 Open Access Article
Open Access ArticleExploring the potential of transition metal tungstates for photoelectrochemical water oxidation: a combined experimental and computational approach
Ana
Caravaca
 ,
Francisco J.
Pastor
,
Francisco J.
Pastor
 ,
Andrés
Parra-Puerto
,
Andrés
Parra-Puerto
 *,
Nathalia C.
Verissimo
*,
Nathalia C.
Verissimo
 ,
Néstor
Guijarro
,
Néstor
Guijarro
 ,
Teresa
Lana-Villarreal
,
Teresa
Lana-Villarreal
 and
Roberto
Gómez
and
Roberto
Gómez
 *
*
Departament de Química Física i Institut Universitari d'Electroquímica, Universitat d'Alacant, Apartat 99, E-03080 Alicante, Spain. E-mail: roberto.gomez@ua.es; andres.parra@ua.es
First published on 5th February 2026
Abstract
The combination of experimental and computational methodologies offers a reliable strategy to not only discover new materials but also rationalize their performance and their intrinsic limitations. Following this concept, fourth-period metal tungstates (RWO4, R = Mn, Fe, Co, Ni, Cu or Zn) have been explored for their application as photoanodes for solar water oxidation. The theoretical screening of these materials reveals large effective masses for both electron and hole carriers, while the density of states (DOS) profiles indicate that only Fe and Cu tungstates show adequate features to be photoanodes. This agrees with experimental data showing that they yield the best photocurrents in the series. In fact, FeWO4 reaches over 0.04 mA cm−2 at 1.23 VRHE under one sun, a value 67% higher than the current record. The still low photoactivity is partly linked to the fact that the electrode deviates from the band edge pinning regime over a wide potential range. Under the same conditions, CuWO4 delivers a photocurrent over 0.3 mA cm−2, in line with the best results found in the literature. Overall, the exploratory analysis performed in this paper not only identifies the parameters that limit the photoelectrochemical response of a wide range of metal tungstates, but it enables the identification of iron and copper tungstates as promising photoanodes for solar water oxidation whose performance may be enhanced by the implementation of some modification strategies.
1 Introduction
Photoelectrochemical (PEC) cells can perform water electrolysis by using sunlight, generating hydrogen with low or ideally zero applied bias. Such unassisted water splitting can be more easily achieved by employing a dual-absorber tandem cell configuration, which involves the assembly of a photoanode and a photocathode (generating hydrogen and oxygen, respectively).1 However, current semiconductor electrodes show several limitations. In the case of photoanodes, they tend to have a rather high onset potential, low photocurrents, and relatively poor chemical stability.2 In the course of the last decades, there have appeared several studies addressing semiconductor composition and synthesis routes oriented to overcome these limitations.3–5 An ideal photoanode must possess some characteristics such as (a) efficient solar light harvesting (band gap around 2.0 eV for a single-photoelectrode device); (b) long-term stability against photocorrosion; (c) low electron–hole recombination (favored by highly mobile carriers and fast electron–hole separation), and (d) efficient hole transfer to water/hydroxide at the electrode/electrolyte interface. Unfortunately, no material has yet been found that satisfactorily fulfils all the requirements to sustain efficient water photooxidation.6In the past years, simple oxides based on earth-abundant materials (i.e., α-Fe2O3,7 WO3,8 or TiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) have been extensively studied due to their scalable synthesis routes and acceptable photoresponse, with photocurrent densities achieving up to 6.5 mA cm−2.10 In this respect, numerous studies have focused on tungsten trioxide,8 as it presents a band gap ranging between 2.6 to 3 eV,11,12 moderate hole diffusion length (around 150 nm) and high electron mobility (µe = 12 cm2 V−1 s−1) compared to hematite and titanium dioxide,13,14 high chemical stability in acid media,15 and suitable band edge positions to drive the water oxidation reaction.16 Also, tungsten is a relatively abundant17 and cost-effective element, which makes it a good candidate to put research effort into improving the activity of tungsten trioxide. However, WO3 is not stable against photocorrosion in alkaline media,18 and it presents weak light absorption in the visible range (α = 104–105 cm−1).19 Thus, several alternatives are emerging to address these issues and enhance the oxide properties. One of the most prominent is the use of related ternary oxides, such as metal tungstates, due to their potentially more favorable band gap and enhanced stability, especially in neutral to basic media (pH above 4).
9) have been extensively studied due to their scalable synthesis routes and acceptable photoresponse, with photocurrent densities achieving up to 6.5 mA cm−2.10 In this respect, numerous studies have focused on tungsten trioxide,8 as it presents a band gap ranging between 2.6 to 3 eV,11,12 moderate hole diffusion length (around 150 nm) and high electron mobility (µe = 12 cm2 V−1 s−1) compared to hematite and titanium dioxide,13,14 high chemical stability in acid media,15 and suitable band edge positions to drive the water oxidation reaction.16 Also, tungsten is a relatively abundant17 and cost-effective element, which makes it a good candidate to put research effort into improving the activity of tungsten trioxide. However, WO3 is not stable against photocorrosion in alkaline media,18 and it presents weak light absorption in the visible range (α = 104–105 cm−1).19 Thus, several alternatives are emerging to address these issues and enhance the oxide properties. One of the most prominent is the use of related ternary oxides, such as metal tungstates, due to their potentially more favorable band gap and enhanced stability, especially in neutral to basic media (pH above 4).
The metal tungstate most widely reported as a photoelectrode is CuWO4, with a band gap between 2.2 to 2.4 eV, high chemical stability, and the ability to carry out the photooxidation of water. However, its photocurrent is usually affected by fast electron–hole recombination. Several strategies have been explored to address this issue.20–22 For instance, Yang et al. and Ikeue et al. reported that Mo doping effectively narrows the band gap and increases the electron carrier density.23,24 The construction of heterojunctions to suppress charge-carrier recombination has also been widely used; WO3 has frequently been employed as an underlayer25–27 or as a part of local heterojunctions.28 Other studies have introduced NiWO4, CdS, and BiVO4 underlayers.29–31 Surface charge-separation efficiency has additionally been improved through the incorporation of cocatalysts, such as Co–Pi, and Ni–Pi.32–34 Furthermore, collection efficiency has been enhanced by tailoring the nanostructure of CuWO4.35 Some studies have also appeared on the potential use as photoanodes of other metal tungstates, such as FeWO4,36 SnWO4,37 MnWO4,38 CoWO4,39 and NiWO4.40
Density Functional Theory (DFT) stands out as a reliable methodology to better understand these materials, predicting both properties and potential drawbacks. This should lead to a more efficient design and optimization of the materials studied. In the literature, one can find DFT calculations for the previously mentioned tungstates,41–43 although it is rare to find a theoretical screening of these materials.44,45 In most cases hybrid functionals or the Hubbard correction have been applied to increase accuracy because the method of the Generalized Gradient Approximations (GGA) functional parametrized by Perdew, Burke, and Ernzerhof (PBE) tends to underestimate semiconductor band gaps.46 Most of these studies focus on fundamental aspects and photocatalytic applications, with only a few of them dealing with photoelectrochemical aspects.47–49
This work focuses on the investigation of a series of metal tungstates, RWO4 (R = Mn, Fe, Co, Ni, Cu, and Zn) synthesized following the same sol–gel procedure. Subsequent theoretical screening provides their electronic structure, including the carrier effective masses, and their band gap values and types. Such computational output enables both rationalizing the experimental results and gaining further physical insights. It is shown how, by combining theory and experiment, the possible drawbacks for these materials are more easily tracked and tackled in an effective way. As an outcome of this study, FeWO4 and CuWO4 are identified as the most promising materials for water splitting photoanodes. Overall, this study shows how tungstates may be used instead of tungsten trioxide, particularly once methods for enhancing carrier mobility are found and implemented.
2 Experimental
2.1 Materials preparation
The materials were synthesized following a sol–gel-based spin coating method.33 Briefly, 0.685 mmol of citric acid are dissolved in 2.5 mL deionized water and 1.25 mL 2-methoxyethanol. Then, 3.746 mmol of the divalent cation precursor (R = Mn2+, Fe2+, Co2+, Ni2+, Cu2+ and Zn2+) are added. Once the previous solution is completely homogenized, 0.312 mmol of ammonium metatungstate (AMT) are introduced (Fig. S1). F:SnO2-coated glass (FTO, Pilkington TEC15) is cleaned by immersion for 5 min in soapy water and for another 5 min in deionized water and then dried in air at ambient temperature. Next, 25 µL of the precursor solution are dropped on a 1 cm2 FTO piece and spin-coated at 1500 rpm for 30 s. The number of layers applied according to this procedure was optimized for both CuWO4 and FeWO4, which revealed that using 8 layers yields the highest photocurrent (Fig. S2). Each layer was pretreated for 8 min at 350 °C in an oven prior to the application of subsequent layers. Unless otherwise stated, all the samples are made of 8 layers. The final film was thermally treated at 500 °C for 3 hours (ramp rate of 3 °C min−1).2.2 Materials characterization
X-ray diffraction (XRD) experiments were carried out using a Bruker Advance D8 with Cu-Kα radiation (λ = 1.5418 Å) generated at 40 kV and 40 mA. Experiments scanned 2θ from 10° to 45° with 0.02° per step. A Shimadzu UV-3600i Plus spectrophotometer with an integrating sphere working in absorbance mode was used for the optical properties. The top-view film morphology was obtained by scanning electron microscopy (SEM) (Jeol IT500HR/LA instrument at an acceleration voltage of 10/15 KV). Furthermore, an energy dispersive X-ray spectrometer (EDS) was employed to analyze the sample surface chemical composition.2.3 Electrochemical characterization
Photoelectrochemical measurements were carried out at room temperature in a three-electrode cappuccino-type cell equipped with a quartz window by using a potentiostat-galvanostat (Autolab, PGSTAT30). An Ag/AgCl/KCl (3 M) was used as a reference electrode (all the potentials converted to the reversible hydrogen electrode, RHE), and an Au wire as a counter electrode. A 0.2 M potassium phosphate (KPi) buffer (pH 7), a 0.1 M potassium borate (KBi) buffer (pH 9), and solutions 0.1 M KOH, 0.1 M H2SO4 were used as working electrolytes. All the experiments were conducted under substrate-electrode (SE) illumination, unless stated otherwise, with an intensity of 0.1 W cm−2 (1 sun) delivered by a solar simulator (SAN-EI ELECTRONIC) equipped with an AM 1.5 G filter. An ozone-free 1000 W Xe (Hg) arc lamp (Newport Instruments) was also used together with a UV filter (Newport model FSQ-KG3, λ > 350 nm) for some measurements with FeWO4 and CuWO4 electrodes. Incident photon to current efficiency (IPCE) was measured using a Xe arc lamp (Newport 66![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 902, Thermo Oriel) together with a monochromator (Cornerstone 260, Oriel Instruments). Measurements were taken every 10 nm in the wavelength range from 600 to 300 nm while keeping the electrode potential at 1.23 V. An optical power meter (model 70
902, Thermo Oriel) together with a monochromator (Cornerstone 260, Oriel Instruments). Measurements were taken every 10 nm in the wavelength range from 600 to 300 nm while keeping the electrode potential at 1.23 V. An optical power meter (model 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 310 from Thermo Oriel) and a photodiode (model 71
310 from Thermo Oriel) and a photodiode (model 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 638, Thermo Oriel) were used to measure the light power at each wavelength.
638, Thermo Oriel) were used to measure the light power at each wavelength.
Mott–Schottky (MS) plots were obtained by performing Electrochemical Impedance Spectroscopy (EIS) measurements at frequencies ranging from 105 Hz to 0.1 Hz with an amplitude of 10 mV and at applied potentials from 0.74 to 1.64 VRHE.
2.4 DFT calculations
DFT calculations were carried out using the Vienna Ab Initio Simulation Package (VASP)50 with the Projector Augmented Wave (PAW) method51,52 at the PBE + U level (employing the Dudarev53 approach). Using this approach an accurate band gap value cannot be obtained as it leads to significant DOS distortion (due to high U values). In our work, we have prioritized obtaining reliable DOS profiles. All calculations were performed following the work done by Pastor et al.54 To sum up, the plane-wave energy cutoff was 520 eV, and spin-polarized calculations were performed by arranging the magnetic moments in antiferromagnetic and ferromagnetic configurations and selecting that with the lowest energy. In all cases, such a configuration was the antiferromagnetic arrangement. As a particular case, ZnWO4 was considered as diamagnetic.55–61 The Ueff values (Table S1) were selected from the bibliography.29,43,50 The structures were obtained from the Crystallography Open Database (COD). All the tungstates present a monoclinic structure (P12/c1) except for CuWO4, which is triclinic (–P1).3 Results and discussion
3.1 Theoretical results
DFT calculations were carried out for the family of oxides RWO4 with R = Mn, Fe, Co, Ni, Cu, and Zn. Fig. 1 shows the profiles for the density of states (DOS) calculated at the PBE + U level for all the structures adopting the U values given in Table S1.The DOS for the valence bands (VBs) (depicted on the left of each panel of Fig. 1) are formed by O2p states hybridized with other states of the two transition metals. Likewise, the DOS corresponding to the conduction bands (CBs) (displayed on the right) show a prominent contribution of W5d levels, except for CuWO4. The band gap (Eg) is defined by R3d states near the edges of the bands for most tungstates (except for NiWO4 and ZnWO4). In any case, the DOS for these compounds and that for tungsten tioxide62 show some common characteristics as the VBs are mainly constituted by O2p states, whereas the CBs are composed of W5d states. The Mn, Fe and Co tungstates show a localized R3d contribution in the upper part of the VB, while CuWO4 shows Cu3d localized states in the lower part of the CB, suggesting the prevalence of hopping-type conductivity. These localized states are associated with the VB and the CB, respectively.
The tungstates can be classified according to their DOS into three different types: (i) MnWO4, FeWO4, and CoWO4 type: it shows localized R3d states just below the Fermi level, which may act as hole traps. Moreover, Mn and Fe tungstates present W states in the CB, which suggests that the photogenerated carriers would be in different atoms. In contrast, CoWO4 also shows R3d states in the CB and thus the photogenerated carriers would not necessarily be spatially separated, which could favor recombination. (ii) NiWO4 and ZnWO4 type: it is characterized by a large value of Eg, as defined by O states in the VB and W states in the CB. (iii) CuWO4 type: it presents R3d localized states associated with the CB, and oxygen states in the VB. The R3d states of CuWO4 would be characterized by a low degree of hybridization with the rest of the atoms in the crystal structure and they may act as electron traps. Tian et al. have reported the DOS for CuWO4 as obtained via XPS VB and O K-edge XAS.47 It is worth noting that it aligns well with the total DOS derived here from DFT. In addition, the XPS VB spectra for iron tungstate63,64 and cobalt tungstate63 show the prevalence of R3d states, as shown in Fig. 1b and c. These results are consistent with previously reported data for Co and Zn tungstates.65,66
Fig. 2 presents the band structure obtained for each RWO4 at the PBE + U level, while Fig. 3 shows the calculated effective masses for the charge carriers, which are key to understanding the limitations of these materials. From Fig. 2, both CB and VB edges are shown to be characterized by a low curvature, which indicates low charge carrier mobility and weak hybridization in the structure.67 Moreover, all samples present an indirect Eg as shown in Fig. 2 and Table S2. Results in Fig. 3 suggest that Mn, Fe, and Co tungstates exhibit rather anisotropic electric conduction, as their electron and hole effective masses vary significantly along the a, b and c directions. In contrast, Ni, Cu, and Zn tungstates show a more isotropic behavior, with effective masses that are relatively similar along the main directions. Furthermore, the relative magnitudes of electron and hole effective masses 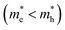 for Ni, Cu, and Zn tungstates indicate a tendency toward n-type conductivity.
for Ni, Cu, and Zn tungstates indicate a tendency toward n-type conductivity.
3.2 Experimental results
With the aim to study the particle nature, the RWO4 samples were analyzed in terms of particle size, composition, and elemental distribution by means of Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS) (Fig. 4, and S3–S5). The morphology of the layer is important as it may play a role in determining the electron–hole recombination rate.69 The sol–gel method normally yields rather compact thin films composed of nano- or microparticles. The top-view images of the MnWO4, CoWO4 and ZnWO4 thin films (Fig. 4a, c and f, respectively) reveal a morphology composed of agglomerated small particles, yielding a smooth surface. In the case of NiWO4 (Fig. 4d), some larger spherical particles also appear on top of the smooth layer. On the other hand, FeWO4 (Fig. 4b) displays larger particles, while CuWO4 (Fig. 4e) exhibits a more defined morphology consisting of spherical particles and some flakes.
The thickness of the different electrodes was measured from cross-sectional images (insets in Fig. 4). The thickness values estimated for each electrode are 3.0 µm (MnWO4), 2.6 µm (FeWO4), 2.5 µm (CoWO4), 3.2 µm (NiWO4), 1.2 µm (CuWO4), and 1.6 µm (ZnWO4). The differences observed in thickness values may be due to the role of metal ions in the sol–gel process,70 which may impact the viscosity of the solution and thus the final thickness of the spin-coated samples. Elemental mapping using EDS is shown in Fig. S5(a–f). All the samples show a homogeneous compositional distribution.
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group, COD no: 00-100-8036) at 2θ = 15.3, 19.0, 22.9, 23.6, 24.1, 25.9, 28.7, 30.2, 30.8, 31.6 and 32.2°, respectively. The peaks observed for thin films (Fig. 5 and S7) are the same as for the powders (Fig. S6). However, for the FeWO4 powder, there appear weak WO3 peaks, which are not shown in the thin film (Fig. 5). In any case, some electrochemical measurements were carried out to verify the absence of significant amounts of surface WO3 in the FeWO4 thin film (Fig. S8).
space group, COD no: 00-100-8036) at 2θ = 15.3, 19.0, 22.9, 23.6, 24.1, 25.9, 28.7, 30.2, 30.8, 31.6 and 32.2°, respectively. The peaks observed for thin films (Fig. 5 and S7) are the same as for the powders (Fig. S6). However, for the FeWO4 powder, there appear weak WO3 peaks, which are not shown in the thin film (Fig. 5). In any case, some electrochemical measurements were carried out to verify the absence of significant amounts of surface WO3 in the FeWO4 thin film (Fig. S8).
 | ||
| Fig. 5 XRD patterns for the different tungstates (as thin films) prepared by sol gel; × → WO3 and ● → FTO (database standard reference information for the XRD patterns are shown in Fig. S7). | ||
Once confirmed that the desired materials were synthesized, the experimental values of Eg were determined from UV-vis spectra (Fig. 6a) by means of Tauc plots, both for direct, EDirg (Fig. 6b) and indirect transitions, EIng (Fig. 6c). As shown in Fig. 6a, CoWO4 presents a secondary absorption band in the range from 640 nm to 500 nm (corresponding to the bluish color of the sample), together with the main absorption below 450 nm. NiWO4 also presents a secondary absorption band at ∼430 nm. Such bands are due to spin-allowed transitions for the Co2+ and Ni2+ ions, which happen between the t2g and eg subshells.54,71–73 A comparison of Fig. 1c and 6a firmly supports this assignment. The main feature in the spectra (below 450 nm) is ascribed to electronic excitations from O2p to W5d orbitals in the [WO4]2− and [WO6]6− groups.71 The materials with the narrowest Eg within this family are FeWO4 and CuWO4, providing the best light harvesting efficiency, whereas Ni and Zn tungstates display a much wider Eg minimizing light harvesting. Table S4 shows a compilation of experimental and theoretical Eg values obtained in this work and from the literature. Both sets of values are quite similar.
The resulting Efb values (0.5–0.7 VRHE except for the case of the Mn tungstate) are similar or slightly higher than for other typical electrode materials reported in the literature, such as hematite74 (Efb between 0.4 to 0.8 VRHE), bismurh vanadate75,76 (Efb between 0.1 to 0.68 VRHE), or tin tungstate77 (Efb of about 0.32 VRHE). To estimate the position of the CB edge, the flat band potential is assumed to be approximately equal to the potential corresponding to the CB edge. Thus, the following equations are used to estimate the value of the CB and VB edges:
| εCB/eV ≅ −4.44 − e(Efbvs. SHE) | (1) |
| εVB = εCB − Eg | (2) |
Fig. 7 gathers the locations of the band edges determined either theoretically or experimentally. The procedure followed for estimating the theoretical band edge positions is given in the SI.
As observed, the VB is lower in energy than the O2/H2O redox couple in all cases, which means that water photooxidation is thermodynamically feasible. Fig. 7 also shows that there are some localized electronic states in the Mn, Fe, and Cu tungstates (in agreement with theoretical calculations) that could be detrimental for their photoactivity as they may favor recombination. It is worth noting that experimental data do not exactly correspond to the expectations from the PBE + U calculations. These calculations are based on considering defect-free, bulk materials in contrast with real materials, with defects and, obviously, surfaces. Moreover, they tend to underestimate Eg values. In any case, there is an acceptable correlation between experimental and theoretical band edge positions (and thus between experimental and theoretical band gaps). This means that the calculations are able to capture the main trends.
To evaluate the photoelectrochemical response of these materials, linear sweep voltammograms (LSV) were recorded both in the dark and under illumination for electrodes with either similar (Fig. S13) or optimized thickness (Fig. 8a). The same trend is observed in both cases. MnWO4, CoWO4, NiWO4 and ZnWO4 (inset) show very modest photocurrents. In contrast, FeWO4 delivers, for the optimal thickness, a significant photocurrent of 41 µA cm−2@1.23 VRHE, which represents a 64% increase over the state-of-the-art (Fig. 8b). CuWO4 is the photoanode with the highest photoresponse, with 315 µA cm−2@1.23 VRHE for the optimized thickness, being this value (Fig. 8b) among the best reported in the literature.78 Table S8 gathers relevant literature for different electrodes based on RWO4 (from Mn to Zn).
It is worth noting that there is a relation between experimental and DFT results. The theoretical approach indicates that CuWO4 and FeWO4 are potential candidates as photoanodes because of their narrower Eg and their electronic state distribution, while the other materials could also be of interest. However, this needs to be and is clarified by means of experimental results, which show that only FeWO4 and CuWO4 yield significant photocurrents.
Only FeWO4 and CuWO4 can be considered as potential candidates for water photooxidation. Further analysis of their behavior was carried out for better understanding the charge carrier dynamics (mobility, effective masses, recombination…). This determines the maximum photocurrent that the material can achieve, and it may give hints on potential approaches to upgrade electrode performance. Experiments with more intense illumination (1 W cm−2) were performed to assess the photocurrent response under increased photon flux (Fig. S14). The spectral distributions of the 1 W cm−2 and 0.1 W cm−2 sources differ. The 1 W cm−2 spectrum shows relevant peaks in the range of 275 to 325 nm, minimized using a UV filter. A ten-fold increase in the photoresponse is expected based on the illumination intensity difference, but the actual photocurrent enhancement is lower, as shown in Fig. S14a and b. The FeWO4 electrode shows a photocurrent of 0.163 mA cm−2@1.23VRHE (4-fold increase relative to 1 sun), while the CuWO4 electrode yielded 2.87 mA cm−2@1.23VRHE (9-fold increase). This fact can be attributed mainly to charge carrier recombination, together with some persisting differences in the spectral distribution of the illumination sources.
Both front illumination (Electrolyte-RWO4-FTO, EE) and back illumination (FTO-RWO4-Electrolyte, SE) were studied. Fig. S15 reveals higher photocurrents for SE illumination than for EE illumination for both FeWO4 and CuWO4. This is rather unexpected, and it shows that these electrodes have limitations derived from poor electron mobility and bulk recombination. In fact, back illumination diminishes the recombination probability.
Assessing the maximum attainable photocurrent requires the utilization of a hole scavenger such as methanol to mitigate surface recombination. Specifically, a 50% v/v ratio of methanol/electrolyte was used.79 In such a medium, the catalytic efficiency is considered to be 100%, which means no surface recombination losses. Fig. 9a and b shows the photocurrent density in the presence (blue) and the absence (red) of methanol for both FeWO4 and CuWO4 electrodes. With the aim to get a straightforward overview of the materials limitations, the surface and bulk separation efficiencies (ηsurf and ηbulk) have been determined and are shown in Fig. 9c. The potential-dependent charge separation efficiency at the semiconductor surface was determined according to:
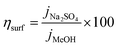 | (3) |
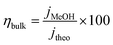 | (4) |
Even though the ηsurf values for these tungstates are high, around 73% (FeWO4) and 65% (CuWO4) at 1.23 V, ηbulk values are lower (around 0.24% for FeWO4 and 6.6% for CuWO4 at 1.23 V in both cases), being the CuWO4 results in line with those in the literature.81 They are also coherent with the high effective masses of the charge carriers and the existence of localized states as observed by DFT.55 It can be seen that ηbulk increases at higher potentials for CuWO4 likely due to a stronger band bending, which favors charge carrier separation and lowers charge recombination. In contrast, ηbulk for FeWO4 does barely change with potential, as expected for electrodes that deviate from the BEP regime. FeWO4 presents a much lower bulk efficiency compared with CuWO4 (by 28 times), which can be further supported based on Fig. 1b, as FeWO4 presents VB-associated Fe3d localized states that may act as holes trap. For CuWO4 the 3d localized states (less localized than for FeWO4) are associated with the CB, acting in this case as electron traps (Fig. 1e), which is expected to have a lower impact as it affects the majority carriers.
The surface efficiency shows an unexpected behavior in the case of FeWO4 as it decreases for increasing potentials above 1.1 V vs. RHE. Surface states seem to facilitate catalytic activity at low applied potentials, probably by mediating charge transfer, while under more oxidative conditions, they tend to behave as recombination centers. These results highlight the dual role of surface states in FeWO4.
Recombination kinetics was also studied by means of photocurrent transients carried out at 1.23 VRHE (Fig. 10a). When the light is on, a peak appears corresponding to surface accumulation of minority carriers, while a cathodic spike is observed upon turning off the light, resulting from the recombination of surface-trapped holes with FTO electrons. We define R as a normalized transient photocurrent density by means of eqn (5).82–84
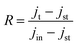 | (5) |
 | ||
Fig. 10 (a) Photocurrent density–time curves of Fe and Cu tungstates measured under 1 sun illumination at 1.23 VRHE. (b) Plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) R vs. time. R vs. time. | ||
Fig. 11 shows the incident photon to current efficiency (IPCE) as a function of wavelength for both FeWO4 and CuWO4 electrodes. FeWO4 starts to show a photoresponse at around 600 nm, which indicates an effective Eg of about 2.1 eV, similar to the optical indirect Eg obtained through UV-vis measurements (Fig. 6b and c). CuWO4 presents the onset at around 550 nm, corresponding to an effective Eg around 2.21 eV, which is comparable to the indirect Eg resulting from Fig. 6c. The integrated IPCE spectra (Fig. S17) yields a short-circuit photocurrent density (JSC) of 44 µA cm−2 for FeWO4 and 200 µA cm−2 for CuWO4. The FeWO4 value is almost identical to the photocurrent measured under simulated sunlight, whereas the value for CuWO4 is 1.58 times lower. Such a difference likely indicates that the recombination processes are more sensitive to light intensity for CuWO4 than for FeWO4.
Finally, as the eventual enhancement of the stability of WO3-related materials in a broad range of media was one of the motivations of this study, tungstates were tested in solutions of different pH (pH values tested: 1, 7, 9 and 13.5) (Fig. S18a and b). The stabilities for CuWO4 and FeWO4 slightly depend on pH, with neutral media being more favorable. Relatively minor photocurrent decays of around 9% and 25% for CuWO4 and FeWO4, respectively, are though recorded after 3 h of continuous illumination. As in the case of WO3, these materials are unstable in highly alkaline media (pH = 13.5–14).
From a practical perspective, sol–gel prepared Cu and Fe tungstates are promising transparent photoanodes due to their relatively narrow band gaps and good chemical stability. These features make them suitable for integration into tandem PEC devices in which they can be the front electrode (photoanode) to be combined with a second semiconductor electrode (photocathode) possessing a narrower band gap. These devices could potentially drive solar water-splitting without the application of a bias.85,86 Further optimization through oxide modification, interface engineering, and optical management will be essential for designing efficient devices.
4 Conclusions
A series of transition metal tungstates (RWO4; R = Mn to Zn) has been synthesized, characterized, and studied both experimentally (photoelectrochemical measurements) and theoretically (DFT). Calculations reveal that these tungstates can be divided into three categories according to the DOS profiles: (i) materials with R3d states in the VB (Mn, Fe and Co tungstates); (ii) those with a wider band gap and no R3d states near the band edges (Ni and Zn tungstates); and (iii) those with R3d states associated with the CB (CuWO4). The band diagrams show indirect Eg for all these materials, with relatively high values of the carrier effective masses. The theoretical results indicate that, out of the series, CuWO4 and FeWO4 are the most promising materials for photoelectrochemical applications despite the presence of d-states at the band edges, which may act as hole and electron traps. Specifically, for FeWO4, minority carriers would be trapped at Fe3d VB states, which can be hypothesized to have a significant impact on its photoelectrochemical bulk efficiency. In the case of CuWO4, the electronic structure points to the existence of electron traps, which are expected to affect to a lesser extent the electrode performance. UV-vis spectroscopy measurements confirm the indirect band gaps as well as the general trends theoretically predicted for the Eg values.Mott–Schottky plots reveal that only CuWO4 electrodes follow the BEP regime for an n-type semiconductor, which poses a limitation in the anodic photocurrents that the rest of the tungstates can deliver. Accordingly, an analysis of the photoelectrochemical performance for water oxidation reveals that the best performing material is CuWO4, with a photocurrent over 0.3 mA cm−2@1.23 VRHE under 1 sun. FeWO4 photoanodes yield a photocurrent over 0.04 mA cm−2@1.23 VRHE, which is relatively modest, but over 60% higher than the current state-of-the-art.
Although the photoresponses for the iron and copper tungstates are remarkable, these materials are still far from reaching their theoretical maximum photocurrents. This is a consequence of (i) the deviation from the BEP regime in the potential region of interest for FeWO4 electrodes, (ii) the low bulk efficiencies due to the presence of electronic states that can act as carrier traps and (iii) the large effective masses of both charge carriers. These limitations may be overcome by tailored nanostructuring as all the materials present anisotropic conductivity (according to the evaluation of carrier effective masses). In addition, doping these materials to increase conductivity could be a good strategy to achieve higher photocurrents and lower recombination rates. In a more general vein, we believe that this study illustrates how a combination of theoretical and experimental results may help advance toward the development of materials useful in the context of artificial photosynthesis.
Author contributions
A. C.: data curation, formal analysis, investigation, methodology, visualization, software, writing – original draft; F. J. P.: software, conceptualization, data curation, formal analysis, methodology, resources, supervision, writing – review & editing; A. P.-P.: data curation, formal analysis, methodology, supervision, writing – review & editing; N. V.: data curation, writing – review & editing; N. G.: methodology, writing – review & editing; T. L.-V.: writing – review & editing, funding acquisition; R. G.: conceptualization, data curation, funding acquisition, methodology, project administration, resources, supervision, validation, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: procedures for materials preparation, further photoelectrochemical characterization, DFT computational details and results (theoretical band edge energies), additional SEM images, flat band potentials, and comparison with literature. See DOI: https://doi.org/10.1039/d5ta08157b.Acknowledgements
These results are part of the project TED2021-132697B-I00, funded by MCIN/AEI/10.13039/501100011033 and by the Union Europea “NextGenerationEU”/PRTR. We are also grateful to the MCINN/AEI/FEDER for financial support through projects PID2021-128876OB-I00 and PID2024-162784OB-I00. A. P.-P. and N. C. V are grateful to the Ministry of Universities, the European Union “NextGenerationEU” and the University of Alicante for the María Zambrano Fellowship. N. G. thanks the Spanish Ministry of Science & Innovation for the “Ramon y Cajal” Program (RYC2018-023888-I) funded by MCIN/AEI/10.13039/501100011033.References
- K. Sivula and R. van de Krol, Nat. Rev. Mater., 2016, 1, 15010 CrossRef CAS.
- K. Sivula, Chimia, 2013, 67, 155 CrossRef CAS PubMed.
- A. Radmilovic, T. J. Smart, Y. Ping and K.-S. Choi, Chem. Mater., 2020, 32, 3262–3270 CrossRef CAS.
- T. T. Nguyen, M. Patel, S. Kim, V.-A. Dao and J. Kim, ACS Appl. Mater. Interfaces, 2021, 13, 10181–10190 CrossRef CAS PubMed.
- T. Higashi, Y. Sasaki, Y. Kawase, H. Nishiyama, M. Katayama, K. Takanabe and K. Domen, Catalysts, 2021, 11, 584 CrossRef CAS.
- C. Ros, T. Andreu and J. R. Morante, J. Mater. Chem. A, 2020, 8, 10625–10669 RSC.
- R.-T. Gao, J. Zhang, T. Nakajima, J. He, X. Liu, X. Zhang, L. Wang and L. Wu, Nat. Commun., 2023, 14, 2640 CrossRef CAS PubMed.
- A. K. Díaz-García and R. Gómez, J. Photochem. Photobiol., A, 2018, 366, 65–71 CrossRef.
- Z. Li, L. Luo, M. Li, W. Chen, Y. Liu, J. Yang, S.-M. Xu, H. Zhou, L. Ma, M. Xu, X. Kong and H. Duan, Nat. Commun., 2021, 12, 6698 CrossRef CAS PubMed.
- K. Hidayatullah, J. Manopo, I. Supu, A. Hadju, C. Ofiyen, M. K. Mahardhika and Y. Darma, Inorg. Chem. Commun., 2025, 179, 114885 CrossRef CAS.
- Y. Wang, X. Li and Y. Yang, RSC Adv., 2022, 12, 26099–26105 RSC.
- Y. Li, Q. Mei, Z. Liu, X. Hu, Z. Zhou, J. Huang, B. Bai, H. Liu, F. Ding and Q. Wang, Appl. Catal., B, 2022, 304, 120995 CrossRef CAS.
- X. Feng, Y. Chen, Z. Qin, M. Wang and L. Guo, ACS Appl. Mater. Interfaces, 2016, 8, 18089–18096 CrossRef CAS PubMed.
- S. Ye, C. Ding, M. Liu, A. Wang, Q. Huang and C. Li, Adv. Mater., 2019, 31, 1902069 CrossRef CAS PubMed.
- S. A. Alves, L. L. Soares, L. A. Goulart and L. H. Mascaro, J. Solid State Electrochem., 2016, 20, 2461–2470 CrossRef CAS.
- W. A. Smith, I. D. Sharp, N. C. Strandwitz and J. Bisquert, Energy Environ. Sci., 2015, 8, 2851–2862 RSC.
- X. WANG, W. qing QIN, F. JIAO, L. yang DONG, J. gen GUO, J. ZHANG and C. ren YANG, Trans. Nonferrous Met. Soc. China, 2022, 32, 2318–2338 CrossRef CAS.
- S. Speldrich, M. Wark and G. Wittstock, ACS Appl. Energy Mater., 2023, 6, 9602–9614 CrossRef CAS.
- J. Cen, Q. Wu, D. Yan, W. Zhang, Y. Zhao, X. Tong, M. Liu and A. Orlov, RSC Adv., 2019, 9, 899–905 RSC.
- Q. Yi, H. Wang and J.-M. Lee, ChemElectroChem, 2025, 12, e202400600 CrossRef CAS.
- H. Abdullah, H. Shuwanto, J. Lie and M. Sillanpää, J. Environ. Chem. Eng., 2023, 11, 109356 CrossRef CAS.
- J. U. Lee, J. H. Kim and J. S. Lee, Catalysts, 2023, 13, 1408 CrossRef CAS.
- K. Ikeue and T. Ueno, Mater. Lett., 2023, 348, 134690 CrossRef CAS.
- J. Yang, C. Li and P. Diao, Electrochim. Acta, 2019, 308, 195–205 CrossRef CAS.
- C. Loka, D. Gelija, S. V. P. Vattikuti and K.-S. Lee, ACS Sustain. Chem. Eng., 2023, 11, 11978–11990 CrossRef CAS.
- J. Li, S. Hu, S. Liu, S. Hou, L. Li and J. Huang, Int. J. Hydrogen Energy, 2024, 61, 967–974 CrossRef CAS.
- D. Wang, P. Bassi, H. Qi, X. Zhao, Gurudayal, L. Wong, R. Xu, T. Sritharan and Z. Chen, Materials, 2016, 9, 348 CrossRef PubMed.
- P. Ostellari, S. Benedoue, D. Zamboni, A. Basagni, S. Silloni, E. Scattolin, M. Lorenzoni, R. Pilot, I. Fortunati, S. Lauciello, M. Wang, M. Prato, J. N. Ndi, F. Arcudi, L. Đorđević, G. Meneghesso, S. Gross, L. Franco, G. Rizzi, T. Gatti and F. Lamberti, Adv. Mater. Interfaces, 2025, 12, e00610 CrossRef CAS.
- M. Zhou, Z. Liu, X. Li and Z. Liu, Ind. Eng. Chem. Res., 2018, 57, 6210–6217 CrossRef CAS.
- L. Fan, J. Sunarso, X. Zhang, X. Xiong, L. He, L. Luo, F. Wang, Z. Fan, C. Wu, D. Han, N. H. Wong, Y. Wang, G. Chen and W. Chen, Int. J. Hydrogen Energy, 2022, 47, 20153–20165 CrossRef CAS.
- W. Ye, F. Chen, F. Zhao, N. Han and Y. Li, ACS Appl. Mater. Interfaces, 2016, 8, 9211–9217 CrossRef CAS PubMed.
- X. Xiong, L. Fan, G. Chen, Y. Wang, C. Wu, D. Chen, Y. Lin, T. Li, S. Fu and S. Ren, Electrochim. Acta, 2019, 328, 135125 CrossRef CAS.
- J. U. Lee, J. H. Kim, K. Kang, Y. S. Shin, J. Y. Kim, J. H. Kim and J. S. Lee, Renewable Energy, 2023, 203, 779–787 CrossRef CAS.
- C. Li, B. Guo, B. Peng, C. Yue and P. Diao, Int. J. Electrochem. Sci., 2019, 14, 9017–9029 CrossRef CAS.
- N. Nasori, D. Cao, Z. Wang, U. Farahdina, A. Rubiyanto and Y. Lei, Molecules, 2021, 26, 2900 CrossRef CAS PubMed.
- X. M. C. Ta, T. K. A. Nguyen, A. D. Bui, H. T. Nguyen, R. Daiyan, R. Amal, T. Tran-Phu and A. Tricoli, Adv. Mater. Technol., 2023, 8, 2201760 CrossRef CAS.
- W. Qiu, Y. Zhang, G. He, L. Chen, K. Wang, Q. Wang, W. Li, Y. Liu and J. Li, ACS Appl. Energy Mater., 2022, 5, 11732–11739 CrossRef CAS.
- A. E. B. Lima, M. Assis, A. L. S. Resende, H. L. S. Santos, L. H. Mascaro, E. Longo, R. S. Santos, L. S. Cavalcante and G. E. Luz, J. Solid State Electrochem., 2022, 26, 997–1011 CrossRef CAS.
- P. Chatterjee and A. K. Chakraborty, Sol. Energy, 2022, 232, 312–319 CrossRef CAS.
- F. Zhan, H. Zhao, G. Wen, Z. Ma, Y. Liu, M. Zhu, Y. Zheng, D. Chen and P. La, New J. Chem., 2023, 47, 1620–1624 RSC.
- N. Doudin, S. Pomp, M. Blatnik, R. Resel, M. Vorokhta, J. Goniakowski, C. Noguera, F. P. Netzer and S. Surnev, Surf. Sci., 2017, 659, 20–30 CrossRef CAS.
- S. Shepard and M. Smeu, Comput. Mater. Sci., 2018, 143, 301–307 CrossRef CAS.
- E. Bandiello, P. Rodríguez-Hernández, A. Muñoz, M. B. Buenestado, C. Popescu and D. Errandonea, Mater. Adv., 2021, 2, 5955–5966 RSC.
- B. Huang and J. N. Hart, Phys. Chem. Chem. Phys., 2020, 22, 1727–1737 RSC.
- S. Dey, R. A. Ricciardo, H. L. Cuthbert and P. M. Woodward, Inorg. Chem., 2014, 53, 4394–4399 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- C. M. Tian, M. Jiang, D. Tang, L. Qiao, H. Y. Xiao, F. E. Oropeza, J. P. Hofmann, E. J. M. Hensen, A. Tadich, W. Li, D. C. Qi and K. H. L. Zhang, J. Mater. Chem. A, 2019, 7, 11895–11907 RSC.
- D. Peeters, O. Mendoza Reyes, L. Mai, A. Sadlo, S. Cwik, D. Rogalla, H.-W. Becker, H. M. Schütz, J. Hirst, S. Müller, D. Friedrich, D. Mitoraj, M. Nagli, M. C. Toroker, R. Eichberger, R. Beranek and A. Devi, J. Mater. Chem. A, 2018, 6, 10206–10216 RSC.
- C. Kim, D. Jeon, N. Kim, J. Ryu and J. H. Lee, Small, 2024, 2400114 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- P. E. Blöchl, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Phys. Rev. B:Condens. Matter Mater. Phys., 1998, 57, 1505–1509 CrossRef CAS.
- F. J. Pastor, M. Contreras, T. Lana-Villarreal, J. M. Orts and R. Gómez, Ceram. Int., 2023, 49, 24812–24823 CrossRef CAS.
- A. Kalinko, A. Kuzmin, P. Roy and R. A. Evarestov, Low Temp. Phys., 2016, 42, 552–555 CrossRef CAS.
- J. B. Forsyth, C. Wilkinson and A. I. Zvyagin, J. Phys.: Condens. Matter, 1991, 3, 8433 CrossRef CAS.
- S. Shanmugapriya, V. D. Nithya, A. Rajalakshmi, K. S. Sivaranjani, P. Bharathi, S. Shalini, P. R. Kasturi and R. K. Selvan, J. Mater. Sci.: Mater. Electron., 2020, 31, 15616–15626 CrossRef CAS.
- A. Maignan, M. Schmidt, Y. Prots, O. I. Lebedev, R. Daou, C.-F. Chang, C.-Y. Kuo, Z. Hu, C.-T. Chen, S.-C. Weng, S. G. Altendorf, L.-H. Tjeng and Y. Grin, Chem. Mater., 2022, 34, 789–797 CrossRef CAS.
- J. Mao, J. Zhao, L. Li, T. Chen, Q. Lv and Y. Li, Chin. J. Phys., 2020, 63, 163–167 CrossRef CAS.
- M. Jeyakanthan, U. Subramanian, R. B. Tangsali and A. Ramesh, Phys. B, 2020, 586, 412151 CrossRef CAS.
- A. Jain, G. Hautier, C. J. Moore, S. Ping Ong, C. C. Fischer, T. Mueller, K. A. Persson and G. Ceder, Comput. Mater. Sci., 2011, 50, 2295–2310 CrossRef CAS.
- A. K. Mohamedkhair, Q. A. Drmosh, M. Qamar and Z. H. Yamani, Catalysts, 2021, 11, 381 CrossRef CAS.
- S. Rajagopal, D. Nataraj, O. Yu. Khyzhun, Y. Djaoued, J. Robichaud and D. Mangalaraj, J. Alloys Compd., 2010, 493, 340–345 CrossRef CAS.
- S. G. Altendorf, D. Takegami, A. Meléndez-Sans, C. F. Chang, M. Yoshimura, K. D. Tsuei, A. Tanaka, M. Schmidt and L. H. Tjeng, Phys. Rev. B, 2023, 108, 85119 CrossRef CAS.
- O. Y. Khyzhun, V. L. Bekenev, V. V Atuchin, E. N. Galashov and V. N. Shlegel, Mater. Chem. Phys., 2013, 140, 588–595 CrossRef CAS.
- G. Bakradze, A. Kalinko and A. Kuzmin, J. Phys. Chem. Solids, 2022, 161, 110425 CrossRef CAS.
- V.-A. Ha, F. Ricci, G.-M. Rignanese and G. Hautier, J. Mater. Chem. C, 2017, 5, 5772–5779 RSC.
- L. Chen, W. Li, W. Qiu, G. He, K. Wang, Y. Liu, Q. Wu and J. Li, ACS Appl. Mater. Interfaces, 2022, 14, 47737–47746 CrossRef CAS PubMed.
- M. Xiao, B. Luo, Z. Wang, S. Wang and L. Wang, Sol. RRL, 2020, 4, 1900509 CrossRef CAS.
- J. H. Lopes, O. M. V. M. Bueno, I. O. Mazali and C. A. Bertran, Mater. Sci. Eng., C, 2019, 97, 669–678 CrossRef CAS PubMed.
- M. Jeyakanthan, U. Subramanian and R. B. Tangsali, J. Mater. Sci.: Mater. Electron., 2018, 29, 1914–1924 CrossRef CAS.
- H. Harshan, K. P. Priyanka, A. Sreedevi, A. Jose and T. Varghese, Eur. Phys. J. B, 2018, 91, 287 CrossRef CAS.
- K. Karoui, A. Ben Rhaiem and F. Jemni, Ionics, 2021, 27, 1511–1524 CrossRef CAS.
- B. Iandolo, H. Zhang, B. Wickman, I. Zorić, G. Conibeer and A. Hellman, RSC Adv., 2015, 5, 61021–61030 RSC.
- Y. Zhang, X. Chen, F. Jiang, Y. Bu and J.-P. Ao, ACS Sustain. Chem. Eng., 2020, 8, 9184–9194 Search PubMed.
- T. Li, J. He, B. Peña and C. P. Berlinguette, Angew. Chem., Int. Ed., 2016, 55, 1769–1772 CrossRef CAS PubMed.
- D. Liu, X. Chen, Y. Qiao, Y. Zhou and Y. Kuang, Adv. Energy Sustainability Res., 2022, 3, 2100146 CrossRef CAS.
- Y. Liu, L. Chen, X. Zhu, H. Qiu, K. Wang, W. Li, S. Cao, T. Zhang, Y. Cai, Q. Wu and J. Li, J. Electroanal. Chem., 2022, 924, 116859 CrossRef CAS.
- A. Venugopal and W. A. Smith, Faraday Discuss., 2019, 215, 175–191 RSC.
- C. A. Mesa, A. Kafizas, L. Francàs, S. R. Pendlebury, E. Pastor, Y. Ma, F. Le Formal, M. T. Mayer, M. Grätzel and J. R. Durrant, J. Am. Chem. Soc., 2017, 139, 11537–11543 CrossRef CAS PubMed.
- K. Wang, L. Chen, X. Liu, J. Li, Y. Liu, M. Liu, X. Qiu and W. Li, Chem. Eng. J., 2023, 471, 144730 CrossRef CAS.
- J. Li, S. K. Cushing, P. Zheng, F. Meng, D. Chu and N. Wu, Nat. Commun., 2013, 4, 2651 CrossRef PubMed.
- X. Zhong, H. He, M. Yang, G. Ke, Z. Zhao, F. Dong, B. Wang, Y. Chen, X. Shi and Y. Zhou, J. Mater. Chem. A, 2018, 6, 10456–10465 RSC.
- D. Tafalla, P. Salvador and R. M. Benito, J. Electrochem. Soc., 1990, 137, 1810 CrossRef CAS.
- B. Liu, S. Wang, G. Zhang, Z. Gong, B. Wu, T. Wang and J. Gong, Chem. Soc. Rev., 2023, 52, 4644–4671 RSC.
- A. Cots, P. Bonete, D. Sebastián, V. Baglio, A. S. Aricò and R. Gómez, ACS Appl. Mater. Interfaces, 2018, 10, 25393–25400 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |









