Stabilising gallium-based liquid metal alloy nanoparticles by carbon encapsulation
Imtisal
Zahid
a,
Karma
Zuraiqi
*a,
Caiden J.
Parker
 a,
Muhammad Hamza
Nazir
a,
Pierre H. A.
Vaillant
b,
Edwin L. H.
Mayes
b,
Ali
Zavabeti
a,
Susanne
Wintzheimer
de,
Vaishnavi
Krishnamurthi
a,
Dan
Yang
c,
Aaron
Elbourne
a,
Muhammad Hamza
Nazir
a,
Pierre H. A.
Vaillant
b,
Edwin L. H.
Mayes
b,
Ali
Zavabeti
a,
Susanne
Wintzheimer
de,
Vaishnavi
Krishnamurthi
a,
Dan
Yang
c,
Aaron
Elbourne
 b,
Ken
Chiang
b,
Ken
Chiang
 *a and
Torben
Daeneke
*a and
Torben
Daeneke
 *a
*a
aDepartment of Chemical and Environmental Engineering, School of Engineering, RMIT University, Melbourne, VIC 3001, Australia. E-mail: Ken.Chiang@rmit.edu.au; Torben.Daeneke@rmit.edu.au
bSchool of Science RMIT University Melbourne, VIC 3001, Australia
cSchool of Engineering, Chemical Engineering, The University of Western Australia (M017), 6009 Perth, Australia
dDepartment of Chemistry and Pharmacy, Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Egerlandstraße 1, 91058 Erlangen, Germany
eFraunhofer-Institute for Silicate Research ISC, Neunerplatz 2, 97082 Würzburg, Germany
First published on 5th January 2026
Abstract
Gallium-based liquid metal (LM) nanoparticles hold an exceptional promise for catalysis, energy storage, and printed electronics due to their high conductivity, fluidity, and dynamic catalytic surfaces. However, maintaining their mechanical and chemical stability remains a major challenge, as LM nanoparticles tend to agglomerate due to their high surface tension and are susceptible to chemical degradation, such as dissolution or leaching in reactive environments. Surface modification and encapsulation techniques are employed to enhance the mechanical and functional stability of these particles. Previously, methane pyrolysis has been considered as a route to produce high-purity hydrogen and carbon. In this work, we employ methane pyrolysis as a controllable route to synthesise carbon-encapsulated Ga-based alloy nanoparticles (NPs), where catalytic activity serves as the driving mechanism for shell formation rather than the ultimate function of the material. During pyrolysis, trimetallic Cu–Pt–Ga NPs act as transient catalytic sites that initiate carbon growth, while the resulting graphitic shell provides mechanical confinement, prevents agglomeration, and enhances resistance to leaching. By tuning alloy composition, the rate and morphology of carbon formation can be modulated, enabling precise control over the resulting core–shell architecture. Overall, the primary contribution of this work is the demonstration of a robust and general method for producing carbon-coated liquid-metal nanomaterials with tailored structural and functional properties for applications beyond catalysis.
1 Introduction
Gallium-based liquid metal (LM) alloys exhibit high conductivity and flexibility, along with the unique functional capability of self-healing, making them ideal for use in flexible electronics, catalysis, and biomedical devices.1–3 Bulk LM exhibits unique catalytic properties; its practical application is constrained by interfacial instability, limited control over active sites, and mass-transfer limitations. To overcome these challenges, LM micro/nanodroplets are produced using ultrasonication and mechanical shear mixing techniques.4,5 The LM NPs naturally form a thin oxide skin (∼3 nm) that prevents coalescence, simultaneously enabling surface modifications to improve colloidal stability and functional versatility.6 However, despite these advances, the inherent fragility of the native oxide skin poses challenges for achieving long-term stability, chemical durability, and multifunctionality required for electrocatalysis and battery applications, especially under extreme pH. To enhance the stability of LM droplets, creating LM cores within an inert shell structure could offer improved mechanical strength, chemical resistance, and surface tunability.7–11 Notably, thin carbon coatings offer a promising alternative, since the inherent insolubility of carbon in Ga prevents alloying while providing mechanical robustness, conductivity, and chemical stability.Methane pyrolysis with LM catalysts takes place at higher temperatures (900–1200 °C), methane decomposes within rising bubbles to yield hydrogen and solid carbon, with the carbon segregating from the molten phase and accumulating at the surface, thereby facilitating continuous removal.12–14 Extending this concept to the nanoscale, carbon-encapsulated LM NPs can be synthesised via methane decomposition, producing a carbon shell that has minimal chemical bonding with LM, analogous to the bulk system where the molten metal promotes carbon segregation and surface accumulation.
In this study, we used methane pyrolysis to stabilise the Ga-based alloy NPs by forming a protective carbon coating on their surface. Moreover, Ga-based alloys containing active transition metals (Cu, Pt, Pd, Ni, etc) are particularly promising for enhancing carbon production. Trimetallic Cu–Pt–Ga NPs serve both as catalytic centers and as the core material for shell formation. The carbon shell preserves the liquid nature of the core and improves resistance to mechanical stress and harsh reaction media. This indicates that the main contribution of this work lies not in creating a fully stable catalyst, but rather in demonstrating a controllable approach for synthesizing carbon-coated liquid-metal nanoparticles with tunable core–shell structures, which could have applications beyond catalysis.
2 Results and discussion
2.1 Carbon deposition on different ternary liquid metal alloy nanoparticles
A preliminary screening study was conducted by introducing Ni, Cu, Pt, W, Co, and Pd into Ga at low concentrations to assess their effects on carbon production rates during methane pyrolysis. All transition-metal additions enhanced catalytic performance relative to pure Ga (see Fig. 1a); however, the improvements observed with W and Co were minor, and these metals were therefore not considered further (see Fig. S2a, SI). Based on these findings, tri-metallic alloys were subsequently designed to investigate the possibility of synergistic effects. These alloys incorporated 3 wt% of transition metal (either Ni or Cu) together with 0.5 wt% of a noble metal (Pt or Pd). The composition was selected to reduce the reaction temperature, lower the alloy's melting point, and enable alloying at low temperatures. Interestingly, all tri-metallic samples containing Ni displayed lower performance than their bimetallic counterparts, suggesting the possibility of negative synergy. Among all catalysts, the Cu–Pt–Ga (3.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5) and Cu–Pd–Ga (3.0
96.5) and Cu–Pd–Ga (3.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5) showed the highest amount of carbon deposition, 0.137 ± 0.01 mg C mgCatalyst−1 and 0.134 ± 0.007 mg C mgCatalyst−1, respectively, as shown in Fig. 1a.
96.5) showed the highest amount of carbon deposition, 0.137 ± 0.01 mg C mgCatalyst−1 and 0.134 ± 0.007 mg C mgCatalyst−1, respectively, as shown in Fig. 1a.
Previous studies showed that Pt provides the lowest barriers for CH4 dissociation and stabilises intermediates, enabling high intrinsic activity but also predisposing the surface to carbide formation under carbon-rich environments.15,16 Pristine Cu (111) can thermodynamically activate methane but faces high kinetic barriers for C–H bond cleavage, while single-atom dopants enhance CH4 adsorption and C–H activation via electron transfer. Over time, accumulated intermediates on Cu surfaces can lead to carbon deposition and graphitisation.17 Ga further modifies these behaviours by altering metal electronic structure and carbon solubility, suppressing stable carbide formation while promoting graphitic or dispersive carbon pathways.18 In binary alloys, Pt–Ga SCALMS maintain isolated Pt sites in a liquid Ga matrix, combining strong C–H bond activation with suppressed coking and surface-localized carbon formation, yielding high hydrogen productivity.19 In contrast, Cu–Ga alloys are less reactive due to weaker CH4 adsorption and higher activation barriers, but alloying and molten-metal interfaces can still promote dissociative methane adsorption and interfacial carbon nucleation.20 These reported studies suggest that the ternary Cu–Pt–Ga catalyst compositions could integrate these effects: Pt provides highly active dehydrogenation pathways, while Cu and Ga reduce carbide stability and steer carbon toward graphitic shell formation. This synergy explains the combination of high initial activity and rapidly increasing carbon growth exhibited by Cu–Pt–Ga and Cu–Pd–Ga as shown in Fig. 1a.
Computational studies suggest that methane pyrolysis in Ga-based SCALMS may proceed through a cooperative rather than single-site mechanism. Atomically dispersed Cu or Pt atoms segregate to the liquid gas interface, where they initiate C–H activation, while the surrounding liquid Ga matrix dissolves and redistributes carbon at the nanoscale, delaying deactivation and stabilizing the single-atom sites.21 Over time, carbon supersaturation promotes the formation of ordered graphitic layers that restrict methane access.22 Thus, methane activation is likely governed by the transition metals, carbon transport by the liquid Ga phase, and catalytic performance by their interplay. Collectively, these results show that the behaviour of LM alloy catalysts is not yet fully understood and emphasise the importance of studying targeted DFT calculations, and molecular dynamics simulations to understand how different elements interact within these alloy systems.
The carbon production rate increases with higher temperatures, as shown in Fig. 1b, indicating an increase in methane conversion rate with temperature. On the other hand, the Ga-only catalyst does not exhibit a monotonic increase in carbon production with temperature, indicating that the process is not solely governed by temperature-dependent methane self-pyrolysis. Carbon production increased with time for all catalysts, with the most significant rise in the initial hours, as shown in Fig. S2 (SI). Among all the catalysts, Cu-based catalysts (Cu–Pt–Ga and Cu–Pd–Ga) showed the highest performance, significantly outperforming their Ni-based counterparts. The slower increase in carbon production over time is likely due to the accumulation of carbon deposits on the surface of the LM NPs, which block active sites and gradually reduce catalytic efficiency. Notably, the Cu–Pt–Ga alloy NPs exhibit the highest carbon deposition, suggesting enhanced catalytic activity or stronger methane cracking capability. These findings highlight the importance of reaction temperature in achieving higher carbon coating for LM nanoparticle stabilisation. When comparing the carbon production rate over different time periods, the graph in Fig. S2c shows that the Cu–Pt–Ga catalyst exhibits pronounced early-stage carbon growth that gradually slows as the reaction progresses. However, as the carbon shell thickens and becomes more ordered, methane molecules experience increasing diffusion resistance, reducing their access to the Cu–Pt–Ga core over 12 h. As a result, although carbon continues to accumulate, the formation rate steadily declines, reflecting the progressive encapsulation of the active alloy surface and the corresponding decrease in methane catalyst interaction over extended operation. The accumulated carbon progressively developed into shell-like architectures that encapsulated the Cu–Pt–Ga core.
2.2 Controlling the carbon shell in the core–shell structures
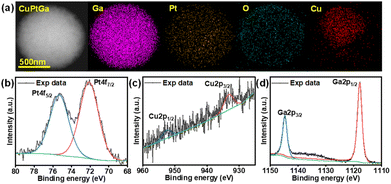
To further investigate the surface composition, morphology, and structural changes of LM NPs, as well as the nature of the carbon shell formed, the LM NPs were examined with different techniques. After methane pyrolysis, the surface and elemental composition of the prepared ternary alloy NPs were studied using different analytical techniques, including TEM, XPS, and SEM. The elemental composition and distribution of Cu and Pt in Ga-based NPs were analyzed using TEM-EDS. Fig. 2a shows NPs with a homogeneous distribution of Ga and Pt with a Cu-rich core at their centre.Fig. 2b–d displays the XPS spectra, revealing the presence of Ga2p, Cu2p and Pt4f spectra. Given the surface-sensitive nature of XPS, the spectra suggest that the solid intermetallic core is deeply embedded within the NPs. Consequently, the Cu concentration detected by XPS is substantially lower, approaching the detection limit, with Cu2p3/2 and Cu2p1/2 peaks observed at approximately 933.3 eV and 952.7 eV, respectively. These binding energy values indicate the presence of Cu metal and are consistent with prior studies.23 XPS spectra show that the surface is mainly composed of the Ga2p region and the Pt4f region with prominent peaks. Ga3+ is the predominant species on the droplet surface, indicating the formation of gallium oxide on the surface upon exposure to air, which aligns with the literature.5Fig. 2d shows that the Ga (2p3/2 and 2p1/2) peaks were detected at 1144.5 and 1117.8 eV, respectively. The observed peaks in the Pt4f region are visible in Fig. 2b as the Pt lies near the surface.24,25 The positions of Pt4f7/2 and Pt4f5/2 peaks were observed at 75.38 eV and 72.08 eV.
In contrast, the active metal Pd is not detectable in Cu–Pd–Ga NPs, and this is attributed to the low Pd concentrations, as shown in Fig. S3a, see SI.26 The pronounced Ga2p3/2 and Ga2p1/2 peaks (1145.9 and 1119.1 eV), along with the Ga![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu ratio, reveal that the surface is predominantly composed of Ga and an oxide layer, as shown in Fig. S3a. This finding aligns with earlier research on Ga-based LM alloyed with Cu.27 Despite the differences in the metal ratios, it can be observed that the Ga and survey scans for every sample in the XPS data set are comparable, as shown in Fig. S3c and d (see SI). While the TEM-EDS shows the presence of solute metals in the NPs (Fig. S3b).
Cu ratio, reveal that the surface is predominantly composed of Ga and an oxide layer, as shown in Fig. S3a. This finding aligns with earlier research on Ga-based LM alloyed with Cu.27 Despite the differences in the metal ratios, it can be observed that the Ga and survey scans for every sample in the XPS data set are comparable, as shown in Fig. S3c and d (see SI). While the TEM-EDS shows the presence of solute metals in the NPs (Fig. S3b).
After carbon encapsulation, the change in elemental composition of the NPs was observed. XPS shows the presence of Ga2p peaks near 1118.8 eV and 1145.7 eV, and Cu was not detected on the surface of Cu–Pd–Ga NPs (as shown in Fig. S4b, see SI). The alloy NPs surface exhibits a minor shift in peaks, primarily gallium oxide (Ga2O3), on the surface of Cu–Pd–Ga after methane pyrolysis. On the other hand, the XPS scan of Pt4f (72.7 eV and 75.9 eV) and Ga2p (1145.6 eV and 1118.7 eV) for the Cu–Pt–Ga NPs confirms that gallium present at the surface exists in the Ga(III) oxidation state, as shown in Fig. S4a. TEM-EDS analysis of Cu–Pd–Ga and Cu–Pt–Ga nanoparticles (minor shape change), along with individual elemental maps of Cu, Pt, Pd, O, and Ga, confirms that all elements are retained in the NPs after the reaction (see SI, Fig. S5a and b). Although after reaction, XPS shows Cu near the detection limit, TEM confirm its presence in both pristine and post-catalyst NPs, and literature reports that liquid Ga can rapidly redistribute or transiently segregate to the interface under reaction conditions.28,29 We therefore hypothesise that Cu may briefly occupy catalytically relevant interfacial sites or electronically modify neighbouring Ga–Pt ensembles during methane pyrolysis, even if it re-dissolves into the Ga matrix or becomes buried beneath carbon by the end of the reaction, explaining its catalytic influence despite low post-reaction surface detectability.
Fig. 3a and b show the SEM images that depict the change in morphology and size distribution of the Cu–Pt–Ga and carbon formation Cu–Pt–Ga NPs obtained after methane pyrolysis. The SEM imaging of Cu–Pt–Ga before the reaction has an average particle size of 825 ± 27 nm. After methane pyrolysis, the carbon encapsulated (CC) Cu–Pt–Ga NPs exhibit both a spherical shape and irregular-shaped agglomeration of NPs, with a few instances of tighter aggregation, as shown in Fig. 3b. To investigate the elemental composition and distribution of the carbon product formed after the reaction, SEM-EDS analysis was carried out. Fig. 3f shows the presence of carbon flake-like structures surrounded by alloy NPs. Moreover, the close-up images reveal different carbon layers stacked together to form the carbon flake-like structure (highlighted with a yellow arrow, see SI, Fig. S7b). The elemental mapping showed the thick carbon matrix surrounded by NPs containing all active metals present in the CC–Cu–Pt–Ga NPs (see Fig. 3f–k). Most of the NPs (CC–Cu–Pd–Ga) retain their spherical shape after the reaction, as observed on the surface in Fig. 3d. As shown in Fig. 3g–q, SEM-EDS confirms that the produced carbon is surrounded by NPs retaining all the constituent alloy elements after the reaction.
All ternary Ni-based nanoparticles showed aggregation or surface deformation after methane pyrolysis, likely causing their low catalytic activity and limited carbon encapsulation (shown in Fig. S5). Moreover, Ni–Cu-Ga showed the highest aggregation, while Ni–Pt–Ga formed spherical agglomerates embedded in the carbon matrix, with SEM-EDS confirming the presence of Ga, Ga2O3, and a uniform Ni/Pt distribution (Fig. S6d). Ni–Pd–Ga displayed surface deformation and Pd clustering near large, porous carbon flakes (15–40 µm, Fig. S6c). Among all the catalysts, the Cu–Pt–Ga and Cu–Pd–Ga NPs showed effective encapsulation during methane pyrolysis, with EDS confirming that the active elements remained inside the NPs post-encapsulation.
Raman spectroscopy was used to provide insights into the nature of the carbon deposits formed over the alloy NPs, e.g., the degree of graphitisation and disorder. Specifically, a lower D/G ratio corresponds to a higher degree of graphitization, meaning that the sp2 carbon domains are more ordered and contain fewer structural defects as reported in literature.30,31 All binary alloy nanoparticles confirmed carbon formation; however, only Pd–Ga showed graphitic carbon, evidenced by a distinct 2D peak in the Raman spectra (see Fig. S9d, SI). The Raman spectra of Cu-based ternary alloy NPs exhibit three distinct peaks: the D-band (∼1350 cm−1), G-band (∼1590 cm−1), and 2D-band (∼2700 cm−1), as seen in Fig. 3e. Notably, the Cu–Pd–Ga NPs show a well-defined G-band at 1590 cm−1, associated with in-plane vibrations of sp2-bonded carbon atoms, and a sharp 2D-band around 2670 cm−1, indicative of graphitic carbon structures. The pronounced 2D-band, an overtone of the D-band, is a hallmark of multilayer graphene and is highly sensitive to the number of layers and degree of graphitisation.32,33 Among the various LM alloy NPs, the Cu-based catalysts, including Cu–Pd–Ga and Cu–Pt–Ga, consistently show more structured and sharper Raman features, suggesting a higher degree of graphitisation in the resulting carbon. For instance, the 2D-band of Cu–Pt–Ga NPs is both broadened and upshifted, pointing to few-layer graphene and interlayer interactions typical of more ordered carbon. The Cu-based NPs (Cu–Pt–Ga and Cu–Pd–Ga) displaying a D/G ratio around 1.12 and 1.13, indicating a moderate defective carbon structure with curved graphene domains, despite still producing distinguishable G and 2D bands.
In contrast, Ni-containing NPs (Ni–Pt–Ga, Ni–Pd–Ga and Ni–Cu-Ga), only Ni–Cu-Ga shows graphitic features (e.g., a 2D-band at 2681 cm−1), but their spectra more closely resemble that of bulk graphite. In Ni-based catalysts, the trend of increasing disorder is even more pronounced. Ni–Pt–Ga exhibits a highest D/G ratio (1.60), accompanied by diffuse Raman features indicative of highly defective or partially amorphous carbon. Ni–Cu-Ga displays a lowest D/G ratio of 0.90, higher degree of graphitization while Ni–Pt–Ga shows D/G ratio 1.05, reflecting moderately disordered carbon as reported.34
This trend highlights that Cu-based alloy NPs tend to catalyse the formation of ordered, well-graphitized carbon structures, as reflected by their sharper peaks and varied ID/IG ratios. The resulting graphene layers encapsulate and stabilise the NPs, forming core–shell-like structure. The addition of nickel disrupts graphitization, increasing disorder. However, introducing a third metal in ternary alloys reduces defects as compared to binary alloys, as seen by moderate D/G ratios (0.99–1.13), indicating partially ordered graphitic structures.35 Furthermore, the SEM-EDS showed the elemental distribution of Cu–Pt–Ga and Cu–Pd–Ga NPs, indicating the thick carbon regions in Fig. S10a and b.
The morphology of encapsulated Cu–Pt–Ga and Cu–Pd–Ga NPs was observed by HR-TEM and SAED after separation. Fig. 4a illustrates the diffraction pattern of multi-layer graphene, showing that the inner spots exhibit greater intensity compared to the outer spots.36 The HR-TEM imaging shows the presence of tri-layer, and five-layer graphene structures formed after methane conversion by the Cu–Pt–Ga alloy NPs (see Fig. 4b, yellow box). The spherical, core–shell structure of ternary NPs wrapped in graphene sheets formed during methane pyrolysis. Previous studies have demonstrated that carbon generated on the catalyst surface subsequently dissolves into the metal and precipitates at particle dislocations, leading to the formation of graphite.37 This indicates that Cu–Pt–Ga (03![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5) NPs are surrounded by graphitic carbon sheets, as observed in Fig. 4a. The crystalline structures of graphite (002), and (110) are shown by selected-area electron diffraction (SAED) patterns, which measure around 0.34 and 0.21 nm−1 (graphite) as shown in Fig. 4c, respectively.38,39 It was also confirmed that the crystal, as shown in Fig. 4c (highlighted in green colour), confirmed the presence of gallium oxide (300) entrapped within graphitic carbon shell. The yellow highlighted box in Fig. 4b shows multilayer graphene sheets with a lattice spacing of 0.34 nm.
96.5) NPs are surrounded by graphitic carbon sheets, as observed in Fig. 4a. The crystalline structures of graphite (002), and (110) are shown by selected-area electron diffraction (SAED) patterns, which measure around 0.34 and 0.21 nm−1 (graphite) as shown in Fig. 4c, respectively.38,39 It was also confirmed that the crystal, as shown in Fig. 4c (highlighted in green colour), confirmed the presence of gallium oxide (300) entrapped within graphitic carbon shell. The yellow highlighted box in Fig. 4b shows multilayer graphene sheets with a lattice spacing of 0.34 nm.
The HR-TEM image shows graphitic lattices on the nanoparticle's surface, indicating a typical graphene structure as reported in the literature.40,41 With a lattice spacing of roughly 3.4 Å, three-to-five-layer graphene can be seen in Fig. 4e (yellow box). Selected-area electron diffraction (SAED) patterns obtained from the red box region were used to further confirm the presence of multi-layer graphene. By comparing with the powder diffraction pattern, it was confirmed from SAED diffraction the presence of graphitic lattices (111), (110) and (100) (see Fig. 4f, red highlighted).38 The number of graphene layers was visually evaluated using TEM analysis to determine whether monolayer or few-layer graphene was present. Similar graphene structures, as reported previously, are observed in TEM imaging, as presented in Fig. 4e.41 The region indicated by the yellow box in the high-resolution TEM image shows clearly the presence of multi-layer graphene with a smooth edge, dominated by many dark lines.41 Thin graphene sheet aggregates, which show the agglomeration of graphitic structures, can be seen in Fig. 4e (highlighted with yellow arrows). The difference in carbon morphology and crystallinity obtained using the various ternary catalysts proves that the catalyst composition significantly influences the carbon produced during the methane pyrolysis reaction.
The TEM images 4 h shows well-defined graphitic layers surrounding the particles, while the diffuse contrast within the shell together with the SAED features (Fig. 4c), indicates that portions of the oxide become incorporated during carbon growth. This creates a stable mixed oxide–carbon interface that acts as a robust template for carbon encapsulation. The resulting hybrid structure could enhance the uniformity and mechanical stability of the nanoparticles. Fig. 4g and h show the thickness of the carbon shell measured at 3 h intervals, revealing an alloy core encapsulated within the carbon shell. Nanoparticles of different size ranges produced carbon coatings of varying thicknesses during methane pyrolysis. Nanoparticles in the 100–300 nm range formed a carbon shell of average thickness 4.5 ± 0.7 nm. Furthermore, the NPs 301–600 nm range formed a shell having an average thickness of 7.3 ± 2.8 nm. Similarly, the average thickness of the carbon shell around 601–900 nm particles was found to be 9.5 ± 1.8 nm, while larger particles (900–1500 nm) exhibited a reduced carbon layer thickness of 6.2 ± 0.8 nm. After a 12 h reaction period, the carbon shells exhibit a slight increase in thickness, as shown in Fig. 4j. During methane pyrolysis, core–shell nanostructures form, stabilising alloy NPs through encapsulation by graphitic carbon. After reaction, a carbon shell forms around the liquid metal NPs, effectively encapsulating and protecting them from agglomeration or structural degradation.
2.3 Stability of the carbon-coated liquid metal nanoparticles
Mechanical stress testing was performed to evaluate the structural stability of fresh Cu–Pt–Ga and carbon-coated Cu–Pt–Ga NPs (CC–Cu–Pt–Ga) under applied force. After applying a force of 1 N under shearing motion, the Cu–Pt–Ga NPs ruptured, agglomerated and metallic gallium can be seen on the surface of the glass slide as shown in Fig. S12a (highlighted in red arrows). On the other hand, the CC–Cu–Pt–Ga withstands the mechanical stress test, showing that the carbon shell protects the NPs from pressure-induced agglomeration, as shown in Fig. S12b.The morphology of NPs observed by SEM imaging showed that uncoated Cu–Pt–Ga particles underwent significant deformation and coalescence due to the weak mechanical resistance of their native Ga2O3 shell, which ruptured easily under minimal pressure (Fig. S13). In contrast, CC–Cu–Pt–Ga maintained their morphology and resisted droplet aggregation even after applied stress, as the carbon shell effectively preserved particle integrity (Fig. S14). These results confirm that the carbon coating provides mechanical reinforcement, allowing the liquid metal core to remain encapsulated and stable under stress conditions. The NPs remained intact during testing, as confirmed by consistent before and after imaging during analysis.
To further confirm the improved mechanical stability of the carbon-coated Cu–Pt–Ga particles in comparison to the noncoated particles, atomic force microscopy (AFM) measurements were carried out. The AFM force curves show the distinct peaks obtained from Cu–Pt–Ga and carbon-coated Cu–Pt–Ga NPs. The AFM force–distance curves for the two samples, Cu–Pt–Ga and carbon-coated Cu–Pt–Ga alloy NPs, showed distinct mechanical interaction profiles in Fig. 5a and b. The resistance to rupturing signifies greater surface stiffness and a robust outer layer. In contrast, the noncoated Cu–Pt–Ga displays much smoother and minimal peak features in Fig. 5a. This suggests a softer, more uniform surface, likely due to the absence of a protective carbon layer and direct exposure of the liquid metal or its native oxide. On the other hand, the CC–Cu–Pt–Ga alloy NPs show a more complex structure with multiple peaks, indicating increased surface interactions and structural resistance during indentation. The first noticeable layer rupture occurred nearly at 400 nm, followed by the second rupture approximately at 335 nm with a lower applied force. These fluctuations in the peak could correspond to the resistance encountered during rupture, indicating the presence of a complex shell likely consisting of a graphitic carbon layer and a gallium oxide layer, as shown in Fig. 5b. This indicates that despite the encapsulation, the LM alloy NPs retain their liquid nature.
The dynamic light scattering (DLS) measurements presented critical insights into the chemical stability of carbon-coated and uncoated Cu–Pt–Ga alloy NPs catalysts after acid treatment (Fig. S16a and b, see SI). Fig. S17a (see SI) shows that for both 0.01 M and 0.1 M HCl, the majority of CC–Cu–Pt–Ga NPs exhibited relatively stable particle size distributions, remaining below approximately 1000 nm. In contrast, a distinct shift was observed under strongly acidic conditions (1 M HCl) after 3 h, where the fresh Cu–Pt–Ga sample shows a significant increase in particle size with a broader distribution, indicating possible surface corrosion, agglomeration, or even reprecipitation effects. The CC–Cu–Pt–Ga samples under the same conditions (1 M HCl, after 3 h) have a consistent particle size distribution around 1200 nm. This stability is further reinforced after 48 h, as shown in Fig. S17b (see SI), where the coated samples across all acid concentrations show minimal change in particle size, while the non-coated samples continue to exhibit signs of growth and instability, most significantly observed after treatment in 1 M HCl solution. The chemical stability of the carbon-coated particles was confirmed by SEM imaging, shown in Fig. S18. The SEM-EDS results indicate that the carbon encapsulation minimises leaching and prevents aggregation, enhancing the overall chemical durability of liquid metal NPs, especially in corrosive or acidic environments common in catalytic and electrochemical applications.
3 Methodology
3.1 Catalyst synthesis
Gallium (99.9%) was purchased from Indium Corporation. Further, supplies were sourced from Sigma-Aldrich and included 99% pure anhydrous sodium acetate (NaOAc) and metal powders of copper, palladium, platinum, nickel, and tungsten, having a purity of 99.9%. Ethanol (C2H5OH) (99.5%) was purchased from Thermo Fisher Scientific.3.2 Liquid metal alloy synthesis
The preparation of different liquid metal alloy catalysts was conducted inside a nitrogen-filled glovebox to prevent metal oxidation and facilitate the alloying process. Alloys investigated included Ni–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97), Pt–Ga (0.5
97), Pt–Ga (0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99.5), Pd–Ga (0.5
99.5), Pd–Ga (0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99.5), Cu–Ga (3
99.5), Cu–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97), Ni–Pt–Ga (3
97), Ni–Pt–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5), Ni–Pd–Ga (3
96.5), Ni–Pd–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5), Cu–Pt–Ga (3
96.5), Cu–Pt–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5) and Cu–Pd–Ga (3
96.5) and Cu–Pd–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5), with weight percentages. Alloying was achieved by mechanical mixing by means of a mortar and pestle at 400 °C. Alloys with higher melting temperature, including W–Ga (3
96.5), with weight percentages. Alloying was achieved by mechanical mixing by means of a mortar and pestle at 400 °C. Alloys with higher melting temperature, including W–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97), Co–Ga (3
97), Co–Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97), and Ni–Cu-Ga (3
97), and Ni–Cu-Ga (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 03
03![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 93), were heated in a tube furnace at 800 °C (at a temperature ramping rate of 10 °C min−1) for 12 h under an argon flow (0.1 L min−1) to prevent oxidation. The alloying temperature was selected based on phase diagrams to ensure that the liquid metal alloy remained in the liquid phase.42 Upon completion of the alloying process, the alloys were cooled to room temperature and stored at 4 °C. These alloys are the precursor materials for preparing alloy NPs.
93), were heated in a tube furnace at 800 °C (at a temperature ramping rate of 10 °C min−1) for 12 h under an argon flow (0.1 L min−1) to prevent oxidation. The alloying temperature was selected based on phase diagrams to ensure that the liquid metal alloy remained in the liquid phase.42 Upon completion of the alloying process, the alloys were cooled to room temperature and stored at 4 °C. These alloys are the precursor materials for preparing alloy NPs.
3.3 Preparation of alloy nanoparticles
Alloy NPs were prepared following a previously published procedure.5 In brief, the materials were broken into nanodroplets in molten sodium acetate (NaOAc) through high-temperature sonication. A hotplate was used to melt 15.3 g of NaOAc in a glass vial at 400 °C. The hotplate was fitted with a custom-made aluminium heating block that featured a cavity matching the shape and size of the glass vial to ensure effective heat transfer and consistent heating as shown in Fig. S1. A 1.5 g solid alloy sample was added to the molten sodium acetate and allowed to stir using a magnetic stirrer.43 The liquid metal was subjected to 30 min of sonication using a SCIENTZ-IID probe sonicator (6 mm titanium tip, 300 W) at 400 °C operated in a 3 s on/off pulsation cycles, while the temperature was constantly monitored by a thermocouple. After the sonication process, the mixture was allowed to cool and solidify. The nanodroplets were purified by washing with deionized water to remove the sodium acetate. The product was recovered via vacuum filtration using a sterlitech polyethylene terephthalate (PETE) membrane (47 mm diameter and 400 nm pore size). The droplets were subsequently re-dispersed in ethanol and then dried in a vacuum oven for storage. The homogeneity of the synthesised nanodroplet compositions obtained using this process highlights the efficacy of sonication as a robust technique for achieving balanced dispersion of metals within multi-element LM alloy systems at the nanoscale.44,453.4 Methane pyrolysis for LM alloy nanoparticles encapsulation
The deposition of carbon on to alloy materials via methane pyrolysis was carried out inside a tube furnace. Quartz tubes (1000 mm long, 32 mm I.D. and 35 mm O.D.) were used as reactors, inside which round ceramic pans containing 20 mg of catalyst were placed. The reaction was carried out at the following reaction conditions: reaction temperature: 600–800 °C, reaction time: 180–720 min, flow rate of 4% methane/argon: 0.1 L min−1 gas. After cooling, carbon products were quantified by measuring the catalyst weight change before and after reaction using a weighing balance and PerkinElmer Thermogravimetric Analyser (TGA 8000).3.5 Characterisation techniques
A JEOL F200 Cold Field Emission Gun (CFEG) transmission electron microscope (TEM) was used to capture the images of alloy NPs at an accelerating voltage of 200 kV. Imaging and analysis were conducted with a Gatan Rio16 4k charge-coupled device camera (model 1816) and the Gatan Digital Micrograph software (version 3.43.3213.0). For energy-dispersive X-ray spectroscopy (EDS), an Oxford X-Maxn 80T X-ray spectrometer was used along with Aztec software, which enabled elemental mapping of each nanoparticle. A Nano Nova scanning electron microscopy (SEM) operating at an accelerating voltage of 3 kV was utilised for imaging. SEM-EDS elemental mapping data were collected using an SEM instrument equipped with an Oxford Instruments Xmax EDS detector.X-ray photoelectron spectroscopy (XPS) data were collected using a Thermo Scientific K-alpha XPS spectrometer, which is equipped with a concentric hemispherical electron analyser and utilizes an X-ray source with an energy of 1486.7 eV (Al Kα). The data was processed using CASA XPS software. Additionally, Raman spectroscopy was conducted with a 532 nm laser using a HORIBA HR Evolution (12912a). The Raman shifts in the range of 900–3000 cm−1 were employed to detect carbon formation, and Origin Pro 2023 was used to plot the resulting data.
The Cu–Pt–Ga and carbon-coated Cu–Pt–Ga alloy NPs shell robustness was investigated using a Cypher ES atomic force microscope (from Oxford Instruments, Asylum Research, Santa Barbara, CA, USA) at room temperature (25 °C). Dynamic Light Scattering (DLS) data were collected using an ALV 5022F, utilizing a laser wavelength of 632.8 nm and measured at a 90° scattering angle.
3.6 Recovery of carbon shell
To separate carbon shells from alloy NPs after methane pyrolysis, a sample was suspended in 5 mL of aqua regia heated at 60 °C for 60 min under continuous stirring. After heating, the samples were washed and filtered to separate the carbon shells for further analysis.3.7 Mechanical and chemical stability of carbon-coated nanoparticles
To assess mechanical stability, the sample was drop-cast onto a silicon wafer mounted on a glass slide and then covered with another glass slide, forming a sandwich structure with the wafer positioned between the two slides. Gallium was added into the vial and weighed until it reached 1 N mass. The two glass slides are then pressed against each together under a constant force of 1 N (as shown in Video S1) and allowed to slide over each other. The morphological changes of the nanodroplets after the sliding test were then examined under the SEM.Force curves of samples are also collected by using an atomic force microscope (AFM). For analysis, a nanodroplet sample was drop-casted onto a pure silicon wafer. The AFM analysis was performed using a Cypher ES instrument (Oxford Instruments, Asylum Research) with a 100% silicon cantilever tip at 29 °C under atmospheric conditions. Force measurements were performed using a Cypher ES atomic force microscope (Oxford Instruments, Asylum Research, Santa Barbara, CA, USA) in contact mode, penetrating the nanodroplet oxide layer and any underlying structures. With a trigger force set at 600 nN (trigger voltage ranging from 2 to 4 V, depending on the cantilever spring constant), individual nanodroplets were targeted and probed across multiple locations on the substrate.46,47 Multiple nanodroplets from one sample were analyzed, and the resulting force curves were processed using Asylum Research software, custom Python scripts, and Gwyddion. The values of Young's Modulus were calculated in Wave Metrics, assuming a conical silicon tip (a Poisson's ratio of 0.17 and a modulus of 150 GPa).
Chemical stability was assessed by preparing hydrochloric acid solutions of 1.0 M (pH 0), 0.1 M (pH 1), and 0.01 M (pH 2) and suspending the alloy samples in each solution. The particle size distribution of the suspensions was then evaluated after 3 h and 48 h of mixing. Before particle size measurements, 5 mL of suspension from each sample was transferred to an 8 mm diameter cylindrical glass cuvette and maintained at a constant temperature of 20 °C. Each particle size measurement was run for 30 s and repeated three times. The resulting correlation functions were analyzed using the CONTIN algorithm to determine the particle size distribution.
4 Conclusions
This study demonstrates a methane pyrolysis-based strategy for synthesising Ga-based liquid metal (LM) nanoparticles encapsulated in graphitic carbon shells. The method effectively prevents agglomeration, preserves the liquid nature of the core, and produces uniform, mechanically robust carbon-metal hybrid nanostructures. During synthesis, the Cu–Pt–Ga (3.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 96.5 wt%) alloy exhibits the highest catalytic activity, enabling efficient methane decomposition and controlled graphitic carbon growth, particularly at 600 °C. Structural characterisation (SEM, HR-TEM, SAED) confirms the formation of multilayer graphitic shells surrounding the LM cores. A key insight of this work is that catalytic activity and carbon-shell formation are intrinsically linked: initial carbon deposition enables catalysis, while continued graphitisation naturally transitions the system into a stable, encapsulated LM nanoparticle. This self-limiting process highlights the primary contribution of the study, a scalable, catalytic route to produce carbon-coated LM nanoparticles with enhanced stability. Overall, the work provides a practical synthesis platform for generating multifunctional LM-based nanomaterials, with transient catalytic behaviour serving as a constructive step toward achieving durable core–shell architectures.
96.5 wt%) alloy exhibits the highest catalytic activity, enabling efficient methane decomposition and controlled graphitic carbon growth, particularly at 600 °C. Structural characterisation (SEM, HR-TEM, SAED) confirms the formation of multilayer graphitic shells surrounding the LM cores. A key insight of this work is that catalytic activity and carbon-shell formation are intrinsically linked: initial carbon deposition enables catalysis, while continued graphitisation naturally transitions the system into a stable, encapsulated LM nanoparticle. This self-limiting process highlights the primary contribution of the study, a scalable, catalytic route to produce carbon-coated LM nanoparticles with enhanced stability. Overall, the work provides a practical synthesis platform for generating multifunctional LM-based nanomaterials, with transient catalytic behaviour serving as a constructive step toward achieving durable core–shell architectures.
Author contributions
Imtisal Zahid: writing—original draft (lead). Karma Zuraiqi: supervision (equal); writing—review & editing (supporting). Caiden J. Parker: resources (supporting); validation (supporting). Muhammad Hamza Nazir: writing—review & editing (supporting). Pierre H. A. Vaillant: data curation (supporting). Edwin L. H. Mayes: resources (supporting); validation (supporting). Ali Zavabeti: data curation (supporting); resources (supporting). Vaishnavi Krishnamurthia: resources (supporting). Dan Yang: validation (supporting). Aaron Elbourne: conceptualization (supporting); data curation (supporting). Ken Chiang: conceptualization; project administration. Susanne Wintzheimer: data curation (supporting); methodology (supporting). Torben Daeneke: conceptualization (equal); supervision (equal).Conflicts of interest
The authors declare no conflict of interest.Data availability
The data that support the findings of this study, including raw and processed characterisation data (SEM, TEM, AFM, Raman spectroscopy, DLS, XRD, XPS, and methane pyrolysis reaction data) are available in the institutional repository of RMIT University at [https://www.microsoft.com/en-au/microsoft-365/onedrive/online-cloud-storage].Supplementary information (SI): full experimental protocols, supplementary figures and supporting tables is included in the supplementary information associated with the manuscript. Additional demonstration videos are also made available with this submission. See DOI: https://doi.org/10.1039/d5ta08460a.
Acknowledgements
The authors acknowledge the facilities, and the scientific and technical assistance of the RMIT University's Microscopy & Microanalysis Facility, a linked laboratory of the Microscopy Australia, enabled by NCRIS. This research has been supported by funding via the Australian Research Council (ARC) Discovery Project scheme (DP220101923, DP240101215). A. Z. would like to acknowledge support received from the ARC-DECRA grant DE240100743. S. W. and T. D., K. C. and K. Z. acknowledge funding by BMFTR (MobiliKat/01DR24007).References
- T. Daeneke, K. Khoshmanesh, N. Mahmood, I. A. De Castro, D. Esrafilzadeh and S. J. Barrow, et al., Liquid metals: fundamentals and applications in chemistry, Chem. Soc. Rev., 2018, 47(11), 4073–4111 RSC.
- D. H. Lee, T. Lim, J. Pyeon, H. Park, S. W. Lee and S. Lee, et al., Self-Mixed Biphasic Liquid Metal Composite with Ultra-High Stretchability and Strain-Insensitivity for Neuromorphic Circuits, Adv. Mater., 2024, 36(16), 2310956 CrossRef CAS PubMed.
- H. Song, T. Kim, S. Kang, H. Jin, K. Lee and H. J. Yoon, Ga-Based liquid metal micro/nanoparticles: recent advances and applications, Small, 2020, 16(12), 1903391 CrossRef CAS PubMed.
- X. Zhao, M. Qiao, Y. Zhou and J. Liu, Liquid metal droplet dynamics, Droplet, 2024, 3(1), e104 CrossRef.
- C. J. Parker, V. Krishnamurthi, K. Zuraiqi, C. K. Nguyen, M. Irfan and F. Jabbar, et al., Synthesis of Planet-Like Liquid Metal Nanodroplets with Promising Properties for Catalysis, Adv. Funct. Mater., 2024, 34(31), 2304248 CrossRef CAS.
- N. J. Morris, Z. J. Farrell and C. E. Tabor, Chemically modifying the mechanical properties of core–shell liquid metal nanoparticles, Nanoscale, 2019, 11(37), 17308–17318 RSC.
- C. Chiew, M. J. Morris and M. H. Malakooti, Functional liquid metal nanoparticles: synthesis and applications, Mater. Adv., 2021, 2(24), 7799–7819 RSC.
- S.-Y. Tang and R. Qiao, Liquid metal particles and polymers: A soft–soft system with exciting properties, Acc. Mater. Res., 2021, 2(10), 966–978 CrossRef CAS.
- G. Zhu, Y. Tian, J. Tan, Q. Wu, L. Tan and X. Ren, et al., Fabrication of core–shell liquid metal@ silica nanoparticles for enhanced mechanical, dielectric and thermal properties of silicone rubber, CrystEngComm, 2023, 25(22), 3299–3307 RSC.
- V. Okatenko, L. Castilla-Amorós, D. C. Stoian, J. Vávra, A. Loiudice and R. Buonsanti, The native oxide skin of liquid metal Ga nanoparticles prevents their rapid coalescence during electrocatalysis, J. Am. Chem. Soc., 2022, 144(22), 10053–10063 CrossRef CAS PubMed.
- H. Bark and P. S. Lee, Surface modification of liquid metal as an effective approach for deformable electronics and energy devices, Chem. Sci., 2021, 12(8), 2760–2777 RSC.
- M. R. Gunarayu, M. F. A. Patah and W. M. A. W. Daud, Advancements in methane pyrolysis: A comprehensive review of parameters and molten catalysts in bubble column reactors, Renewable Sustainable Energy Rev., 2025, 210, 115197 CrossRef CAS.
- D. C. Upham, V. Agarwal, A. Khechfe, Z. R. Snodgrass, M. J. Gordon and H. Metiu, et al., Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon, Science, 2017, 358(6365), 917–921 CrossRef CAS PubMed.
- L. Chen, Z. Song, S. Zhang, C.-K. Chang, Y.-C. Chuang and X. Peng, et al., Ternary NiMo-Bi liquid alloy catalyst for efficient hydrogen production from methane pyrolysis, Science, 2023, 381(6660), 857–861 CrossRef CAS PubMed.
- G. W. Watson, R. P. Wells, D. J. Willock and G. J. Hutchings, A comparison of the adsorption and diffusion of hydrogen on the {111} surfaces of Ni, Pd, and Pt from density functional theory calculations, J. Phys. Chem. B, 2001, 105(21), 4889–4894 CrossRef CAS.
- Z. Al-Auda and K. L. Hohn, Theoretical Study of Methane Dissociation on Pt (111) Surface Using Density Functional Theory (DFT) Calculations, Bull. Chem. React. Eng. Catal., 2023, 18(3), 499–505 CrossRef CAS.
- J. Wang and G.-C. Wang, Promotion effect of methane activation on Cu (111) by the surface-active oxygen species: a combination of DFT and ReaxFF study, J. Phys. Chem. C, 2018, 122(30), 17338–17346 CrossRef CAS.
- N. Raman, S. Maisel, M. Grabau, N. Taccardi, J. Debuschewitz and M. Wolf, et al., Highly effective propane dehydrogenation using Ga–Rh supported catalytically active liquid metal solutions, ACS Catal., 2019, 9(10), 9499–9507 CrossRef CAS PubMed.
- N. Raman, M. Wolf, M. Heller, N. Heene-Würl, N. Taccardi and M. Haumann, et al., GaPt supported catalytically active liquid metal solution catalysis for propane dehydrogenation–support influence and coking studies, ACS Catal., 2021, 11(21), 13423–13433 CrossRef CAS PubMed.
- Y.-F. Huo, L. Zhou, M. Feng and T.-L. Hu, Theoretical study of Cu-based alloy catalysts for oxidative coupling of methane, J. Mater. Chem. A, 2025, 13(21), 15928–15940 RSC.
- X. Sun and H. Li, Recent progress of Ga-based liquid metals in catalysis, RSC Adv., 2022, 12(38), 24946–24957 RSC.
- M. Wolf, N. Raman, N. Taccardi, R. Horn, M. Haumann and P. Wasserscheid, Capturing spatially resolved kinetic data and coking of Ga–Pt supported catalytically active liquid metal solutions during propane dehydrogenation in situ, Faraday Discuss., 2021, 229, 359–377 RSC.
- H. Li, W. Yin, C. K. Ng, R. Huang, S. Du and M. Sharma, et al., Macroporous perovskite nanocrystal composites for ultrasensitive copper ion detection, Nanoscale, 2022, 14(33), 11953–11962 RSC.
- M. H. Nazir, T. C. Le, I. Zahid, K. Zuraiqi, M. P. Aukarasereenont and C. J. Parker, et al., Liquid Metal Electrocatalyst with Ultralow Pt Loading for Ethanol Oxidation, Small Sci., 2025, 5(1), 2400370 CrossRef CAS PubMed.
- A. I. Stadnichenko, E. M. Slavinskaya, O. A. Stonkus and A. I. Boronin, Low-Temperature CO Oxidation by the Pt/CeO2 Based Catalysts, ChemCatChem, 2024, 16(15), e202301727 CrossRef CAS.
- A. G. Shard, Detection limits in XPS for more than 6000 binary systems using Al and Mg Kα X-rays, Surf. Interface Anal., 2014, 46(3), 175–185 CrossRef CAS.
- K. Zuraiqi, A. Zavabeti, J. Clarke-Hannaford, B. J. Murdoch, K. Shah and M. J. Spencer, et al., Direct conversion of CO2 to solid carbon by Ga-based liquid metals, Energy Environ. Sci., 2022, 15(2), 595–600 RSC.
- M. Wolf, A. L. de Oliveira, N. Taccardi, S. Maisel, M. Heller and A. S. Khan, et al., Dry reforming of methane over gallium-based supported catalytically active liquid metal solutions, Commun. Chem., 2023, 6(1), 224 CrossRef CAS PubMed.
- M. Tamtaji, I. I. Goddard, Z. Hu, S. Chen and G. Chen, Dynamic single-atom catalysts on gallium to overcome the scaling relationship limit: AIMD screening for CO2 reduction and hydrogen evolution reactions, JACS Au, 2025, 5(9), 4459–4471 CrossRef CAS PubMed.
- Y. Wang, D. Chen, J. Zhang, M. S. Balogun, P. Wang and Y. Tong, et al., Charge relays via dual carbon-actions on nanostructured BiVO4 for high performance photoelectrochemical water splitting, Adv. Funct. Mater., 2022, 32(13), 2112738 CrossRef CAS.
- K. Ye, Y. Li, H. Yang, M. Li, Y. Huang and S. Zhang, et al., An ultrathin carbon layer activated CeO2 heterojunction nanorods for photocatalytic degradation of organic pollutants, Appl. Catal., B, 2019, 259, 118085 CrossRef CAS.
- Y. Hao, Y. Wang, L. Wang, Z. Ni, Z. Wang and R. Wang, et al., Probing layer number and stacking order of few-layer graphene by Raman spectroscopy, Small, 2010, 6(2), 195–200 CrossRef CAS PubMed.
- A. C. Ferrari, F. Bonaccorso, V. Fal'Ko, K. S. Novoselov, S. Roche and P. Bøggild, et al., Science and technology roadmap for graphene, related two-dimensional crystals, and hybrid systems, Nanoscale, 2015, 7(11), 4598–4810 RSC.
- V. Van Pham, T. Q. Nguyen, H. V. Le and T. M. Cao, Visible light photocatalytic NO x removal with suppressed poisonous NO 2 byproduct generation over simply synthesized triangular silver nanoparticles coupled with tin dioxide, Nanoscale Adv., 2024, 6(9), 2380–2389 RSC.
- Z. Ni, Y. Wang, T. Yu and Z. Shen, Raman spectroscopy and imaging of graphene, Nano Res., 2008, 1, 273–291 CrossRef CAS.
- T. Alves, W. S. Mota, C. Barros, D. Almeida, D. Komatsu and A. Zielinska, et al., Review of scientific literature and standard guidelines for the characterization of graphene-based materials, J. Mater. Sci., 2024, 59(32), 14948–14980 CrossRef CAS.
- J. X. Qian, T. W. Chen, L. R. Enakonda, D. B. Liu, J.-M. Basset and L. Zhou, Methane decomposition to pure hydrogen and carbon nano materials: State-of-the-art and future perspectives, Int. J. Hydrogen Energy, 2020, 45(32), 15721–15743 CrossRef CAS.
- C. Shyam Kumar, C. Possel, S. Dehm, V. S. K. Chakravadhanula, D. Wang and W. Wenzel, et al., Graphitizability of polymer thin films: an in situ TEM study of thickness effects on nanocrystalline graphene/glassy carbon formation, Macromol. Mater. Eng., 2024, 309(1), 2300230 CrossRef CAS.
- H. Calderon, A. Okonkwo, I. Estrada-Guel, V. Hadjiev, F. Alvarez-Ramírez and F. Robles Hernández, HRTEM low dose: the unfold of the morphed graphene, from amorphous carbon to morphed graphenes, Adv. Struct. Chem. Imaging, 2016, 2(1), 10 CrossRef PubMed.
- Y. Hernandez, V. Nicolosi, M. Lotya, F. M. Blighe, Z. Sun and S. De, et al., High-yield production of graphene by liquid-phase exfoliation of graphite, Nat. Nanotechnol., 2008, 3(9), 563–568 Search PubMed.
- Z. Zhang, H. Jin, C. Wu and J. Ji, Efficient production of high-quality few-layer graphene using a simple hydrodynamic-assisted exfoliation method, Nanoscale Res. Lett., 2018, 13, 1–8 Search PubMed.
- D. R. F. West, Ternary Phase Diagrams in Materials Science, CRC press, 2017 Search PubMed.
- C. J. Parker, V. Krishnamurthi, K. Zuraiqi, C. K. Nguyen, M. Irfan and F. Jabbar, et al., Synthesis of planet-like liquid metal nanodroplets with promising properties for catalysis, Adv. Funct. Mater., 2024, 34(31), 2304248 CrossRef CAS.
- F. M. Allioux, S. Nazari, M. B. Ghasemian, A. Zavabeti, Z. Pei and J. Leverett, et al., Atomic Dispersion via High-Entropy Liquid Metal Alloys, Small Struct., 2024, 2400294 CrossRef CAS.
- K. Zuraiqi, A. Zavabeti, F.-M. Allioux, J. Tang, C. K. Nguyen and P. Tafazolymotie, et al., Liquid metals in catalysis for energy applications, Joule, 2020, 4(11), 2290–2321 CrossRef CAS.
- P. H. Vaillant, V. Krishnamurthi, C. J. Parker, R. Kariuki, S. P. Russo and A. J. Christofferson, et al., Beneath the Skin: Nanostructure in the Sub-Oxide Region of Liquid Metal Nanodroplets, Adv. Funct. Mater., 2024, 34(31), 2310147 Search PubMed.
- T. R. Lear, S.-H. Hyun, J. W. Boley, E. L. White, D. H. Thompson and R. K. Kramer, Liquid metal particle popping: Macroscale to nanoscale, Extreme Mech. Lett., 2017, 13, 126–134 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |