Open Access Article
Open Access ArticleFeO-enabled low-temperature CO2-splitting for chemical looping carbon utilization
Huang-Chin Lin†
,
Yi Chen†,
Tzu-Hao Hung,
Ding-Huei Tsai,
Lu-Yu Chueh,
Chun-I Chou,
Kun-Han Lin * and
Yung-Tin (Frank) Pan
* and
Yung-Tin (Frank) Pan *
*
Department of Chemical Engineering, National Tsing Hua University, 101 Sec. 2, Kuang-Fu Rd, Hsinchu City, 300044, Taiwan. E-mail: ytpan@mx.nthu.edu.tw; kunhan.lin@mx.nthu.edu.tw
First published on 6th February 2026
Abstract
Chemical looping integrated with CO2 utilization is an attractive chemical process that can contribute greatly to on-site carbon capture and utilization. To reduce the excess energy consumption, the kinetics of the CO2-splitting half reaction has been revisited on pure iron oxygen carriers. High CO2-splitting rate at a very low temperature of 350 °C is discovered with an unambiguous relationship to the presence of FeO. Through controlled reduction, a nanoporous oxygen carrier enriched with active FeO, capable of 2-dimensional oxide growth with fast kinetics is formed and can be regenerated under cycling conditions of a chemical looping reverse water–gas shift model reaction at 500 °C. Over-reduction leads to the formation of low activity metallic Fe that requires additional 100 °C or more to conduct CO2-splitting, which deactivates rapidly due to severe sintering. Density functional theory calculations reveal the minimum energy pathway involving the dissociation of adsorbed CO2 on the Fe surface, followed by the spillover of CO to the FeO surface for desorption. These findings provide guidance for the design of a reactive iron-based oxygen carrier for low temperature CO2-splitting in all chemical looping carbon capture utilization processes.
Introduction
Decarbonization of the power, transportation, and industrial sectors is vital for the success of a net-zero emission scenario before 2050 to withhold the rise of global temperature within the 2 °C limit. On-site carbon capture and utilization (CCU) would be a preferable way to mitigate carbon emissions from centralized large-scale sources such as fossil fuel power plants. This is because the energy penalty for CCU can potentially be compensated with proper heat integration.1,2 The optimum case would be the use of low-grade waste heat to drive such CCU processes.Hydrogenation of CO2 to value-added chemicals is a plausible approach for CCU in the presence of affordable hydrogen (H2). CO2 can be hydrogenated to form methane via the nickel-catalyzed methanation reaction at relatively low pressure and temperature.3–6 It can also be converted to CO via the reverse water–gas shift (RWGS) reaction at low pressure and higher temperatures in the presence of copper or iron catalysts.7,8 With additional expense for compression, CO2 can also be hydrogenated into methanol using copper-based catalysts at much higher pressures.9,10 For these catalytic conversions of CO2 to be economically feasible, high yield and product selectivity is required to minimize down-stream separation cost. In this aspect, the chemical looping method provides a better opportunity to regulate product selectivity with very high precision.11–17 Since the redox chemistry is now mediated by the oxygen carrier, co-existence of CO2 and H2 in the reactor is not required. The reaction proceeds in two-steps where the reduced-form oxygen carrier splits CO2 into CO and the oxidized-form oxygen carrier oxidizes H2 into H2O, both specifically.18–22 Since the two-steps are temporally and spatially separated, down-stream separation can be omitted. Furthermore, the CO2-splitting half-reaction, in principle, can be coupled with any desirable oxidation half reactions such as the partial oxidation of alkanes.23
The key component of a successful chemical looping process is an active oxygen carrier with good structural reversibility. For the CO2 splitting half reaction, various transition metal oxides,13,19,24–26 spinel oxides,27,28 perovskite oxides,23,29,30 and others31,32 have been investigated. Within the wide variety of elements and crystal structures, iron serves as the critical element that conducts redox chemistry with CO2. For example, earlier studies by Galvita revealed the reversible phase change of iron-based oxygen carriers between Fe3O4/FeO/Fe during H2 temperature programmed reduction (H2-TPR) and CO2 temperature programmed oxidation (CO2-TPO) by in situ XRD.13,21 Based on the TPR/TPO results, constant temperature looping was conducted at 600 °C. A recent study by Xiao demonstrated high CO2-splitting activity using Cu/Mn/Co-doped spinel ferrites at 650 °C.24,28 Li reported the use of Cu-doped LaFeO3 perovskite to conduct CO2 splitting in the presence of O2 at 850 °C.33 Zhao reported the use of Ce-modified SrFeO3-δ to conducted CO2 splitting coupled with ethane oxidative dehydrogenation at 725 °C.29 Obviously, it would be more desirable to conduct the looping process at lower temperatures, but attempts at temperatures below 600 °C remain scarce.
To achieve this goal, we have revisited the redox chemistry and phase behavior of pure iron oxides, seeking opportunities to decipher the key descriptor that enables low-temperature chemical looping CCU. We proved that fast kinetics of CO2 splitting at low temperature is possible and revealed its unambiguous strong correlation with the presence of the meta-stable wüstite FeO. Using the chemical looping reverse water–gas shift (CL-RWGS) as a model reaction, we have identified the optimum matching of CO2 splitting and H2-oxidation half reaction conditions. By applying the general kinetic model for grain boundary nucleation and growth, distinctive 2-D growth and fast oxidation kinetics were discovered for the oxygen carriers treated with short H2 durations. CO2-TPO revealed a highly reactive species, having a peak temperature below 350 °C, which is accountable for the fast CO2-splitting kinetics. Detailed crystallography analysis pointed out the likelihood of FeO being accountable for the high reactivity, for which its generation and regeneration was more effective with shorter H2 steps. With extended H2 step duration, FeO was no longer preserved, and the fast CO2-splitting kinetic feature of the reduced oxygen carrier was lost.
Experimental
Materials and chemicals
Iron(III) nitrate nonahydrate (Fe(NO3)3·9H2O, 98%, Alfa-Aesar); ammonium hydroxide (NH4OH(aq), 30–33%, Honeywell); deionized water (DI water, from Merck Millipore Milli-Q, 18.2 MW resistance); hydrogen gas (H2, 5%, 4N-grade, Sinda Gas Corp.); carbon dioxide (CO2, 10% balanced with argon, 4N-grade, Sinda Gas Corp); argon (Ar, 4N-grade, Sinda Gas Corp.)Preparation of iron oxide oxygen carriers
The pristine iron oxide oxygen carriers in the present study were prepared by a direct precipitation method, as reported in previous studies.18 In a typical process, 0.015 mol of Fe(NO3)3·9H2O was first dissolved in 50 mL of DI water. Under vigorous stirring conditions, the solution was titrated with 1 M ammonium hydroxide to pH 9 and maintained at this pH for another 30 min. The precipitated products were then separated by centrifugation and washed with DI water through repetitive dispersion/centrifugation cycles until the pH of the supernatant approached 7. The samples were then dried in an 80 °C vacuum oven overnight. The dried samples were ground using a mortar and pestle to fine powders and subjected to a 3 h thermal treatment in air at 500 °C to yield the final powder samples.Characterization
Powder X-ray diffraction (PXRD, D8A25, Bruker) was conducted to characterize the crystal phase of the oxygen carriers during different stages of their lifetime. Refinement of XRD patterns was conducted using GSAS. Field emission scanning electron microscopy (FESEM, SU8010, Hitachi) was carried out to image the morphological features of the oxygen carriers at 20 kV. Scanning transmission electron microscopy (STEM, JEM-F200, JEOL) was conducted at an accelerating voltage of 200 kV from the cold field emission gun to image the nanoscale features of the oxygen carriers. A coupled energy dispersive X-ray spectrometer was used to conduct spatial and quantitative elemental analysis.Chemical looping testing, temperature programmed oxidation in CO2, and product analysis
The chemical looping reverse water–gas shift reaction and CO2-TPO were both conducted using a home-built reaction and analysis system. The reaction system includes three mass flow controllers (SLA5850E, Brooks), a 4-inch diameter U-shape quartz tube, and a tube furnace (custom-built, GL-Glass). A packed bed reactor was configured by placing 100 mg of powder oxygen carrier samples inside the U-shape quartz tube and immobilized between two pieces of quartz wool. During a typical chemical looping process, once the packed bed reactor is placed inside the tube furnace, Ar was then purged into the reactor at 300 sccm for 10 min to remove residual air. The reactor was then ramped up to certain temperature with a ramp rate of 10 °C min−1. The chemical looping process was then conducted isothermally by introducing 5%-H2 and 10%-CO2 alternatively with a 3-min Ar purge step in between, all at a flow rate of 300 sccm. The gaseous products exiting the reactor were analyzed using a CO2 non-dispersive infrared spectrometer (NDIR, CI-IR 20, Chang-AI), a CO-NDIR (CI-IR 20, Chang-Ai), and a thermal conductivity detector (TCD, CU-TC 90, Chang-AI) to quantify CO2, CO, and H2 with ms resolution. The CO2-TPO was conducted with a process similar to the chemical looping reverse water–gas shift reaction except that the CO2 steps in certain cycles were carried out with a temperature ramp from 150 °C to 500 °C instead of isothermally at 500 °C.Computational details
All periodic DFT computations were carried out using the Vienna Ab initio Simulation Package (VASP),34–37 while electronic structure analyses were conducted with the post-processing VASPKIT package.38 The Perdew–Burke–Ernzerhof (PBE) functional within the framework of generalized gradient approximation (GGA)39,40 was employed to model the exchange–correlation interactions in pure Fe systems. For the remaining systems, additional corrections—including Hubbard U, spin–orbit coupling (SOC), and D3(BJ) dispersion—were incorporated, as described in detail in the Results and discussion section and SI. Projector augmented wave (PAW) potentials were used to account for electron–ion interactions.41,42 A plane-wave energy cutoff of 520 eV was consistently applied across all calculations. To treat Fermi surface broadening, the Methfessel–Paxton smearing scheme43 was adopted, with a smearing width of 0.20 eV for pure Fe systems, while a Gaussian smearing width of 0.04 eV was applied for Fe surfaces with adsorbates and for FeO systems. Geometric relaxations were performed using the conjugate gradient method, and convergence was reached when all atomic forces dropped below 0.02 eV Å−1. Monkhorst–Pack k-point mesh was adjusted based on the specific slab dimensions, as detailed in the Results and discussion section and SI. A vacuum region of 15 Å was introduced perpendicular to the slab surface in all models to eliminate spurious interactions between periodic images. All reported energy values were extrapolated to 0 K. Spin polarization was explicitly considered for Fe atoms throughout the surface calculations. In addition, Bader charge analysis was performed only for FeO systems, as detailed in the Results and discussion section and SI.44–48 Most of the structural visualizations were generated using Crystal Toolkit developed by A. Jain et al.49Results and discussion
Regenerative CO2-splitting of iron oxygen carriers through optimized looping programs
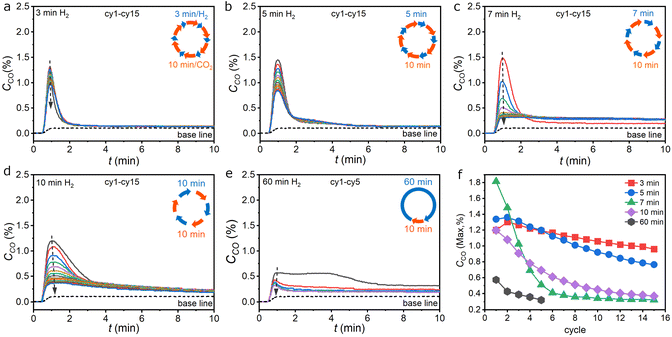
The performance of CL-RWGS using Fe-oxygen carriers is strongly associated with the reversibility of its redox chemistry, which is heavily dependent on the reaction conditions and history. Preliminary studies on the CO2-splitting activity and cycle stability suggested that under a fixed H2/CO2 program of 5 min/10 min, a process temperature of 500 °C is optimal for obtaining the highest instantaneous CO concentration during the CO2-splitting step due to the balanced reduction and oxidation kinetics of the oxidized and reduced Fe-oxygen carriers respectively (Fig. S1). The rather low 500 °C operation is beneficial from the viewpoint of utilizing industrial waste heat to conduct the chemical looping CO2 utilization process. However, to optimize performance and cycle stability, the H2/CO2 program was further studied systematically by varying duration of the H2 step while fixing the CO2 step at 10 min. Using homemade Fe2O3 as the starting material, the program with a 1-min H2 step showed good cycle stability but the amount of CO produced in the CO2-splitting step was too little due to the limited amount of reduced-form Fe species formed during the short exposure to H2. When increasing the H2-step duration to 3 min, the CO2-splitting reaction was completed within two minutes as the CO concentration measured by the non-dispersive infrared (NDIR) detector approached the baseline level (Fig. 1a, S2 and S3). The maximum instantaneous CO concentration can reach up to 1.3%, equivalent to 13% of CO2 conversion and gradually decreased to 0.9% after fifteen cycles. When the H2-step duration was increased to 5 min, a similar behavior of the CO production can be observed but the maximum CO concentration was increased to 1.4% and approached the baseline level after 5 min with a transition stage in between (Fig. 1b). The most apparent difference lies in the width of the CO peak, which increased from 43 s to 68 s and ended up having higher CO2-splitting capacity in each cycle. The CO2-splitting behavior was significantly altered when the H2 step was increased to 7 min. Although the maximum CO concentration can reach up to 1.5% in early stages, a rapid loss of the CO peak concentration in successive cycles occurred and was accompanied by the appearance of slow-kinetic CO production in the range of 0.2–0.3% throughout the later stages (Fig. 1c). A similar behavior was observed when the H2 step duration was extended to 10 min. However, the CO peak width was further increased to 90 s and the slow kinetic CO production started to take place even in the first cycle (Fig. 1d). When the H2 step was further extended to 60 min, the CO2-splitting behavior became drastically different. Except for the first cycle, both the maximum CO concentration and the slow kinetic CO production were significantly smaller than previous cases (Fig. 1e). The dynamic variations of CO production using different programs are summarized in Fig. 1f by plotting the maximum CO concentration against the number of cycles. The CO peak concentration was much better maintained over successive cycles with programs of short H2 steps and can be maintained over 0.48% after 50 cycles for the 5 min/10 min program (Fig. S4). As the H2 step duration exceeded 5 min, rapid loss of the CO peak concentration, i.e., CO2-splitting kinetics, occurred.Oxidation kinetics of iron oxygen carriers in CO2
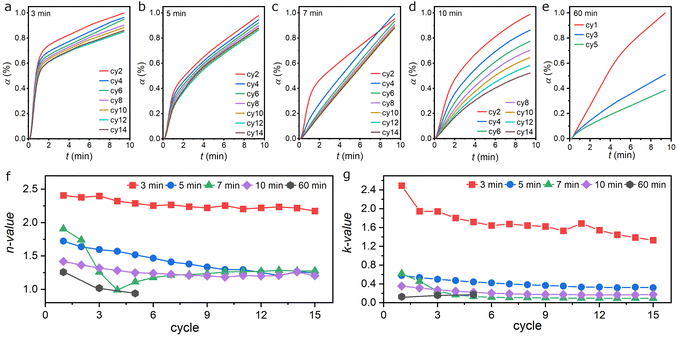
Since the rate of CO2-splitting should be equivalent to the rate of Fe (or FeO) oxidation, kinetic models developed for reactions of Fe with gaseous species can be implemented here to aid the understanding of experimental observations. According to Hancock and Sharp's method,50,51 isothermal solid-state reactions can be compared based on the general equation for nucleation and growth on phase boundaries:| α = 1 − e−ktn | (1) |
ln(−ln(1 − α)) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k + n k + n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) t t
| (2) |
Thermo-chemical properties and their relation to the crystal phase studied via CO2-temperature programmed oxidation and powder X-ray diffraction
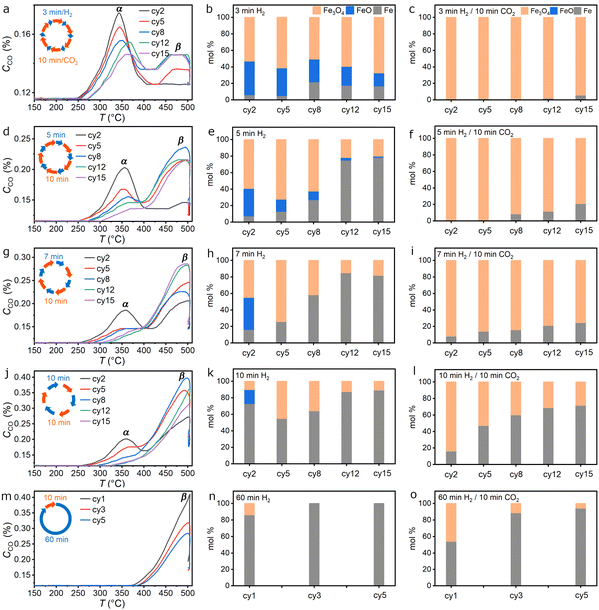
The observed variations in CO2-splitting kinetics with different redox programs are likely due to the dynamic phase composition of the Fe oxygen carriers in different reaction environments. To gain further understanding, CO2-TPO and post-mortem PXRD were conducted on the Fe oxygen carriers after certain cycles of the CL-RWGS process (please refer to Fig. S7, S8 and SI Note 1 for the raw data and smoothing protocol). In general, the CO2-TPO curves are composed of a low temperature α peak around 350 °C and a high temperature β peak around 475 °C, showing a high correspondence to the “fast-kinetic” and “slow-kinetic” species identified previously. The relative ratio between the α and β peaks decreased in the programs with longer H2-steps. This α/β ratio also decreased in successive cycles in individual programs. Corresponding PXRD patterns confirm that CL-RWGS was associated with the redox processes between metallic Fe, wustite FeO, and spinel Fe3O4 (Fig. S9). For the 3 min/10 min program, the β peak was absent at initial stages and appeared after the 5th cycle, accompanied by the increased peak temperature and decreased intensity of the α peak (Fig. 3a). From PXRD refinement results, the reduced-form oxygen carrier in the 3 min/10 min program was originally dominated by FeO, reaching 40.8% after the 2nd cycle and gradually decreased to 15.7% after the 15th cycle (Fig. 3b). The Fe content increased complementary from 6.1% after the 2nd cycle to 21.6% after the 8th cycle and decreased to 16.8% after the 15th cycle. Although decreasing in its fraction, the regenerative formation of FeO was observed even though being reported thermodynamically unstable at 500 °C.54,55 After the CO2 steps, the Fe oxygen carrier was composed of Fe3O4 except with a small fraction of unreacted Fe after the 15th cycle (Fig. 3c). To figure out the oxidation process associated with the α-peak, the CO2-TPO was cut-off before the β-peak appeared. PXRD patterns of the corresponding oxygen carrier at different cycles showed only peaks for Fe3O4 and Fe, confirming that oxidation of FeO is an important contributor to the fast CO2-to-CO kinetics (Fig. S10, Eq. (3)).| 3FeO + CO2 → Fe3O4 + CO | (3) |
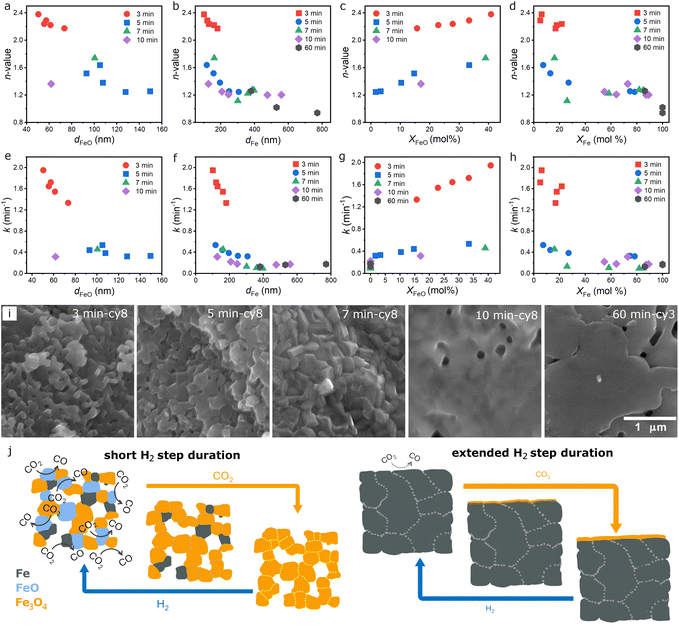
The statement is strengthened from the systematic study of the oxygen carrier experiencing longer H2 steps, where the ratio of α/β peaks in the CO2-TPO correlated positively with the FeO/Fe ratio consistently. For the 5 min/10 min program, the α peak and FeO content in the reduced oxygen carrier gradually decreased to zero as Fe became the dominant phase after the 8th cycle (Fig. 3d and e). However, not all Fe was converted back to Fe3O4, explaining the loss in CO2-splitting capability due to the formation of inactive Fe species over time. In the 7 min/10 min program, Fe had become the dominant phase with no FeO just after the 5th H2-step (Fig. 3g and h). It was unique in that most of the formed Fe here, although showing slow kinetics, remained reactive throughout the 15 cycles (Fig. 3i). The formation of inactive Fe became severe when the H2-step duration passed the 10-min threshold (Fig. 3j–l). Further extension of the H2-step to 60 min eliminated the formation of FeO and led to the formation of nearly inactive Fe after the 3rd cycle (Fig. 3m–o). In addition to the fractional distribution of Fe and FeO, the grain size of each of the phases can also be obtained from the PXRD patterns. The mole fraction and grain size of Fe and FeO, i.e., reduced-form oxygen carriers, are summarized with respect to the number of cycles (Fig. S11, S12 and Table S1, SI Note 2).
Theoretical investigation of CO2-splitting
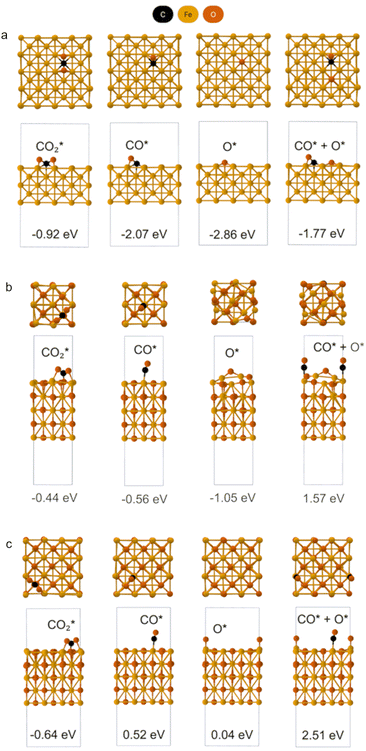
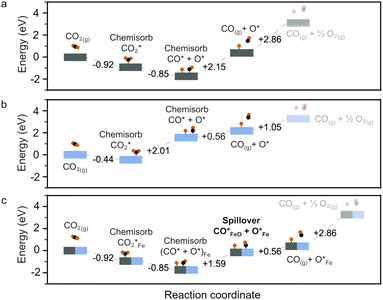
To rationalize the observed irreversibility and the coexistence of fast and slow kinetics in our experiments, we first aimed to establish a reliable theoretical description of the electronic structure of FeO as a prerequisite for surface adsorption modeling. Because FeO is a prototypical Mott insulator, conventional generalized-gradient approximation (GGA) functionals significantly underestimate its band gap and fail to reproduce its insulating nature.We therefore benchmarked the PBE + U approach, with and without spin–orbit coupling (SOC), by systematically varying the Hubbard U parameter and comparing the resulting lattice parameters and band gap against experimental values. Based on this benchmark, we selected a PBE + U (U = 3.7 eV) with SOC setup for the remainder of the calculations. Full computational details and the benchmarking results are provided in SI Note 3 and Note 4 (Tables S2–S4 and Fig. S13, S14). We first examined plausible adsorption configurations of CO2, CO, and O (denoted as 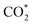 , CO*, and O*) on the Fe(100) and FeO(100) surfaces. Fig. 4 summarizes the most stable adsorption geometries identified for each adsorbate across the investigated surfaces. Full details—including all configurations screened, the definition and evaluation of adsorption energies (Eads), and the dissociative chemisorption energy (ΔEchem)—are provided in SI Note 5. Monkhorst–Pack k-point mesh was adjusted based on the specific slab dimensions, as detailed in Tables S4 and S5. This accurate reproduction of the electronic band structure serves as a proof of method, ensuring the reliability of subsequent adsorption calculations. Fig. 4 and 5 presents the reaction energy diagram for CO2 splitting on Fe(100) and FeO(100) surfaces. CO2 chemisorbs exothermically on both surfaces, with slightly stronger binding on Fe(100) (∼0.48 eV more negative) than on FeO(100).
, CO*, and O*) on the Fe(100) and FeO(100) surfaces. Fig. 4 summarizes the most stable adsorption geometries identified for each adsorbate across the investigated surfaces. Full details—including all configurations screened, the definition and evaluation of adsorption energies (Eads), and the dissociative chemisorption energy (ΔEchem)—are provided in SI Note 5. Monkhorst–Pack k-point mesh was adjusted based on the specific slab dimensions, as detailed in Tables S4 and S5. This accurate reproduction of the electronic band structure serves as a proof of method, ensuring the reliability of subsequent adsorption calculations. Fig. 4 and 5 presents the reaction energy diagram for CO2 splitting on Fe(100) and FeO(100) surfaces. CO2 chemisorbs exothermically on both surfaces, with slightly stronger binding on Fe(100) (∼0.48 eV more negative) than on FeO(100).
Upon adsorption, the chemisorbed CO2 (*CO2) can undergo dissociation into *CO and O*. However, the energetics differ significantly: CO2 splitting on Fe(100) is strongly exothermic, consistent with previous DFT studies,56 while on FeO(100) it is highly endothermic. These contrasting behaviors indicate that CO2 dissociation is more favorable on Fe(100) than on FeO(100).
The adsorption energy profiles and associated configurations are provided in Tables S6, S7 and Fig. S15–S20, while computational details are described in SI Note 4–6. The full CO2 splitting half-cycle includes *CO desorption. The strong exothermicity of *CO2 dissociation on Fe(100) results in a strongly bound *CO, requiring 2.15 eV to desorb (Fig. 5a). An alternative pathway involves *CO spillover from Fe(100) to FeO(100), which is uphill by 1.59 eV—still significantly lower than the direct desorption energy from Fe(100) (Fig. 5a and c). From the FeO(100) surface, *CO desorption requires only 0.56 eV, making this spillover route a plausible and kinetically accessible CO release pathway (Fig. 5b). We assign this as the “fast” process. In contrast, the “slow” process corresponds to direct *CO desorption from Fe(100), which is hindered by its strong binding. Nevertheless, this desorption becomes progressively easier as the surface *CO coverage increases (SI Note 7 and Fig. S21, S22), consistent with previous theoretical findings.57 Finally, the remaining O* can either remain on Fe(100) or migrate to FeO(100). As the surface O* coverage increases, oxidation of Fe and FeO occurs, leading to the formation of Fe3O4 through phase transformation (detailed in SI Note 8 and Fig. S23). Experimentally, when the H2 pulse duration is increased, the FeO content decreases. This suggests that the contribution of the fast CO release pathway is reduced, forcing the system to rely on the slower desorption route, where *CO must accumulate on Fe before desorption becomes thermodynamically favorable. The observed suppression of CO release is attributed to the strong *CO binding on Fe(100), which not only impedes desorption but also passivates the surface, inhibiting further CO2 splitting and thus preventing Fe oxidation to Fe3O4, explaining the experimental results.
Note that our DFT calculations employ ideal, defect-free Fe(100) and FeO(100) slab models, neglect vibrational free-energy contributions, and do not explicitly locate transition states that determine kinetic barriers. We also did not consider surface defects or strain,58,59 both of which could modulate CO2 dissociation barriers and CO adsorption and thereby affect CO spillover at the Fe/FeO interface. A more rigorous description would therefore require incorporating these effects explicitly. Despite these simplifications, the computed adsorption energetics reproduce the qualitative experimental behavior, suggesting that the dominant features governing the chemical-looping process are captured. A systematic assessment of defect and strain effects, and the potential for performance optimization via defect engineering, will be an important direction for future work.
Further discussion on the structural properties of the reduced oxygen carrier and their relations to CO2-splitting performance
The roles of the mole fraction and grain size of Fe and FeO are further discussed by plotting the n-values and k-values with respect to these structural properties. In general, the n-value decreases as the grain size of Fe (dFe) and FeO (dFeO) increases (Fig. 6a and b). It also showed a positive correlation with the abundance of FeO (XFeO) but a negative correlation with the Fe content (XFe) (Fig. 6c and d). The small grains of Fe (dFe) and FeO (dFeO) and the abundance of FeO (XFeO) with adequate amounts of Fe (XFE) in the 3 min/3 min program made it stand out among others, giving rise to its 2D oxide growth during the CO2-splitting step. Similar results were observed for the k-value; it decreases as dFe and dFeO increase (Fig. 6e and f). It showed a positive correlation with XFeO but a negative correlation with XFe (Fig. 6g and h). Our results suggested that large amounts of small size FeO and Fe grains in the oxygen carrier created optimum CO2 dissociation pathways and were responsible for the additional growth dimension and fast kinetics in the short-H2 CL programs.60 Microstructure characterization from scanning electron microscopy (SEM) provided additional insights into the varying behaviors of the oxygen carrier in different programs (Fig. 6i and S24). A significant transition in morphology can be observed as the H2-step duration increased. From the micrograph of the oxygen carrier taken after the 8th H2-step in the 3 min/10 min program, it reveals that the material was not only formed of small-size grains but also exhibited a porous structure. High resolution scanning transmission electron microscopy (STEM) imaging from the same but crushed sample suggested that particles observed in the SEM were composed of a uniform mixture of small grain-size Fe, FeO, and Fe3O4, since the coupled EDS spot analysis of 13 particles was very similar (Fig. S25).Furthermore, a fine nanoporous structure was identified from the dark-field STEM image (Fig. S26). The nanoporosity and small particle size, with narrow size and composition distributions, provide a suitable condition for 2-D oxide growth with fast kinetics.16 When increasing the H2-step duration to 5 min, the oxygen carrier evolved into layered materials formed of larger plate-like particles with much decreased void space. STEM-EDS analysis also revealed the formation of large particles that were dominated by metallic Fe (Fig. S27). This resulted in a significant drop in the n-value and k-value as a significant fraction of the oxygen carriers became large metallic Fe particles, which was only capable of 1-D oxide growth with much slower kinetics. The 7 min/10 min program had evolved the oxygen carrier into densely packed particles of high crystallinity. The absence of void space resulted in 1-D oxide growth with slow kinetics. However, sintering into larger grains seemed to be impeded under this operating condition. Further increase in the H2 step duration to 10 min resulted in the formation of a continuous Fe over-layer covering the more faceted particles. Finally, only sintered large particles can be observed at a long H2-step of 60 min. Based on the detailed characterization of the oxygen carrier, we proposed that the high CO2-splitting kinetics and cycle stability was enabled by the high content of small size FeO and the nanoporous structure of the oxygen carrier created in the CL process with short H2 steps (Fig. 6j). Sintered large Fe particles formed in extended H2 steps can react with CO2 only through their surface layer with limited reactivity while the bulk becomes inaccessible to further oxidation. Although we have identified the critical role of FeO in low temperature CO2 splitting and operational conditions to maximize its regeneration, the issue of performance loss over time is not yet resolved. This is due to the irreversible growth of particle size and loss of nanoporous structure.Conclusions
The ability of pure iron oxygen carriers to conduct CO2-splitting in a chemical looping process is revisited using RWGS as a model reaction. Through thorough kinetic, thermochemical, and structural analysis, the relationship between the crystal/morphological structure of the oxygen carrier with its CO2-splitting performance was mapped out. With controlled reduction, the nanoporous oxygen carrier with high FeO content showed exceptional reactivity towards CO2-splitting at temperatures as low as 350 °C. Kinetic and structural analyses suggest that the nanoporous structure contributes to a 2-D oxide growth, while the high FeO content promotes the intrinsic growth rate. Extended reduction led to the formation of bulky pure metallic iron, which appears to be significantly kinetically impeded towards CO2-splitting under identical conditions. DFT calculation suggested the minimum energy pathway for CO2 dissociation and subsequent CO desorption under the coexistence of metallic Fe and wustite FeO. The findings in this work may provide alternative thinking in the design of the oxygen carriers and CL-CCU operations from a materials aspect focusing on kinetically controlled structural reversibility.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta07934a.Acknowledgements
This research was financially supported by the Industrial Technology Research Institute (ITRI) with grant no. N354DA7210. This research was also supported by the National Science and Technology Council (NSTC) of Taiwan with grant no. 112-2221-E-007-020-MY3 and 112-2628-E-007-011-MY3 on the advanced characterization via EM of the Instrumentation Center at National Tsing Hua University (NTHU). This research was also supported by the National Science and Technology Council (NSTC) of Taiwan with grant no. 114-2628-E-007-002 on computational studies. We thank the National Center for High-performance Computing (NCHC) for providing computational and storage resources.References
- G. G. Esquivel-Patiño, F. Nápoles-Rivera and A. Jiménez-Gutiérrez, Energy Convers. Manag., 2023, 295, 117619 CrossRef.
- Å. Eliasson, E. Fahrman, M. Biermann, F. Normann and S. Harvey, Int. J. Greenhouse Gas Control, 2022, 118, 103689 CrossRef.
- D. Bhalothia, W.-H. Hsiung, S.-S. Yang, C. Yan, P.-C. Chen, T.-H. Lin, S.-C. Wu, P.-C. Chen, K.-W. Wang, M.-W. Lin and T.-Y. Chen, ACS Appl. Energy Mater., 2021, 4, 14043–14058 CrossRef CAS.
- C. Yan, C.-H. Wang, M. Lin, D. Bhalothia, S.-S. Yang, G.-J. Fan, J.-L. Wang, T.-S. Chan, Y.-l. Wang, X. Tu, S. Dai, K.-W. Wang, J.-H. He and T.-Y. Chen, J. Mater. Chem. A, 2020, 8, 12744–12756 RSC.
- T. A. Le, M. S. Kim, S. H. Lee, T. W. Kim and E. D. Park, Catal. Today, 2017, 293–294, 89–96 CrossRef CAS.
- L. Shen, J. Xu, M. Zhu and Y.-F. Han, ACS Catal., 2020, 10, 14581–14591 CrossRef CAS.
- D.-H. Tsai, T.-T. Wu, H.-C. Lin, L.-Y. Chueh, K.-H. Lin, W.-Y. Yu and Y.-T. Pan, Chem.–Asian J., 2024, 19, e202300955 CrossRef CAS PubMed.
- H.-X. Liu, S.-Q. Li, W.-W. Wang, W.-Z. Yu, W.-J. Zhang, C. Ma and C.-J. Jia, Nat. Commun., 2022, 13, 867 CrossRef CAS PubMed.
- Y.-F. Shi, P.-L. Kang, C. Shang and Z.-P. Liu, J. Am. Chem. Soc., 2022, 144, 13401–13414 CrossRef CAS PubMed.
- M. Bowker, ChemCatChem, 2019, 11, 4238–4246 CrossRef CAS PubMed.
- S. Abanades and A. Le Gal, Fuel, 2012, 102, 180–186 CrossRef CAS.
- Y. Feng, N. Wang, X. Guo and S. Zhang, Fuel, 2020, 262, 116489 CrossRef CAS.
- V. V. Galvita, H. Poelman, V. Bliznuk, C. Detavernier and G. B. Marin, Ind. Eng. Chem. Res., 2013, 52, 8416–8426 CrossRef CAS.
- N. S. Yüzbasi, A. Armutlulu, T. Huthwelker, P. M. Abdala and C. R. Müller, J. Mater. Chem. A, 2022, 10, 10692–10700 RSC.
- L. Zeng, Z. Cheng, J. A. Fan, L.-S. Fan and J. Gong, Nat. Rev. Chem., 2018, 2, 349–364 CrossRef CAS.
- A. A. Sunny, Q. Meng, S. Kumar, R. Joshi and L.-S. Fan, Acc. Chem. Res., 2023, 56, 3404–3416 CrossRef CAS PubMed.
- X. Zhu, Q. Imtiaz, F. Donat, C. R. Müller and F. Li, Energy Environ. Sci., 2020, 13, 772–804 RSC.
- W.-Z. Hung, Z. X. Law, D.-H. Tsai, B.-H. Chen, C.-H. Chen, H.-Y. Hsu and Y.-T. Pan, MRS Energy Sustain, 2022, 9, 342–349 CrossRef.
- Y. Fan, B. Jin, Q. Guo and Z. Liang, ACS Sustain. Chem. Eng., 2023, 11, 5999–6010 CrossRef CAS.
- J. Rojas, E. Sun, G. Wan, J. Oh, R. Randall, V. Haribal, I.-h. Jung, R. Gupta and A. Majumdar, ACS Sustain. Chem. Eng., 2022, 10, 12252–12261 CrossRef CAS.
- V. V. Galvita, H. Poelman, C. Detavernier and G. B. Marin, Appl. Catal. B, 2015, 164, 184–191 CrossRef CAS.
- T. Ouyang, B. Jin, Y. Mao, D. Wei and Z. Liang, Appl. Catal. B, 2024, 343, 123531 CrossRef CAS.
- J. Zhang, V. Haribal and F. Li, Sci. Adv., 2017, 3, e1701184 CrossRef PubMed.
- Y. Qiu, L. Ma, M. Li, D. Cui, S. Zhang, D. Zeng and R. Xiao, Chem. Eng. J., 2020, 387, 124150 CrossRef CAS.
- L. Liu and M. R. Zachariah, Energy Fuels, 2013, 27, 4977–4983 CrossRef CAS.
- L.-l. Wang, L.-h. Shen, W. Liu and S. Jiang, Energy Fuels, 2017, 31, 8423–8433 CrossRef CAS.
- F. He, Z. Huang, G. Wei, K. Zhao, G. Wang, X. Kong, Y. Feng, H. Tan, S. Hou, Y. Lv, G. Jiang and Y. Guo, Energy Convers. Manag., 2019, 201, 112157 CrossRef CAS.
- L. Ma, Y. Qiu, M. Li, D. Cui, S. Zhang, D. Zeng and R. Xiao, Ind. Eng. Chem. Res., 2020, 59, 6924–6930 CrossRef CAS.
- X. Tian, C. Zheng and H. Zhao, Appl. Catal. B, 2022, 303, 120894 CrossRef CAS.
- D. Sastre, D. P. Serrano, P. Pizarro and J. M. Coronado, J. CO2 Util., 2019, 31, 16–26 CrossRef CAS.
- F. Li, Z. Sun, S. Luo and L.-S. Fan, Energy Environ. Sci., 2011, 4, 876–880 RSC.
- Y.-Y. Chen, M. Guo, M. Kim, Y. Liu, L. Qin, T.-L. Hsieh and L.-S. Fan, Chem. Eng. J., 2021, 406, 126729 CrossRef CAS.
- Y. e. Zheng, X. Liao, H. Xiao, V. Haribal, X. Shi, Z. Huang, L. Zhu, K. Li, F. Li, H. Wang and X. Chen, Nano Energy, 2020, 78, 105320 CrossRef CAS.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 558–561 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 49, 14251–14269 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- V. Wang, N. Xu, J.-C. Liu, G. Tang and W.-T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- J. P. Perdew and W. Yue, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 8800–8802 CrossRef PubMed.
- J. P. Perdew, J. A. Chevary, S. H. Vosko, K. A. Jackson, M. R. Pederson, D. J. Singh and C. Fiolhais, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6671–6687 CrossRef CAS PubMed.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- M. Methfessel and A. T. Paxton, Phys. Rev. B: Condens. Matter Mater. Phys., 1989, 40, 3616–3621 CrossRef CAS PubMed.
- R. F. W. Bader, Acc. Chem. Res., 1985, 18, 9–15 CrossRef CAS.
- G. Henkelman, A. Arnaldsson and H. Jónsson, Comput. Mater. Sci., 2006, 36, 354–360 CrossRef.
- E. Sanville, S. D. Kenny, R. Smith and G. Henkelman, J. Comput. Chem., 2007, 28, 899–908 CrossRef CAS PubMed.
- W. Tang, E. Sanville and G. Henkelman, J. Phys. Condens. Matter., 2009, 21, 084204 CrossRef CAS PubMed.
- M. Yu and D. R. Trinkle, J. Chem. Phys., 2011, 134, 064111 CrossRef PubMed.
- A. Jain, S. P. Ong, G. Hautier, W. Chen, W. D. Richards, S. Dacek, S. Cholia, D. Gunter, D. Skinner, G. Ceder and K. A. Persson, APL Mater., 2013, 1, 011002 CrossRef.
- M. Avrami, J. Chem. Phys., 1939, 7, 1103–1112 CrossRef CAS.
- J. D. Hancock and J. H. Sharp, J. Am. Ceram. Soc., 1972, 55, 74–77 CrossRef CAS.
- K. Piotrowski, K. Mondal, T. Wiltowski, P. Dydo and G. Rizeg, Chem. Eng. J., 2007, 131, 73–82 Search PubMed.
- Q. Tang and K. Huang, Chem. Eng. J., 2022, 434, 134771 CrossRef CAS.
- W. K. Jozwiak, E. Kaczmarek, T. P. Maniecki, W. Ignaczak and W. Maniukiewicz, Appl. Catal. A, 2007, 326, 17–27 CrossRef CAS.
- M. H. Jeong, D. H. Lee, G. Y. Han, C.-H. Shin, M. K. Shin, C. K. Ko and J. W. Bae, Fuel, 2017, 202, 547–555 CrossRef CAS.
- X. Liu, L. Sun and W.-Q. Deng, J. Phys. Chem. C, 2018, 122, 8306–8314 CrossRef CAS.
- T. Wang, X. Tian, Y.-W. Li, J. Wang, M. Beller and H. Jiao, J. Phys. Chem. C, 2014, 118, 1095–1101 CrossRef CAS.
- A. Chakrabarty, E. T. Bentria, S. A. Omotayo, O. Bouhali, N. Mousseau, C. S. Becquart and F. El Mellouhi, Appl. Surf. Sci., 2019, 491, 792–798 CrossRef CAS.
- D. C. Sorescu, D. L. Thompson, M. M. Hurley and C. F. Chabalowski, Phys. Rev. B: Condens. Matter Mater. Phys., 2002, 66, 035416 CrossRef.
- Q. Jiang, Y. Gao, V. P. Haribal, H. Qi, X. Liu, H. Hong, H. Jin and F. Li, J. Mater. Chem. A, 2020, 8, 13173–13182 RSC.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |