Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tin chloride metal salt modified carbon nitride for efficient hydrogen and hydrogen peroxide production from pure water via piezocatalysis
Ying
Pan
 *a,
Luocheng
Liao
b,
Dan
Qiao
a,
Irene Lamata
Bermejo
a,
Yunya
Liu
*a,
Luocheng
Liao
b,
Dan
Qiao
a,
Irene Lamata
Bermejo
a,
Yunya
Liu
 b,
Ran
Su
*c and
Nieves
López-Salas
b,
Ran
Su
*c and
Nieves
López-Salas
 *a
*a
aDepartment of Chemistry, University of Paderborn, Paderborn 33098, Germany. E-mail: ying.pan@uni-paderborn.de; nieves.lopez.salas@uni-paderborn.de
bKey Laboratory of Low Dimensional Materials and Application Technology of Ministry of Education, School of Materials Science and Engineering, Xiangtan University, Xiangtan 411105, China
cHebei Key Laboratory of Photoelectric Control on Surface and Interface, College of Science, Hebei University of Science and Technology, Shijiazhuang 050018, China. E-mail: suranxida@163.com
First published on 27th January 2026
Abstract
Piezocatalysis presents a sustainable and energy-efficient method for producing hydrogen (H2) and hydrogen peroxide (H2O2), utilizing mechanical energy to drive chemical reactions without the need for external power sources or harmful chemicals. In this study, we used a one-pot synthetic method to modify graphitic carbon nitride (g-C3N4). The obtained catalysts showed enhanced H2 and H2O2 production from pure water without any co-catalysts or sacrificial agents via piezocatalysis. In the cost-effective synthetic method, a metal oxide/g-C3N4 composite structure was constructed through the molten salt method. During thermal pyrolysis, non-noble metal-based SnCl2 provided a molten medium to facilitate the exfoliation of the g-C3N4 layer. The oxidation of SnCl2 and its interaction with g-C3N4 promoted the formation of g-C3N4/Sn-based composites. The synergistic interaction between exfoliated, defect-rich g-C3N4 and non-piezoelectrically active Sn species leads to a significant enhancement of the piezoelectric effect compared to pristine g-C3N4. Notably, the g-C3N4/Sn-based composites achieved superior H2 (3846.46 µmol g−1 h−1) and H2O2 (999.11 µmol g−1 h−1) production rates from pure water. This work provides new insights into the structural and compositional modulation of g-C3N4 without the use of any noble metals and paves the way for further piezocatalytic research using two-dimensional carbon nitride materials.
Introduction
Hydrogen (H2), with its high energy density and clean combustion that yields only water, represents a promising alternative for mitigating CO2 emissions. As a result, the production of H from water has become a key objective in the pursuit of low-carbon energy technologies.1 In parallel, hydrogen peroxide (H2O2) is a widely used green oxidant in synthesis, disinfection, and bleaching. It has also gained attention as a potential fuel cell energy carrier. Direct synthesis of H2O2 from oxygen and water has attracted significant attention for its simplicity, safety, and lower energy demands.2 Given the environmental and practical benefits of both H2 and H2O2, as well as the inherent sustainability of using water as a feedstock, there is growing interest in developing green and efficient methods for their production. In particular, strategies that enable the generation of H2 or H2O2 from water under mild conditions, without the use of noble metals or hazardous reagents, are highly desirable for advancing sustainable energy and chemical manufacturing.The piezocatalytic properties of dielectric materials have recently attracted attention as a novel approach to energy conversion.3,4 When subjected to mechanical vibration, these materials generate an internal electric field due to their non-centrosymmetric crystal structures, which drives the separation of electrons and holes and enables catalytic activity. In 2010, Xu and co-workers first demonstrated piezocatalytic water splitting,5 showing that ZnO microfibers and BaTiO3 microdendrites dispersed in water under ultrasonication could produce H2 and O2.5 Unlike conventional systems that rely on metal redox reactions, these piezocatalysts generate strain-induced charge carriers without undergoing chemical changes.6 Since then, researchers have been rigorously expanding this field by focusing either on piezocatalyst design or on combining piezocatalysis with other catalytic techniques. In terms of catalyst design, most studies have concentrated on the following types of piezoelectric materials, such as ZnO,5 perovskites,6 two-dimensional transition metal sulfides/selenides,7 layered bismuth-based piezoelectrics,8 and their composites.9,10 With respect to catalytic reactions, most researchers have focused on piezo-assisted photocatalysis.11,12 Great improvement has been achieved in this field.13 However, the efficiency of pure piezoelectric water splitting for H2 production remains much lower than that of established methods like photocatalysis.14 To realize the full potential of this approach, further advancements are needed in the design and development of materials with enhanced piezoelectric properties and catalytic performance under mechanical vibration.
Graphitic carbon nitride (g-C3N4), a metal-free polymeric material composed primarily of earth-abundant carbon and nitrogen, has attracted growing interest in piezocatalytic applications. This interest stems from its inherent flexoelectricity and the presence of nanoscale, triangular-shaped, non-centrosymmetric holes that contribute to its piezoelectric behavior.15 Moreover, the conjugated ring structure of g-C3N4 provides abundant pyridinic nitrogen sites, which have been identified as active centers for oxygen adsorption and activation.16,17 In 2019, Wang and co-workers reported the first example of H2O2 production from water and oxygen via piezocatalysis using g-C3N4, achieving a rate of 34 µmol h−1 at an ultrasonic frequency of 53 kHz without the use of scavengers.18 g-C3N4 exhibits inherently lower piezoelectric efficiency compared to conventional metal-based dielectric materials. Structural modification through the introduction of carbon and nitrogen vacancies has been proposed as a strategy to disrupt symmetry in the g-C3N4 lattice, thereby enhancing its piezoelectric response. However, the observed enhancement remains limited, and current modification strategies are largely restricted to liquid-phase or thermal exfoliation methods.19
Molten salt methods have been extensively employed to modify carbon-based materials, particularly in applications such as gas adsorption and energy storage.20 These salt melts provide a liquid media that facilitate the condensation and aromatization of organic precursors at temperatures beyond the capabilities of conventional solvents. Additionally, molten salts can function as structure-directing or templating agents, enabling fine-tuning of the microstructure across a wider range of conditions.21 In contrast to thermally polycondensed melon-type carbon nitride, which is typically amorphous or poorly crystalline, molten salts enhance mass and energy transport during thermal treatment, thereby promoting more efficient recondensation processes.22 Recently, Odziomek and co-workers introduced SnCl2 as a non-innocent molten salt medium for synthesizing nanoporous carbons.23 With its low melting point, SnCl2 serves as a solvent, significantly influencing the development of porosity in the resulting materials.
In this study, we combined the unique properties of g-C3N4 with the advantages of the SnCl2-based molten salt method to synthesize a one-pot modified g-C3N4 composite anchored with Sn-based species. To the best of our knowledge, this work is the first to report the use of reactive SnCl2 salt methods to modify g-C3N4 to enhance its piezoelectric effect for catalytic applications. During the thermal pyrolysis process, SnCl2 acts similarly to conventional molten salt methods, promoting the exfoliation of g-C3N4 layers. Additionally, the oxidation of SnCl2 and its interaction with g-C3N4 facilitate the formation of g-C3N4/Sn-based composites. These composites exhibit a significantly enhanced piezoelectricity compared to pure g-C3N4. Notably, the g-C3N4/Sn-based composites demonstrate superior H2 and H2O2 production rates from pure water. This work provides valuable insights into the structural and compositional modulation of g-C3N4 and may inspire further studies on piezocatalytic applications of two-dimensional carbon nitride-based materials.
Results and discussion
Morphology and structure
As illustrated in Fig. 1a, carbon nitride–metal oxide composites were synthesized via a thermal treatment process involving the mixing of melamine with SnCl2 salt, followed by calcination at 500 °C. Depending on the atmosphere employed during thermal treatment, two different samples were obtained: M-0.5Sn (air atmosphere) and M-0.5Sn–N2 (nitrogen atmosphere). For comparison, pristine g-C3N4 (denoted as M) was also prepared in the absence of SnCl2. Detailed synthesis procedures are provided in the Experimental section. The morphologies of the resulting samples were characterized using scanning electron microscopy (SEM) and transmission electron microscopy (TEM), as shown in Fig. 1 and S1. Pristine g-C3N4 (M) exhibited a bulk, layered structure (Fig. 1b). In contrast, both M-0.5Sn (Fig. 1c) and M-0.5Sn–N2 (Fig. 1d) displayed significantly thinner, exfoliated nanosheet structures. This suggests that SnCl2 facilitates the exfoliation of g-C3N4 during pyrolysis, leading to a marked reduction in layer thickness. In samples treated under air (M-0.5Sn), small particles were observed on the surface of the exfoliated sheets (Fig. 1c). The EDS results confirm the presence of C, N, O, and Sn elements. The Sn element is mainly distributed within Sn-containing nanoparticles (Fig. S1), which are anchored on the surface of the g-C3N4 nanosheets, consistent with the TEM image shown in Fig. 1c (inset). Both the quantity and agglomeration of these particles increased with higher SnCl2 loading (Fig. S1a–d). In contrast, the sample prepared under a nitrogen atmosphere (M-0.5Sn–N2) maintained an exfoliated morphology but showed no detectable particle formation (Fig. 1d), suggesting that metal particles are formed only under oxidative conditions.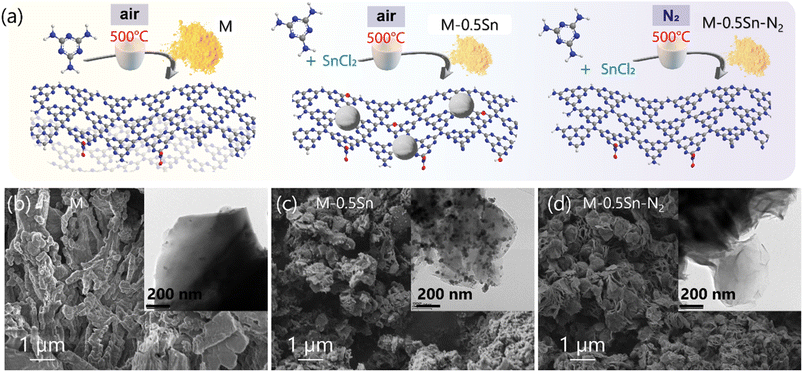 | ||
| Fig. 1 (a) Illustration of the sample preparation process; The SEM and TEM (insert) morphologies of (b) M, (c) M-0.5Sn, and (d) M-0.5Sn–N2. | ||
An additional sample (M-0.5KSn) was synthesized using a KCl–SnCl2 mixture instead of SnCl2 alone to further investigate the role of metal salt composition. As shown in Fig. S2e and f, M-0.5KSn also exhibited an exfoliated morphology relative to pristine g-C3N4, but the layers were thicker compared to M-0.5Sn. This indicates that the KCl–SnCl2 mixture has a weaker exfoliating effect and may also suppress the oxidation of metal salts to some extent. This can be explained by the formation of compounds in the system KCl–SnCl2. Elemental compositions of the samples were analyzed via inductively coupled plasma (ICP) spectroscopy. As summarized in Table S1, samples treated under air contained significant amounts of residual Sn, whereas M-0.5Sn–N2 showed only trace levels. These results can be explained as follows. Under an N2 atmosphere, no oxidation occurs, and the molten salt acts solely as an exfoliating medium, converting melamine into g-C3N4. Most of the SnCl2 is removed during the subsequent acid washing step. In contrast, under air, SnCl2 not only serves as a molten medium to promote melamine decomposition and exfoliation but is also oxidized to form Sn-based oxides. These Sn-based nanoparticles cannot be readily removed by the 1 M HCl solution used in the experimental procedure, leading to the formation of Sn-based/g-C3N4 composites.
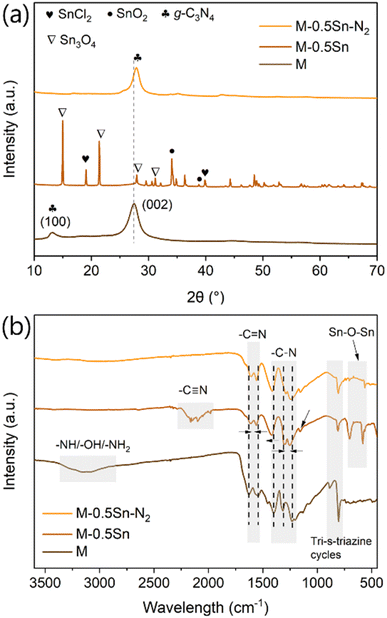
The crystal structures of the samples were characterized by X-ray diffraction (XRD). As shown in Fig. 2a, the pristine g-C3N4 sample (M) exhibits two characteristic diffraction peaks at 2θ = 13.1° and 27.3°. The weak peak at 13.1° corresponds to the in-plane structural packing of the tri-s-triazine units, indexed as the (100) reflection, while the strong peak at 27.3° is attributed to the interlayer stacking of conjugated aromatic systems, corresponding to the (002) reflection typically observed in graphitic materials. These features are consistent with the known crystal structure of g-C3N4.24 In the case of M-0.5Sn–N2, both peaks are present but with significantly reduced intensity, particularly for the (100) reflection, suggesting a loss of in-plane structural order due to increased distortion of the heptazine framework. Additionally, a slight shift of the (002) peak to higher angles was observed, indicating a reduction in interlayer spacing in the remaining crystallites, likely due to enhanced interlayer interactions or compression of the graphitic layers.25–27 Notably, no additional diffraction peaks corresponding to Sn-containing phases were detected, suggesting the absence of crystalline Sn-based species. This observation is consistent with the SEM (Fig. 1d) and ICP (Table S1) results, which show no evidence of residual metal particles or significant Sn content in M-0.5Sn–N2.
The XRD pattern of M-0.5Sn shows the disappearance of the characteristic g-C3N4 peaks at 13.1° and 27.3°, which is not only due to the exfoliation effect but can also be attributed to the emergence of new sharp peaks. These sharp peaks correspond to Sn3O4, SnO2, and unreacted SnCl2 (Fig. S3). The presence of Sn3O4 and SnO2 indicates that SnCl2 underwent partial oxidation during calcination under an air atmosphere, while the residual SnCl2 likely results from incomplete oxidation and its limited solubility, which hinders complete removal during post-synthesis washing.28,29 For the M-0.5KSn sample, no diffraction peaks corresponding to KCl were observed (Fig. S4a), suggesting that the interaction with g-C3N4 primarily involves Sn-containing species rather than KCl. Furthermore, reducing the SnCl2-to-melamine ratio in the precursor mixture led to a decrease in the intensity and number of Sn-related peaks (Fig. S5a), consistent with the lower SnCl2 content in the reaction system.
The Fourier transform infrared (FT-IR) spectra offered valuable insights into the chemical structures of the various samples (Fig. 2b). All samples displayed characteristic vibrational bands near 800 cm−1 and within the 1200–1700 cm−1 range, which are associated with the stretching vibrations of tri-s-triazine rings and C–N heterocycles, respectively.30 Notably, the intensity of the band around 800 cm−1 was significantly reduced in the SnCl2-treated samples compared to M, indicating a higher degree of structural disorder or defects within the carbon nitride framework. A distinct absorption peak at approximately 2180 cm−1 corresponding to the stretching vibration of cyanide (-C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) groups was observed in M-0.5Sn (Fig. 2b) as well as in other samples subjected to oxidative treatment (Fig. S4b and S5b). The presence of this peak is indicative of terminal –C
N) groups was observed in M-0.5Sn (Fig. 2b) as well as in other samples subjected to oxidative treatment (Fig. S4b and S5b). The presence of this peak is indicative of terminal –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N groups resulting from incomplete condensation, and it reflects an increased level of structural defects.31 Broad absorption bands in the 2900–3600 cm−1 region were clearly visible in sample M, which can be attributed to surface-NHx functionalities and physically adsorbed water molecules. In M-0.5Sn and M-0.5Sn–N2, the bands within the 1200–1700 cm−1 region exhibited slight shifts relative to M, likely due to strong interactions between Sn-based species and aromatic C–N structures. The SnCl2-treated samples showed new peaks (Fig. 2b and S5b) in the 570–700 cm−1 region, which are assigned to Sn–O–Sn stretching vibrations. In contrast, M-0.5Sn–N2 exhibited only a weak single peak in this region, suggesting a significantly lower content of residual Sn species. This observation is consistent with other characterizations, including XRD, SEM, and ICP analyses.
N groups resulting from incomplete condensation, and it reflects an increased level of structural defects.31 Broad absorption bands in the 2900–3600 cm−1 region were clearly visible in sample M, which can be attributed to surface-NHx functionalities and physically adsorbed water molecules. In M-0.5Sn and M-0.5Sn–N2, the bands within the 1200–1700 cm−1 region exhibited slight shifts relative to M, likely due to strong interactions between Sn-based species and aromatic C–N structures. The SnCl2-treated samples showed new peaks (Fig. 2b and S5b) in the 570–700 cm−1 region, which are assigned to Sn–O–Sn stretching vibrations. In contrast, M-0.5Sn–N2 exhibited only a weak single peak in this region, suggesting a significantly lower content of residual Sn species. This observation is consistent with other characterizations, including XRD, SEM, and ICP analyses.
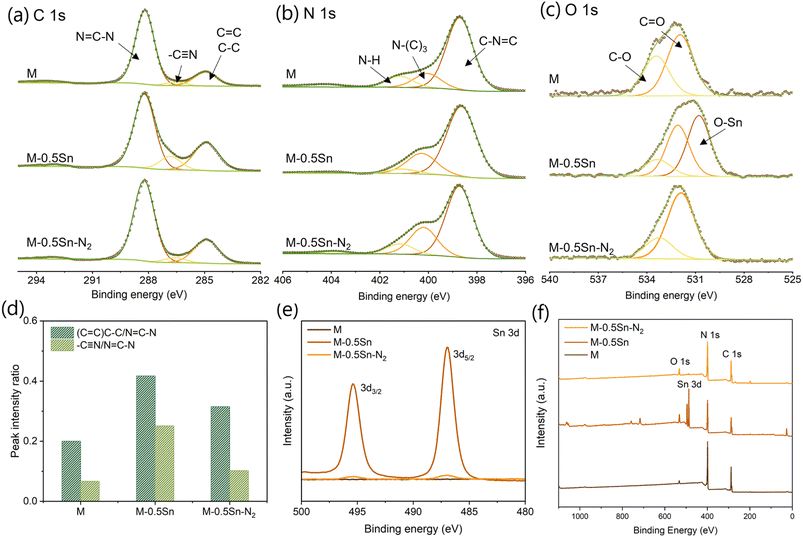
X-ray photoelectron spectroscopy (XPS) was performed to further confirm the structures of the samples (Fig. 3). The survey spectrum (Fig. 3f) confirms the presence of O, N, and C elements in all three samples, with Sn as an additional element in M-0.5Sn. It should be noted that additional minor peaks are observed for the M-0.5Sn–N2 sample, which can also be attributed to Sn-related signals (Fig. 3f). However, these peaks are generally not suitable for quantitative analysis or for distinguishing the chemical states of Sn, as they exhibit relatively weak intensities and small chemical shifts among different Sn oxidation states. Moreover, they are often affected by overlapping Auger features or background contributions. Fig. 3a shows the high-resolution C 1s spectra of the samples. Four peaks at a binding energy of 284.9, 286.5, 288.2, and 293.6 eV are assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(C–C), –C
C(C–C), –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N, N
N, N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–N, and π excitation of heptazine rings, respectively.21 We calculated the relative peak intensity ratios to show the structure variations in different samples. As quantified in Fig. 3d, M shows the lowest ratio of the C
C–N, and π excitation of heptazine rings, respectively.21 We calculated the relative peak intensity ratios to show the structure variations in different samples. As quantified in Fig. 3d, M shows the lowest ratio of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(C–C) peak and the –C
C(C–C) peak and the –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N peak, whereas M-0.5Sn shows the highest ratio for these two peaks among the three samples. The relative intensity of these two peaks indicates the degree of defect in the carbon nitride framework. These results prove that the oxidation of metal salts during sample preparation introduces more defects into the carbon nitride framework, which is well aligned with the FTIR analysis. Fig. 3b shows the high-resolution N 1 s spectra. The binding energies centered at 398.7, 400.1, and 401.2 eV can be ascribed to the C–N
N peak, whereas M-0.5Sn shows the highest ratio for these two peaks among the three samples. The relative intensity of these two peaks indicates the degree of defect in the carbon nitride framework. These results prove that the oxidation of metal salts during sample preparation introduces more defects into the carbon nitride framework, which is well aligned with the FTIR analysis. Fig. 3b shows the high-resolution N 1 s spectra. The binding energies centered at 398.7, 400.1, and 401.2 eV can be ascribed to the C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, N-(C)3, N–H, and π excitation of heptazine rings, respectively.31,32 While the binding energy of various N species in metal salt-treated samples remains largely unchanged compared to M, the relative peak intensity ratios of N–(C)3 and N–H increase and decrease, respectively. This variation in the chemical environment is attributed to the modification induced by the Sn species. Fig. 3c shows the high-resolution O 1s spectra. One extra peak assigned to O–Sn was detected in M-0.5Sn compared to M and M-0.5Sn–N2. Fig. 3e presents the high-resolution XPS spectra of Sn 3d, in which two distinct peaks are clearly attributed to Sn2+/Sn4+ species.33,34 The intensity of the Sn 3d peaks increases markedly when the samples are prepared under an oxidative atmosphere, consistent with the observations from SEM, XRD, and FTIR analyses.
C, N-(C)3, N–H, and π excitation of heptazine rings, respectively.31,32 While the binding energy of various N species in metal salt-treated samples remains largely unchanged compared to M, the relative peak intensity ratios of N–(C)3 and N–H increase and decrease, respectively. This variation in the chemical environment is attributed to the modification induced by the Sn species. Fig. 3c shows the high-resolution O 1s spectra. One extra peak assigned to O–Sn was detected in M-0.5Sn compared to M and M-0.5Sn–N2. Fig. 3e presents the high-resolution XPS spectra of Sn 3d, in which two distinct peaks are clearly attributed to Sn2+/Sn4+ species.33,34 The intensity of the Sn 3d peaks increases markedly when the samples are prepared under an oxidative atmosphere, consistent with the observations from SEM, XRD, and FTIR analyses.
Piezocatalytic performance
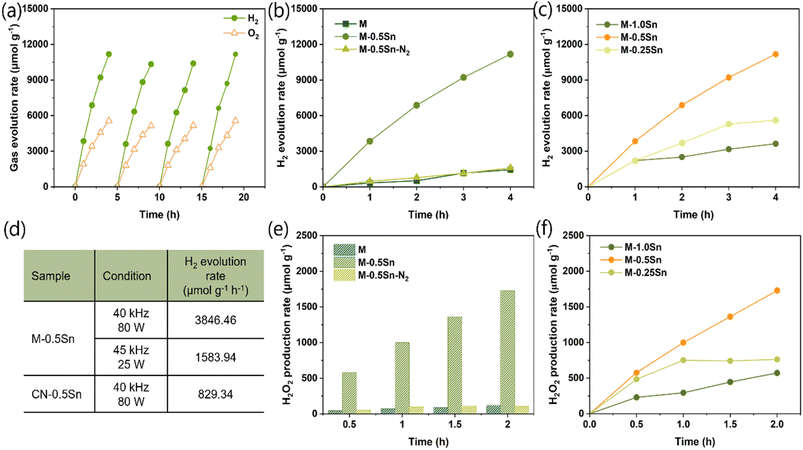
To investigate the role of Sn species in enhancing piezocatalytic activity, the piezocatalytic water splitting performance of various samples was evaluated in an ultrasonication bath (80 W, 40 kHz) without the use of any sacrificial agents or cocatalysts. As shown in Fig. 4a, the amount of H2 produced increased progressively with ultrasonication time within each cycle. Moreover, M-0.5Sn demonstrated excellent stability over four consecutive cycles. The good stability of this catalyst was further confirmed by the negligible changes in its chemical structure before and after the reaction, as evidenced by the FTIR (Fig. S6a), XRD (Fig. S6b), and XPS (Fig. S7) results. Among the tested samples, M-0.5Sn exhibited the highest H2 production rate, achieving 3846.46 µmol g−1 within one hour of ultrasonication: approximately 12 times greater than that of M (328.73 µmol g−1) and about 8 times greater than that of M-0.5Sn–N2 (456.79 µmol g−1) (Fig. 4b). Although the exfoliation effect induced by SnCl2 led to a significant reduction in the thickness of the g-C3N4 layers, sample M showed only a marginal increase in H2 production. This indicates that the mere thinning of the g-C3N4 layers does not substantially enhance piezocatalytic performance. To further elucidate the origin of the performance enhancement observed in M-0.5Sn, the influence of varying SnCl2 content on the piezocatalytic H2 generation was investigated (Fig. 4c). While M-0.5Sn exhibited optimal activity, increasing the SnCl2 content to produce M-1.0Sn led to a decline in H2 evolution. This decrease is attributed to the excessive deposition of Sn-based nanoparticles, which excessively cover the carbon nitride nanosheets (Fig. S2c and d) and hinder their catalytic activity, as neither SnO2 nor SnCl2 typically exhibits any intrinsic piezoelectric properties.13To better understand the role of the interaction between g-C3N4 and Sn species, a comparative sample, CN-0.5Sn, was prepared by pyrolysing the mixture of SnCl2 and the pre-synthesised g-C3N4 (Fig. S8). As shown in Fig. 4d, CN-0.5Sn exhibited a hydrogen production rate of 829.34 µmol g−1, which is approximately twice that of the pristine g-C3N4 sample (M) but still significantly lower than that of M-0.5Sn. These results indicate that while the oxidation of SnCl2 in contact with pre-formed g-C3N4 can moderately improve piezocatalytic performance, the enhancement is substantially more effective when Sn species are introduced during the precursor stage, likely due to stronger interfacial interactions and more uniform distribution within the g-C3N4 matrix. We also investigated the piezocatalytic reaction under two different ultrasonication baths available in the lab: 40 kHz–80 W and 45 kHz–25 W. The H2 production rate decreased with decreasing ultrasonic power. This behavior can be attributed to the fact that higher ultrasonic power induces greater mechanical deformation and generates a stronger internal electric field, which enhances carrier separation and, consequently, improves catalytic performance. The superior performance of our optimal catalyst is further compared with previously published results in Table S2. As shown, M-0.5Sn exhibits comparable or even higher activity than the values reported in the literature. However, we also note that the ultrasonication conditions employed in these studies are not identical. Therefore, a more accurate comparison that considers the influence of ultrasonic energy is necessary for a more standardized evaluation in future work.
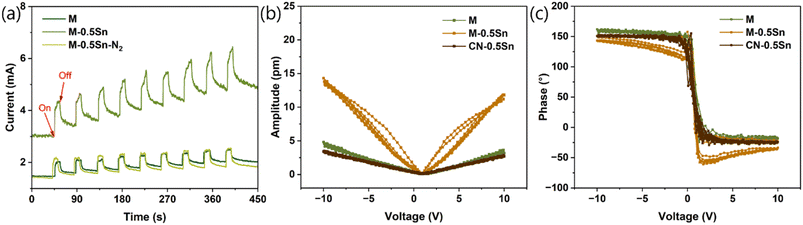
To investigate the influence of the Sn-g-C3N4 interaction on the piezoelectric response, the piezoelectric current responses of M, M-0.5Sn, and M-0.5Sn–N2 were measured under periodic ultrasonic vibration. As shown in Fig. 5a, all three samples exhibit a pronounced and rapid increase in current upon ultrasonication, confirming the generation of piezo-induced charge carriers in the electrodes. Notably, M-0.5Sn shows a significantly enhanced piezoelectric polarization current compared with M and M-0.5Sn–N2, indicating more efficient charge separation and transfer.8,11,12 To further investigate the piezoelectric properties, the ferroelectric switching behaviour of M, M-0.5Sn, and CN-0.5Sn was examined using switching spectroscopy piezo response force microscopy (SSPFM). As shown in Fig. 5b and c, all three samples exhibited a complete 180° phase reversal (Fig. 5b) and a characteristic butterfly-shaped amplitude curve (Fig. 5c), confirming typical ferroelectric switching behaviour. Notably, the calibrated effective piezoelectric coefficient followed the trend: M-0.5Sn > M ≈ CN-0.5Sn. These findings suggest that the strong interaction between the Sn species and the carbon nitride framework formed during the pyrolysis of melamine in the presence of SnCl2 is critical for enhancing piezoelectric performance.
The melting and oxidation of SnCl2 during synthesis play a pivotal role in this enhancement. SnO2 does not exhibit intrinsic ferroelectric behaviour. However, it can be integrated with ferroelectric materials, such as in heterostructures with BaTiO3–TiO2, which enhances specific ferroelectric properties in the resulting composite.35 In this work, SnCl2 serves a dual function: it facilitates the exfoliation of bulk g-C3N4 layers, and it introduces additional structural defects into the g-C3N4 matrix. Simultaneously, the oxidative atmosphere promotes the formation of Sn–O bonds during synthesis. The resulting Sn-based nanoparticles not only increase the specific surface area but also chemically interact with the g-C3N4 framework. Ultrasonic excitation induces periodic mechanical strain in the tin oxide/g-C3N4 composites, generating a piezoelectric potential that promotes the separation and migration of charge carriers at the catalyst surface, thereby enabling pure water splitting. Pristine g-C3N4 exhibits an intrinsic piezoelectric response and can drive water splitting under ultrasonic stimulation; however, the generated piezoelectric potential and charge separation efficiency are relatively limited. Upon incorporation of Sn-based species into the g-C3N4 matrix, the piezoelectric potential is significantly enhanced due to interfacial coupling and strain amplification effects. The strengthened internal electric field accelerates charge transfer and suppresses carrier recombination, ultimately resulting in markedly improved piezocatalytic H2 production.
To investigate the role of the piezoelectric effect in H2O2 production, the samples were evaluated in pure water under ultrasonication, without the addition of any sacrificial agents or cocatalysts. As shown in Fig. 4e, the H2O2 generation rates over two hours followed the order: M-0.5Sn > M-0.5Sn–N2 ≥ M. Notably, M-0.5Sn achieved a production rate of 999.11 µmol g−1, representing a nearly 14-fold increase compared to M (72.03 µmol g−1) and approximately 10 times higher than M-0.5Sn–N2 (99.22 µmol g−1) within one hour. Similarly, the amount of SnCl2 added during catalyst synthesis should have an optimal value, as M-0.5Sn shows the best performance compared to M-0.25Sn and M-1.0Sn (Fig. 4f). To elucidate the underlying reaction mechanisms and identify the active species, the influence of atmospheric conditions (air vs. N2) on H2O2 production was first examined.36 As shown in Fig. S9, the H2O2 yield of M-0.5Sn was significantly higher under an air atmosphere than under N2, indicating that O2 from the air is the primary source of H2O2via reduction. This suggested that the contribution of water oxidation to H2O2 production is minimal during the piezocatalytic reaction. Subsequently, radical scavenging experiments were conducted using isopropyl alcohol (IPA) and benzoquinone (BQ) to probe the roles of hydroxyl radicals (*OH) and superoxide radicals (*O2−), respectively.37 As illustrated in Fig. S9, the addition of BQ led to a marked suppression of H2O2 generation, implying that *O2− is a key intermediate in the piezocatalytic process. In contrast, the presence of IPA had a negligible effect on H2O2 production, suggesting that *OH radicals are not significantly involved in the reaction pathway. Based on these findings, we propose that under ultrasonication, M-0.5Sn facilitates H2O2 production via a two-electron oxygen reduction pathway: adsorbed O2 is reduced by piezoelectrically generated electrons to form *O2−, which is subsequently reduced to yield H2O2.38
Conclusion
In this study, we successfully introduced SnCl2 as a molten salt medium for the synthesis of carbon nitride/metal oxide-based composites, demonstrating a simple yet highly effective strategy to engineer multifunctional piezocatalytic materials. The utilization of SnCl2 under air atmosphere conditions played a critical dual role: it facilitated the thermal exfoliation of bulk graphitic carbon nitride into thinner nanosheets and promoted the formation and anchoring of oxidized Sn-based species onto the g-C3N4 framework. This process also led to the generation of structural defects within the carbon nitride matrix, which are known to enhance charge separation and active site availability. Remarkably, the prepared composites exhibited efficient dual-functional activity for H2 and H2O2 production under ultrasonication, without using any sacrificial agents or additional co-catalysts. The observed enhancements are attributed to the modulation of the local electric field via the piezoelectric effect, which is believed to be strengthened by the SnCl2 melt treatment in an oxidative environment. With optimized SnCl2 loading, the composites achieved a notably high hydrogen evolution rate of 3846.46 µmol g−1 h−1, alongside an impressive hydrogen peroxide production rate of 999.11 µmol g−1 h−1. This work proves the potential of utilizing g-C3N4/metal oxide-based composites for the production of green energy and chemicals via mechanical energy.Author contributions
Ying Pan: conceptualization, data curation, formal analysis, investigation, visualization, methodology, and writing. Luocheng Liao and Yunya Liu: piezoelectric effect testing and data analysis. Dan Qiao: H2O2 quantification. Irene Lamata Bermejo: XRD measurement. Ran Su: data curation, resources, and formal analysis. Nieves Lopez Salas: formal analysis, visualization, and resources. All authors contributed to discussions and commented on the manuscript, and all authors reviewed the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Should any raw data files be needed in another format, they are available from the corresponding author upon reasonable request. Source data are provided with this paper.The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information (SI). Supplementary information: experimental details, characterization data, performance tests, and comparative table. See DOI: https://doi.org/10.1039/d5ta07919e.
Acknowledgements
Ying Pan acknowledges the financial support from the Postdoctoral Fellowship of the University of Paderborn. Nieves Lopez Salas acknowledges funding from DAAD Afriger project (577030859) and Fonds der Chemischen Industrie. Yunya Liu acknowledges funding from the Science and Technology Innovation Program of Hunan Province (2024RC3161). Ran Su acknowledges funding from the Natural Science Foundation of Hebei Province (E2019208243). The authors greatly acknowledge Alexandra Glas for carrying out the XPS measurement on sample M-0.5Sn in Fig. S7.Notes and references
- Q. Qian, Y. Zhu, N. Ahmad, Y. Feng, H. Zhang, M. Cheng, H. Liu, C. Xiao, G. Zhang and Y. Xie, Recent Advancements in Electrochemical Hydrogen Production via Hybrid Water Splitting, Adv. Mater., 2024, 36, 1–37, DOI:10.1002/adma.202306108.
- S. Fukuzumi, Y. M. Lee and W. Nam, Recent progress in production and usage of hydrogen peroxide, Chin. J. Catal., 2021, 42, 1241–1252, DOI:10.1016/S1872-2067(20)63767-6.
- R. Su, H. A. Hsain, M. Wu, D. Zhang, X. Hu, Z. Wang, X. Wang, F. Li, X. Chen, L. Zhu, Y. Yang, Y. Yang, X. Lou and S. J. Pennycook, Nano-Ferroelectric for High Efficiency Overall Water Splitting under Ultrasonic Vibration, Angew. Chem., 2019, 131, 15220–15225, DOI:10.1002/ange.201907695.
- R. Su, J. Zhang, V. Wong, D. Zhang, Y. Yang, Z. D. Luo, X. Wang, H. Wen, Y. Liu, J. Seidel, X. Yang, Y. Pan and F. tang Li, Engineering Sub-Nanometer Hafnia-Based Ferroelectrics to Break the Scaling Relation for High-Efficiency Piezocatalytic Water Splitting, Adv. Mater., 2023, 35, 1–10, DOI:10.1002/adma.202303018.
- K. S. Hong, H. Xu, H. Konishi and X. Li, Direct water splitting through vibrating piezoelectric microfibers in water, J. Phys. Chem. Lett., 2010, 1, 997–1002, DOI:10.1021/jz100027t.
- R. Su, Z. Wang, L. Zhu, Y. Pan, D. Zhang, H. Wen, Z. D. Luo, L. Li, F. Tang Li, M. Wu, L. He, P. Sharma and J. Seidel, Strain-Engineered Nano-Ferroelectrics for High-Efficiency Piezocatalytic Overall Water Splitting, Angew. Chem., Int. Ed., 2021, 60, 16019–16026, DOI:10.1002/anie.202103112.
- S. Li, Z. Zhao, D. Yu, J. Z. Zhao, Y. Su, Y. Liu, Y. Lin, W. Liu, H. Xu and Z. Zhang, Few-layer transition metal dichalcogenides (MoS2, WS2, and WSe2) for water splitting and degradation of organic pollutants: Understanding the piezocatalytic effect, Nano Energy, 2019, 66, 104083, DOI:10.1016/j.nanoen.2019.104083.
- J. Xu, H. Che, C. Tang, B. Liu and Y. Ao, Tandem Fields Facilitating Directional Carrier Migration in Van der Waals Heterojunction for Efficient Overall Piezo-Synthesis of H2O2, Adv. Mater., 2024, 36, 1–9, DOI:10.1002/adma.202404539.
- H. Zeng, C. Liu, B. Lan, M. Tan, C. Yu, Y. Su, L. Qiao and Y. Bai, Bifunctional Bi0.98Sm0.02FeO3/-C3N4 Piezocatalyst for Simultaneous H2 and H2O2 Production, ACS Appl. Mater. Interfaces, 2024, 16, 70566–70574, DOI:10.1021/acsami.4c15127.
- Y. Zheng, Y. Jia, H. Li, Z. Wu and X. Dong, Enhanced piezo-electro-chemical coupling of BaTiO3/g-C3N4 nanocomposite for vibration-catalysis, J. Mater. Sci., 2020, 55, 14787–14797, DOI:10.1007/s10853-020-05001-x.
- W. Li, W. Duan, G. Liao, F. Gao, Y. Wang, R. Cui, J. Zhao and C. Wang, 0.68% of Solar-To-Hydrogen Efficiency and High Photostability of Organic-Inorganic Membrane Catalyst, Nat. Commun., 2024, 15, 6763, DOI:10.1038/s41467-024-51183-2.
- T. Ma, G. Liao, F. Gao, W. Duan, Y. Wang, R. Cui, C. Wang and W. Li, Flexible Hybrid Membrane with Synergistic Exciton Dynamics for Excessive 280 h of Durably Piezo-Photocatalytic H2O-to-H2 Conversion, Small, 2024, 20, 1–12, DOI:10.1002/smll.202408056.
- S. Li, Z. Zhao, J. Zhao, Z. Zhang, X. Li and J. Zhang, Recent Advances of Ferro-, Piezo-, and Pyroelectric Nanomaterials for Catalytic Applications, ACS Appl. Nano Mater., 2020, 3, 1063–1079, DOI:10.1021/acsanm.0c00039.
- J. Ma, C. Peng, X. Peng, S. Liang, Z. Zhou, K. Wu, R. Chen, S. Liu, Y. Shen, H. Ma and Y. Zhang, H2O2 Photosynthesis from H2O and O2 under Weak Light by Carbon Nitrides with the Piezoelectric Effect, J. Am. Chem. Soc., 2024, 146, 21147–21159, DOI:10.1021/jacs.4c07170.
- M. Zelisko, Y. Hanlumyuang, S. Yang, Y. Liu, C. Lei, J. Li, P. M. Ajayan and P. Sharma, Anomalous piezoelectricity in two-dimensional graphene nitride nanosheets, Nat. Commun., 2014, 5, 4284, DOI:10.1038/ncomms5284.
- Z. Li, Y. Zhou, Y. Zhou, K. Wang, Y. Yun, S. Chen, W. Jiao, L. Chen, B. Zou and M. Zhu, Dipole field in nitrogen-enriched carbon nitride with external forces to boost the artificial photosynthesis of hydrogen peroxide, Nat. Commun., 2023, 14, 5742, DOI:10.1038/s41467-023-41522-0.
- J. Barrio, M. Volokh and M. Shalom, Polymeric carbon nitrides and related metal-free materials for energy and environmental applications, J. Mater. Chem. A, 2020, 8, 11075–11116, 10.1039/d0ta01973a.
- K. Wang, D. Shao, L. Zhang, Y. Zhou, H. Wang and W. Wang, Efficient piezo-catalytic hydrogen peroxide production from water and oxygen over graphitic carbon nitride, J. Mater. Chem. A, 2019, 7, 20383–20389, 10.1039/c9ta06251c.
- N. Rono, J. K. Kibet, B. S. Martincigh and V. O. Nyamori, A comparative study between thermal etching and liquid exfoliation of bulk graphitic carbon nitride to nanosheets for the photocatalytic degradation of a model environmental pollutant, Rhodamine B, J. Mater. Sci.: Mater. Electron., 2021, 32, 687–706, DOI:10.1007/s10854-020-04849-8.
- J. Kossmann, T. Heil, M. Antonietti and N. López-Salas, Guanine-Derived Porous Carbonaceous Materials: Towards C1N1, ChemSusChem, 2020, 13, 6643–6650, DOI:10.1002/cssc.202002274.
- Z. Pan, M. Zhao, H. Zhuzhang, G. Zhang, M. Anpo and X. Wang, Gradient Zn-doped poly heptazine imides integrated with a van der Waals homojunction boosting visible light-driven water oxidation activities, ACS Catal., 2021, 11, 13463–13471, DOI:10.1021/acscatal.1c03687.
- M. Cao, K. Wang, I. Tudela and X. Fan, Synthesis of Zn doped g-C3N4 in KCl-ZnCl2 molten salts: The temperature window for promoting the photocatalytic activity, Appl. Surf. Sci., 2020, 533, 147429, DOI:10.1016/j.apsusc.2020.147429.
- X. Zheng, Z. Tian, R. Bouchal, M. Antonietti, N. López-Salas and M. Odziomek, Tin (II) Chloride Salt Melts as Non-Innocent Solvents for the Synthesis of Low-Temperature Nanoporous Oxo-Carbons for Nitrate Electrochemical Hydrogenation, Adv. Mater., 2024, 36, 1–12, DOI:10.1002/adma.202311575.
- S. Yang, Y. Gong, J. Zhang, L. Zhan, L. Ma, Z. Fang, R. Vajtai, X. Wang and P. M. Ajayan, Exfoliated graphitic carbon nitride nanosheets as efficient catalysts for hydrogen evolution under visible light, Adv. Mater., 2013, 25, 2452–2456, DOI:10.1002/adma.201204453.
- J. Zhang, X. Liang, C. Zhang, L. Lin, W. Xing, Z. Yu, G. Zhang and X. Wang, Improved Charge Separation in Poly(heptazine-triazine) Imides with Semi-coherent Interfaces for Photocatalytic Hydrogen Evolution, Angew. Chem., Int. Ed., 2022, 61, e20221084, DOI:10.1002/anie.202210849.
- S. Ganesan, T. Kokulnathan, S. Sumathi and A. Palaniappan, Efficient photocatalytic degradation of textile dye pollutants using thermally exfoliated graphitic carbon nitride (TE-g-C3N4), Sci. Rep., 2024, 14, 2284, DOI:10.1038/s41598-024-52688-y.
- L. Yang, J. Huang, L. Shi, L. Cao, Q. Yu, Y. Jie, J. Fei, H. Ouyang and J. Ye, A surface modification resultant thermally oxidized porous g-C3N4 with enhanced photocatalytic hydrogen production, Appl. Catal., B, 2017, 204, 335–345, DOI:10.1016/j.apcatb.2016.11.047.
- A. R. Kamali, G. Divitini, C. Ducati and D. J. Fray, Transformation of molten SnCl2 to SnO2 nano-single crystals, Ceram. Int., 2014, 40, 8533–8538, DOI:10.1016/j.ceramint.2014.01.067.
- S. Panja, Y. Miao, J. Döhn, J. Choi, S. Fleischmann, S. Guddehalli Chandrappa, T. Diemant, A. Groß, G. Karkera and M. Fichtner, Synthesis, Structural Analysis, and Degradation Behavior of Potassium Tin Chloride as Chloride-Ion Batteries Conversion Electrode Material, Adv. Funct. Mater., 2024, 35, 2413489, DOI:10.1002/adfm.202413489.
- Z. Li, K. Tong, H. Dong, Y. Li, K. Wang, T. Ding, Y. Yun, Y. Zhou and M. Zhu, H-Bonds in Carbon Quantum Dot-anchored C3N5 to Boost Proton-Coupled Electron Transfer for Piezoelectric-Driven Hydrogen Peroxide Synthesis under Ambient Conditions, Angew. Chem., Int. Ed., 2025, 64, e202502390, DOI:10.1002/anie.202502390.
- Q. Deng, H. Li, W. Hu and W. Hou, Stability and Crystallinity of Sodium Poly(Heptazine Imide) in Photocatalysis, Angew. Chem., Int. Ed., 2023, 62, e202314213, DOI:10.1002/anie.202314213.
- W. Wang, H. Zhang, S. Zhang, Y. Liu, G. Wang, C. Sun and H. Zhao, Potassium-Ion-Assisted Regeneration of Active Cyano Groups in Carbon Nitride Nanoribbons: Visible-Light-Driven Photocatalytic Nitrogen Reduction, Angew. Chem., Int. Ed., 2019, 58, 16644–16650, DOI:10.1002/anie.201908640.
- M. Kwoka, L. Ottaviano, M. Passacantando, S. Santucci, G. Czempik and J. Szuber, XPS study of the surface chemistry of L-CVD SnO2 thin films after oxidation, Thin Solid Films, 2005, 490, 36–42, DOI:10.1016/j.tsf.2005.04.014.
- A. Y. Mohamed, S. J. Lee, Y. Jang, J. S. Kim, C. S. Hwang and D. Y. Cho, X-ray spectroscopy study on the electronic structure of Sn-added p-type SnO films, J. Condens. Matter Phys., 2020, 32, 065502, DOI:10.1088/1361-648X/ab4f51.
- R. Li, G. Zhang, Y. Wang, Z. Lin, C. He, Y. Li, X. Ren, P. Zhang and H. Mi, Fast ion diffusion kinetics based on ferroelectric and piezoelectric effect of SnO2/BaTiO3 heterostructures for high-rate sodium storage, Nano Energy, 2021, 90, 106591, DOI:10.1016/j.nanoen.2021.106591.
- H. Yang, C. Li, T. Liu, T. Fellowes, S. Y. Chong, L. Catalano, M. Bahri, W. Zhang, Y. Xu, L. Liu, W. Zhao, A. M. Gardner, R. Clowes, N. D. Browning, X. Li, A. J. Cowan and A. I. Cooper, Packing-induced selectivity switching in molecular nanoparticle photocatalysts for hydrogen and hydrogen peroxide production, Nat. Nanotechnol., 2023, 18, 307–315, DOI:10.1038/s41565-022-01289-9.
- Q. Wu, J. Cao, X. Wang, Y. Liu, Y. Zhao, H. Wang, Y. Liu, H. Huang, F. Liao, M. Shao and Z. Kang, A metal-free photocatalyst for highly efficient hydrogen peroxide photoproduction in real seawater, Nat. Commun., 2021, 12, 483, DOI:10.1038/s41467-020-20823-8.
- W. Zhao, P. Yan, B. Li, M. Bahri, L. Liu, X. Zhou, R. Clowes, N. D. Browning, Y. Wu, J. W. Ward and A. I. Cooper, Accelerated Synthesis and Discovery of Covalent Organic Framework Photocatalysts for Hydrogen Peroxide Production, J. Am. Chem. Soc., 2022, 144, 9902–9909, DOI:10.1021/jacs.2c02666.
| This journal is © The Royal Society of Chemistry 2026 |