 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mass transport behaviour of caesium and strontium in geopolymer cements
Charlotte
Nevin
a,
Daniel A.
Geddes
 a,
Clare L.
Thorpe
a,
Martin C.
Stennett
a,
Clare L.
Thorpe
a,
Martin C.
Stennett
 a,
Tom J.
Wilkinson
a,
Latham T.
Haigh
a,
Dinu
Iuga
b and
Brant
Walkley
a,
Tom J.
Wilkinson
a,
Latham T.
Haigh
a,
Dinu
Iuga
b and
Brant
Walkley
 *a
*a
aSchool of Chemical, Materials, and Biological Engineering, University of Sheffield, UK. E-mail: b.walkley@sheffield.ac.uk
bDepartment of Physics, University of Warwick, Coventry, UK
First published on 29th January 2026
Abstract
Geopolymers are a promising alternative to conventional Portland cement-based wasteforms for immobilising hazardous radioactive fission products such as caesium-137 and strontium-90, offering superior durability and lower leach rates. However, the specific mass transport mechanisms governing radionuclide release in geopolymers remain poorly understood, limiting implementation. This study reveals the incorporation and mass transport mechanisms of caesium (Cs) and strontium (Sr) in metakaolin-based geopolymers. Solid-state characterisation showed Sr incorporation via direct chemical binding in the alkali aluminosilicate hydrate gel in charge-balancing extra-framework sites, replacing K+ ions, and precipitation of SrCO3 and Sr(OH)2, while Cs is predominantly bound within the charge-balancing sites in the alkali aluminosilicate gel. Leach testing confirmed low overall release rates, with all measured leachability indices significantly exceeding the industry minimum of 6 (Li >13 for Cs; Li >18 for Sr), outperforming conventional PC systems. Mass transport modelling revealed distinct mechanisms: Cs release is accurately described by a Diffusion/Surface Exchange Kinetics Model (DSEM), yielding high correlation (R2 > 0.99), however, Sr exhibited a complex, staggered release profile. Standard mass transport models (diffusion, dissolution, surface exchange) could not satisfactorily capture this complex behaviour. We hypothesise this rate resumption is caused by the structural reordering or crystallisation of the amorphous K–A–S–H gel into a zeolitic phase, potentially excluding incorporated Sr. This finding highlights that simple diffusive models, commonly assumed for geopolymers, are inadequate for predicting the long-term performance of Sr-containing geopolymer wasteforms. The new insight presented here is critical development of geopolymers for radioactive waste disposal.
1 Introduction
Radioactive waste, generated from nuclear power and other applications, for example medical isotope production (such as cobalt-60 or technicium-99), poses significant environmental and societal hazards. Safe conditioning and disposal of this waste is therefore essential, particularly for legacy sites requiring remediation.1 Caesium-137 (Cs-137) and strontium-90 (Sr-90) are two particularly abundant radionuclides produced in the fission reaction of uranium-235 (U-235), and are prevalent in wastes streams from civil nuclear operations and from major nuclear accidents. For example, they are found together in waste streams such as complex sludges and slurries at the Sellafield nuclear site in the UK and contaminated groundwater at Fukushima Daichi Nuclear Power Plant in Japan. Their medium-term half-life (∼30 years), high radiotoxicity, water solubility, and biological mimicry2 make them particularly harmful to human health and the environment, and necessitate robust wasteforms to immobilise them for hundreds of years.Currently, in the UK, intermediate level waste (ILW) and low level waste (LLW), including those containing Cs-137 and Sr-90, are encapsulated in specifically formulated Portland cement (PC)/blast furnace slag (BFS) blends. These cement formulations have traditionally been used due to their simplicity, high throughput, low cost, and secure availability. However, PC/BFS systems exhibit limitations including high leaching rates and variable Cs retention depending on their C–S–A ratios.3,4 Furthermore, BFS supply chain vulnerabilities in the UK, due to the closure of blast furnaces in the steel sector, highlight the need for alternative disposal technologies.5
Geopolymer cements have been highlighted as a potential alternative material for the cementation of radioactive waste due to their superior physiochemical properties, their cation binding sites providing capability to immobilise certain cations (e.g. Cs+ and Sr2+), their lower leach rates, and their improved durability in extreme conditions, including high temperatures and acidic environments.5 Geopolymers comprise a structurally disordered, highly cross-linked alkali aluminosilicate gel (denoted (N,K)–A–S–H, as Na+ and K+ are the most common cations) that exhibits a pseudo-zeolitic structure, with the alkali cations in extra-framework sites balancing the negative charge arising from Al3+ ion tetrahedral coordination.6 If energetically favourable, the alkali cations may be substituted with other alkali or alkaline earth cations via ion exchange processes. This provides the ability to chemically bind and immobilise Cs+ and Sr2+ in the alkali aluminosilicate gel, in addition to physical encapsulation of the waste.1,7 The International Atomic Energy Agency (IAEA) standard leach tests assume diffusion as the dominating mass transport mechanisms governing the movement of Cs and Sr in geopolymers and other cements.8 However, the mass transport mechanisms in geopolymers have not been widely studied, especially regarding applications in radioactive waste disposal.
There have been numerous studies assessing leaching of different species from geopolymers (such as cadmium, copper, lead, zinc, chromium, cobalt, strontium, caesium, and other cations) under various conditions, but the mechanisms governing this leaching are rarely explored. Similar studies have been performed for Portland cement and blast furnace slag cements, such as early work done by Côtê9 and more recent studies by Abdel-Rahman10,11 and El-Kamash,12 which found that releases were governed by complex, multi-parametric mass transport. El-Kamash also studied mass transport in zeolites and Portland cement.13 These studies defined mass transport processes (diffusion, dissolution, and surface exchange/reaction) from first principles and fit these to experimental release data, collected from leaching experiments. Generally, it was found that diffusion and surface reactions dominated release in these studies.10 However, similar investigations for geopolymers are lacking.
This work aims to address this knowledge gap by investigating the mass transport mechanisms of Cs and Sr in metakaolin-based geopolymer cements using stable isotopes to represent Sr-90 and Cs-137. It quantifies and elucidates the release mechanisms, contributing to the development of safer and more durable radioactive waste disposal technologies.
2 Experimental methods
2.1 Sample preparation
Geopolymer cements were produced by reaction of a solid metakaolin powder (MetaMax, Lawrence Industries, with composition determined by X-ray fluorescence as shown in Table 1) and a potassium silicate solution (PQ-silicates, solution modulus SiO2/K2O = 1.0). Stoichiometry was designed to obtain a nominal chemical composition of K2O/Al2O3 = 1, SiO2/Al2O3 = 3, H2O/K2O = 13.1,14 Samples containing non-radioactive isotopes of Cs and Sr were prepared by mixing 1 or 3 wt% Sr(OH)2 or CsOH as a solid powder with the metakaolin prior to addition of the activating solution. Samples were subsequently mixed for 10 minutes at 1200 rpm using a high shear mixer to achieve homogeneity, cast in sealed cylindrical containers, and cured at 20 ± 2 °C for 28 days.| SiO2 | Al2O3 | K2O | Na2O | MgO | TiO2 | Fe2O3 | Other | LOI |
|---|---|---|---|---|---|---|---|---|
| 52.5 | 44.5 | 0.2 | 0.2 | < 0.05 | 1.3 | 0.4 | 0.2 | 0.6 |
After curing, samples were removed from sealed containers and formed into cylinders (13 mm length, 14 mm diameter) with the ends sealed with araldite to prevent axial mass transfer. Samples underwent a leach test according to a modified ASTM C1308 standard, wherein the monolithic samples were fully submerged in deionised water which was sampled and fully replaced at 2 hours, 7 hours, 24 hours, every day until 11 days, and then at 14 days, 21 days, 28 days, and 35 days. After the last time point, the monoliths were removed from solution and submerged in isopropanol to remove any bound water through ion exchange to prevent further reaction. The samples were then prepared for characterisation of the solid phase by hand grinding with a pestle and mortar until a talc-like consistency was reached. Samples for SEM or EPMA were left as solid monoliths and prepared as described in Section 2.2.
2.2 Characterisation of the solid phase
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 240 scans collected. Additionally, a nominal 1H decoupling field strength of 80 kHz was employed during acquisition.1
240 scans collected. Additionally, a nominal 1H decoupling field strength of 80 kHz was employed during acquisition.1
133Cs SS MAS NMR experiments were performed at the National High Field NMR facility at The University of Warwick, UK, to probe the local environment of the Cs nuclei. Spectra were acquired on a Bruker Avance NEO HFXY spectrometer with a MAS III spinning speed controller, using the 4 mm HX (low gamma) H13892 probe. 133Cs spectra were acquired at 20.0 T (B0) with a Larmour frequency of 111.5 MHz, using a rotor synchronised double frequency sweep echo pulse, a measured 0.1 s relaxation delay, spinning at 20.0 MHz, and a total of 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 scans.
000 scans.
High-field solid state 39K MAS NMR data were acquired at 20.0 T (ν0 = 39.67 MHz) using a Bruker Avance Neo 850 spectrometer with a Bruker 4.0 mm HX MAS probe, which enabled a spinning rate of 14 kHz to be implemented. Pulse calibration and chemical shift referencing for all 39K data were achieved using KCl(s) (δiso = 47.8 ppm) as a secondary reference to the IUPAC primary reference of 0.1 M KCl(aq) (δiso = 0 ppm). A ‘non-selective’ π/2 pulse of 12 µs was measured allowing for a ‘selective’ 4 µs π/3 to be implemented. Spectra were acquired using a Hahn echo pulse sequence (π/2–τ–π–acquire) using a measured relaxation delay of 0.1 s and acquiring a total of 480![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 transients per spectra.
000 transients per spectra.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 133 eV. The yttrium foil was measured periodically to ensure that there was no energy drift over the course of the experiment. Data reduction and analysis was performed using the Demeter software package, specifically Athena and Artemis.
133 eV. The yttrium foil was measured periodically to ensure that there was no energy drift over the course of the experiment. Data reduction and analysis was performed using the Demeter software package, specifically Athena and Artemis.
Cs K-edge (35.98 keV) XAS spectra were collected on the XMaS beamline at the European Synchrotron Radiation Facility (ESRF) in transmission, covering the 35.7–36.9 keV energy range.
2.3 Characterisation of the aqueous phase
 | (1) |
3 Mass transport models
In order to determine the mass transfer mechanisms leading to leaching of the Cs and Sr from geopolymers, models must be developed describing the main mass transport processes, including diffusion, dissolution, surface exchange kinetics, and combinations thereof. The mass transfer models are outlined subsequently, as developed from previous literature.9–11 Using the curve fit tool from the SciPy package in Python, an optimised fit is achieved between the experimental data from the leach tests and the models outlined subsequently. Statistical analysis is performed to understand the goodness of fit of each model and therefore to determine the best-fitting descriptive model of the CFL behaviour.3.1 Diffusive mass transport
It is commonly assumed that, after the initial period of surface wash-off, the leaching behaviour of certain radionuclides is controlled by diffusion.16 To model this, an assumption of diffusion through a semi-infinite cylinder (no axial diffusion due to the ends of the cylindrical monolit being sealed), and therefore the solution of Fick's law in a semi-infinite medium can be applied (eqn (2)) as follows: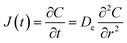 | (2) |
 | (3) |
3.2 Dissolutive mass transport
As the Cs and Sr can form a structurally significant part of the geopolymer due to the potential ion exchange with potassium, the release into solution of these radionuclides can cause a structural change to the matrix. Dissolution behaviour is shown in eqn (4), which assumes a release controlled by the dissolution constant. | (4) |
3.3 Surface exchange kinetics
Surface phenomena contribute to the mass transport of species from a cementitious or glass wasteform, described based on the kinetics of exchanges between the surface of a solid and the aqueous solution. The descriptive model for this is shown in eqn (5). | (5) |
3.4 Combined mass transport
 | (6) |
 | (7) |
 | (8) |
3.5 Leachability index (Li)
In stabilisation/solidification (S/S) of radioactive and other hazardous wastes, the leachability index (L(i)) is an indicative parameter for determining the effectiveness of a waste matrix. Li is the logarithm of the effective diffusivity of the radionuclide of interest and a waste form is considered to have acceptable leachability if Li is greater than 6. The leachability index can be calculated with eqn (9). | (9) |
3.6 Statistical analysis
 | (10) |
4 Results and discussion
4.1 Cumulative fraction leached
Geopolymer cements loaded with 1 and 3 wt% Sr(OH)2 or CsOH were prepared and leached as described, in deionised water held at 20 °C in a temperature-controlled environment chamber. At the discrete leaching time points, aliquots of the leachate were extracted and analysed by ICP-MS analysis to quantify the release of Cs and Sr from the waste matrix. The cumulative fraction leached (CFL) for both cations, shown in Fig. 1, represents the ratio of the released radionuclide concentration to its initial mass within the geopolymer matrix. All measurements were performed in triplicate, and the reported data represent the mean of these replicates. The error bars are small, especially for Cs, indicating high reproducibility and consistency due to adherence to standardised methodology.The pH rose to 11.00 during the first 24 hours, remained above 11 for the 48 hours time point and thereafter dropped a little remaining pH 10 ± 0.5 until the end of the 35 days experiment. In the bulk solution the maximum Cs and Sr concentrations measured were 30 ppm and 0.006 ppm respectively. Solution modelling in PHREEQC-2 confirmed that the bulk solution was undersaturated with regard to Sr and Cs bearing phases throughout the experiment (although this does not rule out supersaturation within the matrix pores or localised to the material surface). The solution was, however, intermittently supersaturated with regard to aluminium hydroxide and aluminium-silicate phases (see SI, Tables 3 and 4) and became increasingly supersaturated as the pH dropped below 10.5.
Overall, the Cs release profile exhibits a smooth increase, characterised by rapid initial release (<200 hours), followed by a plateau. Conversely, the Sr release profile exhibits a more complex staggered release pattern, with a period of release then stabilisation, followed by a subsequent rate resumption period at ∼200 hours and further stabilisation.
The observed CFL profiles reveal a larger release for Cs (total 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 100
000 and 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppb of Cs for the 1 and 3% CsOH samples, respectively) compared to Sr (total 23.5 and 36.1 ppb for the 1 and 3% Sr(OH)2 samples, respectively). This difference likely arises due to the differences in the solubility and incorporation of the cations. Sr may remain present as undissolved Sr(OH)2, partition to SrCO3, or occupy partially Sr-substituted zeolite sites.1,14,17 SrCO3 is highly stable and practically insoluble in water; therefore Sr release may be dependent on the precipitation and dissolution of this phase.
000 ppb of Cs for the 1 and 3% CsOH samples, respectively) compared to Sr (total 23.5 and 36.1 ppb for the 1 and 3% Sr(OH)2 samples, respectively). This difference likely arises due to the differences in the solubility and incorporation of the cations. Sr may remain present as undissolved Sr(OH)2, partition to SrCO3, or occupy partially Sr-substituted zeolite sites.1,14,17 SrCO3 is highly stable and practically insoluble in water; therefore Sr release may be dependent on the precipitation and dissolution of this phase.
Cs tends to occupy the zeolitic domain within the K–A–S–H gel17–19 but previous work has shown that there is selectivity for K+ over Cs+ into these sites.20 Therefore, when K occupies available exchangeable sites, Cs is excluded from these hosts and its retention becomes governed by diffusion or surface exchange, making it more readily leachable. The solid-state characterisation in this work has provided some evidence that Sr is present as discrete Sr-rich phases whilst Cs is evenly distributed through the K–A–S–H matrix and therefore it is likely this that is causing an increase in the leachability of Cs compared with Sr. There is evidence of SrCO3 forming in the higher loaded samples analysed by XAS analysis.
Notably, the Sr release (as a percentage of Sr added) from the lower loading samples (1%) is larger than that of the higher loading sample (3%) after ∼200 hours. In both systems the total amount of strontium released over the experiment is similar (approximately 30% higher in the higher 3% Sr system vs. the 1% Sr system instead of the 300% that might be expected). This observation is unexpected, as a higher loading would typically be expected to result in a higher CFL. However, it is possible that a Sr saturation limit is reached for SrCO3, meaning that a relatively higher proportion of the added Sr is bound in this poorly soluble mineral phase and a lower proportion incorporated into less soluble phases or sorbed to mineral surfaces that would be more easily released from the sample.
The staggered release behaviour of Sr mirrors trends observed in glass systems where crystallisation of secondary phases on the surface of the glass consumes silica from the protective amorphous gel layer and surrounding solution leading to a resumption of dissolution of the glass matrix.21–23 This phenomenon is usually observed in aluminosilicate glass compositions subjected to high pH and high temperatures that promote precipitation of zeolites.23,24 Given the structural similarities between the K–A–S–H gel and aluminosilicate glasses, it is possible that the leaching mechanisms may be somewhat analogous. Although solution replacement throughout the ASTM C1308 leach test is intended to simulate semi-dynamic leaching, the bulk solution was intermittently supersaturated with regard to Al and Si and, furthermore, the test is not agitated meaning that supersaturation and secondary phase formation likely occurred in pores and where localised concentration gradients occurred at the sample surface. It is also possible that crystallisation of the initially amorphous gel layer could cause a release of Sr that was previously incorporated into the K–A–S–H gel but that substitutes to a lesser extent into the newly formed mineral or zeolite phases.
This presents implications for waste disposal, as deviations from expected phases or from expected diffusive leaching behaviour could affect predictive modelling. Deviations from the long-term modelling may result in an uncontrolled, underestimated radionuclide release after repository closure.
Using the CFL, the effective diffusivity coefficient can be calculated from the slope of the straight line (m) of the plot of CFL versus using eqn (11).
using eqn (11).
 | (11) |
Using the relation above, the effective diffusion coefficient and associated Li (calculated from eqn (9)) were calculated and are displayed in Table 2.
| Sample | D e (cm2 s−1) | L i |
|---|---|---|
| Sr_1 | 2.2 × 10−19 ± 3.4 × 10−23 | 18.7 ± 0.00007 |
| Sr_3 | 7.8 × 10−20 ± 1.9 × 10−24 | 19.1 ± 0.00001 |
| Cs_1 | 5.4 × 10−14 ± 1.6 × 10−18 | 13.3 ± 0.00001 |
| Cs_3 | 6.0 × 10−14 ± 3.1 × 10−18 | 13.2 ± 0.00002 |
All Li values significantly exceed the industry minimum expected value of 6, even accounting for error on the calculation of De. Additionally, conventional PC systems typically exhibit Li ≈ 10 for Sr and Li ≈ 7.5 for Cs.25 Therefore, geopolymers show superior performance under these conditions when compared with PC.
4.2 Effect of leaching on the solid phase
Overall, the XRD diffractogram remains consistent after leaching. Therefore, it can be concluded that the leaching process did not significantly alter the bulk crystalline or amorphous phases within the geopolymers. However, as XRD was performed on a small volume of size reduced material, phases present as <1% of the total (e.g. surface precipitates) would not be detected by XRD.
The SEM images (Fig. 2) reveal a relatively homogeneous cross-sectional surface with a high number of pores, displaying no clear observable differences in the bulk sample before and after leaching or due to incorporation of Sr or Cs.
Low-resolution EDX mapping of the bulk of the samples (see SI, Fig. 3–5), indicated a homogeneous element distribution across all samples, with minimal elemental clustering. Post-leaching, an increased Si/Al ratio was observed indicated by the colouring of the maps, particularly in the 3% sample. This is consistent with the NMR findings (Section 4.2.4). Regions of high potassium content, likely due to residual potassium hydroxide or silicate in the pore solution, are present.
Due to oxygen interference with the elemental energy levels of Cs and Sr, EPMA was used for more accurate spatial distribution of elements and mass ratio quantification within the bulk geopolymer. The scans were taken of the samples loaded with 3% CsOH or Sr(OH)2. EPMA scans reveal a relatively homogeneous cross-sectional surface with some element clustering. In the bulk sample, silicon, aluminium, and potassium are evenly distributed, except in the pore regions where measurements were unable to be ascertained. Areas of high aluminium content, corresponding to low potassium content, suggest the presence of unreacted metakaolin which is characterised by a lower Si/Al ratio than a geopolymer. High potassium regions corroborated the EDX findings, indicating residual potassium hydroxide or silicate in the pores.
Sr-containing samples showed Sr distribution in the matrix of approximately 1%, with localised areas up to 40% Sr. These areas were depleted in silicon, aluminium, and potassium, therefore they are likely either undissolved Sr(OH)2 due to its low solubility or the formation of SrCO3 due to carbon present in the porewater. In contrast, Cs distribution was more uniform, likely due to the higher solubility of CsOH under the reaction mixture conditions. Overall, no significant visual differences were observed between the pre- and post-leach samples in the bulk phase.
Elemental quantification was performed on the bulk matrix and specific points of interest. Table 3 presents the average bulk matrix compositions and Table 4 details the point-of-interest compositions. The bulk matrix composition was determined by averaging elemental quantifications from discrete spots within the geopolymer, as shown in the image in Fig. 5.
| Species | Sr_3_Pre | Sr_3_Post | Cs_3_Pre | Cs_3_Post | ||||
|---|---|---|---|---|---|---|---|---|
| Mean (%) | SD | Mean (%) | SD | Mean (%) | SD | Mean (%) | SD | |
| Al | 15.96 | 0.67 | 12.54 | 0.91 | 17.50 | 0.64 | 19.85 | 1.91 |
| Si | 25.52 | 0.81 | 19.57 | 0.90 | 21.79 | 1.07 | 25.90 | 2.28 |
| K | 10.00 | 1.11 | 14.55 | 2.60 | 13.00 | 2.49 | 7.83 | 2.66 |
| O | 46.03 | 0.51 | 51.39 | 1.12 | 43.76 | 0.97 | 43.20 | 2.82 |
| Ti | 0.21 | 0.12 | 0.41 | 0.46 | 0.57 | 0.59 | 1.89 | 4.63 |
| Cs/Sr | 0.97 | 0.18 | 1.49 | 0.49 | 2.84 | 0.45 | 0.90 | 0.40 |
| Species | Point | Sr_3_Pre | Sr_3_Post | Cs_3_Pre | Cs_3_Post | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean (%) | SD | Mean (%) | SD | Mean (%) | SD | Mean (%) | SD | ||
| Al | 1 | 12.41 | 1.47 | 9.91 | 0.47 | 0.717 | — | 5.33 | 5.06 |
| 2 | 16.16 | 1.38 | 4.11 | 0.72 | 9.54 | — | 20.26 | 0.58 | |
| Si | 1 | 18.49 | 1.40 | 17.72 | 1.26 | 0.657 | — | 7.46 | 8.22 |
| 2 | 23.06 | 0.74 | 7.73 | 1.87 | 13.78 | — | 26.14 | 2.42 | |
| K | 1 | 7.64 | 3.19 | 6.035 | 2.67 | 0.993 | — | 2.45 | 2.39 |
| 2 | 14.87 | 6.01 | 2.66 | 9.64 | — | 7.55 | 3.33 | ||
| O | 1 | 38.34 | 0.35 | 38.84 | 0.59 | 39.93 | — | 60.15 | 4.25 |
| 2 | 44.11 | 1.32 | 35.42 | 0.64 | 41.61 | — | 44.27 | 6.57 | |
| Sr/Cs | 1 | 21.98 | 1.70 | 25.80 | 1.37 | 0.0 | — | 0.18 | 1.38 |
| 2 | 1.71 | 0.17 | 46.38 | 0.98 | 2.33 | — | 0.80 | 1.53 | |
In the bulk matrix, oxygen, silicon, aluminium, and potassium have the highest elemental concentrations, as expected for geopolymer gel. The Si/Al ratio is approximately 1.6, decreasing slightly after leaching (this is assessed more accurately in Section 4.2.4). Bulk Cs concentration is just below the expected 3% loading, decreasing significantly after leaching as a significant quantity is lost from the sample, as shown in Section 4.1. However, bulk Sr concentration is lower than expected, at 1% pre-leach, and are the same (within error) after leaching.
There EPMA scans reveal some heterogeneity, and there are some specific points of interest, for which elemental quantification was performed. These include ‘bright spots’, as shown in Fig. 5. The composition of these spots was determined and is shown in Table 4.
The high Sr concentration (up to 40%) of the bright particles (spot 1 on the pre-leach and both spots on the post-leach), alongside low silicon, aluminium, and potassium levels, indicate localised Sr(OH)2 or SrCO3 clusters (the presence of carbon could not be positively ascertained due to the carbon coating used on the samples). However, point 2 on the Sr pre-leach sample closely resembles the bulk composition, with a slightly elevated Sr concentration.
Conversely, for the Cs points of interest, the bright particles show minimal Cs and elevated titanium levels (up to 50%). Titanium, present in the metakaolin precursor, may form TiO2 during dissolution, although its concentration varied considerably as indicated by the high standard deviation. Point 2 on the pre-leach sample, also shows elevated titanium levels, also with an increase in Cs levels. This could be because the Cs sorbed onto the titanium-rich clusters. Finally, the bright region on the post-leach sample (point 2) is largely similar to the bulk matrix.
Overall, the imaging reveals no significant changes to the bulk matrix as a result of leaching. However, it is likely that any significant changes due to dissolution would be visible in approximately the first 10 microns from the outside of the sample. Low resolution scans are included in the SI and do not indicate any significant alteration at this magnitude, but more detailed scans could reveal insights into the potential formation of a leaching front or different phases.
Furthermore, the shoulders within this broad peak, at 1120 and 1160 cm−1, represent more asymmetric stretching of specifically T–O–Si1 and Si–O–Si,29 respectively due to the bonds present in the aluminosilicate gel. This indicates the presence of unreacted metakaolin in the gel. The peaks below 900 cm−1 further confirm geopolymer formation, including: symmetric stretching of Al–O–Si (560 cm−1),29 the formation of a potassium substituted zeolite A, K-LTA (673 cm−1),30 symmetric stretching of T–O–T,7 and the asymmetric stretching of T–O–T bonds linking AlO4 and SiO4 tetrahedral bonds,1 respectively. The small peak at 810 cm−1 which is present on some samples could further indicate the formation of zeolitic material within these samples.31 Finally, the peaks at 1300, 1370, 1470 cm−1 are consistent with the formation of carbonate ions of potassium/strontium/caesium.
A minor shift towards lower wavenumbers (by about 10 cm−1) is observed due to the incorporation of Sr and Cs. This suggests that these cations enhance the charge-balancing capacity of the aluminosilicate gel, resulting in a higher proportion of Si–O–Al bonds compared with Si–O–Si bonds. This shifts the spectra towards lower wavenumbers and indicates that the cations are influencing the gel structure and therefore may be replacing the charge-balancing potassium cations.
Post-leach samples exhibit almost identical FTIR spectra, indicating there has been little observable change in the microstructure of the geopolymer gel during leaching. The main observable difference is that the carbonate peaks (red box included to indicate area where these peaks occur) are more intense in the post-leach samples than in the pre-leach samples, likely due to prolonged atmospheric exposure as these samples were not handled in an inert atmosphere.
 | ||
| Fig. 6 29Si MAS (B0 = 11.7 T, νR = 12.5 kHz, shown in black) NMR and 1H–29Si CPMAS (B0 = 11.7 T, νR = 12.5 kHz and Hartmann–Hahn contact period t = 1.7 ms, shown in red) NMR spectra. | ||
To differentiate between the contributions from unreacted precursor (metakaolin) and the hydrated K–A–S–H gel, 1H–29Si cross polarisation MAS NMR spectra were collected which centred around δiso = −85 ppm before and after leaching with all cation incorporation levels. The 1H–29Si cross polarisation MAS NMR spectra span from δiso = −75 ppm to −95 ppm, all with a consistent lineshape. As the 1H–29Si cross polarisation MAS NMR signal is sensitive to internuclear distance between silicon atoms and nearby protons, the signal of silicon atoms near to protons is preferentially enhanced.1 Therefore, it is possible to differentiate between the hydrated gel sites and the silicon sites in the metakaolin which remained unreacted and present in the geopolymer.
The deconvolutions for each NMR fit for pre- and post-leach samples are shown in Fig. 7 and 8, respectively with 29Si MAS NMR on the left and 1H–29Si CP MAS NMR on the right for each sample. The deconvolution reveals five distinct silicon sites within the geopolymer sample at δiso values of −86 ppm, −90 ppm, 95 ppm, −102 ppm ± 4 ppm, and a broad resonance around −110 ppm.32 These correspond to Q4(4Al), Q4(3Al), Q4(2Al), and Q4(1Al) in the K–A–S–H gel and Q4(0Al) for the sites identified in the metakaolin precursor.32 The sites present in the hydrated K–A–S–H gel were identified using the 1H–29Si CP MAS spectra.32 These sites could then be quantified and compared with the unreacted metakaolin using the 29Si MAS spectra.
For most samples, the Q4(3Al) site is most abundant, followed by Q4(2Al), then Q4(4Al), with minor amounts of Q4(1Al) and unreacted metakaolin. Sr incorporation into the geopolymer increases Q4(3Al) sites and reduces Q4(4Al) sites, slightly increasing the Si/Al ratio.33 Conversely, the effect of incorporating Cs is less consistent: at 1 wt%, Q4(2Al) sites increase and dominate, but at 3 wt% Q4(2Al) decreases while Q4(4Al) increases, indicating unpredictable structural changes with more than one method of incorporation.
After leaching, the control sample shows a slight increase in Q4(3Al) sites and a decrease in Q4(4Al) sites, suggesting structural reordering into a less aluminium-rich gel, with a corresponding rise in the Si/Al ratio. This trend is also observed in the Cs samples at both 1 and 3 wt%. However, the Sr-containing samples exhibit the opposite trend, with an increase in Q4(4Al) sites and a decrease in Q4(3Al) sites (Table 5).
| Site | Q4(4Al) | Q4(3Al) | Q4(2Al) | Q4(1Al) | Si/Al |
|---|---|---|---|---|---|
| C_0_Pre | 23.9 | 38.6 | 32.1 | 5.4 | 1.42 |
| Sr_1_Pre | 18.2 | 44.3 | 31.8 | 5.7 | 1.45 |
| Sr_3_Pre | 17.3 | 47.4 | 33.1 | 2.3 | 1.43 |
| Cs_1_Pre | 21.3 | 36.1 | 37.7 | 4.9 | 1.46 |
| Cs_3_Pre | 26.5 | 38.1 | 29.9 | 5.4 | 1.40 |
| C_0_Post | 21.7 | 41.7 | 32.5 | 4.2 | 1.42 |
| Sr_1_Post | 32.1 | 32.1 | 32.1 | 3.8 | 1.37 |
| Sr_3_Post | 24.6 | 37.3 | 32.8 | 5.3 | 1.42 |
| Cs_1_Post | 19.2 | 46.0 | 30.5 | 4.2 | 1.43 |
| Cs_3_Post | 21.9 | 38.8 | 32.5 | 6.8 | 1.45 |
To assess the aluminium sites in the geopolymer matrix, 27Al MAS NMR data were acquired and are plotted in Fig. 9. The main resonances due to aluminium at δobs = 56, 33, and 8 ppm which are attributed to tetrahedral, pentahedral, and octahedral coordination.1 All samples display one large resonance between δobs = 70 and 50 ppm which is attributed to a AlO4 structure within the K–A–S–H gel, indicating Al in within a fully polymerised tetrahedral site. This is the expected coordination for aluminium to be in due to the excess of aluminium cations within the formulation of the cement.1
 | ||
| Fig. 9 27Al MAS NMR spectra (B0 = 11.7 T, νR = 12.5 kHz) for each geopolymer gel, pre- and post-leach, and for metakaolin. | ||
There is a very consistent lineshape with only a very small shift in the maxima (δobs = 60.0–60.5 ppm). In the pre-leach samples, the maxima are slightly higher for the Sr- containing samples than for the Cs or control samples (δobs ≈ 60.5 ppm and 60.0 ppm, respectively), likely due to slight shielding of the 27Al nucleus as a result of the divalent Sr2+ ion.1 In the post-leach samples, this shielding is not observed to the same degree with the maxima for Sr being in line with that of Cs. This indicates there is some structural reordering during the leaching process, especially in the Sr containing samples. The negligible amount of AlO6 and lack of AlO5 coordination of aluminium for all samples and the lack of any other variation in the 27Al MAS NMR spectra either after incorporation of cations or due to leaching indicates that there is little to no alteration in the local structure of the gel framework.
To further probe the environment of the Cs within the geopolymer gel, 133Cs MAS data were acquired at the national high field facility in Warwick, UK, and is shown in Fig. 10. The black lines indicate samples which were spun at 10 kHz, in an attempt to improve the signal/noise ratio but this resulted in interference from the spinning side bands. Therefore, for the post-leach sample the sample spun at 20 kHz in a smaller rotor is also included in red, with less interference from spinning side bands.
The pre-leach sample has a symmetrical line-shape with a sharp peak and a broad base and is indicative of a Cs ion in a structure similar to that seen in zeolite-A,18 with a chemical shift δobs = 42.7 (a line is included to guide the eye). This indicates that the Cs is bound into the charge-balancing sites of the aluminosilicate gel, in place of the K+ ions in the K–A–S–H gel. However, there is a significant shift towards lower δobs values after leaching, with a chemical shift δ = 35.6. This is more indicative of Cs in a zeolite-X structure,34 which has a different shape to zeolite-A and offers larger pore sizes.35 Furthermore, the peak becomes narrower after leaching, indicating that the Cs environment is more ordered or symmetric, possibly due to leaching of some of the loosely bound Cs causing some structural rearrangement. The 133Cs data for the sample spun at 20 kHz confirms that the Cs is present in only one site.
133Cs multiple-quantum magic angle spinning (MQMAS) data of the post-leach, 3% sample, is in Fig. 11. MQMAS is a powerful technique which can study half-integer quadrupolar nuclear (such as 133Cs which has  ).36 The two-dimensional plot allows resolution of overlapping sites by separating quadrupolar broadening effects.36 The spectrum shows a single, well-defined peak which confirms the presence of a single Cs site, or potentially two very similar overlapping Cs sites, with a relatively symmetrical local environment. This, combined with the insights from the 133Cs MAS NMR, confirms that Cs is likely only present in a zeolitic structure, as discussed in Section 4.1 and corroborated by the 133Cs spectrum for the sample spun at 20 kHz, and is not forming any significantly different sites.
).36 The two-dimensional plot allows resolution of overlapping sites by separating quadrupolar broadening effects.36 The spectrum shows a single, well-defined peak which confirms the presence of a single Cs site, or potentially two very similar overlapping Cs sites, with a relatively symmetrical local environment. This, combined with the insights from the 133Cs MAS NMR, confirms that Cs is likely only present in a zeolitic structure, as discussed in Section 4.1 and corroborated by the 133Cs spectrum for the sample spun at 20 kHz, and is not forming any significantly different sites.
High field 39 K MAS NMR data (Fig. 12) were obtained for the Cs_3 samples before and after leaching. The data exhibit a broad resonance spanning from δobs = 20 ppm to −300 ppm, centred at δobs = −53 ppm. This indicates charge-balancing extra-framework K+ ions within a (N,K)–A–S–H-type gel.33 The width and lineshape of the chemical shift distribution in the 39K MAS NMR data is very similar for the sample before and after leaching, indicating no significant changes to the local environment of K.
 | ||
| Fig. 13 (a) Normalised X-ray absorption spectra for the Sr-loaded geopolymer samples, brewsterite-Sr, and SrCO3. (b) First derivative of the spectra. | ||
The Sr K-edge is characterised by a smoothly rising absorption edge with a single peak at the maximum. A line is included to guide the eye. Although the absorption edge appears relatively featureless, when displayed in normalised intensity (Fig. 13b) the visibility of subtle features is enhanced. There is a slight shift in the peak of the brewsterite first derivative compared to the samples and the SrCO3 or Sr(OH)2. Close inspection reveals a doublet in the peak, the relative intensity of which changes between samples and reference standards. The brewsterite derivative spectrum reveals a doublet with two sharp features, the SrCO3 reveals a doublet with two broad peaks, and the Sr(OH)2 reveals a doublet with one shallow peak and one broad peak. The pre-leach samples have a shallow peak and then a sharp peak, with the post-leach revealing a slightly less intense first peak. One feature (feature A) on Fig. 13a shows a shoulder on the brewsterite-Sr and Sr(OH)2 which is not clearly visible on the SrCO3 or the sample spectra. This is clearer in the derivative plot, where there is a clear, broad peak indicated by the box. The final feature (feature B), present on all samples, is slightly sharper for the brewsterite-Sr.
Direct visual comparison of the samples with the standards can indicate the coordination of the Sr within the geopolymer matrix. The pre-leach spectra are visually similar to the local structure of Sr is in a zeolitic structure, such as brewsterite-Sr, as well as SrCO3. However, the direct visual comparison of those samples with the post-leach samples does reveal some slight changes in the spectra which could indicate a change in the Sr local structure, towards a mixture of zeolitic and SrCO3/Sr(OH)2-type local structures.
4.2.5.1 Extended X-ray absorption fine structure (EXAFS). EXAFS can determine the local coordination environment of the Sr in the geopolymer samples before and after leaching. Fig. 14a shows the k3-weighted EXAFS oscillations and Fig. 14b shows the magnitude of the Fourier transform (FT), which allows a direct, phase-shifted view of the radial structure. Also included are reference spectra for brewsterite, Sr(OH)2, and SrCO3 for comparison.
 | ||
| Fig. 14 (a) k2 weighted χ(k) and (b) Fourier transforms (FT) of k2-weighted χ(k) for the Sr-doped geopolymer and standard samples. The spectra have been stacked to improve clarity. | ||
Visual inspection of Fig. 14 reveal similarities between the spectra and those of brewsterite (a monoclinic zeolite with formula (Sr,Ba)2Al4Si12O32·10H2O), SrCO3, and Sr(OH)2. This suggests the Sr may occupy multiple local environments within the sample.
The amplitude reduction factor (S02) was determined by fitting FEFF-generated structural models of both brewsterite and SrCO3 to the experimental standard data. A value of S02 = 1 was satisfactory and was subsequently used for all sample refinements. The brewsterite model included single scatter paths for nearest-neighbour O, Si, and Al atoms. The SrCO3 model consisted of nearest-neighbour O shell (N = 9), and two further out cation shells (N = 6). Fig. 15 shows the fits to the structural models for the standards. Given the similarities observed between the samples and brewsterite, the Sr K-edge EXAFS data for the samples were modelled using the brewsterite model. Attempts to fit the samples using the SrCO3 and Sr(OH)2 models resulted in poor fits and were discarded.
For all samples, the following fitting approach was used:
• S02 was fixed at 1 based on the standard refinements.
• Path degeneracies (N) were fixed according to the refined brewsterite structure.
• All Debye–Waller factors (σ2) were restrained by element type.
• Multiple single-scatter O paths at similar distances were combined into an effective path.
• The Si/Al ratio was set to the ratio defined in the experiment, but due to CIF and FEFF restrictions of no mixed occupancy sites, this is not a perfect, realistic representation of the system.
Although the overall fits are good, it is acknowledged that the mismatch between the modelled and real Si/Al distribution is likely introducing uncertainty, leading to large uncertainties on the Al paths. This reflects static disorder from the random distribution of Al and Si on the same crystallographic sites, which cannot be fully modelled by FEFF.
The resulting fits are shown in Fig. 16 and the calculated factors in Table 6.
| Sample | R-factor | ΔE0 | ± | R Sr-i | ± | σ 2 | ± | N Sr-i | ± |
|---|---|---|---|---|---|---|---|---|---|
| Sr_1_Pre | 0.035 | −4.33 | 1.52 | — | — | — | — | — | — |
| Sr–O1 | — | — | — | 2.58 | 0.058 | 0.013 | 0.0011 | 9.0 | |
| Sr–Si | — | — | — | 3.73 | 0.088 | 0.024 | 0.013 | 4.5 | |
| Sr–Al | — | — | — | 4.09 | 0.012 | 0.0039 | 0.0063 | 1.5 | |
| Sr–O2 | — | — | — | 4.65 | 0.018 | 0.013 | 0.0011 | 8.0 | |
| Sr_1_Post | 0.035 | −4.18 | 1.59 | — | — | — | — | — | — |
| Sr–O1 | — | — | — | 2.57 | 0.16 | 0.013 | 0.0011 | 9.0 | |
| Sr–Si | — | — | — | 3.77 | 0.012 | 0.020 | 0.0083 | 4.0 | |
| Sr–Al | — | — | — | 4.09 | 0.0087 | 0.0098 | 0.0091 | 2.0 | |
| Sr–O2 | — | — | — | 4.63 | 0.00002 | 0.013 | 0.0011 | 8.0 | |
| Sr_3_Pre | 0.029 | −3.83 | 1.22 | — | — | — | — | — | — |
| Sr–O1 | — | — | — | 2.59 | 0.15 | 0.011 | 0.00090 | 9.0 | |
| Sr–Si | — | — | — | 3.78 | 0.14 | 0.020 | 0.0074 | 4.0 | |
| Sr–Al | — | — | — | 4.10 | 0.0017 | 0.0036 | 0.0034 | 2.0 | |
| Sr–O2 | — | — | — | 4.66 | 0.027 | 0.011 | 0.00090 | 8.0 | |
| Sr_3_Post | 0.030 | −3.70 | 1.31 | — | — | — | — | — | — |
| Sr–O1 | — | — | — | 2.58 | 0.16 | 0.013 | 0.0010 | 9.0 | |
| Sr–Si | — | — | — | 3.78 | 0.14 | 0.013 | 0.00059 | 3.0 | |
| Sr–Al | — | — | — | 4.09 | 0.0087 | 0.0098 | 0.0091 | 3.0 | |
| Sr–O2 | — | — | — | 4.67 | 0.041 | 0.013 | 0.0010 | 8.0 | |
| ⋮ |
For the Sr_1_Pre sample, the best fit was obtained by representing inner shell O paths as a single oxygen path at 2.58 Å (N = 9). A Sr–Si path with a scattering length of 3.73 Å and a Sr–Al path with a scattering length of 4.09 Å, adjusted to 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to reflect the realistic composition, were added. Finally, as with the inner shell, the outer shell oxygens best fit was obtained where all single scatter oxygen paths were represented by a single path at 4.65 Å, providing an overall satisfactory fit.
1 to reflect the realistic composition, were added. Finally, as with the inner shell, the outer shell oxygens best fit was obtained where all single scatter oxygen paths were represented by a single path at 4.65 Å, providing an overall satisfactory fit.
However, in Sr_1_Post, although the scattering lengths are very similar to the Sr_1_Pre sample, with inner shell O, Sr–Si, Sr–Al, and outer shell O scattering lengths of 2.57, 3.77, 4.09, and 4.63 Å, respectively, the Al/Si ratio adjustments were required for a satisfactory fit to be achieved. Overall, the Sr_1_Post sample showed a similar fit to Sr_1_Pre, with slight structural changes as the Sr–Si and Sr–Al contributions alter slightly.
Although still satisfactory, the fits for the samples with 3% loading were slightly inferior, indicated by the larger errors on the Sr–O and Sr–Si distances. Alternative inner shell oxygen paths were attempted, however the best fit was still achieved around the 2.58 Å distance, indicating that the local structure around the Sr absorber is similar to Sr_1_Pre. The larger errors, however, may indicate subtle differences in the long-range structure or larger contribution from other structures, such as Sr(OH)2 or SrCO3. For the Sr_3_Pre sample, the Sr–Si, Sr–Al, and outer shell O paths were 3.78, 4.10, and 4.66 Å, respectively. For the Sr_3_Post sample, the Sr–Si, Sr–Al, and outer shell O paths were 3.78, 4.09, and 4.67 Å, respectively. Both these fits are very similar to the 1% loaded samples, which minimal changes in the bond scattering lengths. However, again the Si/Al ratio had to be adjusted for a satisfactory fit.
Across all samples, the first-shell Sr–O distance (2.57–2.59 Å) and coordination number (N = 9) remains unchanged before and after leaching, indicating that the immediate Sr–O environment is preserved. However, small changes in the other shell contributions reveal:
• At low Sr loading (1%), the change in the Sr–Si and Sr–Al contributions are relatively minimal after leaching.
• At higher Sr loading (3%), NSr–Si decreases from 4 to 3 after leaching, while NSi–Al increases from 2 to 3. This suggests that the local environment around the Sr absorber may be altering slightly, potentially towards a carbonate or hydroxide environment or due to the partial crystallisation of the amorphous gel, as discussed in Section 4.1. However, this is not a significant enough change to cause a large interference with the brewsterite EXAFS signal.
Overall, the EXAFS analysis indicates that a brewsterite-based structural model adequately described Sr coordination, before and after leaching. At higher Sr loadings, slightly inferior model fits indicate partial Sr relocation or precipitation into secondary phases such as carbonate, hydroxide, or re-absorption into a crystalline zeolite site, however this is very minimal. The small structural changes are consistent with the low leaching rates of Sr and somewhat explain the distinct, ‘S-shaped’ curve.
Multi-phase EXAFS fits were attempted, combining brewsterite and SrCO3 models, but these could not be successfully fit. However, the single-model fits using brewsterite, combined with the other solid-state characterisation techniques, provide confidence in the precipitation of some level of Sr to SrCO3 at high waste loadings and the potential crystallisation of the amorphous gel.
4.2.5.2 Cs K-edge X-ray absorption near edge fine structure (XANES). XANES can be utilised to infer the local coordination of Cs within geopolymer samples before and after leaching. Fig. 17 shows the Cs–K edge XANES spectra for geopolymer samples with comparative standards.
 | ||
| Fig. 17 (a) Normalised X-ray absorption spectra of Cs-loaded geopolymer samples, Cs2CO3, CsNO3, Cs-rhodozite, and Cs-pollucite. (b) First derivative of the spectra. | ||
Visual inspection of XANES and derivative plots showed significant similarity between loaded geopolymers before and after leaching experiments with minimal changes in post-edge features, particularly the features at 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 eV and 36
000 eV and 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 025 eV which are present in all loaded geopolymer samples. Intensity and positions of both post-edge features remain consistent between pre and post leaching samples, indicating negligible change in the immediate coordination environment of the Cs.
025 eV which are present in all loaded geopolymer samples. Intensity and positions of both post-edge features remain consistent between pre and post leaching samples, indicating negligible change in the immediate coordination environment of the Cs.
Comparison with standards of Cs2CO3, and CsNO3 show similar features to loaded geopolymers with oscillations also at 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 eV and 36
000 eV and 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 025 eV with no discernible oscillations thereafter.
025 eV with no discernible oscillations thereafter.
The lack of further post-edge oscillations in geopolymer samples is indicative of low-crystallinity or amorphous coordination environments around the Cs absorber, with Cs2CO3 and CsNO3 standards also showing low intensity oscillations after the absorption edge, likely as a result of deliquescence leading to poor crystallinity. This contrasts with the more crystalline Rhodozite and Pollucite samples which display more intense oscillations beyond the absorption edge.
4.3 Leaching characteristics
 function. Goodness of fit was evaluated using R2 values. Fig. 18 illustrates the fits, with corresponding parameters and R2 values summarised in Table 7. Optimal model fits are characterised by the R2 approaching one.
function. Goodness of fit was evaluated using R2 values. Fig. 18 illustrates the fits, with corresponding parameters and R2 values summarised in Table 7. Optimal model fits are characterised by the R2 approaching one.
 in Python
in Python
| Model | Variable | R 2 | |
|---|---|---|---|
| Sr_1 | DIF | D = 1.88 × 10−17 ± 2.1 × 10−18 | 0.872 |
| DIS | U = 2.28 × 10−10 ± 1.3 × 10−11 | 0.893 | |
| SEK | C = 3.91 × 10−6 ± 7.0 × 10−7 | 0.938 | |
| k = 0.00156 ± 0.00042 | |||
| Sr_3 | DIF | D = 9.70 × 10−18 ± 5.0 × 10−19 | 0.950 |
| DIS | U = 1.53 × 10−10 ± 1.5 × 10−11 | 0.594 | |
| SEK | C = 1.61 × 10−6 ± 1.2 × 10−7 | 0.912 | |
| k = 0.00433 ± 0.00068 | |||
| Cs_1 | DIF | D = 7.18 × 10−11 ± 9.9 × 10−12 | 0.467 |
| DIS | U = 3.86 × 10−7 ± 6.4 × 10−8 | 0.467 | |
| SEK | C = 0.00307 ± 7.0 × 10−5 | 0.956 | |
| k = 0.0207 ± 0.0023 | |||
| Cs_3 | DIF | D = 9.92 × 10−11 ± 1.5 × 10−11 | 0.270 |
| DIS | U = 4.49 × 10−7 ± 7.8 × 10−8 | −0.981 | |
| SEK | C = 0.00354 ± 7.3 × 10−5 | 0.953 | |
| k = 0.0270 ± 0.0032 |
The models represent diffusion (DIF), dissolution (DIS), and surface-exchange kinetics (SEK), or reaction. The diffusion model assumes that the Sr or Cs is initially distributed evenly throughout the sample and then diffuses through the pores. The dissolution model assumes that the Sr or Cs is a major structural component of the geopolymer matrix and therefore its release causes a breakdown in the structure. Finally, surface exchange kinetics, or reaction, models the release of the Sr or Cs which is readily soluble at the surface of the geopolymer. This is also sometimes known as surface wash-off.10
For Sr at the lower loading concentrations (1%), diffusion (DIF), dissolution (DIS), and surface exchange kinetics (SEK) models all demonstrated reasonable fits to the experimental CFL data (R2 > 0.85). SEK yields the highest R2 value, indicating that surface exchange kinetics primarily govern the release. The non-negligible contributions from DIF and DIS also indicate more complex changes at the material surface. As described previously, it is hypothesised that the recrystallisation of an initially amorphous secondary alteration phase (e.g. K–A–S–H gel to zeolite) led to a resumption in the release of Sr2+ into solution after 200 hours.
At higher Sr waste loadings (3%), DIF and SEK models exhibited strong correlation with the experimental data (R2 > 0.90). However, DIS demonstrated poor agreement (R2 = 0.59), indicating less contribution from dissolutive processes. This observation suggests that higher Sr loadings favour diffusion-controlled release, as more loosely bound Sr is available, relative to the more structural Sr. This may be because, at higher waste loadings, there is a greater proportion of SrCO3 or Sr(OH)2 compared with structurally bound, zeolitic Sr, allowing it to diffuse more readily rather than relying on dissolution or reactive processes for release.
Cs release was primarily governed by the SEK model (R2 > 0.95), while DIF and DIS show very poor correlation with the experimental data. The Cs experimental release exhibits a rapid initial wash-off phase, followed by a plateau after ∼200 hours, suggesting there are two distinct release phases. The DIF model under-predicts the initial release and over-predicts the later release, whereas the DIS model fails to describe the release behaviour at any stage. The SEK model accurately predicts the first 300 hours, but under-predicts thereafter.
Overall, the observed contributions from multiple release mechanisms suggests that the release of Sr and Cs from the studied geopolymer wasteform involves a complex interplay of interacting processes.
 function, an algorithm designed to calculate the global maximum which is particularly effective for models with multiple parameters, compared to
function, an algorithm designed to calculate the global maximum which is particularly effective for models with multiple parameters, compared to  . Goodness of fit was evaluated using R2 values with the results and calculated model parameters summarised in Table 8.
. Goodness of fit was evaluated using R2 values with the results and calculated model parameters summarised in Table 8.
| Sample | Model | Parameters | R 2 |
|---|---|---|---|
| Sr_1 | DSEM | D = 0.0 ± 7.8 × 10−18C = 3.90 × 10−6 ± 1.1 × 10−6k = 0.00156 ± 0.062 | 0.937 |
| DDM | D = 0.0 ± 5.5 × 10−18U = 2.28 × 10−10 ± 3.2 × 10−11 | 0.860 | |
| DISEM | U = 1.90 × 10−10 ± 2.7 × 10−7C = 3.02 × 10−7 ± 2.2 × 10−7k = 0.483 ± 0.36 | 0.928 | |
| Sr_3 | DSEM | D = 0.0 ± 0.0 C = 1.61 × 10−6 ± 1.1 × 10−7k = 0.00434 ± 0.000876 | 0.900 |
| DDM | D = 0.0 ± 0.0 U = 1.53 × 10−10 ± 2.6 × 10−11 | 0.318 | |
| DISEM | U = 8.69 × 10−11 ± 1.3 × 10−11C = 5.31 × 10−7 ± 1.1 × 10−7k = 0.0872 ± 0.30 | 0.930 | |
| Cs_1 | DSEM | D = 5.05 × 10−12 ± 1.5 × 10−12C = 0.00223 ± 9.25 × 10−5k = 0.0331 ± 0.0032 | 0.996 |
| DDM | D = 7.22 × 10−11 ± 1.3 × 10−11U = 1.51 × 10−10 ± 7.08 × 10−11 | 0.379 | |
| DISEM | U = 6.05 × 10−8 ± 2.3 × 10−8C = 0.00265 ± 0.000254 k = 0.0304 ± 0.29 | 0.992 | |
| Cs_3 | DSEM | D = 5.26 × 10−12 ± 2.22 × 10−12C = 0.00272 ± 0.000124 k = 0.0417 ± 0.0054 | 0.994 |
| DDM | D = 9.88 × 10−11 ± 2.10 × 10−11U = 3.94 × 10−12 ± 8.0 × 10−11 | 0.134 | |
| DISEM | U = 6.18 × 10−8 ± 2.7 × 10−8C = 0.00314 ± 0.00026 k = 0.0378 ± 0.25 | 0.988 |
For the Cs-containing samples, the DSEM and DISEM models accurately predict the release behaviour across the entire dataset. In conjunction with observations from the single-source models, these results suggest that surface exchange kinetics are the dominant release mechanism for Cs release, with diffusion contributing at later time points.
For Sr, the superimposed models provide a significantly improved representation of the experimental data compared to the single-source models, with most fits achieving high R2 values, with the exception of the DDM model for the 3% loaded sample. This observation confirms the complex, multi-parametric nature of Sr mass transport, which cannot be accurately described by a single release mechanism.
Despite achieving high R2 values, the combined models were still unable to accurately predict the S-shaped release profile observed in the experimental data. This could be attributed to a ‘rate resumption’ effect analogous to glass systems, as described in Section 4.1. Specifically, due to the crystallisation of the K–A–S–H gel to a zeolitic structure, evidenced by the presence of zeolitic phases in both NMR and XAS, could influence the Sr mobility. Potentially, the Sr is well incorporated into the amorphous gel but only partially within the crystalline zeolite, causing the Sr to be ‘kicked out’ of the structure as precipitation occurs.
The rate resumption period is more pronounced in the 1% samples, further indicating that diffusion dominates throughout the entire leaching process for the 3% sample. A possible explanation is that, at 1% loading, a greater proportion of Sr is bound into the amorphous gel, whereas at 3% loading, potential saturation of Sr into more stable SrCO3 which remains unaffected by the crystallisation of the gel into zeolite.
Another plausible explanation is the presence of two distinct mass transport phases: an initial phase dominated by diffusion of loosely bound ions, followed by a period dominated by structural effects. To accurately model the two distinct mass transport phases, the experimental data was truncated into time points before 200 hours and after 200 hours, and then fitted to both single-source (Fig. 20) and combined source (Fig. 21) models. The corresponding R2 values are presented in Table 9.
| Sample | Model | R 2 < 200 h | R 2 > 200 h |
|---|---|---|---|
| Sr_1 | DIF | 0.630 | 0.901 |
| DIS | −0.198 | 0.630 | |
| SEK | 0.777 | 0.0 | |
| DSEM | 0.774 | 0.936 | |
| DDM | −0.762 | 0.561 | |
| DISEM | 0.966 | 0.936 | |
| Sr_3 | DIF | 0.821 | 0.802 |
| DIS | 0.202 | −0.966 | |
| SEK | 0.886 | 0.0 | |
| DSEM | 0.882 | 0.942 | |
| DDM | −0.137 | −1.45 | |
| DISEM | 0.994 | 0.949 |
Visual inspection of the model fits revealed no substantial improvement when the data was segmented. Across both time intervals, the DSEM and DISEM models consistently show the most agreement with the experimental release data, yielding high R2 values.
The lack of improvement in fit with the truncated data indicates that it is not two distinct phases. Therefore, it is likely there is another phenomenon causing the distinct curve of the plot, which isn't caused by dissolution, surface exchange kinetics, or dissolution, conforming that this complex mass transport cannot and should not be predicted by these models alone.
| Residuals = observed value − predicited value | (12) |
 | ||
| Fig. 19 Combined source mass transfer models for (top) Sr and (bottom) Cs. Experimental data is plotted as blue dots, models are plotted as lines. | ||
 | ||
| Fig. 20 Single-source mass transport model fits for Sr at 1% (top) and 3% (bottom) waste loading. The plots are split into prior to 200 hours and after 200 hours. | ||
 | ||
| Fig. 21 Combined-source mass transport model fits for Sr at 1% (top) and 3% (bottom) waste loading. The plots are split into prior to 200 hours and after 200 hours. | ||
For the single-source mechanisms, all residual plots exhibited non-random patterns, deviating from scatter around the zero line (Fig. 22). For the 1% Sr loaded samples, a consistent trend was observed across all models: under-prediction at early time points, over-prediction in the mid-range, and under-prediction at later time points. The models exhibited slight variation in the final stages, with DIF under-predicting, DIS over-predicting, and SEK providing an accurate estimation. This discrepancy arises from the inverted, ‘S’ shape of the curve, which the models are unable to fully capture. A similar trend was observed for the 3% Sr loaded samples, although the DIF model over-predicted at the latter time points. Despite the non-random residuals, the magnitude of the residuals is consistently low, indicating little deviation between the observe values and the model predictions.
The residual plots for the Cs-containing samples showed distinct patterns between the models. The DIF and DIS models exhibited under- and over-prediction trends, expected due to their poor overall fits. However, the SEK model exhibits relatively well-distributed residuals, with a slight over- and under-prediction pattern. These are small in magnitude, indicating a reasonable fit. The residuals plots for the combined source models exhibit similar patterns for both Cs and Sr samples (Fig. 23). These consistent trends suggest that the models are not fully capturing the complex release behaviour, and therefore there may be additional mass transport contributing to the release.
5 Conclusions
This study investigated the performance and leaching mechanisms of Cs- and Sr-loaded geopolymers. Leaching tests revealed that Cs exhibits higher leachability than Sr, with distinct, different release patterns: Sr displays a staggered release, while Cs showed a smooth, consistent release. However, all samples out-performed PC/BFS blends under comparable conditions and significantly exceeded the industry minimum accepted value.Structural analysis across XRD, SEM-EDX, EPMA, FTIR, and NMR revealed minimal impact of Cs and Sr incorporation or leaching on the geopolymer bulk structure. While element-specific NMR and XAS showed subtle changes in the immediate environment of the Cs and Sr after leaching, these did not significantly impact the bulk structure over the 35-day time-frame of the leach test and release rates were lower than for Portland cement wasteforms.
Finally, mass transport modelling did not produce a satisfactory fit to Sr data due to processes occurring not accounted for in the models. It is hypothesised that K–A–S–H gel reordering to form crystalline zeolite may account for the sudden increase in Sr release rates observed in the data. This presents implications for modelling the long-term performance of Sr in geopolymers destined for disposal as its long-term performance cannot be modelled using these processes. Nevertheless, even accounting for this rate resumption, Sr release rates remain very low. Further work will aim to understand the behaviour over longer timescales. In contrast, Cs release could be fit with a mixed model combining surface exchange kinetics and diffusion, with negligible contribution from dissolution.
To conclude, the geopolymer wasteforms, when leached in deionised water at 20 °C, are considered safe within the parameters of this study. Future work will explore more realistic disposal scenarios, such as increased temperature, radiation damage, and the use of clinoptilolite (ion exchange material) in disposal. Longer term experiment could explore the hypothesised mechanisms responsible for Sr's staggered release and whether this behaviour persists over longer time frames.
Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting this article have been included as part of the main manuscript.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta08377j.
Acknowledgements
This work has been funded by the Engineering and Physical Sciences Research Council (EPSRC), UK, through EPSRC PhD studentship 2602279, part of the EPSRC CDT GREEN (EP/S022295/1), with co-funding from Sellafield Ltd., and grant EP/Y029208/1. The UK High-Field Solid-State NMR Facility used in this work was funded by EPSRC and BBSRC (grant EP/T015063/1), as well as the University of Warwick, including via part funding through Birmingham Science City Advanced Materials Projects 1 and 2 supported by Advantage West Midlands (AWM), and the European Regional Development Fund (ERDF). We acknowledge Diamond Light Source for time on Beamline B18 under Proposal SP37458-1, and thank Dr Thokozile Kathyola, Beamline Scientist, B18, Diamond Light Source, for assistance with acquiring the XAS data. We thank and acknowledge Dr Khalid Doudin, School of Maths, Physics, and Chemistry, The University of Sheffield, for assistance in acquiring some of NMR data obtained at The University of Sheffield, Dr Sarah Pepper, Royce Discovery Centre, The University of Sheffield, for assistance in acquiring some of the ICP data, and Dr Stuart Creasey-Gray, Sorby Centre for Electron Microscopy, The University of Sheffield, for assistance in acquiring some of the EPMA data.References
- B. Walkley, X. Ke, O. H. Hussein, S. A. Bernal and J. L. Provis, Incorporation of strontium and calcium in geopolymer gels, J. Hazard. Mater., 2020, 382, 121015, DOI:10.1016/j.jhazmat.2019.121015.
- A. M. James, S. Harding, T. Robshaw, N. Bramall, M. D. Ogden and R. Dawson, Selective environmental remediation of strontium and cesium using sulfonated hyper-cross-linked polymers (shcps), ACS Appl. Mater. Interfaces, 2019, 11(25), 464–473, DOI:10.1021/acsami.9b06295.
- E. Duque-Redondo, K. Yamada and H. Manzano, Cs retention and diffusion in c-s-h at different ca/si ratio, Cem. Concr. Res., 2021, 140, 106–294, DOI:10.1016/j.cemconres.2020.106294.
- S. Bagosi and L. J. Csetényi, Caesium immobilisation in hydrated calcium-silicate-aluminate systems, Cem. Concr. Res., 1998, 28(12), 1753–1759, DOI:10.1016/S0008-8846(98)00163-X.
- S. Kearney, A. S. Yorkshire, D. A. Geddes, et al., “Chapter 25 – cement-based stabilization/solidification of radioactive waste”, in Low Carbon Stabilization and Solidification of Hazardous Wastes, ed. D. C. W. Tsang and L. Wang, Elsevier, 2022, pp. 407–431, DOI:10.1016/B978-0-12-824004-5.00005-0.
- J. Provis and J. Van Deventer, Alkali Activated Materials: State-Of-The-Art Report, RILEM TC 224-AAM, Springer, 2014, DOI:10.1007/978-94-007-7672-2.
- Q. Tian, S. Nakama and K. Sasaki, Immobilization of cesium in fly ash-silica fume based geopolymers with different si/al molar ratios, Sci. Total Environ., 2019, 687, 1127–1137, DOI:10.1016/j.scitotenv.2019.06.095.
- A. N. Society, “Ansi/ans-16.1-2003: Measurement of the Leachability of Solidified Low-Level Radioactive Wastes by a Short-Term Test Procedure”, American Nuclear Society, Tech. Rep., 2003, 2003 Search PubMed.
- P. L. Côtê, T. W. Constable and A. Moreira, An evaluation of cement-based waste forms using the results of approximately two years of dynamic leaching, Nucl. Chem. Waste Manage., 1987, 7(2), 129–139, DOI:10.1016/0191-815X(87)90007-6.
- R. O. Abdel Rahman and A. A. Zaki, Comparative study of leaching conceptual models: Cs leaching from different ilw cement based matrices, Chem. Eng. J., 2011, 173(3), 722–736, DOI:10.1016/j.cej.2011.08.038.
- A. Abdel Rahman and R. O. Zaki, Assessment of the leaching characteristics of incineration ashes in cement matrix, Chem. Eng. J., 2009, 155, 698–708, DOI:10.1016/j.cej.2009.09.002.
- A. M. El-Kamash, A. M. El-Dakroury and H. F. Aly, Leaching kinetics of 137cs and 60co radionuclides fixed in cement and cement-based materials, Cem. Concr. Res., 2002, 32(11), 1797–1803, DOI:10.1016/S0008-8846(02)00868-2.
- A. M. El-Kamash, M. R. El-Naggar and M. I. El-Dessouky, Immobilization of cesium and strontium radionuclides in zeolite-cement blends, J. Hazard. Mater., 2006, 136(2), 310–316, DOI:10.1016/j.jhazmat.2005.12.020.
- K. T. O'Donoghue, D. A. Geddes, M. Hayes and B. Walkley, Reaction mechanisms and kinetics in geopolymers incorporating strontium salts, Dalton Trans., 2025, 54(29), 337–348, 10.1039/D5DT01087J.
- R. Snellings, J. Chwast and Ö. Cizer, et al., Rilem tc-238 scm recommendation on hydration stoppage by solvent exchange for the study of hydrate assemblages, Mater. Struct., 2018, 51(6), 172, DOI:10.1617/s11527-018-1298-5.
- E. Hespe, Leach testing of immobilized radioactive waste solids a proposal for a standard method, Atom. Energy Rev., 1971, 9(1), 195–207 Search PubMed.
- E. Mukiza, Q. T. Phung, L. Frederickx, D. Jacques, S. Seetharam and G. De Schutter, Co-immobilization of cesium and strontium containing waste by metakaolin-based geopolymer: Microstructure, mineralogy and mechanical properties, J. Nucl. Mater., 2023, 585, 154–639, DOI:10.1016/j.jnucmat.2023.154639.
- M. Arbel-Haddad, Y. Harnik, Y. Schlosser and A. Goldbourt, Cesium immobilization in metakaolin-based geopolymers elucidated by 133cs solid state nmr spectroscopy, J. Nucl. Mater., 2022, 562, 153–570, DOI:10.1016/j.jnucmat.2022.153570.
- M. Arbel Haddad, E. Ofer-Rozovsky and G. Bar-Nes, et al., Formation of zeolites in metakaolin-based geopolymers and their potential application for cs immobilization, J. Nucl. Mater., 2017, 493, 168–179, DOI:10.1016/j.jnucmat.2017.05.046.
- T. Skorina, Ion exchange in amorphous alkali-activated aluminosilicates: Potassium based geopolymers, Appl. Clay Sci., 2014, 87, 205–211, DOI:10.1016/j.clay.2013.11.003.
- R. G. Newton, Durability of glass - a review, Glass Technol., 1985, 26(1), 21–38 CAS.
- F. Frizon, S. Gin and C. Jegou, “Mass transfer phenomena in nuclear waste packages”, in Advances in Transport Phenomena: 2009, ed. L. Wang, Springer Berlin Heidelberg, Berlin, Heidelberg, 2009, pp. 31–133, DOI:10.1007/978-3-642-02690-4_2.
- P. Frugier, S. Gin and Y. Minet, et al., Son68 nuclear glass dissolution kinetics: Current state of knowledge and basis of the new graal model, J. Nucl. Mater., 2008, 380(1), 8–21, DOI:10.1016/j.jnucmat.2008.06.044.
- A. Ait Chaou, A. Abdelouas, Y. El Mendili and C. Martin, The role of ph in the vapor hydration at 175 °c of the french son68 glass, Appl. Geochem., 2017, 76, 22–35, DOI:10.1016/j.apgeochem.2016.11.006.
- J.-Y. Goo, B.-J. Kim, M. Kang, J. Jeong, H. Y. Jo and J.-S. Kwon, Leaching behavior of cesium, strontium, cobalt, and europium from immobilized cement matrix, Appl. Sci., 2021, 11(18), 8418 CrossRef CAS.
- V. A. Baki, Enhancing the reactivity of natural minerals as supplementary cementitious materials for low-carbon binders, PhD thesis, University of Bath, 2023.
- K. Lee and J. Kim, Immobilization of 137cs as a crystalline pollucite surrounded by amorphous aluminosilicate, Environ. Res., 2023, 221, 115–309, DOI:10.1016/j.envres.2023.115309.
- A. Hajimohammadi, J. L. Provis and J. S. J. van Deventer, The effect of silica availability on the mechanism of geopolymerisation, Cem. Concr. Res., 2011, 41(3), 210–216, DOI:10.1016/j.cemconres.2011.02.001.
- W. K. W. Lee and J. S. J. van Deventer, Use of infrared spectroscopy to study geopolymerization of heterogeneous amorphous aluminosilicates, Langmuir, 2003, 19(21), 8726–8734, DOI:10.1021/la026127e.
- E. Ofer-Rozovsky, M. A. Haddad and G. Bar-Nes, et al., Cesium immobilization in nitrate-bearing metakaolin-based geopolymers, J. Nucl. Mater., 2019, 514, 247–254, DOI:10.1016/j.jnucmat.2018.11.003.
- A. Mekki, A. Benmaati, A. Mokhtar, M. Hachemaoui, F. Zaoui, H. Habib Zahmani and B. Boukoussa, et al., Michael addition of 1,3-dicarbonyl derivatives in the presence of zeolite y as an heterogeneous catalyst, J. Inorg. Organomet. Polym. Mater., 2020, 30(7), 2323–2334, DOI:10.1007/s10904-019-01424-5.
- B. Walkley and J. L. Provis, Solid-state nuclear magnetic resonance spectroscopy of cements, Mater. Today Adv., 2019, 1, 100007, DOI:10.1016/j.mtadv.2019.100007.
- D. A. Geddes, M. C. Stennett, T. J. Wilkinson, B. E. Griffith, J. V. Hanna, J. L. Provis and B. Walkley, et al., Alkali-mediated sr incorporation mechanism and binding capacity of alkali aluminosilicate hydrate in geopolymers, J. Hazard. Mater., 2025, 488, 137426, DOI:10.1016/j.jhazmat.2025.137426.
- F. Yagi, N. Kanuka, H. Tsuji, S. Nakata, H. Kita and H. Hattori, 133cs and 23na mas nmr studies of zeolite x containing cesium, Microporous Mater., 1997, 9(5), 229–235, DOI:10.1016/S0927-6513(96)00113-7.
- A. Materials, Type a and x zeolites, Web Page, n.d .
- A. Medek, J. S. Harwood and L. Frydman, Multiple-quantum magic-angle spinning nmr: A new method for the study of quadrupolar nuclei in solids, J. Am. Chem. Soc., 1995, 117(51), 779–787, DOI:10.1021/ja00156a015.
| This journal is © The Royal Society of Chemistry 2026 |















