view of the pores highlighting which of the fumarate linkers are coplanar (based on orange/purple colour scheme). The side view also highlights the differences in linker orientation between the different conformational isomers. The structures in the second row show the front view of the framework pores, maintaining the same colour scheme as before. From this, we derive the vector diagrams seen inside the pores where the direction of the arrows denotes the orientation of the linker as “up” or “down”. When travelling in the direction of the arrow, one would go “downhill” across the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond in the centre of the fumarate linker. C double bond in the centre of the fumarate linker. | ||
Based on this scheme, we can classify the structures of MIL-53(Ga)-fum reported by Ramaswamy et al., and the original report of MIL-53(Al)-fum (Basolite A520), to be of the isomer, MIL-53(X)-fum(a).47,49 We then find the Ga homologue reported by Zhang et al. to correspond to the MIL-53(Ga)-fum(b) conformational isomer.50 The MIL-53(In)-fum single crystal structure obtained in this study (vide infra) comprises the third isomer, MIL-53(In)-fum(c), which adopts the same conformation as the related structure of MIL-53(Al)-mes reported by Stassin et al.51 Based on our schematic, this final isomer completes the set of possible conformational isomers stemming from the relative orientations of the fumarate linkers within this particular topology.
Experimentally, there are various challenges associated with the structure determination of these isomers of MIL-53(X)-fum. Firstly, difficulties in obtaining single crystals suitable for X-ray diffraction have necessitated the use of powder X-ray diffraction to date, generating data which, in theory, could be sensibly refined to obtain any of the isomers depending on the methods used. In addition, the presence of individual crystals of different conformational isomers within a single synthetic batch (or indeed as domains within a single particle) cannot be excluded as a possibility, and would significantly hinder the identification process from the convolution of Bragg peaks. In cases where single crystal diffraction (either X-ray or electron) is possible, identification could still be obstructed by twinning or solvation effects. Bulk crystals may contain different local regions consisting of these isomers, known as correlated disorder, wherein a spectrum of properties may be observed depending on said composition. Tuning correlated disorder has led to the potential of engineering the properties of MOFs.57,58 For example, the existence of these previously unexamined isomers in MIL-53(X)-fum and potential correlated disorder may explain inconsistent adsorption behaviours and framework properties observed in previous studies.48,59
Prompted by our classification of the conformational isomers of MIL-53(X)-fum and characterisation of the third possible isomer, we investigate, herein, the effects of conformational isomerism, as well as the nature of the linking metal, on the extent of breathing in response to the presence and absence of pore-bound guests. Using ab initio molecular dynamics (AIMD), we study different metal homologues of the isomers, finding enhanced framework stability and rigidity that is unique to the Ga analogue of one isomer, MIL-53(Ga)-fum(c). To determine the caloric potential of MIL-53(Ga)-fum(c), we calculate the resulting adsorption-driven entropy change using grand canonical Monte Carlo (GCMC) simulations. Our work reveals a ΔS = 210(30) J K−1 kg−1 and 150(30) J K−1 kg−1 for ΔP = 0.4 MPa with the adsorption of water and carbon dioxide, respectively. These quantities demonstrate that rigid MOFs are a promising potential class of refrigerants, with comparable performances and an improved efficiency over that of R-134a.
Methodology
The synthesis of MIL-53(In)-fum(c)
In(NO3)3·xH2O (0.0451 g, 0.15 mmol if x = 0) and fumaric acid (0.0172 g, 0.15 mmol) were dissolved in a mixture of N,N-dimethylformamide (DMF, 4 mL) and water (1 mL) in a 25 mL screw top glass jar. Concentrated HCl (37%, 50 µL, ca 0.60 mmol) was added and the jar was sealed. The clear solution was heated to 353 K in a programmable oven and kept at this temperature for 24 h to yield DMF solvated MIL-53(In)-fum(c) as colourless blocks. On cooling, an appropriate single crystal was selected for analysis directly from the reaction mixture without washing. The as-synthesised material was isolated by drying in a vacuum desiccator. Samples were activated by solvent exchange with acetone, followed by heating to 293 K for 24 h under 2 × 10−5 mbar vacuum.The synthesis of MIL-53(Ga)-fum(b)
Ga(NO3)3·xH2O (0.0637 g, 0.25 mmol if x = 0) and fumaric acid (0.029 g, 0.25 mmol) were sonicated in DMF (10 mL) for 10 min. Concentrated HCl (37%, 20 µL, ca 0.25 mmol) was added and the mixture sealed in a Teflon-lined acid digestion vessel (Parr, 23 mL). After heating to 423 K in a programmable oven for 24 h, the mixture was cooled, and the solids isolated by centrifugation were washed three times with DMF (15 mL) before drying in a vacuum desiccator to yield the as-synthesised material. Samples were activated by solvent exchange with methanol, followed by heating to 293 K for 24 h under 2 × 10−5 mbar vacuum.Single crystal X-ray diffraction
Ambient temperature single crystal X-ray diffraction (SCXRD) measurements were performed on the resulting MIL-53(In)-fum(c) crystals. A Bruker D8 VENTURE diffractometer equipped with a Bruker PHOTON II detector with Mo Kα radiation was used.60 The SAINT routine implemented in the Bruker APEX3 software was employed for data integration.60 SHELXL was used for the final structural refinements.61,62Crystal data for MIL-53(In)-fum(c) (C7H10InNO6, M = 318.98 g mol−1): orthorhombic, space group Pnma (no. 62), a = 7.307(2) Å, b = 10.626(3) Å, c = 13.915(5) Å, V = 1080.4(6) Å3, Z = 4, T = 298 K, µ(Mo Kα) = 2.196 mm−1, Dcalc = 1.961 g cm−3, 5946 reflections measured (4.824° ≤ 2θ ≤ 52.89°), 1165 unique (Rint = 0.0511, Rsigma = 0.0420) which were used in all calculations. The final R1 was 0.0362 (I > 2σ(I)) and wR2 was 0.0876 (all data).
The crystal structure is available on the CCDC with the deposition number 2498629.
Powder X-ray diffraction
Powder X-ray diffractograms were collected using a benchtop Rigaku MiniFlex diffractometer at 298 K. Radiation was monochromated CuKα, with a characteristic wavelength of 1.5405 Å. Scans ranged from 3–45° and were collected on spinning zero-background holders. Selected diffractograms were fitted using Jana 2020.63Thermogravimetric analysis
TGA measurements were carried out in air using a Discovery Series TGA 5500, with gas flows of 10 mL min−1 (balance) and 25 mL min−1 (sample). The analysis was completed with a heating rate of 10 K min−1 and a heating range of 298 K to 1073 K.Gas adsorption analysis
N2 adsorption–desorption isotherms (77 K, 0–1 bar) were carried out on activated samples using a Micromeritics 3Flex porosity analyser.Density functional theory calculations and ab initio molecular dynamics
In order to accurately describe the organic–inorganic interactions within the system, a quantum mechanical based approach was taken and the system was simulated using the Quickstep module as implemented in the CP2K software package (version 2023.1).64 The PBE function was chosen with Van der Waals corrections administered by Grimme's D3 corrections.65,66 The atoms were described using the Goedecker-Teter-Hutter (GTH) pseudopotentials and a molecularly optimised double-zeta-valence-potential (DZVP-MOLOPT) basis set with a planewave cutoff energy of 750 Ry applied, in a periodic cell.67It is known that the difference in metal centres can affect the flexibility and behaviours of MOFs with the MIL-53 topology.68,69 To sample this, based on our solved solvated crystal structure of MIL-53(In)-fum(c) with DMF as the guest molecule, gallium and scandium analogues were derived computationally. This was accomplished by changing the “atom type” of the linking metal in the coordinates file. The framework was then vacated by removing the presence of guest atoms from the pores. This was repeated for MIL-53(X)-fum(a) and MIL-53(X)-fum(b), where the initial structures were obtained from previous published data.48,50 Energy minimisation of the structures was achieved using the conjugated gradient (CG) method with a convergence criterion for changes in atomic position and forces as 0.003 Bohr and 0.003 Hartree/Bohr. A self-consistent field (SCF) was used with a convergence limit of 0.00001 Hartree.
Ab initio molecular dynamics (AIMD) simulations with the isobaric-isothermal ensemble (NPT) were used to simulate the systems at ambient pressure and temperature (0.1 MPa and 298 K). Velocity-rescaling was used for the thermostat and a barostat developed by G. J. Martyna et al. was employed with coupling parameters of 100 fs, post equilibration.70,71 The systems were sampled with a timestep of 0.55 fs. To probe the thermal stability of the vacated system, subsequent simulations at ambient pressure (0.1 MPa) and temperatures of 400 to 650 K, in increments of 50 K, were performed. This range was determined from reported thermogravimetric analysis (TGA) measurements performed on MIL-53(Ga)-fum(b) which showed a decomposition temperature of near 673 K.50 To access the closed structure of the frameworks and gauge the effect of the guest on the mechanical stability, the vacant systems were put under 100 MPa of pressure at 298 K.
By tracking how the potential energy varied with the volume of the system during the 100 MPa NPT simulation and binning the points based on volume, a potential energy surface (PES) was obtained. The bins were taken as constant intervals from the maximum to the minimum volume of the system, over the course of the simulation. The values in each bin were averaged and the uncertainty was taken as the standard deviation between the points in every bin. For the MD simulations at high pressures, the resulting PES would give information on the system properties at 100 MPa. The same method was used to generate a PES from the ambient MD outputs. However, as MIL-53(Ga)-fum(c) remains in the open phase, the comparable configurational space, defined by volume, was limited. To extrapolate and gain an understanding of how the potential energy surface would look when the system is not affected by pressure, configurations from each of the volume bins were taken from the high-pressure trajectory. Single point energy calculations were performed on the configurations. The uncertainty associated with each of the points were taken as the SCF convergence criteria. The results of this can be seen in Section 6 of the SI.
The porosities of the frameworks were analysed using the program PoreBlazer v4.0 which calculates the solvent accessible volume of the pores using either nitrogen atoms, helium atoms, or geometric grid estimations.72 As with the GCMC simulations, the framework and nitrogen atom was described using the universal forcefield (UFF) parameters. The settings used can be found in Section 15 of the SI.
Grand canonical Monte Carlo
To probe the adsorption properties of the framework, a grand canonical Monte Carlo (GCMC) approach was used. Using the RASPA2.0 code, 4 × 3 × 3 super cells of the DFT optimised frameworks were simulated and described using parameters from the Universal Force Field (UFF).73,74 As physisorption was the predominant adsorption event, rather than chemisorption, the frameworks were simulated as a rigid body and the accuracy of the force field was deemed sufficient.75–77Adsorbate molecules were given equal possibilities of insertion, deletion, rotation, and translation moves. The intermolecular interactions were modelled using Lennard-Jones (LJ) potentials with a coulombic term to describe the electrostatic contributions. The parameters for the water and carbon dioxide molecules were taken from the TIP5P force field and TraPPE force fields, respectively, chosen for their previous accuracies in predicting isotherms.78–81 The full set of force field parameters was generated by a combination of the above parameters, using the Lorentz–Berthelot mixing rule.82
For each adsorbate, GCMC simulations with increasing external pressures of 1, 5, 10, 50, 100, 500, 1 × 103, 5 × 103, 1 × 104, 5 × 104, 1 × 105, 5 × 105, 1 × 106, 5 × 106, 1 × 107, 5 × 107, 1 × 108, 5 × 108, and 1 × 109 Pa were performed. For each pressure, around 400![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 post-equilibrated production MC steps were obtained. The radial distribution function (RDF) was calculated using the in-built RDF function from RASPA2.0 to give information on adsorption sites. The isosteric heat of adsorption (qis) or the enthalpy of adsorption (ΔHads) was calculated from the GCMC outputs at every pressure using eqn (1).83
000 post-equilibrated production MC steps were obtained. The radial distribution function (RDF) was calculated using the in-built RDF function from RASPA2.0 to give information on adsorption sites. The isosteric heat of adsorption (qis) or the enthalpy of adsorption (ΔHads) was calculated from the GCMC outputs at every pressure using eqn (1).83
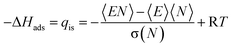 | (1) |
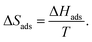 | (2) |
To relate the entropy change (ΔSads) per adsorption event to the overall entropy change as a function of a change in pressure (ΔSads(ΔP)), the change in the number of guests corresponding to the pressure change (ΔN(ΔP)) must be considered. The ΔSads(ΔP) values relate to the ΔN(ΔP) through pressure. As such, the overall entropy change with respect to the change in pressure (ΔStot,ΔP) was derived from the area under the plot of ΔSads(ΔP) against ΔN(ΔP), see Section 11 of the SI. Between two adjacent data points on said plot, the entropy change (ΔStot,P1→P2) can be expressed as an integral of said plot, expressed as a function of ΔSads(ΔP) and ΔN(ΔP):
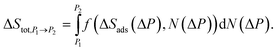 | (3) |
The implication of eqn (3) is that, there must exist a continuous function, f(ΔSads(ΔP), N(ΔP)), between any given two points on a plot of ΔSads(ΔP) against ΔN(ΔP). In this work, a linear function was chosen to correlate ΔSads(ΔP) and ΔN(ΔP) between the two sampled pressure points (P1 and P2), i.e.,
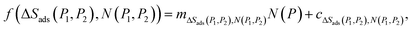 | (4) |
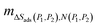 represents the gradient and
represents the gradient and 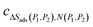 is the y-intercept for the linear fit between the two points at different pressures. When integrated following eqn (4), by definition, the space between two pressure points is split into infinitesimally small steps. Hence, despite using a simple linear regression, the error of the fitting is minimised. To evaluate the integral and calculate ΔStot,ΔP between two points, the trapezoidal rule was used. For changes in pressures that extend beyond two points, the resulting ΔStot,ΔP was calculated as a summation of the integrals (see eqn. (3)) over the constituent pairs of pressure points, implemented using the chained trapezoidal rule.
is the y-intercept for the linear fit between the two points at different pressures. When integrated following eqn (4), by definition, the space between two pressure points is split into infinitesimally small steps. Hence, despite using a simple linear regression, the error of the fitting is minimised. To evaluate the integral and calculate ΔStot,ΔP between two points, the trapezoidal rule was used. For changes in pressures that extend beyond two points, the resulting ΔStot,ΔP was calculated as a summation of the integrals (see eqn. (3)) over the constituent pairs of pressure points, implemented using the chained trapezoidal rule.
The GCMC outputs used to compute the results shown, the analysis on the guest adsorption sites, and the full collection of ΔStot with respects to ΔP upon pressurisation/depressurisation are available in Sections 7–11 of the SI.
Results and discussion
Single crystal X-ray diffraction reveals the structure of MIL-53(In)-fum(c)
The crystal structure of MIL-53(In)-fum(c) was solved at 298 K using SCXRD. MIL-53(In)-fum(c) crystallises in the orthorhombic space group, Pnma, and has unit cell dimensions of a = 7.307(2) Å, b = 10.626(3) Å, and c = 13.915(5) Å. In terms of metal-linker connectivity, the crystal structure of MIL-53(In)-fum(c) is analogous to the previously solved MIL-53 structures.56,69,86 DMF molecules were located within the pores, hydrogen bonded to the µ2-OH units through the formamide oxygen atom (O3⋯O4 = 2.750(10) Å). This hydrogen bond ensures the formamide oxygen atom (O4) is fully occupied in one position, while the remainder of the DMF molecule (other than the formamide nitrogen, N1) displays two-fold positional disorder. It is notable that the previously reported structures of MIL-53(X)-fum, which are reported as the (a) and (b) conformational isomers, all correspond to MOFs that had either been synthesised or worked up under aqueous conditions, suggesting a possible role of DMF in templating the formation of MIL-53(X)-fum(c). The structure refinement parameters for MIL-53(In)-fum(c) can be found in Section 1 of the SI.To our knowledge this is the first experimental characterisation of a MIL-53(X)-fum framework through SCXRD. As previously noted, when refining powder diffraction data, it may be possible to derive sensible structures corresponding to any of the predicted conformational isomers from the same diffractogram, depending on the method used. The significance of obtaining a structure by SCXRD is the fact that we can unequivocally confirm the existence of one specific isomer.
Enhanced rigidity and mechanical stability for MIL-53(Ga)-fum(c) over MIL-53(Ga)-fum(a) and MIL-53(Ga)-fum(b)
As discussed, conformational isomerism can have significant impact on the physical properties of MOFs.52,54,55 Hence, in this work, we focus on simulating the different isomers by using a single unit cell which captures the inherent fumarate orientations of the MIL-53(X)-fum(a/b/c) structures that were experimentally obtained. Complementing the known isomers of MIL-53(Ga)-fum(a/b) studied by Ramaswamy et al. and Zhang et al., by substituting Ga into our structure of MIL-53(In)-fum(c), we are able to complete the series of linker-orientational based conformational isomers for the MIL-53(Ga)-fum frameworks.49,50 This enabled our investigation on the impact of the isomerism on the framework properties, as the only independent variable. This was achieved through AIMD NPT simulations at ambient (298 K and 0.1 MPa) as well as at elevated pressures of 100 MPa.By simply tracking the evolution of the volume of the three systems in response to pressure, we are able to first qualitatively gauge the rigidity of the isomers when the pores are vacated of guest molecules. Of the three, we determine MIL-53(Ga)-fum(b) to be flexible, where the empty framework closes spontaneously under ambient temperature and pressure, with ΔV = ∼50% of the original unit cell. Previously, Zhang et al. concluded MIL-53(Ga)-fum(b) to be rigid after immersing the framework in different solvents and obtaining powder X-ray diffractograms, despite minor differences in the diffractograms of the as-synthesised and water hydrated materials.50 This disparity in predicted flexibility could originate from uncertainty in the pore occupancy. As observed in other MOFs, including the MIL-53 family, it can be very difficult to confirm complete evacuation of guests, post synthesis or solvation.87,88 Moreover, isomeric interconversion or coexistence in the bulk sample may occur.89–91 Another important consideration when comparing experimental results to computational is the crystal size effect. Simulations employ periodic boundary condition which mimic crystalline systems with infinite unit cells. Meanwhile, in experimental settings, the crystal size is a variable that must be considered. For example, Johnson et al. demonstrated smaller crystals of ZIF-8 exhibited higher flexibility compared to the larger ones.92
As the aim is to minimise mechanical stress of refrigerants through framework rigidity, the predicted flexibility of completely evacuated MIL-53(Ga)-fum(b) was ill-suited for our application. Furthermore, as discussed, the opposing ΔSiso from the ΔV to that of the corresponding adsorption event, would suggest that the flexible MIL-53(Ga)-fum(b) would have an inferior barocaloric potential compared to the two more rigid isomers.44 On the other hand, we observed that both MIL-53(Ga)-fum(a) and MIL-53(Ga)-fum(c) remained in the open-phase at ambient conditions. While the results for MIL-53(Ga)-fum(c) have not been verified experimentally, the stability of the open MIL-53(Ga)-fum(a) framework agrees with the high-pressure powder X-ray diffraction experiments and calculated energy profile with respects to volume by Ramaswamy et al.49 They determined the open phase of MIL-53(Ga)-fum(a) as the global minimum whereas the closed phase sits within a local minimum.
To gain further insight into the rigidity of the different isomers, we computed the energy of stabilisation which we define as the difference in potential energy (ΔEpot) between the open and closed phases of each isomer. The larger ΔEpot is, the more favourable it is for the framework to close. To attain the closed phases, we ran subsequent NPT simulations of the isomers with an increase in applied hydrostatic pressure of 100 MPa. Similar to the MIL-53(Al) framework, wherein the structural phase transition was characterised by a ΔV of ∼40% combined with a reduction of the solvent accessible volume (SAV) from ∼393 Å3 to almost zero, we also observe a ΔV of ∼50% with a change in SAV from ∼550 Å3 to 0 Å3.42 As such, we have characterised the observed structural change of the MIL-53(X)-fum(a/b/c) frameworks as a transition between different pore states, analogous to that observed in the MIL-53 frameworks. Between the pore states of the three isomers, we observed decreasing ΔEpot in the order of (b) > (a) > (c), as detailed in Table S3 of the SI. The ΔEpot for MIL-53(Ga)-fum(b) was the largest (−212.40(5) kJ mol−1) which implied that it is the most favourable to close. This agrees with the more flexible nature of the structure as observed in the ambient NPT simulations. Additionally, the trend would imply that it is less favourable for isomer (c) to undergo the structural phase transition compared to (a), with ΔEpot of −104.68(6) and −157.35(2) kJ mol−1, respectively. Hence isomer (c) would possess a more rigid nature.
By constructing potential energy surfaces (PES) with respect to the volume of the framework, we are able to determine how much energy is required for the framework to transition from the open to closed phase. This potential energy difference allows us to indirectly probe the stability of the open-phase, and hence rigidity, of the MIL-53(Ga)-fum(c) isomer (see Fig. 3). At ambient conditions, we observe a single minimum at a volume of ∼950 Å3 which corresponds to the open phase. Fig. 3 infers a potential energy difference of 250 kJ mol−1 or greater for the system to transition towards the closed phase at 298 K and 0.1 MPa. In comparison, MIL-53(Ga)-fum(a) was previously calculated to have a ∼15 kJ mol−1 difference.49 The larger potential energy penalty infers that MIL-53(Ga)-fum(c) is more rigid than its isomeric counterpart with an open-phase that is more stable. At high pressures of 100 MPa, a large change in the potential energy surface was observed for MIL-53(Ga)-fum(c), clearly favouring the system in the closed phase (∼580 Å3), as one might expect.
To understand whether MIL-53(Ga)-fum(c) and its rigidity can withstand operational temperatures, NPT simulations at increased temperature were conducted. For temperatures up to 650 K, we observed no closing of the framework. Unfortunately, all attempts to synthesise MIL-53(Ga)-fum(c) resulted in the isolation of the (b) conformational isomer (see Section 13 of the SI), with characterisation data closely related to that reported by Zhang et al.50 The overall thermal stability predicted for MIL-53(Ga)-fum(c) is in good agreement with the TGA performed on MIL-53(Ga)-fum(b) by Zhang et al. and our own data (see Section 13 of the SI).50 This implies that there is an insignificant effect on the chemical stability of the material associated with the inevitable warming and cooling of the material during a refrigerant cycle.50 This sets MIL-53(Ga)-fum(c) apart from thermally activated frameworks such as MIL-53(Sc)-BDC which close upon heating.41 As the recorded degradation temperature of the metal homologues of MIL-53-fum series exists within the range of 600 to 700 K, this family of framework can be regarded to possess good thermal stability.50,93 The variability and trend of the degradation temperatures strongly suggest a metal-dependent thermal stability, with the Ga-based framework showing the higher stability amongst the metal homologues.
The differences in potential energy and rigidity between the three isomers, predicted in this work, highlights the importance of a robust characterisation method for MOFs with asymmetric linkers, like MIL-53(X)-fum. Depending on the intended application, one may find advantages in one isomer over the other, for example, the choice of rigid over a flexible framework in barocalorics. The combination of the thermal and mechanical stability of MIL-53(Ga)-fum(c) at ambient pressure (0.1 MPa) suggests the suitability of this framework as an adsorption-driven refrigerant that could overcome the challenge of mechanical failure during cyclic operation. Hence, we chose to continue the study with the MIL-53(Ga)-fum(c) isomer over MIL-53(Ga)-fum(a) or MIL-53(Ga)-fum(b), in this work.
The Ga analogues of MIL-53-fum(c) exhibit rigid properties over the flexible Sc and In analogues
To assess the effect of bridging metal on flexibility, we also probed the properties of the In and Sc homologues of the (c) isomer. The latter was achieved by substituting the In ions with Sc ions in the crystal structure of MIL-53(In)-fum(c). Unlike the more rigid Ga homologues, we find that both MIL-53(Sc)-fum(c) and MIL-53(In)-fum(c) frameworks transitioned towards the closed-phase with a smaller potential energy difference. This was statistically confirmed from 20 independent AIMD simulations initiated with random seeds with simulation lengths ranging from 4 ps to 8 ps. The simulation outputs of the metal homologues are shown in Section 3 of the SI. These simulations strongly indicate that the Ga ion was responsible for the observed rigidity of the MIL-53-fum(c) framework. The larger ionic radii of Sc3+ and In3+ compared to Ga3+ may be the reason for the metal-dependent flexibility, which would mirror that seen with MIL-53(X)-BDC series; MIL-53(Sc)-BDC adopts an extremely narrow pore form not seen in other homologues.41,94 It was possible to scale up the synthesis of MIL-53(In)-fum(c) to collect bulk characterisation data (see Section 13 of the SI). After activation through solvent exchange in acetone and heating to 493 K for 24 under 2 × 10−5 vacuum, MIL-53(In)-fum(c) adopted a closed pore configuration. Le Bail fitting of powder X-ray diffraction data showed a unit cell volume of approximately 800 Å3, and MIL-53(In)-fum(c) was non porous to N2 (77 K, 1 bar) in contrast to the literature report of the rigidity and porosity of MIL-53(In)-fum(b).50 These results further underpin the importance of conformational isomerism on the physical properties of MOFs, and validate the simulations.Hence, Ga-based MIL-53-fum frameworks would be most applicable for minimising mechanical stress while maximising the performance during refrigeration cycles. The remainder of this paper aims to uncover the refrigeration capability of MIL-53(Ga)-fum(c).
GCMC simulation can quantify adsorption-driven refrigeration potential of MIL-53(Ga)-fum(c) under low working pressures
As MIL-53(Ga)-fum(c) was determined to be rigid in nature at 0.1 MPa, GCMC simulations were performed to determine its adsorption driven refrigeration potential. The choice of water and carbon dioxide as adsorbates to drive the refrigeration process was motivated by green chemistry principals and safety concerns given the target application of refrigeration; both adsorbates are stable and do not have serious safety hazards associated with them. The simulated H2O and CO2 adsorption isotherms showed spontaneous adsorption of the guests into the pores at ambient conditions, in line with previous experimental work.50 Notably, both adsorbate systems experience a large energetic stabilisation with the adsorption of guest molecules. Saturation of the framework occurred at 10 MPa, with ∼16 water molecules or ∼7 CO2 molecules per unit cell. The difference in the number of molecules at saturation can be explained by the fact that CO2 is a larger molecule and possesses rigid C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds which lead to less flexibility when it comes to packing compared to water. Furthermore, water can form hydrogen bonding networks that stabilise structures with higher loading. This is also observed in the calculated RDFs (see Section 9 of the SI).
O double bonds which lead to less flexibility when it comes to packing compared to water. Furthermore, water can form hydrogen bonding networks that stabilise structures with higher loading. This is also observed in the calculated RDFs (see Section 9 of the SI).
The enthalpies of adsorption (ΔHads) for both adsorbates were determined across a pressure range of 1 Pa to 10 GPa, using eqn (1) (see Fig. 4a). ΔHads is a measure of the energy of stabilisation associated with the adsorption of one guest molecule in the framework. A clear decreasing trend was observed for the calculated ΔHads with increasing external pressure, as with many other systems.9,95–98 At lower pressures, there are fewer guest molecules within the pores which results in less competition to the adsorption sites. At higher pressures, guest molecules compete, and increasing electrostatic repulsion leads to a destabilising effect which lowers the resulting ΔHads; this was also observed in the potential energy (see Fig. S16 and S17 in the SI). Overall, water exhibited a higher ΔHads compared to carbon dioxide which can be attributed to the stronger adsorbent–adsorbate interactions.
Comparing the calculated ΔHads values for CO2 to the experimentally derived values for MIL-53(Al)-fum, our ΔHads values are greater by ∼30 kJ mol−1 and ∼10 kJ mol−1 at lower loading and as the framework reaches saturation, respectively.99 The former implies stronger interactions between the Ga framework and the CO2 molecules in comparison to the Al analogue. The inconsistent differences depending on loading are suggested to be artifacts of using a fully rigid framework in the GCMC simulations. Despite defining the MIL-53(Ga)-fum(c) as “rigid” in that the framework does not close when vacated, local dynamics in the framework atoms and volume fluctuations can still occur (see Sections 3 and 4 of the SI). Research into comparing dynamic and static framework atoms has provided evidence to suggest that static adsorption simulations can overestimate adsorption in MIL-53(X) frameworks.100 For water, our calculated ΔHads values are better matched to the experimental values; our values are greater by just ∼10 kJ mol−1 and ∼5 kJ mol−1 at lower to higher loading.101 From the calculated RDFs, it is strongly implied that the predominant adsorption sites are either at the oxygen or hydrogen atoms of the framework (see Section 9 of the SI). Hence, we expect the ΔHads values between the Ga and Al analogues to be relatively comparable.
Using the chain trapezoidal rule, the change in entropy resulting from a change in pressure (ΔStot,ΔP) was calculated. From Fig. 4b, it can be inferred that, from an applied vapour pressure (ΔP) of 0.4 MPa from atmospheric pressure (0.1 MPa), the associated total entropy change (ΔStot) is 210(30) J K−1 kg−1 and 150(30) J K−1 kg−1 for water and carbon dioxide, respectively. While at the lowest pressure change, water performed better than carbon dioxide, the opposite was true for high pressure changes. Moreover, it was found that a negative ΔP from ambient pressures (shifting to sub-ambient pressures) results in a greater ΔStot. For ΔP = −0.04 MPa, we calculate ΔStot = −410(40) and −340(30) J K−1 kg−1 for H2O and CO2, respectively. At ambient pressure, both systems are already close to saturation hence, higher pressures were required for the adsorption of one more guest into the framework compared to lower pressures.
The roadmap of the Henry Royce Institute states that barocalorics should operate under pressures of 30 MPa to work efficiently and minimise energy consumption.12 While questions on implementation are outwith the scope of this study, the colossal ΔStot in response to the low ΔP shown is competitive with the previously studied flexible MOFs and R-134a (see Table 1).8,21 As such, we conclude MIL-53(Ga)-fum(c) to be good candidate as a green solid-state refrigerant.
| Material | ΔS (J K−1 kg−1) | ΔP* (MPa) | Working mechanism | Reference |
|---|---|---|---|---|
| R-134a | ∼520 | 1.5 | Barocaloric: vapour-compression | 9 |
| NPG | ∼510 | 570 | Solid-state barocaloric: order–disorder transitions | 11 |
| MOF-508b | 438.0 | 2.6 | Breathing framework + CO2 adsorption | 21 |
| MIL-53(Al)-BDC | 311 | 1.6 | Breathing framework + CO2 adsorption | 8 |
| MIL-53(Ga)-fum(c) | 210(30) | 0.4 | H2O adsorption | This work |
| −410(30) | −0.04 | |||
| 150(30) | 0.4 | CO2 adsorption | This work | |
| −340(30) | −0.04 |
Conclusions
We have identified and classified the different conformational isomers of the MIL-53(X)-fum frameworks based on the inherent orientation of the fumarate linkers. By conceiving a vector-based scheme which encodes information on the fumarate linker planarity and relative orientation, we have concluded that there are only three possible isomers based on the combinations of fumarate orientations. We have synthesised the novel MIL-53(In)-fum(c) framework material and, alongside the previous two reported isomers from the literature, completed the set of possible isomers. The importance of conformational isomerism is evident in AIMD simulations, which predict the open-phase of MIL-53(Ga)-fum(c) to be the most stable at ambient conditions, requiring more than 250 kJ mol−1 to transition towards the closed-phase, whilst MIL-53(Ga)-fum(a) and MIL-53(Ga)-fum(b) were either less stable in the open-phase or spontaneously closed under these simulated conditions. The stability of the unique rigid nature of the Ga homologue of the MIL-53(X)-fum(c) isomer against that of the In and Sc homologues was subsequently confirmed by further simulations. From this, we emphasise the need to consider these local isomeric structures and how they can significantly impact the properties of a framework. Our results indicate the need for single crystal structures (either from X-ray or electron diffraction) for MOFs with asymmetric linkers that can display such conformational isomerism.102 The ability to unequivocally characterise the isomer(s) present allows for targeting specific isomers for their unique properties.The enhanced thermal and mechanical properties present MIL-53(Ga)-fum(c) as a promising, long-lasting, and efficient refrigerant. The entropy change associated with adsorption was calculated to be 210(30) J K−1 kg−1 and 150(30) J K−1 kg−1 under 0.4 MPa of applied vapour pressure for water and carbon dioxide, respectively. These values are within the realms of colossal barocaloric materials and R-134a, which solidifies MIL-53(Ga)-fum(c) as a strong candidate for refrigeration applications. The implications of this work highlight the vast potentials in rigid MOFs, where the refrigeration efficiencies are improved while removing the problems associated with mechanical wear.
A sensible next step to this work would be to validate the results of this work with experimental adsorption isotherms and with thermal studies of the different isomers; this would require, however, an in-depth experimental study of the extent of isomerism and conversion within bulk samples, which cannot currently be captured by conventional powder diffraction methods. Our next work focusses on understanding interconversion pathways between the isomers to unlock the potential for property-driven framework engineering. To gain an insight into the correlated disorder and understand the extent of the tuneability of the framework as a result, supercell simulations would be required. More accurate force fields, perhaps in the form of a custom machine learning interatomic potential (MLIPs) which can also facilitate the study of the framework dynamics during adsorption cycles, may facilitate this computational work. Additionally, MLIPs can aid in the determination of a free energy surface to sample the open to closed phase transitions of these correlated disordered frameworks.103–105 Our observations on the isomerism related to the fumarate linker may also help elucidate the behaviour of MOFs linked by other commonly used “stepped” dicarboxylate linkers, such as naphthalene-2,6-dicarboxylate, stilbene-4,4′-dicarboxylate, and azobenzene-4,4′-dicarboxylate, and further enhance the structural versatility amongst MOFs in these phase spaces.
Conflicts of interest
The authors declare no competing financial interest.Data availability
CCDC 2498629 contains the supplementary crystallographic data for this paper.106The data supporting this article have been included in the supplementary information (SI) and are available from the corresponding author upon reasonable request, provided that the request is for legitimate academic or research purposes. The analysis codes used are open-source and available on the GitHub (https://github.com/hobdaylab/Codes_MIL-53-Ga-fum_paper). Supplementary information: SCXRD information and crystal structure for MIL-53(In)-fum(c), CCDC 2498629, outputs from AIMD and GCMC simulations, potential energy surfaces, RDF calculations, and entropy calculations. See DOI: https://doi.org/10.1039/d5ta08920d.
Acknowledgements
We acknowledge financial support from UK Research and Innovation (Future Leaders Fellowship MR/V026070/1 to C. L. H.) and the University of Edinburgh (Chancellor's Fellowship to C. L. H., doctoral studentships to C. C. H). R. S. F., W. L. L. and M. J. L. acknowledge support from the University of Glasgow and EPSRC (EP/T517896/1 and EP/W524359/1). This work used the Cirrus UK National Tier-2 HPC Service at EPCC (https://www.cirrus.ac.uk/) and ARCHER2 UK National Supercomputing Service (https://www.archer2.ac.uk/), funded by the University of Edinburgh and EPSRC (EP/P020267/1). Access to ARCHER2 was obtained via the UKCP consortium and funded by EPSRC grant ref (EP/X035891/1). This work has also made use of the resources provided by the Edinburgh Compute and Data Facility (ECDF) (https://information-services.ed.ac.uk/research-support/research-computing/ecdf), namely Eddie.References
- L. Wang, L. Wang, Y. Li and J. Wang, Decis. Anal. J., 2023, 7, 100237 CrossRef
.
- Refrigerator Market Size, Share, Growth | Industry Trends, 2032 Search PubMed.
- C. Cazorla, Appl. Phys. Rev., 2019, 6, 041316 Search PubMed
.
- B. H. Samset, C. Zhou, J. S. Fuglestvedt, M. T. Lund, J. Marotzke and M. D. Zelinka, Commun. Earth Environ., 2023, 4, 400 Search PubMed
.
- H. Hou, S. Qian and I. Takeuchi, Nat. Rev. Mater., 2022, 7, 633–652 Search PubMed
.
- S. Crossley, N. D. Mathur and X. Moya, AIP Adv., 2015, 5, 067153 CrossRef
.
- X. Moya and N. D. Mathur, Science, 1979, 370, 797–803 Search PubMed
.
- J. García-Ben, J. López-Beceiro, R. Artiaga, J. Salgado-Beceiro, I. Delgado-Ferreiro, Y. V. Kolen’ko, S. Castro-García, M. A. Señarís-Rodríguez, M. Sánchez-Andújar and J. M. Bermúdez-García, Chem. Mater., 2022, 34, 3323–3332 CrossRef PubMed
.
-
M. McLinden, in ASHRAE Handbook, ASHRAE, Atlanta, 2009 Search PubMed
.
- B. Li, Y. Kawakita, S. Ohira-Kawamura, T. Sugahara, H. Wang, J. Wang, Y. Chen, S. I. Kawaguchi, S. Kawaguchi, K. Ohara, K. Li, D. Yu, R. Mole, T. Hattori, T. Kikuchi, S. Yano, Z. Zhang, Z. Zhang, W. Ren, S. Lin, O. Sakata, K. Nakajima and Z. Zhang, Nature, 2019, 567, 506–510 Search PubMed
.
- P. Lloveras, A. Aznar, M. Barrio, Ph. Negrier, C. Popescu, A. Planes, L. Mañosa, E. Stern-Taulats, A. Avramenko, N. D. Mathur, X. Moya and J.-L. Tamarit, Nat. Commun., 2019, 10, 1803 Search PubMed
.
- J. Garcia-Ben, I. Delgado-Ferreiro, J. Salgado-Beceiro and J. M. Bermudez-Garcia, Materials, 2021, 14, 5947 CrossRef CAS PubMed
.
- P. Saikia, P. Gupta, T. R. Nath and N. K. Nath, CrystEngComm, 2024, 26, 4259–4263 Search PubMed
.
- K. Takazawa, J. Inoue, K. Mitsuishi, Y. Yoshida, H. Kishida, P. Tinnemans, H. Engelkamp and P. C. M. Christianen, Sci. Rep., 2021, 11, 3175 CrossRef CAS PubMed
.
- V. Bon, N. Kavoosi, I. Senkovska and S. Kaskel, ACS Appl. Mater. Interfaces, 2015, 7, 22292–22300 CrossRef CAS PubMed
.
- J. M. Pringle, P. C. Howlett, D. R. MacFarlane and M. Forsyth, J. Mater. Chem., 2010, 20, 2056 Search PubMed
.
- F. B. Li, M. Li, X. Xu, Z. C. Yang, H. Xu, C. K. Jia, K. Li, J. He, B. Li and H. Wang, Nat. Commun., 2020, 11, 4190 CrossRef CAS PubMed
.
- J. Seo, J. D. Braun, V. M. Dev and J. A. Mason, J. Am. Chem. Soc., 2022, 144, 6493–6503 Search PubMed
.
- A. Aznar, P. Lloveras, M. Romanini, M. Barrio, J.-L. Tamarit, C. Cazorla, D. Errandonea, N. D. Mathur, A. Planes, X. Moya and L. Mañosa, Nat. Commun., 2017, 8, 1851 Search PubMed
.
- J. Seo, R. D. McGillicuddy, A. H. Slavney, S. Zhang, R. Ukani, A. A. Yakovenko, S.-L. Zheng and J. A. Mason, Nat. Commun., 2022, 13, 2536 CrossRef CAS PubMed
.
- M. Gelpi, J. García-Ben, S. Rodríguez-Hermida, J. López-Beceiro, R. Artiaga, Á. Baaliña, M. Romero-Gómez, J. Romero-Gómez, S. Zaragoza, J. Salgado-Beceiro, J. Walker, C. J. McMonagle, S. Castro-García, M. Sánchez-Andújar, M. A. Señarís-Rodríguez and J. M. Bermúdez-García, Adv. Mater., 2024, 36, 2310499 CrossRef CAS PubMed
.
- Z. Han, Y. Yang, J. Rushlow, J. Huo, Z. Liu, Y.-C. Hsu, R. Yin, M. Wang, R. Liang, K.-Y. Wang and H.-C. Zhou, Chem. Soc. Rev., 2025, 54, 367–395 RSC
.
- Z. Han, K.-Y. Wang, R.-R. Liang, Y. Guo, Y. Yang, M. Wang, Y. Mao, J. Huo, W. Shi and H.-C. Zhou, J. Am. Chem. Soc., 2025, 147, 3866–3873 Search PubMed
.
- P. Deria, J. E. Mondloch, O. Karagiaridi, W. Bury, J. T. Hupp and O. K. Farha, Chem. Soc. Rev., 2014, 43, 5896–5912 Search PubMed
.
- E. Martínez-Ahumada, M. L. Díaz-Ramírez, M. D. J. Velásquez-Hernández, V. Jancik and I. A. Ibarra, Chem. Sci., 2021, 12, 6772–6799 RSC
.
- J.-B. Lin, T. T. T. Nguyen, R. Vaidhyanathan, J. Burner, J. M. Taylor, H. Durekova, F. Akhtar, R. K. Mah, O. Ghaffari-Nik, S. Marx, N. Fylstra, S. S. Iremonger, K. W. Dawson, P. Sarkar, P. Hovington, A. Rajendran, T. K. Woo and G. K. H. Shimizu, Science, 1979, 374, 1464–1469 CrossRef PubMed
.
- C.-H. Ho and F. Paesani, ACS Appl. Mater. Interfaces, 2023, 15, 48287–48295 CrossRef CAS PubMed
.
- W. Xu and O. M. Yaghi, ACS Cent. Sci., 2020, 6, 1348–1354 CrossRef CAS PubMed
.
- I. Abánades Lázaro, X. Chen, M. Ding, A. Eskandari, D. Fairen-Jimenez, M. Giménez-Marqués, R. Gref, W. Lin, T. Luo and R. S. Forgan, Nat. Rev. Methods Primers, 2024, 4, 42 CrossRef
.
- G. Liu, V. Chernikova, Y. Liu, K. Zhang, Y. Belmabkhout, O. Shekhah, C. Zhang, S. Yi, M. Eddaoudi and W. J. Koros, Nat. Mater., 2018, 17, 283–289 CrossRef CAS PubMed
.
- C. E. Wilmer, O. K. Farha, Y.-S. Bae, J. T. Hupp and R. Q. Snurr, Energy Environ. Sci., 2012, 5, 9849 RSC
.
- D. Yang and B. C. Gates, ACS Catal., 2019, 9, 1779–1798 CrossRef CAS
.
- L. Ma, C. Abney and W. Lin, Chem. Soc. Rev., 2009, 38, 1248 RSC
.
- V. F. Yusuf, N. I. Malek and S. K. Kailasa, ACS Omega, 2022, 7, 44507–44531 CrossRef CAS PubMed
.
- D.-W. Lim and H. Kitagawa, Chem. Rev., 2020, 120, 8416–8467 CrossRef CAS PubMed
.
- C.-Y. Sun, X.-L. Wang, X. Zhang, C. Qin, P. Li, Z.-M. Su, D.-X. Zhu, G.-G. Shan, K.-Z. Shao, H. Wu and J. Li, Nat. Commun., 2013, 4, 2717 CrossRef PubMed
.
- Z. Liu, L. Zhang and D. Sun, Chem. Commun., 2020, 56, 9416–9432 RSC
.
- S. Seth and S. Jhulki, Mater. Horiz., 2021, 8, 700–727 RSC
.
- F. Formalik, A. V. Neimark, J. Rogacka, L. Firlej and B. Kuchta, J. Colloid Interface Sci., 2020, 578, 77–88 CrossRef CAS PubMed
.
- M. Alhamami, H. Doan and C.-H. Cheng, Materials, 2014, 7, 3198–3250 Search PubMed
.
- L. Chen, J. P. S. Mowat, D. Fairen-Jimenez, C. A. Morrison, S. P. Thompson, P. A. Wright and T. Düren, J. Am. Chem. Soc., 2013, 135, 15763–15773 Search PubMed
.
- Y. Liu, J.-H. Her, A. Dailly, A. J. Ramirez-Cuesta, D. A. Neumann and C. M. Brown, J. Am. Chem. Soc., 2008, 130, 11813–11818 Search PubMed
.
- S. R. G. Balestra, R. Bueno-Perez, S. Hamad, D. Dubbeldam, A. R. Ruiz-Salvador and S. Calero, J. Mater. Chem., 2016, 28, 8296–8304 Search PubMed
.
- W. K. Feldmann, C. Esterhuysen and L. J. Barbour, ChemSusChem, 2020, 13, 5220–5223 CrossRef CAS PubMed
.
- H. Liu, T. Fu and Y. Mao, ACS Omega, 2022, 7, 14430–14456 Search PubMed
.
- A. López-Olvera, J. A. Zárate, E. Martínez-Ahumada, D. Fan, M. L. Díaz-Ramírez, P. A. Sáenz-Cavazos, V. Martis, D. R. Williams, E. Sánchez-González, G. Maurin and I. A. Ibarra, ACS Appl. Mater. Interfaces, 2021, 13, 39363–39370 Search PubMed
.
- M. Gaab, N. Trukhan, S. Maurer, R. Gummaraju and U. Müller, Microporous Mesoporous Mater., 2012, 157, 131–136 CrossRef CAS
.
- E. Alvarez, N. Guillou, C. Martineau, B. Bueken, B. Van de Voorde, C. Le Guillouzer, P. Fabry, F. Nouar, F. Taulelle, D. de Vos, J. Chang, K. H. Cho, N. Ramsahye, T. Devic, M. Daturi, G. Maurin and C. Serre, Angew. Chem., Int. Ed., 2015, 54, 3664–3668 Search PubMed
.
- P. Ramaswamy, J. Wieme, E. Alvarez, L. Vanduyfhuys, J.-P. Itié, P. Fabry, V. Van Speybroeck, C. Serre, P. G. Yot and G. Maurin, J. Mater. Chem. A, 2017, 5, 11047–11054 RSC
.
- Y. Zhang, B. E. G. Lucier, S. M. McKenzie, M. Arhangelskis, A. J. Morris, T. Friščić, J. W. Reid, V. V. Terskikh, M. Chen and Y. Huang, ACS Appl. Mater. Interfaces, 2018, 10, 28582–28596 Search PubMed
.
- T. Stassin, S. Waitschat, N. Heidenreich, H. Reinsch, F. Pluschkell, D. Kravchenko, J. Marreiros, I. Stassen, J. van Dinter, R. Verbeke, M. Dickmann, W. Egger, I. Vankelecom, D. De Vos, R. Ameloot and N. Stock, Chem. – Eur. J., 2020, 26, 10841–10848 Search PubMed
.
- E. G. Meekel and A. L. Goodwin, CrystEngComm, 2021, 23, 2915–2922 Search PubMed
.
- P. St. Petkov, V. Bon, C. L. Hobday, A. B. Kuc, P. Melix, S. Kaskel, T. Düren and T. Heine, Phys. Chem. Chem. Phys., 2019, 21, 674–680 RSC
.
- E. G. Meekel, P. Partridge, R. A. I. Paraoan, J. J. B. Levinsky, B. Slater, C. L. Hobday and A. L. Goodwin, Nat. Mater., 2024, 23, 1245–1251 CrossRef CAS PubMed
.
- S. Ehrling, E. M. Reynolds, V. Bon, I. Senkovska, T. E. Gorelik, J. D. Evans, M. Rauche, M. Mendt, M. S. Weiss, A. Pöppl, E. Brunner, U. Kaiser, A. L. Goodwin and S. Kaskel, Nat. Chem., 2021, 13, 568–574 CrossRef CAS PubMed
.
- C. Serre, F. Millange, C. Thouvenot, M. Noguès, G. Marsolier, D. Louër and G. Férey, J. Am. Chem. Soc., 2002, 124, 13519–13526 Search PubMed
.
- F. C. N. Firth, M. J. Cliffe, D. Vulpe, M. Aragones-Anglada, P. Z. Moghadam, D. Fairen-Jimenez, B. Slater and C. P. Grey, J. Mater. Chem. A, 2019, 7, 7459–7469 Search PubMed
.
- G. C. Shearer, S. Chavan, S. Bordiga, S. Svelle, U. Olsbye and K. P. Lillerud, J. Mater. Chem., 2016, 28, 3749–3761 CrossRef CAS
.
- M. Froehly, G. Chaplais, H. Nouali, V. Roucoules and T. J. Daou, New J. Chem., 2025, 49, 5694–5706 Search PubMed
.
-
APEX3, SAINT and SADABS, Bruker AXS Inc., Madison, Wisconsin, USA, 2016 Search PubMed
.
- G. M. Sheldrick, Acta Crystallogr., Sect. C:Struct. Chem., 2015, 71, 3–8 Search PubMed
.
-
G. M. Sheldrick, Program for Crystal Structure Solution, University of Göttingen, Göttingen, 2018 Search PubMed
.
- V. Petříček, L. Palatinus, J. Plášil and M. Dušek, Z. Kristallogr. Cryst. Mater., 2023, 238, 271–282 CrossRef
.
- T. D. Kühne, M. Iannuzzi, M. Del Ben, V. V. Rybkin, P. Seewald, F. Stein, T. Laino, R. Z. Khaliullin, O. Schütt, F. Schiffmann, D. Golze, J. Wilhelm, S. Chulkov, M. H. Bani-Hashemian, V. Weber, U. Borštnik, M. Taillefumier, A. S. Jakobovits, A. Lazzaro, H. Pabst, T. Müller, R. Schade, M. Guidon, S. Andermatt, N. Holmberg, G. K. Schenter, A. Hehn, A. Bussy, F. Belleflamme, G. Tabacchi, A. Glöß, M. Lass, I. Bethune, C. J. Mundy, C. Plessl, M. Watkins, J. VandeVondele, M. Krack and J. Hutter, J. Chem. Phys., 2020, 152, 194103 Search PubMed
.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 Search PubMed
.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132(15), 154104 CrossRef PubMed
.
- J.-B. Lu, D. C. Cantu, M.-T. Nguyen, J. Li, V.-A. Glezakou and R. Rousseau, J. Chem. Theory Comput., 2019, 15, 5987–5997 Search PubMed
.
- J.-P. Zhang, H.-L. Zhou, D.-D. Zhou, P.-Q. Liao and X.-M. Chen, Natl. Sci. Rev., 2018, 5, 907–919 Search PubMed
.
- F. Millange and R. I. Walton, Isr. J. Chem., 2018, 58, 1019–1035 Search PubMed
.
- G. Bussi, D. Donadio and M. Parrinello, J. Chem. Phys., 2007, 126, 014101 Search PubMed
.
- G. J. Martyna, D. J. Tobias and M. L. Klein, J. Chem. Phys., 1994, 101, 4177–4189 Search PubMed
.
- L. Sarkisov, R. Bueno-Perez, M. Sutharson and D. Fairen-Jimenez, Chem. Mater., 2020, 32, 9849–9867 Search PubMed
.
- D. Dubbeldam, S. Calero, D. E. Ellis and R. Q. Snurr, Mol. Simul., 2016, 42, 81–101 CrossRef CAS
.
- A. K. Rappe, C. J. Casewit, K. S. Colwell, W. A. Goddard and W. M. Skiff, J. Am. Chem. Soc., 1992, 114, 10024–10035 Search PubMed
.
- Y. Sakanaka, S. Hiraide, I. Sugawara, H. Uematsu, S. Kawaguchi, M. T. Miyahara and S. Watanabe, Nat. Commun., 2023, 14, 6862 Search PubMed
.
- M.-H. Lee, K. Vikrant, S. A. Younis, J. E. Szulejko and K.-H. Kim, J. Clean. Prod., 2020, 250, 119486 Search PubMed
.
- Q. Liu, N. Hoefer, G. Berkbigler, Z. Cui, T. Liu, A. C. Co, D. W. McComb and C. R. Wade, Inorg. Chem., 2022, 61, 18710–18718 Search PubMed
.
- D. D. Do and H. D. Do, Colloids Surf. A Physicochem. Eng. Asp., 2006, 277, 239–248 Search PubMed
.
- M. W. Mahoney and W. L. Jorgensen, J. Chem. Phys., 2000, 112, 8910–8922 CrossRef CAS
.
- S. W. Rick, J. Chem. Phys., 2004, 120, 6085–6093 Search PubMed
.
- L. Zhang and J. I. Siepmann, J. Phys. Chem. B, 2005, 109, 2911–2919 Search PubMed
.
- H. A. Lorentz, Ann. Phys., 1881, 248, 127–136 CrossRef
.
- R. T. Cimino, P. Kowalczyk, P. I. Ravikovitch and A. V. Neimark, Langmuir, 2017, 33, 1769–1779 Search PubMed
.
-
D. P. N. G. Nicholson, in Computer Simulation and the Statistical Mechanics of Adsorption, Academic Press Inc. Ltd, London, 1982, pp. 112–115 Search PubMed
.
-
W. J. Moore, in Physical Chemistry ed. W. J. Moore, Lowe & Brydone (Printers) Ltd, London, 1963, pp. 76–103 Search PubMed
.
- A. U. Ortiz, A. Boutin, A. H. Fuchs and F.-X. Coudert, J. Chem. Phys., 2013, 138, 174703 Search PubMed
.
- S. Leubner, R. Stäglich, J. Franke, J. Jacobsen, J. Gosch, R. Siegel, H. Reinsch, G. Maurin, J. Senker, P. G. Yot and N. Stock, Chem. – Eur. J., 2020, 26, 3877–3883 Search PubMed
.
- J. R. H. Manning, G. Donval, M. Tolladay, T. L. Underwood, S. C. Parker and T. Düren, J. Mater. Chem. A, 2023, 11, 25929–25937 Search PubMed
.
- A. A. Kamin, E. J. Brannan, K. M. Snook, S. Krajewski, P. M. Gannon, W. Kaminsky and D. J. Xiao, CrystEngComm, 2024, 26, 6396–6402 Search PubMed
.
- H. H. Chen, S. Duan, Y. Sun, X. Song and J. Yu, RSC Adv., 2020, 10, 5604–5609 RSC
.
- H. Molavi and M. S. Salimi, Sci. Rep., 2025, 15, 7074 Search PubMed
.
- L. J. W. Johnson, D. Mirani, A. Le Donne, L. Bartolomé, E. Amayuelas, G. A. López, G. Grancini, M. Carter, A. A. Yakovenko, B. A. Trump, S. Meloni, P. Zajdel and Y. Grosu, Nano Lett., 2023, 23, 10682–10686 Search PubMed
.
- L. I. FitzGerald, A. L. Sutton, A. J. Seeber, B. W. Muir and C. M. Doherty, ACS Omega, 2025, 10(18), 19206–19213 Search PubMed
.
- F. Millange and R. I. Walton, Isr. J. Chem., 2018, 58, 1019–1035 Search PubMed
.
- R. Oktavian, R. Goeminne, L. T. Glasby, P. Song, R. Huynh, O. T. Qazvini, O. Ghaffari-Nik, N. Masoumifard, J. L. Cordiner, P. Hovington, V. Van Speybroeck and P. Z. Moghadam, Nat. Commun., 2024, 15, 3898 Search PubMed
.
- J. A. Mason, M. Veenstra and J. R. Long, Chem. Sci., 2014, 5, 32–51 Search PubMed
.
- A. V. Palodkar, K. Anupam, Z. Roy, B. B. Saha and G. N. Halder, Heat Mass Tran., 2017, 53, 3155–3166 Search PubMed
.
- H. Tun and C.-C. Chen, Adsorption, 2021, 27, 979–989 Search PubMed
.
- Z. Li, K. Shi, L. Zhai, Z. Wang, H. Wang, Y. Zhao and J. Wang, Sep. Purif. Technol., 2023, 307, 122725 Search PubMed
.
- Z. Yu, D. M. Anstine, S. E. Boulfelfel, C. Gu, C. M. Colina and D. S. Sholl, ACS Appl. Mater. Interfaces, 2021, 13, 61305–61315 Search PubMed
.
- F. Jeremias, D. Fröhlich, C. Janiak and S. K. Henninger, RSC Adv., 2014, 4, 24073–24082 Search PubMed
.
- R. J. Marshall, C. L. Hobday, C. F. Murphie, S. L. Griffin, C. A. Morrison, S. A. Moggach and R. S. Forgan, J. Mater. Chem. A, 2016, 4, 6955–6963 Search PubMed
.
- O. T. Unke, S. Chmiela, H. E. Sauceda, M. Gastegger, I. Poltavsky, K. T. Schütt, A. Tkatchenko and K.-R. Müller, Chem. Rev., 2021, 121, 10142–10186 Search PubMed
.
- S. Vandenhaute, S. M. J. Rogge and V. Van Speybroeck, Front. Chem., 2021, 9, 718920 Search PubMed
.
- A. Sharma and S. Sanvito, npj Comput. Mater., 2024, 10, 237 CrossRef PubMed
.
-
CCDC 2498629: Experimental Crystal Structure Determination, 2022, DOI:10.5517/ccdc.csd.cc2pw0yk
.
| This journal is © The Royal Society of Chemistry 2026 |



