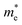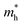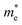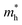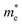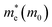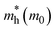Open Access Article
Open Access ArticleQuasi-1D selenohalides: first-principles insights into thermoelectric and photovoltaic applications
Prakash
Govindaraj
 and
Hern
Kim
and
Hern
Kim
 *
*
Department of Energy Science and Technology, Environment Waste Recycle Institute, Myongji University, Yongin, Gyeonggi-do 17058, Republic of Korea. E-mail: hernkim@mju.ac.kr
First published on 11th February 2026
Abstract
The rapid search for advanced functional materials is increasingly essential to meet the rising global energy demands, secure long-term energy solutions, and achieve a sustainable future. In this work, we systematically investigate the structural, thermoelectric, and photovoltaic properties of quasi-one-dimensional selenohalides XSeHa (X = Sb, Bi; Ha = Cl, Br) using density functional theory. The unique chemical environments, consisting of 1D layers stacked via weak van der Waals interactions, give rise to pronounced anisotropic electron and phonon transport properties alongside nearly isotropic optical behavior. The synergistic combination of favorable electronic features and lone-pair electrons yields a high-power factor (1.74 mW m−1 K−2) and a low lattice thermal conductivity (0.31 W m−1 K−1), resulting in a remarkable thermoelectric figure of merit of up to 0.81 at 600 K for BiSeBr. Additionally, strong optical absorption driven by the imaginary dielectric function and favorable excitonic properties achieves a spectroscopic limited maximum efficiency of 31.13% for SbSeBr. Based on these results, BiSeBr and SbSeBr are suggested as promising candidates for experimental exploration in thermoelectric and photovoltaic applications, respectively. This study not only demonstrates the potential of selenohalides but also provides a thorough assessment of their stability and synthesizability to guide future investigations.
1. Introduction
Eco-friendly energy conversion and storage technologies are being actively developed to address the ongoing energy crisis and mitigate the environmental challenges associated with global warming. In this context, thermoelectric (TE) and photovoltaic (PV) technologies have witnessed remarkable progress in recent years.1,2 For thermoelectric devices, the efficiency is primarily governed by the dimensionless figure of merit,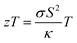 , where σ is the electrical conductivity, S is the Seebeck coefficient, κ is the total thermal conductivity (the sum of the electronic, κe and lattice part, κL) and T is the absolute temperature.3 Typically, zT> 1 is considered as a desirable parameter for practical applications. Several Te-based chalcogenides, such as PbTe (zT = 1.6 at 773 K), GeTe (zT = 2.0 at 700 K), and Bi2Te3 (zT = 1.4 at 380 K)4–6 already demonstrate high performance. Despite these achievements, the scarcity and high cost of Te hinder large-scale commercialization, motivating the search for alternative TE materials composed of earth-abundant elements.7 In recent years, the discovery and optimization of such materials have been propelled by both experimental and computational approaches.8,9 To further enhance zT, two strategies have been widely employed: (i) improving electronic transport through band structure engineering, resonant level doping, and carrier concentration optimization; and (ii) suppressing lattice thermal conductivity via nanostructuring, defect engineering, and grain boundary manipulation.10,11
, where σ is the electrical conductivity, S is the Seebeck coefficient, κ is the total thermal conductivity (the sum of the electronic, κe and lattice part, κL) and T is the absolute temperature.3 Typically, zT> 1 is considered as a desirable parameter for practical applications. Several Te-based chalcogenides, such as PbTe (zT = 1.6 at 773 K), GeTe (zT = 2.0 at 700 K), and Bi2Te3 (zT = 1.4 at 380 K)4–6 already demonstrate high performance. Despite these achievements, the scarcity and high cost of Te hinder large-scale commercialization, motivating the search for alternative TE materials composed of earth-abundant elements.7 In recent years, the discovery and optimization of such materials have been propelled by both experimental and computational approaches.8,9 To further enhance zT, two strategies have been widely employed: (i) improving electronic transport through band structure engineering, resonant level doping, and carrier concentration optimization; and (ii) suppressing lattice thermal conductivity via nanostructuring, defect engineering, and grain boundary manipulation.10,11
In parallel, extensive efforts have been devoted to improving the solar cell conversion efficiency and enabling effective solar energy harvesting.12,13 Lead-based halide perovskites have emerged as outstanding photovoltaic absorbers due to their high absorption coefficients and exceptional conversion efficiencies, which now exceed 26%. However, their intrinsic toxicity and limited long-term chemical and thermal stability disturb their large-scale commercialization.14 Other classes of perovskites have also contributed significantly to photovoltaics; for example, the overall efficiency of this family has improved by nearly 47%, from 14.1% in 2013 to 26.95% in 2025. Current research strategies primarily focus on enhancing the open-circuit voltage, improving stability, and optimizing interfacial engineering between the solar absorber and charge-transport layers. Beyond these experimental approaches, first-principles methods and high-throughput computational screening are playing a central role in accelerating materials discovery and deepening the understanding of structure–property relationships, thereby advancing both TE and PV technologies.15,16
Multi-anion chalcohalides represent an emerging class of semiconductors with promising potential in energy-related technologies, owing to their inherent properties such as structural tunability, phase stability, and unique electronic features. These materials have been explored for diverse applications, including photodetectors, solar cells, and batteries.17–19 From a structural aspect, they typically consist of one or more metals combined with divalent chalcogens and monovalent halogens. Among them, the orthorhombic SbSI and BiSI have gained considerable attention in recent years.20–22 Originally reported by E. Dönges in 1950, these compounds have been investigated for nearly 75 years across various functional applications, with a predominant focus on their electronic and optical properties.23 More recently, Chauhan et al. examined Janus antimony chalcohalide monolayers for piezoelectric, thermoelectric, photovoltaic, and photocatalytic applications, while Bafekry et al. studied the optical and thermoelectric properties of 2D SbXY (X = Se, Te; Y = Br, I).24,25 However, most of the existing studies emphasize the optical properties of low-dimensional chalcohalides, whereas the thermoelectric and photovoltaic characteristics of their parent bulk quasi-1D analogues, XSeHa (X = Sb, Bi; Ha = Cl, Br), remain largely unexplored to the best of our knowledge.
To address this research gap, we present a detailed investigation on the thermoelectric and photovoltaic properties of quasi-one-dimensional selenohalides within the framework of density functional theory (DFT). This study not only explores the structure–property relationships relevant to thermoelectric and photovoltaic applications but also examines the structural peculiarities, diverse stability aspects, and synthesizability that are critical for guiding experimental realization. These factors are comprehensively analyzed to establish a reliable foundation for practical implementation. For clarity, the paper is organized as follows: Section 2 outlines the computational methodology; Section 3 presents the results and discussion, covering structural, stability, and electronic properties (Sections 3.1–3.3), thermoelectric response (Section 3.4), and photovoltaic performance (Section 3.5); and Section 4 concludes with the key findings and outlook.
2. Computational methods
2.1. First-principles calculations
All the density functional theory (DFT) calculations were performed using the Vienna Ab initio Simulation Package (VASP) under the projector augmented wave (PAW) method.26,27 To treat the core and valence electrons, the Perdew, Burke, and Ernzerhof (PBE) functional under generalized gradient approximation was used.28 The van der Waals (vdW) dispersion term (DFT-D3) with Becke-Johnson damping function was used to treat the vdW interlayer interaction.29 Based on the performed convergence test, a kinetic energy cutoff of 550 eV and a Monkhorst–Pack 5 × 3 × 7 k-mesh was applied.30 The tight energy and force convergence criteria were set as 1 × 10−8 eV and 1 × 10−7 eV Å−1, respectively. All the crystal structures were picturized using the VESTA package.31 The electronic structure was plotted using the sumo plotting tool.32 Furthermore, the LOBSTER code was used for crystal orbital Hamiltonian population analysis.33 The convex hull and chemical potential calculations were performed using the pydefect code.342.2. Thermoelectric property calculations
The phonon dispersion spectra were calculated by the finite difference method implemented in the phonopy code.35,36 The single-point calculations on displaced structures were performed using VASP to obtain second-order interatomic force constants. A 3 × 1 × 2 supercell was utilized for phonon related calculations. And a Monkhorst–Pack 3 × 3 × 3 mesh was applied.30 The phonon frequencies and normalized eigenvectors were computed. The eigenvectors were obtained from the band.yaml output file generated with eigenvector printing enabled (EIGENVECTORS = TRUE). These eigenvectors were post-processed using custom Python scripts to evaluate atom-resolved vibrational contributions based on the squared norms of the eigenvector components, which were subsequently used to construct the projected phonon dispersion plots. Furthermore, the lattice thermal conductivity was computed by solving the full linearized Boltzmann transport equation iteratively using the ShengBTE code.37 The thirdorder.py script was used to generate the displaced structures.38 The q-mesh and cutoff radius convergence tests were employed with ΔκLof 0.05 W m−1 K−1. The electron transport properties were calculated by adopting the highly efficient and reliable AMSET code.39 Here, the code solves the semiclassical Boltzmann transport equation using the BoltzTraP2 code.40 Furthermore, the relaxation times of carriers were computed by considering multiple scattering mechanisms, such as acoustic phonons, polar optical phonons and ionized impurities. These calculations were performed by considering deformation potentials, elastic constants and dielectric constants. A converged interpolation factor of 110 was used. BiSeCl was considered as a representative system for all the phonon and electron-related convergence tests, which are given in Fig. S1 and S2 of the SI.2.3. Photovoltaic property calculations
Considering the larger memory requirements for GW calculations, the frequency-dependent dielectric function and optical absorption spectra were obtained through independent particle approximation (IPA) on the top of PBE-D3. The VASPKIT tool was used to post-process the optical properties of the materials.41 The widely used Spectroscopic Limited Maximum Efficiency (SLME) approach was utilized to access the maximum radiative limit of energy conversion with respect to thickness.423. Results and discussion
3.1. Crystal structure and bonding analysis
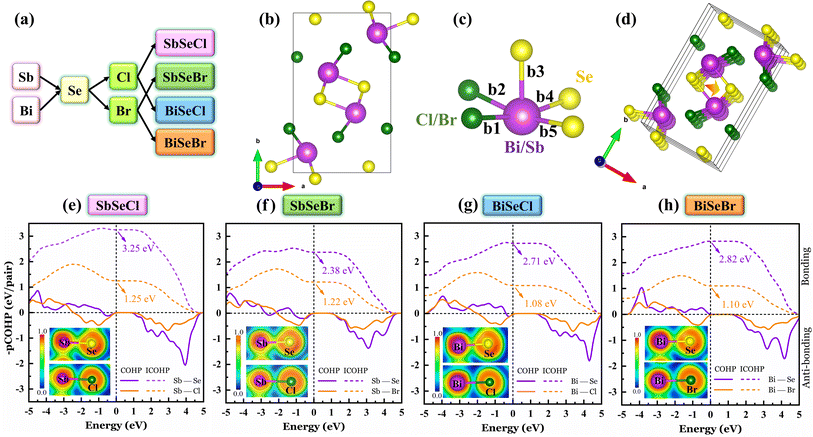
In this work, four selenohalides with chemical formula XSeHa (X = Bi, Sb; Ha = Cl, Br) are studied, as indicated in Fig. 1(a). These selenohalides crystallize in an orthorhombic structure with the Pnam space group (refer to Fig. 1(b)). Experimentally, such selenohalides exhibit a needle-like morphology, which originates from their one-dimensional structural peculiarities i.e., an infinite chain extending along the z-direction.43,44 These chains comprise repeating units of two distorted square pyramids as depicted in Fig. 1(c). Each pyramid contains one Bi/Sb, two halide, and three chalcogen atoms. The bond lengths are listed in Table S1 for better comparison. The chalcogen atoms serve as bridging units between pyramids, linking them together to form infinite chains as represented in Fig. 1(d). The chains are held together by weak van der Waals interactions, resulting in pronounced anisotropic behavior in the material's physical properties. The atomic structures were optimized using the DFT-D3 method, which includes dispersion corrections to accurately capture the weak interchain interactions. The relaxed lattice parameters show better agreement with previous theoretical and experimental reports (see Table 1).| Materials | Lattice parameters (Å) | Methods | Reference | ||
|---|---|---|---|---|---|
| a | b | c | |||
| SbSeCl | 7.554 | 10.752 | 3.939 | Theo. (PBE + D3) | Present work |
| SbSeBr | 8.275 | 10.217 | 3.996 | Theo. (PBE + D3) | Present work |
| 8.300 | 9.750 | 3.950 | Expt. | 47 | |
| 8.300 | 10.200 | 3.950 | — | 48 | |
| BiSeCl | 7.086 | 11.611 | 4.005 | Theo. (PBE + D3) | Present work |
| 7.700 | 9.870 | 4.000 | Expt. | 49 | |
| BiSeBr | 8.220 | 10.365 | 4.105 | Theo. (PBE + D3) | Present work |
| 8.180 | 10.470 | 4.110 | Expt. | 23 | |
To investigate the bonding characteristics of XSeHa, Crystal Orbital Hamiltonian Population (COHP) analysis was performed along with the Electron Localization Function (ELF), as illustrated in Fig. 1(e–h). The COHP method provides insights into the bonding and antibonding interactions between atomic pairs by projecting the Hamiltonian onto the selected orbitals. The integrated COHP (ICOHP) values quantify the bonding strength: higher ICOHP values indicate stronger bonding interactions, while lower values correspond to weaker bonds.45 Here we have considered all the s, p, and d orbitals for cations and anions. Across all the structures, the ICOHP values for Bi/Sb–Se bonds were notably higher than those for Bi/Sb–Cl/Br bonds, suggesting stronger bonding between the cations and Se compared to the halogen atoms.
The insets of Fig. 1(e–h) show the ELF analysis which complements the COHP results by visualizing regions of electron localization, thereby providing deeper insight into the nature of bonding both within and between chains. The ELF values range from 0 to 1, where values of 0, 0.5, and 1 correspond to no localization, partial localization, and complete localization, respectively. The partial electron localization between Sb/Bi and the chalcogen atoms reveals the covalent bonding character, which is consistent with the high ICOHP values. In contrast, significantly lower electron localization was found between Sb/Bi and the halogen atoms, supporting the interpretation of weaker, possibly more ionic interactions. Additionally, 3D ELF isosurfaces plotted along the (001) plane at an isosurface value of 0.0005 reveal the presence of charge density beneath the square pyramids, indicating the existence of lone pair electrons on the cationic centers (refer to Fig. S3). These lone pairs induce significant distortions in the square pyramidal geometry, resulting in unequal bond lengths between the cations and anions (see Table S1). Such structural asymmetry is often associated with enhanced lattice anharmonicity.46
3.2. Phase stability
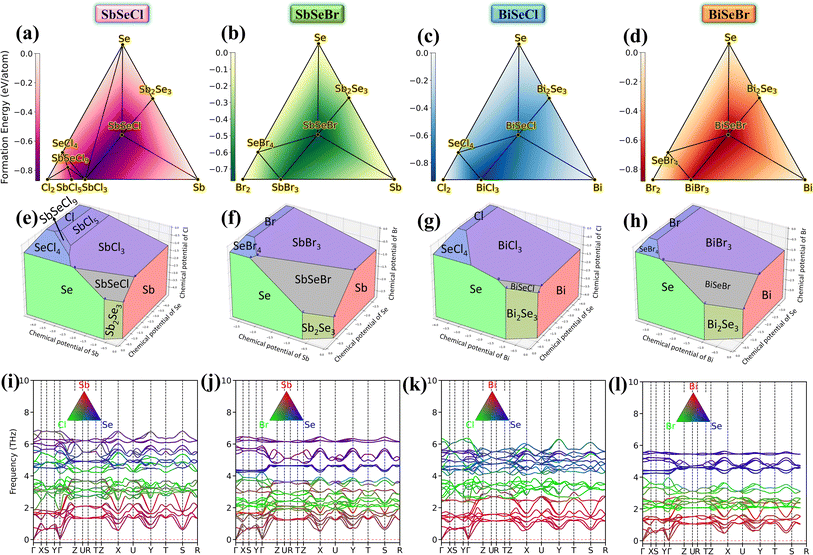
The combined analysis of the convex hull and chemical potential diagram (CPD) will establish the compound's thermodynamic stability and the permissible synthesis window. Hence, the convex hull and chemical potential diagram (CPD) calculations are carried out, and the results are illustrated in Fig. 2(a–h). The convex hull analysis, Fig. 2(a–d), depicts that the compounds lying on the hull are predicted to be thermodynamically stable. For example, SbCl5, SbCl3, SeCl4, SbSeCl9, SbSeCl and Sb2Se3 are found to be stable while constructing convex hull for SbSeCl (refer to Fig. 2(a)). Also, the convex hull illustrates the formation energy of the considered phases. The dark and lighter shades within the different colored convex hull indicate the lower (more favorable) and higher (less favorable) formation energies, respectively. All the selenohalides are observed in the darker shades, except BiSeCl. In the case of BiSeCl, BiCl3 is the most prone phase to be formed during the synthesis process. However, all the interesting selenohalides have negative formation energy from −0.60 to −0.97 eV/atom, indicating their successful formation under experimental conditions. Hence, suitable approaches should be adopted to eradicate the formation of BiCl3 as a secondary phase in BiSeCl synthesis.Furthermore, the CPDs show the stability window of the selenohalides in terms of the corresponding elemental chemical potentials. The shaded polyhedral regions represent the allowed combinations of chemical potentials where each phase is stable. The targeted selenohalides occupy smaller regions, especially in the case BiSeCl. This reflects their sensitivity to growth conditions, implying that precise control of precursor ratios and the growth environment is required to avoid competing phases.
To facilitate the probability of synthesis, the cohesive energy (Ecoh) is calculated using the relation Ecoh=(EPnSeHa − EPn − ESe − EHa)/4, where EPnSeHa, EPn, ESe, and EHa represent the energies of the selenohalide and constituent isolated atoms such as pnictogen (Sb/Bi), selenium, and halogen (Cl/Br), respectively.50 The calculated Ecoh for SbSeCl, SbSeBr, BiSeCl and BiSeBr is −3.82, −3.68, −3.85 and −3.71 eV/atom, respectively. These values are comparatively higher than those of the experimentally reported chalcohalides like BiSeI (−2.14 eV/atom) which is synthesized by physical vapor deposition and ball milling methods. Hence, it is inferred that these selenohalides can also be synthesized.
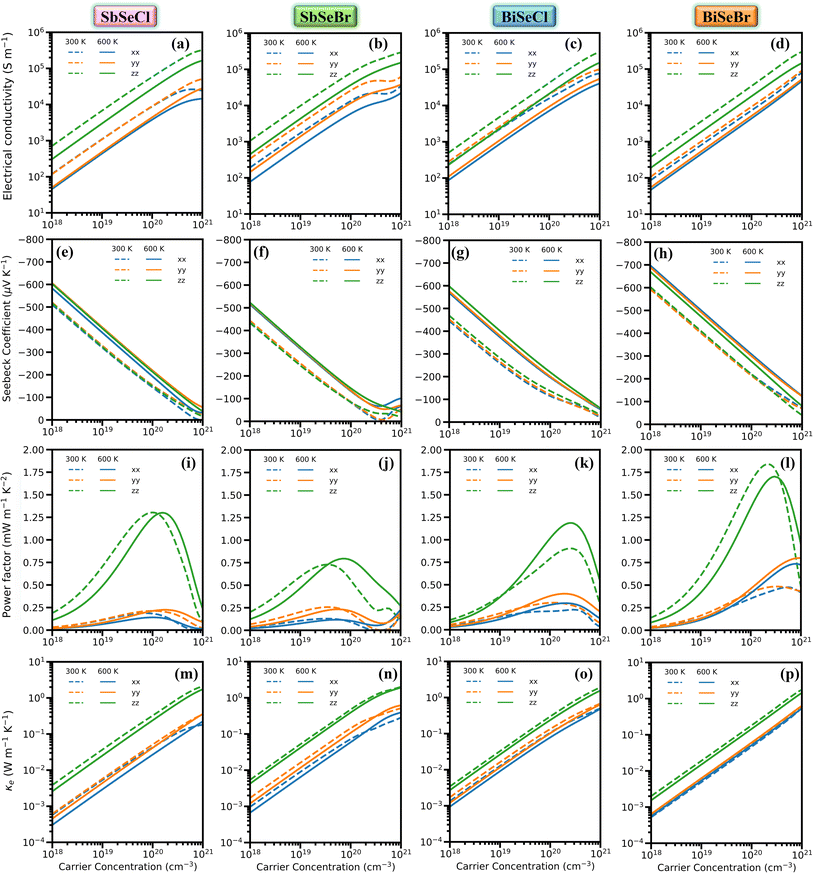
To understand the dynamical stability of the selenohalides, the projected phonon dispersion spectra were calculated and are depicted in Fig. 2(i–l). No imaginary/negative frequency is observed in all the compounds, ensuring their dynamical stability. The unit cell comprises 12 atoms, resulting in 36 phonon modes, including 3 acoustic and 33 optical modes. The highest frequency tends to decrease linearly as the pnictogen/halogen varies from Cl to Br or Sb to Bi, ascribed to the increasing atomic mass of the corresponding constituent elements. The Sb/Bi, Cl/Br and Se atoms predominantly contribute to the low, mid and high frequency modes respectively. For a specific mode, the highest frequency along different crystallographic directions follows the trend x < y < z for all the selenohalides. For instance, in SbSeCl, the highest frequency of the TA mode is 0.57, 0.69 and 1.38 THz along the x-, y-, and z-directions, respectively. This directional variation in phonon frequencies indicates pronounced anisotropy in phonon transport. In crystalline solids, acoustic phonon modes are the primary contributors to heat conduction due to their abrupt variation in frequency. Notably, the frequencies also vary significantly with respect to q-points, particularly near the Γ-point. Consequently, acoustic modes are expected to contribute significantly to the phonon velocity, which generally correlates with higher κL. Additionally, band crossing is avoided between the highest acoustic and lowest optical modes in all the investigated compounds. This feature may reflect the weak bonding characteristics of the crystal, but it could also signify the presence of strong anharmonicity and enhanced phonon–phonon scattering between these modes. A detailed discussion on the implications of this feature in relation to phonon dispersion will be presented in Section 3.4.2.
The mechanical stability of the selenohalides was evaluated by computing their elastic constants, which were obtained using the stress–strain approach and are summarized in Table 2. In 2014, Mouhat et al. reformulated the Born stability criteria for all the crystal systems. For an orthorhombic lattice, the necessary and sufficient conditions are: C11 > 0, C11C22 > C212, C11C22C33 + 2C12C13C23 − C11C223 − C22C213 − C33C212 > 0, C44 > 0, C55 > 0, and C66 > 0.51 All these six conditions are satisfied by the studied selenohalides, confirming their mechanical stability. Additional mechanical properties are provided in Section S1 and Table S2 of the SI. Notably, the Pugh's ratio (B/G) for all compounds exceeds the critical value of 1.75, indicating their ductile nature.21 Furthermore, their Poisson's ratios lie in the range of 0.3–0.4, suggesting mixed bonding characteristics.52
| Elastic constants | SbSeCl | SbSeBr | BiSeCl | BiSeBr |
|---|---|---|---|---|
| C 11 | 24.08 | 30.16 | 23.59 | 32.35 |
| C 12 | 20.15 | 19.21 | 20.08 | 20.78 |
| C 13 | 13.67 | 12.87 | 13.31 | 14.50 |
| C 21 | 20.15 | 19.21 | 20.08 | 20.78 |
| C 22 | 23.84 | 24.74 | 28.02 | 22.84 |
| C 23 | 15.64 | 13.29 | 22.69 | 16.59 |
| C 31 | 13.67 | 12.87 | 13.31 | 14.50 |
| C 32 | 15.64 | 13.29 | 22.69 | 16.59 |
| C 33 | 68.22 | 58.71 | 72.08 | 56.36 |
| C 44 | 16.87 | 15.25 | 18.91 | 13.95 |
| C 55 | 15.24 | 12.86 | 19.36 | 13.51 |
| C 66 | 6.73 | 5.42 | 7.58 | 4.75 |
3.3. Electronic structure and effective mass
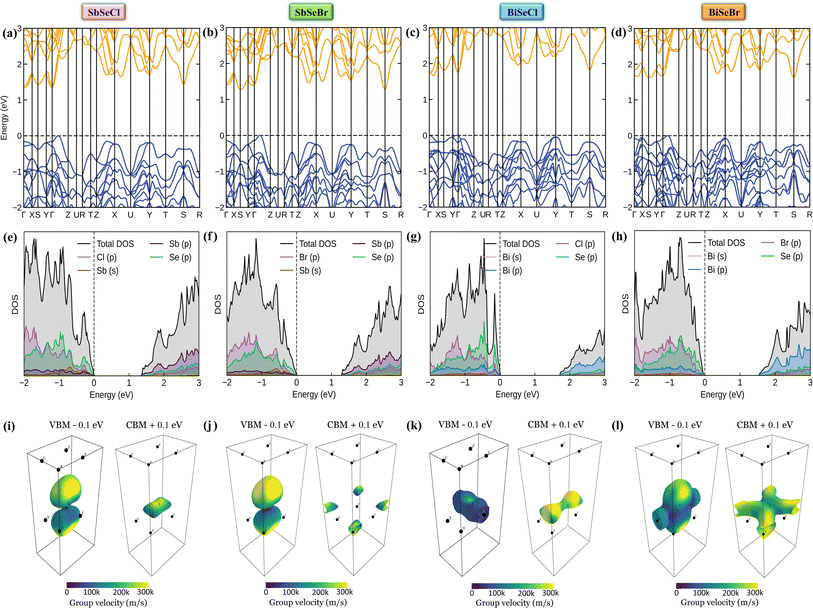
Based on the calculation results, no significant changes in the band edges were observed while considering the hybrid exchange correlational functional (HSE06), except for an increase in the band gap (refer to Fig. S4). Hence, the electronic properties of the studied selenohalides are calculated using the PBE functional with van der Waals correction. Fig. 3 displays the calculated electronic band structures alongside the projected density of states (DOS) as well as the Fermi surfaces, to assess the electronic transport and optical characteristics. The VBM and CBM are located at Γ-Z and Γ for SbSeCl, Γ-Z and S for SbSeBr, Γ and Γ-X for BiSeCl, and Γ-Z and S–Y for BiSeBr, confirming the presence of indirect band gaps in all the investigated selenohalides (see Fig. 3(a–d)). The calculated band gap spans from 1.28 to 1.66 eV, which are well consistent with earlier reports (refer to Table 3). The sharp band edges result in smaller carrier effective masses rather than flat edges, resulting in high carrier mobility and electrical conductivity. As inferred from the projected DOS, shown in Fig. 3(e and h), the VBM of all the selenohalides is primarily contributed by the Se p-orbital while the CBM is dominated by the Sb/Bi p-orbital.For further insights, the Fermi surface below and above 0.1 eV from the band edges is displayed in Fig. 3(i–l). The color scale represents the carrier group velocities. In SbSeCl, a complete hole pocket is observed, extending from Γ to Z, with a corresponding downward dispersion arising from the symmetry of the Brillouin zone (BZ). Similarly, a pillow-shaped electron pocket is noted from Γ to X. In comparison, the Fermi surface of BiSeBr is widely spread over the entire BZ. Holes have the highest group velocity in antimony selenohalides, while in bismuth selenohalides it is electrons. Furthermore, the carrier effective masses along different crystallographic directions are calculated using the AMSET code (refer to Table 4).39 The calculated carrier effective masses reveal a clear asymmetry between electrons (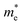 ) and holes (
) and holes (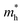 ), which has direct relationship to charge transport. Across all selenohalides and transport directions (Γ-X, Γ-Y, Γ-Z), holes are consistently heavier than electrons, except for BiSeBr (x-direction). For instance, in BiSeCl,
), which has direct relationship to charge transport. Across all selenohalides and transport directions (Γ-X, Γ-Y, Γ-Z), holes are consistently heavier than electrons, except for BiSeBr (x-direction). For instance, in BiSeCl, 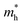 ranges from 1.69 to 2.43 m0, whereas
ranges from 1.69 to 2.43 m0, whereas 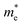 varies only between 0.17 and 0.65 m0. Here m0 is the rest mass of the electron. This difference indicates that the mobility of electrons will be much higher than holes, favoring n-type conduction. In case of electrons, the favorable transport channels occur along Γ-Z direction for all the four selenohalides: 0.29 m0 (SbSeCl), 0.23 m0 (SbSeBr), 0.17 m0 (BiSeCl), and 0.36 m0 (BiSeBr). These values are significantly lower than those along the other directions, highlighting z-axis as the most favorable conduction pathway for electrons. In the case of holes, SbSeCl has heavy holes along Γ-X (2.41 m0) and Γ-Y (3.39 m0) than Γ-Z (0.95 m0). In SbSeBr, largest hole mass of 3.92 m0 is observed along Γ-X, while the smallest value of 1.21 m0 along Γ-Z. These results suggest that z-direction not only favors n-type transport but also for p-type. Among the studied compounds, SbSeBr and BiSeCl are particularly essential. SbSeBr exhibits heavy hole effective mass (up to 3.92 m0), indicating of limited hole mobility, while retaining relatively light electrons along Γ-Z (0.23 m0), favoring efficient n-type conduction. On the other hand, BiSeCl displays the lowest electron effective mass within series (0.17 m0 along Γ-Z) together with moderately heavy holes, suggesting an intrinsic preference for electron-dominated transport.
varies only between 0.17 and 0.65 m0. Here m0 is the rest mass of the electron. This difference indicates that the mobility of electrons will be much higher than holes, favoring n-type conduction. In case of electrons, the favorable transport channels occur along Γ-Z direction for all the four selenohalides: 0.29 m0 (SbSeCl), 0.23 m0 (SbSeBr), 0.17 m0 (BiSeCl), and 0.36 m0 (BiSeBr). These values are significantly lower than those along the other directions, highlighting z-axis as the most favorable conduction pathway for electrons. In the case of holes, SbSeCl has heavy holes along Γ-X (2.41 m0) and Γ-Y (3.39 m0) than Γ-Z (0.95 m0). In SbSeBr, largest hole mass of 3.92 m0 is observed along Γ-X, while the smallest value of 1.21 m0 along Γ-Z. These results suggest that z-direction not only favors n-type transport but also for p-type. Among the studied compounds, SbSeBr and BiSeCl are particularly essential. SbSeBr exhibits heavy hole effective mass (up to 3.92 m0), indicating of limited hole mobility, while retaining relatively light electrons along Γ-Z (0.23 m0), favoring efficient n-type conduction. On the other hand, BiSeCl displays the lowest electron effective mass within series (0.17 m0 along Γ-Z) together with moderately heavy holes, suggesting an intrinsic preference for electron-dominated transport.
3.4. Thermoelectric properties
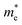 varies between 0.17 and 0.65 m0, while
varies between 0.17 and 0.65 m0, while 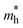 lies between 1.69 and 2.43 m0. The lighter electrons can move faster, and thus higher n-type electrical conductivity is observed. For example, along the z-direction, σ = 42
lies between 1.69 and 2.43 m0. The lighter electrons can move faster, and thus higher n-type electrical conductivity is observed. For example, along the z-direction, σ = 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 244 S m−1 at 300 K for n-type, while p-type has only 4772 S m−1. Furthermore, the anisotropy in σ mirrors the trend observed in directional effective mass. For example, in SbSeBr, the
244 S m−1 at 300 K for n-type, while p-type has only 4772 S m−1. Furthermore, the anisotropy in σ mirrors the trend observed in directional effective mass. For example, in SbSeBr, the 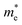 is lowest along Γ-Z (
is lowest along Γ-Z (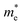 =0.23 m0), which corresponds to the highest conductivity along the z-direction at 300 K (78
=0.23 m0), which corresponds to the highest conductivity along the z-direction at 300 K (78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 757 S m−1). Similarly, SbSeCl exhibits moderate
757 S m−1). Similarly, SbSeCl exhibits moderate 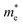 values (0.29–1.24 m0), resulting in intermediate conductivity values ranging from 8826 to 57
values (0.29–1.24 m0), resulting in intermediate conductivity values ranging from 8826 to 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 910 S m−1. Interestingly, temperature has a pronounced effect on conductivity, with all compounds showing a substantial decrease in σ, when heated from 300 to 600 K. This reduction can be attributed to enhanced carrier scattering, which restricts the mobility of the carrier. For instance, the n-type conductivity of BiSeCl drops from 42
910 S m−1. Interestingly, temperature has a pronounced effect on conductivity, with all compounds showing a substantial decrease in σ, when heated from 300 to 600 K. This reduction can be attributed to enhanced carrier scattering, which restricts the mobility of the carrier. For instance, the n-type conductivity of BiSeCl drops from 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 244 S m−1 (300 K, z-direction) to 21
244 S m−1 (300 K, z-direction) to 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 064 S m−1 (600 K, z-direction). A similar trend is also evident in p-type materials, where the σ of SbSeCl decreases from 11
064 S m−1 (600 K, z-direction). A similar trend is also evident in p-type materials, where the σ of SbSeCl decreases from 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 137 S m−1 at 300 K to 4670 S m−1 at 600 K, along the z-direction. Overall, SbSeBr and BiSeCl show high σ, consistent with their relatively small
137 S m−1 at 300 K to 4670 S m−1 at 600 K, along the z-direction. Overall, SbSeBr and BiSeCl show high σ, consistent with their relatively small 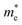 along specific transport directions. The σ values for all selenohalides are given in the table for clarity (refer to Table S3 and S4). SbSeCl shows moderate performance, while the conductivity of BiSeBr is affected by its larger carrier effective masses. The correlation between directional effective mass and anisotropic conductivity highlights the critical role of band structure engineering in optimizing the thermoelectric performance.
along specific transport directions. The σ values for all selenohalides are given in the table for clarity (refer to Table S3 and S4). SbSeCl shows moderate performance, while the conductivity of BiSeBr is affected by its larger carrier effective masses. The correlation between directional effective mass and anisotropic conductivity highlights the critical role of band structure engineering in optimizing the thermoelectric performance.
The calculated Seebeck coefficient as a function of carrier concentration is displayed in Fig. 4(e–h). For n-type BiSeBr, along the x-direction, at T = 300 K and n = 1 × 1020 cm−3, the S value ranges from −83 to −220 µV K−1, while at 600 K, the magnitude increases significantly, reaching as high as −308 µV K−1. This trend confirms that the thermal excitation of the carriers affects the S of the n-type system. Among the selenohalides, BiSeBr exhibits the largest negative S value, suggesting superior thermoelectric potential upon n-type doping, while SbSeCl and SbSeBr show relatively lower magnitudes (refer to Table S5 and S6). For the p-type case, at 300 K, the S values lie between 147 and 298 µV K−1, while at 600 K, they increase markedly to 227–386 µV K−1. Notably, p-type BiSeCl exhibits the highest S value at 300 K (∼298 µV K−1 along the y-direction), which exceeds 370 µV K−1 (along the y- and z-directions) at 600 K. The observed temperature dependence of S aligns well with the general behavior of semiconductors, where thermal excitation broadens the carrier distribution and alters the conductivity. Anisotropy effects are considerable, suggesting direction-dependent electron transport properties, which are common in layered materials.
To examine the coupling effect of S and σ, the power factor is calculated as a function of carrier concentration and displayed in Fig. 4(i–l). It reveals clear distinctions between n- and p-type doping accompanied by strong anisotropy across crystallographic directions. In Bi-based systems, n-type systems consistently exhibit higher PF than their counterparts, both at 300 and 600 K, highlighting the superior role of lighter electrons in facilitating higher electrical transport. At 300 K, for n-type doping, the highest PF occurs predominantly along the z-direction, with SbSeCl, SbSeBr, BiSeCl, and BiSeBr reaching 1.30, 0.56, 0.79 and 1.64 mW m−1 K−2, respectively. This noticeable PF along the z-direction can be related to the interplay between the intermediate electron effective mass and favorable transport along the chain direction. In contrast, PF along the x- and y-directions remains below 0.42 mW m−1 K−2, underscoring the highly anisotropic nature of charge transport. At elevated temperature (600 K), the PF of n-type systems increases, except for SbSeCl and BiSeBr (z-direction), but the directional trends are retained. For example, the z-direction remains the most favorable in SbSeCl (1.27 mW m−1 K−2), SbSeBr (0.78 mW m−1 K−2), BiSeCl (1.01 mW m−1 K−2), and BiSeBr (1.35 mW m−1 K−2). In contrast, the p-type systems display significantly smaller PF values along all directions and temperatures. At 300 K, the maximum p-type PF is observed for SbSeBr along the y-direction (0.55 mW m−1 K−2), while other compounds exhibit considerably lower values, typically below 0.38 mW m−1 K−2. Increasing the temperature to 600 K, alters the PF further, with most values falling in the range of 0.06 to 0.45 mW m−1 K−2. The comparatively lower performance in p-type systems can be directly linked to the heavier hole effective mass, which suppresses the mobility and limits the electrical conductivity. Refer to Tables S7 and S8 for better comparison.
The calculated κe as a function of carrier concentration is shown in Fig. 4(m–p). The obtained κe reveals significant temperature dependence, carrier-type dependence, and notable directional anisotropy. Across all selenohalides and directions, κe decreases considerably as temperature increases. This behavior is ascribed to the enhanced phonon–electron scattering at higher temperatures, thereby reducing the contribution of charge carriers to thermal transport. In n-type SbSeBr (y-direction), κe drops from 0.144 W m−1 K−1 at 300 K to 0.108 W m−1 K−1 at 600 K (∼25% reduction). Similarly, in p-type SbSeBr (y-direction), κe decreases from 0.136 W m−1 K−1 to 0.066 W m−1 K−1 (∼51% reduction). A strong directional dependence of κe is observed in most compounds, particularly for n-type doping. For n-type BiSeCl at 300 K, κe along the z-direction is 0.298 W m−1 K−1, which is ∼2.8 times higher than that along the x-direction i.e., 0.105 W m−1 K−1. Similarly, for p-type SbSeCl, at 300 K, κe is 0.069 W m−1 K−1 along the y-direction, while κe is 0.023 W m−1 K−1 along the x-direction, showing strong anisotropy. For the studied compounds, n-type systems exhibit significantly higher κe compared to their counterparts, especially along the z-direction, associated with the behavior of σ. In n-type SbSeBr, at 300 K, along the z-direction, the observed κe is 0.473 W m−1 K−1, which is nearly seven times higher than in p-type i.e., 0.066 W m−1 K−1. This trend is consistent at 600 K, although the absolute values tend to decrease. The higher electronic thermal conductivity in n-type materials indicates that electrons have better mobility than holes, likely originating from conduction-band curvature differences and lower effective masses of electrons. The κe values for all selenohalides are listed in Table S9 and S10.
To uncover the origin of κL further, the individual phonon mode contribution to κL is computed. For all the selenohalides, along all the directions, the percentage of acoustic mode contribution to κL is illustrated in Fig. 5(i–l) and that for optical modes is shown in Fig. S7. As expected, irrespective of the systems and directions, the acoustic modes show dominant contribution to κL, ascribed to the higher phonon group velocities. For example, the three acoustic modes collectively contribute almost 60% of κL in the case of SbSeCl at 300 K along the x-direction. It is worth noting that the influence of the TA1 mode to κL along the y-direction is much higher than in the considered cases. In addition, the TA1 mode exhibits the strongest anisotropy, for example, in SbSeCl, significant differences are observed along each direction, i.e., 31% along x, 41% along y and 21% along z, highlighting the crucial role of transverse vibrations. Low-frequency optical mode e.g., O1 show appreciable contributions (≈9–3% each), reflecting their coupling with the acoustic modes and indicating their importance in the phonon–phonon scattering process. Mid-frequency optical modes like O6 and O7 modes yield larger portion of κL along the z-direction. The contributions from other modes are not appreciable, except for O10 (6.9%), O11 (6.5%) and O17 (5.4%) of BiSeCl along the x-direction. These results suggest that the absolute magnitudes of mode contributions vary slightly between the materials, but the overall vibrational anisotropy and mode dependence remain remarkably similar. In each case, acoustic modes dominate heat-carrying capacity, whereas optical modes provide scattering channels that limit phonon transport. This consistent trend indicates a common underlying mechanism controlling their lattice dynamics, implying that the difference in lattice thermal conductivity among these compounds is primarily governed by secondary factors like mass contrast, bonding strength, and anharmonicity.
To demonstrate the effect of nanostructuring on κL, the cumulative lattice thermal conductivity (κcum) as a function of phonon mean free path (MFP) at 600 K was calculated. As depicted in Fig. 5(m–p), phonon transport is dominated along the z-direction, due to the presence of chains. For all the four systems, the majority of κL is accumulated below 50 nm MFPs, indicating that the phonon transport is largely governed by short- to intermediate-range phonons. Interestingly, κL along the z-axis reaches its maximum contribution within a short range, indicating the need for fine-nanostructuring to utilize the systems at their maximum potential.
3.5. Photovoltaic properties
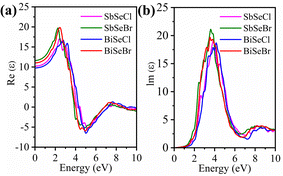
The imaginary part, shown in Fig. 7(b), is directly related to the optical absorption coefficient and provides information about the inter-band electronic transitions. All compounds exhibit a sharp absorption onset near 1.5 eV, which aligns with their semiconducting band gaps, confirming their effective capability to absorb visible light. The highest peaks are observed for SbSeBr (Im(ε) = 21.1) and BiSeBr (19.6) followed by SbSeCl (19) and BiSeCl (18.7) suggesting the strong absorption in Br-based compounds. The stronger optical transitions in Br-containing compounds can be attributed to the reduced band dispersion, leading to increased oscillator strength. Multiple peaks between 3.0 and 5.0 eV indicate the presence of complex inter-band transitions. The broader response around 8–9 eV suggests the potential of these materials to harvest photons across a wide spectral range, making them promising for both visible-light photovoltaics and optoelectronic devices.
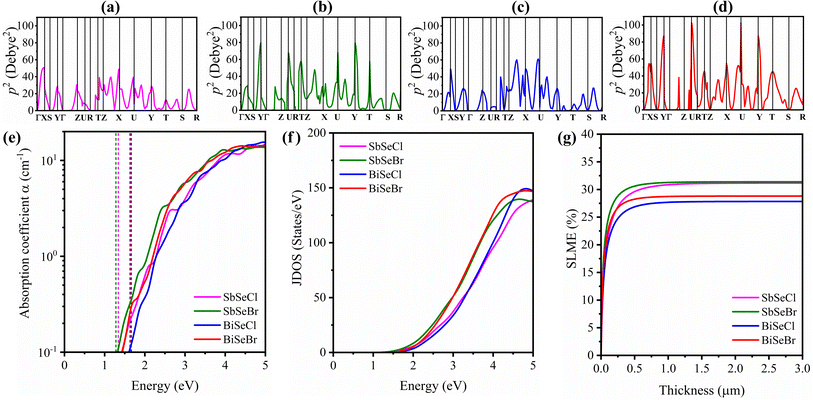
The optical absorption coefficient is a critical parameter in evaluating the light-harvesting potential of solar absorbers. To this end, the optical absorption spectra are derived from the frequency-dependent dielectric function for all the selenohalides, as shown in Fig. 8(e). Generally, a sharp increase in absorption is observed when the incident photon energy approaches the Eg of the material, a key feature of an efficient photovoltaic material. A pronounced exponential rise in absorption is evident near their respective band edges, consistent with the calculated Eg values. This behaviour is closely linked with the joint density of states (JDOS), where flatter band edges lead to a steeper rise in JDOS, near the band gap, thereby enhancing the absorption (Fig. 8(f)). Hence, the absorption trends are strongly correlated with the JDOS profile. Interestingly, selenohalides with Br halogen have higher absorption than Cl-based compounds, which can be associated with the behaviour of P2.
To assess the photovoltaic potential of the selenohalides, spectroscopic limited maximum efficiency (SLME) calculations as a function of film thickness are performed and depicted in Fig. 8(g). This SLME approach accounts for various energetic sequences (dipole forbidden, dipole allowed and indirect Eg), photon absorptivity, and dependence of radiative recombination losses. This also facilitates the understanding of photovoltaic efficiency as a function of absorption layer thickness. However, a major limitation of this method is the exclusion of non-radiative recombination, which significantly limits the efficiency of most of the indirect band gap absorbers. In these calculations, the dielectric response computed using the PBE + D3 functional. For all the compounds, SLME increases rapidly with thickness and saturates thereafter. Notably, under 1-sun illumination AM1.5 G, the Sb-based selenohalides achieve higher SLME values up to ∼31% at a thickness of 0.5 µm, while Bi-based compounds yield slightly lower efficiencies, around 27% at the same thickness, within the radiative limit. This performance difference can be attributed to the stronger optical transition intensities and absorption features observed in Sb-based systems. However the calculated SLME values are larger than those of the established candidates such as Si (26.1%) and CuInSe2 (∼23%).61,62
4. Conclusions
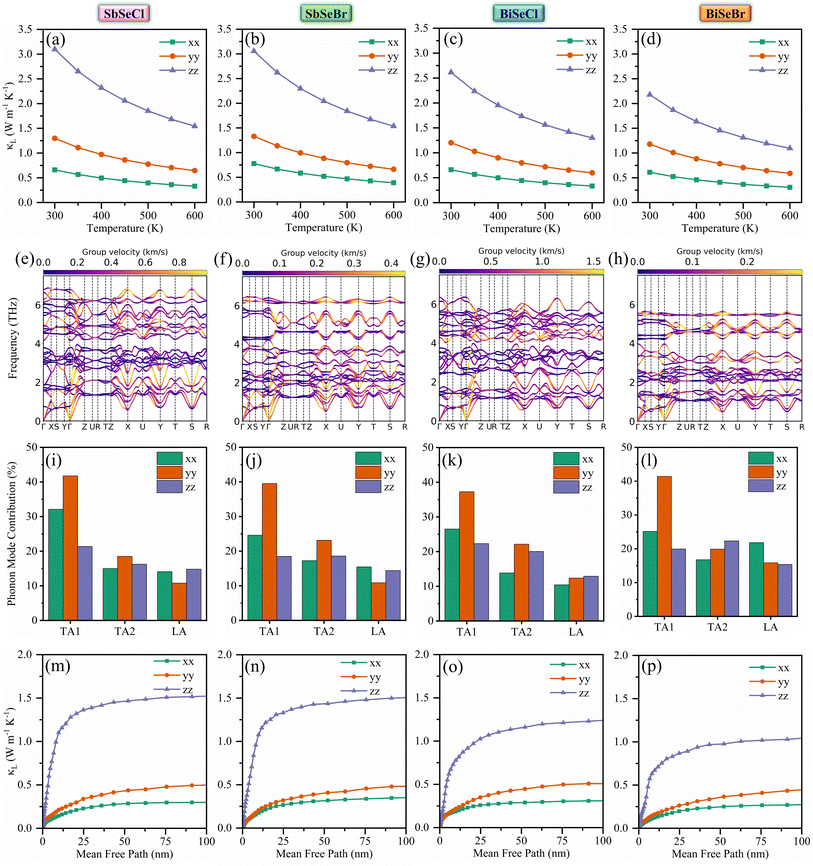
This work systematically explored the thermoelectric and photovoltaic potential of quasi-1D selenohalides using first-principles calculations. The weak van der Waals interlayer interactions combined with heterogeneous intralayer bonding give rise to distinct electron and phonon transport characteristics. Stability was confirmed through formation energy, convex hull analysis, phonon spectra, and elastic constants, while chemical potential diagrams suggest their synthesizability. The calculated band gaps (1.28–1.66 eV) and favorable carrier effective masses underscore their suitability for energy applications. From a thermoelectric perspective, n-type transport dominates, with attractive electron transport characteristics along the z-direction, facilitated by extended bonding pathways. Despite mixed bonding characteristics, lattice thermal conductivity remains below 2 W m−1 K−1 at 600 K. Notably, n-type BiSeBr exhibits the best performance, achieving a zT of 0.81 at 600 K along the z-direction, driven by its high-power factor (1.36 W m−1 K−1) and moderate lattice thermal conductivity (1.09 W m−1 K−1). On the photovoltaic side, SbSeBr demonstrates small effective masses, moderate exciton binding energies, and strong optical absorption, leading to a spectroscopic limited maximum efficiency of ∼31% at a film thickness of 0.5 µm under 1-sun illumination AM1.5 G. Overall, quasi-1D selenohalides emerge as promising candidates for thermoelectric and photovoltaic applications, warranting further experimental validation.Author contributions
Prakash Govindaraj: conceptualization, software, formal analysis, investigation, writing – original draft, writing – review & editing, visualization, data curation, validation, methodology. Hern Kim: supervision, resources, project administration, data curation, funding acquisition, conceptualization.Conflicts of interest
The authors declare no conflict of interest.Data availability
The computational results that predominantly support our claims were calculated using the Vienna Ab Initio Simulation Package (VASP) and can be found at https://www.vasp.at/ with https://doi.org/10.1103/PhysRevB.54.11169 and https://doi.org/10.1103/PhysRevB.59.1758. The version of the code employed for this study is version 5.4.Supplementary information (SI): the convergence test of q-point, cut-off radius and interpolation factor to calculate phonon and electron transport properties; 3D ELF, bond length and mechanical properties; validating the suitability of exchange correlational functional; electron transport properties of hole doped selenohalides; Gruneisen projected phonon dispersion; phonon mode contribution to KL, and peak zT along different directions. See DOI: https://doi.org/10.1039/d5ta08032k.
Acknowledgements
This work was supported by the National Research Foundation (NRF) grants funded by the Ministry of Education (RS-2020-NR049576), Republic of Korea.References
- T. Kirchartz, G. Yan, Y. Yuan, B. K. Patel, D. Cahen and P. K. Nayak, Nat. Rev. Mater., 2025, 1–20 Search PubMed.
- X.-L. Shi, N.-H. Li, M. Li and Z.-G. Chen, Chem. Rev., 2025, 16, 7525–7724 CrossRef PubMed.
- G. J. Snyder and E. S. Toberer, Nat. Mater., 2008, 7, 105–114 CrossRef CAS PubMed.
- H. He, J. Song, R. Liang, G. Yan, Y. Geng, L. Hu, F. Liu, W. Ao and C. Zhang, Small, 2025, 21, 2408864 CrossRef CAS PubMed.
- F. Xu, B. Liu and R. Ang, Acta Mater., 2025, 121266 CrossRef CAS.
- K. Imasato, S. Fujimoto, Y. Ikuta, M. Miyata, N. Saitoh, N. Yoshizawa, A. Yamamoto, T. Ishida, M. Koyano and M. Ohta, ACS Appl. Mater. Interfaces, 2025, 17, 25478–25488 CrossRef CAS PubMed.
- H. Ming, Z.-Z. Luo, Z. Zou and M. G. Kanatzidis, Chem. Rev., 2025, 125, 3932–3975 CrossRef CAS PubMed.
- T. Deng, P. Qiu, T. Yin, Z. Li, J. Yang, T. Wei and X. Shi, Adv. Mater., 2024, 36, 2311278 CrossRef CAS PubMed.
- D. Singh and R. Ahuja, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1547 CAS.
- H. Han, L. Zhao, X. Wu, B. Zuo, S. Bian, T. Li, X. Liu, Y. Jiang, C. Chen and J. Bi, J. Mater. Chem. A, 2024, 12, 24041–24083 RSC.
- C. Wu, X.-L. Shi, L. Wang, W. Lyu, P. Yuan, L. Cheng, Z.-G. Chen and X. Yao, ACS Nano, 2024, 18, 31660–31712 CrossRef CAS PubMed.
- X. Han, Y. Ji and Y. Yang, Adv. Funct. Mater., 2022, 32, 2109625 CrossRef CAS.
- B. Anand, R. Shankar, S. Murugavelh, W. Rivera, K. M. Prasad and R. Nagarajan, Renewable Sustainable Energy Rev., 2021, 141, 110787 CrossRef CAS.
- H. Zhang, Y. Xia, Y. Zhang, U. V. Ghorpade, M. He, S. W. Shin, X. Hao and M. P. Suryawanshi, Advanced Science, 2025, 12, 2413131 CrossRef CAS PubMed.
- S. Luo, T. Li, X. Wang, M. Faizan and L. Zhang, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2021, 11, e1489 CAS.
- B. Kozinsky and D. J. Singh, Annu. Rev. Mater. Res., 2021, 51, 565–590 CrossRef CAS.
- J. Li, S. S. Han and S. P. Guo, Eur. J. Inorg. Chem., 2022, 2022, e202200419 CrossRef CAS.
- S. Shyamal and N. Pradhan, ACS Energy Lett., 2023, 8, 3902–3926 CrossRef CAS.
- X. H. Li, Z. H. Shi, M. Yang, W. Liu and S. P. Guo, Angew. Chem., 2022, 134, e202115871 CrossRef.
- B. Peng, K. Xu, H. Zhang, Z. Ning, H. Shao, G. Ni, J. Li, Y. Zhu, H. Zhu and C. M. Soukoulis, Adv. Theory Simul., 2018, 1, 1700005 CrossRef.
- P. Govindaraj and K. Venugopal, J. Alloys Compd., 2022, 929, 167347 CrossRef CAS.
- D. Tiwari, F. Cardoso-Delgado, D. Alibhai, M. Mombrú and D. J. Fermín, ACS Appl. Energy Mater., 2019, 2, 3878–3885 CrossRef CAS.
- V. Donges, Z. Anorg. Allg. Chem., 1950, 263, 280–291 CrossRef.
- P. Chauhan, J. Singh and A. Kumar, J. Mater. Chem. A, 2024, 12, 16129–16142 RSC.
- A. Bafekry, M. Faraji, M. Fadlallah, D. Hoat, H. Jappor, I. A. Sarsari, M. Ghergherehchi and S. Feghhi, Phys. Chem. Chem. Phys., 2021, 23, 25866–25876 RSC.
- G. Kresse and J. Furthmüller, Phys. Rev. B, 1996, 54, 11169 CrossRef CAS PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B, 1999, 59, 1758 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B, 1976, 13, 5188 CrossRef.
- K. Momma and F. Izumi, Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- A. M. Ganose, A. J. Jackson and D. O. Scanlon, J. Open Source Softw., 2018, 3, 717 CrossRef.
- R. Nelson, C. Ertural, J. George, V. L. Deringer, G. Hautier and R. Dronskowski, J. Comput. Chem., 2020, 41, 1931–1940 CrossRef CAS PubMed.
- Y. Kumagai, N. Tsunoda, A. Takahashi and F. Oba, Phys. Rev. Mater., 2021, 5, 123803 CrossRef CAS.
- A. Togo, L. Chaput, T. Tadano and I. Tanaka, J. Phys.: Condens. Matter, 2023, 35, 353001 CrossRef CAS PubMed.
- A. Togo, J. Phys. Soc. Jpn., 2023, 92, 012001 CrossRef.
- W. Li, J. Carrete, N. A. Katcho and N. Mingo, Comput. Phys. Commun., 2014, 185, 1747–1758 CrossRef CAS.
- W. Li, L. Lindsay, D. A. Broido, D. A. Stewart and N. Mingo, Phys. Rev. B Condens. Matter, 2012, 86, 174307 CrossRef.
- A. M. Ganose, J. Park, A. Faghaninia, R. Woods-Robinson, K. A. Persson and A. Jain, Nat. Commun., 2021, 12, 2222 CrossRef CAS PubMed.
- G. K. Madsen, J. Carrete and M. J. Verstraete, Comput. Phys. Commun., 2018, 231, 140–145 CrossRef CAS.
- V. Wang, N. Xu, J.-C. Liu, G. Tang and W.-T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- L. Yu and A. Zunger, Phys. Rev. Lett., 2012, 108, 068701 CrossRef PubMed.
- I. Caño, A. Navarro-Güell, E. Maggi, A. Gon Medaille, D. Rovira, A. Jimenez-Arguijo, O. Segura, A. Torrens, M. Jimenez and C. López, Small, 2025, e05430 CrossRef PubMed.
- R. S. Nielsen, Á. L. Álvarez, A. G. Medaille, I. Caño, A. Navarro-Güell, C. L. Álvarez, C. Cazorla, D. R. Ferrer, Z. J. Li-Kao and E. Saucedo, J. Mater. Chem. A, 2025, 13, 31727–31739 RSC.
- B. Wan, Z. Gao, X. Huang, Y. Yang, L. Chen, Q. Wang, C. Fang, W. Shen, Y. Zhang and H. Ma, ACS Appl. Energy Mater., 2022, 5, 9549–9558 CrossRef CAS.
- P. Acharyya, K. Pal, A. Ahad, D. Sarkar, K. S. Rana, M. Dutta, A. Soni, U. V. Waghmare and K. Biswas, Adv. Funct. Mater., 2023, 33, 2304607 CrossRef CAS.
- P. Rentzeperis, Prog. Cryst. Growth Charact. Mater., 1991, 21, 113–138 CrossRef CAS.
- E. Wlaźlak, A. Blachecki, M. Bisztyga-Szklarz, S. Klejna, T. Mazur, K. Mech, K. Pilarczyk, D. Przyczyna, M. Suchecki and P. Zawal, Chem. Commun., 2018, 54, 12133–12162 RSC.
- C. Fong and F. Wooten, Experimental and Theoretical Study of the Feasibility of the Gunn Effect in BiSCl, BiSBr, BiSI, BiSeI, BiSeBr and BiSeCl, 1981.
- P. Govindaraj, H. Kim and K. Venugopal, J. Mater. Chem. C, 2025, 13, 13986–14000 RSC.
- F. Mouhat and F.-X. Coudert, Phys. Rev. B, 2014, 90, 224104 CrossRef.
- M.-M. Zhong, X.-Y. Kuang, Z.-H. Wang, P. Shao, L.-P. Ding and X.-F. Huang, J. Phys. Chem. C, 2013, 117, 10643–10652 CrossRef CAS.
- S. Yang, H. Shi, Y. Hu, J. Si, C. Chen, J. Yang, H. Qu, X. Hu, F. Zhang and S. Zhang, J. Phys. Chem. Lett., 2024, 15, 5721–5727 CrossRef CAS PubMed.
- S. Li, Photoelectric Behavior and Photovoltaic Performance of Bismuth Chalcohalides, PhD thesis, Kagawa University Publication, 2021.
- A. Das, K. Debnath, I. Maria, S. Das, P. Dutta, D. Swain, U. V. Waghmare and K. Biswas, J. Am. Chem. Soc., 2024, 146, 30518–30528 CrossRef CAS PubMed.
- Z. Chen, H. Ming, Z. Li, S. N. Girard, C. D. Morris, W. Guo, M. Wu, Y. Yu, C. Wolverton and Z. Z. Luo, Angew. Chem., 2025, 137, e202501667 CrossRef.
- R. Wu and A. M. Ganose, J. Mater. Chem. A, 2023, 11, 21636–21644 RSC.
- G. C. La Rocca, Thin Films Nanostruct., 2003, 31, 97–128 Search PubMed.
- M. Bokdam, T. Sander, A. Stroppa, S. Picozzi, D. Sarma, C. Franchini and G. Kresse, Sci. Rep., 2016, 6, 28618 CrossRef CAS PubMed.
- X.-J. Wu, Y.-F. Ding, B. Liu, J.-L. Yang and M.-Q. Cai, Appl. Phys. Lett., 2022, 121, 211103 CrossRef CAS.
- D. Zhang, B. Wang, Y. Gan, J. Zhou and Z. Sun, J. Phys. Chem. C, 2024, 128, 1582–1590 CrossRef CAS.
- L. Yu, R. S. Kokenyesi, D. A. Keszler and A. Zunger, Adv. Energy Mater., 2013, 3, 43–48 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |