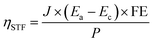Activating SnOx-coated Cu2O photocathodes for efficient photoelectrochemical CO2 reduction and unassisted tandem device integration
Lili
Wan†
ab,
Linxiao
Wu†
a,
Yumeng
Han
a,
Fusong
Kang
a,
Dongfeng
Du
a,
Jinshui
Cheng
a and
Jingshan
Luo
 *acd
*acd
aInstitute of Photoelectronic Thin Film Devices and Technology, State Key Laboratory of Photovoltaic Materials and Cells, Tianjin Key Laboratory of Efficient Solar Energy Utilization, Ministry of Education Engineering Research Center of Thin Film Photoelectronic Technology, Nankai University, Tianjin 300350, China. E-mail: jingshan.luo@nankai.edu.cn
bInner Mongolia Key Laboratory of Rare Earth Catalysis, School of Chemistry and Chemical Engineering, Inner Mongolia University, 235 West College Road, Hohhot, 010021, Inner Mongolia, China
cFrontiers Science Center for New Organic Matter, Nankai University, Tianjin 300071, China
dAcademy for Advanced Interdisciplinary Studies, Nankai University, Tianjin 300071, China
First published on 21st November 2025
Abstract
Efficient and selective photoelectrochemical CO2 reduction remains a significant challenge for solar fuel production. Here, we fabricated a heterostructured Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 photocathode featuring a pristine SnO2 layer deposited via atomic layer deposition. Upon photoelectrochemical activation, the SnO2 layer transformed into uniformly distributed SnOx nanoparticles across the photocathode surface. Systematic characterization revealed the SnO2 layer's structural evolution. The Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode significantly enhanced the charge transfer characteristics and PEC CO2 reduction performance, delivering a current density of −2.5 mA cm−2 at −0.2 V vs. RHE and a formate faradaic efficiency of 75.4% at −0.1 V vs. RHE. Coupling with a semitransparent BiVO4/NiCo LDH photoanode enables unassisted, solar-driven CO2 reduction and ethylene glycol (EG) oxidation, achieving a current density of 0.11 mA cm−2 and producing 0.13 µmol cm−2 formate yield in the first 0.5 hour. This work presents a scalable and earth-abundant material-based strategy for efficient PEC CO2 reduction and underscores the potential of tandem architectures for sustainable solar fuel generation.
1. Introduction
The production of solar fuels from CO2 and water with sunlight, often termed artificial photosynthesis, represents a promising and innovative approach to address global energy and environmental challenges.1,2 Photoelectrochemical (PEC) CO2 reduction (CO2R) utilizes photoelectrodes to harness solar energy directly, driving CO2 reduction reactions through photo-generated electrons and converting CO2 into valuable carbon-based fuels or chemcials.3,4 However, photocathodes used in PEC-CO2R systems face key limitations, including low energy efficiencies and poor product selectivity.5,6The requirement for the conduction band level to be higher than the equilibrium potential of CO2 reduction limits the number of semiconductors suitable for CO2 conversion.7 Prominent examples include p-GaP,8 p-InP,9 p-Si10–12 and p-Cu2O.13,14 Among them, p-Cu2O, a metal oxide semiconductor with a band gap of approximately 2.0 eV, offers a theoretical photovoltage of 1.6 V and a theoretical photocurrent density of −14.7 mA cm−2 under the AM 1.5 G illumination, rendering it an attractive choice for PEC-CO2R.15 Highly efficient heterostructure Cu2O photocathodes for the PEC hydrogen evolution reaction (HER) have been developed with a buried p–n junction, consisting of the Cu2O photo-absorber, buffer layer and protection layer.16–18 However, the application of such heterostructures in CO2R remains underexplored.
Grätzel and co-workers demonstrated PEC CO2 reduction using a Cu2O-based heterostructure photocathode modified with an electrochemically deposited Sn/SnOx catalyst layer, achieving faradaic efficiencies (FEs) of 42.9% for H2, 22.7% for CO, and 34.4% for formate.19 Despite this progress, two major challenges hinder the practical implementation of Cu2O-based PEC-CO2R systems: (1) the thermodynamic competition between HER and CO2 reduction pathways, and (2) inadequate control over product selectivity. Advancing the viability of these systems requires strategic improvements in charge carrier dynamics and rational interface engineering to enhance catalytic selectivity.
Beyond photocathode development, constructing a solar-driven, unassisted PEC device necessitates pairing with a suitable photoanode or dark anode for the oxidation half-reaction.20,21 However, water oxidation suffers from sluggish kinetics. In contrast, ethylene glycol (EG) offers a favorable alternative, with a significantly lower oxidation potential (∼0.2 V vs. RHE),22,23 thereby reducing the anodic energy barrier and facilitating overall device operation.
In this study, we developed a heterostructure Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 photocathode featuring the SnO2 pristine catalyst layer deposited by atomic layer deposition (ALD). After activation, the homogeneous SnO2 pristine catalyst layer transformed into SnOx, resulting in a densely and uniformly dispersed nanoparticle layer across the photoelectrode surface. The structural evolution of the SnO2 layer and its structure–property correlation with PEC performance were unambiguously elucidated. The Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode demonstrated superior charge transfer characteristics and enhanced photoelectrochemical performance, delivering a current density of −2.5 mA cm−2 at −0.2 V vs. RHE. Notably, its FE of formate reached 75.4% at −0.1 vs. RHE.
Furthermore, we integrated the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode with a semi-transparent BiVO4 photoanode to construct a tandem PEC device. This system achieved a solar-to-fuel (STF) efficiency of 0.074% for the co-production of formate, CO, and H2. Operating in a mixed electrolyte of 1 M KHCO3 and 1 M EG under AM 1.5 G illumination, CO2 reduction to formate on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode and EG oxidation to formate on the BiVO4 photoanode occurred simultaneously. The device sustained an average current density of 0.11 mA cm−2 and yielded 0.13 µmol cm−2 of formate within the first 0.5 hour. These results underscore the potential of unassisted all-oxide tandem Cu2O-BiVO4 PEC devices for efficient, solar-driven CO2 conversion and sustainable valuable chemical production.
2. Results and discussion
The heterostructure Cu2O/Ga2O3/ZnGeOx/TiO2 photocathodes without a catalyst layer were fabricated according to the previous report (refer to the SI for detailed information).17 Then the SnO2 pre-catalyst layer was deposited onto it by atomic layer deposition (ALD). The morphology and structure of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathodes were characterized by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). As illustrated in Fig. 1a, the distinct structure of the assembled cubic particles of Cu2O was obscured by multiple overlayers. TEM imaging of focused ion beam (FIB)-sliced cross-sections and corresponding energy-dispersive X-ray (EDX) mapping confirmed a well-defined layered architecture (Fig. 1b–d, S1a–i and S2). The SnO2 film conformed well to the Cu2O photocathode surface, and its thickness was precisely controlled by the number of ALD cycles. Unless otherwise specified, the “Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 photocathode” referred to a sample with a 60 nm SnO2 layer.The SnO2 layer on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathodes functioned as a pre-catalyst layer, requiring additional activation. To activate it, 10 cycles of linear sweep voltammetry (LSV) were conducted over a potential range of 0.3–0 V vs. RHE in CO2-saturated 1 M KHCO3 under AM 1.5 G light illumination. As illustrated in Fig. S3, small nanoparticles emerged on the surface of the Cu2O photocathode after two LSV cycles, and their size gradually increased to an average of 100–200 nm as the number of LSV cycles increased. During the LSV treatment, the SnO2 layer was reduced and reconstructed into nanoparticles, which became densely and uniformly dispersed across the Cu2O photocathode surface (Fig. 1e). FIB-TEM images and corresponding EDX mapping demonstrated that the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode retained the same well-defined layer structure as the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode (Fig. 1f, j and S1j–r), but the continuous SnO2 layer had transformed into nanoparticles after activation (Fig. 1g and h). The TEM images further demonstrated that these nanoparticles were evenly dispersed on the surface of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode (Fig. 1i).
Grazing incidence X-ray diffraction (GIXRD) was performed to evaluate structural changes. Diffraction peaks observed in the pattern of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode at 0.1° incident angle were attributed to metallic Sn (Fig. S4). However, no peaks appeared in the pattern of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode under the same test conditions, as the SnO2 synthesized via ALD was amorphous in nature. When the incident angle increased to 1°, diffraction peaks corresponding to Au and Cu2O were present in both samples, but metallic Sn peaks were only observed in the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode (Fig. 1l). Furthermore, the lattice spacing of the nanoparticles on the photocathode, as shown in the HR-TEM image, was consistent with the (101) and (200) planes of metallic Sn (Fig. 1k). X-ray photoelectron spectroscopy (XPS) was carried out to confirm the chemical states of Sn. In the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode, only Sn4+ was detected, while in the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode, Sn existed in the metallic Sn0, Sn2+ and Sn4+ states (Fig. 1m). Consequently, the SnO2 layer originally present on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode underwent transformation into mixed-valence SnOx (x = 0, +2 or +4) following photocathode activation. As demonstrated by Mayer et al., SnO2 retains its characteristic oxidation states even at applied potentials as negative as −0.9 V vs. RHE.24 The Sn2+ and Sn4+ species detected on the activated Cu2O photocathode originated partly from residual unreduced SnO2 and partly from the atmospheric oxidation of metallic Sn during air exposure. The full coverage of 60 nm SnO2 on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode surface masked the underlying TiO2 layer, preventing detection of the Ti 2p peak in the XPS spectrum (Fig. 1n). After activation, the continuous SnO2 layer transformed into discrete nanoparticles, exposing the underlying TiO2 layer and resulting in a clear Ti 2p peak corresponding to Ti4+ (Fig. 1n).
Kelvin probe force microscopy (KPFM) was employed to spatially resolve the surface potential of the photocathode samples under dark and illuminated conditions. Atomic force microscopy (AFM) images of the pristine and activated Cu2O photocathodes revealed a textured surface with smooth corners, and further revealed the presence of small nanoparticles on the surface of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode (Fig. 2a and d), confirming the findings from SEM analysis. The average contact potential difference under dark and light conditions ranged from 250 to 312 mV on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode, whereas it increased from 170 to 589 mV on Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathodes (Fig. 2b, c, e and f). The 6.7-fold increase in the contact potential difference on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode compared to the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode indicated more efficient charge separation and transfer on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode under light illumination. Interestingly, the contact potential difference of the valleys on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode was higher than that of the peaks. In contrast, the contact potential difference of valleys was significantly lower than that of the SnOx nanoparticles on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode under light illumination conditions, suggesting that electrons accumulate in the valleys of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode and on the SnOx nanoparticles of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode (Fig. 2g and h). Photoelectrochemical impedance spectroscopy (PEIS) measurements performed at 0.1 V vs. RHE in CO2 saturated 1 M KHCO3 under AM 1.5 G light illumination revealed a reduced semicircle diameter for the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode compared to the pristine one (Fig. S5), indicating improved charge transfer efficiency and enhanced carrier separation capability in the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode. In summary, the SnOx nanoparticles on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode enhance charge separation and facilitate electron transfer to the photocathode surface.
The performance of PEC CO2 reduction on Cu2O photocathodes was evaluated in an H-type two-chamber cell using a three-electrode configuration (Fig. 3a). The LSV curves of pristine and activated Cu2O photocathodes, measured in CO2-saturated 1 M KHCO3 under chopped-light illumination, are shown in Fig. 3b. The Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode exhibited an onset potential of 0.4 V vs. RHE and a plateau photocurrent density of −2.5 mA cm−2, better than the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode. The Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode showed significant degradation in CO2 reduction performance, yielding a FE for formate of only 33.0–41.6% and a total FE of 65.6–68.0% at 0–0.1 V vs. RHE (Fig. S6a). Under these applied potentials, two main reactions occurred on the Cu2O/Ga2O3/ZnGeOx/TiO2/SnO2 (pristine) photocathode: the reduction of SnO2 and the CO2 reduction reaction. The competition between these reactions for available electrons led to the low overall product FE. In contrast, the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode exhibited enhanced CO2 reduction performance, with the FE for H2 as low as ∼10%, while the formate FE reached 67.8–75.4% across a broad potential range of −0.1 to 0.2 V versus RHE (Fig. 3c). The production rate of formate reached 28 µmol cm−1 h−1, with an average current density of −1.7 mA cm−2 at −0.1 V vs. RHE (Fig. 3d). Chronoamperometry measurements revealed a stark contrast in stability: the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathodes showed a notable stability (Fig. 3e), whereas the pristine one degraded at 0 V and 0.1 V vs. RHE (Fig. S6b). We ascribe this instability to the sparse dispersion of SnOx nanoparticles on the photocathode surface (Fig. S7), a feature particularly pronounced at 0 V vs. RHE and distinct from the activated samples (Fig. S12), as it likely impeded efficient electron transport. The efficiency of incident photon-to-current efficiency (IPCE) of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode reached 33% under 375 nm light illumination, with the integrated current density of approximately 2.25 mA cm−2 over the whole spectrum range.
The Cu2O photocathodes with varying SnO2 layer thicknesses were further investigated. Samples with 20 nm SnO2 and 40 nm SnO2 were denoted as Cu2O-20 and Cu2O-40 photocathodes, respectively. The Cu2O photocathode and activated Cu2O photocathode represented the samples with 60 nm SnO2 and derived from 60 nm SnO2, respectively. The LSV curves for Cu2O photocathodes during activation are illustrated in Fig. S8. Compared with Cu2O-20 and Cu2O-40 photocathodes, the Cu2O photocathode exhibited a more positive onset potential, higher current density and increased fill factor, suggesting higher activity in the PEC CO2 reduction reaction. LSV measurements of both pristine and activated photocathodes with different SnO2 thicknesses further demonstrated that the 60 nm SnO2 layer led to higher catalytic activity (Fig. S9a, b and 3b). The PEC CO2 reduction performance of activated Cu2O-20 and Cu2O-40 photocathodes was assessed under the same test conditions as the activated Cu2O photocathode, with FE analysis confirming the superior activity of the activated Cu2O photocathode over its counterparts (Fig. S10). SEM analysis revealed that increasing SnO2 thickness correlated with enlarged dimensions and higher population density of SnOx nanoparticles on activated photocathodes (Fig. S11a, b and 1e). However, the SnOx nanoparticles on the activated Cu2O photocathode were more densely dispersed. The KPFM results identify these SnOx nanoparticles as essential electron transfer sites, thus SnO2 thickness consequently determines the electron transfer capability and the resultant CO2 reduction performance. SEM imaging combined with EDX analysis demonstrated that the activated Cu2O photocathode retained enhanced structural stability after 1800 s of testing in CO2-saturated 1 M KHCO3, likely attributed to the improved retention of SnOx particles on its surface compared to Cu2O-20 and Cu2O-40 counterparts (Fig. S9c, d, S11c, d, S12 and Table S1).
To identify the active phase of Sn on the photocathode, the Sn XPS spectrum of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photoelectrode after CO2 reduction testing was collected. As shown in Fig. S13, Sn exhibits a mixed phase of metallic Sn0, Sn2+, and Sn4+ species, a phase composition similar to that of the fresh Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode (Fig. 1m). The previously reported studies systematically indicated that Sn–O (Snδ+) species are the active sites for formate production.25,26 Mayer et al. also confirmed the persistence of SnOδ (δ = +2 or +4) even at applied potentials as negative as −0.9 V vs. RHE by in situ X-ray absorption spectroscopy.24 Therefore, we conclude that the Snδ+ species is the active phase for the formate production in our work.
A prolonged stability test was conducted on the activated Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx photocathode at 0 V vs. RHE. As shown in Fig. S14, the current density retained about 77% of its initial value after 1800 s but decreased to approximately 38% by 3600 s. To investigate the origin of this performance degradation, we performed SEM and XRD characterization on the sample after the 3600 s test. The SEM image (Fig. S15) shows that the SnOx nanoparticles became sparsely dispersed on the photocathode surface, indicating a substantial loss of active sites essential for electron transfer and CO2 reduction. This observation was further corroborated by the XRD results (Fig. S16), in which the characteristic peaks corresponding to Sn species became nearly negligible after 3600 s of operation. Consequently, we attribute the performance degradation primarily to the loss of SnOx species.
For unassisted solar-driven PEC operation, the Cu2O photocathode was paired with a semitransparent BiVO4/NiCo LDH photoanode27 (Fig. 4a). To match the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode of CO2 reduction, the semitransparent BiVO4 photoanode decorated with NiCo LDH catalysts (BiVO4/NiCo LDH) was prepared for the anodic reaction (Fig. S17). The BiVO4/NiCo LDH photoanode was first investigated for its potential to drive water oxidation. The LSV curves of these two photoelectrodes were collected using three-electrode configuration in CO2-saturated 1 M KHCO3. The BiVO4/NiCo LDH photoanode was back-illuminated during LSV testing, while the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode was positioned posterior to the photoanode and irradiated by transmitted light passing through the BiVO4/NiCo LDH (Fig. 4a). As shown in Fig. 4b, the BiVO4/NiCo LDH photoanode exhibited a positive onset potential and sluggish current rise in LSV, indicating inefficient water oxidation kinetics. The LSV curves of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode and BiVO4/NiCo LDH photoanode intersected at a point characterized by very low current density and high potential, highlighting an energy-mismatched operating point in the tandem system. Under AM 1.5 G light illumination, the Cu2O-BiVO4 tandem device operated in the CO2-saturated 1 M KHCO3 electrolyte for 1 h, exhibiting an average current density of 0.03 mA cm−2 (Fig. 4c). The FE of formate reached 31.1% during the initial 0.5 h. However, formate production became negligible in the latter half hour, with hydrogen evolution dominating the product distribution (Fig. 4b and S18). The solar-to-fuel (STF) efficiency for the first half hour was calculated to be 0.022%, 0.029%, and 0.078% accounting for formate alone, formate combined with CO, and the total sum of formate/CO/H2 products, respectively (Fig. 4d).
To lower the energy consumption of the anodic reaction, EG oxidation was further examined as a substitute for water oxidation. The LSV curves of the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode for CO2 reduction and BiVO4/NiCo LDH photoanode for EG oxidation were collected in a CO2-saturated mixture of 1 M KHCO3 and 1 M EG. The LSV curve of the BiVO4/NiCo-LDH photoanode exhibited a more negative onset potential and a significantly steeper current density increase during ethylene glycol (EG) oxidation compared to water oxidation, demonstrating its superior catalytic activity toward EG oxidation (Fig. 4e). The LSV curves intersected at 0.3 V vs. RHE (0.16 mA cm−2), demonstrating the potential of the Cu2O-BiVO4 tandem device for co-driven CO2 reduction and EG oxidation. The EG oxidation activity of the BiVO4/NiCo LDH photoanode was further measured in a CO2-saturated mixture of 1 M KHCO3 and 1 M EG in a H-type two-chamber cell with three-electrode configuration. The FEs of formate from EG oxidation were 9.4% and 11.0% at 0.3 V and 0.4 V vs. RHE, respectively (Fig. S19a), accompanied by relatively stable current density (Fig. S19b). As reported by Yan et al., the electrooxidation of EG exhibits high formate selectivity in alkaline media, whereas glycolaldehyde becomes the dominant product under weakly alkaline to neutral conditions.28 Specifically, at pH = 8, the FE of glycolaldehyde exceeds 60%, while the FE for formate drops below 10%. In this work, 1 M KHCO3 was adopted as the electrolyte, which under CO2-saturated conditions reaches a pH of approximately 7.8. This explains why the BiVO4/NiCo LDH anode exhibited a low formate FE of only about 10%. The FE of formate for the Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode achieved 48.2% at 0.3 V vs. RHE in a mixture of 1 M KHCO3 and 1 M EG (Fig. S19c). The BiVO4/NiCo LDH photoanode and Cu2O/Ga2O3/ZnGeOx/TiO2/SnOx (activated) photocathode both demonstrated formate selectivity at the intersection potential of the LSV curves (0.3 V vs. RHE), confirming the feasibility of simultaneous CO2 reduction and EG oxidation in the Cu2O-BiVO4 tandem device.
The Cu2O-BiVO4 tandem device retained stable operation for three hours in a CO2-saturated mixture of 1 M KHCO3 and 1 M EG under AM 1.5 G light illumination, yielding a time-dependent formate production of 0.13 µmol cm−2 within the initial 0.5 h and a cumulative yield of 0.34 µmol cm−2 by the end of the test (Fig. 4f). The average current density of the tandem device was 0.105 mA cm−2 during the first 0.5 hours of operation and stabilized at 0.071 mA cm−2 over 3 hours. As shown in Fig. S20, the FE of H2 progressively increased, which may indicate catalyst degradation. The Cu2O-BiVO4 PEC tandem device successfully demonstrated the feasibility of driving CO2 reduction and EG oxidation for the production of formate under solar illumination (Fig. 4a). However, further improvements are required to enhance the device's efficiency and stability.
3. Conclusions
We developed a heterostructure Cu2O photocathode with an ALD-deposited SnO2 overlayer that transforms into uniformly distributed SnOx nanoparticles upon activation. This structural evolution enhances interfacial charge transfer and boosts CO2 reduction performance, achieving −2.5 mA cm−2 at −0.2 V vs. RHE and a 75.4% formate faradaic efficiency at −0.1 V vs. RHE in CO2-saturated 1 M KHCO3. Integration with a semitransparent BiVO4/NiCo-LDH photoanode enabled an unassisted tandem PEC device with 0.074% solar-to-fuel efficiency. In a KHCO3/EG electrolyte, the system produced 0.13 µmol cm−2 of formate in 0.5 h under AM 1.5 G illumination, coupling CO2 reduction with ethylene glycol oxidation. This work highlights a scalable strategy for activating earth-abundant photocathodes and establishes a viable tandem platform for solar-driven CO2 utilization and value-added chemical synthesis.4. Experimental
4.1. Preparation of the activated Cu2O photocathodes
The Cu2O-x samples were pre-activated by running 10 cycles of linear sweep voltammetry (LSV) at a scan rate of 10 mV s−1 in the potential range of 0.3 V vs. RHE to 0 V vs. RHE in CO2-saturated 1 M KHCO3 (99.99% metal basis, Aladdin) under AM 1.5 G light illumination. The LSV measurements were performed with a scan rate of 10 mV s−1 in the range of 0.6 V vs. RHE to −0.2 V vs. RHE on the samples before and after LSV activation when necessary. The Cu2O-x samples after activation were denoted as activated Cu2O-x. The activated Cu2O photocathode represented the photocathode derived from the 60 nm SnO2 photocathode unless otherwise indicated.4.2. Materials characterization
The morphologies of the samples were imaged by SEM (JSM-7800F), and corresponding EDX spectroscopic mapping was collected when required. FIB-TEM images were acquired on an FEI Talos F200X G2 operated at a 300 kV accelerating voltage and collected corresponding EDX mapping when required. GIXRD was performed on an Aeris X-ray diffractometer (Malvern Panalytical) over a 2θ range of 20–80° at a scan rate of 10° min−1. XPS was performed on a Thermo Scientific ESCALAB 250 Xi XPS spectrometer, with an Al Ka X-ray source operating at 1486.6 eV. The spectra were obtained with an analyser pass energy of 100 eV with an energy spacing of 1 eV. AFM and KPFM measurements were performed on a Bruker Dimension Icon under dark and illumination conditions. The contact potential difference (CPD) measured by KPFM represents the difference between the probe's and the sample's surface potential.4.3. Photoelectrochemical CO2 reduction
Photoelectrochemical CO2 reduction was carried out in a two-chamber H-type cell separated by an anion exchange membrane. The activated Cu2O-x (around 0.25 cm2), platinum gauze (1 cm2) and Ag/AgCl electrode were adopted as the working electrode, counter electrode and reference electrode, respectively. 15 mL of CO2-saturated 1 M KHCO3 (pH 7.8) was used as an electrolyte for both chambers. CO2 gas was bubbled into the electrolyte with a flow rate of 10 sccm during the test continuously. A Xe lamp with a light intensity of 100 mW cm−2 illuminated the activated Cu2O-x during the chronoamperometry tests in the potential range of 0.2 to −0.1 V vs. RHE. Each test was carried out for 30 min and repeated three times.The gas products were detected using an online GC (GC9790 Plus, Fuli Instruments) equipped with two flame ionization detector (FID) and one thermal conductivity detectors (TCD), which enables the detection of hydrogen, carbon monoxide, methane, ethane and ethylene. The gaseous products were automatically sampled every 10 min. The liquid products were quantified by high performance liquid chromatography (HPLC, Agilent 1260) equipped with a refractive index detector (RID), a variable wavelength ultraviolet detector (VWD) and an Aminex HPX-87H Column (Bio-rad, 300 × 7.8 mm). The catholyte was collected after performance tests, and 900 µL catholyte and 100 µL 4.5 M H2SO4 were mixed to balance the pH of the catholyte and 5 mM H2SO4 was utilized as a carrier liquid.
Photoelectrochemical impedance spectroscopy (PEIS) was carried out on the pristine and activated Cu2O photocathode in CO2 saturated 1 M KHCO3 under light illumination conditions at 0.1 V vs. RHE. The IPCE was measured by comparing the photoresponse of the activated Cu2O photocathode and silicon diode at every 10 nm in the range of 300–700 nm under light from a xenon lamp through a monochromator (7IS151W, Newport).
4.4. Photoelectrochemical ethylene glycol oxidation
Photoelectrochemical ethylene glycol (EG) oxidation was carried out in a two-chamber H-type cell separated by an anion exchange membrane. The BiVO4/NiCo LDH (1 cm2), platinum gauze (1 cm2) and Ag/AgCl electrode were adopted as the working electrode, counter electrode and reference electrode, respectively. 15 mL of CO2 saturated 1 M KHCO3 containing 1 M EG was used as the anolyte and 1 M KHCO3 was used as the catholyte. CO2 gas was bubbled into the electrolyte with a flow rate of 10 sccm during the test continuously. A Xe lamp with a light intensity of 100 mW cm−2 illuminated the BiVO4-NiCo LDH from the backside during the chronoamperometry tests in the potential range of 0.3 to 0.4 V vs. RHE. Each test was carried out for 30 min. The anolyte was collected after each test for liquid product detection. HPLC was used to quantify the mass of formate.4.5. Construction of the solar-driven unassisted PEC device
The LSV curves for the activated Cu2O photocathode and the BiVO4/NiCo LDH photoanode were recorded in a single-chamber quartz cell. During the LSV measurement of the BiVO4/NiCo LDH photoanode, the BiVO4/NiCo LDH served as the working electrode, while a platinum gauze (1 cm2) and an Ag/AgCl electrode were employed as the counter and reference electrodes, respectively. Light illumination was applied from the backside of the BiVO4/NiCo LDH photoanode. For the LSV measurement of the activated Cu2O photocathode, a three-electrode configuration was used. The BiVO4/NiCo LDH photoanode was placed in front of the activated Cu2O to replicate the light illumination conditions of the tandem device. Light passing through the BiVO4/NiCo LDH illuminated the activated Cu2O photocathode. CO2-saturated 1 M KHCO3 was used as the electrolyte for water oxidation measurements. For the evaluation of EG oxidation, a CO2-saturated mixture of 1 M KHCO3 and 1 M EG was utilized.The activated Cu2O photocathode and BiVO4/NiCo LDH photoanode were coupled as an unassisted device for the tandem device. The activated Cu2O and BiVO4/NiCo LDH were connected using a copper conductor, and two electrodes were fixed face-to-face with a distance of 5 mm. 15 mL of CO2-saturated 1 M KHCO3 or 15 mL of a CO2-saturated mixture of 1 M KHCO3 and 1 M EG were used as the electrolyte for water oxidation and EG oxidation, respectively, and CO2 gas was continuously bubbled into the electrolyte with a flow rate of 10 sccm during the test. A Xe lamp with a light intensity of 100 mW cm−2 illuminated on the back side of BiVO4/NiCo LDH, and then the light that penetrated the photoanode further illuminated the activated Cu2O during the running process. Photocurrent was recorded using an electrochemical workstation under bias-free conditions. The electrolyte was collected every 30 min during the device running, and the liquid product was detected by HPLC. The gas products H2 and CO were detected by online GC.
4.6. Calculation of the solar-to-fuel (STF) efficiency
where J is the photocurrent density of the Cu2O-BiVO4 tandem device; Ea is the equilibrium potential of water oxidation; Eb is the equilibrium potential of CO2 reduction to formate; FE is the faradaic efficiency of formate on the activated Cu2O photocathode; P is the incident irradiance in mW cm−2.
Conflicts of interest
The authors declare no competing conflicts.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed description of systhesis method of Cu2O photocathode and semitransparent BiVO4 photoanode. SEM, TEM and correspongding EDX mapping images, XRD patterns, PEIS curves, XPS spetra, LSV and I–t curves, FE of reduction or oxidation products and tables of elements percentage. See DOI: https://doi.org/10.1039/d5ta07811c.Acknowledgements
L. W. acknowledges the funding support from the National Natural Science Foundation of China (22209081) and the start-up funding of Inner Mongolia University (No. 10000-23112101/131). J. L. acknowledges the funding support from the National Key Research and Development Program of China (Grant No. 2019YFE0123400), the National Natural Science Foundation of China (Grant No. 52072187 and 22122903), and the Tianjin Distinguished Young Scholar Fund (Grant No. 20JCJQJC00260). D. D. acknowledges the funding support from the National Natural Science Foundation of China (22309089).References
- L. Hammarstrom, Acc. Chem. Res., 2009, 42, 1859–1860 CrossRef CAS PubMed.
- Y. Zhang, B. Liu, L. Xu, Z. Ding, R. Yang and S. Wang, ChemSusChem, 2025, 18, e202401420 CrossRef CAS.
- C. Pornrungroj, V. Andrei and E. Reisner, J. Am. Chem. Soc., 2023, 145, 13709–13714 CrossRef CAS PubMed.
- I. Roh, S. Yu, C. K. Lin, S. Louisia, S. Cestellos-Blanco and P. Yang, J. Am. Chem. Soc., 2022, 144, 8002–8006 CrossRef CAS.
- M. Kan, C. Yang, Q. Wang, Q. Zhang, Y. Yan, K. Liu, A. Guan and G. Zheng, Adv. Energy Mater., 2022, 12, 2201134 CrossRef CAS.
- C. Kim, A. J. King, S. Aloni, F. M. Toma, A. Z. Weber and A. T. Bell, Energy Environ. Sci., 2023, 16, 2968–2976 RSC.
- S. Ma, S. Wang, X. Wang and S. Wang, Energy Technol., 2025, 13, 2400136 CrossRef CAS.
- M. Halmann, Nature, 1978, 275, 115–116 CrossRef CAS.
- B. A. Parkinson and P. F. Weaver, Nature, 1984, 309, 148–149 CrossRef CAS.
- S. K. Choi, U. Kang, S. Lee, D. J. Ham, S. M. Ji and H. Park, Adv. Energy Mater., 2014, 4, 1301614 CrossRef.
- Y. Hu, F. Chen, P. Ding, H. Yang, J. Chen, C. Zha and Y. Li, J. Mater. Chem. A, 2018, 6, 21906–21912 RSC.
- C. J. Sheehan, S. Suo, S. Jeon, Y. Zheng, J. Meng, F. Zhao, Z. Yang, L. Xiao, S. Venkatesan and A. S. Metlay, J. Am. Chem. Soc., 2025, 147, 9629–9639 CrossRef CAS PubMed.
- G. Liu, F. Zheng, J. Li, G. Zeng, Y. Ye, D. M. Larson, J. Yano, E. J. Crumlin, J. W. Ager and L. W. Wang, Nat. Energy, 2021, 6, 1124–1132 CrossRef CAS.
- M. Schreier, P. Gao, M. T. Mayer, J. Luo, T. Moehl, M. K. Nazeeruddin, S. D. Tilley and M. Grätzel, Energy Environ. Sci., 2015, 8, 855–861 RSC.
- R. Wick and S. D. Tilley, J. Phys. Chem. C, 2015, 119, 26243–26257 CrossRef CAS.
- J. Luo, L. Steier, M.-K. Son, M. Schreier, M. T. Mayer and M. Grätzel, Nano Lett., 2016, 16, 1848–1857 CrossRef CAS PubMed.
- J. Cheng, L. Wu and J. Luo, Nat. Commun., 2023, 14, 7228 CrossRef CAS.
- L. Pan, J. H. Kim, M. T. Mayer, M.-K. Son, A. Ummadisingu, J. S. Lee, A. Hagfeldt, J. Luo and M. Grätzel, Nat. Catal., 2018, 1, 412–420 CrossRef CAS.
- M. Xia, L. Pan, Y. Liu, J. Gao, J. Li, M. Mensi, K. Sivula, S. M. Zakeeruddin, D. Ren and M. Grätzel, J. Am. Chem. Soc., 2023, 145, 27939–27949 CrossRef CAS.
- V. Andrei, R. A. Jagt, M. Rahaman, L. Lari, V. K. Lazarov, J. L. MacManus-Driscoll, R. L. Hoye and E. Reisner, Nat. Mater., 2022, 21, 864–868 CrossRef CAS PubMed.
- M. Rahaman, C. Pulignani, M. Miller, S. Bhattacharjee, A. Bin Mohamad Annuar, R. R. Manuel, I. A. Pereira and E. Reisner, J. Am. Chem. Soc., 2025, 147, 8168–8177 CrossRef CAS.
- D. Du, F. Kang, S. Yang, B. Shao and J. Luo, Sci. China: Chem., 2024, 67, 1539–1544 CrossRef CAS.
- Y. Qin, W. Zhang, F. Wang, J. Li, J. Ye, X. Sheng, C. Li, X. Liang, P. Liu and X. Wang, Angew. Chem., Int. Ed., 2022, 61, e202200899 CrossRef CAS.
- L. C. Pardo Pérez, A. Arndt, S. Stojkovikj, I. Y. Ahmet, J. T. Arens, F. Dattila, R. Wendt, A. Guilherme Buzanich, M. Radtke, V. Davies, K. Höflich, E. Köhnen, P. Tockhorn, R. Golnak, J. Xiao, G. Schuck, M. Wollgarten, N. López and M. T. Mayer, Adv. Energy Mater., 2022, 12, 2103328 CrossRef.
- Y. Jiang, J. Shan, P. Wang, L. Huang, Y. Zheng and S. Z. Qiao, ACS Catal., 2023, 13, 3101–3108 CrossRef CAS.
- Y. Wang, G. Li, J. Feng, X. Wang, Q. Xue, A. Cheng, B. Liu, S. Zeng, M. Wang and X. Zhang, J. Am. Chem. Soc., 2025, 147, 40126–40135 CrossRef CAS PubMed.
- F. Kang, Q. Wang, D. Du, L. Wu, D. W. F. Cheung and J. Luo, Angew. Chem., Int. Ed., 2025, 64, e202417648 CrossRef CAS.
- X. Jiang, K. Zhao, H. Feng, L. Ke, X. Wang, Y. Liu, L. Li, P. Sun, Z. Chen, Y. Sun, Z. Wang, L. Yu and N. Yan, J. Am. Chem. Soc., 2025, 147, 13471–13482 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |