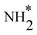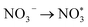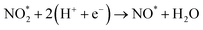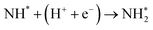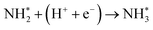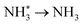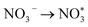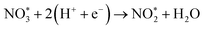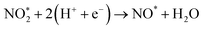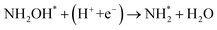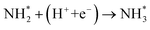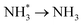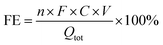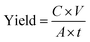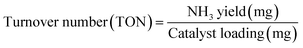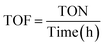Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Efficient ammonia synthesis via electrocatalytic nitrate reduction over a [8 + 2]-connected three-dimensional metal-bipyridine covalent organic framework
Tsukasa
Irie†
 a,
Ayumu
Kondo†
a,
Kai
Sun†
b,
Kohki
Sasaki
a,
Mika
Nozaki
a,
Shiho
Tomihari
a,
Kotaro
Sato
c,
Tokuhisa
Kawawaki
a,
Ayumu
Kondo†
a,
Kai
Sun†
b,
Kohki
Sasaki
a,
Mika
Nozaki
a,
Shiho
Tomihari
a,
Kotaro
Sato
c,
Tokuhisa
Kawawaki
 a,
Yu
Zhao
a,
Yu
Zhao
 *d,
Saikat
Das
*d,
Saikat
Das
 *a and
Yuichi
Negishi
*a and
Yuichi
Negishi
 *a
*a
aInstitute of Multidisciplinary Research for Advanced Materials, Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai 980-8577, Japan. E-mail: das.saikat.c4@tohoku.ac.jp; yuichi.negishi.a8@tohoku.ac.jp
bDepartment School of Materials and Energy, and LONGi Institute of Future Technology, Lanzhou University, Lanzhou 730000, China
cCarbon Value Research Center, Research Institute for Science & Technology, Tokyo University of Science, Kagurazaka, Shinjuku-ku, Tokyo 162-8601, Japan
dZhejiang Engineering Laboratory for Green Syntheses and Applications of Fluorine-Containing Specialty Chemicals, Institute of Advanced Fluorine-Containing Materials, Zhejiang Normal University, 321004 Jinhua, China. E-mail: zhaoyu@zjnu.edu.cn
First published on 2nd February 2026
Abstract
Covalent organic frameworks (COFs) have recently emerged as promising platforms for electrocatalytic nitrate reduction to ammonia (NO3RR), yet most reported systems are limited to two-dimensional architectures. Herein, we present TU-82, a structurally distinct 3D COF featuring an intricate [8 + 2]-connected bcu topology derived from the reticulation of an octatopic D2h-symmetric tetragonal prism node and a C2-symmetric bipyridyl linker. TU-82 exhibits high crystallinity, permanent porosity, and robust structural integrity, enabling precise postsynthetic metalation at bipyridyl coordination sites to yield catalytically active TU-82-Fe and TU-82-Cu frameworks. Among them, TU-82-Fe demonstrates superior NO3RR performance, delivering a faradaic efficiency (FE) of 88.1% at −0.6 V (RHE) and an ammonia yield rate of 2.87 mg h−1 cm−2 at −0.8 V (RHE), together with a turnover frequency of 7.2 h−1 and excellent operational stability. Density functional theory calculations reveal that the enhanced activity of TU-82-Fe originates from a lower energy barrier (0.354 eV) for the rate-determining NO* → NHO* step along the NHO-mediated reaction pathway. This work pioneers a structural blueprint for deploying 3D COFs in electrocatalysis, fostering deeper insights into framework-controlled reactivity and offering new routes to sustainable nitrate management.
1. Introduction
Ammonia (NH3) is an indispensable chemical for fertilizers, pharmaceuticals, and emerging energy technologies due to its high hydrogen content (17.6 wt%) and carbon-free nature.1,2 Despite its central role, industrial ammonia synthesis remains dominated by the Haber–Bosch (HB) process, a century-old method responsible for about 1.5–2.0% of global CO2 emissions and consuming about 2% of the world's energy supply.3 In contrast, the electrochemical nitrate reduction reaction (NO3RR) has gained momentum as a green and decentralized strategy for ammonia synthesis.4 Not only does NO3RR bypass the high-pressure, high-temperature conditions of the HB process, but it also addresses the global nitrate pollution crisis by converting NO3−, a common contaminant from agricultural runoff and industrial waste, into value-added NH3.5 Compared to N2, NO3− is more soluble, chemically reactive, and easier to reduce due to its lower bond dissociation energies.6 Furthermore, recent innovations in plasma and oxidation chemistry have enabled the efficient oxidation of atmospheric N2 into nitrogen oxides (NOx), which can be further transformed into stable nitrate feedstocks.7 Therefore, electrochemical NO3RR offers a dual benefit of sustainable ammonia generation and environmental remediation, positioning it as a key technology for a circular nitrogen economy.Efficient NO3RR is an eight-electron, nine-proton process complicated by sluggish kinetics and competition with the hydrogen evolution reaction (HER), necessitating the development of robust and selective catalysts.8 A wide range of catalyst systems has been explored to date, including metal nanoparticles, alloys, metal oxides/hydroxides, nanoclusters, single-atom catalysts (SACs), and molecular complexes.9,10 Metal nanoparticles and alloys have been among the earliest and most extensively investigated materials. Bimetallic catalysts such as Ni–Cu, Ru–Cu, and Pd–Ru have shown synergistic effects that enhance NO3− adsorption, intermediate stabilization, and NH3 selectivity.11 For instance, Liu et al. reported a PdCu alloy catalyst that achieved an impressive NH3 faradaic efficiency (FE) of 85% and an NH3 yield rate of 3058 µg h−1 mg−1 at −0.25 V vs. reversible hydrogen electrode (RHE).12 Cu–Pt nanostructure developed by Das et al. achieved a FE of 72.33% for NH3 at −0.1 V vs. RHE, along with substantial ammonia output of 0.71 mg h−1 mgcat−1 at −0.3 V.13 Metal oxides and hydroxides, such as TiO2, Fe3O4, and Cu2O, offer distinct advantages due to their structural stability and redox-active surfaces. For example, Zhang and colleagues reported that TiO2 nanotubes exhibited an FE of 66.3% and an NH3 yield of 0.024 mmol h−1 mg−1, which increased to 85.0% and 0.045 mmol h−1 mg−1, respectively, upon introducing oxygen vacancies.14 However, these materials often suffer from limited conductivity and moderate catalytic turnover rates, which has motivated the exploration of more tunable systems. Metal nanoclusters, such as Cu14 developed by our group, achieved 78% FE at −0.4 V vs. RHE—2.7 times higher than counterparts without exposed Cu sites and over 5.6 times greater than conventional Cu nanoparticles.15 SACs represent a breakthrough in NO3RR electrocatalysis due to their atomically dispersed metal sites, which maximize active site utilization and often exhibit unique reaction pathways. Fe SACs on nitrogen-doped carbon, prepared via two-step pyrolysis, showed ∼75% FE with enhanced NH3 selectivity attributed to the suppression of N–N coupling.16 However, pyrolytic methods often lead to uncontrolled coordination environments, low metal loading, and defect-rich supports, hindering the establishment of clear structure-activity relationships. Molecular electrocatalysts, including porphyrins and phthalocyanines, offer a complementary approach by combining well-defined coordination environments with structural modularity.17 Their rigid π-conjugated backbones can stabilize isolated transition metals and facilitate selective nitrate-to-ammonia conversion. These systems allow for precise tuning of the metal center electronic structure and have the added advantage of synthetic accessibility and compositional versatility. However, drawbacks include aggregation of the active molecules under operating conditions, limited conductivity, and challenges in immobilization on electrodes. Collectively, while substantial progress has been made in designing NO3RR catalysts, limitations such as poor structural tunability, low active site exposure, and insufficient control over coordination environments persist across most systems. This underscores the need for new catalyst platforms that combine structural modularity, high porosity, programmable active site integration, and long-range order.
Covalent organic frameworks (COFs) are an emerging class of crystalline, porous polymers built from reticulated organic building blocks via strong covalent bonds.18–31 Their high surface areas, ordered porosity, and modular backbones make them ideal scaffolds for incorporating redox-active moieties and coordinating catalytic centers in well-defined environments. COFs have already demonstrated their potential in electrocatalysis, particularly in CO2 reduction, oxygen evolution, and hydrogen evolution reactions. In 2015, Yaghi et al. first demonstrated that metalloporphyrin-based COFs could serve as efficient electrocatalysts for CO2 reduction, highlighting the advantages of site isolation, periodicity, and framework stability.32 More recently, the potential of COFs for NO3RR has garnered growing attention. Most of the reported examples are two-dimensional (2D) COFs constructed from metal-porphyrin or metal-bipyridine linkers.33–39 Lu and co-workers developed a series of metal-porphyrin COFs, where Fe−, Co−, Ni, and Cu-centered porphyrin nodes were incorporated into 2D layered frameworks.33 These COFs achieved FEs of up to 85.4% for NH3 and an ammonia yield rate of 1883.6 µmol h−1 mg−1 COF, with high selectivity attributed to the suppression of N–N coupling and stabilization of key NO3RR intermediates. Similarly, Li et al. developed a highly crystalline, mesoporous 2D nickel porphyrin COF (NiPr-TPA-COF) that exhibited ∼90% NH3 selectivity, a production rate of 2.5 mg h−1 cm−2, and a turnover frequency (TOF) of 3.5 s−1 for electrochemical nitrate reduction in neutral media.34 Nejati et al. developed a vapor-phase strategy to construct Cu- and Co-porphyrin COFs, with CuPOR-COF achieving a peak FE of ca. 86% and an NH3 production rate of 6.0 mg h−1 cm−2 under an applied potential range of −1.4 to −1.8 V vs. Ag/AgCl.35 In parallel, metal-bipyridine COFs have also emerged as promising NO3RR catalysts, offering a more compact and modular metal-binding environment compared to porphyrins. For instance, Lu and co-workers synthesized isomeric bipyridine-based COFs, incorporating Fe, Co, or Ni ions within mesoporous 2D backbones.36 Identified as the most efficient for electrocatalytic nitrate reduction, the Fe-coordinated framework (Py-Bpy-Fe) achieved 91.2% FE at −1.6 V vs. SCE and ∼3909 µmol h mg−1 NH3 yield at −2.0 V vs. SCE. A structural isomer with dual bipyridine sites (Py-PyIm-Fe) further boosted the NH3 yield to ∼4300 µmol h−1 mg−1. These frameworks benefit from the ability to precisely localize metal centers within a tunable π-conjugated system, which facilitates efficient electron transport and substrate activation. However, despite their promise, most COF-based NO3RR catalysts reported to date remain confined to 2D architectures. In contrast, three-dimensional (3D) COFs remain virtually unexplored for NO3RR, representing an untapped frontier with transformative potential. Unlike their 2D counterparts, which often suffer from interlayer π–π stacking that can restrict ion diffusion and reduce site accessibility, 3D COFs feature an extended framework that allows isotropic diffusion of electrolyte ions and uniform exposure of active sites. The inherently higher connectivity and crosslinking in 3D COFs enhances structural robustness, charge delocalization, and mechanical stability under electrochemical conditions. Furthermore, their topological diversity enables the integration of high densities of metal-binding sites within a rigid and spatially addressable scaffold. High-connectivity 3D nets, such as bcu, pts, ffc, or scu, are particularly appealing for NO3RR, as they provide interconnected channels that facilitate reactant transport and enable cooperative catalysis through spatially ordered active sites. Importantly, the precise and periodic positioning of these sites enables better understanding of structure–activity relationships, which is often elusive in amorphous or disordered systems. Despite these advantages, only one 3D COF has been recently reported for NO3RR,40 underscoring the nascency of this field and revealing a critical gap in current catalyst development strategies. Bridging this gap offers the potential to not only improve catalytic performance but also unlock new mechanistic insights through rational framework engineering.
In this study, we report a topologically intricate 3D COF as an electrocatalyst for selective nitrate-to-ammonia transformation. The COF, termed TU-82, is built through [8 + 2] topological design by integrating a D2h-symmetric octatopic tetragonal prismatic building unit and a C2-symmetric bipyridyl linker, establishing a highly ordered, intrinsically porous three-periodic bcu network. The COF's bipyridyl pockets facilitate controlled metal coordination, enabling generation of Fe- and Cu-functionalized analogues with well-defined active centers. Exhibiting robust performance metrics toward nitrate reduction, TU-82-Fe outperforms its Cu analogue, exhibiting an impressive FE of 88.1% (at −0.6 V vs. RHE), elevated NH3 productivity of 2.87 mg h−1 cm−2 (at −0.8 V vs. RHE), and prolonged cycling stability. Mechanistic modeling reveals that both Fe and Cu sites favor the NHO-mediated reduction pathway, with the Fe center offering a lower activation barrier (0.354 eV) for the rate-determining step (NO* → NHO*). This study not only introduces a new design paradigm by leveraging the structural precision and connectivity of 3D COFs but also sets the stage for understanding structure–activity relationships in reticular electrocatalysts. The findings open avenues for developing next-generation, architecturally programmable COF-based catalysts for sustainable nitrogen cycle engineering.
2. Results and discussion
The 3D COF TU-82 was synthesized via a solvothermal Schiff-base condensation reaction between 4′,5′-bis(3,5-diformylphenyl)-3′,6′-dimethyl-[1,1′:2′,1″-terphenyl]-3,3″,5,5″-tetracarbaldehyde (DPTB-Me) and 2,2′-bipyridine-5,5′-diamine (Bpy), in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 molar ratio. Optimal reaction conditions were identified following extensive parameter screening, wherein a mixed solvent system of o-dichlorobenzene and n-butanol (1
4 molar ratio. Optimal reaction conditions were identified following extensive parameter screening, wherein a mixed solvent system of o-dichlorobenzene and n-butanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
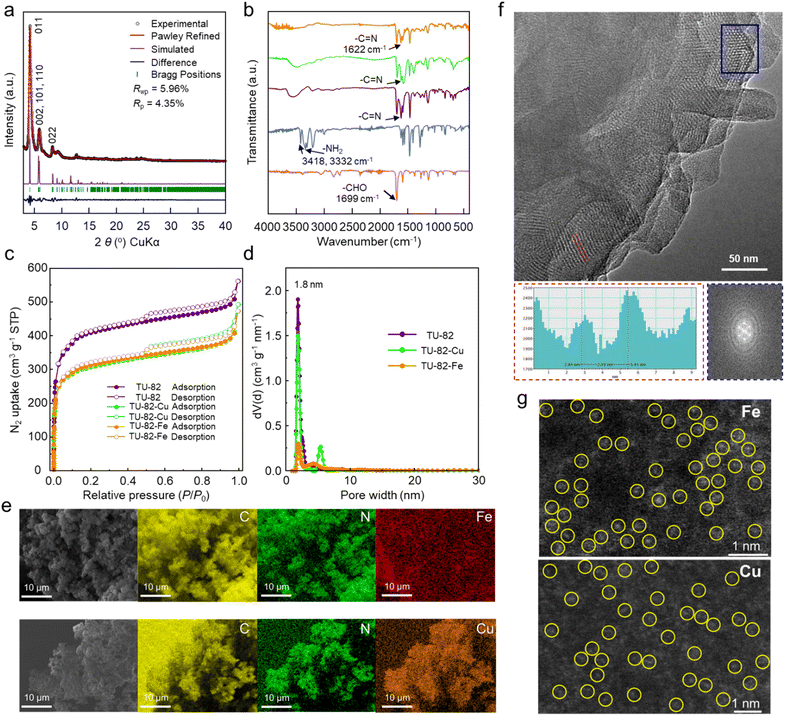
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v), with 6 M aqueous acetic acid as the catalyst, afforded TU-82 as a yellow crystalline solid after heating at 120 °C for 72 h (Fig. 1a). The incorporation of bipyridine moieties within the framework was strategic, enabling postsynthetic metalation due to their strong chelating affinity for transition metals—a critical feature for engineering catalytically competent sites in electrocatalytic NO3RR (Fig. 1c and d). Postsynthetic coordination with Fe2+ and Cu+/Cu2+ ions was achieved by refluxing TU-82 in methanolic solutions of the corresponding metal acetates at 60 °C for 12 h under an inert argon atmosphere, yielding TU-82-Fe and TU-82-Cu, respectively (Fig. 1a). Crystallographic elucidation of TU-82 was accomplished through detailed analysis of powder X-ray diffraction (PXRD) data in comparison with simulated patterns derived from energy-minimized structural models (Fig. 2a). Geometrical optimization was performed using the Forcite module in Materials Studio 7.0,(ref. 41) employing classical force-field-based methods. The refined structure revealed that TU-82 adopts a non-interpenetrated bcu topology (Fig. 1b), crystallizing in the orthorhombic space group Imm2 (No. 44), with unit cell parameters of a = 17.4656 Å, b = 29.2450 Å, and c = 33.0385 Å (α = β = γ = 90°; Table S5). The experimental PXRD pattern exhibited distinct Bragg reflections at 2θ values of 4.14°, 5.84°, and 8.29°, which were indexed to the (011), a multiplet comprising the overlapping (002), (101), and (110) reflections, and the (022) crystallographic planes, respectively. Pawley refinement yielded excellent agreement between the experimental and simulated patterns, with low residuals (Rp = 4.35%, Rwp = 5.96%), confirming the structural fidelity of the proposed model. The close match between observed and calculated patterns, along with minimal deviation in the difference profile, underscores the long-range periodicity and high structural order of the synthesized TU-82 framework. Furthermore, the PXRD profiles of TU-82-M (M = Fe, Cu) exhibited identical peak positions to the pristine TU-82, confirming that the incorporation of metal ions did not disrupt the underlying framework integrity and that the crystallinity of the COF was fully retained post-metalation (Fig. S2).
3, v/v), with 6 M aqueous acetic acid as the catalyst, afforded TU-82 as a yellow crystalline solid after heating at 120 °C for 72 h (Fig. 1a). The incorporation of bipyridine moieties within the framework was strategic, enabling postsynthetic metalation due to their strong chelating affinity for transition metals—a critical feature for engineering catalytically competent sites in electrocatalytic NO3RR (Fig. 1c and d). Postsynthetic coordination with Fe2+ and Cu+/Cu2+ ions was achieved by refluxing TU-82 in methanolic solutions of the corresponding metal acetates at 60 °C for 12 h under an inert argon atmosphere, yielding TU-82-Fe and TU-82-Cu, respectively (Fig. 1a). Crystallographic elucidation of TU-82 was accomplished through detailed analysis of powder X-ray diffraction (PXRD) data in comparison with simulated patterns derived from energy-minimized structural models (Fig. 2a). Geometrical optimization was performed using the Forcite module in Materials Studio 7.0,(ref. 41) employing classical force-field-based methods. The refined structure revealed that TU-82 adopts a non-interpenetrated bcu topology (Fig. 1b), crystallizing in the orthorhombic space group Imm2 (No. 44), with unit cell parameters of a = 17.4656 Å, b = 29.2450 Å, and c = 33.0385 Å (α = β = γ = 90°; Table S5). The experimental PXRD pattern exhibited distinct Bragg reflections at 2θ values of 4.14°, 5.84°, and 8.29°, which were indexed to the (011), a multiplet comprising the overlapping (002), (101), and (110) reflections, and the (022) crystallographic planes, respectively. Pawley refinement yielded excellent agreement between the experimental and simulated patterns, with low residuals (Rp = 4.35%, Rwp = 5.96%), confirming the structural fidelity of the proposed model. The close match between observed and calculated patterns, along with minimal deviation in the difference profile, underscores the long-range periodicity and high structural order of the synthesized TU-82 framework. Furthermore, the PXRD profiles of TU-82-M (M = Fe, Cu) exhibited identical peak positions to the pristine TU-82, confirming that the incorporation of metal ions did not disrupt the underlying framework integrity and that the crystallinity of the COF was fully retained post-metalation (Fig. S2).
The formation of the imine-linked framework in TU-82 was confirmed through a combination of Fourier-transform infrared (FT-IR) spectroscopy and solid-state 13C cross-polarization magic angle spinning (CP/MAS) NMR analysis. In the FT-IR spectrum of TU-82, a characteristic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching band appeared prominently at 1622 cm−1, verifying the establishment of imine linkages. Concurrently, significant attenuation of the N–H stretching vibrations of the bipyridyl diamine monomer (originally at 3418 and 3332 cm−1) and the aldehyde C
N stretching band appeared prominently at 1622 cm−1, verifying the establishment of imine linkages. Concurrently, significant attenuation of the N–H stretching vibrations of the bipyridyl diamine monomer (originally at 3418 and 3332 cm−1) and the aldehyde C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O band of DPTB-Me (at 1699 cm−1) indicated complete consumption of the starting functional groups, supporting the successful progression of Schiff-base polymerization (Fig. 2b). Notably, TU-82-Fe and TU-82-Cu retained similar vibrational profiles in the FT-IR spectra, affirming that the framework's integrity was preserved following metal coordination. Complementary 13C CP/MAS NMR spectroscopy further substantiated COF formation, with a distinct resonance at ∼161 ppm attributed to the imine carbon (C
O band of DPTB-Me (at 1699 cm−1) indicated complete consumption of the starting functional groups, supporting the successful progression of Schiff-base polymerization (Fig. 2b). Notably, TU-82-Fe and TU-82-Cu retained similar vibrational profiles in the FT-IR spectra, affirming that the framework's integrity was preserved following metal coordination. Complementary 13C CP/MAS NMR spectroscopy further substantiated COF formation, with a distinct resonance at ∼161 ppm attributed to the imine carbon (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) (Fig. S3). Elemental analysis of TU-82 (C160H100N32) revealed measured values (C: 72.11; H: 4.27; N: 14.28) in close proximity to theoretical predictions (C: 77.80; H: 4.05; N: 18.15), validating the expected framework stoichiometry. Porosity and surface area properties of TU-82 and its metalated analogues were evaluated by nitrogen adsorption–desorption measurements at 77 K following thermal activation at 100 °C for 8 h. The nitrogen sorption isotherms of TU-82 exhibited a steep uptake at low relative pressures (P/P0 < 0.1), indicative of intrinsic microporosity, along with a hysteresis loop at higher relative pressures, suggesting the presence of mesopores (Fig. 2c and S3). The Brunauer–Emmett–Teller (BET) surface area was determined to be 1527 m2 g−1 based on multipoint fitting in the low-pressure region. Upon postsynthetic metalation, the resulting TU-82-Fe and TU-82-Cu frameworks maintained the porous nature of the parent COF, albeit with moderately reduced BET surface areas of 1168 and 1163 m2 g−1, respectively. This reduction is ascribed to partial pore volume occupation and increased framework density due to coordinated metal ions. Quenched solid density functional theory (QSDFT) analysis employing a cylindrical pore model revealed a narrow pore size distribution centered at ∼1.8 nm for all three materials (Fig. 2d), in excellent agreement with the structural model (Fig. S9). Importantly, the preservation of pore size and isotherm profile across TU-82, TU-82-Fe, and TU-82-Cu further corroborates that the metalation process did not compromise the intrinsic porosity or framework architecture.
N) (Fig. S3). Elemental analysis of TU-82 (C160H100N32) revealed measured values (C: 72.11; H: 4.27; N: 14.28) in close proximity to theoretical predictions (C: 77.80; H: 4.05; N: 18.15), validating the expected framework stoichiometry. Porosity and surface area properties of TU-82 and its metalated analogues were evaluated by nitrogen adsorption–desorption measurements at 77 K following thermal activation at 100 °C for 8 h. The nitrogen sorption isotherms of TU-82 exhibited a steep uptake at low relative pressures (P/P0 < 0.1), indicative of intrinsic microporosity, along with a hysteresis loop at higher relative pressures, suggesting the presence of mesopores (Fig. 2c and S3). The Brunauer–Emmett–Teller (BET) surface area was determined to be 1527 m2 g−1 based on multipoint fitting in the low-pressure region. Upon postsynthetic metalation, the resulting TU-82-Fe and TU-82-Cu frameworks maintained the porous nature of the parent COF, albeit with moderately reduced BET surface areas of 1168 and 1163 m2 g−1, respectively. This reduction is ascribed to partial pore volume occupation and increased framework density due to coordinated metal ions. Quenched solid density functional theory (QSDFT) analysis employing a cylindrical pore model revealed a narrow pore size distribution centered at ∼1.8 nm for all three materials (Fig. 2d), in excellent agreement with the structural model (Fig. S9). Importantly, the preservation of pore size and isotherm profile across TU-82, TU-82-Fe, and TU-82-Cu further corroborates that the metalation process did not compromise the intrinsic porosity or framework architecture.
Scanning electron microscopy (SEM) images revealed that TU-82 crystallizes into well-defined, isometric microcrystals with uniform morphology (Fig. S5). Upon postsynthetic metalation, SEM coupled with energy-dispersive X-ray spectroscopy (EDS) elemental mapping confirmed the successful incorporation and homogeneous spatial distribution of Fe and Cu within the TU-82-Fe and TU-82-Cu frameworks, respectively (Fig. 2e). The quantitative metal content was further determined by inductively coupled plasma mass spectrometry (ICP-MS), yielding average loadings of 6.42 wt% Fe and 8.95 wt% Cu across three independent measurements (Table S1), consistent with efficient coordination at bipyridyl sites. High-resolution transmission electron microscopy (HR-TEM) analysis provided further structural insights into the crystallinity of TU-82 (Fig. 2f and S6). Clear lattice fringes were observed with a d-spacing of 2.59 nm (Fig. 2f), which corresponds well to the (001) plane as predicted from the crystallographic model (Fig. 1b). We note that HRTEM probes only local ultrathin regions (<10 nm) and depends on specific domain orientations under the electron beam, which can enable the visualization of well-oriented (001) planes. In contrast, PXRD reflects the bulk-averaged structure, where (001) reflections of low abundance, weak crystallinity, or overlap with stronger peaks (e.g., (100)/(110)) may render them less discernible in the diffraction pattern (Fig. 2a). Fast Fourier transform (FFT) analysis of the HR-TEM images further affirmed the long-range periodicity of the framework. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images of TU-82-Fe and TU-82-Cu showed bright, atomically dispersed contrast features—attributable to the heavier Fe and Cu atoms—distributed uniformly throughout the framework (Fig. 2g). Although occasional larger bright spots and lighter contrast features are visible, these can be ascribed to overlapping atomic columns or local thickness variations of the COF matrix rather than metal aggregation, since no lattice fringes corresponding to metallic Fe or Cu nanocrystals were observed. Furthermore, X-ray photoelectron spectroscopy (XPS) and extended X-ray absorption fine structure (EXAFS) analyses revealed the metals to be present in well-defined coordination environments without signatures of metallic bonding or cluster formation (described later). These results confirm that the observed contrast originates from isolated Fe and Cu centers uniformly embedded within the COF lattice, thereby excluding the presence of aggregated nanoparticles and validating the single-site catalytic behavior of TU-82-Fe and TU-82-Cu. The thermal stability of the pristine and metalated frameworks was evaluated by thermogravimetric analysis (TGA) under nitrogen. TU-82 demonstrated excellent thermal robustness with negligible weight loss up to ∼420 °C (Fig. S7), indicative of a thermally resilient network. The metalated analogues, while showing early-stage mass loss attributable to residual solvent and weakly coordinated species, retained appreciable stability under operating conditions. To evaluate the chemical robustness of the framework, TU-82 was immersed in a variety of protic and aprotic solvents relevant to NO3RR electrochemistry. PXRD patterns collected after 24 h of solvent exposure confirmed that the framework crystallinity remained intact (Fig. S8), underscoring the high chemical stability of the COF scaffold.
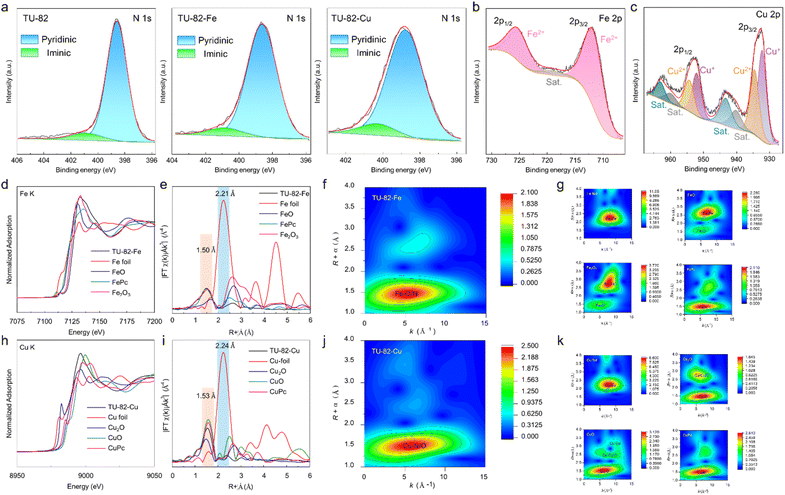
To elucidate the electronic environment and coordination states of the metal centers in TU-82-Fe and TU-82-Cu, comprehensive XPS and X-ray absorption spectroscopy (XAS) analyses were conducted. The wide-scan XPS spectra confirmed the presence of C, N, and the respective metal species (Fe or Cu) in the frameworks (Fig. S10). High-resolution C 1s spectra of TU-82 and its metalated analogues displayed nearly identical peak profiles, indicating that the backbone structure of the COF remained chemically stable upon metal coordination (Fig. S11). The N 1s XPS spectrum of pristine TU-82 exhibited two distinct peaks at 398.5 eV and 400.9 eV, corresponding to pyridinic and imine nitrogen species, respectively (Fig. 3a).36 Upon metalation, these binding energies shifted slightly due to coordination effects: TU-82-Fe showed peaks at 398.7 eV (pyridinic N) and 401.0 eV (imine N), while TU-82-Cu exhibited similar features at 398.8 eV and 400.4 eV, respectively, corroborating the formation of metal-nitrogen coordination bonds at the bipyridyl sites. Further insights into the oxidation state and coordination configuration were gained from the high-resolution Fe 2p and Cu 2p XPS spectra. For TU-82-Fe, two prominent peaks appeared at 712.1 eV (Fe 2p3/2) and 725.7 eV (Fe 2p1/2), indicative of Fe(II).36 In the case of TU-82-Cu, characteristic Cu 2p peaks were observed at 932.2 eV and 952.2 eV, assigned to Cu(I), alongside additional signals at 934.5 eV and 954.3 eV corresponding to Cu(II), suggesting the coexistence of mixed valence states.37,42,43 To further investigate the local coordination environment of the metal sites, X-ray absorption near-edge structure (XANES) and EXAFS analyses were performed. The Fe K-edge XANES spectrum of TU-82-Fe displayed an absorption edge beyond those of FeO and Fe2O3 (Fig. 3d). This coordination configuration of Fe atoms in TU-82-Fe was probed through EXAFS fitting of the Fe K-edge data that revealed a primary coordination number of 5.3 ± 0.3 (Table S2, Fig. S14), suggesting a five-coordinate geometry, likely comprising two nitrogen atoms from the bipyridine ligands and three oxygen atoms from acetate groups originating from the Fe(OAc)2 precursor. This mixed N/O coordination geometry is analogous to previously reported structures such as BiPy-COF-Mn, which also featured five-coordinate metal centers. The Fourier-transformed (FT) EXAFS spectrum of TU-82-Fe exhibited a main peak at ∼1.50 Å (phase uncorrected), attributed to Fe–N/O scattering, and the absence of a signal near 2.21 Å confirmed the lack of Fe–Fe bonds, ruling out the presence of Fe aggregates or nanoparticles (Fig. 3e). This was further corroborated by wavelet transform (WT) analysis of the EXAFS data, where the intensity maximum centered around 4.5 Å−1 (in k-space) and 1.5 Å (in R-space) was characteristic of light atom scatterers (N or O), validating the isolated single-site nature of Fe centers (Fig. 3f and g). Analogous characterization of TU-82-Cu by Cu K-edge XANES placed the absorption edge between those of Cu2O and CuO, and closer to CuO (Fig. 3h). This coordination state was understood through EXAFS fitting that revealed a Cu coordination number of 4.1 ± 0.2 (Table S2, Fig. S15), consistent with a square-planar or tetrahedral geometry involving bipyridine N-donors and acetate O-ligands. The FT-EXAFS spectrum of TU-82-Cu showed a prominent peak at ∼1.53 Å, again attributable to Cu–N/O coordination, and lacked any significant signal near 2.24 Å, confirming the absence of Cu–Cu interactions (Fig. 3i). WT-EXAFS plots reinforced these findings, displaying intensity maxima characteristic of light-atom coordination environments (Fig. 3j and k). Together, these spectroscopic analyses definitively establish that both Fe and Cu centers in TU-82-M are atomically dispersed, occupy well-defined coordination environments involving bipyridine nitrogen and acetate oxygen ligands, and are free from metallic clustering. These structural attributes are critical to the observed catalytic activity and selectivity in nitrate electroreduction.
To evaluate the electrocatalytic efficacy of TU-82-Fe and TU-82-Cu toward nitrate reduction to ammonia, a series of electrochemical measurements were performed in an H-type two-compartment cell. Each metalated COF was homogenously mixed with carbon black and uniformly cast onto a 1 × 1 cm2 carbon paper substrate, achieving a consistent catalyst loading of 0.4 mg cm−2. All potentials were referenced to RHE. Linear sweep voltammetry (LSV) measurements carried out in an electrolyte solution of 0.1 M KNO3 and 0.1 M KOH revealed distinct onset potentials and activity profiles for each catalyst (Fig. 4a). TU-82-Fe exhibited a significant increase in current density at approximately −0.21 V, while TU-82-Cu required a more negative onset of −0.23 V to elicit a comparable response, indicative of superior catalytic kinetics on the Fe site. TU-82-Fe consistently outperformed its Cu counterpart across the potential window, reaching a current density of 53 mA cm−2 at −0.80 V, compared to 43 mA cm−2 for TU-82-Cu. Chronoamperometric (i–t) measurements were employed to quantify the ammonia generated, using the indophenol blue method and UV-vis absorption at 654 nm (Fig. S16). As shown in Fig. 4b, TU-82-Fe exhibited a volcano-type trend in FE with increasing overpotential, peaking at 88.1% at −0.60 V, while the yield rate increased monotonically, attaining 2.87 mg h−1 cm−2 at −0.80 V. In contrast, TU-82-Cu did not display a well-defined volcano profile. Instead, the FE fluctuated across the tested potentials, with a notably low efficiency of 42.5% at −0.50 V. This reduced selectivity may stem from heightened hydrogen evolution activity at this intermediate potential, which competes for protons and electrons, thereby diminishing nitrate-to-ammonia conversion. The highest FE achieved for TU-82-Cu was 62.8% at −0.60 V, accompanied by a yield rate of 2.19 mg h−1 cm−2 at −0.80 V. Partial current densities for NH3 production, calculated from FE and total current density (Fig. 4c), further confirmed the superior performance of TU-82-Fe. At −0.80 V, TU-82-Fe achieved an NH3 partial current density of 36.19 mA cm−2, substantially surpassing that of TU-82-Cu (27.56 mA cm−2). Byproduct formation was assessed using the Griess assay to quantify NO2− (Fig. S17). The results indicated trace NO2− generation over the entire potential range for TU-82-Fe, while TU-82-Cu exhibited comparatively higher NO2− formation (Fig. S18), further substantiating the higher selectivity of the Fe-based catalyst. To ensure comprehensive selectivity evaluation, gaseous byproducts were quantified using gas chromatography equipped with a thermal conductivity detector (GC-TCD) and calibration curves. Only trace amounts of N2 (Fig. S19) and H2 (Fig. S20) were detected across all applied potentials, confirming that TU-82-Fe effectively suppresses competing pathways and maintains high selectivity toward NH3. Control experiments were designed to clarify the catalytic origin of the activity. LSV in 0.1 M KOH devoid of nitrate revealed a positive shift in the onset potential from −0.21 V to −0.65 V for TU-82-Fe, along with a sharp decline in current density (Fig. 4d). This confirms that the observed electrocatalytic response originates from NO3− reduction rather than competing hydrogen evolution. The introduction of 0.1 M KSCN, a strong chelator of single metal sites, led to pronounced suppression in current and a delayed onset potential (∼−0.57 V), underscoring the critical role of coordinated Fe centers in the bipyridyl motifs as the primary active sites.
To conclusively verify the nitrogen source in the reduced product, isotope-labeling experiments were conducted using 15NO3− in place of 14NO3−. The resultant NH3 was characterized by 1H NMR spectroscopy (Fig. 4e). The spectrum of 14NH3 displayed a triplet pattern with a 1J (N–H) coupling constant of 52 Hz, characteristic of coupling with a spin-1 14N nucleus. In contrast, the spectrum of 15NH3 exhibited a doublet pattern with a coupling constant of 72 Hz, consistent with spin-½ 15N. This distinct shift in splitting patterns and coupling constants provides unambiguous evidence that the nitrogen in the NH3 product originates exclusively from the nitrate feedstock, not from adventitious contamination. Electrochemical double-layer capacitance (Cdl), derived from cyclic voltammetry in the non-faradaic region (Fig. S21), was used as a proxy for electrochemical surface area (ECSA). TU-82-Fe exhibited a substantially higher Cdl (16.03 mF cm−2) than TU-82-Cu (9.07 mF cm−2), suggesting more abundant or accessible active sites, consistent with its higher NO3RR performance (Fig. 4f). The operational durability of TU-82-Fe was assessed over 10 continuous electrolysis cycles at −0.60 V (Fig. 4g). The catalyst retained an average FE of ∼77.0% and an NH3 yield of ∼1.79 mg h−1 cm−2, demonstrating excellent long-term stability and resilience under operating conditions. To assess industrial relevance, chronopotentiometric measurements were carried out at a high current density of 120 mA cm−2, where TU-82-Fe displays a stable potential profile under sustained NO3RR operation (Fig. S22). Furthermore, long-term electrolysis over 100 h at −0.6 V vs. RHE reveals consistent catalytic performance (Fig. S23), demonstrating excellent durability beyond the initial cycling tests. The catalytic activity of TU-82-Fe was further examined across a wide pH range (1–13) to assess its robustness under conditions relevant to real wastewater. As shown in Fig. S24, the catalyst remains active under acidic and neutral conditions and achieves its highest FE and NH3 yield in alkaline media, demonstrating broad pH tolerance and strong operational versatility. XRD and FT-IR analyses (Fig. S25) confirmed that the material retained its crystallinity and chemical integrity after cycling tests, indicating excellent structural stability. Post-NO3RR XPS analysis confirms that the bipyridine-Fe coordination environment in TU-82-Fe remains fully preserved after electrolysis. The N 1s (Fig. S26a) and Fe 2p (Fig. S26b) spectra showed negligible shifts in binding energies and retention of the Fe(II) oxidation state, demonstrating that neither ligand degradation nor metal demetalation occured during catalysis. A radar chart summarizing and comparing key performance metrics—FE, yield rate, NH3 partial current density, and Cdl—for TU-82-Fe and TU-82-Cu (Fig. 4h) clearly highlights the superior electrochemical behavior of the Fe-based catalyst. Notably, TU-82-Fe exhibits performance benchmarks that rival or exceed those of leading state-of-the-art NO3RR catalysts (Table S3), underscoring its promise for practical and scalable ammonia electrosynthesis.
To elucidate the structure–activity relationship of TU-82-M in electrocatalytic NO3RR, density functional theory (DFT) calculations were performed. Four possible mechanistic pathways were constructed, each describing the transformation of NO3− to NH3via a series of adsorbed intermediates. The free energy diagrams for TU-82-Fe and TU-82-Cu (Fig. 5a and Table 1) reveal that the key divergence between pathways lies in the NO* to 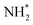 transformation, where two main routes involving either NOH* (Path 1, dotted lines) or NHO* (Path 2, solid lines) intermediates are energetically feasible. For TU-82-Fe, the activation energy for the NO* → NHO* step is significantly lower (0.354 eV) than that for NO* → NOH* (1.131 eV), indicating that the reaction preferentially proceeds via the NHO-mediated pathway. A similar trend is observed for TU-82-Cu, where the NO* → NHO* step requires 0.814 eV, while NO* → NOH* is substantially less favorable (1.531 eV). These findings suggest that both catalytic sites favor the NHO* intermediate pathway, wherein NHO* is sequentially hydrogenated to NHOH*, followed by protonation to NH2OH*, which undergoes dehydration to form
transformation, where two main routes involving either NOH* (Path 1, dotted lines) or NHO* (Path 2, solid lines) intermediates are energetically feasible. For TU-82-Fe, the activation energy for the NO* → NHO* step is significantly lower (0.354 eV) than that for NO* → NOH* (1.131 eV), indicating that the reaction preferentially proceeds via the NHO-mediated pathway. A similar trend is observed for TU-82-Cu, where the NO* → NHO* step requires 0.814 eV, while NO* → NOH* is substantially less favorable (1.531 eV). These findings suggest that both catalytic sites favor the NHO* intermediate pathway, wherein NHO* is sequentially hydrogenated to NHOH*, followed by protonation to NH2OH*, which undergoes dehydration to form 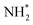 . The final protonation yields NH3. Moreover, the lower energy barrier for the rate-determining NO* → NHO* step on TU-82-Fe rationalizes its superior experimental NO3RR performance relative to TU-82-Cu. Notably, the TU-82-Fe site offers a more favorable energy landscape throughout the entire reaction coordinate, reinforcing its catalytic advantage. The complete reaction pathway, consistent with both theoretical predictions and experimental product distribution, is proposed as:
. The final protonation yields NH3. Moreover, the lower energy barrier for the rate-determining NO* → NHO* step on TU-82-Fe rationalizes its superior experimental NO3RR performance relative to TU-82-Cu. Notably, the TU-82-Fe site offers a more favorable energy landscape throughout the entire reaction coordinate, reinforcing its catalytic advantage. The complete reaction pathway, consistent with both theoretical predictions and experimental product distribution, is proposed as: 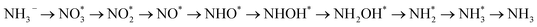 (Fig. 1d). This mechanism also explains the detection of NO2− as a main byproduct, attributable to the facile desorption of
(Fig. 1d). This mechanism also explains the detection of NO2− as a main byproduct, attributable to the facile desorption of 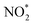 from the catalytic surface. The most stable adsorption configurations of each intermediate on TU-82-Fe and TU-82-Cu were fully optimized (Fig. 5b and c), revealing strong metal-adsorbate interactions at the bipyridyl-coordinated metal centers. These configurations reflect the geometric adaptability of the 3D COF scaffold, which offers a well-defined coordination environment that supports stable binding and transformation of reactive intermediates. A complete thermodynamic profile including electronic energies (E), zero-point energy corrections (ZPE), entropic contributions (T.S), and Gibbs free energies (G = E + ZPE − T S) of each intermediate is summarized in Table S4. These data provide quantitative support for the proposed reaction mechanism and further validate the superior kinetic and thermodynamic profile of TU-82-Fe. Overall, the DFT results highlight the critical role of metal identity in determining the reaction pathway and kinetics and underscore the structural advantages of TU-82's 3D framework in supporting active, isolated metal sites. This study offers a compelling demonstration of how topologically sophisticated COF platforms can be tailored to promote selective and efficient nitrogen-cycle electrocatalysis.
from the catalytic surface. The most stable adsorption configurations of each intermediate on TU-82-Fe and TU-82-Cu were fully optimized (Fig. 5b and c), revealing strong metal-adsorbate interactions at the bipyridyl-coordinated metal centers. These configurations reflect the geometric adaptability of the 3D COF scaffold, which offers a well-defined coordination environment that supports stable binding and transformation of reactive intermediates. A complete thermodynamic profile including electronic energies (E), zero-point energy corrections (ZPE), entropic contributions (T.S), and Gibbs free energies (G = E + ZPE − T S) of each intermediate is summarized in Table S4. These data provide quantitative support for the proposed reaction mechanism and further validate the superior kinetic and thermodynamic profile of TU-82-Fe. Overall, the DFT results highlight the critical role of metal identity in determining the reaction pathway and kinetics and underscore the structural advantages of TU-82's 3D framework in supporting active, isolated metal sites. This study offers a compelling demonstration of how topologically sophisticated COF platforms can be tailored to promote selective and efficient nitrogen-cycle electrocatalysis.
3. Conclusions
This work presents a transformative advancement in the development of COF-based electrocatalysts by introducing TU-82, a novel 3D COF platform applied to the electrochemical NO3RR. Unlike conventional 2D COF systems, TU-82 leverages a high-connectivity [8 + 2] topology and a rigid bcu network derived from rational topological design using an octatopic tetragonal prismatic node and a linear bipyridyl linker. This deliberate architectural strategy enables spatially precise metalation of bipyridyl coordination sites, thereby establishing uniformly distributed, catalytically active Fe and Cu centers within a robust and porous 3D scaffold. The superior performance of TU-82-Fe in NO3RR—evidenced by its high FE (88.1% at −0.6 V vs. RHE), notable ammonia yield (2.87 mg h−1 cm−2 at −0.8 V vs. RHE), and long-term durability—demonstrates the power of 3D framework engineering in tuning electrocatalytic function. Theoretical calculations corroborate these findings, revealing that Fe sites within the TU-82 scaffold facilitate a more energetically favorable NHO-mediated reaction pathway, with a notably low barrier for the rate-determining NO* → NHO* step. More broadly, this contribution establishes a conceptual and structural foundation for expanding the application of 3D COFs in electrocatalysis. By demonstrating how deliberate lattice design and coordination site engineering in 3D frameworks can drive selective multi-electron transformations, this work paves the way for the development of next-generation porous materials tailored for sustainable nitrogen conversion, clean energy processes, and programmable catalytic systems.4. Experimental section
4.1. Synthesis of TU-82
A finely ground mixture of DPTB-Me (19.04 mg, 0.03 mmol) and Bpy (22.35 mg, 0.12 mmol) was introduced into a Pyrex tube (ID: 8 mm; OD: 10 mm). To the mixture was added 0.25 mL of o-dichlorobenzene and 0.75 mL of n-butanol. Following 10 min of sonication, 0.2 mL of 6 M aqueous acetic acid was added dropwise. The resulting mixture was sonicated for an additional 10 min to ensure a homogenous suspension. The tube was flash-frozen in liquid nitrogen (77 K), degassed by three freeze–pump–thaw cycles, flame-sealed under vacuum, and heated at 120 °C for 72 h. Upon cooling to room temperature, the resulting solid was isolated via centrifugation, washed with THF (5 × 10 mL), Soxhlet extracted in THF for 24 h, and dried in vacuo at 100 °C for 12 h to afford TU-82 as a yellow powder (73% yield). Anal. Calcd. for C160H100N32: C: 77.80; H: 4.05; N: 18.15. Found: C: 72.11; H: 4.27; N: 14.28.4.2. Preparation of TU-82-M (M = Fe, Cu)
To prepare the metalated COF, TU-82 (40 mg) and the corresponding metal acetate salt (Fe(OAc)2 or Cu(OAc)2, 20 mg) were dispersed in methanol (40 mL) and stirred under reflux at 60 °C for 12 h under a continuous flow of argon. After completion, the resulting solid was collected by filtration, washed with methanol, and dried overnight in a desiccator under reduced pressure. For TU-82-Fe, the dried solid was further subjected to thermal treatment under a hydrogen atmosphere at 200 °C for 1 h, affording TU-82-Fe as a dark orange powder. For TU-82-Cu, the solid was thermally treated under vacuum at 100 °C for 4 h to yield TU-82-Cu as a dark green powder.4.3. Preparation of catalyst slurry
To prepare the catalyst slurry, TU-82-M (3.75 mg) and carbon black (3.75 mg) were dispersed in a mixed solvent consisting of 2-propanol (1.27 mL), ultrapure water (215 µL), and 5 wt% Nafion™ dispersion (15 µL). The mixture was sonicated for 30 min to ensure homogeneous dispersion of the COF catalyst and carbon black (denoted as TU-82-M/CB), forming a uniform catalyst ink. Carbon black was introduced as a conductive additive to improve the electron transport within the catalyst layer and to ensure uniform dispersion of the COF on the electrode surface. The 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mass ratio of TU-82-M to carbon black was chosen to balance electrical conductivity and active site exposure, a ratio that has also been employed in previous studies on COF-based electrocatalysts for nitrate reduction.44 Therefore, the prepared catalyst slurry is denoted as TU-82-M/CB, highlighting that carbon black serves as a conductivity enhancer and dispersant rather than an independent catalytic phase.
1 mass ratio of TU-82-M to carbon black was chosen to balance electrical conductivity and active site exposure, a ratio that has also been employed in previous studies on COF-based electrocatalysts for nitrate reduction.44 Therefore, the prepared catalyst slurry is denoted as TU-82-M/CB, highlighting that carbon black serves as a conductivity enhancer and dispersant rather than an independent catalytic phase.
4.4. Electrochemical measurements
All electrochemical measurements for nitrate reduction (NO3RR) were carried out using an ECstat-302 electrochemical workstation (EC FRONTIER, Japan) in a two-compartment H-type cell (EC FRONTIER, Japan) separated by an AMVN membrane. The working electrode was prepared by drop-casting 16 µL aliquots of the catalyst slurry onto a 1 cm × 1 cm piece of carbon paper (SIGRACET®GDL 22BB), followed by drying at room temperature. This process was repeated 10 times to ensure sufficient catalyst loading. A platinum coil served as the counter electrode, and a silver/silver chloride (Ag/AgCl) electrode was used as the reference electrode. All potentials reported herein were converted to the reversible hydrogen electrode (RHE) scale using the equation:45| ERHE = EAg/AgCl + 0.0591 × pH + 0.1976 |
The electrolyte in both chambers consisted of 0.1 M KNO3 and 0.1 M KOH (pH ≈ 13). Prior to each electrolysis, the electrolyte was purged with Ar gas for 20 min. Chronoamperometry (CA) was then conducted at the desired potential for 60 min. After each electrolysis cycle, the electrolyte was replaced with fresh solution before performing the next experiment.
where n is the number of electrons transferred per molecule of product (8 for NH3, 2 for NO2−), F is the Faraday constant (96,485 C mol−1), C is the concentration of product (mol L−1), V is the volume of electrolyte (L), and Qtot is the total charge passed, determined by integrating the current over the electrolysis time.
The NH3 and NO2− yields are calculated according to the following equation:
| ECSA = Cdl/Cs |
4.5. Computational method
All density functional theory (DFT) calculations were performed using the Vienna Ab initio Simulation Package (VASP),47,48 adopting the generalized gradient approximation (GGA) with the Perdew–Burke–Ernzerhof (PBE)49 functional. The projected augmented wave (PAW)50,51 potentials were utilized to describe the ionic cores and take valence electrons into account using a plane wave basis set with a kinetic energy cutoff of 450 eV. The DFT-D3 empirical correction method was used to account for the van der Waals interactions. Geometry optimizations were carried out with the force convergency smaller than 0.02 eV Å−1.To simulate the local environment of the catalytic site, a cluster model was extracted from the fully optimized 3D COF crystal structure. The terminal carbon atoms were saturated with hydrogen atoms. The terminal C–H bonds were optimized with all hydrogen atoms relaxed, while the rest of the cluster framework atoms were kept fixed. For adsorption calculations, the terminal atoms (including C atoms saturated by H and a complete benzene ring) were fixed, while the remaining framework atoms and adsorbates were allowed to relax. The COF fragment was placed in an orthorhombic simulation box of dimensions 20 Å × 20 Å × 30 Å. A (1 × 1 × 1) k-point sampling with the gamma-centered scheme was employed for the Brillouin zone integration.
The free energy changes (ΔG) for each elementary step of the nitrate reduction reaction (NO3RR) were calculated using the computational hydrogen electrode (CHE) model. This model assumes that the chemical potential of an electron–proton pair is equal to that of ½ H2 in the gas phase. The free energies of adsorption are then calculated by the following equation:
| ΔG = ΔE + ΔEZPE − TΔS |
The adsorption free energy of NO3−, denoted as Gad (NO3−), was determined according to Fig. S1.52,53 ΔG1 corresponds to the reaction free energy from gas HNO3(g) to adsorbed 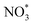 and (H+ + e−), and can be evaluated based on the CHE model. The free energy change from HNO3(l) to HNO3(g) was computed based on the difference in standard Gibbs free energy of formation between HNO3 in liquid and gas, which is 0.075 eV. The formation of HNO3(l) from NO3− in an aqueous solution is associated with a Gibbs free energy of 0.317 eV. All Gibbs free energy values were sourced from the CRC Handbook of Chemistry and Physics.54 The final adsorption free energy is then expressed as:
and (H+ + e−), and can be evaluated based on the CHE model. The free energy change from HNO3(l) to HNO3(g) was computed based on the difference in standard Gibbs free energy of formation between HNO3 in liquid and gas, which is 0.075 eV. The formation of HNO3(l) from NO3− in an aqueous solution is associated with a Gibbs free energy of 0.317 eV. All Gibbs free energy values were sourced from the CRC Handbook of Chemistry and Physics.54 The final adsorption free energy is then expressed as:
| Gad(NO3−) = ΔG1 + 0.075 + 0.317 |
Author contributions
Y. Z. S. D and Y. N. conceived the study and supervised the project. T. I carried out the synthesis, characterization and application with assistance from A. K. K. S. K. S and M. N. S. T. K. S. and T. K. contributed to the NO3RR experiments. S. D drafted the manuscript. All authors discussed the results and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are provided in the main text and supplementary information (SI). Additional data are available from the corresponding author upon reasonable request. Supplementary information: materials and methods, PXRD of metalated COFs, solid-state 13C CP-MAS NMR spectroscopy, BET surface area plot, SEM and TEM characterization, ICP-MS, TGA, chemical stability analysis, simulated structural model of TU-82, XPS, XANES analysis results, EXAFS fitting analysis results, electrochemical measurements, DFT calculations, and unit cell information and fractional atomic coordinates. See DOI: https://doi.org/10.1039/d5ta07989f.Acknowledgements
This study was supported by the Scientific Research on Innovative Areas “Aquatic Functional Materials” (grant no. 22H04562), JSPS KAKENHI (grant no. 23H00289 and 23KK0098), the Yazaki Memorial Foundation for Science and Technology, and the FUSO Innovative Technology Fund.Notes and references
- L. Zhai, S. Liu and Z. Xiang, Ind. Chem. Mater., 2023, 1, 332–342 RSC.
- J. Lim, C. A. Fernández, S. W. Lee and M. C. Hatzell, ACS Energy Lett., 2021, 6, 3676–3685 CrossRef CAS.
- W. I. F. David, G. D. Agnew, R. Bañares-Alcántara, J. Barth, J. B. Hansen, P. Bréquigny, M. de Joannon, S. Fürstenberg Stott, C. Fürstenberg Stott, A. Guati-Rojo, M. Hatzell, D. R. MacFarlane, J. W. Makepeace, E. Mastorakos, F. Mauss, A. Medford, C. Mounaïm-Rousselle, D. A. Nowicki, M. A. Picciani, R. S. Postma, K. H. R. Rouwenhorst, P. Sabia, N. Salmon, A. N. Simonov, C. Smith, L. Torrente-Murciano and A. Valera-Medina, J. Phys. Energy, 2024, 6, 021501 CrossRef.
- W. Song, L. Yue, X. Fan, Y. Luo, B. Ying, S. Sun, D. Zheng, Q. Liu, M. S. Hamdy and X. Sun, Inorg. Chem. Front., 2023, 10, 3489–3514 RSC.
- W. Chen, X. Yang, Z. Chen, Z. Ou, J. Hu, Y. Xu, Y. Li, X. Ren, S. Ye, J. Qiu, J. Liu and Q. Zhang, Adv. Funct. Mater., 2023, 33, 2300512 CrossRef CAS.
- G.-F. Chen, Y. Yuan, H. Jiang, S.-Y. Ren, L.-X. Ding, L. Ma, T. Wu, J. Lu and H. Wang, Nat. Energy, 2020, 5, 605–613 CrossRef CAS.
- Z. Qu, J. Hong, Y. Gao, J. Sun, J. Huang, M. Zhang, M. Zhu, T. Li, X. Wang, D. Gan, Q. Song, T. Zhang, R. Zhou, D. Liu, P. J. Cullen and R. Zhou, Adv. Sci., 2025, 12, 2411783 CrossRef CAS PubMed.
- W. Qiu, Y. Liu, M. Xie, Z. Jin, P. Li and G. Yu, EES Catal., 2024, 2, 202–219 RSC.
- Z. Wang, D. Richards and N. Singh, Catal. Sci. Technol., 2021, 11, 705–725 Search PubMed.
- C. Wu, Y. Shen, L. Lv, X. Meng, X. Yang, X. Wang, X. Jiang, Q. Ai, Y. Shuai and Z. Zhou, J. Mater. Chem. A, 2025, 13, 6631–6643 RSC.
- R. Zhang, X. Ma, S. Zhang, Q. Li, Y. Zhao and C. Zhi, ChemElectroChem, 2025, 12, e202400499 CrossRef CAS.
- L. Sun, H. Yao, F. Jia, Y. Wang and B. Liu, Adv. Energy Mater., 2023, 13, 2302274 CrossRef CAS.
- D. K. K. Kori and A. K. Das, ACS Appl. Eng. Mater., 2023, 1, 2386–2396 CrossRef CAS.
- R. Jia, Y. Wang, C. Wang, Y. Ling, Y. Yu and B. Zhang, ACS Catal., 2020, 10, 3533–3540 CrossRef CAS.
- S. Tomihari, M. Kamiyama, H. Minhas, T. Kawawaki, K. Takemae, Y. Shingyouchi, Z. Chen, B. Pathak and Y. Negishi, ACS Catal., 2025, 15, 20853–20863 CrossRef CAS.
- Z.-Y. Wu, M. Karamad, X. Yong, Q. Huang, D. A. Cullen, P. Zhu, C. Xia, Q. Xiao, M. Shakouri, F.-Y. Chen, J. Y. Kim, Y. Xia, K. Heck, Y. Hu, M. S. Wong, Q. Li, I. Gates, S. Siahrostami and H. Wang, Nat. Commun., 2021, 12, 2870 Search PubMed.
- L. Xie, Y. Wang, Q. Kong and R. Cao, ChemCatChem, 2024, 16, e202400956 CrossRef CAS.
- O. M. Yaghi, M. J. Kalmutzki and C. S. Diercks, Introduction to Reticular Chemistry: Metal–Organic Frameworks and Covalent Organic Frameworks, Wiley-VCH, Weinheim, Germany, 2019, ch. 7–11, pp. 177–283 Search PubMed.
- T. Irie, S. Das, Q. Fang and Y. Negishi, J. Am. Chem. Soc., 2025, 147, 1367–1380 CrossRef CAS PubMed.
- K. T. Tan, S. Ghosh, Z. Wang, F. Wen, D. Rodríguez-San-Miguel, J. Feng, N. Huang, W. Wang, F. Zamora, X. Feng, A. Thomas and D. Jiang, Nat. Rev. Methods Primers, 2023, 3, 1 CrossRef CAS.
- F. Haase and B. V. Lotsch, Chem. Soc. Rev., 2020, 49, 8469–8500 RSC.
- S.-Y. Ding and W. Wang, Chem. Soc. Rev., 2013, 42, 548–568 RSC.
- D. Blätte, F. Ortmann and T. Bein, J. Am. Chem. Soc., 2024, 146, 32161–32205 CrossRef PubMed.
- S. Kandambeth, K. Dey and R. Banerjee, J. Am. Chem. Soc., 2019, 141, 1807–1822 CrossRef CAS PubMed.
- X. Zhao, P. Pachfule and A. Thomas, Chem. Soc. Rev., 2021, 50, 6871–6913 RSC.
- Y. Song, Q. Sun, B. Aguila and S. Ma, Adv. Sci., 2019, 6, 1801410 CrossRef PubMed.
- X. Han, C. Yuan, B. Hou, L. Liu, H. Li, Y. Liu and Y. Cui, Chem. Soc. Rev., 2020, 49, 6248–6272 RSC.
- Y. Li, W. Chen, G. Xing, D. Jiang and L. Chen, Chem. Soc. Rev., 2020, 49, 2852–2868 RSC.
- X. Li, P. Yadav and K. P. Loh, Chem. Soc. Rev., 2020, 49, 4835–4866 RSC.
- C. Qian, L. Feng, W. L. Teo, J. Liu, W. Zhou and Y. Zhao, Nat. Rev. Chem., 2022, 6, 881–898 CrossRef CAS PubMed.
- J. Á. Martín-Illán, D. Rodríguez-San-Miguel and F. Zamora, Coord. Chem. Rev., 2023, 495, 215342 CrossRef.
- S. Lin, C. S. Diercks, Y.-B. Zhang, N. Kornienko, E. M. Nichols, Y. Zhao, A. R. Paris, D. Kim, P. Yang, O. M. Yaghi and C. J. Chang, Science, 2015, 349, 1208–1213 CrossRef CAS PubMed.
- H. Hu, R. Miao, F. Yang, F. Duan, H. Zhu, Y. Hu, M. Du and S. Lu, Adv. Energy Mater., 2024, 14, 2302608 CrossRef CAS.
- F. Lv, M. Sun, Y. Hu, J. Xu, W. Huang, N. Han, B. Huang and Y. Li, Energy Environ. Sci., 2023, 16, 201–209 RSC.
- S. I. G. P. Mohamed, S. Namvar, T. Zhang, H. Shahbazi, Z. Jiang, A. M. Rappe, A. Salehi-Khojin and S. Nejati, Adv. Mater., 2024, 36, 2309302 CrossRef CAS PubMed.
- S. Lu, F. Yang, H. Hu, D. Li, H. Hu, J. Wang and M. Du, J. Mater. Chem. A, 2025, 13, 9265–9273 Search PubMed.
- Y. Zhu, H. Duan, C. G. Gruber, W. Qu, H. Zhang, Z. Wang, J. Zhong, X. Zhang, L. Han, D. Cheng, D. D. Medina, E. Cortés and D. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202421821 CrossRef CAS PubMed.
- J. Zhou, J. Zhao, D. Song, J. Liu, W. Xu, J. Li and N. Wang, Inorg. Chem., 2024, 63, 15177–15185 CrossRef CAS PubMed.
- M. Teng, J. Yuan, Y. Li, C. Shi, Z. Xu, C. Ma, L. Yang, C. Zhang, J. Gao and Y. Li, J. Colloid Interface Sci., 2024, 654, 348–355 CrossRef CAS PubMed.
- S. Yang, S. Bi, L. Zhai and Q. Xu, Chem. Sci., 2025, 16, 15611–15619 RSC.
- Materials Studio, Ver. 7.0, Accelrys Inc., San Diego, CA, 2013 Search PubMed.
- X. Zhao, J. Chen, X. Mao, C. Li, L. He, F. Zhang, M. Zhang, J. Diwu, G. Wu, Z. Chai and S. Wang, Inorg. Chem., 2024, 63, 12333–12341 CrossRef CAS PubMed.
- J. Yang, W. Liu, M. Xu, X. Liu, H. Qi, L. Zhang, X. Yang, S. Niu, D. Zhou, Y. Liu, Y. Su, J.-F. Li, Z.-Q. Tian, W. Zhou, A. Wang and T. Zhang, J. Am. Chem. Soc., 2021, 143, 14530–14539 Search PubMed.
- B.-R. Wulan, S.-S. Yi, S.-J. Li, Y.-X. Duan, J.-M. Yan, X.-B. Zhang and Q. Jiang, Mater. Chem. Front., 2018, 2, 1799–1804 Search PubMed.
- A. Chaturvedi, S. Gaber, S. Kaur, K. C. Ranjeesh, T. C. Nagaiah and D. Shetty, ACS Energy Lett., 2024, 9, 2484–2491 CrossRef CAS.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 49, 14251–14269 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- P. E. Blöchl, O. Jepsen and O. K. Andersen, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 49, 16223–16233 Search PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- Y. Huang, J. Long, Y. Wang, N. Meng, Y. Yu, S. Lu, J. Xiao and B. Zhang, ACS Appl. Mater. Interfaces, 2021, 13, 54967–54973 Search PubMed.
- J.-X. Liu, D. Richards, N. Singh and B. R. Goldsmith, ACS Catal., 2019, 9, 7052–7064 CrossRef CAS.
- CRC Handbook of Chemistry and Physics, ed. D. R Lide, CRC Press, 90th edn(Taylor and Francis Group), Boca Raton, FL. 2009 Search PubMed.
- H. Hu, R. Miao, F. Yang, F. Duan, H. Zhu, Y. Hu, M. Du and S. Lu, Adv. Energy Mater., 2024, 14, 2302608 Search PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |