 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Oxygen vacancy-rich In2O3-ZrO2 catalysts synthesized via DBD plasma for enhanced CO2-to-CO conversion
Sai
Li
*a,
Yuhang
Wang
a,
Kui
Zhang
b,
Haiyan
Zhu
c,
Shaobo
Jia
c,
Dongyuan
Yang
a,
Peng
Ren
d,
Zekai
Ma
a,
Shuoshuo
Wang
a,
Haixia
Wu
a,
Yameng
Ma
a,
Qi
Chen
a,
Jiahao
Zhouhuang
a,
Qiuliang
Yu
g,
Lihui
Zeng
h,
Rui
Tan
 e,
Zhiming
Feng
e,
Zhiming
Feng
 *f and
Qing
Feng
*d
*f and
Qing
Feng
*d
aSchool of Chemistry and Chemical Engineering, Xi'an University of Science and Technology, Xi'an, 710054, China. E-mail: saili@xust.edu.cn
bSchool of Engineering, Newcastle University, Newcastle Upon Tyne NE1 7RU, UK
cShaanxi Key Laboratory for Theoretical Physics Frontiers, Institute of Modern Physics, Northwest University, Xi'an, Shaanxi 710069, China
dXi'an Tai Jin Xin Neng Technology Co., Ltd, Xi'an, 710201, China. E-mail: fengqing0715@126.com
eDepartment of Chemical Engineering, Swansea University, Swansea, SA1 8EN, UK
fDepartment of Chemical Engineering, University of Manchester, Manchester, M13 9PL, UK. E-mail: zhiming.feng@manchester.ac.uk
gShaanxi Anchuang Electronic Technology Co., Ltd, Xi'an, 710201, China
hKaili Catalyst New Materials Co., Ltd, Xi'an, 710201, China
First published on 6th January 2026
Abstract
The efficient utilization of CO2 as a carbon feedstock is vital for achieving carbon neutrality while enabling sustainable production of C1 chemicals. Plasma-assisted catalytic conversion has emerged as a promising strategy under mild conditions, yet its progress is limited by the lack of highly active and plasma-tolerant catalysts. In this work, an In2O3-ZrO2 composite catalyst with high catalytic activity, excellent thermal stability and long service life was successfully prepared by combining the chemical precipitation method with plasma technology. The In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst exhibited the best performance, reaching a CO2 conversion of 26.3% and CO selectivity above 90% at an SIE of 104 kJ L−1. Compared with pure In2O3, the composite showed markedly improved thermal stability, sustaining continuous operation for 450 min, three times longer than In2O3. Plasma modification induced a higher concentration of oxygen vacancies (1.69 × 1013 spins per g), increased surface area (56.7 m2 g−1), and a narrowed bandgap (2.49 eV), which synergistically enhanced catalytic activity. Mechanistic studies and DFT calculations further revealed that the strong plasma-catalyst interaction facilitates CO2 activation pathways. This study demonstrates not only the durability of In-Zr composites but also highlights plasma modification as an effective strategy to design next-generation catalysts for plasma-assisted CO2 utilization. Meanwhile, the In-Zr catalyst successfully developed in this study, with its outstanding performance, stability and durability, is a highly promising candidate material for high-temperature industrial catalytic processes.
1) catalyst exhibited the best performance, reaching a CO2 conversion of 26.3% and CO selectivity above 90% at an SIE of 104 kJ L−1. Compared with pure In2O3, the composite showed markedly improved thermal stability, sustaining continuous operation for 450 min, three times longer than In2O3. Plasma modification induced a higher concentration of oxygen vacancies (1.69 × 1013 spins per g), increased surface area (56.7 m2 g−1), and a narrowed bandgap (2.49 eV), which synergistically enhanced catalytic activity. Mechanistic studies and DFT calculations further revealed that the strong plasma-catalyst interaction facilitates CO2 activation pathways. This study demonstrates not only the durability of In-Zr composites but also highlights plasma modification as an effective strategy to design next-generation catalysts for plasma-assisted CO2 utilization. Meanwhile, the In-Zr catalyst successfully developed in this study, with its outstanding performance, stability and durability, is a highly promising candidate material for high-temperature industrial catalytic processes.
1. Introduction
In recent years, the persistent rise in carbon dioxide (CO2) emissions intensified concerns over global warming and climate risk, posing a major challenge to sustainable development.1–5 Meanwhile, the global transition from fossil-fuel-based systems to low-carbon and hydrogen-enabled energy technologies is accelerating.6–8 Therefore, converting CO2 into high-value products, such as CO, CH4, and CH3OH,9via carbon capture and utilization (CCU) is regarded as one of the most effective scientific strategies.10–13 Carbon monoxide (CO) possesses significant commercial value as a C1 resource, serving as a precursor to produce various value-added chemicals.10,14 Consequently, the direct conversion of CO2 into CO and O2 constitutes a significant industrial process.15–17| CO2 = CO + 1/2O2, ΔH = 283 kJ mol−1 | (1) |
There are several methods for CO2 conversion: pyrolysis, electrocatalysis, photocatalysis, and plasma catalysis. Nonetheless, due to the dissociation energy of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O being 5.5 eV, the pyrolysis process requires elevated temperatures approaching 2000 K to facilitate the breakdown of CO2. The catalysts employed in electrocatalytic and photocatalytic processes are often precious metal catalysts, which are relatively expensive. Conversely, non-thermal plasma technology (NTP) produces high-energy electrons ranging from 1 to 10 eV at ambient temperature and pressure,18–20 which can cleave the C
O being 5.5 eV, the pyrolysis process requires elevated temperatures approaching 2000 K to facilitate the breakdown of CO2. The catalysts employed in electrocatalytic and photocatalytic processes are often precious metal catalysts, which are relatively expensive. Conversely, non-thermal plasma technology (NTP) produces high-energy electrons ranging from 1 to 10 eV at ambient temperature and pressure,18–20 which can cleave the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond, thereby facilitating the decomposition and conversion of CO2. Thus, NTP is considered a viable alternative to conventional conversion methods. Common plasma generation methods include glow discharge, microwave discharge, and dielectric barrier discharge (DBD).21 Among them, DBD is more appropriate for CO2 conversion owing to its benefits of stable discharge, homogeneous gas distribution, and effective gas tightness.22–25
O bond, thereby facilitating the decomposition and conversion of CO2. Thus, NTP is considered a viable alternative to conventional conversion methods. Common plasma generation methods include glow discharge, microwave discharge, and dielectric barrier discharge (DBD).21 Among them, DBD is more appropriate for CO2 conversion owing to its benefits of stable discharge, homogeneous gas distribution, and effective gas tightness.22–25
The NTP technique for CO2 processing offers advantages such as (1) operation at ambient temperature and pressure, (2) reduced energy input requirements, and (3) enhanced CO selectivity. Nevertheless, the limited utilization of high-energy electrons produced by NTP prevents a single plasma from attaining high CO2 conversion efficiency. Consequently, the integration of plasma technology with catalysts to collaboratively facilitate the breakdown of CO2 is the primary research focus in this domain.26 In 2020,27 Ray et al. introduced a g-C3N4 – based catalyst into a DBD system and demonstrated that 5% ZnO + g-C3N4 enhanced the catalytic conversion of acidic gases, such as CO2, owing to the abundance of alkaline sites. In 2023,28 Rao et al. investigated the synergistic catalytic effects of several alkali metal oxides with DBD, revealing a trend of SrO > Al2O3 > MgO > CaO. In 2024,29 Hadi et al. confirmed that oxygen vacancies in metal oxides enhanced the catalytic breakdown of CO2.
Cubic-phase indium oxide (c-In2O3) is notable among many metal oxides that tend to produce oxygen vacancies, owing to its stable thermodynamic configuration and superior catalytic performance.30,31 We previously synthesized In2O3 nanocatalysts with elevated oxygen vacancies using chemical precipitation coupled with DBD technology and confirmed their efficacy in synergistically enhancing the catalytic conversion of CO2 when used with DBD.32,33 In2O3 has remarkable catalytic activity at lower temperatures (225 °C), but at elevated temperatures, it experiences a significant decrease in activity, resulting in catalyst deactivation.
To address the issues of short lifespan and thermal deactivation of In2O3 catalysts, our research team selected ZrO2 as a second component owing to its excellent thermal stability and catalytic potential, based on preliminary screening experiments. In this study, In2O3-ZrO2 composite catalysts with different indium–zirconium molar ratios were successfully prepared by combining chemical precipitation and DBD technology and placed in a DBD reactor to explore the effects of synergistic interactions between catalysts and DBD plasma on the catalytic conversion of CO2. It is important to promote the application of plasma catalysis technology in CO2 conversion. This work aims to promote the application of plasma-catalysis technology in CO2 conversion. The catalytic performances of In2O3, ZrO2 and In2O3-ZrO2 catalysts with different indium–zirconium ratios were compared in a DBD reactor. In addition, the composite catalysts were analyzed to gain insight into improving the reaction mechanism for the catalytic conversion of CO2 in the DBD reactor and the possible reasons for its high lifetime.
2. Experimental
2.1 Plasma reactor setup
Fig. 1 illustrates the interior configuration of the experimental apparatus and DBD reactor. The reaction gas utilized was high-purity CO2 (Shanxi Lizhe Gas Corporation, purity: 99.99%), with the flow rate regulated between 10–80 mL min−1 using a pressure regulator valve and a mass flow meter (ACU10FD-LC, produced by Beijing AccuFlow Technology Co., Ltd). After flow regulation, the gas was introduced at one end of the DBD reactor, traversed the plasma region to undergo the reaction, and subsequently, the reaction products were collected at the opposite end. The gaseous products of the reaction were analyzed using a GC9790 II gas chromatograph equipped with a thermal conductivity detector (TCD) to determine their composition after reaction. The experiment utilized a high-voltage AC power supply as the power source. The detailed setup is shown in Fig. 1a.Fig. 1b illustrates the internal structure of the DBD reactor. The primary structure of the DBD reactor comprises a quartz tube with an internal diameter of 15 mm. A stainless-steel rod, measuring 13 mm in diameter and 150 mm in length, is positioned along the axis within the quartz tube as the inner electrode. A 120-mm-long stainless-steel mesh was positioned around the outer surface of the quartz tube to serve as the outer electrode. The reactor's discharge gap measured 1 mm, while the discharge length was 120 mm. The catalyst was introduced into the DBD reactor via the incorporated powder catalyst (IPC) approach. The plasma power signal was measured using a TM10 oscilloscope (Tecman Electronic Instrument Holdings Limited).34
2.2 Catalyst preparation
The catalysts were synthesized using a chemical precipitation process integrated with DBD plasma technology. Using the catalyst with an In2O3-ZrO2 molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (designated as In-Zr (1
1 (designated as In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)) as a case study: initially, 1.86 g of indium nitrate and 1.07 g of zirconium nitrate were dissolved in 50 mL of deionized water to create the precursor solution, followed by the dissolution of sodium carbonate in deionized water to prepare a sodium carbonate solution at a concentration of 0.2 mol L−1. The catalyst was synthesized using chemical impregnation while stirring at 80 °C and 800 rpm, using a syringe pump to maintain a flow rate of 120 mL min−1. Subsequently, the sodium carbonate solution was gradually added to the precursor solution via a syringe pump at 120 mL min−1 under continuous stirring (80 °C, 800 rpm) until the pH reached 10. Following a three-hour aging period at 80 °C, the suspension was filtered, rinsed with hot deionized water, and then dried at 80 °C for 12 hours, followed by calcination in a muffle furnace (heating rate of 5 °C min−1, calcination temperature of 450 °C, duration of 3 hours). The resultant powder samples were positioned in a DBD reactor and treated in batches at 60 W for 20 minutes, ultimately yielding a light yellow powdered In-Zr (1
1)) as a case study: initially, 1.86 g of indium nitrate and 1.07 g of zirconium nitrate were dissolved in 50 mL of deionized water to create the precursor solution, followed by the dissolution of sodium carbonate in deionized water to prepare a sodium carbonate solution at a concentration of 0.2 mol L−1. The catalyst was synthesized using chemical impregnation while stirring at 80 °C and 800 rpm, using a syringe pump to maintain a flow rate of 120 mL min−1. Subsequently, the sodium carbonate solution was gradually added to the precursor solution via a syringe pump at 120 mL min−1 under continuous stirring (80 °C, 800 rpm) until the pH reached 10. Following a three-hour aging period at 80 °C, the suspension was filtered, rinsed with hot deionized water, and then dried at 80 °C for 12 hours, followed by calcination in a muffle furnace (heating rate of 5 °C min−1, calcination temperature of 450 °C, duration of 3 hours). The resultant powder samples were positioned in a DBD reactor and treated in batches at 60 W for 20 minutes, ultimately yielding a light yellow powdered In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst. The specific raw material dosages and reaction parameters of other catalysts prepared by the chemical precipitation method are shown in Table 1.
1) catalyst. The specific raw material dosages and reaction parameters of other catalysts prepared by the chemical precipitation method are shown in Table 1.
| Catalyst type | Dosage of In(NO3)3 | Dosage of Zr(NO3)4 | Stirring rate | Calcination conditions |
|---|---|---|---|---|
| In2O3 | 3.00 g | 0 g | 800 rpm | 450–5 °C min−1 |
In-Zr (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
2.61 g | 0.64 g | 800 rpm | 450–5 °C min−1 |
In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
1.86 g | 1.07 g | 800 rpm | 450–5 °C min−1 |
In-Zr (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) 7) |
1.12 g | 1.50 g | 800 rpm | 450–5 °C min−1 |
| ZrO2 | 0 g | 3.00 g | 800 rpm | 450–5 °C min−1 |
A mechanical grinding method was adopted to prepare the physically mixed composite catalyst. The experimental procedures are as follows: 2.77 g of In2O3 sample and 1.23 g of ZrO2 sample were dried and uniformly mixed, followed by grinding in a mortar for 10 minutes to ensure sufficient mixing. Finally, the target physically mixed composite catalyst was obtained.
2.3 Chromatographic analysis and parameter definition
The CO2 conversion rate, CO yield, and CO selectivity were defined as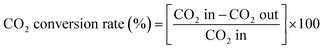 | (2) |
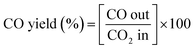 | (3) |
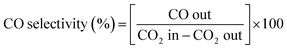 | (4) |
The specific input energy (SIE) is defined as.
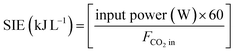 | (5) |
The energy efficacy of the reaction is defined as.
 | (6) |
The residence time of CO2 in the DBD device was determined by the following equation.
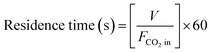 | (7) |
2.4 Catalyst characterization
The various c-catalyst samples were individually analyzed using X-ray diffraction (MiniFlex 600), and spectra were produced within the range of 2θ = 0°–90°. The microstructures of various catalysts were examined with a Hitachi SU8230 field emission scanning electron microscope and an FEI Tecnai F20 transmission electron microscope, while the elemental compositions of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts were analyzed using an X-Twin energy-dispersive X-ray spectrometer (EDS). The surface chemical compositions and oxidation states of the various catalysts were ascertained using X-ray photoelectron spectroscopy (Thermofisher ESCALAB Xi+). The quantity of bulk oxygen vacancies in the samples was assessed using electron paramagnetic resonance (EPR) spectroscopy (Bruker A300 Germany). A Shimadzu 3600-Plus UV-Vis spectrometer was employed to acquire diffuse reflectance UV-Vis spectra spanning 200–800 nm, and the bandgap energies were determined using the Tauc plot technique. Ultimately, CO2 adsorption and desorption studies were conducted utilizing a kinetic gas adsorption analyzer (Conta Autosorb-iQ) at 273 K, with CO2 as the adsorbate gas. All samples were degassed under vacuum at 120 °C for 7 hours prior to analysis. The surface area of each catalyst was determined using the BET (Brunauer–Emmett–Teller) technique, while the pore volume and pore size of each catalyst were measured from the BJH (Barrett–Joyner–Halenda) desorption curve.
1) catalysts were analyzed using an X-Twin energy-dispersive X-ray spectrometer (EDS). The surface chemical compositions and oxidation states of the various catalysts were ascertained using X-ray photoelectron spectroscopy (Thermofisher ESCALAB Xi+). The quantity of bulk oxygen vacancies in the samples was assessed using electron paramagnetic resonance (EPR) spectroscopy (Bruker A300 Germany). A Shimadzu 3600-Plus UV-Vis spectrometer was employed to acquire diffuse reflectance UV-Vis spectra spanning 200–800 nm, and the bandgap energies were determined using the Tauc plot technique. Ultimately, CO2 adsorption and desorption studies were conducted utilizing a kinetic gas adsorption analyzer (Conta Autosorb-iQ) at 273 K, with CO2 as the adsorbate gas. All samples were degassed under vacuum at 120 °C for 7 hours prior to analysis. The surface area of each catalyst was determined using the BET (Brunauer–Emmett–Teller) technique, while the pore volume and pore size of each catalyst were measured from the BJH (Barrett–Joyner–Halenda) desorption curve.
The specific parameters of the Density Functional Theory (DFT) simulation calculations are as follows:
Computational software and procedures: all first-principles calculations were performed using the Vienna Ab initio Simulation Package (VASP). Modelling and visualisation were conducted using Materials Studio and VESTA software.
Exchange-correlation functional: The Perdew–Burke–Ernzerhof (PBE) functional under the generalized gradient approximation (GGA) was employed.
Dispersion correction: to accurately describe intermolecular weak interactions, the DFT-D3 method was employed for dispersion correction.
Planar wave cutoff energy: set to 520 eV.
Electronic self-consistency iterations: energy convergence criterion set to EDIFF = 10−5 eV.
Atomic geometry optimisation: force convergence criterion on each atom set to EDIFFG = 0.02 × eV Å−1.
3. Results and discussion
3.1 Microscopic morphology of the catalyst
The surface morphology of In2O3, ZrO2, and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was characterized using scanning electron microscopy (SEM), as seen in Fig. 1c–e. Fig. 1c and d show the SEM images of In2O3 and ZrO2, respectively. In2O3 exists as nanoparticles with an average size of around 100 nm, while ZrO2 exhibits a distinct morphology characterized as ‘amorphous,’ with particle sizes ranging from 200 to 400 nm. The particle size ranges from 200 to 400 nanometers. Fig. 1e illustrates the surface morphology of In-Zr (1
1) was characterized using scanning electron microscopy (SEM), as seen in Fig. 1c–e. Fig. 1c and d show the SEM images of In2O3 and ZrO2, respectively. In2O3 exists as nanoparticles with an average size of around 100 nm, while ZrO2 exhibits a distinct morphology characterized as ‘amorphous,’ with particle sizes ranging from 200 to 400 nm. The particle size ranges from 200 to 400 nanometers. Fig. 1e illustrates the surface morphology of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite catalysts, revealing the nanoparticle morphology of In2O3 alongside the droplet-like structure of ZrO2. A distinct interface between the various morphologies is evident, signifying that the structure is heterogeneous, comprising In2O3 and ZrO2. The presence of multiple heterogeneous interfaces increases the exposure of active sites, thereby enhancing catalytic activity.
1) composite catalysts, revealing the nanoparticle morphology of In2O3 alongside the droplet-like structure of ZrO2. A distinct interface between the various morphologies is evident, signifying that the structure is heterogeneous, comprising In2O3 and ZrO2. The presence of multiple heterogeneous interfaces increases the exposure of active sites, thereby enhancing catalytic activity.
The catalysts' morphological characteristics were examined and analyzed by transmission electron microscopy. Fig. 1f presents the TEM image of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst, whereas Fig. 1h displays the HRTEM image corresponding to its designated location. The assessment of the catalysts through Fourier and inverse Fourier transformations (Fig. S1), along with lattice spacing measurements, indicates that the In-Zr (1
1) catalyst, whereas Fig. 1h displays the HRTEM image corresponding to its designated location. The assessment of the catalysts through Fourier and inverse Fourier transformations (Fig. S1), along with lattice spacing measurements, indicates that the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst displays two lattice fringes corresponding to the (111) crystallographic plane of ZrO2 and the (222) crystallographic plane of In2O3, thereby reaffirming the heterogeneous structure of the In-Zr (1
1) catalyst displays two lattice fringes corresponding to the (111) crystallographic plane of ZrO2 and the (222) crystallographic plane of In2O3, thereby reaffirming the heterogeneous structure of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst. Meanwhile, we have performed SAED analysis of the composite sample, with the results presented in Fig. S1. It can be seen that the diffraction pattern consists of diffraction rings rather than diffraction spots, indicating that this region has a polycrystalline structure. Additionally, through the analysis of these diffraction rings, they can be indexed to different crystal planes of In2O3 and ZrO2, which further confirms the component composition of the composite catalyst.
1) catalyst. Meanwhile, we have performed SAED analysis of the composite sample, with the results presented in Fig. S1. It can be seen that the diffraction pattern consists of diffraction rings rather than diffraction spots, indicating that this region has a polycrystalline structure. Additionally, through the analysis of these diffraction rings, they can be indexed to different crystal planes of In2O3 and ZrO2, which further confirms the component composition of the composite catalyst.
Fig. 1i presents the HAADF-STEM and elemental mapping images of the catalysts, accompanied by energy dispersive X-ray spectroscopy (EDS) and elemental analysis (Fig. 1g and S2). Fig. 1e illustrates that, among the three constituents In, Zr, and O in the catalyst, the O element predominates, while the molar ratio of In to Zr is approximately 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. This observation aligns with the design of the composite catalyst, In-Zr (1
1. This observation aligns with the design of the composite catalyst, In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), where the molar ratio of In2O3 to ZrO2 is also 1
1), where the molar ratio of In2O3 to ZrO2 is also 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The elemental mapping image (Fig. 1g) reveals a homogeneous distribution of the elements In, Zr, and O throughout the catalyst. This confirms that the In-Zr (1
1. The elemental mapping image (Fig. 1g) reveals a homogeneous distribution of the elements In, Zr, and O throughout the catalyst. This confirms that the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst consists of a heterogeneous composition, with ZrO2 and In2O3 interwoven. This heterogeneous structure enhances catalytic activity, while the interaction of the dioxide phases improves the overall performance of the catalyst through synergistic effects.
1) catalyst consists of a heterogeneous composition, with ZrO2 and In2O3 interwoven. This heterogeneous structure enhances catalytic activity, while the interaction of the dioxide phases improves the overall performance of the catalyst through synergistic effects.
We also conducted SEM and TEM characterizations on the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite catalyst after DBD treatment, and the detailed results are presented in Fig. S3. A comparison of the SEM images before and after treatment reveals that the catalyst's overall morphology, particle size, and distribution remained consistent. Low-magnification TEM images further confirm that DBD treatment did not cause obvious sintering, fragmentation, or agglomeration of the particles. To investigate potential effects on the crystal structure, we conducted HRTEM analysis. It can be observed that the clear lattice fringes in the catalyst were completely retained after treatment. These data strongly indicate that the DBD plasma treatment we employed is a mild and highly selective surface modification technique. This technique enables precise regulation of the catalyst's surface properties without altering its bulk morphology or crystal structure.
1) composite catalyst after DBD treatment, and the detailed results are presented in Fig. S3. A comparison of the SEM images before and after treatment reveals that the catalyst's overall morphology, particle size, and distribution remained consistent. Low-magnification TEM images further confirm that DBD treatment did not cause obvious sintering, fragmentation, or agglomeration of the particles. To investigate potential effects on the crystal structure, we conducted HRTEM analysis. It can be observed that the clear lattice fringes in the catalyst were completely retained after treatment. These data strongly indicate that the DBD plasma treatment we employed is a mild and highly selective surface modification technique. This technique enables precise regulation of the catalyst's surface properties without altering its bulk morphology or crystal structure.
3.2 Catalytic activity evaluation
To validate the catalytic conversion of CO2 by various catalysts in the DBD reactor, 0.2 g of each catalyst was introduced into the DBD reactor using the IPC method. The CO2 inlet flow rate was maintained at 20 mL min−1, while the specific energy input (SIE) of the system was regulated by modulating the input power. The test outcomes are illustrated in Fig. 2. Fig. 2a and b illustrate that an increase in the specific energy input of the DBD system resulted in a substantial enhancement in CO2 conversion and CO production. This occurs because, at elevated SIE, the plasma generates more energetic electrons, thereby increasing the collision likelihood between CO2 and these electrons, ultimately improving CO2 conversion.35 The catalyst-filled DBD exhibits a substantial enhancement in CO2 conversion and CO yield relative to the catalyst-free DBD. This results from the synergistic interaction between the catalyst and plasma discharge in CO2 conversion.19,36 The total CO2 conversion efficacy follows the sequence: In2O3 > In-Zr (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) > In-Zr (1
3) > In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) > In-Zr (3
1) > In-Zr (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) > ZrO2. From this, In2O3 demonstrates a greater contribution to CO2 conversion than ZrO2. Fig. 2d illustrates that the maximum CO yield is achieved in the DBD system containing In-Zr in a 1
7) > ZrO2. From this, In2O3 demonstrates a greater contribution to CO2 conversion than ZrO2. Fig. 2d illustrates that the maximum CO yield is achieved in the DBD system containing In-Zr in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. In conjunction with the CO selectivity illustrated in Fig. 2c, it is evident that the CO selectivity of the DBD reactors diminished to varied extents following the introduction of the catalysts, although the CO selectivity of the In-Zr (1
1 ratio. In conjunction with the CO selectivity illustrated in Fig. 2c, it is evident that the CO selectivity of the DBD reactors diminished to varied extents following the introduction of the catalysts, although the CO selectivity of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) synergistic DBD consistently remained elevated (>90%). This explains the high CO yield observed with the In-Zr (1
1) synergistic DBD consistently remained elevated (>90%). This explains the high CO yield observed with the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) DBD system at increased SIE. The In-Zr (1
1) DBD system at increased SIE. The In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst therefore selectively promotes the conversion of CO2 to CO within the DBD reactor.
1) catalyst therefore selectively promotes the conversion of CO2 to CO within the DBD reactor.
Fig. 2d illustrates the comparative energy efficiency of several catalysts over different SIEs. The energy efficiency of DBD systems containing catalysts surpasses those of the empty DBDs, indicating that the presence of catalysts enhances the utilization of energy for CO2 conversion rather than for heat dissipation.
The catalyst's stability is similarly crucial in practical applications. In the operation of DBD, the gas temperature within the system rises with increasing input power37 (Fig. S4), and excessive temperatures might adversely influence the catalytic activity of the catalyst. To assess the thermal stability of several catalysts, 0.2 g of each catalyst was introduced into the DBD reactor using the IPC technique and evaluated at an input power of 80 W (SIE = 237 kJ L−1, T = 251.8 °C). The findings are presented in Fig. 2e. During the early phase of the reaction, the CO2 conversion in all reactors progressively rose over time, indicating that the catalyst is undergoing activation. At 50 minutes, the CO2 conversion in the DBD reactor containing In2O3 and In-Zr (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) began to decrease gradually, ultimately stabilizing at approximately 16%, which is lower than that of the catalyst-free DBD. In contrast, the CO2 conversion in the DBD reactor filled with In-Zr (1
3) began to decrease gradually, ultimately stabilizing at approximately 16%, which is lower than that of the catalyst-free DBD. In contrast, the CO2 conversion in the DBD reactor filled with In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), In-Zr (3
1), In-Zr (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and ZrO2 catalysts continued to rise, eventually stabilizing at a level exceeding that of the catalyst-free DBD. The likely explanation for this behaviour is that ZrO2 possesses significant thermal stability and its physicochemical properties remain stable at elevated temperatures, while the incorporation of ZrO2 helps to preserve the catalytic activity of the catalysts under high-temperature conditions.
7), and ZrO2 catalysts continued to rise, eventually stabilizing at a level exceeding that of the catalyst-free DBD. The likely explanation for this behaviour is that ZrO2 possesses significant thermal stability and its physicochemical properties remain stable at elevated temperatures, while the incorporation of ZrO2 helps to preserve the catalytic activity of the catalysts under high-temperature conditions.
To investigate the service life of the composite catalyst, 0.2 g of In2O3 catalyst, ZrO2 catalyst and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst, identified as the most effective among the composite catalysts, was introduced into a DBD reactor via the IPC method. The service life of the catalyst was measured under the condition of SIE = 104 kJ L−1, where the system energy efficiency reaches its maximum. The results are shown in Fig. 2f. During the initial 0–80 minutes, the In2O3 catalyst undergoes an activation phase, during which the conversion of CO2 progressively rises over time. During the 80–180 minute interval, the In2O3 catalyst remains in the stabilization phase, exhibiting negligible variation in its CO2 conversion rate. After 180 minutes, the CO2 conversion of In2O3 starts to decline and ultimately drops below that of the empty-loaded DBD. After activation, the ZrO2 catalyst can also maintain a high CO2 conversion rate. Although slightly lower than that of the In-Zr (1
1) catalyst, identified as the most effective among the composite catalysts, was introduced into a DBD reactor via the IPC method. The service life of the catalyst was measured under the condition of SIE = 104 kJ L−1, where the system energy efficiency reaches its maximum. The results are shown in Fig. 2f. During the initial 0–80 minutes, the In2O3 catalyst undergoes an activation phase, during which the conversion of CO2 progressively rises over time. During the 80–180 minute interval, the In2O3 catalyst remains in the stabilization phase, exhibiting negligible variation in its CO2 conversion rate. After 180 minutes, the CO2 conversion of In2O3 starts to decline and ultimately drops below that of the empty-loaded DBD. After activation, the ZrO2 catalyst can also maintain a high CO2 conversion rate. Although slightly lower than that of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite catalyst, no obvious downward trend is observed, demonstrating excellent stability. In comparison, the activation duration of the In-Zr (1
1) composite catalyst, no obvious downward trend is observed, demonstrating excellent stability. In comparison, the activation duration of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst significantly exceeds that of the pure In2O3 catalyst, with the former exhibiting an activation period of 0–150 minutes and consistently achieving superior CO2 conversion rates during this timeframe. During the 150 to 450 minute interval, the In-Zr (1
1) catalyst significantly exceeds that of the pure In2O3 catalyst, with the former exhibiting an activation period of 0–150 minutes and consistently achieving superior CO2 conversion rates during this timeframe. During the 150 to 450 minute interval, the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst exhibited stability, with CO2 conversion remaining constant across time and no observable deactivation phase. This signifies that the catalyst's lifespan was significantly enhanced with the incorporation of ZrO2.
1) catalyst exhibited stability, with CO2 conversion remaining constant across time and no observable deactivation phase. This signifies that the catalyst's lifespan was significantly enhanced with the incorporation of ZrO2.
Based on these findings, it was determined that the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst exhibited not only elevated CO2 conversion and CO yield but also the capability to selectively break down CO2 into CO, with superior thermal stability and longevity compared to the pure In2O3 catalyst. Consequently, the ensuing composite catalysts were investigated utilizing In-Zr (1
1) catalyst exhibited not only elevated CO2 conversion and CO yield but also the capability to selectively break down CO2 into CO, with superior thermal stability and longevity compared to the pure In2O3 catalyst. Consequently, the ensuing composite catalysts were investigated utilizing In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts.
1) catalysts.
3.3 The effect of SIE and residence time
In order to investigate the effect of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst dosage on the catalytic conversion performance of CO2, different dosages of In-Zr (1
1) catalyst dosage on the catalytic conversion performance of CO2, different dosages of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst were placed in the DBD reactor using the IPC method, and the flow rate of CO2 gas inlet was controlled at 20 mL min−1, while the tests were carried out at different SIEs, and the results of the experiments are shown in Fig. 3a–d. Fig. 3a–c show the CO2 conversion, CO yield and energy efficiency at different In-Zr (1
1) catalyst were placed in the DBD reactor using the IPC method, and the flow rate of CO2 gas inlet was controlled at 20 mL min−1, while the tests were carried out at different SIEs, and the results of the experiments are shown in Fig. 3a–d. Fig. 3a–c show the CO2 conversion, CO yield and energy efficiency at different In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) dosages, respectively. At low SIE, the CO2 conversion, CO yield and energy efficiency of the catalyst-filled DBD are higher than those of the catalyst-free DBD due to the synergistic effect between the catalyst and the plasma, which promotes the conversion of CO2. However, the CO2 conversion of the DBD reactors filled with 0.3 g and 0.4 g of In-Zr (1
1) dosages, respectively. At low SIE, the CO2 conversion, CO yield and energy efficiency of the catalyst-filled DBD are higher than those of the catalyst-free DBD due to the synergistic effect between the catalyst and the plasma, which promotes the conversion of CO2. However, the CO2 conversion of the DBD reactors filled with 0.3 g and 0.4 g of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was ultimately lower than that of the catalyst-free DBD reactor at high SIE. This is due to the stacking of excess catalysts in the reactor, which makes it difficult for the catalyst to fully contact CO2 and reduces its utilization rate, while a large amount of catalyst stacking hinders the plasma discharge, which ultimately leads to a decrease in the CO2 conversion performance. Although the CO2 conversion of 0.1 g In-Zr (1
1) was ultimately lower than that of the catalyst-free DBD reactor at high SIE. This is due to the stacking of excess catalysts in the reactor, which makes it difficult for the catalyst to fully contact CO2 and reduces its utilization rate, while a large amount of catalyst stacking hinders the plasma discharge, which ultimately leads to a decrease in the CO2 conversion performance. Although the CO2 conversion of 0.1 g In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at high SIE was slightly higher than that of empty DBD, its CO yield at high SIE was lower than that of empty DBD, which also matched with the lower CO selectivity of 0.1 g catalyst at high SIE (Fig. 3d). The possible reason for this situation is that the catalyst dosage is too low, and the catalysis is not sufficient. The overall trend of catalyst dosage is 0.2 g > 0.1 g > 0.3 g > 0.4 g. This indicates that excess catalysts tend to accumulate in the DBD reactor, which not only has a low utilization rate but also affects the plasma discharge, while insufficient dosage may lead to reduced catalytic activity. Therefore, the amount of catalyst needs to be strictly controlled.
1) at high SIE was slightly higher than that of empty DBD, its CO yield at high SIE was lower than that of empty DBD, which also matched with the lower CO selectivity of 0.1 g catalyst at high SIE (Fig. 3d). The possible reason for this situation is that the catalyst dosage is too low, and the catalysis is not sufficient. The overall trend of catalyst dosage is 0.2 g > 0.1 g > 0.3 g > 0.4 g. This indicates that excess catalysts tend to accumulate in the DBD reactor, which not only has a low utilization rate but also affects the plasma discharge, while insufficient dosage may lead to reduced catalytic activity. Therefore, the amount of catalyst needs to be strictly controlled.
The residence time of CO2 gas in the DBD system influences its overall conversion efficiency. The residence time is determined using eqn (7), and the test results are illustrated in Fig. 3e and f. The CO2 conversion rate rises with an increase in residence duration, irrespective of the presence of a catalyst (Fig. 3e). The duration of CO2 gas presence in the discharge zone correlates positively with the likelihood of it being struck by energetic electrons. The CO2 conversion in the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) system exceeds that of the DBD reactor without catalyst at extended residence times, whereas the CO2 conversion in the catalyst-free DBD exceeds that of the catalyst-filled DBD at shorter residence times. This is likely because the catalyst needs sufficient time to absorb CO2; an inadequate residence time hinders CO2 from adhering to the catalyst's surface, thus limiting its catalytic effect. The catalyst may also influence plasma discharge, resulting in reduced CO2 conversion. Fig. 3f illustrates the correlation between energy efficiency and varying residence times, revealing a declining trend in energy efficiency as residence time increases. This phenomenon occurs because prolonged CO2 presence in the system facilitates its heating by high-energy electrons. A greater amount of energy is expended as heat, leading to reduced energy efficiency. The energy efficiency of the reactor containing In-Zr (1
1) system exceeds that of the DBD reactor without catalyst at extended residence times, whereas the CO2 conversion in the catalyst-free DBD exceeds that of the catalyst-filled DBD at shorter residence times. This is likely because the catalyst needs sufficient time to absorb CO2; an inadequate residence time hinders CO2 from adhering to the catalyst's surface, thus limiting its catalytic effect. The catalyst may also influence plasma discharge, resulting in reduced CO2 conversion. Fig. 3f illustrates the correlation between energy efficiency and varying residence times, revealing a declining trend in energy efficiency as residence time increases. This phenomenon occurs because prolonged CO2 presence in the system facilitates its heating by high-energy electrons. A greater amount of energy is expended as heat, leading to reduced energy efficiency. The energy efficiency of the reactor containing In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was significantly enhanced at the longest residence time of 22.9 seconds, suggesting that CO2 and In-Zr (1
1) was significantly enhanced at the longest residence time of 22.9 seconds, suggesting that CO2 and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) achieved optimal contact during this duration, allowing high-energy electrons to facilitate CO2 conversion rather than dissipating energy as heat.
1) achieved optimal contact during this duration, allowing high-energy electrons to facilitate CO2 conversion rather than dissipating energy as heat.
3.4 Mechanistic analysis
To examine the catalytic mechanisms of the catalysts In2O3, ZrO2, In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), and In-Zr (1
1), and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD (the latter being derived from In-Zr (1
1)-DBD (the latter being derived from In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts treated for 20 minutes in a DBD reactor under an air atmosphere at 160 V), XRD, XPS, EPR, and BET characterization tests were conducted. The findings are illustrated in Fig. 4. Fig. 4a presents the XRD diffractograms of several catalysts, demonstrating that the In2O3 and ZrO2 samples align well with their respective PDF cards. In the In-Zr (1
1) catalysts treated for 20 minutes in a DBD reactor under an air atmosphere at 160 V), XRD, XPS, EPR, and BET characterization tests were conducted. The findings are illustrated in Fig. 4. Fig. 4a presents the XRD diffractograms of several catalysts, demonstrating that the In2O3 and ZrO2 samples align well with their respective PDF cards. In the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composition, the typical peaks of the ZrO2 sample are largely aligned, and the distinctive peaks of the In2O3 (211) crystal plane emerge at a 2θ diffraction angle of 21.5°.38 This demonstrates that the In-Zr (1
1) composition, the typical peaks of the ZrO2 sample are largely aligned, and the distinctive peaks of the In2O3 (211) crystal plane emerge at a 2θ diffraction angle of 21.5°.38 This demonstrates that the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite catalyst was effectively synthesized. In contrast, the comparison of the In-Zr (1
1) composite catalyst was effectively synthesized. In contrast, the comparison of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts prior to and following DBD treatment did not provide a discernible change, thus confirming that the DBD treatment does not influence the crystallinity of the catalysts.
1) catalysts prior to and following DBD treatment did not provide a discernible change, thus confirming that the DBD treatment does not influence the crystallinity of the catalysts.
Fig. 4b–g presents the XPS test results for several catalysts. Fig. 4b and c present the characterization test results for In 3d and Zr 3d, respectively. Notably, binding energies of 452.4 eV and 444.8 eV correspond to In3d3/2 and In3d5/2, while binding energies of 183.5 eV and 181.1 eV correspond to Zr3d3/2 and Zr3d5/2. This observation is attributed to the characteristic spin–orbit splitting of In3+ and Zr4+ ions. Comparing In2O3 and ZrO2, both In3d3/2 and In3d5/2 in In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts shift to lower binding energies, whereas Zr3d3/2 and Zr3d5/2 shift to higher binding energies. This indicates an electron transfer from Zr4+ to In3+ during the formation of In-Zr (1
1) catalysts shift to lower binding energies, whereas Zr3d3/2 and Zr3d5/2 shift to higher binding energies. This indicates an electron transfer from Zr4+ to In3+ during the formation of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalysts.39 This indicates that the In-Zr composite catalyst is not a simple physical mixture, but rather that chemical interactions occur between In2O3 and ZrO2. To verify that such interactions are conducive to enhancing its catalytic performance, a physically mixed In-Zr composite catalyst was prepared and subjected to a series of tests and characterizations, with the results presented in Fig. S5. As can be seen from the XRD and TEM patterns in Fig. S5, the physically mixed In-Zr composite catalyst was successfully synthesized. Meanwhile, in comparison with the physically mixed In-Zr composite catalyst, the chemically combined In-Zr composite catalyst exhibits more oxygen vacancies, stronger CO2 catalytic conversion capability, and superior thermal stability. All the above research results confirm that the interactions between In2O3 and ZrO2 in the In-Zr composite catalyst have improved its catalytic conversion performance. In Fig. 4b and c, comparing In-Zr (1
1) catalysts.39 This indicates that the In-Zr composite catalyst is not a simple physical mixture, but rather that chemical interactions occur between In2O3 and ZrO2. To verify that such interactions are conducive to enhancing its catalytic performance, a physically mixed In-Zr composite catalyst was prepared and subjected to a series of tests and characterizations, with the results presented in Fig. S5. As can be seen from the XRD and TEM patterns in Fig. S5, the physically mixed In-Zr composite catalyst was successfully synthesized. Meanwhile, in comparison with the physically mixed In-Zr composite catalyst, the chemically combined In-Zr composite catalyst exhibits more oxygen vacancies, stronger CO2 catalytic conversion capability, and superior thermal stability. All the above research results confirm that the interactions between In2O3 and ZrO2 in the In-Zr composite catalyst have improved its catalytic conversion performance. In Fig. 4b and c, comparing In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) with In-Zr (1
1) with In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD reveals no significant alteration in the binding energy of either In3+ or Zr4+, indicating that the DBD treatment during catalyst synthesis does not influence the electronic states of In and Zr species.
1)-DBD reveals no significant alteration in the binding energy of either In3+ or Zr4+, indicating that the DBD treatment during catalyst synthesis does not influence the electronic states of In and Zr species.
Fig. 4d–g shows the O 1s spectra of the catalysts, indicating that oxygen exists in three forms for each catalyst. The species with a binding energy of around 530 eV corresponds to lattice oxygen (Olattice), the peak near 531 eV is attributed to surface oxygen vacancies (SOV), and the peak exceeding 532 eV is associated with the adsorbed oxygen (Oads)40 on the catalyst surface. ZrO2 exhibits a greater number of oxygen defects than In2O3, indicating its enhanced propensity for oxygen vacancy formation. The proportion of oxygen defects in the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composition is lower than that in ZrO2 but significantly higher than in In2O3, suggesting that the incorporation of ZrO2 promotes the development of oxygen vacancies in the catalyst. Comparing the catalysts pre- and post-DBD treatment, the fraction of O-defects is significantly increased, indicating that DBD treatment promotes the creation of oxygen vacancies in the catalysts. XPS primarily indicates the existence of surface oxygen vacancies; hence, the formation of bulk oxygen vacancies (BOV) resulting from the migration of oxygen vacancies to the interior is not detectable in the XPS spectra.41 To examine the potential migration of surface oxygen vacancies inside the catalysts, electron paramagnetic resonance spectroscopy (EPR) was employed for analysis, with the findings presented in Fig. 4h. The results indicate that both catalyst types exhibit distinctive signals associated with bulk oxygen vacancies at g = 2.003.42Table 2 presents the quantitative data on oxygen vacancies, illustrating the general findings. The order of oxygen vacancy concentration is In-Zr (1
1) composition is lower than that in ZrO2 but significantly higher than in In2O3, suggesting that the incorporation of ZrO2 promotes the development of oxygen vacancies in the catalyst. Comparing the catalysts pre- and post-DBD treatment, the fraction of O-defects is significantly increased, indicating that DBD treatment promotes the creation of oxygen vacancies in the catalysts. XPS primarily indicates the existence of surface oxygen vacancies; hence, the formation of bulk oxygen vacancies (BOV) resulting from the migration of oxygen vacancies to the interior is not detectable in the XPS spectra.41 To examine the potential migration of surface oxygen vacancies inside the catalysts, electron paramagnetic resonance spectroscopy (EPR) was employed for analysis, with the findings presented in Fig. 4h. The results indicate that both catalyst types exhibit distinctive signals associated with bulk oxygen vacancies at g = 2.003.42Table 2 presents the quantitative data on oxygen vacancies, illustrating the general findings. The order of oxygen vacancy concentration is In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD > ZrO2 > In-Zr (1
1)-DBD > ZrO2 > In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) > In2O3, with the highest concentration of oxygen vacancies, 1.69 × 1013, seen in the In-Zr (1
1) > In2O3, with the highest concentration of oxygen vacancies, 1.69 × 1013, seen in the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD catalysts. This may result from the DBD treatment, where high-energy electrons produced by the plasma impact the catalyst,41,43 leading to the removal of oxygen from its surface to create oxygen vacancies, while some surface oxygen vacancies migrate inward to form bulk oxygen vacancies. The presence of oxygen vacancies significantly enhances the catalytic conversion of CO2,44 elucidating the high efficiency of the In-Zr (1
1)-DBD catalysts. This may result from the DBD treatment, where high-energy electrons produced by the plasma impact the catalyst,41,43 leading to the removal of oxygen from its surface to create oxygen vacancies, while some surface oxygen vacancies migrate inward to form bulk oxygen vacancies. The presence of oxygen vacancies significantly enhances the catalytic conversion of CO2,44 elucidating the high efficiency of the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD catalysts in this process. In addition, the overall performance of DBD-assisted catalysis is excellent.28
1)-DBD catalysts in this process. In addition, the overall performance of DBD-assisted catalysis is excellent.28
| BOV (spins per g) | Average pore width (nm) | Pore volume (cm3 g−1) | Surface area (m2 g−1) | CO2 adsorption (cm3 g−1) | E g (eV) | |
|---|---|---|---|---|---|---|
| In2O3 | 1.254 × 1013 | 0.627 | 0.0129 | 50.154 | 5.68 | 2.7 |
| ZrO2 | 1.612 × 1013 | 0.573 | 0.0144 | 47.716 | 4.57 | 4.58 |
In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
1.289 × 1013 | 0.573 | 0.0176 | 54.694 | 5.54 | 2.58 |
In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD 1)-DBD |
1.69 × 1013 | 0.6 | 0.0181 | 56.689 | 5.86 | 2.49 |
Fig. 4i–k illustrate the pore size distribution, cumulative pore volume, and specific surface area plots of several catalysts, respectively. Fig. 4i illustrates the pore size distribution of several catalysts. Additionally, in combination with Table 2, it is observed that the average pore size of In2O3 is around 0.627 nm, while both ZrO2 and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) exhibit an average pore size of 0.573 nm, and In-Zr (1
1) exhibit an average pore size of 0.573 nm, and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD has an average pore size of 0.6 nm. All catalysts possess pore diameters in the microporous range, exceeding the kinetic diameter of the CO2 molecule (0.48 nm), hence confirming their capacity to collect CO2. While smaller pore sizes are unfavorable for CO2 adsorption, excessively large pore sizes lead to the desorption of CO2, making it easily dislodged. Therefore, In-Zr (1
1)-DBD has an average pore size of 0.6 nm. All catalysts possess pore diameters in the microporous range, exceeding the kinetic diameter of the CO2 molecule (0.48 nm), hence confirming their capacity to collect CO2. While smaller pore sizes are unfavorable for CO2 adsorption, excessively large pore sizes lead to the desorption of CO2, making it easily dislodged. Therefore, In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD, characterized by moderate pore sizes, is most conducive to CO2 adsorption, corroborating the data presented in Table 2 and Fig. S6. Fig. 4j and k illustrate the cumulative pore volume and specific surface area plots of various catalysts, respectively. When considered alongside Table 2, it is evident that both the pore volume and surface area of the composite catalysts exceed those of the pure catalysts. This enhancement can be ascribed to the heterogeneous structure of the composite catalysts, which induces surface defects, thereby augmenting pore volume and specific surface area.45 The In-Zr (1
1)-DBD, characterized by moderate pore sizes, is most conducive to CO2 adsorption, corroborating the data presented in Table 2 and Fig. S6. Fig. 4j and k illustrate the cumulative pore volume and specific surface area plots of various catalysts, respectively. When considered alongside Table 2, it is evident that both the pore volume and surface area of the composite catalysts exceed those of the pure catalysts. This enhancement can be ascribed to the heterogeneous structure of the composite catalysts, which induces surface defects, thereby augmenting pore volume and specific surface area.45 The In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD catalyst exhibited the greatest pore volume of 0.181 cm3 g−1 and the highest specific surface area of 56.689 m2 g−1. This is likely due to a substantial quantity of high-energy electrons produced by the DBD impacting the catalyst surface, resulting in an increased number of defects on the catalyst surface. The combination of elevated pore volume and increased specific surface area enhances CO2 capture by the catalysts, elucidating why the peak CO2 adsorption (5.86 cm3 g−1) in Table 2 and Fig. S6 is seen in In-Zr (1
1)-DBD catalyst exhibited the greatest pore volume of 0.181 cm3 g−1 and the highest specific surface area of 56.689 m2 g−1. This is likely due to a substantial quantity of high-energy electrons produced by the DBD impacting the catalyst surface, resulting in an increased number of defects on the catalyst surface. The combination of elevated pore volume and increased specific surface area enhances CO2 capture by the catalysts, elucidating why the peak CO2 adsorption (5.86 cm3 g−1) in Table 2 and Fig. S6 is seen in In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD. Meanwhile, in Fig. S6, the incomplete closure of the desorption curve indicates strong adsorption of CO2 by the catalyst under the tested conditions, correlating with its excellent adsorption performance.
1)-DBD. Meanwhile, in Fig. S6, the incomplete closure of the desorption curve indicates strong adsorption of CO2 by the catalyst under the tested conditions, correlating with its excellent adsorption performance.
Both In2O3 and ZrO2 are classified as semiconductor materials. The forbidden bandwidth (Eg) is a critical performance metric for semiconductors. To investigate the Eg of various catalysts, UV reflectance spectra were transformed over the range of 200–800 nm to establish the correlation between (αhv)2 and the optical energy (hv). The bandgap value was determined from the intersection of the tangent line with the horizontal axis,46 with results presented in Fig. S7 and Table 2. The bandgap of In2O3 is 2.7 eV, that of ZrO2 is 4.58 eV, and that of the composite catalyst In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) is 2.58 eV. The likely cause of this scenario is that the composite material has a greater number of defects (e.g., oxygen vacancies), which possess energy levels that reduce the energy gap between the valence and conduction bands. Conversely, In-Zr (1
1) is 2.58 eV. The likely cause of this scenario is that the composite material has a greater number of defects (e.g., oxygen vacancies), which possess energy levels that reduce the energy gap between the valence and conduction bands. Conversely, In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)-DBD exhibits the lowest band gap, Eg = 2.49 eV, indicating that the bombardment of the catalyst by high-energy electrons produced by the plasma has induced a greater number of defects within the catalyst. This facilitates electron transitions from the valence band to the conduction band, thereby augmenting the catalyst's electrical conductivity.47 The reduced band gap further implies an increased susceptibility to excitation by the plasma, both of which enhance the catalyst's efficacy in CO2 reduction.
1)-DBD exhibits the lowest band gap, Eg = 2.49 eV, indicating that the bombardment of the catalyst by high-energy electrons produced by the plasma has induced a greater number of defects within the catalyst. This facilitates electron transitions from the valence band to the conduction band, thereby augmenting the catalyst's electrical conductivity.47 The reduced band gap further implies an increased susceptibility to excitation by the plasma, both of which enhance the catalyst's efficacy in CO2 reduction.
3.5 Density functional theory (DFT) simulations
To further elucidate the underlying mechanism, DFT simulations were conducted using the VASP program. Based on the XRD analysis results, the oxygen vacancy formation energies and CO2 adsorption energies corresponding to the crystal planes of the strongest peaks and second-strongest peaks of In2O3, ZrO2, and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were calculated. Meanwhile, the structural schematics of CO2 adsorption on different crystal planes were simulated, charge distribution of CO2 adsorbed on the heterojunction surface of In-Zr composite catalysts with and without oxygen vacancies, as shown in Fig. 5, and the specific calculation results are presented in Table 3. The equations used to calculate the oxygen vacancy formation energy (Evac), the CO2 adsorption energy (Eads) and the charge density (Δρ) were as follows.
1) were calculated. Meanwhile, the structural schematics of CO2 adsorption on different crystal planes were simulated, charge distribution of CO2 adsorbed on the heterojunction surface of In-Zr composite catalysts with and without oxygen vacancies, as shown in Fig. 5, and the specific calculation results are presented in Table 3. The equations used to calculate the oxygen vacancy formation energy (Evac), the CO2 adsorption energy (Eads) and the charge density (Δρ) were as follows.| Evac = ED + 1/2EO2 − EP | (8) |
| Eads = E(slab-CO2) − Eslab − ECO2 | (9) |
| Δρ = ρtotal − ρsubstrate − ρadsorbate | (10) |
 | ||
Fig. 5 Schematic structure of CO2 adsorption on different crystalline surfaces of In2O3, ZrO2 and In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). 1). | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)
| In2O3 (222) | In2O3 (440)\ | ZrO2 (220) | ZrO2 (111) | In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) In2O3 face 1) In2O3 face |
In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) ZrO2 face 1) ZrO2 face |
|
|---|---|---|---|---|---|---|
| E O2 | −9.893 | |||||
| E CO2 | −22.95 | |||||
| E D | −841.839 | −425.727 | −651.834 | −8783787 | −298.191 | −297.414 |
| E P | −849.335 | −433.285 | 656.006 | −883.475 | −303.285 | −304.541 |
| E CO2-slab | −866.13 | −450.104 | −676.709 | −903.519 | −323.524 | −323.113 |
| E vac | 2.549 | 2.611 | −0.774 | −0.259 | 0.148 | 2.181 |
| E ads | −1.341 | −1.427 | −1.925 | −1.782 | −2.383 | −2.749 |
According to the data in Table 3, it can be seen that Evac follows the trend of ZrO2 < In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) < In2O3. In general, smaller oxygen vacancy formation energy indicates that it is easier to form oxygen vacancies,48 from which it can be inferred that the oxygen vacancy content roughly follows the trend of ZrO2 > In-Zr (1
1) < In2O3. In general, smaller oxygen vacancy formation energy indicates that it is easier to form oxygen vacancies,48 from which it can be inferred that the oxygen vacancy content roughly follows the trend of ZrO2 > In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) > In2O3. In addition, the Eads was −2.749 eV at the ZrO2 end face and −2.383 eV at the In2O3 end face, which were much smaller than those of the pure In2O3 and ZrO2, suggesting that compared to the In2O3 and ZrO2, the composite In-Zr (1
1) > In2O3. In addition, the Eads was −2.749 eV at the ZrO2 end face and −2.383 eV at the In2O3 end face, which were much smaller than those of the pure In2O3 and ZrO2, suggesting that compared to the In2O3 and ZrO2, the composite In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst was more favorable for the thermodynamics of the CO2 adsorption.49 In-Zr (1
1) catalyst was more favorable for the thermodynamics of the CO2 adsorption.49 In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) not only exhibits a lower oxygen vacancy formation energy but also has the lowest CO2 adsorption energy. Although the actual formation of oxygen vacancy and CO2 adsorption is influenced by external kinetic factors and sample morphology, these results still explain the excellent CO2 catalytic conversion performance of In-Zr (1
1) not only exhibits a lower oxygen vacancy formation energy but also has the lowest CO2 adsorption energy. Although the actual formation of oxygen vacancy and CO2 adsorption is influenced by external kinetic factors and sample morphology, these results still explain the excellent CO2 catalytic conversion performance of In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite catalysts.
1) composite catalysts.
The differential charge density distributions clearly show distinct charge-transfer behaviors for the two systems. On the pristine In2O3-ZrO2 surface (Fig. 5), only limited electron accumulation is observed around the adsorbed CO2 molecule, corresponding to a small electron transfer of 0.171|e|. In contrast, the introduction of an oxygen vacancy leads to a much stronger electronic interaction between CO2 and the catalyst surface (Fig. 5), resulting in a substantial electron transfer of 0.875|e|. This pronounced charge enrichment on the defective surface indicates that the oxygen vacancy greatly enhances the electron donation ability of the catalyst, thereby strengthening the activation of the adsorbed CO2. The results demonstrate that oxygen vacancies play a critical role in strengthening electron donation to CO2, thereby boosting its activation and improving the overall catalytic performance of the In2O3-ZrO2 heterojunction.
3.6 Evaluation of catalytic efficiency
Fig. 6 illustrates a comparison between the present study and previous studies conducted over the past five years, demonstrating that the In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) catalyst developed herein achieves the highest CO2 conversion rate at moderate input power. The catalyst formulated in this work shows a distinct advantage, underscoring its potential for future applications.
1) catalyst developed herein achieves the highest CO2 conversion rate at moderate input power. The catalyst formulated in this work shows a distinct advantage, underscoring its potential for future applications.
 | ||
| Fig. 6 Benchmarking of catalytic CO2 conversion performance: current study vs. state-of-the-art systems (2019–2023).26,29,50–55 | ||
This work presents a schematic mechanism for the improved conversion of CO2 using In-Zr composite catalysts inside the DBD reaction system, integrating experimental characterization and DFT simulations (Fig. 7). Plasma catalysis can generate both homogeneous and heterogeneous reactions.56 In the catalyst-free DBD reactor, the predominant homogeneous reaction involves high-energy electrons dissociating CO2 molecules via electron impact, which is the principal route for CO2 dissociation in the DBD plasma reactor. Upon introducing the catalyst into the DBD reactor, both homogeneous reactions in the gas phase and heterogeneous reactions on the catalyst occur simultaneously,54 and this interplay enhances the overall CO2 conversion efficiency of the system. Furthermore, introducing an In-Zr composite catalyst into the discharge zone of the DBD reactor facilitates the emergence of novel CO2 decomposition reaction pathways, supplementing the existing high-energy electron excitation mechanism. There are primarily three pathways to enhance the CO2 conversion efficiency of the system. (1) A significant quantity of oxygen vacancies, as demonstrated in our prior research and existing literature, can significantly facilitate the conversion of CO2 in the DBD reactor. Oxygen vacancies on the catalyst will sequester CO2 gas molecules within the reactor, extending their residence time in the discharge zone, thereby increasing their likelihood of collision with high-energy electrons and eventually improving the total catalytic conversion efficiency.57 (2) The BET analysis indicates that the In-Zr composite catalyst possesses the highest specific surface area and the greatest pore volume. A larger specific surface area facilitates enhanced interaction between CO2 gas molecules and the catalyst, while an increased pore volume allows for greater adsorption of CO2 gas molecules by the catalyst. (3) Upon plasma activation, the reduced bandgap promotes the separation of electron–hole pairs inside the catalyst, allowing electrons to be transported to the adsorbed CO2 molecules on the catalyst's surface, thereby facilitating the breakdown of CO2. The reduced bandgap facilitates the separation of hole–electron pairs while impeding their recombination, which greatly benefits the catalytic conversion of CO2. Moreover, introducing a catalyst into the DBD reactor alters the plasma discharge from filamentary to a hybrid filamentary-surface mode. This modification augments the electric field strength in the discharge region, thereby stabilizing the discharge and enhancing CO2 conversion.
 | ||
| Fig. 7 Mechanistic analysis of the superior comprehensive catalytic performance in In-Zr composite catalysts. | ||
4. Conclusion
In summary, a series of In2O3-ZrO2 composite catalysts were developed through co-precipitation and post-modified using DBD plasma for CO2 decomposition in a plasma-catalytic system. The In-Zr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composition exhibited optimal performance, achieving a CO2 conversion rate of 26.3% with CO selectivity exceeding 90%, along with excellent thermal stability and a prolonged catalytic lifetime of up to 450 minutes. Comprehensive characterization and DFT analysis confirmed that the superior performance originates from (i) the generation of abundant oxygen vacancies, (ii) enhanced specific surface area and pore volume, and (iii) a reduced bandgap promoting charge separation. These factors collectively improve CO2 adsorption, activation, and electron transfer under plasma conditions. The results offer an engineering-oriented strategy for designing robust and efficient plasma-catalyst systems suitable for scalable CO2 conversion applications.
1) composition exhibited optimal performance, achieving a CO2 conversion rate of 26.3% with CO selectivity exceeding 90%, along with excellent thermal stability and a prolonged catalytic lifetime of up to 450 minutes. Comprehensive characterization and DFT analysis confirmed that the superior performance originates from (i) the generation of abundant oxygen vacancies, (ii) enhanced specific surface area and pore volume, and (iii) a reduced bandgap promoting charge separation. These factors collectively improve CO2 adsorption, activation, and electron transfer under plasma conditions. The results offer an engineering-oriented strategy for designing robust and efficient plasma-catalyst systems suitable for scalable CO2 conversion applications.
Author contributions
Sai Li: conceptualization, methodology, investigation, writing – original draft. Yuhang Wang: data curation, validation, writing – original draft. Kui Zhang: conceptualization, methodology. Shaobo Jia: writing – review & editing. Dongyuan Yang: conceptualization, supervision. Peng Ren: writing – review & editing. Zekai Ma: methodology. Shuoshuo Wang: methodology, validation. Haixia Wu: data curation. Yameng Ma: supervision. Qi Chen: methodology. Jiahao Zhouhuang: investigation. Qiuliang Yu: investigation. Lihui Zeng: validation. Rui Tan: writing – review & editing. Zhiming Feng: methodology, supervision, writing – review & editing. Qing Feng: supervision, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data included in this article are available upon request by contacting the corresponding author.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta08108d.
Acknowledgements
This work was financially supported by the Natural Science Foundation of China (NSFC, No. 22109126), Shaanxi Province key research and development plan item (2024CY2-GJHX-72), Yulin City science and technology plan project (2023-CXY-189), Key Laboratory of Coal Resources Exploration and Comprehensive Utilization, Ministry of Land Resources (SMDZ-KF2024-3), and Shaanxi Province Key Point Research and Development Project (2022GY-378).References
- T. P. Hughes, J. T. Kerry, A. H. Baird, S. R. Connolly, T. J. Chase, A. Dietzel, T. Hill, A. S. Hoey, M. O. Hoogenboom, M. Jacobson, A. Kerswell, J. S. Madin, A. Mieog, A. S. Paley, M. S. Pratchett, G. Torda and R. M. Woods, Nature, 2019, 568, 387–390 CrossRef CAS PubMed.
- D. Li, J. Liu, X. Chen, Z. Feng, S. Wang, Y. Wang, N. Lin, J. Wu and Y. Feng, Appl. Catal., B, 2024, 365, 124824 CrossRef.
- S. Z. Xingpeng Tian, H. Hongqing, R. R. Gaddam, R. C. Dutta, H. Wang, B. Wu, N. P. Brandon and R. Tan, Chain, 2025, 2, 38–54 Search PubMed.
- D. Li, S. Xie, J. Liang, B. Ma, J. Fu, J. Wu, Y. Feng and Z. Feng, Sep. Purif. Technol., 2024, 340, 126545 CrossRef CAS.
- Z. Feng, G. Gupta and M. Mamlouk, Int. J. Hydrogen Energy, 2023, 48, 25830–25858 CrossRef CAS.
- Y. Luo, Y. Qiao, H. He, M. Chen, G. Liu, X. Liu, L. Pan, Z. Feng and R. Tan, J. Power Sources, 2025, 631, 236297 CrossRef CAS.
- S. Li, Z. Xin, J. Han, Z. Feng, G. Liao, S. Wang, K. Liu, Q. Li, T. X. Liu and M. Bayati, Ionics, 2024, 30, 7213–7222 CrossRef CAS.
- Z. Feng, S. Zheng, H. Che and Z. Gao, J. Alloys Compd., 2025, 1044, 184360 Search PubMed.
- S. C. Roy, O. K. Varghese, M. Paulose and C. A. Grimes, ACS Nano, 2010, 4, 1259–1278 CrossRef CAS PubMed.
- R. M. Cuéllar-Franca and A. Azapagic, J. CO2 Util., 2015, 9, 82–102 CrossRef.
- Z. Zhang, T. Wang, M. J. Blunt, E. J. Anthony, A.-H. A. Park, R. W. Hughes, P. A. Webley and J. Yan, Appl. Energy, 2020, 278, 115627 Search PubMed.
- S. Ian, G. Andrey, A. D. Ibadillah, L. Xueqian, A. A. Harry, A. V. David and X. Chengxiang, Nat. Catal., 2021, 4, 952–958 Search PubMed.
- Y. Zhao, R. Tan, J. Yang, K. Wang, R. Gao, D. Liu, Y. Liu, J. Yang and F. Pan, J. Power Sources, 2017, 340, 160–166 Search PubMed.
- A. H. Khoja, M. Tahir and N. A. S. Amin, Energy Convers. Manage., 2019, 183, 529–560 CrossRef CAS.
- A. George, B. Shen, M. Craven, Y. Wang, D. Kang, C. Wu and X. Tu, Renewable Sustainable Energy Rev., 2020, 135, 109702 CrossRef.
- S. Li, M. Ongis, G. Manzolini and F. Gallucci, Chem. Eng. J., 2021, 410, 128335 CrossRef CAS.
- J. Liu, S. Xie, D. Li, Z. Feng, H. Lu, X. Li, J. Liang, B. Ma, J. Fu, J. Xie, X. Zhi and Y. Feng, Appl. Catal., B, 2025, 379, 125700 CrossRef CAS.
- M. F. Mustafa, X. Fu, Y. Liu, Y. Abbas, H. Wang and W. Lu, J. Hazard. Mater., 2018, 347, 317–324 CrossRef CAS PubMed.
- M. Alliati, D. Mei and X. Tu, J. CO2 Util., 2018, 27, 308–319 CrossRef CAS.
- V. Maslova, R. Nastase, G. Veryasov, N. Nesterenko, E. Fourré and C. Batiot-Dupeyrat, Prog. Energy Combust. Sci., 2024, 101, 101096 Search PubMed.
- V. Damideh, O. H. Chin, H. A. Gabbar, S. J. Ch'ng and C. Y. Tan, Vacuum, 2020, 177, 109370 Search PubMed.
- H. Guo, Z. Li, Y. Zhang, N. Jiang, H. Wang and J. Li, Sep. Purif. Technol., 2020, 253, 117540 Search PubMed.
- Y. Wang, Z. Liao, S. Mathieu, F. Bin and X. Tu, J. Hazard. Mater., 2021, 404, 123965 Search PubMed.
- L. Wang, Y. Yi, H. Guo and X. Tu, ACS Catal., 2017, 8, 90–100 Search PubMed.
- A. H. Babikir, M. G. M. Ekanayake, O. Oloye, J. D. Riches, K. Ostrikov and A. P. O'Mullane, Adv. Funct. Mater., 2023, 34, 117540 Search PubMed.
- N. Lu, N. Liu, C. Zhang, Y. Su, K. Shang, N. Jiang, J. Li and Y. Wu, Chem. Eng. J., 2021, 417, 129283 Search PubMed.
- D. Ray, P. Chawdhury and C. Subrahmanyam, Environ. Res., 2020, 183, 109286 Search PubMed.
- M. Umamaheswara Rao, K. Bhargavi, G. Madras and C. Subrahmanyam, Chem. Eng. J., 2023, 468, 143671 Search PubMed.
- H. Hadi, K. Mohammadreza and S. Babak, Results Eng., 2024, 24, 102884 Search PubMed.
- Y.-X. Pan, Y. You, S. Xin, Y. Li, G. Fu, Z. Cui, Y.-L. Men, F.-F. Cao, S.-H. Yu and J. B. Goodenough, J. Am. Chem. Soc., 2017, 139, 4123–4129 CrossRef CAS PubMed.
- D. Cai, Y. Cai, K. B. Tan and G. Zhan, Materials, 2023, 16, 2803 Search PubMed.
- S. Li, Y. Wang, K. Zhang, H. Zhu, G. Liao, Q. Li, S. Jia, Q. Feng, J. Pan, R. Tan, Z. Feng and D. Yang, J. Alloys Compd., 2025, 183304, DOI:10.1016/j.jallcom.2025.183304.
- Q. Li, G. Liao, N. Liu, Z. Feng, H. Zhu, S. Jia, Y. Wang, Z. Ma, K. Zhang, Q. Feng, D. Yang and S. Li, J. Phys. D: Appl. Phys., 2025, 58, 375205 Search PubMed.
- G. Liao, S. Li, Y. Luo, Z. Xin, Q. Li, R. Tan, K. Zhang and Z. Feng, J. Alloys Compd., 2025, 1013, 178599 Search PubMed.
- P. Chen, J. Shen, T. Ran, T. Yang and Y. Yin, Plasma Sci. Technol., 2017, 19, 125505 CrossRef.
- D. H. Mei, S. Y. Liu and X. Tu, J. CO2 Util., 2017, 21, 314–326 Search PubMed.
- L. Na, Z. Chuke, S. Kefeng, J. Nan, L. Jie and W. Yan, J. Phys. D: Appl. Phys., 2019, 52, 224003 Search PubMed.
- M. N. Shabbir, R. Alabada, M. Aadil, Z. Ahmad, A. A. Alothman, N. A. Almuhous, W. A. Khan and R. M. K. Mohamed, Ceram. Int., 2024, 50, 4404–4414 Search PubMed.
- L. Wang, B. Cheng, L. Zhang and J. Yu, Small, 2021, 17, 2103447 Search PubMed.
- A. Tsoukalou, P. M. Abdala, A. Armutlulu, E. Willinger, A. Fedorov and C. R. Müller, ACS Catal., 2020, 10, 10060–10067 Search PubMed.
- S. Li, G. Liao, D. Bildan, Z. Feng, H. Cai, J. Han, Z. Xin, K. Liu, Q. Li, J. Shang and T. X. Liu, Int. J. Hydrogen Energy, 2024, 67, 448–457 Search PubMed.
- R. Bian, S. An, X. Wang, Y. Xue, J. Tian, Z. Liang and Z. Song, Int. J. Hydrogen Energy, 2024, 51, 787–795 Search PubMed.
- X. Hu, J. Wang, J. Wang, Y. Deng, H. Zhang, T. Xu and W. Wang, Appl. Catal., B, 2022, 318, 121879 CrossRef CAS.
- X. Liu, M. Wang, C. Zhou, W. Zhou, K. Cheng, J. Kang, Q. Zhang, W. Deng and Y. Wang, Chem. Commun., 2018, 54, 140–143 RSC.
- X. Sun, J. Wu, F. Tian, W. Zhang and Q. Li, Mater. Res. Bull., 2018, 103, 247–258 CrossRef CAS.
- D. Triyono, U. Hanifah and H. Laysandra, Results Phys., 2020, 16, 102995 CrossRef.
- T. Xiong, W. Cen, Y. Zhang and F. Dong, ACS Catal., 2016, 6, 2462–2472 CrossRef CAS.
- H. Zhu, X. Song, X. Han, X. Zhang, J. Bao, N. Zhang and G. He, Environ. Sci. Technol., 2020, 54, 8601–8611 CrossRef CAS PubMed.
- J. Li, X. Zhai, C. Ma, S. Zhu, F. Yu, B. Dai, G. Ge and D. Yang, Nanomaterials, 2019, 9, 1595 CrossRef CAS PubMed.
- H. Hatami, M. Khani, S. A. Razavi Rad and B. Shokri, Heliyon, 2024, 10, e26280 CrossRef CAS PubMed.
- P. Kaliyappan, A. Paulus, J. D'Haen, P. Samyn, Y. Uytdenhouwen, N. Hafezkhiabani, A. Bogaerts, V. Meynen, K. Elen, A. Hardy and M. K. Van Bael, J. CO2 Util., 2021, 46, 101468 Search PubMed.
- L. Xia, M. Wu, Y. Li, H. Guo, Y. Cheng and G. Meng, Energy Fuels, 2023, 38, 565–575 Search PubMed.
- X. Ma, S. Li, M. Ronda-Lloret, R. Chaudhary, L. Lin, G. van Rooij, F. Gallucci, G. Rothenberg, N. Raveendran Shiju and V. Hessel, Plasma Chem. Plasma Process., 2018, 39, 109–124 CrossRef.
- K. Zhang, G. Zhang, X. Liu, A. N. Phan and K. Luo, Ind. Eng. Chem. Res., 2017, 56, 3204–3216 CrossRef CAS.
- W. Taobo, L. Hongxia, X. Xiang and F. Xinxin, IOP Conf. Ser. Earth Environ. Sci., 2017, 52, 012100 CrossRef.
- X. Fan, T. Zhu, Y. Sun and X. Yan, J. Hazard. Mater., 2011, 196, 380–385 CrossRef CAS PubMed.
- Y. Lu, X. Li, X. Jing, Y. Huang, Y. Qiang, G. Cao, R. Qin, Q. Cao, J. Cao and S. C. Lee, Surf. Interfaces, 2024, 53, 104957 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




