Advances and challenges for Mg3(Sb,Bi)2-based thermoelectric materials and devices
Yu
Yan
a,
Wen
Zhang
a,
Huijun
Kang
 *ab,
Zongning
Chen
ab,
Enyu
Guo
ab,
Rongchun
Chen
*a and
Tongmin
Wang
*ab,
Zongning
Chen
ab,
Enyu
Guo
ab,
Rongchun
Chen
*a and
Tongmin
Wang
 *ab
*ab
aKey Laboratory of Solidification Control and Digital Preparation Technology (Liaoning Province), School of Materials Science and Engineering, Dalian University of Technology, Dalian, 116024, China. E-mail: kanghuijun@dlut.edu.cn; crc618@dlut.edu.cn; tmwang@dlut.edu.cn
bNingbo Institute of Dalian University of Technology, Ningbo, 315000, China
First published on 17th November 2025
Abstract
Mg3(Sb,Bi)2-based thermoelectric (TE) materials have attracted considerable attention due to their earth-abundant constituent elements, excellent environmental compatibility, and outstanding TE properties in the low- and medium-temperature range. However, optimizing TE properties remains challenging for Mg3(Sb,Bi)2, as does device structure design. Given the rapid development of Mg3(Sb,Bi)2-based materials and their devices in recent years, there is an urgent need for a systematic and critical review of the progress, challenges, and prospects. We systematically summarize and analyze various current strategies for improving TE properties, including component modulation, defect engineering, band engineering, grain boundary engineering, and mixed scattering optimization of ionized impurities and phonons. In addition, the key challenges for practical applications are comprehensively analyzed, particularly focusing on connection technology, barrier layer material screening, and device structure design. Finally, we outline the significant potential and current challenges of Mg3(Sb,Bi)2-based materials in low- and medium-temperature waste heat recovery and solid-state cooling applications, and provide future insights regarding TE properties modulation, preparation scale-up, and novel application prospects.
1. Introduction
With the rapid development of society and the increasing consumption of fossil fuels, the search for clean, efficient, and renewable energy materials has emerged as a global priority. Thermoelectric (TE) materials have gained widespread attention, attributed to their environmental friendliness and capability for solid-state cooling and power generation.1,2 Advancements in near-room-temperature TE materials are being critically driven by the demand for energy harvesting and low-grade waste heat recovery.3–7 The TE conversion efficiency (η) is defined by the dimensionless figure-of-merit, zT = S2σT/κ, where S, σ, κ, and T represent Seebeck coefficient, electrical conductivity, total thermal conductivity, and absolute temperature, respectively.8,9 Recently, Mg3(Sb,Bi)2 has garnered significant research attention because of its benefits such as abundance, low toxicity, and cost-effectiveness.10–12 Compositional modulation strategies enable Mg3(Sb,Bi)2 to demonstrate promising properties for low-grade heat recovery and room-temperature cooling, positioning it as a viable next-generation candidate.13–16Mg3(Sb,Bi)2 is a typical Zintl compound that crystallizes in a CaAl2Si2-type structure, as shown in Fig. 1a. Its crystal structure features a three-dimensional chemical bonding network comprising Mg2+ cations in octahedral coordination and (Mg2Sb2)2− anionic layers in tetrahedral coordination, establishing a robust basis for excellent TE properties.17–21 As shown in Fig. 1b, the intrinsic band structure of Mg3(Sb,Bi)2 indicates that Mg3Sb2 is an indirect bandgap (Eg) semiconductor (0.4–0.8 eV) with relatively low carrier concentration (nH), whereas Mg3Bi2 is a type-II topological nodal-line semimetal characterized by the absence of Eg and high nH.22–25Fig. 1c shows the density of states (DOS) for Mg3Sb2 and Mg3Bi2. The valence band maximum (VBM) of Mg3Sb2 and Mg3Bi2 is dominated by the Sb/Bi 5p orbitals, while the conduction band minimum (CBM) is dominated by the Mg 3s orbitals.26,27 This band structure indicates that the electronic states of anions and cations dominate the electrical properties of p-type and n-type conduction samples, respectively. The Eg can be regulated by changing the Bi/Sb ratio through Mg3Sb2 and Mg3Bi2 alloying, enabling optimization of electrical transport properties.28–30 However, the volatility and high vapor pressure of Mg during synthesis result in a large number of Mg vacancies, significantly inhibiting electron transport properties.31,32 The breakthrough came when Tamaki et al.33 first synthesized n-type Mg3.2Sb1.5Bi0.49Te0.01 using excess Mg and Te-doping, achieving a remarkable zT of 1.5 at 723 K. This immediately attracted significant research interest. Subsequent studies demonstrated that interstitial Mn doping effectively suppresses the formation of Mg vacancies. This approach led to the first identification of a positive temperature-dependent Eg in Mg3+δSbxBi2−x, directly confirming the suppression of the bipolar effect at medium temperatures. Ultimately, Mg3+δSb1.5Bi0.49Te0.01Mn0.01 achieved an average zT (zTave) of 1.05 between 323 and 523 K. Notably, its favorable mechanical toughness highlights Mg3+δSbxBi2−x materials as promising candidates for TE applications near-room-temperature.34 Further demonstrating potential, a zT value of 0.9 was achieved at 350 K for the n-type Mg3.2Bi1.998−xSbxTe0.002 reported by Mao et al.,35 demonstrating its significance as a substitute for Bi2Te3 in low-grade waste heat recovery applications. Furthermore, in a cooling device with p-type Bi0.5Sb1.5Te3, it achieved a temperature difference (ΔT) of 91 K at a hot-side temperature (Th) of 350 K, exceeding the performance of commercial counterparts. A room-temperature analysis of the electronic quality factor (BET/κl) confirms that n-type Mg3Sb2-based alloys exhibit highly competitive TE properties compared to commercial Bi2Te3 alloys, solidifying their status as strong contenders for TE cooling and power generation near-room-temperature.36 Given this remarkable progress, a systematic and critical review is essential to summarize the advancements, challenges, and prospects of Mg3(Sb,Bi)2.
Currently, defect engineering,37 additive manufacturing,38 and device applications39,40 cover various aspects of TE materials. To facilitate the further development of Mg3(Sb,Bi)2, we systematically summarize the past advancements in TE performance regulation and device structure design. In this review, we commence by introducing the fundamental properties of Mg3(Sb,Bi)2, including crystal and electronic structures, to elucidate the intrinsic foundations of its transport behavior. Next, we systematically categorize strategies for enhancing properties, including component modulation, band engineering, grain boundary (GB) engineering, composite engineering, and mixed scattering optimization of ionized impurities and phonons. We then discuss connection techniques, the screening of barrier layer materials, and the design of device structures for TE modules of Mg3(Sb,Bi)2, addressing critical steps toward practical applications. Ultimately, this review provides valuable insights into the development of Mg3(Sb,Bi)2-based materials and promotes their application in low-grade heat recovery and solid-state cooling.
2. Synthesis and progress
2.1. Preparation method
Fabricating advanced Mg3(Sb,Bi)2-based materials demands precise control over composition, microstructure, and defect states. Common synthesis techniques encompass melt-based processing, solid-state reactions, single crystal growth, mechanical alloying, and emerging additive manufacturing approaches. Careful selection of the fabrication route is therefore crucial for optimizing TE and mechanical properties, especially considering the volatility of Mg.High-temperature melting/reaction is a traditional synthesis method valued for its simplicity and suitability for large-scale production.41 For instance, Y-doped ingots achieving grain sizes > 100 µm and room-temperature carrier mobility (µH) > 100 cm2 V−1 s−1 were prepared by high-temperature reactive melting with subsequent annealing.42 However, significant Mg volatilization during melting remains a challenge. This can be mitigated by adding excess Mg during sintering, which effectively compensates for Mg loss in conventional n-type Mg3(Bi, Sb)2 alloys.43 Recently, kilogram-level Mg3(Sb,Bi)2 powder was synthesized in merely 5 minutes via the self-propagating high-temperature synthesis method.44 This technique also achieves an oxygen content below 0.3%, offering a viable pathway for the industrial-scale production of powder.
As shown in Fig. 2a, solid-state synthesis in sealed tubes suppresses Mg volatilization and impurity formation through high-temperature reactions.45–47 Using this method, Ponnambalam et al.45 synthesized single-phase p-type Mg3SbBi, achieving a zT value of 0.4 at 825 K. The Mg3.2Bi1.4975Sb0.5Te0.0025 compound, prepared by sealing in tantalum tubes followed by melt annealing and directional solidification, exhibits record grain sizes larger than 500 µm and room-temperature µH of 220 cm2 V−1 s−1 among Mg3(Sb,Bi)2-based materials.48 Although offering low-cost sintering and minimal sintering equipment requirements, the high-temperature synthesis step necessitates specialized equipment and currently lacks scalability for industrial production.
 | ||
| Fig. 2 (a) Mg-flux method using a sealed tantalum tube.47 Copyright 2020, Royal Society of Chemistry. (b) Photograph of as-grown Mg3Bi1.49Sb0.5Te0.01 and Mg3.1Li0.003Bi1.49Sb0.5Te0.01 single crystals.51,52 Copyright 2020, Elsevier. Copyright 2024, American Chemical Society. (c) Synthesis process for mechanical alloying and sintering of Mg3(Sb,Bi)2.66,67 Copyright 2024, American Chemical Society. Copyright 2020, Tsinghua University Press. (d) Schematic diagram of the used 3D printer with a picture of the printed TE legs and their assembly into the device.63 Copyright 2025, The American Association for the Advancement of Science. | ||
Single crystals, with fewer grain boundaries, are expected to exhibit excellent near-room-temperature properties in principle, as GB effects can significantly limit carrier mobility µH and TE properties below 500 K.49,50 As shown in Fig. 2b, the Bridgman method enables the controlled growth of high-quality Mg3(Sb,Bi)2 single crystals with precise stoichiometry and low defect densities.51–53 For example, Mg3.1Li0.003Bi1.49Sb0.5Te0.01 achieved an excellent zT of 1.05 at 300 K by suppressing Mg inclusion during single crystal growth, highlighting their competitiveness for cooling applications.52 However, compared to polycrystalline synthesis, this method requires precise temperature control and slow growth rates, leading to higher energy consumption and limited productivity.
As illustrated in Fig. 2c, the combination of mechanical alloying (MA) with either hot pressing (HP) or discharge plasma sintering (SPS) offers a relatively simple and controllable approach for synthesizing Mg3(Sb,Bi)2.54–57 Single-phase n-type Mg3(Sb,Bi)2 were successfully synthesized using a multi-step ball milling process, achieving a remarkable peak zT value of 1.5 at 716 K.33 Particularly in Bi-rich compositions, the nanostructures induced by MA promote interface potential scattering of phonons, significantly suppressing the bipolar effect at high-temperatures.54 Nevertheless, like other specialized techniques, this method faces challenges for large-scale implementation due to high processing requirements.
Despite the many advantages of these preparation methods, developing novel Mg3(Sb,Bi)2-based materials with superior formability and excellent TE performance still faces many obstacles. Given the excellent plasticity of Mg alloys,58,59 deformation processing (e.g., stretching, rolling) offers a promising strategy to further optimize the microstructure and TE properties. Specifically, rolling refines grain boundaries, reduces porosity, and induces texture. These are critical for improving µH and mechanical strength. Furthermore, the enhanced plasticity and mechanical properties of Mg3Bi2 provide a solid foundation for exploring its potential in flexible TE device applications.60–62 Leveraging these properties enables future integration of Mg3Bi2 into flexible platforms, boosting η and device flexibility.
Beyond established methods, emerging additive manufacturing methods such as 3D printing (Fig. 2d) may offer significant potential for fabricating complex and gradient structures in future research, as evidenced in the Bi2(Sb,Te)3 system.63,64 This approach uniquely enables the precise construction of complex geometries and facilitates direct multi-material integration of TE materials, electrodes, and substrates, thereby advancing compact, fully integrated device development.65
2.2. Progress in thermoelectric figure of merit
As summarized in Fig. 3, the maximum zT values of reported Mg3(Sb,Bi)2-based materials since 2018 highlight rapid advancements in the field and underscore their potential for practical applications. The peak zT of Mg3(Sb,Bi)2 has remained essentially in the range of 1 to 2 in recent years. While most Mg3Sb2-based materials achieve zT values greater than 1, several Mg3Bi2-based compositions report lower performance. Among them, the Mg3Bi1.49Sb0.5Te0.01 single crystals grown by the Bridgman method exhibited a peak zT value of 1.43 at 523 K. The n-type 0.1Nb/Mg3Sb1.5Bi0.49Te0.01 achieved a peak zT value of 2.04 at 798 K by introducing Nb/Ta impurities at GB modulation of charge carrier and phonon transport.68 Similarly, n-type Mg3.2In0.02Bi1.4Sb0.595Te0.005 with the homogenizing composition exhibited a zT of 1.27 at 500 K.69 Overall, the TE properties of p-type Mg3(Sb,Bi)2 are relatively weak due to the lower VB degeneracy.70 Despite this, p-type (Mg3.18Ag0.02Sb2)0.5(YbZn2Sb2)0.5 achieved a zT of 1.17 at 773 K through optimized preparation techniques.71 A synergistic strategy of band engineering combined with entropy engineering then boosted the peak zT of p-type (Mg3.07Ag0.03Sb2)0.5(YbZn1.2Cd0.8Sb2)0.5 to 1.34 and a zTave of 0.77 between 323 and 773 K, demonstrating unprecedented performance for p-type Mg3Sb2-based materials.72 In conclusion, n-type Mg3(Sb,Bi)2 demonstrates TE properties nearly twice those of its p-type counterpart, highlighting the need for greater optimization efforts for p-type materials. | ||
| Fig. 3 The relationship between the peak zT values of Mg3(Sb,Bi)2-based materials and the year of publication.35,51,52,68,74–98 | ||
In recent years, the rapid development of Mg3(Sb,Bi)2 has made it a promising alternative to Bi2Te3. Compared with Bi2Te3, Mg3(Sb,Bi)2 exhibits a lower cost due to its reduced need for the expensive Te element. Meanwhile, n-type Mg3(Sb,Bi)2 shows a good zT value of 0.9 at 300 K,73 making it more promising for solid-state cooling applications compared to Bi2Te3−xSex. Furthermore, Sb-rich compositions have good potential for power generation at medium temperatures, while Bi-rich compositions hold even more potential for cooling applications. The high cooling performance of Mg3Bi2-based alloys motivates future exploration of TE materials for cooling.
3. Optimization strategies for Mg3(Sb,Bi)2-based materials thermoelectric properties
Optimizing TE properties of Mg3(Sb,Bi)2 requires enhancing the power factor (PF) while suppressing κ. Considering the crystal structure, band structure, fundamental TE properties, and fabrication methods, key strategies include Mg content tuning, band structure engineering, optimizing mixed scattering of ionized impurities and phonons, and complemented are introduced by extensive external element doping. Specifically, TE properties can be improved by optimizing nH and µH to enhance σ, controlling Mg-related defects and tailoring electronic structures to increase the S, and introducing point defects and disordered structures to reduce lattice thermal conductivity (κl). These approaches collectively maximize the TE properties of Mg3Sb2−xBix materials, providing a systematic framework for achieving high η in waste heat recovery and solid-state cooling applications.3.1. Electrical conductivity
The increase in σ is mainly based on nH or µH.99,100 The σ can be described as eqn (1):101| σ = neµ | (1) |
 | ||
| Fig. 4 (a) The nH as a function of nominal composition Mg3+xSb1.5Bi0.5.105 (b) Illustration of the reduction in Eg due to alloying with Mg3Bi2.107 Copyright 2020, Elsevier. (c) Schematic nH dependence of zT for Mg3Sb2−xBix using the SPB model.109 Copyright 2020, The American Association for the Advancement of Science. (d) The µH and nH at 300 K as a function of the electronegativity difference |χQ − χMg| in n-type Mg3Sb1.5Bi0.5.74 Copyright 2018, John Wiley and Sons. (e) Doping atoms versus nH of Mg3(Bi,Sb)2-based compounds at room-temperature. (f) Temperature-dependent σ of Mg3(Sb,Bi)2-based materials.52,80,86,89,114,115 Band structures of (g) Mg24Sb16, (h) Pr1Mg23Sb15Se1, and (i) Nd1Mg23Sb15Se1.86 Copyright 2022, Elsevier. | ||
Chalcogen element doping (e.g., S, Se, Te) represents a common approach to enhance the properties of n-type Mg3(Sb,Bi)2. For example, S-doped Mg3Sb1.5Bi0.5 achieved a peak zT of 1.0 at 725 K, though its properties are inferior to Se- and Te-doped systems due to lower nH and µH, as shown in Fig. 4d.74 The decreasing electronegativity difference (|χQ − χMg|) from S to Te leads to enhanced nH and µH, ultimately improving TE properties. Subsequent studies revealed that Se doping offers environmental benefits, obtaining a peak zT of 1.24 at 498 K in Mg3.2Bi1.4Sb0.59Se0.01 and demonstrating its widespread prospect for low-temperature TE applications.87 Additionally, the combination of Te doping with defect control and GB optimization significantly enhances TE properties, underscoring the pivotal role of chalcogen doping in improving σ.112
In addition to anion doping with chalcogen elements, both rare-earth elements doping and single-crystal design enhance the σ of Mg3(Sb,Bi)2.56,78,81,113Fig. 4e demonstrates the higher nH achievable with rare-earth element doping, underscoring its potential for σ enhancement. Fig. 4f compares the temperature-dependent σ of single crystal and rare-earth elements doping.52,80,86,89,114,115 Elements like La and Ce act as promising dopants, enabling nH to approach the theoretical doping limit.89 In addition, the σ of Li–Te co-doped single crystals increased to 13 × 104 S m−1 from 6.2 × 104 S m−1 for Mg3.1Bi1.49Sb0.5Te0.01 at 300 K.52 To further understand the electron transport mechanism, the band structures of Mg24Sb16, Pr1Mg23Sb15Se1, and Nd1Mg23Sb15Se1 were calculated by first-principles calculation.86Fig. 4g shows that the CBM and VBM of pristine Mg3Sb2 are at different locations, which corresponds to indirect Eg semiconductors. As shown in Fig. 4h and i, Pr–Se and Nd–Se significantly increase the DOS near the EF, narrow the Eg, and enhance carrier transport, thereby improving the σ and PF. Ultimately, Mg3.17Pr0.03Sb1.5Bi0.49Se0.01 and Mg3.17Nd0.03Sb1.5Bi0.49Se0.01 achieved peak zT values of 1.67 and 1.74 at 700 K, which are 38% and 43% higher than that of Se-only doped sample, respectively. In conclusion, the optimized treatment of band engineering, doping, and defect control is crucial for improving nH and electrical transport properties in Mg3(Sb,Bi)2. The trade-off between nH optimization and bipolar effect suppression remains a key challenge, especially in high-temperature environments. Therefore, introducing deep-level dopants and leveraging energy filtering effects offer strategies to mitigate detrimental bipolar effects.
 | ||
| Fig. 5 (a) Temperature-dependent µH of Mg3.1A0.1Sb1.5Bi0.49Te0.01 (A = Fe, Co, Hf, and Ta).77 (b) Predicted upper-limit of the zT when grain boundary phase resistance is considered negligible (solid line).49 Copyright 2018, Royal Society of Chemistry. (c) SPS displacement as a function of sintering time. (d) Microstructural evolution of Mg3.2Bi1.5Sb0.498Te0.002Cu0.01.85 Copyright 2022, Springer Nature. (e) The EBSD orientation maps for Mg3.2Sb1.5Bi0.49Te0.01 samples sintered at 873 K and 1123 K.129 Copyright 2018, AIP Publishing. (f) and (g) The APT characterization of Nb0.1Mg3.05Sb1.99Te0.01.131 Copyright 2021, John Wiley and Sons. | ||
GB scattering severely limits room-temperature µH in Mg3(Sb,Bi)2.126,127 GB resistance reduces µH, leading to a decrease in the PF, restricting TE properties. Simulation of GB band offsets indicates that eliminating GB resistance could enhance the room-temperature zT of Mg3.2Sb1.5Bi0.49Te0.01 by over 60% (Fig. 5b), emphasizing the significance of GB engineering in enhancing zT.49 Optimizing sintering temperatures and incorporating a small amount of Cu have proven effective in GB engineering. As shown in Fig. 5c and d, optimal sintering produces dense microstructures with large grains and minimal porosity. Significant increase in grain size at 1073 K compared to 723 K sintering. The room-temperature peak zT of Mg3.2Bi1.5Sb0.48Te0.002Cu0.01 increased significantly to 0.9 due to the complete elimination of thermal GB resistance at low temperatures.85 Two-dimensional nanomaterials such as graphene have shown remarkable potential as GB additives. The introduction of graphene (G) can effectively regulate the space charge region at grain boundaries, reducing carrier transport barriers. For instance, in Mg3.24Sb1.5Bi0.49Te0.01/1.0 vol% G, the Eb decreased from 42 meV to 18 meV, enhancing room-temperature µH to 68 cm2 V−1 s−1.93 Additionally, graphene nanoplatelets (GNPs) have been reported to increase interface thermal resistance, reduce the temperature at grain boundaries, and optimize the interfacial S.128 Chemical doping and control of grain size effectively mitigate GB scattering. For example, indium doping significantly suppressed GB scattering and enhanced room-temperature µH.88 Moreover, the addition of transition metals such as Mo and Nb promotes grain growth via GB segregation, which reduces scattering and increases both µH and σ.68,95 For instance, Mo doping increased the average grain size of Mg3.16Mo0.04Sb1.5Bi0.49Te0.01 from 9.46 µm for Mg3.2Sb1.5Bi0.49Te0.01 to 18.07 µm.95 Similarly, introducing Nb impurities into Mg3Sb1.5Bi0.49Te0.01 improved the room-temperature zT by more than 200%, reaching an unprecedented high zT of 2.04 at 798 K.68 Grain size optimization also effectively suppresses GB scattering. As shown in Fig. 5e, the average grain size of Mg3.2Sb1.5Bi0.49Te0.01 increased from 1.0 µm to 7.8 µm,129 significantly reduced GB scattering, enhanced µH, and maintained stable κ.
The synergistic effects of grain size and texturing improved the σ of the layered Mg3.24Sb1.5Bi0.49Te0.01 structure, achieving a maximum zT of 1.71 and a high zTave of 1.12 perpendicular to the pressing direction (⊥P).94 Increasing the sintering temperature further enlarged grain sizes, reducing GB scattering and increasing the room-temperature σ by over 300%.130 Notably, the enhanced µH can be attributed to the weakening of GB scattering due to the grain growth and coarsening. Fig. 5f presents the scanning electron microscope (SEM) image of an atom probe tomography (APT) sample. Fig. 5g illustrates the three-dimensional distribution of Mg, Sb, Nb, and Te. These results indicate that the Nb fails to incorporate into the Mg3Sb2 matrix after doping but instead segregates along the GB in a metallic state. This segregation leads to the formation of a Nb-rich wetting layer, which promotes grain growth, therefore enhancing σ.131 Notably, the elimination of GB scattering in single crystals leads to superior TE properties.132 Demonstrating this advantage, Imasato et al.133 first synthesized n-type Te-doped Mg3Sb2 single crystals by employing a flux method followed by Mg vapor annealing. The elimination of GB resistance led to a remarkable improvement of over 100% in both µW and zT near-room-temperature.
While the aforementioned strategies have successfully enhanced the µH in Mg3(Sb,Bi)2, recent research has shifted toward gaining deeper, atomic-level insights into how defects and carriers interact locally. Fig. 6a–c show high-angle annular dark field (HAADF) and annular bright field (ABF) scanning transmission electron microscopy (STEM) images, along with the integrated differential phase contrast (iDPC) views of Mg3Sb2 single crystals. The iDPC imaging visualizes both Mg and Sb atoms in Mg3Sb2 single crystals.134 Furthermore, Fig. 6f illustrates the Fourier difference electron density maps (Fo − Fc) difference electron density map, confirming the presence of Mg cation vacancy defects and Frenkel defects.135 Meanwhile, the origin of the excellent TE properties is further explored by angle-resolved photoelectron spectroscopy (ARPES). Fig. 6g shows the constant energy diagrams of the ARPES of Mg3Bi2, Mg3Bi1.25Sb0.75, and Mg3Sb2 single crystals. The electronic states close to EF disappear from Mg3Bi2 to Mg3Sb2, and the valence band tops move significantly to higher binding energies, leading to a significant widening of the Eg.47 To elucidate the effect of doping on the local structure, synchrotron X-ray pair distribution function (X-PDF) was performed on Mg3.2(Bi0.7Sb0.3)1.99Te0.01, Mg3.2(Bi0.7Sb0.3)1.99Te0.01–Cu2Se 1.0%, and Mg3.2(Bi0.7Sb0.3)1.99Te0.01–Ag2Se 1.0%, as shown in Fig. 6f–h.136 In comparison to Mg3.2(Bi0.7Sb0.3)1.99Te0.01, the enhanced short-range ordering in the Cu2Se and Ag2Se composites, as evidenced by the narrowing of the characteristic peaks, provides a superior channel for carrier transport. These studies emphasize the crucial role played by GB engineering, defect control, and single-crystal methods in achieving superior µH. Since the cost-effective and controllable approach to achieve defect-free single crystals is still a major challenge, GB engineering combined with defect modulation strategies offers a preferred path to balance performance and cost.
 | ||
| Fig. 6 STEM images of Mg3Sb2: (a) HAADF, (b) ABF, and (c) iDPC images along [001] direction.134 Copyright 2021, Elsevier. (d) The Fo − Fc difference electron density map of n-type Mg3Sb2 before refining the interstitial position.135 Copyright 2023, American Chemical Society. (e) Series constant energy maps of Mg3(Sb,Bi)2.47 Copyright 2020, Royal Society of Chemistry Synchrotron X-PDF data as a function of r for (f) Mg3.2(Bi0.7Sb0.3)1.99Te0.01, (g) Mg3.2(Bi0.7Sb0.3)1.99Te0.01–Cu2Se 1.0 wt%, and (h) Mg3.2(Bi0.7Sb0.3)1.99Te0.01–Ag2Se 1.0 wt%.136 Copyright 2025, Elsevier. | ||
3.2. Seebeck coefficient
In addition to high σ, an excellent S value is essential to achieve excellent PF. The S can be expressed by eqn (2):137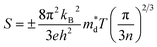 | (2) |
 | ||
| Fig. 7 (a) Schematic diagrams of the band structures and DOS for band convergence and band anisotropy.138 Copyright 2021, Springer Nature. Electrical properties of (b) n-type and (c) p-type Mg3Sb2.109,138,140 Copyright 2020, The American Association for the Advancement of Science. Copyright 2021, Springer Nature. (d) Strain-dependent m* at conduction band edge.142 Copyright 2020, The American Association for the Advancement of Science. (e) Defect formation energies of the lanthanide substitution on the Mg1 site.157 Copyright 2020, John Wiley and Sons. (f) Temperature-dependent S of Mg3(Sb,Bi)2-based materials.52,80,86,89,114,115 (g) Schematic diagram of Eb near the GB and metal–semiconductor interface region.68 Copyright 2023, Springer Nature. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10,29 have limited the commercial development of Mg3(Sb,Bi)2 TE devices. Strategies have primarily focused on optimizing the band structure and implementing effective doping to enhance nH.
10,29 have limited the commercial development of Mg3(Sb,Bi)2 TE devices. Strategies have primarily focused on optimizing the band structure and implementing effective doping to enhance nH.
As illustrated in Fig. 7d, under the influence of strain, the m* of the DOS at the CBM of Mg3Sb2 exhibited a linear decreasing trend.142 Theoretically, a reduction in m* leads to a lower S. However, when acoustic phonon scattering dominates, the maximum PF is proportional to the ratio of Nv/m*. Thus, a smaller m* corresponds to a higher µ and a higher PF.107 Meanwhile, biaxial strain can effectively enhance band degeneracy. For example, p-type Mg3Sb2 achieved a maximum S in the plane from 375 µV K−1 under unstrained to 623 µV K−1 under 2.5% biaxial strain at 700 K.143 Furthermore, in p-type Mg3(Sb,Bi)2, the introduction of elements such as Li,144 Zn,31 Cd,32 Ag,145 and Na146 at the cationic sites to replace Mg can effectively regulate the nH and µH. Meanwhile, multiple co-doping at the Mg sites simultaneously enhances both carrier transport and phonon scattering.32,147 For instance, a peak zT value of 0.87 was achieved in Mg1.95Na0.01Zn1Sb2 at 773 K through Na–Zn co-doping.98 First-principles calculations revealed that Ag–Bi co-doping shifts the EF into the VB and narrows the Eg. This facilitates hole generation, increasing nH from 3.50 × 1017 cm−3 to 7.88 × 1019 cm−3. As a result, Mg2.94Ag0.06Sb1.9Bi0.1 achieved an impressive PF of 778.9 µW m−1 K−2 at 723 K.92 Nanostructured bulk materials fabricated using high-energy ball milling combined with SPS exhibit uniform microstructures that enhance S.148
Band structure modulation via doping effectively enhances the S.154–156 The calculated formation energies for various dopants are summarized in Fig. 7e. La and Ce act as effective n-type dopants, exhibiting high donor transition energy levels significantly above the CBM.157Fig. 7f compares the S for single crystals and rare earth element doping.52,80,86,89,114,115 The S of all rare-earth doping increases with temperature, indicating degenerate semiconductor behavior. Moreover, the S of single-crystal Mg3.1Li0.003Bi1.49Sb0.5Te0.01 decreases with rising temperature owing to the presence of bipolar effect.52 The significant enhancement of S in Mn-doped Mg3Sb1.5Bi0.5 was attributed to the presence of multiple conduction bands with high band degeneracy.75 Furthermore, the addition of transition metals such as Nb/Ta can scatter low-energy carriers more efficiently by changing the interfacial barrier (Eb). As shown in Fig. 7g, the interfacial barrier decreases from Eb1 to Eb2, improving the tendency for more high-energy carriers to pass through, which leads to an increase in the S.68 In summary, achieving an optimal S not only requires precise control of nH but also demands a comprehensive strategy involving band engineering, defect chemistry, and interface optimization. Future research should further explore the interplay between band structure modulation and microstructural design to achieve an ideal balance between the S and σ.
3.3. Lattice thermal conductivity
The relatively high κ of Mg3.2Bi2 limits its TE properties. The minimum value of κ is derived through Cahill and Ball using eqn (3):158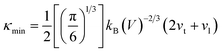 | (3) |
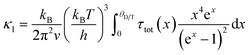 | (4) |
 | ||
| Fig. 8 Neutron dynamical structure factor S(|Q|, ω) of (a) Mg3Sb2 and (b) Mg3Bi2.161 Copyright 2020, The American Association for the Advancement of Science. (c) Atom-projected phonon DOS of Mg3Sb2 and Mg3Bi2.160 Copyright 2023, American Chemical Society. (d) The Grüneisen parameter, and (e) scattering rate of anharmonic for 300 K of Mg3Sb2.160 Calculated phonon spectra of (f) Mg24Sb16, (g) YMg23Sb15Cl, (h) YMg23Sb14ClTe, and (i) the sound velocity comparison.162 Copyright 2025, Elsevier. | ||
In Mg3.2Bi2, partial substitution of Bi with Sb effectively reduces κl through alloy scattering. The κl reduced from 9.6 W m−1 K−1 for Mg3.2Bi2 to 3.2 W m−1 K−1 for Mg3.2Bi1.298Sb0.7Te0.002, leading to a high zT value of 0.9 at 350 K.35 Additionally, alloy scattering induced by compositional variations reduced the κl of Mg3.2Bi1.4Sb0.595Se0.01 to 0.6–0.7 W m−1 K−1 in the low-temperature range, demonstrating excellent room-temperature TE properties.87 Furthermore, the Sb–Bi lattice disorder introduced during alloying can further reduce κl to 0.44 W m−1 K−1 at 716 K, approaching the Cahill minimum κl.33 Point defects and lattice defects also significantly contribute to κl reduction by scattering phonons of different wavelengths. For instance, introducing heavy-mass Co enhanced phonon scattering, enabling Mg3.15Co0.05(Sb0.3Bi0.7)1.99Te0.01 to achieved a minimum κl of 0.64 W m−1 K−1 at 450 K and a high peak zT of 1.03 at 525 K.79 Furthermore, Mg3.18Y0.02Sb1.5Bi0.49Se0.01 leveraged point defects and dense grain boundaries to enhance phonon scattering, achieving a κl reduction. This effect yielded a theoretical η of 13.8% and an excellent zT of 1.87 under a 450 K temperature gradient.91 Doping with rare-earth elements such as Gd and Pr can further optimize nH and κ. By synergistic tuning the electronegativity difference, mass fluctuations, and point defects, Gd-doped Mg3.065Sb1.3Bi0.7Gd0.015 achieved a high zT of 1.55 at 700 K.82 Similarly, Pr-doping not only increased nH but also reduced κl to 0.42 W m−1 K−1, leading to a peak zT of 1.70 for Mg3.2Pr0.02Sb1.5Bi0.5 at 725 K.76
Nanostructured materials, through the introduction of interfaces and defects, can effectively scatter a wide range of phonons while maintaining favorable electronic transport properties.148,169,170 For instance, nanostructured Mg3Sb2 and Mg3Sb1.8Bi0.2 exhibited respective peak zT enhancements of 54% and 56% at 773 K, primarily due to preferential phonon scattering at interfaces.81,84Fig. 9a demonstrates a transmission electron microscopy (TEM) image of Mg2.94Ag0.06Sb1.9Bi0.1, indicating the presence of nano-precipitates and lattice distortion. Furthermore, lattice distortion was verified by high-resolution transmission electron microscopy (HRTEM) (Fig. 9b). The κl was effectively decreased to 0.48 W m−1 K−1 by phonon scattering at point defects, distorted lattices, and nanoprecipitates, as illustrated in the schematic diagram Fig. 9c.92 Similarly, Bi nanoprecipitation and lattice distortion significantly enhance phonon scattering. Combined with shorter relaxation times, this ultimately reduced κl for Mg3.18Ce0.02SbBi0.97Te0.03 to 0.51 W m−1 K−1 at 673 K.171 In Mg3.24Sb1.5Bi0.49Te0.01/1 vol% G, an equivalent nanoparticle scattering mechanism resulted in a moderate reduction in κl, achieving a peak zT value of 1.30 at 773 K.93 Furthermore, Mn doping suppresses Mg evaporation during the sintering process, helping to decouple the electrical and thermal transport. As shown in Fig. 9d and e, low-magnification TEM imaging reveals the grains fractured along the crystals. Fig. 9f presents an HRTEM image that allows the observation of nanoprecipitates and nanostructured GB grain boundaries, both contributing to lower κl.34 Dislocation and strain engineering have shown remarkable success in further optimizing phonon transport mechanisms. The aggregation and evolution of vacancies caused by Bi defects into dislocations enhanced phonon scattering, resulting in a peak zT of 1.82 for Mg3.16Y0.01Sb1.5Bi0.48 at 473 K.90 The presence of dislocations and strains in Mg3.2Sb1.5Bi0.49Te0.01–4% Mg and Mg3.2In0.005Sb1.5Bi0.49Te0.01 reduced the phonon relaxation time, leading to a remarkable decrease in κl.67,172 Phonon transport is further constrained by strain-induced disorder and lattice anharmonicity, which reduce phonon relaxation times. Additionally, as shown in Fig. 9g, dislocation strain and defect-induced mass fluctuations further suppress phonon transport. Mg3.22Ho0.03Sb1.5Bi0.5 sintered at 1073 K achieved a low κl of 0.47 W m−1 K−1 at 750 K.130
 | ||
Fig. 9 HRTEM images of (a) Mg2.94Ag0.06Sb1.9Bi0.1 and (b) rectangular area indicated in (a). (c) Schematic diagram of point defects strengthening phonon scattering.92 Copyright 2025, Elsevier. (d) and (e) Low-magnification TEM image of the sample with Sb content of Mg3.02Mn0.01Sb1.2Bi0.79Te0.01, with an inset depicting the electron diffraction patterns along the [1![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 1 1![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ] direction. (f) HAADF image of the coarse-grained boundary.34 Copyright 2018, John Wiley and Sons. (g) Schematic diagram demonstrating the combined effect of strain engineering and scattering mechanisms on the modulation of phonon and charge carrier transport.130 (h) The lph dependence of accumulated κl and schematic diagram of all-scale hierarchical structures. Copyright 2024, Elsevier. ] direction. (f) HAADF image of the coarse-grained boundary.34 Copyright 2018, John Wiley and Sons. (g) Schematic diagram demonstrating the combined effect of strain engineering and scattering mechanisms on the modulation of phonon and charge carrier transport.130 (h) The lph dependence of accumulated κl and schematic diagram of all-scale hierarchical structures. Copyright 2024, Elsevier. | ||
Under the Debye model approximation, the lph can be expressed as eqn (5):
| lph = vsτ(x) | (5) |
Fig. 9h illustrates the relationship between the lph and the accumulated κl. Lattice defects of different scales scatter phonons of various frequencies and lead to extremely low κl. To effectively employ a multi-scale defects, the dimensions of the introduced scattering centers should be smaller than the κl and larger than the carrier mean free path.173 These studies reveal the complex mechanism of phonon transport in Mg3(Sb,Bi)2 materials and offer a theoretical basis for reducing κl by modulating phonon scattering. Future studies should integrate multiscale defect optimization with advanced characterization techniques to simultaneously balance electrical/thermal properties and advance TE performance limits.
4. Mg3(Sb,Bi)2-based thermoelectric device
Despite high zT values and promising TE properties of Mg3(Sb,Bi)2-based materials, there remain numerous challenges to overcome before achieving large-scale practical applications. A high zT value alone does not ensure outstanding device performance, particularly when sustaining high η over a wide temperature range is required. The η of the device is determined by the zTave across the entire operating ΔT, expressed by eqn (6):174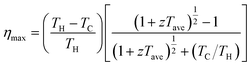 | (6) |
The manufacture of TE devices entails more intricate scientific and engineering issues than optimization of material properties. Appropriate interface and structural design strategies, including the connection technology, selection of barrier layers, multi-stage optimization, and size matching between n-type and p-type TE legs, are essential for enhancing device performance.150,175–178Fig. 10a illustrates a single-leg structure consisting of a Cu electrode, hot- and cold-side solders, a metal contact layer, a barrier layer, and a TE leg that can be used to test the performance of a single material. The traditional module (Fig. 10b) is suitable for η in terms of a combination of n- and p-type materials over a wide temperature range. However, optimization of leg size matching and interfacial stability between different materials is required. Though thermal expansion compatibility and interfacial contact resistance must be addressed, the segmented module (Fig. 10c) maximizes the η of the device. Additionally, issues such as thermal stability, mechanical properties, device assembly techniques, and manufacturing costs are key obstacles to address for large-scale applications.175,179–181Fig. 10d summarizes the η of Mg3(Sb,Bi)2-based TE devices at different ΔT. Mg3.2Sb1.5Bi0.49Te0.01 + 0.25 vol% TiO2−n single-leg achieved a η of 15% at ΔT of 470 K, which fully demonstrates its excellent potential for application.11,68,182–186
 | ||
| Fig. 10 Schematic diagram of the structure of (a) single-leg, (b) traditional module, and (c) segmented module. (d) The η of Mg3(Sb,Bi)2-based TE devices as a function of ΔT.11,68,182–188 | ||
4.1. Connection technology
The advancement of high-performance TE modules critically depends on achieving low-resistance and thermally stable interconnects between TE materials and electrodes.183,189–192Fig. 11a shows the joining of Al–Si–Cu alloys by brazing combined with pressure-assisted sintering under 830 K and 10 MPa. However, high-temperature leads to the volatilization of Mg and Zn, resulting in the formation of cracks in the TE legs. The Mg3.2Bi1.497Sb0.5Te0.003/Bi0.4Sb1.6Te3 device using SnBi solder retained 98.3% of its original performance after 3000 current cycles, demonstrating the excellent thermal fatigue resistance of the solder.193 In a separate case, the Pb-free commercial solder Ag56Cu22Zn17Sn5 was used to bond Fe7Mg2Ti and Fe7Mg2Cr to Cu electrodes at approximately 903 K. The resulting joints maintained shear strength greater than 30 MPa and contact resistivity below 10 µΩ cm2 after aging at 673 K for 15 days, indicating exceptional high-temperature stability.194 | ||
| Fig. 11 (a) Schematic of conventional soldering technique.197 Copyright 2023, Oxford University Press. (b) Mg3Sb2-based modules assembled by Ag composite paste under low-temperature and low-pressure conditions.197 (c) Mass change and heat flow of Ag composite paste during thermal cycling.197 (d) Analysis of the resistance changes observed before and after soldering with Ag composite paste.197 (e) Schematic diagram of TE device assembly by Ag nanoparticle sintered bonding.183 Copyright 2024, Springer Nature. (f) and (g) Characterization of the nano-Ag sintered connection layer.183 | ||
Compared with the traditional soldering technique, the novel composite Ag paste soldering technique effectively avoids the problem of device failure due to TE arm damage caused by pressure and temperature limitations. As shown in Fig. 11b, the Ag paste was uniformly applied to the electrode before heating and heated at 523 K in argon for 1.5 h. The connection between the TE legs and Cu electrode was realized after heating and exhibits good thermal stability (Fig. 11c).195,196Fig. 11d compares the resistance change before and after soldering of the new Ag composite paste to avoid impairing the TE material and interface, and the resistivity is consistent with the predicted values.197 This low-temperature, low-pressure connection technique utilizing Ag composite adhesive demonstrates effective performance. In addition, the efficiency of n-type single-leg Mg3Sb2-based devices prepared using a low-temperature nano-Ag sintering process is up to 13.3%.183 Nano-Ag sintering was performed at 573 K and 10 MPa, as shown in Fig. 11e. The Ag nanoparticles exhibited an almost spherical morphology and were encapsulated by an organic layer that inhibits oxidation and self-adhesion (Fig. 11f). The sintered nano-Ag network structure was homogeneously dispersed and well-connected at the interface (Fig. 11g). Also, this process can be combined with phase diagram engineering, which exhibits great potential for interconnect packaging of a wide range of high-performance TE power generation devices.
4.2. Barrier layer
To maintain the robust operation of TE modules, a stable interface barrier layer with low contact resistance is required.198–203 However, interface losses and failures between TE materials and electrodes remain significant challenges.192 In the preparation of cooling modules for Mg3(Sb,Bi)2, the selected diffusion barrier materials should exhibit excellent chemical stability, compatible thermal expansion coefficients (CTE), robust bonding strength, low contact resistance, and comparatively low processing temperatures to avoid degradation of Mg3(Sb,Bi)2. Wu et al.194 systematically investigated interfacial reactions and diffusion behavior through high-throughput experiments. Their study identified Fe, Ni, Mo, W, and Co as pure TE interface materials with excellent thermal stability. However, when bonding performance with Mg3Sb1.5Bi0.5 was evaluated after 15 minutes at 773 K, Nb, Mo, W, and Co were excluded due to their tendency to form intermetallic compounds and inert diffusion characteristics, which led to inferior interfaces.The Fe/Ni layers have been confirmed as proper barrier layers for Mg3(Sb,Bi)2.188,204,205 Taking the Fe contact layer as an example, the microstructure and compositional line scan of the Mg3.2Sb1.5Bi0.49Te0.01/Fe interface show that the mixed region is primarily enriched with Fe and Mg (Fig. 12a). Due to the minimal solubility of Fe and Mg, the excess Mg in the mixed layer is residual and destabilizing. Aging the Mg3.2Sb1.5Bi0.49Te0.01/Fe interface at 523 K for 400 h further elucidated its thermal chemical stability. As shown in Fig. 12b, the thickness of the Fe–Sb layer and the Sb-rich Mg3Sb2 layer increased to 1 µm and 0.7 µm, respectively. The overall thickness of the reaction layer exhibits a favorable linear relationship with the square root of time (Fig. 12c), indicating that the interface layer growth is governed by diffusion. Subsequent studies found that Mg2Cu was a suitable barrier layer for Mg3(Sb,Bi)2 compared to Ni/Fe electrodes.206 The ternary phase diagram of Cu–Mg–Sb(Bi) indicated that Mg2Cu is thermodynamically stable within the Mg–Mg2Cu–Mg3Bi1.5Sb0.5 phase region, as shown in Fig. 12d. Thus, it effectively prevents the diffusion of Cu into Mg3(Sb,Bi)2. The contact resistivity (ρc) of the Mg3.2Bi1.4975Sb0.5Te0.0025 legs, measured using the four-probe method, was as low as 12 mΩ cm2 (Fig. 12e).207Fig. 12f demonstrates the maximum thermal stress (70 MPa) in the contact layer of Cu/Mg2Cu/Mg3.2Bi1.4975Sb0.5Te0.0025 at a constant ΔT and optimized current, indicating that the material is used as an optimal buffer layer to relieve the thermal stresses.
 | ||
| Fig. 12 The composition profile of Mg3.2Sb1.5Bi0.49Te0.01/Fe interface after an aging treatment of (a) 0 h and (b) 400 h. (c) The thickness of the reaction layer as a function of aging time.206 Copyright 2020, Elsevier. (d) Mg–Mg2Cu–Mg3Bi1.5Sb0.5 three-phase region. (e) Contact resistance measurement of the interfacial layers. (f) Simulated thermal stress distribution near the barrier layers of the module during operation.207 Copyright 2022, Elsevier. (g) The energy dispersive spectroscopy mapping and line scanning results showing the elemental distribution of Ti foil sintered with Mg3(Sb,Bi)2. (h) Changes in power generation performance of a Mg3Sb2-based single-leg during thermal cycling testing.208 Copyright 2024, Springer Nature. | ||
Through the dynamical transition from a high-temperature substable phase to a low-temperature stable phase, the limitations of thermodynamic equilibrium can be overcome, and ideal materials for TE barrier layers are identified. By comparing the kinetic properties of the reaction and diffusion behavior of Mg3(Sb,Bi)2 and Ti during sintering and operation, the Ti foil/Mg3(Sb,Bi)2 connectors present excellent sintering activity and inert behavior.208 As shown in Fig. 12g, the interface reaction product between the Ti foil and Mg3(Sb,Bi)2 was a MgTiSb compound with a thickness of 1 µm when held at 1023 K for 15 min. Therefore, it can function effectively as a barrier layer for Mg3Sb2-based materials. As illustrated in Fig. 12h, the power generation performance of a Mg3Sb2-based single-leg TE device remained stable during 200 thermal cycles lasting 330 h, with the heat source temperature cycling between 473 K and 748 K, exhibiting excellent stability and reliability. The interface ρc reduced to below 5 µΩ cm2, allowing the Mg3Sb2-based module to achieve a high η of 11% at a ΔT of 440 K, surpassing most advanced medium-temperature TE modules. Advances in novel connection techniques and the development of stabilizing barrier layers have greatly improved the reliability, efficiency, and performance of Mg3(Sb,Bi)2-based TE devices. These strategies address critical issues related to interfacial loss, contact resistance, and thermal stress.
4.3. Thermoelectric modules
From a device design perspective, output power and reliability are the two core factors influencing practical applications. High output power ensures superior η and expands the applicability of TE modules, while good stability guarantees device reliability and long-term operation under diverse environments.209–211 One of the main challenges to the thermal stability of Mg3Sb2−xBix lies in the loss of Mg at elevated temperatures.212,213 Significant progress has been achieved in enhancing the thermal stability of Mg3(Sb,Bi)2 through various thermal treatments, chemical reactions, and surface modification techniques. For instance, BN-coated Mg3.2Sb1.5Bi0.49Te0.01 achieved a high η of 11.1% under a ΔT of 473 K.182 Additionally, annealing processes have been demonstrated to significantly mitigate material degradation. Applying Mg vapor annealing, surface coatings, and low-temperature annealing effectively suppresses non-equilibrium defects, thereby enhancing structural stability.214,215 Traditional TE technology at room temperature has long been dominated by Bi2Te3-based materials. However, the scarcity of Te poses significant limitations for future large-scale applications.216 Developing the next generation of Te-free materials is therefore crucial to mitigating potential raw material supply bottlenecks and achieving sustainable development. The one-step hot-pressing method has been shown to realize strong bonding between the barrier layers and Mg3Sb2-based TE materials, effectively reducing contact resistance, as shown in Fig. 13a. This method enabled a η of 10.6% within a ΔT range of 100 to 500 °C.11 As shown in Fig. 13b, the 0.1Nb/Mg3Sb1.5Bi0.49Te0.01 single-leg apparatus can run continuously for more than 120 h at ΔT = 470 K with η higher than 14%.68 In addition, Mg3(Sb,Bi)2 and p-type MgAgSb modules designed by GB engineering, and geometrical optimization achieved η of 7.3% and 8.2%, respectively,185,217 and the performance remained stable after 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 thermal cycles.218 These studies further demonstrate the reliability and high η values of the Te-free modules, achieving competitive performance in the low temperature range.
000 thermal cycles.218 These studies further demonstrate the reliability and high η values of the Te-free modules, achieving competitive performance in the low temperature range.
 | ||
| Fig. 13 (a) Single-leg η measurement architecture under high ΔT, with operational setup visualization.11 Copyright 2019, Elsevier. (b) Comparison of the maximum η among the single-leg devices in 0.1Nb/Mg3Sb1.5Bi0.49Te0.01 with other TE modules.68 Copyright 2023, Springer Nature. (c) Module fabrication and cooling measurement setup for n-type Mg3.2Bi1.5Sb0.498Te0.002Cu0.01 and p-type α-Mg0.99Cu0.01Ag0.97Sb0.99.85 Copyright 2022, Springer Nature. (d) Cooling performance of p-type MgAg0.97Sb0.99 and n-type Mg3.6Y0.003Sb0.6Bi1.4.218 Copyright 2022, Royal Society of Chemistry. (e) Design of the setup used to characterize the cooling performance of the Mg3Bi2-based double-stage TEC.9 Copyright 2025, Innovation Press. (f) and (g) Solid-state sensor device and applications of Mg3.19Co0.01Sb0.5Bi1.49Te0.01.228 Copyright 2024, Elsevier. | ||
The CTE mismatch between the two TE legs can lead to stress accumulation during repeated thermal cycles, ultimately degrading performance.219,220 For medium- and high-temperature applications, minimizing the CTE mismatch is an important strategy for improving device performance. For example, p-type MgAgSb and n-type Mg3.49Y0.01Sb1.5Bi0.5 exhibited a CTE mismatch of only 0.95%, demonstrating good compatibility between the two leg materials within TE modules.221 Furthermore, Peltier modules mediated by ultrafine grains and nanoporous structures in α-MgAgSb and Mg3.2(Bi,Sb)2 achieved a maximum ΔT of 52 K and a coefficient of performance (COP) of 8.3 under high-temperature conditions.222 For medium- and high-temperature modules, using n- and p-type legs from the same material system can reduce CTE mismatches and simplify electrode material selection.223 For example, n-type Mg3.2Sb1.5Bi0.49Te0.01Mn0.01 and p-type Na0.005Mg2.995Sb1.5Bi0.5 exhibited comparable thermal expansion properties, achieving a theoretical η of approximately 9.5% under a ΔT of 300 K to 773 K.224
Besides power generation, TE cooling is equally important.225–227 Full-scale TE cooling devices with 7 pairs, 31 pairs, and 71 pairs were fabricated by joining an n-type Mg3.2Bi1.4975Sb0.5Te0.0025 layer with an effective and reliable Mg2Cu diffusion barrier to a p-type (Sb0.75Bi0.25)2(Te0.97Se0.03)3.207 Nevertheless, despite the progress in high-performance single-pair and full-scale TE coolers, the development of TE devices entirely free of (Bi, Sb)2Te3-based TE devices remains limited. Inspired by this, a cooling module was constructed by combining n-type Mg3.2Bi1.5Sb0.5 with p-type α-MgAgSb through microstructure design (Fig. 13c). It achieved an η value of 2.8% (ΔT = 95 K), a maximum cooling ΔT of 56.5 K, and a maximum COP (COPmax) of 2.6 (ΔT = 10 K).85 Furthermore, a TE device composed of high-performance n-type Mg3.6Y0.003Sb0.6Bi1.4 and p-type MgAg0.97Sb0.99 compounds exhibits a maximum ΔT of 72 K with Th of 347 K (Fig. 13d).218 A 7-pair TE cooling module fabricated using p-type Bi0.5Sb1.5Te3 and n-type MgAg0.97Sb0.99 reached a maximum ΔT of 100 K at Th = 400 K, exhibiting COPmax of 8.9, 7.5, and 3.2 at Tc of 0 K, 5 K, and 10 K, respectively.48 The subsequently developed multistage refrigeration device follows the design of heat distribution where the cooling capacity of the next stage is larger than the heat dissipation of the previous stage for realizing the cooling stage by stage.9 The temperature field simulation results show that the seven-stage refrigeration device of Mg3.1Sb0.497Bi1.5Te0.003/(Bi,Sb)2Te3 can realize step-by-step refrigeration by the layer-by-layer stacked “pyramid” configuration. As shown in Fig. 13e, the Mg3.1Sb0.497Bi1.5Te0.003/(Bi,Sb)2Te3 two-stage refrigeration device achieved a maximum cooling capacity of 3.0 W and a maximum ΔT of 103.2 K, which is comparable to the commercial Bi2Te3 alloy-based device.
The potential interdisciplinary applications of Mg3(Sb,Bi)2 have also been explored. Fig. 13f shows an infrared thermogram of a respiratory mask equipped with Mg3.19Co0.01Sb0.5Bi1.49Te0.01-based TE sensor, demonstrating its potential for respiratory monitoring applications. Additionally, Fig. 13g reveals that five common liquids have different contact angles on the polyurethane film, indicating the TE sensor can be used for liquid identification.228 These advancements suggest that Mg3(Sb,Bi)2-based modules have the potential to rival, or even surpass, commercial Bi2Te3 systems in performance, offering an environmentally friendly and efficient alternative that represents a significant step forward in the sustainable development of TE technology.
5. Challenge and outlooks
Mg3(Sb,Bi)2-based materials, as a resource-abundant and environmentally friendly class of Zintl-phase materials, have attracted widespread focus and discussion owing to their excellent thermoelectric (TE) properties and low cost. Compared to conventional Bi2(Te,Se)3-based TE materials, Mg3(Sb,Bi)2 exhibits high conversion efficiency (η) near-room-temperature while addressing the scarcity and high cost of Te, presenting great potential for both research and practical applications. We summarize the key factors for Mg3(Sb,Bi)2-based materials and devices in Fig. 14. Before large-scale commercialization, Mg3(Sb,Bi)2 must overcome several key challenges in practical implementation, as outlined below: | ||
| Fig. 14 Challenges and optimization aspects of Mg3(Sb,Bi)2.181,186,215 Copyright 2023, John Wiley and Sons. Copyright 2023, Oxford University Press. Copyright 2024, Springer Nature. Copyright 2023, Elsevier. | ||
(1) In terms of fabrication, methods such as mechanical alloying, discharge plasma sintering, and hot-pressing have been widely employed for producing Mg3(Sb,Bi)2 bulk materials. These techniques not only improve material densification but also effectively enhance lattice uniformity and TE properties. Furthermore, advancements in single-crystal fabrication technologies have drastically reduced electrical resistance caused by grain boundary scattering, further improving carrier mobility (µH) and η. Current fabrication techniques are mostly limited to laboratory-scale production, which cannot meet the demands of large-scale industrial manufacturing. Developing cost-effective and environmentally friendly synthesis methods is essential for the commercialization of these materials.
(2) By adjusting the Bi/Sb ratio, the bandgap of Mg3(Sb,Bi)2 can be flexibly tuned to optimize carrier concentration and µH, thereby enhancing electrical transport properties. Additionally, doping with rare-earth elements has further improved the Seebeck coefficient (S) and power factor, with some studies reporting a peak zT exceeding 2.0 across a wide temperature range from room temperature to medium- and high-temperatures. Achieving the ideal balance between electrical conductivity with S and minimizing undesirable effects such as bipolar effects remains a key challenge in realizing the full potential of practical TE applications.
(3) The introduction of multiscale defects significantly reduces lattice thermal conductivity (κl) by effectively suppressing thermal conduction through phonon scattering, resulting in outstanding TE properties in the low- and medium-temperature range. Critically, while enhanced phonon scattering reduces κl, charge carrier mobility µH must be considered to maintain excellent electrical transport properties. Future research should screen Mg3(Sb,Bi)2-based compositions with intrinsically low κl through combined first-principles calculations and high-throughput methods, while exploring emerging phonon transport mechanisms like liquid-like ionic diffusion and topological phonon states.
(4) Mg3(Sb,Bi)2-based TE devices exhibit significant prospects for both power generation and cooling applications. To narrow the gap between theoretical and actual η, researchers have systematically developed interface barrier layer design and connection process optimization strategies. While Fe/Ni, Mg2Cu, and Ti barrier materials exhibit excellent contact resistance, chemical stability, and thermal expansion coefficient matching, long-term reliability under thermal cycling remains challenging due to interfacial reactions. Novel connection processes like low-temperature Ag sintering and composite Ag paste soldering prevent degradation from temperature-pressure coupling while enabling robust bonding, yet face scalability challenges requiring improved uniformity and long-term thermal cycling reliability.
(5) Enhancing the long-term stability of TE modules and optimizing device structural design are central to the practical utilization of TE devices. By optimizing p-type MgAgSb and n-type Mg3(Sb,Bi)2 with matched thermal expansion coefficients, the accumulation of thermal stresses can be significantly reduced. The combination of geometrical optimization and surface coating technology improves the long-term stability of the modules. In addition, emerging manufacturing technologies such as 3D printing are expected to overcome the bottleneck of mass production. With its excellent performance and scalable applications, Mg3(Sb,Bi)2 is expected to be a core candidate for the next generation of TE materials, which will support the advancement of clean energy technology.
Author contributions
Yu Yan: writing – original draft, data curation, formal analysis, conceptualization. Wen Zhang: validation, visualization. Huijun Kang: writing – original draft, writing – review & editing, supervision, investigation, conceptualization. Zongning Chen: writing – review & editing, investigation. Enyu Guo: writing – review & editing, investigation. Rongchun Chen: writing – original draft, writing – review & editing, supervision. Tongmin Wang: writing – original draft, writing – review & editing, supervision, investigation, conceptualization.Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 52271025, 52501042, U25A20222 and U22A20174), the Postdoctoral Fellowship Program of China Postdoctoral Science Foundation (GZC20240173), the Science and Technology Planning Project of Liaoning Province (2023JH2/101700295 and 2025-BS-0002), and the Innovation Foundation of Science and the Technology of Dalian (No. 2023JJ12GX021).References
- X. L. Shi, J. Zou and Z. G. Chen, Chem. Rev., 2020, 120, 7399–7515 Search PubMed.
- R. Moshwan, L. Yang, J. Zou and Z. G. Chen, Adv. Funct. Mater., 2017, 27, 1703278 Search PubMed.
- E. S. Toberer and G. J. Snyder, Nat. Mater., 2008, 7, 105–114 CrossRef PubMed.
- Z. Han, J.-W. Li, F. Jiang, J. Xia, B.-P. Zhang, J.-F. Li and W. Liu, J. Materiomics, 2022, 8, 427–436 CrossRef.
- Z. Soleimani, S. Zoras, B. Ceranic, S. Shahzad and Y. Cui, Sustainable Energy Technologies and Assessments, 2020, 37, 100604 Search PubMed.
- L. Yang, Z. G. Chen, M. S. Dargusch and J. Zou, Adv. Energy Mater., 2017, 8, 1701797 CrossRef.
- J. Ding, W. Zhao, W. Jin, C. Di and D. Zhu, Adv. Funct. Mater., 2021, 31, 2010383 Search PubMed.
- Y. Pei, H. Wang and G. J. Snyder, Adv. Mater., 2012, 24, 6125–6135 CrossRef CAS PubMed.
- H. Yang, C. Lin, K. Liang, X. Ma, L. Yin, S. Ye, P. Zhao, X. Wu, F. Cao, Q. Zhang and J. Mao, Innovation Mater., 2025, 3, 100060 Search PubMed.
- X. Shi, X. Wang, W. Li and Y. Pei, Small Methods, 2018, 2, 1800012 CrossRef.
- Q. Zhu, S. Song, H. Zhu and Z. Ren, J. Power Sources, 2019, 414, 393–400 CrossRef CAS.
- J. Mao, G. Chen and Z. Ren, Nat. Mater., 2020, 20, 454–461 CrossRef.
- Y. Chen, X. Hou, C. Ma, Y. Dou and W. Wu, Adv. Mater. Sci. Eng., 2018, 2018, 1210565 Search PubMed.
- M. Hong, Z.-G. Chen and J. Zou, Chin. Phys. B, 2018, 27, 048103 Search PubMed.
- Z. Han, Z. Gui, Y. B. Zhu, P. Qin, B. P. Zhang, W. Zhang, L. Huang and W. Liu, Research, 2020, 2020, 1672051 CAS.
- X. Ma, C. Lin, H. Yang, Y. Fu, K. Liang, X. Bao, S. Ye, J. Wang, P. Zhao, J. Chen, S. Zhi, L. Wu, S. Duan, F. Cao, Q. Zhang and J. Mao, Innovation, 2025, 6, 100635 Search PubMed.
- A. Balvanz, J. Qu, S. Baranets, E. Ertekin, P. Gorai and S. Bobev, Chem. Mater., 2020, 32, 10697–10707 CrossRef CAS.
- J. Lee, H. Sa, H. Jo, D. Moon, K. M. Ok and T.-S. You, Cryst. Growth Des., 2020, 20, 4503–4511 CrossRef CAS.
- K. Imasato, M. Wood, S. Anand, J. J. Kuo and G. J. Snyder, Adv. Energy Sustainability Res., 2022, 3, 2200031 CrossRef.
- J. Li, S. Zhang, S. Zheng, Z. Zhang, B. Wang, L. Chen and G. Lu, J. Phys. Chem. C, 2019, 123, 20781–20788 CrossRef CAS.
- J. Zhang, L. Song, M. Sist, K. Tolborg and B. B. Iversen, Nat. Commun., 2018, 9, 4716 CrossRef PubMed.
- M. Ullah, G. Murtaza, S. M. Ramay and A. Mahmood, Mater. Res. Bull., 2017, 91, 22–30 CrossRef CAS.
- T.-R. Chang, I. Pletikosic, T. Kong, G. Bian, A. Huang, J. Denlinger, S. K. Kushwaha, B. Sinkovic, H.-T. Jeng, T. Valla, W. Xie and R. J. Cava, Adv. Sci., 2019, 6, 1800977 Search PubMed.
- X. Shi, C. Sun, Z. Bu, X. Zhang, Y. Wu, S. Lin, W. Li, A. Faghaninia, A. Jain and Y. Pei, Adv. Sci., 2019, 6, 1901546 Search PubMed.
- T. Feng, P. Wang, Z. Han, L. Zhou, W. Zhang, Q. Liu and W. Liu, Adv. Mater., 2022, 34, 2200342 CrossRef.
- R. Wang, Z. Guo, Q. Zhang, J. Cai, G. Liu, X. Tan and J. Jiang, J. Mater. Chem. A, 2022, 10, 11131–11136 RSC.
- J. Lee, B. Monserrat, I. D. Seymour, Z. Liu, S. E. Dutton and C. P. Grey, J. Mater. Chem. A, 2018, 6, 16983–16991 RSC.
- K. Imasato, S. D. Kang and G. J. Snyder, Energy Environ. Sci., 2019, 12, 965–971 RSC.
- J. Zhang, L. Song and B. B. Iversen, npj Comput. Mater., 2019, 5, 76 CrossRef.
- W. Zhang, J.-F. Halet and T. Mori, J. Mater. Chem. A, 2023, 11, 24228–24238 RSC.
- L. Huang, T. Liu, X. Mo, G. Yuan, R. Wang, H. Liu, X. Lei, Q. Zhang and Z. Ren, Mater. Today Phys., 2021, 21, 100538 Search PubMed.
- S. Xiao, K. Peng, Z. Zhou, H. Wang, S. Zheng, X. Lu, G. Han, G. Wang and X. Zhou, J. Magnesium Alloys, 2023, 11, 2486 CrossRef CAS.
- H. Tamaki, H. K. Sato and T. Kanno, Adv. Mater., 2016, 28, 10182 CrossRef CAS PubMed.
- R. Shu, Y. Zhou, Q. Wang, Z. Han, Y. Zhu, Y. Liu, Y. Chen, M. Gu, W. Xu, Y. Wang, W. Zhang, L. Huang and W. Liu, Adv. Funct. Mater., 2019, 29, 1807235 CrossRef.
- J. Mao, H. Zhu, Z. Ding, Z. Liu, G. A. Gamage, G. Chen and Z. Ren, Science, 2019, 365, 495 CrossRef CAS.
- X. Zhang, Z. Bu, X. Shi, Z. Chen, S. Lin, B. Shan, M. Wood, A. H. Snyder, L. Chen, G. J. Snyder and Y. Pei, Sci. Adv., 2020, 6, eabc0726 CrossRef CAS PubMed.
- C. Wu, X.-L. Shi, L. Wang, W. Lyu, P. Yuan, L. Cheng, Z.-G. Chen and X. Yao, ACS Nano, 2024, 18, 31660 CrossRef CAS.
- J. Bi, Z. Liu, B. Li, S. Li, Z. Yang, M. D. Starostenkov and G. Dong, J. Mater. Sci., 2023, 59, 359 CrossRef.
- T. Cao, X.-L. Shi, M. Li, B. Hu, W. Chen, W.-D. Liu, W. Lyu, J. MacLeod and Z.-G. Chen, eScience, 2023, 3, 100056 CrossRef.
- Q.-Y. Liu, X.-L. Shi, T.-Y. Cao, W.-Y. Chen, L. Li and Z.-G. Chen, Prog. Mater. Sci., 2025, 150, 101206 CrossRef.
- J. Zhang, L. Song, A. Mamakhel, M. R. V. Jørgensen and B. B. Iversen, Chem. Mater., 2017, 29, 5371 CrossRef CAS.
- X. M. Shi, T. T. Zhao, X. Y. Zhang, C. Sun, Z. W. Chen, S. Q. Lin, W. Li, H. Gu and Y. Z. Pei, Adv. Mater., 2019, 31, 1903387 CrossRef.
- Y. Liu, Y. Geng, Y. Dou, X. Wu, L. Hu, F. Liu, W. Ao and C. Zhang, Small, 2023, 19, 2301063 CrossRef PubMed.
- X. Peng, J. Guo, X. Su, J. Wu, Q. Zhang and X. Tang, ACS Appl. Energy Mater., 2024, 7, 2552–2560 CrossRef CAS.
- V. Ponnambalam and D. T. Morelli, J. Electron. Mater., 2013, 42, 1307 CrossRef CAS.
- F. Lv, Q. Zhang, W. Fan, X. Yang, J. Fan, H. Zhang, S. Chen, W. Wang and X. Tang, J. Mater. Sci., 2018, 53, 8039 CrossRef CAS.
- Y. Pan, M. Yao, X. Hong, Y. Zhu, F. Fan, K. Imasato, Y. He, C. Hess, J. Fink, J. Yang, B. Büchner, C. Fu, G. J. Snyder and C. Felser, Energy Environ. Sci., 2020, 13, 1717 RSC.
- N. Chen, H. Zhu, G. Li, Z. Fan, X. Zhang, J. Yang, T. Lu, Q. Liu, X. Wu, Y. Yao, Y. Shi and H. Zhao, Nat. Commun., 2023, 14, 4932 CrossRef CAS.
- J. J. Kuo, S. D. Kang, K. Imasato, H. Tamaki, S. Ohno, T. Kanno and G. J. Snyder, Energy Environ. Sci., 2018, 11, 429 RSC.
- M. Wood, J. J. Kuo, K. Imasato and G. J. Snyder, Adv. Mater., 2019, 31, 1902337 CrossRef.
- Q.-Q. Wang, K.-F. Liu, Y.-Y. Su, X.-C. Liu, Q. Liu, S. Zhou, J. Liu and S.-Q. Xia, Acta Mater., 2023, 255, 118256 Search PubMed.
- S. Zhou, Y.-R. Yin, Y. Tian, X.-C. Liu, Q. Liu, B. Li and S.-Q. Xia, ACS Appl. Mater. Interfaces, 2024, 16, 28886 CrossRef CAS.
- Q.-Q. Wang, F. Li, S.-Q. Xia, J. Liu, X.-C. Liu, L. Chen and C.-Q. Zhang, Adv. Electron. Mater., 2022, 8, 2101149 CrossRef.
- J. Li, Y. Pan, C. Wu, F. Sun and T. Wei, Sci. China: Technol. Sci., 2017, 60, 1347 CrossRef.
- M. Calderon-Cueva, W. Peng, S. M. Clarke, J. Ding, B. L. Brugman, G. Levental, A. Balodhi, M. Rylko, O. Delaire, J. P. S. Walsh, S. M. Dorfman and A. Zevalkink, Chem. Mater., 2021, 33, 567 CrossRef CAS.
- J. Xin, G. Li, G. Auffermann, H. Borrmann, W. Schnelle, J. Gooth, X. Zhao, T. Zhu, C. Felser and C. Fu, Mater. Today Phys., 2018, 7, 61 CrossRef.
- M. T. Agne, K. Imasato, S. Anand, K. Lee, S. K. Bux, A. Zevalkink, A. J. E. Rettie, D. Y. Chung, M. G. Kanatzidis and G. J. Snyder, Mater. Today Phys., 2018, 6, 83 CrossRef.
- W. Huang, J. Chen, H. Yan, W. Xia and B. Su, J. Mater. Sci. Technol., 2022, 101, 187 CrossRef CAS.
- F. Ji, T. Xin, Y. Zhao, W. Yang, G. Bai, S. Tang, E. Guo, M. Zhou, Q. Shi, L. Cui, L.-Q. Chen and B. He, Int. J. Plast., 2024, 181, 103788 Search PubMed.
- P. Zhao, W. Xue, Y. Zhang, S. Zhi, X. Ma, J. Qiu, T. Zhang, S. Ye, H. Mu, J. Cheng, X. Wang, S. Hou, L. Zhao, G. Xie, F. Cao, X. Liu, J. Mao, Y. Fu, Y. Wang and Q. Zhang, Nature, 2024, 631, 777 CrossRef CAS.
- M. Hu, J. Yang, Y. Wang, J. Xia, Q. Gan, S. Yang, J. Xu, S. Liu, W. Yin, B. Jia, L. Xie, H. Li and J. He, Nat. Commun., 2025, 16, 676 CrossRef.
- Z. Zhang, Z. Gao, T. Deng, Q. Song, L. Chen and S. Bai, J. Mater. Chem. A, 2024, 12, 8893 RSC.
- S. Xu, S. Horta, A. Lawal, K. Maji, M. Lorion and M. Ibáñez, Science, 2025, 387, 845 CrossRef CAS.
- D. Zhang, X. J. G. Lim, X. Li, K. Saglik, S. F. D. Solco, X. Y. Tan, Y. Leow, W. Zhai, C. K. I. Tan, J. Xu and A. Suwardi, ACS Energy Lett., 2022, 7, 332 Search PubMed.
- M. R. Burton, S. Mehraban, D. Beynon, J. McGettrick, T. Watson, N. P. Lavery and M. J. Carnie, Adv. Energy Mater., 2019, 9, 1901217 Search PubMed.
- M. Ozen, A. B. Burcak, D. Zavanelli, M. Heo, M. Yahyaoglu, Y. Oz, U. Burkhardt, H.-S. Kim, G. J. Snyder and U. Aydemir, ACS Appl. Mater. Interfaces, 2024, 16, 52501 CrossRef CAS PubMed.
- L. Wang, S. Xu, J. Yang, X. Tang, X. Wang, G. Qiao and G. Liu, J. Adv. Ceram., 2025, 14, 319 Search PubMed.
- J.-W. Li, Z. Han, J. Yu, H.-L. Zhuang, H. Hu, B. Su, H. Li, Y. Jiang, L. Chen, W. Liu, Q. Zheng and J.-F. Li, Nat. Commun., 2023, 14, 787 CrossRef.
- L. Wang, A. Li, J. Li, N. Kawamoto, D. H. Nguyen and T. Mori, Nano Energy, 2025, 137, 108392 CrossRef.
- J. Lei, H. Wuliji, Q. Ren, X. Hao, H. Dong, H. Chen, T.-R. Wei, J. Zhang, P. Qiu, K. Zhao and X. Shi, Energy Environ. Sci., 2024, 17, 1416 RSC.
- H. Zhu, Y. Zhang, Y. Chen, X. Ren, S. Liu, X. Mo, B. Zhang, X. Lu, G. Han, G. Wang and X. Zhou, ACS Appl. Mater. Interfaces, 2025, 17, 60635 CrossRef CAS.
- K. Bartholomé, B. Balke, D. Zuckermann, M. Köhne, M. Müller, K. Tarantik and J. König, J. Electron. Mater., 2013, 43, 1775 CrossRef.
- Q. Zhang, Z. Fan, X. Zhang, N. Chen, K. Guo, Q. Zhao, Y. Wang, C. Pan, S. Qi, X. Zhou, G. Li, Y. Liu, C. Liu, Y. Liu, J. Xu, H. Zhao and H. Zhu, Mater. Today Phys., 2025, 54, 101286 Search PubMed.
- J. Zhang, L. Song, K. A. Borup, M. R. V. Jorgensen and B. B. Iversen, Adv. Energy Mater., 2018, 8, 1800244 Search PubMed.
- X. Chen, H. Wu, J. Cui, Y. Xiao, Y. Zhang, J. He, Y. Chen, J. Cao, W. Cai, S. J. Pennycook, Z. Liu, L.-D. Zhao and J. Sui, Nano Energy, 2018, 52, 246 CrossRef CAS.
- J. Li, F. Jia, S. Zhang, S. Zheng, B. Wang, L. Chen, G. Lu and L. Wu, J. Mater. Chem. A, 2019, 7, 19316 RSC.
- J. Mao, J. Shuai, S. Song, Y. Wu, R. Dally, J. Zhou, Z. Liu, J. Sun, Q. Zhang, C. dela Cruz, S. Wilson, Y. Pei, D. J. Singh, G. Chen, C.-W. Chu and Z. Ren, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 10548 CrossRef CAS.
- J. Li, S. Zhang, F. Jia, S. Zheng, X. Shi, D. Jiang, S. Wang, G. Lu, L. Wu and Z.-G. Chen, Mater. Today Phys., 2020, 15, 100255 CrossRef.
- Y. Chen, C. Wang, Z. Ma, L. Li, S. Li and J. Wang, Curr. Appl. Phys., 2021, 21, 25 CrossRef.
- H. Shang, J. Zhang, H. Gu, S. Song, C. L. Chen, J. F. Lee, K. Shih, Z. Ren and F. Ding, Mater. Today Phys., 2021, 21, 100527 CrossRef.
- K. Kihou, H. Kunioka, H. Nishiate and C. H. Lee, J. Mater. Res. Technol., 2021, 10, 438 CrossRef CAS.
- J. Lei, H. Wuliji, K. Zhao, T.-R. Wei, Q. Xu, P. Li, P. Qiu and X. Shi, J. Mater. Chem. A, 2021, 9, 25944 RSC.
- B. Zhu, X. Lu, J. D. Li, W. H. Xie, L. F. Wang, Q. Zhang, J. F. Fan and Y. Zheng, ChemNanoMat, 2022, 8, e202200098 CrossRef.
- L. Huang, J. Liao, G. Yuan, T. Liu, X. Lei, C. Wang and Q. Zhang, Mater. Today Energy, 2022, 25, 100992 Search PubMed.
- Z. Liu, W. Gao, H. Oshima, K. Nagase, C.-H. Lee and T. Mori, Nat. Commun., 2022, 13, 616 CrossRef.
- L. Yu, W. Li, Z. Zhang, S. Wei, J. Li, Z. Ji, J. Zhuo, G. Lu, W. Song and S. Zheng, Mater. Today Phys., 2022, 26, 100933 Search PubMed.
- X. Mo, J. Liao, G. Yuan, S. Zhu, X. Lei, L. Huang, Q. Zhang, C. Wang and Z. Ren, J. Magnesium Alloys, 2022, 10, 1024 CrossRef CAS.
- W. Zhu, W. Fang, J. Zou, S. Zhu and J. Si, J. Electron. Mater., 2022, 51, 1591 CrossRef CAS.
- F. Zhang, C. Chen, S. Li, L. Yin, B. Yu, J. Sui, F. Cao, X. Liu, Z. Ren and Q. Zhang, Adv. Electron. Mater., 2020, 6, 1900915 Search PubMed.
- J. W. Li, W. S. Liu, W. Xu, H. L. Zhuang, Z. J. Han, F. Jiang, P. Zhang, H. H. Hu, H. B. Gao, Y. L. Jiang, B. W. Cai, J. Pei, B. Su, Q. Li, K. Hayashi, H. Z. Li, Y. Miyazaki, X. Z. Cao, Q. Zheng and J. F. Li, Adv. Mater., 2023, 35, 2300031 Search PubMed.
- J.-S. Liang, X.-L. Shi, Y. Peng, W.-D. Liu, H.-Q. Yang, C.-Y. Liu, J.-L. Chen, Q. Zhou, L. Miao and Z.-G. Chen, Adv. Energy Mater., 2022, 12, 2202247 CrossRef.
- Y.-B. Zhang, J.-S. Liang, C. Liu, Q. Zhou, Z. Xu, H.-B. Chen, F.-C. Li, Y. Peng and L. Miao, J. Mater. Sci. Technol., 2024, 170, 25 CrossRef CAS.
- Q. Zhang, J. Li, N. S. Chauhan, L. Wang, Z. Huang, W. Fan, K. Hayashi, S. Chen, J. Fan and Y. Miyazaki, J. Mater. Chem. A, 2023, 11, 18811 RSC.
- L. Hu, Q. Zhang, Z. Shan, L. Wang, Y. Zheng and J. Fan, Scr. Mater., 2023, 235, 115540 CrossRef.
- L. Wang, N. Sato, Y. Peng, R. Chetty, N. Kawamoto, D. H. Nguyen and T. Mori, Adv. Energy Mater., 2023, 13, 2300928 Search PubMed.
- Q. Xu, K. Zhao, H. Huang, S. Wan, Q. Ren, X. Hao, H. Wuliji, J. Lei, T.-R. Wei and X. Shi, Acta Mater., 2024, 274, 120040 CrossRef CAS.
- J. Wang, H. Gao, K. Zhao, H. Wuliji, B. Zhao, J. Ma, X. Chen, J. Zhang, Y. Sui, T.-R. Wei, M. Zhu and X. Shi, Sci. Adv., 2025, 11, eadt6298 CrossRef CAS PubMed.
- J. Hu, J. Zhu, F. Guo, H. Qin, Y. Liu, Q. Zhang, Z. Liu, W. Cai and J. Sui, Research, 2022, 2022, 12 CrossRef.
- D. Wu, Y. Pei, Z. Wang, H. Wu, L. Huang, L.-D. Zhao and J. He, Adv. Funct. Mater., 2014, 24, 7763 CrossRef CAS.
- X.-L. Shi, N.-H. Li, M. Li and Z.-G. Chen, Chem. Rev., 2025, 125, 7525 CrossRef CAS PubMed.
- M. Zhu, X.-L. Shi, H. Wu, Q. Liu and Z.-G. Chen, Chem. Eng. J., 2023, 473, 145745 Search PubMed.
- T. Zhu, Y. Liu, C. Fu, J. P. Heremans, J. G. Snyder and X. Zhao, Adv. Mater., 2017, 29, 1605884 CrossRef.
- F. Jiang, X. Wu, Y. Zhu, C. Xia, Z. Han, H. Yu, C. Chen, T. Feng, J. Mao, Y. Chen and W. Liu, Acta Mater., 2024, 265, 115410 CrossRef.
- Z. Zhou, G. Han, X. Lu, G. Wang and X. Zhou, J. Magnesium Alloys, 2022, 10, 1719 CrossRef CAS.
- J. Shuai, B. Ge, J. Mao, S. Song, Y. Wang and Z. Ren, J. Am. Chem. Soc., 2018, 140, 1910 CrossRef CAS.
- X. Shi, X. Zhang, A. Ganose, J. Park, C. Sun, Z. Chen, S. Lin, W. Li, A. Jain and Y. Pei, Mater. Today Phys., 2021, 18, 100383 Search PubMed.
- K. Imasato, S. D. Kang, S. Ohno and G. J. Snyder, Mater. Horiz., 2018, 5, 59 RSC.
- J. Li, S. Zhang, B. Wang, S. Liu, L. Yue, G. Lu and S. Zheng, J. Mater. Chem. A, 2018, 6, 20454 RSC.
- A. Li, C. Fu, X. Zhao and T. Zhu, Research, 2020, 2020, 7163710 Search PubMed.
- J. Liang, H. Yang, C. Liu, L. Miao, J. Chen, S. Zhu, Z. Xie, W. Xu, X. Wang, J. Wang, B. Peng and K. Koumoto, Appl. Mater. Interfaces, 2020, 12, 21799–21807 CrossRef CAS.
- K. Jin, H. Nie, F. Gao, J. J. Ma, M. Shu, X. Yan, Z. H. Ge, X. Gao, W. Zhao, M. Liu, Y. Yu, J. Ma, B. Xu and L. Fu, Adv. Funct. Mater., 2024, 35, 2418041 CrossRef.
- J. Mao, Y. Wu, S. Song, J. Shuai, Z. Liu, Y. Pei and Z. Ren, Mater. Today Phys., 2017, 3, 1 CrossRef.
- X. Shi, C. Sun, X. Zhang, Z. Chen, S. Lin, W. Li and Y. Pei, Chem. Mater., 2019, 31, 8987 CrossRef CAS.
- L. Yu, X.-L. Shi, Y. Mao, M. Li, W.-D. Liu, Z. Ji, S. Wei, Z. Zhang, W. Song, S. Zheng and Z.-G. Chen, Chem. Eng. J., 2024, 482, 147755 Search PubMed.
- J. Li, S. Zhang, S. Zheng, K. Han, B. Sun, Z. Bi and X. Wang, Adv. Theory Simul., 2022, 5, 2200071 Search PubMed.
- J. Liang, Q. Zhou, Z. Pan, Z. Zhang, F. Mao, S. Zhao, S. Zhu, J.-L. Chen, J. Gao and L. Miao, Mater. Today Phys., 2025, 52, 101121 Search PubMed.
- F. Zhang, X. Zhao, R. Li, S. He, X. Tan, J. Zhu and R. Ang, J. Mater. Chem. A, 2022, 10, 23521 RSC.
- J. Li, Z. Chen, X. Zhang, H. Yu, Z. Wu, H. Xie, Y. Chen and Y. Pei, Adv. Sci., 2017, 4, 1700341 CrossRef.
- Q. Xiong, G. Han, G. Wang, X. Lu and X. Zhou, Adv. Funct. Mater., 2024, 34, 2408709 Search PubMed.
- C. Hu, K. Xia, C. Fu, X. Zhao and T. Zhu, Energy Environ. Sci., 2022, 15, 1406 RSC.
- F. Zhang, C. Chen, H. Yao, F. Bai, L. Yin, X. Li, S. Li, W. Xue, Y. Wang, F. Cao, X. Liu, J. Sui and Q. Zhang, Adv. Funct. Mater., 2020, 30, 1906143 CrossRef CAS.
- J. Mao, Y. Wu, S. Song, Q. Zhu, J. Shuai, Z. Liu, Y. Pei and Z. Ren, ACS Energy Lett., 2017, 2, 2245 CrossRef CAS.
- H. Zhu, B. Zhang, G. Wang, K. Peng, Y. Yan, Q. Zhang, X. Han, G. Wang, X. Lu and X. Zhou, J. Mater. Chem. A, 2019, 7, 11690 RSC.
- H. Su, W. Shi, Y. Zhang, Y. Lin and Y. Liu, ACS Appl. Energy Mater., 2025, 8, 3942 CrossRef CAS.
- J. Shuai, J. Mao, S. Song, Q. Zhu, J. Sun, Y. Wang, R. He, J. Zhou, G. Chen, D. J. Singh and Z. Ren, Energy Environ. Sci., 2017, 10, 799 RSC.
- X. Chen, J. Zhu, D. Qin, N. Qu, W. Xue, Y. Wang, Q. Zhang, W. Cai, F. Guo and J. Sui, Sci. China Mater., 2021, 64, 1761 CrossRef CAS.
- X. Li, Y. Du, K. Y. Yang, S. Li, J. Zhang, L. Li, J. Xing, G. H. Rao, J. T. Zhao and K. Guo, Adv. Funct. Mater., 2025, 35, 2409947 Search PubMed.
- Y. Lin, M. Wood, K. Imasato, J. J. Kuo, D. Lam, A. N. Mortazavi, T. J. Slade, S. A. Hodge, K. Xi, M. G. Kanatzidis, D. R. Clarke, M. C. Hersam and G. J. Snyder, Energy Environ. Sci., 2020, 13, 4114 RSC.
- T. Kanno, H. Tamaki, H. K. Sato, S. D. Kang, S. Ohno, K. Imasato, J. J. Kuo, G. J. Snyder and Y. Miyazaki, Appl. Phys. Lett., 2018, 112, 033903 CrossRef.
- S. Chen, J. Wei, Z. Kang, X. Miao, D. An, W. Fan and C. Dun, Chem. Eng. J., 2024, 490, 125756 Search PubMed.
- T. Luo, J. J. Kuo, K. J. Griffith, K. Imasato, O. Cojocaru-Miredin, M. Wuttig, B. Gault, Y. Yu and G. J. Snyder, Adv. Funct. Mater., 2021, 31, 2103344 Search PubMed.
- S. Kim, C. Kim, Y.-K. Hong, T. Onimaru, K. Suekuni, T. Takabatake and M.-H. Jung, J. Mater. Chem. A, 2014, 2, 12311 RSC.
- K. Imasato, C. Fu, Y. Pan, M. Wood, J. J. Kuo, C. Felser and G. J. Snyder, Adv. Mater., 2020, 32, 2002587 CrossRef PubMed.
- P. Nan, A. Li, L. Cheng, K. Wu, Z. Liang, F. Lin, C. Fu, T. Zhu and B. Ge, Mater. Today Phys., 2021, 21, 100528 CrossRef.
- X. Ma, H. Yao, S. Zhi, P. Zhao, S. Ye, S. Duan, L. Yin, J. Li, X. Bao, J. Sui, F. Cao, Q. Zhang and J. Mao, Chem. Mater., 2023, 35, 5640 CrossRef CAS.
- Y. Yan, S. Cao, X. Shi, W. Zhang, X. Wang, J. Liang, H. Kang, Z. Chen, E. Guo, R. Chen and T. Wang, Acta Mater., 2025, 298, 129959 CrossRef.
- J. V. Heremans and E. Toberer, Science, 2008, 321, 554–557 CrossRef CAS PubMed.
- A. Li, C. Hu, B. He, M. Yao, C. Fu, Y. Wang, X. Zhao, C. Felser and T. Zhu, Nat. Commun., 2021, 12, 1509 CrossRef.
- Y. Goto, H. Usui, M. Murata, J. E. Goldberger, J. P. Heremans and C.-H. Lee, Chem. Mater., 2024, 36, 2018 CrossRef CAS.
- J. Liu, A. Li, C. Fu and T. Zhu, Innovation Mater., 2021, 12, 100158 Search PubMed.
- X. Sun, X. Li, J. Yang, J. Xi, R. Nelson, C. Ertural, R. Dronskowski, W. Liu, G. J. Snyder, D. J. Singh and W. Zhang, J. Comput. Chem., 2019, 40, 1693 CrossRef CAS PubMed.
- Z. Han, Z. Gui, Y. B. Zhu, P. Qin, B.-P. Zhang, W. Zhang, L. Huang and W. Liu, Research, 2020, 2020, 6072893 Search PubMed.
- S. Ning, S. Huang, Z. Zhang, N. Qi, M. Jiang, Z. Chen and X. Tang, J. Materiomics, 2022, 8, 1086 CrossRef.
- C. Chen, X. Li, S. Li, X. Wang, Z. Zhang, J. Sui, F. Cao, X. Liu and Q. Zhang, J. Mater. Sci., 2018, 53, 16001–16009 CrossRef CAS.
- S. Xie, X. Wan, Y. Wu, C. Li, F. Yan, Y. Ouyang, H. Ge, X. Li, Y. Liu, R. Wang, M. Y. Toriyama, G. J. Snyder, J. Yang, Q. Zhang, W. Liu and X. Tang, Adv. Mater., 2024, 36, e2400845 CrossRef PubMed.
- Z. Liang, C. Xu, S. Song, X. Shi, W. Ren and Z. Ren, Adv. Funct. Mater., 2022, 33, 2210016 CrossRef.
- M. Tiadi, V. Trivedi, S. Kumar, P. K. Jain, S. K. Yadav, R. Gopalan, D. K. Satapathy and M. Battabyal, Appl. Mater. Interfaces, 2023, 15, 20175–20190 CrossRef CAS PubMed.
- A. Bhardwaj, N. S. Chauhan and D. K. Misra, J. Mater. Chem. A, 2015, 3, 10777 RSC.
- M. Ozen, M. Yahyaoglu, C. Candolfi, I. Veremchuk, F. Kaiser, U. Burkhardt, G. J. Snyder, Y. Grin and U. Aydemir, J. Mater. Chem. A, 2021, 9, 1733 RSC.
- J. Yang, S. Zhao, X. Liu, L. Chen and L. M. Wu, Adv. Funct. Mater., 2024, 35, 2309736 Search PubMed.
- Q. Zhang, X. Su, Y. Yan, H. Xie, T. Liang, Y. You, X. Tang and C. Uher, ACS Appl. Mater. Interfaces, 2016, 8, 3268 CrossRef CAS PubMed.
- C. Xu, Z. Liang, H. Shang, D. Wang, H. Wang, F. Ding, J. Mao and Z. Ren, Mater. Today Phys., 2021, 17, 100341 CrossRef.
- J. Shuai, B. Ge, J. Mao, S. Song, Y. Wang and Z. Ren, J. Am. Chem. Soc., 2018, 140, 1910 CrossRef CAS PubMed.
- S. W. Song, J. Mao, M. Bordelon, R. He, Y. M. Wang, J. Shuai, J. Y. Sun, X. B. Lei, Z. S. Ren, S. Chen, S. Wilson, K. Nielsch, Q. Y. Zhang and Z. F. Ren, Mater. Today Phys., 2019, 8, 25 CrossRef.
- H. Cho, S. Y. Back, N. Sato, Z. Liu, W. Gao, L. Wang, H. D. Nguyen, N. Kawamoto and T. Mori, Adv. Funct. Mater., 2024, 34, 2405223 Search PubMed.
- R. Wang, S. Luo, X. Mo, H. Liu, T. Liu, X. Lei, Q. Zhang, J. Zhang and L. Huang, Adv. Sustainable Syst., 2023, 7, 2300081 Search PubMed.
- J. Zhang, L. Song and B. B. Iversen, Adv. Sci., 2020, 7, 2002626 Search PubMed.
- D. R. Jaishi, N. Sharma, B. Karki, B. P. Belbase, R. P. Adhikari and M. P. Ghimire, AIP Adv., 2021, 11, 025018 CrossRef.
- J. Callaway, Phys. Rev., 1959, 113, 1046 CrossRef CAS.
- P. Ouyang, M.-H. Yuan, P. Tang, Q. Zhang, S. Liu, J. Shuai and X.-G. Li, ACS Appl. Mater. Interfaces, 2023, 15, 37554 CrossRef CAS.
- J. Ding, T. Lanigan-Atkins, M. Calderon-Cueva, A. Banerjee, D. L. Abernathy, A. Said, A. Zevalkink and O. Delaire, Sci. Adv., 2021, 7, eabf1412 Search PubMed.
- J.-X. Liang, S.-T. Luo, Z.-B. Wei, T. Wang, Y.-T. Jiang, L.-X. Dong, S.-Q. Zheng, W.-Y. Song and H.-C. Wang, Mater. Today Phys., 2025, 55, 101738 Search PubMed.
- S. Han, S. Dai, J. Ma, Q. Ren, C. Hu, Z. Gao, M. D. Le, D. Sheptyakov, P. Miao, S. Torii, T. Kamiyama, C. Felser, J. Yang, C. Fu and T. Zhu, Nat. Phys., 2023, 19, 1649 Search PubMed.
- Y. Yan, W. Zhang, Q. Zhang, X. Shi, H. Song, F. Guo, H. Kang, Z. Chen, E. Guo, R. Chen, J. Zhu and T. Wang, Mater. Today Chem., 2025, 48, 101630 Search PubMed.
- Y. Z. Li, Q. Zhang, K. Liu, Y. J. Lin, N. Lin, Y. Yu, F. Liu, X. B. Zhao, B. H. Ge, O. Cojocaru-Mirédin, C. G. Fu and T. J. Zhu, Mater. Today Nano, 2023, 22, 100283 Search PubMed.
- J. Yang, X. Zhang, G. Liu, L. Zhao, J. Liu, Z. Shi, J. Ding and G. Qiao, Nano Energy, 2020, 74, 104847 Search PubMed.
- C. X. Zhao, Z. Li, T. J. Fan, C. Xiao and Y. Xie, Research, 2020, 2020, 7281527 Search PubMed.
- W. Peng, G. Petretto, G.-M. Rignanese, G. Hautier and A. Zevalkink, Joule, 2018, 2, 1879 Search PubMed.
- M. Nomura, Mater. Trans., 2016, 57, 1022 CrossRef CAS.
- Y. Zhu, Z. Han, B. Han, F. Jiang, X. Wu, C.-G. Han, Y. Deng and W. Liu, ACS Appl. Mater. Interfaces, 2021, 13, 56164 CrossRef CAS PubMed.
- L. Zhai, H. Liu, L. Su, Y. Kuang, F. Chen, Y. Zhang, W. Fan and Z. Sun, Energy Environ. Mater., 2025, 25, e70066 CrossRef.
- L. Wang, W. Zhang, S. Y. Back, N. Kawamoto, D. H. Nguyen and T. Mori, Nat. Commun., 2024, 15, 6800 CrossRef CAS PubMed.
- B. Jiang, Y. Yu, H. Chen, J. Cui, X. Liu, L. Xie and J. He, Nat. Commun., 2021, 12, 3234 CrossRef CAS.
- X. Li, C. Chen, L. Yin, X. Wang, J. Mao, F. Cao and Q. Zhang, Energy Environ. Sci., 2023, 16, 6147–6154 RSC.
- C. Xu, Z. Liang, S. Song and Z. Ren, Adv. Funct. Mater., 2023, 33, 2309323 Search PubMed.
- Q. Yan and M. G. Kanatzidis, Nat. Mater., 2021, 21, 503 CrossRef PubMed.
- F. Jiang, T. Feng, Y. Zhu, Z. Han, R. Shu, C. Chen, Y. Zhang, C. Xia, X. Wu, H. Yu, C. Liu, Y. Chen and W. Liu, Mater. Today Phys., 2022, 27, 100722 Search PubMed.
- J. Cheng, W. Xue, T. Zhang, X. Li, Y. Zhu, L. Yin, H. Yao, Z. Fu, L. Wu, C. Chen, P. Zhao, X. Ma, F. Jiang, X. Wang, M. Li, J. Mao, Y. Wang, F. Cao and Q. Zhang, Joule, 2025, 9, 934 CrossRef.
- H. Shang, Q. Zou, L. Zhang, Z. Liang, S. Song, B. Hong, H. Gu, Z. Ren and F. Ding, Nano Energy, 2023, 109, 108343 CrossRef.
- Z. Liang, M. Jian, C. Xu, B.-H. Lei, X. Shi, H. Shang, S. Song, W. Ren, F. Ding, D. J. Singh, Z. Feng and Z. Ren, Acta Mater., 2023, 247, 118480 CrossRef.
- Y. Geng, Z. Li, Z. Lin, Y. Liu, Q. Lai, X. Wu, L. Hu, F. Liu, Y. Yu and C. Zhang, Small, 2023, 20, 2305608 Search PubMed.
- H. Shang, Z. Liang, C. Xu, S. Song, D. Huang, H. Gu, J. Mao, Z. Ren and F. Ding, Acta Mater., 2020, 201, 572 CrossRef CAS.
- L. Yin, X. Li, X. Bao, J. Cheng, C. Chen, Z. Zhang, X. Liu, F. Cao, J. Mao and Q. Zhang, Nat. Commun., 2024, 15, 1468 CrossRef CAS PubMed.
- J. W. Li, H. Gao, Z. Han, J. Yu, H. L. Zhuang, L. Chen, H. Li, Y. Jiang, Z. Wang, Q. Zheng and J. F. Li, Adv. Mater., 2025, 37, 2307607 Search PubMed.
- P. Ying, H. Reith, K. Nielsch and R. He, Small, 2022, 18, 2203713 CrossRef PubMed.
- Z. Zhang, C. Ming, Q. Song, L. Wang, H. Chen, J. Liao, C. Wang, L. Chen and S. Bai, Acta Mater., 2025, 287, 117353 CrossRef.
- L. Xie, L. Yin, Y. Yu, G. Peng, S. Song, P. Ying, S. Cai, Y. Sun, W. Shi, H. Wu, N. Qu, F. Guo, W. Cai, H. Wu, Q. Zhang, K. Nielsch, Z. Ren, Z. Liu and J. Sui, Science, 2023, 382, 921–928 CrossRef CAS PubMed.
- J. Lei, K. Zhao, J. Liao, S. Yang, Z. Zhang, T.-R. Wei, P. Qiu, M. Zhu, L. Chen and X. Shi, Nat. Commun., 2024, 15, 6588 CrossRef CAS PubMed.
- Y. Fu, Q. Zhang, Z. Hu, M. Jiang, A. Huang, X. Ai, S. Wan, H. Reith, L. Wang, K. Nielsch and W. Jiang, Energy Environ. Sci., 2022, 15, 3265 RSC.
- X. Qi, J. Chen, K. Guo, S. He, J. Yang, Z. Li, J. Xing, J. Hu, H. Luo, W. Zhang and J. Luo, Chem. Eng. J., 2019, 374, 494 CrossRef CAS.
- M. F. Aamir, R. Chetty, J. Babu and T. Mori, J. Mater. Chem. C, 2025, 13, 10567 RSC.
- S. Bano, R. Chetty, J. Babu and T. Mori, Device, 2024, 2, 100028 CrossRef.
- C. Lin, X. Ma, K. Liang, Y. Xu, H. Yang, J. Wang, S. Ye, J. Qiu, L. Song, S. Zhi, J. Chen, P. Zhao, S. Duan, Z. Wu, L. Yin, L. Wu, F. Jiang, Z. Tang, F. Cao, Q. Zhang and J. Mao, Nat. Commun., 2025, 16, 7799 CrossRef PubMed.
- X. Wu, Z. Han, Y. Zhu, B. Deng, K. Zhu, C. Liu, F. Jiang and W. Liu, Acta Mater., 2022, 226, 117616 CrossRef CAS.
- F. Yang, B. Hu, Y. Peng, C. Hang, H. Chen, C. Lee, J. Wei and M. Li, J. Mater. Sci.: Mater. Electron., 2019, 30, 5526 CrossRef CAS.
- B.-G. Park, K.-H. Jung and S.-B. Jung, J. Alloys Compd., 2017, 699, 1186 CrossRef CAS.
- M. Jiang, Y. Fu, Q. Zhang, Z. Hu, A. Huang, S. Wang, L. Wang and W. Jiang, Natl. Sci. Rev., 2023, 10, nwac225 CrossRef PubMed.
- J. Shuai, J. Mao, S. Song, Q. Zhang, G. Chen and Z. Ren, Mater. Today Phys., 2017, 1, 74 CrossRef.
- M. Liu, X. Zhang, J. Tang, Z. Chen, W. Li and Y. Pei, Sci. Bull., 2023, 68, 2536 CrossRef PubMed.
- J. Zhang, P. Wei, H. Zhang, L. Li, W. Zhu, X. Nie, W. Zhao and Q. Zhang, ACS Appl. Mater. Interfaces, 2023, 15, 22705 CrossRef CAS PubMed.
- S. Hu, C. Huang, C. Li, L. Yang, Z. Chen, B. Sa, W. Li and Y. Pei, J. Materiomics, 2025, 11, 435 Search PubMed.
- M. Guo, A. Zhang, C. Wu, W. Fan, Q. Zhang and S. Chen, ACS Appl. Energy Mater., 2025, 8, 3837 CrossRef CAS.
- H. Zhang, Y. Wang, Z. A. Munir, Y. Zhang, W. Fan and S. Chen, J. Mater. Sci. Technol., 2024, 168, 208 CrossRef CAS.
- Z. Liang, C. Xu, H. Shang, Q. Zhu, F. Ding, J. Mao and Z. Ren, Mater. Today Phys., 2021, 19, 100404 CrossRef.
- A. Nozariasbmarz, B. Poudel, W. Li, H. B. Kang, H. Zhu and S. Priya, iScience, 2020, 23, 101108 CrossRef PubMed.
- L. Yin, C. Chen, F. Zhang, X. Li, F. Bai, Z. Zhang, X. Wang, J. Mao, F. Cao, X. Chen, J. Sui, X. Liu and Q. Zhang, Acta Mater., 2020, 198, 25 CrossRef CAS.
- J. Yang, G. Li, H. Zhu, N. Chen, T. Lu, J. Gao, L. Guo, J. Xiang, P. Sun, Y. Yao, R. Yang and H. Zhao, Joule, 2022, 6, 193–204 CrossRef CAS.
- Y. Fu, X. Ai, Z. Hu, S. Zhao, X. Lu, J. Huang, A. Huang, L. Wang, Q. Zhang and W. Jiang, Nat. Commun., 2024, 15, 9355 CrossRef CAS PubMed.
- T. Cao, X.-L. Shi and Z.-G. Chen, Prog. Mater. Sci., 2023, 131, 101008 CrossRef.
- D.-l. Shi, Z.-m. Geng, L. Shi, Y. Li and K.-h. Lam, J. Mater. Chem. C, 2020, 8, 10221 RSC.
- W.-S. Liu, Q. Zhang, Y. Lan, S. Chen, X. Yan, Q. Zhang, H. Wang, D. Wang, G. Chen and Z. Ren, Adv. Energy Mater., 2011, 1, 577 CrossRef CAS.
- X. Wu, X. Ma, H. Yao, K. Liang, P. Zhao, S. Hou, L. Yin, H. Yang, J. Sui, X. Lin, F. Cao, Q. Zhang and J. Mao, ACS Appl. Mater. Interfaces, 2023, 15, 50216 CrossRef CAS PubMed.
- Y. Wang, J. Chen, Y. Jiang, M. Ferhat, S. Ohno, Z. A. Munir, W. Fan and S. Chen, ACS Appl. Mater. Interfaces, 2022, 14, 33419 CrossRef CAS PubMed.
- J. Zhang, L. R. Jorgensen, L. Song and B. B. Iversen, ACS Appl. Mater. Interfaces, 2022, 14, 31024 CrossRef CAS PubMed.
- C. C. Xu, M. M. Jian, Z. X. Liang, B. H. Lei, S. W. Song, F. H. Zhang, D. J. Singh, Z. Z. Feng and Z. F. Ren, Nano Energy, 2023, 106, 108036 CrossRef CAS.
- W. Xu, B. A. Al-Maythalony, J. Li, X. Li, L. Fu and B. Xu, Adv. Funct. Mater., 2024, 35, 2414194 CrossRef.
- Z. Liu, N. Sato, W. Gao, K. Yubuta, N. Kawamoto, M. Mitome, K. Kurashima, Y. Owada, K. Nagase, C.-H. Lee, J. Yi, K. Tsuchiya and T. Mori, Joule, 2021, 5, 1196–1208 CrossRef CAS.
- P. Ying, L. Wilkens, H. Reith, N. P. Rodriguez, X. Hong, Q. Lu, C. Hess, K. Nielsch and R. He, Energy Environ. Sci., 2022, 15, 2557–2566 RSC.
- C. Gayner, K. K. Kar and W. Kim, Mater. Today Energy, 2018, 9, 359–376 CrossRef.
- Z.-G. Chen, X. Shi, L.-D. Zhao and J. Zou, Prog. Mater. Sci., 2018, 97, 283–346 CrossRef CAS.
- Y. Sun, J. Fu, Y. Ohishi, K. Toh, K. Suekuni, K. Kihou, U. Anazawa, C.-H. Lee and K. Kurosaki, ACS Appl. Mater. Interfaces, 2023, 15, 23246–23254 CrossRef CAS PubMed.
- L. Xie, J. Yang, Z. Liu, N. Qu, X. Dong, J. Zhu, W. Shi, H. Wu, G. Peng, F. Guo, Y. Zhang, W. Cai, H. Wu, H. Zhu, H. Zhao, Z. Liu and J. Sui, Mater. Today, 2023, 65, 5–13 CrossRef CAS.
- R. Santos, S. Aminorroaya Yamini and S. X. Dou, J. Mater. Chem. A, 2018, 6, 3328–3341 RSC.
- J. Hu, F. Guo, M. Guo, J. Zhu, C. Chen, Q. Zhang, W. Cai and J. Sui, J. Materiomics, 2020, 6, 729–735 CrossRef.
- J. Shi, D. Han, Z. Li, L. Yang, S.-G. Lu, Z. Zhong, J. Chen, Q. M. Zhang and X. Qian, Joule, 2019, 3, 1200–1225 CrossRef.
- J. Ballard, M. Hubbard, S.-J. Jung, V. Rojas, R. Ung, J. Suh, M. Kim, J. Lee, J. M. Pierce and R. Venkatasubramanian, Nat. Commun., 2025, 16, 4421 CrossRef CAS PubMed.
- C. Liu, D. Liang, T. Chen, X. Ye, D. He, W. Zhu, X. Nie, P. Wei, W. Zhao and Q. Zhang, Adv. Funct. Mater., 2024, 35, 2415368 CrossRef.
- B. Tian, H. Ma, X. An, Y. Mao, Q. Deng, Q. Sun and R. Ang, Device, 2024, 2, 100524 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |

