In situ ammonia synthesis and energy generation via aqueous Zn–NO3− batteries
Akansha
Chaturvedi
,
Shivangi
Mehta
and
Tharamani C.
Nagaiah
 *
*
Department of Chemistry, Indian Institute of Technology Ropar, Rupnagar, Punjab-140001, India. E-mail: tharamani@iitrpr.ac.in
First published on 7th January 2026
Abstract
The sustainable generation of electricity and ammonia (NH3) is essential for modern industrial advancement. The electrochemical nitrate reduction reaction (NO3RR) provides a sustainable pathway for NH3 production, complemented by pollution mitigation. Nevertheless, this process is constrained by limited nitrate adsorption, multiple competing reactions, and sluggish kinetics involving coupled proton–electron transfer steps, thereby reducing NH3 selectivity. Herein, we have utilised FeCu(1:2)Sx as a cathode catalyst in Zn–NO3− battery to produce NH3via the NO3RR, simultaneously generating electricity. The assembled Zn–NO3− battery demonstrated a remarkable faradaic efficiency (F.E.) of 98.96% for NH3 and a power density of 5.2 mW cm−2. Real time NH3 production examined by in situ electrochemical Raman and attenuated total reflectance-Fourier transform infrared (ATR FT-IR) spectroscopy revealed that Cu promotes NO3− adsorption and reduction to NO2−, while Fe facilitates hydrogenation resulting in an NH3 yield of 3.69 mg h−1 cm−2 at −0.9 V vs. RHE. As a proof of concept, two Zn–NO3− batteries connected in series powered 62 LEDs for over 70 h.
Introduction
The rising global energy demands are increasingly outpacing the limited availability of fossil fuel resources, thereby intensifying the need for advancements and innovations in energy storage technologies.1,2 Among emerging systems, the zinc-based battery has garnered substantial interest as a promising next-generation energy storage system, owing to its high theoretical energy density and exceptional current output capabilities.3,4 The economic feasibility of these batteries is further enhanced by the widespread availability and low cost of zinc (Zn), making them an appealing option for sustainable energy solutions.5,6 However, in such batteries, the electrocatalytic oxygen evolution and reduction reactions (OER and ORR) occur at the cathode during charging and discharging, respectively and are significantly impeded by the intricate gas–solid–liquid triple-phase interface and sluggish kinetics; moreover, these processes yield no commercially valuable product.7 Thus, replacing air with nitrate (NO3−) in these batteries could potentially provide higher output voltage than O2-based batteries.6,7 Moreover, the Zn–NO3− battery is considered environmentally advantageous due to its ability to generate electricity while converting NO3− from wastewater into value-added NH3.5,6,8 Thus compared to the well-established Zn–air (oxygen) battery, the Zn–NO3− battery offers a ‘one stone-three birds’ strategy: pollutant remediation with NH3 production, energy storage & conversion.7 Moreover, the produced NH3 is the second most widely synthesised chemical in the world, with an annual demand exceeding 200 million metric tonnes, with its crucial role as a necessary component in the production of fertilisers and other advantageous chemical compounds.8–10 Despite these advantages, the Zn–NO3− battery faces significant challenges in meeting the performance criteria required to power modern electrical appliances. The primary limitation in the NO3RR arises from its complex eight-electron transfer mechanism, which is hindered by slow kinetics and the substantial energy barrier associated with breaking N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds (204 kJ mol−1).11–14 Furthermore, the reaction often leads to the generation of undesirable by-products, including NO2−, N2H4, and NH2OH, thereby diminishing both selectivity and overall efficiency.12,15
O bonds (204 kJ mol−1).11–14 Furthermore, the reaction often leads to the generation of undesirable by-products, including NO2−, N2H4, and NH2OH, thereby diminishing both selectivity and overall efficiency.12,15
Another significant challenge lies in the co-existence of desorbed and non-desorbed intermediates, which complicates the favourable adsorption of specific intermediates, such as NO2−, due to the limitations imposed by scaling relationships.16–18 These factors collectively underscore the difficulty and necessity of designing an efficient electrocatalyst capable of promoting selective adsorption and conversion of certain intermediates such as NO2−.
Transition metal-based catalysts, owing to their earth abundance and cost-effectiveness, have become a feasible choice for the NO3RR to NH3, particularly due to their superior adsorption strength to NO3− through N and O atoms.19–21 Among these, copper-based catalysts have gained immense attention due to the similar energies of Cu d-orbital and π* (LUMO) molecular orbital of NO3−.22–25 While this compatibility facilitates strong NO3− adsorption and promotes its initial conversion, however, it concurrently results in the accumulation of NO2− by-products. The limited ability of Cu to further cleave the N–O bond in NO2− stems from its weak adsorption of NO2− intermediates. Moreover, the electroreduction of NO3−, governed by complex proton-coupled electron transfer (PCET) reactions, is further constrained by Cu's poor catalytic activity for hydrogen atom evolution, thereby impeding efficient PCET progression. As a result, monometallic Cu systems often fail to achieve high NH4+ selectivity, likely due to suboptimal hydrogenation steps. Inspired by the catalytic activity of iron (Fe) in nitrate reductase enzymes in plants, Fe has been employed in electrochemical NH3 synthesis.26–28 Its favourable binding energies with N-containing intermediates make it particularly effective.12,29–31 Additionally, Fe exhibits sufficient catalytic properties for hydrogen evolution, thereby enhancing the stepwise PCET reactions involved in the NO3RR.
Thus, a bimetallic Fe and Cu-based catalyst is predicted to enhance electrochemical NO3− reduction activity towards NH3 production. Inspired by the aforementioned consideration in mind, we synthesised FeCu(a:b)Sx as a catalyst for the NO3RR. The optimised catalyst FeCu(1:2)Sx gave a high NH3 F.E. of 96.5% at a potential of −0.6 V vs. RHE. Furthermore, in situ Raman spectra and ATR FT-IR analyses showed formation of NO2− and NH3 during the NO3RR. To evaluate the influence of rational catalyst design on catalytic activity, we assessed the performance of monometallic control catalysts. The FeCu(1:2)Sx catalyst was further utilised as a cathode material in the Zn–NO3− battery with a Zn plate as the anode (Scheme 1). The Zn–NO3− battery assembled with FeCu(1:2)Sx as the cathode demonstrated a power density of 5.2 mW cm−2 and high F.E. of 98.96% for NH3 production. This research offers profound insights into the design of efficient electrocatalysts for the NO3RR and Zn–NO3− battery.
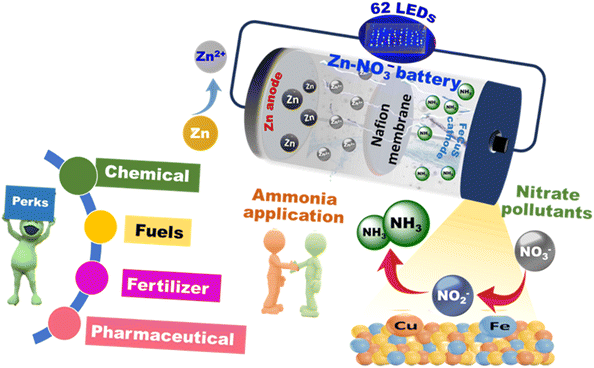 | ||
| Scheme 1 Schematic representation of the assembled Zn–NO3− battery with catalyst design and ammonia application. | ||
Results and discussion
Electrochemical nitrate reduction (NO3RR)
The potential applicability of the designed FeCu(1:2)Sx catalyst towards the NO3RR was assessed in 0.5 M K2SO4 electrolyte using 0.1 M KNO3 as the nitrate source. Initially, linear sweep voltammetry (LSV) experiments were performed in Ar-saturated 0.5 M K2SO4 electrolyte containing 0.1 M KNO3. As seen from Fig. 1a, a rapid increase in the reduction current density was observed attributed to the NO3RR. In the absence of 0.1 M KNO3, a similar behaviour was observed, but at a more reductive potential. These observations indicate the electrocatalytic activity of the designed catalyst towards the NO3RR (inset, Fig. 1a and S1).To understand the role of each element of the designed catalyst towards NO3RR, control experiments were performed using the monometallic catalyst viz. CuSx and FeS2. The comparative LSV curves in Fig. 1a displayed an onset potential of −0.47 V vs. RHE (@10 mA cm−2) for FeCu(1:2)Sx with a maximum current density (cd) of 224 mA cm−2 @ −1.0 V vs. RHE. Conversely, CuSx and FeS2, exhibited a cd of 90 mA cm−2 and 112 mA cm−2, respectively, at @ −1.0 V vs. RHE and with the onset potentials of −0.52 V and −0.51 vs. RHE (@10 mA cm−2), signifying a superior activity of the FeCu(1:2)Sx catalyst towards the NO3RR. To gain a deeper understanding on the product formation during the NO3RR, chronoamperometric (CA) measurements were conducted at a fixed potential of −0.6 V vs. RHE for 1 h, and the obtained products were quantified using colorimetric methods. Notably, FeCu(1:2)Sx demonstrated a significantly enhanced NH3 yield rate of 1542 µg h−1 cm−2, surpassing those of monometallic FeS2 (746 µg h−1 cm−2) and CuSx (566 µg h−1 cm−2) (Fig. 1b and S1–S4). In contrast, CuSx achieved the highest NO2− yield rate of 1149 µg h−1 cm−2, with a F.E. of 13.08%, which was superior to that of FeS2 (686 µg h−1 cm−2, 6.83%) and FeCu(1:2)Sx (518 µg h−1 cm−2, 2.99%) (Fig. 1b and S2).
To provide more compelling evidence for the synthesis of NH3 from NO3− and the role of each metal, potential dependent in situ electrochemical ATR-FTIR spectroscopic analysis was performed for CuSx and FeCu(1:2)Sx catalysts. The FTIR spectra of FeCu(1:2)Sx displayed peaks at ∼3851, ∼3752 and ∼1106 cm−1 corresponding to intermediates NH, NH2, and NH3, respectively (Fig. 1c and d).32 As the sample was polarized to a more negative potential the intensity of these bands increases with the increase in the potential, which highlights the progression of NH3 production. Amid the finding, a pair of characteristic peaks at ∼1548 cm−1 and 1360 cm−1 represent the asymmetric and symmetric vibration modes of the absorbed NO2− intermediates.33 These observations unequivocally underscore the remarkable potential of FeCu(1:2)Sx catalysts in facilitating the hydrogenation of NO3− to NH3via the NO2− intermediate. Conversely, for CuSx ATR-FT-IR measurements exhibit peaks at ∼1106 cm−1 and ∼1360 cm−1, corresponding to NH3 and adsorbed NO2−. However, the minimal presence of hydrogenation intermediates (*NH2 and *NH) indicates the limited efficacy of CuSx for hydrogenation to NH3. In the case of FeS2, ATR-FT-IR measurements display a pronounced peak at ∼1106 cm−1 attributed to NH3, accompanied by a negligible hump at ∼1360 cm−1 for NO2− (Fig. S5). This observation clearly indicates that FeS2 facilitates more complete hydrogenation and suppresses intermediate accumulation compared to CuSx. Additional peaks at ∼1646 cm−1 and ∼3371 cm−1 are assigned to water.
To gain deeper insight into the NO3RR, Raman spectra were recorded over a range of potentials (−0.4 V to −0.9 V vs. RHE) and at open-circuit potential (OCP) (Fig. 1e). As anticipated, at OCP, two distinct peaks were observed at ∼1043 cm−1 and ∼979 cm−1 corresponding to the presence of NO3− and SO42−, respectively.34,35 The emergence of a peak at ∼1439 cm−1, attributed to the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of the bridged nitro group, confirmed the formation of NO2− during the NO3RR.36 Additional peaks at ∼1315 cm−1 and ∼1586 cm−1 were associated with the symmetric and antisymmetric bending vibrations of HNH of NH3, respectively, and exhibited increasing intensity with a more negative applied potential.34,37 The pronounced NH3 peak intensity, accompanied by a diminished NO2− hump, signifies the catalyst's selective facilitation of NH3 formation on its surface. Based on in situ Raman spectroscopic analysis, it was conclusively established that NO3RR intermediates, including NO2−, and NHN, predominantly contribute to the electrochemical production of NO2− and NH3 on the catalyst surface.
O stretching vibration of the bridged nitro group, confirmed the formation of NO2− during the NO3RR.36 Additional peaks at ∼1315 cm−1 and ∼1586 cm−1 were associated with the symmetric and antisymmetric bending vibrations of HNH of NH3, respectively, and exhibited increasing intensity with a more negative applied potential.34,37 The pronounced NH3 peak intensity, accompanied by a diminished NO2− hump, signifies the catalyst's selective facilitation of NH3 formation on its surface. Based on in situ Raman spectroscopic analysis, it was conclusively established that NO3RR intermediates, including NO2−, and NHN, predominantly contribute to the electrochemical production of NO2− and NH3 on the catalyst surface.
The electrochemical activity of FeCu(1:2)Sx was further tested at various potentials to quantify NO3RR products (Fig. S6). Then the product was evaluated by UV-Vis spectral analysis via an Indophenol blue method (Fig. S6). With an increase in the negative potential during the NO3RR, an increasing trend in NH3 yield rate was observed, indicating higher NH3 synthesis at larger negative potential,6 and the highest NH3 yield rate of 3.69 mg h−1 cm−2 was attained at −0.9 V vs. RHE (Fig. S6). Furthermore, the F.E. of NH3 was studied at various potentials, revealing a F.E. of 96.5% and NH3 yield rate of 1.54 mg h−1 cm−2 (Fig. 1d) at −0.6 V vs. RHE with a turn over frequency (TOF) of 0.154 h−1, suggesting its efficient performance in carrying out the NO3RR in a neutral medium. This prompted further experiments with other variants to discern the influence of the Cu and Fe ratio in FeCu(a:b)Sx catalysts. The activity was found to be in the order FeCu(1:2)Sx > FeCu(1:1)Sx > FeCu(2:1)Sx (Fig. S7). Furthermore, to compare the NH3 production rates and efficiency of other bimetallic variants with CuFe(1:2)Sx, the CA experiments were performed at a potential of −0.6 V vs. RHE under similar experimental conditions (Fig. S5b). Amongst these, FeCu(1:2)Sx showed the highest NH3 yield rate and F.E., indicating its superior NO3RR activity (Fig. S7d). In order to substantiate the synthesis of NH3 from NO3−, time-dependent in situ ATR FTIR analyses were performed on FeCu(1:2)Sx (Fig. 2a). These exhibited peaks indicating intermediates NH, NH2, and NH3 for ammonia (Fig. 2a).32 The progressive increase in the intensity of these bands over time underscores the advancement of NH3 production. Among these, a pair of characteristic peaks comprises vibration modes of the absorbed NO2− intermediates.33 Further analysis identifies peaks at ∼1646 cm−1 and ∼3371 cm−1, which originate from the O–H stretching and bending modes of water, signifying its role as a proton source for subsequent hydrogenation reactions.32,33 These observations clearly highlight the significant potential of FeCu(1:2)Sx catalysts in promoting the hydrogenation of NO3− to NH3via the NO2− intermediate.
 | ||
| Fig. 2 (a) In situ time-dependent ATR FT-IR spectra of the FeCu(1:2)Sx catalyst with the zoomed parts in i and ii. (b) Schematic illustration for the reaction mechanism. | ||
In light of our comprehensive in situ analytical observations, coupled with corroborative electrochemical data and pertinent literature, it can be conclusively inferred that Cu is highly effective in promoting NO3− adsorption and its reduction to NO2−, whereas Fe plays a pivotal role in facilitating the hydrogenation to NH3.11,38 The electrochemical reduction of NO3− to NH3, governed by the reaction NO3− + 6H2O + 8e− → NH3 + 9OH−, proceeds through a well-defined mechanistic pathway (Fig. 2b).11,38 This pathway is initially characterized by a sequence of deoxygenation reactions (*NO3− → *NO2 → *NO → *N), followed by a series of hydrogenation steps (*N → *NH → *NH2 → *NH3). Such mechanistic elucidation highlights the intricate interplay of deoxygenation and hydrogenation processes pivotal to the NO3− reduction pathway.
The structural features of the synthesised variants of FeCu(a:b)Sx along with its monometallic variant FeS2 and CuSx were analysed via powder X-ray diffraction (P-XRD). Fig. 3a demonstrates the P-XRD pattern of FeCu(1:2)Sx, FeS2 and CuSx. The P-XRD pattern of the synthesised FeS2 displays diffraction peaks in agreement with JCPDS No. 42-1340. Additionally, the P-XRD pattern of CuSx shows a combination of peaks attributed to CuS (JCPDS No. 06-0464) and Cu2S (JCPDS No. 53-0522), with the most prominent peak at 31.785° assigned to the (103) plane of CuS, and a major peak at 46.105° corresponding to the (220) plane of Cu2S. The P-XRD pattern of the synthesised FeCu(a:b)Sx catalysts indicates the coexistence of FeS2, CuS, Cu2S and CuFeS2 in all the variants, consistent with their respective JCPDS standards (Fig. 3a and S8).
The field emission scanning electron microscopy (FE-SEM) images of FeCu(1:2)Sx reveal microflower-shaped morphology (Fig. 3b and c). Energy dispersive X-ray spectroscopy (EDS) elemental dot mapping also confirms the coexistence and uniform distribution of Cu, Fe, and S within the microflowers (Fig. S8). The high-resolution transmission electron microscopy (HR-TEM) images of the FeCu(1:2)Sx catalyst show distinct lattice fringes with measured interplanar spacings of 0.303 nm, 0.189 nm, and 0.163 nm, which can be indexed to the (112) plane of CuFeS2, the (110) plane of CuS, and the (311) plane of FeS2, respectively (Fig. S9a and b). The corresponding selected area electron diffraction (SAED) patterns further corroborate these assignments, displaying diffraction spots consistent with the planes of CuFeS2, CuS and FeS2, thereby confirming the structural coherence between HR-TEM and SAED observations (Fig. S9c).
The survey XPS (X-ray photoelectron spectroscopy) spectra of FeCu(1:2)Sx indicate the presence of Fe, Cu, and S (Fig. S10a). Fe 2p XPS spectra unveil binding energies of Fe 2p3/2 (710.9 eV) and Fe 2p1/2 (724.5 eV), attributed to Fe3+ in CuFeS2.39 Additionally, two peaks at 707.7 eV and 720.7 eV correspond to the Fe 2p3/2 and Fe 2p1/2 binding energies of Fe2+ in FeS2, respectively (Fig. 3d).39,40 The deconvoluted Cu 2p XPS spectra shown in Fig. 3e revealed BE peaks at 932.0 eV and 951.9 eV corresponding to 2p3/2 and 2p1/2, respectively, corresponding to Cu+. Furthermore, peaks at 932.9 eV and 954.5 eV corresponded to 2p3/2 and 2p1/2 of Cu2+.41,42 Thus, we can conclude that Fe is present in the +2 and +3 oxidation state and Cu in both the Cu(I) and Cu(II) oxidation state in the FeCu(1:2)Sx catalyst. Moreover, the deconvoluted S 2p XPS spectrum shows peaks at 161.0 eV and 162.0 eV, corresponding to S 2p3/2 and S 2p1/2 bonded with metals. Additionally, peaks at 163.5 eV and 168.5 eV indicate the presence of divalent sulfides and sulfates, respectively (Fig. S10b).43 Furthermore, the XPS spectra of FeCu(1:2)Sx exhibits a decrease of ∼0.2 eV in the Cu 2p binding energy and a concomitant positive shift of ∼0.3 eV in the Fe 2p binding energy relative to their respective monometallic counterparts. These shifts provide strong evidence for effective Cu–Fe electronic coupling within FeCu(1:2)Sx (Fig. S11a and b). Comparative XPS analysis thus highlights a pronounced chemical interaction between Cu and Fe, consistent with electron transfer from Fe to Cu atoms within FeCu(1:2)Sx.6,44,45 To further clarify the oxidation state of Cu, Auger emission (AE) spectra reveal a distinct Auger Cu LMM peak corresponding to Cu2+ and Cu1+, located at 568.5 eV and 569.8 eV, respectively (Fig. S11c).46,47 Notably, no signal is observed at 568 eV, confirming the absence of Cu0.47 The Raman spectrum exhibits a distinct peak at ∼470 cm−1, which can be attributed to the Cu–S vibrational modes characteristic of the chalcopyrite CuFeS2 phase (Fig. S12).48,49 The powder X-ray diffraction (P-XRD) patterns of FeCu(a:b)Sx catalysts with varying Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu ratios indeed show noteable differences. Specifically, when the Fe
Cu ratios indeed show noteable differences. Specifically, when the Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu ratio is increased from 1
Cu ratio is increased from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, the diffraction peaks corresponding to CuS at 2θ = 32.85° and 47.94° display enhanced intensity, indicating enrichment of copper-containing phases. Conversely, when the Fe
2, the diffraction peaks corresponding to CuS at 2θ = 32.85° and 47.94° display enhanced intensity, indicating enrichment of copper-containing phases. Conversely, when the Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu ratio is adjusted from 1
Cu ratio is adjusted from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 2
1 to 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the characteristic CuS peak at 2θ = 47.94° diminishes, reflecting a relative decline in copper content. Thus the structural features are governed by the dominant elemental composition (Fig. S13). Fig. S14 depicts the FE-SEM images and EDS dot mapping for the FeCu(1:1)Sx and FeCu(2:1)Sx catalysts.
1, the characteristic CuS peak at 2θ = 47.94° diminishes, reflecting a relative decline in copper content. Thus the structural features are governed by the dominant elemental composition (Fig. S13). Fig. S14 depicts the FE-SEM images and EDS dot mapping for the FeCu(1:1)Sx and FeCu(2:1)Sx catalysts.
The superior activity of FeCu(1:2)Sx could be due to the faster kinetics at the electrode–electrolyte interface, which was evidenced by the lower Tafel slopes compared to the other catalyst variants (Fig. S15 and Table S1). This was further supported by electrochemical impedance spectroscopy (EIS) analysis, where in Fig. S16, a lower charge transfer resistance (Rct) was observed for the FeCu(1:2)Sx catalyst (Table S2), signifying fast charge transfer kinetics at the electrode–electrolyte interface.8 The FeCu(1:2)Sx catalyst shows a higher conductivity of 5.32 mS cm−1 compared to its monometallic variants (Table S2). The facile kinetics could also be attributed to the increased number of accessible electrochemically active sites on the catalyst surface which was further evaluated using double-layer capacitance in the non-faradaic region. The electrochemical surface area (ECSA) of the FeCu(1:2)Sx catalyst was found to be 329 cm2 which is superior to that of other variants (Fig. S17 and Table S1). Thus, larger ECSA of FeCu(1:2)Sx justified its superior activity towards the NO3RR. The energy efficiency was found to be 28.47% at a potential of −0.4 V vs. RHE (details in the SI).
Furthermore, the validation of NH3 generated and reliability of the data for the same have emerged as critical and significant criteria. As a result, a strict methodology described in the literature was followed, including cleaning of cell components (Scheme S1).50 Also, the control experiments were performed by conducting CA measurement in Ar-saturated 0.5 M K2SO4 (in the absence of NO3−) at a potential of −0.6 V vs. RHE, on a bare electrode in the presence of NO3−. The UV-Vis spectra obtained for the electrolyte for NH3 production, indicated negligible NH3 production, thereby endorsing that the source of N is exclusively from the added NO3− and no existence of potential interferences (Fig. 4a). Furthermore, to confirm that NH3 generated in our experiment comes from the NO3RR only, isotope labelling experiments were performed using 14NO3− and 15NO3− as the N source. For the quantitative analysis of 15NH4+/14NH4+ acquired from the isotope labelling studies, the calibration curves were extracted for different known concentrations of 15NH4+/14NH4+ using 1H NMR spectroscopy (Fig. S18–S20). After electrolysis, 1H NMR spectra of 14NO3− and 15NO3− solutions showed a triplet and doublet with coupling constants of 52 Hz and ∼72 Hz, respectively, and no doublet or triplet was observed in the absence of NO3−, indicating the reliability of produced data (Fig. 4b and S20). Fig. 4c demonstrates the NH3 yields for both 15NH4+ and 14NH4+ obtained using the Indophenol blue method and 1H NMR spectra showing negligible change in the NH3 yield. NH3 yield was further quantified spectrophotometrically using Nessler's reagent, yielding 1.53 mg h−1 cm−2 at −0.6 V vs. RHE, validating the results from the Indophenol blue method (Fig. S21). Apart from NH3, other possible reduction products produced during the NO3RR such as NO2−, H2, N2H4, and NH2OH, were quantified, after the CA test at −0.6 V vs. RHE for 1 h. The by-products NO2−, N2H4 and NH2OH were tested and quantified by UV-Vis spectroscopy as illustrated in Fig. S22 and S23 (detailed in the SI). It is worth noting that N2H4 and NH2OH were not detected (Fig. S16 and S17); however, NO2− was detected with a yield rate of 1.29 mg h−1 cm−2 and F.E. of 2.99%. Thus, the relatively high selectivity for NH3 synthesis by the FeCu(1:2)Sx catalyst with a total F.E. for the NO3RR at −0.6 V vs. RHE was found to be 99.49%, with F.E. for NH3, and NO2− to be 96.5% and 2.99% respectively (Fig. S24).
Along with the electrochemical activity, stability is an important parameter for evaluating the performance of the catalyst. The electrochemical stability of FeCu(1:2)Sx towards the NO3RR was then investigated using consecutive electrolysis for 10 cycles at a potential of −0.6 V vs. RHE (Fig. S25). The CA curves revealed a negligible change in the current response and its corresponding UV-Vis spectra for 10 consecutive cycles. A steady F.E. and NH3 yield was maintained throughout the duration of 10 h of the stability test pointing towards reproducibility of the catalyst's performance (Fig. S25c). The changes in the catalyst after the stability test were also investigated by P-XRD, FE-SEM, EDS and XPS techniques. The XRD pattern showed that crystalline structures are well preserved (Fig. S26a) and, post XPS analysis (Fig. S26b–d) showed no change in the chemical composition of elements present in FeCu(1:2)Sx, endorsing the high stability of the catalyst for the NO3RR. Furthermore, the FE-SEM image (Fig. S26e), showed that morphology is well maintained. Also, EDS spectra and dot mapping images showed the presence of all the elements in the catalyst even after the stability test (Fig. S26f–i). Table S3 shows the atomic % of the elements present in FeCu(1:2)Sx, demonstrating almost negligible change in the composition. The aforementioned results show appreciable activity towards the NO3RR in neutral media in comparison to reported literature (Table S4).
Zn–NO3− battery
Motivated by the superior performance of FeCu(1:2)Sx towards the NO3RR, a Zn–NO3− battery was assembled to demonstrate that the battery could be used for producing electric current and NH3.The assembled Zn–NO3− battery comprise FeCu(1:2)Sx coated over Ni foam as the cathode in Ar saturated 0.5 M K2SO4 solution containing 0.1 M KNO3 and a polished Zn plate was employed as the anode in 1 M KOH containing 0.02 M Zn(CH3COO)2. During the discharge process, the anodic Zn gets oxidised, followed by spontaneous production of zincate anions which upon reaching saturation convert to ZnO (eqn (2)). At the cathode, NO3− reduction takes place assisted by electrons released by Zn metal oxidation (eqn (3))6,51
Anode:
| 4Zn + 8OH− → 4ZnO + 4H2O + 8e− | (1) |
Cathode:
| NO3− + 7H2O + 8e− → NH4OH + 9OH− | (2) |
Overall reaction
| 4Zn + NO3− + 3H2O → 4ZnO + NH4OH + OH− | (3) |
The assembled battery showed an open circuit voltage (OCV) of 1.32 V vs. Zn/Zn2+ (calculations detailed in the SI) and it remained stable for more than 20 h (Fig. 5a). Furthermore, as shown in the discharge polarisation curve (Fig. 5b), a maximum current density of 30 mA cm−2 was obtained with a maximum power density of 5.2 mW cm−2 @ a current density of 16 mA cm−2 in the assembled battery, which is comparable to that in reported literature in neutral media (Table S5). Moreover, the sequential chronopotentiometric measurements at various discharge current densities ranging from −0.1 mA cm−2 to −2.5 mA cm−2 (Fig. 5c) were recorded and a stable response was obtained with negligible change in the potential over a time scale of 1 h indicating fast mass transfer and conductivity.6 Furthermore, the production and quantification of NH3 during battery discharging was analysed by recording chronopotentiometry measurements at different current densities ranging from −2 to −10 mA cm−2 (Fig. S27a) and then quantified by UV-Vis spectroscopy (Fig. S27b). Interestingly, the highest F.E. of 98.96% was obtained at a current density of −4 mA cm−2 with an NH3 yield of 158 µg h−1 cm−2 and a maximum NH3 yield of 257 µg h−1 cm−2 at a current density of −10 mA cm−2 (Fig. 5d). Moreover, upon discharging at −1 mA cm−2, the battery exhibited a remarkable energy density of 620.12 Wh kg−1 w.r.t. the weight of Zn consumed. Again, after mechanical polishing of the Zn anode and replacing the anodic electrolyte, an energy density of 614.74 Wh kg−1 was achieved, indicating only a slight decrease and stable response of FeCu(1:2)Sx as the cathode (Fig. S28). To illustrate the practical utility of the Zn–NO3− battery system, two home-made Zn–NO3− batteries were connected in series, forming an energy unit capable of powering an array of 62 blue light-emitting diodes (LEDs). These LEDs were configured in a parallel arrangement to ensure uniform voltage distribution and stable operation. Remarkably, the system sustained uninterrupted illumination of the LEDs for a prolonged duration exceeding 70 h, as depicted in Fig. 5e–h. This compelling demonstration highlights the battery's potential for real-world energy storage and delivery applications, emphasising its robustness and prolonged operational efficacy in powering small-scale electronic devices. These results prove that FeCu(1:2)Sx could be an efficient and cost-effective cathode material for NH3 production and electricity generation simultaneously.
Conclusions
In conclusion, we have designed FeCu(1:2)Sx as an effective catalyst for the NO3RR showing the highest NH3 yield of 3.69 mg h−1 cm−2 at a potential of −0.9 V vs. RHE and a high NH3 F.E. of 96.5% at −0.6 V vs. RHE. In situ studies and electrochemical analyses validate the distinct roles of Fe and Cu in the NO3RR. Cu effectively promotes NO3− adsorption and reduction to NO2−, while Fe is essential for hydrogenation to NH3. Benefitting from superior electrochemical performance of FeCu(1:2)Sx a Zn–NO3− battery was assembled utilising the FeCu(1:2)Sx catalyst as the cathode. It exhibited a high OCV of 1.32 V with a power density of 5.2 mW cm−2 and a maximum NH3 F.E. of 98.96%. The two home-made Zn–NO3− batteries connected in series could power 62 blue LEDs for more than 70 h. This work enriches the use of Zn-based batteries for electrocatalysis and widens the scope of bimetallic catalysts for electrochemical NH3 synthesis.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, additional characterization data, and figures that validate the findings presented in the main text. See DOI: https://doi.org/10.1039/d5ta08241b.Acknowledgements
The authors thank the Central Research Facility (CRF), IIT Ropar, for FE-SEM, XPS, and Raman spectroscopy facilities. T. C. Nagaiah thanks the Anusandhan National Research Foundation (ANRF/PAIR/2025/000006/PAIR-A) for financial support. A. Chaturvedi thanks I. I. T Ropar for a fellowship and research facilities. S. Mehta thanks CSIR (09/1005(0045)/2020-EMR-I) for a fellowship.References
- H. Jiang, Y. Zhang, Y. Liu, J. Yang, L. Xu, P. Wang, Z. Gao, J. Zheng, C. Meng and Z. Pan, J. Mater. Chem. A, 2020, 8, 15130–15139 RSC.
- L. Li, P. Wang, Q. Shao and X. Huang, Chem. Soc. Rev., 2020, 49, 3072–3106 RSC.
- Y. Niu, X. Teng, S. Gong and Z. Chen, J. Mater. Chem. A, 2020, 8, 13725–13734 RSC.
- N. Thakur, M. Kumar, D. Mandal and T. C. Nagaiah, ACS Appl. Mater. Interfaces, 2021, 13, 52487–52497 CrossRef CAS PubMed.
- Y. Guo, R. Zhang, S. Zhang, Y. Zhao, Q. Yang, Z. Huang, B. Dong and C. Zhi, Energy Environ. Sci., 2021, 14, 3938–3944 RSC.
- R. Zhang, Y. Guo, S. Zhang, D. Chen, Y. Zhao, Z. Huang, L. Ma, P. Li, Q. Yang and G. Liang, Adv. Energy Mater., 2022, 12, 2103872 Search PubMed.
- T. Zhou, N. Zhang, C. Wu and Y. Xie, Energy Environ. Sci., 2020, 13, 1132–1153 RSC.
- A. Chaturvedi, D. Gupta, S. Kaur, K. Garg and T. C. Nagaiah, J. Mater. Chem. A, 2023, 11, 18280–18290 Search PubMed.
- G. Soloveichik, Nat. Catal., 2019, 2, 377–380 Search PubMed.
- G. Qing, R. Ghazfar, S. T. Jackowski, F. Habibzadeh, M. M. Ashtiani, C.-P. Chen, M. R. Smith and T. W. Hamann, Chem. Rev., 2020, 120, 5437–5516 Search PubMed.
- Y. Wang, A. Xu, Z. Wang, L. Huang, J. Li, F. Li, J. Wicks, M. Luo, D.-H. Nam, C.-S. Tan, Y. Ding, J. Wu, Y. Lum, C.-T. Dinh, D. Sinton, G. Zheng and E. H. Sargent, J. Am. Chem. Soc., 2020, 142, 5702–5708 Search PubMed.
- S. Ye, Z. Chen, G. Zhang, W. Chen, C. Peng, X. Yang, L. Zheng, Y. Li, X. Ren, H. Cao, D. Xue, J. Qiu, Q. Zhang and J. Liu, Energy Environ. Sci., 2022, 15, 760–770 RSC.
- X. Fan, J. Liang, L. Zhang, D. Zhao, L. Yue, Y. Luo, Q. Liu, L. Xie, N. Li, B. Tang, Q. Kong and X. Sun, Carbon Neutralization, 2022, 1, 6–13 CrossRef.
- A. Chaturvedi, S. Gaber, S. Kaur, K. C. Ranjeesh, T. C. Nagaiah and D. Shetty, ACS Energy Lett., 2024, 9, 2484–2491 Search PubMed.
- Y. Wang, C. Wang, M. Li, Y. Yu and B. Zhang, Chem. Soc. Rev., 2021, 50, 6720–6733 RSC.
- Z. Wang, D. Richards and N. Singh, Catal. Sci. Technol., 2021, 11, 705–725 RSC.
- E. Murphy, Y. Liu, I. Matanovic, M. Rüscher, Y. Huang, A. Ly, S. Guo, W. Zang, X. Yan, A. Martini, J. Timoshenko, B. R. Cuenya, I. V. Zenyuk, X. Pan, E. D. Spoerke and P. Atanassov, Nat. Commun., 2023, 14, 4554 Search PubMed.
- A. Chaturvedi, K. Garg and T. C. Nagaiah, Small, 2025, e09526 CrossRef CAS PubMed.
- Z. Tang, Z. Bai, X. Li, L. Ding, B. Zhang and X. Chang, Processes, 2022, 10, 751 CrossRef CAS.
- J. Wang, Y. Wang, C. Cai, Y. Liu, D. Wu, M. Wang, M. Li, X. Wei, M. Shao and M. Gu, Nano Lett., 2023, 23, 1897–1903 CrossRef CAS PubMed.
- A. Chaturvedi, S. Kaur, K. Garg and T. C. Nagaiah, J. Mater. Chem. A, 2025, 13, 9923–9932 Search PubMed.
- H. Xu, Y. Ma, J. Chen, W.-x. Zhang and J. Yang, Chem. Soc. Rev., 2022, 51(7), 2710–2758 Search PubMed.
- M. Teng, J. Ye, C. Wan, G. He, H. J. I. Chen and E. C. Research, J. Ind. Eng. Chem., 2022, 61, 14731–14746 Search PubMed.
- J. Zhao, L. Liu, Y. Yang, D. Liu, X. Peng, S. Liang and L. Jiang, ACS Sustain. Chem. Eng., 2023, 11, 2468–2475 Search PubMed.
- Y. Xu, K. Ren, T. Ren, M. Wang, Z. Wang, X. Li, L. Wang and H. Wang, Appl. Catal., B, 2022, 306, 121094 Search PubMed.
- Z.-Y. Wu, M. Karamad, X. Yong, Q. Huang, D. A. Cullen, P. Zhu, C. Xia, Q. Xiao, M. Shakouri, F.-Y. Chen, J. Y. Kim, Y. Xia, K. Heck, Y. Hu, M. S. Wong, Q. Li, I. Gates, S. Siahrostami and H. Wang, Nat. Commun., 2021, 12, 2870 CrossRef CAS PubMed.
- P. Li, Z. Jin, Z. Fang and G. Yu, Energy Environ. Sci., 2021, 14, 3522–3531 RSC.
- Q. Liu, Q. Liu, L. Xie, Y. Ji, T. Li, B. Zhang, N. Li, B. Tang, Y. Liu, S. Gao, Y. Luo, L. Yu, Q. Kong and X. Sun, ACS Appl. Mater. Interfaces, 2022, 14, 17312–17318 CrossRef CAS PubMed.
- W. Guo, K. Zhang, Z. Liang, R. Zou and Q. Xu, Chem. Soc. Rev., 2019, 48, 5658–5716 RSC.
- T. Kandemir, M. E. Schuster, A. Senyshyn, M. Behrens and R. Schlögl, Angew. Chem., Int. Ed., 2013, 52, 12723–12726 CrossRef CAS PubMed.
- W. H. Campbell, Annu. Rev. Plant Physiol. Plant Mol. Biol., 1999, 50, 277–303 CrossRef CAS PubMed.
- W. Wen, P. Yan, W. Sun, Y. Zhou and X.-Y. Yu, Adv. Funct. Mater., 2023, 33, 2212236 CrossRef CAS.
- K. Fan, W. Xie, J. Li, Y. Sun, P. Xu, Y. Tang, Z. Li and M. Shao, Nat. Commun., 2022, 13, 7958 CrossRef CAS PubMed.
- W. Gao, K. Xie, J. Xie, X. Wang, H. Zhang, S. Chen, H. Wang, Z. Li and C. Li, Adv. Mater., 2023, 35, 2202952 CrossRef CAS PubMed.
- W. He, J. Zhang, S. Dieckhöfer, S. Varhade, A. C. Brix, A. Lielpetere, S. Seisel, J. R. C. Junqueira and W. Schuhmann, Nat. Commun., 2022, 13, 1129 Search PubMed.
- J.-Y. Fang, Q.-Z. Zheng, Y.-Y. Lou, K.-M. Zhao, S.-N. Hu, G. Li, O. Akdim, X.-Y. Huang and S. S. Shi-Gang, Nat. Commun., 2022, 13(1), 7899 Search PubMed.
- J.-Y. Fang, Q.-Z. Zheng, Y.-Y. Lou, K.-M. Zhao, S.-N. Hu, G. Li, O. Akdim, X.-Y. Huang and S.-G. Sun, Nat. Commun., 2022, 13, 7899 CrossRef CAS PubMed.
- J.-X. Liu, D. Richards, N. Singh and B. R. Goldsmith, ACS Catal., 2019, 9, 7052–7064 Search PubMed.
- B. Li, L. Huang, M. Zhong, Z. Wei and J. Li, RSC Adv., 2015, 5, 91103–91107 RSC.
- M. Wang, C. Xing, K. Cao, L. Zhang, J. Liu and L. Meng, J. Mater. Chem. A, 2014, 2, 9496–9505 Search PubMed.
- M. Goswami, S. Kumar, N. Singh, N. Sathish, M. Ashiq and S. Kumar, Ionics, 2021, 27, 5277–5285 Search PubMed.
- P. V. F. de Sousa, A. F. de Oliveira, A. A. da Silva and R. P. Lopes, Environ. Sci. Pollut. Res., 2019, 26, 14883–14903 Search PubMed.
- M. Lu, N. Gao, X.-J. Zhang and G.-S. Wang, RSC Adv., 2019, 9, 5550–5556 RSC.
- W. Yu, J. Yu, M. Huang, Y. Wang, Y. Wang, J. Li, H. Liu and W. Zhou, Energy Environ. Sci., 2023, 16, 2991–3001 RSC.
- Y. Cao, D. Li, C. Ding, S. Ye, X. Zhang, H. Chi, L. Liu, Y. Liu, J. Xiao and C. Li, ACS Catal., 2023, 13, 11902–11909 CrossRef CAS.
- G. B. Murdoch, M. Greiner, M. G. Helander, Z. B. Wang and Z. H. Lu, Appl. Phys. Lett., 2008, 93(8), 083309 CrossRef.
- H. Younan, S. Yue, L. Kai, W. J. Yuan, C. Y. Shen, C. Yixin, F. Chao and L. Xiaomin, Studies on XPS valence state analysis of copper materials, 2016 IEEE 37th International Electronics Manufacturing Technology (IEMT) & 18th Electronics Materials and Packaging (EMAP) Conference, 2016, pp. 1–2, DOI:10.1109/IEMT.2016.7761945.
- P. Rupa Ranjani, P. M. Anjana and R. B. Rakhi, J. Energy Storage, 2021, 33, 102063 CrossRef.
- G. Urbano, I. Lázaro, I. Rodríguez, J. L. Reyes, R. Larios and R. Cruz, Int. J. Min. Met. Mater., 2016, 23, 127–136 Search PubMed.
- D. Gupta, A. Kafle, S. Kaur, T. S Thomas, D. Mandal and T. C. Nagaiah, ACS Appl. Mater. Interfaces, 2023, 15, 4033–4043 Search PubMed.
- S. Chen, K. Lian, W. Liu, Q. Liu, G. Qi, J. Luo and X. Liu, Nano Res., 2023, 9214–9230 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




