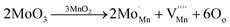Inhibiting Jahn–Teller distortion of δ-MnO2 cathode using 3d and 4d block elements doping to improve the aqueous zinc ion battery performance
Priya
Yadav
a,
Pragati
a,
Amit
Kumar
b,
S. A.
Hashmi
 b,
Satendra Pal
Singh
b,
Satendra Pal
Singh
 c,
Palash J.
Thakuria
d,
Ankur K.
Guha
c,
Palash J.
Thakuria
d,
Ankur K.
Guha
 d,
Arun Kumar
Rai
e and
Alok Kumar
Rai
d,
Arun Kumar
Rai
e and
Alok Kumar
Rai
 *a
*a
aDepartment of Chemistry, University of Delhi, Delhi 110007, India. E-mail: alokkumarrai1@gmail.com
bDepartment of Physics and Astrophysics, University of Delhi, Delhi 110007, India
cDepartment of Physics, University of Lucknow, Lucknow 226007, India
dAdvanced Computational Chemistry Centre, Department of Chemistry, Cotton University, Panbazar, Guwahati, Assam 781001, India
eRaja Ramanna Centre for Advanced Technology, Indore, Madhya Pradesh 452013, India
First published on 23rd December 2025
Abstract
The δ-MnO2 polymorph is a safe, low-cost cathode with high energy density for rechargeable aqueous zinc-ion batteries. However, its structural instability arises from the formation of the intermediate Mn3+ during the reduction of Mn4+. Mn3+ is unstable in octahedral symmetry due to the presence of a single electron in the eg orbital, leading to Jahn–Teller distortion and substantial capacity fading. To address this, we adopted a doping strategy using suitable 3d (Fe3+) and 4d (Mo6+, Rh3+) block elements to substitute Mn3+ and suppress Jahn–Teller distortion. This approach aims to ensure morphological stability, expand interlayer spacing, and facilitate Zn2+ diffusion for enhanced electrochemical performance. Fe3+ (0.55 Å) and Mo6+ (0.59 Å), with ionic radii close to that of Mn3+ (0.58 Å), are more likely to substitute Mn3+ effectively than Rh3+ (0.67 Å). Electrochemical results reveal that Rh3+-doped electrodes show rapid capacity decay, indicating failed substitution and persistent distortion. Mo6+-doped δ-MnO2 maintains high capacity up to 600 cycles but declines gradually with further cycling. In contrast, Fe3+-doped δ-MnO2 demonstrates excellent stability, high capacity at 2C, and low charge transfer resistance, confirming successful mitigation of Jahn–Teller distortion, which is also verified by theoretical evidence obtained via DFT calculation. This doping strategy opens new avenues for developing stable, high-performance aqueous zinc-ion batteries.
1. Introduction
The need for energy storage technology in recent years is significantly growing as human needs continue to increase. Among many energy storage technologies, lithium-ion batteries (LIBs) are found to be one of the most suitable choices due to their advantageous position in terms of their high energy density, excellent cycle life, light weight property, etc.1 However, there are several obstacles in the future development of LIBs such as the use of flammable and toxic components, expensive manufacturing cost, and lack of lithium resources.2 Over the past few years, multivalent metal ion batteries (e.g. Mg, Zn, Ca, and Al) have also been investigated and look promising for advanced energy storage technology due to their higher theoretical volumetric capacity, limited dendrite formation and lower material cost.3,4 However, compared to monovalent ions, the intercalation of multivalent ions into their host matrices is much more sluggish owing to the higher diffusion barriers, largely restricting the development of these batteries. On the other hand, the selection and identification of suitable electrolytes is another major challenging task, which can facilitate the reversible intercalation of multivalent ions into a cathode. However, among all multivalent batteries, rechargeable aqueous zinc ion batteries (ZIBs) are attractive as zinc exhibits higher water compatibility due to its high overpotential for hydrogen evolution and excellent stability compared to alkaline metals, making it a suitable candidate for multivalent charge transport.5 More importantly, the conductivity of water is not only two times higher than their organic counterparts but it is also safe and cheap, which may therefore satisfy the high-rate capacity and specific power demands.6 Thus, it is believed that an aqueous rechargeable battery based on Zn2+ intercalation chemistry will be highly desirable due to the utilization of the Zn metal as an anode, which is also low-cost, highly abundant, non-toxic, and easily recyclable and has high theoretical capacity (820 mAh g−1) with relatively low redox potential (0.76 V vs. standard hydrogen electrode [SHE]).7 Therefore, the design of cathode materials with high structural integrity and excellent stability, along with high energy density and good cyclability, would be very impressive.8,9 The major obstacle to investigate an appropriate cathode material is the strong electrostatic repulsion of divalent Zn2+ ions, resulting in the sluggish transport kinetics that significantly hamper the development of aqueous ZIBs.10,11In comparison to toxic vanadium oxide-based cathodes, which exhibit low discharge platforms and Prussian blue cathodes that have limited capacity, the manganese-based compounds are one of the most promising materials because of their abundant reserves, low toxicity, stable structure, rich manganese valency, high redox potential and excellent zinc storage capacity.12–14 In addition, the open crystal structure with large crystal tunnels or layered spacings and a high operating voltage of ∼1.3 V with a theoretical capacity of ∼308 mAh g−1 based on the discharge product of Zn0.5MnO2 make manganese oxide polymorphs a promising cathode material of aqueous ZIBs.15 There are different crystals of MnO2 such as tunnel structures (α-MnO2, β-MnO2, γ-MnO2, R-MnO2), layered structures (δ-MnO2) and 3D structures (λ-MnO2 and ε-MnO2), which are formed due to the different corner- and/or edges-sharing manners of basic units of MnO6 octahedra.16 Since the layered δ-MnO2 polymorph has a large interlayer spacing (∼7 Å) and shows high potential towards the storage and transport of Zn2+ ions, it has been considered as a promising cathode for aqueous ZIBs. However, a significant challenge in employing MnO2 as a cathode material is the dissolution of Mn2+, arising from the reduction of Mn3+. Mn3+ is inherently unstable in an octahedral coordination environment due to the presence of a single electron in the eg orbital. This configuration induces a pronounced Jahn–Teller distortion, which compromises the structural integrity of the material and ultimately leads to rapid capacity fading during the electrochemical cycling.17,18 Thus, finding effective methods to inhibit Jahn–Teller distortion and to strengthen the layered structure of the δ-MnO2 cathode with excellent cycling stability and rate capability is still a major challenge. To address the structural instability of manganese-based cathode materials (particularly the issue of Jahn–Teller distortion arising from the formation of Mn3+), various modification strategies have been explored to enhance their electrochemical performance. These approaches include doping, surface coating, the creation of oxygen vacancies, composite formation with other inorganic or organic materials and the engineering of tailored morphologies.18 Among these strategies, doping has attracted considerable attention due to its simplicity, which requires no additional processing steps, as well as its effectiveness in modulating both the electronic environment and atomic structure.18–20 This method has been found to be helpful to stabilize the Mn-based frameworks by suppressing Jahn–Teller distortion. As a result, a wide variety of metal ions such as Ca2+, Ni2+, Cu2+, Co3+, V5+ and Fe3+ have been successfully incorporated into MnO2 lattices.5,21–25 The selection of dopants is typically guided by factors such as suitable ionic radius, strong bonding affinity with oxygen, and chemical inertness during redox processes. These characteristics collectively contribute to suppressing the Jahn–Teller distortion, enhancing the structural integrity and ultimately improving the cycling stability during repeated Zn2+ (de)intercalation.25
Since the Mn3+-rich region induces the Jahn–Teller distortion (t32g–e1g) in the δ-MnO2 cathode, we have opted for three dopants of different ionic radii with respect to Mn3+ in this work to study the effect of Jahn–Teller distortion on the electrochemical performances.17,26 The objective is to investigate whether the close value of the dopant's ionic radius could efficiently replace the unstable Jahn–Teller Mn3+ ions in comparison to a dopant with a large ionic radius, and study their influence on the electrochemical performance after mitigation of the Jahn–Teller distortion. Thus, iron (Fe3+; 3d block element), molybdenum (Mo6+; 4d block element) and rhodium (Rh3+; 4d block element)-doped δ-MnO2 cathodes have been synthesized using a straightforward hydrothermal synthesis process and their electrochemical performances have been compared against that of pure δ-MnO2 for rechargeable aqueous ZIBs. Since the ionic radii for the dopants Fe3+ and Mo6+ are 0.55 Å and 0.59 Å, respectively, which are very close to that of Mn3+ (0.58 Å) in comparison to that of Rh3+ (ionic radius: 0.67 Å), it is anticipated that Fe3+ and Mo6+ doping will certainly substitute Mn3+ and reduce the Jahn–Teller active content, resulting in an enhanced number of Mn4+ to maintain the charge neutrality.27,28 Although the ionic radius of Mn4+ (0.53 Å) is smaller than that for Mn3+, the lattice shrinkage is also obvious after suppression of the Jahn–Teller distortion, which increases the structural stability of the active material. The obtained electrochemical results show that the Mo6+- and Fe3+-doped δ-MnO2 cathodes demonstrate excellent capacity even at a high current rate of 2C in comparison to Rh3+ doping, which could not retain the host structure of the δ-MnO2 cathode during repeated Zn2+ intercalation/de-intercalation under long-range cycling, resulting in the structural collapse of the host material. While the Mo6+-doped δ-MnO2 electrode demonstrates high capacity, the capacity trend is unstable and decreases with increasing cycle numbers, despite the Shannon radius of Mo6+ (0.59 Å) being nearly identical to that of Mn3+ (0.58 Å). In contrast, after introducing Fe3+ into the δ-MnO2 cathode, it seems that the dopant preferentially replaces the Mn3+ sites, resulting in the Jahn–Teller effect of Mn3+ being alleviated, followed by improvement of the material stability and electronic conductivity.17 Hence, to better understand the inherent issues of the δ-MnO2 cathode, including its poor electrical conductivity, low Zn2+ ion diffusivity and Jahn–Teller distortion, the current work has been performed with detailed investigations using suitable dopants from 3d and 4d block elements.
2. Results and discussion
2.1 Structural identification and morphological studies
Fig. 1 illustrates the synchrotron XRD patterns of the calcined samples of pure (δ-MnO2), Fe3+-, Mo6+- and Rh3+-doped MnO2 samples. All four patterns exhibit characteristic diffraction peaks that can be perfectly indexed to the birnessite framework layered structure of δ-MnO2 (JCPDS Card No. 80-1098). The presence of well-defined peaks in the patterns clearly indicate the highly crystalline nature of all the samples. While the pure samples crystallize into the monoclinic structure in the C2/m space group, the other three doped samples crystallize in the monoclinic structure in the C2/m space group, along with the formation of a secondary Mn0.98O2 phase with a tetragonal structure in the I4/m space group (ICDD No. 01-081-1947). A broadening of peaks in the doped samples can be easily seen in comparison to pure δ-MnO2, which can be attributed to the incorporation of dopant ions into the δ-MnO2 lattice, resulting in the formation of smaller nano crystallites. In order to precisely determine the crystal structure and lattice parameter, a single phase Lebail refinement using Fullprof Program29 for the pure sample and two-phase refinement for the doped samples were carried out. This resulted in a very good fit between the experimental and calculated patterns. For the Lebail refinement, initial structural models were adopted from Invernizzi et al.30 and the refinement was carried out in the 2θ range of 3.5° to 35°, as shown in Fig. 1. The wavelengths used in the synchrotron XRD measurements for the pure, Fe-doped, and Rh-doped samples and for the Mo-doped sample were 0.6606 Å and 0.7293 Å, respectively. During the refinement, a Pseudo-Voigt function for profile shape and a linear interpolation between a set background point with refinable heights for the background were used. All the structural parameters were varied during the refinement. The structures of all three doped compounds were found to be nearly identical with very small changes in the cell volume due to the very small differences in the ionic radii. The cell volume as obtained after Rietveld refinement was found to be 103.3(1), 113.9(4), 129.4(1) and 127.7(5) for the pure, Fe3+-, Mo6+- and Rh3+-doped MnO2 samples, respectively. Table S1 (provided in the SI) summarizes the data of the Rietveld refinement.The detailed morphological investigation of the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 samples was carried out by FE-SEM analysis at different magnifications, and the results are displayed in Fig. 2a, b, e, f, i, j, m and n, respectively. It can be seen that the FE-SEM images of the pure, Fe3+ and Rh3+ electrodes exhibit nanoflower-like morphology of several nanometers in size, which is in fact composed of the self-assembly of crumpled nanosheets or flake-like 2D nanosheet layers, whereas the Mo6+-doped δ-MnO2 sample revealed a well-defined hawthorn-like morphology, consisting of randomly arranged small nanorods embedded within agglomerated clumps. These clumps are primarily formed through the self-assembly of smaller particles that coalesce into larger structures. Furthermore, it is believed that the nanoflower-like morphology is generated from the high surface energy and interlayer van der Waals attraction of MnO2.8 The dopants have clearly affected the crystal growth and agglomeration processes, resulting in distinct morphological differences. However, out of the three doped samples, Fe3+ doping clearly exhibits excellent hierarchical structure with high porosity, which is possibly beneficial for effective electron transport. The unique architecture obtained in the Mo6+-doped δ-MnO2 sample is also believed to reduce surface energy, while simultaneously enhancing the porosity. The porous nature significantly increases the surface area, promoting faster ion diffusion and better electrolyte penetration, which are favourable for high-performance ZIBs applications. In contrast, the Rh3+ doping demonstrates a compact morphology with dense packing of 2D nanosheets, which shows less porosity, suggesting a possible reduction of the available surface area in comparison to the other Fe3+- and Mo6+-doped δ-MnO2 samples. As shown in Fig. 2c, g, k and o, the FE-TEM images are perfectly analogous to the FE-SEM observations, clearly demonstrating the nanoflower-like morphology for the pure, Fe3+- and Rh3+-doped δ-MnO2 samples and confirming a distinctive hawthorn-like morphology for the Mo6+-doped δ-MnO2 cathode. As we have discussed above, the interconnected and well-aligned 2D nanosheets are clearly evident with porous architecture. The nanoflower-like skeleton structure can expose more surface area and increase the ion transportation and diffusion in the electrolyte. Since the doping effect can be easily observed, it seems that the TEM image of the pure δ-MnO2 sample (Fig. 2c) shows the presence of small pore sizes with rough surfaces. Meanwhile, the Fe3+-doped δ-MnO2 sample (Fig. 2g) exhibits thin, sharp and comparatively transparent nanosheets. The introduction of Fe appears to promote the formation of an elongated, anisotropic structure. As can be seen in Fig. 2k, the morphology of the Mo6+-doped δ-MnO2 sample consists of loosely packed, agglomerated clusters with rooted nanorods, confirming the open and mesoporous architecture observed in the SEM images. Fig. 2o illustrates the effect of Rh3+ doping, which offers a compact needle-like structure with brighter elongated nanosheets, as compared to Fe3+ doping. More importantly, the insets of Fig. 2c, g and o also display the zoomed TEM images of their corresponding samples. The high-resolution TEM images of all four pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 samples are provided in Fig. 2d, h, l and p, respectively, showing the high crystallinity of the samples with clear lattice fringes. The common interplanar distances of 0.241 nm, 0.70 nm and 0.352 nm of all four samples corresponding to the (−111), (001) and (002) planes, respectively, are perfectly matched with the standard JCPDS data of the pristine δ-MnO2 structure.
Energy-dispersive X-ray spectroscopy (EDS) coupled with FE-SEM was carried out to validate the elemental composition and confirm the successful incorporation of dopant ions into the δ-MnO2 lattice. A detailed explanation of the EDS analysis, along with Fig. S1, is presented in the SI.
Furthermore, inductively coupled plasma (ICP) analysis was performed to precisely quantify the percentage of doped metal ions in the δ-MnO2 samples. The obtained results, as given in Table S2 of the SI, confirm the successful incorporation of Fe, Mo, and Rh into the δ-MnO2 lattice. It can be seen that each doped sample contains a measurable concentration of the respective dopant, along with manganese, indicating effective substitution without the presence of excess or unreacted dopant species.
To further prove that doping occurred and investigate the electronic state and the valency of ions within the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 samples, XPS analysis was carried out, and the result is shown in Fig. 3. The survey spectrum of pure δ-MnO2 (Fig. 3a) confirms the presence of manganese (Mn 2p), and oxygen (O 1s), indicating the high purity of the pristine sample. The Mn-2p spectra in all four, pure and doped samples exhibit two peaks at around 642.4 ± 0.3 eV and 654.1 ± 0.3 eV, which correspond to Mn 2p3/2 and Mn 2p1/2, demonstrating the existence of Mn4+ in the samples.18,31 Deconvolution of these peaks clearly reveals the coexistence of Mn3+ and Mn4+ oxidation states, with the binding energy gap of ∼11.4 eV indicating the mixed-valent nature of manganese in the sample.24 The MnO2 charge storage mechanism indicates that the multiple valence states (Mn3+ and Mn4+) of the Mn ions in MnO2 introduce ion defects, thereby shortening the ion diffusion paths and promoting the redox reactions.32 The increased Mn3+ content within the Mn 2p spectral region can be attributed to the intercalation of dopants. A low ratio of Mn4+/Mn3+ was observed in the doped samples in comparison to pure δ-MnO2. The other three doped samples show iron, molybdenum and rhodium spectra, confirming that doping occurred. The Fe-2p spectra (Fig. 3d) in the iron-doped sample have been deconvoluted into two peaks of Fe 2p3/2 at 711.5 eV and Fe 2p1/2 at 725.1 eV, suggesting the existence of Fe3+ in this sample, which can substitute Mn in the [MnO6] octahedron sites.27 The Mo-3d spectrum (Fig. 3g) displays two well-defined peaks at 231.7 eV and 234.9 eV, which are ascribed to Mo 3d5/2 and Mo 3d3/2, respectively.33 These binding energies confirm the presence of Mo in the +6 oxidation state within the δ-MnO2 lattice without the formation of secondary Mo oxide phases. On the other hand, Fig. 3j displays two major peaks located at 307.6 eV and 312.4 eV with a spin energy separation of 4.8 eV, which can be allotted to the Rh 3d5/2 and Rh 3d3/2 spin states, respectively.34 The obtained binding energy indicates the successful incorporation of Rh3+ ions into the lattice of δ-MnO2. More importantly, the O 1s spectra of all four samples exhibit three individual peaks at 529.7 ± 0.2 eV, 530.6 ± 0.4 eV and 531.9 ± 0.6 eV, which can be assigned to Mn–O–Mn bonds, Mn–OH bonds and H–O–H bonds, respectively.33–36 The Mn–O–H bonds are indicative of oxygen vacancies, which suggests that the incorporation of Fe3+, Mo6+ and Rh3+ increases oxygen vacancies because of their different valences compared to Mn4+. This results in a higher concentration of oxygen vacancies, which is known to enhance the material conductivity.
X-ray fluorescence (XRF) analysis was also carried out to determine the elemental composition of the samples and the obtained results are displayed in Fig. S2 of the SI. In all the spectra, the strong Mn-Kα and Mn-Kβ peaks validate the Mn-rich composition of the host oxide lattice. More importantly, the additional peaks corresponding to all dopants (Fe3+, Rh3+, and Mo6+) clearly appear in the spectra of the doped samples. The presence of these element-specific signals confirms the successful incorporation of Fe, Mo, and Rh into the bulk of the MnO2 material.33 This observation is consistent with the XPS data (Fig. 3), which also suggests partial substitution of Mn4+ within the MnO2 lattice. It is reasonable to suggest that a small amount of dopant can substitute Mn ions within the MnO2 lattice. However, the excessive doping may disrupt the birnessite structure, leading to reduced crystallinity and compromised structural integrity.37 Overall, the XRF analysis certifies the elemental composition and confirms that the intended dopants have been successfully incorporated into the δ-MnO2 host material.
To investigate the porous features of the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 samples, N2 adsorption–desorption isotherms have been recorded, as presented in Fig. S3 of the SI. The obtained type IV hysteresis loop indicates the existence of abundant mesopores in the samples. The internal and external mesoporous structures including pore sizes exhibit substantial influence on the performance of electrode materials.36 A detailed discussion of the surface area and average pore volume is provided in the SI.
2.2 Electrochemical performances
Zinc ion storage properties have been investigated using cyclic voltammetry (CV) measurements. Fig. 4a–d displays the initial four CV curves of the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 cathodes at a constant scan rate of 0.1 m Vs−1 within the potential range of 0.6 to 1.8 V versus Zn/Zn2+, respectively. Despite variations in the dopant, there are no notable differences between the CV profiles of the pure and doped electrodes, indicating that the doping has not affected the Zn2+ storage mechanism of the δ-MnO2 cathode. Since the CV scan starts from the open circuit voltage of 1.4 V in the pure, Fe3+-, and Rh3+-doped δ-MnO2 cathodes, the pure and Fe3+-doped δ-MnO2 cathodes display a broad cathodic peak in comparison to the Rh3+-doped electrode at ∼1.2 V, which is split into two peaks in later cycles. In addition, a significant change in the profile particularly, the enhanced reversibility and less polarization can be clearly seen after doping, indicating the pronounced impact of doping.31 The increased area under the curve after the first cycle indicates electrode activation.38 After the first cycle, the two asymmetric cathodic peaks located at ∼1.2 V and ∼1.4 V for all four electrodes are evidently observed, corresponding to the reversible insertion/extraction of H+ and Zn2+, accompanied by the reduction of Mn (from Mn4+ to Mn3+) respectively.37 It can be also observed that the current density of the redox peaks progressively increases with each successive CV cycle, indicating enhanced electrochemical activity and improved utilization of the doped MnO2 electrode. This behaviour is attributed to the gradual activation of the electrode material, where continued cycling leads to increase exposure of electrochemically active sites and improved ion diffusion pathways, thereby facilitating more efficient redox reactions.33 On the other hand, compared to a broad oxidation peak observed at ∼1.6 V for the pure δ-MnO2 cathode, two oxidation peaks appear at ∼1.57 V and ∼1.61 V for all doped electrodes, attributed to the extraction of inserted ions (H+ and Zn2+).38 It can be also seen that the subsequent CV curves are almost coincident with stable peak positions, implying the superior reversibility of the electrochemical reactions and high structural integrity of the electrodes. | ||
| Fig. 4 Cyclic voltammetry curves at the constant current density of 0.1 mV s−1 of (a) pure δ-MnO2, (b) Fe-doped δ-MnO2, (c) Mo-doped δ-MnO2 and (d) Rh-doped δ-MnO2 electrodes. | ||
The galvanostatic charge/discharge profiles of the 1st, 2nd, 10th, 50th and 100th cycles of the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 cathodes are shown in Fig. 5a–d at the constant current rate of 1C in the voltage range of 0.6–1.8 V versus Zn/Zn2+. There is one discharge plateau at around ∼1.24 V in the first cycle of all four electrodes, which corresponds to the one reduction peak observed in the CV curve of the initial cycle, indicating the insertion of Zn2+ ions into the host. The pure δ-MnO2 electrode exhibits a first discharge capacity of 120.4 mAh g−1 at 1C, which is much lower than the capacities of 139.1 mAh g−1, 365.8 mAh g−1 and 342.3 mAh g−1 of the Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 electrodes, respectively. After the first cycle, the discharge plateaus are more evident with a slight shift at ∼1.28 V in the subsequent cycles induced by the insertion of Zn2+ ions. It can be seen that the first and second cycles of all of the doped electrodes are almost overlapped, implying that there is no formation of a solid electrolyte interphase (SEI) film on the electrode surfaces.33,39 However, with increasing cycles, the Fe3+-doped electrode discharge/charge capacity increases and reaches up to ∼175.2/171.1 mAh g−1 for the 100th cycle, while the Rh3+-doped and pure δ-MnO2 electrode specific capacities decline at 295.7/295.1 mAh g−1 and 113.2/114.1 mAh g−1 for the same cycle, respectively. Notably, the Mo6+-doped electrode maintains nearly identical discharge/charge profiles over extended cycling, delivering ∼364/368 mAh g−1, ∼318/325 mAh g−1 and ∼272/274 mAh g−1 for the 10th, 50th and 100th cycles respectively, with nearly 100% coulombic efficiency, reflecting outstanding electrochemical reversibility and minimal polarization. This stable behaviour is attributed to the effect of Mo6+ doping and the hawthorn-like morphology, which collectively facilitate the rapid ion diffusion and buffer structural strain during the repeated cycling. On the other hand, Mn4+ (0.53 Å) might be reduced to Mn3+ (0.58 Å) in the presence of the high valence Mo6+ ions to ensure charge balance, resulting in the lattice expansion along the c-direction, which is also more conducive to the transport of Zn2+. Therefore, the enlarged interlayer spacing induced by doping, combined with the presence of interconnected porous channels, collectively enhances the electrochemical performances. Moreover, the increased interlayer spacing helps alleviate internal stress, thereby reducing structural degradation. Furthermore, the minimal voltage hysteresis between the discharge and charge plateaus confirms efficient redox kinetics and robust structural integrity.18,24
Fig. 5e highlights the comparison of the cyclic performance of all four electrodes of the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 cathodes at 1C up to 500 cycles. It can be observed that the Rh3+-doped δ-MnO2 cathode initially exhibited a higher capacity compared to the Mo6+-doped electrode for up to 60 cycles. However, this was followed by a significant decline in the performance. In the later cycles, the Mo6+-doped cathode demonstrated the highest capacity among all four electrodes, although its performance also showed a downward trend with increasing cycle numbers. The Mo6+-doped δ-MnO2 cathode delivers the highest charge capacity of 249.5 mAh g−1 after 500 cycles, followed by the Rh3+-doped δ-MnO2 electrode with 210.2 mAh g−1 and then the Fe3+-doped electrode with a capacity of 154.2 mAh g−1. The pure δ-MnO2 cathode delivers only 78.9 mAh g−1 after 500 cycles, which is obvious due to the high charge transfer resistance and structural collapse by volume expansion/contraction during repeated cycling. Since the Mo6+- and Rh3+-doped δ-MnO2 cathodes show poor capacity retention, the Fe3+-doped δ-MnO2 cathode stands out by maintaining excellent capacity retention with stable performance over 500 cycles. This improvement is likely due to its enhanced structural robustness and high electrochemical reversibility, highlighting the advantages of doping. Substitution of Mn by suitable dopants plays a critical role in suppressing the Jahn–Teller distortion, which is typically induced by the presence of Mn3+ ions. Since Mn3+ (d4) is Jahn–Teller active and leads to structural instability upon repeated Zn2+ insertion/extraction, its partial suppression via doping stabilizes the crystal lattice and mitigates phase degradation during prolonged cycling.18 Doping with Fe3+, Mo6+ and Rh3+ into the δ-MnO2 lattice can enhance the electronic conductivity by facilitating improved charge transfer within the material. Moreover, the dopants can introduce defects or vacancies in the crystal lattice, which generate additional active sites or pathways for ion transport. This leads to faster and more efficient ion diffusion, thereby improving the rate capability and overall electrochemical kinetics. A representative substitution reaction of Mo6+ doping can be described as follows, where V′′′Mn denotes a Mn4+ vacancy in the lattice:
To gain more insights into the doping effect, the rate capability test was performed at different C-rates ranging from 0.1C to 2C and the obtained results are summarized in Fig. 5f. All four electrodes were cycled for a minimum of 10 cycles at each current rate to scrutinize the structural stability of the electrode materials. As per the obtained cyclability data in Fig. 5e, it has become obvious that the Mo6+- and Rh3+-doped δ-MnO2 cathodes initially deliver superior capacity when compared to the Fe3+-doped and pure electrodes. It also can be seen that both Mo6+- and Rh3+-doped electrode capacities are overlapped, especially at higher current rates. The Mo6+-doped δ-MnO2 cathode delivered charge capacities of 761.9, 566.4, 416.7, 384.5, and 266.3 mAh g−1 at current rates of 0.1C, 0.2C, 0.5C, 1C and 2C, respectively. These values are comparable to those of the Rh3+-doped electrode, which exhibited capacities of 664.4, 639.6, 494.9, 384.4 and 266.3 mAh g−1 at the same current rates. However, the capacities obtained for both Mo6+- and Rh3+-doped electrodes are significantly higher than those of the Fe3+-doped δ-MnO2 electrode (654.4, 504.8, 338.7, 220.1 and 156.9 mAh g−1) and the pure δ-MnO2 electrode (399.3, 325.1, 229.3, 156.2 and 92.9 mAh g−1) under the same rates. Furthermore, when the current rate is reverted back to 0.1C, the Mo6+-, Rh3+- and Fe3+-doped cathodes, as well as the pure δ-MnO2 cathode, recover capacities of 667.4 mAh g−1 (87% retention), 547.3 mAh g−1 (82% retention), 404.8 mAh g−1 (62% retention) and 306.7 mAh g−1 (77% retention), respectively. The rate capability results indicate that Mo6+ and Rh3+ doping effectively enhances the kinetics of the electrochemical reactions within the δ-MnO2 structure, enabling fast charge transfer and improved ion diffusion, which is crucial for high-rate applications.23,24 However, the serious capacity decline at each current rate can be also seen for the Rh3+-doped δ-MnO2 cathode. Meanwhile, the other two doped electrode capacities are quite stable until the end of the measured C-rate performance.
We have now seen that doping is an efficient strategy to enhance the specific capacity of the δ-MnO2 cathode and the small doping amount plays a key role for both specific capacity and capacity retention. Thus, all four electrodes were evaluated again at a high current rate of 2C for 530 cycles to further confirm the superiority of the doped δ-MnO2 cathode over the pure one, and the obtained results are displayed in Fig. 5g. The cyclability data obtained at 2C especially reveal a key contrast between the Mo6+-, Rh3+- and Fe3+-doped δ-MnO2 electrodes, highlighting the trade-off between the initial capacity and long-term stability. As observed, the Mo6+-doped δ-MnO2 electrode exhibits a substantially higher capacity at 2C compared to all other electrodes, clearly demonstrating the enhanced electrochemical performance imparted by Mo doping. This remarkable cycling performance can be ascribed to the presence of Mo6+ ions, which enter the δ-MnO2 lattice by substituting a fraction of Mn4+ ions and generating Mn4+ vacancies to maintain electrostatic balance, thereby minimizing the extent of Jahn–Teller distortion. This results in improved lattice rigidity, lower internal strain and enhanced structural integrity over extended cycling.19,25 However, despite the superior initial capacities of the Mo6+- and Rh3+-doped δ-MnO2 cathodes, they exhibit significant capacity degradation over extended cycling, ultimately declining to 155.2 mAh g−1 and 83.03 mAh g−1, respectively, after 530 cycles. In contrast, the Fe3+-doped δ-MnO2 electrode maintains a relatively stable capacity for up to 530 cycles with the capacity of 100.5 mAh g−1, indicating excellent long-term stability. The formation of an excess Mn0.98O2 phase, as revealed by Rietveld refinement analysis in the Mo-doped δ-MnO2 sample, may be one of the factors contributing to its high initial capacity. It is believed that this excess phase likely provides additional active sites or boosts the kinetics, resulting in enhanced electrochemical performances. However, it seems that the obtained high amount of extra phase alters the structural integrity of the material, reducing its ability to accommodate the volume changes associated with repeated Zn2+ insertion and extraction. As a result, the Mo-doped sample experiences capacity decay during long-term cycling. In contrast, the Fe-doped δ-MnO2 sample appears to contain an optimal amount of secondary phase, which is higher than that in the Rh-doped sample and lower than that in the Mo-doped sample, contributing to improved structural stability and a more consistent capacity over extended cycling. The good retention of the Fe3+-doped electrode implies that its kinetics and stability were synergistically improved by Fe3+ doping. The foremost reason behind the long range cyclability of the Fe3+-doped δ-MnO2 cathode and superior specific capacity of the Mo6+-doped δ-MnO2 is possibly due to the reduction of Jahn–Teller distortion driven by the presence of Mn3+. As mentioned above, the ionic radii of Fe3+ and Mo6+ are relatively close to that of Mn3+. Thus, they can substitute Mn3+ within the lattice more effectively than Rh3+, whose ionic radius is larger. The Mn3+ substitution reduces the concentration of Jahn–Teller active Mn3+ sites, resulting in a decrease in the Jahn–Teller distortion. The dilution of Mn3+ by the doping of Fe3+ and Mo6+ may promote the Mn4+ content, which maintains charge balance during cycling.22,39–41 Since Mn4+ has a smaller ionic radius compared to Mn3+, the alleviation of Jahn–Teller distortion causes the lattice to shrink, followed by the enhanced structural stability of the δ-MnO2 cathode material. Though Rh3+ doping may be inefficient to mitigate the Jahn–Teller distortion, the limited electrochemical reaction kinetics may volunteer structural instability during repeated Zn2+ intercalation/de-intercalation, causing the host material to collapse. Opposite to this, the substitution of Fe3+ and Mo6+ in the Mn-site may be conducive to avoid the collapse of the δ-MnO2 crystal structure induced by Jahn–Teller distortion, resulting comparable capacity at this high rate to the reported studies.42–46 Finally, the pure δ-MnO2 cathode retains only 60.6 mAh g−1 after 530 cycles, corresponding to just 56% of its initial capacity. This significant capacity loss indicates considerable structural degradation under prolonged cycling conditions.
To further validate the suppression of the Jahn–Teller distortion (JTD), a theoretical investigation was conducted using density functional theory (DFT). The obtained theoretical results were found to be consistent with the experimental observations. Fe3+ doping in the δ-MnO2 cathode effectively eliminates the JTD effect, as indicated by the nearly identical axial and equatorial bond lengths. Since Mn4+ and Mn2+ do not exhibit JTD, it is believed that Fe3+ either oxidizes Mn3+ to the JTD-free Mn4+ state or reduces Mn3+ to Mn2+. In contrast, the other dopants, Rh3+ and Mo6+, fail to induce a similar effect due to the destruction of structural unity, followed by the loss of unit-cell symmetry, which is because of their ionic radius mismatch. A detailed discussion of the DFT results, along with the relevant figures and calculations, is provided in Fig. S4–S6 in the SI of the manuscript.47–49
To distinguish the conductivity, internal resistance and the reasons for the improved long-term stability of the Fe3+-doped δ-MnO2 cathode, electrochemical impedance spectroscopy (EIS) was carried out after cycling and the results are shown in Fig. 6a–d. The equivalent circuit diagram used to fit the EIS data is incorporated in the insets of the figure. The conductivity refers to the ability of the bulk material to transport charge carriers (electrons or ions), directly affecting how easily charges move within the electrode material. On the other hand, charge transfer resistance arises at the electrode–electrolyte interface and reflects the difficulty of electron exchange during redox reactions. The Nyquist plot of the cycled electrodes clearly distinguishes the charge transfer resistance of all four electrodes. At the high and medium frequency regions, the EIS plot of the pure δ-MnO2 electrode exhibits large impedance with a bigger depressed semicircle in comparison to other doped cathodes, suggesting the poor conductivity of the pure electrode with high charge transfer resistance. Compared with the Mo6+- and Rh3+-doped δ-MnO2 electrodes, the smaller diameter of the semicircle at the high and medium frequency regions of the Fe3+-doped δ-MnO2 cathode indicates its lower charge transfer resistance and consequent faster reaction kinetics. The results of the fitting analysis indicate that the charge-transfer resistances were 274.3 Ω, 28.8 Ω, 132 Ω and 62.7 Ω, for the pure, Fe3+-, Mo6+- and Rh3+-doped δ-MnO2 electrodes, respectively. Additionally, at the low frequency region, the increased steepness of the slope for the Fe3+-doped δ-MnO2 cathode in comparison to the other two electrodes implies that Fe3+ doping promotes a higher ion diffusion coefficient, which can be attributed to the enlargement in the surface area that is electrochemically active and accessible for redox reactions.38,39 As anticipated, the Fe3+ substitutes Mn3+ to mitigate Jahn–Teller distortion, resulting in an increase the concentration of holes, which is advantageous to reduce the charge transfer resistance and increase the electrical conductivity, diffusion coefficient and kinetic properties during the charge/discharge process.
Since the different reaction mechanisms, such as H+/Zn2+ insertion, conversion reaction mechanism and deposition–dissolution reaction mechanism have been reported so far for aqueous ZIBs, ex situ XRD analysis of all electrodes was performed after 200 cycles in the discharge state at 1C to confirm the storage mechanism and the obtained results are displayed in Fig. 7. Ex situ XRD data of the pure δ-MnO2 presented in Fig. 7a perfectly match well with that for Mn3O4 (JCPDS no. 001-1127). It can be seen that the proton-coupled electron transfer leads to the formation of MnOOH, in which Mn is in the +3 oxidation state. This Mn(III), which is Jahn–Teller active and thermodynamically unstable in aqueous media, tends to be disproportionate to Mn2+.40 The Mn2+ accumulates with the residual MnO2 and yields spinel Mn3O4 (hausmannite), as shown in the following equations:
| 2MnOOH → MnO2 + Mn2+ + 2OH− |
| MnO2 + 2Mn2+ + 2H2O ↔ Mn3O4 + 4H+ |
 | ||
| Fig. 7 Ex situ XRD images of the (a) pure δ-MnO2, (b) Fe-doped δ-MnO2, (c) Mo-doped δ-MnO2 and (d) Rh-doped δ-MnO2 electrodes. | ||
Additionally, the diffraction peaks of ZnMn2O4 (JCPDS card no. 024-1133) can be seen, which is due to the Zn2+ intercalation into the δ-MnO2 layer during the discharge process. Fig. 7b elucidates the ex situ XRD of the Fe-doped δ-MnO2 cathode, which was conducted under the same parameters as that of pure δ-MnO2. It can be seen that all of the peaks matched very well with ZnMn2O4 (JCPDS-24-1133), which clearly confirmed the reversible insertion/de-insertion of Zn2+ ions into the δ-MnO2 cathode. It is believed that the layered structure of the δ-MnO2 cathode transformed into the spinel phase of ZnMn2O4 upon Zn2+ insertion/de-insertion reactions.18Fig. 7c represents the ex situ XRD data of the Mo-doped δ-MnO2 electrode, which clearly exhibit sharp crystalline peaks corresponding to MoMnO4 (JCPDS-01-078-0221), along with a few peaks from ZnMn2O4. This confirms the reversible insertion and de-insertion of H+/Zn2+ ions into the δ-MnO2 cathode, followed by its conversion into the spinel phase of MnMoO4. During discharge, the partial insertion of H+ into MnO2 takes place, resulting in the formation of MnOOH. During charging, this H+ is extracted. Meanwhile, the presence of Mo in the MnO2 lattice promotes the 2-electron MnO2 ↔ Mn2+ redox chemistry, enhancing the capacity by facilitating lattice oxygen redox and defect chemistry.41 During discharge, MnO2 generates Mn2+ ions in the electrolyte via dissolution. Meanwhile, Mo from the doped lattice exists as MoO42− (Mo6+). When Mn2+ encounters MoO42−, insoluble manganese molybdate (MoMnO4) precipitates on the cathode surface. The process is favoured toward the end of discharge, as the concentration of Mn2+ ions is highest during discharge.
| 2MnOOH → MnO2 + Mn2+ + 2OH− |
| Mn2+ + MoO42− → MnMoO4 |
The ex situ XRD result of the Rh-doped δ-MnO2 cathode is shown in Fig. 7d. It can be seen that the diffraction peaks that appear at 18.0 and 32.9 can be well indexed with the spinel ZnMn2O4 (JCPDS No. 024-1133), which confirmed the reversible insertion/extraction of Zn2+. Furthermore, the rest of the reflections mainly belong to Zn4SO4(OH)6·5H2O (JCPDS No. 039-0688). The partial hydrolysis of Zn2+ under conditions of high local Zn2+ concentration at the electrode/electrolyte interface can further contribute to the OH− availability. Once the OH− concentration exceeds the solubility threshold in the presence of Zn2+ and SO42−, Zn4SO4(OH)6·5H2O precipitates as per the following equations: 31
| 2MnO2 + Zn2+ + 2e− → ZnMn2O4 |
| 4Zn2+ + SO42− + 6OH− + 2e− + 5H2O → Zn4SO4(OH)6·5H2O |
During discharge, Zn2+ ions intercalate into the layered Rh-doped δ-MnO2 structure, acting as support pillars, followed by the deposition of Zn4SO4(OH)6·5H2O, which is the main reason for the capacity fading observed in this electrode.24,40
In addition, ex situ FE-SEM analysis has been conducted on cycled electrodes to monitor the morphological evolution of all four cathodes, as demonstrated in Fig. 8a–d. To perform this analysis, the half-cells were initially dismantled and thoroughly washed with DI and ethanol to remove electrolyte salt on the electrode surfaces. After drying the electrodes in a vacuum oven, the material was scraped off from the current collector for recording the FE-SEM images. The pure δ-MnO2 electrode (Fig. 8a) exhibits a distinct transformation from the original nanoflower-like morphology into interconnected, thin nanoflakes after repeated cycling. This morphological change is consistent with ex situ XRD results, which reveal the formation of a Mn3O4 spinel phase. The emergence of Mn3O4 suggests that the structural conversion of δ-MnO2 into a spinel framework underlies the observed textural reconstruction. More importantly, the obtained nanoflakes are very-well knitted even after prolonged cycling, which indicates that the electrochemical reactions are reversible in the pure electrode. In contrast, Fig. 8b displays the ex situ image of Fe3+-doped δ-MnO2 sample, which clearly exhibits retention of its hierarchical nanoflower-like morphology even after prolonged cycling, highlighting its structural robustness. As discussed above, ex situ XRD confirmed the presence of ZnMn2O4 in the discharged state, which arises from a Zn2+ insertion-driven conversion process. The original morphological framework of the Fe-doped sample is nearly preserved, indicating that Fe-doping effectively stabilizes the δ-MnO2 host lattice and mitigates severe morphological degradation, thereby promoting reversible Zn2+ intercalation and enhancing the cycling stability. For the Mo-doped δ-MnO2 electrode (Fig. 8c), a more pronounced morphology change is observed with an increase of the electrochemical cycling: the initial hawthorn-like structures transform into open, crumpled nanosheets, accompanied with nanoparticles. Ex situ XRD confirms the formation of MnMoO4, indicating a dissolution–reconstruction pathway in which Mn2+ is released during discharge and reacts with the molybdate species to nucleate MnMoO4. The resultant nanosheet morphology is therefore consistent with the newly formed phase, explaining both the structural transformation and the altered surface chemistry of the electrode. Finally, the Rh3+-doped δ-MnO2 electrode (Fig. 8d) shows the most severe structural degradation with the original morphology completely replaced by broad, flake-like sheets after cycling. Ex situ XRD identifies these nanoflakes as Zn(SO4)(OH)6·5H2O (ZSH), formed as a result of interfacial alkalization during Zn2+/H+ (de)insertion, followed by the subsequent precipitation of Zn2+ and SO42− with OH− from the electrolyte.31 The growth of insulating ZSH layers not only masks the electrode surface, but also contributes to the significant loss of electrochemical activity and rapid capacity fading observed in the Rh-doped electrode. Overall, the combined ex situ XRD and FE-SEM results clearly demonstrate that the dopant type plays a decisive role in determining the structural reconstruction pathways of the δ-MnO2 cathode. While the pristine electrode undergoes conversion to Mn3O4, Fe-doping favours reversible Zn2+ insertion with minimal degradation. Mo doping induces phase reconstruction into MnMoO4 nanosheets, whereas Rh-doping results in the accumulation of ZSH, severely compromising the electrode stability. These findings underscore the crucial role of dopant chemistry in tailoring the phase stability and morphology of δ-MnO2-based cathodes for aqueous zinc-ion batteries. A detailed comparative table between the specific capacities of our work and previously reported δ-MnO2 electrodes is shown in the SI section (Table S3).
 | ||
| Fig. 8 Ex situ FE-SEM images of the (a) pure δ-MnO2, (b) Fe-doped δ-MnO2, (c) Mo-doped δ-MnO2 and (d) Rh-doped δ-MnO2 electrodes. | ||
3. Conclusion
Herein, we describe a systematic doping approach to suppress Jahn–Teller distortion and enhance the electrochemical performance of the δ-MnO2 cathode for rechargeable aqueous zinc-ion batteries. In fact, three dopants were purposely taken into consideration in which two of the dopants' ionic radii nearly match with that of Mn3+, which is the main cause of Jahn–Teller distortion, while the other dopant's ionic radius do not match with that of Mn3+. Since the motivation was to replace intermediate Mn3+ ions and observe the effect of the Jahn–Teller distortion on the electrochemical performances, Fe3+ (3d block), Mo6+ (4d block) and Rh3+ (4d block) metal ions have been doped on the Mn-site of the δ-MnO2 cathode. More importantly, between Fe3+ and Mo6+, the ionic radius of Mo6+ is almost identical to that of Mn3+, suggesting that the substitution of Mn3+ by Mo6+ is highly favourable. In contrast, although the ionic radius of Fe3+ is relatively close to that of Mn3+, the capacity of the Fe3+-doped δ-MnO2 cathode is expected to be moderately lower than that of the Mo6+-doped δ-MnO2. Furthermore, since the ionic radius of Rh3+ does not closely match that of Mn3+, the corresponding doped cathode is anticipated to deliver a relatively poor capacity. Although the Rh3+- and Mo6+-doped δ-MnO2 cathodes initially demonstrate superior charge capacities at 1C and 2C rates, their capacity declines progressively with increasing cycle numbers. Both Rh3+- and Mo6+-doped δ-MnO2 cathodes initially offer high charge capacities of 338.1 and 302.5 mAh g−1 at 2C, respectively, which are much higher than the initial capacities of the Fe3+-doped (91.1 mAh g−1) and pure δ-MnO2 (91.6 mAh g−1) electrodes. However, the Rh3+-doped electrode does not withstand long-term cycling and the capacity decreases to 83.03 mAh g−1 after 530 cycles. This can be mainly ascribed to the structural collapse and Jahn–Teller distortion due to the inability of Rh3+ to replace Mn3+, as both have unmatched ionic radii. In contrast, the Mo6+-doped δ-MnO2 electrode delivers the highest capacity among all tested electrodes. However, it also experiences a significant capacity decline with increasing cycle numbers, ultimately stabilizing at 155.2 mAh g−1. Meanwhile, the Fe3+-doped δ-MnO2 cathode shows remarkable stability during cycling and maintained a capacity of 100.5 mAh g−1 after 530 cycles even at high current rate of 2C, which can be credited to its ability to alleviate the Jahn–Teller distortion effect by preferentially replacing the Mn3+ ion sites due to their close ionic radii, thereby enhancing the structural integrity and stability of the electrode material. To theoretically confirm the suppression of Jahn–Teller distortion (JTD), density functional theory (DFT) calculations were performed. The results show that Fe3+ doping eliminates the JTD effect and renders the axial and equatorial bond lengths similar, owing to the comparable ionic radii. In contrast, Rh3+ and Mo6+ doping fails to stabilize the geometries, leading to a complete loss of unit-cell symmetry due to the mismatch between the guest and host ionic radii. Thus, the overall findings of this work highlight the importance of elemental doping to overcome the major limitations of Mn-based cathodes for various applications.4. Experimental
4.1. Material synthesis
In a typical hydrothermal synthesis, 1.50 g of KMnO4 (Emplura, 99%, India) and 0.30 g of MnSO4·H2O (Alfa Aesar, 99%) were first dissolved separately in 50 mL and 20 mL of deionized water, respectively, using continuous stirring. The above two solutions were mixed and stirred continuously for 10 minutes. To synthesize the Fe3+, Mo6+ and Rh3+-doped δ-MnO2 cathodes, a 0.078 g (5 wt%) of ferric (III) nitrate nonahydrate (Alfa Aesar, 99%), sodium molybdate (Alfa Aesar, 99%), and rhodium(III) chloride (Sigma Aldrich, 99.98%) were separately added to the above solution. After vigorously stirring for 30 minutes, the obtained homogeneous solutions were transferred to a Teflon-lined stainless-steel autoclave and heated up to 150 °C for 14 hours. Afterwards, the resultant solid product was centrifuged and washed with deionized water and ethanol for several times to remove the organic impurities, followed by drying overnight at 75 °C. Finally, the obtained as-prepared sample was calcined at 360 °C for 2 h to obtain the doped δ-MnO2 samples. For comparison, the pristine δ-MnO2 cathode was also prepared using the same process, except with the addition of the dopant precursors (rhodium, molybdenum and iron).4.2 Material characterisation
To confirm the crystallinity and single-phase formation of the calcined sample, the extreme conditions X-ray diffraction (ECXRD) beamline at the Indus-2 (2 gigabyte) synchrotron source in RRCAT, Indore was used. Detailed microstructural studies on the surface morphology, particle size, and identification of the lattice fringes of the samples were carried out by field emission-scanning electron microscopy (FE-SEM, Zeiss Gemini SEM 500) and field emission-transmission electron microscopy (FE-TEM, Philips TecnaiF20 at 200 keV). Inductively coupled plasma (ICP) analysis was carried out using the Agilent 8900 Triple Quadrupole ICP-MS. X-ray fluorescence analysis (XRF) was conducted with a Bruker-made portable Benchtop/countertop analyzer. The surface area and porosity analysis of the samples were measured by Brunauer–Emmett–Teller (BET) using the nitrogen adsorption/desorption isotherm (BELSORP-maxII (BELSORP-series) from Microtrac BEL Corp). Additionally, X-ray photoelectron spectroscopy (XPS) was conducted using the angle-integrated beamline at the Indus 1 (450 Mega Electron Volt) synchrotron source, India to analyze the surface elemental states and chemical composition.4.3 Electrochemical measurements
To test the electrochemical performances of the pure and doped δ-MnO2 electrodes, the working electrodes were fabricated by mixing the active materials, carbon black and polyvinylidene fluoride with a weight ratio of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20
20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, using N-methyl 2-pyrrolidone as the common solvent. Then, the obtained uniform mixture slurry was directly drop-cast onto the spacer (current collector) using a micro pipette, followed by heating on a hot plate at 100 °C and vacuum drying overnight. The 2032 coin-type half-cells were assembled using zinc metal foil as the reference electrode, glass fibre as a separator, and 2 M ZnSO4 + 0.1 M MnSO4 solution as the electrolyte in an open-air environment. Before the electrochemical measurements, the cells were kept for aging. Cyclic voltammetry and the charge/discharge measurements were performed using the CH-Instruments electrochemical workstation and Neware battery tester (China), respectively, within the potential range of 0.6–1.8 V at constant and different current densities.
10, using N-methyl 2-pyrrolidone as the common solvent. Then, the obtained uniform mixture slurry was directly drop-cast onto the spacer (current collector) using a micro pipette, followed by heating on a hot plate at 100 °C and vacuum drying overnight. The 2032 coin-type half-cells were assembled using zinc metal foil as the reference electrode, glass fibre as a separator, and 2 M ZnSO4 + 0.1 M MnSO4 solution as the electrolyte in an open-air environment. Before the electrochemical measurements, the cells were kept for aging. Cyclic voltammetry and the charge/discharge measurements were performed using the CH-Instruments electrochemical workstation and Neware battery tester (China), respectively, within the potential range of 0.6–1.8 V at constant and different current densities.
Author contributions
Conceptualization, P. Y. and A. K. R.; methodology, P. Y. and P.; software, A. K., S. P. S., Arun K. R., P. J. T and A. K. G.; validation, A. K. R.; formal analysis, P. Y. and A. K. R.; investigation, P. Y. and P.; resources, Arun K. R., P. J. T and A. K. G.; data curation, P. Y. and A. K.; writing – original draft preparation, P. Y.; writing – review and editing, A. K. R.; visualization, S. A. H.; supervision, A. K. R.; project administration, A. K. R.; funding acquisition, A. K. R. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
All data generated or examined in this study are included in this article. The datasets used in the current study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta07864d.
Acknowledgements
One of the authors (Priya Yadav) would like to thank CSIR, New Delhi, for the award of the Junior Research Fellowship (09/0045(11332)/2021-EMR-I). This work was mainly supported by the Faculty Research Programme Grant (Ref. No./IoE/2024-25/12/FRP), sanctioned by the Institution of Eminence, University of Delhi, and partially supported by the Council of Scientific and Industrial Research (CSIR) Research Grant (File No. 01(3015)/21/EMR-II).References
- S. Islam, M. H. Alfaruqi, V. Mathew, J. Song, S. Kim, S. Kim, J. Jo, J. P. Baboo, D. T. Pham, D. Y. Putro, Y. Sun and J. Kim, J. Mater. Chem. A, 2017, 5, 23299–23309 RSC.
- W. Li, J. R. Dahn and D. S. Wainwright, Science, 1994, 264, 1115–1118 CrossRef CAS PubMed.
- T. Leisegang, F. Meutzner, M. Zschornak, W. Münchgesang, R. Schmid, T. Nestler, R. A. Eremin, A. A. Kabanov, V. A. Blatov and D. C. Meyer, Front. Chem., 2019, 7, 268 Search PubMed.
- M. Mokhtar, M. Z. M. Talib, E. H. Majlan, S. M. Tasirin, W. M. F. W. Ramli, W. R. W. Daud and J. Sahari, J. Ind. Eng. Chem., 2015, 32, 1–20 CrossRef CAS.
- W. Lao-atiman, T. Julaphatachote, P. Boonmongkolras and S. Kheawhom, J. Electrochem. Soc., 2017, 164, A859–A863 CrossRef CAS.
- H. Wang, Y. Zeng, K. Huang, S. Liu and L. Chen, Electrochim. Acta, 2007, 52, 5102–5107 CrossRef CAS.
- M. H. Alfaruqi, J. Gim, S. Kim, J. Song, J. Jo, S. Kim, V. Mathew and J. Kim, J. Power Sources, 2015, 288, 320–327 CrossRef CAS.
- C. Guo, H. Liu, J. Li, Z. Hou, J. Liang, J. Zhou, Y. Zhu and Y. Qian, Electrochim. Acta, 2019, 304, 370–377 CrossRef CAS.
- S. Hosseini, A. Abbasi, L. O. Uginet, N. Haustraete, S. Praserthdam, T. Yonezawa and S. Kheawhom, Sci. Rep., 2019, 9, 14958 CrossRef.
- S. D. Han, S. Kim, D. Li, V. Petkov, H. D. Yoo, P. J. Phillips, H. Wang, J. J. Kim, K. L. More, B. Key, R. F. Klie, J. Cabana, V. R. Stamenkovic, T. T. Fister, N. M. Markovic, A. K. Burrell, S. Tepavcevic and J. T. Vaughey, Chem. Mater., 2017, 29, 4874–4884 CrossRef CAS.
- C. Guo, Q. Zhou, H. Liu, S. Tian, B. Chen, J. Zhao and J. Li, Electrochim. Acta, 2019, 324, 134867 Search PubMed.
- D. Liu, M. Song, F. Xu, A. Wang, C. Xu, J. Zheng, C. Yun, B. Zhang, Z. Li, Y. Liu, P. Wang, S. Wang and J. Guo, Chem.–Eur. J., 2025, 31, e202404600 CrossRef CAS PubMed.
- F. Xu, J. Zheng, D. Liu, A. Wang, Z. Li, C. Xu, M. Song, B. Zhang, Z. Bai and Z. Chen, Inorg. Chem. Front., 2024, 11(23), 8526–8534 RSC.
- H. Tian, T. Gao, X. Li, X. Wang, C. Luo, X. Fan, C. Yang, L. Suo, Z. Ma, W. Han and C. Wang, Nat. Commun., 2017, 8, 14083 Search PubMed.
- W. Liu, X. Zhang, Y. Huang, B. Jiang, Z. Chang, C. Xu and F. Kang, J. Energy Chem., 2021, 56, 365–373 Search PubMed.
- Y. Guo, Y. Zhang and H. Lu, Battery Energy, 2022, 1, 20210014 CrossRef CAS.
- X. Li, Y. Xu and C. Wang, J. Alloys Compd., 2009, 479, 310–313 CrossRef CAS.
- P. Yadav, D. Putro, N. Kumari, J. Kim and A. K. Rai, Phys. Chem. Chem. Phys., 2023, 25, 21082–21088 RSC.
- Y. Zhang, Y. Liu, Z. Liu, X. Wu, Y. Wen, H. Chen, X. Ni, G. Li, J. Huang and S. Peng, J. Energy Chem., 2022, 64, 23–32 CrossRef CAS.
- T. Li, N. Zhang, B. Liu, P. Wang, Z. Liu, Y. Wang, D. Xu, H. Tian, Q. Zhang and T. F. Yi, Adv. Funct. Mater., 2025, 35, 2423755 CrossRef CAS.
- Q. Gao, J. Wang, B. Ke, J. Wang and Y. Li, Ceram. Int., 2018, 44, 18770–18775 CrossRef CAS.
- T. Xiong, Z. G. Yu, H. Wu, Y. Du, Q. Xie, J. Chen, Y.-W. Zhang, S. J. Pennycook, W. S. V. Lee and J. Xue, Adv. Energy Mater., 2019, 9, 1803815 Search PubMed.
- R. Poonguzhali, N. Shanmugam, R. Gobi, A. Senthilkumar, G. Viruthagiri and N. Kannadasan, J. Power Sources, 2015, 293, 790–798 CrossRef CAS.
- D. Xie, Y. Wang, L. Tian, H. Huang, J. Sun, D. Kim, J. Zhao and J. Mao, Adv. Funct. Mater., 2025, 35, 2501135 CrossRef.
- J. Liang, Y. Zhao, L. Ren, M. Li, Q. Zhang, Y. Wang, X. Sun, M. Chuai, X. Wang and W. Liu, Adv. Funct. Mater., 2025, 2501135 CrossRef CAS.
- P. Yadav, N. Kumari and A. K. Rai, J. Power Sources, 2023, 555, 232385 CrossRef CAS.
- J. W. Xu, Q. L. Gao, Y. M. Xia, X. S. Lin, W. L. Liu, M. M. Ren, F. G. Kong, S. J. Wang and C. Lin, J. Colloid Interface Sci., 2021, 598, 419–429 CrossRef CAS PubMed.
- D. K. Bhat, H. Bantawal and U. S. Shenoy, Nanoscale Adv., 2020, 2, 5688–5698 RSC.
- J. Rodriguez-Carvajal, FullProf; Laboratoire Léon Brillouin, CEA-CNRS CEA/Saclay: 91191, Gif sur Yvette CedexFrance, 2008 Search PubMed.
- R. Invernizzi, V. M. Kovrugin, L. Molini, A. Iadecola, M. Duttine, L. Bourgeois, J. Olchowka and L. Guerlou-Demourgues, J. Mater. Chem. A, 2024, 12, 3373–3385 RSC.
- P. Yadav, D. Putro, J. Kim and A. K. Rai, Batteries, 2023, 9, 133 CrossRef CAS.
- G. Wang, Z. Ma and G. Zhang, et. al. , Electrochim. Acta, 2015, 182, 1070–1077 CrossRef CAS.
- A. Xia, C. Zhao, W. Yu, Y. Han, J. Yi and G. Tan, J. Appl. Electrochem., 2020, 50, 733–744 CrossRef CAS.
- S. Ida, K. Sato, T. Nagata, H. Hagiwara, M. Watanabe, N. Kim, Y. Shiota, M. Koinuma, S. Takenaka, T. Sakai, E. Ertekin and T. Ishihara, Angew. Chem., Int. Ed., 2018, 57, 9073–9077 CrossRef CAS PubMed.
- M. Toupin, T. Brousse and D. Belanger, Chem. Mater., 2004, 16, 3184–3190 CrossRef CAS.
- Q. Gao, J. Wang, B. Ke, J. Wang and Y. Li, Ceram. Int., 2018, 44, 18770–18775 CrossRef CAS.
- Z. Li, Z. Ye, G. Liu, Z. Jiang, W. Jiang, J. Luo, S. Liu and H. Hu, Batteries Supercaps, 2024, e202400660 Search PubMed.
- J. Wang, J. G. Wang, H. Liu, C. Wei and F. Kang, J. Mater. Chem. A, 2019, 7, 13727–13735 RSC.
- J. Singh, P. Yadav, R. Nagarajan, J. Kim and A. K. Rai, ACS Appl. Nano Mater., 2023, 6, 17748–17759 CrossRef CAS.
- F. Ji, J. Yu, S. Hou, J. Hu and S. Li, Materials, 2024, 17(13), 3327 Search PubMed.
- X. Xia, Y. Zhao, Y. Zhao, M. Xu, W. Liu and X. Sun, Nano Res., 2023, 16(2), 2511–2518 CrossRef CAS.
- R. D. Corpuz, L. M. Z. D. Juan, S. Praserthdam, R. Pornprasertsuk, T. Yonezawa, M. Thanhnguyen and S. Khawhom, Sci. Rep., 2019, 9, 15107 CrossRef.
- D. Gao, T. Zhu, L. Yu, P. Chen, C. Li and Y. Liu, IOP Conf. Ser. Earth Environ. Sci., 2021, 844, 012007 Search PubMed.
- S. Khamsanga, R. Pornprasertsuk, T. Yonezawa, A. A. Mohamad and S. Kheawhom, Sci. Rep., 2019, 9, 8441 CrossRef PubMed.
- S. Khamsanga, M. T. Nguyen, T. Yonezawa, P. Thamyongkit, R. Pornprasertsuk, P. Pattananuwat, A. Tuantranont, S. Siwamogsatham and S. Kheawhom, Int. J. Mol. Sci., 2020, 21, 4689 CrossRef CAS.
- P. Wang, H. Yu, C. Zou, Y. Wu and Z. Chen, Solids, 2025, 6, 45 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, and D. J. Fox, Gaussian 16, Revision B.01, Gaussian, Inc., Wallingford CT, 2016 Search PubMed.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |