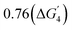Open Access Article
Open Access ArticleUnderstanding dissolution mechanism of oxide perovskites and activity tuning for O2 evolution by surface doping
Shagun
Singh
and
Naiwrit
Karmodak
 *
*
Department of Chemistry, Shiv Nadar Institution of Eminence, Greater Noida, India 201314. E-mail: naiwrit.karmodak@snu.edu.in
First published on 22nd January 2026
Abstract
The oxygen evolution reaction (OER) is the major bottleneck in electrochemical water splitting. Development of efficient electrocatalysts from earth-abundant materials is essential. This study reports a design principle for tuning the catalytic efficiency and stability of perovskite oxides (ABO3), focusing on SrMnO3, SrFeO3, SrCoO3, and SrNiO3 through B-site surface doping with Mn, Fe, Co, and Ni metal ions. Owing to multiple active sites and tunable oxidation states, these oxide surfaces exhibit a considerable reduction in thermodynamic overpotential (ηTD) upon doping. The dissolution free energy calculations are performed across a wide pH window (acidic to alkaline) and oxidative potentials to determine the Pourbaix stability and surface dissolution pathways. We find that SrCoO3 and SrNiO3 show the highest stability on doping with different transition metal atoms. Reaction free energy analysis indicates that the lattice oxygen mechanism generally outperforms the adsorbate evolution mechanism on the undoped surfaces. Surface doping results in a wide variation in the preferred reaction pathway. Mn-doped SrNiO3 emerges as the most active system with ηTD of 0.34 V, while Fe doping enhances the activity of SrCoO3 the most. Crystal orbital Hamilton population and electronic structure analyses show that modulation of the d-band center and surface structure deformation upon doping, influences the catalytic activity and stability. Using the activity descriptor Gmax(η) at an applied η of 0.3 V, the corresponding rate-determining steps (RDS) are identified. While most sites follow a single-step RDS, Fe- and Co-doped SrNiO3 exhibit multistep contributions. On varying the doping percentages of Fe on the SrCoO3 surface, we find that 25% surface doping shows the highest catalytic activity for OER.
Introduction
With the depletion of fossil fuels and the enhancement of environmental pollution, the demand for clean and sustainable energy sources has surged significantly in recent years. Renewable energy generation from solar, wind, and hydro power has shown significant advantages.1 However, these renewable energy options often face challenges due to seasonal natural cycles and regional variability. To address these limitations, there has been growing interest over the past few decades in developing energy storage and conversion technologies to store energy intermittently and use it as needed.2–5The water-splitting reaction could allow for the generation of renewable fuels.6–8 Electrochemical water-splitting reactions involve two half-cell reactions: the hydrogen evolution reaction (HER: 2H+ + 2e− → H2) at the cathode and the oxygen evolution reaction (OER: 2H2O → O2 +4H++ 4e−) at the anode.3,9–11 H2 serves as a clean fuel but while the formation of H2 is more important, the OER at the anodic half-cell is the primary bottleneck of the process. The high overpotential associated with the OER reduces the overall reaction efficiency.12–16
An efficient electrocatalyst could reduce the overpotential for O2 evolution. In the past decades, increasing attention has been given to developing electrocatalysts for OER. While developing efficient catalysts, three crucial factors must be considered: activity, stability, and cost-effectiveness.1,17–20 The Pt-group metal-based electrocatalysts, such as iridium oxide (IrO2) or ruthenium oxide (RuO2), are found with the highest efficiency in an acidic medium for the OER.21–23 However, due to their expensive nature, these catalysts would be unsuitable for industrial-scale utilization. The other catalysts with good activity for OER face several inherent challenges. The catalytic surfaces undergo surface reactions under applied potentials and solvent pH, resulting in the gradual degradation of the catalytic sites.24–29 This leads to a reduction in the catalytic efficiency under operating conditions over time. Inspired by the recent advancements in developing effective synthetic strategies and understanding catalytic efficiencies of transition metal-based catalysts, tremendous research attention has been drawn to investigate the OER activity of the transition metal oxide surfaces.30–34
Transition metal oxides have emerged as a promising alternative to traditional Pt-group metal-based electrocatalysts for OER. Among these, perovskite oxides, with the general formula ABO3, offer unique advantages.35–41 Typically, the A-site is occupied by non-transition metal ions, whereas the B-site hosts transition metal ions. The A-site shows the 12-fold coordination with oxygen, and the B-site has a 6-fold coordination.34,35 The oxidation states of the transition metal site in perovskite oxides could be further tuned by varying the A-site metal ions and the creation of the O vacancies.42–44 Furthermore, in recent studies, precise synthetic techniques have been developed for precisely doping the B-site metal ions, with multiple transition metals with varying oxidation states. This flexibility could allow for precisely modulating the catalytic properties.32,32,34,45–53
In addition to catalytic activity, stability is a crucial factor in evaluating the efficiency of an electrocatalyst.26–29 The perovskite oxide catalysts show a performance degradation often linked with the surface oxidation and aqueous dissolution.9,54 To enhance the surface activity and stability under the OER environment, strategies such as doping with different transition metals have been employed for spinel and antispinel oxide surfaces.55 Bimetallic and multi-metallic spinel oxide surfaces show multiple active sites with tunable oxidation states.56–63 Furthermore, the presence of different metal centers with varying electronegativity helps in redistributing the metal–O electron density and improves the resistance to aqueous dissolution under various oxidative electrochemical conditions.58,64–67
Recent studies have reported several oxide perovskite materials with comparable activity for OER as the Pt-group metal-based catalysts.32,32,50–53 The electrochemical stability of the perovskite oxides have been a major issue retarding the electrochemical activity for OER.9,54 Understanding the aqueous dissolution pathways of the oxide perovskite surfaces will allow devising an effective approach to improve their electrocatalytic activity for OER and unleash a design principle. The OER activity of perovskite oxides is closely linked to their electronic structure and frontier energy states, pinning near the Fermi energy level.9,32,34,45,49–53,68 The frontier energy states could also regulate the surface reactivity with the aqueous environment. Therefore, a strategy to modulate the frontier energy states will lead to an optimum strategy to tune both the activity and stability of these surfaces. Inspired by this idea, in this study, we employ a B-site surface doping of the four perovskite oxides surfaces (SrMnO3, SrFeO3, SrNiO3, and SrCoO3) with different transition metal atoms (Mn, Fe, Ni, and Co).46,69,70 Modulating the d-band center of the oxide surfaces, we tune the stability and electrocatalytic activity for the OER.71 The surface Pourbaix analysis has been performed to understand the surface aqueous dissolution reactions and stable surface configurations under the oxidative potentials and varying pH from acidic to alkaline domains. Doping with transition metals is found to enhance the stability and activity of the surfaces of both SrCoO3 and SrNiO3. We find that SrCoO3 with Fe-dopant and SrNiO3 with Mn-surface dopant, the OER overpotential is considerably reduced compared to the undoped surfaces.
Computational details
The computational calculations are performed with density functional theory (DFT), using the Vienna Ab initio Simulation Package (VASP).72–74 The electron exchange–correlation effects are treated within the Generalized Gradient Approximation (GGA) using the revised Perdew–Burke–Ernzerhof (RPBE) functional.75 The projector augmented-wave (PAW) method76 is employed to describe the electron–ion interactions, and a plane-wave basis set with an energy cutoff of 400 eV is used. The Brillouin zone is sampled using a 2 × 2 × 1 Monkhorst–Pack k-point mesh.77The electronic and ionic convergence criteria are set to 10−6 eV for energy and 10−3 eV Å−1 for forces, respectively. To account for non-covalent interactions between adsorbates and surfaces, van der Waals corrections are included. Solvent effects are incorporated using an implicit solvation model as implemented in the VASPSol package.78–80 The implicit solvation model is employed to describe the electrochemical interface. In this setup, the perovskite surfaces and adsorbed intermediates are treated quantum mechanically, while the aqueous environment is represented by a polarizable continuum (PC) medium with a dielectric constant of 80. Electrolyte interactions are incorporated through a linearized Poisson–Boltzmann approach, with a Debye screening length of 3 Å. Non-electrostatic contributions to the solvation free energy arising from surface tension are excluded, as they are less significant in interfacial two-dimensional systems.81,82 Consequently, the TAU parameter, which corresponds to the effective surface tension (eV Å−2) in the VASPsol package and accounts for cavity formation free energy, is neglected in the present calculations.
All calculations are performed with spin polarization to account for the unpaired electron and variable spin states of the surface systems. The Atomistic Simulation Environment (ASE) package is used for generating VASP input files and post-processing of the computational results.83 We have considered a 2 × 2 supercell of the perovskite surfaces to carry out the reaction free energy calculations. On the other hand, for the surface Pourbaix analysis, we have considered both 2 × 2 and 2 × 3 supercells of the surfaces to obtain the different surface vacancy structures. In all the simulations, a four-layer surface has been taken, with the bottom two layers kept fixed to mimic the bulk configuration. The upper two layers, along with the adsorbates, are allowed to minimize to obtain the most preferred geometries. A gap of 20 Å is kept along the non-periodic direction to minimize the furious interactions between the adjacent periodic cells. A detailed explanation of the computational method for reaction free energy and surface Pourbaix energy calculations is given in Sections S-I and S-II of the SI, respectively.71
Results and discussion
We perform the dissolution free energy calculations of the four perovskite oxides (SrMnO3, SrFeO3, SrCoO3, and SrNiO3), varying the pH from the acidic to alkaline range and the potential in the oxidative region from 1 to 2.0 V vs. SHE to understand the dissolution mechanism.9 The corresponding stability is determined under varied electrochemical environments. In this study, we have considered the 001 surface termination with B-site ions on the surfaces. The 001 BO-site terminated (Fig. 1a) surfaces have been identified as the most stable surface terminations in the previous studies.13,39,84–86 Furthermore, the 001 surfaces have been analyzed and found to be most active for OER. Therefore, we have built our model surfaces using the 001 BO surface terminations for the stability and catalytic studies.Electrochemical stability of the pristine and doped 001 surface
Depending upon the positioning of the O-p band center with respect to the Fermi level (EFermi), the perovskite oxides show different dissolution pathways.9Fig. 1b shows the projected density of states (PDOS) of the 001 surface of the four perovskite oxides. The red region shows the energy states of the O-p bands. The O-p band center (denoted by the green line) is positioned relatively away from the EFermi for all the surfaces (varying within the range 2.99 to 3.50 eV). Therefore, based on previous studies, the dissolution mechanism will primarily involve the leaching of surface B-site transition metal ions.54 Furthermore, on the 001 surfaces, the transition metal ions are in direct contact with the aqueous medium compared to the cationic Sr2+ ions; therefore, leaching of the surface transition metals will be more feasible than the A-site ion leaching.The dissolution free energies (ΔGpbx) for the transition metal ions are computed considering the stable transition metal ion phase separation to metal oxides at different oxidation states, as shown in eqn (1) (Section S-II in the SI gives a detailed overview of the calculation method):
| SrMO3 + zyH2O → SrM1−yO3 + yMOz + 2zyH+ + 2zye− | (1) |
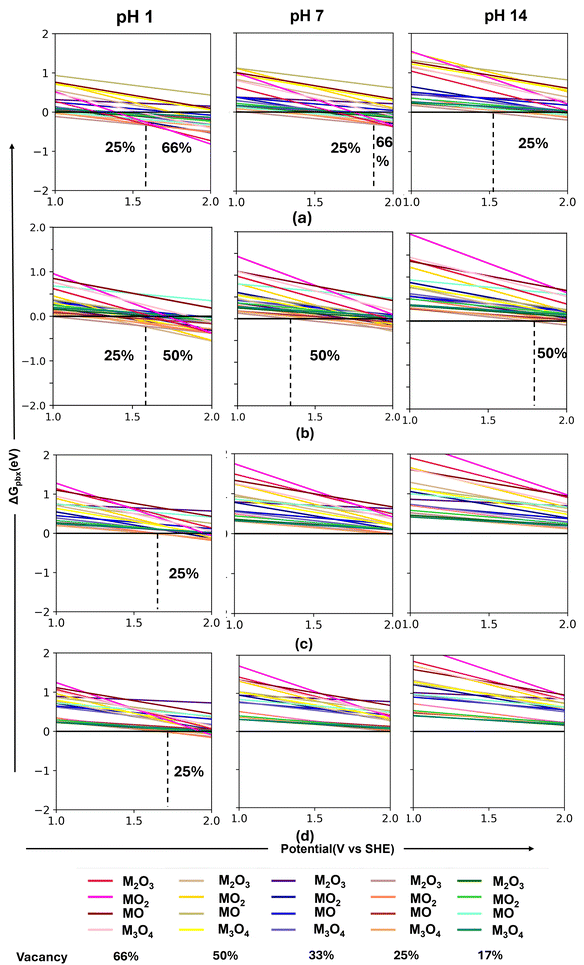
The ΔGpbx for the four perovskite 001 surfaces is shown in Fig. 2(a)–(d) at three different pH levels. The y-axis corresponds to the ΔGpbx values, and the x-axis denotes the variation in the oxidative potential with reference to SHE (VSHE). The stable configurations are found to vary considerably depending on the potential ranges at each pH. The black vertical dotted lines give the potential ranges when we found a stability switching of the configuration on varying the pH values.
On the SrMnO3 001 surface (Fig. 2(a)), at pH = 1, the dissolution of the surface Mn atoms up to the 25% surface vacancy formation is found to be most suitable up to oxidative potentials of 1.6 VSHE. Beyond this potential, the surface leaching of 66% surface atoms is more suitable. Shifting to neutral pH, the most stable surface configuration corresponds to 25% surface vacancy up to 1.8 VSHE. The formation of 66% surface vacancy is more preferred beyond that potential range. The alkaline pH shows a greater stability of the pristine surface at low oxidative potential (around 1.5 VSHE), beyond that, the 25% vacancy formation shows more feasibility.
A differing stability trend is observed for the SrFeO3 surface (Fig. 2(b)). The most stable surface configuration corresponds to the leaching of the surface Fe atoms to 25% at the potential range up to 1.6 VSHE at pH = 1. The stability of the pristine surface increases in the neutral and alkaline regions at low oxidative potentials. The relative stability of the surfaces with 50% surface vacancy shows a greater preference with the gradual increase in the potential. However, the potential window defining the corresponding stability region for these vacancy formations considerably varies. At pH 7, the pristine surface shows the highest stability up to 1.4 VSHE, whereas at pH 14, the pristine surface shows the highest stability up to 1.7 VSHE. Beyond that, the 50% vacancy formation is more preferable at both pH values.
SrCoO3 and SrNiO3 show a similar stability ordering of the surfaces (Fig. 2(c) and (d), respectively). At pH 1, the pristine surface shows the highest stability at a lower potential range (up to 1.6 and 1.7 VSHE for SrCoO3 and SrNiO3, respectively). An increase in the potential up to 2 VSHE, the surface with 25% vacancy becomes the most stable surface configuration. At pH 7 and 14, we found that the pristine surface has the most negative ΔGpbx for all potential ranges, corresponding to the most stable structural configuration for both SrCoO3 and SrNiO3.
Next, we study the effect of doping with the different transition metal atoms on these four perovskite surfaces. We have considered 25% surface doping and calculated the electrochemical stability of the doped surfaces. Mn surface positions in SrMnO3 have been doped with Fe, Co, and Ni atoms. For SrFeO3, the doped surfaces have been created by doping the surface Fe atom with Mn, Co, and Ni. On the other hand, Co and Ni atoms on SrCoO3 and SrNiO3 surfaces, respectively, are doped with Mn, Fe, and Ni dopants, and Mn, Fe, and Co dopants. The doped surfaces are denoted as M*(M). Here, M* corresponds to the dopant metal, and M denotes the host metal. For example, Fe-doped SrMnO3 surface will be denoted as Fe(Mn).
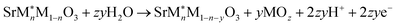 | (2) |
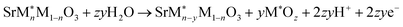 | (3) |
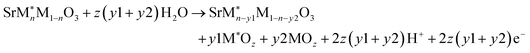 | (4) |
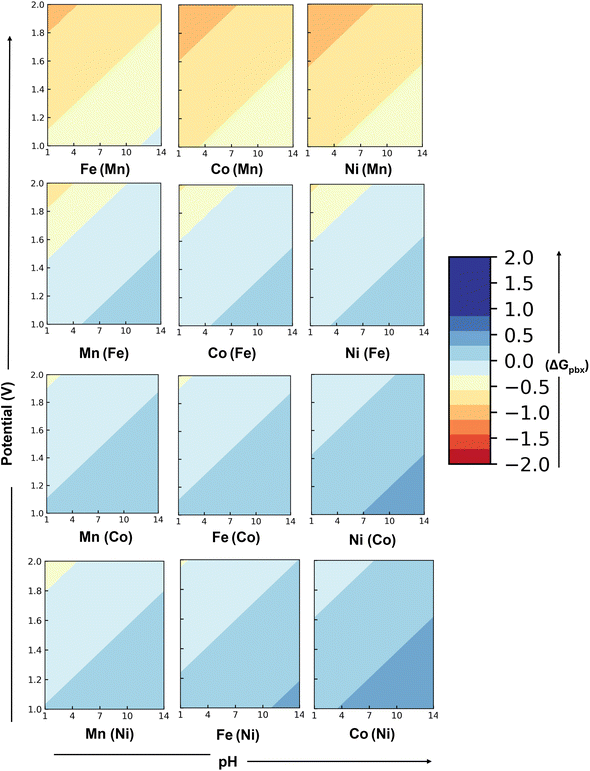
Fig. 3 shows the two-dimensional stability plots of the doped perovskite surfaces. The Pourbaix energies are calculated considering the most stable surface configuration detected for each surface. In the presence of the surface dopants, the dissolution of the surface metal ions could follow three different pathways depending upon the type of metal leaching (eqn (2)–(4)). The formation of metal oxides from both the host (M) and dopant (M*) metal atoms of the surface could lead to the generation of surface vacancies. On the other hand, either the host or the dopant metal atom could leach out and form a single phase of the metal oxide. Further details for the computations are provided in Section S-II in the SI.
On doping the different transition metal dopants, both SrMnO3 and SrFeO3, show considerable negative free energies for the creation of the surface vacancies. Both these surfaces will lead to the dissolution of the surface metal atoms. For SrFeO3, we find a narrow stability region. For Mn- and Co-doped SrFeO3, the stability region corresponds to a pH value varying between 4.5 and 14, and the potential range between 1 and 1.4 VSHE. With Ni as the surface dopant, the stability region corresponds to 3.9 to 14 pH and potential 1 to 1.6 VSHE. On the other hand, SrNiO3 and SrCoO3 show a considerable range of stable regions. At acidic pH, the stable region corresponds to lower oxidative potentials. However, at neutral and alkaline pH, the stability region corresponds to both lower and higher oxidative potentials.
To understand the differing stability trend, we analyze the electronic d-band centers of the surface host metals (Fig. 4(a)). For the undoped surfaces, a shift in the d-band center to more negative energies is observed on moving from SrMnO3 to SrFeO3 to SrCoO3 to SrNiO3. The shaded bars show the d-band center values of the surfaces without the dopant in Fig. 4a. The d-band center for the Mn in SrMnO3 is −0.59 eV from the Fermi energy, whereas the corresponding values are −1.66 eV for SrFeO3, −1.75 eV for SrCoO3, and −2.07 eV for SrNiO3. A shift in the d-band centers to more negative energies implies lower reactivity of the surfaces with the electrolyte and aqueous molecules. This results in an increasing electrochemical stability of the pristine surfaces across a wide range of pH and potential values of the undoped surfaces from SrMno3 to SrFeO3 to SrCoO3 to SrNiO3 (Fig. 2a–(d)).
To further elucidate the effect of d-band center positioning with respect to the Fermi level in governing surface stability, we compare the d-band center values with the free energies associated with the formation of 25% surface vacancies on the pristine surfaces. The tendency for surface vacancy formation varies significantly depending on the surface structure, solution pH, and applied electrochemical potential. The direct comparison of individual key dissolution steps across different surfaces is challenging. Nevertheless, our analysis shows that the formation of 25% surface vacancies is thermodynamically feasible for all the surfaces considered. Therefore, we compare the dissolution free energies associated with the formation of 25% surface vacancies with the corresponding d-band center positions. Fig. 4(b) presents this comparative analysis. The resulting linear correlation indicates that as the d-band center shifts to more negative energies, the dissolution process becomes increasingly endothermic. This trend demonstrates that the position of the d-band center plays a crucial role in determining surface stability. Specifically, a less negative d-band center strengthens interactions with solvent molecules, thereby promoting surface dissolution.
A comparison of the average d-band centers of the host metal centers on the doped surfaces (Fig. 4a) shows great similarity to the undoped surfaces. For SrMnO3, the d-band center values vary within the range −0.54 to −0.81 eV. The doping with Co slightly reduces the d-band center values to the more negative side (−0.81 eV). With SrFeO3, the d-band center values marginally vary in the presence of the different dopants. The values correspond to −1.60 for Mn(Fe), −1.58 eV for Co(Fe), and −1.49 eV for Ni(Fe). Both the SrCoO3 and SrNiO3 doped surfaces show the highest negative values for the d-band centers. The corresponding values vary within the range −1.89 to −2.22 eV for SrCoO3, and −1.90 eV to −2.18 eV for SrNiO3. In the case of SrCoO3, the d-band center values for the doped surfaces are slightly more negative than the undoped surfaces, with the highest negative value (−2.22 eV) obtained for Mn(Co). On the other hand, for SrNiO3, the d-band center value for Mn(Ni) is only greater than the undoped surface. For both Fe(Ni) and Co(Ni), the d-band center value becomes less negative (−1.93 eV and −1.90 eV, respectively) compared to the undoped surface.
The d-band center of the B-site metal ions has been known to modulate the catalytic activity and choice of the particular reaction pathway. In this study, we find that the relative stability of the perovskite surfaces could also be governed by the alignment of the d-band center relative to the Fermi energy level. A strong correlation between surface dissolution free energy values and the position of the d-band center has been obtained (Fig. 4b). Both SrCoO3 and SrNiO3 show good stability in both doped and undoped forms due to the deeper positioning of the d-band center with reference to the Fermi energy level.
Doping influences the catalytic activity of the perovskite surfaces
Having determined the electrochemical stability of the surfaces, we analyze the catalytic activity of the pristine and doped surfaces for OER. The stability and surface configurations of the surfaces will largely modulate the catalytic activity. On the perovskite oxide surfaces, among the different mechanisms, the adsorbate evolution mechanism (AEM) and lattice oxygen mechanism (LOM) compete during the oxidation of water to produce O2.13,32Fig. 5a shows the two mechanisms. AEM (black pathway in Fig. 5a) involves the adsorption of the oxygenated intermediates at the surface metal sites. The evolution of O2 occurs via the formation of the different intermediates OH*, O*, and OOH*. In contrast, the LOM pathway (red pathway in Fig. 5a) enables lattice oxygen atoms to directly participate in O–O bond formation. The strength of the lattice O bond with the surface transition metal atoms determines the reaction thermodynamics and the overpotential to drive the oxidation reaction of H2O to molecular O2. The evolution of the O2 creates a vacancy on the surface (vacO2, Fig. 5a). In the next step, the adsorption of H2O leads to the formation of adsorbed OH* at the vacancy site (vacOH* in Fig. 5a, red pathway). The removal of a proton and an electron from vacOH* regenerates the catalytic site in the fourth electrochemical step.The reaction free energies of the key OER intermediates on the pristine surfaces of the four perovskite materials are computed to elucidate the relative ordering of catalytic activity and the preferred reaction pathways. The reaction free energy values are calculated using the computational hydrogen electrode method.12 The computational details are provided in Section S-I in the SI. Fig. S1 presents the free-energy diagrams for the pristine surfaces along their most favorable reaction pathways, while Table ST1 summarizes the corresponding reaction free energies and overpotential values. SrMnO3 and SrFeO3 favor the AEM, with calculated overpotentials (ηTD) of 0.94 and 0.7 V, respectively.
On pristine SrCoO3 and SrNiO3 surface sites, the AEM is found to have higher ηTD compared to LOM. For LOM, the corresponding ηTD values are 0.51 and 1.06 V, respectively. On the other hand, for AEM, the ηTD values are 0.66 V and 1.16 V (Table ST6, SI), respectively. The formation of the OOH* intermediate shows the highest endothermicity on SrCoO3 and corresponds to the PDS for the AEM pathway. On the SrNiO3 surface, the second step, formation of O* intermediate, corresponds to the PDS.
The selectivity of the mechanistic pathways for OER on pristine SrCoO3 and SrNiO3 surfaces is in good agreement with previous studies.13,16,87–89 The binding strength of the O* intermediate is the determining factor, responsible for the preferential selectivity of the LOM vs. AEM mechanism. To understand the preferences in the mechanistic pathway, we analyse the metal–O bond strength of the O* intermediate and plot the crystal orbital Hamilton population (COHP) density in the bonding and antibonding metal–O bands.90Fig. 5b and c show the COHP plots for the O* intermediates on SrCoO3 and SrNiO3. For SrMnO3 and SrFeO3 surfaces, the corresponding COHP plots are given in Fig. S3 in the SI. The vertical line in black differentiates the bonding and antibonding energy states; on the left-hand side of each plot, the antibonding energy states are shown, whereas the bonding energy states are on the right-hand side of the vertical line. The black horizontal line denotes the Fermi energy.
Analysing the COHP plots, we find that for the O* intermediate on both SrCoO3 and SrNiO3 surfaces, the antibonding metal–O* bands are filled up compared to SrMnO3 and SrFeO3 surfaces. This results in a slight elongation of the metal–O* bond, thereby enhancing the possibility of bonding with the lattice O atoms. The occupancy of the antibonding bands destabilizes the O* intermediate in comparison to the 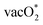 , resulting in the preference for LOM (Fig. S3 in the SI). The corresponding antibonding states are less occupied for SrMnO3, and SrFeO3 surfaces, and the integrated crystal orbital Hamilton population (ICOHP) values in Fig. S3 (SI) are greater than the values shown in Fig. 5(b) and (c). This indicates greater metal–O* bond strength on SrMnO3 and SrFeO3 001 surfaces and preference for AEM over LOM.13
, resulting in the preference for LOM (Fig. S3 in the SI). The corresponding antibonding states are less occupied for SrMnO3, and SrFeO3 surfaces, and the integrated crystal orbital Hamilton population (ICOHP) values in Fig. S3 (SI) are greater than the values shown in Fig. 5(b) and (c). This indicates greater metal–O* bond strength on SrMnO3 and SrFeO3 001 surfaces and preference for AEM over LOM.13
The relative trend in the catalytic activity of the pristine surfaces shows good coherence with previous experimental and theoretical reports.13,16,87–89 Comparing the catalytic activity of SrMnO3, SrFeO3, SrCoO3, and SrNiO3 (Table ST1 in SI), we find that the pristine SrCoO3 surface is most active for OER.
Since SrCoO3 and SrNiO3 show the highest stability, in the proceeding discussion, we will only consider these surfaces with and without the surface dopants to study the electrocatalytic activity. The reaction free energy values for the AEM and LOM pathways on the doped surfaces for SrCoO3 and SrNiO3 are shown in Table 1. For comparison, we have given the corresponding values for the undoped surfaces of SrCoO3 and SrNiO3. The potential determining steps (PDS) are denoted in bold, and overpotential (ηTD) values are in column 8 in Table 1. For each surface, the catalytic site with the lowest ηTD values is given in Table 1. We denote the active surface sites using the nomenclature M1M2(Co) and M1M2(Ni), where M1 corresponds to the dopant, and M2 denotes the active site among the dopant or the host. Within the parentheses, we write the host metal atom. The CoCo(Co) and NiNi(Ni) for SrCoO3 and SrNiO3 denotes the undoped surfaces. The reaction free energies for the other sites with higher ηTD values are given in Table ST6 in the SI.
When considering the LOM pathway, we analyse oxygen vacancy formation at all unique oxygen sites surrounding both the host and dopant metal atoms. For the dopant, we identify a single unique oxygen site bridging the host and dopant metal centers. On the otherhand, two distinct lattice oxygen sites associated with the host metal participate in vacancy formation: the oxygen bridging two host metal atoms and the oxygen bridging the host and dopant metal centers (Fig. S2, SI). We have considered all these lattice O to evaluate the minimum energy pathways. In Table ST6 (SI), reactions at the host metal site are distinguished based on the participating lattice oxygen, denoted as [OH] and [OD] in column 2. [OH] corresponds to the oxygen bridging two host metal atoms, whereas [OD] corresponds to the oxygen bridging the host and dopant metal atoms.
A considerable reduction in the ηTD values has been observed with the surface doping. On SrCoO3 surfaces, we find that for all the doped surfaces, the active site with the lowest ηTD values corresponds to the doped sites. LOM shows a slightly greater preference compared to the AEM with all the surface dopants. The 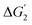 corresponding to the formation of the
corresponding to the formation of the 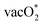 intermediate has a lower thermodynamic barrier compared to O* formation. This favors the LOM pathway.
intermediate has a lower thermodynamic barrier compared to O* formation. This favors the LOM pathway.
The PDS shows a wide variation depending on the active sites and the surface dopants. The MnMn(Co) show the formation of OH* as having the highest endothermicity. For FeFe(Co), the formation of 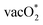 (step 3 for LOM, Fig. 5a) is the PDS, whereas the NiNi(Co) site shows the highest endothermicity for the formation of the vacOH* intermediate.
(step 3 for LOM, Fig. 5a) is the PDS, whereas the NiNi(Co) site shows the highest endothermicity for the formation of the vacOH* intermediate.
The SrNiO3 surface with the different surface dopants shows a differing trend. Unlike the NiNi site (undoped surface), the AEM is found to be the most preferred pathway with Fe and Co surface doping (FeFe(Ni) and CoCo(Ni)). Though 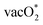 formation shows more preference than O* formation, on these sites, the regeneration of the catalytic site (the fourth step, ΔG*) has been found with high endothermicity while following the LOM.
formation shows more preference than O* formation, on these sites, the regeneration of the catalytic site (the fourth step, ΔG*) has been found with high endothermicity while following the LOM.
Compared to the undoped surface (NiNi(Ni)), both FeFe(Ni) and CoCo(Ni) sites show an increment in the catalytic activity. The PDS for FeFe(Ni) corresponds to the formation of the vacOH* intermediate with ηTD value of 0.54 V. In contrast, on the CoCo(Ni) site, the PDS corresponds to the formation of the OH* intermediate. The corresponding ηTD value is 0.61 V. With Mn-doping, we have seen the highest catalytic activity. On both MnNi(Ni) and MnMn(Ni) sites, the ηTD values are considerably lower than the NiNi site (Table 1 and ST6 in the SI). Both sites show higher preference for LOM. The most active site is MnNi (Ni), with a PDS corresponding to 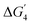 (ηTD value of 0.34 V).
(ηTD value of 0.34 V).
Surface oxygen vacancy formation is often associated with significant structural distortion and, in some cases, surface collapse of perovskite oxides. Previous studies have shown that lattice oxygen participation in the oxygen evolution reaction, accompanied by the formation of oxygen vacancies, can accelerate surface degradation and metal dissolution.91–94
In the present study, however, we do not observe such structural instability for either SrCoO3 or SrNiO3 (with or without surface dopants) upon surface O-vacancy formation. Fully relaxed surface geometries exhibit only minimal distortions following lattice oxygen participation in O2 evolution (Fig. S7 in SI). These results indicate that both SrCoO3 and SrNiO3 surfaces maintain structural stability under the considered reaction conditions.
Fig. 6(a) and (b) show the comparison of the reaction free energies on the doped surfaces with the undoped surfaces. The ideal catalysts, with ΔG values of 1.23 eV for each step, are also plotted in these diagrams (black lines). Compared to the undoped surfaces, a considerable reduction in the ΔG value of the PDS is observed for the most active doped surfaces.
The difference between the center of the d-band and p-band has been known to influence the preference of the mechanism in the OER.32,52,53,95–97 The decrease in the gap between the d-band and p-band center promotes the increase in the covalency of the M–O bond and could enhance the preference for LOM. In this study, we analyze the d-band centers of the host (Mnd) and dopant (Dnd) B-site ions and determine the difference with the O p-band centers of the different types of lattice O available in the surroundings of these metal ions. Fig. S6 shows the different O types present surrounding the dopant and the host metal center, and Table ST7 in the SI gives the differences in the band centers. For most of the cases, the differences in the d-band and p-band centers are minimal except for SrCoO3 with the Mn dopant. We have obtained that formation of 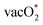 is more favorable than that of the O* intermediate, corresponding to favoring the lattice O participating in the reaction. However, in most of the cases,
is more favorable than that of the O* intermediate, corresponding to favoring the lattice O participating in the reaction. However, in most of the cases, 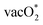 formation does not correspond to the potential determining step (PDS). Therefore, the specificity towards the preference of the mechanism is not solely dependent on the
formation does not correspond to the potential determining step (PDS). Therefore, the specificity towards the preference of the mechanism is not solely dependent on the 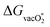 values.
values.
To understand the differing trends in the reactivity of the doped surfaces, we analyze the electronic structure of the surfaces. For SrCoO3, Mn-doping leads to stabilizing the d-bands of the active site to the most negative values (−2.22 eV). This results in the lowest catalytic activity and the highest ηTD values compared to the undoped surface. With the Fe doping to SrCoO3 surface, the catalytic activity is enhanced the most. The Fe site exhibits the least negative value for the d-band center (−1.46 eV), resulting in the greatest reduction in the endothermicity of the OH* intermediate. This is the possible reason for enhancing the catalytic activity of this site. The FeCo(Co) site, though, has an almost similar d-band center as the undoped surface (−1.88 eV and −1.76 eV for FeCo(Co) and CoCo, respectively); nevertheless, an increase in the activity is observed. We find considerable surface structure distortion on Fe doping (Fig. 6d). An increase in the binding strength of Fe with lattice O is observed. The Co–Olattice has been elongated compared to the undoped surface. We have calculated the ICOHP values to determine the metal–Olattice bond strength, as shown in Fig. 6(d) and compared to the undoped surface (Fig. 6c). The corresponding ICOHP values are lower than those of the undoped surface, reflecting the lowering of the bond strength. The structural distortions allow the creation of dangling valences on the surface metal sites and increase its activity for OER. An almost similar activity is obtained for the CoFe(Co) and FeFe(Co) sites. For the other doped surfaces, we have not observed a significant deviation of the ICOHP values in comparison to the undoped surfaces. Table ST8 in the SI shows these ICOHP values.
Similar structural distortions and alignment of the d-band centers of the SrNiO3 surface, on doping with the different transition metals, have been found to tune the catalytic activity. While following the LOM mechanism, we find that the vacOH* intermediate shows extensive stability on both the undoped and doped surfaces, except for MnNi(Ni) and MnMn(Ni) sites. This increases the endothermicity of 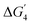 (corresponding to catalytic site regeneration), leading to an enhancement of the ηTD value. Therefore, to enhance the activity of these sites, it is important to reduce the binding affinity of the H to the lattice O site. Therefore, the lattice O–metal bonds need to be strengthened to reduce the endothermicity of the fourth step. Stronger binding of lattice O to the metal atoms will reduce the O–H bond strength in vacOH* and hence the endothermicity of
(corresponding to catalytic site regeneration), leading to an enhancement of the ηTD value. Therefore, to enhance the activity of these sites, it is important to reduce the binding affinity of the H to the lattice O site. Therefore, the lattice O–metal bonds need to be strengthened to reduce the endothermicity of the fourth step. Stronger binding of lattice O to the metal atoms will reduce the O–H bond strength in vacOH* and hence the endothermicity of 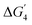 .
.
The ICOHP values of the lattice O with the metal centers increase on doping with Fe, Co, and Mn in comparison to the undoped SrNiO3 surface. A maximum increment in the binding strength of the lattice O is observed for the Mn-doped surface. Therefore, for MnNi(Ni) and MnMn(Ni) sites, formation of vacOH* shows higher endothermicity than other doped surfaces. A decrease in binding strength has been observed, providing an advantage for these sites. The thermodynamic barrier of the fourth step 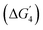 is reduced, thereby the ηTD values have been reduced to a great extent for MnNi(Ni) and MnMn(Ni) sites. Fig. 6(f) shows the corresponding ICOHP values for lattice O bonded to MnNi(Ni) and MnMn(Ni) sites. For the other surfaces (doped and undoped SrNiO3 surfaces), the ICOHP values are shown in Table ST8 in the SI.
is reduced, thereby the ηTD values have been reduced to a great extent for MnNi(Ni) and MnMn(Ni) sites. Fig. 6(f) shows the corresponding ICOHP values for lattice O bonded to MnNi(Ni) and MnMn(Ni) sites. For the other surfaces (doped and undoped SrNiO3 surfaces), the ICOHP values are shown in Table ST8 in the SI.
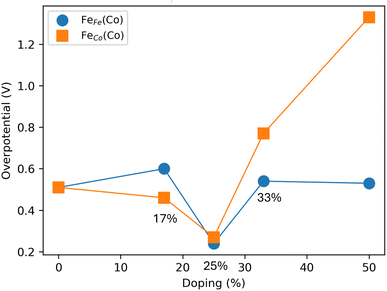
To understand the effect of dopant concentration on the OER activity, we perform a systematic activity analysis of the most active doped surface: Fe-doped SrCoO3 surfaces with Fe concentrations of 17%, 25%, 33%, and 50%. The 25% doping level is achieved by substituting one surface Co atom in a 2 × 2 (001) surface supercell of SrCoO3. The other doping concentrations 17%, 33%, and 50% are obtained by substituting one, two, and three surface Co atoms, respectively, in a 2 × 3 surface supercell of SrCoO3.
We have considered both the host and dopant atoms as potential active sites and compared the AEM and LOM pathways across different dopant concentrations. Fig. 7 shows a comparison of the theoretical ηTD values as a function of Fe doping percentage. In all cases, the LOM pathway exhibits lower ηTD values than AEM. At a Fe doping concentration of 17%, the FeCo(Co) site exhibits the lowest ηTD. However, as the dopant concentration increases, the FeFe(Co) site becomes the most favorable active site for OER. Among all doping percentages studied, the 25% Fe-doped surface shows the lowest ηTD values for both FeCo(Co) and FeFe(Co) sites. Although the difference in the corresponding ηTD values is minimal at the 25% surface doping, the FeFe(Co) site is slightly more favorable.
Recent studies have shown that B-site doping of perovskite surfaces can effectively modulate the positions of the O-p band and metal d-band centers. By varying the Mn doping concentration on the SrCoO3 surface, the electronic structure can be systematically tuned for different applications.98 More prominently, Fe doping of SrCoO3 surfaces has been reported to significantly enhance the O2 evolution reaction (OER) activity.99 Several Sr–Fe–Co–O perovskite compositions have been synthesized by varying the Fe/Co ratio. Sr2Co1.5Fe0.5O6−δ exhibits the highest OER performance, with an overpotential of 318 mV at 10 mA cm−2 and a Tafel slope of 44.8 mV dec−1.99 These experimental findings are in good agreement with our computational results. In particular, we find that maintaining the Fe dopant concentration at approximately 25% yields the maximum O2 evolution activity on the Fe-doped SrCoO3 surface (Fig. 7). Among the different active sites, FeFe(Co) shows the highest activity. Although doping of the other perovskite surfaces has not yet been reported for OER application, our results demonstrate that tuning the dopant concentration on SrNiO3 and SrFeO3 surfaces can significantly enhance their catalytic activity for the oxygen evolution reaction.
The kinetics of the elementary electrochemical steps are important in defining catalytic activity. ηTD is evaluated at zero applied potential and corresponds to the elementary step with the largest thermodynamic free energy. However, in multistep electrochemical pathways, this step is not always the rate-determining step (RDS), particularly in cases where the potential-determining step (PDS) differs from the RDS.100
To address this, Exner and co-workers introduced a potential-dependent reactivity descriptor, Gmax(η), which directly identifies the RDS under an applied potential.100 The descriptor is derived from the free energy changes of the elementary steps (ΔGi, i = 1, 2, 3, 4) under an applied overpotential. Unlike ηTD, Gmax(η) scales linearly with the free energy of the RDS, providing a more accurate reflection of catalytic activity.101–103
For OER catalysts, values of Gmax(η) less than or equal to 1.0 eV at η = 0.3 V are typically associated with good catalytic performance.101 Accordingly, we have used this criterion in our analysis (Table 1) to identify the RDS and evaluate the efficiency of various active sites with different dopants. The specific ΔGi values that govern the RDS are given in column 7.
The trend in Gmax values indicates that, for the majority of active sites obtained in our study, the RDS is governed by a single elementary step. However, the corresponding step determining the RDS shows wide variation depending upon the active sites. The CoCo(Co), MnMn(Co) sites show ΔG1 as the RDS. On the other hand, 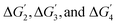 are found to be the RDS for FeFe(Co), NiNi(Co) and NiNi(Ni) respectively. This suggests that the reaction kinetics in these sites are largely dictated by the energetics of the specific step with the largest Gmax values.
are found to be the RDS for FeFe(Co), NiNi(Co) and NiNi(Ni) respectively. This suggests that the reaction kinetics in these sites are largely dictated by the energetics of the specific step with the largest Gmax values.
In contrast, a different pattern emerges for certain dopants, most notably Fe and Co doped to the SrNiO3 surfaces, where Gmax is not confined to a single ΔGi value. These surfaces instead show the cumulative contribution of multiple steps (e.g., ΔG1 + ΔG2 + ΔG3). The kinetic limitation is distributed over several transitions rather than a localized single barrier on these surface sites. The multistep character in defining the RDS reflects a more complex kinetic landscape, where the reaction energetics of multiple intermediates determine the catalytic activity.
To assess the experimental viability of the doped surfaces, we have calculated the formation energies (Eform) for all perovskites with the different surface dopants using bulk metals as references for the dopants and the pristine surface as the reference for the host perovskite (Fig. S4 in the SI). In Section S-V in the SI, the details for the formation energy of the doped surfaces are shown.
A clear trend is observed in which the Eform becomes progressively less favorable when moving from early to late transition-metal dopants. While all dopants on SrMnO3 surfaces show endothermic Eform, SrFeO3 shows exothermic stabilization only for Mn doping. In contrast, most dopants with SrCoO3 and SrNiO3 surfaces exhibit exothermic formation energies, indicating enhanced thermodynamic stability. Notably, Fe-doped SrCoO3 and Mn-doped SrNiO3 surfaces, which exhibit good catalytic activity, display exothermic formation energies. Therefore, these compositions are experimentally viable.
Conclusions
In this study, we have analyzed the catalytic activity and reaction mechanism for OER on four perovskite oxide surfaces, namely SrMnO3, SrFeO3, SrCoO3, and SrNiO3. The electrochemical stability and catalytic activity for OER have been analyzed, focusing on the (001) surface termination of the pristine surface, and doped with different transition metal ions at the B-site. We have calculated the dissolution free energies across a wide pH range (acidic to alkaline) and oxidative potentials of 1–2 V vs. SHE to determine the electrochemical stability under these varying conditions. PDOS analysis of the (001) surfaces shows the O-p band center located 2.99–3.50 eV below EFermi, indicating that dissolution mainly proceeds through leaching of surface B-site transition metal ions.Dissolution free energies (ΔGpbx) are computed for surface vacancy creation of 17–66%. For SrMnO3, Mn dissolution is most favorable (up to 66%) in acidic media, while neutral and alkaline pH favor only 25% or pristine surfaces depending on potential. SrFeO3 surfaces are most stable with 50% vacancy formation at acidic pH, whereas pristine surfaces dominate stability at neutral and alkaline pH under low potentials. In SrCoO3 and SrNiO3, pristine surfaces are most stable at low potentials across pH 1–14. At higher oxidation potential and acidic pH, 25% vacancy formation is found to be possible. However, at neutral and alkaline pH, the pristine surfaces show extensive stability for the entire potential range.
With 25% surface doping of transition metals (Mn, Fe, Co, and Ni) wide variation in the electrocatalytic activity and stability is observed. Pourbaix free energy (ΔGpbx) calculations of these doped systems reveal many dissolution pathways depending on whether the host, dopant, or both ions leach. SrMnO3 do not show stability regions with any of the surface dopants. SrFeO3 show a narrow stable region at the alkaline pH and low oxidative potential. However, SrCoO3 and SrNiO3 show stable regions at the neutral to alkaline pH. The positioning of the metal d-band centers with respect to the Fermi energy strongly influences the stability of the surfaces.
Next, we analyze the catalytic activity of pristine and doped SrCoO3 and SrNiO3 surfaces. OER is found to occur via two competing pathways: Adsorbate Evolution Mechanism (AEM) and the Lattice Oxygen Mechanism (LOM). The calculations show that undoped SrCoO3 and SrNiO3 favor LOM with ηTD values of 0.51 and 1.06 V, respectively. Analysis of COHP plots reveals progressive weakening of metal–O bonds from SrCoO3 to SrNiO3, promoting LOM. Surface doping further tunes catalytic activity: Fe-doping in SrCoO3 reduces ηTD to as low as 0.24 V at the FeFe(Co) site. In SrNiO3, Mn-doping significantly lowers ηTD to 0.34 V at MnNi(Ni) site. We found that surface distortion and the alignment of d-band center of the active sites considerably determine the relative activity of the surface sites. On varying the Fe-dopant percentages on SrCoO3 surface, we find that 25% surface doping shows the highest catalytic activity for OER.
The activity descriptor Gmax(η) at the applied η of 0.3 V is used to analyse the rate-determining step (RDS). While most active sites exhibit a single step as the RDS, Fe- and Co-doped SrNiO3 display a multistep process for determining the reaction rate. The design principle shown in this study to tune the catalytic activity and stability of perovskite oxide surfaces by surface doping will be of great importance for efficient catalyst development for OER.
Author contributions
S. S.: data curation, formal analysis, investigation, methodology, and visualization. N. K.: conceptualization, funding acquisition, methodology, project administration, resources, software, supervision, and visualization. Both authors contributed to writing the original draft, reviewing, and editing.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data related to computations will be available from the authors. Most of the data used to set up the figures has been provided in the manuscript and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta07376f.Acknowledgements
N. K. and S. S. acknowledge the Shiv Nadar Institution of Eminence and Shiv Nadar Foundation for financial assistance and computational support. The calculations are performed at the supercomputer (Magus).Notes and references
- Z. W. Seh, J. Kibsgaard, C. F. Dickens, I. Chorkendorff, J. K. Nørskov and T. F. Jaramillo, Science, 2017, 355, eaad4998 CrossRef PubMed.
- Y. Zhu, W. Zhou and Z. Shao, Small, 2017, 13, 1603793 CrossRef PubMed.
- L. Han, S. Dong and E. Wang, Adv. Mater., 2016, 28, 9266–9291 CrossRef CAS PubMed.
- B.-J. Kim, D. F. Abbott, X. Cheng, E. Fabbri, M. Nachtegaal, F. Bozza, I. E. Castelli, D. Lebedev, R. Schaublin and C. Copéret, et al. , ACS Catal., 2017, 7, 3245–3256 CrossRef CAS.
- R. S. Haszeldine, Science, 2009, 325, 1647–1652 CrossRef CAS PubMed.
- N.-T. Suen, S.-F. Hung, Q. Quan, N. Zhang, Y.-J. Xu and H. M. Chen, Chem. Soc. Rev., 2017, 46, 337–365 Search PubMed.
- Y. Jin, H. Wang, J. Li, X. Yue, Y. Han, P. K. Shen and Y. Cui, Adv. Mater., 2016, 28, 3785–3790 CrossRef CAS PubMed.
- Y. Zheng, Y. Jiao, Y. Zhu, L. H. Li, Y. Han, Y. Chen, M. Jaroniec and S.-Z. Qiao, J. Am. Chem. Soc., 2016, 138, 16174–16181 CrossRef CAS PubMed.
- B. Han, M. Risch, Y.-L. Lee, C. Ling, H. Jia and Y. Shao-Horn, Phys. Chem. Chem. Phys., 2015, 17, 22576–22580 RSC.
- O. Diaz-Morales, I. Ledezma-Yanez, M. T. Koper and F. Calle-Vallejo, ACS Catal., 2015, 5, 5380–5387 CrossRef CAS.
- W. Li, H. Tian, L. Ma, Y. Wang, X. Liu and X. Gao, Mater. Adv., 2022, 3, 5598–5644 RSC.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jonsson, J. Phys. Chem. B, 2004, 108, 17886–17892 CrossRef PubMed.
- J. S. Yoo, X. Rong, Y. Liu and A. M. Kolpak, ACS Catal., 2018, 8, 4628–4636 CrossRef CAS.
- H. Li, Y. Chen, J. Z. Y. Seow, C. Liu, A. C. Fisher, J. W. Ager and Z. J. Xu, Small Sci., 2022, 2, 2100048 CrossRef CAS PubMed.
- Y. Zhao, D. P. Adiyeri Saseendran, C. Huang, C. A. Triana, W. R. Marks, H. Chen, H. Zhao and G. R. Patzke, Chem. Rev., 2023, 123, 6257–6358 Search PubMed.
- I. C. Man, H.-Y. Su, F. Calle-Vallejo, H. A. Hansen, J. I. Martínez, N. G. Inoglu, J. Kitchin, T. F. Jaramillo, J. K. Nørskov and J. Rossmeisl, ChemCatChem, 2011, 3, 1159–1165 CrossRef CAS.
- K. S. Exner, Acc. Chem. Res., 2024, 57, 1336–1345 CrossRef CAS PubMed.
- S. Singh and N. Karmodak, ACS Appl. Energy Mater., 2024, 7, 7854–7863 CrossRef CAS.
- M. Sokolov and K. S. Exner, Chem Catal., 2024, 4, year Search PubMed.
- N. Karmodak and O. Andreussi, ACS Energy Lett., 2020, 5, 885–891 CrossRef CAS.
- N. Danilovic, R. Subbaraman, K.-C. Chang, S. H. Chang, Y. J. Kang, J. Snyder, A. P. Paulikas, D. Strmcnik, Y.-T. Kim and D. Myers, et al. , J. Phys. Chem. Lett., 2014, 5, 2474–2478 CrossRef CAS PubMed.
- J. Rossmeisl, A. Logadottir and J. K. Nørskov, Chem. Phys., 2005, 319, 178–184 CrossRef CAS.
- S. Cherevko, S. Geiger, O. Kasian, N. Kulyk, J.-P. Grote, A. Savan, B. R. Shrestha, S. Merzlikin, B. Breitbach and A. Ludwig, et al. , Catal. Today, 2016, 262, 170–180 CrossRef CAS.
- Y. Zhu, W. Zhou, J. Yu, Y. Chen, M. Liu and Z. Shao, Chem. Mater., 2016, 28, 1691–1697 CrossRef CAS.
- G. K. K. Gunasooriya and J. K. Nørskov, ACS Energy Lett., 2020, 5, 3778–3787 CrossRef CAS.
- R. Frydendal, E. A. Paoli, B. P. Knudsen, B. Wickman, P. Malacrida, I. E. Stephens and I. Chorkendorff, ChemElectroChem, 2014, 1, 2075–2081 CrossRef CAS.
- N. Hodnik, P. Jovanovic, A. Pavlisic, B. Jozinovic, M. Zorko, M. Bele, V. S. Selih, M. Sala, S. Hocevar and M. Gaberscek, J. Phys. Chem. C, 2015, 119, 10140–10147 CrossRef CAS.
- K. S. Exner, ChemElectroChem, 2021, 8, 46–48 CrossRef CAS.
- V. M, S. Singh, F. Bononi, O. Andreussi and N. Karmodak, J. Chem. Phys., 2023, 159, 111001 CrossRef CAS PubMed.
- D. A. Kuznetsov, B. Han, Y. Yu, R. R. Rao, J. Hwang, Y. Román-Leshkov and Y. Shao-Horn, Joule, 2018, 2, 225–244 CrossRef CAS.
- N. Karmodak, L. Bursi and O. Andreussi, J. Phys. Chem. Lett., 2021, 13, 58–65 CrossRef PubMed.
- A. Grimaud, O. Diaz-Morales, B. Han, W. T. Hong, Y.-L. Lee, L. Giordano, K. A. Stoerzinger, M. T. Koper and Y. Shao-Horn, Nat. Chem., 2017, 9, 457–465 CrossRef CAS PubMed.
- Z. Wang, Y.-R. Zheng, I. Chorkendorff and J. K. Nørskov, ACS Energy Lett., 2020, 5, 2905–2908 CrossRef CAS.
- J. Xu, C. Chen, Z. Han, Y. Yang, J. Li and Q. Deng, Nanomaterials, 2019, 9, 1161 CrossRef CAS PubMed.
- M. De Graef and M. E. McHenry, Structure of Materials: an Introduction to Crystallography, Diffraction and Symmetry, Cambridge University Press, 2012 Search PubMed.
- J. Hwang, R. R. Rao, L. Giordano, Y. Katayama, Y. Yu and Y. Shao-Horn, Science, 2017, 358, 751–756 CrossRef CAS PubMed.
- R. E. Cohen, Nature, 1992, 358, 136–138 CrossRef CAS.
- K. Huang, R. S. Tichy and J. B. Goodenough, J. Am. Ceram. Soc., 1998, 81, 2565–2575 CrossRef CAS.
- J. Suntivich, H. A. Gasteiger, N. Yabuuchi, H. Nakanishi, J. B. Goodenough and Y. Shao-Horn, Nat. Chem., 2011, 3, 546–550 CrossRef CAS PubMed.
- E. Fabbri, M. Nachtegaal, T. Binninger, X. Cheng, B.-J. Kim, J. Durst, F. Bozza, T. Graule, R. Schäublin and L. Wiles, et al. , Nat. Mater., 2017, 16, 925–931 CrossRef CAS PubMed.
- B.-J. Kim, X. Cheng, D. F. Abbott, E. Fabbri, F. Bozza, T. Graule, I. E. Castelli, L. Wiles, N. Danilovic and K. E. Ayers, et al. , Adv. Funct. Mater., 2018, 28, 1804355 CrossRef.
- W. T. Hong, M. Risch, K. A. Stoerzinger, A. Grimaud, J. Suntivich and Y. Shao-Horn, Energy Environ. Sci., 2015, 8, 1404–1427 RSC.
- H. Over, Y. D. Kim, A. Seitsonen, S. Wendt, E. Lundgren, M. Schmid, P. Varga, A. Morgante and G. Ertl, Science, 2000, 287, 1474–1476 CrossRef CAS PubMed.
- J. Kim, X. Yin, K.-C. Tsao, S. Fang and H. Yang, J. Am. Chem. Soc., 2014, 136, 14646–14649 CrossRef CAS PubMed.
- A. Grimaud, K. J. May, C. E. Carlton, Y.-L. Lee, M. Risch, W. T. Hong, J. Zhou and Y. Shao-Horn, Nat. Commun., 2013, 4, 2439 CrossRef PubMed.
- A. Swarnkar, W. J. Mir and A. Nag, ACS Energy Lett., 2018, 3, 286–289 CrossRef CAS.
- W. Zhang, H. Liu, Y. Qu, J. Cui, W. Zhang, T. Shi and H.-L. Wang, Adv. Mater., 2024, 36, 2309193 CrossRef CAS PubMed.
- Y. Liang, F. Li, X. Cui, C. Stampfl, S. P. Ringer, X. Yang, J. Huang and R. Zheng, Sci. Adv., 2025, 11, eads7054 CrossRef CAS PubMed.
- Z. Li, P. Wei and G. Wang, Energy Fuels, 2022, 36, 11724–11744 CrossRef CAS.
- C. E. Beall, E. Fabbri and T. J. Schmidt, ACS Catal., 2021, 11, 3094–3114 CrossRef CAS.
- S. Sen and T. K. Mandal, ChemCatChem, 2025, e00535 CrossRef CAS.
- J. T. Mefford, X. Rong, A. M. Abakumov, W. G. Hardin, S. Dai, A. M. Kolpak, K. P. Johnston and K. J. Stevenson, Nat. Commun., 2016, 7, 11053 CrossRef CAS PubMed.
- N. Zhang and Y. Xiong, J. Phys. Chem. C, 2023, 127, 2147–2159 CrossRef CAS.
- B.-J. Kim, E. Fabbri, M. Borlaf, D. F. Abbott, I. E. Castelli, M. Nachtegaal, T. Graule and T. J. Schmidt, Mater. Adv., 2021, 2, 345–355 RSC.
- L.-B. Liu, C. Yi, H.-C. Mi, S. L. Zhang, X.-Z. Fu, J.-L. Luo and S. Liu, Electrochem. Energy Rev., 2024, 7, 14 CrossRef CAS PubMed.
- Y. Ma, M. Zha, Y. Dong, L. Li and G. Hu, Mater. Res. Express, 2019, 6, 115033 CrossRef CAS.
- Y.-F. Li and A. Selloni, ACS Catal., 2014, 4, 1148–1153 CrossRef CAS.
- F. Song, L. Bai, A. Moysiadou, S. Lee, C. Hu, L. Liardet and X. Hu, J. Am. Chem. Soc., 2018, 140, 7748–7759 CrossRef CAS PubMed.
- Z.-D. He, R. Tesch, M. J. Eslamibidgoli, M. H. Eikerling and P. M. Kowalski, Nat. Commun., 2023, 14, 3498 CrossRef CAS PubMed.
- I. Hussain, U. Sajjad, O. J. Kewate, U. Amara, F. Bibi, A. Hanan, D. Potphode, M. Ahmad, M. S. Javed and P. Rosaiah, et al. , Mater. Today Phys., 2024, 42, 101382 CrossRef CAS.
- H. Wang, W. Xu, S. Richins, K. Liaw, L. Yan, M. Zhou and H. Luo, Electrochim. Acta, 2019, 296, 945–953 CrossRef CAS.
- H. Wang, J. Wang, Y. Pi, Q. Shao, Y. Tan and X. Huang, Angew. Chem., Int. Ed., 2019, 131, 2338–2342 CrossRef.
- J. Bian, R. Su, Y. Yao, J. Wang, J. Zhou, F. Li, Z. L. Wang and C. Sun, ACS Appl. Energy Mater., 2019, 2, 923–931 Search PubMed.
- M. Bajdich, M. García-Mota, A. Vojvodic, J. K. Nørskov and A. T. Bell, J. Am. Chem. Soc., 2013, 135, 13521–13530 CrossRef CAS PubMed.
- O. N. Avcı, L. Sementa and A. Fortunelli, ACS Catal., 2022, 12, 9058–9073 CrossRef PubMed.
- N. Karmodak and O. Andreussi, J. Phys. Chem. C, 2021, 125, 10397–10405 CrossRef CAS.
- A. Li, S. Kong, C. Guo, H. Ooka, K. Adachi, D. Hashizume, Q. Jiang, H. Han, J. Xiao and R. Nakamura, Nat. Catal., 2022, 5, 109–118 Search PubMed.
- J. Suntivich, K. J. May, H. A. Gasteiger, J. B. Goodenough and Y. Shao-Horn, Science, 2011, 334, 1383–1385 CrossRef CAS PubMed.
- A. S. Raman, R. Patel and A. Vojvodic, Faraday Discuss., 2021, 229, 75–88 RSC.
- B. Luo, F. Li, K. Xu, Y. Guo, Y. Liu, Z. Xia and J. Z. Zhang, J. Mater. Chem. C, 2019, 7, 2781–2808 RSC.
- The computational details are in the SI.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 48, 13115–13118 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 Search PubMed.
- S. Maintz, V. L. Deringer, A. L. Tchougréeff and R. Dronskowski, J. Comput. Chem., 2013, 34, 2557–2567 Search PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758 CrossRef CAS.
- K. Mathew, R. Sundararaman, K. Letchworth-Weaver, T. Arias and R. G. Hennig, J. Chem. Phys., 2014, 140, 084106 CrossRef PubMed.
- K. Mathew, V. Kolluru, S. Mula, S. N. Steinmann and R. G. Hennig, J. Chem. Phys., 2019, 151, 234101 Search PubMed.
- J. A. Gauthier, S. Ringe, C. F. Dickens, A. J. Garza, A. T. Bell, M. Head-Gordon, J. K. Nørskov and K. Chan, ACS Catal., 2018, 9, 920–931 CrossRef.
- J. A. Gauthier, C. F. Dickens, S. Ringe and K. Chan, ChemPhysChem, 2019, 20, 3074–3080 Search PubMed.
- O. Andreussi, I. Dabo and N. Marzari, J. Chem. Phys., 2012, 136, 064102 CrossRef PubMed.
- A. H. Larsen, J. J. Mortensen, J. Blomqvist, I. E. Castelli, R. Christensen, M. Dułak, J. Friis, M. N. Groves, B. Hammer and C. Hargus, et al. , J. Condens. Matter Phys., 2017, 29, 273002 CrossRef PubMed.
- Y. Choi, D. S. Mebane, M.-C. Lin and M. Liu, Chem. Mater., 2007, 19, 1690–1699 CrossRef CAS.
- R. Evarestov, E. Kotomin, Y. A. Mastrikov, D. Gryaznov, E. Heifets and J. Maier, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 72, 214411 CrossRef.
- Y.-L. Lee, J. Kleis, J. Rossmeisl and D. Morgan, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 80, 224101 CrossRef.
- L. Trotochaud, J. K. Ranney, K. N. Williams and S. W. Boettcher, J. Am. Chem. Soc., 2012, 134, 17253–17261 CrossRef CAS PubMed.
- J. O. Bockris and T. N. Veziroglu, Int. J. Hydrogen Energy, 2007, 32, 1605–1610 Search PubMed.
- M. D. Bhatt and J. Y. Lee, Energy Fuels, 2020, 34, 6634–6695 Search PubMed.
- R. Dronskowski and P. E. Bloechl, J. Phys. Chem., 1993, 97, 8617–8624 CrossRef CAS.
- S. Geiger, O. Kasian, M. Ledendecker, E. Pizzutilo, A. M. Mingers, W. T. Fu, O. Diaz-Morales, Z. Li, T. Oellers and L. Fruchter, et al. , Nat. Catal., 2018, 1, 508–515 CrossRef CAS.
- O. Kasian, J.-P. Grote, S. Geiger, S. Cherevko and K. J. Mayrhofer, Angew. Chem., Int. Ed., 2018, 57, 2488–2491 Search PubMed.
- C. W. Song, J. Lim, H. B. Bae and S.-Y. Chung, Energy Environ. Sci., 2020, 13, 4178–4188 RSC.
- F.-Y. Chen, Z.-Y. Wu, Z. Adler and H. Wang, Joule, 2021, 5, 1704–1731 Search PubMed.
- N. Zhang and Y. Chai, Energy Environ. Sci., 2021, 14, 4647–4671 Search PubMed.
- J. Zaanen, G. Sawatzky and J. Allen, Phys. Rev. Lett., 1985, 55, 418 CrossRef CAS PubMed.
- A. Grimaud, W. T. Hong, Y. Shao-Horn and J.-M. Tarascon, Nat. Mater., 2016, 15, 121–126 CrossRef CAS PubMed.
- W. Zhang, Y. Wang, Y. Li and X. Zhang, J. Ind. Eng. Chem., 2024, 139, 199–212 Search PubMed.
- S. R. Ede, C. N. Collins, C. D. Posada, G. George, H. Wu, W. D. Ratcliff, Y. Lin, J. Wen, S. Han and Z. Luo, ACS Catal., 2021, 11, 4327–4337 Search PubMed.
- S. Razzaq and K. S. Exner, ACS Catal., 2023, 13, 1740–1758 Search PubMed.
- K. S. Exner, ACS Catal., 2020, 10, 12607–12617 CrossRef CAS.
- M. Usama, S. Razzaq, C. Hättig, S. N. Steinmann and K. S. Exner, Nat. Commun., 2025, 16, 6137 Search PubMed.
- K. Dhaka and K. S. Exner, J. Catal., 2025, 443, 115970 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



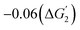
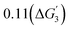
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)