Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Influence of europium ion doping on photoinduced properties of 2D cobalt hydroxide: photocatalytic degradation and negative photoconductivity studies
Viktorija
Pankratova
 a,
Rossella
Greco
a,
Rossella
Greco
 *a,
Takashi
Yamamoto
*a,
Takashi
Yamamoto
 b,
Joanna
Hoszowska
b,
Joanna
Hoszowska
 c,
Meng
Zhang
c,
Meng
Zhang
 *d,
Ali M.
Huerta-Flores
*d,
Ali M.
Huerta-Flores
 e,
Miikka
Willman
a,
Kimmo
Sirkka
e,
Miikka
Willman
a,
Kimmo
Sirkka
 f,
Vladimir
Pankratov
f,
Vladimir
Pankratov
 g,
Rafal
Sliz
g,
Rafal
Sliz
 h,
Yang
Bai
h,
Yang
Bai
 f and
Wei
Cao
f and
Wei
Cao
 a
a
aNano and Molecular Systems Research Unit, University of Oulu, Oulu FIN-90014, Finland
bDepartment of Science and Technology, Tokushima University, Tokushima, 770 8506, Japan
cDepartment of Physics, University of Fribourg, Fribourg CH-1700, Switzerland
dSchool of Physics, East China University of Science and Technology, Shanghai 200237, China
eResearch Unit of Sustainable Chemistry, University of Oulu, Oulu FIN-90014, Finland
fMicroelectronics Research Unit, Faculty of Information Technology and Electrical Engineering, University of Oulu, Oulu FIN-90014, Finland
gInstitute of Solid State Physics, University of Latvia, Riga LV-1063, Latvia
hOptoelectronics and Measurement Techniques Unit, University of Oulu, Oulu FIN-90014, Finland
First published on 10th December 2025
Abstract
Transition metal hydroxides have been extensively used in the electrocatalytic oxygen evolution reaction; however, their use in photocatalytic processes remains less common. In this study, we report the successful synthesis of europium-doped (2.8 wt%) cobalt hydroxide nanoplates (Co(OH)2:Eu) via one-pot hydrothermal synthesis for photocatalytic and photoconductive applications. The structural and electronic properties of europium-doped cobalt hydroxide were studied and compared with undoped Co(OH)2 prepared via a similar procedure. Co(OH)2:Eu exhibited increased stability compared to undoped Co(OH)2. Potential applications of Co(OH)2:Eu for photocatalytic pollutant degradation were evaluated on the basis of methylene blue degradation. The band structure of Co(OH)2:Eu was proposed based on the photoconductivity behaviour. The mechanism of the driving forces of the photocatalytic degradation reaction was studied using band structure analysis and radical scavenger experiments. Furthermore, density functional theory (DFT) calculations provided a deeper understanding of the reason for the improved photocatalytic efficiency of Co(OH)2:Eu. We established that Co(OH)2:Eu possesses the local state above the conduction band edge, which induced a marked increase in the negative photoconductivity of Co(OH)2:Eu due to the binding of electrons, highlighting the potential use of rare-earth doping in optoelectronic switches.
Introduction
Photocatalytic reactions using renewable and infinite solar energy provide a sustainable approach within a fossil fuel-based economy. These processes include the use of a photocatalyst, which is usually a transition metal semiconductor. The use of photons to excite the semiconductor materials promotes the excitation of electrons from the valence band to the conduction band, leaving a positive hole in the valence band. The photoexcited electron and hole can behave as a redox pair and, after migration to the surface of the semiconductor, react with molecules adsorbed on the surface. Such photocatalytic reactions can be used to produce hydrogen and oxygen via water splitting and for wastewater treatment by oxidation of the pollutants in water. Currently, one of the environmental crises is the presence of pollutants in wastewater, such as pesticides, antibiotics, and other persistent pollutants, posing serious risks to human health and ecosystems due to their resistance to natural degradation. High energy consumption in current water treatment trends is the reason for the search for new methods.1 Photocatalytic water treatment is considered a promising alternative due to its use of renewable solar energy as the power source.2,3 Ideally, this approach does not require a lot of electricity, but only the energy coming from the sun would be sufficient. This is true in the case of highly efficient photocatalysts, which are still a subject under intense study due to some intrinsic drawbacks of these materials.Common drawbacks of photocatalysts include charge–carrier recombination, poor stability, or wide bandgaps,4–7 which can be improved through changes in crystallinity and dimensions, creating heterostructures, or doping.8 An appropriate bandgap (less than 3 eV) is necessary to ensure the electron–hole pair generation and thus photocatalytic activity under sunlight. Wide-bandgap semiconductors such as titania do not satisfy this requirement, as they require ultraviolet light irradiation for the reaction to be possible, while only 5% of the sunlight is in the ultraviolet region.9–11 Crystal growth determines not only the phase of the synthesized material but also the size of the photocatalyst crystals and, consequently, the surface area, along with the number of active sites.12 For example, dangling bonds at the surface of the materials are a great active site for chemisorption and, therefore, subsequent reaction at the surface.9 Substitution with heteroatoms13,14 and introduction of vacancies15 into the structure are also among other surface defect creation methods for improved reactivity. Interestingly, the dimensions of the catalyst will even tune the bandgap of the material due to the surface plasmonic effect in smaller dimensionalities.16 Two or more semiconductors joined together in a heterostructure prevent charge recombination, therefore limiting the processes competing with photocatalysis, such as radiative and nonradiative recombination. While also modifying the electronic structure, doping techniques act differently from the heterostructure production. Non-metal and transition metal doping have been widely used for increasing photocatalytic activity by enhancing the light absorbance in the visible light spectra.17–21 Rare-earth doping of photocatalysts is also used for bandgap engineering and structure stabilization, inhibiting phase changes and stabilizing the material at increased temperatures.22–24 Rare-earth metal introduction promotes the electron transfer of the reactive species and improve the redox properties of the host materials.25,26
Transition metals, specifically, nickel,27–29 cobalt,30–35 copper,12,36–38 and iron,39–42 are considered good options for producing photocatalysts, mainly because of their high abundance and low price, especially when compared to those of noble metals. Hydroxides of transition metals are known to be good photocatalyst matrices for the oxygen evolution reaction (OER), CO2 reduction, and degradation of organic pollutants.19,28,43,44 Furthermore, transition metal hydroxides were found to increase the yield of the hydrogen evolution reaction (HER) when modifying existing photocatalysts such as TiO2, ZnIn2S4, and others.29,45–48 Hence, the unique features of transition metal hydroxides are very much in demand to mitigate climate, energy and water crises.
Cobalt hydroxide has been previously identified as an electrocatalyst and photocatalyst for the OER.31,33,49–53 On the other hand, the basicity of the hydroxyl groups makes the adsorption of water at the surface challenging, which significantly affects the water splitting to obtain hydrogen.54 Doping cobalt hydroxide with rare-earth metals can improve its photocatalytic activity by increasing its internal resistance and favouring the charge transfer at the surface, a crucial factor in any photocatalytic applications.55 Furthermore, cobalt hydroxide has been proven to be unstable by itself under irradiation, and only the presence of secondary species could avoid this undesired event.30
The study of photoexcited charge carriers and their behaviour is possible by monitoring the photoconductivity of the semiconductor, or the change in electrical conductivity upon light illumination. The cause of the conductivity change is the creation of electron–hole pairs under photoexcitation, the initial step being the same as in photocatalysis. Depending on how the photo-excited charge carriers migrate, the photoconductivity may increase or decrease, and this phenomenon constitutes the basis of several applications of semiconductors, such as photoresistors and photodetectors.
Negative photoconductivity, where the conductivity of the material decreases under illumination, is less commonly observed than positive photoconductivity. This process is attributed to charge carrier trapping. For example, in low-dimensional materials, trap centres and numerous surface defects are responsible for trapping electrons, causing the charge carrier flow and subsequently the conductivity to decrease.56,57 The applications of semiconductors with negative photoconductive properties include optoelectronic sensors, optical memory devices, optical switches, and more.58–60 To date, the photoconductivity of Co(OH)2 has not been well studied, as previous research has focused more on layered hydroxides61 or other cobalt-containing semiconductors62,63 that are typically used as photocatalysts. Indeed, photoconductivity studies complement the search for the semiconductors that would be ideal for photocatalytic applications and help to understand the behaviour of charge carriers.64
In this work, we report a novel approach to enhance the structural and functional properties of β-Co(OH)2 by doping it with Eu3+. The introduction of Eu ions into the Co(OH)2 lattice significantly improves the material's thermal stability, as demonstrated by thermogravimetric analysis. Importantly, the photocatalytic activity of Co(OH)2 is preserved, as is demonstrated through methylene blue degradation experiments as a proof of concept. Computational studies confirmed the findings obtained by photocatalytic degradation, observing an easier desorption of radicals responsible for the reaction from the europium-doped Co(OH)2. Exceptionally, by photoconductivity measurements, we could demonstrate the europium-doped material to exhibit an enhanced negative photoconductivity response, which could open its application as an optoelectronic detector or switch.60 Overall, we open the path for a new family of materials, such as rare-earth doped transition metals hydroxides, to be applied in photomediated processes.
Experimental methods
Synthesis
The synthesis was carried out in a similar manner as previously reported by our research group.30 Cobalt nitrate hexahydrate (Co(NO3)2·6H2O, ≥98.0%, Sigma-Aldrich), polyethylene glycol-6000 (H(OCH2CH2)nOH, Alfa Aesar), europium nitrate hydrate (Eu(NO3)3·H2O, 99.99%, Sigma-Aldrich), absolute ethanol (99.5%, ETAX), and deionized water were used. All reactants were used without any further purification. 0.5 mmol of Co(NO3)2·6H2O and 0.5 g of polyethylene glycol-6000 (PEG-6000) were stirred in 20 mL of H2O at 500 rpm for 30 min. The necessary volume of 3 mM Eu(NO3)3 aqueous solution was then added dropwise to the stirring reaction mixture to reach the nominal 3 wt% of europium. The aqueous solution of NaOH (10 w/v%) was added dropwise until the pH reached 12. Subsequently, the solution was stirred for 10 minutes and transferred to a 40 mL Teflon-lined stainless-steel autoclave. The autoclave was heated at 180 °C for 24 hours for the hydrothermal reaction to occur and then the autoclave was naturally cooled down to room temperature. The mixture with the precipitate was transferred to a centrifuge tube and was centrifuged at 4000 rpm for 15 min and then washed twice with water and ethanol at 4000 rpm for 15 min. The pink color precipitate was dried under vacuum overnight.Characterization
The phase content of the synthesized samples was confirmed by powder X-ray diffractometry (XRD). Powder XRD measurements were performed utilizing a Rigaku SmartLab 9 kW system using Co Kα radiation (λ = 1.79 Å, 40 kV, 135 mA) and a Rigaku MiniFlex 600 using Cu Kα radiation (λ = 1.54 Å, 40 kV, 15 mA). Europium content was determined using inductively coupled plasma-optical emission spectroscopy (ICP-OES), Agilent 5110 VDV (Agilent Technologies Inc., Santa Clara, California, USA). Prior to the analysis, 6.2 mg of Co(OH)2:Eu were dissolved in approximately 1 mL of aqua regia and diluted with deionized water until the total volume was 5 mL. Differential scanning calorimetry-thermogravimetry (DSC-TG) measurements were performed using a Netzsch STA 409 in the temperature range of 38–803 °C in the air with a rate of 10 K min−1.Scanning electron microscopy (SEM) images were obtained using a Zeiss Sigma FESEM (field emission scanning electron microscope). The samples were suspended in ethanol and the suspensions were dried on a silicon wafer prior to the experiment. Transmission electron microscopy (TEM) images were taken using a JEOL JEM-2200FS EFTEM/STEM (energy filtered transmission electron microscopy/scanning transmission electron microscopy) setup. The suspensions of the samples in ethanol were applied dropwise on the C/Cu TEM grids and dried. X-ray photoelectron spectroscopy (XPS) measurements were performed employing an ESCALAB 250Xi using Al Kα radiation and a Au grid.
Electrochemical impedance spectroscopy (EIS) was conducted using a Metrohm Vionic electrochemical station (Metrohm). The frequency range was set from 5 MHz to 0.01 Hz with an amplitude of 10 mV. The measurements were performed at room temperature. Fitting of the Nyquist plot was performed on data recorded at −0.3 V vs. Ag/AgCl under illumination across a frequency spectrum ranging from 0.1 Hz to 1 MHz using a potentiostat–galvanostat (Corrtest Instruments Model CS310M) with EIS capabilities.
Diffuse reflectance spectra of the undoped and doped powders in the ultraviolet-visible (UV-Vis) region were measured using a Shimadzu UV-2600 spectrophotometer using the diffuse reflectance of BaSO4 (Nacalai Tesque Inc.) as a reference.
Kelvin probe force microscopy (KPFM) images were recorded using a NanoSurf FlexAFM system equipped with a C3000i Controller (Nanosurf AG, Switzerland). The work function of the Multi75-G tip was calibrated using highly oriented pyrolytic graphite (HOPG) on a support (Nanosurf AG). Image analysis was performed using the Gwyddion software.65 The sample was prepared via drying the sample suspension in ethanol on a Si wafer.
Commercially produced Co(OH)2 nanoplates (FUJIFILM Wako Pure Chemical Corporation) were used for comparison.
X-ray absorption spectroscopy study
X-ray absorption spectra were recorded with a laboratory-type spectrometer R-XAS Looper66 (Rigaku) in transmission mode at the Co K- and Eu L3-edge. Curved monochromator crystals Ge(220) and Si(400) were used for extended X-ray absorption fine structure (EXAFS) and X-ray absorption near edge structure (XANES) experiments, respectively. The data reduction was performed with the REX2000 program.67 The radial structure function (RSF) was obtained by Fourier transformation of k3-weighted EXAFS in the range of ca. 2–9 Å−1. Note that the available k-range for Co(OH)2:Eu samples at the Co K-edge is restricted up to ca. 9.5 Å−1 due to interference of the Eu L1 edge. The phase shift and backscattering amplitudes were calculated using the FEFF8.4 program.68 The lab-based XAS spectrometer was selected so that the radiation damage of the hydroxides30 can be avoided.Photoluminescence
Photoluminescent properties of the materials were studied under synchrotron radiation excitation. The experiments were carried out at the Superlumi setup installed at the P66 beamline of the PETRA III storage ring at the DESY synchrotron (Hamburg, Germany). The details of the setup can be found elsewhere.69,70 The experiments were carried out under ultra-high vacuum conditions at 12 K. The emission spectra were measured under excitation with 4.1 eV photons.Methylene blue degradation
Methylene blue (MB) dye was purchased from Sigma-Aldrich. The photocatalytic activity of the studied samples was evaluated by testing the photocatalytic methylene blue degradation under white-light LED illumination (0.495 W), which was provided by a PerfectLight PCX50B photoreactor. All experiments were carried out in glass reactors with quartz bottoms. During the photocatalytic experiment, 5.5 mg of the catalyst was added to 25 mL of solution containing 5 mL of 50 mg L−1 MB and 20 mL of deionized water, leading to a final MB concentration of 10 mg L−1. The amount of the catalyst used was chosen to be similar to that reported elsewhere.27,30 At given time intervals, the solutions were filtered through 0.22 µm filters (Electron Microscopy Sciences). The MB degradation behaviour was monitored by observing the changes in the absorbance of the solution in the visible range using a Shimadzu UV-2600 spectrophotometer (λ = 665 nm). The photolysis reaction of MB was conducted under similar conditions, omitting only the catalyst. The effect of adsorption was studied by stirring the MB solution with the added catalyst in the dark and observing the changes in absorbance in the same way.The mechanism of the photocatalytic degradation reaction of MB was studied by adding 20 µL of isopropanol (Sigma-Aldrich) to the initial solutions to assess the role of OH˙ radicals. The degradation of MB was observed via HPLC using a Shimadzu Prominence LC system with a fluorescence detector (λ = 290 nm). The chromatographic separation was performed using an ACQUITY UPLC® BEH C18 1.7 µm column (2.1 mm × 100 mm). 0.1% formic acid aqueous solution and 0.1% formic acid solution in acetonitrile were used as a mobile phase with a flow rate of 0.9 mL min−1 and a composition of 15% of 0.01% formic acid in acetonitrile, which increased to 85% for the first 8 minutes, was held at 85% for one minute and then decreased back to 15% for the last 2 minutes.
To assess the role of oxygen in the photocatalytic degradation reaction, the reaction system was purged for 30 minutes with argon. The photocatalytic activity of Co(OH)2:Eu was also studied in basic media (pH 9), which was adjusted with 10 wt% NaOH solution. Finally, the photocatalytic degradation of other concentrations of MB was also studied (5 and 15 mg L−1). The reaction was monitored by observing the absorbance changes as mentioned above.
Photoelectrochemical characterization
Electrodes composed of active materials (Co(OH)2 and Co(OH)2:Eu) were prepared through the following procedure: 5 mg of the powder materials was dispersed in 500 µL of absolute ethanol along with 0.5 µL of ethanolamine. This mixture was subjected to ultrasonic treatment for 5 minutes. Subsequently, 0.1 mL of the resulting solution was deposited onto a fluorine-doped tin oxide (FTO) conductive substrate and spin-coated at a rotation speed of 2000 rpm for 45 seconds. The film was then dried at 80 °C for 30 min. An electrical connection to the film was established using a copper wire and silver paint, with the active area of the film measured to be 1 cm2.Photoelectrochemical tests were conducted in a quartz cell utilizing a potentiostat–galvanostat (Corrtest Instruments Model CS310M) with EIS capabilities. The electrodes containing photoactive material served as the working electrode, while a platinum wire acted as the counter electrode and a silver/silver chloride (Ag/AgCl) electrode functioned as the reference electrode. A 0.1 M sodium sulfate (Na2SO4) solution (pH 7) was employed as the electrolyte. Illumination of the electrodes was achieved from the front side using a UV-Vis lamp with a measured spectral irradiance of 1109.21 W m−2. Chronoamperometric tests were carried out over a duration of 100 seconds at an applied potential of 0.3 V vs. Ag/AgCl, with the light shutter being opened and closed every 10 seconds. Mott–Schottky plots were generated at a frequency of 500 Hz across a potential range of 0.3 to −1 V vs. Ag/AgCl.
To convert the flat band potential values measured electrochemically to energy in eV vs. vacuum, the following eqn (1) was used, where 4.645 eV is the absolute potential of the Ag/AgCl (3.5 M KCl) reference electrode with respect to the vacuum energy level.71
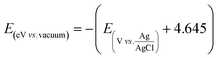 | (1) |
Photoconductivity measurements
Interdigitated Au electrodes were first deposited on a glass substrate via sputtering. 25 mg of Co(OH)2 and Co(OH)2:Eu were suspended individually in 2.5 mL of ethanol (96% v/v). The suspensions were then drop-casted onto the substrates with electrodes and dried at 80 °C on a hot plate. Current–voltage (I–V) curve measurements were performed at room temperature using a source measure unit (2450, Keithley, USA). The samples were measured in a dark room without illumination and under the illumination of lasers (Coherent Inc., USA) operating at 20 mW. The nominal wavelengths of the lasers were 660 nm, 552 nm, and 405 nm, corresponding to photon energies of 1.88 eV, 2.25 eV, and 3.06 eV, respectively. Homogeneity of the sample coverage on the electrodes and the elemental composition were evaluated using a Zeiss ULTRA plus FESEM equipped with an EDS (energy dispersive X-ray spectroscopy) detector. The samples were coated with carbon prior to the FESEM and EDS analyses.Computational methods
The computational work was executed by employing the first-principles approach incorporated in the Vienna Ab initio Simulation Package (VASP).72 In the process of geometry optimization, the Perdew–Burke–Ernzerhof (PBE) functional, which falls under the category of the Generalized Gradient Approximation (GGA), was harnessed to depict the exchange–correlation interactions among electrons.73,74 A plane-wave cutoff energy of 400 eV was established, and the Brillouin zone (BZ) was sampled utilizing a 2 × 2 × 1 k-point grid in accordance with the Monkhorst–Pack scheme.75 The convergence criteria were determined as 10−5 eV for the total energy and 0.02 eV Å−1 for atomic forces. To take into consideration the van der Waals interactions, the DFT-D3 correction method was put into effect.76 In addition, a vacuum layer measuring 20 Å along the z-axis was incorporated with the aim of eliminating periodic interactions. The DFT + U method was employed to characterize the localized Eu 4f and Co 3d states, with U values of 7 eV and 4.5 eV, respectively.Results and discussion
The successful synthesis of the β-cobalt hydroxide was proved by XRD (Fig. 1a). The visible peaks can be indexed to the (001), (100), (101), (002), (102), (110), (003), (200), (103), (021), and (112) planes of β-Co(OH)2 (ICDD card number 00-045-0031). Minor Co3O4 peaks that can be attributed to the (311) and (440) planes can be observed in the spectra, as cobalt hydroxide is metastable and tends to convert to cobalt oxide.30,77 In contrast, separate europium compound signals are not present, signifying the inclusion of europium in the structure of cobalt hydroxide and the absence of bigger Eu-based aggregates, such as nanoparticles. The cobalt hydroxide peaks show a uniform 0.3° shift to the higher angle values, reflecting the increase in the interplanar distance after the inclusion of Eu ions with a larger radius. The changes in the lattice parameters after Eu doping are shown in Table S1. As expected, doping with a larger ion results in an increase in the crystal parameters. The europium content in Co(OH)2:Eu, 2.8 wt%, was determined by ICP-OES, underlining that all the nominal Eu was introduced into the Co(OH)2 structure.In order to first distinguish between the two materials, the stability of the synthesized materials was analyzed using the DSC-TG method (Fig. 1b and c) and compared with each other. The derivative of the thermogravimetric curve is shown for clearer analysis (Fig. 1b, inset). It can be observed that the thermal behaviour of both samples can be categorized into similar stages. The mass of both pure and doped Co(OH)2 gradually decreases by less than one percent until 150 °C due to dehydration and loss of absorbed water molecules. Further thermal degradation of pure Co(OH)2, that is, 16.7% of weight loss after the main reaction at 210 °C, occurred following the pathway of transformation through CoOOH to Co3O4, as already described in the literature.78–80 It is worth noting that the thermal degradation of Co(OH)2:Eu is similar; however, the above-mentioned transitions happened at 20 °C higher than the corresponding temperature for the undoped hydroxide. Additionally, both the thermogravimetry (Fig. 1b) and differential scanning calorimetry (Fig. 1c) results show that Co(OH)2:Eu exhibits fewer phase transitions, signified by the lack of peak shoulders. After heating the materials at 250 °C and 450 °C, XRD was performed (Fig. S1) to confirm the better thermal stability of Co(OH)2:Eu compared to Co(OH)2. Indeed, at 450 °C both materials transform into Co3O4. However, after heating at 250 °C, a clear difference between the hydroxide and the doped material is evident – while Co(OH)2 after thermal degradation has transformed into Co3O4 and CoOOH completely, thermally degraded Co(OH)2:Eu still contains a significant amount of β-Co(OH)2 according to the quantitative XRD analysis. This leads to the conclusion that europium ions are stabilizing the β-Co(OH)2 structure by delaying its conversion to CoOOH or Co3O4, which are less efficient materials in photocatalysis due to the low electrical conductivity and the unexceptional dimensionality.55 It is important to note that at both studied temperatures, the XRD spectra of Co(OH)2:Eu exhibited unidentified peaks, which we suppose are associated with a europium-containing phase.
To confirm the presence of Eu and to prove the even element distribution in the sample, the materials' morphology was examined using TEM coupled with EDS and SEM. Fig. 2 shows the distribution of the elements across the as-synthesized material obtained by EDS mapping. Europium was found to be distributed across the whole surface of the cobalt hydroxide nanoplates, and no europium agglomeration was detected, in line with the observation on the XRD pattern. However, the elemental mapping showed a higher europium signal at the edges of the particles. Additional SEM images can be found in the SI (Fig. S2). The STEM (Fig. 2a–d) and TEM (Fig. 2e) images showed a deviation from a typical hexagonal morphology of the pristine cobalt hydroxide (Fig. S2). Although the particles still possess the two-dimensional structure previously reported,20,79,81 the edges of the nanoplates appear rounded and less regular. The two-dimensional sheets are approximately 100–150 nm in width and ∼10 nm in thickness, as determined by HRTEM in Fig. 2f, with a similar size distribution shown in Fig. S3.
The SAED (selected area electron diffraction) pattern (Fig. 2g) agrees with the XRD pattern and shows a predominant Co(OH)2 phase. No separate europium-containing compound was detected. The shadows near the Co(OH)2 signals correspond to continuously growing Co3O4 formed under the electron beam, which enhances the oxidation process.77
Furthermore, the chemical environment of Co(OH)2:Eu was studied using XPS, which revealed additional information about the changes induced by doping. The XPS survey (Fig. 3a) shows cobalt, oxygen, and europium as the main elements of the material. The indium 3d signal in the survey is associated with the sample holder used. In the Co 2p spectrum (Fig. 3b), Co 2p3/2 peaks at 780.9 eV and 783.0 eV are associated with Co2+ ions, which is consistent with the nature of Co(OH)2. A low-intensity shoulder at 777.7 eV, usually attributed to metallic cobalt, appears due to metal formation on the surface of the sample after reduction by the probe beam.82 Co 2p1/2 peaks at 795.9 eV and 796.9 eV are also present and arise from spin–orbit coupling. Additionally, shake-up satellite peaks are present in the Co 2p spectrum. The cobalt peaks show a small shift to lower energies when compared to pristine Co(OH)2 in the literature83 as well as Co(OH)2 synthesized using the same method. The positive shift in the values of the cobalt peaks (Table S2) is consistent with doping with europium – an element with a lower electronegativity than cobalt. This results in an increase in the electron density around cobalt and a subsequent decrease in cobalt binding energy. The Eu 3d spectrum (Fig. 3c) shows spin–orbit coupling into Eu 3d5/2 and 3d3/2 peaks, as well as a shake-up satellite and two shake-down satellites. The corresponding energies of these peaks are 1135.0 eV and 1164.6 eV, respectively, which are typical for Eu3+, and no shifts in binding energy are observed compared to the values in Eu2O3 (ref. 84) (Table S2). The O 1s spectrum (Fig. 3d) shows three peaks and mainly consists of the XPS peak with the energy of 530.5 eV. It is associated with the oxygen that is bonded to hydrogen, i.e., the hydroxide group (O–H). The higher energy peak at 531.9 eV is typically assigned to the oxygen in the adsorbed water molecules. The peak at 528.5 eV is attributed to the metal–oxygen (M–O) bond in the lattice.85
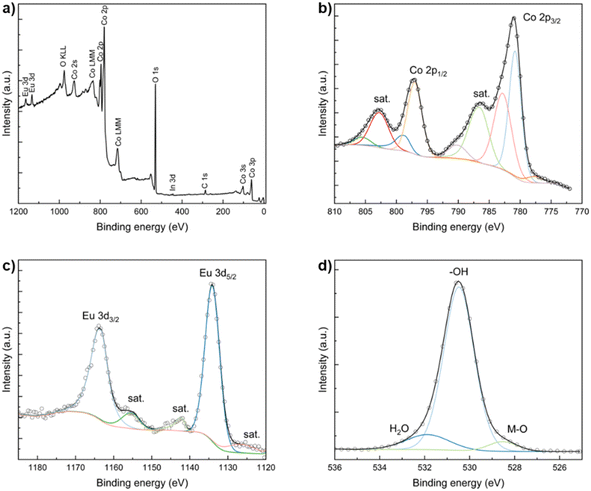 | ||
| Fig. 3 XPS survey of Co(OH)2:Eu (a) and XPS spectra of Co 2p (b), Eu 3d (c), and O 1s (d). The experimental data are shown as circles and the fitted data are shown as solid lines. | ||
The chemical state and the local structure of the synthesized Co(OH)2:Eu nanoplates were studied using in-house X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure spectroscopy (Fig. 4). The Co K-edge XANES (Fig. 4a) experiments were performed, and the results of the synthesized europium-doped cobalt hydroxide were compared with the commercially available reference compounds, Co(OH)2 and CoO (Wako Pure Chemical Industries, Ltd). The absorption edge of Co(OH)2:Eu is the same as that of Co(OH)2, indicating that the Co species is divalent. This is consistent with the XPS results discussed above. The Eu L3-edge XANES (Fig. 4b) experiments were performed and compared with Eu2O3 and EuS to confirm the existence of Eu3+ species in the synthesized cobalt hydroxide. Strong intensity of the “white line” peak of the Eu L3-edge is caused by the difference in the amount of unoccupied Eu 5d states compared to Eu2O3. Further data analysis and calculations were performed using the Co K-edge XANES spectra to get more insights into the sample structure.
 | ||
| Fig. 4 Co K-edge (a) and Eu L3-edge (b) XANES spectra, Co K-edge EXAFS spectra (c), and Fourier transforms (d) of the synthesized Co(OH)2:Eu and the commercially produced Co(OH)2 and CoO samples. | ||
Fig. 4c depicts the EXAFS spectra. The Fourier transforms of the Co K-edge EXAFS oscillations, shown in Fig. 4d, establish the similarity of the structure of the synthesized europium-doped cobalt hydroxide to that of an undoped commercial powder and show the absence of a relevant amount of cobalt oxides. The EXAFS parameters summarized in Table S3 support this proposal by confirming similar Co–O coordination numbers in Co(OH)2 and Co(OH)2:Eu, but different ones in the case of Co–Co.
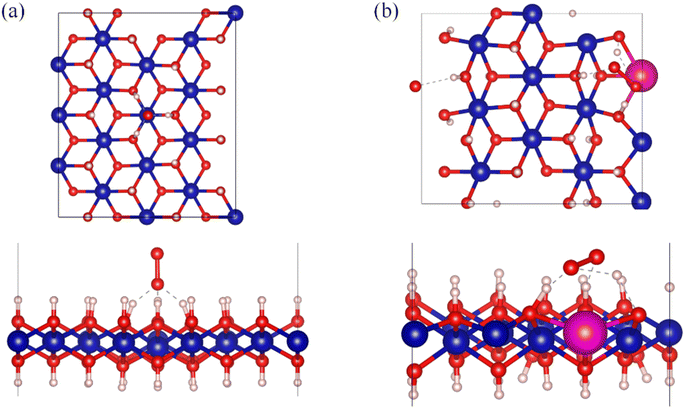
Computational geometry optimization provides more insight into the structural changes occurring in Co(OH)2 upon doping with europium. Fig. 5a and b present the geometrically optimized unit cells of β-Co(OH)2 and Co(OH)2:Eu, respectively. The β-Co(OH)2 unit cell crystallizes in the hexagonal system with space group P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1. In this structure, all Co atoms are six-coordinated, forming a hexagonal arrangement with O atoms. The lattice constants are determined as a = b = 3.18 Å, c = 3.62 Å, α = β = 90°, γ = 120°, consistent with previous studies.86,87 Notably, prior research has demonstrated that the (001) facet of β-Co(OH)2 exhibits lower surface energy,88 which justifies our focus on this specific facet for subsequent investigations. Upon Eu doping in Co(OH)2:Eu, structural changes emerge, i.e., the Eu–O bond length increases to 2.37 Å, significantly longer than the Co–O bond length of 2.04 Å. Furthermore, the bond angles involving Eu–O and O–H show a marked enlargement compared to the undoped system. These modifications highlight the lattice distortion induced by Eu incorporation, which may influence the material's electronic and catalytic properties.
m1. In this structure, all Co atoms are six-coordinated, forming a hexagonal arrangement with O atoms. The lattice constants are determined as a = b = 3.18 Å, c = 3.62 Å, α = β = 90°, γ = 120°, consistent with previous studies.86,87 Notably, prior research has demonstrated that the (001) facet of β-Co(OH)2 exhibits lower surface energy,88 which justifies our focus on this specific facet for subsequent investigations. Upon Eu doping in Co(OH)2:Eu, structural changes emerge, i.e., the Eu–O bond length increases to 2.37 Å, significantly longer than the Co–O bond length of 2.04 Å. Furthermore, the bond angles involving Eu–O and O–H show a marked enlargement compared to the undoped system. These modifications highlight the lattice distortion induced by Eu incorporation, which may influence the material's electronic and catalytic properties.
 | ||
| Fig. 5 Top view (upper) and side view (lower) of β-Co(OH)2 (a) and Co(OH)2:Eu (b), with one Eu atom displayed as a periodic mirror image. | ||
After establishing the elemental and structural composition of the synthesized samples, we studied the electrical and optical properties of Co(OH)2:Eu in order to confirm its applicability in photocatalysis, as well as its interaction with light in the ultraviolet and visible range. Diffuse reflectance spectra (Fig. 6a) reveal the absorption of Co2+ ions in the octahedral crystal field and contain the band characteristic of the 4T1g(F)–4T1g(P) (υ3) transition at around 530 nm.89,90 High absorption of both doped and undoped samples in the ultraviolet region has been associated with the ligand-to-metal charge transfer (LMCT) process. The diffuse reflectance spectra were then transformed into the Kubelka–Munk function using eqn (2):
| (ℏνF(R∞))2 = A(ℏν − Eg) | (2) |
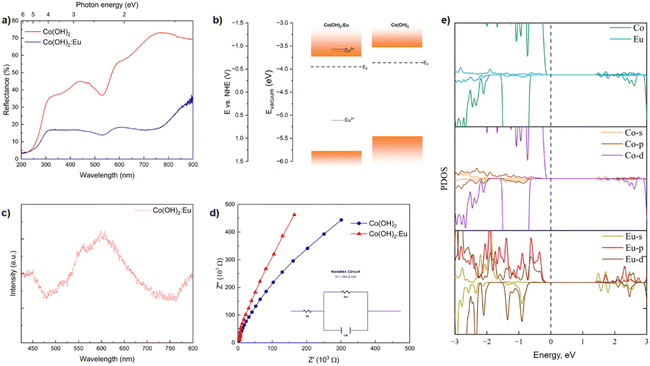 | ||
| Fig. 6 Diffuse reflectance spectra of undoped and Eu-doped Co(OH)2 (a), proposed band structure of Co(OH)2:Eu in comparison with that of undoped Co(OH)2 (ref. 30) (b), luminescence spectra of Co(OH)2:Eu at 12 K (luminescence excitation 4.1 eV) (c), Nyquist plots of Co(OH)2 and Co(OH)2:Eu (d), and the PDOS (partial density of states) of Eu and Co atoms, and their s, p, and d states in Co(OH)2:Eu (e). | ||
The data were then analysed using the Tauc equation to determine the bandgap of the materials. The resulting Tauc plot and fitting are shown in Fig. S4, illustrating a nominal bandgap of 2.05 eV in Eu-doped Co(OH)2 (Fig. S4a), as opposed to 2.17 eV in undoped Co(OH)2 synthesized using the same method (Fig. S4b) and approximately 1.8–1.9 eV for β-Co(OH)2 synthesized using other methods.30,91–93 These values are not far off from the density functional theory calculations of the band structure of monolayer Co(OH)2 (1.52 eV)94 and bulk Co(OH)2 (2.00 eV).95 The decrease in the bandgap after doping confirms the inclusion of the europium ions in the lattice and their displacement of Co2+ ions. The XPS valence band spectra (Fig. S4c), calibrated at the O 2s peak value, were used to determine the value of the valence band of the material (−5.78 eV vs. vacuum). The lowest energy of the conduction band was then calculated based on the known valence band and the bandgap energies, which −3.73 eV. The Fermi level of the doped hydroxide was calculated based on the contact potential difference from the KPFM images (Fig. S5), which turned out to be −3.95 eV. The latter value confirms the doped material as a negative semiconductor, as is the case for undoped Co(OH)2. Both the valence and the conduction band values (Fig. 6b) show a slight change compared to undoped Co(OH)2 introduced by the inclusion of Eu3+ in the structure. Indeed, doping with transition and rare-earth metal ions is a well-known bandgap engineering method.96,97 In heavily doped semiconductors, the density of states changes with the introduction of the dopants, slightly changing the positions of the valence band maximum and the conduction band minimum. When the dopant states interact with each other, they form the impurity band around the dopant energy levels. The broadening of the dopant band and the formation of the band tails may cause bandgap narrowing and valence band shift towards the majority carrier band.98,99
Photocatalysis and photoluminescence are competing processes, resulting from different behaviours of photo-excited electrons and holes. For photocatalysis, reactions occur at the surface of the material, while for photoluminescence, radiative recombination occurs inside the material. Therefore, we performed photoluminescence experiments to study the relaxation mechanism after the excitation. Cobalt-containing compounds are connected to low luminescence intensity, and thus cobalt is considered a luminescence “killer” along with iron and nickel.100,101 Luminescence quenching properties of cobalt are apparent even when present in the structure with a low doping concentration. The photoluminescence spectra of europium-doped cobalt hydroxide (Fig. 6c) show a broad and shapeless luminescence band in the yellow region of the spectra, but the undoped cobalt hydroxide was not found to emit light. The lack of photoluminescence of undoped Co(OH)2 signifies that no radiative recombination takes place. The shape of the luminescence band of Co(OH)2:Eu is a characteristic of surface defect radiative recombination in nanoparticles. However, the spectra of the doped Co(OH)2 lack the characteristic Eu3+ f–f lines at 580–620 nm of high intensity.102–104 The shape of the spectra, along with the established presence of Eu3+ ions proven by XPS and XAS, provides the possibility that the excited electron level of europium is located well above the bottom of the conduction band. Indeed, for the radiative recombination to successfully occur, both ground and excited 4f electron levels of Eu3+ should be located inside the bandgap, which is utilized in europium-doped luminophores such as titania, molybdates, phosphates, vanadates, and many others.102,103,105–109 While the energy difference between the ground and excited levels varies among different compounds where the lanthanides act as the dopants, in the case of Eu3+, it is larger than the determined bandgap of Co(OH)2.110,111 This explains different optical and luminescence properties than expected from materials doped with trivalent europium ions. Therefore, we can assume that a similar process of non-radiative recombination occurs in Co(OH)2, possibly through the existence of deep levels in the bandgap acting as traps. The influence of different energy levels in the doped and undoped Co(OH)2 is further explained below in relation to photoconductivity.
Even though the inclusion of trivalent europium in the material does not cause the radiative 4f–4f recombination, it does include more unfilled levels into the conduction band of the material (Fig. 6b), possibly contributing to the creation of the superoxide radical O2−˙ (ref. 112) that is responsible for the photocatalytic degradation of organic compounds after transformation to OH˙.113
To confirm our hypothesis regarding the band structure of the materials, we used DFT calculations. As shown in Fig. 6e, the introduction of trivalent Eu incorporates more unfilled energy levels into the conduction band of the material, altering its electronic structure and increasing the density of electronic states in the conduction band. We primarily considered the energy level contributions of Co and Eu to Co(OH)2:Eu. The valence band top of Co(OH)2:Eu is mainly contributed by the d orbitals of Co and the p and d orbitals of Eu, while the conduction band bottom is primarily contributed by the s orbitals of Co and Eu. Due to the self-interaction errors in the strongly localized orbitals, f-elements are notoriously difficult to describe with DFT. Therefore, it is regrettable that the contribution of the Eu 4f orbitals was not considered in this analysis of atomic orbital contributions. However, the Eu 4f contribution can be visualized in the photoconductivity analysis discussed below.
Electrochemical impedance spectroscopy (Fig. 6d) was used to determine the ionic conductivity changes in Co(OH)2 upon doping with europium. The ionic conductivities derived from EIS are 1.61 × 10−8 S cm−1 for pure Co(OH)2 and 3.98 × 10−9 S cm−1 for europium-doped Co(OH)2. Higher ionic conductivity and a smaller semicircle in the Nyquist plot (Fig. S6, with the Randles circuit model fitting results in Table S4) in the case of undoped hydroxide signify a lower charge transfer resistance (Rct) and a more effective charge carrier separation, and, as a result, a lower degree of recombination processes. The Rct parameter relates to the facility of the photogenerated charges to reach the electrolyte. In Co(OH)2:Eu, the larger Rct values imply a more complex interfacial charge transfer kinetics, due to charge trapping induced by Eu3+ doping. These trap sites temporarily hold photogenerated charges and reduce recombination, promoting a higher photocurrent. From the point of view of the EIS measurements, the undoped hydroxide would be a better photocatalyst than the doped one. This seems to confirm the previously described defect-related luminescence of Co(OH)2:Eu compared to the lack of luminescence in the case of pure Co(OH)2.
The band structures examined above are clearly reflected by the photoconductivity results. Fig. 7a shows an example of such results for Eu-doped Co(OH)2. It can be seen that as the photon energy increased, the same electric field induced a smaller current density, indicating negative photoconductivity. In fact, negative photoconductivity is observed in both undoped and doped Co(OH)2, as shown in Fig. 7b, but the evolution of conductivity as a function of incident photon energy experienced a bifurcation after 2.25 eV. The decrease in conductivity stopped at an incident photon energy of 2.25 eV for the undoped samples, whereas for the doped samples, the conductivity continued to decrease when the incident photon energy increased to 3.06 eV. Despite the actual applied voltage during the I–V curve measurement reaching a high value of 100 V, no signs of ion migration or secondary phase formation were observed after the measurement (Fig. S7). Therefore, the conductivity response must be attributed to the band structures of the samples, as has been previously reported for negative photoconductivity in two-dimensional materials caused by trap centers in the bandgap.56
Fig. 7c and d propose the band structures in undoped and doped Co(OH)2, respectively, during the photoconductivity measurement. Under dark conditions, the isolated in-gap state, which is approximately 1.7 eV or 1.5 eV above the valence band top (according to Fig. S4, see the edges of the absorption tails), does not contribute to the total conductivity. The origin of this in-gap state is attributed either to a low amount of Co3O4 phase found detected by XRD or to an intrinsic isolated trap state. The total conductivity (σtotal) is defined by the dark conductivity (σdark) and photoconductivity (σphoto) viaeqn (3):
| σtotal = σdark + σphoto = q(ne + Δne)µe + q(nh + Δnh)µh | (3) |
For undoped Co(OH)2, no local state exists above the conduction band bottom. When the incident photon energy increases to 3.06 eV, Δne is largely recovered by the contribution of electrons that are excited to higher energy levels. Due to this, σtotal stops decreasing. On the other hand, the state above the conduction band bottom of the doped Co(OH)2 may continue to localize the higher-energy excited electrons under photons of 3.06 eV, leading to a continuous decrease of σtotal. This characteristic offers the possibility of using undoped and europium-doped Co(OH)2 as a pair for photodetection or photo-switching under violet or UV light, with the unfilled 4f states above the conduction band edge to influence it to the full extent.
Fig. S8a shows the chronoamperometry profiles of Co(OH)2 and Co(OH)2:Eu obtained at an applied potential of 0.3 V vs. Ag/AgCl. The photocurrent response is higher for the modified material, Co(OH)2:Eu. The net photocurrent is affected by the light absorption, charge generation, separation, and surface kinetics. The smaller band gap of Co(OH)2:Eu provides better visible light absorption and more photogenerated charges, which leads to a higher photocurrent response. From Mott–Schottky analysis (Fig. S8b), the flat band potential (Fb) and donor density (Nd) were obtained. These values are summarized in Table S4. The Mott–Schottky test served as additional confirmation that both materials possess the properties of n-type semiconductors, as demonstrated in Fig. 6b.
To further study the difference in the photoinduced processes upon Eu3+ doping, photocatalytic degradation of the methylene blue (MB) pigment was performed and compared with the degradation of the pigment under the same conditions without the doped Co(OH)2 being present. Changes in the rate of MB degradation when using both undoped and europium-doped Co(OH)2 are shown in Fig. 8a.
Higher degradation efficiency in the presence of Co(OH)2:Eu (Fig. 8b) compared to photolysis proves the photocatalytic properties of the synthesized material. However, the efficiency of the degradation compared to undoped Co(OH)2 is slightly better despite the EIS and photoluminescence results suggesting the opposite, which could confirm the presence of non-radiative recombination in pure Co(OH)2. The adsorption properties of Co(OH)2:Eu did not influence the experiment, as proven by the degradation experiment of methylene blue in the presence of the photocatalyst and in the absence of light irradiation.
The same degradation reaction also occurred when using other concentrations of methylene blue. As shown in Fig. 8c, the photocatalytic degradation of a 15 mg L−1 methylene blue solution followed a similar trend, even though at a lower rate, explained by exceeding the limit of the photocatalyst capabilities and covering of the active sites of the photocatalyst with dye molecules.114 The photocatalytic degradation of the methylene blue solution, which was twice more diluted than the original solution studied (5 mg L−1), followed a similar trend as well; however, the methylene blue determination proved to be more difficult as the absorption values were approaching the quantification limit.
Radical scavenger experiments were used to confirm the influence of OH˙ radicals on the reaction (Fig. 8b) and to compare the mechanism using the undoped and doped Co(OH)2. In Fig. S9a and b, it is demonstrated that upon addition of isopropanol, which acts as a scavenger of the hydroxyl radical, the degradation of methylene blue slows down already during the first hour of the experiment. Considering the band edge positions (valence band maximum 1.34 V vs. NHE), Co(OH)2 will not be able to oxidize water to hydroxyl radicals directly (redox potential 2.80 V at pH 0),115 which are demonstrated to be responsible for photocatalytic degradation. In this case, the O2−˙ radicals are formed at the valence band and are then converted to OH˙ radicals in solution, as confirmed in previous studies.30 As mentioned in Table S4, Co(OH)2:Eu exhibits a more negative flat band potential, related to a stronger reducing power of electrons. Hence, it is more inclined to transform O2 → O2−˙ → H2O2 → OH˙, which are the main species involved in organic dye degradation. A higher Nd provides better electronic conductivity and carrier density. Therefore, even when Rct is higher in this material, more charges reach the surface with a higher driving force, resulting in a higher photocurrent and photocatalytic organic dye degradation efficiency.
Experiments involving the scavenging of O2−˙ radicals were not conducted because the redox reaction between methylene blue and K2Cr2O7, used to inhibit superoxide radical activity, interfered with the results. Instead, the methylene blue degradation reaction in an argon atmosphere was conducted and monitored (Fig. 8b). The anoxic atmosphere prevented the first step of oxygen adsorption on the surface of the photocatalyst, and, consequently, no superoxide radicals were formed, as confirmed by the decreased rate of methylene blue photocatalytic degradation.
The methylene blue photocatalytic degradation by Co(OH)2:Eu was also checked in basic media (pH 9). As demonstrated by Fig. S9c, both photolysis and the photocatalytic reaction of methylene blue reached almost full degradation after an hour under light irradiation. At higher pH values, the cationic methylene blue molecule does not compete with H+ for adsorption to the surface (negatively charged in basic solutions); thus, the subsequent degradation reaction occurs faster than at lower pH.114,116–118 The higher stability of hydroxyl radicals and hydroxide ions at high pH values also increases the photocatalytic and photolysis rate. The photocatalytic degradation experiments were not performed in acidic media due to the instability of Co(OH)2:Eu in acidic solutions.
DFT calculations helped us understand why the behaviours in photocatalytic degradation of doped and undoped samples were opposite to each other when compared to our expectations based on the photoluminescence and EIS results. Indeed, we could notice a higher degradation rate when using Co(OH)2:Eu despite having an alleged higher recombination rate, as confirmed by photoluminescence and EIS. We ascribe this incongruence to a different capacity of the two materials to generate the designated radicals active in the degradation process. To explore the capacity of Co(OH)2 to generate O2−˙ both before and after Eu doping, we initially constructed the structures of Co(OH)2 adsorbing O2 and Co(OH)2:Eu adsorbing O2, as depicted in Fig. 9. When Co(OH)2 adsorbs O2, the O atom is directly opposite to the Co atom, with the two O atoms vertically aligned. Conversely, when Co(OH)2:Eu adsorbs O2, the two O atoms are arranged diagonally, with one O atom positioned above the Eu atom. The O–O bond lengths are 1.38 Å and 1.40 Å, respectively, which differ from the O–O bond length of 1.21 Å in O2 and are close to the O–O bond length of O2−, which is 1.33 Å.
To further validate that the optimized adsorbed species is O2−˙ rather than O2, we calculated the differential charge density to ascertain the charge-transfer scenario. As presented in Fig. 10, the regions surrounding the oxygen atoms are yellow, denoting electron accumulation, while the outer regions are cyan, signifying electron depletion. This indicates that in both the cases of Co(OH)2 and Co(OH)2:Eu, electrons are transferred to O2. To further quantify the charge transfer, Bader charge analyses were conducted. The results revealed that Co(OH)2 transferred 1.01|e| to O2, and Co(OH)2:Eu transferred 1.33|e| to O2, further corroborating that both Co(OH)2 and Co(OH)2:Eu adsorb O2−˙. Additionally, when Co(OH)2:Eu adsorbs O2, Eu loses 1.43|e|, and Eu functions as an electron donor to facilitate the reduction of O2 to O2−˙.
 | ||
| Fig. 10 Differential charge density of O2 adsorption on Co(OH)2 and Co(OH)2:Eu, where yellow and cyan represent electron accumulation and depletion, respectively. | ||
Furthermore, we calculated the adsorption energy (Eads) using the formula Eads = EAB − EA − EB, where EAB represents the total energy of O2 adsorption on Co(OH)2 (or Co(OH)2:Eu), EA refers to the energy of Co(OH)2 (or Co(OH)2:Eu), and EB refers to the energy of O2. The results indicate that the adsorption energy of O2 on Co(OH)2 is −3.89 eV, whereas that on Co(OH)2:Eu is −11.37 eV. The more negative the adsorption energy, the more stable the adsorption. Co(OH)2:Eu exhibits strong chemical adsorption of O2, which can be stably immobilized on the surface. This provides ample time for electron transfer and prevents the premature desorption of O2, which needs to be adsorbed in order to be reduced by the catalyst.
In summary, the adsorption energy of O2 on Co(OH)2:Eu (−11.37 eV) is markedly stronger than that on the undoped system (−3.89 eV). Moreover, the Bader charge analysis indicates that Co(OH)2:Eu transfers 1.33|e| to O2, which is close to the theoretical charge of the superoxide radical O2− (−1|e|). Although the undoped system can also generate O2−˙ (with a charge transfer of 1.01|e|), its weak adsorption leads to the facile desorption of O2, resulting in lower actual catalytic efficiency due to a lower production of OH˙ from O2−˙. The differential charge density further discloses that Eu doping significantly enhances the electron-supply capacity and the stability of O2−˙ by forming a Co–Eu synergistic electron transfer pathway, thereby more efficiently promoting the generation of superoxide radicals and the following production of OH˙.
Radical scavenger experiments in combination with the DFT calculations of O2−˙ adsorption and desorption reveal that the key to more efficient photocatalytic properties of the doped system lies in the promotion of the superoxide radical and hence the hydroxyl radical production after irradiation with light. Despite the undoped hydroxide possessing ionic conductivity and a lack of radiative recombination, europium-doped cobalt hydroxide is still better suited to photocatalytic degradation as is proven on the basis of methylene blue degradation.
Conclusions
In this work, we have successfully synthesized europium-doped Co(OH)2 nanoplates via a one-pot hydrothermal synthesis route for the first time. We have excluded the presence of bigger Eu-based aggregates and have confirmed the ions to be Eu3+. More importantly, the comparative characterization has demonstrated a higher stability of Co(OH)2:Eu for potentially increasing the time of use in photocatalysis, as is reflected by the increase in thermal stability by 30 °C. The photoconductivity studies showed that the introduction of trivalent europium ions into the crystal structure of cobalt hydroxide results in a 1.5-fold increase in the material's negative photoconductivity, offering the opportunity for further studies on rare-earth doped hydroxides in negative photoconductivity applications. Additionally, doping of Co(OH)2 with trivalent europium ions increases the photocatalytic production of hydroxyl radicals, which are then actively used in degradation reactions, as demonstrated by the increase in methylene blue photocatalytic degradation from 66% to 72% compared to undoped Co(OH)2. This work paves the way for the novel higher applicability of doped transition metal hydroxides in photo-mediated processes. Future studies may focus on the use of different dopants and/or hydroxides.Author contributions
Viktorija Pankratova: investigation, formal analysis, visualization, writing – original draft, review & editing; Rossella Greco: methodology, formal analysis, writing – review & editing; Takashi Yamamoto – investigation, formal analysis, writing – original draft; Joanna Hoszowska: writing – review & editing; Meng Zhang: investigation, formal analysis, writing – original draft; Ali M. Huerta-Flores: investigation, formal analysis, visualization, writing – review & editing; Miikka Willman: investigation, visualization; Kimmo Sirkka: investigation, writing – original draft; Vladimir Pankratov: investigation, writing – review & editing; Yang Bai: investigation, formal analysis, visualization, writing – review & editing; Wei Cao: funding acquisition, conceptualization, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data reported here can be found on Zenodo at https://doi.org/10.5281/zenodo.17130017 or obtained upon reasonable request from the corresponding author.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta07316b.
Acknowledgements
Viktorija Pankratova, Rossella Greco, and Wei Cao acknowledge funding from the European Research Council (ERC) under the European Union's Horizon 2020 Research and Innovation Programme (Grant Agreement No. 101002219). Kimmo Sirkka and Yang Bai acknowledge the funding from the European Union (ERC, UNIFY, 101039110). Ali M. Huerta-Flores acknowledges the University of Oulu & the Research Council of Finland Profi 352788, as well as the Nano and Molecular Systems Research Unit and the Sustainable Chemistry Unit for the acquisition of the Corrtest Instrument CS310M potentiostat and its consumables. The work has been partially supported by ERDF Project No. 1.1.1.3/1./24/A/077. Views and opinions expressed are, however, those of the authors only and do not necessarily reflect those of the European Union or the European Research Council. Neither the European Union nor the granting authority can be held responsible for them. The Center of Material Analysis (CMA), University of Oulu and Research Council of Finland (decision number 348700) are acknowledged. We acknowledge DESY (Hamburg, Germany), a member of the Helmholtz Association HGF, for the provision of experimental facilities. Parts of this research were carried out at Petra III. Data were collected using the P66 beamline Superlumi provided by DESY Photon Science. We would like to thank Dr Aleksei Kotlov for his assistance during the experiments. Beamtime was allocated for proposal I-20221411 EC. We are thankful to Dr Edgars Elsts (Institute of Solid State Physics, University of Latvia) for XRD measurements.References
- M. Grzegorzek, K. Wartalska and B. Kaźmierczak, Int. Commun. Heat Mass Tran., 2023, 143, 106674 CrossRef.
- J. M. Quimbayo, S. Ojala, S. Urpelainen, M. Huuhtanen, W. Cao, M. Huttula and R. L. Keiski, Advanced Oxidation Processes for Wastewater Treatment, 2022, pp. 249–270 Search PubMed.
- S. Mishra, L. Acharya, B. Marandi, K. Sanjay and R. Acharya, Diam. Relat. Mater., 2024, 142, 110834 CrossRef CAS.
- L. Xie, J. G. Hao, H. Q. Chen, Z. X. Li, S. Y. Ge, Y. Mi, K. Yang and K. Q. Lu, Catal. Commun., 2022, 162, 106371 CrossRef CAS.
- C. Prasad, H. Tang, Q. Q. Liu, S. Zulfiqar, S. Shah and I. Bahadur, J. Mol. Liq., 2019, 289, 111114 CrossRef CAS.
- K. Maeda, K. Ishimaki, Y. Tokunaga, D. Lu and M. Eguchi, Angew. Chem., Int. Ed., 2016, 55, 8309–8313 CrossRef CAS PubMed.
- Z. Gao, Z. W. Yu, F. Q. Liu, C. Yang, Y. H. Yuan, Y. Yu and F. Luo, ChemSusChem, 2019, 12, 4623–4628 CrossRef CAS PubMed.
- G. Liu, L. Wang, H. G. Yang, H. M. Cheng and G. Q. Lu, J. Mater. Chem., 2010, 20, 831–843 RSC.
- R. Botella, W. Cao, J. Celis, J. Fernández-Catalá, R. Greco, L. Lu, V. Pankratova and F. Temerov, J. Phys.: Condens. Matter, 2024, 36, 141501 CrossRef CAS PubMed.
- Z. You, D. Lu, K. K. Kondamareddy, W. Gu, Y. Su, J. Pan, J. Yang, P. Cheng and W. Ho, Sep. Purif. Technol., 2025, 361, 131293 CrossRef CAS.
- E. Kabir, P. Kumar, S. Kumar, A. A. Adelodun and K. H. Kim, Renew. Sustain. Energy Rev., 2018, 82, 894–900 CrossRef.
- Z. Shan, Y. Yang, H. Shi, J. Zhu, X. Tan, Y. Luan, Z. Jiang, P. Wang and J. Qin, Front. Chem., 2021, 9, 755836 CrossRef CAS PubMed.
- J. Xiao, Y. Chen, C. Cai, S. Lai, L. Cheng, J. Zhang, W. Zhu, Y. Guo, M. Hou, L. Ma, W. Chen, X. Chen and C. P. Wong, Small, 2025, 21, 2503335 CrossRef CAS.
- X. Xu, Z. Peng, H. Xu and D. Cheng, J. Catal., 2022, 416, 47–57 CrossRef CAS.
- B. Wang, J. Liu, S. Yao, F. Liu, Y. Li, J. He, Z. Lin, F. Huang, C. Liu and M. Wang, J. Mater. Chem. A, 2021, 9, 17143–17172 RSC.
- S. Ida and T. Ishihara, J. Phys. Chem. Lett., 2014, 5, 2533–2542 Search PubMed.
- R. Marschall and L. Wang, Catal. Today, 2014, 225, 111–135 CrossRef CAS.
- F. M. Sanakousar, C. Vidyasagar, V. M. Jiménez-Pérez and K. Prakash, Mater. Sci. Semicond. Process., 2022, 140, 106390 CrossRef CAS.
- S. Samuei, S. S. Akbari, E. Ülker and F. Karadas, Catal. Sci. Technol., 2022, 12, 3044–3053 Search PubMed.
- L. Chen, H. Zhang, L. Chen, X. Wei, J. Shi and M. He, J. Mater. Chem. A, 2017, 5, 22568–22575 RSC.
- C. Wang, W. Li, A. A. Kistanov, H. Singh, Y. Kayser, W. Cao and B. Geng, J. Colloid Interface Sci., 2022, 628, 398–406 CrossRef CAS PubMed.
- B. Zheng, J. Fan, B. Chen, X. Qin, J. Wang, F. Wang, R. Deng and X. Liu, Chem. Rev., 2022, 122, 5519–5603 CrossRef CAS.
- Z. M. El-Bahy, A. A. Ismail and R. M. Mohamed, J. Hazard. Mater., 2009, 166, 138–143 CrossRef CAS PubMed.
- Y. Zhang, H. Zhang, Y. Xu and Y. Wang, J. Solid State Chem., 2004, 177, 3490–3498 CrossRef CAS.
- Z. Liang, Y. Zhang, G. Zhang, J. Liu, Y. Cai, Y. Wang, Y. Zhao, G. Li and K. Bei, Int. J. Hydrogen Energy, 2023, 48, 18644–18656 CrossRef CAS.
- H. Liu, D. Wierzbicki, R. Debek, M. Motak, T. Grzybek, P. Da Costa and M. E. Gálvez, Fuel, 2016, 182, 8–16 CrossRef CAS.
- P. Talebi, H. Singh, E. Rani, M. Huttula and W. Cao, RSC Adv., 2021, 11, 2733–2743 RSC.
- P. Talebi, R. Greco, T. Yamamoto, M. Zeynali, S. Asgharizadeh and W. Cao, Mater. Adv., 2024, 5, 2968–2973 RSC.
- R. Chen, H. Zhu, W. Liu, D. Zhan, Q. Fu, J. Tian, Y. Huang and C. Han, J. Am. Ceram. Soc., 2024, 107, 5201–5211 CrossRef CAS.
- R. Greco, L. Baxauli-Marin, F. Temerov, M. Daboczi, S. Eslava, Y. Niu, A. Zakharov, M. Zhang, T. Li and W. Cao, Chem. Eng. J., 2023, 471, 144569 CrossRef CAS.
- P. Bai, J. Xie, H. Wang, X. Kang and X. Wang, Appl. Surf. Sci., 2023, 640, 158305 CrossRef CAS.
- T. Y. Ma, S. Dai, M. Jaroniec and S. Z. Qiao, J. Am. Chem. Soc., 2014, 136, 13925–13931 CrossRef CAS.
- H. Li, C. Sun, Y. Zhao, X. Xu and H. Yu, J. Nanopart. Res., 2018, 20, 1–13 CrossRef.
- X. Ma, Y. Liu, Y. Wang and Z. Jin, Int. J. Hydrogen Energy, 2021, 46, 33809–33822 CrossRef CAS.
- C. Gao, Q. Meng, K. Zhao, H. Yin, D. Wang, J. Guo, S. Zhao, L. Chang, M. He, Q. Li, H. Zhao, X. Huang, Y. Gao and Z. Tang, Adv. Mater., 2016, 28, 6485–6490 CrossRef CAS PubMed.
- J. Fernández-Catalá, L. Jussila, M. Daboczi, F. Temerov, S. Eslava, R. Greco and W. Cao, Cryst. Growth Des., 2023, 23, 8828–8837 CrossRef PubMed.
- J. Fernández-Català, J. Chávez-Caiza, R. Greco, M. Navlani-García, A. Berenguer-Murcia, W. Cao and D. Cazorla-Amorós, Inorg. Chem. Commun., 2025, 178, 114424 CrossRef.
- G. Sorekine, G. Anduwan, M. N. Waimbo, H. Osora, S. Velusamy, S. Kim, Y. S. Kim and J. Charles, J. Mol. Struct., 2022, 1248, 131487 CrossRef CAS.
- M. Gunawan, M. Priest, D. Gunawan, S. Nie, A. Satriyatama, J. Vongsvivut, Z. Hameiri, Q. Zhang, S. Zhou and R. Amal, Energy Environ. Sustain., 2025, 1, 100019 Search PubMed.
- R. Greco, J. Fernandez-Catalá, J. Quimbayo and R. Botella, Advanced Energy and Sustainability Research, 2025, p. 2500123 Search PubMed.
- K. G. M. Laurier, F. Vermoortele, R. Ameloot, D. E. De Vos, J. Hofkens and M. B. J. Roeffaers, J. Am. Chem. Soc., 2013, 135, 14488–14491 CrossRef CAS.
- G. Jayanthi, S. Sumathi, K. Kannan, V. Andal and S. Murugan, Adv. Mater. Sci. Eng., 2022, 6607683 CAS.
- M. Gao, W. Sheng, Z. Zhuang, Q. Fang, S. Gu, J. Jiang and Y. Yan, J. Am. Chem. Soc., 2014, 136, 7077–7084 CrossRef CAS PubMed.
- S. Anantharaj, K. Karthick and S. Kundu, Mater. Today Energy, 2017, 6, 1–26 CrossRef.
- H. Wang, B. Shao, Y. Chi, S. Lv, C. Wang, B. Li, H. Li, Y. Li and X. Yang, Nanomaterials, 2022, 12, 946 CrossRef CAS PubMed.
- L. Mao, Q. Ba, X. Jia, S. Liu, H. Liu, J. Zhang, X. Li and W. Chen, RSC Adv., 2019, 9, 1260–1269 RSC.
- Y. Li, Y. Hou, Q. Fu, S. Peng and Y. H. Hu, Appl. Catal., B, 2017, 206, 726–733 CrossRef CAS.
- Y. Zhang, Z. Jin, X. Yan, H. Wang and G. Wang, Catal. Lett., 2019, 149, 1174–1185 CrossRef CAS.
- Y. Zhan, G. Du, S. Yang, C. Xu, M. Lu, Z. Liu and J. Y. Lee, ACS Appl. Mater. Interfaces, 2015, 7, 12930–12936 CrossRef CAS.
- L. Huang, J. Jiang and L. Ai, ACS Appl. Mater. Interfaces, 2017, 9, 7059–7067 CrossRef CAS PubMed.
- G. Zhang, S. Zang and X. Wang, ACS Catal., 2015, 5, 941–947 CrossRef CAS.
- L. J. Zhang, R. Zheng, S. Li, B. K. Liu, D. J. Wang, L. L. Wang and T. F. Xie, ACS Appl. Mater. Interfaces, 2014, 6, 13406–13412 CrossRef CAS PubMed.
- S. Liu, R. T. Gao, M. Sun, Y. Wang, T. Nakajima, X. Liu, W. Zhang and L. Wang, Appl. Catal., B, 2021, 292, 120063 CrossRef CAS.
- Y. Zhao, X. Jia, G. I. N. Waterhouse, L. Z. Wu, C. H. Tung, D. O'Hare and T. Zhang, Adv. Energy Mater., 2016, 6, 1501974 CrossRef.
- M. Suksomboon, K. Kongsawatvoragul, S. Duangdangchote and M. Sawangphruk, ACS Omega, 2021, 6, 20804–20811 CrossRef CAS PubMed.
- B. Cui, Y. Xing, J. Han, W. Lv, W. Lv, T. Lei, Y. Zhang, H. Ma, Z. Zeng and B. Zhang, Chin. Phys. B, 2021, 30, 028507 CrossRef CAS.
- N. K. Tailor, C. A. Aranda, M. Saliba and S. Satapathi, ACS Mater. Lett., 2022, 4, 2298–2320 CrossRef CAS.
- M. Y. Hassan and D. S. Ang, ACS Appl. Mater. Interfaces, 2019, 42339–42348 CrossRef CAS PubMed.
- S. Rahman, S. Samanta, A. Kuzmin, D. Errandonea, H. Saqib, D. L. Brewe, J. Kim, J. Lu and L. Wang, Adv. Sci., 2019, 6, 1901132 CrossRef CAS PubMed.
- B. Sreelakshmi and R. Thamankar, Mater. Adv., 2024, 5, 5912–5921 RSC.
- B. Schwenzer, J. R. Neilson, K. Sivula, C. Woo, J. M. J. Fréchet and D. E. Morse, Thin Solid Films, 2009, 517, 5722–5727 CrossRef CAS.
- K. Chatterjee, A. Dutta, S. Mishra, B. Bairy, M. B. Sen, A. Gorai, S. K. Saha and A. J. Akhtar, Ceram. Int., 2023, 49, 32768–32778 CrossRef CAS.
- U. Basak, P. Ghosh, D. P. Chatterjee, G. Mahapatra, A. Banerjee and A. K. Nandi, J. Mater. Chem. A, 2025, 13, 7813–7833 RSC.
- A. A. Minnekhanov, N. T. Le, E. A. Konstantinova and P. K. Kashkarov, Appl. Magn. Reson., 2017, 48, 335–345 CrossRef CAS.
- D. Nečas and P. Klapetek, Cent. Eur. J. Phys., 2012, 10, 181–188 Search PubMed.
- T. Taguchi, J. Harada, A. Kiku, K. Tohji and K. Shinoda, J. Synchrotron Radiat., 2001, 8, 363–365 CrossRef CAS.
- T. Taguchi, T. Ozawa and H. Yashiro, Phys. Scr., T, 2005, 115, 205–206 CrossRef.
- A. L. Ankudinov, B. Ravel, J. J. Rehr and S. D. Conradson, Phys. Rev. B:Condens. Matter Mater. Phys., 1998, 58, 7565 CrossRef CAS.
- V. Pankratov and A. Kotlov, Nucl. Instrum. Methods Phys. Res., Sect. B, 2020, 474, 35–40 CrossRef CAS.
- Y. Smortsova, O. Chukova, M. Kirm, V. Nagirnyi, V. Pankratov, A. Kataev and A. Kotlov, J. Synchrotron Radiat., 2025, 32, 1539–1548 CrossRef CAS PubMed.
- X. Chen, S. Shen, L. Guo and S. S. Mao, Chem. Rev., 2010, 110, 6503–6570 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- J. A. White and D. M. Bird, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 4954 CrossRef CAS PubMed.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B, 1976, 13, 5188 CrossRef.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 43 CrossRef PubMed.
- J. Feng and H. C. Zeng, Chem. Mater., 2003, 15, 2829–2835 CrossRef CAS.
- Z. P. Xu and H. C. Zeng, J. Mater. Chem., 1998, 8, 2499–2506 RSC.
- H. Ebrahimzade, G. R. Khayati and M. Schaffie, Adv. Powder Technol., 2017, 28, 2779–2786 CrossRef CAS.
- J. Yang, H. Liu, W. N. Martens and R. L. Frost, J. Phys. Chem. C, 2010, 114, 111–119 CrossRef CAS.
- E. Higuchi, H. Otsuka, M. Chiku and H. Inoue, J. Power Sources, 2014, 248, 762–768 CrossRef CAS.
- T. Baird, K. C. Campbell, P. J. Holliman, R. W. Hoyle, D. Stirling, B. P. Williams and M. Morris, J. Mater. Chem., 1997, 7, 319–330 RSC.
- K. M. Cole, D. W. Kirk and S. J. Thorpe, Surf. Sci. Spectra, 2020, 27, 024013 CrossRef CAS.
- J. P. Baltrus and M. J. Keller, Surf. Sci. Spectra, 2019, 26(1), 014001 CrossRef CAS.
- M. C. Biesinger, B. P. Payne, A. P. Grosvenor, L. W. M. Lau, A. R. Gerson and R. S. C. Smart, Appl. Surf. Sci., 2011, 257, 2717–2730 CrossRef CAS.
- D. Hunt, G. Garbarino, J. Alberto Rodríguez-Velamazán, V. Ferrari, M. Jobbagy and D. A. Scherlis, Phys. Chem. Chem. Phys., 2016, 18, 30407–30414 RSC.
- N. Di, J. Kubal, Z. Zeng, J. Greeley, F. Maroun and P. Allongue, Appl. Phys. Lett., 2015, 106, 122405 CrossRef.
- Y. Zhang, T. Gao, F. Zhang, X. Qu, Y. Luo, P. Zhang, J. Liang, Y. Song, F. Fang, F. Wang, D. Sun and Y. Liu, Adv. Energy Mater., 2024, 14, 2401834 CrossRef CAS.
- M. N. Taran and G. R. Rossman, Am. Mineral., 2001, 86, 889–895 CrossRef CAS.
- N. V. Kosova, V. F. Anufrienko, T. V. Larina, A. Rougier, L. Aymard and J. M. Tarascon, J. Solid State Chem., 2002, 165, 56–64 CrossRef CAS.
- C. P. Roshni, K. Jithesh, P. M. Anjana, K. Govind Raj and R. B. Rakhi, Next Mater., 2024, 4, 100199 CrossRef.
- M. Wang, J. Q. Wang, C. Xi, C. Q. Cheng, C. Q. Zou, R. Zhang, Y. M. Xie, Z. L. Guo, C. C. Tang, C. K. Dong, Y. J. Chen and X. W. Du, Angew. Chem., Int. Ed., 2020, 59, 11510–11515 CrossRef CAS PubMed.
- L. Wang, Z. Fang, T. Qi, L. Xing, J. Liu, S. Zhang, P. Xu and P. Ning, J. Colloid Interface Sci., 2020, 571, 90–99 CrossRef CAS.
- M. Nagli and M. Caspary Toroker, Solid State Ionics, 2018, 314, 149–155 CrossRef CAS.
- J. Chen and A. Selloni, J. Phys. Chem. C, 2013, 117, 20002–20006 CrossRef CAS.
- J. M. Ogiegło, A. Katelnikovas, A. Zych, T. Jüstel, A. Meijerink and C. R. Ronda, J. Phys. Chem. A, 2013, 117, 2479–2484 CrossRef.
- Y. Ou, W. Zhou, F. Ma, C. Liu, R. Zhou, F. Su, Y. Huang, P. Dorenbos and H. Liang, J. Rare Earths, 2020, 38, 514–522 CrossRef CAS.
- F. T. Geldasa, M. A. Kebede, M. W. Shura and F. G. Hone, AIP Adv., 2022, 12, 115302 CrossRef CAS.
- D. H. Neuhaus, P. P. Altermatt, R. P. Starre, A. Schenk and A. G. Aberle, Conference Record of the IEEE Photovoltaic Specialists Conference, 2000, pp. 104–107 Search PubMed.
- S. Shionoya, W. M. Yen and H. Yamamoto, Phosphor Handbook, CRC Press, Boca Raton, 2nd edn, 2006 Search PubMed.
- M. Tabei, S. Shionoya and H. Ohmatsu, Jpn. J. Appl. Phys., 1975, 14, 240–247 CrossRef CAS.
- F. Baur and T. Jüstel, Opt. Mater.:X, 2019, 1, 100015 CAS.
- V. Serga, R. Burve, A. Krumina, V. Pankratova, A. I. Popov and V. Pankratov, J. Mater. Res. Technol., 2021, 13, 2350–2360 CrossRef CAS.
- Y. Liu, W. Luo, H. Zhu and X. Chen, J. Lumin., 2011, 131, 415–422 CrossRef CAS.
- Ž. Antić, R. M. Krsmanović, M. G. Nikolić, M. Marinović-Cincović, M. Mitrić, S. Polizzi and M. D. Dramićanin, Mater. Chem. Phys., 2012, 135, 1064–1069 CrossRef.
- D. Sofich, S. G. Dorzhieva, O. D. Chimitova, B. G. Bazarov, Y. L. Tushinova, Z. G. Bazarova and R. Y. Shendrik, Phys. Solid State, 2019, 61, 844–846 CrossRef CAS.
- A. Kuzmanoski, V. Pankratov and C. Feldmann, Solid State Sci., 2015, 41, 56–62 CrossRef CAS.
- R. Skaudzius, A. Katelnikovas, D. Enseling, A. Kareiva and T. Jüstel, J. Lumin., 2014, 147, 290–294 CrossRef CAS.
- L. Shirmane, C. Feldmann and V. Pankratov, Phys. B, 2017, 504, 80–85 CrossRef CAS.
- P. Dorenbos, J. Lumin., 2004, 108, 301–305 CrossRef CAS.
- P. Dorenbos, J. Phys.: Condens. Matter, 2003, 15, 8417 CrossRef CAS.
- S. Talukdar and R. K. Dutta, RSC Adv., 2015, 6, 928–936 RSC.
- G. Cohen and R. E. Heikkila, J. Biol. Chem., 1974, 249, 2447–2452 CrossRef CAS PubMed.
- I. Khan, K. Saeed, I. Zekker, B. Zhang, A. H. Hendi, A. Ahmad, S. Ahmad, N. Zada, H. Ahmad, L. A. Shah, T. Shah and I. Khan, Water, 2022, 14, 242 CrossRef CAS.
- Y. Deng and R. Zhao, Curr. Pollut. Rep., 2015, 1, 167–176 CrossRef CAS.
- T. N. T. Thu, N. N. Thi, V. T. Quang, K. N. Hong, T. N. Minh and N. L. T. Hoai, J. Exp. Nanosci., 2016, 11, 226–238 CrossRef.
- N. R. Hendekhale and A. Mohammad-Khah, J. Environ. Chem. Eng., 2020, 8, 104065 CrossRef CAS.
- R. Hejazi, A. R. Mahjoub, A. H. C. Khavar and Z. Khazaee, J. Photochem. Photobiol., A, 2020, 400, 112644 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |