Tailoring electrolyte solvation for improved Na-based supercapacitor efficiency: an operando characterization approach
Raissa
Venâncio
 a,
Manuel J.
Pinzón C
b,
João Pedro
Aguiar dos Santos
a,
Manuel J.
Pinzón C
b,
João Pedro
Aguiar dos Santos
 ac,
Isabela
Galantini
ac,
Isabela
Galantini
 a,
Hugo
Cruz
a,
Hugo
Cruz
 d,
Carlos A.
Rufino
Jr.
e,
Gustavo
Doubek
d,
Carlos A.
Rufino
Jr.
e,
Gustavo
Doubek
 f,
Luís
Branco
f,
Luís
Branco
 d,
Débora V.
Franco
g,
Leonardo M.
Da Silva
*ag,
Josué M.
Gonçalves
d,
Débora V.
Franco
g,
Leonardo M.
Da Silva
*ag,
Josué M.
Gonçalves
 *hi and
Hudson
Zanin
*hi and
Hudson
Zanin
 *a
*a
aAdvanced Energy Storage Division, Center for Innovation on New Energies, BREnergies, School of Electrical and Computer Engineering, Universidade Estadual de Campinas, Av. Albert Einstein 400, Campinas, SP 13083-852, Brazil. E-mail: hzanin@unicamp.br
bCenter for Cooperative Research on Alternative Energies (CIC energiGUNE), Basque Research and Technology Alliance (BRTA), Alava Technology Park, Albert Einstein 48, 01510 Vitoria-Gasteiz, Spain
cEldorado Research Institute, Av. Alan Turing 400, Campinas, SP 13083-898, Brazil
dLAQV, REQUIMTE, Departamento de Química, Faculdade de Ciências e Tecnologia, Universidade Nova de Lisboa, 2829-516 Monte de Caparica, Portugal
eCARISSMA Institute for Electric, Connected and Safe Mobility (C-ECOS), Technische Hochschule Ingolstadt, 85049 Ingolstadt, Germany
fAdvanced Energy Storage Division, Laboratory of Advanced Batteries (LAB), Center for Innovation on New Energies, School of Chemical Engineering, Universidade Estadual de Campinas (UNICAMP), Campinas, São Paulo 13083-852, Brazil
gDepartment of Chemistry, Laboratory of Fundamental and Applied Electrochemistry, Federal University of Jequitinhonha and Mucuri's Valley, Rodovia MGT 367, km 583, 5000, Alto da Jacuba, Diamantina, MG 39100-000, Brazil. E-mail: leonardo.morais@ufvjm.edu.br
hMackenzie Institute for Research in Graphene and Nanotechnologies (MackGraphe), Mackenzie Presbyterian Institute, Consolação Street 930, São Paulo, São Paulo 01302-907, Brazil
iMackenzie School of Engineering, Mackenzie Presbyterian University, Consolação Street 930, São Paulo, São Paulo 01302-907, Brazil. E-mail: josue.goncalves@mackenzie.br
First published on 29th December 2025
Abstract
The development of safe and high-performance sodium-based supercapacitors critically depends on the choice of electrolyte, which governs ionic transport, the material's electrochemical stability, and the device's long-term durability. In this work, we present a comprehensive comparative study of four distinct classes of sodium-based electrolytes: highly concentrated aqueous solutions (WiSE), organic carbonate solvents, ionic liquids (IL), and deep eutectic solvents (DES). By combining classical electrochemical characterization studies (CV, GCD, and EIS) with operando electrochemical mass spectrometry (OEMS), we provide novel insights into the interplay between electrolyte composition, ion accessibility in porous carbon electrodes, and gas evolution processes. Our results reveal that the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] ionic liquid system delivers the highest specific capacitance and energy, while the NaOTf
[C3mpip][NTf2] ionic liquid system delivers the highest specific capacitance and energy, while the NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 deep eutectic solvent demonstrates intermediate behavior with moderate stability improvements. Conversely, the 17 m NaClO4 aqueous system, despite its superior ionic conductivity, is prone to the gas evolution reaction and instabilities at the electrode/electrolyte interface. The OEMS integrated analysis allowed us to verify a direct correlation between the gas evolution rate and electrolyte degradation mechanisms, shedding light on the formation of a solid electrolyte interface (SEI) and solvent stability. The integrated method establishes a framework for rational electrolyte design, highlighting the importance of balancing ionic transport with interface stability. The novelty of this work primarily lies in bridging electrochemical performance with real-time gas evolution analysis using various electrolytes, thereby providing a roadmap for the development of next-generation sodium-based energy storage devices.
PEG400 deep eutectic solvent demonstrates intermediate behavior with moderate stability improvements. Conversely, the 17 m NaClO4 aqueous system, despite its superior ionic conductivity, is prone to the gas evolution reaction and instabilities at the electrode/electrolyte interface. The OEMS integrated analysis allowed us to verify a direct correlation between the gas evolution rate and electrolyte degradation mechanisms, shedding light on the formation of a solid electrolyte interface (SEI) and solvent stability. The integrated method establishes a framework for rational electrolyte design, highlighting the importance of balancing ionic transport with interface stability. The novelty of this work primarily lies in bridging electrochemical performance with real-time gas evolution analysis using various electrolytes, thereby providing a roadmap for the development of next-generation sodium-based energy storage devices.
1 Introduction
Energy storage devices have evolved remarkably, pushed by the increasing demand for efficient and sustainable energy solutions.1–3 The quest for high-capacity, fast-charging, and long-lasting storage systems has been ongoing from the early days of lead-acid batteries to advancements in lithium-ion technology.4,5 This journey has witnessed the emergence of various devices like batteries, fuel cells, and supercapacitors (SCs), each with unique advantages and limitations.6,7 However, among these innovations, supercapacitors have gained substantial attention for their ability to bridge the gap between traditional capacitors and batteries. SCs exhibit rapid charge/discharge capabilities, excellent power density, and prolonged cycling life, making them vital in applications requiring quick bursts of energy, such as regenerative braking in vehicles and renewable energy integration.8–10 Concerning SCs, there has been a long-standing interest in sodium-based SCs due to the abundance and low cost of sodium compared to other materials used in energy storage.11 These supercapacitors utilize sodium-ion mechanisms, enabling efficient energy storage and release. One significant advantage of Na-based supercapacitors is their potential for scalability and affordability in large-scale applications, aligning with the growing demand for sustainable energy solutions, due to the safety and cost-effectiveness.12–14 Their ability to withstand numerous charge/discharge cycles with minimal degradation makes them attractive for long-term and reliable energy storage needs. Consequently, Na-based supercapacitors hold immense potential to revolutionize energy storage technologies, paving the way for a more sustainable and efficient energy future.Sodium perchlorate (NaClO4) is an inorganic salt that has been explored for its potential use in SCs, primarily due to its high solubility, high conductivity, and stability.15 Previous works have used sodium perchlorate-based electrolytes with aqueous, organic, and ionic solvents.16–18 However, in practical applications, the choice of solvent involves a trade-off between ionic conductivity, voltage range, capacitance, power, safety, stability, and cost. Since sodium perchlorate is highly soluble in water, several previous studies have been conducted using very high molalities (e.g., 17 m, m = mol kg−1) for increasing the stored energy and capacitance by expanding the working voltage window (WVW ≈ 1.2 V to 1.8 V) without provoking the solvent decomposition.19,20 Organic solvents, such as ethylene carbonate (EC), dimethyl carbonate (DMC), and diethyl carbonate (DEC), are commonly used in conjunction with sodium perchlorate for SCs. These solvents are flammable in atmospheric environments and toxic, but can offer a wider voltage window compared to water-based electrolytes, thus enabling higher energy densities with lower molarities of sodium perchlorate (e.g., 1 M, M = mol dm−3). At least, ionic liquids have garnered interest for their wide electrochemical stability window and low volatility, making them potentially suitable for Na-based SCs. However, their high cost and potential toxicity may limit their widespread use.
Previous studies21–23 have explored various strategies to enhance the performance of Na-based ILs and DEES electrolytes, although typically by addressing isolated aspects of their behavior. For instance, Amara et al.24 demonstrated that amide-based DESs formulated with LiFSI or NaFSI salts can achieve superionic conductivities of 3–4 mS cm−1 at room temperature, increasing substantially at elevated temperatures, while maintaining efficient ion transport and stable capacitive behavior on porous carbon electrodes, as reflected by fill factors of 75–85%. Their work highlighted the environmental compatibility and compositional tunability of DESs through simple HBA/HBD combinations, positioning them as promising candidates for sustainable next-generation supercapacitors. In a different approach, Guillemin et al.25 fabricated solid-state-like 3D micro-supercapacitors using ionogels based on EMIM-TFSI incorporating lithium or sodium salts, achieving high areal energy densities (≈10 µW h cm−2), excellent cycling durability (>50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles with ∼85% retention), and remarkable thermal robustness up to 100 °C without signs of electrolyte degradation or leakage. More recently, Thior et al.26 introduced a hybrid electrolyte system combining EMIM-DCA with highly concentrated NaNO3, revealing synergistic interactions that significantly improved ionic conductivity and molecular organization. When tested with N,S-doped activated carbon electrodes, this electrolyte delivered high capacitance (257 F g−1), extended voltage operation (2.3 V), and strong cycling stability (>10
000 cycles with ∼85% retention), and remarkable thermal robustness up to 100 °C without signs of electrolyte degradation or leakage. More recently, Thior et al.26 introduced a hybrid electrolyte system combining EMIM-DCA with highly concentrated NaNO3, revealing synergistic interactions that significantly improved ionic conductivity and molecular organization. When tested with N,S-doped activated carbon electrodes, this electrolyte delivered high capacitance (257 F g−1), extended voltage operation (2.3 V), and strong cycling stability (>10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles), outperforming conventional aqueous nitrate-based systems.
000 cycles), outperforming conventional aqueous nitrate-based systems.
Despite these advances, most reports evaluate IL and DES electrolytes primarily through electrochemical performance metrics, such as conductivity, capacitance, voltage window, or cycling retention, without addressing their long-term chemical stability or decomposition pathways under sustained polarization. Critically, the literature still lacks operando investigations linking gas evolution, molecular degradation, and electrochemical response during floating tests, particularly for amide-based DESs containing FSI− anions. These knowledge gaps limit the rational design of safe, durable electrolytes for practical supercapacitor applications. Addressing this gap, our work introduces an integrated methodology that incorporates operando gas analysis with a novel combined electrochemical protocol applied under long-term potentiostatic polarization conditions. As a result, we can obtain a direct correlation between electrochemical behavior, ion transport phenomena, and chemical stability information. This powerful integrated approach provides mechanistic insights into degradation processes in IL- and DES-based electrolytes that have not been previously elucidated in the literature, establishing a more robust framework for designing the next generation of high-voltage sustainable electrolytes.
Our research aims to investigate the electrochemical variations arising from the use of NaClO4 salt in aqueous solutions at the limit of solubility and in organic solvents within SCs, with the goal of determining the most suitable trade-off among key features for practical applications. The research question of the study is that while aqueous solvents, such as water-based electrolytes containing sodium perchlorate, demonstrate higher ionic conductivity and relative safety, they might exhibit limitations concerning the operating voltage range and could cause electrolyte decomposition at high WVWs. Conversely, organic, ionic, and eutectic solvents, when combined with sodium perchlorate, could potentially widen the operational voltage window, resulting in increased energy densities. Furthermore, we propose coupling rigorous electrochemical testing with operando characterization using mass spectrometry (MS) and Fourier transform infrared (FTIR) spectroscopy. This integrated approach enables simultaneous monitoring of gaseous products, molecular transformations, structural changes, and compositional evolution during device operation. With that, we aim to identify a balanced compromise between these contrasting features to recommend an optimal electrolyte configuration for practical SC applications, considering performance, safety, stability, and cost-effectiveness.
Our initial hypothesis suggested that aqueous solvent, due to its inherent ionic conductivity, would deliver superior power output, especially at varying concentrations of sodium perchlorate. Meanwhile, our second hypothesis proposed that the use of an ionic liquid (IL), particularly 1-methyl-1-propylpyrrolidinium bis(trifluoromethanesulfonyl)imide ([C3mpip][NTf2]) with NaClO4, would yield higher energy densities, primarily attributable to an extended operational voltage window rather than solely reliant on high capacitance. Our predictions aligned with these hypotheses, anticipating that experimental results suggest an advantage of aqueous solvents for maximal power output and highlight the use of ILs to elevate energy density through a widened voltage window (e.g., WVW ≈ 4 V), without sacrificing power density. The superior performance of IL is a consequence of the lack of solvent dielectric screening, which leads to a dense EDL, enhancing charge storage per unit area. At the same time, the interfacial stabilization observed for ILs SCs is beneficial for suppressing parasitic reactions, thereby contributing to extended electrochemical stability and WVW. The findings obtained from rigorous electrochemical analyses and performance evaluations not only validated these hypotheses and predictions but also provided crucial insights into selecting optimal electrolyte configurations for practical supercapacitor applications, balancing performance, safety, stability, and cost-effectiveness.
To test our hypothesis, we investigate the electrochemical performance of sodium perchlorate-based SCs when utilizing aqueous solutions of different concentrations, organic IL, and DES solvents, aiming to discern a practical balance between key features crucial for their application. Following a rigorous methodology, we adhered to standard protocols, employing a suite of electrochemical characterization techniques: electrochemical impedance spectroscopy (EIS), single potential step chronoamperometry (SPSC), cyclic voltammetry (CV), and galvanostatic charge–discharge (GCD) measurements. In addition, to further analyze electrolyte stabilities, we determine the gas fractions, potential degradation products, and molecular structures under polarization conditions using a mass spectrometer and an FTIR spectrometer. These techniques were instrumental in not only identifying the correct potential window but also in analyzing ion dynamics, diffusion resistance, electrolyte stability, and overall energy storage capabilities. Concurrently, we meticulously correlated these electrochemical, compositional, and molecular investigations with the porous and structural properties of the activated carbon electrodes assembled in symmetrical cell configurations.
2 Experimental session
2.1 Electrode fabrication
Conductive ink was prepared with 80% wt. of activated carbon (Kuraray), 10% wt. of PVDF binder (Sigma-Aldrich), and 10% wt. of black pearls 2000 (CABOT), respectively. This solution was stirred for 24 h at 600 rpm. Then, the homogenized mixture was coated onto etched aluminum or stainless-steel foil and dried at 80 °C for 2 h. After, the electrodes were cut into 16 mm discs with 4.5 mg and vacuum dried at 120 °C for 24 h.2.2 Cell assembly
Water-based electrolytes were prepared in three concentrations (1, 5, and 17 molal) using sodium perchlorate (NaClO4) as the salt, following the methodology reported by Vicentini et al.27 In contrast, the organic-based supercapacitors were assembled inside a Mbraun glovebox under an argon atmosphere with moisture and oxygen levels maintained below 0.1 ppm. They were prepared by dissolving NaClO4 (1.0 molal) in different solvents mixtures: dimethyl carbonate (DMC, Sigma-Aldrich, >99%), diethyl carbonate (DEC, Sigma-Aldrich, >99%), and ethylene carbonate (EC, Sigma-Aldrich, 99%), resulting in the following electrolyte formulations: NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC, NaClO4-EC
DMC, NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC, and NaClO4-EC
DEC, and NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC
DEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC.
DMC.
Additionally, the ionic liquid 1-methyl-1-propylpyrrolidinium bis(trifluoromethanesulfonyl)imide ([C3mpip][NTf2], Sigma-Aldrich, >99%) and the deep eutectic solvent (DES) composed of sodium trifluoromethanesulfonate (NaOTf) and polyethylene glycol (PEG 400) were preheated at 80 °C for 4 h. Subsequently, NaClO4 was added to the ionic liquid at a molar ratio of 0.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.2 (IL
0.2 (IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaClO4). All supercapacitor cells were assembled using Celgard® separators.
NaClO4). All supercapacitor cells were assembled using Celgard® separators.
Nevertheless, symmetrical supercapacitor cells were assembled using activated carbon electrodes and 160 µL of electrolyte, which was drop-cast onto a polypropylene (PP) separator (Celgard®, 25 µm thickness).
2.3 Electrochemical analyses
The electrochemical tests were performed with a potentiostat from Biologic® to determine the cell characteristics, such as the working voltage window (WVW), and specific power, energy, and capacitance as a function of the pore size distribution and electrolyte composition. By applying the methodology proposed by some of the present authors to determine the WVW,20 we incorporated cyclic voltammetry (CV) experiments at a low scan rate (10 mV s−1) for the voltage range of 1.0 to 4.2 V. To confirm the WVW values, single-step chronoamperometric (SSC) tests were conducted at different voltages to monitor the transitory current response governed by the characteristic time constant (τ = RC). The current decay follows the so-called Heaviside's voltage step, whose transitory current response is I = I0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ), where I0 = Ustep/Resr is the initial current at t = 0, and τ = CedlResr is the relaxation time constant due to the coupling between the equivalent series resistance (Resr) and the electrical double-layer capacitance (Cedl). This current transient reflects both the ideal capacitive response of the electrical double layer (EDL) and the parallel parasitic/Faradaic processes involving the solvent degradation for those cases where I/I0 > 0.01, i.e., when an almost constant current plateau persists after a brief elapsed time in the ca. 1–25 s interval. By contrast, if the current reduction percentage is less than 0.1%, it confirms the practical absence of electrolyte degradation. To complete the analysis, electrochemical impedance spectroscopy (EIS) experiments with an amplitude of 10 mV were conducted in the frequency range of 10 kHz to 10 mHz, using the same voltages as those employed in the SSC study.
exp(−t/τ), where I0 = Ustep/Resr is the initial current at t = 0, and τ = CedlResr is the relaxation time constant due to the coupling between the equivalent series resistance (Resr) and the electrical double-layer capacitance (Cedl). This current transient reflects both the ideal capacitive response of the electrical double layer (EDL) and the parallel parasitic/Faradaic processes involving the solvent degradation for those cases where I/I0 > 0.01, i.e., when an almost constant current plateau persists after a brief elapsed time in the ca. 1–25 s interval. By contrast, if the current reduction percentage is less than 0.1%, it confirms the practical absence of electrolyte degradation. To complete the analysis, electrochemical impedance spectroscopy (EIS) experiments with an amplitude of 10 mV were conducted in the frequency range of 10 kHz to 10 mHz, using the same voltages as those employed in the SSC study.
Once the WVW for each electrolyte system was determined, the discharging curves from the galvanostatic charge–discharge (GCD) experiments were obtained at different specific/gravimetric currents (e.g., 0.02, 0.1, 0.25, 0.5, 1.0, and 5.0 A g−1) and then numerically differentiated to obtain the time domain distributed specific capacitance using eqn (1):
 | (1) |
GCD findings were also applied to calculate the specific energy, power, and maximum power:28
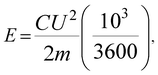 | (2) |
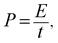 | (3) |
 | (4) |
In addition, based on the GCD findings, we construct the Ragone plot by applying the numerical iteration procedure proposed by Vicentini et al.,28 where the power (PL) and energy (EL) values can be obtained using the following equation:
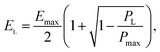 | (5) |
Finally, we investigated the self-discharge process by adopting the procedure where the coin cell was progressively charged at 1.0 mV s−1 until obtaining the WVW. Firstly, the cell voltage (or WVW) value was maintained at 1.0 h. Afterward, the open-circuit voltage (OCV) conditions (absence of external polarization) were maintained for 100 h to verify the presence of leakage current/voltage decrease.
2.4 Analysis of gas evolution by operando electrochemical mass spectroscopy
The residual gas analysis was collected in the four electrochemical cells assembled with carbon-based symmetrical electrodes, and 80 µL of 17 molal NaClO4 WISE, 1.0 M NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC, NaClO4
DEC, NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2], and NaOTf
[C3mpip][NTf2], and NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG 400, respectively. The cell was connected to a high-vacuum chamber using the same setup previously reported by Venâncio et al.29 The electrochemical behavior was controlled by the Biologic© SP-150 potentiostat, in which the cell was polarized in a Floating Time (FT) test of 40 h. Firstly, the cell was preconditioned for 10 h of charging and discharging at the WVW values using the CV technique at 1 mV s−1. Then the FT was alternated among CV, EIS, SPSC, and GCD sequentially and cyclically over 18 h. As the cell fails or deactivates, we measure the evolved gases using a Pfeiffer Quadrupole Mass Spectrometer (QMS-200) analyzer with a Faraday detector, coupled to an ultra-high vacuum (UHV) stainless steel chamber maintained at 6 µbar.
PEG 400, respectively. The cell was connected to a high-vacuum chamber using the same setup previously reported by Venâncio et al.29 The electrochemical behavior was controlled by the Biologic© SP-150 potentiostat, in which the cell was polarized in a Floating Time (FT) test of 40 h. Firstly, the cell was preconditioned for 10 h of charging and discharging at the WVW values using the CV technique at 1 mV s−1. Then the FT was alternated among CV, EIS, SPSC, and GCD sequentially and cyclically over 18 h. As the cell fails or deactivates, we measure the evolved gases using a Pfeiffer Quadrupole Mass Spectrometer (QMS-200) analyzer with a Faraday detector, coupled to an ultra-high vacuum (UHV) stainless steel chamber maintained at 6 µbar.
3 Results and discussion
3.1 Electrochemical analysis and the working voltage window for the different electrolytes
A comprehensive electrochemical analysis was conducted to assess the effect of solvent composition on the charge-storage properties of symmetrical two-electrode (coin cell) devices. In which the electrolytes are composed of 1.0 M NaClO4 prepared in deionized water, and mixtures of EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1
DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), EC
1), EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1
DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), and EC
1), and EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC
DMC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1
DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). A water-in-salt electrolyte (WiSE) was prepared using a 17-molal NaClO4 solution. In addition, other electrolytes composed of NaClO4 dissolved in 1-propyl-1-methylpiperidinium bis(trifluoromethylsulfonyl)amide ([C3mpip][NTf2]) in a molar ratio of 4
1). A water-in-salt electrolyte (WiSE) was prepared using a 17-molal NaClO4 solution. In addition, other electrolytes composed of NaClO4 dissolved in 1-propyl-1-methylpiperidinium bis(trifluoromethylsulfonyl)amide ([C3mpip][NTf2]) in a molar ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and sodium trifluoromethanesulfonate (NaOTf) salts dissolved in polyethylene glycol 400 (PEG400) in 1
1, and sodium trifluoromethanesulfonate (NaOTf) salts dissolved in polyethylene glycol 400 (PEG400) in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 molar ratio were prepared, respectively.
6 molar ratio were prepared, respectively.
The electrochemical findings focusing on determining the WVW as a function of the electrolyte composition are gathered in Fig. 1 and S1 (please check the SI).
All coin-cell-based SC prototypes containing different electrolyte systems exhibited the so-called electrical double-layer supercapacitor (EDLC) behavior, as determined by the various electrochemical techniques employed in this study. For instance, see the almost symmetrical rectangular voltammetric profiles in the first column of Fig. 1(a, e, i, m and q). For the different cases, it is interesting to note that almost constant voltammetric currents are achieved at approximately 0.2 A g−1. As expected, the aqueous-based SC presents a well-behaved voltammetric profile, which can be ascribed to facilitated ion insertion into the porous electrode structure. Besides, the ionic mobility can be enhanced from the specific conductivity increase from ca. 20 mS cm−1 (1.0 molal NaClO4) to 108 mS cm−1 (17 molal NaClO4).30 By contrast, it was verified that the organic-based, ionic liquid, and eutectic-based SCs presented a slight voltammetric asymmetry, which can be associated with the greater size of the hydrated ionic species or solvation shell and a mismatch of these charged structures with the average pore size.
The initial evaluation of the WVW using the CV technique revealed maximum voltages for NaClO4 in water (1.0 molal), WiSE (17 molal), EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1.0 M), in [C3mpip][NTf2] (4
DEC (1.0 M), in [C3mpip][NTf2] (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio), and NaOTf in PEG400 (1
1 molar ratio), and NaOTf in PEG400 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 molar ratio) of 1.2 V, 1.8 V, 3.0 V, 3.8 V, and 4.0 V, respectively. Generally, the corresponding CV profiles retained the RC response expected for well-behaved EDLCs, represented as follows:
6 molar ratio) of 1.2 V, 1.8 V, 3.0 V, 3.8 V, and 4.0 V, respectively. Generally, the corresponding CV profiles retained the RC response expected for well-behaved EDLCs, represented as follows:
 | (6) |
As the voltage increases beyond the stability region (ΔU > WVW), the solvent starts to decompose due to the presence of irreversible electrochemical oxidation processes, which are commonly accompanied by the formation of new dissolved organics and/or gas evolution (e.g., O2, CO2, etc.). To a first approximation, the linear charging process in CVs allied to the irreversible solvent decomposition during the anodic scan can be described by incorporating the Butler–Volmer–Erdey-Grúz kinetic model as follows:20
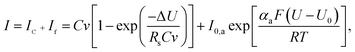 | (7) |
As can be seen, a cathodic peak was observed at 0.42 V in the voltammogram of the less concentrated (1.0 molal) aqueous electrolyte. These findings are commonly ascribed to the oxygen reduction reaction (ORR) involving the adsorbed oxygen O2(ads.) formed in the oxygen evolution reaction (OER) during the anodic voltage scan when ΔU > WVW. In the case of the IL electrolyte, minor CV distortions were also detected in the 2.23 V to 1.20 V cathodic scan interval. The latter findings can be indicative of a slight electrode passivation, possibly involving the redox activity of the ionic species subjected to abusive voltages (ΔU > WVW), probably with the formation of a more stable interfacial layer, akin to the solvent-blocked interface mechanism often reported in IL systems.31–34 This interfacial stabilization is beneficial for suppressing parasitic reactions and may contribute to the extended electrochemical stability of IL-based electrolytes. In a similar way, there is a small peak at ca. 1.8 V for the eutectic solvent electrolyte (DES). The latter can indicate the presence of a weak faradaic process superimposed on the dominant double-layer charging behavior. Based on the highly polar and partially ionic nature of the DES,34–36 these findings can be attributed to transient strong interactions between Na+ ions and oxygen-containing surface functionalities (e.g., carbonyl or hydroxyl groups) present on the activated carbon electrode.
In the case of NaOTf/PEG400 electrolyte system, PEG400 can act as a soft donor ligand capable of coordinating Na+ ions through ether oxygens, forming stable solvation shells.37,38 Upon electrode polarization, the electric field may disrupt these complexes, especially under cathodic potentials (reverse scan), facilitating minor ion–surface or ion–solvent reorganizations that manifest as a broadened or shallow cathodic peak, i.e., thus, the observed minor cathodic band can serve as an indirect signature of strong interactions between Na+-PEG complexes and the carbon surface, enabled by the highly polar, coordinatively active nature of the eutectic solvent matrix.
After applying Heaviside's voltage step function in SSC experiments to SCs outside the solvent stability domain (Ustep > WVW), a capacitive current transient is then followed by a leakage/faradaic current due to the irreversible solvent decomposition (Tafel's behavior) with a very small residual/spurious faradaic current (Ires) as follows:
 | (8) |
Fig. 1(b, f, j, n and r) shows the almost stationary leakage current over time as a function of different applied step voltages (Ustep) used to determine the WVW. The presence of a very small residual current is commonly found in the SSC experiments due to the presence of traces of impurities in the used chemicals, where Ires < 0.01% of the measured current. For ideal SCs, the second term on the right side of eqn (8) is absent since irreversible solvent decomposition does not occur. Therefore, it is expected that a current decrease to negligible residual values will occur after a short transient (t < 20 s), which amounts to less than ca. 0.01% of its initial (maximum) value at t = 0.
From the above discussion, the SSC analysis can provide unequivocal experimental pieces of evidence for the accurate determination of the WVW in SCs after the initial evaluation of the maximum voltages using the CV technique (see the previous discussion). We verified with the aid of SSC the following WVW values: 1.6 V (1.0 molal NaClO4), 2.0 V (17 molal NaClO4), 3.0 V (EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC), 3.6 V (NaOTf/PEG400), and 4.0 V ([C3mpip][NTf2]). As can be seen, a 1.0-molal aqueous solution roughly matches the theoretical limit of 1.23 V obtained from thermodynamics (e.g., using unitary activities and fugacities). At the same time, the WVW is extended to 2.0 V in the presence of the WISE system (17 molal), since the latter can effectively reduce water activity at the electrode/solution interface with strong suppression of water splitting. For the non-aqueous electrolytes, we verified progressively higher WVWs in the 3.0 V to 4.0 V interval due to the presence of highly stable organic-based solvents. As verified, the use of SSC for monitoring leakage currents incurred by solvent degradation (aqueous or not) provides an essential experimental approach for accurate determination of the WVW in SCs.
DEC), 3.6 V (NaOTf/PEG400), and 4.0 V ([C3mpip][NTf2]). As can be seen, a 1.0-molal aqueous solution roughly matches the theoretical limit of 1.23 V obtained from thermodynamics (e.g., using unitary activities and fugacities). At the same time, the WVW is extended to 2.0 V in the presence of the WISE system (17 molal), since the latter can effectively reduce water activity at the electrode/solution interface with strong suppression of water splitting. For the non-aqueous electrolytes, we verified progressively higher WVWs in the 3.0 V to 4.0 V interval due to the presence of highly stable organic-based solvents. As verified, the use of SSC for monitoring leakage currents incurred by solvent degradation (aqueous or not) provides an essential experimental approach for accurate determination of the WVW in SCs.
In the absence of solvent decomposition, the maximum initial current verified in the SSC experiments at t = 0 is strictly governed by Ohm's Law (Ustep = Rs × I0) Fig. 1(b, f, j, n and r). According to the Maximum Power Transfer theorem, the maximum instantaneous power that an SC can deliver is inversely proportional to Rs according to Pmax = Ustep2/Rs. By nearly an order of magnitude, the significantly higher initial currents observed in aqueous electrolytes compared to non-aqueous systems (e.g., organic, IL, and DES) can be ascribed to the superior ionic conductivity promoted by higher dielectric constants (D ≈ 70−81) allied to low viscosities (ν ≈ 1−5 mPa s), which facilitate rapid ion mobility and effective pore penetration dynamics inside carbon-based electrodes. In contrast, most organic electrolytes can exhibit higher Rs values due to reduced salt dissociation caused by lower dielectric solvent constants (D ≈ 18−40) and higher viscosities (ν ≈ 10−100 mPa s). In particular, ionic liquids and deep eutectic solvents suffer from severe ion transport limitations imposed by their inherently high viscosities (e.g., ν(ILs) ≈ 40 mPa s to 1500 mPa s). As a result, severe internal energy losses (Joule's effect) can be present in SCs when high currents are applied in some organics and ILs-based electrolytes.
Following the adopted methodology, the SSC and CV findings are then confronted with the impedance results, as shown in Fig. 1(c, g, k, o and s). The impedance response of well-behaved SCs features an almost vertical line in the complex-plane plot (−ZImvs. ZRe) characterizing the strong capacitive behavior. The presence of surface inhomogeneities and specific ionic adsorption processes is the primary source of deviations from the theoretical phase angle of ca. −90° predicted for an RC circuit with an imaginary impedance part given by Z = 1/jωC. In this sense, we must use the so-called constant phase element (CPE) given by Zcpe = 1/Y0(jω)n, where C ≅ Y0 for n ≥ 0.90, to incorporate the inclined capacitive lines verified in practice. When irreversible faradaic processes, such as solvent decomposition, occur at the electrode–electrolyte interface, we must introduce a charge-transfer resistance (Rct) in parallel with the CPE to represent the poorly developed semicircles tending to intercept the real axis at the extrapolated zero frequency. Therefore, the distinction between inclined and curved capacitive–resistive responses in the complex-plane plot can serve as a diagnostic to detect the presence of leakage currents.
Even so, in the special case of DES- and IL-based electrolytes, such RC curvatures in the complex plane may be incorrectly associated with a leakage current arising from electrolyte decomposition.
As shown in Fig. 1(o, p, s and t), the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] and NaOTf
[C3mpip][NTf2] and NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 electrolytes show complex-plane impedance plots with reduced phase angles at low frequencies in the −60° to −90° interval. This electrochemical behavior is commonly ascribed to the unique structural and migration transport characteristics of highly concentrated electrolytes and solvent-free electrolyte systems. In ionic liquids, the lack of a conventional solvent with a given dielectric constant close to the interface leads to an EDL structure that is markedly different from the classical Gouy–Chapman–Stern (GCS) model, typically valid for diluted aqueous-based electrolyte solutions, as well as Helmholtz–Perrin's model, mainly applied for concentrated electrolytes.39–41 By contrast, for the aforementioned electrolytes used in this study, the charge accumulation process occurs by a crowding of ions in the electrode surface, in which the charged ions alternate in layers with strong ion–ion interactions. The latter phenomenon, combined with the complex ion pathways, increases the overall impedance with a reduction in the phase angle, as shown in the Bode plots given in Fig. 1(d, h, i, p and t).42–44 For the DES-based electrolyte (NaOTf
PEG400 electrolytes show complex-plane impedance plots with reduced phase angles at low frequencies in the −60° to −90° interval. This electrochemical behavior is commonly ascribed to the unique structural and migration transport characteristics of highly concentrated electrolytes and solvent-free electrolyte systems. In ionic liquids, the lack of a conventional solvent with a given dielectric constant close to the interface leads to an EDL structure that is markedly different from the classical Gouy–Chapman–Stern (GCS) model, typically valid for diluted aqueous-based electrolyte solutions, as well as Helmholtz–Perrin's model, mainly applied for concentrated electrolytes.39–41 By contrast, for the aforementioned electrolytes used in this study, the charge accumulation process occurs by a crowding of ions in the electrode surface, in which the charged ions alternate in layers with strong ion–ion interactions. The latter phenomenon, combined with the complex ion pathways, increases the overall impedance with a reduction in the phase angle, as shown in the Bode plots given in Fig. 1(d, h, i, p and t).42–44 For the DES-based electrolyte (NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400), additional contributions to this non-ideal behavior stem from high viscosity and strong hydrogen bonding within the PEG-based matrix. These effects slow down ion diffusion and increase the interfacial resistance at the electrode–electrolyte interface, further deviating the system from ideal capacitive behavior.
PEG400), additional contributions to this non-ideal behavior stem from high viscosity and strong hydrogen bonding within the PEG-based matrix. These effects slow down ion diffusion and increase the interfacial resistance at the electrode–electrolyte interface, further deviating the system from ideal capacitive behavior.
Furthermore, the impedance data suggest that both electrolytes exhibit pronounced resistive behavior (or real part) at low frequencies, indicative of hindered ion transport and a more complex, voltage-sensitive interfacial structure. Even so, the EIS findings support the WVW values of 4.0 V and 3.4 V for IL and DES, respectively. The large WVW observed for DES is a consequence of the charge-stabilizing process in a highly polar medium comprised of the Na+/OTf− salt embedded in PEG. The latter mitigates solvent breakdown while allowing the charge–discharge process to occur reversibly. In the case of DES-based systems, hydrogen bonding and ion–dipole interactions can transiently stabilize more weakly bounded ionic species.36–38,45,46 These findings highlight the importance of considering bulk electrolyte properties and interface EDL dynamics for designing and interpreting SC characteristics for ILs and DES electrolyte systems.
In a different way, the aqueous- and organic-based devices exhibited slightly higher phase angles of ca. −80°, which is normally found for typical laboratory-made EDLCs. When the voltage increases beyond WVW, the phase angle starts to decrease due to the occurrence of electrolyte degradation. Thus, as expected from the adopted protocol analysis, the three different electrochemical techniques (CV, SSC, and EIS) unequivocally confirm the WVW values of 1.2 V, 1.8 V, and 3.0 V for 1.0 m NaClO4, 17 m NaClO4, and 1.0 M NaClO4 in EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1
DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively. Notably, when we compare organic-based and carbonate-based electrolytes, the overall electrochemical response is quite different (please see Fig. S1). By applying the same methodology using the aforementioned techniques, the EC
1), respectively. Notably, when we compare organic-based and carbonate-based electrolytes, the overall electrochemical response is quite different (please see Fig. S1). By applying the same methodology using the aforementioned techniques, the EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC and EC
DMC and EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC
DEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC electrolyte systems exhibited a characteristic SC behavior, while their WVWs are found to be 2.6 V and 3.0 V, respectively. Thus, we observed that the presence of DMC does not favor improvement in the energy-storage capabilities as for the DEC-based electrolyte system.
DMC electrolyte systems exhibited a characteristic SC behavior, while their WVWs are found to be 2.6 V and 3.0 V, respectively. Thus, we observed that the presence of DMC does not favor improvement in the energy-storage capabilities as for the DEC-based electrolyte system.
The nature of ion–solvent interactions significantly influences the shell solvation structure, ion transport dynamics, and interfacial stability of the electrolyte system. In carbonate-based electrolytes, EC plays a critical role due to its high dielectric constant (89.8) and strong coordinating properties with Na+.10 This behavior was verified by Shakourian-Fard et al.,47 where the presence of EC as a primary solvent promoted strong coordination in the primary solvation shell. The latter was evidenced by shorter Na+⋯O bond lengths during strong interactions in Na+(EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC) complexes (d = 2.187 Å) compared to Na+(EC
DEC) complexes (d = 2.187 Å) compared to Na+(EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC) (d = 2.392 Å), i.e., a compellingly stronger interaction with the carbonyl oxygen atom. These interactions lead to more stable shell solvation structures, thus reducing the occurrence of side reactions (solvent degradation) at the electrode/solution interface with improvements in the WVW values.
DMC) (d = 2.392 Å), i.e., a compellingly stronger interaction with the carbonyl oxygen atom. These interactions lead to more stable shell solvation structures, thus reducing the occurrence of side reactions (solvent degradation) at the electrode/solution interface with improvements in the WVW values.
The combination of EC and diethyl carbonate in the Na![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC-DEC system appears to facilitate an optimal balance between solvation shell characteristics and ionic mobility. We verified that DEC is characterized by low viscosity (0.75 cP) and reduced dielectric constant (2.8) compared to EC.10 As a result, the former contributes to a lesser extent in the Na+ coordination process, thereby allowing EC to dominate the primary solvation shell. This preferential coordination with EC facilitates the formation of more stable and less saturated states of the solvation clusters, which enhance interfacial stability and enable efficient ion desolvation at the electrode surface. Conversely, while dimethyl carbonate exhibits a stronger binding energy with Na+ (−127.40 kcal mol−1), it can facilitate pore confinement effects, which hinder ion desolvation and mobility.47–49 Moreover, in the ternary mixture EC
EC-DEC system appears to facilitate an optimal balance between solvation shell characteristics and ionic mobility. We verified that DEC is characterized by low viscosity (0.75 cP) and reduced dielectric constant (2.8) compared to EC.10 As a result, the former contributes to a lesser extent in the Na+ coordination process, thereby allowing EC to dominate the primary solvation shell. This preferential coordination with EC facilitates the formation of more stable and less saturated states of the solvation clusters, which enhance interfacial stability and enable efficient ion desolvation at the electrode surface. Conversely, while dimethyl carbonate exhibits a stronger binding energy with Na+ (−127.40 kcal mol−1), it can facilitate pore confinement effects, which hinder ion desolvation and mobility.47–49 Moreover, in the ternary mixture EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC
DMC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC, the presence of multiple cosolvents may lead to competitive coordination processes involving Na+, resulting in different solvation shell structures and an ion pairing process, thus disrupting the optimal solvation characteristics for SCs. Overall, the enhanced electrochemical performance verified for the Na-EC
DEC, the presence of multiple cosolvents may lead to competitive coordination processes involving Na+, resulting in different solvation shell structures and an ion pairing process, thus disrupting the optimal solvation characteristics for SCs. Overall, the enhanced electrochemical performance verified for the Na-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC system can be rationalized by considering its characteristic solvation shell structure, i.e., while EC ensures strong and selective coordination for ions, one has that DEC enhances the fluidity and ionic conductivity of the electrolyte medium without significant changes in ion–solvent interactions. Collectively, these factors lead to larger WVWs and improved charge-storage performance.
DEC system can be rationalized by considering its characteristic solvation shell structure, i.e., while EC ensures strong and selective coordination for ions, one has that DEC enhances the fluidity and ionic conductivity of the electrolyte medium without significant changes in ion–solvent interactions. Collectively, these factors lead to larger WVWs and improved charge-storage performance.
The previous discussion is supported by the electrolyte bulk properties gathered in Table 1. As can be seen, an inverse correlation between viscosity and conductivity is observed for the studied systems. Regarding the aqueous electrolytes, the 17 molal NaClO4 system exhibited the highest ionic conductivity (103.1 mS cm−1) and relatively low viscosity (3.51 cP), which can aid us in explaining its superior ion mobility and low ionic resistance affecting the ESR. Conversely, both NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] and NaOTf
[C3mpip][NTf2] and NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 electrolyte systems show higher viscosities of 611.58 cP and 48.95 cP, respectively. At the same time, these systems exhibited reduced conductivities of 0.4 and 1.6 mS cm−1, respectively. These bulk electrolyte properties result in restricted ion transport, higher ESR values, and less effective charge-storage capabilities (please see Bode plots in Fig. 1).
PEG400 electrolyte systems show higher viscosities of 611.58 cP and 48.95 cP, respectively. At the same time, these systems exhibited reduced conductivities of 0.4 and 1.6 mS cm−1, respectively. These bulk electrolyte properties result in restricted ion transport, higher ESR values, and less effective charge-storage capabilities (please see Bode plots in Fig. 1).
| Electrolyte | Conductivity (mS cm−1) | Viscosity (cP) |
|---|---|---|
| 1.0 molal NaClO4 | 51.7 | 1.0033 |
| 17 molal NaClO4 | 103.1 | 2.7389 |
1.0 molal NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1 DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
6.39 | 3.4121 |
1.0 molal NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1 DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
9.82 | 3.4792 |
1.0 molal NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC DEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1 DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
6.50 | 3.4718 |
NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] [C3mpip][NTf2] |
0.86 | 611.58 |
NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG600 PEG600 |
— | 48.948 |
This inverse relationship between viscosity and conductivity, known as Walden's rule (Λ × η ≈ K), particularly in concentrated systems such as WiSE and DES, can be directly linked to increased ionic associations and reduced solvent availability.50 In highly concentrated electrolytes, the limited number of free solvent molecules and stronger ion pairing preclude the total salt dissociation, leading to a reduced fraction of the “free ions” for charge transport or ionic current. However, this reduction in conductivity does not necessarily completely translate into poor charge-storage characteristics in SCs. As previously discussed in this work, WiSEs, despite their elevated viscosity and reduced ionic mobility, commonly benefit from solvent-blocked interfacial structures, incurring larger WVWs with improved energy and power characteristics. Similarly, DES and IL electrolyte systems present their peculiar electrochemical properties with the formation of stable interfaces, which overcompensate for the reduced ion mobility. Therefore, while viscosity critically governs ionic conductivity, the overall performance of SCs depends on a more nuanced interplay between bulk electrolyte transport properties and interfacial electrochemical stability.
3.2 Evaluation of the figures-of-merit for supercapacitors
Besides the WVW values, the other key parameters to be determined for SCs are the specific capacitance, energy, and power. Initially, we determined the distributed capacitance in the time domain using the discharge curves available from GCD experiments. This innovative approach proposed earlier by some of the present authors can permit discrimination of the charge-storage dynamic characteristics involving the progressive ion penetration inside different interconnected pore structures present in AC-based and other carbon-based electrode materials.Fig. 2 shows the experimental discharging curves differentiated using eqn (1) and normalized for the maximum time (t/t0). The charge-storage dynamic profiles strongly depend on electrolyte composition and applied gravimetric current. As the gravimetric current increases, ions experience limited migration inside pores because they do not have sufficient time to fully access the narrow/deep pore structures that comprise the inner surface regions. This limited access to narrow micropores hinders the ions from achieving optimal performance, resulting in a well-defined capacitance plateau with reduced capacitance. In moderate (1.0 molal) and highly concentrated (17 molal or WiSE) NaClO4 aqueous solutions, there is a significant increase in capacitance during the early stages of discharge (t/t0 < 0.4). These findings suggest that ions can rapidly access the readily available mesopores and macropores located near the surface (outer surface regions). As time progresses, the ions become saturated on the surface and attempt to migrate into the more narrow/deep meso- and micro-porous regions, resulting in a capacitance plateau. As the ions access the nanoporous structures, more pronounced capacitance slopes appear until saturation is reached, i.e., these findings confirm that a 1.0 molal NaClO4 solution (Fig. 2(a)) exhibits better ionic conductivity compared to the organic, IL, and eutectic solutions. For the highly concentrated aqueous electrolyte (WiSE), the capacitance plateau ends at 0.5 t/t0, after which the capacitance continues to linearly increase. The latter behavior is attributed to the increased difficulty of ion migration due to ion pairing present in narrow/deep pores located close to the outer surface regions (Fig. 3(b)).
The EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC electrolyte (Fig. 2(c)) displays a pronounced and significantly delayed capacitance increase, especially at high gravimetric currents. This behavior implies slower ion transport kinetics, likely due to a higher viscosity and a low dielectric constant within different pores, which hinders fast migration and delays ion adsorption at the walls of deeper pores that comprise the inner surface regions. The capacitance values are also lower compared to aqueous-based NaClO4 electrolytes, confirming limited accessibility within the porous electrode matrix, which is composed of hierarchically interconnected pores with different characteristic dimensions. When we examine the NaClO4
DEC electrolyte (Fig. 2(c)) displays a pronounced and significantly delayed capacitance increase, especially at high gravimetric currents. This behavior implies slower ion transport kinetics, likely due to a higher viscosity and a low dielectric constant within different pores, which hinders fast migration and delays ion adsorption at the walls of deeper pores that comprise the inner surface regions. The capacitance values are also lower compared to aqueous-based NaClO4 electrolytes, confirming limited accessibility within the porous electrode matrix, which is composed of hierarchically interconnected pores with different characteristic dimensions. When we examine the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2]-based electrolyte system, the capacitance exhibits a gradual and delayed rise during discharge, particularly at 1.0 A g−1. These findings can be attributed to the viscosity and absence of solvent in ionic liquids, which in turn significantly decreases ion mobility. As a result, ion redistribution on carbon surfaces forming the electrical double-layer became more sluggish, and screening is governed by ion layering rather than migration transport. Collectively, these events reduce ion accessibility inside more internal or inner surface regions with a limited pore occupancy. Even for the lowest gravimetric current of 0.02 A g−1, the capacitance does not show complete saturation with the elapsed polarization time, suggesting only partial utilization of the internal/inner surface area.
[C3mpip][NTf2]-based electrolyte system, the capacitance exhibits a gradual and delayed rise during discharge, particularly at 1.0 A g−1. These findings can be attributed to the viscosity and absence of solvent in ionic liquids, which in turn significantly decreases ion mobility. As a result, ion redistribution on carbon surfaces forming the electrical double-layer became more sluggish, and screening is governed by ion layering rather than migration transport. Collectively, these events reduce ion accessibility inside more internal or inner surface regions with a limited pore occupancy. Even for the lowest gravimetric current of 0.02 A g−1, the capacitance does not show complete saturation with the elapsed polarization time, suggesting only partial utilization of the internal/inner surface area.
In a different way, the capacitance findings obtained for the electrolyte system denoted as NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400, shown in Fig. 2(e), reveal an intermediate charge-storage behavior when compared to the aqueous and IL electrolytes. The distributed capacitance increased progressively with the normalized discharge time, reflecting a gradual penetration of ions into the various porous structures present in activated carbon. Although the PEG400 is moderately polar and can solvate Na+ ions, its relatively high viscosity and extensive hydrogen bonding network decrease ion mobility, especially at higher gravimetric currents. From these considerations, we verified that the DES system exhibited inferior capacitances compared to the IL-based system (NaClO4
PEG400, shown in Fig. 2(e), reveal an intermediate charge-storage behavior when compared to the aqueous and IL electrolytes. The distributed capacitance increased progressively with the normalized discharge time, reflecting a gradual penetration of ions into the various porous structures present in activated carbon. Although the PEG400 is moderately polar and can solvate Na+ ions, its relatively high viscosity and extensive hydrogen bonding network decrease ion mobility, especially at higher gravimetric currents. From these considerations, we verified that the DES system exhibited inferior capacitances compared to the IL-based system (NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2]), since the latter displays more efficient ion packing and enhanced surface affinity, enabling higher charge storage.
[C3mpip][NTf2]), since the latter displays more efficient ion packing and enhanced surface affinity, enabling higher charge storage.
From the above considerations, it was verified that the charge-storage performance of SCs can be optimized by matching the size of the solvation shell structure with the average pore sizes present in the carbon-based electrode materials. Even considering that higher capacitances and larger WVWs indicate superior device performance, it is crucial to align the material's properties with specific applications that demand different discharging rates dictated by the gravimetric currents.
We summarized the overall performance of the EDLCs using different electrolyte systems in Fig. 3.
Fig. 3, which shows the dependence of cell capacitance on gravimetric current for various sodium-based electrolytes, allowed us to make vital observations about the studied electrolyte systems. As can be seen, the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] system delivered the highest gravimetric capacitances, exceeding 60 F g−1 at 0.02 A g−1, and retained a relatively large value with the gravimetric current increase. Thus, this system is quite suitable for practical applications demanding fast and high charges (or high-power characteristics). This superior power performance is a consequence of the lack of solvent dielectric screening in ILs, which leads to a dense and compact electric double-layer structure, thus enhancing the overall charge-storage process.
[C3mpip][NTf2] system delivered the highest gravimetric capacitances, exceeding 60 F g−1 at 0.02 A g−1, and retained a relatively large value with the gravimetric current increase. Thus, this system is quite suitable for practical applications demanding fast and high charges (or high-power characteristics). This superior power performance is a consequence of the lack of solvent dielectric screening in ILs, which leads to a dense and compact electric double-layer structure, thus enhancing the overall charge-storage process.
Another observation of the above findings regarding the EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC-based SCs, permitted us to verify well-balanced overall characteristics involving charge-storage and power performance. While Na+ ions form weaker interactions with solvents compared to Li+ ions due to their larger size, their coordination in carbonates, typically through a single carbonyl group (–CO), supports efficient solvation and desolvation processes, thus aiding ion migration during charge/discharge. In the case of the NaOTf
DEC-based SCs, permitted us to verify well-balanced overall characteristics involving charge-storage and power performance. While Na+ ions form weaker interactions with solvents compared to Li+ ions due to their larger size, their coordination in carbonates, typically through a single carbonyl group (–CO), supports efficient solvation and desolvation processes, thus aiding ion migration during charge/discharge. In the case of the NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 system, the latter showed lower gravimetric capacitances compared to the IL system. Even so, this system still outperforms most aqueous and organic carbonate systems at low gravimetric currents (reduced power values).
PEG400 system, the latter showed lower gravimetric capacitances compared to the IL system. Even so, this system still outperforms most aqueous and organic carbonate systems at low gravimetric currents (reduced power values).
At the same time, the presence of moderately high viscosity and a strong hydrogen bonding network limits Na+ mobility; however, the eutectic structure provides a more stable ion–solvent coordination, enabling better access of Na+ ions into the AC pores at low rates, as obtained under reduced power conditions. However, for higher specific currents, the presence of limited ionic migration transport in the PEG400 system becomes more evident, leading to a pronounced decline in the gravimetric capacitance. Surprisingly, the 17 molal NaClO4 WiSE system, even considering its high ionic conductivity, exhibited lower gravimetric capacitances than expected for this type of aqueous-based electrolyte system. This behavior can be explained by the crowded ionic environment present at very high concentrations or WiSE conditions, which disrupts the EDL structure and hinders both ion desolvation and its insertion into the AC micropores. Moreover, strong ion–ion correlations present in WiSE systems may impair the ion mobility and effective ion packing in the porous surface regions.
Self-discharge in SCs represents the spontaneous energy losses characterized by a voltage decrease due to the presence of an undesirable leakage current across the EDL structure under the device's resting conditions. Therefore, a search for strategies aiming to mitigate leakage currents to negligible values by increasing charge-transfer resistance to very high values under open-circuit voltage conditions is mandatory in real applications.
Generally, the conditions to obtain well-behaved and stable SCs can be achieved by properly stabilizing the EDL structure to effectively attain the so-called blocked interface conditions, which are practically represented from the impedance analysis as Rct → ∞, and ϕ → −90° at 10 mHz. Therefore, electrolyte compositions that exhibit a poorly developed semicircle in the complex-plane plot, instead of an almost vertical straight line, do not make sense for practical purposes, since the self-charge process will be strongly pronounced.
The presence of a leakage current mainly manifests through three primary events, often intertwined, given as follows: (i) faradaic reactions for unblocked interfaces (e.g., finite values of Rct, and ϕ < −90°); (ii) internal charge redistribution involving interconnected pores as an spontaneous self-searching for the most stable charge distribution at a given equilibrium voltage, and (iii) current leakage across the EDL structure causing Joule's effect with entropy increase accompanying charge redistribution.
From these considerations, electrolyte chemistry influences the dominant pathways and charge mobility rates due to local differences in electrochemical potential (µ), as proposed by Butler and Guggenheim, to explain the stability under thermodynamic equilibrium (or open circuit voltages), as well as electrochemical stability (absence of solvent electrolysis), viscosity, and conductivity governed by individual ionic mobilities for different solvation shell structures and radii. Therefore, the entropy production-driven processes stem from an initial inhomogeneous charge distribution within the complex, porous electrode structures after the device charging is complete. Fast charging leads to preferential ion accumulation in more easily accessible macropores and mesopores near the porous separator. At the same time, micropores and deeper regions (or inner surface regions) remain sub-occupied with a high/unstable Gibbs' surface energy. As a result, under open-circuit conditions, ions spontaneously diffuse from high-concentration (or high electrochemical potential) to low-concentration (or low electrochemical potential) regions, internally redistributing the overall charge thanks to a reduction in the overall Gibbs free energy and a reduction in the equilibrium cell voltage (Fig. 3(b–d)).
The overall charge transport kinetics are governed by the Nernst–Planck equation, due to the presence of concentration and potential gradients, since convective processes are absent in stagnant electrolytes. While fast charge–discharge capability is a key advantage for high-power electrochemical systems or EDLCs, it can be a drawback for practical applications that require sustained energy output with stable voltages during rest periods, as a fraction of the stored energy can be internally dissipated. As such, Fig. 3(b) shows the self-discharge profiles over 100 h. At first glance, it is observed that all curves representing the charge leakage process across the EDL decrease almost exponentially with time. These findings suggest that the self-discharge spontaneous process strongly depends on the electrolyte composition.
As seen, the worst scenario is verified for the 1.0 molal NaClO4 in EC-DMC-DEC electrolyte and characterized by a sudden voltage decrease in the t < 40 h interval. This means that when the circuit is abruptly opened after the cell is fully charged, there is not enough time for charges to reorganize properly, and the local ion concentration differences then support charge transport involving different pores present in the so-called inner surface regions.51,52
Nevertheless, all cells maintain an energy retention above 70% over 100 h. The IL-based cell maintains the highest residual voltage, indicating minimal leakage current and strong ion immobilization at the interface. The DES also shows slow voltage decay, likely due to its low ionic diffusivity and the strong association between Na+ and the PEG matrix. In contrast, aqueous electrolytes, especially at low concentration (1.0 molal), undergo rapid voltage loss, driven by high ion mobility and low electrochemical stability windows.
When we look at the Ragone plot (Fig. 3(c)), we see a clear picture of how each electrolyte balances the specific energy and power values. Again, NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] stands out, delivering the highest specific energy and maintaining impressive power performance, especially at longer discharge times. This result highlights the IL's ability to sustain high energy storage without sacrificing delivery speed, a tradeoff often encountered in supercapacitor systems. At the same time, despite its lower potential, the EC
[C3mpip][NTf2] stands out, delivering the highest specific energy and maintaining impressive power performance, especially at longer discharge times. This result highlights the IL's ability to sustain high energy storage without sacrificing delivery speed, a tradeoff often encountered in supercapacitor systems. At the same time, despite its lower potential, the EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC system also performs remarkably well; its stable coordination structure enables efficient ion movement at low-to-moderate discharge rates, resulting in a large energy–power profile. Interestingly, the 17 molal NaClO4 WiSE electrolyte, which would typically be expected to perform well due to its high ionic conductivity, showed a pronounced voltage drop with time. Lastly, the 1.0 molal NaClO4 aqueous system delivers the lowest performance in the Ragone plot, as expected due to its low WVW, remembering that energy ∝ U2 (see eqn (2)).
DEC system also performs remarkably well; its stable coordination structure enables efficient ion movement at low-to-moderate discharge rates, resulting in a large energy–power profile. Interestingly, the 17 molal NaClO4 WiSE electrolyte, which would typically be expected to perform well due to its high ionic conductivity, showed a pronounced voltage drop with time. Lastly, the 1.0 molal NaClO4 aqueous system delivers the lowest performance in the Ragone plot, as expected due to its low WVW, remembering that energy ∝ U2 (see eqn (2)).
After all, when we examine the summarized EDLC parameters in Table 2, the IL system clearly demonstrates the best performance for high output energy. At the same time, the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] system is capable of delivering the highest cell capacitance (e.g., 136 F g−1) and maximum energy density (18.96 W h kg−1), thanks to its wider WVW of 4.0 V. However, this comes with a trade-off in terms of high internal resistance (ESR = 14.36 Ω) and moderate self-discharge (e.g., 2.87 V after 1 h), which limits high-power applications. On the other hand, the aqueous electrolytes composed of 1.0 molal NaClO4 and 17 molal NaClO4 WiSE systems offered lower specific energies, associated with reduced ESR values and rapid charge/discharge or high-power characteristics. Collectively, these findings reveal that the latter systems are quite suitable for several practical applications, prioritizing power behavior over specific energy content. Notably, the WiSE system resulted in better specific capacitance (97 F g−1) and energy (2.74 W h kg−1) compared to the 1.0 molal case, albeit with slightly higher ESR and self-discharge.
[C3mpip][NTf2] system is capable of delivering the highest cell capacitance (e.g., 136 F g−1) and maximum energy density (18.96 W h kg−1), thanks to its wider WVW of 4.0 V. However, this comes with a trade-off in terms of high internal resistance (ESR = 14.36 Ω) and moderate self-discharge (e.g., 2.87 V after 1 h), which limits high-power applications. On the other hand, the aqueous electrolytes composed of 1.0 molal NaClO4 and 17 molal NaClO4 WiSE systems offered lower specific energies, associated with reduced ESR values and rapid charge/discharge or high-power characteristics. Collectively, these findings reveal that the latter systems are quite suitable for several practical applications, prioritizing power behavior over specific energy content. Notably, the WiSE system resulted in better specific capacitance (97 F g−1) and energy (2.74 W h kg−1) compared to the 1.0 molal case, albeit with slightly higher ESR and self-discharge.
| Electrolyte | U (V) | C edl (F g−1) | ESR (Ω) | E m (W h kg−1) | P m (kW kg−1) | SD-100h/U-ret. |
|---|---|---|---|---|---|---|
| a Where U is the WVW, Cedl is the EDL capacitance at 0.1 A g−1, ESR is the equivalent series resistance obtained by EIS at the resonance frequency, Em is the maximum specific energy, Pm is the maximum specific power, SD-1h is the open circuit voltage after 1.0 h of self-discharge, and U-ret is the voltage retention after 100 h of self-discharge. | ||||||
1.0 m NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O H2O |
1.20 | 112 | 0.14 | 1.40 | 88.1 | 0.76 V/58% |
17 m NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O H2O |
1.80 | 97 | 0.39 | 2.74 | 81.1 | 1.39 V/77% |
1.0 M NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1 DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
3.00 | 93 | 2.10 | 7.24 | 70.5 | 2.30 V/77% |
1.0 M NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1 DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
2.60 | 77 | 1.14 | 4.50 | 88.2 | 2.12 V/82% |
1.0 M NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EC EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC DEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1 DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
3.00 | 73 | 9.00 | 5.72 | 12.5 | 2.03 V/68% |
NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] [C3mpip][NTf2] |
4.00 | 136 | 14.4 | 19.0 | 63.3 | 2.77 V/69% |
NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 PEG400 |
3.40 | 73 | 2.36 | 7.30 | 27.8 | 2.47 V/73% |
As seen, among the organic carbonate-based systems, NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1
DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) conferred a balance involving moderate specific energy (4.50 W h kg−1), high power (88.2 kW kg−1), and low self-discharge (2.19 V). At the same time, NaClO4-EC
1) conferred a balance involving moderate specific energy (4.50 W h kg−1), high power (88.2 kW kg−1), and low self-discharge (2.19 V). At the same time, NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC
DEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMC (1
DMC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) delivers high power (12.5 kW kg−1) allied to moderate specific energy (5.72 W h kg−1). Regarding the DES system (NaOTf
1) delivers high power (12.5 kW kg−1) allied to moderate specific energy (5.72 W h kg−1). Regarding the DES system (NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG600), which does not feature top performance for isolated parameters, we verified a high specific energy (7.30 W h kg−1) allied to similar self-discharge characteristics (2.65 V/78%), revealing promising findings comprising stability and charge-storage performance.
PEG600), which does not feature top performance for isolated parameters, we verified a high specific energy (7.30 W h kg−1) allied to similar self-discharge characteristics (2.65 V/78%), revealing promising findings comprising stability and charge-storage performance.
3.3 Residual gas analysis using operando mass spectrometry
To further analyze the charge accumulation process and the stability of the electrolyte compounds, we determine the gas fractions under polarization conditions. The setup applied was the same as described in ref. 29. The cell was configured with the working electrode positioned at the base and the counter electrode at the top, both fully immersed in the electrolyte. To protect the mass spectrometer's high-vacuum chamber (p = 6 µbar) from electrolyte intrusion, a membrane barrier was employed. The gases analyzed were CO (m/z = 28), CO2 (m/z = 44), O2 (m/z = 32), H2 (m/z = 2), H2O (m/z = 18), HF (m/z = 20), and CH4 (m/z = 16), and the mass-to-charge ratio signals were collected during the experiments. The gases generated are drawn into the chamber by the pressure difference. Gases produced at the top electrode will reach the detector faster than those produced at the bottom, as these gases must first diffuse through the separator or move through a volume of electrolyte to reach the detector. However, this delay could result in extended dwell time in the cell, allowing secondary reactions to occur. At this stage, we aim to explore the performance of the concentrated aqueous solution (17 molal NaClO4), the organic solution 1.0 molal NaClO4-EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC, NaClO4
DEC, NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2], and NaOTf
[C3mpip][NTf2], and NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400, respectively.
PEG400, respectively.
Fig. 4 presents the various findings from the mass spectrometry analysis involving the evolved gases.
Fig. 4(a) shows the 17 molal NaClO4 performance as the cell is cycled. The electrochemical testing includes CV, SPCA, EIS, and GCD at operational limits for 40 h, alongside tracking transient current and voltage responses. At the early stages, as the EDLC charges, the current spikes cause system disturbances, making the gases rapidly evolve. When it reaches the equilibria, the gases start to fade, which is consistent with the solvent-blocked interface formation once reported.27,50 Under polarization, electrolytes interact with the functional groups on the carbon surface, creating ionic aggregations that disrupt the hydrogen-bond network, affecting the physicochemical structure of the bulk liquid phase. This results in a protective, insulating passivation layer that reduces further water decomposition.
Thermodynamically, the high salt concentration induces a strong salting-out effect, significantly reducing the activity and partial pressure of dissolved gases, such as O2 and H2, within the electrolyte matrix. This effect reduces the effective chemical potential of water, thereby decreasing the thermodynamic driving force for electrolysis and shifting the onset of gas evolution to higher cell voltages than the standard 1.23 V predicted under ideal dilute conditions.50,53
However, despite this apparent stability, mass spectrometry reveals that gas evolution, particularly H2 and minor fragments of O2, emerges gradually after a floating time or prolonged cycling, indicating that water reduction and minor oxidative decomposition are not completely suppressed but kinetically hindered. At the molecular level, this kinetic suppression is rooted in the strong electrolyte–electrode interactions characteristic of WISE systems. The high concentration of salt promotes extensive ion pairing and contact-ion networks that reorganize at the carbon surface, interacting specifically with oxygen-containing functional groups such as OH, –COOH, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O.54,55 This delay is attributed to the formation of a densely packed, ion-saturated interfacial layer (analogous to the solid-electrolyte interphase, SEI), which increases the activation energy for electron transfer. It is composed predominantly of ion aggregates, reduced anion fragments, and tightly coordinated water clusters, which are electronically insulating but selectively permeable to cations, restricting electron tunneling and suppressing direct water access to electrochemically active edge sites. As this layer thickens and becomes more ordered, it increases the activation barrier for both proton reduction and water-splitting pathways. According to the Butler–Volmer kinetics, this translates into a higher overpotential required to initiate hydrogen evolution, especially within the compact Helmholtz layer, where ion crowding restricts solvent accessibility. As a result, while WiSE systems enable extended electrochemical stability windows, the slow and progressive gas evolution observed underscores the eventual breakdown of interfacial passivation, likely linked to mechanical or chemical SBI disruption under repeated cycling or higher applied currents.
O.54,55 This delay is attributed to the formation of a densely packed, ion-saturated interfacial layer (analogous to the solid-electrolyte interphase, SEI), which increases the activation energy for electron transfer. It is composed predominantly of ion aggregates, reduced anion fragments, and tightly coordinated water clusters, which are electronically insulating but selectively permeable to cations, restricting electron tunneling and suppressing direct water access to electrochemically active edge sites. As this layer thickens and becomes more ordered, it increases the activation barrier for both proton reduction and water-splitting pathways. According to the Butler–Volmer kinetics, this translates into a higher overpotential required to initiate hydrogen evolution, especially within the compact Helmholtz layer, where ion crowding restricts solvent accessibility. As a result, while WiSE systems enable extended electrochemical stability windows, the slow and progressive gas evolution observed underscores the eventual breakdown of interfacial passivation, likely linked to mechanical or chemical SBI disruption under repeated cycling or higher applied currents.
When we look at the WISE mass spectra, the hydrogen fraction is the dominant gaseous product during discharge phases. This indicates that water reduction is a primary degradation route despite the high salt concentration intended to suppress water activity. The evolution of H2 closely follows the voltage oscillations, confirming its electrochemical origin via the hydrogen evolution reaction (HER) shown below. Over time, the intensity of H2 production gradually decreases, suggesting either partial passivation of the electrode surface or a reduction in the availability of free water near the electrode interface.
 | (9) |
Simultaneously, sharp peaks in carbon dioxide (CO2) and carbon monoxide (CO) are also detected. These species indicate oxidative degradation of the carbon electrode, particularly at higher anodic potentials. CO2 is typically associated with complete oxidation of carbon, while CO indicates partial oxidation or decomposition of oxygen-containing functional groups (like C–OH, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, COOH). The gradual decline in their signal intensities may be attributed to the formation of resistive surface layers that limit further attack on the electrode.
O, COOH). The gradual decline in their signal intensities may be attributed to the formation of resistive surface layers that limit further attack on the electrode.
| C + 2H2O → CO2 + 4H+ + 4e− | (10) |
| C + 2OH− → CO + H2O + 2e− | (11) |
Traces of oxygen (O2) are present, albeit at lower levels than expected from classical water oxidation. This suggests that the high concentration of NaClO4, which reduces water activity and mobility, may suppress the oxygen evolution reaction (OER) to some extent. Nonetheless, small amounts of O2 evolution indicate that some water oxidation still occurs at high potentials. The presence of methane (CH4) and water vapor (H2O) signals, especially during the initial hours, is likely linked to the decomposition of surface-bound organics or reactions involving radicals generated during perchlorate breakdown. Perchlorate (ClO4−), though thermodynamically stable, may partially degrade under extreme anodic stress, producing reactive oxygen species (ROS) such as –OH and atomic oxygen. These highly reactive intermediates can interact with carbon surfaces or residual organic species, leading to CH4 evolution.
 | (12) |
Taken together, the gas evolution spectra suggest that the WiSE electrolyte undergoes multiple degradation pathways during extended operation. Water electrolysis (both HER and OER), carbon corrosion, and possible anion decomposition all contribute to the formation of gaseous byproducts. These processes not only impact the long-term stability and safety of the supercapacitor but also provide crucial information for optimizing electrolyte formulation and voltage window limits in future designs.
Following this, one can examine the mass spectra from EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC-based EDLC in Fig. 4(b). First, CO2 and CH4 evolve periodically, maintaining the same current levels through the cycles. The H2 appears in cycles, but its signal amplitude is intensified at the beginning of the cycle. These gases originate from the electrochemical decomposition of the organic solvents ethylene carbonate (EC) and diethyl carbonate (DEC).56 Under anodic conditions, EC and DEC can undergo oxidative cleavage, producing CO2 as a primary product. The CH4 signal, which follows a similar periodic trend, suggests alkyl group fragmentation, likely from the DEC component. Formaldehyde (HCHO), a key intermediate in DEC degradation, may undergo further electrochemical oxidation to CO and CO2, often accompanied by hydrogen evolution.29,57,58 The recurrence of these gas peaks during each electrochemical cycle indicates that solvent degradation is repetitive and triggered by polarization, particularly during high-voltage charging events; however, the low signal amplitude suggests that it is an inherent process. Interestingly, while CO2 and CH4 signals are intense and highly modulated by the electrochemical regime, CO shows a more subtle and continuous presence. This suggests that CO forms through a parallel degradation route, possibly involving incomplete oxidation or decarboxylation of carbonate intermediates. Additionally, glycol-type fragments generated from EC ring-opening may suffer further oxidation or cleavage, contributing to the formation of CH4, CO, and CO2.56,59,60 Unlike CO2 and CH4, which appear in bursts, the CO signal persists with lower amplitude, pointing to ongoing, less energetically demanding decomposition reactions at the electrode surface, possibly during low-current or rest periods. In this way, we proposed the following mechanism in Fig. 5.
DEC-based EDLC in Fig. 4(b). First, CO2 and CH4 evolve periodically, maintaining the same current levels through the cycles. The H2 appears in cycles, but its signal amplitude is intensified at the beginning of the cycle. These gases originate from the electrochemical decomposition of the organic solvents ethylene carbonate (EC) and diethyl carbonate (DEC).56 Under anodic conditions, EC and DEC can undergo oxidative cleavage, producing CO2 as a primary product. The CH4 signal, which follows a similar periodic trend, suggests alkyl group fragmentation, likely from the DEC component. Formaldehyde (HCHO), a key intermediate in DEC degradation, may undergo further electrochemical oxidation to CO and CO2, often accompanied by hydrogen evolution.29,57,58 The recurrence of these gas peaks during each electrochemical cycle indicates that solvent degradation is repetitive and triggered by polarization, particularly during high-voltage charging events; however, the low signal amplitude suggests that it is an inherent process. Interestingly, while CO2 and CH4 signals are intense and highly modulated by the electrochemical regime, CO shows a more subtle and continuous presence. This suggests that CO forms through a parallel degradation route, possibly involving incomplete oxidation or decarboxylation of carbonate intermediates. Additionally, glycol-type fragments generated from EC ring-opening may suffer further oxidation or cleavage, contributing to the formation of CH4, CO, and CO2.56,59,60 Unlike CO2 and CH4, which appear in bursts, the CO signal persists with lower amplitude, pointing to ongoing, less energetically demanding decomposition reactions at the electrode surface, possibly during low-current or rest periods. In this way, we proposed the following mechanism in Fig. 5.
Hydrogen evolution is also evident, although it presents a more irregular profile. Its presence is typically associated with trace water reduction or proton release during solvent degradation. While the EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC mixture is nominally aprotic, small amounts of water or protic species, either from contamination or reaction intermediates, can be reduced at the negative electrode. The persistence of H2 throughout the cycling process suggests that parasitic reduction reactions continue even after initial polarization and that the system does not fully passivate over time.
DEC mixture is nominally aprotic, small amounts of water or protic species, either from contamination or reaction intermediates, can be reduced at the negative electrode. The persistence of H2 throughout the cycling process suggests that parasitic reduction reactions continue even after initial polarization and that the system does not fully passivate over time.
Taken together, the gas evolution pattern reflects a multi-pathway degradation mechanism. Upon electrochemical polarization, EC and DEC are activated and undergo oxidative decomposition, releasing CO2 and CH4 during anodic sweeps. Simultaneously, at the cathodic interface, H2 evolution occurs due to residual water or protonated species. The relatively stable production of CO suggests partial degradation of carbonate solvents through a separate channel that remains active even at lower potentials or during open-circuit phases.
At the electrode–electrolyte interface, these degradation phenomena correlate with the formation of a fragile, carbonate-derived SEI-like layer whose composition is driven by the reductive decomposition of EC and DEC. XPS analysis (please refer to Fig. S2 in the SI) confirms the presence of functional groups in the electrode that can react with these compounds. These interactions initially generate a thin passivation film; however, its intrinsic mechanical fragility and poor ionic cohesion render it highly prone to cracking under cycling. Each rupture re-exposes reactive edge sites, triggering renewed solvent decomposition and sustaining the continuous gas evolution observed.
This behavior is consistent with the formation of an SEI at the carbon electrode, although in supercapacitors this layer is typically thinner and less protective than in batteries. The repeated breakdown of solvent molecules suggests that the SEI is continuously disrupted during cycling. In contrast to aqueous systems, where water splitting dominates the degradation process, solvent oxidation and reduction are the primary pathways here, leading to the evolution of hydrocarbon and carbon dioxide gases. In summary, the mass spectra provide direct evidence that 1.0 M NaClO4 in EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC (1
DEC (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), is chemically unstable under prolonged operation.
1), is chemically unstable under prolonged operation.
Conversely, the NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] complex doesn't exhibit constant behavior (please see Fig. 4(c)); CO, CO2, H2, and CH4 start to evolve in the early stages of the dynamic process. It reveals a high degree of chemical and electrochemical stability, even with continuous cycling, the corresponding gas signals of CO2, CH4, O2, CO, and H2 remain minimal and decrease progressively over time. This absence of strong periodic gas peaks, in contrast to what is often observed in less stable systems, suggests that the functional groups present in the [C3mpip]+ cation and [NTf2]− anion are relatively resistant to electrochemical decomposition under the applied conditions. The initial release of CO2 and CH4 may be linked to minor surface reactions or residual decomposition of carbon-based functional groups (eqn (10) and (11)), possibly from the electrode or trace impurities. However, the rapid decline in their intensity supports the idea that a thin but effective passivating layer forms early in the cycling, limiting further breakdown. Similarly, O2 and CO evolution is observed mainly during the first few hours and corresponds to the oxidative removal of surface oxides or decomposition of labile oxygenated species. The minimal and sporadic H2 signal reinforces the notion that water content is negligible and that proton reduction is not a dominant pathway in this system. Overall, the data imply that the ionic liquid system promotes a stable interfacial environment, and the low gas evolution is primarily a result of limited functional group reactivity and strong electrochemical resilience of the ionic liquid components.
[C3mpip][NTf2] complex doesn't exhibit constant behavior (please see Fig. 4(c)); CO, CO2, H2, and CH4 start to evolve in the early stages of the dynamic process. It reveals a high degree of chemical and electrochemical stability, even with continuous cycling, the corresponding gas signals of CO2, CH4, O2, CO, and H2 remain minimal and decrease progressively over time. This absence of strong periodic gas peaks, in contrast to what is often observed in less stable systems, suggests that the functional groups present in the [C3mpip]+ cation and [NTf2]− anion are relatively resistant to electrochemical decomposition under the applied conditions. The initial release of CO2 and CH4 may be linked to minor surface reactions or residual decomposition of carbon-based functional groups (eqn (10) and (11)), possibly from the electrode or trace impurities. However, the rapid decline in their intensity supports the idea that a thin but effective passivating layer forms early in the cycling, limiting further breakdown. Similarly, O2 and CO evolution is observed mainly during the first few hours and corresponds to the oxidative removal of surface oxides or decomposition of labile oxygenated species. The minimal and sporadic H2 signal reinforces the notion that water content is negligible and that proton reduction is not a dominant pathway in this system. Overall, the data imply that the ionic liquid system promotes a stable interfacial environment, and the low gas evolution is primarily a result of limited functional group reactivity and strong electrochemical resilience of the ionic liquid components.
Finally, the gas evolution profile for the NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 electrolyte (Fig. 4(d)) shows a distinct pattern of instability and active degradation throughout the electrochemical cycling. Unlike the relatively stable ionic liquid system, this deep eutectic solvent exhibits high-frequency, pulsatile gas emissions, notably HF, CO2, CH4, O2, CO, and H2, indicating a dynamic and chemically reactive interfacial environment. The pronounced and early signal of HF is particularly significant, pointing to decomposition of the triflate anion (OTf−). This behavior is consistent with known reactions on highly reactive surfaces, where C–F and C–S bond scission leads to the formation of NaF, CF2, and SO3 species. This decomposition is less likely on inert substrates (e.g., graphite), but in the context of supercapacitors with carbon electrodes, localized hot spots or radical formation under polarization may promote partial OTf− breakdown.61–63
PEG400 electrolyte (Fig. 4(d)) shows a distinct pattern of instability and active degradation throughout the electrochemical cycling. Unlike the relatively stable ionic liquid system, this deep eutectic solvent exhibits high-frequency, pulsatile gas emissions, notably HF, CO2, CH4, O2, CO, and H2, indicating a dynamic and chemically reactive interfacial environment. The pronounced and early signal of HF is particularly significant, pointing to decomposition of the triflate anion (OTf−). This behavior is consistent with known reactions on highly reactive surfaces, where C–F and C–S bond scission leads to the formation of NaF, CF2, and SO3 species. This decomposition is less likely on inert substrates (e.g., graphite), but in the context of supercapacitors with carbon electrodes, localized hot spots or radical formation under polarization may promote partial OTf− breakdown.61–63
Interfacial interaction analysis further reinforces the absence of a stable SEI-like film in this DES system. XPS data (Fig. S6 in SI) confirms the natural presence of oxygen-containing functional groups on the activated carbon, such as C–O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and COOH, which are known to interact with electrolyte components. In the NaOTf
O and COOH, which are known to interact with electrolyte components. In the NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 environment, these functional groups form only weak and dynamic interactions with PEG chains and OTf− fragments (e.g., hydrogen bonding or loose dipole–ion coordination). Unlike organic carbonates or ionic liquids, these interactions are insufficient to yield a compact, electronically insulating interphase. Instead, they generate a highly fragile, discontinuous passivation layer that is repeatedly disrupted during cycling. Each rupture exposes fresh carbon edge sites, triggering renewed OTf− cleavage and PEG oxidation, which directly contributes to the pulsatile and multi-gas degradation pattern observed in the mass spectrometry signals. Thus, the interfacial chemistry of DES systems inherently favors intermittent passivation rather than SEI formation, explaining their lower stability and accelerated degradation.
PEG400 environment, these functional groups form only weak and dynamic interactions with PEG chains and OTf− fragments (e.g., hydrogen bonding or loose dipole–ion coordination). Unlike organic carbonates or ionic liquids, these interactions are insufficient to yield a compact, electronically insulating interphase. Instead, they generate a highly fragile, discontinuous passivation layer that is repeatedly disrupted during cycling. Each rupture exposes fresh carbon edge sites, triggering renewed OTf− cleavage and PEG oxidation, which directly contributes to the pulsatile and multi-gas degradation pattern observed in the mass spectrometry signals. Thus, the interfacial chemistry of DES systems inherently favors intermittent passivation rather than SEI formation, explaining their lower stability and accelerated degradation.
The CO2 and CH4 signals suggest oxidative degradation of the PEG400 matrix, likely involving terminal –OH groups and ether linkages undergoing cleavage, especially during repeated voltage excursions. Their intermittent emission pattern indicates a non-uniform passivation layer, where localized rupture and reformation cycles repeatedly expose fresh electrode surface to the electrolyte. The detection of O2 and CO further supports partial oxidation and rearrangement of organic species and anion fragments, possibly enhanced by trace water or reactive oxygen intermediates.64–66 The H2 evolution profile, although not dominant, is sustained and aligned with cathodic cycles, indicating the reduction of either trace water or hydrogen-containing fragments from PEG decomposition.
This complex gas behavior is further explained by the coordination chemistry of Na+ in DES: while OTf− is a weakly coordinating anion under dilute conditions, at higher concentrations or in viscous media like PEG400, ion-pair formation becomes significant. This leads to stronger Na+–OTf− interactions, formation of contact ion pairs, and potentially larger ionic aggregates. These interactions increase viscosity and reduce ionic mobility, which not only limits electrochemical performance but also concentrates reactive species near the interface, contributing to localized decomposition and elevated gas evolution.61
Altogether, the mass spectrometry data highlight that NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400, despite its initial promise as a safe and stable electrolyte, suffers from poor interfacial stability under prolonged cycling. The high reactivity of the OTf− anion under reductive and oxidative stresses, combined with the susceptibility of the PEG solvent to degradation, leads to fragmented passivation, continuous gas evolution, and a likely disordered, inhomogeneous layer. These findings underscore the importance of molecular-level tuning of DES components or additive strategies to stabilize the interfacial chemistry and mitigate electrolyte decomposition in sodium-based energy storage systems.
PEG400, despite its initial promise as a safe and stable electrolyte, suffers from poor interfacial stability under prolonged cycling. The high reactivity of the OTf− anion under reductive and oxidative stresses, combined with the susceptibility of the PEG solvent to degradation, leads to fragmented passivation, continuous gas evolution, and a likely disordered, inhomogeneous layer. These findings underscore the importance of molecular-level tuning of DES components or additive strategies to stabilize the interfacial chemistry and mitigate electrolyte decomposition in sodium-based energy storage systems.
4 Conclusions
This study systematically investigated the influence of solvent systems on the electrochemical behavior of sodium-based supercapacitors, with a focus on optimizing performance through electrolyte composition properties. By employing aqueous, organic, ionic liquid, and deep eutectic solvent systems in combination with Na-based salts, we were able to assess key performance metrics, including capacitance, power, and energy densities, as well as stability and gas evolution, through operando techniques. Our findings confirmed that ionic liquids, particularly NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2], delivered the highest energy density (19 W h kg−1) and maximum capacitance (136 F g−1) due to their extended electrochemical stability window and efficient ion packing. However, their high internal resistance and moderate self-discharge point to limitations for high-power applications.
[C3mpip][NTf2], delivered the highest energy density (19 W h kg−1) and maximum capacitance (136 F g−1) due to their extended electrochemical stability window and efficient ion packing. However, their high internal resistance and moderate self-discharge point to limitations for high-power applications.
In contrast, the water-in-salt electrolyte (17 molal NaClO4) exhibited fast ionic mobility and excellent conductivity, making it favorable for high-power output, despite showing a reduced energy density due to constrained voltage windows. Organic carbonates, such as EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DEC, provided a balanced electrolyte performance, offering a wider stability window than aqueous systems and a well-defined solvation structure that supports stable charge/discharge cycles. Meanwhile, the NaOTf
DEC, provided a balanced electrolyte performance, offering a wider stability window than aqueous systems and a well-defined solvation structure that supports stable charge/discharge cycles. Meanwhile, the NaOTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEG400 DES, although not the best performer in absolute metrics, showed promising intermediate behavior with good voltage stability and moderate energy retention, underlining its potential for sustainable electrolyte design.
PEG400 DES, although not the best performer in absolute metrics, showed promising intermediate behavior with good voltage stability and moderate energy retention, underlining its potential for sustainable electrolyte design.
Operando electrochemical mass spectrometry revealed significant insights into the degradation mechanisms, confirming that aqueous and carbonate-based systems are more prone to gas evolution, particularly H2 and CO2, associated with water electrolysis and solvent oxidation. Conversely, the IL systems exhibited minimal and delayed gas evolution, suggesting more robust interfacial chemistry and potential for improved long-term stability. Unlike the others, the DES electrolyte exhibited poor cyclability, losing performance more rapidly than the aqueous/organic electrolytes and releasing toxic gases, such as HF.
Overall, the results demonstrate that tailoring the solvation environment through careful solvent selection enables fine-tuning of the electrochemical properties of sodium-based supercapacitors. The NaClO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [C3mpip][NTf2] system emerges as the most promising candidate for applications demanding high energy density and extended operating voltage, while the 17 molal aqueous solution is ideal for power-oriented use. These findings offer a practical roadmap for electrolyte engineering in next-generation sodium-ion-based energy storage devices.
[C3mpip][NTf2] system emerges as the most promising candidate for applications demanding high energy density and extended operating voltage, while the 17 molal aqueous solution is ideal for power-oriented use. These findings offer a practical roadmap for electrolyte engineering in next-generation sodium-ion-based energy storage devices.
Conflicts of interest
There are no conflicts to declare.Data availability
All data are available upon request from the corresponding author.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta07938a.
Acknowledgements
The authors are very grateful for the financial support from Shell and acknowledge they ANP (Brazil’s National Oil, Natural Gas, and Biofuels Agency) through the R&D levy regulation. They also extend their appreciation to the Brazilian funding agencies FAPESP (2017/11958-1, 2017/11986-5, 2014/02163-7 & 2023/17560-0), CAPES, and CNPq (405643/2022-5 & 302180/2022-2). L.M. Da Silva wishes to thank FAPEMIG (Financial support for the LMMA/UFVJM Laboratory) and CNPq (PQ-2 grant: Process 301095/2018-3).References
- A. B. Gallo, J. R. Simões-Moreira, H. K. M. Costa, M. M. Santos and E. Moutinho dos Santos, Energy storage in the energy transition context: A technology review, Renewable Sustainable Energy Rev., 2016, 65, 800–822, DOI:10.1016/j.rser.2016.07.028
.
- K. Fic, A. Platek, J. Piwek and E. Frackowiak, Sustainable materials for electrochemical capacitors, Mater. Today, 2018, 21, 437–454, DOI:10.1016/j.mattod.2018.03.005
.
- F. Fuso Nerini, J. Tomei, L. S. To, I. Bisaga, P. Parikh, M. Black, A. Borrion, C. Spataru, V. Castán Broto, G. Anandarajah, B. Milligan and Y. Mulugetta, Mapping synergies and trade-offs between energy and the Sustainable Development Goals, Nat. Energy, 2018, 3, 10–15, DOI:10.1038/s41560-017-0036-5
.
- É. A. Santos, M. C. Policano, M. J. Pinzón, I. Galantini, V. A. Gonçalves, F. C. B. Maia, L. J. A. Macedo, G. Doubek, R. G. Freitas and H. Zanin, Operando FTIR study on water additive in lithium-sulfur batteries to mitigate shuttle effect, J. Energy Chem., 2024, 98, 702–713, DOI:10.1016/J.JECHEM.2024.06.060
.
- S. G. Booth, A. J. Nedoma, N. N. Anthonisamy, P. J. Baker, R. Boston, H. Bronstein, S. J. Clarke, E. J. Cussen, V. Daramalla, M. De Volder, S. E. Dutton, V. Falkowski, N. A. Fleck, H. S. Geddes, N. Gollapally, A. L. Goodwin, J. M. Griffin, A. R. Haworth, M. A. Hayward, S. Hull, B. J. Inkson, B. J. Johnston, Z. Lu, J. L. MacManus-Driscoll, X. Martínez De Irujo Labalde, I. McClelland, K. McCombie, B. Murdock, D. Nayak, S. Park, G. E. Pérez, C. J. Pickard, L. F. J. Piper, H. Y. Playford, S. Price, D. O. Scanlon, J. C. Stallard, N. Tapia-Ruiz, A. R. West, L. Wheatcroft, M. Wilson, L. Zhang, X. Zhi, B. Zhu and S. A. Cussen, Perspectives for next generation lithium-ion battery cathode materials, APL Mater., 2021, 9, 109201, DOI:10.1063/5.0051092/123081
.
- Y. A. Kumar, N. Roy, T. Ramachandran, M. Hussien, M. Moniruzzaman and S. W. Joo, Shaping the future of energy: The rise of supercapacitors progress in the last five years, J. Energy Storage, 2024, 98, 113040, DOI:10.1016/J.EST.2024.113040
.
- S. Banerjee, B. Mordina, P. Sinha and K. K. Kar, Recent advancement of supercapacitors: A current era of supercapacitor devices through the development of electrical double layer, pseudo and their hybrid supercapacitor electrodes, J. Energy Storage, 2025, 108, 115075, DOI:10.1016/J.EST.2024.115075
.
- Z. S. Iro, C. Subramani and S. S. Dash, A Brief Review on Electrode Materials for Supercapacitor, Int. J. Electrochem. Sci., 2016, 11, 10628–10643, DOI:10.20964/2016.12.50
.
- E. Frackowiak, Carbon materials for supercapacitor application, Phys. Chem. Chem. Phys., 2007, 9, 1774–1785, 10.1039/B618139M
.
- M. M. Amaral, R. Venâncio, A. C. Peterlevitz and H. Zanin, Recent advances on quasi-solid-state electrolytes for supercapacitors, J. Energy Chem., 2022, 67, 697–717, DOI:10.1016/j.jechem.2021.11.010
.
-
A. K. S. S. M. Riyaz, A. M. Shanmugharaj, M. D. Kumar and R. K. Pai, Advances in Lithium-Ion and Sodium-Ion-Based Supercapacitors: Prospects and Challenges, Nanostructured Materials for Energy Storage I-IV, 2024, pp. 1293–1342, DOI:10.1002/9783527838851.ch35
.
- W. Raza, F. Ali, N. Raza, Y. Luo, K. H. Kim, J. Yang, S. Kumar, A. Mehmood and E. E. Kwon, Recent advancements in supercapacitor technology, Nano Energy, 2018, 52, 441–473, DOI:10.1016/J.NANOEN.2018.08.013
.
- X. Tian, Q. Zhu and B. Xu, “Water-in-Salt” Electrolytes for Supercapacitors: A Review, ChemSusChem, 2021, 14, 2501–2515, DOI:10.1002/CSSC.202100230
.
- P. Lannelongue, R. Bouchal, E. Mourad, C. Bodin, M. Olarte, S. le Vot, F. Favier and O. Fontaine, “Water-in-Salt” for Supercapacitors: A Compromise between Voltage, Power Density, Energy Density and Stability, J. Electrochem. Soc., 2018, 165, A657–A663, DOI:10.1149/2.0951803JES/DATA
.
- S. Gharouel and F. Béguin, Revisiting the performance of electrical double-layer capacitors implementing a sodium perchlorate water-in-salt electrolyte, Electrochim. Acta, 2023, 450, 142212, DOI:10.1016/J.ELECTACTA.2023.142212
.
- K. Mahankali, N. K. Thangavel, Y. Ding, S. K. Putatunda and L. M. R. Arava, Interfacial behavior of water-in-salt electrolytes at porous electrodes and its effect on supercapacitor performance, Electrochim. Acta, 2019, 326, 134989, DOI:10.1016/J.ELECTACTA.2019.134989
.
- R. Väli, A. Laheäär, A. Jänes and E. Lust, Characteristics of non-aqueous quaternary solvent mixture and Na-salts based supercapacitor electrolytes in a wide temperature range, Electrochim. Acta, 2014, 121, 294–300, DOI:10.1016/J.ELECTACTA.2013.12.149
.
- S. Suh, J. Lee, J. Park and W. Kim, The strong correlations between the performance of a KB supercapacitor and the properties of NaClO4 and LiFSI electrolytes over wide concentration ranges, Int. J. Energy Res., 2022, 46, 18676–18688, DOI:10.1002/ER.8485
.
- X. Bu, L. Su, Q. Dou, S. Lei and X. Yan, A low-cost “water-in-salt” electrolyte for a 2.3 V high-rate carbon-based supercapacitor, J. Mater. Chem. A, 2019, 7, 7541–7547, 10.1039/C9TA00154A
.
- W. G. Nunes, B. G. A. Freitas, R. M. Beraldo, R. M. Filho, L. M. Da Silva and H. Zanin, A rational experimental approach to identify correctly the working voltage window of aqueous-based supercapacitors, Sci. Rep., 2020, 10, 19195, DOI:10.1038/s41598-020-75851-7
.
- H. de A. Chagas, G. Colherinhas and E. E. Fileti, Unveiling the Potential of Natural Deep Eutectic Solvents in Electrochemical Energy Storage Applications, ACS Phys. Chem. Au, 2025 DOI:10.1021/ACSPHYSCHEMAU.5C00063
.
- K. Pithaksinsakul, P. Chatsiri, J. Deng, T. Burton, C. Pareseecharoen, Y. Zhu, V. Promarak and O. Fontaine, Developing carbazole-based redox ionic liquids for enhanced charge retention and stability in porous carbon electrode, Discover Electrochem., 2025, 2(1 2), 50, DOI:10.1007/S44373-025-00064-4
.
- F. T. Zohura, A. H. Reaz, M. M. A. Khan, M. R. Islam, R. A. Mahmood, C. K. Roy and S. H. Firoz, Strategic modulation of hydrogen bonding networks in aqueous electrolytes using green deep eutectic solvents: Experimental and computational insights for enhanced capacitive performance in symmetric supercapacitors, Electrochim. Acta, 2025, 532, 146455, DOI:10.1016/J.ELECTACTA.2025.146455
.
- S. Amara, W. Zaidi, L. Timperman, G. Nikiforidis and M. Anouti, Amide-based deep eutectic solvents containing LiFSI and NaFSI salts as superionic electrolytes for supercapacitor applications, J. Chem. Phys., 2021, 154, 164708, DOI:10.1063/5.0048392
.
- T. Guillemin, C. Douard, K. Robert, B. Asbani, C. Lethien, T. Brousse and J. Le Bideau, Solid-state 3D micro-supercapacitors based on ionogel electrolyte: Influence of adding lithium and sodium salts to the ionic liquid, Energy Storage Mater., 2022, 50, 606–617, DOI:10.1016/J.ENSM.2022.05.041
.
- S. Thior, V. N. Kitenge, N. F. Diop, K. O. Otun, V. M. Maphiri, R. A. M. Adam, B. D. Ngom and N. Manyala, Enhancing potential window with ionic liquid/water in salt mixture in co-doped activated carbon electrodes for high-energy supercapacitors, J. Energy Storage, 2026, 141, 119425, DOI:10.1016/J.EST.2025.119425
.
- R. Vicentini, R. Venâncio, W. Nunes, L. M. Da Silva and H. Zanin, New Insights on the Sodium Water-in-Salt Electrolyte and Carbon Electrode Interface from Electrochemistry and Operando Raman Studies, ACS Appl. Mater. Interfaces, 2021, 13, 61139–61153, DOI:10.1021/acsami.1c18777
.
- R. Vicentini, J. P. Aguiar, R. Beraldo, R. Venâncio, F. Rufino, L. M. Da Silva and H. Zanin, Ragone Plots for Electrochemical Double-Layer Capacitors, Batteries Supercaps, 2021, 4, 1291–1303, DOI:10.1002/batt.202100093
.
- R. Venâncio, R. Vicentini, M. Pinzón, D. A. Corrêa, A. N. de Miranda, A. C. Queiroz, F. T. Degasperi, L. J. A. Siqueira, L. M. Da Silva and H. Zanin, Combining Electrochemical, Molecular Simulation and Operando techniques to investigate the stability of electrodes and organic electrolytes used in EDLCs, Energy Storage Mater., 2023, 62, 102943, DOI:10.1016/j.ensm.2023.102943
.
- R. Sakamoto, M. Yamashita, K. Nakamoto, Y. Zhou, N. Yoshimoto, K. Fujii, T. Yamaguchi, A. Kitajou and S. Okada, Local structure of a highly concentrated NaClO4 aqueous solution-type electrolyte for sodium ion batteries, Phys. Chem. Chem. Phys., 2020, 22, 26452–26458, 10.1039/D0CP04376A
.
- F. Endres and S. Zein El Abedin, Air and water stable ionic liquids in physical chemistry, Phys. Chem. Chem. Phys., 2006, 8, 2101–2116, 10.1039/B600519P
.
- A. Yochelis, M. B. Singh and I. Visoly-Fisher, Coupling Bulk and Near-Electrode Interfacial Nanostructuring in Ionic Liquids, Chem. Mater., 2015, 27, 4169–4179, DOI:10.1021/ACS.CHEMMATER.5B00780
.
- S. A. Forsyth, J. M. Pringle and D. R. MacFarlane, Ionic liquids - An overview, Aust. J. Chem., 2004, 57, 113–119, DOI:10.1071/CH03231
.
- D. R. MacFarlane, M. Forsyth, P. C. Howlett, M. Kar, S. Passerini, J. M. Pringle, H. Ohno, M. Watanabe, F. Yan, W. Zheng, S. Zhang and J. Zhang, Ionic liquids and their solid-state analogues as materials for energy generation and storage, Nat. Rev. Mater., 2016, 1, 1–15, DOI:10.1038/NATREVMATS.2015.5
.
- A. Lewandowski and A. Świderska-Mocek, Ionic liquids as electrolytes for Li-ion batteries—An overview of electrochemical studies, J. Power Sources, 2009, 194, 601–609, DOI:10.1016/J.JPOWSOUR.2009.06.089
.
- A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Novel solvent properties of choline chloride/urea mixtures, Chem. Commun., 2003, 70–71, 10.1039/B210714G
.
- W. Xiong, X. Zhang, Z. Tu, X. Hu and Y. Wu, Novel Deep Eutectic Electrolyte Induced by Na⋯N Interactions for Sodium Batteries, Ind. Eng. Chem. Res., 2023, 62, 51–61, DOI:10.1021/ACS.IECR.2C03313
.
- Q. Zhang, K. De Oliveira Vigier, S. Royer and F. Jérôme, Deep eutectic solvents: syntheses, properties and applications, Chem. Soc. Rev., 2012, 41, 7108–7146, 10.1039/C2CS35178A
.
-
J. O. Bockris and A. K. N. Reddy, Modern Electrochemistry 2B: Electrodics in Chemistry, Engineering, Biology and Environmental Science, Springer Science & Business Media, 1998 Search PubMed
.
- E. A. Arkhipova, A. S. Ivanov, K. I. Maslakov and S. V. Savilov, Nitrogen-doped mesoporous graphene nanoflakes for high performance ionic liquid supercapacitors, Electrochim. Acta, 2020, 353, 136463, DOI:10.1016/J.ELECTACTA.2020.136463
.
- R. Venâncio, D. A. C. da Silva, L. O. Borges, I. A. Silva, L. M. Da Silva, G. Doubek, L. J. A. Siqueira and H. Zanin, High performance of supercapacitors containing an ionic liquid electrolyte by means of electrochemical and computational studies, Electrochim. Acta, 2025, 146569, DOI:10.1016/J.ELECTACTA.2025.146569
.
- M. Z. Bazant, B. D. Storey and A. A. Kornyshev, Double layer in ionic liquids: Overscreening versus crowding, Phys. Rev. Lett., 2011, 106, 046102, DOI:10.1103/PhysRevLett.106.046102
.
- M. Z. Bazant, M. S. Kilic, B. D. Storey and A. Ajdari, Towards an understanding of induced-charge electrokinetics at large applied voltages in concentrated solutions, Adv. Colloid Interface Sci., 2009, 152, 48–88, DOI:10.1016/j.cis.2009.10.001
.
- M. McEldrew, Z. A. H. Goodwin, S. Bi, M. Z. Bazant and A. A. Kornyshev, Theory of ion aggregation and gelation in super-concentrated electrolytes, J. Chem. Phys., 2020, 152, 234506, DOI:10.1063/5.0006197
.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Deep Eutectic Solvents (DESs) and Their Applications, Chem. Rev., 2014, 114, 11060–11082, DOI:10.1021/cr300162p
.
- X. Chen, X. Zhang, H. Li and Q. Zhang, Cation−Solvent, Cation−Anion, and Solvent−Solvent Interactions with Electrolyte Solvation in Lithium Batteries, Batteries Supercaps, 2019, 2, 128–131, DOI:10.1002/batt.201800118
.
- M. Shakourian-Fard, G. Kamath, K. Smith, H. Xiong and S. K. R. S. Sankaranarayanan, Trends in Na-Ion Solvation with Alkyl-Carbonate Electrolytes for Sodium-Ion Batteries: Insights from First-Principles Calculations, J. Phys. Chem. C, 2015, 119, 22747–22759, DOI:10.1021/ACS.JPCC.5B04706
.
- R. Sharma, A. Sharma and R. C. Thakur, Effect of Deep Eutectic Solvent (DES) on physicochemical and electrochemical properties of triethylmethylammonium tetrafluoroborate in propylene carbonate and aqueous propylene carbonate solvent systems for energy storage applications: A comparative analysis, J. Mol. Liq., 2024, 416, 126494, DOI:10.1016/J.MOLLIQ.2024.126494
.
- X. Wang, Y. Wang, M. Dong, Z. Fang, Y. Hu, K. Xue, Z. Ye and X. Peng, A two-dimensional nanochannel facilitates ionic conductivity of a deep eutectic solvent for an efficient supercapacitor, Mater. Today Energy, 2023, 34, 101285, DOI:10.1016/J.MTENER.2023.101285
.
- D. A. C. Da Silva, M. J. Pinzón C., A. Messias, E. E. Fileti, A. Pascon, D. V. Franco, L. M. Da Silva and H. G. Zanin, Effect of conductivity, viscosity, and density of water-in-salt electrolytes on the electrochemical behavior of supercapacitors: molecular dynamics simulations and in situ characterization studies, Mater. Adv., 2022, 3, 611–623, 10.1039/D1MA00890K
.
- Q. Zhang, C. Cai, J. Qin and B. Wei, Tunable self-discharge process of carbon nanotube based supercapacitors, Nano Energy, 2014, 4, 14–22, DOI:10.1016/J.NANOEN.2013.12.005
.
- K. Liu, C. Yu, W. Guo, L. Ni, J. Yu, Y. Xie, Z. Wang, Y. Ren and J. Qiu, Recent research advances of self-discharge in supercapacitors: Mechanisms and suppressing strategies, J. Energy Chem., 2021, 58, 94–109, DOI:10.1016/J.JECHEM.2020.09.041
.
- P. Kulkarni, D. Ghosh and R. G. Balakrishna, Recent progress in ‘water-in-salt’ and ‘water-in-salt’-hybrid-electrolyte-based high voltage rechargeable batteries, Sustainable Energy Fuels, 2021, 5, 1619–1654, 10.1039/D0SE01313G
.
- J. Meng, G. Jia, H. Yang and M. Wang, Recent advances for SEI of hard carbon anode in sodium-ion batteries: A mini review, Front. Chem., 2022, 10, 986541, DOI:10.3389/FCHEM.2022.986541
.
- G. Huang, J. Hu, Z. Wang, F. Gao, Y. Zhou, S. Fu, Y. Wang, Z. Shang, C. Gao, L. Luo, Y. Chen and Y. Wang, Synergistic Dual-Engineering of Hard Carbons via Graphene Oxide
Nanosheets: Enabling Ultrahigh ICE Sodium-Ion Batteries through Edge Passivation and Catalytic SEI Design, Adv. Funct. Mater., 2025, 36, e13551, DOI:10.1002/ADFM.202513551
.
- Y. Liao, H. Zhang, Y. Peng, Y. Hu, J. Liang, Z. Gong, Y. Wei and Y. Yang, Electrolyte Degradation During Aging Process of Lithium-Ion Batteries: Mechanisms, Characterization, and Quantitative Analysis, Adv. Energy Mater., 2024, 14, 2304295, DOI:10.1002/AENM.202304295
.
- M. Hahn, A. Würsig, R. Gallay, P. Novák and R. Kötz, Gas evolution in activated carbon/propylene carbonate based double-layer capacitors, Electrochem. Commun., 2005, 7, 925–930, DOI:10.1016/j.elecom.2005.06.015
.
- M. He, K. Fic, E. Frackowiak, P. Novák and E. J. Berg, Ageing phenomena in high-voltage aqueous supercapacitors investigated by in situ gas analysis, Energy Environ. Sci., 2016, 9, 623–633, 10.1039/c5ee02875b
.
- G. Gachot, S. Grugeon, M. Armand, S. Pilard, P. Guenot, J. M. Tarascon and S. Laruelle, Deciphering the multi-step degradation mechanisms of carbonate-based electrolyte in Li batteries, J. Power Sources, 2008, 178, 409–421, DOI:10.1016/J.JPOWSOUR.2007.11.110
.
- S. Laruelle, S. Pilard, P. Guenot, S. Grugeon and J.-M. Tarascon, Identification of Li-Based Electrolyte Degradation Products Through DEI and ESI High-Resolution Mass Spectrometry, J. Electrochem. Soc., 2004, 151(8), A1202, DOI:10.1149/1.1760992
.
- N. Ortiz-Vitoriano, I. R. de Larramendi, G. Åvall, R. Cid, M. Enterría, P. Johansson and R. Bouchal, Unlocking the role of electrolyte concentration for Na-O2 batteries, Energy Storage Mater., 2024, 70, 103501, DOI:10.1016/J.ENSM.2024.103501
.
- M. Rezaei and A. Groß, Degradation of a water-in-salt electrolyte at graphite and Na metal electrodes from first principles, J. Power Sources, 2025, 640, 236583, DOI:10.1016/J.JPOWSOUR.2025.236583
.
- X. Sun, Y. Li, Y. Wang, Z. Liu, K. Dong and S. Zhang, Effect of Interlayer Spaces and Interfacial Structures on High-Performance MXene/Ionic Liquid Supercapacitors: A Molecular Dynamics Simulation, Langmuir, 2024, 40, 2220–2229, DOI:10.1021/ACS.LANGMUIR.3C03277
.
- J. Wang, G. Chen and S. Song, Na-ion conducting gel polymer membrane for flexible supercapacitor application, Electrochim. Acta, 2020, 330, 135322, DOI:10.1016/J.ELECTACTA.2019.135322
.
- D. Martin-Vosshage and B. V. R. Chowdari, X-Ray Photoelectron Spectroscopy Studies on Poly-(ethylene oxide) with Sodium Triflate, J. Electrochem. Soc., 1993, 140, 3531–3536, DOI:10.1149/1.2221122/XML
.
- X. Yu, Y. Feng, J. Tian, X. Liu, B. Wang, Y. Zhang, T. Zhang, G. Li, X. Li, H. Jin, W. Zhou, W. Li, Z. Zeng, L. Li, D. Zhao and D. Chao, Unveil the Failure of Alkali Ion-Sulfur Aqueous Batteries: Resolving Water Migration by Coordination Regulation, Angew. Chem., Int. Ed., 2025, 64, e202503138, DOI:10.1002/ANIE.202503138
.
| This journal is © The Royal Society of Chemistry 2026 |





