Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Microwave-assisted one-pot sol–gel synthesis of tungsten silicate microspheres with dispersed WOx and their activity in ethanol dehydration
David
Skoda
 *a,
Tomas
Pokorny
*a,
Tomas
Pokorny
 b,
Barbora
Hanulikova
b,
Barbora
Hanulikova
 a,
Ales
Styskalik
a,
Ales
Styskalik
 *b,
Zuzana
Hlavenkova
*b,
Zuzana
Hlavenkova
 c,
Lucie
Simonikova
c,
Lucie
Simonikova
 b,
Jan
Varada
b,
Jan
Varada
 b,
Lucie
Leonova
b,
Lucie
Leonova
 b,
Ivo
Kuritka
b,
Ivo
Kuritka
 a,
Claude
Poleunis
d and
Damien P.
Debecker
a,
Claude
Poleunis
d and
Damien P.
Debecker
 *d
*d
aCentre of Polymer Systems, Tomas Bata University in Zlín, tr. Tomase Bati 5678, Zlín, CZ-76001, Czech Republic. E-mail: dskoda@utb.cz
bDepartment of Chemistry, Faculty of Science, Masaryk University, Kotlarska 2, Brno, CZ-61137, Czech Republic. E-mail: styskalik@chemi.muni.cz
cCentral European Institute of Technology, Masaryk University, Kamenice 5, Brno, CZ 62500, Czech Republic
dInstitute of Condensed Matter and Nanosciences (IMCN), Université catholique de Louvain (UCLouvain), Place Louis Pasteur 1, 1348 Louvain-La-Neuve, Belgium. E-mail: damien.debecker@uclouvain.be
First published on 13th March 2026
Abstract
This study introduces a novel microspherical W–SiO2 heterogeneous catalyst for alcohol dehydration, prepared via an innovative microwave-assisted condensation synthesis method. The process involves the microwave-assisted preparation of a hybrid tungsten naphthalene dicarboxylate-based precursor solution, which is subsequently condensed with (3-aminopropyl)triethoxysilane in a single step—eliminating the need for separate silica support preparation, as required in conventional impregnation methods. After calcination at 550 °C, the resulting amorphous and porous microspheres contain highly dispersed tungsten species (with loadings of 2, 6, and 12 wt%), with no crystalline WO3 phase detected, even at the highest loading. Compared to a 12 wt% WO3/SiO2 catalyst prepared via conventional impregnation, the W–SiO2 microspheres exhibit higher catalytic activity and ethylene selectivity in ethanol dehydration at 420 °C. Notably, the 2W–SiO2 catalyst achieved the highest initial ethylene productivity per mole of tungsten (520 mmol mmolW−1 h−1), maintaining 230 mmol mmolW−1 h−1 after 1000 minutes on stream, indicating high long-term stability. In terms of mass-specific performance, the 12W–SiO2 catalyst reached 133 mmol g−1 h−1, outperforming other comparable previously reported tungsten–silica catalysts.
1 Introduction
Ethylene is one of the world's most important chemical commodities, serving as a key building block for the synthesis of major derivatives such as polyethylene, vinyl chloride, ethylene oxide, ethylbenzene, and poly(ethylene terephthalate). Currently, ethylene production relies predominantly on the steam cracking of petroleum-based feedstocks.1 However, the growing global demand for alkene compounds is driving increased consumption of fossil fuels and oil resources. This situation underscores the urgent need for sustainable alternatives to conventional alkene production. One promising route is the dehydration of (biomass-derived) alcohols to produce alkenes. Ethanol, in particular, stands out as a highly sustainable and readily available feedstock for the ethanol-to-ethylene (ETE) process.1,2 Traditionally, alcohol dehydration requires elevated temperatures and strong acids, making the development of efficient heterogeneous catalysts essential for enabling milder reaction conditions and reducing energy consumption in the ETE process. Utilizing biomass-derived ethanol for ethylene production can significantly lower greenhouse gas emissions and reduce dependence on finite fossil resources,3 contributing to the achievement of Sustainable Development Goals (SDGs), namely SDG7 (Affordable and Clean Energy), SDG 9 (Industry, Innovation and Infrastructure), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action).4Tungsten-containing catalysts have demonstrated notable activity and selectivity in alcohol dehydration to alkenes,1,5–8 olefin metathesis,9,10 and dehydration of glucose11 and glycerol.12 The catalytic performance of these systems depends on several factors, including the nature of the tungsten species, the choice of support material, and reaction conditions. In particular, tungsten oxide (WOx) supported on silica exhibits unique properties that promote high ethanol dehydration activity.6 The interaction between tungsten and silica stabilizes the active species and boosts overall catalytic activity. For example, WO3 supported on mesoporous structures such as mesocellular foam silica (MCF-Si) and SBA-15 has shown excellent performance in the ETE process. Among these, WO3/MCF-Si catalysts have achieved ethylene yields of up to 96.6% under vapor-phase conditions at 400 °C, even with dilute ethanol feeds (70% v/v), and maintain stability over extended operation without regeneration.6,13
A common synthesis method for WOx/SiO2 heterogeneous catalysts is impregnation, where a precursor solution is deposited onto a silica support followed by calcination.6,8,13–17 For instance, WO3/SiO2 catalysts are typically prepared via incipient wetness impregnation of ammonium tungstate onto silica, followed by calcination at around 550 °C.17 Calcination not only ensures a well-dispersed active phase but also enhances thermal stability, which is crucial for sustained catalytic performance. Beyond classical impregnation, innovative techniques such as microwave-assisted grafting, aerosol-assisted, and non-hydrolytic preparation methods have emerged.18–20 These methods facilitate the rapid and uniform distribution of WO3 on silica substrates, enhancing catalyst activity and selectivity in reactions such as glycerol dehydration to acrolein or olefin metathesis.18–20 Specifically, in the case of olefin metathesis the isolated and highly dispersed W(VI)–dioxo sites are of tremendous importance to achieve high catalytic performance.9 Microwave-assisted methods, in particular, optimize the grafting of metal oxides onto colloidal silica, significantly enhancing functional performance compared to conventional heating. Additional advanced methods include aerosol processes combined with surfactant-templated sol–gel synthesis,21 and template-free sol–gel methods assisted by supercritical CO2 drying.22
Tungsten-based catalysts can exhibit both Brønsted and Lewis acidity,12,23–25 with each type contributing to the catalytic activity in alcohol dehydration reactions.26–28 The mechanism for ethanol dehydration over these catalysts generally involves ethanol adsorption, protonation, and subsequent water elimination, ultimately forming ethylene.29,30 This process typically follows a unimolecular elimination (E1) pathway, where the initial cleavage of the C–O bond is the rate-determining step, followed by β-hydrogen elimination to generate the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond.31 The distribution and strength of Brønsted and Lewis acid sites on the catalyst surface play a crucial role in determining product selectivity, with optimal conditions favoring high ethylene yields.29 The amount of tungsten loaded onto the silica support also significantly impacts catalytic performance; an optimal loading maximizes the number of active sites while minimizing steric hindrance.32 Additionally, the stability and durability of tungsten-silica catalysts under specific reaction conditions are essential for their practical implementation. Catalyst deactivation, often due to coking or sintering, presents a significant challenge in maintaining catalytic performance over time.33 It has also been reported that the hydrophobicity and hydrophilicity of heterogeneous catalysts used in reactions that generate water play an important role in catalyst stability and overall performance.34 Recent advancements in catalyst design—such as the incorporation of mesoporous structures and hybrid materials—have been explored to enhance the stability and activity of tungsten-based catalysts.35,36 These innovations aim to create more robust catalytic frameworks capable of withstanding the demanding conditions typical of ethanol dehydration processes. Despite these advances, the rapid and one-step synthesis of stable, reusable catalysts with a homogeneous distribution of tungsten, high activity, and selectivity at moderate temperatures remains a significant challenge.
C bond.31 The distribution and strength of Brønsted and Lewis acid sites on the catalyst surface play a crucial role in determining product selectivity, with optimal conditions favoring high ethylene yields.29 The amount of tungsten loaded onto the silica support also significantly impacts catalytic performance; an optimal loading maximizes the number of active sites while minimizing steric hindrance.32 Additionally, the stability and durability of tungsten-silica catalysts under specific reaction conditions are essential for their practical implementation. Catalyst deactivation, often due to coking or sintering, presents a significant challenge in maintaining catalytic performance over time.33 It has also been reported that the hydrophobicity and hydrophilicity of heterogeneous catalysts used in reactions that generate water play an important role in catalyst stability and overall performance.34 Recent advancements in catalyst design—such as the incorporation of mesoporous structures and hybrid materials—have been explored to enhance the stability and activity of tungsten-based catalysts.35,36 These innovations aim to create more robust catalytic frameworks capable of withstanding the demanding conditions typical of ethanol dehydration processes. Despite these advances, the rapid and one-step synthesis of stable, reusable catalysts with a homogeneous distribution of tungsten, high activity, and selectivity at moderate temperatures remains a significant challenge.
We have recently introduced a facile preparation technique for vanadium- or molybdenum-silicate microspheres and shown their application as heterogeneous catalysts for epoxidation reactions and propylene metathesis, respectively.37–39 We have demonstrated that the developed synthetic procedure leads to a highly homogeneous dispersion of molybdate or vanadate species in the microspheres compared to the catalysts prepared by incipient wetness impregnation. Based on these findings we developed an original preparation method for tungsten-silicate microspheres with homogeneously dispersed tungstate species.
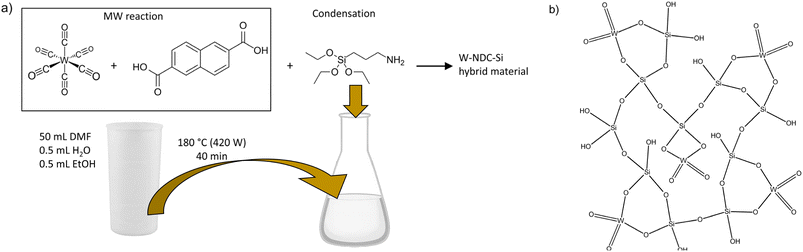
A microwave-assisted sol–gel condensation method allows the fabrication of W–SiO2 catalysts tailored for alcohol dehydration. The synthesis begins with the microwave-assisted preparation of a hybrid precursor solution comprising tungsten hexacarbonyl and 2,6-naphthalene dicarboxylic acid, which is subsequently condensed with (3-aminopropyl)triethoxysilane. This reaction leads to the precipitation of a hybrid tungsten silicate solid precursor. Upon calcination, the final inorganic W–SiO2 microspheres are obtained. This streamlined one-pot approach eliminates the need for separate silica support synthesis. Moreover, the method enables the formation of highly dispersed and homogeneously distributed tungsten active sites within the silica matrix, which significantly enhances the catalytic performance of the resulting W–SiO2 microspheres.
2 Experimental
2.1 Chemicals
Tungsten hexacarbonyl (W(CO)6, Mw 351.9 g mol−1, 99%), 2,6-naphthalenedicarboxylic acid (H2NDC, Mw 216.19 g mol−1, 95%), (3-aminopropyl)triethoxysilane (H2N(CH2)3Si(OC2H5)3, APTES, Mw 221.37 g mol−1, 99%), ammonium metatungstate hydrate ((NH4)6H2W12O40·xH2O, Mw 2956.3 g mol−1, 99%), and a silica support (Normasil 60) were supplied from Merck. N,N′-Dimethylformamide (DMF, p.a., 99.5%) and absolute ethanol (UV-vis spectroscopy grade, 99.8%) were purchased from Penta Chemicals (Czech Republic).The W–NDC–Si solid precursors were calcined in a muffle furnace at 550 °C (heating ramp rate of 5 °C min−1) for 3 hours under an air atmosphere to generate inorganic W–SiO2-based samples, which were utilized as catalysts for alcohol dehydration. Three samples denoted as 12W–SiO2, 6W–SiO2, and 2W–SiO2 were prepared in total with the numbers at the beginning of the sample label referring to different W loadings.
2.2 Characterization techniques
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3. The W4f – 4f7/2 and 4f5/2 peaks were generally all constrained to have an equal FWHM.
3. The W4f – 4f7/2 and 4f5/2 peaks were generally all constrained to have an equal FWHM.
3 Results and discussion
3.1 Catalyst characterization
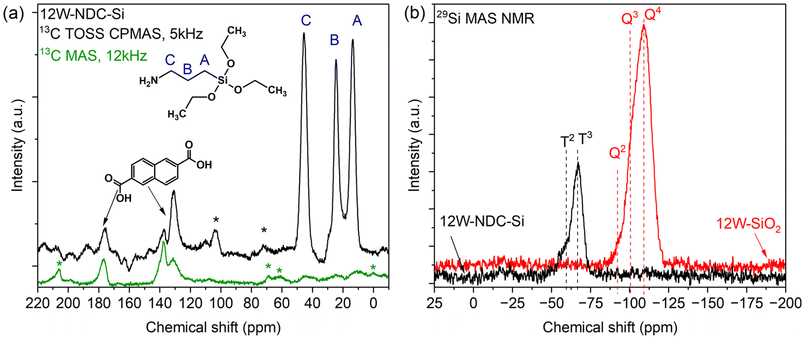
In this work, a novel sol–gel synthetic procedure for W–SiO2 microspheres is introduced and their catalytic activity in alcohol dehydration is demonstrated. The fine dispersion of tungstate species in microspheres was achieved through a preparation technique involving the microwave-assisted heating of W(CO)6 and 2,6-naphthalene dicarboxylic acid precursors, followed by condensation with APTES. Within this procedure hybrid metallosilicate solid precursors (Fig. 1a) with different W loadings were prepared (FTIR and TGA analyses of 12W–NDC–Si are given in SI; Fig. 2S and 3S) and, after subsequent calcination at 550 °C, they were transformed to inorganic W–SiO2 materials (Fig. 1b) containing 2, 6, and 12 wt% of W. The described synthesis was inspired by our previous work on Mo–SiO2 and V–SiO2 microspheres, which have exhibited highly homogeneous metal dispersions and outstanding catalytic activities in olefin metathesis, olefin epoxidation, and ethyl lactate oxidation.37–39 However, here we have applied a different linker: previously applied 4,4′-biphenyl dicarboxylic acid has been replaced by cost-effective 2,6-naphthalene dicarboxylic acid. For the sake of comparison, the pure SiO2 microspheres prepared via the same methodology were impregnated with ammonium metatungstate hydrate. This sample is labeled as 4W–SiO2-imp.Solid-state NMR spectroscopy was used to identify functional groups in both the as-prepared (12W–NDC–Si) and calcined (12W–SiO2) samples. This experiment was performed to confirm the elimination of ethoxy groups during the sol–gel condensation. The 13C TOSS CPMAS and 13C MAS NMR spectra are shown in Fig. 2a. The signals for different functional groups are distinguishable. The carbon atoms from the 3-aminopropyl group appear at 14, 25, and 46 ppm. Since the signals of ethoxy groups from APTES did not significantly appear in the 13C TOSS CPMAS NMR spectrum, it can be concluded that ethoxy groups are released in the form of ethanol as the product of the condensation between APTES and W–NDC species in the precursor solution. The NDC linker is identified by distinct carbonyl resonance observed at 176 ppm. Additionally, the 13C MAS NMR spectrum was recorded as well (Fig. 2a). As expected, the intensity of signals originating in the NDC linker is increased compared to the 13C TOSS CPMAS NMR spectrum due to the absence of signal intensity enhancement via cross-polarization. The ratio between the aminopropyl groups and the aromatic rings (from the linker) is approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.87. This corresponds to a Si
0.87. This corresponds to a Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NDC molar ratio of 1.15
NDC molar ratio of 1.15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in 12W NDC Si. This value is consistent with the mass losses recorded within the TGA analysis of 12W NDC Si (Fig. 3S).
1 in 12W NDC Si. This value is consistent with the mass losses recorded within the TGA analysis of 12W NDC Si (Fig. 3S).
 | ||
| Fig. 2 13C TOSS CPMAS and 13C NMR spectra of the 12W–NDC–Si sample (a). 29Si MAS NMR spectra of 12W–NDC–Si and 12W–SiO2 samples (b). Rotational sidebands are marked with an asterisk. | ||
The 29Si MAS NMR spectrum of the 12W–NDC–Si sample is shown in Fig. 2b. The spectrum displays signals at chemical shifts of −60 and −67 ppm, corresponding to T2 and T3 type groups, respectively. In this case the T2 type is attributed to LSi(OR)(OSi)2 and T3 type is characteristic of LSi(OSi)3 species, where L: aminopropyl group, and R: W or NDC linker. The results confirm the condensation of APTES and are consistent with the findings from the 13C TOSS CPMAS NMR analysis. Additionally, the 29Si MAS NMR spectrum of the 12W–SiO2 sample, shown in Fig. 2b, revealed signals corresponding to Qn sites in silicates. This demonstrates the successful conversion of organosiloxanes into fully inorganic silicates after the calcination of 12W–NDC–Si. The features denoted as Q4, Q3, and Q2 correspond to the signals of Si(OSi)4, (WO)Si(OSi)3/HO–Si(OSi)3, and (WO)2Si(OSi)2/(HO)2Si(OSi)2 species, respectively. As anticipated, because the MAS NMR spectra were acquired without using cross-polarization, the Q4 sites (−110 ppm) exhibited the strongest signal intensity. The peaks corresponding to Q3 (WO)Si(OSi)3/HO–Si(OSi)3 and Q2 (WO)2Si(OSi)2/(HO)2Si(OSi)2 environments appeared at −101 ppm and −92 ppm, respectively.24
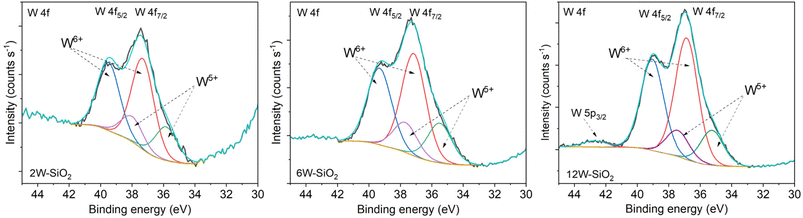
It was found that all W–SiO2 microspheres are amorphous, showing no diffraction lines of WO3 in their powder X-ray diffraction patterns (Fig. 3a). Moreover, Raman spectra of W–SiO2 microspheres (Fig. 3b) did not show the presence of the bands indicating WO3 crystalline species and therefore, the presence of the WO3 crystalline phase can be safely excluded.32,45 The band with a Raman shift of 960 cm−1 may be ascribed to the W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond.46 It should be noted that in the case of supported WOx/SiO2 catalysts, separation of WO3 oxide occurs already at a tungsten content of around 8–10 wt%.32 However, our preparation technique can maintain the amorphous form of the tungstate species even for the microsphere sample with 12 wt% of tungsten. In contrast, the impregnated pure silica microspheres (4W–SiO2-imp) and the 12W–SiO2–WI sample prepared via the impregnation technique shows the presence of crystalline WO3 in the PXRD diffractogram (JCPDS card no. 01-071-0305) and Raman spectrum (Fig. 3b).18,32,47 These results demonstrate that the method, which integrates microwave-assisted heating with one-step sol–gel condensation, yields samples containing a high population of amorphous and well-dispersed tungstate species.45
O bond.46 It should be noted that in the case of supported WOx/SiO2 catalysts, separation of WO3 oxide occurs already at a tungsten content of around 8–10 wt%.32 However, our preparation technique can maintain the amorphous form of the tungstate species even for the microsphere sample with 12 wt% of tungsten. In contrast, the impregnated pure silica microspheres (4W–SiO2-imp) and the 12W–SiO2–WI sample prepared via the impregnation technique shows the presence of crystalline WO3 in the PXRD diffractogram (JCPDS card no. 01-071-0305) and Raman spectrum (Fig. 3b).18,32,47 These results demonstrate that the method, which integrates microwave-assisted heating with one-step sol–gel condensation, yields samples containing a high population of amorphous and well-dispersed tungstate species.45
Electron microscopy was employed to study the morphology of the W–SiO2 samples derived from W–NDC–Si solid precursors and 4W–SiO2-imp microspheres prepared via impregnation of pure silica microspheres. As illustrated by SEM (Fig. 4S–7S), all samples exhibited well defined spherical morphology. The average size of microspheres determined via graphical analysis of the SEM images was 265 ± 39 nm for 12W–SiO2, 385 ± 38 nm for 6W–SiO2, and 659 ± 33 nm for 2W–SiO2 (Fig. 8S). It can be argued that the dispersion of tungsten in this kind of heterogeneous catalyst plays an important role in the catalytic activity. Therefore, the elemental distribution of silicon, tungsten, and oxygen in microspheres was investigated by STEM-EDS. Fig. 4 unambiguously shows that tungsten is homogeneously dispersed throughout the microspheres. Conversely, the STEM-EDS images of the 4W–SiO2-imp sample (Fig. 9S) show an inhomogeneous dispersion of tungsten species and their agglomeration into larger particles, which is consistent with the PXRD and Raman spectroscopy results.
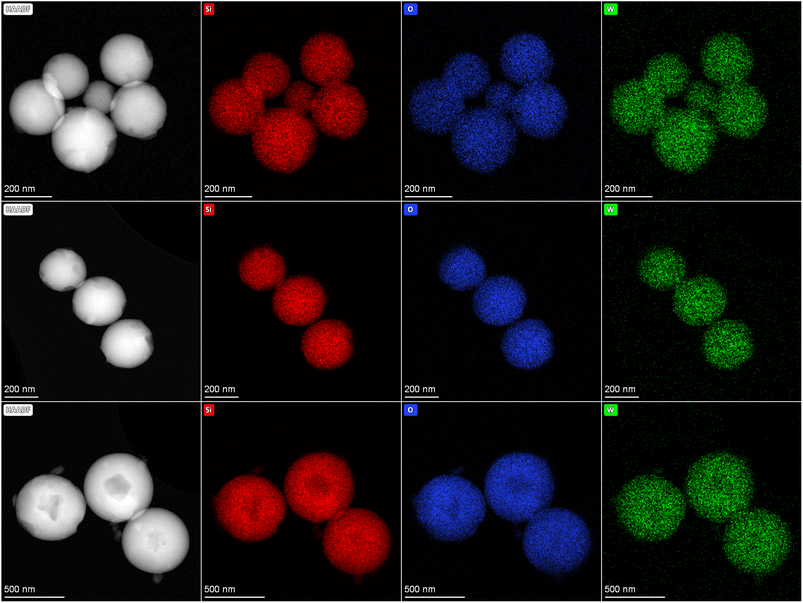 | ||
| Fig. 4 HAADF images and STEM-EDS elemental maps of W–SiO2 microspheres (12W–SiO2 – upper row, 6W–SiO2 – middle row, and 2W–SiO2 – bottom row). Si – red, O – blue, and W – green. | ||
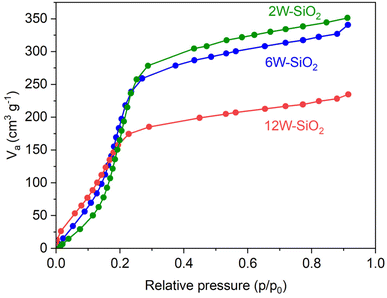
Nitrogen adsorption/desorption isotherms of W–SiO2 microspheres are displayed in Fig. 5a (and in the case of the sample 12W–SiO2–WI in the SI in Fig. 10S). According to the IUPAC classification, all isotherms of microspherical samples revealed a Type I shape that is indicative of microporous materials. Based on the BET method, high surface areas of 416, 512, and 594 m2 g−1 were achieved for 12W–SiO2, 6W–SiO2, and 2W–SiO2 microspheres (Table 1). The NLDFT method was used for pore size distribution calculation and confirmed a microporous character with a pore size distribution centered on a diameter of 1.1 nm for all samples (Fig. 5b). We attribute this behavior to the well-organized hybrid silicate framework crosslinked by the NDC linker, which, upon its removal during calcination, gives rise to the porous structure. SA and pore volume values, together with the tungsten contents and microsphere diameters, are listed in Table 1.
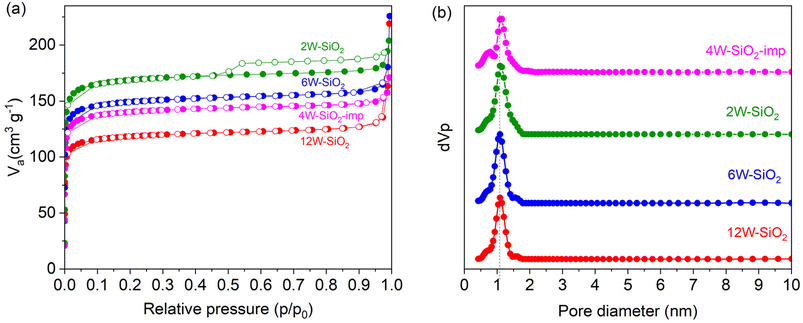 | ||
| Fig. 5 Nitrogen adsorption/desorption isotherms of W–SiO2 microspheres (a). Pore size distribution according to the NLDFT method (b). | ||
| Sample | Wa [wt%] | SA(BET) [m2 g−1] | Pore volume [cm3 g−1] | Pore diameter [nm] | Microsphere diameterb [nm] | W surface densityc/d [W nm−2] |
|---|---|---|---|---|---|---|
| a Determined by the ICP-OES method. b Calculated from SEM images. c Surface density was calculated as the number of W atoms per square nanometer of the catalyst21 using the W content determined by XPS. d Surface density was calculated as the number of W atoms per square nanometer of the catalyst21 using the W content determined by ICP-OES. | ||||||
| 2W–SiO2 | 1.96 ± 0.01 | 594 | 0.31 | 1.1 | 659 ± 33 | 0.05/0.10 |
| 6W–SiO2 | 6.46 ± 0.01 | 512 | 0.30 | 1.1 | 385 ± 38 | 0.18/0.41 |
| 12W–SiO2 | 11.83 ± 0.01 | 416 | 0.28 | 1.1 | 266 ± 39 | 0.62/0.93 |
| 4W–SiO2-imp | 4.01 ± 0.01 | 498 | 0.25 | 1.1 | 684 ± 129 | 0.17/0.26 |
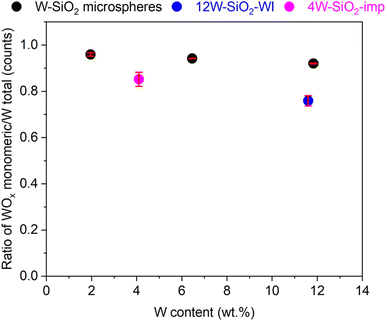
X-ray photoelectron spectroscopy (XPS) was employed to identify the tungsten oxidation state and functional groups on the surface of W–SiO2 microspheres. Survey scans over the whole binding energy (BE) range are included in the SI (Fig. 11S). As illustrated in Fig. 6, the high-resolution W 4f spectra of W–SiO2 microspheres revealed W 4f5/2 and W 4f7/2 contributions of the W6+ and W5+ valence states after deconvolution and fitting of the recorded data.5,11,48 In the case of sample 12W–SiO2, the W 4f5/2 and W 4f7/2 spin–orbit doublets of the W6+ and W5+ states appear at 39.0 and 36.8, and 37.5 and 35.3 eV, respectively. These positions align with published data on WOx/SiO2 catalysts.5,25,47,48 In contrast, the W 4f spectrum of 12W–SiO2–WI (Fig. 12S) shows W6+ contributions at 37.4 eV (W 4f5/2) and 35.3 eV (W 4f7/2), characteristic of bulk WO3.49 Specifically, shifts to higher binding energy regions in the tungsten 4f XPS peaks when WOx is dispersed on silica compared to its bulk WO3 form have been reported in several studies.25,50–52 It is suggested that these shifts could be attributed to changes in electron density resulting from metal–support interactions. Furthermore, Howell et al. noted that the tungsten oxide dispersion on a silica support influences catalytic behavior and is reflected in shifts of the 4f peaks toward higher binding energies.53 Thus, the observed high W 4f5/2 and W 4f7/2 binding energies indicate a well dispersed WOx in W–SiO2 microspheres in accordance with the other analyses (DRUV-Vis spectroscopy and STEM-EDS). XPS spectra of the 4W–SiO2-imp sample are given in the SI (Fig. 13S and 14S).
Based on the XPS results the tungsten content on 12W–SiO2, 6W–SiO2, and 2W–SiO2 microsphere surfaces is 7.9, 2.8, and 0.9 wt%, respectively (Table 2S), which is lower than the corresponding bulk contents determined by the ICP-OES method. This may be attributed to the more rapid polycondensation reactions occurring around tungsten centers during synthesis, which lead to a comparatively lower tungsten concentration on the surface than within the interior of the walls. Hence, the surface Si and O concentration is complementarily higher. O 1s high-resolution XPS spectra are given in the SI (Fig. 15S–17S). As illustrated, the contributions of W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and Si–O species at a BE of 530.9 and 532.8 eV, respectively, are found in the spectra. Moreover, Si–OH and C–O moieties with a BE of 533.6 and 538.8 eV were evidenced. In the Si 2p high-resolution XPS spectra (Fig. 13S–15S), the characteristic contributions of Si–O–Si and Si–OH moieties were evidenced. Notably, the number of adsorbed species (adventitious carbon) on the catalyst surface is very low as expected (Fig. 11S, 15S–17S and Table 2S).
O and Si–O species at a BE of 530.9 and 532.8 eV, respectively, are found in the spectra. Moreover, Si–OH and C–O moieties with a BE of 533.6 and 538.8 eV were evidenced. In the Si 2p high-resolution XPS spectra (Fig. 13S–15S), the characteristic contributions of Si–O–Si and Si–OH moieties were evidenced. Notably, the number of adsorbed species (adventitious carbon) on the catalyst surface is very low as expected (Fig. 11S, 15S–17S and Table 2S).
Time-of-flight secondary ion mass spectrometry (ToF-SIMS) offered detailed information on the tungstate species present at the outermost surface of the catalysts.54–56 Table 3S lists all quantified W-containing secondary anions detected and identified during analysis. The overall proportion of W-containing clusters in the W–SiO2-based microspheres (determined as the sum of the normalized intensities of each W-containing anion) logically increased with the W loading (Fig. 18S). At the same time, it can be seen that the 12W–SiO2–WI sample prepared by the impregnation technique exhibits the highest proportion of tungsten clusters on the surface, as expected. Detected ions were categorized based on W nuclearity: (1) ions with one W atom (WO3−, WO4−, WO5Si−, and WO7Si2−), and (2) ions with multiple W atoms (W2O6− and W3O9−). The relative intensities of clusters with one W atom serve as a useful indicator of WOx dispersion. Conversely, a high proportion of clusters with multiple W atoms suggests the presence of condensed species, such as poly-tungstates or crystalline WO3. As illustrated in Fig. 7, the ratio of monomeric W-containing clusters to total counts of detected W-containing clusters was significantly higher in W–SiO2 as compared to both 4W–SiO2-imp and 12W–SiO2–WI samples (Fig. 7). Noteworthily, ToF-SIMS shows that the proportion of highly dispersed WOx species at the surface of the W–SiO2 microspheres remains similar whatever the W loading. Thus, ToF-SIMS showed a better W dispersion in W–SiO2 samples in comparison to impregnated materials at the outermost surface of the tungsten silicates complementing the XPS, STEM-EDS, XRD, and Raman analyses.
Diffuse reflectance UV-vis spectroscopy was conducted in order to evaluate the local structure (isolated monomer, dimer, polymeric chain, cluster, or three-dimensional structure) of tungsten species via the ligand-to-metal charge transfer (LMCT) band position and the corresponding edge energy (Eg) value. DRUV-vis spectroscopy measurements (Fig. 8a) were performed on the samples dried under vacuum (150 °C, flushed with nitrogen). A distinctive characteristic of DRUV-vis spectra for isolated WO4 reference compounds is the presence of a single ligand-to-metal charge transfer (LMCT) band in the range of approximately 218–274 nm, with most band maxima occurring between 220 and 250 nm. In contrast, bulk WO3 displays two LMCT transitions at 251 nm and 338 nm.45 The corresponding band edge energy values, Eg, for regular isolated WO4 structures are 5.2–5.6 eV, whereas the energies of 4.0–4.5 eV are reported for the distorted WO4 structures.45,57 For example, Na2WO4, in which W is present as isolated tetrahedral centers exhibits Eg = 4.7 eV.53 As demonstrated in Fig. 8b, the band edge values for 12W–SiO2, 6W–SiO2, and 2W–SiO2 are 4.00, 4.10, and 4.58 eV. These values can support the presence of the distorted WO4 species, especially for 12W–SiO2 and 6W–SiO2 microspheres. In the case of the 2W–SiO2 sample, the sharp band in DRUV-vis spectra with a maximum at 215 nm and band edge value of 4.58 eV may be assigned to isolated WO4 species.32,45 The Eg value of the 4W–SiO2-imp microspherical sample was 3.92 eV, lower than that of all W–SiO2 samples, a hint of worse dispersion of tungsten species in samples prepared via impregnation. The position of the maxima in the DRUV-vis spectra suggests the presence of a certain proportion of dispersed tungstate species. However, significant absorption in the 320–450 nm region (Fig. 8a) demonstrates a poorer distribution of tungsten in this sample, further supported by PXRD, Raman, and STEM-EDS analyses. In the case of the 12W–SiO2–WI sample, the band is shifted to a significantly higher wavelength indicating poor tungsten dispersion and the presence of tungsten oxide species (in agreement with XRD and Raman spectroscopy). Accordingly, the band edge energy reaches 3.34 eV, which corresponds to isopolytungstate species containing WO6 clusters—consistent with literature values reported between 3.0 and 3.6 eV.11,45
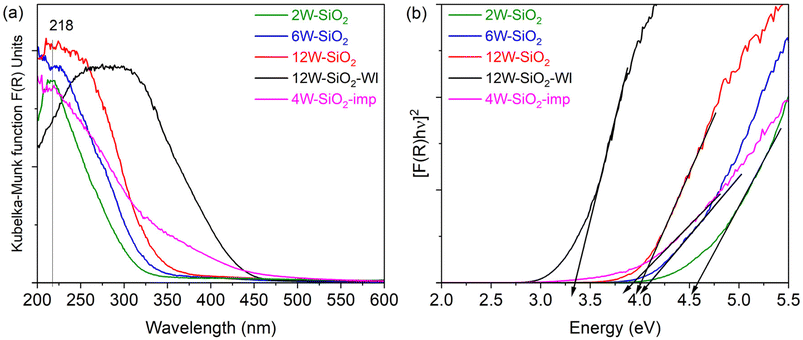 | ||
| Fig. 8 Diffuse reflectance UV-vis spectra of W–SiO2 microspheres (a) and Tauc plots with band edge energy values (Eg) (b). | ||
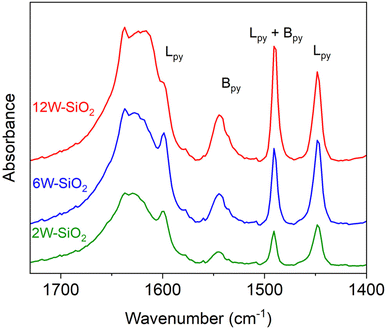
Pyridine adsorption tests were performed to investigate the surface acidity of W–SiO2 microspheres. Pyridine was adsorbed on dried samples (150 °C, vacuum), and acid sites were monitored by FTIR spectroscopy. As illustrated in Fig. 9, all W–SiO2 microspheres contain both Lewis and Brønsted acid sites characterized by vibrational bands at 1449 and 1545 cm−1, respectively.11,58 Both types of sites have been shown to be active in ethanol dehydration. Specifically, Lewis sites, characterized by tungsten di-oxo species, help stabilize reaction intermediates and participate in the elimination mechanism, while Brønsted sites are responsible for the protonation of ethanol.26–28,59,60
The Lpy (1449 cm−1) and Bpy (1545 cm−1) bands were deconvoluted and integrated, and the Lpy/Bpy ratio was calculated taking into account the integrated molar extinction coefficients (IMECs) reported by Emeis,61 Zholobenko,62 and Gallo63 for silica–alumina-based catalysts. The IMEC values used for pyridine adsorbed on Lewis and Brønsted acid sites were 2.22 and 1.67 cm µmol−1, respectively. The absorption band areas for pyridine adsorbed on Lewis and Brønsted acid sites, along with the Lpy/Bpy area ratios, are presented in Table 2. Based on these data, it can be concluded that the 12W–SiO2 catalyst (Lpy/Bpy = 0.95) exhibited a significantly higher population of Brønsted acid sites compared to the 6W–SiO2 and 2W–SiO2 microspheres. Conversely, the Lpy/Bpy ratios of 1.50 and 2.00 obtained for 6W–SiO2 and 2W–SiO2, respectively, indicate a higher population of Lewis acid sites.
For deeper insights, TG analysis of the samples with adsorbed pyridine was performed in a nitrogen atmosphere. As illustrated in Fig. 19S, the mass losses assigned to pyridine desorption are observed at temperatures above 160 °C. According to the derivatives of the TG curves, the mass loss attributed to the adsorbed pyridine was evidenced. The amount of pyridine adsorbed on 12W–SiO2, 6W–SiO2, and 2W–SiO2 catalysts, determined by TG analysis, reached 4.54, 3.66, and 2.04 wt%, respectively (Table 2), which corresponds to the tungsten content in the samples. The pyridine adsorption study for the 12W–SiO2–WI and 4W–SiO2-imp samples is presented in the SI (Fig. 20S). A noticeably lower intensity of absorption bands associated with the Lewis acid sites is observed compared to the microspherical W–SiO2 samples. Additionally, thermogravimetric (TG) analysis indicates a pyridine mass loss of 1.27% for 12W–SiO2–WI (0.16 mmol g−1 of pyridine), the lowest among all W–SiO2 samples prepared herein. TGA analysis of the 4W–SiO2-imp sample after pyridine adsorption (Fig. 20S) exhibited mass loss in the region of 160–290 °C corresponding to 3.1 wt% (0.39 mmol g−1) of pyridine (Table 4S). Interestingly, when comparing the dTG curve of 4W–SiO2-imp-py with the dTG curves of W–SiO2-py microspheres, the maxima are found at 236 and 247 °C, respectively, indicating that pyridine is released at slightly higher temperatures in the case of W–SiO2 microspheres prepared via the condensation approach, which may suggest a stronger interaction between pyridine and these microspheres.
In conjunction with the TG analysis of pyridine-adsorbed samples, the populations of Lewis and Brønsted acid sites can be correlated with the observed mass loss (Fig. 19S). According to the Lpy/Bpy ratios derived from FTIR, the quantities of Lpy and Bpy sites are summarized in Table 2. For example, the 12W–SiO2 catalyst contained 0.29 mmol g−1 of Bpy sites, whereas the 2W–SiO2 sample contained only 0.09 mmol g−1. Furthermore, the Lpy content in 12W–SiO2 was 0.28 mmol g−1, while the 2W–SiO2 sample exhibited 0.17 mmol g−1.
To evaluate the hydrophilic or hydrophobic character of the W–SiO2 microspheres, water vapor sorption measurements were performed.35,64–66 The resulting adsorption isotherms are presented in Fig. 10 and, according to the IUPAC classification, exhibit a Type V profile. All isotherms display a steep increase in adsorption at low relative pressures, which is characteristic of hydrophilic materials.66 Moreover, the individual W–SiO2 microsphere samples differ in the specific relative pressure range at which water uptake occurs. As illustrated in Fig. 10, for the 12W–SiO2 sample, water uptake begins at the lowest relative pressure (p/p0). As the tungsten content decreases, the onset of the steep adsorption shifts toward higher p/p0 values. This trend indicates that 12W–SiO2 exhibits a more hydrophilic character compared to 6W–SiO2 and 2W–SiO2.35,67,68 The values of water volume adsorbed on the W–SiO2 microsphere surface corresponds with the nitrogen adsorption/desorption isotherms (Fig. 5).
The results from water sorption experiments correlate with the water contact angles measured on the surface of W–SiO2 microspheres in the form of pellets. As illustrated in Fig. 21S, the water contact angle decreases with increasing tungsten content. The values for 2W–SiO2, 6W–SiO2, and 12W–SiO2 were 38.9, 29.6, and 13.3°, respectively. Due to high hydrophilicity, the quality of the drop captured on the surface of the 12W–SiO2 sample is poor.
3.2 Catalytic studies
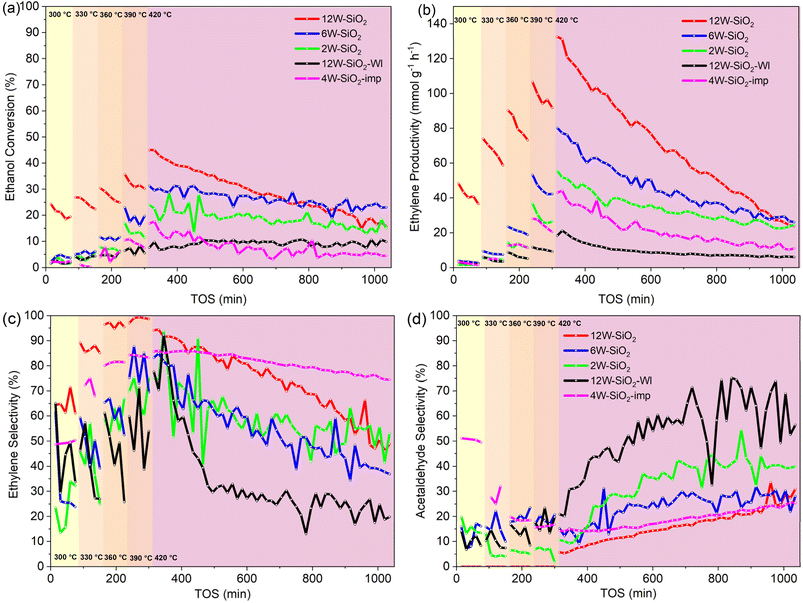
The potential application of W–SiO2 microspheres in catalysis was demonstrated on ethanol dehydration to ethylene. First, the ethanol dehydration reactions were performed at two different WHSV values (14.19 and 7.09 h−1) to check the occurrence of a mass transfer limitation (Fig. 22S). As illustrated, the initial conversion doubled when WHSV was decreased to 7.09 h−1.69 Also, Weisz–Prater and Mears criteria were well below 0.3 and 0.15, respectively, excluding possible mass transfer limitations (Tables 5S and 6S). Thus, catalytic studies were performed at 14.19 h−1 to ensure that conversion is well below 100% (i.e., under the kinetic regime).70,71The average ethanol conversions and ethylene selectivity from each five injections at the temperatures of 300–390 °C (Fig. 11a) are given in SI (Fig. 23S). As illustrated in Fig. 11a, the conversion of ethanol increased with temperature, reaching the highest initial conversion of 43% at 420 °C for the 12W–SiO2 sample. However, the 12W–SiO2 sample exhibited significant deactivation over time on stream (TOS), primarily due to coking, as discussed later. For the 6W–SiO2 and 2W–SiO2 catalysts, which have lower tungsten loadings, the conversion at 420 °C was 31% and 24%, respectively. These samples showed better stability with less prominent deactivation compared to the 12W–SiO2 sample.
The initial ethylene selectivity was fluctuating close to 100% for the 12W–SiO2 sample. Although ethylene selectivity decreased (in favor of acetaldehyde72) over time-on-stream (from 100% to 65% in 14 h), it remained higher than that of the 6W–SiO2 and 2W–SiO2 microspheres (Fig. 11c). Trace amounts of diethyl ether were detected across all samples, regardless of tungsten loading or reaction temperature; however, its selectivity remained negligible (<1%).
The high potential of W–SiO2 microspheres for ethanol dehydration is further demonstrated in Fig. 11b, which shows the initial ethylene productivity. The 12W–SiO2 sample achieved the highest initial ethylene productivity of 132.6 mmol g−1 h−1. With time on stream, however, ethanol conversion decreases and acetaldehyde selectivity increases. 6W–SiO2 and 2W–SiO2 samples with initial ethylene productivities of 80.2 and 55.6 mmol g−1 h−1, respectively, exhibited more stable ethanol conversion, so that the ethylene productivity across all catalyst samples plateaued at nearly the same value (25 mmol g−1 h−1) after 17 h TOS. A high initial ethylene productivity in the 12W–SiO2 catalyst can be ascribed to the high concentration of Brønsted acid sites as the initial ethylene productivities for 2W–SiO2, 6W–SiO2, and 12W–SiO2 samples correlate with the number of Brønsted acid sites (Table 2 and Fig. 24S).
The results obtained for W–SiO2 catalysts prepared by the microwave-assisted sol–gel technique contrast with the catalytic performance of the 12W–SiO2–WI catalyst prepared by the wet impregnation technique, which simulates an industrial catalyst. 12W–SiO2–WI reached only 10% ethanol conversion, with ethylene selectivity dropping rapidly over time on stream from ∼80 to 20% in parallel with an increase in acetaldehyde selectivity. Overall, maximum productivity of 12W–SiO2–WI reached only 21 mmol g−1 h−1 at 420 °C with a decreasing trend during TOS (6 mmol g−1 h−1 after 14 h) (Fig. 11b). Similarly, the 4W–SiO2-imp sample prepared via impregnation of pure silica microspheres synthesized via MW-assisted sol–gel technique exhibited lower initial ethanol conversion (17%) at 420 °C as well as lower initial ethylene productivity (43 mmol g−1 h−1) than W–SiO2 microspheres prepared via the one-pot approach (Fig. 11). Similarly, the ethylene productivity decline during TOS was evidenced resulting in a value of 11 mmol g−1 h−1, after 12 h (Fig. 11c). Interestingly, this sample shows relatively high and stable selectivity to ethylene.
Clearly, the well-dispersed active sites in W–SiO2 microspheres play a crucial role in achieving high catalytic activity as shown by a correlation between initial ethylene productivity normalized to the tungsten molar content and DRUV-vis band-edge energy (Fig. 25S). Samples prepared via impregnation exhibited lower band-edge energies and lower ethylene productivities than W–SiO2 catalysts prepared in one-pot. The tungsten surface density does not appear to be decisive (Fig. 25S).
If we normalize the activity of the catalysts to the molar content of tungsten (Fig. 26S), it is obvious that the space-time yield of ethylene is the highest in the case of the sample with the lowest tungsten content (520 mmol mmolW−1 h−1). This further confirms the need for highly dispersed tungsten species in silica (Fig. 8) and a balanced ratio of Lewis and Brønsted acid sites (Fig. 9) for catalytic purposes. However, due to the increasing acetaldehyde selectivity with time-on-stream, the decrease in space-time yield is evident (Fig. 26S). The increased selectivity toward acetaldehyde for 6W–SiO2 and, in particular, for 2W–SiO2 can be attributed to the competitive dehydrogenation of ethanol that could be promoted by the presence of Lewis acid sites.12,72–74
To enhance the stability of ethanol conversion and ethylene productivity over 12W–SiO2, inspired by Pampararo et al.,75 the reaction system was co-fed with O2 (20% of O2 in N2). The catalytic performance under these conditions was compared in terms of ethanol, ethylene, and acetaldehyde peak area evolution during TOS with the catalytic experiment performed in pure N2. As illustrated in Fig. 27S, the co-feeding with O2 led to significantly higher stability of ethanol and ethylene peak areas during TOS at 420 °C. However, as shown in Fig. 27S, a significant increase in acetaldehyde production has been observed, probably originating in the oxidative ethanol dehydrogenation pathway that has been activated in the presence of O2.76
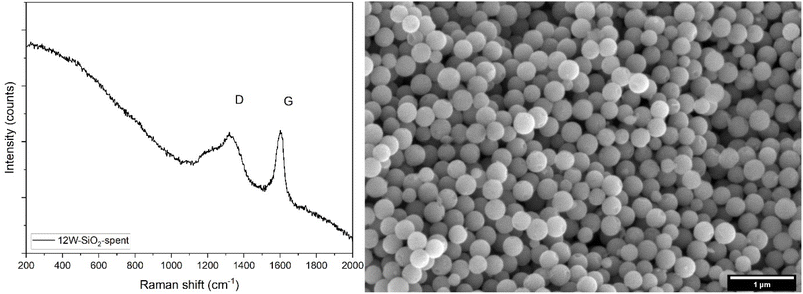
The spent catalysts were analyzed by Raman spectroscopy, XRD, SEM, XPS, and TG analysis to describe their structure and morphology after the catalytic reaction as well as their tendency to coke. The SEM image of the 12W–SiO2 spent catalyst (Fig. 12 and 28S) shows that the microspherical morphology remained unchanged in comparison to the fresh catalyst (Fig. 4S). The Raman spectra of spent 12W–SiO2 catalysts displayed the D and G bands characteristic of coke (Fig. 12). The Raman spectra of the spent 6W–SiO2 and 2W–SiO2 catalysts were affected by high fluorescence; however, weak D and G bands of carbon were observable, similar to 12W–SiO2 (Fig. 29S). All these data suggest that coking is the primary source of deactivation in the W–SiO2 materials presented herein, in agreement with the literature on tungsten silicate catalysts.77
The PXRD analysis of the spent 12W–SiO2 catalyst revealed weak diffraction maxima corresponding to crystalline triclinic WO3 (Fig. 29S; JCPDS card no. 01-071-0305), whereas the spent 6W–SiO2 and 2W–SiO2 catalysts exhibited diffraction patterns without any WO3-related signals. The presence of WO3 in the spent 12W–SiO2 catalyst can be considered as an additional factor that causes deactivation of the catalyst and pronounced ethanol conversion drop. In contrast, the 6W–SiO2 and 2W–SiO2 catalysts without WO3 diffractions exhibit more stable ethanol conversion.
The TG analysis of W–SiO2 spent microsphere catalysts was performed to follow the decomposition of the coke generated on the catalyst surface. As illustrated in Fig. 30S the spent catalysts exhibited the maximum weight loss between 350 and 600 °C. Specifically, the mass loss for 12W–SiO2, 6W–SiO2, and 2W–SiO2 spent catalyst was 12.4, 5.9, and 4.7%, respectively. According to the decomposition temperatures, the deposited carbon is mainly divided into soft coke (composed mainly of aliphatic compounds) with decomposition temperatures of 300–480 °C, and hard coke (composed mainly of aromatic compounds) with decomposition temperatures of 480–800 °C.78 Based on this definition we can assume the presence of both soft and hard coke on the surface of spent catalysts after dehydration reactions. XPS analysis of spent catalysts (Fig. 31S–33S) shows a higher carbon content on the surface compared to the fresh catalysts (Tables 2S and 7S). The carbon content decreases with the decreasing W content. This trend corresponds to the results from TGA of spent catalysts where the highest amount of coke was observed in the case of the 12W–SiO2 catalyst. In addition, the TGA curve of the spent 12W–SiO2–WI catalyst (Fig. 34S) exhibited a mass loss of 6.58% showing that, even despite its low catalytic activity, coking occurred as well.
Furthermore, catalyst deactivation can be correlated with coke formation on the surface of the spent catalysts (Fig. 30S) as well as with the Brønsted acid site content in the fresh catalysts (Fig. 13).60 It is well known that Brønsted acid sites are highly active in ethanol dehydration,26–28,59,60 and they are largely responsible for the high initial ethanol conversion observed for the 12W–SiO2 sample, for which the Lpy/Bpy ratio is 0.95 (Table 2). As the tungsten loading decreases, the relative concentration of Lewis acid sites with respect to Brønsted sites increases, leading to improved stability of ethanol conversion and lower coking.
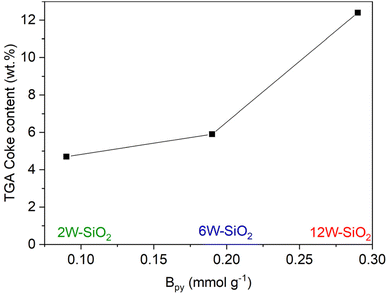 | ||
| Fig. 13 Correlation between the number of Brønsted acid sites (Bpy) and coke content in the spent catalysts. | ||
Interestingly, the W 4f XPS spectra of spent catalysts (Fig. 33S) show a higher contribution of lower oxidation states (W5+)11 than in the case of fresh catalysts (Fig. 6). Moreover, the increase in W5+ states (lower W6+/W5+ ratio) is more prominent in the case of 6W–SiO2 and 2W–SiO2 spent catalysts (Fig. 33S and Table 8S). This behavior may be explained as a consequence of exposure of W–SiO2 catalysts to a reductive environment (ethanol in nitrogen). Notably, samples with a lower W6+/W5+ ratio (Table 8S) exhibited a higher selectivity to acetaldehyde at the end of catalytic tests, i.e., were more active in ethanol dehydrogenation to acetaldehyde (Fig. 11d).
An additional parameter of critical relevance is the hydrophilicity of the fresh catalysts.35,79 Because ethanol dehydration inherently generates water as a co-product, the presence of water may adversely affect catalytic activity through competitive adsorption at active sites. As shown in Fig. 10, the water adsorption isotherms confirm the intrinsically hydrophilic nature of the W–SiO2 microspheres. Notably, water sorption measurements reveal a systematic decrease in hydrophilicity with decreasing tungsten loading (Fig. 10), which is further supported by water contact angle measurements (Fig. 21S). This behavior follows the observed catalytic performance and deactivation trends. However, it has to be considered that the hydrophilicity of the catalysts increases with the tungsten loading and with the number of acid sites. Thus, the effect of hydrophilicity cannot be unambiguously confirmed. Based on the obtained results it can be argued that catalysts with a higher ratio of Lewis to Brønsted acid sites, combined with lower hydrophilicity, exhibit enhanced catalytic stability and reduced susceptibility to deactivation under specific reaction conditions.
To investigate the possible regeneration of the 12W–SiO2 catalyst, three cycles of ethanol dehydration reactions at 420 °C with a WHSV of 7.09 h−1 were conducted in pure N2, each of them lasting for 12 h. After the first and second runs, the catalyst was heated in a reactor bed at 500 °C in the presence of oxygen (20%) for 2 hours to remove carbon residues and re-activate the catalyst. As illustrated in Fig. 14, the catalytic activity was almost fully recovered after each regeneration step. This supports the assumption that the deactivation is caused mainly by coking. In addition, the formation of crystalline WO3 species in 12W–SiO2 (Fig. 29S) during the catalytic reaction must also be considered as a reason for the slightly decreasing initial ethanol conversion within each regeneration step. As frequently suggested in the literature,35,60,79,80 the total amount of surface acid sites as well as the ratio between Lewis and Brønsted acid sites can significantly affect the catalyst's activity, selectivity, stability, and susceptibility to coking. Notably, Müller et al. demonstrated that a higher Lewis-to-Brønsted acid site ratio in zeolites enhanced catalytic performance in alcohol dehydration.60 Herein, based on the presented results, it can be inferred that the Brønsted/Lewis vibrational band ratio shown in Fig. 9 and Table 2 is the highest for the 12W–SiO2 microspheres as well as the content of Brønsted acid sites (Fig. 13), which may be connected with a greater tendency toward coking27,28,81 in this sample (Fig. 30S and Table 7S).
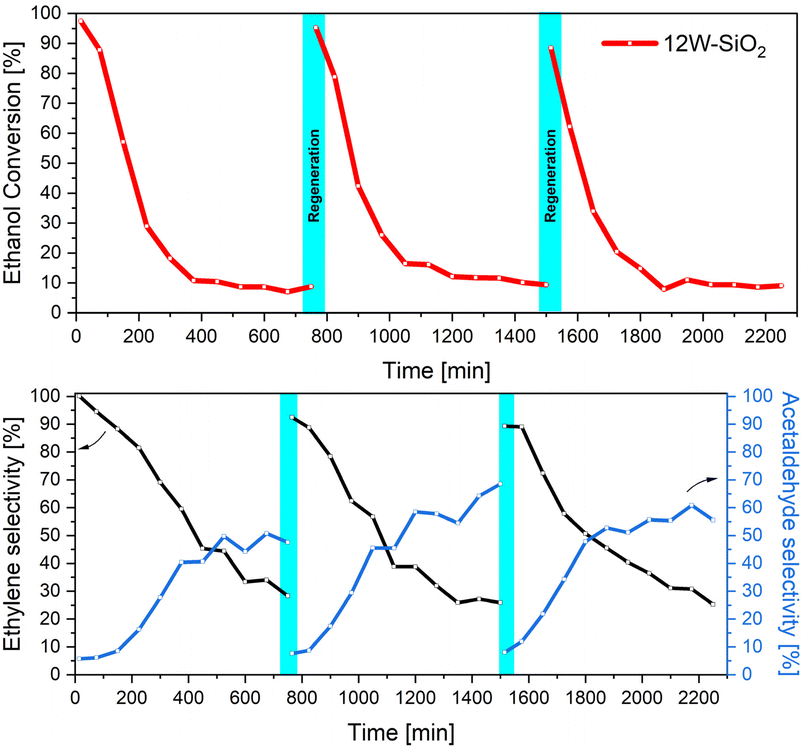 | ||
| Fig. 14 Regeneration of the 12W–SiO2 catalyst and ethanol conversions and product selectivity at 420 °C with a WHSV of 7.09 h−1. | ||
Table 3 summarizes the performance of W–SiO2 microspheres in ethanol dehydration (Fig. 11), compared with other relevant catalysts. As illustrated, the reactions were conducted under varying conditions, which must be taken into account when evaluating the performance of different catalysts. A detailed evaluation of the data reveals that W–SiO2 microspheres exhibit high catalytic performance in comparison to other tungsten-based catalysts. This is particularly evident when ethylene productivity is normalized either per gram of catalyst or per millimole of tungsten content. Specifically, the 2W–SiO2 sample demonstrates the highest initial ethylene productivity based on tungsten molar content, reaching 520 mmol mmolW−1 h−1. Notably, it maintains a high productivity of 230 mmol mmolW−1 h−1 even after 1000 minutes on stream (TOS). In terms of productivity per gram of catalyst, the 12W–SiO2 sample achieves the highest initial value of 133 mmol g−1 h−1, outperforming all other W-based catalysts evaluated. Overall, all W–SiO2 microspheres show high ethylene productivity when normalized to tungsten molar content.
| Catalyst | W content [wt%] | Catalyst amount [g] | Reaction temperature [°C] | WHSV [h−1] | Initial EtOH conversion [%] | Initial ethylene selectivity [%] | Ethylene productivity [mmol g−1 h−1] | Ethylene productivity [mmol mmolW−1 h−1] | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| a After 1000 min at TOS. b Tungsten content calculated from the 10WO390SiO2 formula. c Reaction performed at 1 MPa. | |||||||||
| 12W–SiO2 | 11.83 | 0.05 | 420 | 14.19 | 43 | 100 | 132.62 | 206 | This work |
| 6W–SiO2 | 6.46 | 0.05 | 420 | 14.19 | 31 | 83 | 80.21 | 226 | This work |
| 2W–SiO2 | 1.96 | 0.05 | 420 | 14.19 | 24 | 75 | 55.61 | 520 | This work |
| 2W–SiO2a | 1.96 | 0.05 | 420 | 14.19 | 15 | 53 | 24.60 | 230 | This work |
| W10KIT-6b | 23.7 | 1.5 | 360 | 9.47 | 55.3 | 80.3 | 43.14 | 34 | 83 |
| 36-STA/SiO2c | 27.6 | n.a. | 240 | 9.6 | 86.9 | 60 | 108.73 | 72 | 1 |
| WO3/MCF-Si | 13.5 | 0.1 | 400 | 3.1 | 99.7 | 95 | 63.81 | 87 | 13 |
| WO3/SBA-15 | 13.5 | 0.1 | 400 | 3.1 | 99 | 94 | 62.75 | 85 | 6 |
| SiAl(HIPE) | — | — | 400 | 11.8 | 99 | 94 | 327.99 | — | 84 |
The comparison with other common catalysts is not straightforward. Zeolites, alumina, and amorphous aluminosilicates have mostly been tested at lower temperatures and WHSV,27,79,80,82 hampering direct quantitative comparison. Here, hierarchically porous aluminosilicate monoliths (SiAl-(HIPE)) were tested in ethanol dehydration under reaction conditions that most closely matched those applied to the W–SiO2 (Table 3) and exhibited a better ethylene productivity than W–SiO2 catalysts presented herein. In addition, commercial γ-Al2O3 was used for comparison of catalytic activity under the same conditions as those applied to W–SiO2 at temperatures of 300–390 °C (Fig. 23S). As expected, γ-Al2O3 exhibited a higher catalytic activity in terms of ethanol conversion, reaching 92% at 390 °C. Interestingly, however, the 12W–SiO2 catalyst showed a higher selectivity toward ethylene under these conditions (98 vs. 94% for alumina and 12W–SiO2 at 390 °C, respectively).
In summary, although ethanol dehydration is most commonly promoted by aluminosilicate- or alumina-based catalysts,85 the W–SiO2 microspheres presented here demonstrate stimulating catalytic behavior in this reaction. As demonstrated in this work, the highly homogeneous dispersion of WOx species together with the well-defined surface acidity play a crucial role in achieving high catalytic activity compared with tungsten-based catalysts prepared via conventional impregnation methods (Table 3). Arguably, given that tungsten-containing catalysts are widely applied in a broad range of reactions,8–12,46,86 the properties of the W–SiO2 microspheres reported here may also stimulate interest in their potential use in other catalytic processes, such as olefin metathesis, olefin epoxidation, and in the upgrading of glucose or glycerol.
4 Conclusions
In this study, we report the successful one-pot sol–gel synthesis of tungsten silicate microspheres. The synthesis approach involves microwave-assisted preparation of a tungsten 2,6-naphthalene dicarboxylate precursor solution derived from tungsten hexacarbonyl, followed by room-temperature condensation with (3-aminopropyltriethoxy)silane. This process yields hybrid tungsten silicate microspheres, which, upon calcination, form inorganic, microporous silica-based structures containing homogeneously distributed amorphous tungstate species possessing Lewis and Brønsted acidity. The uniform dispersion of tungstate is attributed to the optimized reaction conditions and sol–gel condensation, offering significant advantages over conventional impregnation methods.Catalysts with tungsten loadings ranging from 2 to 12 wt% were evaluated for ethanol dehydration to ethylene. All samples exhibited notable activity and high selectivity, with initial ethanol conversion rates of 43%, 31%, and 24% for 12W–SiO2, 6W–SiO2, and 2W–SiO2, respectively. Ethylene selectivity initially reached 100%, gradually decreasing to 65% over time on stream (TOS). Based on tungsten molar content, the 2W–SiO2 sample showed the highest initial ethylene productivity of 520 mmol mmolW−1 h−1 (14.6 g mmolW−1 h−1) and maintained a high value of 230 mmol mmolW−1 h−1 (6.44 g mmolW−1 h−1) after 1000 minutes TOS, indicating good long-term catalytic stability. The 12W–SiO2 sample achieved the highest initial ethylene productivity per gram of catalyst (133 mmol g−1 h−1), below that of conventional (silica-)alumina-based dehydration catalysts, but clearly outperforming other tungsten–silica-based catalysts reported in the literature.
The most significant deactivation was observed for 12W–SiO2, primarily due to coking, caused by the high amount of Brønsted acid sites. Catalysts with a lower tungsten content exhibited a lower hydrophilicity, higher Lpy/Bpy ratio and more stable catalytic performance. Importantly, the homogeneous dispersion of active sites was found to be a key factor in catalytic efficiency providing significantly higher activity of W–SiO2 microspherical catalysts obtained from one-step MW-assisted sol–gel condensation compared to the 12W–SiO2–WI and 4W–SiO2-imp samples prepared via impregnation. These findings underscore the promising performance of W–SiO2 microspheres and their potential as efficient catalysts for ethanol dehydration. Furthermore, the presence of highly dispersed tungstate active sites in these materials suggests promising applicability in heterogeneous catalysis, for example in olefin metathesis, expanding their utility beyond ethanol conversion.
Author contributions
David Skoda: conceptualization, methodology, formal analysis, investigation, validation, visualization, writing – original draft, funding acquisition, resources, project administration. Tomas Pokorny: conceptualization, methodology, investigation, formal analysis, visualization, validation, writing – review & editing. Barbora Hanulikova: investigation, formal analysis, writing – review & editing. Ales Styskalik: methodology, investigation, formal analysis, validation, writing – review & editing, resources. Zuzana Hlavenkova: investigation, visualization. Lucie Simonikova: investigation. Jan Varada: investigation. Lucie Leonova: investigation, formal analysis, visualization. Ivo Kuritka: writing – review & editing, funding acquisition, resources. Claude Poleunis: investigation, formal analysis. Damien P. Debecker: formal analysis, conceptualization, methodology, writing – review & editing, resources.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta08046k.Acknowledgements
This work was supported by project LUAUS23085, funded by the Ministry of Education, Youth and Sports of the Czech Republic (MEYS CR) within the INTER-EXCELLENCE II program. The support by the MEYS CR within the DKRVO (RP/CPS/2024-28/007) project is gratefully acknowledged. The authors acknowledge the project Quantum Materials for Applications in Sustainable Technologies (QM4ST), CZ.02.01.01/00/22_008/0004572 by Program Johannes Amos Comenius, Excellent Research call. We also acknowledge CF CryoEM and CF NMR of CIISB, Instruct-CZ Centre, supported by MEYS CR (LM2023042) and European Regional Development Fund-Project “UP CIISB” (No. CZ.02.01.01/00/23_015/0008175). CzechNanoLab project LM2023051, funded by MEYS CR, is gratefully acknowledged for the financial support of the XPS measurements at CEITEC Nano Research Infrastructure. This work was financially supported by the Grant Agency of Masaryk University (GAMU) under the grant number MUNI/A/1298/2022.References
- H. Li, Z. Yu, Z. Zhang, S. Li, Z. Shi, C. Ni, L. Cao, H. Du, Q.-X. Luo and F. Wang, Ind. Eng. Chem. Res., 2024, 63, 7624–7635 CrossRef CAS.
- G. Pampararo and D. P. Debecker, in Encyclopedia of Green Chemistry, Elsevier, 2025, pp. 304–323 Search PubMed.
- P. T. Benavides, U. R. Gracida-Alvarez, K. Richa, J. Port and T. R. Hawkins, Bioresour. Technol., 2025, 430, 132565 CrossRef CAS PubMed.
- C. D'Alessandro and H. Besada, in The Palgrave Handbook of Contemporary International Political Economy, Palgrave Macmillan UK, London, 2019, vol. 16301, pp. 377–389 Search PubMed.
- M. Duan, C. Hu, H. Li, Y. Chen, R. Chen, W. Gong, Z. Lu, N. Zhang, R. Long, L. Song and Y. Xiong, JACS Au, 2022, 2, 1160–1168 CrossRef CAS PubMed.
- P. Trongjitraksa, Y. Klinthongchai, P. Praserthdam and B. Jongsomjit, J. Taiwan Inst. Chem. Eng., 2023, 152, 105168 CrossRef CAS.
- R. Al-Faze, A. Finch, E. F. Kozhevnikova and I. V. Kozhevnikov, Appl. Catal., A, 2020, 597, 117549 CrossRef CAS.
- P. Sudarsanam, N. K. Gupta, B. Mallesham, N. Singh, P. N. Kalbande, B. M. Reddy and B. F. Sels, ACS Catal., 2021, 11, 13603–13648 CrossRef CAS.
- T. Z. H. Gani, Z. J. Berkson, R. Zhu, J. H. Kang, J. R. Di Iorio, K. W. Chan, D. F. Consoli, S. K. Shaikh, C. Copéret and Y. Román-Leshkov, Nature, 2023, 617, 524–528 CrossRef CAS PubMed.
- S. Lwin and I. E. Wachs, ACS Catal., 2014, 4, 2505–2520 CrossRef CAS.
- J. J. Wiesfeld, R. Gaquere and E. J. M. Hensen, ACS Sustain. Chem. Eng., 2019, 7, 7552–7562 CrossRef CAS.
- N. V. Vlasenko, G. R. Kosmambetova, E. V. Senchylo, P. Kúš, K. Veltruská and P. E. Strizhak, ChemSusChem, 2025, 18(5), e202401800 CrossRef CAS PubMed.
- P. Trongjitraksa, M. Yazdanpanah, M. Fereidooni, P. Praserthdam and B. Jongsomjit, S. Afr. J. Chem. Eng., 2025, 51, 180–187 Search PubMed.
- C. v. Schalkwyk, A. Spamer, D. J. Moodley, T. Dube, J. Reynhardt and J. M. Botha, Appl. Catal., A, 2003, 255, 121–131 CrossRef.
- P. Promchana, K. Choojun, W. Limphirat, Y. Poo-arporn and T. Sooknoi, Appl. Catal., A, 2023, 650, 118972 CrossRef CAS.
- S.-H. Chai, B. Yan, L.-Z. Tao, Y. Liang and B.-Q. Xu, Catal. Today, 2014, 234, 215–222 CrossRef CAS.
- M. P. Conley, V. Mougel, D. V. Peryshkov, W. P. Forrest, D. Gajan, A. Lesage, L. Emsley, C. Copéret and R. R. Schrock, J. Am. Chem. Soc., 2013, 135, 19068–19070 CrossRef CAS PubMed.
- Y. Zengin, B. Kaya, M. Safak Boroglu and I. Boz, Ind. Eng. Chem. Res., 2023, 62, 1852–1864 CrossRef CAS.
- S. Maksasithorn, P. Praserthdam, K. Suriye and D. P. Debecker, Microporous Mesoporous Mater., 2015, 213, 125–133 CrossRef CAS.
- S. Maksasithorn, P. Praserthdam, K. Suriye, M. Devillers and D. P. Debecker, Appl. Catal., A, 2014, 488, 200–207 CrossRef CAS.
- D. P. Debecker, M. Stoyanova, U. Rodemerck, F. Colbeau-Justin, C. Boissère, A. Chaumonnot, A. Bonduelle and C. Sanchez, Appl. Catal., A, 2014, 470, 458–466 CrossRef CAS.
- Y. Tao, O. De Luca, B. Singh, A. J. Kamphuis, J. Chen, P. Rudolf and P. P. Pescarmona, Mater. Today Chem., 2020, 18, 100373 CrossRef CAS.
- A. Guntida, K. Suriye, J. Panpranot and P. Praserthdam, Catal. Today, 2020, 358, 354–369 CrossRef CAS.
- S. Boonpai, S. Wannakao, J. Panpranot, B. Jongsomjit and P. Praserthdam, J. Phys. Chem. C, 2020, 124, 15935–15943 CrossRef CAS.
- J. González, J. A. Wang, L. F. Chen, M. E. Manríquez and J. M. Dominguez, J. Phys. Chem. C, 2017, 121, 23988–23999 CrossRef.
- P. Kerdnoi, C. Autthanit, N. Chitpong and B. Jongsomjit, Bull. Chem. React. Eng. Catal., 2020, 15, 96–103 CrossRef CAS.
- A. Styskalik, V. Vykoukal, L. Fusaro, C. Aprile and D. P. Debecker, Appl. Catal. B Environ., 2020, 271, 118926 CrossRef CAS.
- C. P. Nash, A. Ramanathan, D. A. Ruddy, M. Behl, E. Gjersing, M. Griffin, H. Zhu, B. Subramaniam, J. A. Schaidle and J. E. Hensley, Appl. Catal., A, 2016, 510, 110–124 CrossRef CAS.
- V. Zacharopoulou and A. Lemonidou, Catalysts, 2017, 8, 2 CrossRef.
- P. González-Navarrete, M. Schlangen, X. Wu and H. Schwarz, Chem.–Eur. J., 2016, 22, 3077–3083 CrossRef PubMed.
- M. Duan, C. Hu, H. Li, Y. Chen, R. Chen, W. Gong, Z. Lu, N. Zhang, R. Long, L. Song and Y. Xiong, JACS Au, 2022, 2, 1160–1168 CrossRef CAS PubMed.
- S. Lwin, Y. Li, A. I. Frenkel and I. E. Wachs, ACS Catal., 2016, 6, 3061–3071 CrossRef CAS.
- R. B. Demuner, J. G. Soares Santos Maia, A. R. Secchi, P. A. Melo, R. W. do Carmo and G. S. Gusmão, Ind. Eng. Chem. Res., 2019, 58, 2717–2726 CrossRef CAS.
- A. I. Osman, J. K. Abu-Dahrieh, A. Abdelkader, N. M. Hassan, F. Laffir, M. McLaren and D. Rooney, J. Phys. Chem. C, 2017, 121, 25018–25032 CrossRef CAS.
- A. Styskalik, I. Kordoghli, C. Poleunis, A. Delcorte, Z. Moravec, L. Simonikova, V. Kanicky, C. Aprile, L. Fusaro and D. P. Debecker, J. Mater. Chem. A, 2020, 8, 23526–23542 RSC.
- B. T. Loc, L. N. Q. Tu and N. Q. Long, Vietnam J. Catal. Adsorpt., 2021, 10, 79–83 CrossRef.
- D. Skoda, B. Hanulikova, A. Styskalik, V. Vykoukal, P. Machac, P. Urbanek, E. Domincova Bergerova, L. Simonikova and I. Kuritka, J. Ind. Eng. Chem., 2022, 107, 320–332 CrossRef CAS.
- D. Skoda, R. Zhu, B. Hanulikova, A. Styskalik, V. Vykoukal, P. Machac, L. Simonikova, I. Kuritka, C. Poleunis, D. P. Debecker and Y. Román-Leshkov, ACS Catal., 2023, 13, 12970–12982 CrossRef CAS PubMed.
- D. Skoda, K. Kuzelova, R. B. Pricilla, B. Hanulikova, M. Urbanek, A. Styskalik, T. Pokorny, I. Doroshenko, L. Simonikova and I. Kuritka, J. Ind. Eng. Chem., 2025, 148, 502–513 CrossRef CAS.
- R. López and R. Gómez, J. Sol-Gel Sci. Technol., 2012, 61, 1–7 CrossRef.
- M. Jacquemin, M. J. Genet, E. M. Gaigneaux and D. P. Debecker, ChemPhysChem, 2013, 14, 3618–3626 CrossRef CAS PubMed.
- N. Fairley, V. Fernandez, M. Richard-Plouet, C. Guillot-Deudon, J. Walton, E. Smith, D. Flahaut, M. Greiner, M. Biesinger, S. Tougaard, D. Morgan and J. Baltrusaitis, Appl. Surf. Sci. Adv., 2021, 5, 100112 CrossRef.
- S. Lowell, J. E. Shields, M. A. Thomas and M. Thommes, in Characterization of Porous Solids and Powders: Surface Area, Pore Size and Density, Springer Netherlands, 2004, pp. 58–81 Search PubMed.
- M. Luisa Ojeda, J. Marcos Esparza, A. Campero, S. Cordero, I. Kornhauser and F. Rojas, Phys. Chem. Chem. Phys., 2003, 5, 1859 RSC.
- E. I. Ross-Medgaarden and I. E. Wachs, J. Phys. Chem. C, 2007, 111, 15089–15099 CrossRef CAS.
- E. Mazoyer, N. Merle, A. De Mallmann, J.-M. Basset, E. Berrier, L. Delevoye, J.-F. Paul, C. P. Nicholas, R. M. Gauvin and M. Taoufik, Chem. Commun., 2010, 46, 8944 RSC.
- T. Takkawatakarn, K. Suriye, B. Jongsomjit, J. Panpranot and P. Praserthdam, Catal. Today, 2020, 358, 345–353 CrossRef CAS.
- A. V. Colusso, A. McDonagh and M. B. Cortie, Surf. Interface Anal., 2018, 50, 1384–1388 CrossRef CAS.
- Y. Liu, S. Shrestha and W. E. Mustain, ACS Catal., 2012, 2, 456–463 CrossRef CAS.
- K. M. McCreary, A. T. Hanbicki, G. G. Jernigan, J. C. Culbertson and B. T. Jonker, Sci. Rep., 2016, 6, 19159 CrossRef CAS PubMed.
- N. Minh Vuong, D. Kim and H. Kim, Sci. Rep., 2015, 5, 11040 CrossRef PubMed.
- Q. Xin, L. Jiang, S. Yu, S. Liu, D. Yin, L. Li, C. Xie, Q. Wu, H. Yu, Y. Liu and Y. Liu, J. Phys. Chem. C, 2021, 125, 18170–18179 CrossRef CAS.
- J. G. Howell, Y.-P. Li and A. T. Bell, ACS Catal., 2016, 6, 7728–7738 CrossRef CAS.
- D. P. Debecker, B. Schimmoeller, M. Stoyanova, C. Poleunis, P. Bertrand, U. Rodemerck and E. M. Gaigneaux, J. Catal., 2011, 277, 154–163 CrossRef CAS.
- R. De Clercq, M. Dusselier, C. Poleunis, D. P. Debecker, L. Giebeler, S. Oswald, E. Makshina and B. F. Sels, ACS Catal., 2018, 8, 8130–8139 CrossRef CAS.
- L.-T. Weng, Appl. Catal., A, 2014, 474, 203–210 CrossRef CAS.
- E. L. Lee and I. E. Wachs, J. Phys. Chem. C, 2007, 111, 14410–14425 CrossRef CAS.
- C. Martín, P. Malet, G. Solana and V. Rives, J. Phys. Chem. B, 1998, 102, 2759–2768 CrossRef.
- B. Li, K. Leng, Y. Zhang, J. J. Dynes, J. Wang, Y. Hu, D. Ma, Z. Shi, L. Zhu, D. Zhang, Y. Sun, M. Chrzanowski and S. Ma, J. Am. Chem. Soc., 2015, 137, 4243–4248 CrossRef CAS PubMed.
- J. M. Müller, G. C. Mesquita, S. M. Franco, L. D. Borges, J. L. de Macedo, J. A. Dias and S. C. L. Dias, Microporous Mesoporous Mater., 2015, 204, 50–57 CrossRef.
- C. A. Emeis, J. Catal., 1993, 141, 347–354 CrossRef CAS.
- V. Zholobenko, C. Freitas, M. Jendrlin, P. Bazin, A. Travert and F. Thibault-Starzyk, J. Catal., 2020, 385, 52–60 CrossRef CAS.
- J. M. R. Gallo, C. Bisio, G. Gatti, L. Marchese and H. O. Pastore, Langmuir, 2010, 26, 5791–5800 CrossRef CAS PubMed.
- G. Rother, A. G. Stack, S. Gautam, T. Liu, D. R. Cole and A. Busch, J. Phys. Chem. C, 2020, 124, 15188–15194 CrossRef CAS.
- S. W. Rutherford, Langmuir, 2025, 41, 25086–25099 CrossRef CAS PubMed.
- S. W. Rutherford, Langmuir, 2025, 41, 20502–20515 CrossRef CAS PubMed.
- K. Zhang, R. P. Lively, J. D. Noel, M. E. Dose, B. A. McCool, R. R. Chance and W. J. Koros, Langmuir, 2012, 28, 8664–8673 CrossRef CAS PubMed.
- M. Thommes, S. Mitchell and J. Pérez-Ramírez, J. Phys. Chem. C, 2012, 116, 18816–18823 CrossRef CAS.
- P. B. Weisz and C. D. Prater, Interpretation of Measurements in Experimental Catalysis, in Advances in Catalysis, 1954, pp. 143–196 Search PubMed.
- F. Schüth, M. D. Ward and J. M. Buriak, Chem. Mater., 2018, 30, 3599–3600 CrossRef.
- C. Perego, Catal. Today, 1999, 52, 133–145 CrossRef CAS.
- J. F. DeWilde, C. J. Czopinski and A. Bhan, ACS Catal., 2014, 4, 4425–4433 CrossRef CAS.
- Y. Zhang, L. Qi, Y. Li, T. Yang, D. M. Meira, C. Dun, H. Hu, H. Chen, S. Xu, J. J. Urban, A. D. Sadow, T. Kobayashi, L. Qi, P. Tian and A. T. Bell, ACS Catal., 2024, 14, 15204–15220 CrossRef CAS.
- Q. Meng, F. Chen, X. Li, C. Qiu, Q. Yuan and T. Wang, J. Catal., 2026, 453, 116571 CrossRef CAS.
- G. Pampararo, Z. Hlavenková, A. Styskalik and D. P. Debecker, Catal. Sci. Technol., 2024, 14, 4912–4926 RSC.
- J. Pang, M. Yin, P. Wu, X. Li, H. Li, M. Zheng and T. Zhang, Green Chem., 2021, 23, 7902–7916 RSC.
- O. Verdes, V. Sasca, A. Popa, M. Suba and S. Borcanescu, Catal. Today, 2021, 366, 123–132 CrossRef CAS.
- Y. Sun, L. Wei, Z. Zhang, H. Zhang and Y. Li, Energy Fuels, 2023, 37, 1657–1677 CrossRef CAS.
- L. Leonova, Z. Moravec, P. Sazama, J. Pastvova, L. Kobera, J. Brus and A. Styskalik, ChemCatChem, 2023, 15(13), e202300449 CrossRef CAS.
- T. K. Phung and G. Busca, Catal. Commun., 2015, 68, 110–115 CrossRef CAS.
- J. Bi, X. Guo, M. Liu and X. Wang, Catal. Today, 2010, 149, 143–147 CrossRef CAS.
- A. Styskalik, I. Kordoghli, C. Poleunis, A. Delcorte, C. Aprile, L. Fusaro and D. P. Debecker, Microporous Mesoporous Mater., 2020, 297, 110028 Search PubMed.
- H. Zhu, A. Ramanathan, J.-F. Wu and B. Subramaniam, ACS Catal., 2018, 8, 4848–4859 CrossRef CAS.
- D. P. Debecker, C. Boissière, G. Laurent, S. Huet, P. Eliaers, C. Sanchez and R. Backov, Chem. Commun., 2015, 51, 14018–14021 RSC.
- T. K. Phung, L. Proietti Hernández, A. Lagazzo and G. Busca, Appl. Catal., A, 2015, 493, 77–89 CrossRef CAS.
- D. Hoegaerts, B. F. Sels, D. E. De Vos, F. Verpoort and P. A. Jacobs, Catal. Today, 2000, 60, 209–218 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |